Journal Description
Cancers
Cancers
is a peer-reviewed, open access journal of oncology published semimonthly online. The North-East German Society for Gynecological Oncology (NOGGO), Irish Association for Cancer Research (IACR), Spanish Association for Cancer Research (ASEICA), Biomedical Research Centre (CIBM), British Neuro-Oncology Society (BNOS) and more are affiliated with Cancers and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Oncology) / CiteScore - Q1 (Oncology)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.1 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 18 topical sections.
- Companion journals for Cancers include: Radiation and Onco.
- Journal Clusters of Oncology: Cancers, Current Oncology, Onco and Targets.
Impact Factor:
4.4 (2024);
5-Year Impact Factor:
4.8 (2024)
Latest Articles
Association of Autophagy-Related Gene Expression Profiles with Survival in Diffuse Astrocytic Tumors
Cancers 2026, 18(8), 1215; https://doi.org/10.3390/cancers18081215 - 10 Apr 2026
Abstract
Background: The aim of this study is to investigate the relationship between the expression levels of autophagy-related genes (SQSTM1, Beclin1, Atg5, and Atg7) in diffuse astrocytic tumors and clinicopathological parameters, including tumor grade, IDH mutation status, and survival outcomes. Materials and Methods:
[...] Read more.
Background: The aim of this study is to investigate the relationship between the expression levels of autophagy-related genes (SQSTM1, Beclin1, Atg5, and Atg7) in diffuse astrocytic tumors and clinicopathological parameters, including tumor grade, IDH mutation status, and survival outcomes. Materials and Methods: A total of 150 histopathologically confirmed diffuse astrocytic tumor cases were retrospectively analyzed. Clinical data were extracted from patient records. Gene expression levels were determined using qRT-PCR and evaluated by the 2−ΔCt method, where lower ΔCt values indicate higher gene expression. IDH1 R132H mutation status was evaluated by immunohistochemistry. Results: No statistically significant differences were observed in the expression levels of SQSTM1, Beclin1, Atg5, and Atg7 across WHO tumor grades (p > 0.05). However, when analyzed by IDH status, IDH-mutant tumors exhibited significantly higher gene expression levels (demonstrated by lower ΔCt values) of Beclin1 (p = 0.046) and Atg5 (p = 0.027) compared to IDH wild-type tumors. In multivariate Cox regression analysis, age and WHO tumor grades were confirmed as independent prognostic factors. Crucially, higher SQSTM1 expression independently predicted worse clinical outcomes, specifically poorer overall survival (OS) (p = 0.004) and shorter progression-free survival (PFS) (p = 0.031). Additionally, elevated Beclin1 expression was identified as an independent predictor of worse OS (p = 0.023). Conclusions: This study demonstrates that increased expression of autophagy-related genes, particularly SQSTM1 and Beclin1, serves as a robust indicator of poor prognosis and shorter survival times in diffuse astrocytic tumors. Furthermore, the elevated expression of Beclin1 and Atg5 in IDH-mutant cases highlights a complex metabolic interplay that warrants further investigation as potential therapeutic targets.
Full article
(This article belongs to the Section Tumor Microenvironment)
►
Show Figures
Open AccessReview
Therapeutic Potential of Peptides in Cancer Treatment: Focus on Peptide and Aptamer-Decorated Exosomes
by
Prakash Gangadaran, Aswini Suresh Kumar, Kasinathan Kumaran, Kruthika Prakash, Sanjana Dhayalan, Ramya Lakshmi Rajendran, Vasanth Kanth Thasma Loganathbabu, Janani Balaji, Radhika Baskaran, Raksa Arun, Vanshikaa Karthikeyan, Sreyee Biswas, Chae Moon Hong, Kandasamy Nagarajan ArulJothi and Byeong-Cheol Ahn
Cancers 2026, 18(8), 1214; https://doi.org/10.3390/cancers18081214 - 10 Apr 2026
Abstract
Traditional cancer therapies such as surgery, chemotherapy, and antibody-based treatments often face significant barriers, including systemic toxicity, a lack of selectivity, and the emergence of drug resistance. These issues demand innovative and targeted solutions. Peptide-based therapeutics have gained prominence for their ability to
[...] Read more.
Traditional cancer therapies such as surgery, chemotherapy, and antibody-based treatments often face significant barriers, including systemic toxicity, a lack of selectivity, and the emergence of drug resistance. These issues demand innovative and targeted solutions. Peptide-based therapeutics have gained prominence for their ability to disrupt cancer pathways and facilitate targeted drug delivery, offering structural flexibility, precise targeting, and low immunogenicity with minimal effects on healthy tissues. Concurrently, aptamers, which are structured nucleic acid molecules capable of high-affinity molecular recognition, are being developed as both direct therapeutic agents and as targeting ligands for the improved delivery of anticancer drugs. Combining peptide and aptamer technologies with engineered exosomes provides a modular drug delivery system that enhances targeting specificity, stability, and the ability to cross complex biological barriers such as the blood–brain barrier. The emergence of peptide-decorated, aptamer-decorated exosomes represents a new frontier in precision oncology, promising highly selective, biocompatible, and tunable cancer therapies. Further advances are required to overcome challenges in pharmacokinetics, scalable production, and regulatory compliance, but ongoing bioengineering and nanotechnology research continues to accelerate the translation of these innovative strategies toward improved cancer diagnostics and treatment outcomes. This review discusses the synergistic integration of peptides and aptamers with exosome-based delivery systems, highlighting their current applications and future possibilities.
Full article
(This article belongs to the Special Issue Smart Nanotechnology for Drug Delivery in Cancer Therapy)
►▼
Show Figures

Figure 1
Open AccessArticle
Dynamics of Pulmonary Perfusion and Function Following Radical Treatment for Lung Tumors: A Prospective Comparative Study of Surgery, Radiotherapy, and Thermal Ablation
by
Aurimas Mačionis, Ieva Balčiūnaitė, Grytė Galnaitienė, Rūta Dubeikaitė, Gertrūda Maziliauskienė, Ieva Dimienė, Irena Nedzelskienė, Edita Mišeikytė-Kaubrienė, Lina Padervinskienė, Skaidrius Miliauskas, Donatas Vajauskas and Marius Žemaitis
Cancers 2026, 18(8), 1213; https://doi.org/10.3390/cancers18081213 - 10 Apr 2026
Abstract
Background/Objectives: Accurate assessment of pulmonary function is essential before planning radical lung cancer treatment. While spirometry reflects global lung function, perfusion imaging provides detailed information on regional perfusion patterns. This study aimed to characterize the pre-treatment profile of patients and compare the
[...] Read more.
Background/Objectives: Accurate assessment of pulmonary function is essential before planning radical lung cancer treatment. While spirometry reflects global lung function, perfusion imaging provides detailed information on regional perfusion patterns. This study aimed to characterize the pre-treatment profile of patients and compare the impact of surgical resection, radiotherapy, and thermal ablation on global pulmonary function and regional perfusion using SPECT/CT. Methods: In this prospective study of 68 patients, pre- and post-treatment assessments were conducted using lung perfusion SPECT/CT. While the entire cohort underwent imaging, longitudinal global pulmonary function (spirometry and gas diffusion) was analyzed for 45 patients who completed the three-month follow-up. Quantitative analysis included perfusion percentages and lung volumes, while a semi-quantitative scoring system evaluated the severity of perfusion defects. Results: In the overall cohort, the affected lung perfusion and volume significantly decreased (p = 0.002). Subgroup analysis revealed that the surgical resection group experienced significant reductions in perfusion (from 54.0% to 41.0%, p = 0.002) and volume (p < 0.001) of the affected lung, whereas no statistically significant changes were observed in the thermal ablation and radiotherapy groups (p > 0.05). Notably, 60.3% of patients presented with perfusion defects before treatment. Post-treatment spirometry parameters, particularly FEV1% (threshold 83.5%, AUC = 0.783), served as reliable predictors of persistent perfusion impairment. Conclusions: Radiotherapy and thermal ablation are lung-perfusion-sparing treatments compared to surgical resection. The high prevalence of pre-existing perfusion defects emphasizes the importance of incorporating lung perfusion SPECT/CT into routine pre-treatment evaluation to optimize treatment selection.
