Journal Description
Scientia Pharmaceutica
Scientia Pharmaceutica
is an international, peer-reviewed, open access journal related to the pharmaceutical sciences, published quarterly online. It is the official journal of the Austrian Pharmaceutical Society (ÖPhG). Society members receive discounts on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: CiteScore - Q2 (Pharmaceutical Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 22.8 days after submission; acceptance to publication is undertaken in 3.1 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Journal Clusters-Pharmaceutical Science: Scientia Pharmaceutica, Marine Drugs, Pharmaceuticals, Pharmaceutics, Pharmacy, Future Pharmacology, Pharmacoepidemiology, Drugs and Drug Candidates and Journal of Pharmaceutical and BioTech Industry.
Impact Factor:
2.5 (2024);
5-Year Impact Factor:
3.3 (2024)
Latest Articles
Ocular Irritation Potential and Cytotoxicity of Selected Surfactants and Cosurfactants: Identifying Suitable Concentrations for Topical Ophthalmic Formulations
Sci. Pharm. 2026, 94(2), 46; https://doi.org/10.3390/scipharm94020046 (registering DOI) - 5 Jun 2026
Abstract
The cornea and conjunctiva are particularly susceptible to injury and adverse effects, either induced by topically applied drugs or excipients used in ophthalmic formulations. Surfactants and cosurfactants are important for producing topical eye formulations of poorly water-soluble drugs, yet they have not been
[...] Read more.
The cornea and conjunctiva are particularly susceptible to injury and adverse effects, either induced by topically applied drugs or excipients used in ophthalmic formulations. Surfactants and cosurfactants are important for producing topical eye formulations of poorly water-soluble drugs, yet they have not been always used in concentrations that are nontoxic and non-irritating to the ocular surface. This study systematically compared the cytotoxicity and ocular irritation potential of commonly used ophthalmic surfactants and cosurfactants under standardized experimental conditions using complementary in vitro and ex vivo ocular safety models. The ocular irritation of Tween 80, Cremophor EL, polyethylene glycol 400 (PEG 400) and propylene glycol (PG) was examined using the HET-CAM (conjunctival) and BCOP (corneal) eye assays. The toxic effect of the four excipients after 24 h on HLE-B3 cell growth was investigated and found to be dose-dependent. The highest tolerable concentrations of Tween 80 and Cremophor EL were 0.25% (w/w), whereas PEG 400 and PG were non-toxic at 5% (w/w). Tween 80 and Cremophor EL at 0.25% (w/w) and PEG 400 and PG at 5% (w/w) were all devoid of conjunctival and corneal irritation. This study systematically compared the cytotoxicity and ocular irritation potential of commonly used ophthalmic surfactants and cosurfactants under standardized experimental conditions using complementary in vitro and ex vivo ocular safety models. Interestingly, there is strong agreement between the results obtained using the HET-CAM and BCOP assays, where both have been successfully used to evaluate the potential for ocular irritation caused by the aforementioned excipients.
Full article
(This article belongs to the Special Issue Innovative Perspectives in Ocular Drug Research)
Open AccessArticle
Preparation, Kinetic Stability, and Dissolution Study of Amorphous Norfloxacin
by
Alexander Gerasimov, Dar’ya Khabibulina, Liana Zubaidullina, Elina Mirgazieva, Nikolay Lyadov, Ruslan Nagrimanov and Semen Lapuk
Sci. Pharm. 2026, 94(2), 45; https://doi.org/10.3390/scipharm94020045 - 2 Jun 2026
Abstract
►▼
Show Figures
Obtaining amorphous forms of drugs is one of the ways to increase bioavailability. This is especially important for active pharmaceutical ingredients belonging to class II and IV according to the biopharmaceutical classification. These compounds include the currently widely used fluoroquinolone antibiotics. They have
[...] Read more.
Obtaining amorphous forms of drugs is one of the ways to increase bioavailability. This is especially important for active pharmaceutical ingredients belonging to class II and IV according to the biopharmaceutical classification. These compounds include the currently widely used fluoroquinolone antibiotics. They have low solubility in water and are therefore typically used as hydrochlorides. The presence of a strong acid and a charged active pharmaceutical ingredient in the drug increases solubility, but can also lead to additional side effects and decreased permeability. One way to improve the properties of active pharmaceutical ingredients is to convert them to amorphous form. In this study, an amorphous form of the fluoroquinolone antibiotic norfloxacin was obtained, its stability was determined, and its solubility was studied. It was shown that the resulting amorphous form has good temporal stability. The optimal models describing the cold crystallization process are the Nakamura and Sbirrazzuoli models. Despite the slower dissolution kinetics compared to the crystalline sample, the amorphous form shows higher equilibrium solubility values. These results can be used in pharmaceutical engineering to produce amorphous forms of active pharmaceutical ingredients and determine their stability.
Full article

