Journal Description
Vaccines
Vaccines
is an international, peer-reviewed, open access journal on laboratory and clinical vaccine research, utilization and immunization, published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Medicine, Research and Experimental) / CiteScore - Q1 (Pharmacology (medical))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 18.1 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.4 (2024);
5-Year Impact Factor:
3.7 (2024)
Latest Articles
The Maternal Vaccine Study Protocol: A Victorian Cohort Study Evaluating Infant and Childhood Safety and Health and Developmental Outcomes After Vaccination Against Respiratory Viruses in Pregnancy
Vaccines 2026, 14(5), 449; https://doi.org/10.3390/vaccines14050449 (registering DOI) - 18 May 2026
Abstract
Objectives: Changes in public policy are eroding vaccine confidence. Previously accepted peer-reviewed evidence around vaccination and developmental outcomes for children is being questioned. Robust, methodologically sound safety data are more needed than ever to maintain consumer confidence. Establishing further safety data on infant
[...] Read more.
Objectives: Changes in public policy are eroding vaccine confidence. Previously accepted peer-reviewed evidence around vaccination and developmental outcomes for children is being questioned. Robust, methodologically sound safety data are more needed than ever to maintain consumer confidence. Establishing further safety data on infant health, development, and allergies after COVID-19 and influenza vaccination in pregnancy may improve confidence and acceptance. Methods: This is a state-wide multi-centre prospective cohort study conducted as a sub-study of the Generation Victoria birth cohort. It will examine the risk difference for infant health, developmental, and allergy outcomes between groups of mother–baby pairs who will be examined according to exposure (vaccination against a respiratory virus during pregnancy) and comparator (no vaccination against a respiratory virus). Results: Data contributing to the analysis include GenV-collected developmental, health, and allergy outcomes to 12 months of age, as well as data from state-wide linked datasets. Conclusions: This linked-data longitudinal study will provide information on health, allergy, and developmental outcomes for infants in the first year of life after influenza and COVID-19 vaccination during pregnancy. Implications for Public Health: The reporting of developmental data will be a new contribution to knowledge around outcomes after vaccination during pregnancy.
Full article
(This article belongs to the Section Vaccines and Public Health)
Open AccessArticle
Increasing HPV Vaccination Among Early Adolescents Using a Game-Based Digital Intervention: A Randomized Controlled Trial
by
Angela Chia-Chen Chen, Lihong Ou, Elizabeth Reifsnider, Kimberly Arcoleo, Ashish Amresh and Michael Todd
Vaccines 2026, 14(5), 448; https://doi.org/10.3390/vaccines14050448 (registering DOI) - 18 May 2026
Abstract
Background/Objectives: Human papillomavirus (HPV) vaccination coverage among adolescents remains below public health targets despite strong evidence of vaccine effectiveness in preventing HPV-related cancers. Digital interventions (e.g., serious games) may improve HPV vaccine uptake, but evidence for effects on vaccination behavior remains limited. Methods:
[...] Read more.
Background/Objectives: Human papillomavirus (HPV) vaccination coverage among adolescents remains below public health targets despite strong evidence of vaccine effectiveness in preventing HPV-related cancers. Digital interventions (e.g., serious games) may improve HPV vaccine uptake, but evidence for effects on vaccination behavior remains limited. Methods: This secondary analysis of a randomized controlled trial evaluated a co-designed, game-based digital intervention to increase HPV vaccine initiation among unvaccinated youth aged 11–14 years and their parents. The sample included 64 parent–adolescent dyads (33 intervention and 31 usual care dyads). The primary outcome was HPV vaccine initiation at 2-month follow-up. Results: A significantly greater proportion of adolescents in the intervention group initiated HPV vaccination compared with controls (88.5% vs. 46.2%; χ2(1) = 10.58, p = 0.001; risk difference = 0.423, 95% CI = [0.196, 0.650]). No significant between-group baseline differences were observed in parent HPV vaccination intention, knowledge, or psychosocial perceptions, although adolescent vaccination intention was higher in the intervention group. In adjusted logistic regression controlling for adolescent baseline HPV vaccination intention, intervention participants remained significantly more likely to initiate vaccination than controls (OR = 9.31, 95% CI = 2.13–40.70, p = 0.003). Intervention acceptability was high, with most parents and adolescents reporting that the game was easy to use, engaging, and relevant to vaccination decision-making. Conclusions: These findings provide preliminary evidence that a brief, family-centered, game-based digital intervention may help increase HPV vaccination initiation among adolescents. Larger trials with longer follow-up are needed to assess vaccine series completion and effectiveness across diverse settings.
Full article
(This article belongs to the Special Issue Research and Significance of Vaccines Against Sexually Transmitted Diseases)
Open AccessArticle
Social Marketing to Enhance Community Empowerment and Ownership for a Successful Implementation of the “Big Catch-Up” in Togo in 2025: A Mixed-Methods Study
by
Soliou Badarou, Aimé Serge Dali, Kokou Herbert Gounon, Lorraine Shamalla-Hannah, Amevegbe Kodjo Boko, Xavier Richard Sire and Erinna Corinne Dia
Vaccines 2026, 14(5), 447; https://doi.org/10.3390/vaccines14050447 - 18 May 2026
Abstract
Introduction: The COVID-19 pandemic disrupted immunization services in Togo, resulting in 69,672 “zero-dose” and 24,846 “under-vaccinated” children by the end of 2023. This study assessed the effectiveness, acceptability, and feasibility of a social marketing approach deployed during the 2025 Big Catch-Up initiative in
[...] Read more.
Introduction: The COVID-19 pandemic disrupted immunization services in Togo, resulting in 69,672 “zero-dose” and 24,846 “under-vaccinated” children by the end of 2023. This study assessed the effectiveness, acceptability, and feasibility of a social marketing approach deployed during the 2025 Big Catch-Up initiative in Togo. Methods: A convergent mixed-methods study was conducted in 17 priority health districts. The quantitative component compared vaccination coverage before and after the intervention using administrative data. Chi-squared test for linear trend compared district-level coverages, and statistical significance was set at p < 0.05 for all tests. The qualitative component used in-depth interviews with key informants to collect data, followed by thematic content analysis. The intervention was grounded on the social marketing framework with 4 pillars (4Ps): Product, Price, Place, and Promotion. Results: Coverage increased dramatically: Penta1 from 1% to 64%, Penta3 from 1% to 45%, MR1 from 4% to 50%, and MR2 from 6% to 49% (all p < 0.001). Togo ranked 3rd out of 24 African countries for Penta1 progress. The approach demonstrated high community acceptability, with Vaccination Monitoring Committees praised as being culturally appropriate. Key concerns included sustainability and resource constraints. Conclusions: Social marketing was associated with increased community adherence and immunization coverage improvement. However, long-term sustainability requires the institutionalization of community structures with domestic funding and continued health system strengthening.
