Journal Description
Electrochem
Electrochem
is an international, peer-reviewed, open access journal on electrochemistry published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 20.7 days after submission; acceptance to publication is undertaken in 3.8 days (median values for papers published in this journal in the second half of 2025).
- Journal Rank: CiteScore - Q1 (Materials Chemistry)
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Journal Cluster of Chemical Reactions and Catalysis: Catalysts, Chemistry, Electrochem, Inorganics, Molecules, Organics, Oxygen, Photochem, Reactions, Sustainable Chemistry.
Latest Articles
TiO2-Doped Hydrochar Derived from Phoenix dactylifera: Synthesis and Electrocatalytic Performance for Alkaline Hydrogen Production
Electrochem 2026, 7(2), 12; https://doi.org/10.3390/electrochem7020012 - 14 May 2026
Abstract
►
Show Figures
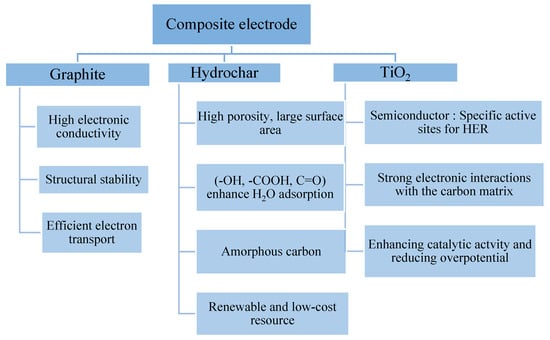
Cost-effective, durable, and environmentally friendly electrocatalysts to be used for the alkaline hydrogen evolution reaction (HER) represent one of the key challenges facing green hydrogen generation. In this context, a TiO2-doped hydrochar derived from Phoenix dactylifera L. Deglet Nour (date pits)
[...] Read more.
Cost-effective, durable, and environmentally friendly electrocatalysts to be used for the alkaline hydrogen evolution reaction (HER) represent one of the key challenges facing green hydrogen generation. In this context, a TiO2-doped hydrochar derived from Phoenix dactylifera L. Deglet Nour (date pits) was synthesized and incorporated into a graphite-based electrode to improve HER performance in a 1 M KOH solution. Three TiO2 loadings (1, 3, and 6 wt%) were systematically studied and compared using electrochemical techniques to evaluate the influence of oxide incorporation on HER kinetics. In parallel, physicochemical characterization analyses were performed to acquire an in-depth understanding of the morphology, composition, and surface properties of biomass-derived carbonaceous materials and to establish correlations with their electrochemical behavior. The G/HC-3% TiO2 electrode exhibited the most pronounced electrocatalytic performance, with an overpotential of 194 mV at −10 mA·cm−2 and a Tafel slope of 67 mV·dec−1, indicating favorable interfacial charge transfer kinetics. The present work demonstrates that biomass-derived TiO2-doped hydrochar has significant potential as a sustainable and high-performance electrocatalyst in alkaline water electrolysis.
Full article
Open AccessCorrection
Correction: Zhang et al. Graphene-Oxide-Coated CoP2@C Anode Enables High Capacity of Lithium-Ion Batteries. Electrochem 2023, 4, 473–484
by
Wei Zhang, Hangxuan Xie, Zirui Dou, Zhentao Hao, Qianhui Huang, Ziqi Guo, Chao Wang, Kanghua Miao and Xiongwu Kang
Electrochem 2026, 7(2), 11; https://doi.org/10.3390/electrochem7020011 - 8 May 2026
Abstract
The authors would like to make the following corrections about the published paper [...]
Full article
Open AccessArticle
Frequency Dependence of Effective Capacitance Cec for Polyaniline Membrane-Based pH Sensor and its Extension to the Gouy–Chapman–Stern Model
by
Tingting Han, Tao Song, Junyu Gan, Dongxue Han and Li Niu
Electrochem 2026, 7(2), 10; https://doi.org/10.3390/electrochem7020010 - 7 May 2026
Abstract
►▼
Show Figures
This study proposed an effective capacitance (Cec) for bare and conducting polymer-covered electrodes using electrochemical impedance spectroscopy (EIS). Bare electrodes show three regimes: potential-dependent Helmholtz capacitance, Gouy–Chapman–Stern diffusion capacitance (1 MHz–10 Hz), and complex low-frequency responses, deviating from semi-infinite Warburg
[...] Read more.
This study proposed an effective capacitance (Cec) for bare and conducting polymer-covered electrodes using electrochemical impedance spectroscopy (EIS). Bare electrodes show three regimes: potential-dependent Helmholtz capacitance, Gouy–Chapman–Stern diffusion capacitance (1 MHz–10 Hz), and complex low-frequency responses, deviating from semi-infinite Warburg diffusion (1 Hz–10 MHz). Polyaniline (PANI) and poly(3,4-ethylenedioxythiophene) PEDOT-based electrodes exhibit larger potential-dependent diffusion pseudocapacitance (1 MHz–10 Hz) and the absence of a Warburg tail or a nearly horizontal low-frequency slope at 0.01–0.026 (1 Hz–10 MHz). A high-frequency Cec of a PANI membrane correlates with bulk electrolyte concentration, while bare electrodes are less affected and dominated by Helmholtz capacitance. The equivalent circuit of the time-dependent EIS impedance spectrum for bare electrodes and PANI and PEDOT-based electrodes shows parallel capacitor behavior in combination with high-frequency capacitance (1 MHz–10 Hz) and a low-frequency response (1 Hz–10 MHz). The mathematical simulation of effective capacitance Cec with respect to time period t (f−1) follows two time constants (τ = RC), representing double-layer capacitance or pseudocapacitance (τ1) and complex low-frequency responses or Warburg diffusion (τ2) for bare electrodes and conducting polymer-based electrodes, respectively. This simulation analysis also elucidates the frequency dependence of the Warburg characteristic frequency (ω) and the extension of the double-layer capacitance diffuse distance LD for H+ with GC electrodes to approximately 0.74~1.51 mm over a time interval of ca. 1 s (t = f−1). The diffusion coefficient Di of K+ ion transfer through a PEDOT solid contact from 1 mC (0.1 µm) to 10 mC (1 µm) is in the range of 0.57–12 × 10−14 cm2·s−1, following a power law with an exponent of 1.75 with respect to the polymerization time of PEDOT, which is inconsistent with Fick’s law.
Full article

