-
 Investigation of Storage Conditions and Quality Control Markers for Metabolites and Lipids in Human Feces
Investigation of Storage Conditions and Quality Control Markers for Metabolites and Lipids in Human Feces -
 1H NMR-Based Metabolomics in Pediatric Acute Lymphoblastic Leukemia: A Pilot Study of Plasma and Cerebrospinal Fluid Profiles
1H NMR-Based Metabolomics in Pediatric Acute Lymphoblastic Leukemia: A Pilot Study of Plasma and Cerebrospinal Fluid Profiles -
 Sex-Specific Plasma Metabolomic Signatures in COPD Reveal Creatine, Purine/Urate, and Bile-Acid Axes
Sex-Specific Plasma Metabolomic Signatures in COPD Reveal Creatine, Purine/Urate, and Bile-Acid Axes -
 Metabolomic Study of 7-Ethyl-9-(N-methyl)aminomethyl-10-hydroxycamptothecin Derivative (NMe)—The Chemotherapeutic Drug Candidate Versus Irinotecan (IR) on a Mouse Model
Metabolomic Study of 7-Ethyl-9-(N-methyl)aminomethyl-10-hydroxycamptothecin Derivative (NMe)—The Chemotherapeutic Drug Candidate Versus Irinotecan (IR) on a Mouse Model -
 Anthropometric Indices and Markers of Atherothrombotic Risk in Subjects with Primary Hyperparathyroidism
Anthropometric Indices and Markers of Atherothrombotic Risk in Subjects with Primary Hyperparathyroidism
Journal Description
Metabolites
Metabolites
is an international, peer-reviewed, open access journal of metabolism and metabolomics, published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Biochemistry and Molecular Biology) / CiteScore - Q2 (Endocrinology, Diabetes and Metabolism)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 16.7 days after submission; acceptance to publication is undertaken in 3.6 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.7 (2024);
5-Year Impact Factor:
4.1 (2024)
Latest Articles
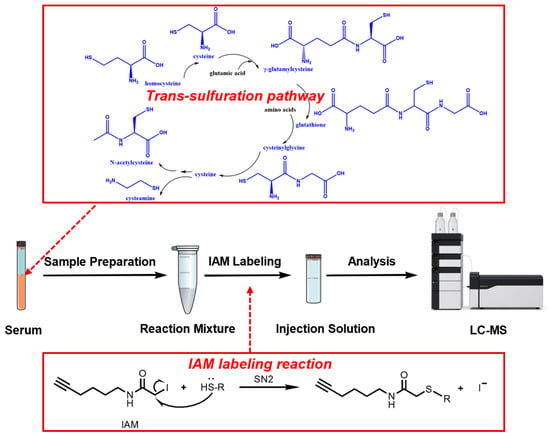
Iodoacetamine-Alkyne Derivatization-Based Liquid Chromatography–Mass Spectrometry Method for Quantification of Thiol Metabolites in Serum Samples of Hepatocellular Carcinoma Patients
Metabolites 2026, 16(5), 345; https://doi.org/10.3390/metabo16050345 - 20 May 2026
Abstract
Background/Objectives: The dysregulation of thiol metabolites is strongly linked to hepatocellular carcinoma (HCC) pathogenesis. However, quantifying these highly polar and oxidation-prone thiols in clinical serum samples via conventional liquid chromatography–mass spectrometry (LC-MS) remains challenging due to their poor sensitivity and reproducibility. Methods
[...] Read more.
Background/Objectives: The dysregulation of thiol metabolites is strongly linked to hepatocellular carcinoma (HCC) pathogenesis. However, quantifying these highly polar and oxidation-prone thiols in clinical serum samples via conventional liquid chromatography–mass spectrometry (LC-MS) remains challenging due to their poor sensitivity and reproducibility. Methods: We developed a sensitive and robust iodoacetamine-alkyne (IAM) derivatization–based LC-MS method for quantification of seven trans-sulfuration pathway thiols in human serum. Results: IAM derivatization markedly improved the method’s specificity due to enhanced chromatographic retention and diagnostic MS/MS fragments containing both the alkyne tag and analyte backbone. Sensitivity increased 33-to-160-fold versus underivatized analytes, with limits of detection of 0.02–0.1 nM. All analytes exhibited good linearity, acceptable precision with intra-day and inter-day relative standard deviations in the range of 1.2–13.8%, and high recovery from 88.6% to 102.9%. Conclusions: From the thiol quantification in human serum from 40 HCC patients and 40 healthy controls, it was found that levels of cysteine, homocysteine, glutathione, and cysteinylglycine were significantly lower in HCC patients (p < 0.05). A two-variable logistic regression model using cysteine and cysteinylglycine achieved 90.0% specificity and 80.0% sensitivity for robust HCC discrimination between HCC patients and healthy controls to some extent, with an area under the receiver operating characteristic curve of 0.88 (95% confidence interval: 0.792–0.968).
Full article
(This article belongs to the Special Issue Derivatization Techniques in Mass Spectrometry: Unlocking the Low-Abundance Metabolome)
►
Show Figures
Open AccessArticle
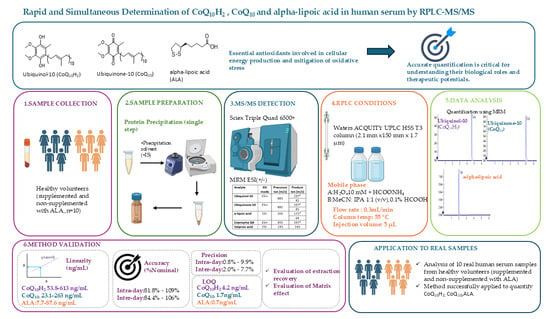
Integrated Redox Profiling: Simultaneous Determination of Ubiquinol-10, Ubiquinone-10, and Alpha-Lipoic Acid in Serum by LC-MS/MS
by
Domniki Gallou, Olga Begou, Georgios Theodoridis and Helen Gika
Metabolites 2026, 16(5), 344; https://doi.org/10.3390/metabo16050344 - 20 May 2026
Abstract
Background: Coenzyme Q10 and Alpha-lipoic acid are two essential antioxidants involved in numerous physiological processes, including cellular energy production and the mitigation of oxidative stress. Their accurate quantification is critical for understanding their biological roles and therapeutic potential. Herein, an RPLC-MS/MS
[...] Read more.
Background: Coenzyme Q10 and Alpha-lipoic acid are two essential antioxidants involved in numerous physiological processes, including cellular energy production and the mitigation of oxidative stress. Their accurate quantification is critical for understanding their biological roles and therapeutic potential. Herein, an RPLC-MS/MS method for the rapid and simultaneous determination of ubiquinone-10 (CoQ10), the reduced form ubiquinol-10 (CoQ10H2), and Alpha-lipoic acid (ALA) in human serum was developed and validated. Methods: Chromatographic separation was performed on a Waters ACQUITY UPLC HSS T3 column (2.1 mm × 150 mm, i.d. 1.7 μm). Detection was performed on a SCIEX Triple Quad 6500+ system, applying multiple reaction monitoring (MRM). Single-phase protein precipitation was selected as the sample preparation protocol, providing satisfactory recovery for the analytes. Results: The method was linear over the concentration of 53.8–613 ng/mL for CoQ10H2, 23.1–263 ng/mL for CoQ10 and 7.7–87.6 ng/mL for ALA. Intra- and inter-day accuracy was found to be between 81.8 and 109% and 84.4 to 106%, respectively, for all analytes, while intra- and inter-day precision was found to vary from 0.8% to 9.9% %RSD and 2.0% to 7.7% %RSD, respectively. A limit of quantitation (LOQ) of 4.2 ng/mL was found for CoQ10H2, 1.7 ng/mL for CoQ10 and 0.7 ng/mL for ALA. Conclusions: The developed LC-MS/MS method enables rapid, sensitive and simultaneous quantification of CoQ10H2, CoQ10, and ALA in human serum with satisfactory accuracy, precision and sensitivity. The method is suitable for bioanalytical applications and was successfully applied to the analysis of 10 real samples obtained from healthy volunteers.