Full article
(This article belongs to the Special Issue Clinical Trials and Outcomes for Non-Small Cell Lung Cancer)
►▼
Show Figures
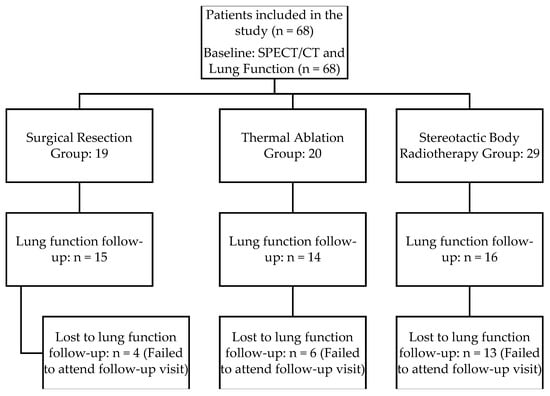
Figure 1
Open AccessArticle
Obstructive Sleep Apnea After Supracricoid Laryngeal Surgery (OPHL II): A Monocentric Prospective Pilot Study
by
Massimo Mesolella, Salvatore Allosso, Fabio Perrotta, Carlo Iadevaia, Carmela Cirillo, Nicola Serra, Pasquale Capriglione, Martina Ricciardiello, Anna Leoni and Anna Rita Fetoni
Cancers 2026, 18(8), 1212; https://doi.org/10.3390/cancers18081212 - 10 Apr 2026
Abstract
Background/Objectives: Obstructive sleep apnea (OSA) is increasingly observed in patients undergoing supracricoid laryngeal surgery; however, the impact of postoperative anatomical changes on sleep-disordered breathing remains insufficiently characterized. This pilot study aimed to assess the incidence and severity of OSA after Open Partial
[...] Read more.
Background/Objectives: Obstructive sleep apnea (OSA) is increasingly observed in patients undergoing supracricoid laryngeal surgery; however, the impact of postoperative anatomical changes on sleep-disordered breathing remains insufficiently characterized. This pilot study aimed to assess the incidence and severity of OSA after Open Partial Horizontal Laryngectomy type II (OPHL II) and to evaluate correlations between polysomnographic parameters and radiologic measurements of the neolarynx. Methods: A prospective observational cohort study was conducted on ten patients who underwent OPHL II between 2019 and 2024 and were evaluated at least one year postoperatively. The sample size was determined using a conservative estimate appropriate for a pilot prospective study, which required a long postoperative follow-up period of at least one year. All patients completed Stop-Bang, Berlin, and Epworth questionnaires and underwent overnight polysomnography. Cervical CT scans were used to measure airway length to the vocal cords (ALVC), supralaryngeal tract horizontal (SVTH) and vertical (SVTV) segments, and the base-of-tongue–to–cervical-body distance (BTCB). Results: OSA was detected in all patients: 40% mild, 30% moderate, and 30% severe. Mean AHI was 25.5 ± 18.9 events/h, and OSA severity strongly correlated with AHI (rho = 0.94; p < 0.0001). Among radiologic parameters, SVTV showed a positive correlation with OSA severity (rho = 0.82; p = 0.0035), while BTCB demonstrated a significant negative correlation (rho = −0.71; p = 0.0207). No significant associations were found for ALVC or SVTH. Conclusions: Supracricoid laryngectomy produces anatomical changes that predispose patients to OSA. Radiologic metrics—particularly SVTV and BTCB—appear to be meaningful predictors of OSA severity. A multidisciplinary approach is essential for early diagnosis and management. Due to the small number of patients enrolled larger multicenter studies are needed to confirm these findings and define radiologic criteria associated with postoperative OSA.
Full article
(This article belongs to the Special Issue Targeted Therapy in Head and Neck Cancer)
►▼
Show Figures

Figure 1
Open AccessArticle
Clinicopathological Characteristics and BAP1 Expression in an Enucleation-Based Uveal Melanoma Cohort: A Single-Center Croatian Experience with Long-Term Follow-Up
by
Domagoj Vlašić, Mira Knežić Zagorec, Antonia Jakovčević, Dina Lešin Gaćina, Marijana Ćorić and Tomislav Jukić
Cancers 2026, 18(8), 1211; https://doi.org/10.3390/cancers18081211 - 10 Apr 2026
Abstract
Background/Objectives: Loss of nuclear BAP1 (BRCA1-associated protein 1) expression is a well-established adverse prognostic marker in uveal melanoma (UM). However, data from Central and Southeastern European populations are limited. This descriptive study aimed to evaluate BAP1 immunohistochemical expression in a Croatian enucleation-based UM
[...] Read more.
Background/Objectives: Loss of nuclear BAP1 (BRCA1-associated protein 1) expression is a well-established adverse prognostic marker in uveal melanoma (UM). However, data from Central and Southeastern European populations are limited. This descriptive study aimed to evaluate BAP1 immunohistochemical expression in a Croatian enucleation-based UM cohort, characterize its associations with clinicopathological parameters, and contextualize the findings within the published literature. Methods: Formalin-fixed, paraffin-embedded tumor tissue from 58 consecutive patients with primary choroidal and ciliary body melanoma treated with enucleation at University Hospital Centre Zagreb (2006–2016) was analyzed immunohistochemically for BAP1 nuclear expression. Associations with clinicopathological parameters were assessed using chi-square and Fisher’s exact tests. Survival analysis was performed using Kaplan–Meier estimation, log-rank tests, and Cox proportional hazards regression with a median follow-up of 11.2 years. Results: Loss of nuclear BAP1 expression was observed in 53/58 (91.4%) specimens, resulting in a severely imbalanced distribution (53 versus 5 patients) precluding meaningful comparative survival analysis. Five-year and 10-year overall survival rates were 72.4% and 51.7%, respectively, with a median overall survival of 14.5 years. BAP1 loss was associated with longer disease-free survival (log-rank p = 0.020); however, this finding likely reflects a statistical artifact attributable to the extremely small BAP1-retained group (n = 5) harboring concurrent adverse features and should not be interpreted biologically. The study was underpowered to draw prognostic inferences regarding BAP1 status. Exploratory survival analyses are presented for transparency but should not be interpreted inferentially. Conclusions: The exceptionally high prevalence of BAP1 loss reflects the selection bias inherent in enucleation-based cohorts, which are enriched for large, molecularly high-risk tumors. This study provides the first comprehensive BAP1 immunohistochemical data from Croatia, contributing to the growing evidence that enucleation cohorts represent a distinct, biologically high-risk subgroup in which BAP1 immunohistochemistry offers limited discriminatory value. The extended follow-up of 11.2 years confirms the prolonged natural history of UM. Future multi-center studies incorporating molecular validation and diverse treatment modalities are needed to establish the prognostic utility of BAP1 across the full spectrum of UM disease.
