-
 Chronic Histamine Exposure Promotes Melanogenesis via ORAI1-STIM1-Mediated Calcium Signaling Remodeling
Chronic Histamine Exposure Promotes Melanogenesis via ORAI1-STIM1-Mediated Calcium Signaling Remodeling -
 Unraveling the Enigma of Melanoma Brain Metastasis: New Molecular Insights and Therapeutic Directions
Unraveling the Enigma of Melanoma Brain Metastasis: New Molecular Insights and Therapeutic Directions -
 GLP-1 Receptor Agonists at the Crossroads of Circadian Biology, Sleep, and Metabolic Disease
GLP-1 Receptor Agonists at the Crossroads of Circadian Biology, Sleep, and Metabolic Disease -
 Apple-Derived Vesicles Orchestrate Bone Regeneration: In Vitro Proof of Concept
Apple-Derived Vesicles Orchestrate Bone Regeneration: In Vitro Proof of Concept -
 Molecular and Mechanistic Divergence of Seizures in Pediatric Acute Lymphoblastic Leukemia: CNS Infiltration Versus Chemotherapy-Induced Neurotoxicity
Molecular and Mechanistic Divergence of Seizures in Pediatric Acute Lymphoblastic Leukemia: CNS Infiltration Versus Chemotherapy-Induced Neurotoxicity
Journal Description
International Journal of Molecular Sciences
International Journal of Molecular Sciences
is an international, peer-reviewed, open access journal providing an advanced forum for biochemistry, molecular and cell biology, molecular biophysics, molecular medicine, and all aspects of molecular research in chemistry, and published semimonthly online by MDPI. The Epigenetics Society, European Chitin Society (EUCHIS), Spanish Society for Cell Biology (SEBC) and others are affiliated with IJMS and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, MEDLINE, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Biochemistry and Molecular Biology) / CiteScore - Q1 (Organic Chemistry)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.8 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our editors and authors say about IJMS.
- Companion journals for IJMS include: Biophysica, Stresses, Lymphatics, SynBio and Inflammation Journal.
Impact Factor:
4.9 (2024);
5-Year Impact Factor:
5.7 (2024)
Latest Articles
Phase-Dependent MoS2 Nanosheets-Embedded Urinary Catheter for Advanced Photothermal Sterilization
Int. J. Mol. Sci. 2026, 27(11), 4806; https://doi.org/10.3390/ijms27114806 - 26 May 2026
Abstract
The high prevalence of catheter-associated urinary tract infections (CAUTIs) has become a significant concern in the healthcare system, prompting the development of antibacterial urinary catheters to effectively prevent these infections in clinical settings. In this work, metallic phase and semiconducting phase molybdenum disulfide
[...] Read more.
The high prevalence of catheter-associated urinary tract infections (CAUTIs) has become a significant concern in the healthcare system, prompting the development of antibacterial urinary catheters to effectively prevent these infections in clinical settings. In this work, metallic phase and semiconducting phase molybdenum disulfide (MoS2) embedded polydimethylsiloxane (PDMS) were developed as antibacterial urinary catheters with photothermal sterilization. The metallic phase MoS2 (1T-MoS2) was synthesized using a facile hydrothermal method, and an annealing process transformed it into the semiconducting phase (2H-MoS2). The optical and structural characterizations confirmed the successful preparations of 1T-MoS2 nanosheets (1T-MoS2 NSs) and 2H-MoS2 NSs. The increase in the contents of 1T-MoS2 NSs and 2H-MoS2 NSs in PDMS resulted in enhanced photothermal conversion, a slight decrease in the water contact angle, and no significant changes in the mechanical properties of the samples. The bacterial growth curves demonstrated the remarkable ability of phase-dependent 1T- and 2H-MoS2 NSs-embedded PDMS urinary catheters to inhibit the growth of E. coli and S. aureus with near-infrared (NIR) laser irradiation. In the agar plate test, exposing PDMS with 0.3% 1T-MoS2 or 2H-MoS2 to NIR for 10 min demonstrated excellent antibacterial effects, completely eradicating E. coli and eliminating over 99.9% of S. aureus. The SEM image results highlighted the significant photothermal antibacterial effect of 1T-MoS2 PDMS and 2H-MoS2 PDMS urinary catheters, effectively damaging and eradicating both E. coli and S. aureus. The 1T-MoS2 PDMS and 2H-MoS2 PDMS urinary catheters, with excellent photothermal effects, good hydrophobicity, and superior mechanical properties, demonstrated their potential as photothermal antibacterial catheters for clinical applications.