Graphical abstract
Open AccessArticle
Machine Learning-Based Modeling and Multi-Objective Optimization of Direct Urea–Hydrogen Peroxide Fuel Cell
by
Phan Khanh Thinh Nguyen, Thi Thu Ha Tran and Tamirat Redae Gebreselassie
Electrochem 2026, 7(2), 9; https://doi.org/10.3390/electrochem7020009 - 15 Apr 2026
Abstract
►▼
Show Figures
Direct urea–hydrogen peroxide fuel cells (DUHPFCs) are promising for sustainable power generation, but their performance is governed by highly nonlinear material and operating interactions. This study develops a machine-learning framework employing a multi-output artificial neural network (ANN) to predict cell voltage, power density
[...] Read more.
Direct urea–hydrogen peroxide fuel cells (DUHPFCs) are promising for sustainable power generation, but their performance is governed by highly nonlinear material and operating interactions. This study develops a machine-learning framework employing a multi-output artificial neural network (ANN) to predict cell voltage, power density (PD), and substrate-based energy efficiency (SEE) of DUHPFCs. The ANN exhibits excellent predictive accuracy, achieving coefficients of determination (R2) above 0.995 and normalized root mean square errors (NRMSE) below 1.75 × 10−2 for all outputs. Model interpretability is enhanced by using Shapley additive explanations and partial dependence plots, which identify current density as the dominant factor affecting DUHPFC performance, followed by temperature and anolyte composition. The ANN is coupled with a multi-objective Pareto-search algorithm optimization (PAO) to resolve the trade-offs among competing performance metrics. Under different optimization objectives, a DUHPFC with an Ni0.2Co0.8/Ni-foam anode is predicted to achieve a maximum PD of 45.6 mW/cm2 with a low SEE of 2.6% or a maximum SEE of 15.2% with a moderate PD of 40.9 mW/cm2. Additionally, a balanced operating regime is identified, achieving a PD of 43.1 mW/cm2 and an SEE of 13.9%. Overall, the proposed framework provides an effective decision-support tool for optimizing DUHPFC performance under competing objectives.
Full article

Figure 1
Open AccessArticle
Non-Idealities in Mott–Schottky Analysis of CdSe (Photo)electrodes
by
Dionysios S. Karousos, Panagiotis Priftis and Mirtat Bouroushian
Electrochem 2026, 7(2), 8; https://doi.org/10.3390/electrochem7020008 - 7 Apr 2026
Abstract
►▼
Show Figures
CdSe-coated electrodes, formed by electrodeposition of CdSe barrier layers on metallic Ti or porous TiO2 substrates, were characterized by electrochemical impedance spectroscopy in a (photo)cell using aqueous redox electrolytes based on the sulfide/polysulfide or ferro/ferricyanide couples. The influence of electrode material properties,
[...] Read more.
CdSe-coated electrodes, formed by electrodeposition of CdSe barrier layers on metallic Ti or porous TiO2 substrates, were characterized by electrochemical impedance spectroscopy in a (photo)cell using aqueous redox electrolytes based on the sulfide/polysulfide or ferro/ferricyanide couples. The influence of electrode material properties, electrolyte contact, thermal annealing, and measurement conditions (illumination, frequency, potential-scan speed) on the shape and features of Mott–Schottky plots was investigated. The obtained information was evaluated on the basis of the ideal Schottky diode model and photocurrent voltammetry data. Deviations from linear diode behavior and uncertainties in the determination of energetic parameters were examined and attributed to the presence of donor density gradients and surface states in the semiconductor electrode, further complicated by chemical corrosion. The origin of the observed non-idealities is inquired into, and specific aspects of the measuring procedure related to the non-stationary character of the interface are discussed.
Full article

