Journal Description
Molbank
Molbank
is an international, peer-reviewed, open access journal comprised of a unique collection of one-compound-per-paper short notes on synthetic compounds and natural products published bimonthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Reaxys, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 12.7 days after submission; acceptance to publication is undertaken in 2.7 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
0.4 (2024)
Latest Articles
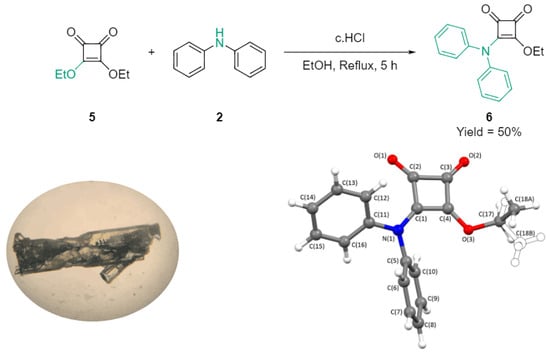
3-(Diphenylamino)-4-ethoxycyclobut-3-ene-1,2-dione
Molbank 2026, 2026(3), M2182; https://doi.org/10.3390/M2182 - 25 May 2026
Abstract
The title compound, 3-(diphenylamino)-4-ethoxycyclobut-3-ene-1,2-dione (6), was prepared by reaction of diphenylamine (2) with diethyl squarate (DES; 5) as part of our ongoing studies on monosquarate-amides. Following purification and recrystallisation, the product was isolated as a green crystalline solid.
[...] Read more.
The title compound, 3-(diphenylamino)-4-ethoxycyclobut-3-ene-1,2-dione (6), was prepared by reaction of diphenylamine (2) with diethyl squarate (DES; 5) as part of our ongoing studies on monosquarate-amides. Following purification and recrystallisation, the product was isolated as a green crystalline solid. Its structure was established by spectroscopic methods, including FTIR, 1H NMR, 13C NMR and HRMS, and was unambiguously confirmed by single-crystal X-ray diffraction. This work provides access to a previously unreported diphenylamino-substituted squaric acid derivative.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►
Show Figures
attachment
Supplementary material:
Supplementary File 1 (ZIP, 6153 KB)
Supplementary File 2 (MOL, 2 KB)
Supplementary File 4 (MOL, 2 KB)
Supplementary File 1 (ZIP, 6153 KB)
Supplementary File 2 (MOL, 2 KB)
Supplementary File 4 (MOL, 2 KB)
Open AccessCommunication
Eco-Friendly Synthesis of Perimidine Derivatives Using Recyclable Fe3O4@Nano-Cellulose/Ti(IV)
by
Ghaffar Pasdar, Abdolhamid Bamoniri and Bi Bi Fatemeh Mirjalili
Molbank 2026, 2026(3), M2181; https://doi.org/10.3390/M2181 (registering DOI) - 21 May 2026
Abstract
A novel bio-capable method has been implemented for the synthesis of newly substituted derivatives of perimidine using Fe3O4@nano-cellulose/Ti(IV) as a magnetic, sustainable, and eco-friendly Lewis acid nanocatalyst. The catalyst was thoroughly characterized by XRD, FESEM, and TGA analyses, confirming
[...] Read more.
A novel bio-capable method has been implemented for the synthesis of newly substituted derivatives of perimidine using Fe3O4@nano-cellulose/Ti(IV) as a magnetic, sustainable, and eco-friendly Lewis acid nanocatalyst. The catalyst was thoroughly characterized by XRD, FESEM, and TGA analyses, confirming its crystalline structure, uniform nanoscale morphology, and high thermal stability. The reaction proceeded smoothly in eco-friendly solvents, providing outstanding yields under mild and rapid conditions, especially with ultrasonics. The catalyst, derived from renewable materials, exhibited remarkable activity, easy magnetic recovery, and excellent reusability over several cycles without significant loss of efficiency. Spectral characterization, IR, 1H NMR, 13C NMR, 19F NMR, and HRMS analyses verified that perimidine derivatives were synthesized properly. This sustainable and efficient approach demonstrates the prospect of green Lewis acid nanocatalysts for the sustainable synthesis of valuable heterocyclic compounds.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures

Figure 1
Open AccessCommunication
Synthesis and Characterization of Piperazine-Linked Eugenol Derivative
by
Munishama Gowda Yenagunte Narayanaswamy, Sujeet Kumar, Siddamsetty Ramachandra Setty, Basavaraj Metikurki and Chaluvaraju Kalamuddanadoddi Chaluvegowda
Molbank 2026, 2026(3), M2180; https://doi.org/10.3390/M2180 (registering DOI) - 20 May 2026
Abstract
In the present work, we synthesize 2-(4-allyl-2-methoxyphenoxy)-1-(4-phenylpiperazin-1-yl)ethan-1-one, a semisynthetic derivative of the natural product eugenol. The compound was synthesized via a three-step synthetic pathway involving esterification, hydrolysis, and subsequent coupling with 4-phenylpiperazine, as confirmed by FTIR, 1H NMR, 13C NMR, and
[...] Read more.