Full article
(This article belongs to the Special Issue Advances in Uveal Melanoma)
►▼
Show Figures

Figure 1
Open AccessArticle
The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases
by
Saif-Eldin Abedellatif, Marija Janjic, Logman Khalafov, Harun Asoglu, Juliane Dittmer, Muriel Heimann, Mohammed Jaber, Haitham Alenezi, Marieta Ioana Toma, Matthias Schneider, Hartmut Vatter, Motaz Hamed and Mohammed Banat
Cancers 2026, 18(8), 1210; https://doi.org/10.3390/cancers18081210 - 10 Apr 2026
Abstract
Background: The prognostic assessment of patients with spinal metastases is primarily based on clinical and radiological parameters. Biological tumor characteristics such as the proliferation marker Ki-67 have prognostic relevance in various metastatic settings. This study aimed to evaluate the prognostic impact of the
[...] Read more.
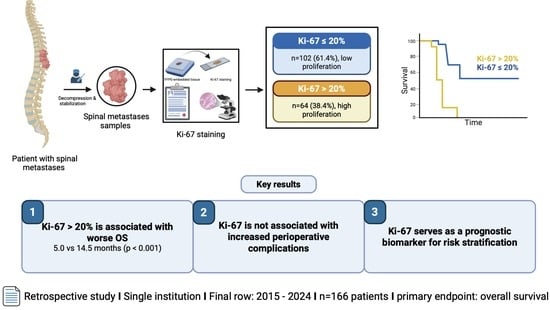
Background: The prognostic assessment of patients with spinal metastases is primarily based on clinical and radiological parameters. Biological tumor characteristics such as the proliferation marker Ki-67 have prognostic relevance in various metastatic settings. This study aimed to evaluate the prognostic impact of the Ki-67 proliferation index on survival outcomes in patients undergoing surgery for spinal metastases. Methods: We included 166 patients who underwent surgical treatment for spinal metastases at our university clinic between 2015 and 2024. Clinical, functional, tumor-related, and perioperative variables were collected. Receiver operating characteristic (ROC) analysis was performed to evaluate the discriminatory ability of Ki-67, and comparisons were made between patient groups according to Ki-67 expression (≤20% vs. >20%). Results: Based on ROC analysis, Ki-67 demonstrated a moderate but significant predictive ability for 1-year mortality (area under the curve [AUC]: 0.69, p = 0.001). Patients with a Ki-67 index of >20% showed a significantly shorter overall survival than those with a lower Ki-67 index of ≤20% (median overall survival: 5.0 vs. 14.5 months, p < 0.001). One-year mortality was significantly higher in the high Ki-67 group (78.9% vs. 41.8%, p = 0.001). High Ki-67 expression was associated with more aggressive tumor characteristics but was not associated with increased perioperative morbidity. Conclusions: The Ki-67 proliferation index is a significant prognostic biomarker in surgically treated patients with spinal metastases. A Ki-67 index threshold of 20% identifies patients at increased risk of early mortality and significantly reduced overall survival.
Full article
(This article belongs to the Special Issue Cancer Metastasis in 2025–2026)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Ex Vivo Characterization Studies Identify Candidate Therapies for the Individualized Care of NF2-Related Schwannomatosis
by
Ethan W. Hass, Anna Nagel, Alexandra J. Scott, Robert Allaway, Haley M. Hardin, Hollie M. Hayes, Lenna Huelbes, Alexander W. Sutton, Sofia A. Oliveira, Michelle Pei, Fred F. Telischi, John Ragheb, McKay McKinnon, Ziad Khatib, Mislen Bauer, Christine T. Dinh and Cristina Fernandez-Valle
Cancers 2026, 18(8), 1209; https://doi.org/10.3390/cancers18081209 - 10 Apr 2026
Abstract
Background/Objectives: NF2-related schwannomatosis (NF2-SWN) is a genetic tumor predisposition syndrome of the nervous system caused by pathogenic variants in NF2 encoding the merlin tumor suppressor. Truncating variants in NF2 cause severe phenotypes with higher tumor burden, early mortality, and
[...] Read more.
Background/Objectives: NF2-related schwannomatosis (NF2-SWN) is a genetic tumor predisposition syndrome of the nervous system caused by pathogenic variants in NF2 encoding the merlin tumor suppressor. Truncating variants in NF2 cause severe phenotypes with higher tumor burden, early mortality, and a lifetime need for multiple surgeries due to lack of medications that control schwannoma growth. Methods: We developed a functional precision medicine (FPM)-inspired workflow to identify drug sensitivities in cells isolated from a pediatric severe NF2-SWN patient’s spinal and peripheral schwannomas. Transcriptomic profiling, high-content drug sensitivity assays, tissue and isolated cell immunostaining, flow cytometry, and capillary-based immunoblotting were used to study the available tissues. Results: Aberrant merlin-dependent pathway expression was conserved between the spinal schwannoma and its cultured primary cells. Drug sensitivity screens in 2- and 3-dimensional formats revealed cytotoxic effects of fimepinostat in primary cells; dasatinib with brigatinib was the most effective cytostatic combination. Ineffective therapies attempted in the patient were also ineffective ex vivo. Conclusions: These data support the idea of using the FPM workflow to improve and individualize the standard of care for severe NF2-SWN patients using surgical samples.
Full article
(This article belongs to the Special Issue Targeted Therapies for Pediatric Nervous System Tumors)
►▼
Show Figures

Figure 1
Open AccessComment
Comment on Rivas et al. Unexpectedly Low Rate of Metastasis and Death Among Patients Treated for Uveal Melanoma with Brachytherapy, Vitrectomy, and Silicone Oil. Cancers 2025, 17, 2683
by
Zelia M. Correa, J. William Harbour, Brian P. Marr, Prithvi Mruthyunjaya, David Reichstein, Miguel A. Materin, Amy C. Schefler and Alison Skalet
Cancers 2026, 18(8), 1208; https://doi.org/10.3390/cancers18081208 - 10 Apr 2026
Abstract
As the executive leadership of the Collaborative Ocular Oncology Group (COOG), which has conducted the largest prospective clinical studies ever performed in patients with uveal melanoma, we read with concern the recent report by Rivas, Samlowski and McCannel, which claims that brachytherapy combined
[...] Read more.
As the executive leadership of the Collaborative Ocular Oncology Group (COOG), which has conducted the largest prospective clinical studies ever performed in patients with uveal melanoma, we read with concern the recent report by Rivas, Samlowski and McCannel, which claims that brachytherapy combined with vitrectomy and silicone oil injection results in an “unexpectedly low rate” of metastasis and death in patients with uveal melanoma [...]
Full article
(This article belongs to the Special Issue Advances in Treatment of Uveal Melanoma)
Open AccessArticle
Dosimetric Comparison of Automated Noncoplanar VMAT (HyperArc) Versus CyberKnife for Single-Fraction Vestibular Schwannoma Stereotactic Radiosurgery
by
Zhenyu Xiong, Yin Zhang, Lili Zhou, Keying Xu, Xinxin Zhang, Loren Bell, Fredrick Warburton, David Huang, Sabin B. Motwani, Charles S. Cathcart, Ke Nie, Ning Yue and Xiao Wang
Cancers 2026, 18(8), 1207; https://doi.org/10.3390/cancers18081207 - 10 Apr 2026
Abstract
Background: Vestibular schwannoma (VS) stereotactic radiosurgery (SRS) requires high target conformality and rapid dose falloff to spare adjacent organs at risk (OARs), particularly the brainstem. HyperArc (HA) is an automated noncoplanar volumetric-modulated arc therapy (VMAT) approach designed to standardize and streamline cranial SRS
[...] Read more.
