Journal Description
Organics
Organics
is an international, peer-reviewed, open access journal on organic chemistry published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 22.3 days after submission; acceptance to publication is undertaken in 4.7 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Journal Cluster of Chemical Reactions and Catalysis: Catalysts, Chemistry, Electrochem, Inorganics, Molecules, Organics, Oxygen, Photochem, Reactions, Sustainable Chemistry.
Impact Factor:
1.6 (2024);
5-Year Impact Factor:
1.8 (2024)
Latest Articles
Integrated In Silico and Chromatographic Evaluation of the Biological Properties of Novel Bis-Substituted Thiocarbohydrazone Derivatives
Organics 2026, 7(2), 19; https://doi.org/10.3390/org7020019 - 12 May 2026
Abstract
►
Show Figures
Thiocarbohydrazone derivatives represent a highly significant class in medicinal chemistry, characterized by a versatile scaffold defined with a thiocarbonyl (C=S) core and one or two imine (–C=N–) functionalities, allowing for precise modulation of their physicochemical and biological properties. The biological potential of a
[...] Read more.
Thiocarbohydrazone derivatives represent a highly significant class in medicinal chemistry, characterized by a versatile scaffold defined with a thiocarbonyl (C=S) core and one or two imine (–C=N–) functionalities, allowing for precise modulation of their physicochemical and biological properties. The biological potential of a series of novel bis-substituted thiocarbohydrazone derivatives was predicted and evaluated through comprehensive in silico analysis. All investigated compounds complied with Lipinski’s Rule of 5, with most also satisfying the Rule of 3 while simultaneously exhibiting favorable pharmacokinetic properties and low predicted ecotoxicity. To substantiate these findings and elucidate the influence of para-substituents, chromatographic behavior of the studied derivatives was evaluated using reversed-phase thin-layer chromatography (RP-TLC). Initial linear regression analysis revealed statistically significant correlations between chromatographic parameters and in silico-derived descriptors of lipophilicity, pharmacokinetics, and ecotoxicity. Furthermore, cluster analysis and principal component analysis provided a robust and unambiguous interpretation of the structure–property relationships, highlighting substituent polarity as the leading factor controlling the bioactivity of bis-substituted thiocarbohydrazones, although the contribution of electronic effects cannot be neglected. Moreover, RM0 correlates with lipophilicity and pharmacokinetics, whereas m reflects ecotoxicity. Collectively, these findings emphasize the critical role of subtle structural variations in shaping the overall properties of these novel derivatives.
Full article
Open AccessArticle
Synthesis of 23,23-Difluoro-24-nor- and 24′,24′-Difluoro-24-Homovitamin D3 Analogues and Unexpected Structure-Activity Relationships
by
Fumihiro Kawagoe, Hiroya Tabuchi, Taiyo Ideguchi, Yuki Okamoto, Souma Murata, Tomofumi Yatsu, Syota Yamada, Kaori Yasuda, Yusuke Akagi, Masashi Takano, Toshie Fujishima, Yoshiki Miyata, Ken’ichi Aoki, Toshiyuki Sakaki and Atsushi Kittaka
Organics 2026, 7(2), 18; https://doi.org/10.3390/org7020018 - 27 Apr 2026
Abstract
►▼
Show Figures
We synthesized two vitamin D3 analogues, 3 and 4, which have a shortened or elongated fluoro-side-chain based on 24,24-difluoro-25-hydroxyvitamin D3 (5) using an efficient convergent approach and studied their preliminary biological activity. Both analogues exhibited greater resistance to
[...] Read more.
We synthesized two vitamin D3 analogues, 3 and 4, which have a shortened or elongated fluoro-side-chain based on 24,24-difluoro-25-hydroxyvitamin D3 (5) using an efficient convergent approach and studied their preliminary biological activity. Both analogues exhibited greater resistance to CYP24A1-mediated metabolism than the natural 25-hydroxyvitamin D3 (6), although their stability was lower than that of 5. Analogue 3 showed an approximately 100-fold lower human vitamin D receptor (hVDR)-binding affinity compared with 5 and 6. Despite this marked reduction in VDR-binding affinity, it demonstrated an approximately 1.5-fold increase in VDR-ligand binding domain (LBD) transcriptional activation of the natural ligand 6. In contrast, analogue 4 displayed moderate VDR-binding affinity and VDR-LBD transactivation compared with 5 and 6. We found that compound 3 is a unique vitamin D analogue with a fluorinated and shortened side-chain, exhibiting low binding affinity for hVDR but potent transcriptional activity through VDR-LBD with its long half-life; thus, 3 may serve as a basic structural skeleton for advancing medicinal chemistry and drug discovery.
Full article

