Journal Description
Pharmacoepidemiology
Pharmacoepidemiology
is an international, peer-reviewed, open access journal on high-quality epidemiological, clinical research across the fields of clinical pharmacology and epidemiology, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 21.3 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Pharmacoepidemiology is a companion journal of Pharmaceuticals and Journal of Clinical Medicine.
- Journal Clusters-Pharmaceutical Science: Scientia Pharmaceutica, Marine Drugs, Pharmaceuticals, Pharmaceutics, Pharmacy, Future Pharmacology, Pharmacoepidemiology, Drugs and Drug Candidates and Journal of Pharmaceutical and BioTech Industry.
Latest Articles
Adjunctive Vortioxetine in Major Depressive Disorder with Inadequate Response to Antidepressants: A Prospective Real-World Pilot Study from Malaysia
Pharmacoepidemiology 2026, 5(2), 14; https://doi.org/10.3390/pharma5020014 - 20 May 2026
Abstract
►
Show Figures
Background: A significant percentage of patients with major depressive disorder (MDD) fail to achieve remission with antidepressant monotherapy and frequently experience residual mood and cognitive symptoms that impair their functional recovery. Thus, an augmentation with vortioxetine, a multimodal antidepressant with reported cognitive
[...] Read more.
Background: A significant percentage of patients with major depressive disorder (MDD) fail to achieve remission with antidepressant monotherapy and frequently experience residual mood and cognitive symptoms that impair their functional recovery. Thus, an augmentation with vortioxetine, a multimodal antidepressant with reported cognitive benefits, might be a useful strategy for such patients. Methods: We conducted a 12-week naturalistic, prospective observational study in a Malaysian university hospital; 40 adults with MDD and inadequate response to at least eight weeks of antidepressant therapy received either adjunctive vortioxetine or optimization of their existing antidepressant as part of treatment-as-usual care. Depressive symptoms were assessed using the Montgomery–Åsberg Depression Rating Scale (MADRS), cognitive symptoms using the Perceived Deficits Questionnaire-5 (PDQ-D5), and global improvement using the Clinical Global Impressions—Improvement (CGI-I) scale. Results: Both groups demonstrated significant improvements in MADRS and PDQ-D5 scores over 12 weeks (p < 0.001). Remission rates at Week 12 were high in both groups (93.8% adjunctive vortioxetine vs. 86.7% control). Both groups demonstrated significant improvements in depressive and cognitive symptoms over 12 weeks. Although between-group differences were not statistically significant, descriptive trends toward earlier symptomatic improvement were observed in the adjunctive vortioxetine group in several core depressive symptoms, including apparent sadness, suicidal ideation, and appetite disturbance. Greater clinician-rated global improvement was observed in the vortioxetine group at Week 12 (87.5% vs. 40.0%, p < 0.001). Conclusions: In this outpatient clinical setting, adjunctive vortioxetine was associated with earlier improvement of core depressive symptoms and greater global clinical improvement compared with optimization of existing antidepressant therapy. Collectively, these findings suggest adjunctive vortioxetine as a clinically relevant option for patients with MDD who show an inadequate response to antidepressant monotherapy; however, findings are exploratory and not causal, and thus larger RCTs are needed for affirmation.
Full article
Open AccessArticle
Household Pharmaceutical Accumulation in Southeastern Mexico: A Multidimensional Pharmacoepidemiological Risk Assessment Framework
by
Rafael Manuel de Jesús Mex-Álvarez, María Magali Guillen-Morales, Patricia Garma-Quen, David Yanez-Nava, Diana Andrea Luna-Salazar and Roger Enrique Chan-Martínez
Pharmacoepidemiology 2026, 5(2), 13; https://doi.org/10.3390/pharma5020013 - 29 Apr 2026
Abstract
►▼
Show Figures
Background/Objectives: The accumulation of unused and expired pharmaceuticals in households is a growing public health concern with implications for patient safety, rational drug use, and environmental health. However, systematic risk characterization integrating clinical and environmental perspectives at the community level remains limited,
[...] Read more.
Background/Objectives: The accumulation of unused and expired pharmaceuticals in households is a growing public health concern with implications for patient safety, rational drug use, and environmental health. However, systematic risk characterization integrating clinical and environmental perspectives at the community level remains limited, particularly in low- and middle-income settings. This study aimed to develop and apply a composite risk index, grounded in an eco-pharmacovigilance framework, for the assessment of health risks associated with accumulated household pharmaceuticals in southeastern Mexico. Methods: A cross-sectional study was conducted in 526 randomly selected households using stratified sampling. Guided in-home medication inventories were performed with participant collaboration, and pharmaceuticals were classified according to the Anatomical Therapeutic Chemical (ATC) system. A composite risk index (CRI = Fr × PR) was developed within an eco-pharmacovigilance framework. The frequency of accumulation (Fr) for each therapeutic group was multiplied by a potential risk score (PR) derived through a structured multidisciplinary expert consensus process integrating clinical toxicity, environmental persistence, and antimicrobial resistance potential. Results: A total of 2184 pharmaceutical units were recorded during the household inventories, of which 28.7% were expired. Expired medications were primarily retained rather than actively used, representing a latent risk for inappropriate self-medication and accidental exposure. The therapeutic groups with the highest CRI values were antihypertensives (CRI = 42.3), antidiabetics (CRI = 37.8), and antibiotics (CRI = 31.5), indicating a relatively higher contribution within the composite risk index framework to overall household pharmaceutical risk. These findings highlight priority therapeutic groups driven by the combined effect of high accumulation frequency, distinct accumulation patterns, and intrinsic hazard. Conclusions: Household pharmaceutical accumulation can be characterized using a composite, eco-pharmacovigilance-based approach that integrates exposure and hazard dimensions. The proposed framework functions as a prioritization tool rather than a precise quantitative measure, enabling the identification of therapeutic groups requiring targeted intervention. Findings should be interpreted as indicative of relative risk patterns rather than precise estimates, given the exploratory design and guided data collection approach. The proposed framework provides a practical tool for prioritizing interventions aimed at improving rational drug use, reducing accumulation, and mitigating environmental impact. Further validation in diverse settings is warranted to strengthen its applicability.
Full article

Figure 1
Open AccessReview
High Tibial Osteotomy (HTO) Versus Unicompartmental Knee Arthroplasty (UKA) in Medial-Compartment Knee Osteoarthritis (KOA): A Critical Narrative Review of Comparative Costs and Cost-Effectiveness
by
Furkan Yapıcı
Pharmacoepidemiology 2026, 5(2), 12; https://doi.org/10.3390/pharma5020012 - 29 Apr 2026
Abstract
►▼
Show Figures
Background: Medial-compartment knee osteoarthritis (KOA) carries substantial disability and long-term cost. High tibial osteotomy (HTO) and unicompartmental knee arthroplasty (UKA) are key joint-preserving or joint-replacing options for selected patients, but their comparative economic ranking remains uncertain. Methods: This critical narrative review
[...] Read more.