Full article
(This article belongs to the Section Molecular Nanoscience)
Open AccessArticle
RUNX1 Alterations in Pediatric Myeloid Malignancies: Divergent Germline and Somatic Trajectories
by
Ana Maria Bicǎ, Andra Daniela Marcu, Cristina Georgiana Jercan, Letiția Elena Radu, Irina Avramescu, Cerasela Jardan, Dumitru Jardan, Onda Tabita Cǎlugǎru, Cristina Mambet and Anca Colițǎ
Int. J. Mol. Sci. 2026, 27(11), 4805; https://doi.org/10.3390/ijms27114805 - 26 May 2026
Abstract
RUNX1 alterations contribute to pediatric myeloid malignancies through both germline predisposition syndromes and somatic leukemogenic events, but their clinical and biological significance in children remains incompletely defined. This retrospective single-center study evaluated six pediatric patients with myelodysplastic syndromes or acute leukemias harboring RUNX1
[...] Read more.
RUNX1 alterations contribute to pediatric myeloid malignancies through both germline predisposition syndromes and somatic leukemogenic events, but their clinical and biological significance in children remains incompletely defined. This retrospective single-center study evaluated six pediatric patients with myelodysplastic syndromes or acute leukemias harboring RUNX1 variants, integrating clinical, cytogenetic, and targeted next-generation sequencing data, with germline status confirmed using non-hematopoietic tissues. Three patients carried germline RUNX1 variants, characterized by antecedent cytopenias, dysplastic features, and increased treatment-related toxicity, including severe infections, persistent cytopenias, and transplant-related mortality. In contrast, somatic RUNX1 alterations were associated with overt high-risk disease, frequently accompanied by complex cytogenetics or monosomy 7, and demonstrated heterogeneous outcomes ranging from sustained remission to post-transplant relapse. Mixed-phenotype acute leukemia was observed in both groups. These findings support a model of RUNX1-driven leukemogenesis, in which germline and somatic alterations represent distinct yet interconnected trajectories, while highlighting the importance of distinguishing variant origin for risk stratification, donor selection, and therapeutic decision-making in pediatric myeloid malignancies. Given the small cohort size, the findings remain descriptive and require validation in larger prospective studies.
Full article
(This article belongs to the Special Issue Molecular Research Advances in Common and Rare Pediatric Diseases)
►▼
Show Figures
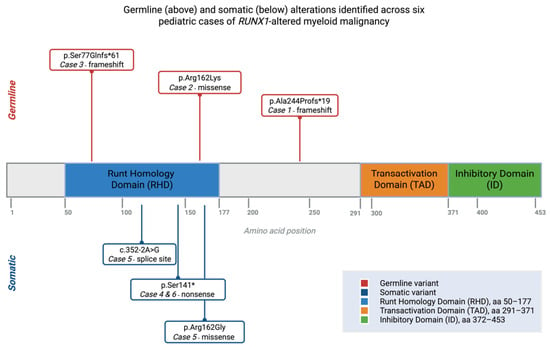
Figure 1
Open AccessReview
Medicinal Plants: A Promising Therapeutic Approach for Addressing Antimicrobial Resistance
by
Huanxin Zhou, Jinkang Du, Meiyan Jia, Yine Li and Kaiyun Zheng
Int. J. Mol. Sci. 2026, 27(11), 4804; https://doi.org/10.3390/ijms27114804 - 26 May 2026
Abstract
Antimicrobial resistance (AMR) is a critical global public health crisis that is being exacerbated by widespread misuse of antibiotics and rapid bacterial adaptation. The progressive decrease in antibiotic efficacy is also compounded by a stagnating drug-discovery pipeline and underscores the urgent need for
[...] Read more.