Full article
(This article belongs to the Section Epidemiology and Vaccination)
►▼
Show Figures
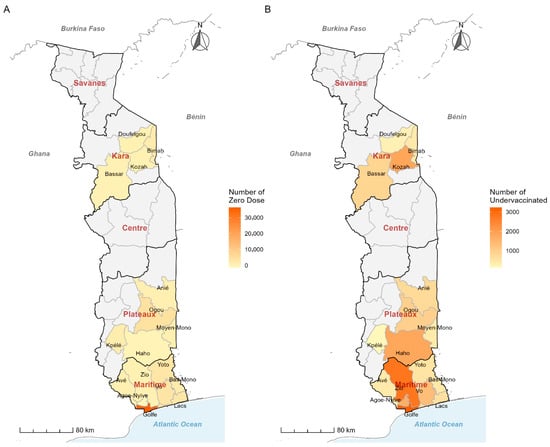
Figure 1
Open AccessArticle
Evaluation of Safety and Immunogenicity of High-Dose Quadrivalent Seasonal Influenza Split Vaccine: A Preclinical Study
by
Lanxin Jia, Ran Qiu, Jing Liu, Bo Liu, Xuanxuan Nian, Yang Le, Xixin Han, Qingmei Zhang, Xuedan Li, Zheng Gong, Ailin Shen, Zhegang Zhang, Ying Zhao and Jiayou Zhang
Vaccines 2026, 14(5), 446; https://doi.org/10.3390/vaccines14050446 - 17 May 2026
Abstract
Objectives: Seasonal influenza leads to substantial global morbidity and mortality, especially in adults aged 65 years and older, who present poor immune responses to standard-dose influenza vaccines. This study aimed to systematically evaluate the preclinical safety and immunogenicity of a high-dose quadrivalent seasonal
[...] Read more.
Objectives: Seasonal influenza leads to substantial global morbidity and mortality, especially in adults aged 65 years and older, who present poor immune responses to standard-dose influenza vaccines. This study aimed to systematically evaluate the preclinical safety and immunogenicity of a high-dose quadrivalent seasonal influenza split vaccine (HD-QIV), providing preclinical evidence for its clinical application in the elderly. Methods: Following GLP guidelines, we performed single-dose and repeated-dose toxicity tests in Sprague–Dawley rats, active systemic anaphylaxis assays in guinea pigs, and immunogenicity assessments in young and aged BALB/c mice. Safety indicators included general clinical signs, hematology, blood biochemistry, histopathology and allergic reactions; immunogenicity was evaluated via hemagglutination inhibition (HI) antibody titers and antigen-specific cellular immune responses. Results: HD-QIV only caused mild and reversible local irritation in rats without obvious systemic toxicity, and no dose-related systemic anaphylaxis was observed in guinea pigs. HD-QIV induced robust and dose-dependent humoral immune responses, and showed significantly higher HI antibody titers, earlier seroconversion and longer antibody persistence than standard quadrivalent influenza vaccine in aged mice. Cellular immunity was slightly enhanced but not the dominant protective response. Conclusions: The HD-QIV demonstrates favorable preclinical safety and superior immunogenicity, supporting its further clinical development and use as a priority vaccine for the elderly population.
Full article
(This article belongs to the Section Vaccine Advancement, Efficacy and Safety)
Open AccessArticle
Measles in the Post-COVID Era: Incidence Trends, Vaccination Coverage, Demographic and Subnational Distribution in Saudi Arabia, 2015–2024
by
Lama Alzamil
Vaccines 2026, 14(5), 445; https://doi.org/10.3390/vaccines14050445 - 16 May 2026
Abstract
Background/Objectives: The COVID-19 pandemic disrupted routine immunisation globally. Saudi Arabia presents a unique epidemiological context for measles, combining high vaccination coverage with mass pilgrimages and a large expatriate workforce. This study examined measles incidence trends, vaccination coverage, and demographic and geographic burden distribution
[...] Read more.
Background/Objectives: The COVID-19 pandemic disrupted routine immunisation globally. Saudi Arabia presents a unique epidemiological context for measles, combining high vaccination coverage with mass pilgrimages and a large expatriate workforce. This study examined measles incidence trends, vaccination coverage, and demographic and geographic burden distribution in Saudi Arabia (2015–2024), with comparative analysis against GCC countries, the Eastern Mediterranean Region (EMR), and global data. Methods: Annual incidence and vaccination coverage data were obtained from the WHO Global Health Observatory and WHO/UNICEF WUENIC; monthly, regional, age- and nationality-stratified data from the Saudi Ministry of Health Annual Statistical Book (2015–2024). Incidence was expressed per 1,000,000 population across three epochs: pre-COVID-19 (2015–2019), pandemic disruption (2020–2021), and post-COVID-19 rebound (2022–2024). Descriptive analyses included period means, percentage changes, rate ratios, and rate differences. Results: Pre-COVID-19 incidence (mean 19.7/1,000,000) remained below EMR and global averages. The pandemic produced near-complete suppression (−96.6% to 1.1/1,000,000 in 2020), exceeding global (−82.2%) and EMR (−61.2%) declines. A marked rebound occurred in 2023 (67.8/1,000,000), surpassing the pre-pandemic peak despite MCV1/MCV2 coverage above 96%. Non-Saudi nationals bore disproportionate burden in 2021 (20.7 vs. 1.1/1,000,000) and 2023 (70.4 vs. 64.8/1,000,000). Children under 15 accounted for 71.6–90.6% of annual cases, with the 5–<15-year group’s contribution rising from 12.7% (pre-COVID mean) to 27.7% in 2024. Geographic burden shifted annually with no consistently dominant region. Conclusions: Saudi Arabia’s post-pandemic rebound despite high national coverage implicates sub-population susceptibility gaps among non-national residents and school-age children, alongside importation risks from mass pilgrimage. Targeted strategies addressing demographic and geographic heterogeneity are essential to meet WHO 2030 elimination targets.