Background: Medial-compartment knee osteoarthritis (KOA) carries substantial disability and long-term cost. High tibial osteotomy (HTO) and unicompartmental knee arthroplasty (UKA) are key joint-preserving or joint-replacing options for selected patients, but their comparative economic ranking remains uncertain. Methods: This critical narrative review synthesized comparative economic evidence on HTO versus UKA for isolated medial-compartment KOA. PubMed and Web of Science were searched as primary sources for English-language studies published from 1 January 2000 to 15 January 2026, while Google Scholar and citation tracking were used supplementarily to identify potentially missed records. Eligible studies were direct economic evaluations or comparative cost/resource studies with clear decision relevance to the HTO–UKA choice. Burden and cost-of-illness studies were used for contextual framing only and were not included in the core comparative synthesis. Results: The direct evidence base was small and methodologically heterogeneous and was dominated by decision-analytic models that differed in perspective, time horizon, utility metric, and assumptions regarding reoperation, revision, and conversion to total knee arthroplasty (TKA). These structural differences largely explain why a U.S. lifetime societal model favored HTO, a UK age-stratified 10-year model produced age-dependent findings, and a recent Canadian public-payer model favored UKA. Observational studies suggest that UKA episode costs can fall substantially in outpatient or ambulatory pathways, whereas HTO costs may rise when reoperations and technique-specific resource use are explicitly captured. Conclusions: Current evidence does not support a context-free economic ranking of HTO and UKA. Because the available studies are heterogeneous and incremental utility differences are often small, the findings should be interpreted cautiously and as scenario-dependent rather than definitive. Future comparative analyses should use contemporary pathway data, transparent and standardized costing, and explicit downstream event definitions for both procedures.
Full article

Graphical abstract
Open AccessArticle
The Possible Relationship Between Adverse Drug Reactions and Potential Drug–Drug Interactions in Patients with NSCLC Treated with EGFR Inhibitors
by
Ivanka Mutafova, Evgeni Grigorov, Violeta Getova-Kolarova and Kaloyan D. Georgiev
Pharmacoepidemiology 2026, 5(2), 11; https://doi.org/10.3390/pharma5020011 - 26 Mar 2026
Abstract
►▼
Show Figures
Background: The introduction of targeted therapy in oncology has led to several challenges. These medicines are relatively new in clinical practice and are not well known to specialists with regard to adverse drug reactions (ADRs) and potential drug–drug interactions (pDDIs). In addition, cancer
[...] Read more.
Background: The introduction of targeted therapy in oncology has led to several challenges. These medicines are relatively new in clinical practice and are not well known to specialists with regard to adverse drug reactions (ADRs) and potential drug–drug interactions (pDDIs). In addition, cancer affects multiple body systems, including weight loss, anemia, liver and kidney function, depression, and pain. Patients frequently have comorbidities, leading to polypharmacy and the use of special foods, nutritional supplements, and herbal products for self-medication. Identification of pDDIs is essential, as concomitant use of multiple medicinal products increases the risk of ADRs and may compromise treatment. Objective: This study aims to retrospectively review and analyze data on ADRs and pDDIs in the treatment of non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) inhibitors and to evaluate the relationship between them. Method: EudraVigilance and UpToDate® Lexidrug™ application were used to screen suspected ADRs and pDDIs, respectively. Descriptive statistical analysis was performed. Results: After reviewing Line Listing Reports (LLRs) from 2021 to 2023 in EudraVigilance, the number of suspected adverse drug reactions (ADRs) reported was higher when drug interactions classified as risk categories D and X were identified, compared with cases involving EGFR inhibitor monotherapy or other drug combinations. Of the 144 cases involving category D and/or X interactions, 63 demonstrated a possible association with the reported ADRs of EGFR inhibitors. The most common pDDIs detected were erlotinib–ranitidine (14 cases, category D) and osimertinib–amiodarone (13 cases, category D). Conclusions: Although EGFR inhibitors improve overall and progression-free survival in NSCLC, screening for pDDIs before treatment is essential to improve safety and quality of life.
Full article

Figure 1
Open AccessArticle
Impact of Long-Term Statin Therapy on Influenza Incidence and Overall Mortality: A Real-World Data Analysis
by
Diana Toledo, Àurea Cartanyà-Hueso, Constança Pagès-Fernández, Rosa Morros, Maria Giner-Soriano, Àngela Domínguez, Carles Vilaplana-Carnerero, Alba Tor-Roca and María Grau
Pharmacoepidemiology 2026, 5(2), 10; https://doi.org/10.3390/pharma5020010 - 26 Mar 2026
Abstract
►▼
Show Figures
Background/Objectives: The study’s goal is to assess the association between long-term statin therapy and influenza incidence, influenza severity, and all-cause mortality. Methods: Two population-based dynamic cohorts (exposed and unexposed to statins) were followed from 2010 to 2019. Participants were 60 years
[...] Read more.
Background/Objectives: The study’s goal is to assess the association between long-term statin therapy and influenza incidence, influenza severity, and all-cause mortality. Methods: Two population-based dynamic cohorts (exposed and unexposed to statins) were followed from 2010 to 2019. Participants were 60 years or older; frail patients were excluded. The primary outcomes were influenza incidence, influenza-related intensive care unit (ICU) admission as a proxy for severity, and all-cause mortality. The exposed cohort comprised new statin users with a minimum of two pharmacy invoices within 90 days of enrollment. Adjusted risk ratios (aRRs) for influenza incidence, ICU admission, and mortality rate were calculated using Poisson regression. Results: The initial study population of 639,564 individuals was evenly split into exposed (319,782) and unexposed (319,782) cohorts; mean age was 71 years (standard deviation: 8 years), and 57% were women. Compared to non-users, new statin users showed a higher influenza incidence [9.39 (95% confidence interval: 9.36–9.42) vs. 7.64 (7.61–7.66) per 1000 person-years], ICU admission [1.65 (1.65–1.66) vs. 1.36 (1.35–1.36) per 1000 person-years], and overall mortality rate [97.09 (96.75–97.43) vs. 94.15 (93.82–94.47) per 1000 person-years]. Adjusted analysis revealed no significant association between statin use and influenza incidence [aRR: 1.04 (0.98–1.10)] or influenza-related ICU admission [aRR: 1.03 (0.89–1.19)] and shifted the effect on mortality from harmful to beneficial [aRR: 0.88 (0.87–0.89)]. Conclusions: Despite new users’ greater vulnerability at the start of treatment, our findings indicate that statins do not influence influenza incidence or severity but reduce all-cause mortality, warranting further exploration of their anti-inflammatory properties.