Figure 1
Open AccessArticle
The Optimization of Non-Uniformity for Copper Electrodeposition with a Tertiary Current Distribution Numerical Model
by
Cheng-Xuan Xiao, Chun-Gu Liu, Cao-Feng Chen, Ping-Feng Yang, Jen-Kuang Fang and Hou-Chien Chang
Electrochem 2026, 7(2), 7; https://doi.org/10.3390/electrochem7020007 - 26 Mar 2026
Abstract
►▼
Show Figures
This study investigates the non-uniformity (NU%) of copper deposition in a three-dimensional panel electroplating cell using COMSOL Multiphysics® 6.1 (COMSOL Inc., Burlington, MA, USA). To ensure the accuracy of the simulated current efficiency, the modeling was initially conducted on the
[...] Read more.
This study investigates the non-uniformity (NU%) of copper deposition in a three-dimensional panel electroplating cell using COMSOL Multiphysics® 6.1 (COMSOL Inc., Burlington, MA, USA). To ensure the accuracy of the simulated current efficiency, the modeling was initially conducted on the electrodeposition of nanoscale metal wires (Nanowires, NWs) using the Finite Element Method (FEM) in COMSOL. After verifying that the simulation accurately reflected the current efficiency at the nanoscale, the model was scaled up to simulate full-sized panel-level electroplating. Various simulation conditions were explored, including two dimensional and three dimensional, electrode kinetics equations, electrolyte compositions, and current densities. The effects of these parameters on current efficiency and deposition uniformity were analyzed to develop a highly accurate COMSOL model. In terms of electrode kinetics, the study compares the advantages and limitations of secondary current distribution and tertiary current distribution models found in the previous literature, and evaluates their simulation results. Furthermore, to reflect the experimental condition where a pre-deposited copper seed layer was applied to reduce internal cathode resistance, the electrode shell physics module in COMSOL was implemented to simulate the potential distribution across the cathode surface. The results confirm that the numerical model using the tertiary current distribution provides more accurate predictions compared to the conventional secondary current distribution approach.
Full article

Figure 1
Open AccessArticle
Mitigating Galvanic Corrosion of Molybdenum Diffusion Barriers in Chemical Mechanical Planarization of Copper Interconnects: A Case Study Using Imidazole in a Citrate Slurry of Neutral pH
by
Kassapa U. Gamagedara and Dipankar Roy
Electrochem 2026, 7(1), 6; https://doi.org/10.3390/electrochem7010006 - 14 Mar 2026
Abstract
►▼
Show Figures
Molybdenum (Mo) is currently considered as a potential diffusion barrier material for copper (Cu) interconnects, and these interconnect structures are generally processed using the technique of chemical mechanical planarization (CMP). While a limited number of publications on Mo CMP are presently available, the
[...] Read more.
Molybdenum (Mo) is currently considered as a potential diffusion barrier material for copper (Cu) interconnects, and these interconnect structures are generally processed using the technique of chemical mechanical planarization (CMP). While a limited number of publications on Mo CMP are presently available, the considerations for mitigating CMP-induced galvanic corrosion of Mo have remained largely underexplored. Using a model CMP system in pH-neutral slurries of citric acid with silica abrasives, the present work demonstrates how Mo barrier lines in contact with Cu wires in the CMP environment can develop CMP defects of galvanic corrosion. Including imidazole in the slurry considerably reduces the galvanic current of this corrosion process. The mechanisms of galvanic inhibition and material removal are examined by employing strategic tribo-electrochemical measurements. Open-circuit potential and potentiodynamic polarization measurements performed under surface abrasion aid the characterization of CMP-enabling surface reactions. The slurry’s surface chemistry initiates the primary modes of material wear for CMP, and corrosion-induced propagation of subsurface wear mostly governs the measured material removal rates for both Mo and Cu. Although the Cu:Mo selectivity of material removal is affected as the galvanic corrosion of Mo is suppressed, this effect can be controlled by varying the slurry content of imidazole.
Full article