In the present work, we synthesize 2-(4-allyl-2-methoxyphenoxy)-1-(4-phenylpiperazin-1-yl)ethan-1-one, a semisynthetic derivative of the natural product eugenol. The compound was synthesized via a three-step synthetic pathway involving esterification, hydrolysis, and subsequent coupling with 4-phenylpiperazine, as confirmed by FTIR, 1H NMR, 13C NMR, and mass spectrometric data.
Full article
(This article belongs to the Section Natural Product Chemistry)
►▼
Show Figures

Scheme 1
Open AccessCommunication
Preparative Enzymatic Desymmetrization of (Acetyl-Leu-Pro-Lys)2-R110 Using Bovine Trypsin Variant D189S
by
Sarah Stoppe, Marianne Hahn, Martin Dauner and Frank Bordusa
Molbank 2026, 2026(3), M2179; https://doi.org/10.3390/M2179 (registering DOI) - 20 May 2026
Abstract
Rhodamine 110 (R110) peptide conjugates are widely used fluorogenic substrates in proteolytic assays; however, their inherent symmetry results in two identical hydrolysis sites, complicating their application as well-defined substrates. Here, we report a preparative enzymatic strategy for the desymmetrization of the symmetric derivative
[...] Read more.
Rhodamine 110 (R110) peptide conjugates are widely used fluorogenic substrates in proteolytic assays; however, their inherent symmetry results in two identical hydrolysis sites, complicating their application as well-defined substrates. Here, we report a preparative enzymatic strategy for the desymmetrization of the symmetric derivative (Acetyl-Leu-Pro-Lys)2-R110 using the bovine trypsin variant D189S. Due to pronounced differences in the rates of the two sequential hydrolysis steps, a mono-substituted intermediate accumulates under controlled reaction conditions. On a preparative scale, Acetyl-Leu-Pro-Lys-R110 was generated by partial hydrolysis and isolated by preparative HPLC in 28.8% yield and 95.8% purity. The structure of the asymmetric product was fully characterized by NMR and high-resolution mass spectrometry. This work demonstrates that selective enzymatic hydrolysis provides a simple and effective preparative route to asymmetric Rhodamine 110 derivatives, offering a practical alternative to conventional multistep synthetic approaches and enabling improved substrate design for kinetic studies.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Figure 1
Open AccessShort Note
2-Methoxy-4-[5-(2-oxo-1,3-dioxolan-4-yl)-4,5-dihydroisoxazol-3-yl]phenyl 4-n-decyloxybenzoate
by
Itamar Luís Gonçalves, Aloir Antonio Merlo, Bruna Thatise Batistel, Leonardo Rossner Wbatuba and Henrique de Aguiar Mello
Molbank 2026, 2026(3), M2178; https://doi.org/10.3390/M2178 (registering DOI) - 13 May 2026
Abstract
This study addresses the synthesis of a new liquid crystalline compound featuring a 3,5-disubstituted isoxazoline and a 1,3-dioxolan-2-one ring, and renewable aromatic building blocks derived from vanilin and benzoic acid. The target compound was synthesized through a multistep synthetic route involving alkylation, esterification,
[...] Read more.