Background: Vestibular schwannoma (VS) stereotactic radiosurgery (SRS) requires high target conformality and rapid dose falloff to spare adjacent organs at risk (OARs), particularly the brainstem. HyperArc (HA) is an automated noncoplanar volumetric-modulated arc therapy (VMAT) approach designed to standardize and streamline cranial SRS planning and delivery. We compared CyberKnife (CK) with HA for single-fraction VS SRS and evaluated the impact of multileaf collimator (MLC) leaf width. Methods: Fifteen VS cases previously treated with single-fraction CK SRS (12.5 Gy) were retrospectively replanned using HA. HA plans used four preconfigured noncoplanar partial arcs and were created with either a standard 5.0 mm MLC (HA-SMLC) or a 2.5 mm high-definition MLC (HA-HDMLC). HA plans were normalized to match the prescription dose target coverage of the corresponding CK plan for each of the patients. Endpoints included planning target volume (PTV) dosimetric statistics (Dmean, Dmin, Dmax, D98%), Paddick conformity index (PCI), Paddick gradient index (GI), ICRU Report 83 homogeneity index (HI), brain V12Gy, and brainstem Dmax. Because plans were generated for the same patients, paired comparisons were performed using two-sided Wilcoxon signed-rank tests (p < 0.05). Results: Both HA techniques achieved a higher near-minimum target dose than CK, with significantly higher PTV D98% (CK 12.35 ± 0.52 Gy; HA-SMLC 12.54 ± 0.28 Gy; HA-HDMLC 12.57 ± 0.35 Gy; p < 0.05). HA reduced target hotspots, with lower PTV Dmax than CK (CK 15.25 ± 0.32 Gy; HA-SMLC 14.70 ± 0.39 Gy; HA-HDMLC 14.73 ± 0.32 Gy; p < 0.05), and improved homogeneity and dose falloff as reflected by HI and GI (p < 0.05). CK achieved the highest conformity by PCI (p < 0.05), while HA-HDMLC improved PCI compared with HA-SMLC (p < 0.05). Brain V12Gy and brainstem Dmax were low and did not differ significantly among techniques. Conclusions: HA provides dosimetric performance comparable to CK for single-fraction VS SRS, with improved near-minimum PTV dose, reduced hotspots, and steeper dose gradients. Although CK showed the highest PCI overall, conformity improved with HA when a high-definition MLC was used. Overall, these findings support HA, particularly HA-HDMLC, as an efficient and clinically practical option for VS SRS treatment planning.
Full article
(This article belongs to the Special Issue Radiation Therapy in Oncology)
►▼
Show Figures
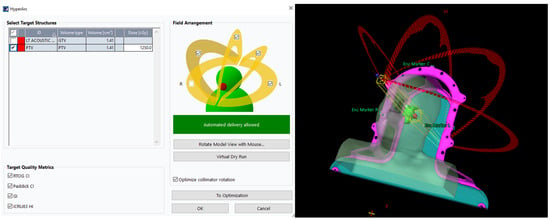
Figure 1
Open AccessReview
Histo-Molecular Intratumoral Heterogeneity in Meningiomas: A Narrative Review
by
Nourou Dine Adeniran Bankole, Tuan Le Van, Luc Kerherve, Edouard Morlaix, Jean-François Bellus, Kerima Belhajali, Julian Lopez, Pierre De Buck, Alia Sayda Houidi, Walid Farah, Maxime Lleu, Olivier Baland, Cathy Cao, Ahmed El Cadhi, Jacques Beaurain, Thiebaud Picart and Moncef Berhouma
Cancers 2026, 18(8), 1206; https://doi.org/10.3390/cancers18081206 - 10 Apr 2026
Abstract
Background: Meningiomas, the most common primary intracranial tumors, are predominantly benign, but high-grade variants show marked aggressiveness, histo-molecular heterogeneity, and treatment resistance. Although the 2021 WHO CNS classification integrates molecular and histopathologic criteria, substantial inter- and intratumoral variability still limits prognostic accuracy
[...] Read more.
Background: Meningiomas, the most common primary intracranial tumors, are predominantly benign, but high-grade variants show marked aggressiveness, histo-molecular heterogeneity, and treatment resistance. Although the 2021 WHO CNS classification integrates molecular and histopathologic criteria, substantial inter- and intratumoral variability still limits prognostic accuracy and treatment effectiveness. The goal was to provide insight regarding the histo-molecular intratumoral heterogeneity (ITH) of meningioma and examine its clinical implications. Methods: A narrative review was performed in accordance with PRISMA guidelines. PubMed and Google Scholar were screened for studies on “meningioma” and “intratumoral heterogeneity” published up to 28 July 2025. Eligible studies included original human research reporting histological or molecular heterogeneity with clinical relevance. Results: Eighteen studies comprising 2952 meningioma patients (mean age 59.4 ± 14.8 years, range 16–85) were included. Integrated cytogenetic, molecular, and spatial analyses, including FISH, karyotyping, scRNA-seq, CNV profiling, and spatial transcriptomics, revealed multilayered histo-molecular heterogeneity. Histologically, regional variations in morphology and proliferative index increased with tumor grade. Genomic diversity, marked by recurrent losses of 1p, 14q, and 22q and transcriptionally distinct subclones, defined a complex tumor architecture. Spatial and temporal analyses demonstrated subclonal expansion, stepwise clonal evolution, and therapy resistance, particularly in recurrent tumors. Functionally, SULT1E1+ subclones and COL6A3-mediated macrophage–tumor interactions emerged as potential key drivers of malignancy, recurrence, and radioresistance. Conclusions: Histo-molecular diversity underlies meningioma progression, recurrence, and therapeutic resistance. Standardization of ITH assessment, integration of AI-based spatial analytics, and the development of subclone-specific therapies are essential next steps toward advancing precision neuro-oncology.
Full article
(This article belongs to the Special Issue Heterogeneity in Intra-Cranial Tumors: Diagnostic, Prognostic, and Therapeutic Implications)
►▼
Show Figures
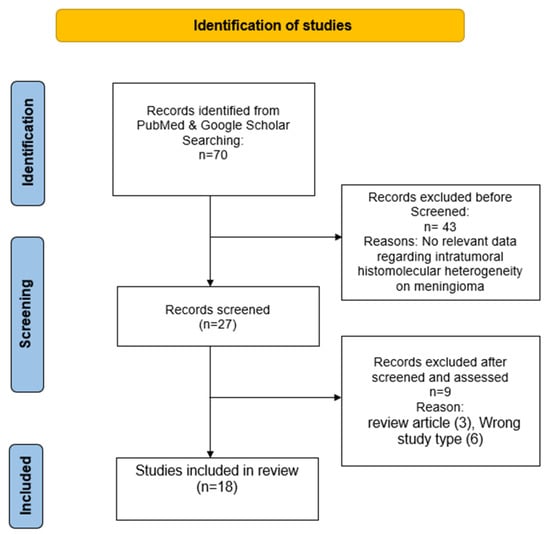
Figure 1
Open AccessArticle
An Interpretable Multimodal Machine-Learning Model for Non-Invasive Preoperative Glioma Grading
by
Xianfeng Rao, Min Yang, Hao Chen, Guanhao Li, Li Wu, Liudong Gong, Minchun Yang, Haiyang Wang, Ye Ding, Guanxi Chen, Xianjun Rao, Na Zhang, Xiaoxiong Wang and Lei Teng
Cancers 2026, 18(8), 1204; https://doi.org/10.3390/cancers18081204 - 10 Apr 2026
Abstract
Background: Gliomas are the most common primary malignant tumors of the central nervous system. Accurate preoperative grading is essential for individualized surgical planning and treatment selection; however, reliable non-invasive prediction tools integrating multimodal preoperative data remain limited. This study aimed to develop
[...] Read more.