Full article
(This article belongs to the Special Issue Mass Spectrometry-Based Metabolomics for Advancing Personalized Medicine)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Metabolomic Signature of Visceral Adiposity: Insights from a Population-Based Cohort
by
Khaled Naja, Najeha Anwardeen, Shamma Almuraikhy, Mohamed A. Elrayess and Ahmed Malki
Metabolites 2026, 16(5), 343; https://doi.org/10.3390/metabo16050343 - 19 May 2026
Abstract
Background: Visceral adipose tissue (VAT) is a key determinant of cardiometabolic risk, yet its underlying molecular mechanisms remain incompletely characterized. Metabolomics offers an opportunity to identify circulating biomarkers that capture VAT-related biology beyond conventional clinical measures. Methods: We conducted a cross-sectional analysis
[...] Read more.
Background: Visceral adipose tissue (VAT) is a key determinant of cardiometabolic risk, yet its underlying molecular mechanisms remain incompletely characterized. Metabolomics offers an opportunity to identify circulating biomarkers that capture VAT-related biology beyond conventional clinical measures. Methods: We conducted a cross-sectional analysis of 2526 participants from the Qatar Biobank using untargeted serum metabolomics profiling. VAT was quantified using DXA-derived estimates and analyzed both as a continuous variable and by comparing individuals in the highest quartile to the remainder quartiles. Associations between metabolites and VAT were assessed using multivariate partial least squares discriminant analysis and adjusted linear regression models controlling for age, sex, and BMI, with Bonferroni correction for multiple testing. Results: Continuous VAT was associated with 106 metabolites, while the Q4 versus Q1–Q3 contrast identified 23 metabolites, with overlapping metabolites defining a robust core VAT signature. Higher VAT was characterized by coordinated elevation of branched-chain amino acids and their keto/hydroxy acid derivatives, glutamate, and central carbon intermediates, consistent with impaired mitochondrial oxidative decarboxylation. We further identified 4-hydroxyglutamate as a novel collagen-derived metabolite positively associated with VAT, suggesting a potential link between extracellular matrix remodeling and glutamate-centered metabolism. Additionally, greater VAT was associated with lower concentrations of glycine and glycine conjugates and reduced levels of unsaturated sphingomyelins and plasmalogens. Conclusions: These findings provide potential mechanistic insights into VAT-related metabolic dysfunction and identify candidate circulating biomarkers that may enable non-invasive assessment of visceral fat-associated cardiometabolic risk. Longitudinal and mechanistic studies are warranted to establish causality and clinical utility.
Full article
(This article belongs to the Special Issue Research on Biomarkers for Cardiometabolic Risk in Metabolic Syndrome—2nd Edition)
►▼
Show Figures

Figure 1
Open AccessReview
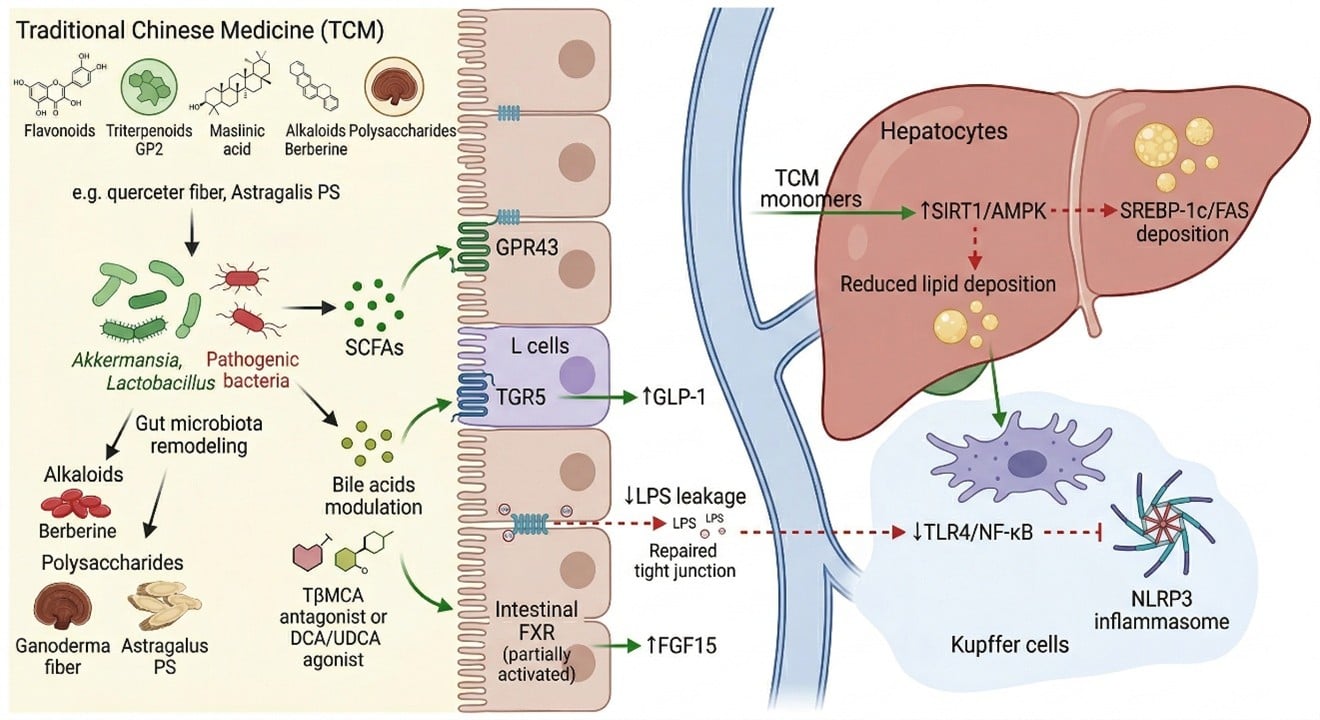
TCM-Derived Natural Compounds Targeting the Gut Microbiota in Metabolic Dysfunction-Associated Steatotic Liver Disease: Gut–Liver Axis Mechanisms, Safety Considerations, and Translational Challenges
by
Huailin Deng and Ruiqiu Zhang
Metabolites 2026, 16(5), 342; https://doi.org/10.3390/metabo16050342 - 19 May 2026
Abstract
The occurrence and development of metabolic dysfunction-associated steatotic liver disease (MASLD) are closely related to intestinal flora imbalance, intestinal barrier damage, and gut-liver axis dysfunction. Due to their multi-target regulatory effects and advantages in intestinal microecological intervention, Chinese herbal monomers have shown promising
[...] Read more.
The occurrence and development of metabolic dysfunction-associated steatotic liver disease (MASLD) are closely related to intestinal flora imbalance, intestinal barrier damage, and gut-liver axis dysfunction. Due to their multi-target regulatory effects and advantages in intestinal microecological intervention, Chinese herbal monomers have shown promising application prospects in the prevention and treatment of MASLD. However, basic research on their toxicity still lags behind, and issues related to safety and clinical translation urgently need attention. This article systematically reviews the research progress on how flavonoids, triterpenoids, alkaloids, and polysaccharides improve hepatic steatosis, inflammatory responses, and metabolic disorders from a toxicological perspective by reshaping the intestinal microbiota, repairing the intestinal mucosal barrier, regulating short-chain fatty acid and bile acid metabolism, and synergistically acting on signaling pathways such as TLR4/NF-kB, FXR, TGR5, SIRT1, and the NLRP3 inflammasome. Furthermore, by combining methods such as 16S rRNA sequencing, metagenomics, metabolomics, and multi-omics integration, the article analyzes their application value and limitations in toxicological mechanism research, and discusses the translational bottlenecks faced by Chinese herbal monomers in pharmacokinetics, bioavailability, quality standardization, targeted delivery, and toxicological safety. Existing evidence indicates that Chinese herbal monomers have a three-in-one intervention advantage of microecological remodeling-metabolic regulation-inflammation inhibition, but their long-term medication safety, toxic target organs, dose-effect/toxicity relationships, and potential drug interactions still need further clarification. This article aims to provide a systematic reference for the safety evaluation and clinical translational research of Chinese herbal monomers in the prevention and treatment of MASLD.