This study addresses the synthesis of a new liquid crystalline compound featuring a 3,5-disubstituted isoxazoline and a 1,3-dioxolan-2-one ring, and renewable aromatic building blocks derived from vanilin and benzoic acid. The target compound was synthesized through a multistep synthetic route involving alkylation, esterification, oxime formation, and a 1,3-dipolar cycloaddition reaction. The synthesized compound, 2-methoxy-4-[5-(2-oxo-1,3-dioxolan-4-yl)-4,5-dihydroisoxazol-3-yl]phenyl 4-n-decyloxybenzoate, was isolated and fully characterized by spectroscopic techniques. Liquid crystal behavior was evaluated by DSC and POM. The monotropic mesomorphic behavior of the title compound was dictated by the interplay between molecular architecture and intermolecular organization, with the methoxy substituent and the 1,3-dioxolan-2-one ring critically influencing phase stability and texture morphology. These findings suggest a structure–property relationship and guide ongoing synthetic optimization toward achieving a stable enantiotropic liquid-crystalline phase and further ion-conduction experiments.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Figure 1
attachment
Supplementary material:
Supplementary File 1 (ZIP, 897 KB)
Supplementary File 2 (MOL, 1 KB)
Supplementary File 3 (INCHI, 395 B)
Supplementary File 4 (MOL, 1 KB)
Supplementary File 5 (MOL, 1 KB)
Supplementary File 6 (INCHI, 425 B)
Supplementary File 7 (MOL, 1 KB)
Supplementary File 8 (CIF, 232 KB)
Supplementary File 9 (CIF, 297 KB)
Supplementary File 1 (ZIP, 897 KB)
Supplementary File 2 (MOL, 1 KB)
Supplementary File 3 (INCHI, 395 B)
Supplementary File 4 (MOL, 1 KB)
Supplementary File 5 (MOL, 1 KB)
Supplementary File 6 (INCHI, 425 B)
Supplementary File 7 (MOL, 1 KB)
Supplementary File 8 (CIF, 232 KB)
Supplementary File 9 (CIF, 297 KB)
Open AccessCommunication
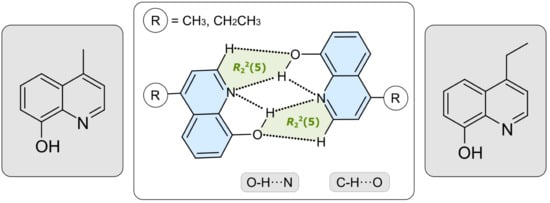
Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines
by
Sara Braun, Anke Schwarzer and Monika Mazik
Molbank 2026, 2026(3), M2176; https://doi.org/10.3390/M2176 (registering DOI) - 11 May 2026
Abstract
4-Methyl- (1) and 4-ethyl-8-hydroxyquinoline (2) crystallize from a mixture of diethyl ether and chloroform in the triclinic space group P
4-Methyl- (1) and 4-ethyl-8-hydroxyquinoline (2) crystallize from a mixture of diethyl ether and chloroform in the triclinic space group P
(This article belongs to the Section Structure Determination)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
2,2′-(Methylenebis(3,4-dimethoxy-6,1-phenylene))diacetic Acid
by
Savina Stoyanova and Milen G. Bogdanov
Molbank 2026, 2026(3), M2177; https://doi.org/10.3390/M2177 (registering DOI) - 11 May 2026
Abstract
The formation of side products is often unavoidable in organic synthesis; however, analyzing these secondary species provides practical insights into reaction pathways, mechanisms, and competing processes. This understanding is essential for optimizing reaction conditions, increasing product yields, and improving overall safety and efficiency.
[...] Read more.
The formation of side products is often unavoidable in organic synthesis; however, analyzing these secondary species provides practical insights into reaction pathways, mechanisms, and competing processes. This understanding is essential for optimizing reaction conditions, increasing product yields, and improving overall safety and efficiency. Additionally, side products can sometimes reveal unexpected molecular structures with valuable properties. In this study, we present the characterization of a compound that formed as a side product in a modified Pictet-Spengler reaction. The molecular structure of 2,2′-(methylenebis(3,4-dimethoxy-6,1-phenylene))diacetic acid was elucidated using a combination of NMR, FTIR and UV–Vis spectroscopic techniques, as well as HRMS analysis.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures

Figure 1
Open AccessCommunication
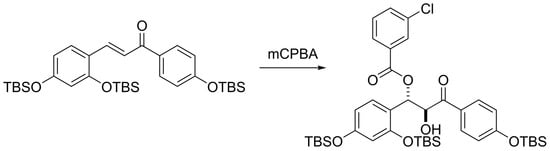
Formation of a β-(3-Chlorobenzoyloxy)-α-hydroxyketone from a TBS-Protected Chalcone upon Oxidation with m-Chloroperbenzoic Acid
by
Sonia Berenguel-Gómez, Irene Moreno-Gutiérrez, Jenifer Acien-García, Manuel Muñoz-Dorado, Míriam Álvarez-Corral and Ignacio Rodríguez-García
Molbank 2026, 2026(3), M2175; https://doi.org/10.3390/M2175 (registering DOI) - 9 May 2026
Abstract
A new tert-butyldimethylsilyl-protected polyoxygenated chalcone was prepared by Claisen–Schmidt condensation of suitably protected acetophenone and benzaldehyde derivatives. Treatment of this chalcone with m-chloroperbenzoic acid (mCPBA) afforded β-(3-chlorobenzoyloxy)-α-hydroxyketone 5, which was fully characterized by spectroscopic methods. The structure of 5 is consistent
[...] Read more.