Antimicrobial resistance (AMR) is a critical global public health crisis that is being exacerbated by widespread misuse of antibiotics and rapid bacterial adaptation. The progressive decrease in antibiotic efficacy is also compounded by a stagnating drug-discovery pipeline and underscores the urgent need for innovative and sustainable antimicrobial strategies. This review systematically delineates the core molecular mechanisms driving bacterial resistance, including enzymatic drug inactivation, target modification, reduced membrane permeability, and multidrug efflux pump overexpression. Furthermore, the potential of (flavonoids, alkaloids, and phenolics) as structurally diverse plant-derived compounds with multi-target activity is comprehensively assessed. The features of multi-target activity make them promising dual-function agents that may be capable of both direct antimicrobial action and resistance modulation. These natural products have distinct mechanisms from conventional antibiotics, low propensity for resistance, and versatile bioactivity as biofilm disruptors, enzyme inhibitors, and efflux pump blockers. Numerous phytochemicals exhibit potent synergistic effects with available antibiotics by effectively resensitizing resistant pathogens and extending the clinical utility of current antimicrobials. By integrating mechanistic understanding with translational potential, this review discusses phytochemicals as a sustainable resource for developing next-generation antimicrobial strategies as a complementary approach to revitalize therapeutic pipelines and combat multidrug-resistant infections.
Full article
(This article belongs to the Special Issue Advances in Understanding the Molecular Basis of Antimicrobial Properties in Natural Products, Functional Additives, and Food-Derived Compounds)
►▼
Show Figures
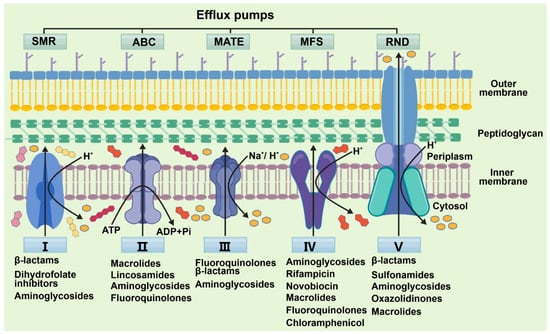
Figure 1
Open AccessArticle
Physics-Based Modeling of Sparse Single-Cell Hi-C Uncovers Structural and Epigenetic Variability
by
Francesca Vercellone, Sumanta Kundu, Andrea Esposito, Andrea M. Chiariello, Mattia Conte, Alex Abraham, Andrea Fontana, Florinda Di Pierno, Sougata Guha, Ciro Di Carluccio, Matteo Olimpo, Mario Nicodemi, Francesco Paolo Casale and Simona Bianco
Int. J. Mol. Sci. 2026, 27(11), 4803; https://doi.org/10.3390/ijms27114803 - 26 May 2026
Abstract
Chromatin conformation capture technologies have revealed the complex 3D organization of the genome and its key regulatory role. Single-cell Hi-C (scHi-C) maps this architecture at single-cell level, but its sparse nature makes data interpretation challenging, and tools for their analysis remain limited. Here,
[...] Read more.