Full article
(This article belongs to the Special Issue Adipose Thermogenesis and Crosstalk: Signaling Networks in Obesity, Metabolic Disease and Beyond)
►▼
Show Figures
Graphical abstract
Open AccessReview
The Impact of Maternal Obesity and Diabetes on the Development of Congenital Heart Defects (CHDs) in Offspring: A Narrative Review
by
Marek Zubrzycki, Mariusz Kuśmierczyk, Jan Fritz Gummert, Angelika Costard-Jäckle, Lech Paluszkiewicz, Tobias Hecht, Ingvild Birschmann, Anna Zubrzycka and Maria Zubrzycka
Metabolites 2026, 16(5), 341; https://doi.org/10.3390/metabo16050341 - 19 May 2026
Abstract
Congenital heart disease (CHD) is the most common anatomical malformation occurring in live-born infants and an increasing cause of morbidity and mortality all over the world. Population-based observations have described associations between maternal cardiometabolic disorders and the risk of CHD in offspring. The
[...] Read more.
Congenital heart disease (CHD) is the most common anatomical malformation occurring in live-born infants and an increasing cause of morbidity and mortality all over the world. Population-based observations have described associations between maternal cardiometabolic disorders and the risk of CHD in offspring. The present article is a narrative review. The aim of this study was to review the epidemiological evidence and clinical observations relating maternal obesity and diabetes mellitus to the risk of CHD in offspring, with particular attention paid to first trimester disturbances of fetal cardiac development and the influence of genetic, epigenetic and environmental factors. Studies have shown that maternal diabetes is a risk factor associated with nearly all subtypes of CHDs in offspring, while obesity and overweight are associated with increased risk for complex defects and outflow tract obstruction and decreased risk for ventricular septal defects. Diabetes and obesity share several phenotypes, which could be transmissible from mother to fetus via the placenta. This means that an increase in maternal glucose could be responsible for the prevalence of CHD in newborns of obese women. On the other hand, maternal diabetes may induce epigenetic modifications in the developing fetus. DNA methylation changes can impact gene expression patterns relevant to heart development. The abovementioned studies are heterogenous, express different opinions and are often difficult to compare. Therefore, the results from these meta-analyses must be interpreted with caution. Optimal diabetes control is responsible for the prevention of oxidative stress in diabetic pregnancies, and a deeper understanding of maternal risk factors holds the potential to improve both prenatal detection of CHDs by identifying at-risk pregnancies and primary prevention of diseases by improving preconception management.
Full article
(This article belongs to the Section Thematic Reviews)
Open AccessArticle
Pyrroloquinoline Quinone Mitigates Type 2 Diabetes-Induced Cardiac Injury Through Mitochondrial Quality Control and Inhibition of NLRP3-Dependent Pyroptosis
by
Xue Zhang, Wei Liu, Zhijing Fu, Zhuoling Chen, Qixin Chen, Yanan Shen, Yukai Jin, Dengfeng Xu, Yin Wang, Xuefeng Qu and Yangjunna Zhang
Metabolites 2026, 16(5), 340; https://doi.org/10.3390/metabo16050340 - 19 May 2026
Abstract
Background: Pyrroloquinoline quinone (PQQ), a naturally occurring redox cofactor with potent antioxidant and anti-inflammatory properties, has been shown to protect against cardiac injury. However, its therapeutic potential in diabetic cardiomyopathy (DCM) induced by Type 2 diabetes mellitus (T2DM) and the underlying mechanisms
[...] Read more.
Background: Pyrroloquinoline quinone (PQQ), a naturally occurring redox cofactor with potent antioxidant and anti-inflammatory properties, has been shown to protect against cardiac injury. However, its therapeutic potential in diabetic cardiomyopathy (DCM) induced by Type 2 diabetes mellitus (T2DM) and the underlying mechanisms remain poorly understood. Methods: A T2DM mouse model was established via a high-fat diet and low-dose STZ. We investigated the cardioprotective effects of 12-week oral PQQ administration, assessing fasting blood glucose, oral glucose tolerance, cardiac function, myocardial histopathology, blood biochemistry, mitophagy, and NLRP3 inflammasome activation. In vitro experiments using AC16 cardiomyocytes exposed to palmitic acid and high glucose were also conducted. Results: Results showed PQQ significantly improved cardiac function, attenuated remodeling, and reduced proinflammatory cytokines in mice with T2DM, regulated key mitophagy-related proteins (Parkin, Beclin-1, LC3B-II, p62), and downregulated NLRP3 inflammasome pathway components (Caspase-1, NLRP3, IL-1β, IL-18). In vitro experiments demonstrated that PQQ reduced reactive oxygen species (ROS) production, improved mitochondrial membrane potential, promoted mitophagy, and inhibited NLRP3 inflammasome-mediated pyroptosis. Conclusions: PQQ alleviates DCM in mice with T2DM by improving mitochondrial quality control, promoting mitophagy, and subsequently inhibiting NLRP3 inflammasome-mediated pyroptosis, highlighting its potential as a promising therapeutic agent for T2DM-associated cardiomyopathy.
Full article
(This article belongs to the Section Endocrinology and Clinical Metabolic Research)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Polysaccharide from Gleditsia sinensis Seed Endosperm Ameliorates Type 2 Diabetes and Its Associated Cardiorenal Injuries by Modulating TLR4/MyD88/NF-κB Pathway and Gut Microbiota
by
Mei Liu, Wenping Liao, Hongyun Liu, Feng Xu, Yanyan Zhang, Xiangpei Wang and Hongmei Wu
Metabolites 2026, 16(5), 339; https://doi.org/10.3390/metabo16050339 - 18 May 2026
Abstract
Background: Type 2 diabetes mellitus (T2DM) represents a pressing global health challenge, underscoring the urgency of developing effective dietary interventions derived from natural resources. Zaojiaomi polysaccharide (ZJMP) from the endosperm of Gleditsia sinensis seeds (zaojiaomi), a traditional edible product, exhibits largely underexplored potential
[...] Read more.
Background: Type 2 diabetes mellitus (T2DM) represents a pressing global health challenge, underscoring the urgency of developing effective dietary interventions derived from natural resources. Zaojiaomi polysaccharide (ZJMP) from the endosperm of Gleditsia sinensis seeds (zaojiaomi), a traditional edible product, exhibits largely underexplored potential in T2DM management. Methods: In the present study, the antidiabetic effects and underlying mechanisms of ZJMP were investigated using a rat model of T2DM induced by a high-fat diet (HFD) combined with streptozotocin (STZ). Relevant biochemical indicators were detected, and histopathological examination was performed. The expression levels of key components of the TLR4/MyD88/NF-κB signaling pathway, as well as the inflammatory cytokines IL-6 and IL-1β in renal tissues, were further analyzed. Additionally, gut microbiota composition and the levels of short-chain fatty acids were determined. Results: ZJMP treatment significantly ameliorated hyperglycemia and dyslipidemia, elevated serum insulin levels, reduced intestinal mucosal permeability, and attenuated histopathological lesions in the heart, kidney, and pancreas of T2DM rats. Meanwhile, ZJMP notably alleviated renal inflammation by suppressing the production of IL-1β and IL-6, as well as inhibiting the TLR4/MyD88/NF-κB pathway. Furthermore, ZJMP administration effectively modulated gut microbiota composition and increased fecal concentrations of acetic acid and propionic acid. Conclusions: Collectively, these findings elucidate the novel bioactivity of ZJMP and highlight its potential as a promising functional food ingredient or dietary supplement for T2DM management.