Full article

Figure 1
Open AccessArticle
Patterns of Furosemide Use and Associated Adverse Drug Events in Primary Healthcare Settings: A Retrospective Pharmacoepidemiological Study
by
Fitim B. Alidema, Lirim Shefki Mustafa, Arieta Hasani Alidema, Mirlinda Havolli and Fellenza Abazi
Pharmacoepidemiology 2026, 5(1), 9; https://doi.org/10.3390/pharma5010009 - 17 Mar 2026
Abstract
►▼
Show Figures
Background: Furosemide is one of the most frequently prescribed loop diuretics for cardiovascular conditions, particularly in the management of volume overload and acute elevations in blood pressure. However, detailed real-world data describing its utilization characteristics and documented safety outcomes in primary healthcare settings
[...] Read more.
Background: Furosemide is one of the most frequently prescribed loop diuretics for cardiovascular conditions, particularly in the management of volume overload and acute elevations in blood pressure. However, detailed real-world data describing its utilization characteristics and documented safety outcomes in primary healthcare settings remain limited, especially in underrepresented health systems. Objective: This study aimed to describe real-world patterns of furosemide utilization, including indications and concomitant treatment patterns, and to document associated adverse drug events and short-term clinical outcomes in routine primary healthcare practice. Methods: A retrospective pharmacoepidemiological observational study was conducted between January and December 2025 in a primary healthcare center. Medical records of 1300 adult patients who received furosemide for cardiovascular indications were reviewed. Indications included arterial hypertension, hypertensive crises, and conditions related to volume overload. Utilization characteristics were operationalized as indication distribution, monotherapy versus combination therapy, and recurrence patterns within the study period. Data collected included demographic characteristics, primary and comorbid diagnoses, blood pressure values recorded before and after administration, furosemide dose and route of administration, concomitant antihypertensive therapy, documented adverse drug events as recorded in routine clinical documentation, recurrent presentations related to hypertensive crises, and the need for hospital referral. Descriptive statistics and paired comparative analyses were performed, with statistical significance set at p < 0.05. Results: The mean patient age was 62.4 ± 11.8 years, with a male predominance (54.1%). Arterial hypertension was the most frequent recorded indication (78.6%), while 32.4% of patients had multiple cardiovascular diagnoses. A statistically significant reduction in systolic blood pressure (from 176.3 ± 18.5 mmHg to 148.7 ± 16.2 mmHg, p < 0.001) and diastolic blood pressure (from 101.2 ± 11.4 mmHg to 89.6 ± 9.8 mmHg, p < 0.001) was observed between measurements recorded before and after administration during the same clinical episode. Recurrent presentations related to hypertensive crises were documented in 27.9% of patients during the study period. Adverse drug events were documented in 9.6% of cases, most commonly dehydration and suspected electrolyte disturbances as noted in routine clinical records. Hospital referral was required in 6.8% of patients. Conclusions: In this real-world primary healthcare cohort, furosemide was commonly used across a heterogeneous mix of cardiovascular indications, predominantly in combination with other antihypertensive agents. Observed temporal reductions in blood pressure and documented adverse events reflect routine clinical practice rather than controlled treatment effects. These findings provide descriptive pharmacoepidemiological evidence from a primary care setting and underscore the importance of careful monitoring, documentation, and rational prescribing in patients receiving loop diuretics.
Full article

Figure 1
Open AccessArticle
Antibiotic Prescribing for Group B Streptococcus Coverage in Preterm Prelabour Rupture of Membranes: A Retrospective Cohort Study
by
Elaf Abuelgasim, Mark McIntyre and Najla Tabbara
Pharmacoepidemiology 2026, 5(1), 8; https://doi.org/10.3390/pharma5010008 - 7 Mar 2026
Abstract
Background: Preterm prelabour rupture of membranes (PPROM) is the spontaneous rupture of fetal membranes prior to 37 weeks of pregnancy. Latency antibiotics, including macrolides with or without group B streptococcus (GBS)-covering antibiotics, are recommended as part of expectant management. Currently, there is
[...] Read more.
Background: Preterm prelabour rupture of membranes (PPROM) is the spontaneous rupture of fetal membranes prior to 37 weeks of pregnancy. Latency antibiotics, including macrolides with or without group B streptococcus (GBS)-covering antibiotics, are recommended as part of expectant management. Currently, there is no consensus on whether GBS-covering antibiotics should be prescribed. The primary objective of this retrospective cohort study was to characterize practice variation in GBS-covering antibiotic prescribing in PPROM. The secondary objective was to explore the association between maternal characteristics and GBS-covering antibiotic prescribing. Methods: Pregnant women with PPROM prescribed azithromycin (institutional standard antibiotic regimen) in 2024 and not in active labour were included. Maternal characteristics, stratified by GBS status, were compared. The association between antibiotic prescribing for GBS coverage and maternal factors was assessed using odds ratios. Two-sided p-values < 0.05 were considered statistically significant. Results: Out of the 181 admissions assessed for eligibility, 146 patients were included. Their GBS status at PPROM diagnosis was negative (19/146; 13%), positive (8/146; 5%), or unknown (119/146; 82%). The frequency of GBS-covering antibiotics prescribing was 5/8 (63%) in the positive group, 4/19 (21%) in the negative group, and 65/119 (55%) in those with an unknown GBS status. Aminopenicillin-based and penicillin regimens accounted for (69/74; 93%) of antibiotic regimens. Half (38/74; 51%) of the GBS-covering antibiotics were prescribed for 3–7 days, with a 33/74 (45%) completion rate as prescribed at PPROM diagnosis. The main reason for antibiotic discontinuation was negative GBS recto-vaginal swabs or urine cultures collected in those with an unknown GBS status at PPROM diagnosis, highlighting the role of microbiology laboratory testing in adjusting antibiotic therapy and facilitating antimicrobial stewardship. Aside from GBS status, no maternal characteristics were associated with GBS-covering antibiotic prescribing. Conclusions: At PPROM diagnosis, GBS coverage was prescribed in 21%, 63%, and 55% of patients with a negative, positive, and unknown GBS status, respectively. Only GBS status was associated with GBS-covering antibiotic prescribing. Further research is required to determine the impact of GBS coverage on perinatal outcomes.