Figure 1
Open AccessArticle
Green-Synthesized Silver Nanoparticles Derived from Calotropis procera as a Multifunctional Nanotherapeutic Platform Targeting Helicobacter pylori, Oxidative Stress, Inflammation, and Gastric Cancer
by
Mounishwaran Kamalesan, Mohanraj Raja, Rameshkumar Neelamegam, Muthukalingan Krishnan, Kayalvizhi Nagarajan and Douglas J. H. Shyu
Sci. Pharm. 2026, 94(2), 44; https://doi.org/10.3390/scipharm94020044 - 29 May 2026
Abstract
Green synthesis of silver nanoparticles (CP-AgNPs) using Calotropis procera (CP) offers a sustainable approach to producing multifunctional therapeutic nanomaterials. This study aimed to synthesize CP-AgNPs and evaluate their antimicrobial, antioxidant, anti-inflammatory, and anticancer potential, with a focus on Helicobacter pylori and gastric cancer
[...] Read more.
Green synthesis of silver nanoparticles (CP-AgNPs) using Calotropis procera (CP) offers a sustainable approach to producing multifunctional therapeutic nanomaterials. This study aimed to synthesize CP-AgNPs and evaluate their antimicrobial, antioxidant, anti-inflammatory, and anticancer potential, with a focus on Helicobacter pylori and gastric cancer cells. CP-AgNPs were prepared by phytochemical reduction using CP leaf extract and characterized by UV–Vis, XRD, FTIR, SEM, EDX, TEM, and Zeta. Antibacterial activity against H. pylori, time-kill kinetics, and SEM imaging of membrane damage were performed. Antioxidant (DPPH, ABTS) and anti-inflammatory assays, together with cytotoxicity studies in AGS cells (DAPI, AO/EtBr, and SEM), were also conducted. CP-AgNPs exhibited an SPR peak at 432 nm, face-centered cubic crystallinity, and spherical morphology (8–32 nm). They showed strong, dose-dependent antibacterial activity against H. pylori, surpassing metronidazole at higher doses. Time-kill assays and SEM confirmed membrane disruption. Antioxidant activity was notable (IC50: 40 µg/mL for DPPH; 60 µg/mL for ABTS). CP-AgNPs demonstrated significant anti-inflammatory effects and dose-dependent cytotoxicity in AGS cells, inducing apoptosis and morphological alterations. The broad biological activity of CP-AgNPs likely arises from the synergy between silver ions and CP phytochemicals. Their superior antibacterial effects, combined with antioxidant and anti-inflammatory properties, indicate strong therapeutic potential for gastric diseases. Anticancer activity in AGS cells suggests additional biomedical relevance, which may involve ROS-associated and apoptosis-related pathways, as suggested by previous studies. CP-AgNPs represent a promising natural nanoplatform for managing H. pylori infection, oxidative stress, inflammation, and gastric cancer, warranting further mechanistic and in vivo studies.
Full article
Open AccessArticle
Discovery of Potential Antihypertensive Agents from the Marine Microalga Phaeodactylum tricornutum Through Metabolite Profiling and In Silico Analysis
by
Miguel Ernesto Guzmán-Rodríguez, Marco Antonio Valdez-Flores, Cinthia Ayón-Fernandez, José Juan Ordaz-Ortiz, Alma Marlene Guadrón-Llanos, Javier Magaña-Gómez, Alberto Kousuke de la Herrán-Arita, Josué Camberos-Barraza, Verónica Judith Picos-Cárdenas, Juan Fidel Osuna-Ramos, Claudia Desireé Norzagaray-Valenzuela and Loranda Calderón-Zamora
Sci. Pharm. 2026, 94(2), 43; https://doi.org/10.3390/scipharm94020043 - 21 May 2026
Abstract
►▼
Show Figures
Hypertension remains a leading cause of global morbidity and mortality, and angiotensin-converting enzyme (ACE) represents a central therapeutic target within the renin–angiotensin–aldosterone system. Marine microalgae, particularly Phaeodactylum tricornutum, provide an underexplored reservoir of structurally diverse metabolites with potential cardiovascular relevance. In this
[...] Read more.
Hypertension remains a leading cause of global morbidity and mortality, and angiotensin-converting enzyme (ACE) represents a central therapeutic target within the renin–angiotensin–aldosterone system. Marine microalgae, particularly Phaeodactylum tricornutum, provide an underexplored reservoir of structurally diverse metabolites with potential cardiovascular relevance. In this in silico study, we characterized metabolites putatively annotated by UPLC-ESI-HRMS and evaluated their predicted ACE inhibitory potential. We performed molecular docking with AutoDock 4 and assessed pharmacokinetic and toxicological properties using the SwissADME, PASS, and ProTox platforms. Several metabolites showed favorable binding orientations within the ACE catalytic pocket, including interactions with key residues and proximity to the zinc-binding motif. Lehualide G, Val–Asn–Pro, tanariflavanone B, hydroxyterbinafine, and anhydro-vitamin A exhibited the most favorable docking profiles. PASS predictions indicated vascular-related bioactivity signals for selected compounds, whereas ADMET modeling revealed heterogeneous but classifiable pharmacokinetic and safety characteristics. The convergence of predicted binding compatibility, bioactivity signals, and stratified safety margins supports P. tricornutum as a promising source of candidate molecules for further experimental validation in antihypertensive research.
Full article
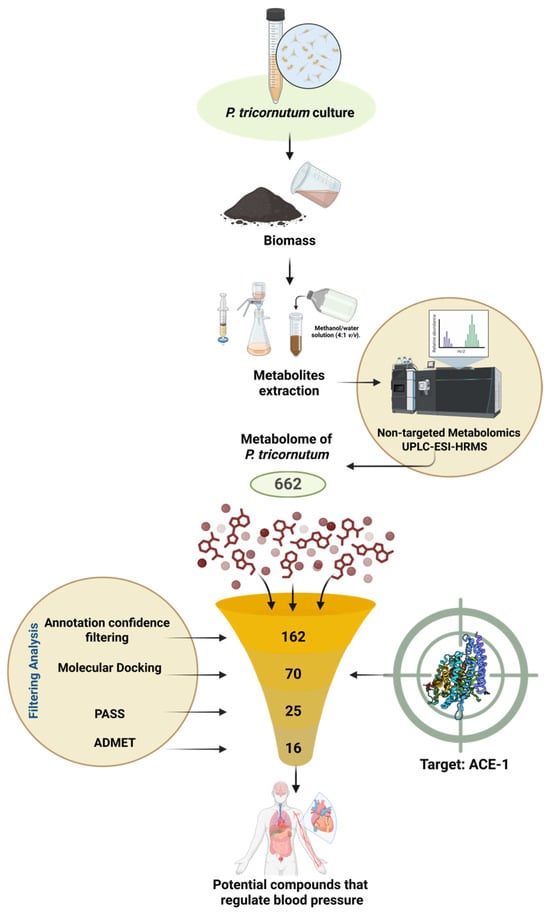
Figure 1
Open AccessArticle
Multivariate Robustness Modeling of Cannabidiol and Δ9-Tetrahydrocannabinol Quantification Using Two-Level Full Factorial Design
by
Athip Maha, Thanapat Songsak, Surang Leelawat and Chaowalit Monton
Sci. Pharm. 2026, 94(2), 42; https://doi.org/10.3390/scipharm94020042 - 20 May 2026
Abstract
►▼
Show Figures
The present study aimed to establish a robustness modeling framework for the determination of cannabidiol (CBD) and Δ9-tetrahydrocannabinol (THC) in cannabis extract using a multivariate approach. A two-level full factorial design was implemented to examine four critical analytical factors, including methanol
[...] Read more.
The present study aimed to establish a robustness modeling framework for the determination of cannabidiol (CBD) and Δ9-tetrahydrocannabinol (THC) in cannabis extract using a multivariate approach. A two-level full factorial design was implemented to examine four critical analytical factors, including methanol concentration (80–85% v/v), flow rate (0.8–1.2 mL/min), column temperature (23–27 °C), and detection wavelength (208–212 nm). Seven analytical responses for each compound were assessed, including peak area, retention time, resolution, asymmetry factor, number of theoretical plates, capacity factor, and peak area difference relative to the reference method. Statistical analysis demonstrated that both main effects and interaction effects significantly influenced the measured responses. Design space construction was performed based on predefined acceptance criteria to ensure method robustness: resolution > 1.5, asymmetry < 1.5, number of theoretical plates > 2000, capacity factor > 2, and peak area difference within −5% to 5%. Predictive performance of the developed models was verified by comparing predicted and experimental results. Good agreement was observed under most conditions, whereas deviation was noted for THC quantification at a detection wavelength of 212 nm. Furthermore, CBD and THC contents determined under three selected operating conditions within the established design space were statistically comparable to those obtained using the reference method, except for the condition employing 212 nm detection. The Analytical GREEnness Metric Approach (AGREE) assessment indicated moderate greenness performance of the analytical procedure. Overall, the multivariate two-level full factorial design proved to be an effective tool for robustness modeling of the HPLC method for simultaneous quantification of CBD and THC.
Full article
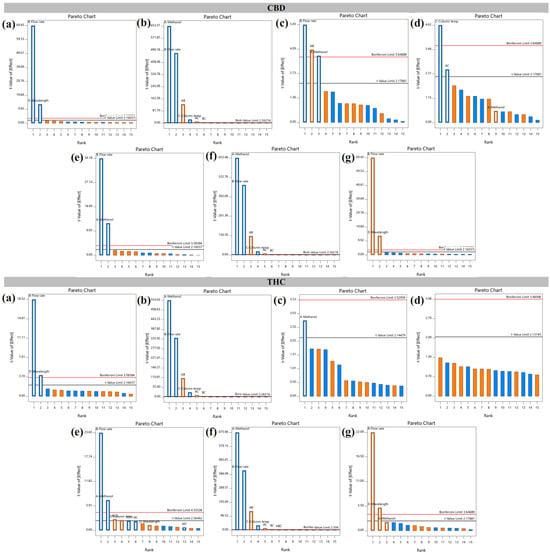
Figure 1
Open AccessReview
Active Pharmaceutical Ingredients in Medical Cannabis: Manufacturer Profiling, Standardization Challenges, and Technological Compatibility
by
Liliia Vyshnevska, Maryana Yaromiy, Iryna Pestun, Kaloyan D. Georgiev, Iliya Zhelev Slavov and Oleh Koshovyi
Sci. Pharm. 2026, 94(2), 41; https://doi.org/10.3390/scipharm94020041 - 18 May 2026
Abstract
►▼
Show Figures
The pharmaceutical development of cannabis-based medicinal products is challenged by significant variability in the quality, composition, and standardization of plant-derived active pharmaceutical ingredients (APIs). In Ukraine, despite recent legislative liberalization, a substantial shortage of standardized raw materials continues to limit the development of
[...] Read more.
The pharmaceutical development of cannabis-based medicinal products is challenged by significant variability in the quality, composition, and standardization of plant-derived active pharmaceutical ingredients (APIs). In Ukraine, despite recent legislative liberalization, a substantial shortage of standardized raw materials continues to limit the development of innovative dosage forms. This study analyses international practices among API manufacturers to identify technological parameters necessary to overcome domestic market barriers and support the implementation of advanced drug delivery systems. Content analysis was conducted on regulatory documentation, professional literature, and manufacturers’ technical specifications. Candidate evaluation followed predefined inclusion and exclusion criteria. The study assessed compliance with Good Manufacturing Practice (GMP) requirements, extraction and purification technologies, the extent of analytical characterization, and batch-to-batch reproducibility. Purposive sampling enabled a comparative analysis of various technological approaches. Marked heterogeneity was observed in API standardization and analytical control indicators among manufacturers. Possession of a GMP certificate was found necessary but may be insufficient to ensure the pharmaceutical equivalence of materials. Differences in extraction methods and purification levels may affect stability profiles, pharmaceutical development strategies, and risk management related to final product quality. The findings demonstrate that manufacturer selection is a critical decision point in pharmaceutical development, with substantiated supplier choice directly influencing dosage form development and regulatory compliance.
Full article