Chromatin conformation capture technologies have revealed the complex 3D organization of the genome and its key regulatory role. Single-cell Hi-C (scHi-C) maps this architecture at single-cell level, but its sparse nature makes data interpretation challenging, and tools for their analysis remain limited. Here, we present a physics-based framework that combines polymer modeling with computational methods to reconstruct full 3D genome structures from sparse scHi-C data. Using both artificial and experimental data, we show that our approach imputes missing contacts and recovers accurate structures validated against independent Hi-C and established polymer models. Applied to scHi-C from a 15 Mb region of human HeLa-S3 cells as a case study, the method uncovers distinct structural classes defined by the spatial distribution of chromatin binding domains. The reconstructed models enable robust downstream analyses, including the identification of single-cell topologically associated domains (TADs), which appear highly variable across cells yet tend to accumulate around those observed in bulk. Importantly, the inferred 3D polymer models capture diverse epigenetic signatures, with active chromatin domains exhibiting greater structural variability than repressive ones across single cells. Overall, our study provides a mechanistic and interpretable framework to analyze sparse scHi-C data, highlighting how polymer physics can be leveraged to uncover genome architecture and its functional variability at single-cell resolution.
Full article
(This article belongs to the Special Issue Molecular Modelling in Material Science)
►▼
Show Figures
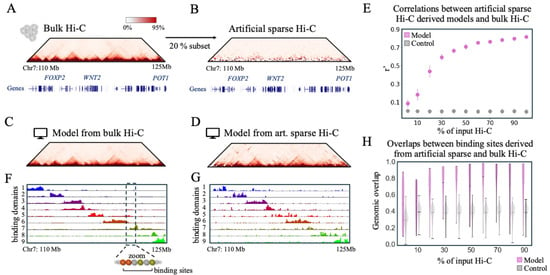
Figure 1
Open AccessReview
The Role of the Rhizosphere, Endophytes, and the Influence of Plant-Growth-Promoting Bacteria: Take the Cannabis Microbiome as an Example
by
Piotr Stanisław Wiszpolski and Mariusz Jerzy Stolarski
Int. J. Mol. Sci. 2026, 27(11), 4802; https://doi.org/10.3390/ijms27114802 - 26 May 2026
Abstract
Cannabis sativa L. is a multipurpose crop of increasing agricultural and medical relevance, whose productivity and phytocannabinoid profile are influenced not only by genotype and environmental factors but also by the composition of its microbiota. This review synthesizes current knowledge (2020–2026) on the
[...] Read more.
Cannabis sativa L. is a multipurpose crop of increasing agricultural and medical relevance, whose productivity and phytocannabinoid profile are influenced not only by genotype and environmental factors but also by the composition of its microbiota. This review synthesizes current knowledge (2020–2026) on the rhizosphere and endophytic microbiota of hemp, with particular emphasis on plant growth-promoting bacteria (PGPB) and their mechanisms of action. Molecular studies indicate that hemp-associated bacterial communities are dominated by Proteobacteria, Actinobacteriota, Firmicutes and Bacteroidota, with genotype-, tissue- and developmental-stage-dependent variation. PGPB influence plant performance through direct mechanisms, including biological nitrogen fixation, phosphate solubilization, siderophore production and phytohormone synthesis (indole-3-acetic acid (IAA), gibberellins, cytokinins, and 1-aminocyclopropane-1-carboxylate (ACC) deaminase), as well as indirect mechanisms such as antibiosis, enzyme-mediated pathogen inhibition and induction of systemic tolerance to abiotic stress. Experimental studies demonstrate that inoculation with selected strains or consortia can enhance biomass accumulation, improve germination and root architecture, increase resistance to Fusarium oxysporum and modulate cannabinoid and terpene profiles. Importantly, plant responses are cultivar-specific, highlighting the need for genotype-tailored microbial formulations.
Full article
(This article belongs to the Section Molecular Plant Sciences)
Open AccessReview
Metabolic Memory-Mediated Epigenetic Regulation of EMT in Diabetic Kidney Disease: Mechanisms and Therapeutic Implications
by
Xinning Ran, Yidan Xu, Ruonan Liang, Yuqi Duan, Wanying Jia, Yuhong Bian, Chenduo Li and Mingxing Zhang
Int. J. Mol. Sci. 2026, 27(11), 4801; https://doi.org/10.3390/ijms27114801 - 26 May 2026
Abstract
Diabetic kidney disease (DKD) is a leading cause of end-stage renal disease, with renal fibrosis as its core pathological hallmark. A central driver of this fibrosis is epithelial–mesenchymal transition (EMT), during which renal tubular epithelial cells transform into matrix-producing myofibroblasts. Endothelial–mesenchymal transition (EndMT)
[...] Read more.