Full article
(This article belongs to the Special Issue Addressing Immunity Gaps: Strengthening Vaccine Confidence and Coverage)
►▼
Show Figures
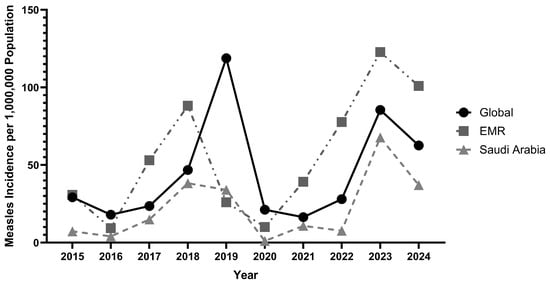
Figure 1
Open AccessArticle
Safety and Immunogenicity of the Cytomegalovirus Vaccine mRNA-1647 in Healthy Adults: Results from a Phase 2, Randomized, Controlled, Dose-Finding Trial with Long-Term Extension Follow-Up Through Month 48
by
Carlos Fierro, Daniel Brune, Richard Leggett, James Peterson, Benjamin Lorenz, Renato Calabro Calheiros, Jiang Lin, Anita S. Iyer, Kai Wu, Xin Cao, Alaknanda Kondapally, Sheila Marsh, Shiva Kalidindi, Jennifer Husson and Lori Panther
Vaccines 2026, 14(5), 444; https://doi.org/10.3390/vaccines14050444 - 16 May 2026
Abstract
Background/Objectives: No licensed vaccine against cytomegalovirus (CMV) is currently available, despite the significant risk of mother-to-infant transmission leading to serious neurodevelopmental impairment and the substantial morbidity caused by CMV infection in immunocompromised persons. We report results from a phase 2 trial of the
[...] Read more.
Background/Objectives: No licensed vaccine against cytomegalovirus (CMV) is currently available, despite the significant risk of mother-to-infant transmission leading to serious neurodevelopmental impairment and the substantial morbidity caused by CMV infection in immunocompromised persons. We report results from a phase 2 trial of the investigational CMV mRNA vaccine mRNA-1647 and a long-term extension study (NCT04232280; NCT04975893). Methods: This randomized, observer-blind, placebo-controlled phase 2 study, conducted at 9 US sites, enrolled participants in two parts. In the first part, healthy adults aged 18–40 years were stratified by baseline CMV status into CMV-seronegative and CMV-seropositive parallel cohorts and randomized 3:1 to receive mRNA-1647 (50, 100, or 150 μg) or placebo. In the second part, healthy female participants aged 18–40 years were randomized 3:1 to receive 100 μg mRNA-1647 or placebo. In both parts, vaccine or placebo was administered at Months 0, 2, and 6. Participants completing the Primary Trial through Month 18 were eligible to enroll in the extension study, wherein safety and immunogenicity were assessed every 6 months until all participants reached Month 48 (interim analysis) and a subset had Month 54 immunogenicity samples available. Primary objectives were to assess safety and neutralizing antibody responses. Results: Solicited adverse reactions were mostly grade 1 or 2 in severity, and no notable dose-related safety trends were identified. Neutralizing antibody and antigen-specific binding IgG responses were induced in CMV-seronegative participants and boosted in CMV-seropositive participants, with durability of responses through Month 48 and up to Month 54. Conclusions: The investigational vaccine mRNA-1647 was generally well tolerated and induced durable humoral immune responses across baseline CMV serostatus, with persistence supported through Month 48 and by available Month 54 data.
Full article
(This article belongs to the Section Vaccine Advancement, Efficacy and Safety)
►▼
Show Figures

Figure 1
Open AccessArticle
Structure-Guided Glycosylation of Hemagglutinin Enhances Stability and Modulates Immunogenicity of Influenza Vaccines
by
Zheng Zhang, Zhiying Xiao, Xu Zhang, Qian Ye, Xin Zhang and Wen-Song Tan
Vaccines 2026, 14(5), 443; https://doi.org/10.3390/vaccines14050443 - 15 May 2026
Abstract
Background: Antigenic drift limits the protective efficacy of influenza vaccine. Glycosylation of hemagglutinin (HA) represents a promising immunofocusing strategy that enhances neutralizing antibody responses by masking immunodominant non-neutralizing epitopes. Methods: B-cell epitopes of influenza viruses were retrieved from the Immune Epitope Database and
[...] Read more.
Background: Antigenic drift limits the protective efficacy of influenza vaccine. Glycosylation of hemagglutinin (HA) represents a promising immunofocusing strategy that enhances neutralizing antibody responses by masking immunodominant non-neutralizing epitopes. Methods: B-cell epitopes of influenza viruses were retrieved from the Immune Epitope Database and were mapped onto the HA structure of A/Puerto Rico/8/1934 (H1N1). Structure-guided analysis identified residues 136 and 137 as candidate sites for N-linked glycosylation (NLG). Single-site mutants (136NLG and 137NLG) were generated using reverse genetics and evaluated for stability, receptor binding, viral replication, and immunogenicity in a murine model with inactivated whole-virus vaccines. Results: Both mutants exhibited increased thermostability at 42 °C. Glycosylation reduced the HA–sialic acid affinity, resulting in decreased viral adsorption and internalization efficiency in MDCK cells, and delayed viral replication at low multiplicity of infection (MOI). In vivo, all vaccine groups provided complete protection against lethal challenge; notably, the 136NLG group exhibited reduced weight loss, indicating improved protective efficacy compared with wild-type (WT). Conclusions: Targeted glycosylation at residue 136 in the HA head domain effectively enhances the viral stability and elicits a 1.78-fold increase in hemagglutination inhibition titer (GMT) relative to the WT, thereby improving vaccine performance. These findings establish a rational and structure-based design strategy for developing more stable and effective influenza vaccines.
Full article
(This article belongs to the Section Influenza Virus Vaccines)
►▼
Show Figures

Figure 1
Open AccessArticle
Rv2656c: A Potential Candidate Antigen Associated with Latent Tuberculosis Infection
by
Yunjie Du, Pu He, Wenrui Dang, Ting Zhou, Yinjuan Song, Xiaoping Li, Yuhao Zhao, Fei Li, Aizhen Guo and Bingdong Zhu
Vaccines 2026, 14(5), 442; https://doi.org/10.3390/vaccines14050442 - 15 May 2026
Abstract
Background/Objectives: Several subunit vaccines for tuberculosis (TB), such as MVA85A and H4:IC31, have not demonstrated ideal protective efficacy in clinical trials, which may be attributed to their limited antigenic profile and lack of effective latency-associated antigens. In this study, we combined bioinformatics with
[...] Read more.