Figure 1
Open AccessArticle
Synthesis and Biological Evaluation of Thiazolyl-Benzene/Camphor Sulfonamide Derivatives as Antibacterial, Antioxidant, and Antidiabetic Compounds
by
Sreenivas Avula, Satish Koppireddi, Micky D. Tortorella and Cleopatra Neagoie
Sci. Pharm. 2026, 94(2), 40; https://doi.org/10.3390/scipharm94020040 - 14 May 2026
Abstract
Thiazolyl-benzene/camphor sulfonamide derivatives (series 4a–k, 5a–j and 6a–i) were synthesized by reaction of various aryl sulfonyl chlorides and camphor sulfonyl chlorides with 2-amino-4-phenylthiazole. The compounds were evaluated for antibacterial, antioxidant, and α-glucosidase/α-amylase inhibitory activities. Biological screening showed that 4h, 5g
[...] Read more.
Thiazolyl-benzene/camphor sulfonamide derivatives (series 4a–k, 5a–j and 6a–i) were synthesized by reaction of various aryl sulfonyl chlorides and camphor sulfonyl chlorides with 2-amino-4-phenylthiazole. The compounds were evaluated for antibacterial, antioxidant, and α-glucosidase/α-amylase inhibitory activities. Biological screening showed that 4h, 5g and 5i displayed significant activity against most Gram-positive bacteria (MICs 4.68–18.75 µg/mL), while 4b and 5i were active against most Gram-negative bacteria with similar MIC ranges. In the DPPH assay, 4e, 4f, 4g and 4h exhibited slightly stronger radical-scavenging activity than ascorbic acid (IC50 ≈ 3.5–3.8 µM vs. 4.14 µM); 5f emerged as the best dual carbohydrate-digesting enzyme inhibitor, and 5b and 5e demonstrated selectivity toward α-amylase.
Full article
(This article belongs to the Special Issue Heterocyclic Chemistry in Drug Design 3.0)
►▼
Show Figures
Figure 1
Open AccessArticle
Machine Learning Integration of In-Silico QSAR, Graph Neural Networks and Docking Reveal Natural Products Inhibitors Against Mycobacterium tuberculosis
by
Sakthidhasan Periasamy, Rajesh Ramasamy, Rajasekar Chinnaiyan and Arun Sridhar
Sci. Pharm. 2026, 94(2), 39; https://doi.org/10.3390/scipharm94020039 - 14 May 2026
Abstract
►▼
Show Figures
Background/Objectives: Tuberculosis (TB), caused by Mycobacterium tuberculosis, remains a major global health challenge, exacerbated by the emergence of multidrug-resistant strains and limited efficacy of existing therapies. Given the involvement of multiple essential mycobacterial proteins, multitarget drug discovery represents a rational therapeutic strategy.
[...] Read more.
Background/Objectives: Tuberculosis (TB), caused by Mycobacterium tuberculosis, remains a major global health challenge, exacerbated by the emergence of multidrug-resistant strains and limited efficacy of existing therapies. Given the involvement of multiple essential mycobacterial proteins, multitarget drug discovery represents a rational therapeutic strategy. Methods: In this study, an integrated in silico pipeline combining machine learning–based quantitative structure–activity relationship modeling, graph neural network–driven drug–target affinity prediction, molecular docking, molecular dynamics (MD) simulations, and pharmacokinetic–toxicity profiling was employed to identify potential antitubercular leads from natural products. Results: A curated library of over 0.69 million compounds from the COCONUT database was systematically screened against seven essential M. tuberculosis protein targets. Machine learning and heterogeneous graph neural network models effectively captured complex ligand–protein interaction patterns, enabling high-confidence multitarget prioritization. Structure-based docking and MM-GBSA analyses revealed favorable binding affinities, further supported by 100 ns Molecular Dynamics simulations demonstrating stable binding and conformational integrity. In silico ADMET and toxicity predictions identified pharmacokinetically balanced candidates, while density functional theory calculations corroborated favorable electronic properties. Conclusions: Notably, a myricetin-based flavonoid glycoside exhibited consistent multitarget binding and dynamic stability across all targets. Overall, this study underscores the potential of integrated artificial intelligence and structure-based approaches in accelerating natural product-based antitubercular drug discovery and supports further experimental validation of prioritized leads.
Full article