A new tert-butyldimethylsilyl-protected polyoxygenated chalcone was prepared by Claisen–Schmidt condensation of suitably protected acetophenone and benzaldehyde derivatives. Treatment of this chalcone with m-chloroperbenzoic acid (mCPBA) afforded β-(3-chlorobenzoyloxy)-α-hydroxyketone 5, which was fully characterized by spectroscopic methods. The structure of 5 is consistent with initial epoxidation of the enone double bond followed by in situ nucleophilic opening of the transient epoxide by m-chlorobenzoate generated in the reaction medium. This work reports the preparation of the chalcone precursor and the characterization of the unexpected oxidation product 5.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
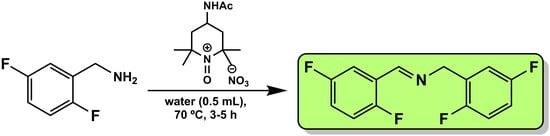
N-(2,5-Difluorobenzylidene)-1-(2,5-difluorobenzyl)methanimine
by
Tiffany L. Chen, Manisha Sharma and Nicholas E. Leadbeater
Molbank 2026, 2026(3), M2174; https://doi.org/10.3390/M2174 - 8 May 2026
Abstract
The conversion of 2,5-difluorobenzylamine to N-(2,5-difluorobenzylidene)-1-(2,5-difluorobenzyl)methanimine using an oxoammonium salt bearing the nitrate anion is reported. The reaction is operationally simple, and the product is obtained with good yield.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
(R)-10-((((2-(3,4-Dihydroxyphenyl)-2-hydroxyethyl)(methyl)carbamoyl)oxy)methyl)-1,3,5,5,7,9-hexamethyl-5H-dipyrrolo[1,2-c:2′,1′-f][1,3,2]diazaborinin-4-ium-5-uide
by
Mikhail A. Panfilov, Alexey Yu. Vorob’ev and Alexander E. Moskalensky
Molbank 2026, 2026(3), M2173; https://doi.org/10.3390/M2173 - 7 May 2026
Abstract
►▼
Show Figures
The BODIPY core has emerged as a versatile scaffold for the design of photoremovable protecting groups (PPGs). Herein, we report the synthesis of a novel BODIPY–epinephrine conjugate linked via a carbamate moiety, enabling light-triggered release of the active compound (epinephrine, also known as
[...] Read more.
The BODIPY core has emerged as a versatile scaffold for the design of photoremovable protecting groups (PPGs). Herein, we report the synthesis of a novel BODIPY–epinephrine conjugate linked via a carbamate moiety, enabling light-triggered release of the active compound (epinephrine, also known as adrenaline). The structure of the obtained product was confirmed by 1H and M 13C NMR spectroscopy as well as high-resolution mass spectrometry (HRMS). The described conjugate represents a potential tool for the photoactivated modulation of biologically relevant processes.