Background/Objectives: Several subunit vaccines for tuberculosis (TB), such as MVA85A and H4:IC31, have not demonstrated ideal protective efficacy in clinical trials, which may be attributed to their limited antigenic profile and lack of effective latency-associated antigens. In this study, we combined bioinformatics with experimental validation to screen for latency-associated antigens that have immune-protective effects. Methods: Highly expressed antigens were identified from models related to latent infections, such as hypoxia and nutritional starvation. Their physicochemical properties and immunogenicity were predicted using online tools such as Expasy-ProParam, IEBD, and VaxiJen. The immunogenicity of these antigens was then evaluated in multiple mycobacterium infection models. Finally, a systematic evaluation of the immune response and protective effects induced by the candidate antigens was performed in a mouse model using intracellular cytokine detection, mycobacterium growth inhibition assays (MGIAs), antibody-dependent cellular phagocytosis (ADCP), and a latent tuberculosis infection (LTBI) mouse model. Results: The antigen Rv2656c is highly expressed in the nutritional starvation model and demonstrates strong immunogenicity in both infected humans and cattle. Moreover, Rv2656c exerted a significant inhibitory effect against Mycobacterium tuberculosis (M. tuberculosis) and Mycobacterium avium (M. avium) infections in MGIA. The humoral immune response elicited by Rv2656c enhanced the phagocytosis and killing of Mycobacteria by macrophages in vitro. Furthermore, in a mouse model of LTBI established using the attenuated M. tuberculosis H37Ra strain, treatment with Rv2656c significantly decreased the bacterial load in the lungs of the mice. Conclusions: Latency-associated Rv2656c may serve as an immune-protective antigen, offering potential for the development of novel multi-stage antigen subunit vaccine against TB.
Full article
(This article belongs to the Special Issue Tuberculosis Diagnosis and Vaccines Research)
►▼
Show Figures

Figure 1
Open AccessArticle
TTV Viremia and Immune Responses Following Vaccination Against Mpox and Dengue Viruses
by
Claudia Minosse, Pietro Giorgio Spezia, Sara Belladonna, Aurora Bettini, Giulia Matusali, Francesca Colavita, Stefania Notari, Linda Petrone, Marta Tiberi, Alessandro Rosario Cavasio, Valentina Mazzotta, Luigi Rosa, Eleonora Cimini, Daniele Focosi, Delia Goletti, Emanuele Nicastri, Andrea Antinori and Fabrizio Maggi
Vaccines 2026, 14(5), 441; https://doi.org/10.3390/vaccines14050441 - 15 May 2026
Abstract
Background: Torquetenovirus (TTV) viremia is increasingly recognized as a biomarker of host immune competence. We assessed the association between baseline TTV DNA levels and immune responses to the Mpox virus (MPXV) and dengue virus (DGV) vaccines in two prospective cohorts. Methods:
[...] Read more.
Background: Torquetenovirus (TTV) viremia is increasingly recognized as a biomarker of host immune competence. We assessed the association between baseline TTV DNA levels and immune responses to the Mpox virus (MPXV) and dengue virus (DGV) vaccines in two prospective cohorts. Methods: A total of 248 individuals were enrolled, and TTV DNA was quantified before vaccination. Humoral and cellular responses to MVA-BN (for MPXV) and QDENGA (for DGV) vaccines were measured by using serology, neutralization assays, and interferon-γ ELISpot, and correlations with TTV viremia were investigated. Results: TTV DNA was detected in 81.2% of individuals, with a significantly higher prevalence and viral loads in the Mpox-Vac group than in the DGV-Vac group. Between both groups, the only significant association observed was an inverse correlation between pre-vaccination TTV load and DGV neutralizing antibody titers in the DGV-Vac group and was limited to the subset of TTV-positive individuals; no additional correlations with antibody and T responses were identified. For the Mpox-Vac group, stratified analyses in people living with HIV (PLWH) confirmed this lack of association. Conclusions: TTV viremia does not predict vaccine immunogenicity in immunocompetent or mildly immunosuppressed individuals. These results, which derive from within-cohort analyses and do not rely on direct comparisons between heterogeneous vaccine populations, support the role of TTV as a marker of immune status along a continuum of immunosuppression, with predictive value likely confined to populations with more severe immune impairment.
Full article
(This article belongs to the Special Issue Research on Immune Response and Vaccines: 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessReview
Bacterial Membrane Vesicles as Versatile Platforms for Systemic and Mucosal Vaccines
by
Si Hyun Park and Young Min Son
Vaccines 2026, 14(5), 440; https://doi.org/10.3390/vaccines14050440 - 15 May 2026
Abstract
Bacterial membrane vesicles (BMVs), encompassing outer membrane vesicles (OMVs) released from Gram-negative bacteria and extracellular vesicles (EVs) released from Gram-positive bacteria, have emerged as promising vaccine platforms owing to their intrinsic immunostimulatory properties and capacity to deliver a wide range of antigens. Although
[...] Read more.
Bacterial membrane vesicles (BMVs), encompassing outer membrane vesicles (OMVs) released from Gram-negative bacteria and extracellular vesicles (EVs) released from Gram-positive bacteria, have emerged as promising vaccine platforms owing to their intrinsic immunostimulatory properties and capacity to deliver a wide range of antigens. Although conventional vaccines effectively prevent infectious diseases, their long-term efficacy is often limited by antigenic variation and reliance on a restricted number of licensed adjuvants. BMVs, as self-adjuvanting systems, enable both antigen delivery and innate immune activation. BMVs are nanoscale lipid bilayer structures enriched with pathogen-associated molecular patterns (PAMPs), facilitating their recognition and uptake by antigen-presenting cells. This leads to the activation of pattern recognition receptors and the induction of pro-inflammatory cytokines, type I interferons, and adaptive immune responses, including antibody production and Th1- and Th17-biased cellular immunity. Recent studies highlight the versatility of BMVs as vaccine platforms across bacterial, fungal, and viral infection models. BMVs induce protective immunity by promoting both systemic and mucosal immune responses, thereby reducing bacterial burden and limiting pathogen colonization across diverse infection models. These properties have supported their application in viral vaccine development, including influenza and SARS-CoV-2, with the potential to enhance mucosal immunity. Despite these advantages, challenges remain in standardization, safety, and antigen-loading efficiency. Engineered BMVs incorporating protein or mRNA antigens may further enhance antigen presentation and CD8+ T cell responses. This review summarizes the biological features, immunological mechanisms, and future potential of BMVs in vaccine development.