Graphical abstract
Open AccessReview
Experimental Design in Pharmaceutical Formulation Development: Achievements, Limitations and the Transition Toward Intelligent Optimization
by
Ayşe Türkdoğan, Tarek Alloush and Burcu Demiralp
Sci. Pharm. 2026, 94(2), 38; https://doi.org/10.3390/scipharm94020038 - 13 May 2026
Abstract
►▼
Show Figures
Historically, pharmaceutical formulation development relied heavily on trial-and-error experimentation, which was useful for empirical progress but often provided limited mechanistic understanding and insufficient efficiency for increasingly complex drug products. The introduction of Design of Experiments (DoE) and Quality by Design (QbD) established a
[...] Read more.
Historically, pharmaceutical formulation development relied heavily on trial-and-error experimentation, which was useful for empirical progress but often provided limited mechanistic understanding and insufficient efficiency for increasingly complex drug products. The introduction of Design of Experiments (DoE) and Quality by Design (QbD) established a more systematic framework for studying formulation variables, manufacturing parameters, and Critical Quality Attributes (CQAs). Approaches such as factorial designs, response-surface methodology, and mixture designs have therefore become central to modern pharmaceutical development because they improve experimental efficiency and support the definition of design space. However, as formulations become more nonlinear, high-dimensional, and multi-objective, these classical approaches may no longer be sufficient on their own. This review examines the evolution of experimental design in pharmaceutical research, from one-factor-at-a-time experimentation to structured DoE/QbD strategies, and then to emerging intelligent optimization methods. Its central objective is to clarify when conventional DoE/QbD remains appropriate and when it should be complemented by machine learning, Bayesian optimization, digital twins, and closed-loop experimental systems. The review first summarizes the foundations and strengths of classical experimental design; then, it discusses its practical limitations in complex formulation settings, and finally evaluates how data-driven and hybrid approaches can extend pharmaceutical development. Evidence from tablets, capsules, nanocarriers, transdermal patches, and biotherapeutic systems suggests that intelligent optimization can improve predictive performance and experimental efficiency when used alongside, rather than instead of, established pharmaceutical development principles.
Full article
Figure 1
Open AccessArticle
Design, Synthesis and Biological Activity of Regioisomeric 3,5-Disubstituted Isoxazoles and 5-(Hydroxy)Isoxazolines with Aryl and Either (Diterpenylfuran-2-Carbonyl) or (Methylfuran-2-Carbonyl) Moiety
by
Maksim E. Mironov, Dmitry S. Baev, Mohammad S. Hamad, Sergey A. Borisov, Vyacheslav I. Krasnov, Tatyana V. Rybalova, Maksim P. Pitukhin, Irina V. Sorokina, Tatyana G. Tolstikova, Andrey G. Pokrovsky, Anastasia I. Poltanovich and Elvira E. Shults
Sci. Pharm. 2026, 94(2), 37; https://doi.org/10.3390/scipharm94020037 - 12 May 2026
Abstract
►▼
Show Figures
Alkyn-1,2-diones have gained great attention as useful building blocks in organic synthesis. Regioselective synthetic routes towards 3,5-disubstituted isoxazoles, containing the methylfuroyl or diterpenylfuroyl moiety at the C-3 or C-5 position from alkyne-1,2-diones 1, 2, 3, are reported. The reaction with
[...] Read more.
Alkyn-1,2-diones have gained great attention as useful building blocks in organic synthesis. Regioselective synthetic routes towards 3,5-disubstituted isoxazoles, containing the methylfuroyl or diterpenylfuroyl moiety at the C-3 or C-5 position from alkyne-1,2-diones 1, 2, 3, are reported. The reaction with hydroxylamine hydrochloride 6 in ethanol afforded the 1,2-addition products: 5-aryl-3-(methylfuran-2-carbonyl)isoxazoles (yield 61–94%) or 16-(5-arylisoxazole-3-carbonyl)labdatrienes (yield 48–97%). The reaction of alkynediones 1–3 with 6 in THF in the presence of triethylamine led to 5-hydroxy-4,5-dihydroisoxazoles and subsequent dehydration afforded regioisomeric 3-aryl-5-(methylfuran-2-carbonyl)isoxazoles or 16-(3-arylisoxazole-5-carbonyl)labda-trienes (yield 65–98%). New heterocyclic compounds exhibited significant analgesic action in acetic acid writhing and hot-plate tests, and the activity was comparable to reference drugs diclofenac sodium and celecoxib. Isoxazoles, which possessed the most analgesic activity, reduced the concanavalin A-induced inflammation by 34–51%; the effect was comparable to the drug indomethacin. The results of in vitro biological assays (MTT test) revealed that isoxazoles were non-toxic against the normal epithelial VERO cells, and 16-(3-aryl-5-hydroxyisoxazoline-5-carbonyl)labdatrienes 20–24 exhibited selective cytotoxicity against the breast adenocarcinoma MCF 7 (GI50 = 4.7–8.3 μM) and cervical cancer cells C33 A (GI50 = 3.4–4.7 μM). Molecular docking analysis to determine the binding potential of new molecules to the active site of human COX-1 and COX-2 enzymes was conducted.
Full article