Diabetic kidney disease (DKD) is a leading cause of end-stage renal disease, with renal fibrosis as its core pathological hallmark. A central driver of this fibrosis is epithelial–mesenchymal transition (EMT), during which renal tubular epithelial cells transform into matrix-producing myofibroblasts. Endothelial–mesenchymal transition (EndMT) has also emerged as a critical contributor, and together with EMT, accounts for the progressive accumulation of myofibroblasts and extracellular matrix. A major clinical challenge in halting DKD progression is “metabolic memory”, a phenomenon whereby renal injury persists and EMT/EndMT remain activated even after glycemic control is achieved. The molecular basis underlying this sustained activation remains incompletely understood. Emerging evidence indicates that metabolic memory is largely mediated by epigenetic mechanisms, including histone modifications, DNA methylation, and non-coding RNA dysregulation. These stable epigenetic imprints maintain the persistent activation of key pro-fibrotic signaling pathways, especially TGF-β, thereby continuously driving EMT, EndMT, and excessive extracellular matrix deposition. Although targeting epigenetic regulators has shown promising anti-fibrotic effects, a systematic review that integrates how metabolic memory orchestrates both EMT and EndMT through a multi-layered epigenetic network remains lacking. This review comprehensively summarizes the epigenetic mechanisms by which metabolic memory sustains EMT and EndMT in DKD, highlights key therapeutic targets, and discusses their translational and clinical implications.
Full article
(This article belongs to the Section Molecular Endocrinology and Metabolism)
►▼
Show Figures
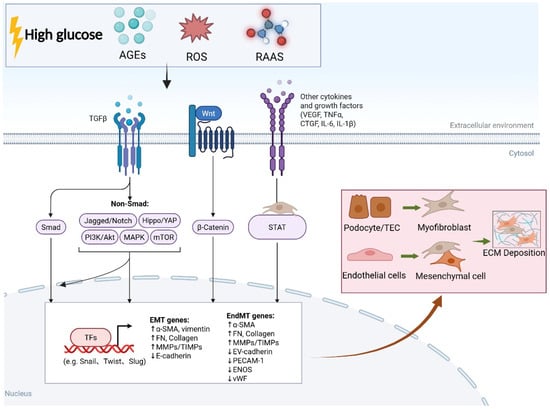
Figure 1
Open AccessReview
The Role of Matrix Metalloproteinases in Orthodontics, Dental Trauma, Restorative Dentistry, and Endodontics: Molecular Mechanisms and Clinical Implications
by
Renata Ławicka, Kinga Królikowska, Katarzyna Błaszczak, Zuzanna Borawska, Monika Zbucka-Krętowska, Sławomir Ławicki and Magdalena Nowosielska
Int. J. Mol. Sci. 2026, 27(11), 4800; https://doi.org/10.3390/ijms27114800 - 26 May 2026
Abstract
Matrix metalloproteinases (MMPs) are zinc-dependent proteolytic enzymes involved in extracellular matrix remodelling in oral and dental tissues, including the periodontal ligament, alveolar bone, dentin, dental pulp, and periapical tissues. This narrative review summarises selected evidence on the role of MMPs and tissue inhibitors
[...] Read more.