Full article
(This article belongs to the Special Issue Vaccine Advancement, Efficacy and Safety: Feature Papers)
►▼
Show Figures

Figure 1
Open AccessArticle
Barriers to Childhood Immunisation in Selected Zero-Dose Communities in Gauteng Province: A Qualitative Study
by
Thobelani Nompilo Majola, Ntombifuthi Blose, Emma Shuvai Chikovore, Zinhle Mtwane, Algernon Africa, James Michael Burnett, Maanda Mudau, Noluthando Ndlovu, Bontle Motloung, Janine Simon-Meyer and Ashnie Padarath
Vaccines 2026, 14(5), 439; https://doi.org/10.3390/vaccines14050439 - 14 May 2026
Abstract
Background/objective: The Immunisation Agenda 2030, led by the World Health Organization, aims to ensure that people of all ages benefit from vaccination. South Africa remains committed to these goals, strengthening childhood immunisation largely through the Expanded Programme on Immunisation. However, despite progress,
[...] Read more.
Background/objective: The Immunisation Agenda 2030, led by the World Health Organization, aims to ensure that people of all ages benefit from vaccination. South Africa remains committed to these goals, strengthening childhood immunisation largely through the Expanded Programme on Immunisation. However, despite progress, the number of unvaccinated and partially vaccinated children continues to rise in some urban settings. This study sought to identify barriers to childhood immunisation in selected zero-dose urban communities in Gauteng Province. Methods: A qualitative exploratory–descriptive design was used to examine factors influencing childhood immunisation. Data were collected through seven focus group discussions and fifteen key informant interviews with purposively selected caregivers, community leaders, community health workers and healthcare workers involved in routine immunisation services at public healthcare facilities across the Cities of Johannesburg, Ekurhuleni, and Tshwane. Thematic analysis was conducted using NVivo® software. Results: The study identified multiple demand-side and supply-side barriers. Demand-side barriers included limited parental knowledge, lack of trust in the government and immunisation services, religious beliefs, migration, and perceptions of caregiver substance use. Supply-side barriers involved distance to the facility, negative healthcare worker attitudes, long queues and waiting times, and frequent vaccine stock-outs. These barriers collectively contributed to missed opportunities and decreased uptake of immunisation services. Conclusions: Childhood immunisation remains a critical and cost-effective public health intervention. The findings demonstrate the complex interplay of individual and system-level factors influencing vaccine demand, uptake and persistent zero-dose status in urban Gauteng communities. Addressing these barriers requires a comprehensive approach that strengthens community trust, improves caregiver vaccine literacy, and ensures accessible, responsive, and respectful immunisation services.
Full article
(This article belongs to the Special Issue Integrating Vaccination/Immunization Services into Universal Health Coverage)
Open AccessCorrection
Correction: Watts et al. Multi-Antigen Elephant Endotheliotropic Herpesvirus (EEHV) mRNA Vaccine Induces Humoral and Cell-Mediated Responses in Mice. Vaccines 2024, 12, 1429
by
Jessica R. Watts, Jennifer L. Spencer Clinton, Jeroen Pollet, Rongsheng Peng, Jie Tan and Paul D. Ling
Vaccines 2026, 14(5), 438; https://doi.org/10.3390/vaccines14050438 - 14 May 2026
Abstract
►▼
Show Figures
The authors would like to make the following corrections to this published paper [...]
Full article
Figure 1
Open AccessSystematic Review
A Possible Unmet Need: Pneumococcal Vaccination in the Workplaces—A Systematic Review of Invasive Pneumococcal Disease Among Shipyard Workers
by
Matteo Riccò, Luca Pipitò, Claudio Costantino, Silvio Tafuri, Chiara Noviello, Marco Bottazzoli, Paolo Manzoni, Daniel Fiacchini, Marco Falcone, Pasquale Gianluca Giuri, Davide Gori and Antonio Cascio
Vaccines 2026, 14(5), 437; https://doi.org/10.3390/vaccines14050437 - 13 May 2026
Abstract
Background: Workplace-related outbreaks of invasive pneumococcal disease (IPD) have been increasingly reported among shipyard workers, yet their epidemiological and clinical features remain incompletely characterized. This systematic review and meta-analysis aimed to synthesize available evidence on IPD outbreaks in shipyard settings. Methods:
[...] Read more.
Background: Workplace-related outbreaks of invasive pneumococcal disease (IPD) have been increasingly reported among shipyard workers, yet their epidemiological and clinical features remain incompletely characterized. This systematic review and meta-analysis aimed to synthesize available evidence on IPD outbreaks in shipyard settings. Methods: A systematic search of PubMed/MEDLINE, Scopus, EMBASE, and medRxiv was conducted up to March 2026. Observational studies reporting IPD outbreaks in shipyards were included. Pooled incidence rates and clinical outcomes were estimated using random-effects models, with heterogeneity assessed by I2 statistics. Risk of bias was evaluated using the Newcastle–Ottawa Scale. Results: Eight studies describing six outbreaks across four European countries (France, Norway, Northern Ireland, Finland; 2015–2025) were included, encompassing 131 cases among 35,623 workers. The pooled incidence was 368.9 cases per 100,000 workers with an attack rate of 2.36 per 1000 person-months for total cases, compared to 200.49 cases per 100,000 workers (95%CI 103.54–387.85) and 1.10 cases per 1000 person-months (95% CI 0.17–2.03) for laboratory confirmed cases, with considerable heterogeneity across studies. Most cases occurred in men (97.7%), with the median age ranging from 39 to 48 years. Hospitalizations occurred in 79.1% of cases, intensive care unit admission in 13.7%, and the case fatality ratio was 0.8%. Serotype 4 accounted for 67.2% of characterized isolates. Occupational exposures and shared accommodation may have contributed to transmission, although this could not be formally assessed. Conclusions: IPD outbreaks in shipyard settings are characterized by high incidence but relatively favorable outcomes, likely reflecting workforce demographics. However, considerable heterogeneity and methodological limitations across studies constrain the interpretation of pooled estimates. Preventive strategies, including vaccination and workplace-targeted interventions, should be considered as plausible public health measures, with a proactive role for occupational health services.
Full article
Open AccessArticle
Enhanced Anti-Lung Cancer Efficacy of Neo-BCV Combined with Cisplatin: Immune Activation and Tumor Microenvironment Remodeling
by
Quexun Cai, Qianli Yang, Kangrui Zhang, Zhengyue Fei, Ruochen Zhao, Tao Lu, Kecheng Xu, Zhenyi Wang and Peihua Lu
Vaccines 2026, 14(5), 436; https://doi.org/10.3390/vaccines14050436 - 13 May 2026
Abstract
Background: Lung cancer is the top cause of cancer-related mortality globally, and chemo-immunotherapy is a core therapeutic strategy for it. The novel bacterial composite vaccine (Neo-BCV) we developed previously can activate anti-tumor immunity. This study explored its synergistic anti-tumor effect with cisplatin (CDDP),
[...] Read more.