Figure 1
Open AccessArticle
Revised Formal Total Synthesis of Dehydro-δ-Viniferin and Anigopreissin A
by
Alessandro Santarsiere, Marianna Volgare and Lucia Chiummiento
Organics 2026, 7(2), 17; https://doi.org/10.3390/org7020017 - 16 Apr 2026
Abstract
►▼
Show Figures
This work presents a revised total synthesis of two pharmacologically relevant benzofurans using newly developed environmentally friendly methodologies. In particular, we focused on establishing improved synthetic routes to stilbene dimers under milder and more sustainable reaction conditions. During our investigations, we optimized an
[...] Read more.
This work presents a revised total synthesis of two pharmacologically relevant benzofurans using newly developed environmentally friendly methodologies. In particular, we focused on establishing improved synthetic routes to stilbene dimers under milder and more sustainable reaction conditions. During our investigations, we optimized an efficient Sonogashira coupling carried out in water, which, followed by a Suzuki-like reaction conducted in dimethyl carbonate (DMC) in the absence of any transition metals, served as the key step for the synthesis of the benzofuran core.
Full article

Figure 1
Open AccessReview
Recent Developments in Chemical Synthesis and Biological Activities of Aloe-Emodin Derivatives
by
Jeltzlin Semerel, Nigel John, Pedro Fardim and Wim Dehaen
Organics 2026, 7(2), 16; https://doi.org/10.3390/org7020016 - 10 Apr 2026
Abstract
Aloe-emodin is an anthraquinone with a wide range of medicinal applications, including anti-angiogenic, anticancer, antimicrobial, antiviral, anti-inflammatory, and antioxidant activities. In this review, the functionalization of aloe-emodin using various synthetic methods, including alkylation, condensation, esterification, the Finkelstein reaction, and the Kabachnik–Fields reaction was
[...] Read more.
Aloe-emodin is an anthraquinone with a wide range of medicinal applications, including anti-angiogenic, anticancer, antimicrobial, antiviral, anti-inflammatory, and antioxidant activities. In this review, the functionalization of aloe-emodin using various synthetic methods, including alkylation, condensation, esterification, the Finkelstein reaction, and the Kabachnik–Fields reaction was reported. The biological activity of the synthesized aloe-emodin derivatives is discussed, with a focus on their potential future applications as anticancer agents, enzyme inhibitors, anti-inflammatory agents, and antimicrobial agents. This review also discusses the structure–activity relationship (SAR) and the mechanism of action (e.g., molecular docking studies, cell membrane-disrupting capacity, and apoptosis studies). This review highlights the many contributions made towards the design and development of novel, biologically active aloe-emodin derivatives.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
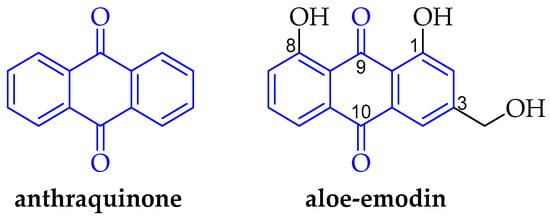
Figure 1
Open AccessArticle
Synthesis, Characterization, and Multidimensional In Silico Evaluation of Novel Etodolac-Based 1,3,4-Oxadiazole Derivatives as Potential Anticancer Agents
by
Tiba M. Hameed, Rafid M. Hashim, S. J. Abed, Raneen Hashim Ridha and O. Al-Mohammed Baqer
Organics 2026, 7(2), 15; https://doi.org/10.3390/org7020015 - 7 Apr 2026
Abstract
A new series of eight novel etodolac-based 1,3,4-oxadiazoles was synthesized, characterized, and tested in silico in multidimensional routes, starting with etodolac, a well-known nonsteroidal anti-inflammatory medication (NSAID). In silico studies were performed prior to synthesis using the molecular docking technique in CCDC GOLD
[...] Read more.
A new series of eight novel etodolac-based 1,3,4-oxadiazoles was synthesized, characterized, and tested in silico in multidimensional routes, starting with etodolac, a well-known nonsteroidal anti-inflammatory medication (NSAID). In silico studies were performed prior to synthesis using the molecular docking technique in CCDC GOLD suite software (2025.3) to assess the interactions with two key targets involved in cancer pathogenesis: the crystal structure of the epidermal growth factor receptor EGFR tyrosine kinase domain (PDB ID: 4HJO) and the matrix metalloproteinase (MMP-9) complex (PDB ID: 5CUH). ADME studies were performed to assess the physicochemical properties of the synthesized molecules. Importantly, biotransformation prediction also indicated that the derivatives possess high metabolic stability, with hydroxylation of the thio-ether group as the primary predicted biotransformation route. All compounds were characterized using melting point, FT-IR, 1H-NMR, and 13C-NMR spectroscopy. In vitro and/or in vivo experiments are needed to confirm this preliminary anticancer study.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures

Figure 1
Open AccessArticle
Salophen-Type Ni(II) Schiff Base Complexes Derived from Naphthalene Aldehydes and Their Application as Catalysts for the Methanol Electro-Oxidation Reaction
by
Fabiola Hernández-García, Emanuel Pérez-Martínez, Raúl Colorado-Peralta, Jesús Antonio Cruz-Navarro and David Morales-Morales
Organics 2026, 7(1), 14; https://doi.org/10.3390/org7010014 - 19 Mar 2026
Abstract
►▼
Show Figures
Salophen-type Schiff base ligands derived from salicylaldehyde and naphthalene aldehydes were synthesized and coordinated to Ni(II) to obtain three nickel complexes (NiL1–NiL3), which were evaluated as heterogeneous electrocatalysts for the methanol electro-oxidation reaction (MOR) in alkaline media. The ligands and complexes were fully
[...] Read more.
Salophen-type Schiff base ligands derived from salicylaldehyde and naphthalene aldehydes were synthesized and coordinated to Ni(II) to obtain three nickel complexes (NiL1–NiL3), which were evaluated as heterogeneous electrocatalysts for the methanol electro-oxidation reaction (MOR) in alkaline media. The ligands and complexes were fully characterized by FT-IR, 1H NMR, EPR, DART-MS, and elemental analysis, confirming tetradentate coordination through imine nitrogen and phenoxide oxygen donors. Electrochemical studies were carried out using carbon paste electrodes modified with 15 wt % of each complex. Cyclic voltammetry revealed that the electrocatalytic activity is mediated by the Ni(II)/Ni(III) redox couple, with Ni(III) oxohydroxide species acting as the active sites for methanol oxidation. Among the evaluated systems, NiL1@CPE showed superior performance at low methanol concentrations, while NiL2@CPE and NiL3@CPE exhibited higher current densities at elevated methanol concentrations. Scan-rate studies indicated that the oxidation process is diffusion-controlled, and a linear response to methanol concentration was observed over a wide concentration range. The results demonstrate that ligand structure and coordination geometry play a crucial role in modulating the electrocatalytic behavior of Ni(II) Schiff base complexes, highlighting their potential as cost-effective molecular catalysts for alkaline methanol oxidation.
Full article

Figure 1
Open AccessArticle
Frontier Orbitals and Charges Approaches in Electrophilic Aromatic Substitution: The Cases of Anisole and Benzaldehyde
by
Lucia Emanuele, Rocco Racioppi and Maurizio D’Auria
Organics 2026, 7(1), 13; https://doi.org/10.3390/org7010013 - 4 Mar 2026
Abstract
►▼
Show Figures
The study aimed to verify the possible use of DFT calculation in the prediction of the orientation in electrophilic aromatic substitution. An activated ortho/para orienting substrate, and a deactivated meta orienting substrate, were used in DFT calculations using B3LYP, B3PW91, BPV86, CAM-B3LP, HCTH,
[...] Read more.
The study aimed to verify the possible use of DFT calculation in the prediction of the orientation in electrophilic aromatic substitution. An activated ortho/para orienting substrate, and a deactivated meta orienting substrate, were used in DFT calculations using B3LYP, B3PW91, BPV86, CAM-B3LP, HCTH, HSEH1PBE, LSDA, MPW1PW91, PBEPBE, TPSSTPSS, and WB97XD functionals. The results showed that the reactivity of anisole can be adequately described considering charge control in reaction performed in hard conditions (nitration), while frontier orbital control can play a role in reactions performed in softer conditions (chlorination). Nitration of benzaldehyde can be rationalized through Hirshfeld charges analysis. Neither the frontier orbital nor Mulliken charges approach adequately account for behavior observed in chlorination of benzaldehyde. The effect of different basis sets was tested performing calculations with B3LYP functional and aug-cc-pVDZ, 6-311G+(d,p), aug-cc-pVQZ, DGTZVP, and LanL2DZ basis sets. For anisole, all basis sets provided a HOMO electron density distribution consistent with experimental reactivity; Hirshfeld charges analysis consistently reproduced the observed reactivity of anisole across all tested basis sets. All the basis sets were able to explain the observed reactivity of benzaldehyde in hard experimental condition, while they failed to give a correct description when a softer reagent was used.
Full article