Full article
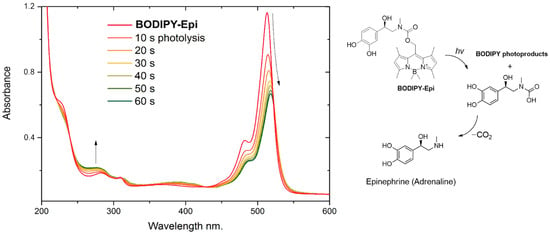
Figure 1
attachment
Supplementary material:
Supplementary File 1 (ZIP, 4554 KB)
Supplementary File 2 (MOL, 8 KB)
Supplementary File 3 (INCHI, 3 KB)
Supplementary File 4 (MOL, 8 KB)
Supplementary File 5 (MOL, 8 KB)
Supplementary File 6 (INCHI, 3 KB)
Supplementary File 7 (MOL, 8 KB)
Supplementary File 8 (MOL, 8 KB)
Supplementary File 9 (INCHI, 3 KB)
Supplementary File 10 (MOL, 8 KB)
Supplementary File 1 (ZIP, 4554 KB)
Supplementary File 2 (MOL, 8 KB)
Supplementary File 3 (INCHI, 3 KB)
Supplementary File 4 (MOL, 8 KB)
Supplementary File 5 (MOL, 8 KB)
Supplementary File 6 (INCHI, 3 KB)
Supplementary File 7 (MOL, 8 KB)
Supplementary File 8 (MOL, 8 KB)
Supplementary File 9 (INCHI, 3 KB)
Supplementary File 10 (MOL, 8 KB)
Open AccessCommunication
Synthesis of Three Bent Bis(imido-ferrocidiphenols)
by
Pascal Pigeon
Molbank 2026, 2026(3), M2170; https://doi.org/10.3390/M2170 - 6 May 2026
Abstract
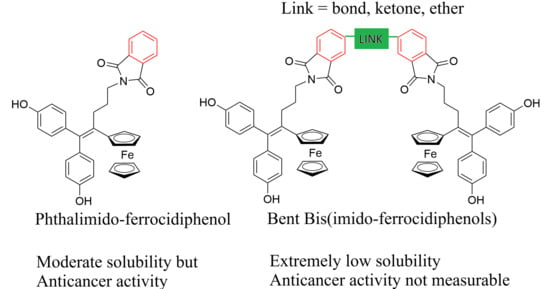
The ferrocidiphenol family brings together anticancer molecules featuring a [ferrocene-alkene-(p-phenol)2] motif that can form upon oxidation a quinone methide metabolite targeting cellular proteins. Adding an imide group (imido-ferrocidiphenol) dramatically enhanced the anticancer activity of ferrocidiphenol. We aimed to explore
[...] Read more.
The ferrocidiphenol family brings together anticancer molecules featuring a [ferrocene-alkene-(p-phenol)2] motif that can form upon oxidation a quinone methide metabolite targeting cellular proteins. Adding an imide group (imido-ferrocidiphenol) dramatically enhanced the anticancer activity of ferrocidiphenol. We aimed to explore whether molecules with two ferrociphenol motifs connected by bisimide moieties could provide even more effective compounds. Using amino-ferrocidiphenol and commercial bisanhydrides at reflux, for the first time, three symmetrical and bent bis(imido-ferrocidiphenols) were synthesized, with moderate yields, and characterized. However, these compounds were insoluble in most common organic solvents and unstable. This likely explains why their anticancer activity was nil.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
4-(6-Chloropyridin-3-yl)-6-cyclopropylpyrimidin-2-amine
by
Yusen Wang, Jian Lv and Yukun Qin
Molbank 2026, 2026(3), M2172; https://doi.org/10.3390/M2172 - 6 May 2026
Abstract
A novel nitrogen-containing heterocyclic compound, 4-(6-Chloropyridin-3-yl)-6-cyclopropylpyrimidin-2-amine, was designed and synthesized using 6-chloropyridin-3-aldehyde and cyclopropyl methyl ketone as starting materials. The structure of the target compound was characterized by 1H NMR, 13C NMR and HRMS, and the spectral data were consistent with
[...] Read more.
A novel nitrogen-containing heterocyclic compound, 4-(6-Chloropyridin-3-yl)-6-cyclopropylpyrimidin-2-amine, was designed and synthesized using 6-chloropyridin-3-aldehyde and cyclopropyl methyl ketone as starting materials. The structure of the target compound was characterized by 1H NMR, 13C NMR and HRMS, and the spectral data were consistent with the expected structure, confirming the correctness of the product.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
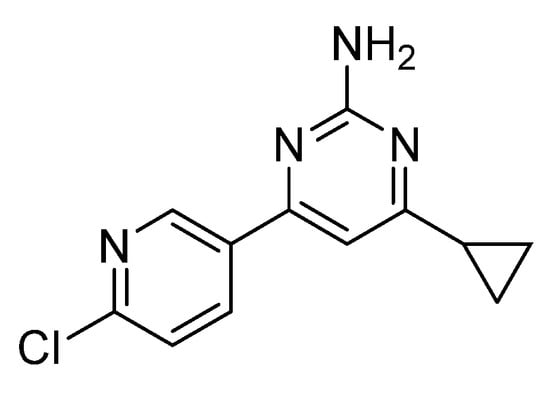
Figure 1
Open AccessCommunication
Synthesis of (Z)-6-Heneicosen-11-one, a Possible Pheromone Component of the Hickory Tussock Moth, Lophocampa caryae
by
Peter Mayo and Sumudu Deepa Abeysekera
Molbank 2026, 2026(3), M2171; https://doi.org/10.3390/M2171 - 6 May 2026
Abstract
The compound (Z)-6-heneicosen-11-one, a possible pheromone component of the hickory tussock moth, Lophocampa caryae, and a known pheromone component of the Douglas fir tussock moth, Orgyia pseudotsugata, was synthesized by a new four-step procedure with a 39% overall yield
[...] Read more.