Background: Lung cancer is the top cause of cancer-related mortality globally, and chemo-immunotherapy is a core therapeutic strategy for it. The novel bacterial composite vaccine (Neo-BCV) we developed previously can activate anti-tumor immunity. This study explored its synergistic anti-tumor effect with cisplatin (CDDP), along with the underlying immunomodulatory mechanisms and molecular regulatory networks. Methods: A murine Lewis lung cancer (LLC) model was established to evaluate the efficacy of the combination therapy. Flow cytometry and multiplex cytokine assay were used to detect immune cell subsets and functional molecules in the spleen, serum and tumor tissues. RNA-sequencing (RNA-seq) was used to elucidate the molecular regulatory networks following the combination therapy in the tumor tissues. Body weight, blood indexes, serum biochemistry and H&E staining were monitored to verify biosafety. Results: Neo-BCV combined with CDDP achieved an 87.77% tumor growth inhibition rate, showing the most significant anti-tumor effect. The combination promoted DC maturation, enhanced effector immune cell infiltration, reduced immunosuppressive cells, upregulated Th1-type cytokines and downregulated CD8+ T cell surface PD-1. RNA-seq confirmed enrichment of multiple immune effector pathways, supporting tumor immune microenvironment remodeling. The combination alleviated CDDP-induced weight loss, had no obvious adverse effects on physiological indicators, and exhibited good biosafety. Conclusions: Neo-BCV combined with CDDP achieves enhanced anti-tumor efficacy and favorable biosafety in murine lung cancer models by regulating immune cell subsets and activating immune-related molecular pathways, providing a solid preclinical basis for its clinical translation in lung cancer treatment.
Full article
(This article belongs to the Section Vaccination Against Cancer and Chronic Diseases)
►▼
Show Figures
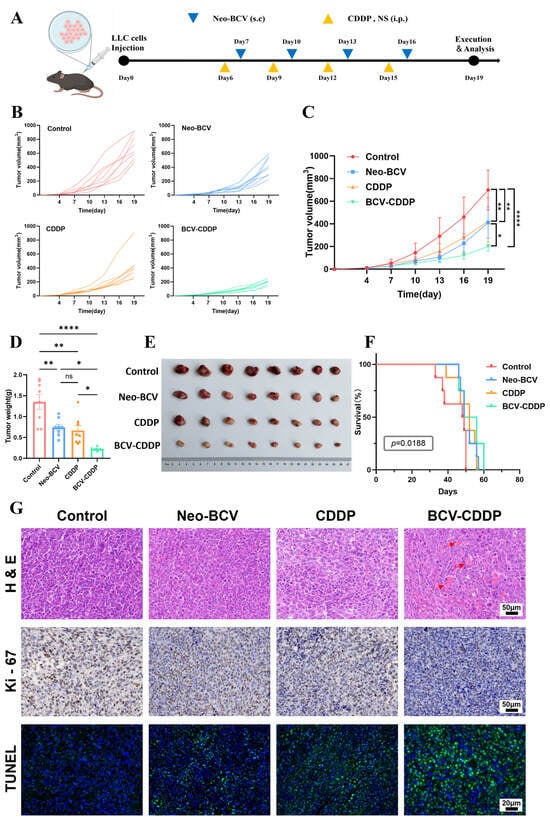
Figure 1
Open AccessReview
Immune Cell Signaling in Feline Infectious Peritonitis Virus Infection and Implications for Vaccine Design
by
Hye-Mi Lee
Vaccines 2026, 14(5), 435; https://doi.org/10.3390/vaccines14050435 - 13 May 2026
Abstract
Feline infectious peritonitis virus (FIPV) remains one of the most challenging viral diseases in veterinary medicine, largely owing to the absence of a consistently effective and safe vaccine. Despite widespread feline coronavirus infection, only a subset of infected cats progresses to feline infectious
[...] Read more.
Feline infectious peritonitis virus (FIPV) remains one of the most challenging viral diseases in veterinary medicine, largely owing to the absence of a consistently effective and safe vaccine. Despite widespread feline coronavirus infection, only a subset of infected cats progresses to feline infectious peritonitis, indicating that host immune responses are key determinants of disease outcomes. Accumulating evidence indicates that disease severity is driven not only by viral replication but also by macrophage- and monocyte-centered immune signaling, leading to excessive inflammation and systemic immunopathology in the host. Previous vaccine approaches against FIPV have failed to provide consistent protection and, in some cases, have been associated with enhanced disease. These outcomes suggest that vaccine-induced immune responses that recapitulate pathogenic signaling patterns may exacerbate disease rather than confer protection. In this review, we discuss the current knowledge of immune cell signaling pathways implicated in FIPV infection, including innate sensing through Toll-like receptors, downstream mitogen-activated protein kinases and NF-κB signaling, cytokine production profiles, Fc receptor-associated processes, and intracellular pathways such as autophagy, and how these mechanisms shape vaccine-induced immunity. By integrating insights from immune signaling kinetics, antibody functionality, adjuvant-driven pathway engagement, and platform-specific immune signatures, this review emphasizes the need to reframe FIPV vaccine development strategies that actively shape host immune responses. Rather than maximizing immunogenicity, successful vaccine design is likely to depend on limiting sustained macrophage activation and pro-inflammatory cytokine amplification while supporting antiviral immune functions, thereby reducing the risk of antibody-dependent enhancement and immunopathology. Beyond feline diseases, these considerations provide broader lessons for vaccine design in settings where immune-mediated pathology contributes to disease severity.
Full article
(This article belongs to the Section Pathogens-Host Immune Boundaries)
►▼
Show Figures
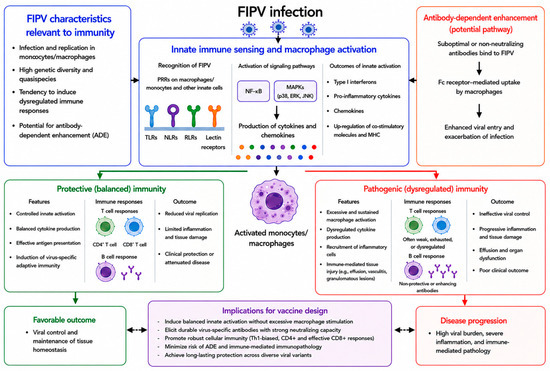
Figure 1
Open AccessEditorial
Closing Editorial—Special Issue on “Vaccines and Vaccination: HIV, Hepatitis Viruses and HPV”
by
Sonia Moretti and Ivan Schietroma
Vaccines 2026, 14(5), 434; https://doi.org/10.3390/vaccines14050434 - 12 May 2026
Abstract
We are pleased to present this closing editorial for the Special Issue “Vaccines and Vaccination: HIV, Hepatitis Viruses and HPV”, which we had the privilege of coordinating [...]