Figure 1
Open AccessArticle
Linear Stepwise Synthesis of 2-(Naphthalen-1-yl)-2,3,5,6-tetrahydro-1H-isoquinolino[8,1,2-hij]quinazoline: A Novel Fused Heteroaromatic Framework
by
Augusto Rivera, Álvaro Castillo, Jaime Ríos-Motta and Diego Quiroga
Organics 2026, 7(1), 12; https://doi.org/10.3390/org7010012 - 3 Mar 2026
Abstract
►▼
Show Figures
In the present work, we describe the synthesis of a new heterocyclic derivative, 2-(naphthalen-1-yl)-2,3,5,6-tetrahydro-1H-isoquinolino[8,1,2-hij]quinazoline 1, using the reaction between the aminal 1,3,6,8-tetraazatricyclo[4.4.1.13,8]dodecane 2 (TATD) and 1-naphthylamine 3 as the first scaffold of a four-step linear synthetic
[...] Read more.
In the present work, we describe the synthesis of a new heterocyclic derivative, 2-(naphthalen-1-yl)-2,3,5,6-tetrahydro-1H-isoquinolino[8,1,2-hij]quinazoline 1, using the reaction between the aminal 1,3,6,8-tetraazatricyclo[4.4.1.13,8]dodecane 2 (TATD) and 1-naphthylamine 3 as the first scaffold of a four-step linear synthetic route. In the first step, a condensation catalyzed by acetic acid in 96% ethanol was carried out, leading to the formation of the intermediate 3-(naphthalen-1-yl)-1,2,3,4-tetrahydrobenzo[h]quinazoline 4. Subsequently, this intermediate was acylated with 2-chloroacetyl chloride in the presence of triethylamine and under an inert atmosphere, obtaining the compound 2-chloro-1-(3-(naphthalen-1-yl)-3,4-dihydrobenzo[h]quinazolin-1(2H)-yl)ethan-1-one 5. In the third step, an intramolecular Friedel–Crafts cyclization was carried out using aluminum trichloride as a catalyst, yielding 2-(naphthalen-1-yl)-1,2,3,6-tetrahydro-5H-isoquinolino[8,1,2-hij]quinazolin-5-one 6. Finally, the reduction of this lactam with phosphorus pentachloride and sodium borohydride under anhydrous conditions led to the further closure of the polycyclic system, yielding the final product 1. The proposed route demonstrates the feasibility of using TATD 2 as a versatile precursor for constructing condensed heterocyclic systems of structural interest and potential relevance in advanced organic synthesis.
Full article

Figure 1
Open AccessCommunication
Synthesis and Evaluation of Cytotoxic Activity of 2-Aryl-2-(3-Indolyl)Propionic Acid Derivatives
by
Alexander V. Aksenov, Nicolai A. Aksenov, Nikolai A. Arutiunov, Dmitrii A. Aksenov, Anna M. Zatsepilina, Daria I. Murashkina, Maksim O. Shcheglov and Sergei N. Ovcharov
Organics 2026, 7(1), 11; https://doi.org/10.3390/org7010011 - 3 Mar 2026
Abstract
►▼
Show Figures
2-Aryl-2-(3-indolyl)acetohydroxamic acids have emerged as promising antitumor agents; however, their poor pharmacokinetic profile remains a significant drawback. To address this limitation, we have synthesized a homolog of such acids—specifically 2-aryl-2-(3-indolyl)propionic acid (IC50 > 100 mM (U87)), along with several other derivatives: ethyl
[...] Read more.
2-Aryl-2-(3-indolyl)acetohydroxamic acids have emerged as promising antitumor agents; however, their poor pharmacokinetic profile remains a significant drawback. To address this limitation, we have synthesized a homolog of such acids—specifically 2-aryl-2-(3-indolyl)propionic acid (IC50 > 100 mM (U87)), along with several other derivatives: ethyl ester (IC50 > 100 mM (U87)), hydroxamate (IC50 21.2 ± 1.0 mM (U87)) and hydrazide (IC50 > 100 mM (U87)). The cytotoxicity of these compounds against glioblastoma cell lines was evaluated and compared to that of the parent acetohydroxamic acid derivatives.
Full article

Graphical abstract
Open AccessArticle
Systematic Investigation of the Solvation Structure in THF-Based Localized High-Concentration Electrolytes
by
Yoonha Hwang, Yeo Jin An, Soohyun Sim and Minjeong Shin
Organics 2026, 7(1), 10; https://doi.org/10.3390/org7010010 - 14 Feb 2026
Abstract
►▼
Show Figures
Understanding Li+ solvation structure is critical for the rational design of high- and localized high-concentration electrolytes. Here, we present a systematic investigation of tetrahydrofuran (THF)-based electrolytes with lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) using Raman spectroscopy and 7Li nuclear magnetic resonance to investigate the
[...] Read more.
Understanding Li+ solvation structure is critical for the rational design of high- and localized high-concentration electrolytes. Here, we present a systematic investigation of tetrahydrofuran (THF)-based electrolytes with lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) using Raman spectroscopy and 7Li nuclear magnetic resonance to investigate the local solvation structures. By varying the THF:LiTFSI molar ratio, we observed a transition of Li+ solvation from solvent-separated ion pairs to contact ion pairs and aggregates, accompanied by increased structural heterogeneity and constrained local dynamics. Raman spectroscopy captures the evolution of Li+–anion coordination with increasing salt concentration, while 7Li NMR chemical shifts, line widths, and relaxation times provide complementary insight into changes in the electronic environment and symmetry of Li+ coordination. Electrolyte structure is further examined by introducing a hydrofluoroether co-solvent into a concentrated (THF)2–LiTFSI electrolyte. Raman results show that the local Li+–TFSI− coordination structure is preserved upon 1,1,2,2-tetrafluoroethyl-2,2,3,3-tetrafluoropropyl ether (TTE) addition, whereas NMR reveals subtle modifications of the ion-rich solvation clusters. These results provide fundamental insight into Li+ solvation and electrolyte localization, offering general design principles for advanced electrolyte systems.
Full article