Full article
(This article belongs to the Special Issue Gut Microbiota-Host Metabolic Axis: From Diet to Systemic Health)
Open AccessReview
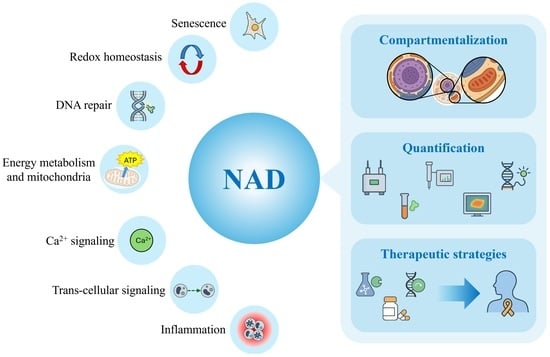
Targeting NAD Homeostasis: Compartmentalization, Quantification, and Modulation
by
Marta Nobile, Veronica Fontanini, Simone Serrao, Johannes Burtscher, Francesca Re and Giuseppe Paglia
Metabolites 2026, 16(5), 338; https://doi.org/10.3390/metabo16050338 - 18 May 2026
Abstract
Nicotinamide adenine dinucleotide (NAD+) and its reduced form, NADH, are essential coenzymes that play central roles in cellular redox homeostasis, energy metabolism, DNA repair, and signaling. Cellular NAD+ levels are maintained by a dynamic balance between the de novo Preiss–Handler,
[...] Read more.
Nicotinamide adenine dinucleotide (NAD+) and its reduced form, NADH, are essential coenzymes that play central roles in cellular redox homeostasis, energy metabolism, DNA repair, and signaling. Cellular NAD+ levels are maintained by a dynamic balance between the de novo Preiss–Handler, and salvage synthesis pathways, and consumption by enzymes like sirtuins, PARPs, and CD38. Among these, the nicotinamide Phosphoribosyltransferase (NAMPT)-driven salvage pathway represents the predominant route of NAD+ synthesis. The specific regulation of NAD (NAD+ and NADH) levels across distinct subcellular compartments has emerged as a critical determinant of cellular function but it remains poorly understood. Dysregulation of NAD metabolism is a hallmark of aging and various pathologies, including cancer, neurodegenerative disorders, and metabolic diseases, making strategies to modulate NAD levels a promising therapeutic frontier. This review provides the first integrated overview of NAD concentrations across cellular compartments (cytosol, mitochondria, nucleus, endoplasmic reticulum, Golgi, peroxisomes, and the extracellular space) together with measurement and modulation strategies. We summarize current knowledge on NAD distribution within organelles, address key challenges in accurate quantification, and highlight established and emerging approaches for both global and compartment-specific analysis. Finally, we discuss therapeutic strategies, from NAD+ precursor supplementation to enzyme modulators and gene therapy, highlighting both their translational potential and current limitations in treating diverse diseases and prolonging life and health span.
Full article
(This article belongs to the Section Cell Metabolism)
►▼
Show Figures
Graphical abstract
Open AccessPerspective
Hypothesis of the Causal Mechanisms Between Gut Microbiota and Neurodegenerative Diseases: An Elucidation from Evolutionary Perspective and Metabolic Consideration
by
Guangyan Tang, Liwen Guo, Zhiwei Liu and Yuan Quan
Metabolites 2026, 16(5), 337; https://doi.org/10.3390/metabo16050337 - 18 May 2026
Abstract
Growing evidence links gut microbiota dysbiosis to neurodegenerative diseases (NDs) such as Alzheimer’s disease and Parkinson’s disease, yet the field remains dominated by correlational observations rather than experimentally validated causal mechanisms. In this hypothesis-generating Perspective, we propose that causal inference in microbiota-associated neurodegeneration
[...] Read more.
Growing evidence links gut microbiota dysbiosis to neurodegenerative diseases (NDs) such as Alzheimer’s disease and Parkinson’s disease, yet the field remains dominated by correlational observations rather than experimentally validated causal mechanisms. In this hypothesis-generating Perspective, we propose that causal inference in microbiota-associated neurodegeneration may be strengthened by combining two complementary lenses: evolutionary biomedicine and microbial metabolism. Because evolutionary information carries intrinsic temporal and causal structure, it can provide biological prior knowledge for inferring causal mechanisms of diseases. Human Accelerated Regions (HARs), genomic loci conserved across mammals but rapidly divergent in the human lineage, offer an anchor for identifying human-specific host–microbe co-evolutionary units relevant to NDs. We further hypothesize that microbial metabolites represent one class of mechanistically testable intermediates linking host genetic background, gut microbial ecology, and neurodegenerative phenotypes. This integrated evolutionary-metabolic perspective offers a tractable path from correlation toward mechanism in gut microbiota–ND research.
Full article
(This article belongs to the Section Bioinformatics and Data Analysis)
Open AccessArticle
GDF15 Improves Renal Injury Induced by Ectopic Lipid Deposition via AMPK/SIRT1 Pathway-Mediated Autophagy
by
Qiang Zhang, Xidong Yang, Yuxuan Yang, Min Wang, Yulin Wu, Xin Xie, Yongjun Jin, Ming Yang and Meizi Yang
Metabolites 2026, 16(5), 336; https://doi.org/10.3390/metabo16050336 - 18 May 2026
Abstract
Objectives: Obesity precipitates excessive lipid accumulation within the kidney, culminating in ectopic lipid deposition that compromises target organ function through lipotoxicity. Given the pivotal role of GDF15 in lipid metabolism, this study aims to determine whether GDF15 can ameliorate ectopic lipid deposition and
[...] Read more.
Objectives: Obesity precipitates excessive lipid accumulation within the kidney, culminating in ectopic lipid deposition that compromises target organ function through lipotoxicity. Given the pivotal role of GDF15 in lipid metabolism, this study aims to determine whether GDF15 can ameliorate ectopic lipid deposition and mitigate the resulting renal injury. Methods: C57BL/6J mice were used to establish a high-fat diet-induced obesity model. Based on Lee’s index, the mice were categorized into a diet-induced obesity group and an obesity-resistant group. Subsequently, the diet-induced obesity group received an injection of AAV-shGFRAL to knock down the GFRAL receptor. Results: In obesity resistant mice, ectopic lipid deposition in the kidneys was markedly reduced, accompanied by decreased expression of the renal injury marker KIM-1 and significantly elevated levels of GDF15. Modulation of the GDF15-GFRAL axis demonstrated that reduced autophagy levels led to increased lipid accumulation and exacerbated renal injury. Conversely, GDF15 activates the AMPK/SIRT1 signaling pathway to promote cellular autophagy, thereby mitigating renal damage induced by ectopic lipid deposition. Consistent with this mechanism, the suppression of autophagy results in the aggravation of renal injury caused by ectopic lipid accumulation. Conclusions: GDF15 ameliorates renal injury induced by ectopic lipid deposition in the kidney primarily through activation of autophagy via the AMPK/SIRT1 signaling pathway.