Full article
(This article belongs to the Special Issue Women’s Special Issue Series: Pharmacoepidemiology)
►▼
Show Figures

Figure 1
Open AccessArticle
Real-World Safety of Acalabrutinib in Mexico: A Postmarketing Surveillance Study
by
Akemi Ishikawa-Ichikawa, Jorge Alberto Islas-Martínez, Eduardo Rios-Garcia, Luis Fernando Tejado-Gallegos and Pamela Monserrat Ramírez-Marín
Pharmacoepidemiology 2026, 5(1), 7; https://doi.org/10.3390/pharma5010007 - 28 Feb 2026
Abstract
►▼
Show Figures
Background: Acalabrutinib is a selective Bruton tyrosine kinase inhibitor widely used for chronic lymphocytic leukemia and mantle cell lymphoma. Real-world safety evidence from Latin America remains limited, which restricts local benchmarking and pharmacovigilance planning. In this study we aimed to assess exposure-adjusted
[...] Read more.
Background: Acalabrutinib is a selective Bruton tyrosine kinase inhibitor widely used for chronic lymphocytic leukemia and mantle cell lymphoma. Real-world safety evidence from Latin America remains limited, which restricts local benchmarking and pharmacovigilance planning. In this study we aimed to assess exposure-adjusted adverse events in routine care in Mexico. Methods: We analyzed postmarketing surveillance datasets and spontaneous reports from March 2020 to August 2024, classifying events with MedDRA and summarizing seriousness, severity, and incidence per 100 patient-years. Results: A total of 266 patients were registered; 193 had evaluable exposure and safety data, contributing 242.73 patient-years. The overall adverse event incidence was 24.71 per 100 patient-years. Twenty-eight individual case safety reports documented 60 events. Forty-four events were serious. Among 33 events with reported severity, 14 were severe, 14 moderate, and five mild. Frequently affected system organ classes were blood and lymphatic, vascular, and infections. Seven deaths were reported; most were associated with COVID-19 complications or disease progression. Conclusions: The adverse event profile observed aligns with published trial experience and supports the tolerability of acalabrutinib in Mexican practice. These country-level, exposure-adjusted estimates provide actionable context for clinicians, institutional pharmacists and pharmacovigilance teams and point to the value of strengthening report completeness to improve signal detection in routine oncology care.
Full article

Figure 1
Open AccessArticle
Pharmacovigilance from the Patient’s Perspective: Self-Reported Adverse Drug Reactions in Kosovo’s Elderly Population
by
Fitim Alidema and Arieta Hasani Alidema
Pharmacoepidemiology 2026, 5(1), 6; https://doi.org/10.3390/pharma5010006 - 30 Jan 2026
Abstract
Background: Pharmacovigilance is a critical component of patient safety, particularly among older adults with chronic diseases who are frequently exposed to polypharmacy. In Kosovo, adverse drug reactions (ADRs) reported by patients remain insufficiently recognized within the healthcare system. Polypharmacy, limited access to pharmaceutical
[...] Read more.
Background: Pharmacovigilance is a critical component of patient safety, particularly among older adults with chronic diseases who are frequently exposed to polypharmacy. In Kosovo, adverse drug reactions (ADRs) reported by patients remain insufficiently recognized within the healthcare system. Polypharmacy, limited access to pharmaceutical counseling, and self-medication practices may contribute to increased medication-related harm. Capturing ADRs directly from patients provides valuable insight into medication safety challenges and communication gaps in clinical care. Objective: To assess the frequency, characteristics, and reporting behavior of adverse drug reactions among adults aged 60–75 years with chronic diseases in Kosovo, and to identify factors associated with awareness and reporting practices. Methods: A multicenter cross-sectional study was conducted between January and September 2025 in four major cities in Kosovo (Prishtina, Prizren, Peja, and Gjilan). A total of 1024 patients receiving continuous therapy for at least one chronic condition were surveyed using a structured questionnaire covering demographic characteristics, drug exposure, ADR experience, and reporting behavior. Statistical analyses included descriptive statistics, chi-square testing, and multivariable logistic regression to identify predictors of ADR reporting. Results: Overall, 47.3% of participants reported experiencing at least one ADR in the preceding 12 months. Among those, 39.5% reported the event to a healthcare professional, whereas 60.5% did not seek professional advice. The most frequently implicated drug classes were antihypertensives (32.8%), analgesics and non-steroidal anti-inflammatory drugs (27.4%), and antirheumatic agents (14.6%), with mainly gastrointestinal (24.1%) and cardiovascular (18.9%) manifestations. Approximately 19.8% of participants reported discontinuing medication due to adverse effects. Female patients were more likely to report ADRs compared to males (p < 0.01). Lack of prior counseling about potential side effects was independently associated with lower reporting (OR = 2.17; 95% CI: 1.41–3.33). Patients using more than six medications had a higher prevalence of ADRs (61.2%). Conclusion: Adverse drug reactions were frequently reported by older patients, while formal reporting to healthcare professionals remained limited. Strengthening patient education, improving patient–provider communication, and integrating clinical pharmacists into primary care may enhance pharmacovigilance practices and medication safety.
Full article
Open AccessSystematic Review
Intraocular Inflammation Following Intravitreal Faricimab: A Systematic Review and Meta-Analysis
by
Jumanah Qedair, Asmaa A. Youssif, Reham Shehada and Hashem Abu Serhan
Pharmacoepidemiology 2026, 5(1), 5; https://doi.org/10.3390/pharma5010005 - 26 Jan 2026
Abstract
►▼
Show Figures
Background/Objectives: To evaluate the incidence, characteristics, and clinical outcomes of intraocular inflammation (IOI) associated with intravitreal faricimab (IVF) in patients with neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME). Methods: Following PRISMA guidelines, a comprehensive search of PubMed, Web of Science,
[...] Read more.
Background/Objectives: To evaluate the incidence, characteristics, and clinical outcomes of intraocular inflammation (IOI) associated with intravitreal faricimab (IVF) in patients with neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME). Methods: Following PRISMA guidelines, a comprehensive search of PubMed, Web of Science, Scopus, Embase, and CENTRAL databases was performed from their inception to February 2025. Using the random-effects model, weighted proportions, standardized mean differences, and weighted log odds ratios (OR) were pooled and calculated. A two-tailed p-value of <0.05 was considered statistically significant. The χ2 (z) test and the Higgins I2 test were used to assess studies heterogeneity. Results: We conducted a systematic review and meta-analysis of 24 studies (4761 patients; 5652 eyes). The most common diagnoses were nAMD (n = 4782, 94.6%) and DME (n = 845, 37.1%). The pooled proportion for IOI incidence in eyes receiving IVF was 3.0% (95% CI: 1.0–6.0). The odds of developing IOI did not differ significantly between the DME and nAMD groups (OR: 1.13, p = 0.78). Unspecified IOI was the most common sign (n = 210, 2.9% [95% CI: 1.2–7.3]), followed by anterior uveitis (n = 80, 1.9% [95% CI: 0.1–34.8]), vitritis (n = 63, 2.9% [95% CI: 0.2–32.1]), retinal hemorrhage (n = 27, 0.7% [95% CI: 0.0–15.3]), and endophthalmitis (n = 8, 0.5% [95% CI: 0.3–1.1]). Conclusions: While IVF demonstrates therapeutic efficacy, our findings highlight a clinically relevant risk of IOI. We, therefore, recommend vigilant clinical monitoring in patients receiving this therapy.