Figure 1
Open AccessArticle
Phosphate Monoester Hydrolysis by Supramolecular Phosphatases Functionalized with Lewis Acidic Moieties in Two-Phase Solvent Systems
by
Hirokazu Okamoto, Ayane Nomoto, Dahiru Umar Liman, Akib Bin Rahman, Toshifumi Tojo and Shin Aoki
Organics 2026, 7(1), 9; https://doi.org/10.3390/org7010009 - 13 Feb 2026
Abstract
►▼
Show Figures
Protein phosphorylation and dephosphorylation reactions of intracellular molecules catalyzed by enzymes such as kinases and phosphatases are essential reactions in a lot of cellular functions such as intracellular signal transduction in living systems. The design and synthesis of artificial enzyme mimics are important
[...] Read more.
Protein phosphorylation and dephosphorylation reactions of intracellular molecules catalyzed by enzymes such as kinases and phosphatases are essential reactions in a lot of cellular functions such as intracellular signal transduction in living systems. The design and synthesis of artificial enzyme mimics are important research topics in bioorganic and bioinorganic chemistry. In this paper, we report on the construction of artificial phosphatases via the supramolecular self-assembly of compounds such as an amphiphilic bis(Zn2+-cyclen) (cyclen = 1,4,7,10-tetraazacyclododecane) complex, barbital derivatives modified with benzocrown ethers and boronophenyl groups, and a copper(II) ion in a two-phase solvent system. We have developed a hypothesis whereby a mono(4-nitrophenyl)phosphate (MNP) substrate coordinates to the Cu2(µ-OH)2 core in supramolecular complexes and is activated either by Lewis acidic units such as alkali metal (Li+, Na+ and K+)-benzocrown ether complexes or by boronophenyl moieties. The findings suggest that supramolecular phosphatase functionalized with a benzo-12-crown-4-Li+ complex shows a higher level of activity in the MNP hydrolysis of a two-phase solvent system compared with that of our previous supramolecular phosphatases in terms of hydrolysis activity and catalytic turnover.
Full article

Graphical abstract
Open AccessArticle
A Quantum Chemical Study on the Relative Stability of Diaminodinitroethylene Isomers
by
Krzysztof K. Zborowski and Urszula Lelek-Borkowska
Organics 2026, 7(1), 8; https://doi.org/10.3390/org7010008 - 10 Feb 2026
Abstract
►▼
Show Figures
This study aims to investigate the relative stability of the diaminodinitroethylene isomers (cis, trans, and gem). To achieve this goal, calculations at several levels of theory were carried out. The B3LYP, PBE0, and CAM-B3LYP functionals, based on density functional theory (DFT), were used.
[...] Read more.
This study aims to investigate the relative stability of the diaminodinitroethylene isomers (cis, trans, and gem). To achieve this goal, calculations at several levels of theory were carried out. The B3LYP, PBE0, and CAM-B3LYP functionals, based on density functional theory (DFT), were used. G4 and MP2 calculations were also executed. All calculation methods predicted that the gem isomer is the most stable, while the cis isomer is the least stable. The energy order obtained for the isomers studied was rationalized by analysis of the detected intramolecular hydrogen bonding, electron delocalization, charge distribution, and changes in atomic energies in the structures studied. The origins of the superior stability of the gem isomer are demonstrated and justified.
Full article