Full article
(This article belongs to the Special Issue Animal Nutritional Metabolism and Toxicosis Disease, 2nd Edition)
►▼
Show Figures
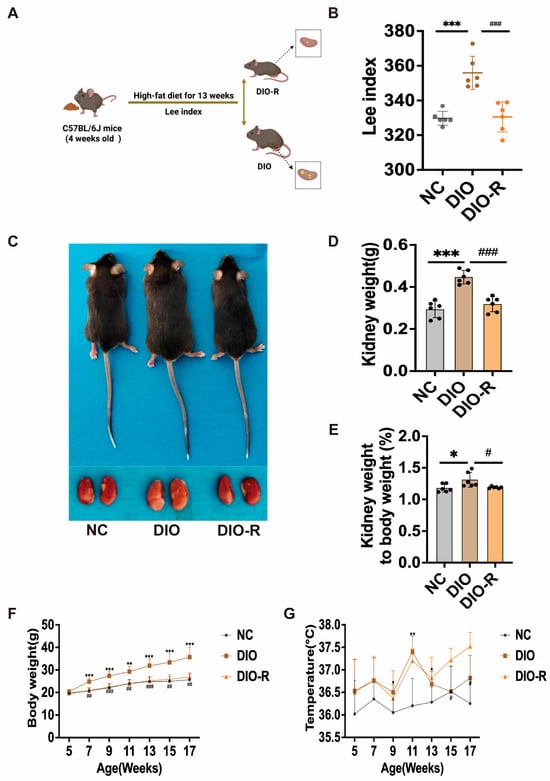
Figure 1
Open AccessArticle
The Effect of Angiotensin (1-7) on Serum Metabolomics in Obese Type 2 Diabetic Mice
by
Qiyuan Chen, Mingjin Sun, Hanqin Wang and Chunli Lu
Metabolites 2026, 16(5), 335; https://doi.org/10.3390/metabo16050335 - 15 May 2026
Abstract
Background: To investigate the effect of angiotensin-(1-7) [Ang-(1-7)] on serum metabolomics in obese type 2 diabetic (T2DM) mice. Methods: Four-week-old male C57BL/6 mice were fed a high-fat diet and intraperitoneally injected with streptozotocin (35 mg/kg) to establish an obese T2DM model.
[...] Read more.
Background: To investigate the effect of angiotensin-(1-7) [Ang-(1-7)] on serum metabolomics in obese type 2 diabetic (T2DM) mice. Methods: Four-week-old male C57BL/6 mice were fed a high-fat diet and intraperitoneally injected with streptozotocin (35 mg/kg) to establish an obese T2DM model. Mice were randomized into control, T2DM and T2DM+Ang-(1-7) groups (n = 6). Body weight and blood glucose were recorded weekly. At 10 weeks, blood glucose, serum inflammatory factors, lipid profiles, and pancreatic β-cell insulin secretion were detected; serum metabolite alterations were analyzed via untargeted metabolomics. Results: 1. Ang-(1-7) intervention decreased blood glucose (p < 0.05) and CRP levels (p < 0.01), and alleviated dyslipidemia (p < 0.05 or p < 0.01), as well as β-cell morphology and insulin expression in obese T2DM mice. 2. Non-targeted metabolomics analysis suggested that Ang-(1-7) may alleviate abnormal amino acid metabolic pathways by regulating levels of metabolites such as L-valine, L-proline, L-histidine, and glutamic acid. This intervention also tended to reduce multiple lipid metabolites, including Omega-3 Arachidonic Acid Ethyl Ester, phosphatidylcholine, and glycerophosphocholine, thereby participating in the modulation of lipid metabolism balance. KEGG enrichment analysis further indicated that Ang-(1-7) was involved in the regulation of protein digestion and the absorption pathway, as well as the HIF-1 signaling pathway related to oxidative stress, bile acid metabolism pathway, and other signaling pathways, and improving the insulin secretion pathway, pyrimidine metabolism, and TCA cycle energy metabolism pathway. Conclusions: Ang-(1-7) may partially improve metabolic disturbances in obese T2DM mice, which is potentially associated with the modulation of multiple metabolic processes, including amino acid metabolism, lipid metabolism, insulin secretion, and TCA cycle energy metabolism.
Full article
(This article belongs to the Section Endocrinology and Clinical Metabolic Research)
►▼
Show Figures

Figure 1
Open AccessReview
Ganoderma lucidum as a Functional Bioactive Candidate for Glycemic Regulation: Mechanisms, Preclinical Evidence, and Clinical Translation
by
Bogdan Florea, Doru Morar, Corina Marina Kracunovic, Simina Velescu, Vlad Iorgoni, Paula Nistor, Janos Degi, Ionica Iancu, Maria-Larisa Ardelean (Rusu), Romeo Teodor Cristina, Alexandra Pocinoc and Eugenia Dumitrescu
Metabolites 2026, 16(5), 334; https://doi.org/10.3390/metabo16050334 - 15 May 2026
Abstract
Type 2 diabetes mellitus (T2DM) is a major global health challenge that has intensified interest in multi-target nutraceuticals with potential adjunctive benefits. Ganoderma lucidum (Lingzhi/Reishi) is a medicinal mushroom traditionally used in East Asia and is increasingly investigated for its role in glycemic
[...] Read more.
Type 2 diabetes mellitus (T2DM) is a major global health challenge that has intensified interest in multi-target nutraceuticals with potential adjunctive benefits. Ganoderma lucidum (Lingzhi/Reishi) is a medicinal mushroom traditionally used in East Asia and is increasingly investigated for its role in glycemic regulation and metabolic disturbances. This review critically synthesizes current evidence on its hypoglycemic effects, focusing on bioactive compounds, molecular mechanisms, and translational limitations. Unlike broader reviews on Ganoderma bioactivity and health-related benefits, this review specifically evaluates the alignment between taxonomic authentication, chemical standardization, preclinical mechanisms, and human clinical evidence in the context of glycemic regulation. This narrative review was based on a targeted literature search conducted in PubMed/MEDLINE, Web of Science, and Scopus for studies published up to October 2025, supplemented by Google Scholar. The included studies comprised in vitro experiments, in vivo animal models, and human clinical trials evaluating glycemic and metabolic outcomes of Ganoderma preparations. In vitro and animal studies indicate that polysaccharides, including β-(1→3)/(1→6)-glucans and proteoglycans such as FYGL, may improve insulin sensitivity via AMPK (AMP-activated protein kinase) and PI3K/Akt pathways, promote GLUT4 (glucose transporter type 4) translocation, suppress hepatic gluconeogenesis, protect pancreatic β-cells, and modulate gut microbiota. In enzyme assays and preclinical models, lanostane-type triterpenoids act primarily by inhibiting α-glucosidase and α-amylase, thereby potentially reducing postprandial glucose excursions. Despite consistent preclinical evidence, clinical findings remain heterogeneous, with the largest randomized controlled trial reporting no significant glycemic benefit. Overall, Ganoderma lucidum shows strong mechanistic plausibility but insufficient clinical evidence for antidiabetic efficacy. Future research should prioritize species authentication, chemical standardization, and adequately powered clinical trials.
Full article
(This article belongs to the Special Issue Functional Foods and Natural Bioactive Compounds: Strategies to Face Metabolic Syndrome and Related Non-Communicable Diseases—2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
A Multi-Omics Approach Uncovers Divergent Mechanisms of Asthma in Normal Weight and Obese Children
by
Ilhame Diboun, Harshita Shailesh, Shana Jacob, Mohamed A. Elrayess, Stefan Worgall, Younes Mokrab and Ibrahim Janahi
Metabolites 2026, 16(5), 333; https://doi.org/10.3390/metabo16050333 - 15 May 2026
Abstract
Background: Children with obesity-related asthma exhibit poorer symptom control and more frequent exacerbations than their normal-weight peers, but the underlying metabolic mechanisms are unclear. This study aimed to identify drivers of obesity-related asthma through untargeted plasma metabolomic and lipidomic profiling. Methods:
[...] Read more.