Figure 1
Open AccessArticle
Reaction Diffusion Modelling of 3D Pillar Electrodes in Single-Catalyst CO2 Reduction Cascades
by
Pablo Fernandez, Marisé García-Batlle, Bo Shang, Hailiang Wang, Gregory N. Parsons, James F. Cahoon and Rene Lopez
Electrochem 2026, 7(1), 5; https://doi.org/10.3390/electrochem7010005 - 28 Feb 2026
Cited by 1
Abstract
Effective electrochemical CO2 reduction to liquid fuels requires that the local catalytic environment facilitates the desired reactivity, yet a microscopic understanding of this environment is difficult to achieve from experiment alone. In this work, a 3D reaction-diffusion model was developed to explore
[...] Read more.
Effective electrochemical CO2 reduction to liquid fuels requires that the local catalytic environment facilitates the desired reactivity, yet a microscopic understanding of this environment is difficult to achieve from experiment alone. In this work, a 3D reaction-diffusion model was developed to explore the effects of electrode surface area and local geometry on the performance of a heterogeneous catalyst that performs a two-step CO2 reduction cascade reaction to CO and then CH3OH under aqueous conditions. Kinetic parameters for the model were inspired by experimental results using a cobalt phthalocyanine (CoPc) catalyst. Three-dimensional architectures composed of arrays of square pillars with varying dimensions and either smooth or periodically modulated surfaces were tested, revealing the extent to which geometry modulates the performance of the cascade reactions. Although structural variations modulate local concentration gradients, we find that electrochemically active surface area predominantly governs the overall cascade reaction. Moreover, the results suggest that supersaturation of CO, with concentrations up to ten-fold higher than the equilibrium solubility limit, might be critical for more efficient conversion to CH3OH. For any given geometry, the spatially averaged ratio of [CO] to [CO2] is dictated by the electrochemically active surface area and determines the yield of CH3OH. For a fixed surface area, geometries that spatially confine the electrolyte yield moderate local [CO] to [CO2] ratios within small volumes. In contrast, less confining geometries result in a broader distribution of local ratios spread over larger volumes, with both configurations yielding the same spatially averaged [CO] to [CO2] ratio. These insights provide valuable design principles—highlighting the critical importance of surface area and possibly CO supersaturation—for engineering advanced electrode architectures that leverage intermediate trapping and CO supersaturation to enhance overall performance in tandem CO2 reduction systems.
Full article
(This article belongs to the Topic Electrocatalytic Advances for Sustainable Energy)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Anodic Thin Films on Gadolinium, Al/Gd and Al/Nb/Gd Systems: Morphology, Growth Mechanisms and Niobia Cork-like Effect
by
Andrei Pligovka, Sergey Zavadski, Andrei Lazavenka and Vadim Bogush
Electrochem 2026, 7(1), 4; https://doi.org/10.3390/electrochem7010004 - 20 Feb 2026
Abstract
►▼
Show Figures
The high reactivity of lanthanide metals poses a challenge to the electrochemical anodizing of surfaces for nanostructured coatings. This paper presents the first systematic experimental investigation of anodic oxidation of lanthanide gadolinium in aqueous solutions of citric, boric, oxalic, and tartaric acids. The
[...] Read more.
The high reactivity of lanthanide metals poses a challenge to the electrochemical anodizing of surfaces for nanostructured coatings. This paper presents the first systematic experimental investigation of anodic oxidation of lanthanide gadolinium in aqueous solutions of citric, boric, oxalic, and tartaric acids. The voltage-current-time responses of anodizing of gadolinium, Al/Gd and Al/Nb/Gd systems were investigated. Anodic thin films were characterized using modern analysis techniques: SEM, FIB, and EDX. Morphology and voltage-current-time response analysis of anodized Al/Nb/Gd systems made it possible to establish the niobia cork-like effect and to develop a growth model.
Full article

Graphical abstract
Open AccessArticle
Yttrium-Enhanced Passive Films in Austenitic Stainless Steel
by
Maksym Bichev, Denis Miroshnichenko, Sergey Nesterenko, Leonid Bannikov, Leonid Saienko, Volodymyr Tertychnyi, Vladislav Reivi, Kyrylo Serkiz and Mariia Shved
Electrochem 2026, 7(1), 3; https://doi.org/10.3390/electrochem7010003 - 16 Jan 2026
Cited by 1
Abstract
►▼
Show Figures
It has been demonstrated that a monomolecular surface film with semiconducting characteristics forms on an austenitic, corrosion- and heat-resistant chromium–nickel steel with 0.10 wt.% C, 20 wt.% Cr, 9 wt.% Ni, and 6 wt.% Mn (10Kh20N9G6), microalloyed with yttrium, in aqueous 1 M
[...] Read more.
It has been demonstrated that a monomolecular surface film with semiconducting characteristics forms on an austenitic, corrosion- and heat-resistant chromium–nickel steel with 0.10 wt.% C, 20 wt.% Cr, 9 wt.% Ni, and 6 wt.% Mn (10Kh20N9G6), microalloyed with yttrium, in aqueous 1 M H2SO4. This passive layer exhibits semiconducting behavior, as confirmed by electrochemical impedance and capacitance measurements. For the first time, key electronic parameters, including the flat-band potential, the thickness of the semiconductor layer, and the Fermi energy, have been determined from experimental Mott–Schottky plots obtained for the interphase boundary between the yttrium-microalloyed austenitic Cr–Ni steel (10Kh20N9G6) and aqueous 1 M H2SO4. The results reveal a systematic shift in the flat-band potential toward more negative values with increasing yttrium content in the alloy, indicating a modification of the electronic structure of the passive film. Simultaneously, a decrease in the Fermi energy is observed, suggesting an increase in the work function of the metal surface due to the presence of yttrium. These findings contribute to a deeper understanding of passivation mechanisms in yttrium-containing stainless steels. The formation of a semiconducting passive film is essential for enhancing the electrochemical stability of stainless steels, and the role of rare-earth microalloying elements, such as yttrium, in this process is of both fundamental and practical interest.
Full article

Figure 1
Open AccessArticle
Ageing and Water Detection in Hydroscopic Organic Electrolytes
by
Eva Alonso-Muñoz, Janwa El Maiss, Wejdene Gongi, Divya Balakrishnan, Delphine Faye, Karine Mougin and César Pascual García
Electrochem 2026, 7(1), 2; https://doi.org/10.3390/electrochem7010002 - 16 Jan 2026
Abstract
Electrolyte degradation and trace water contamination critically affect the lifetime and safety of lithium-ion batteries. In organic-based electrolytes such as acetonitrile (MeCN), even small amounts of water can trigger
Electrolyte degradation and trace water contamination critically affect the lifetime and safety of lithium-ion batteries. In organic-based electrolytes such as acetonitrile (MeCN), even small amounts of water can trigger
(This article belongs to the Special Issue Feature Papers in Electrochemistry)
►▼
Show Figures