Graphical abstract
Open AccessArticle
Investigation of the Efficacy of Qin Pi Extract in Alleviating Dry Eye Disease in Murine Models and Its Association with Suppression of Lymphangiogenesis
by
Feiyun Wang, Jing Hao, Mengjie Li, Yuying Zhu and Jiange Zhang
Sci. Pharm. 2026, 94(2), 36; https://doi.org/10.3390/scipharm94020036 - 5 May 2026
Abstract
►▼
Show Figures
Qin Bing eye drops, a traditional Chinese medicine-based in-hospital preparation, were historically indicated for the treatment of conjunctivitis, keratitis, and photokeratitis. This study aimed to develop Qin Pi extract (QP-E) using a proprietary extraction method, and to evaluate the therapeutic efficacy of QP-E
[...] Read more.
Qin Bing eye drops, a traditional Chinese medicine-based in-hospital preparation, were historically indicated for the treatment of conjunctivitis, keratitis, and photokeratitis. This study aimed to develop Qin Pi extract (QP-E) using a proprietary extraction method, and to evaluate the therapeutic efficacy of QP-E alone, QP-E combined with Bing Pian (BP), and an ophthalmic formulation (QP-D) comprising both constituents in a preclinical model of dry eye disease (DED). DED was induced in mice via subcutaneous scopolamine administration alone, whereas a more robust dry eye phenotype was established in rats through combined treatment with scopolamine and environmental stressors. Ocular surface evaluation included measurement of tear secretion volume and corneal fluorescein staining scores. The results demonstrated that both QP-E monotherapy and the QP-E–BP combination significantly ameliorated key pathological features of DED, including tear film instability and corneal epithelial damage. QP-D—formulated with rationally optimized concentrations of QP-E and BP—significantly enhanced basal tear secretion and attenuated corneal epithelial injury in both murine and rat dry eye models. Mechanistic investigations revealed that QP-E treatment markedly inhibited VEGF-C secretion from classically activated (M1) macrophages, suppressed phosphorylation-dependent activation of the VEGF-C/VEGFR-3 signaling axis, and consequently impaired lymphatic endothelial cell migration and in vitro tube formation. These correlative findings indicate that QP-E may partially alleviate DED by suppressing lymphangiogenesis; however, direct causal evidence—such as genetic ablation of VEGF-C or pharmacological inhibition of VEGFR-3—was not established in the present study. Collectively, our data yield a testable mechanistic hypothesis and propose a novel therapeutic strategy targeting lymphatic remodeling for DED intervention.
Full article
Figure 1
Open AccessCase Report
Personalized Dosage System as a Tool for Detecting Adverse Drug Reactions: Pharmaceutical Intervention in a Polymedicated Patient
by
Adama Peña-Vera, Sandra Dévora-Gutiérrez, Chaxiraxi Morales Marrero, Isabel V. Figueiredo and Susana Abdala Kuri
Sci. Pharm. 2026, 94(2), 35; https://doi.org/10.3390/scipharm94020035 - 30 Apr 2026
Abstract
►▼
Show Figures
Adverse Drug Reactions (ADRs) represent a major public health concern due to their impact on patient safety. In Spain, the Spanish Agency of Medicines and Medical Devices, through the FEDRA database, coordinates the reporting of suspected ADRs under real-world conditions of use, contributing
[...] Read more.
Adverse Drug Reactions (ADRs) represent a major public health concern due to their impact on patient safety. In Spain, the Spanish Agency of Medicines and Medical Devices, through the FEDRA database, coordinates the reporting of suspected ADRs under real-world conditions of use, contributing to the continuous updating of safety information. In this context, community pharmacist, through Professional Pharmaceutical Care Services, plays a key role in the early detection of ADRs and Drug-Related Problems (DRPs). This article describes the case of a 70-year-old polymedicated woman included in a Personalized Dosage System (PDS). Following the substitution of diazepam with clonazepam and an increase in the dose of semaglutide, the patient developed urinary incontinence, nausea and abdominal pain. Coordinated intervention between the community pharmacy and primary care enabled adjustment of the clonazepam dose, optimization of semaglutide administration and discontinuation of unnecessary naproxen use. These measures resulted in improved treatment tolerance and safety, as well as optimization of pharmacotherapy.
Full article

Figure 1
Open AccessArticle
Leveraging ChatGPT for Vancomycin Therapeutic Drug Monitoring: Simulation Using Bayesian Estimation and Hyperparameter Optimization
by
Akira Kageyama, Takahiko Aoyama, Rikuya Maehara, Dai Harada, Takashi Kawakubo and Yasuhiro Tsuji
Sci. Pharm. 2026, 94(2), 34; https://doi.org/10.3390/scipharm94020034 - 29 Apr 2026
Abstract
►▼
Show Figures
The usefulness of ChatGPT, a large language model, has recently been explored in medical research. However, no studies have examined its reproducibility or applicability to therapeutic drug monitoring (TDM), a core task of clinical pharmacists. In this simulation study, we evaluated the feasibility
[...] Read more.
The usefulness of ChatGPT, a large language model, has recently been explored in medical research. However, no studies have examined its reproducibility or applicability to therapeutic drug monitoring (TDM), a core task of clinical pharmacists. In this simulation study, we evaluated the feasibility of using ChatGPT for vancomycin (VCM) TDM based on Bayesian estimation. A total of 1000 virtual patients were generated by Monte Carlo simulations using a population pharmacokinetic model of VCM. Bayesian-estimated pharmacokinetic parameters and predicted concentrations were input into ChatGPT, and dosage regimens were compared among the three conditions, using temperature as a hyperparameter (T = 0.1, 0.5, and 1.0). Reproducibility was evaluated using the mode percentage in repeated runs. The reproducibility of the ChatGPT output was higher at T = 0.1 than at T = 0.5 and T = 1.0. When ChatGPT simulated the mode-recommended regimen (T = 0.1), the target attainment rate of the area under the serum concentration (AUC) (400–600 mg·h/L) improved from 25.5% (pre-optimization AUC (fixed-dose regimen)) to 71.5% (post-optimization AUC (ChatGPT-guided regimen)). These findings demonstrate that ChatGPT-based TDM using Bayesian estimation can enhance dose optimization. Adjusting the hyperparameter temperature to 0.1 improved reproducibility, suggesting that a reliable ChatGPT-assisted TDM support system may be clinically useful.
Full article