Background: Gliomas are the most common primary malignant tumors of the central nervous system. Accurate preoperative grading is essential for individualized surgical planning and treatment selection; however, reliable non-invasive prediction tools integrating multimodal preoperative data remain limited. This study aimed to develop and internally validate an interpretable machine-learning model for non-invasive glioma grading. Methods: Clinical and imaging data from 400 patients with pathologically confirmed gliomas were retrospectively collected. Twenty-four preoperative variables were analyzed. The dataset was randomly divided into training and validation cohorts (7:3). Feature selection was performed using a combination of the Boruta algorithm and logistic regression analyses, followed by correlation filtering. Seventeen machine-learning algorithms were benchmarked using five-fold cross-validation, and the optimal model was evaluated in the independent validation cohort using ROC analysis, calibration assessment, precision–recall curves, and decision curve analysis. Model interpretability was examined using SHAP. Results: Eight key predictors were identified, including age, focal neurological deficits, midline shift, tumor laterality, tumor lobar location, enhancing tumor volume, and MRS-derived Cho/NAA and Cho/Cr ratios. The Random Forest model achieved an area under the ROC curve of 0.946 (95% CI: 0.902–0.989) in the validation cohort. Calibration analysis demonstrated reasonable agreement between predicted and observed outcomes, and the precision–recall curve yielded an average precision of 0.98. Decision curve analysis indicated net clinical benefit across relevant probability thresholds. Conclusions: A multimodal machine-learning model integrating clinical, structural imaging, and MRS-derived metabolic features was developed and internally validated for non-invasive preoperative glioma grading. The model showed good discrimination and calibration and provided individualized probability estimates, suggesting potential value for preoperative risk stratification. However, clinical deployment remains premature, and further external validation is required.
Full article
(This article belongs to the Section Cancer Pathophysiology)
►▼
Show Figures
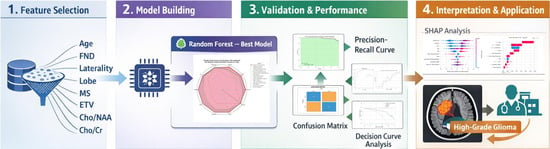
Graphical abstract
Open AccessReview
Sarcopenia as a Marker of Immunometabolic Vulnerability in Pancreatic Ductal Adenocarcinoma
by
Mukund Karthik, Sara Shahrestani, Jin-soo Park, Christian Ratnayake and Charbel Sandroussi
Cancers 2026, 18(8), 1205; https://doi.org/10.3390/cancers18081205 - 9 Apr 2026
Abstract
Despite advances in surgical technique and perioperative care, pancreatic ductal adenocarcinoma (PDAC) remains associated with poor survival. Sarcopenia is highly prevalent in PDAC and is consistently associated with inferior survival and reduced tolerance of systemic therapy. However, interventions primarily aimed at increasing muscle
[...] Read more.
Despite advances in surgical technique and perioperative care, pancreatic ductal adenocarcinoma (PDAC) remains associated with poor survival. Sarcopenia is highly prevalent in PDAC and is consistently associated with inferior survival and reduced tolerance of systemic therapy. However, interventions primarily aimed at increasing muscle mass through nutritional supplementation and resistance-based exercise have yielded limited improvements in clinically meaningful postoperative outcomes. This has prompted increasing interest in sarcopenia as a marker of broader biological vulnerability rather than isolated physical deconditioning. Emerging clinical, translational, and experimental evidence demonstrates that skeletal muscle and adipose tissue function as active immunometabolic organs, and that cancer-associated inflammatory pathways drive early muscle loss, immune dysfunction, and impaired physiological recovery. Across multiple clinical cohorts, sarcopenia is reproducibly associated with worse overall survival and failure to complete adjuvant therapy, but not consistently with increased postoperative complications, suggesting that its prognostic relevance lies in impaired recovery and oncological fitness rather than immediate surgical risk. Translational studies further indicate that sarcopenia identifies patients with reduced antitumor immune competence, particularly in early-stage disease. This review synthesizes current evidence linking sarcopenia, immune dysfunction, and surgical outcomes in PDAC and examines implications for perioperative care. We propose that immunometabolic-informed prehabilitation, integrated with existing nutritional and exercise strategies, may represent a more effective approach to improving recovery, treatment tolerance, and durable oncological outcomes following PDAC resection.
Full article
(This article belongs to the Special Issue Improving Surgical Care for Patients with Hepatobiliary and Pancreatic Cancers)
Open AccessReview
Light-Activated Iron Oxide Nanoparticles in Cancer Treatment: Synergistic Roles in Photothermal and Photodynamic Therapy
by
Aynura Karimova, Habiba Shirinova, Toghrul Sadikhov, Javahir Hajibabazade, Sabina Hajizada, Yerkeblan Tazhbayev, Abdumutolib A. Atakhanov, Samir N. Babayev, Christoph Reissfelder and Vugar Yagublu
Cancers 2026, 18(8), 1203; https://doi.org/10.3390/cancers18081203 - 9 Apr 2026
Abstract
Iron oxide nanoparticles have emerged as multifunctional compounds with prominent potential in cancer theranostics, particularly in photothermal therapy (PTT) and photodynamic therapy (PDT). Their unique electronic and crystal structures, such as the dispersion of Fe2+ and Fe3+ ions and d-orbital splitting,
[...] Read more.
Iron oxide nanoparticles have emerged as multifunctional compounds with prominent potential in cancer theranostics, particularly in photothermal therapy (PTT) and photodynamic therapy (PDT). Their unique electronic and crystal structures, such as the dispersion of Fe2+ and Fe3+ ions and d-orbital splitting, contribute to their magnetic and catalytic properties. In PTT, Fe3O4 nanoparticles exhibit moderate near-infrared (NIR) absorption and photothermal conversion efficiency, which can be enhanced through adjustments in particle size, surface modification, and combinations with other components. In PDT, Fe3O4 nanoparticles demonstrate intrinsic peroxidase-like catalytic activity, facilitating Fenton and photo-Fenton reactions that generate reactive oxygen species (ROS), including hydroxyl radicals (•OH), thereby amplifying oxidative stress in cancer cells. These nanoparticles can also function as carriers for photosensitisers (PS), promoting targeted delivery and enhanced ROS generation. Multifunctional nanomaterials that integrate Fe3O4 with other therapeutic agents and targeting ligands have demonstrated synergistic antitumour effects through amplified photothermal, photodynamic, chemodynamic, and chemotherapeutic mechanisms. Despite certain drawbacks, such as relatively low NIR absorption and challenges in optimising delivery and light activation, ongoing improvements in Fe3O4-based nanoplatforms present significant potential for enhancing treatment outcomes and the precision of cancer therapy. This article systematically explores the synergistic role of Fe3O4 nanoparticles in PTT and PDT, encompassing their magnetic and catalytic characteristics. Additionally, it focuses on multifunctional hybrid nanoplatforms that combine Fe3O4 with targeting or imaging agents, highlighting their potential to enhance therapeutic precision.
Full article
(This article belongs to the Section Molecular Cancer Biology)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Segmentectomy Versus Lobectomy in Patients with Stage IA Lung Adenocarcinoma: Long-Term Survival in a Propensity Score-Matched Cohort
by
Zhangfeng Huang, Tenglong Luo, Zuhan Geng, Qi Gao and Yongde Liao
Cancers 2026, 18(8), 1202; https://doi.org/10.3390/cancers18081202 - 9 Apr 2026
Abstract
Background: Whether there are differences in clinical outcomes between segmentectomy and lobectomy in patients with early-stage lung adenocarcinoma (LUAD) remains uncertain. This study aimed to compare all-cause mortality and lung cancer-specific mortality in patients with lung tumors ≤ 20 mm undergoing these
[...] Read more.