Figure 1
Open AccessArticle
Synthesis and Structural Characterization of Ni/Mn-Doped Co-RGO Composites for Supercapacitor Electrodes
by
Andriono Manalu, Moraida Hasanah, Winfrontstein Naibaho, Mario Geraldi Simanjuntak and Maren Sius Girsang
Electrochem 2026, 7(1), 1; https://doi.org/10.3390/electrochem7010001 - 24 Dec 2025
Abstract
►▼
Show Figures
In this study, Ni/Mn-doped cobalt–reduced graphene oxide (Co-RGO) composites were successfully synthesized as advanced electrode materials for supercapacitors. The structural and morphological properties of the composites were characterized using FTIR, XRD, SEM, TEM, and UV–Vis spectroscopy. Their electrochemical performance was evaluated through electrochemical
[...] Read more.
In this study, Ni/Mn-doped cobalt–reduced graphene oxide (Co-RGO) composites were successfully synthesized as advanced electrode materials for supercapacitors. The structural and morphological properties of the composites were characterized using FTIR, XRD, SEM, TEM, and UV–Vis spectroscopy. Their electrochemical performance was evaluated through electrochemical impedance spectroscopy (EIS), cyclic voltammetry (CV), and galvanostatic charge–discharge (GCD). Among the prepared samples, Co-RGO doped with Ni/Mn at a 40:10 ratio exhibited the most outstanding capacitive behavior, achieving a specific capacitance of 7414 F g−1 at a current density of 10 A g−1, along with a high energy density of 565 Wh kg−1 and a power density of 4998 W kg−1. The high capacitance arises from faradaic pseudocapacitive reactions rather than electric double-layer capacitance, eliminating the need for a large surface area. These results confirm that Ni doping significantly enhances pseudocapacitance and conductivity in the Co-RGO matrix, making Ni/Mn (40:10)–Co-RGO a potential material for advanced energy storage systems.
Full article

Graphical abstract
Open AccessArticle
Experimental-Based Optimal Parameter Extraction for PEM Fuel Cell Semi-Empirical Model Using the Cloud Drift Optimization Algorithm
by
Mohamed A. El-Hameed, Mahmoud M. Elkholy, Mahfouz Saeed, Adnan Kabbani, Essa Al-Hajri and Mohammed Jufaili
Electrochem 2025, 6(4), 45; https://doi.org/10.3390/electrochem6040045 - 17 Dec 2025
Abstract
►▼
Show Figures
Accurate modeling of proton exchange membrane fuel cells (PEMFCs) is essential for predicting system performance under diverse operating conditions. This study introduces a refined semi-empirical modeling that combines experimental validation with an enhanced parameter estimation method based on the Cloud Drift Optimization (CDO)
[...] Read more.
Accurate modeling of proton exchange membrane fuel cells (PEMFCs) is essential for predicting system performance under diverse operating conditions. This study introduces a refined semi-empirical modeling that combines experimental validation with an enhanced parameter estimation method based on the Cloud Drift Optimization (CDO) algorithm. The approach focuses on identifying seven key parameters of the nonlinear PEMFC model by minimizing the difference between experimentally measured and simulated cell voltages. To assess its effectiveness, the proposed CDO-based estimator was compared with several established metaheuristic algorithms, including the particle swarm optimizer and the tetragonula carbonaria optimization algorithm. The evaluation was performed using three commercial PEMFC stacks rated at 250 W, 500 W, and the NedStack PS6, as well as experimental data obtained from the Renewable Energy Laboratory at A’Sharqiyah University. Results demonstrate that the CDO algorithm consistently produced the lowest sum of squared errors (SSE) of 1.0337 and exhibited stable convergence across multiple independent runs with a standard deviation of 1.2114 × 10−7. Its reliable performance under both normal and degraded conditions confirms the algorithm’s robustness and adaptability, establishing CDO as an efficient and dependable technique for PEMFC modeling and parameter identification.
Full article
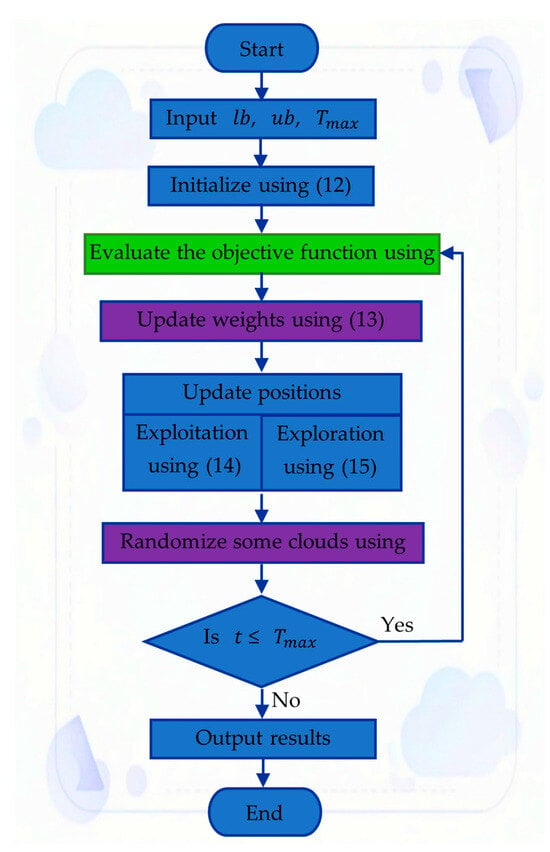
Figure 1
Open AccessReview
A Review of Recent Advances in Multivalent Ion Batteries for Next Generation Energy Storage
by
Raj Shah, Kate Marussich and Vikram Mittal
Electrochem 2025, 6(4), 44; https://doi.org/10.3390/electrochem6040044 - 10 Dec 2025
Cited by 1
Abstract
►▼
Show Figures
As demand for high-performance energy storage grows across grid and mobility sectors, multivalent ion batteries (MVIBs) have emerged as promising alternatives to lithium-based systems due to their potential for higher volumetric energy density and material abundance. This review comprehensively examines recent breakthroughs in
[...] Read more.
As demand for high-performance energy storage grows across grid and mobility sectors, multivalent ion batteries (MVIBs) have emerged as promising alternatives to lithium-based systems due to their potential for higher volumetric energy density and material abundance. This review comprehensively examines recent breakthroughs in magnesium, zinc, aluminum, and calcium-based battery chemistries, with a focus on overcoming barriers related to slow ion transport, limited reversibility, and electrode degradation. Advances in aqueous and non-aqueous electrolyte formulations, including solvation shell engineering, interfacial passivation, and dual-zone ion transport, are discussed for their role in improving compatibility and cycling stability. Particular focus is placed on three high-impact innovations: solvation-optimized Mg-ion systems for improved mobility and retention, interface-engineered Zn-ion batteries enabling dendrite-free operation, and sustainable Al-ion technologies targeting grid-scale deployment with eco-friendly electrolytes and recyclable materials. Cross-cutting insights from operando characterization techniques and AI-guided materials discovery are also evaluated for their role in accelerating MVIB development. By integrating fundamental materials innovation with practical system design, multivalent ion batteries offer a compelling path toward next-generation, safer, and more sustainable energy storage platforms.
Full article