Background: Children with obesity-related asthma exhibit poorer symptom control and more frequent exacerbations than their normal-weight peers, but the underlying metabolic mechanisms are unclear. This study aimed to identify drivers of obesity-related asthma through untargeted plasma metabolomic and lipidomic profiling. Methods: Plasma was obtained from normal weight (NW) asthmatic (n = 95) and non-asthmatic (n = 67) and overweight/obese (OO) asthmatic (n = 99) and non-asthmatic (n = 100) children (6–17 years). We assessed metabolic and lipidomic differences between asthmatics and controls within each BMI group using orthogonal partial least squares discriminant analysis (OPLS-DA), examined overlap with the adult Qatar Biobank cohort, and mapped metabolic–clinical interactions using Gaussian Graphical Models. Results: In the fitted OPLS-DA models, separation between asthmatic and control groups was stronger in the NW group (R2Y = 0.72/0.52) than in OO (R2Y = 0.65/0.63) children. Asthma was associated with altered tricarboxylic acid (TCA) intermediates, ether-linked phosphatidylethanolamines, and sphingomyelins (SM) in NW, and with phosphatidylcholines, lysophosphatidylcholines, and phosphatidylethanolamines in OO. Integrating metabolomic, lipidomic, and clinical data revealed connections between altered SMs and interleukins, and TCA intermediates and electrolytes, all associated with elevated leptin in NW. An increased residual volume to total lung capacity ratio in OO was associated with phospholipid shifts. The overall dynamics in lipid metabolism with asthma, conditioned on BMI, was also observed in the adult Qatar Biobank cohort. Conclusions: Among NW children with asthma, we found enhanced TCA cycle activity and inflammation linked to altered SM metabolism, whereas in OO, the findings suggest oxidative stress arising from chronic obesity-related inflammation. These data reveal BMI-specific metabolic mechanisms of pediatric asthma that might inform precision approaches to disease management.
Full article
(This article belongs to the Special Issue Metabolic Signatures of Pediatric Endocrine and Metabolic Disorders)
►▼
Show Figures
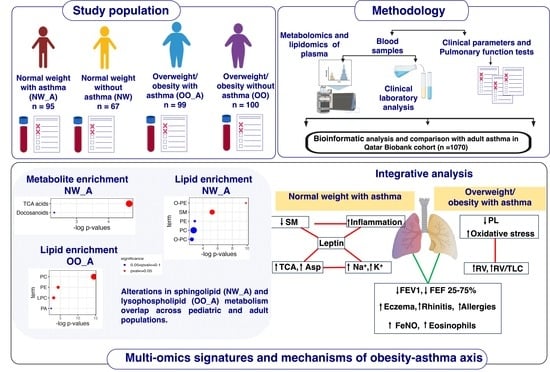
Graphical abstract
Open AccessArticle
Mechanistic Investigation of Vitexin in Ameliorating Ovarian Fibrosis in PCOS Mice via the NR4A1/NLRP3 Signaling Pathway
by
Haoran Sun, Jiejing Xu, Chengxue Pan, Jia-Le Song and Yanyuan Zhou
Metabolites 2026, 16(5), 332; https://doi.org/10.3390/metabo16050332 - 15 May 2026
Abstract
Objective: In this study, Dehydroepiandrosterone (DHEA-induced Polycystic Ovary Syndrome (PCOS) mice were used as models to evaluate the improvement effect of Vitexin (Vit) on ovarian fibrosis and explore the mechanism of action of the NR4A1/NLRP3 signaling pathway. Method: Sixty 4-week-old female ICR mice
[...] Read more.
Objective: In this study, Dehydroepiandrosterone (DHEA-induced Polycystic Ovary Syndrome (PCOS) mice were used as models to evaluate the improvement effect of Vitexin (Vit) on ovarian fibrosis and explore the mechanism of action of the NR4A1/NLRP3 signaling pathway. Method: Sixty 4-week-old female ICR mice of the same batch number were selected and their systems were divided into 6 groups (n = 10): normal (Control, Ctrl) group, model (Polycystic Ovary Syndrome, PCOS) group, treatment (Vitexin, The Vit group, normal NR4A1 gene silencing group (Ctrl NR4A1-/-), NR4A1 gene silencing model group (PCOS NR4A1-/-), and NR4A1 gene silencing treatment group (Vit NR4A1-/-). Silencing gene modeling was performed by tail vein injection of adeno-associated virus (serotype AAV-8), and the mouse genotypes were detected by qRT-PCR technology 14 days after injection. After the genotype was determined, the PCOS group and the PCOS NR4A1-/- group were administered dehydroepandrosterone (6 mg/100 g/d) by gavage for 28 consecutive days for modeling, while the Vit group and the Vit NR4A1-/- group were treated with dehydroepandrosterone + vitexin (10 mg/kg/d) by gavage for 28 consecutive days. All mice were raised with pure water and regular maintenance food. After 4 weeks of drug intervention, the mice were euthanized and samples were collected. The pathological changes in ovarian tissue were observed by H&E staining, and the degree of ovarian tissue fibrosis was observed by Masson staining. The levels of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), malondialdehyde (MDA), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C) in mouse serum were detected by biochemical kits. The levels of inflammatory factors (IL-1β, IL-6, IL-18, TNF-α) in mouse serum were determined by enzyme-linked immunosorbent assay. Real-time fluorescence quantitative PCR (qRT-PCR) was used to detect oxidative kinase (Gsta4, Prdx3, Mgst1, Gpx3, Gsr), inflammatory factors (Nlrp3, Caspase-1, Asc, Il-1β, Il-18, Tnf-α) and fibrotic pathway-related genes (Tgf-β1, Smad3, Collagen1, CTGF, α-SMA, Mmp-13, and β-catenin) in ovarian tissues. The levels of inflammatory factors (NLRP3, Caspase-1, ASC, IL-1β, IL-18, TNF-α, IκBα) and fibrosis in mice were determined by Western blot method, and statistical description and analysis were performed using SPSS software. Result: In the wild-type genotype group, compared with the PCOS group, Vit treatment could effectively regulate the metabolic abnormalities of PCOS mice, including inhibiting excessive weight gain, restoring normal glucose tolerance, and reducing body fat content. After Vit treatment, the levels of MDA, TC, TG, LDL, IL-1β, IL-6, IL-18 and TNF-α in the serum of PCOS mice were significantly reduced, while the levels of SOD and HDL in the serum of PCOS mice were increased. The staining results indicated that Vit treatment could significantly inhibit the process of ovarian fibrosis in PCOS mice. The results of WB and PCR demonstrated that after Vit gavage treatment in mice, inflammatory and fibrotic factors such as Nlrp3, Caspase-1, Asc, Il-1β, Il-18, Tgf-β1, Smad3, Collagen1, CTGF, and α-SMA in ovarian tissues could be significantly down-regulated, and the fibrotic level of ovarian tissues could be reduced. Among the same measurement indicators, the silenced NR4A1 group showed a certain degree of increase compared with the wild genotype group, but there was no significant difference. Conclusions: Vit intervention can restore the sex hormone levels and follicular development in ovarian tissues of PCOS mice, regulate reproductive endocrine disorders and abnormal lipid metabolism levels, and regulate the expression of Collagen I, a-SMA and CTGF in the ovaries by inhibiting the NR4A1/NLRP3 signaling pathway, thereby improving the ovarian fibrosis level of PCOS mice. It is suggested that it may play a key role in the treatment of PCOS and the prevention and delay of its long-term complications.
Full article
(This article belongs to the Section Plant Metabolism)
►▼
Show Figures

Figure 1
Open AccessArticle
Annexin A2 Is Associated with Dietary Cholesterol-Induced Metabolic Dysregulation and the Progression of Hepatic Fibrosis
by
Jiayang Liu, Ling Ou, Haiyan Tai, Yinghan Chai, Lirong Tan, Jie Lin, Bing Li, Ying Cao and Tingting Zhu
Metabolites 2026, 16(5), 331; https://doi.org/10.3390/metabo16050331 - 15 May 2026
Abstract
Background/Objectives: Dietary cholesterol intake significantly influences liver health, yet the specific molecular mechanisms by which it accelerates fibrogenesis remain incompletely defined. This study aimed to characterize the dose-dependent effects of dietary cholesterol on hepatic injury and fibrogenesis, identify cholesterol-responsive gene networks through
[...] Read more.