Graphical abstract
Open AccessCommunication
Chemo- and Regioselective 1,3-Dipolar Cycloaddition of Nitrile Imines to 5-Arylmethylene-2-methylthiohydantoins
by
Maria E. Filkina, Lev A. Lintsov, Victor A. Tafeenko, Maxim E. Kukushkin and Elena K. Beloglazkina
Organics 2026, 7(1), 7; https://doi.org/10.3390/org7010007 - 3 Feb 2026
Abstract
►▼
Show Figures
1,3-Dipolar cycloaddition reactions of nitrile imines are a powerful tool for the construction of spirocyclic frameworks, yet controlling chemoselectivity remains challenging when dipolarophiles contain multiple reactive sites. In this study, we investigated the cycloaddition of nitrile imines with 5-arylmethylene-2-methylthiohydantoins, which possess both exocyclic
[...] Read more.
1,3-Dipolar cycloaddition reactions of nitrile imines are a powerful tool for the construction of spirocyclic frameworks, yet controlling chemoselectivity remains challenging when dipolarophiles contain multiple reactive sites. In this study, we investigated the cycloaddition of nitrile imines with 5-arylmethylene-2-methylthiohydantoins, which possess both exocyclic C=C and endocyclic C=N bonds. Nitrile imines were generated from hydrazonoyl chlorides under basic conditions and reacted with the thiohydantoin substrates under optimized reaction conditions. The cycloaddition proceeded smoothly, affording spiro-fused thiohydantoin–pyrazoline derivatives. In all cases, the reaction occurred selectively at the exocyclic C=C bond, while the C=N bond remained unreactive even in the presence of excess dipole. This chemoselectivity is attributed to the greater steric accessibility of the exocyclic double bond. These results clarify key factors governing nitrile imine chemoselectivity and provide a reliable approach to structurally complex spirocyclic thiohydantoin derivatives.
Full article

Figure 1
Open AccessArticle
Design of Sustainable Copper-Based Hybrid Catalyst Using Aqueous Extract of Curcuma longa L. for One-Pot Synthesis of 1,2,3-Triazole
by
Felipe Pinto, Isadora Barbosa Frederico, Conceição F. A. Olguin, Gabrielle Peiter, Julia C. M. Willig, Helio A. Stefani, Giancarlo V. Botteselle and Flavia Manarin
Organics 2026, 7(1), 6; https://doi.org/10.3390/org7010006 - 23 Jan 2026
Abstract
►▼
Show Figures
A sustainable hybrid material, CuO/Cu2O, was synthesized using an aqueous extract of Curcuma longa L. as a reducing and stabilizing agent. The material was characterized by UV-Vis spectroscopy, FTIR, XRD, SEM, EDX, and TEM. XRD analysis revealed peaks corresponding to CuO
[...] Read more.
A sustainable hybrid material, CuO/Cu2O, was synthesized using an aqueous extract of Curcuma longa L. as a reducing and stabilizing agent. The material was characterized by UV-Vis spectroscopy, FTIR, XRD, SEM, EDX, and TEM. XRD analysis revealed peaks corresponding to CuO and Cu2O phases with crystallite sizes of 15.88 nm and 16.71 nm, respectively. TEM images showed nearly spherical particles with some agglomeration and an average particle diameter of 8.17 nm. The hybrid material exhibited catalytic activity toward the synthesis of 1,2,3-triazoles in water, under low catalyst loading and mild reaction conditions. This work highlights the potential of Curcuma longa-mediated synthesis as a low-cost, eco-friendly alternative for producing efficient catalysts, contributing to the advancement of green chemistry and sustainable nanomaterial applications in organic synthesis.
Full article

Graphical abstract
Open AccessArticle
Aqueous Radical Photopolymerization Catalyzed by Resorufin
by
Wenqiao Zhou and Chunming Liu
Organics 2026, 7(1), 5; https://doi.org/10.3390/org7010005 - 15 Jan 2026
Cited by 1
Abstract
►▼
Show Figures
Commercially available resorufin was shown to function as an organic photocatalyst for visible-light-induced aqueous radical polymerization under low-irradiance illumination. Polymers with narrow molecular weight distributions and high monomer conversions were successfully synthesized from acrylate and acrylamide monomers. The photopolymerization catalyzed by resorufin was
[...] Read more.
Commercially available resorufin was shown to function as an organic photocatalyst for visible-light-induced aqueous radical polymerization under low-irradiance illumination. Polymers with narrow molecular weight distributions and high monomer conversions were successfully synthesized from acrylate and acrylamide monomers. The photopolymerization catalyzed by resorufin was consistent with a reductive quenching mechanism. Good temporal control of the reaction was achieved by toggling visible light irradiation.
Full article
Figure 1
Open AccessArticle
Metalloenzyme-like Catalytic System for the Epoxidation of Olefins with Dioxygen Under Ambient Conditions
by
Lin Lei, Linjian Wu, Yongjian Qiu and Yaju Chen
Organics 2026, 7(1), 4; https://doi.org/10.3390/org7010004 - 7 Jan 2026
Abstract
►▼
Show Figures
The development of a metalloenzyme-like catalytic system for the efficient oxidation of olefins under a dioxygen (O2) atmosphere at room temperature is of significant interest in the field of catalysis. Herein, we present a highly active and selective aerobic epoxidation of
[...] Read more.
The development of a metalloenzyme-like catalytic system for the efficient oxidation of olefins under a dioxygen (O2) atmosphere at room temperature is of significant interest in the field of catalysis. Herein, we present a highly active and selective aerobic epoxidation of olefins using metalloenzyme-like catalysts based on a non-heme ligand, tris(2-pyridylmethyl)amine (TPA). Notably, manganese chloride complexed with TPA (Mn(TPA)Cl2) demonstrated excellent activity for the epoxidation of trans-stilbene using O2 as the oxidant in the presence of a co-reductant at 30 °C. A quantitative conversion of 99% and high yield of 98%, as determined by gas chromatography using an external standard method, were achieved under optimum reaction conditions. Furthermore, Mn(TPA)Cl2 exhibited a good substrate tolerance to styrene derivatives with electron-withdrawing or electron-donating groups, cyclic olefins with different substituents and substitution degrees, as well as long-chain olefins. Coupled with a high turnover frequency (TOF) of up to 30,720 h−1, these results underscore the potential of Mn(TPA)Cl2 as a promising metalloenzyme-like catalytic platform for the aerobic synthesis of diverse epoxides from olefins under ambient conditions.
Full article