Figure 1
Open AccessArticle
Electrochemical Behavior of Yttrium–Magnesium Intermediate Alloy Preparation Process by Molten Salt Electrolysis
by
Wenchang Shu, Fang Zhang, Jun Peng, Quanjun Zhang, Yubao Liu and Baige Sun
Electrochem 2025, 6(4), 43; https://doi.org/10.3390/electrochem6040043 - 4 Dec 2025
Abstract
►▼
Show Figures
Yttrium–magnesium alloys are commonly employed as processing additives in magnesium alloy materials. Incorporating yttrium into magnesium alloys via Y-Mg intermediate alloys not only minimizes oxidation and burn-off loss but also simplifies operational procedures. Utilizing yttrium–magnesium alloys ensures a stable composition and reliable quality
[...] Read more.
Yttrium–magnesium alloys are commonly employed as processing additives in magnesium alloy materials. Incorporating yttrium into magnesium alloys via Y-Mg intermediate alloys not only minimizes oxidation and burn-off loss but also simplifies operational procedures. Utilizing yttrium–magnesium alloys ensures a stable composition and reliable quality of magnesium alloy products, while contributing to reduced production costs and minimized environmental pollution. In this study, a molten salt co-reduction method was developed for the preparation Y-Mg intermediate alloys. The electrochemical co-reduction behaviors of Y(III) and Mg(II), as well as the transient states of Y-Mg intermediate alloys, were systematically investigated by transient electrochemical techniques. Results indicated that the reduction of Y(III) at the molybdenum (Mo) cathode is a reversible electrochemical process, whereas the reduction of Mg(II) is irreversible and diffusion-controlled. The diffusion coefficient of Y(III) and Mg(II) in the fluoride salt at 1000 °C were determined to be 3.98 × 10−5 cm2/s and 1.16 × 10−3 cm2/s, respectively. Electrochemical calculations revealed that the reduction of Y(III) involves a single-step transfer of three electrons, while Mg(II) involves a single-step transfer of two electrons. The corresponding electrode reactions are Y(III) + 3e−→Y and Mg(II) + 2e−→Mg, respectively. A Y-Mg alloy sample prepared by constant-current molten salt electrolysis primarily consists of the MgY phase with a composition of 88.38 wt% yttrium and 11.62 wt% magnesium.
Full article