Background/Objectives: Dietary cholesterol intake significantly influences liver health, yet the specific molecular mechanisms by which it accelerates fibrogenesis remain incompletely defined. This study aimed to characterize the dose-dependent effects of dietary cholesterol on hepatic injury and fibrogenesis, identify cholesterol-responsive gene networks through transcriptomic analysis, and investigate Annexin A2 (ANXA2) as a candidate molecular mediator linking dietary cholesterol to hepatic fibrosis progression. Methods: A CCl4-induced liver fibrosis mouse model was established and supplemented with dietary cholesterol (1–2%). Liver injury and fibrosis were assessed by liver-to-body weight ratios, serum biochemical markers, histological analysis, and fibrogenic gene expression. RNA sequencing combined with multiple hepatic fibrosis database analyses was performed to identify potential molecular mediators. Results: Dietary cholesterol supplementation aggravated CCl4-induced hepatic fibrosis in mice, with dose-dependent increases in liver-to-body weight ratios and serum AST and ALT levels. Histological analysis showed enhanced collagen deposition and upregulation of fibrogenic genes. By integrating RNA-sequencing with multiple hepatic fibrosis database analysis and correlation analysis, we identified Annexin A2 (ANXA2) as a cholesterol-responsive gene associated with fibrosis. Conclusions: Dietary cholesterol promotes liver fibrosis progression, and ANXA2 may act as a potential mediator linking cholesterol metabolism to hepatic fibrogenesis.
Full article
(This article belongs to the Special Issue Human Nutrition and Metabolic Health)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
Lipidomic Signatures in Feline Disease: A PRISMA-Guided Systematic Review
by
Ana Carolina Fontes, Carolina Santos Silva, Ana Carolina Matos, Isabel Ribeiro Dias, Francisco Peixoto, Maria Manuel Oliveira, Maria Rosario Domingues and Carlos Antunes Viegas
Metabolites 2026, 16(5), 330; https://doi.org/10.3390/metabo16050330 - 15 May 2026
Abstract
Background/Objectives: Lipidomics has become a key component of systems biology, enabling comprehensive characterisation of lipid species and their roles in health and disease. As regulators of membrane architecture, energy balance, inflammation, and cellular signalling, lipids offer a powerful framework for understanding metabolic
[...] Read more.
Background/Objectives: Lipidomics has become a key component of systems biology, enabling comprehensive characterisation of lipid species and their roles in health and disease. As regulators of membrane architecture, energy balance, inflammation, and cellular signalling, lipids offer a powerful framework for understanding metabolic dysfunction. In veterinary medicine, however, lipidomics remains comparatively underdeveloped. In cats, lipid metabolism is central to disorders such as hepatic lipidosis, cystitis, obesity, diabetes mellitus, and chronic inflammatory enteropathies, yet available data remain limited. This systematic review synthesised current evidence on lipidomics and lipid-focused profiling in feline disease and identified lipid alterations with potential clinical relevance. Methods: Following PRISMA 2020 guidelines, PubMed, ScienceDirect, and Scopus were searched for original studies (1994–2026) evaluating lipidomics or lipid-focused profiling in cats. Eligible studies assessed lipid species, fatty acids, lipid mediators, or lipoproteins in disease or physiological states. Owing to methodological heterogeneity, findings were synthesised narratively. Results: Seventeen studies met inclusion criteria, covering hepatic, urinary, gastrointestinal, renal, neurological, oncological, metabolic, and pharmacologically modulated conditions. Recurring alterations involved lipoproteins, triglycerides, phospholipids, sphingolipids, fatty acids, and oxylipins. More consistent patterns emerged in hepatic lipidosis, where lipoprotein disturbances may aid diagnosis; in lower urinary tract disease, where PUFA-derived oxylipins differentiated bacterial from idiopathic cystitis; and in obesity, where phospholipid and triglyceride shifts reflected metabolic risk. Fatty acid remodelling in chronic enteropathies aligned with mucosal inflammation, while sphingolipid changes in neurological disease correlated with severity. Heterogeneity in analytical platforms, dietary control, and study design limited comparability. Conclusions: Feline lipidomics reveals biologically meaningful alterations with emerging diagnostic and prognostic value. Although still developing, lipid-focused approaches may enhance disease characterisation and support translational research. Larger, standardised studies and robust reference datasets are needed to validate lipid signatures for clinical implementation.
Full article
(This article belongs to the Topic New Perspectives on Metabolism and Metabolic Disease in Wildlife, Domestic, and Exotic Pets and Livestock)
►▼
Show Figures

Figure 1
Open AccessArticle
Regulation of Mitophagy by Low-Intensity Pulsed Ultrasound Attenuates Endothelial Dysfunction
by
Yucong Shi, Baotian Zhao, Yuhong Wei, Dongxu Lu, Haixia Liu and Yinzhu Chu
Metabolites 2026, 16(5), 329; https://doi.org/10.3390/metabo16050329 - 15 May 2026
Abstract
Background: Diabetic vascular complications are a major cause of poor prognosis in patients with diabetes mellitus (DM). Mitophagy activation is a potential therapeutic target for type 2 diabetes mellitus (T2DM), but the role of low-intensity pulsed ultrasound (LIPUS) in this context remains
[...] Read more.
Background: Diabetic vascular complications are a major cause of poor prognosis in patients with diabetes mellitus (DM). Mitophagy activation is a potential therapeutic target for type 2 diabetes mellitus (T2DM), but the role of low-intensity pulsed ultrasound (LIPUS) in this context remains unclear. Methods: The biological effects of LIPUS on endothelial cells under high glucose conditions were systematically evaluated using high glucose-treated human umbilical vein endothelial cells (HUVECs) and aortic tissues from diabetic rats as models, in combination with bioinformatics analysis and standard molecular and cellular biology techniques. Histological staining was further used to assess the protective role of LIPUS in the aortas of diabetic rats. Results: Bioinformatics analysis predicted that high glucose induces mitochondrial dysfunction, suppresses autophagy in HUVECs, impairs endothelial cell function, and activates fibroblasts. In vitro results were in agreement with these predictions. LIPUS treatment significantly counteracted these effects, restoring migration (p < 0.001) and angiogenesis (p < 0.05), increasing proliferation (p < 0.001), and decreasing apoptosis (p < 0.05). Mechanistically, LIPUS enhanced mitophagy, and its therapeutic effects were markedly diminished upon addition of the autophagy inhibitor 3-Methyladenine (3-MA). In vivo, LIPUS attenuated aortic endothelial damage and reduced collagen deposition in diabetic rats (p < 0.01). Conclusions: LIPUS may ameliorate hyperglycemia-induced endothelial cell dysfunction by activating mitophagy, and it also attenuates pathological damage in the abdominal aorta of diabetic rats, thereby providing experimental evidence for its application in the treatment of diabetic macrovascular complications.
Full article
(This article belongs to the Special Issue Metabolic Modulators in Cardiovascular Disease Management)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Exploratory Associations of Inflammatory Cytokines, Brain-Derived Neurotrophic Factor, and Vascular Endothelial Growth Factor with Clinical Outcomes in Patients with Bipolar Disorder
by
Fumito Hamada, Leo Gotoh, Yuko Tomiyama, Hiroko Sugawara, Muneaki Ogata, Hiroki Kumagai, Ryo Asada, Ryusei Hatae, Kiyohiro Yasumatsu and Hikaru Hori
Metabolites 2026, 16(5), 328; https://doi.org/10.3390/metabo16050328 - 14 May 2026
Abstract
Background/Objectives: Bipolar disorder is characterized by psychosocial dysfunction, cognitive impairment, and incomplete recovery. Although inflammatory and neurotrophic mechanisms have been implicated, their relationships with multidimensional recovery outcomes remain unclear. We examined the relationships of inflammatory cytokines, brain-derived neurotrophic factor (BDNF), and vascular endothelial
[...] Read more.