The compound (Z)-6-heneicosen-11-one, a possible pheromone component of the hickory tussock moth, Lophocampa caryae, and a known pheromone component of the Douglas fir tussock moth, Orgyia pseudotsugata, was synthesized by a new four-step procedure with a 39% overall yield and a six-step procedure incorporating a protecting group with a 28% overall yield. This new synthesis is comparable to other similar syntheses for this molecule in the literature.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures
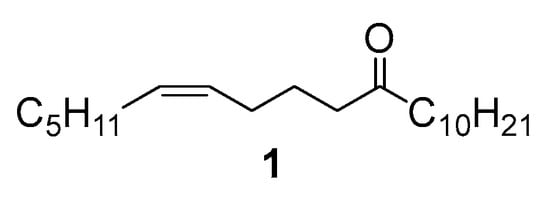
Figure 1
attachment
Supplementary material:
Supplementary File 1 (ZIP, 2220 KB)
Supplementary File 2 (MOL, 4 KB)
Supplementary File 3 (INCHI, 1 KB)
Supplementary File 4 (MOL, 4 KB)
Supplementary File 5 (MOL, 4 KB)
Supplementary File 6 (INCHI, 1 KB)
Supplementary File 7 (MOL, 4 KB)
Supplementary File 8 (MOL, 4 KB)
Supplementary File 9 (INCHI, 1 KB)
Supplementary File 10 (MOL, 4 KB)
Supplementary File 11 (MOL, 4 KB)
Supplementary File 12 (INCHI, 1 KB)
Supplementary File 13 (MOL, 4 KB)
Supplementary File 1 (ZIP, 2220 KB)
Supplementary File 2 (MOL, 4 KB)
Supplementary File 3 (INCHI, 1 KB)
Supplementary File 4 (MOL, 4 KB)
Supplementary File 5 (MOL, 4 KB)
Supplementary File 6 (INCHI, 1 KB)
Supplementary File 7 (MOL, 4 KB)
Supplementary File 8 (MOL, 4 KB)
Supplementary File 9 (INCHI, 1 KB)
Supplementary File 10 (MOL, 4 KB)
Supplementary File 11 (MOL, 4 KB)
Supplementary File 12 (INCHI, 1 KB)
Supplementary File 13 (MOL, 4 KB)
Open AccessCommunication
New Pyridinium Salt Bioconjugates of Cholesterol and Methylpyridine Derivatives: Synthesis and Characterization
by
José María Peña-Martínez, Jesús Alberto Rojas Morales, Luis Ramiro Caso-Vargas, Elizabeth Bautista-Rodríguez, Joel L. Terán and Alan Carrasco-Carballo
Molbank 2026, 2026(3), M2169; https://doi.org/10.3390/M2169 - 2 May 2026
Abstract
The synthesis of three novel, valuable bioconjugates obtained by coupling cholesterol bromoacetate with pyridine derivatives via an SN2 reaction was successfully carried out. Each of the products was fully characterized by magnetic nuclear resonance (1H, 13C, APT, 1H−1
[...] Read more.
The synthesis of three novel, valuable bioconjugates obtained by coupling cholesterol bromoacetate with pyridine derivatives via an SN2 reaction was successfully carried out. Each of the products was fully characterized by magnetic nuclear resonance (1H, 13C, APT, 1H−1H COSY, 1H–13C HMBC, 1H–13C HSQC), infrared spectroscopy (IR), and high-resolution mass spectrometry (HRMS).