Full article

Figure 1
Open AccessArticle
Incidence of Adverse Drug Reactions at the University Hospital Center of Libreville, Gabon: From Data Collection to a Risk Minimization Plan
by
Pierre Constant Ntoutoume Nzoghe, Rim Lakhmiri, Sophie Coniquet, Solange Ntsame, Ihsane Hmamouchi, Yahia Cherrah and Samira Serragui
Pharmacoepidemiology 2026, 5(1), 4; https://doi.org/10.3390/pharma5010004 - 16 Jan 2026
Cited by 2
Abstract
►▼
Show Figures
Background: According to the literature, adverse drug reactions (ADRs) account for 5–10% of hospital admissions and affect 25–30% of hospitalized patients, but no data are available for Gabon. Objectives: To estimate the incidence of ADRs among hospitalized patients at the Libreville University Hospital
[...] Read more.
Background: According to the literature, adverse drug reactions (ADRs) account for 5–10% of hospital admissions and affect 25–30% of hospitalized patients, but no data are available for Gabon. Objectives: To estimate the incidence of ADRs among hospitalized patients at the Libreville University Hospital Center (CHUL) and to classify them according to their frequency, severity, mechanism and preventability, while proposing appropriate risk minimization strategies. Patients and Methods: A 14-month, single-center, prospective study included all patients experiencing ADRs, excluding those without ADRs or with intentional overdoses. ADRs were analyzed using the World Health Organization (WHO) causality assessment, the ATC classification, and Rawlins and Thompson criteria. Data were actively collected from patients and hospital records. Results: Among 4999 patients, 105 experienced 177 adverse events (incidence: 3.5%, 95% CI: 1.7–2.5%). Among the identified ADRs, 42% were serious. Nausea and vomiting were the most frequent ADRs, mainly caused by analgesics (nefopam, tramadol) and antibiotics (amoxicillin–clavulanic acid). The gastrointestinal and nervous systems were the most affected. According to the Rawlins and Thompson classification, 90% of ADRs were type A, 8% type B, and 2% type E (withdrawal syndrome). Overall, 90% of ADRs were preventable. Conclusions: This study highlights the importance of pharmacovigilance at CHUL, Gabon, and emphasizes the role of healthcare professionals in ADR reporting and risk minimization.
Full article

Figure 1
Open AccessReview
Clinical Use, Population-Level Impact, and Antimicrobial Resistance Considerations of Probiotics and Microbiome-Based Therapeutics: Review
by
Monthon Lertcanawanichakul, Phuangthip Bhoopong, Husna Madoromae and Tuanhawanti Sahabuddeen
Pharmacoepidemiology 2026, 5(1), 3; https://doi.org/10.3390/pharma5010003 - 15 Jan 2026
Abstract
Probiotics and microbiome-based therapeutics are increasingly used to prevent antibiotic-associated diarrhea (AAD) and support gut microbiota health across children, adults, and elderly populations. Evidence synthesized in this narrative review from randomized controlled trials and meta-analyses (>20,000 participants) suggests that early probiotic administration, particularly
[...] Read more.
Probiotics and microbiome-based therapeutics are increasingly used to prevent antibiotic-associated diarrhea (AAD) and support gut microbiota health across children, adults, and elderly populations. Evidence synthesized in this narrative review from randomized controlled trials and meta-analyses (>20,000 participants) suggests that early probiotic administration, particularly Lactobacillus rhamnosus GG, Bifidobacterium species, multistrain formulations, and Saccharomyces boulardii, is associated with a 30–40% relative reduction in AAD incidence across heterogeneous studies, with absolute risk reductions of approximately 5–12% depending on baseline risk, strain, dose, and timing. Probiotics are generally well tolerated, with mild gastrointestinal adverse effects reported in 3–5% of users and rare serious events mainly in immunocompromised individuals. However, heterogeneity in formulations, populations, and limited long-term real-world data underscores the need for further pharmacoepidemiological studies, microbiome surveillance, and evaluation of antimicrobial resistance implications.
Full article
(This article belongs to the Special Issue Exploring Herbal Medicine: Applying Epidemiology Principles)
Open AccessArticle
Prevalence Rate of Adverse Drug Reactions from Sodium-Glucose Cotransporter-2 Inhibitors: A Retrospective Cohort Study
by
Pichitra Srimaya, Tossapol Warong, Sudarat Kingdang, Titawadee Pradubkham and Wiraphol Phimarn
Pharmacoepidemiology 2026, 5(1), 2; https://doi.org/10.3390/pharma5010002 - 31 Dec 2025
Abstract
►▼
Show Figures
Background/Objectives: Sodium-glucose cotransporter-2 (SGLT2) inhibitors are widely used in type 2 diabetes mellitus for glycemic control and cardiovascular–renal protection, but adverse effects such as acute kidney injury (AKI), urinary tract infection (UTI), euglycemic diabetic ketoacidosis (Eu-DKA), and acute pancreatitis remain concerns. We
[...] Read more.
Background/Objectives: Sodium-glucose cotransporter-2 (SGLT2) inhibitors are widely used in type 2 diabetes mellitus for glycemic control and cardiovascular–renal protection, but adverse effects such as acute kidney injury (AKI), urinary tract infection (UTI), euglycemic diabetic ketoacidosis (Eu-DKA), and acute pancreatitis remain concerns. We aimed to determine the prevalence of adverse drug reactions (ADRs) associated with SGLT2 inhibitor use. Methods: This retrospective study assessed the prevalence of these adverse events and identified factors associated with UTI among SGLT2 inhibitor users at Suddhavej Hospital (1 January 2019–15 August 2023). Data were extracted from the hospital electronic medical record system (BMS-HOSxP). Results: We analyzed 293 patients (59.73% male; mean age 63.08 ± 0.667 years; 62.08% aged >60). Dapagliflozin had the highest prevalence of AKI (11.42%) and UTI (13.40%). No acute pancreatitis cases were reported. Logistic regression identified female sex (odds ratios [OR] 2.31, 95% confidence intervals [CI] 1.08–4.96; p = 0.032), AKI diagnosis (OR 3.31, 95% CI 1.10–9.89; p = 0.032), age ≥ 60 years (OR 2.78, 95% CI 1.09–7.09; p = 0.033), and SGLT2 inhibitor use <6 months (OR 5.78, 95% CI 2.74–14.18; p = 0.017) as significant risk factors for UTI. Conclusions: Dapagliflozin was associated with the highest prevalence of AKI and UTIs. Female sex, AKI diagnosis, age ≥ 60 years, and SGLT2 inhibitor use <6 months were significant risk factors for UTI among SGLT2 inhibitor users.