Figure 1
Open AccessArticle
An Integrated In Vitro and In Silico Approach Demonstrates Promising Anticancer Potential of Novel Cyclopenta[d]pyrimidine Derivatives
by
Valmik Sopan Aware, Shreya Rajesh Rao, Sanjay Pundalik Khairnar, Arati Prabhu, Hetal Abhay Shah and Sonal M. Manohar
Sci. Pharm. 2026, 94(2), 33; https://doi.org/10.3390/scipharm94020033 - 29 Apr 2026
Abstract
Background: Cancer is a leading cause of mortality worldwide. Discovery of small molecules as anticancer agents is an active area of research, as these molecules possess the remarkable ability to interact with specific targets within cancer cells. Objectives: In vitro anticancer activity of
[...] Read more.
Background: Cancer is a leading cause of mortality worldwide. Discovery of small molecules as anticancer agents is an active area of research, as these molecules possess the remarkable ability to interact with specific targets within cancer cells. Objectives: In vitro anticancer activity of six hit derivatives from a series of 2-phenyl-substituted 4-amino–6, 7-dihydro-5H-cyclopenta[d]pyrimidines was tested against human cancer cell lines, viz., A549 (human lung cancer) and A431 (human skin cancer). Methods: Cytotoxicity was evaluated for six hits by the standard MTT assay. Further, their effect on clonogenic potential and cell cycle was tested using colony forming assay and flow cytometric analysis, respectively. Apoptosis-inducing potential was confirmed using Caspase-3/7 Glo assay and detection of cleaved caspase-3 by immunofluorescence. The effect on cell migration was tested using a wound healing assay. Target analysis, Molecular docking and ADMET simulations were performed to identify molecular targets, interactions and assess pharmacokinetic profiles. Results: Specific derivatives showed good to moderate cytotoxicity against A549 and A431 (with average IC50 in the range of ~30 µM), and these hits led to apoptosis and G1 arrest in these cell lines, respectively. Furthermore, identified hits inhibited cell migration in A549 cells. Computational consensus target analysis identified EGFR and CDK2 as high-confidence targets. Docking studies indicated favorable interactions and stability, whereas the ADMET analysis confirmed the drug-likeness and optimal pharmacokinetic and safety profiles of the small molecules. Conclusions: Our current study demonstrates the anticancer potential of novel pyrimidine derivatives. We envisage the use of these small molecules as promising anticancer agents, particularly in skin and non-small cell lung cancer.
Full article
(This article belongs to the Special Issue Pharmaceutical Applications of Heterocyclic Compounds)
►▼
Show Figures

Figure 1
Open AccessArticle
Analytical Method Optimization, Evaluation and Characterization of Ivermectin Solid Dispersion Formulations Using Hydrophilic Carriers
by
Sajib Chandra Roy, Md. Jahid Hossain, Uttom Kumar, Sreedam Chandra Das, Fatema Moni, Deepankar Sutradhar, Faria Tasneem and A. S. M. Monjur Al Hossain
Sci. Pharm. 2026, 94(2), 32; https://doi.org/10.3390/scipharm94020032 - 22 Apr 2026
Abstract
►▼
Show Figures
Improving the dissolution and solubility of poorly water-soluble drugs remains a major challenge in drug development. Solid dispersion (SD) techniques offer an effective strategy by which to enhance the bioavailability of BCS Class II drugs such as ivermectin (IVM). This study aimed to
[...] Read more.
Improving the dissolution and solubility of poorly water-soluble drugs remains a major challenge in drug development. Solid dispersion (SD) techniques offer an effective strategy by which to enhance the bioavailability of BCS Class II drugs such as ivermectin (IVM). This study aimed to develop and validate stability-indicating analytical methods for the quantification of IVM and to evaluate the performance of the formulated SDs. A novel RP-HPLC and a UV spectrophotometric method were developed and validated in accordance with ICH guidelines. IVM SDs were prepared via physical mixing (PM), solvent evaporation (SE), and melt fusion (MF) using Poloxamer 188, Kollicoat® IR, and PEG 6000 at respective ratios of 1:1, 1:3, and 1:5. Dissolution studies showed a marked enhancement in drug release from SDs prepared by SE and MF methods compared with pure IVM. Among all formulations, the Poloxamer 188-based binary SD prepared by the SE method at a 1:5 ratio exhibited the highest dissolution (98.55% at 60 min), with release kinetics following anomalous (non-Fickian) transport (n = 0.681) according to the Korsmeyer–Peppas model. Solid-state characterization evidenced by FTIR, DSC, TGA, and SEM confirmed the transformation of IVM from its crystalline form to an amorphous state. Future studies will focus on the in vivo evaluation of the optimized IVM SD formulations.
Full article