Background: Whether there are differences in clinical outcomes between segmentectomy and lobectomy in patients with early-stage lung adenocarcinoma (LUAD) remains uncertain. This study aimed to compare all-cause mortality and lung cancer-specific mortality in patients with lung tumors ≤ 20 mm undergoing these two procedures. Methods: Patients with stage IA lung adenocarcinoma (≤20 mm) who underwent segmentectomy or lobectomy were identified from the SEER database (2008–2022). Propensity score matching (PSM) was applied to balance baseline characteristics. Kaplan–Meier curves depicted overall survival and lung cancer-specific survival. Multivariable Cox proportional hazards models were used to evaluate associations between surgical procedures and mortality, reporting hazard ratios (HRs) with 95% confidence intervals (CIs). Results: Among 9641 patients, 1065 (11.0%) underwent segmentectomy. After 1:1 PSM, 2028 patients had well-balanced covariates. The median follow-up was 43.0 months. In the lobectomy group, 158 all-cause deaths (35.1 per 1000 person-years) and 66 lung cancer-specific deaths (14.7 per 1000 person-years) occurred, compared with 176 and 80 events in the segmentectomy group (39.9 and 18.1 per 1000 person-years, respectively). Multivariable Cox regression demonstrated that segmentectomy, compared with lobectomy, was not associated with a higher risk of all-cause mortality (adjusted HR [aHR], 1.07, 95% CI 0.86–1.34) or lung cancer-specific mortality (aHR, 1.18, 95% CI 0.84–1.64). The results were consistent across tumor differentiation subgroups. Conclusions: Among patients with early-stage (≤20 mm) LUAD, segmentectomy was not associated with an increased risk of all-cause or lung cancer-specific mortality compared with lobectomy. Further studies with larger sample sizes are warranted to validate these findings.
Full article
(This article belongs to the Special Issue Advances in Thoracic Oncology Research)
►▼
Show Figures

Graphical abstract
Open AccessReview
The Spectrum of Venetoclax-Based Treatments in Acute Myeloid Leukemia
by
Elvira Pelosi, Germana Castelli and Ugo Testa
Cancers 2026, 18(8), 1201; https://doi.org/10.3390/cancers18081201 - 9 Apr 2026
Abstract
Background/Objectives: In recent years there has been a consistent development of clinical studies surrounding the incorporation of the B-cell lymphoma 2 (BCL-2) inhibitor venetoclax (VEN) into the treatment of acute myeloid leukemia (AML) Methods: A search of the literature showed a
[...] Read more.
Background/Objectives: In recent years there has been a consistent development of clinical studies surrounding the incorporation of the B-cell lymphoma 2 (BCL-2) inhibitor venetoclax (VEN) into the treatment of acute myeloid leukemia (AML) Methods: A search of the literature showed a tremendous development of experimental and clinical studies evaluating the impact of VEN-based regimens in the treatment of AML patients. This review comprehensively analyzes the available scientific evidence—including prospective clinical trials, retrospective cohorts, and real-world studies—to summarize current knowledge on the efficacy and safety of venetoclax-based regimens in AML patients. Results: Recent studies have evaluated VEN-based regimens in newly diagnosed (ND) and refractory/relapsed (R/R) AML patients, showing the efficacy of these treatments. VEN with hypomethylating agents (HMAs) became the standard-of-care for elderly/unfit AML patients. Recent studies strongly support the effectiveness of VEN-based regimens in frontline treatment of adult AML patients eligible for intensive treatments. VEN-based therapies were also used in combination with targeted therapies, thus generating triplet therapeutic regimens that are under evaluation for the treatment of some AML subtypes. However, the response to VEN+HMAs is highly variable and in part depends on tumor genetics; some patients are resistant or relapse following VEN-based treatments and future studies will be required to develop therapeutic strategies able to circumvent resistance and to identify patients at high risk of relapse. Prospective randomized trials are required to establish the real efficacy of VEN in various clinical settings and to refine maintenance and discontinuation strategies, aiming to improve long-term outcomes and to make more safe treatments based on VEN.
Full article
(This article belongs to the Special Issue Precision Medicine in Acute Myeloid Leukemia)
Open AccessArticle
Total Neoadjuvant Therapy Outcomes and Watch-and-Wait Feasibility in Locally Advanced Rectal Cancer: A Single-Institution Retrospective Cohort Study
by
Manuel Ramanović, Franc Anderluh, Ana Jeromen Peressutti, Petar Korošec, Irena Oblak, Ajra Šečerov Ermenc and Vaneja Velenik
Cancers 2026, 18(8), 1200; https://doi.org/10.3390/cancers18081200 - 9 Apr 2026
Abstract
Background/Objectives: Total neoadjuvant therapy (TNT), integrating systemic chemotherapy and radiotherapy before surgery or surveillance, has become a standard approach for locally advanced rectal cancer (LARC). However, optimal sequencing strategies and long-term outcomes of watch-and-wait (W&W) following sandwich TNT remain insufficiently characterized. We
[...] Read more.
Background/Objectives: Total neoadjuvant therapy (TNT), integrating systemic chemotherapy and radiotherapy before surgery or surveillance, has become a standard approach for locally advanced rectal cancer (LARC). However, optimal sequencing strategies and long-term outcomes of watch-and-wait (W&W) following sandwich TNT remain insufficiently characterized. We evaluated oncologic outcomes and treatment response in patients treated with an institutional sandwich TNT protocol. Methods: We conducted a retrospective cohort study of consecutive patients with LARC treated with sandwich TNT (induction chemotherapy followed by hypofractionated intensity-modulated radiotherapy with simultaneous integrated boost [IMRT-SIB] chemoradiotherapy and consolidation chemotherapy) at the Institute of Oncology Ljubljana between 2016 and 2023. The primary endpoint was an overall complete response (CR; pathological [pCR] and clinical [cCR]). Secondary endpoints included tumor regression grade (TRG), major pathological response (MPR), R0 resection rate, organ preservation, overall survival (OS), and disease-free survival (DFS). Results: Among 205 patients (median age 61 years), overall CR was 29.5% (pCR 19.3% and cCR 10.2%). Major pathological response (TRG 3–4) occurred in 37.6%. R0 resection was achieved in 94.5%. In the W&W cohort (n = 21), local regrowth occurred in 33.3% (95% CI, 14.6–57.0%) over a median follow-up of 4.96 years. Total mesorectal excision (TME)-free survival at 5 years was 73.1% (95% CI, 55.0–97.2%). Estimated 5-year OS was 81.1% (95% CI, 75.5–87.2%) and 5-year DFS was 75.2% (95% CI, 69.0–82.0). In multivariable analysis, non-R0 resection (HR 6.06, 95% CI, 1.99–18.42), MRI circumferential resection margin positivity (HR 3.11, 95% CI, 1.53–6.33), and MRI extramural vascular invasion positivity (HR 1.97, 95% CI, 1.05–3.91) remained independent predictors of DFS. Conclusions: Institutional sandwich TNT yields meaningful tumor response and durable survival in MRI-defined high-risk LARC. Structured W&W offers organ preservation with acceptable oncologic control under intensive surveillance.