Full article

Figure 1
Open AccessArticle
Disparity of Prescribed Psychotropics in Alzheimer’s Disease with Neuropsychiatric Symptoms
by
Samuel I. Nathaniel, Maggie Oliver, Thomas I. Nathaniel, Laurie Marie Theriot Roley, Richard L. Goodwin and Adebobola Imeh-Nathaniel
Pharmacoepidemiology 2026, 5(1), 1; https://doi.org/10.3390/pharma5010001 - 22 Dec 2025
Abstract
►▼
Show Figures
Objective: The objective of this study was to determine whether Non-Hispanic Black (NHB) or Non-Hispanic White (NHW) Alzheimer dementia patients with neuropsychiatric symptoms (ADNPS) differ regarding treatment with second-generation antipsychotics (SGAs), central acetylcholinesterase inhibitors (CAIs), and selective serotonin reuptake inhibitors (SSRIs). Methods:
[...] Read more.
Objective: The objective of this study was to determine whether Non-Hispanic Black (NHB) or Non-Hispanic White (NHW) Alzheimer dementia patients with neuropsychiatric symptoms (ADNPS) differ regarding treatment with second-generation antipsychotics (SGAs), central acetylcholinesterase inhibitors (CAIs), and selective serotonin reuptake inhibitors (SSRIs). Methods: Pharmacologic and demographic factors associated with male and female ADNPS were examined using retrospective data collected from a registry from 2016 and 2020 in a regional AD care center. The logistic regression model was developed to generate odds ratios (OR) to determine factors that were associated with male or female ADNPS. Results: A total of 7031 AD patients were identified. Overall, 6237 patients were NHWs, and 794 were NHBs. Among the NHW AD patients, 1909 presented with behavioral disturbances or neuropsychiatric symptoms (NPS), and 168 NHB AD patients presented with NPS. In the adjusted analysis, NHW ADNPS patients were more likely to be treated with galantamine (OR = 1.538, 95% CI, 1.001–2.364, p = 0.049), memantine (OR = 1.222, 95% CI, 1.086–1.375, p < 0.001), olanzapine (OR = 2.323, 95% CI, 1.794–3.009, p < 0.001), risperidone (OR = 4.181, 95% CI, 3.539–4.939, p < 0.001), and escitalopram (OR = 1.401, 95% CI, 1.225–1.602, p < 0.001). In contrast, NHB ADNPS patients were more likely to be treated with memantine (OR = 2.601, 95% CI, 1.746–3.875, p < 0.001) and risperidone (OR = 5.526, 95% CI, 3.411–8.951, p < 0.001). Conclusions: Our findings show the use of memantine and risperidone to treat both NHB and NHW ADNPS patients. NHW ADNPS patients were more likely to be treated with galantamine, memantine, olanzapine, risperidone, and escitalopram. In contrast, NHB patients with ADNPS were more likely to be treated with memantine and risperidone.
Full article

Figure 1
Open AccessArticle
Identifying Myocardial Infarction and Ischemic Stroke Events in China Real-World Data: A Validation Study in Tianjin Regional Healthcare Database
by
Jiamei Liu, Zizhao Zhang, Yin Liu, Liming Zhao, Zhenna Huang, Xuxiao Ye, Jeff L. Lange, Nafeesa Dhalwani, Fan Yang, Kangyin Chen, Hao Zhang and Jifang Zhou
Pharmacoepidemiology 2025, 4(4), 28; https://doi.org/10.3390/pharma4040028 - 15 Dec 2025
Cited by 2
Abstract
►▼
Show Figures
Objectives: Real-world evidence that supports decision-making must meet numerous criteria, including validated identification of clinical outcomes. This study aimed to develop and validate a method for identifying new cases of myocardial infarction (MI) and ischemic stroke (IS) within real-world clinical data in China.
[...] Read more.
Objectives: Real-world evidence that supports decision-making must meet numerous criteria, including validated identification of clinical outcomes. This study aimed to develop and validate a method for identifying new cases of myocardial infarction (MI) and ischemic stroke (IS) within real-world clinical data in China. Methods: Algorithms to identify MI and IS events were developed using ICD-10-CM codes and Chinese diagnosis keywords within the Tianjin Regional Healthcare Database. Validation followed predefined criteria: MI required cardiac troponin elevation and ischemic symptoms or cardiac troponin elevation and electrocardiogram changes; IS required clinical symptoms and neuroimaging confirmation of cerebral Magnetic Resonance Imaging (MRI) or Computerized Tomography (CT) reports. Positive predictive value (PPV) with 95% confidence intervals (CI) was calculated for each outcome. Results: Among 304 MI and 302 IS cases randomly selected, approximately half were identified using ICD-10-CM codes and half through Chinese diagnosis keywords. Overall PPV for MI was 69% (95% CI: 63–74%), with similar PPVs across identification methods. PPV increased to 88% for inpatient MI and 97% for primary inpatient MI. For IS, overall PPV was 65% (95% CI: 58–71%), with higher PPV for cases identified by ICD-10-CM codes (76%) compared to keyword-only cases (56%). PPV increased to 76% for inpatient IS and 91% for primary inpatient IS. Conclusions: The use of ICD-10-CM codes and Chinese diagnosis keywords in primary inpatient diagnoses provides a validated approach for the identification of clinical outcomes of MI and IS within real-world clinical data in China.
Full article

Figure 1
Open AccessArticle
Antipsychotic Drugs and Diabetic Ketoacidosis: A Disproportionality Analysis of the FDA Adverse Event Reporting System
by
Nisrine Haddad, Abdallah Alami, Christopher A. Gravel, Derek Tsui, Yue Chen, Franco Momoli, Donald Mattison, Nawal Farhat and Daniel Krewski
Pharmacoepidemiology 2025, 4(4), 27; https://doi.org/10.3390/pharma4040027 - 25 Nov 2025
Cited by 2
Abstract
►▼
Show Figures
Objectives: To evaluate reports of diabetic ketoacidosis (DKA) associated with antipsychotic drug (APD) use submitted to the U.S. Food and Drug Administration’s Adverse Event Reporting System (FAERS). Methods: A retrospective pharmacovigilance analysis was conducted using FAERS data from January 2000 to
[...] Read more.