Figure 1
Open AccessArticle
Development and Characterization of Melatonin-Loaded Glycerol–Gelatin-Based Vaginal Suppositories for Localized Delivery
by
Regina Julieta Delgadillo Hernández, Gregorio Guadalupe Carbajal Arízaga, José Alfonso Cruz Ramos, Rodolfo Hernández Gutiérrez, José Armando Hernández Díaz, Ana Alejandra Arias García, Norma Morales-Hernández, José Nabor Haro-González, Zaira Yunuen García Carvajal and Moisés Martínez Velázquez
Sci. Pharm. 2026, 94(2), 31; https://doi.org/10.3390/scipharm94020031 - 17 Apr 2026
Abstract
This research aimed to develop glycerol–gelatin vaginal suppositories loaded with melatonin to enhance the localized effects of antineoplastic agents. The solubility of melatonin in different solvents was determined, and glycofurol, which is approved for pharmaceutical use, presented the highest solubilizing capacity. Furthermore, the
[...] Read more.
This research aimed to develop glycerol–gelatin vaginal suppositories loaded with melatonin to enhance the localized effects of antineoplastic agents. The solubility of melatonin in different solvents was determined, and glycofurol, which is approved for pharmaceutical use, presented the highest solubilizing capacity. Furthermore, the cytotoxicity of melatonin incorporated into suppositories against HeLa cells was evaluated using MTT assays, individually and in combination with cisplatin. The results indicate that melatonin enhances the cytotoxic effects of cisplatin. The optimal formulation obtained from an experimental design was 33% gelatin, 1% PVA, 1% PEG 6000, 10% glycerol, 15% glycofurol, and 40% water. To ensure that the vaginal suppositories presented the necessary physical properties for optimal handling and application, tests were performed to determine weight uniformity, texture, surface features and disintegration time. Vaginal suppositories weighted around 1.43 g, showed Young’s modulus values of 7389.6 N/m2 and hardness around 1100 gf, and they disintegrated after 30 min at pH 4.2. Additionally, for in vitro melatonin release, FTIR and XRD tests confirmed the presence of melatonin in the formulation. It is concluded that the developed vaginal suppositories can be explored as potential vehicles for localized delivery of melatonin to the tumor site to enhance therapeutic outcomes.
Full article
(This article belongs to the Topic Complementary Strategies in Drug Delivery: From Particle Engineering to System Optimization)
►▼
Show Figures

Figure 1
Open AccessArticle
In Silico ADMET Profiling and Drug-Likeness Evaluation of Novel Thiopyrano[2,3-d]thiazole Derivatives as Potential Anticonvulsants
by
Maryna Stasevych, Mykhailo Hoidyk, Viktor Zvarych, Andriy Karkhut, Svyatoslav Polovkovych and Roman Lesyk
Sci. Pharm. 2026, 94(2), 30; https://doi.org/10.3390/scipharm94020030 - 9 Apr 2026
Abstract
►▼
Show Figures
The development of novel antiepileptic agents requires early identification of pharmacokinetic limitations to mitigate risks at later stages. This study aimed to perform in silico profiling of a library containing 448 novel 2H,5H-chromeno[4’,3’:4,5]thiopyrano[2,3-d]thiazol-2-one derivatives to select lead
[...] Read more.
The development of novel antiepileptic agents requires early identification of pharmacokinetic limitations to mitigate risks at later stages. This study aimed to perform in silico profiling of a library containing 448 novel 2H,5H-chromeno[4’,3’:4,5]thiopyrano[2,3-d]thiazol-2-one derivatives to select lead compounds with an optimal balance of safety and efficacy. The study was conducted using the ADMET-AI platform, based on a graph neural network, to predict physicochemical, pharmacokinetic, and toxicological properties. The methodology involved calculating drug-likeness descriptors for primary screening and a comparative statistical analysis of the top 20 selected structures against 16 approved antiepileptic drugs and four reference compounds. Based on drug-likeness descriptors and predicted ADMET (absorption, distribution, metabolism, excretion, toxicity) related parameters, 20 structures were prioritized for further analysis. Their predicted profiles suggested high intestinal absorption and blood–brain barrier (BBB) permeability, which may be relevant for central nervous system (CNS) directed agents. In comparison with the reference thiazolidinones, the prioritized compounds showed comparatively more favorable predicted mutagenicity and carcinogenicity profiles. Elevated predicted risks of hepatotoxicity and cardiotoxicity were observed for several structures, indicating the need for further structural optimization. The results suggest that the thiopyranothiazolidinone scaffold merits further anticonvulsant-oriented investigation at the stage of early compound prioritization. Experimental validation will be required to confirm the actual pharmacokinetic, toxicological, and anticonvulsant properties of the prioritized compounds.
Full article

Graphical abstract
Open AccessReview
Three Decades of Taxanes: Exploring the Next Frontier
by
Rita I. L. Catarino, Maria Fernanda C. Leal, Adriana M. Pimenta, Maria Renata S. Souto and Francisco A. M. Silva
Sci. Pharm. 2026, 94(2), 29; https://doi.org/10.3390/scipharm94020029 - 8 Apr 2026
Abstract
►▼
Show Figures
Taxanes, such as paclitaxel and docetaxel, are microtubule-stabilizing agents widely used in oncology, either as monotherapy or in combination regimens. While highly effective, these first-generation taxanes face important limitations, including significant toxicity, reduced water solubility, and the emergence of multidrug resistance. To address
[...] Read more.
Taxanes, such as paclitaxel and docetaxel, are microtubule-stabilizing agents widely used in oncology, either as monotherapy or in combination regimens. While highly effective, these first-generation taxanes face important limitations, including significant toxicity, reduced water solubility, and the emergence of multidrug resistance. To address these challenges, semi-synthetic taxoids have been developed, aiming to improve pharmacological profiles and overcome therapeutic barriers. Central to these efforts is the understanding of structure-activity relationships, which guides the rational design of taxane analogues with enhanced efficacy and safety. This review explores recent advances in taxoid development, highlights findings from clinical trials, and evaluates how these new agents compare with traditional taxanes in terms of therapeutic potential and tolerability. While novel delivery systems offer improved outcomes with existing drugs, the development of new taxane analogues remains a promising approach to address drug resistance, albeit with challenges related to toxicity, high costs, and historically low success rates in drug development. Furthermore, taxanes are already used in certain cardiovascular conditions and show emerging potential in neurodegenerative diseases, although current evidence remains largely limited to preclinical or early-phase clinical studies. These developments mark an important evolution in the field and offer new opportunities for future therapeutic strategies.
Full article