Full article
(This article belongs to the Special Issue Vaccines and Vaccination: HIV, Hepatitis Viruses, and HPV)
Open AccessCorrection
Correction: Loayza et al. Monitoring the Performance of National Immunization Programs: Innovative Methodology and Tool for Countries’ Self-Assessment. Vaccines 2026, 14, 258
by
Sergio Loayza, Bertha Capistran, Marcela Contreras, Martha Velandia and Daniel Salas
Vaccines 2026, 14(5), 433; https://doi.org/10.3390/vaccines14050433 - 12 May 2026
Abstract
The authors would like to make the following correction to this published paper [...]
Full article
(This article belongs to the Section Vaccines and Public Health)
Open AccessReview
Impact of the COVID-19 Pandemic on HPV Vaccination in Low- and Middle-Income Countries: A Scoping Review
by
Joyce Omondi, Robert Ambogo, Candy Ochieng, Marwa Farag and George Mutwiri
Vaccines 2026, 14(5), 432; https://doi.org/10.3390/vaccines14050432 - 12 May 2026
Abstract
Background: The COVID-19 pandemic caused disruptions in HPV vaccination and may have severely undermined global cervical cancer prevention, posing long-term risks to controlling cervical cancer and other HPV-related diseases. Objective: We conducted a scoping review to map and synthesize available evidence on how
[...] Read more.
Background: The COVID-19 pandemic caused disruptions in HPV vaccination and may have severely undermined global cervical cancer prevention, posing long-term risks to controlling cervical cancer and other HPV-related diseases. Objective: We conducted a scoping review to map and synthesize available evidence on how the COVID-19 pandemic has affected human papillomavirus (HPV) vaccination programs in low- and middle-income countries (LMICs) focusing on changes in vaccine delivery and coverage, determinants of uptake, economic and programmatic consequences and vaccine hesitancy. Methods: Inclusion criteria were limited to studies published in the English language between January 2020 to May 2025, and followed JBI and Arksey & O’Malley’s scoping review guidelines. The review proceeded through three stages: database searches, gray literature and citation tracking and used a PRISMA-ScR checklist to guide narrative and tabular synthesis. Results: A total of 1063 records, 57 studies were included in the final analysis, and these were spread out across 37 low- and middle-income countries (LMICs) mainly in Africa, Asia, and Latin America. Our analysis revealed that HPV vaccination coverage declined substantially during the COVID-19 pandemic, with reductions of up to 90% reported across the included studies, in the context of school closures, workforce redeployment, and supply-chain disruptions. Recovery efforts also faced major barriers including vaccine hesitancy, misinformation about COVID-19 vaccines, and travel restrictions. Strategies like digital tools, mobile clinics, and community health workers showed promise alongside integrated school- and facility-based approaches, although there is limited evidence on cost-effectiveness and long-term sustainability of these strategies. Conclusions: HPV vaccination in LMICs was significantly disrupted by the COVID-19 pandemic due to unreliable vaccine supply chains, health-worker shortages, and challenges tied to school-based vaccine delivery. Although recovery methods show potential, longer observation periods are needed to determine their full effectiveness.
Full article
(This article belongs to the Special Issue Strategies to Increase the Uptake and Delivery of HPV Vaccination in LMICs)
►▼
Show Figures
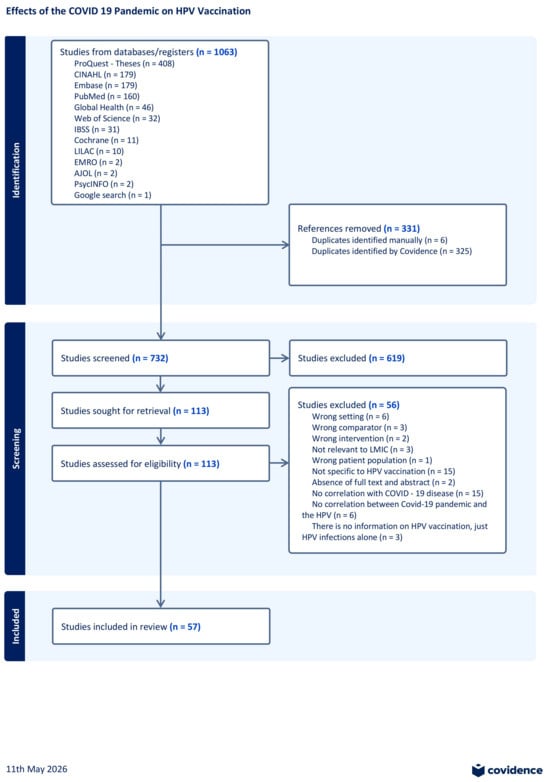
Figure 1
Open AccessArticle
Support for Tying Polio Vaccination Status to Child Tax Credit Eligibility in the United States
by
Matthew R. Boyce and Rebecca Katz
Vaccines 2026, 14(5), 431; https://doi.org/10.3390/vaccines14050431 - 12 May 2026
Abstract
Background/Objectives: In a context characterized by persistent vaccine hesitancy, shifting mandates, and stagnating immunization coverage rates, novel policy tools may be required to bolster immunization coverage in the United States. We conducted a national survey to characterize public support for a new tax
[...] Read more.
Background/Objectives: In a context characterized by persistent vaccine hesitancy, shifting mandates, and stagnating immunization coverage rates, novel policy tools may be required to bolster immunization coverage in the United States. We conducted a national survey to characterize public support for a new tax policy that would require parents to prove that their children are age-appropriately immunized against polio to be eligible for the federal child tax credit. Methods: The survey was conducted in November 2025. Respondents were asked to provide demographic information and use a Likert-scale to indicate their support for the proposed policy. Chi-squared tests and ordinal logistic regression models were used to compare support for the proposed policy across subpopulations. Results: 980 individuals were included in the analysis. 55.8% of respondents supported adding age-appropriate polio immunization to the child tax credit eligibility criteria. 20.9% of respondents opposed the policy proposal. Relative levels of support for the policy differed according to respondent gender, age, 2024 presidential election behavior, and geographic region. However, support did not differ significantly according to race, ethnicity, educational attainment, income, or partisanship. Conclusions: Results show that most survey respondents would support a policy that would add polio immunization status to the eligibility criteria for the federal child tax credit. Further, support did not differ across key demographic and political subgroups. Larger surveys should validate these findings and investigate support for adding additional vaccines to the eligibility criteria.