Objectives: To evaluate reports of diabetic ketoacidosis (DKA) associated with antipsychotic drug (APD) use submitted to the U.S. Food and Drug Administration’s Adverse Event Reporting System (FAERS). Methods: A retrospective pharmacovigilance analysis was conducted using FAERS data from January 2000 to December 2022. DKA cases were identified using the MedDRA preferred term “diabetic ketoacidosis” in reports listing antipsychotic drugs as suspect medications. Disproportionality analyses, including the proportional reporting ratio (PRR) and empirical Bayes geometric mean (EBGM), were used to assess reporting patterns. Multiple analyses were performed, including those restricted to primary suspect listed drugs only, expanded to incorporate secondary suspect drugs, and sensitivity analyses excluding reports submitted by legal professionals. Results: Among 19,961 DKA reports in FAERS, 2489 (12.5%) listed atypical antipsychotics as the primary suspect drug, whereas reports involving typical APDs were rare. The majority of reports were submitted by healthcare professionals (74.1%), and nearly half originated from the United States (45.4%). Hospitalization was a frequent outcome, reported in 74.3% of cases. Quetiapine and olanzapine were the most frequently reported atypical APDs, with disproportionality analyses demonstrating strong safety signals when compared to all other drugs in FAERS: olanzapine PRR 13.2 (95% CI: 12.4–14.2) and quetiapine PRR 11.8 (95% CI: 11.1–12.5). The findings remained consistent across multiple sensitivity analyses, including incorporating secondary suspect drugs, when the comparator group was restricted to only psychotropic drugs, and excluding reports submitted by lawyers. Conclusions: This pharmacovigilance analysis highlights a potential safety signal for DKA with atypical antipsychotic drugs, notably quetiapine and olanzapine. While these findings do not establish causality, they underscore the need for further investigation using clinical and epidemiological data.
Full article
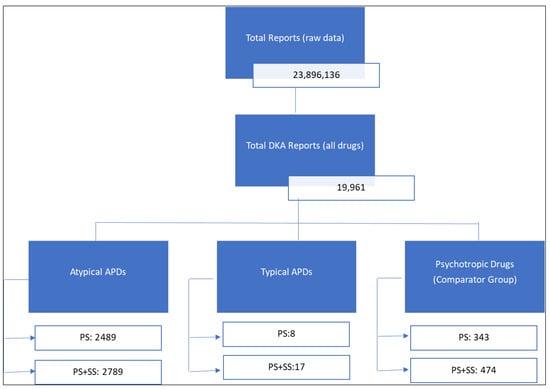
Figure 1
Open AccessReview
Idealized Framework for Assisting Pharmacovigilance Reporting in an Ambulatory Primary Care and Chronic Disease Management Clinic
by
Patrick J. Silva, Sara L. Rogers, Zoya Hassan-Toufique, Jian Tao, Scott A. Bruce, Paula K. Shireman and Kenneth S. Ramos
Pharmacoepidemiology 2025, 4(4), 26; https://doi.org/10.3390/pharma4040026 - 21 Nov 2025
Abstract
►▼
Show Figures
Pharmacovigilance approaches have conventionally focused on the use of epidemiological data to detect emergent adverse drug reactions (ADRs). Recent advances in the use and availability of real-world data have expanded opportunities to detect ADR signals in medical records. We provide a limited review
[...] Read more.
Pharmacovigilance approaches have conventionally focused on the use of epidemiological data to detect emergent adverse drug reactions (ADRs). Recent advances in the use and availability of real-world data have expanded opportunities to detect ADR signals in medical records. We provide a limited review of pharmacovigilance practices and tools we have specifically considered implementing into our comprehensive medication management clinic and associated research programs. Use of pharmacogenomic variants has proven useful only on a limited scale as such data are reliant on low-dimensional approaches matching variants to drugs, often with small effect sizes. As such, most ADRs go unrecognized, undocumented, and unactionable. We posit that an idealized pharmacovigilance framework that relies on artificial-intelligence-assisted reporting with adjudication by pharmacovigilance experts and new models of ambulatory pharmaceutical practice would establish the following attributes: (1) all metadata relating to medication use would be available in the medical record in computable and interoperable data models, (2) digital surveillance tools would detect most ADR events with attributed pharmacological contributions, (3) all events would be characterized using standard adjudication rubrics, and (4) all events would iteratively inform an ADR knowledgebase and improve models to advance detection and prediction of ADR during the course of patient care with a focus on having the necessary tools for clinicians to prevent ADRs. This review provides a limited and focused framework for more systematic documentation of ADRs and tactics to mitigate the idiopathic nature of most ADRs.
Full article
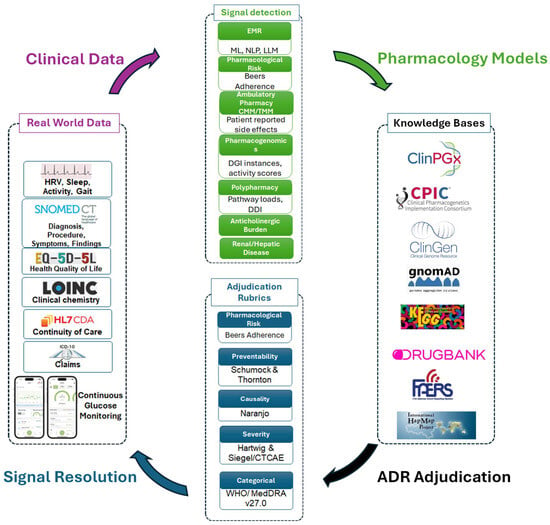
Figure 1
Open AccessArticle
Post-Marketing Pharmacovigilance Study of Darunavir in the United Kingdom: An Analysis of Adverse Drug Reactions Reported to the MHRA
by
Pono Pono, Vicky Cheng, Victoria Skerrett and Alan M. Jones
Pharmacoepidemiology 2025, 4(4), 25; https://doi.org/10.3390/pharma4040025 - 6 Nov 2025
Abstract
Background/Objectives: Human immunodeficiency virus (HIV) continues to be a global public health concern. Several antiretroviral drugs have been approved for the treatment, post-exposure, and pre-exposure prophylaxis of HIV. Darunavir (DRV) is a protease inhibitor (PI) approved for the management of HIV globally.
[...] Read more.