Full article
(This article belongs to the Section Cancer Survivorship and Quality of Life)
►▼
Show Figures
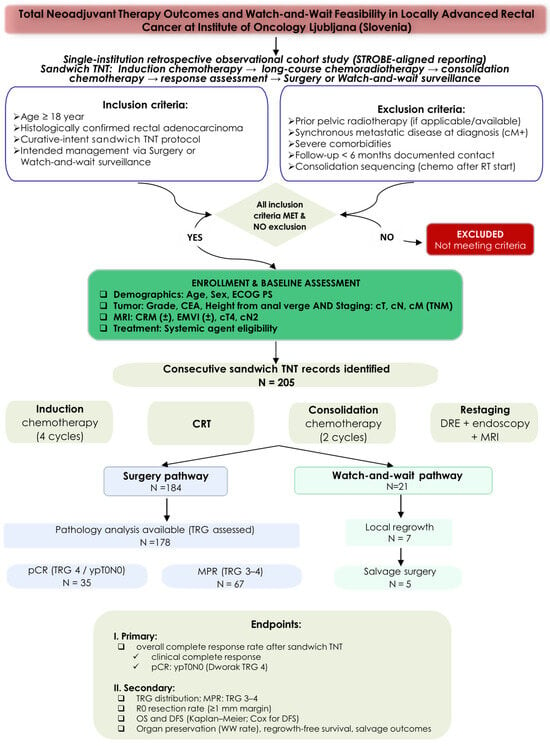
Figure 1
Open AccessArticle
Impact of Postoperative Liver Injury on the Oncological Short- and Long-Term Outcome After Liver Resection for Hepatocellular Carcinoma
by
Katharina Lang, Oliver Beetz, Iakovos Amygdalos, Clara A. Weigle, Bengt A. Wiemann, Julian Palzer, Sebastian Cammann, Georg Wiltberger, Thomas Vogel, Florian W. R. Vondran, Franziska A. Meister and Felix Oldhafer
Cancers 2026, 18(8), 1199; https://doi.org/10.3390/cancers18081199 - 9 Apr 2026
Abstract
Background: Postoperative liver injury remains a major challenge after curative-intended liver resection for hepatocellular carcinoma (HCC), impacting both early complications and long-term survival. The recently introduced SAAR (Sum of AST/ALT Ratios) score may serve as a novel marker for predicting postoperative outcomes. Methods:
[...] Read more.
Background: Postoperative liver injury remains a major challenge after curative-intended liver resection for hepatocellular carcinoma (HCC), impacting both early complications and long-term survival. The recently introduced SAAR (Sum of AST/ALT Ratios) score may serve as a novel marker for predicting postoperative outcomes. Methods: This retrospective single-center study included 213 patients undergoing liver resection for HCC between January 2007 and October 2024. The SAAR score was calculated using AST/ALT ratios on postoperative days 1 and 3 and correlated with post-hepatectomy liver failure (PHLF), disease-free survival (DFS), and overall survival (OS). The predictive performance of the SAAR score was compared with that of the combined APRI + ALBI score. Results: Patients with SAAR ≥ 2 showed significantly higher rates of PHLF (OR = 2.5, p = 0.019) and impaired long-term outcomes. Median DFS and OS were significantly reduced in the SAAR ≥ 2 group (7 vs. 24 months, p < 0.001; 12 vs. 32 months, p = 0.004, respectively). Multivariate Cox regression confirmed SAAR ≥ 2 as an independent predictor for DFS (HR = 2.1) and OS (HR = 1.9). The APRI + ALBI score also demonstrated strong preoperative predictive value for PHLF (AUC = 0.854) but not for long-term outcome.
Full article
(This article belongs to the Special Issue Advancements in Surgical and Multidisciplinary Management of Hepatopancreaticobiliary Cancers)
►▼
Show Figures
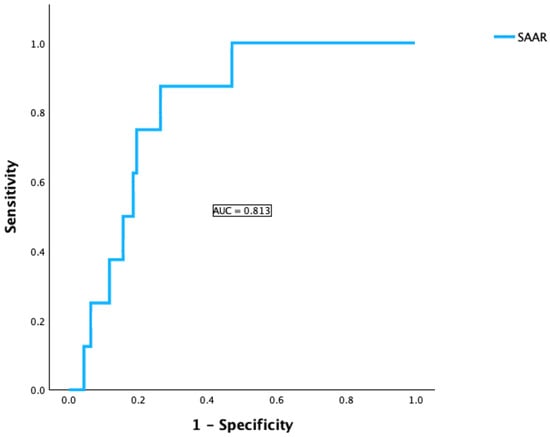
Figure 1
Open AccessArticle
Perioperative Blood Transfusion Impairs Overall Survival Following Radical Resection for Colorectal Cancer: A Propensity Score-Matched Analysis
by
Xiaoran Wang, Zesong Meng, Guangjun Wang, Guiying Wang and Lihua Liu
Cancers 2026, 18(8), 1198; https://doi.org/10.3390/cancers18081198 - 9 Apr 2026
Abstract
Background/Objectives: Perioperative blood transfusion (BTF) remains controversial regarding its impact on oncological outcomes in colorectal cancer (CRC). This study aimed to evaluate the association between BTF, transfusion volume, and long-term prognosis in CRC patients undergoing radical resection. Methods: We conducted a retrospective cohort
[...] Read more.
Background/Objectives: Perioperative blood transfusion (BTF) remains controversial regarding its impact on oncological outcomes in colorectal cancer (CRC). This study aimed to evaluate the association between BTF, transfusion volume, and long-term prognosis in CRC patients undergoing radical resection. Methods: We conducted a retrospective cohort study of 1777 CRC patients who underwent radical surgery at the Fourth Hospital of Hebei Medical University between December 2007 and April 2015. Propensity score matching (PSM) was applied to minimize selection bias between BTF and non-BTF groups. Logistic regression identified factors associated with BTF receipt. Cox proportional hazards models assessed the association between BTF and 5-year overall survival (OS). X-tile analysis determined optimal cut-off values for transfusion volume stratification. Results: Among 1777 patients, 729 (41.02%) received BTF. After PSM, 524 well-matched pairs showed balanced baseline characteristics. Intestinal obstruction was independently associated with BTF requirement (p < 0.001). The BTF group demonstrated significantly inferior 1-, 3-, and 5-year OS compared with non-BTF groups in both overall and PSM cohorts (all p < 0.01). Multivariate Cox analysis identified BTF as an independent adverse prognostic factor (HR = 1.44, 95% CI 1.09–1.89, p = 0.01). X-tile analysis stratified patients into non-BTF, small-volume (≤4 units), and massive-volume (>4 units) groups. Massive-volume transfusion showed the poorest survival outcomes (p < 0.0001) and was independently associated with worse OS (HR = 1.61, 95% CI 1.18–2.20, p = 0.003). Supplementary analyses indicated that no independent association was observed between survival outcomes and either preoperative inflammatory markers or the specific timing and type of transfusion. Conclusions: BTF, particularly when exceeding 4 units, independently predicts inferior long-term survival in CRC patients following radical resection. These findings support the implementation of restrictive transfusion strategies in perioperative CRC management. Given its retrospective observational design, this study shows an association but does not establish causality, and our findings should be interpreted in light of the aforementioned limitations.
Full article
(This article belongs to the Section Clinical Research of Cancer)
►▼
Show Figures

Figure 1
Open AccessArticle
A Multigene Signature for Prognostic Stratification of Nasopharyngeal Carcinoma
by
Yingying Liang, Zhiwen Mo and Muy-Teck Teh
Cancers 2026, 18(8), 1197; https://doi.org/10.3390/cancers18081197 - 9 Apr 2026
Abstract
Background: We previously pioneered a multigene mRNA test, qMIDSV2, validated through an international multicohort study with geographically and ethnically diverse oral squamous cell carcinoma (OSCC) patients from Europe and Asia. This study aimed to repurpose the qMIDSV2 test for
[...] Read more.