Full article
(This article belongs to the Special Issue New Challenges in Vaccines and Vaccination: A Public Health Perspective)
Open AccessArticle
The Yellow Fever Virus Type-Specific Epitope Recognized by Monoclonal Antibody 2D12 Neutralizes Wild Type, but Not Live Attenuated 17D or French Neurotropic Vaccine Strains
by
Clairissa A. Hansen, Shawn Rast, Jill K. Thompson, Haiping Hao, Daniel Jupiter, Stephen Higgs, Nigel Bourne and Alan D. T. Barrett
Vaccines 2026, 14(5), 430; https://doi.org/10.3390/vaccines14050430 - 12 May 2026
Abstract
Background/Objectives: The envelope (E) protein of orthoflaviviruses contains antigenic sites that are composed of one or more epitopes, which can vary in antigenic specificity, including between viral species, strains, and even substrains. Monoclonal antibodies (mAbs) that bind these epitopes vary in functionality
[...] Read more.
Background/Objectives: The envelope (E) protein of orthoflaviviruses contains antigenic sites that are composed of one or more epitopes, which can vary in antigenic specificity, including between viral species, strains, and even substrains. Monoclonal antibodies (mAbs) that bind these epitopes vary in functionality based on their specificity. This makes mAbs useful to study the differences in phenotypes between strains of viruses, such as the wild type (WT) and live attenuated vaccine strains of yellow fever virus (YFV). mAb 2D12 was raised against the 17D-204 YFV vaccine substrain virus (YF VAX®) by Schlesinger et al. in 1983. However, it only neutralizes Asibi WT virus, not the 17D-204 vaccine substrain virus. Results: We confirmed these results and demonstrated that mAb 2D12 fails to neutralize all 17D vaccine substrains (17D-204, 17DD, and 17D-213), indicating that the minor differences between these virus substrains do not affect the epitope or functionality of mAb 2D12. In addition, mAb 2D12 was found to neutralize WT strain of French viscerotropic virus (FVV), with statistically indistinguishable neutralization from the WT strain Asibi. All but one of the live attenuated French neurotropic vaccine (FNV) derivative viruses had significantly lower neutralization than WT strains Asibi and FVV. FVV, Asibi, 17D, and FNV have many amino acid differences in the membrane (M) and E proteins. It is unclear which of them contributes to this differential neutralization. However, FNV and 17D have common amino acid substitutions from WT FVV and Asibi at positions M-36 and E-331, suggesting that one or both of these residues may contribute to the 2D12 epitope. Conclusions: Overall, mAb 2D12 is a valuable tool to distinguish WT virulent strains of YFV from live attenuated vaccine strains.
Full article
(This article belongs to the Special Issue Arboviral Infections (Dengue, Chikungunya, Zika, and Other Mosquito-Borne Diseases): Immune Response and Vaccine Development)
►▼
Show Figures
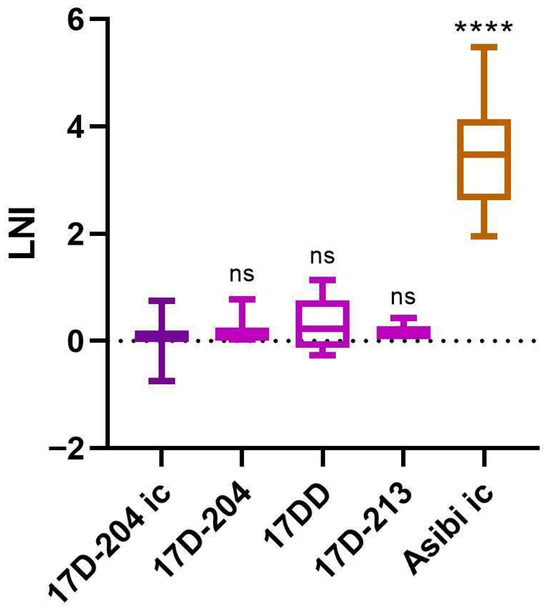
Figure 1

Journal Menu
► ▼ Journal Menu-
- Vaccines Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomolecules, Cells, IJMS, Pathogens, Vaccines, Biologics, Microorganisms, Biomedicines
Advances in Vaccines and Antimicrobial Therapy—2nd Edition
Topic Editors: Raffaele D’Amelio, Roberto PaganelliDeadline: 30 September 2026
Topic in
Animals, Arthropoda, Insects, Vaccines, Veterinary Sciences, Pathogens
Ticks and Tick-Borne Pathogens: 2nd Edition
Topic Editors: Alina Rodriguez-Mallon, Alejandro Cabezas-CruzDeadline: 31 March 2027
Topic in
Biomedicines, Cancers, IJMS, Vaccines, Viruses, JMP
Advances in HPV-Driven Head and Neck Cancer: Pathogenesis, Treatment, and Prevention
Topic Editors: Shilpi Gupta, Bhudev Chandra Das, Prabhat KumarDeadline: 20 April 2027
Topic in
Animals, Aquaculture Journal, Biology, Fishes, Pathogens, Vaccines, IJMS
Novel Insights and Advanced Research in Aquatic Animal Diseases and Immunology
Topic Editors: Erlong Wang, Zihao YuanDeadline: 30 July 2027

Conferences
Special Issues
Special Issue in
Vaccines
Vaccination and Treatments Against Viral Hepatitis: Achievements, Challenges and Perspectives, 2nd Edition
Guest Editors: Bárbara Vieira Do Lago, Vinicius MelloDeadline: 20 May 2026
Special Issue in
Vaccines
Innovative Vaccines and Technologies to Address Tropical Parasitic Diseases
Guest Editors: Cecilia Maria Perez Brandan, Iván Marcipar, Fernando Sanchez-ValdezDeadline: 30 May 2026
Special Issue in
Vaccines
Innovative Vaccines That Modulate the Immune System to Enhance Cancer Therapy
Guest Editor: Ruchi RoyDeadline: 31 May 2026
Special Issue in
Vaccines
Clinical Strategies to Improve Efficacy, Effectiveness, and Safety of Vaccination in Humans
Guest Editor: Kay Choong SeeDeadline: 31 May 2026
Topical Collections
Topical Collection in
Vaccines
Research on Monoclonal Antibodies and Antibody Engineering
Collection Editor: Tatsuya Yamazaki
Topical Collection in
Vaccines
Factors Associated with Vaccine Hesitancy
Collection Editor: Brian D. Poole
Topical Collection in
Vaccines
COVID-19 Vaccine Hesitancy: Correlates and Interventions
Collection Editors: Manoj Sharma, Kavita Batra