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Figure 1
Open AccessCommunication
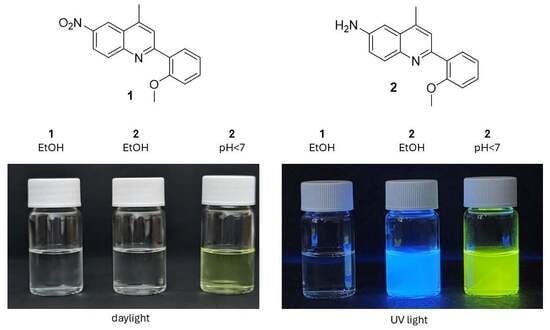
Synthesis of 2-(2-Methoxyphenyl)-4-methyl-6-nitroquinoline and 2-(2-Methoxyphenyl)-4-methylquinolin-6-amine
by
Rumen Lyapchev, Maria Ivanova, Iskra Z. Koleva and Dimitar Shandurkov
Molbank 2026, 2026(3), M2168; https://doi.org/10.3390/M2168 - 30 Apr 2026
Abstract
A new compound, 2-(2-methoxyphenyl)-4-methyl-6-nitroquinoline, was obtained via Suzuki reaction. An unexpected side product, 2-(2-methoxyphenyl)-4-methylquinolin-6-amine, was isolated. The structures of the novel compounds were confirmed by 1H, 13C and 2D-NMR. Their optical properties were also studied.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures
Graphical abstract
Open AccessCommunication
Synthesis of (R)-(+)-3-(1-Hydroxyethylidene)-1-(1-phenylethyl)piperidine-2,4-dione, a Novel Tetramic Acid Analog
by
Alan Aguilar-Aguilar, Ángel Palillero-Cisneros, Félix May-Moreno, Jorge R. Juarez-Posadas, Joel L. Terán and David M. Aparicio
Molbank 2026, 2026(3), M2167; https://doi.org/10.3390/M2167 - 27 Apr 2026
Abstract
Herein, starting from (R)-(+)-α-methylbenzylamine, we report an efficient synthesis and full characterization of a new (R)-3-(1-hydroxyethylidene)-1-(1-phenylethyl)piperidine-2,4-dione, a new tetramic acid analog. The key steps involved a non-classical Corey–Chaykovsky intramolecular cyclization reaction to access the corresponding zwitterion, followed by a
[...] Read more.
Herein, starting from (R)-(+)-α-methylbenzylamine, we report an efficient synthesis and full characterization of a new (R)-3-(1-hydroxyethylidene)-1-(1-phenylethyl)piperidine-2,4-dione, a new tetramic acid analog. The key steps involved a non-classical Corey–Chaykovsky intramolecular cyclization reaction to access the corresponding zwitterion, followed by a sequential desulfurization/reduction and condensation procedure. The titled product was obtained in five steps, and the desired product 7 with an overall 58% yield.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
Synthesis of (S)-4-Benzyl-3-butyl-1-(2-cycloheptylethyl)imidazolidine
by
Matevž Schweiger, Luka Ciber, Nejc Petek, Franc Požgan, Jurij Svete, Bogdan Štefane and Uroš Grošelj
Molbank 2026, 2026(2), M2166; https://doi.org/10.3390/M2166 - 16 Apr 2026
Abstract
LiAlH4 reduction of tert-butyl (S)-butyl(1-((2-cycloheptylethyl)amino)-1-oxo-3-phenylpropan-2-yl)carbamate (1) gave imidazolidine 2, while treatment with lithium diisopropylamide furnished the β-elimination product, cinnamamide 3. Both products were fully characterized. Reductive cyclization of N-alkylated-N-Boc-protected amino acid amides
[...] Read more.
LiAlH4 reduction of tert-butyl (S)-butyl(1-((2-cycloheptylethyl)amino)-1-oxo-3-phenylpropan-2-yl)carbamate (1) gave imidazolidine 2, while treatment with lithium diisopropylamide furnished the β-elimination product, cinnamamide 3. Both products were fully characterized. Reductive cyclization of N-alkylated-N-Boc-protected amino acid amides with LiAlH4 may be a viable synthetic method for trisubstituted chiral imidazolidines.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Figure 1
Open AccessCommunication
A Bulky Aryl–Substituted Acridinium Salt: 10-(3,5-Di-tert-butylphenyl)-9-mesitylacridinium Tetrafluoroborate
by
Yuki Itabashi and Kei Ohkubo
Molbank 2026, 2026(2), M2164; https://doi.org/10.3390/M2164 - 14 Apr 2026
Abstract
9-Mesitylacridinium salts are widely recognized as efficient organic photoredox catalysts owing to their strong excited-state oxidizing power and stability under visible-light irradiation. In this study, a new mesityl acridinium derivative bearing a di-tert-butylphenyl substituent on the nitrogen atom was synthesized. The
[...] Read more.