Background/Objectives: Human immunodeficiency virus (HIV) continues to be a global public health concern. Several antiretroviral drugs have been approved for the treatment, post-exposure, and pre-exposure prophylaxis of HIV. Darunavir (DRV) is a protease inhibitor (PI) approved for the management of HIV globally. This study aims to generate safety signals for DRV through data mining and analysis of adverse events (AEs) reported to the United Kingdom (UK) Medicines and Healthcare products Regulatory Agency (MHRA) Yellow Card Scheme. Methods: Disproportionality analysis was conducted using reporting odds ratio (ROR), proportional reporting ratio (PRR), and Bayesian confidence propagation neural network (BCPNN) approaches to identify potential safety signals. Results: The MHRA database contained n = 779 reports (n = 1791 AEs) attributed to DRV. The majority of AEs were reported for males. Positive safety signals were identified at both the system organ class (SOC, n = 5) and preferred term level (PT, n = 95). At SOC level, endocrine disorders emerged as a signal of interest n = 33 cases (ROR: 8.17, 95% CI: 5.78–11.56; PRR:7.96, 95% CI: 5.68–11.15; and IC: 2.85, IC025: 2.51). Among the results, 40 new potential safety signals are not listed on the product labelling in the UK. These include serious AEs such as cerebrovascular accident, brain injury, thrombosis, and pregnancy, puerperium, and perinatal AEs. Conclusions: This study provides additional real-world safety data for DRV in the UK and paves the way for future observational studies to investigate the identified safety signals.
Full article
(This article belongs to the Special Issue Pharmacoepidemiology and Pharmacovigilance in the UK)
Open AccessArticle
Exploratory Signal Detection of Maternal and Perinatal Adverse ART Drug Events in EudraVigilance: Insights from Network and Cluster Analyses
by
Bárbara Costa and Nuno Vale
Pharmacoepidemiology 2025, 4(4), 24; https://doi.org/10.3390/pharma4040024 - 4 Nov 2025
Cited by 1
Abstract
►▼
Show Figures
Background: Medication safety in pregnancy, puerperium, and perinatal periods is underexplored because these populations are excluded from clinical trials. EudraVigilance offers post-marketing evidence, but disproportionality analyses focus on isolated drug event pairs and may miss syndromic patterns. We applied a network- and
[...] Read more.
Background: Medication safety in pregnancy, puerperium, and perinatal periods is underexplored because these populations are excluded from clinical trials. EudraVigilance offers post-marketing evidence, but disproportionality analyses focus on isolated drug event pairs and may miss syndromic patterns. We applied a network- and cluster-based framework to EudraVigilance reports on antiviral use in pregnancy to improve surveillance and identify meaningful constellations. Methods: We retrieved all individual case safety reports (ICSRs) from January 2015 to June 2025, including pregnancy, puerperium, or perinatal terms, focusing on suspect antivirals. After parsing terms, disproportionality metrics were computed as a benchmark. A bipartite drug–event network was built and projected to event–event co-occurrence networks; Louvain community detection identified clusters. Clusters were characterized by size, drug mix, seriousness, overlap with disproportionality signals, and stratification across periods. Results: The dataset comprised 106,924 ICSRs and 232,067 unique pairs. Disproportionality yielded 6142 signals, mainly involving antiretrovirals (ritonavir, lamivudine, zidovudine, emtricitabine/tenofovir). Network analysis revealed clusters grouping maternal and fetal/neonatal outcomes (e.g., fetal death, low birth weight), and transplacental transfer, highlighting structures not visible in pairwise analyses. Several clusters combined high-frequency exposures with clinically relevant outcomes, suggesting early-warning potential. Conclusions: Combining disproportionality with network- and cluster-based pharmacovigilance adds value for monitoring pregnancy medication safety. Beyond individual signals, this approach reveals meaningful clusters and “bridge” reactions connecting adverse-event domains, offering a richer framework for perinatal surveillance. Despite spontaneous-reporting limits, findings generate hypotheses for mechanistic and pharmacoepidemiologic follow-up and support network methods as complements to traditional pharmacovigilance.
Full article

Figure 1
Open AccessCase Report
Management of Acute Moderate Iron Poisoning with Oral Chelation and Antioxidant Therapy: A Case Report
by
Mary Isabel Vanegas-Rincón, María A. Barón-Bolívar, Javier A. Aguilar-Mejía, Diana Patricia Amador-Munoz and Luis Carlos Rojas-Rodríguez
Pharmacoepidemiology 2025, 4(4), 23; https://doi.org/10.3390/pharma4040023 - 1 Nov 2025
Abstract
►▼
Show Figures
Introduction: Acute iron poisoning is a potentially life-threatening condition that primarily affects the gastrointestinal, hepatic and cardiovascular systems. While it most often occurs accidentally in children, intentional overdoses in adolescents and adults remain an important clinical concern. Case description: We report
[...] Read more.
Introduction: Acute iron poisoning is a potentially life-threatening condition that primarily affects the gastrointestinal, hepatic and cardiovascular systems. While it most often occurs accidentally in children, intentional overdoses in adolescents and adults remain an important clinical concern. Case description: We report the case of a 14-year-old male patient with a history of depression who intentionally ingested 100 ferrous sulfate tablets (equivalent to 118 mg/kg of elemental iron). The patient was admitted to the emergency department three hours after ingestion. He presented with vomiting tablet remnants, headache, and mild abdominal pain. Supportive measures included intestinal irrigation with polyethylene glycol (PEG), gastric protection, and N-acetylcysteine intravenous administration. The iron chelator therapy with deferoxamine was not possible because the medication was unavailable, so treatment with the oral iron chelator (deferasirox) was initiated. The iron levels gradually decreased, with no evidence of liver or cardiovascular involvement. The patient was discharged on day 20 post-ingestion with outpatient psychiatric follow-up. Discussion: This case highlights the importance of early initiation of gastrointestinal decontamination with PEG to limit systemic iron absorption. The use of deferasirox as an alternative chelating agent in the absence of deferoxamine has been associated with a favorable response. Conclusions: The rational use of oral chelators, gastrointestinal decontamination, and hepatoprotective therapies in acute iron poisoning might prevent major complications and improve prognosis. Alternative therapies can be valuable when an antidote is not immediately available; however, further clinical research is required before making a recommendation.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Special Issues
Special Issue in
Pharmacoepidemiology
Recent Advances in the Pharmacoepidemiology of Antirheumatic Medication
Guest Editors: Roberta Foti, Beatrice Maranini, Veronica VenturelliDeadline: 30 June 2026
Special Issue in
Pharmacoepidemiology
Pharmacoepidemiology and Pharmacovigilance in the UK
Guest Editor: Tanja MuellerDeadline: 30 June 2026
Special Issue in
Pharmacoepidemiology
Women’s Special Issue Series: Pharmacoepidemiology
Guest Editors: Carlotta Franchi, Li-Chia Chen, Carlotta LunghiDeadline: 30 June 2026
Special Issue in
Pharmacoepidemiology
The Pharmacoepidemiology of Antipsychotics: Utilization, Safety and Effectiveness in Real-World Settings
Guest Editor: Yi ChaiDeadline: 30 September 2026