Matrix metalloproteinases (MMPs) are zinc-dependent proteolytic enzymes involved in extracellular matrix remodelling in oral and dental tissues, including the periodontal ligament, alveolar bone, dentin, dental pulp, and periapical tissues. This narrative review summarises selected evidence on the role of MMPs and tissue inhibitors of metalloproteinases (TIMPs) in orthodontic tooth movement, dental trauma and root resorption, restorative adhesive dentistry, and pulp/periapical disease. Particular attention is given to signalling pathways that regulate MMP/TIMP activity, including nuclear factor kappa B (NF-κB), mitogen-activated protein kinase (MAPK), Wnt/β-catenin, and transforming growth factor beta (TGF-β)/Smad-related mechanisms. The review also discusses the biomarker potential and translational status of MMP-targeted strategies. Across clinical contexts, MMP activity contributes to both matrix degradation and tissue repair, and its biological effect depends on local stimuli, TIMP-mediated regulation, pathway crosstalk, and the stage of disease or treatment.
Full article
(This article belongs to the Section Molecular Biology)
Open AccessReview
Plant-Derived Terpenes as Emerging Therapeutics Against Schistosomiasis
by
Célia Faustino, Lídia Pinheiro and Noélia Duarte
Int. J. Mol. Sci. 2026, 27(11), 4799; https://doi.org/10.3390/ijms27114799 - 26 May 2026
Abstract
Schistosomiasis remains one of the most significant neglected tropical diseases (NTDs) worldwide, sustained by the complex biology of Schistosoma species and the host’s immunopathological responses to tissue-trapped eggs. Despite decades of reliance on praziquantel (PZQ) as the sole chemotherapeutic option, major limitations persist,
[...] Read more.
Schistosomiasis remains one of the most significant neglected tropical diseases (NTDs) worldwide, sustained by the complex biology of Schistosoma species and the host’s immunopathological responses to tissue-trapped eggs. Despite decades of reliance on praziquantel (PZQ) as the sole chemotherapeutic option, major limitations persist, including its lack of activity against juvenile worms, incomplete protection against reinfection, and concerns regarding emerging tolerance. These challenges, together with persistent hotspots of transmission and uneven global progress toward disease elimination, underscore the urgent need for alternative or complementary therapies. Plant-derived terpenes have emerged as promising antischistosomal candidates due to their structural diversity, broad-spectrum bioactivity, and favourable safety profiles. Evidence from in vitro and in vivo studies demonstrates that monoterpenes, sesquiterpenes, diterpenes, triterpenes, and triterpenoid saponins exert multimodal effects on Schistosoma, including tegumental disruption, interference with metabolic and redox pathways, inhibition of oviposition, and modulation of host immune and fibrotic responses. Advances in mechanistic studies, supported by omics and computational approaches, further highlight their potential as leads for drug development. Additionally, nano-enabled delivery systems offer strategies to overcome pharmacokinetic limitations and enhance therapeutic performance. This review integrates current knowledge on schistosome biology, treatment challenges, and the growing evidence supporting terpenoids as viable components of a diversified antischistosomal therapeutic arsenal.
Full article
(This article belongs to the Special Issue Bioactive Food Ingredients and Precision Nutrition: From Molecular Innovation to Clinical Relevance)
Open AccessArticle
PSMB4/MHC-I Signaling in the Cerebrospinal Fluid-Contacting Nucleus Mediates Neuroinflammatory Depression in Mice
by
Yi-Jun Zhang, Yu-Wei Ma, Bin Gui, Xin-Ling Wang, Jin Qian, Yu Peng and Li-Cai Zhang
Int. J. Mol. Sci. 2026, 27(11), 4798; https://doi.org/10.3390/ijms27114798 - 26 May 2026
Abstract
Neuroinflammation is increasingly implicated in depression pathogenesis, yet the underlying mechanisms are still unclear. This study explores whether PSMB4/MHC-I signaling in the cerebrospinal fluid (CSF)-contacting nucleus mediates neuroinflammatory depression. A persistent neuroinflammation-associated depression model was established in mice by repeated intracerebroventricular lipopolysaccharide (LPS)
[...] Read more.