Graphical abstract
Open AccessArticle
Evaluation of Natural Dye Extracts from African Plants for the Photooxygenation of α-Terpinene to the Anthelmintic Ascaridole
by
Chinyere Chidimma Enyi, Gloria Ihuoma Ndukwe, Godswill Kuta Fekarurhobo and Michael Oelgemöller
Organics 2026, 7(1), 3; https://doi.org/10.3390/org7010003 - 5 Jan 2026
Abstract
►▼
Show Figures
In this study, the singlet oxygen photosensitization potential of three natural African plant extracts was investigated using the photooxygenation of α-terpinene (1). Utilizing visible light, the Carpolobia lutea extract achieved high conversions towards the anthelmintic ascaridole (2) of >60%
[...] Read more.
In this study, the singlet oxygen photosensitization potential of three natural African plant extracts was investigated using the photooxygenation of α-terpinene (1). Utilizing visible light, the Carpolobia lutea extract achieved high conversions towards the anthelmintic ascaridole (2) of >60% after 90 min of irradiation, while the extracts of Hibiscus sabdariffa and Justicia secunda failed to induce significant photoreactivity. Quenching using 1,4-diazabicyclo[2.2.2]octane (DABCO) confirmed a singlet oxygen pathway for irradiation with the C. lutea extract. Further separation of the C. lutea extract and subsequent photooxygenation screening established several active fractions for ascaridole generation. Advanced HPLC–MS analyses of these active fractions revealed several photosensitizing constituents. These findings establish C. lutea extract as a sustainable and effective photosensitiser with comparable performance to commercial dyes.
Full article

Graphical abstract
Open AccessArticle
Helical Molecular Cages with sp-Conjugated Linkages
by
Wei Wu, Takahiro Kojima and Hiroshi Sakaguchi
Organics 2026, 7(1), 2; https://doi.org/10.3390/org7010002 - 25 Dec 2025
Abstract
►▼
Show Figures
A conjugated helical cage, comprising two 1,3,5-tris(phenylethynyl)benzene units connected by diyne linkers, was successfully synthesized. X-ray crystallography revealed helical molecular structures with large twisted angles and a 1:1 mixture of P- and M-enantiomers. Variable-temperature-NMR measurement indicated the racemization process between the enantiomers
[...] Read more.
A conjugated helical cage, comprising two 1,3,5-tris(phenylethynyl)benzene units connected by diyne linkers, was successfully synthesized. X-ray crystallography revealed helical molecular structures with large twisted angles and a 1:1 mixture of P- and M-enantiomers. Variable-temperature-NMR measurement indicated the racemization process between the enantiomers occurs rapidly on the NMR timescale. The rapid interconversion is attributed to the flexible diyne linkages, even though they were believed to be rigid.
Full article

Graphical abstract
Open AccessArticle
Photodynamic Agents of Synthetic Curcuminoids with Antibacterial and Anticancer Activities
by
Sung-Jen Hung, Lo-Yun Chiang, Yi-An Hong, Kai-Chih Chang, Yang-Je Cheng, Hsin-Ying Wu, Hussana Hamid, Anren Hu, Tzenge-Lien Shih and Hao-Ping Chen
Organics 2026, 7(1), 1; https://doi.org/10.3390/org7010001 - 23 Dec 2025
Abstract
►▼
Show Figures
Our previous study demonstrated that thiophene-substituted synthetic curcumin analogs possessed better antibacterial activity and stability than natural curcumin, demethoxycurcumin, or bisdemethoxycurcumin in antibacterial photodynamic therapy (aPDT). In addition, the activity of the furan-substituted analogs was weaker than that of the thiophene-substituted compounds. As
[...] Read more.
Our previous study demonstrated that thiophene-substituted synthetic curcumin analogs possessed better antibacterial activity and stability than natural curcumin, demethoxycurcumin, or bisdemethoxycurcumin in antibacterial photodynamic therapy (aPDT). In addition, the activity of the furan-substituted analogs was weaker than that of the thiophene-substituted compounds. As oxygen, sulfur, and selenium belong to the same group in the periodic table, the antibacterial and anticancer activities of these three different elemental analogs were compared and investigated. The thiophene-substituted analog (compound 3) exhibited the most potent antibacterial activity in aPDT experiments. However, the furan-substituted analog (compound 1) exhibited the most potent anticancer activity. These results indicate that the differences in atomic radii or energy levels in these compounds produce different cell-attack results on generated free radicals. Ruthenium(II) complexes have a good reputation for use in PDT for cancer treatment. Our results show that complexation of ruthenium(II) with thiophene-substituted curcumin analogs does not enhance their antibacterial or anticancer activity.
Full article