9-Mesitylacridinium salts are widely recognized as efficient organic photoredox catalysts owing to their strong excited-state oxidizing power and stability under visible-light irradiation. In this study, a new mesityl acridinium derivative bearing a di-tert-butylphenyl substituent on the nitrogen atom was synthesized. The introduction of tert-butyl groups on the N-aryl moiety was primarily aimed at improving solubility and chemical stability of the acridinium salt. Starting from a 9(10H)-acridinone precursor, the target compound was obtained in high overall yield through a concise synthetic sequence. The synthesis consists of a copper-catalyzed C–N coupling reaction to install the aryl substituent on the nitrogen atom, followed by a Grignard reaction and subsequent acid treatment to afford the corresponding acridinium salt. All transformations proceeded smoothly, providing efficient access to the desired novel acridinium derivative. This work presents a practical example of the structural modification of mesitylacridinium derivatives directed toward enhanced solubility and stability, and provides a useful synthetic platform for the preparation of structurally diverse acridinium salts.
Full article
(This article belongs to the Collection Molecules from Catalytic Processes)
►▼
Show Figures

Figure 1
Open AccessCommunication
Synthesis of 2-Aminonicotinonitriles via Photodecarboxylation of Azirine-2-Carboxylic Acids
by
Julia I. Pavlenko, Mikhail S. Novikov and Anastasiya V. Agafonova
Molbank 2026, 2026(2), M2165; https://doi.org/10.3390/M2165 - 14 Apr 2026
Abstract
2-Aminonicotinonitriles represent an important class of heterocycles with diverse biological activities. Herein, we report an unexpected photochemical transformation of azirine-2-carboxylic acids leading to the formation of 2-aminonicotinonitrile derivatives. Optimization of the reaction conditions enabled the synthesis of the target products in moderate yields.
[...] Read more.
2-Aminonicotinonitriles represent an important class of heterocycles with diverse biological activities. Herein, we report an unexpected photochemical transformation of azirine-2-carboxylic acids leading to the formation of 2-aminonicotinonitrile derivatives. Optimization of the reaction conditions enabled the synthesis of the target products in moderate yields. The structure of the obtained product was confirmed by NMR spectroscopy, HRMS, and single-crystal X-ray diffraction analysis.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
Synthesis of 2-Methylcamalexin
by
Yordan Stremski, Maria Bachvarova, Stela Statkova-Abeghe and Plamen Angelov
Molbank 2026, 2026(2), M2163; https://doi.org/10.3390/M2163 - 13 Apr 2026
Abstract
2-methylcamalexin, a novel derivative of the phytoalexin Camalexin, was synthesized for the first time, using a convenient two-step approach. The approach realizes coupling of two aromatic heterocyclic moieties (2-methylindole and thiazole) by sequential α-amidoalkylation/oxidative re-aromatization. The target product was obtained in a cost-effective
[...] Read more.
2-methylcamalexin, a novel derivative of the phytoalexin Camalexin, was synthesized for the first time, using a convenient two-step approach. The approach realizes coupling of two aromatic heterocyclic moieties (2-methylindole and thiazole) by sequential α-amidoalkylation/oxidative re-aromatization. The target product was obtained in a cost-effective manner, with 88% yield over two steps. The structure of the synthesized product was unequivocally determined on the basis of NMR, HRMS and FTIR spectral measurments.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures
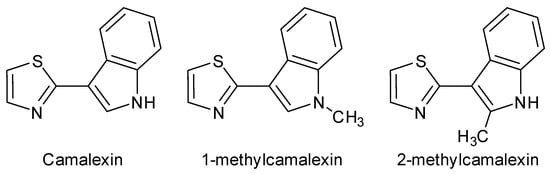
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Molbank
Carbonylation Chemistry in the Synthesis of High Value Added Compounds
Guest Editors: Bartolo Gabriele, Raffaella MancusoDeadline: 30 November 2026
Topical Collections
Topical Collection in
Molbank
Molecules from Catalytic Processes
Collection Editor: Nicola Della Ca’