Figure 1
Open AccessReview
Orodispersible Tablets for Paediatric Use: A Systematic Review and Outlook for Future Research
by
Samia Farhaj, Omar Hamid, Noman Ahmad, Barbara R. Conway and Muhammad Usman Ghori
Sci. Pharm. 2026, 94(2), 28; https://doi.org/10.3390/scipharm94020028 - 5 Apr 2026
Abstract
►▼
Show Figures
Children are often underserved by adult-oriented oral medicines, leading to off-label use and dosage-form manipulation that may compromise dosing accuracy. This review summarises recent advances in paediatric orodispersible tablets (ODTs), focusing on manufacturing technologies, superdisintegrants, taste masking, and in vitro disintegration testing. Following
[...] Read more.
Children are often underserved by adult-oriented oral medicines, leading to off-label use and dosage-form manipulation that may compromise dosing accuracy. This review summarises recent advances in paediatric orodispersible tablets (ODTs), focusing on manufacturing technologies, superdisintegrants, taste masking, and in vitro disintegration testing. Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidance and a protocol registered with the International Platform of Registered Systematic Review and Meta-analysis Protocols (registration number INPLASY2025110022), we searched PubMed, EMBASE, MEDLINE, Scopus, and Google Scholar for experimental studies on paediatric-relevant ODT formulation and evaluation. Two reviewers screened studies and extracted data on manufacturing methods, excipients, disintegration/dissolution testing, and key outcomes. Risk of bias was assessed using a six-domain framework. Overall, 65 studies met the inclusion criteria for this review. Direct compression was the dominant method, with freeze-drying, sublimation, spray-drying, nanoparticle-in-tablet systems, and semi-solid extrusion/3D printing also reported. Crospovidone, croscarmellose sodium, and sodium starch glycolate were the most common superdisintegrants, while natural and co-processed disintegrants showed promise as cost-effective alternatives. Disintegration was usually assessed using pharmacopoeial methods, with some modified set-ups to better simulate oral conditions. Paediatric ODT development is advancing rapidly. Broader translation requires harmonised disintegration testing, age-stratified acceptability reporting, and GMP-ready workflows, alongside benchmarking of superdisintegrants and attention to dose flexibility, packaging, and affordability.
Full article

Figure 1
Open AccessReview
Kalanchoe daigremontiana from Ornamental to Pharmaceutical Applications
by
Cecilia Guadalupe de Loza-García, Ana Belem Rubio-García, Salvador Hernández-Estrada, Luis Alfonso Hernández-Villaseñor, Luis Antonio Ramirez-Contreras, Jorge Manuel Silva-Jara, Jorge L. Mejía-Méndez, Zuamí Villagrán, Eugenio Sánchez-Arreola, Napoleón González-Silva and Luis Miguel Anaya-Esparza
Sci. Pharm. 2026, 94(2), 27; https://doi.org/10.3390/scipharm94020027 - 31 Mar 2026
Abstract
►▼
Show Figures
Kalanchoe daigremontiana, a succulent herbaceous plant in the Crassulaceae family from Madagascar, has gained global popularity as an ornamental and medicinal species. This review examines the traditional uses, phytochemical composition, biological properties, toxicological aspects, and regulatory challenges of K. daigremontiana. The
[...] Read more.
Kalanchoe daigremontiana, a succulent herbaceous plant in the Crassulaceae family from Madagascar, has gained global popularity as an ornamental and medicinal species. This review examines the traditional uses, phytochemical composition, biological properties, toxicological aspects, and regulatory challenges of K. daigremontiana. The traditional medicinal uses of its leaves and roots include treating burns, rheumatic disorders, hypertension, diabetes, kidney pain, diarrhea, cough, fever, gastric issues, anxiety, inflammation, and cancer. Chemical compounds identified include phenolic acids, flavonoids, tannins, alkaloids, glycosides, saponins, sterols, terpenes, and fatty acids, with phenolic compounds and bufadienolides being predominant. In vitro studies of the crude extracts, bufadienolide-rich fractions, and isolated compounds have shown antioxidant, antibacterial, antifungal, antiviral, antiparasitic, anthelmintic, anti-inflammatory, anticoagulant, anti-aging, cytotoxic, antitumoral, and antiproliferative properties. In vivo studies have demonstrated hepatoprotective, skincare, and cardiac-glycoside-like effects. While crude extracts and bufadienolide-rich fractions have shown toxic effects in 2-week-old chicks, guinea pigs, and Artemia salina, no toxicity has been reported in goats, broiler chickens, laying hens, or human erythrocytes. Although K. daigremontiana-based products are commercially available as dietary supplements with various health claims, these lack scientific validation. Despite the potential pharmaceutical applications of K. daigremontiana, further research is needed to determine its effects, dosage, mechanisms, long-term safety, and side effects, with clinical studies essential to validate its therapeutic potential.
Full article

Figure 1

Journal Menu
► ▼ Journal MenuJournal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, IJMS, Sci. Pharm., Molecules, Future Pharmacology, Biomolecules
Natural Products and Drug Discovery—2nd Edition
Topic Editors: Sonia Piacente, Marta MenegazziDeadline: 30 September 2026
Topic in
Bioengineering, Molecules, Pharmaceuticals, Pharmaceutics, Processes, Sci. Pharm.
Complementary Strategies in Drug Delivery: From Particle Engineering to System Optimization
Topic Editors: Barbara R. Conway, Hisham Al-ObaidiDeadline: 30 October 2026
Topic in
Antibiotics, CIMB, IJMS, Pharmaceutics, Sci. Pharm., SynBio, Pharmaceuticals
Design, Synthesis, and Development of Antimicrobial Drugs
Topic Editors: Hao Wang, Wenxuan ZhangDeadline: 30 November 2026
Topic in
Biomedicines, IJMS, Medicines, Molecules, Pharmaceuticals, Sci. Pharm.
New Compounds Discovery and Development in Medicine — Advances in Research on Potential Therapeutic Agents and Drug Candidates, 2nd Edition
Topic Editors: Monika Wujec, Anna Bogucka-Kocka, Przemysław Kołodziej, Jacek BoguckiDeadline: 31 December 2026

Special Issues
Special Issue in
Sci. Pharm.
Novel Bioanalytical Approaches in Therapeutic Drug Monitoring and Environmental Health
Guest Editor: Linden RafaelDeadline: 30 June 2026
Special Issue in
Sci. Pharm.
Monoclonal Antibodies in the Treatment of Diseases: Focus on Stability, Storage, and Administration
Guest Editor: Stefano RugaDeadline: 31 October 2026
Special Issue in
Sci. Pharm.
Future Trends in Nano Drug Delivery
Guest Editors: Marta Slavkova, Sebastian Schwaminger, Borislav S. TzankovDeadline: 31 October 2026
Special Issue in
Sci. Pharm.
Pharmaceutical Applications of Heterocyclic Compounds
Guest Editor: Thierry BessonDeadline: 31 October 2026