Neuroinflammation is increasingly implicated in depression pathogenesis, yet the underlying mechanisms are still unclear. This study explores whether PSMB4/MHC-I signaling in the cerebrospinal fluid (CSF)-contacting nucleus mediates neuroinflammatory depression. A persistent neuroinflammation-associated depression model was established in mice by repeated intracerebroventricular lipopolysaccharide (LPS) administration. Depressive-like behaviors were evaluated using established assays. Neuroinflammatory responses and target protein expression were assessed by immunofluorescence, Western blotting, RT-qPCR, and laser capture microdissection. Neuronal activity was mapped by c-Fos staining and manipulated using chemogenetics, alongside pharmacological and genetic interventions. Repeated LPS administration induced significant depressive-like behaviors and obvious neuroinflammation in the CSF-contacting nucleus. Under these conditions, neuronal activity in this nucleus was selectively enhanced. Crucially, chemogenetic activation of these neurons alleviated depressive phenotypes, whereas their inhibition induced depression. Molecularly, LPS significantly upregulated PSMB4 and MHC-I expression. Pharmacological suppression of upstream neuroinflammation reversed this PSMB4 upregulation, and targeted PSMB4 knockdown reduced MHC-I expression, ultimately ameliorating depressive-like behaviors. These findings identify the CSF-contacting nucleus as a critical node in neuroinflammation-induced depression and reveal a novel PSMB4/MHC-I signaling axis linking central inflammatory responses to behavioral deficits.
Full article
(This article belongs to the Special Issue Molecular Crosstalk in Stress-Induced Psychopathology)
Open AccessArticle
A Metabolic-Related Gene Signature for Predicting Biochemical Recurrence After Radical Prostatectomy: An Integrative Analysis and Targeted Therapeutic Validation
by
Wankun Wang, Xiujuan Hong, Xiaoqi Wang, Ganpei Jiao, Hongjie Cai, Junxiang Zhao, Zhibing Wu and Jun Chen
Int. J. Mol. Sci. 2026, 27(11), 4797; https://doi.org/10.3390/ijms27114797 - 26 May 2026
Abstract
Biochemical recurrence (BCR) after radical prostatectomy (RP) remains a major clinical challenge. Although metabolic reprogramming drives prostate cancer (PCa) progression, its predictive value for BCR and its interplay with the tumor immune microenvironment (TIME) remain incompletely understood. By integrating weighted gene co-expression network
[...] Read more.
Biochemical recurrence (BCR) after radical prostatectomy (RP) remains a major clinical challenge. Although metabolic reprogramming drives prostate cancer (PCa) progression, its predictive value for BCR and its interplay with the tumor immune microenvironment (TIME) remain incompletely understood. By integrating weighted gene co-expression network analysis (WGCNA) with machine learning, we identified four metabolic-related hub genes (GDPD1, PLA2G7, PTGDS, and SRD5A2) and developed an XGBoost-Cox model that accurately stratified BCR risk (training 5-year AUC: 0.858; validation 5-year AUC: 0.745). SHAP analysis enhanced the model’s interpretability, while immunohistochemistry (IHC) validated differential protein expression of these targets across 32 clinical specimens. Furthermore, immune profiling demonstrated that these genes are closely linked to M2 macrophage-mediated immunosuppression and altered T-cell infiltration. To translate these biomarkers into therapeutic targets, we employed in silico screening, molecular docking, and molecular dynamics simulations, identifying (-)-epigallocatechin gallate (EGCG) as a promising multi-target candidate. Subsequent in vitro assays confirmed that EGCG binds stably to GDPD1, PTGDS, and SRD5A2, effectively suppressing malignant PCa phenotypes and prostate-specific antigen (PSA) secretion. In summary, we established a robust and interpretable model for predicting BCR after RP, and our in vitro validation suggests that EGCG holds promise as a therapeutic agent to delay PCa progression.