Background/Objectives: Bipolar disorder is characterized by psychosocial dysfunction, cognitive impairment, and incomplete recovery. Although inflammatory and neurotrophic mechanisms have been implicated, their relationships with multidimensional recovery outcomes remain unclear. We examined the relationships of inflammatory cytokines, brain-derived neurotrophic factor (BDNF), and vascular endothelial growth factor (VEGF) with depressive symptoms, psychosocial functioning, cognitive performance, personal recovery, and quality of life (QOL) in patients with bipolar disorder. Methods: This cross-sectional study of 24 patients with bipolar disorder assessed depressive symptoms, psychosocial functioning, cognitive functions, personal recovery, and QOL. Plasma tumor necrosis factor alpha, interleukin (IL)-6, IL-1β, IL-2, BDNF, and VEGF-A were measured by assay. Results: Subjective cognitive dysfunction was significantly associated with depressive symptom severity (rho = 0.53, p = 0.0083) and reduced QOL (rho = −0.56, p = 0.0042). Depressive symptoms were also associated with lower WHO-QOL-26 scores (rho = −0.43, p = 0.038). Significant interrelationships were observed among objective cognitive measures, and after false discovery rate (FDR) correction, the associations between FAST and PDQ-5-D, Symbol Check and Codebreaker, and Codebreaker and Trail remained statistically significant. High plasma IL-6 levels were associated with worse executive function (rho = 0.43, p = 0.0068). Higher VEGF levels were associated with better attentional performance (rho = −0.42, p = 0.042). Plasma IL-1β levels were positively associated with QOL (rho = 0.54, p = 0.02). After FDR correction, only the association between IL-1β and QOL remained statistically significant. Conclusions: This pilot study suggests that there may be associations between cognitive impairment and cytokines, as well as between quality of life and VEGF, in bipolar disorder. Further studies with larger sample sizes are needed.
Full article
(This article belongs to the Section Endocrinology and Clinical Metabolic Research)
Open AccessArticle
Unveiling the Brain-Penetrating Material Basis of Dragon’s Blood: Identification of Active Metabolites and Metabolic Pathways for Ischemic Stroke Therapy
by
Yu Zhu, Jiahui Ren, Meijia Chen, Jianglong Chen and Guang Li
Metabolites 2026, 16(5), 327; https://doi.org/10.3390/metabo16050327 - 14 May 2026
Abstract
Background: Dragon’s blood (dried resin of Dracaena cochinchinensis (Lour.) S.C.Chen) is a classic traditional medicine for treating ischemic stroke, yet its bioactive components capable of penetrating the blood–brain barrier (BBB) remain ill-defined. This study aims to elucidate its material basis and the
[...] Read more.
Background: Dragon’s blood (dried resin of Dracaena cochinchinensis (Lour.) S.C.Chen) is a classic traditional medicine for treating ischemic stroke, yet its bioactive components capable of penetrating the blood–brain barrier (BBB) remain ill-defined. This study aims to elucidate its material basis and the synergistic mechanism of Borneol as a “guide drug.” Methods: A systematic strategy integrating UHPLC-Q-TOF-MS/MS and metabolomics was employed to map the chemical profile of dragon’s blood and identify its migrating constituents in rats. Results: A total of 96 compounds were characterized in vitro. In vivo analysis of the cerebrospinal fluid (CSF) revealed a brain-penetrating profile that was significantly enriched by Borneol, with the number of detected constituents increasing from 11 in the DB group to 16 in the DB + B group. The results demonstrated that demethylation, glycoside hydrolysis, and oxidation are primary metabolic pathways, validating a “pro-drug” mechanism where aglycones and hydroxylated derivatives act as the central effectors. Notably, Borneol not only enhanced the BBB permeability of lipophilic flavonoids but also facilitated unique metabolic transformations, such as the cyclization of berberrubine to coptisine. Conclusions: This study elucidates the brain-penetrating material basis of dragon’s blood and reveals the dual synergistic mechanism of Borneol involving both physical permeation enhancement and metabolic modulation, offering scientific evidence for its clinical application in central nervous system diseases.
Full article
(This article belongs to the Section Pharmacology and Drug Metabolism)
►▼
Show Figures

Figure 1
Open AccessArticle
Untargeted Metabolomics Reveals Region-Specific Metabolic Signatures and Discriminative Markers in Goji Berry (Lycium barbarum L.)
by
Yan Yan, Wei Ma, Yage Li, Chen Zhang, Fang Li, Tianqing Huang, Beibei Gao, Huihui Meng, Yunfei Hu and Huan Wu
Metabolites 2026, 16(5), 326; https://doi.org/10.3390/metabo16050326 - 14 May 2026
Abstract
Background/Objectives: Goji berry (Lycium barbarum L.), renowned as a typical medicinal and edible plant, is mainly cultivated across four agroclimatic zones in China, including semi-arid, arid, monsoon, and high-altitude regions. Ningxia has long been recognized as the daodi production area for
[...] Read more.
Background/Objectives: Goji berry (Lycium barbarum L.), renowned as a typical medicinal and edible plant, is mainly cultivated across four agroclimatic zones in China, including semi-arid, arid, monsoon, and high-altitude regions. Ningxia has long been recognized as the daodi production area for goji berries. However, the metabolic diversity of goji berries from other core cultivation regions and how these differences are shaped by local environments remain poorly understood. Methods: In this study, untargeted metabolomics was employed to comprehensively investigate the metabolic difference in goji across seven production regions. By integrating multivariate analysis with KEGG pathway enrichment (p < 0.05), 49 discriminative markers enriched in 10 key pathways were putatively identified, and their roles in plant stress tolerance were elucidated. In addition, we conducted targeted quantification of key bioactive components and antioxidant capacity. Results: Significant regional differences were revealed. Redundancy analysis further identified rainfall, temperature, and UV radiation as the key climatic drivers of this variation. Conclusions: These findings provide insights into the metabolic adaptation of goji to local environments and serve as a basis for further functional studies.
Full article
(This article belongs to the Special Issue Metabolites and Plant Stress Resistance)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Metabolites Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cancers, Diagnostics, JCM, Metabolites, Targets
Biomarkers of Disease: Discovery and Clinical Applications
Topic Editors: Andreas Tsakalof, Ioannis KanakisDeadline: 30 June 2026
Topic in
Dairy, Foods, Microorganisms, Nutrients, Metabolites
The Efficacy of Probiotics and Their Functional Metabolites in Fermented Foods
Topic Editors: Rina Wu, Wenjun Liu, Zhen Wu, Feiyu AnDeadline: 31 July 2026
Topic in
Nutrients, Metabolites, Healthcare, Children, Obesities, Life
Non-Communicable Diseases Silent Killer: Metabolic and Obesity Risks of Sedentary Behaviors
Topic Editors: Kotsedi Daniel Monyeki, Machoene Derrick SekgalaDeadline: 30 September 2026
Topic in
Animals, Antioxidants, Metabolites, Pets, Veterinary Sciences
Research on Companion Animal Nutrition
Topic Editors: Baichuan Deng, Lian Li, Yun JiDeadline: 26 October 2026

Conferences
Special Issues
Special Issue in
Metabolites
Metabolomics of Human Nutrition: The Dot of Human Nutrition and the Circle of Soil, Plants, Animals and Microbes in Relation to It, 2nd Edition
Guest Editor: Shoma BerkemeyerDeadline: 21 May 2026
Special Issue in
Metabolites
Targeted and Non-Targeted LC- and GC-HRMS Workflows for the Analysis of Emerging Contaminants and the Environmental Exposome
Guest Editors: Lapo Renai, Massimo Del BubbaDeadline: 21 May 2026
Special Issue in
Metabolites
Phenological Regulation of Secondary Metabolism
Guest Editors: Iván De-la-Cruz-Chacón, Alma Rosa González-Esquinca, Xin FangDeadline: 21 May 2026
Special Issue in
Metabolites
Bioactive Metabolites from Fungal Endophytes Associated with Plants
Guest Editors: Zhong-Hua Tang, Chun-Hao ChangDeadline: 22 May 2026
Topical Collections
Topical Collection in
Metabolites
Feature Papers Related to "Metabolomic Profiling Technology" Section
Collection Editor: Thusitha Rupasinghe
Topical Collection in
Metabolites
Feature Papers Related to "Environmental Metabolomics" Section
Collection Editor: David J. Beale
Topical Collection in
Metabolites
Advances in Metabolomics
Collection Editors: Leonardo Tenori, Edoardo Saccenti