Graphical abstract
Open AccessArticle
Tranexamic Acid-Phenol Smart Scaffolds with Imine Linker: Unlocking Antimicrobial Potential Through In Vitro and In Silico Insights
by
Jovana S. Dragojević, Žiko Milanović, Kristina Milisavljević, Nevena Petrović, Jelena Petronijević, Nenad Joksimović, Vera M. Divac, Marijana Kosanić and Marina D. Kostić
Organics 2025, 6(4), 54; https://doi.org/10.3390/org6040054 - 16 Dec 2025
Cited by 1
Abstract
►▼
Show Figures
A novel series of Schiff bases (3a–3k), incorporating tranexamic acid (TXA) and phenol-derived aldehydes via imine linkers, was synthesized and structurally characterized. The antimicrobial activity of the compounds was evaluated against a range of clinically and environmentally relevant bacterial
[...] Read more.
A novel series of Schiff bases (3a–3k), incorporating tranexamic acid (TXA) and phenol-derived aldehydes via imine linkers, was synthesized and structurally characterized. The antimicrobial activity of the compounds was evaluated against a range of clinically and environmentally relevant bacterial and fungal strains. Among them, derivatives 3i and 3k, bearing bromine and chlorine substituents on the phenol ring, exhibited the most potent antimicrobial effects, particularly against Penicillium italicum and Proteus mirabilis (MIC as low as 0.014 mg/mL). To elucidate the underlying mechanism of action, in silico molecular docking studies were conducted, revealing strong binding affinities of 3i and 3k toward fungal sterol 14α-demethylase (CYP51B), with predicted binding energies surpassing those of the reference antifungal ketoconazole. Additionally, UV-Vis and fluorescence spectroscopy assays demonstrated good stability of compound 3k in PBS and its effective binding to human serum albumin (HSA), respectively. ADMET and ProTox-II predictions further supported the drug-likeness, low toxicity (Class 4), and favorable pharmacokinetic profile of compound 3k. Collectively, these findings highlight TXA–phenol imine derivatives as promising scaffolds for the development of next-generation antimicrobial agents, particularly targeting resistant fungal pathogens.
Full article

Graphical abstract
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Sci, Spectroscopy Journal, Chemistry, Organics, Catalysts, Molecules, Photochem
Photogenerated Intermediates: Spectral Capture, Theoretical Characterization and Chemical Reactivity
Topic Editors: Rui Fausto, Licinia L. G. JustinoDeadline: 31 December 2026
Topic in
Atoms, Crystals, Molecules, Organics, Symmetry, Inorganics
Advances in Molecular Symmetry and Chirality Research
Topic Editors: Ralph N. Salvatore, Guzman Gil-RamirezDeadline: 18 January 2027
Topic in
Molecules, Organics, Chemistry, Plants, Polysaccharides, Foods, Pharmaceuticals, Sci
Biological and Pharmacological Activity of Natural Products
Topic Editors: Laura De Martino, Yajun ZhengDeadline: 31 January 2027
Topic in
Agriculture, Chemistry, Foods, Molecules, Organics, Sustainability, Sustainable Chemistry, Sci
Valorization of Natural Products and Agro-Food Residues
Topic Editors: Isabel Maria Duque Martins, Madalena M. DiasDeadline: 31 July 2027

Conferences
Special Issues
Special Issue in
Organics
Advanced Oxidation Processes for Efficient Removal of Organic Pollutants in Water Treatment
Guest Editor: Yan WangDeadline: 10 June 2026
Special Issue in
Organics
Organic Supramolecular Chemistry of Natural Products
Guest Editor: Ruilong ShengDeadline: 30 September 2026
Special Issue in
Organics
Recent Advances in Selective Oxidation
Guest Editors: Lei Liu, Xigong LiuDeadline: 30 September 2026
Special Issue in
Organics
Recent Advances in Asymmetric Transfer Hydrogenation
Guest Editors: Zbigniew Czarnocki, Zuzanna MolędaDeadline: 30 September 2026
Topical Collections
Topical Collection in
Organics
Advanced Research Papers in Organics
Collection Editors: Wim Dehaen, Michal Szostak, Huaping Xu