Background: We previously pioneered a multigene mRNA test, qMIDSV2, validated through an international multicohort study with geographically and ethnically diverse oral squamous cell carcinoma (OSCC) patients from Europe and Asia. This study aimed to repurpose the qMIDSV2 test for nasopharyngeal carcinoma (NPC). A molecular test independent of Epstein–Barr virus (EBV) status would be clinically useful for risk stratification in NPC patients with undetectable or low levels of EBV. Methods: This study investigated a Chinese cohort of 62 participants (18 donated normal nasopharyngeal mucosa (NPM) and 44 donated NPC tissue samples). Messenger RNA levels of 16 genes in each sample were quantified using the qPCR method, and an algorithm computed a malignancy index for cancer risk stratification. Results: We identified a unique 10-gene panel (containing eight target genes, namely NEK2, INHBA, FOXM1, TOP2A, BIRC5, CXCL8, NR3C1, and IVL, relative to two reference genes, YAP1 and POLR2A, collectively named qMIDSNPC) that demonstrated the best overall diagnostic performance in segregating NPM from NPC, with AUC = 0.909 and positive/negative predictive values of 91% PPV and 78% NPV, respectively. Furthermore, we demonstrated prognostic value of qMIDSNPC in segregating NPM from NPC stage III + IV, with AUC = 0.936, 92% PPV, and 84% NPV. Conclusions: Here, we present a simple qPCR-based 10-gene mRNA test, qMIDSNPC, with potential clinical utilities for rapid (1 h) prognostic stratification of NPC. Further studies involving geographically and ethnically independent NPC cohorts would be needed to validate the clinical use of qMIDSNPC in non-endemic NPC populations.
Full article
(This article belongs to the Section Cancer Biomarkers)
►▼
Show Figures
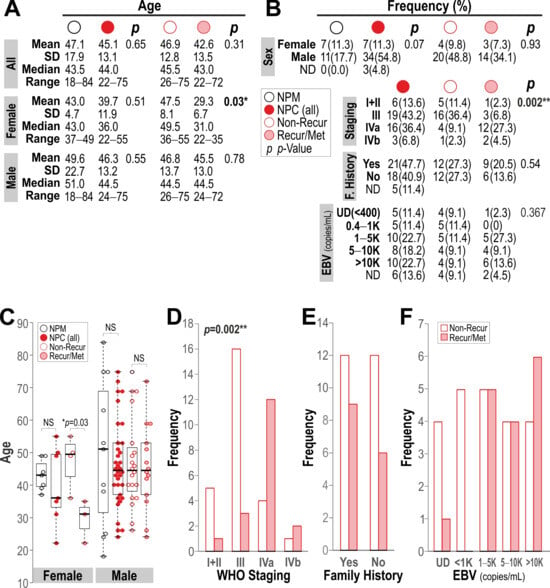
Figure 1
Open AccessReview
Triptolide: A Narrative Review of Its Traditional Use, Derivatives, Pharmacology, Antitumor Effect, and Clinical Applications
by
Yibo Geng, Bettina Kritzer and Javad Nazarian
Cancers 2026, 18(8), 1196; https://doi.org/10.3390/cancers18081196 - 9 Apr 2026
Abstract
Triptolide (TPL), a diterpenoid derived from the Chinese medicinal plant Tripterygium wilfordii, exhibits broad-spectrum biological and pharmacological activities, although its clinical translation is limited by systemic toxicity. Recent advances in the development of TPL derivatives have created new therapeutic opportunities. This review
[...] Read more.
Triptolide (TPL), a diterpenoid derived from the Chinese medicinal plant Tripterygium wilfordii, exhibits broad-spectrum biological and pharmacological activities, although its clinical translation is limited by systemic toxicity. Recent advances in the development of TPL derivatives have created new therapeutic opportunities. This review summarizes current knowledge of triptolide, with a focus on TPL’s toxicity profile, derivative strategies, and antitumor mechanisms across different tumor types, including glioma, pancreatic tumor, leukemia, lung cancer, gastric cancer and others. We also summarize the plant’s origin and traditional uses, TPL’s pharmacokinetics (PKs), and relevant clinical trials against tumors. The main mechanism of the TPL antitumor effect is to interfere with ATPase of XPB by covalently binding to it, as well as inducing the rapid depletion of RPB1 via hyperphosphorylation and ubiquitination. We also reviewed systemic toxicity including neuro-, cardio-, oto-, nephron-, hepato-, and hemato-toxicity, as well as digestive and reproductive toxicity. Finally, we searched clinical trial databases across three platforms for tumors and concluded that Minnelide has strong clinical potential for solid tumors. By critically evaluating TPL from multiple dimensions, specifically its traditional use, chemical derivatization, pharmacokinetics, antitumor mechanisms, toxicity, and clinical trials, this review aims to inform future strategies that maximize therapeutic efficacy while minimizing adverse effects.
Full article
(This article belongs to the Special Issue Preclinical and Clinical Research on the Efficacy of Anticancer Drugs)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Cancers Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cancers, JCM, Nanomaterials, Pharmaceutics, JNT
Application of Nanomaterials and Nanobiotechnology in Cancer
Topic Editors: Ayan Kumar Barui, Susheel Kumar NethiDeadline: 31 May 2026
Topic in
Cancers, Diagnostics, Medicina, Current Oncology
Prostate Cancer: Symptoms, Diagnosis & Treatment—3rd Edition
Topic Editors: Ana Faustino, Lúcio Lara Santos, Paula OliveiraDeadline: 30 June 2026
Topic in
Cancers, Current Oncology, JCM, Medicina, Onco
Cancer Biology and Radiation Therapy: 2nd Edition
Topic Editors: Chang Ming Charlie Ma, Ka Yu Tse, Ming-Yii Huang, Mukund SeshadriDeadline: 25 July 2026
Topic in
Biomedicines, Biomolecules, Cancers, Cells, Hematology Reports, IJMS
Advances in Molecular Pathogenesis and Targeted Therapies for Multiple Myeloma
Topic Editors: Chung Hoow Kok, Cindy H. S. Lee, Claudio CerchioneDeadline: 3 August 2026

Conferences
Special Issues
Special Issue in
Cancers
Clinical and Molecular Biomarkers in Breast Cancer Management
Guest Editor: Zahi I. MitriDeadline: 15 April 2026
Special Issue in
Cancers
State-of-the-Art Surgical Treatment for Lung Cancers
Guest Editors: Andrea Axtell, Malcolm DeCampDeadline: 15 April 2026
Special Issue in
Cancers
Recent Advances in Radiological Imaging for Head and Neck Squamous Cell Carcinoma
Guest Editors: Mintu Nath, Fergus McKiddieDeadline: 15 April 2026
Special Issue in
Cancers
The Bone Marrow Microenvironment in Myelodysplastic Syndromes—Pathogenesis, Immune Evasion, and Therapeutic Opportunities
Guest Editor: Ciro Roberto RinaldiDeadline: 15 April 2026
Topical Collections
Topical Collection in
Cancers
Women’s Cancers Risk: Breast Cancer, Ovarian Cancer and Endometrial Cancer
Collection Editor: Ranjit Manchanda
Topical Collection in
Cancers
Molecular Signaling Pathways and Networks in Cancer
Collection Editors: Shihori Tanabe, You Song
Topical Collection in
Cancers
Combination Therapies in Cancers
Collection Editors: Patrycja Nowak-Sliwinska, Arjan W. Griffioen
Topical Collection in
Cancers
Emerging Therapeutics in Advanced Melanoma
Collection Editors: Jeremy S. Bordeaux, Luke D. Rothermel