Figure 1
Open AccessArticle
Influence of a Plasma Nitriding Treatment on the Corrosion Behavior of API 5L X70 Steel in Simulated Soil Solution
by
O. A. González Noriega, A. Flores Nicolás, J. Uruchurtu Chavarín, A. Torres Islas, E. C. Menchaca Campos and H. Martínez Valencia
Electrochem 2025, 6(4), 42; https://doi.org/10.3390/electrochem6040042 - 27 Nov 2025
Cited by 1
Abstract
In this work, plasma nitriding was carried out to improve the corrosion resistance of API 5L X70 steel. The process was conducted at different treatment times, 4, 6, 8, and 10 h, to determine which one provides greater resistance to corrosion. The conditions
[...] Read more.
In this work, plasma nitriding was carried out to improve the corrosion resistance of API 5L X70 steel. The process was conducted at different treatment times, 4, 6, 8, and 10 h, to determine which one provides greater resistance to corrosion. The conditions under which the nitriding was carried out were as follows: a mixture of 20% N2 and 80% H2 at 3 torr pressure, a current of 2.6 × 10−6 A, a voltage of 360 V, and the temperature inside the plasma chamber was 550 °C. The blank and nitrided materials were characterized using dispersive energy spectroscopy and scanning microscopy to study their morphology and chemical composition. In addition, open potential circuit, electrochemical impedance spectroscopy, and potentiodynamic polarization curves in simulated soil solution were performed to evaluate the materials’ corrosion resistance. The treatment achieved at 10 h presented the greatest corrosion resistance, reducing the corrosion current density up to three orders of magnitude. The thickness reached 678.75 µm for this condition.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
A New Hope for All-Diamond Electrodes? The Interdigitated Double Diamond Electrode
by
Manuel Zulla, Carolin Messerschmidt, Hanadi Ghanem, Johannes Bähr, Lukas Hegemann and Stefan Rosiwal
Electrochem 2025, 6(4), 41; https://doi.org/10.3390/electrochem6040041 - 22 Nov 2025
Cited by 1
Abstract
Nowadays, the development of efficient water treatment processes is increasingly driven by the need to provide solutions for contaminants of emerging concern. Electrochemical advanced oxidation processes (EAOPs) based on diamond electrodes can be part of innovative removal concepts. However, expensive substrates, energy-intensive chemical
[...] Read more.
Nowadays, the development of efficient water treatment processes is increasingly driven by the need to provide solutions for contaminants of emerging concern. Electrochemical advanced oxidation processes (EAOPs) based on diamond electrodes can be part of innovative removal concepts. However, expensive substrates, energy-intensive chemical vapor deposition (CVD) of diamond, and market availability complicate matters for diamond electrodes to gain traction in the water treatment sector. In addition, it has to be stated that the mining and complex processing of necessary substrates like Si, Ti, Nb, or Ta need a significant amount of fresh water, which counteracts the need for more sustainability in the field of EAOPs. In this context, a ceramic-based boron-doped diamond (BDD) electrode is presented, which addresses this dilemma. The presented concept of the so-called interdigitated double diamond electrode (iDDE) consumes 14–46% less energy in batch-mode experiments to degrade an organic model molecule compared to standard BDD technology in a poorly conductive electrolyte (κ < 350 µS/cm). Laser-induced micro-structuring of the BDD layer reduces the interelectrode spacing (IES) of the iDDE to below 50 µm. The structuring approach at the micrometer scale enables the treatment of electrically low-conductivity electrolytes more energy efficiently, while reducing the need for a supporting electrolyte or a proton exchange membrane. Degradation experiments and Raman measurements reveal different properties of an iDDE compared to standard BDD technology. The iDDE concept highlights the need to understand the significance of non-uniform current density distributions on the general electrochemical activity of BDD electrodes.
Full article
(This article belongs to the Special Issue Feature Papers in Electrochemistry)
►▼
Show Figures

Graphical abstract
Open AccessReview
Exploring DNA Nanostructures as Surface Engineering Techniques for Optimizing Nucleic Acid Biosensor Performance
by
Kepler Pyle, Naz Savranoğlu, Selin Naz Avdan and Soha Ahmadi
Electrochem 2025, 6(4), 40; https://doi.org/10.3390/electrochem6040040 - 20 Nov 2025
Abstract
►▼
Show Figures
Surface modification of nucleic acid-based electrochemical biosensors has been at the forefront of research since their inception. Effective modification ensures the optimization of the sensitivity, specificity, and stability of modern biosensors. Recent advances in DNA nanotechnology have enabled the development of novel electrochemical
[...] Read more.
Surface modification of nucleic acid-based electrochemical biosensors has been at the forefront of research since their inception. Effective modification ensures the optimization of the sensitivity, specificity, and stability of modern biosensors. Recent advances in DNA nanotechnology have enabled the development of novel electrochemical biosensor interfaces with precise assembly and high biocompatibility. In this review, we explore three strategies for enhancing biosensor performance: the integration of tetrahedral DNA nanostructures (TDNs), self-assembled monolayers (SAMs), and DNA-based hydrogels. TDNs offer well-defined geometry and controlled spatial presentation of capture probes, significantly reducing background noise and improving target accessibility. SAMs provide a robust and tunable platform for anchoring these nanostructures, enabling reproducible and chemically stable interfaces. DNA hydrogels serve as a responsive and flexible scaffold capable of signal amplification and analyte retention. These surface architectures enhance sensitivity and minimize non-specific adsorption (NSA). We discuss recent applications and experimental outcomes, highlighting how each component is driving the next generation of nucleic acid-based biosensors.
Full article

Figure 1
Open AccessArticle
Defect Engineering and Na-Ion Transport in NaMnPO4: A Computational Perspective
by
G. M. P. Dananjana Galappaththi, Poobalasingam Abiman, Poobalasuntharam Iyngaran and Navaratnarajah Kuganathan
Electrochem 2025, 6(4), 39; https://doi.org/10.3390/electrochem6040039 - 10 Nov 2025
Abstract
►▼
Show Figures
Rechargeable sodium-ion batteries (SIBs) have attracted considerable attention owing to the natural abundance and accessibility of sodium. Maricite NaMnPO4, a phosphate-based cathode material with high theoretical capacity, suffers from blocked sodium-ion diffusion channels. In this study, atomistic simulations using pair potentials
[...] Read more.
Rechargeable sodium-ion batteries (SIBs) have attracted considerable attention owing to the natural abundance and accessibility of sodium. Maricite NaMnPO4, a phosphate-based cathode material with high theoretical capacity, suffers from blocked sodium-ion diffusion channels. In this study, atomistic simulations using pair potentials and density functional theory (DFT) are employed to investigate intrinsic defect mechanisms, sodium-ion migration pathways, and the role of dopant incorporation at Na, Mn, and P sites in generating Na vacancies and interstitials. Among the intrinsic defects, the Na–Mn anti-site cluster emerges as the most favorable, exhibiting a very low formation energy of 0.12 eV, while the Na Frenkel pair (1.93 eV) is the next most stable defect, indicating that sodium diffusion is primarily facilitated by vacancy formation. Nevertheless, sodium-ion mobility in NaMnPO4 remains limited, as reflected by the relatively high migration activation energy of 1.28 eV. Among the isovalent substitutions, K is predicted to be the most favorable dopant at the Na site, whereas Ca and Cu are the most favorable at the Mn site. Thallium is identified as a promising dopant at the Mn site for generating Na vacancies that facilitate Na-ion migration, while Ge substitution at the P site is predicted to enhance the sodium content in the material.
Full article

Figure 1
Open AccessReview
Water Management Strategies for Proton Exchange Membrane Fuel Cells: A Comprehensive Review
by
Mahfouz Saeed, Mohamed A. El-Hameed, Essa Al-Hajri and Adnan Kabbani
Electrochem 2025, 6(4), 38; https://doi.org/10.3390/electrochem6040038 - 27 Oct 2025
Cited by 6
Abstract
►▼
Show Figures
Proton exchange membrane fuel cells (PEMFCs) are a promising clean energy technology due to their zero gas emissions, low operating temperature, and high efficiency. This review synthesizes research from 2015–2025 on (i) materials-level approaches (advanced/modified PFSA membranes and composite membranes) that improve water
[...] Read more.
Proton exchange membrane fuel cells (PEMFCs) are a promising clean energy technology due to their zero gas emissions, low operating temperature, and high efficiency. This review synthesizes research from 2015–2025 on (i) materials-level approaches (advanced/modified PFSA membranes and composite membranes) that improve water retention and ionic conduction, (ii) engineered gas diffusion layers and hydrophobic/hydrophilic gradients (including Janus and asymmetric GDL architectures) that facilitate directional water transport and have been shown to increase peak power density in some reports (e.g., from ≈1.17 to ≈1.89 W·cm−2 with Janus GDL designs), (iii) flow-field design strategies. This review examines the key aspects of water management in PEMFCs, including their impact on cell performance, the underlying causes of related issues, and the mechanisms of water transport within these cells. Additionally, it discusses the methods and materials used to enhance water management, highlighting recent advancements and potential directions for future research. Topics such as water transport, water flooding, and water control strategies in PEMFCs are also addressed. Both excess water (flooding) and water depletion (dehydration) can negatively influence fuel cell performance and lifespan. Particular attention is given to water dehydration, with a detailed discussion of its effects on the cathode, Anode, gas diffusion layer, catalyst layer, and flow channels.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Electrochem, Materials, Molecules, Nanomaterials, Electronic Materials, Reactions
Electrocatalytic Advances for Sustainable Energy
Topic Editors: Yang Liu, Bangwei DengDeadline: 31 December 2026
Topic in
Analytica, AppliedChem, Biosensors, Chemosensors, Electrochem, Sensors
Electrochemical Analysis/Electrochemical Sensors in Bioanalytical Applications and Environmental Monitoring
Topic Editors: Stella Girousi, Rigini M. Papi, Pavlos TziourrouDeadline: 31 March 2027
Topic in
Batteries, Electrochem, Molecules, Chemistry, Sustainable Chemistry, Liquids, AppliedChem
Ionic Liquid/Deep Eutectic Electrolytes/Poly-Based Electrolytes for Batteries
Topic Editors: Dengfeng Yu, Bo Jiang, Gongyuan ZhaoDeadline: 30 November 2027

Special Issues
Special Issue in
Electrochem
Lithium-Ion Battery Second-Life Applications and Recycling
Guest Editors: Qi Zhang, Yunlong Shang, Zhongkai ZhouDeadline: 30 June 2026
Special Issue in
Electrochem
Advanced Electrochemical Materials for Next-Generation High-Performance Batteries
Guest Editor: Tao HuangDeadline: 31 January 2027
Special Issue in
Electrochem
Electrochemically-Mediated Approaches for the Capture, Conversion, and Extraction of Environmentally Relevant Species
Guest Editor: Kyumin JangDeadline: 28 February 2027
Special Issue in
Electrochem
Electrochemical Engineering-Driven Fuel Cell Design: From Fundamentals to Industrial Systems
Guest Editors: Srikanth Ponnada, Anandhan SrinivasanDeadline: 31 March 2027
Topical Collections
Topical Collection in
Electrochem
Recent Advances in Electrochemical Sensors
Collection Editor: Masato Sone