Full article
(This article belongs to the Special Issue Identification of Predictive Molecular Markers for Cancer Progression, Response to Therapy, and Disease Outcome)
Open AccessReview
The Therapeutic Potential of Mesenchymal Stem Cells in Post-Stroke Depression
by
Manru Fan, Que Deng, Zhimin Li, Guibin Wang and Ming Lu
Int. J. Mol. Sci. 2026, 27(11), 4796; https://doi.org/10.3390/ijms27114796 - 26 May 2026
Abstract
Post-stroke depression (PSD) is the most prevalent neuropsychological disorder among stroke survivors, affecting over 30% of patients. It significantly impairs patients’ quality of life and imposes a substantial burden on individuals, families, and society. Currently, the primary treatment for PSD focuses on conventional
[...] Read more.
Post-stroke depression (PSD) is the most prevalent neuropsychological disorder among stroke survivors, affecting over 30% of patients. It significantly impairs patients’ quality of life and imposes a substantial burden on individuals, families, and society. Currently, the primary treatment for PSD focuses on conventional antidepressant therapies, with a lack of innovative approaches. Therefore, there is an urgent need to develop novel targeted therapies for PSD. This review synthesizes PSD pathogenesis as a multi-system network disorder involving monoamine deficits, neuroinflammation, HPA axis dysfunction, and neurotrophic imbalance. Within this framework, mesenchymal stem cells (MSCs) transplantation, as an emerging therapeutic strategy, may exert beneficial effects through anti-inflammatory, neuroprotective mechanisms, and the provision of neurotrophic factors. This review provides a preclinical framework that highlights the potential of MSC-based strategies, while emphasizing the need for further validation in PSD-specific models before clinical translation.
Full article
(This article belongs to the Special Issue Role of Mesenchymal Stem/Stromal Cells (MSCs) in Human Diseases and Their Treatment)
Open AccessArticle
Genotoxicity Integration into Bioprocess Optimization Reveals Progressive DNA Damage During Bioreactor Expansion of Adipose-Derived Stem Cells
by
Vinícius Augusto Simão, Rafaela Choi Peng So, Jaci Leme, Rafael Guilen de Oliveira, Gabriel Adan Araújo Leite, Luiz Gustavo de Almeida Chuffa, Aldo Tonso and João Tadeu Ribeiro-Paes
Int. J. Mol. Sci. 2026, 27(11), 4795; https://doi.org/10.3390/ijms27114795 - 26 May 2026
Abstract
Mesenchymal stromal cells derived from adipose tissue (ASCs) are widely used in regenerative medicine, requiring scalable expansion strategies that preserve both cellular function and biological quality. However, current bioprocess optimization approaches are primarily guided by proliferation and phenotypic stability, often overlooking genomic integrity
[...] Read more.
Mesenchymal stromal cells derived from adipose tissue (ASCs) are widely used in regenerative medicine, requiring scalable expansion strategies that preserve both cellular function and biological quality. However, current bioprocess optimization approaches are primarily guided by proliferation and phenotypic stability, often overlooking genomic integrity as a critical attribute. In this study, we developed a stirred-tank bioreactor system for ASC expansion on microcarriers and applied a genotoxicity-informed optimization strategy by integrating growth kinetics, metabolic profiling, and DNA damage assessment across multiple operational conditions (B1–B5), including variations in dissolved oxygen, agitation, inoculum density, and medium renewal. Optimized culture conditions (B5) enabled high cell productivity within a reduced cultivation period (9 days), while maintaining high viability (>90%), mesenchymal immunophenotype, and differentiation capacity. Distinct metabolic profiles were associated with enhanced proliferation, with increased glycolytic activity observed under optimized conditions. Despite these favorable outcomes, genotoxic analyses revealed a progressive, time-dependent accumulation of DNA damage and increased micronucleus frequency during expansion. Notably, these alterations did not impair cell proliferation, phenotype, or differentiation potential, indicating that conventional optimization metrics may not fully capture underlying genomic changes. Collectively, our findings demonstrate that bioprocess optimization based solely on classical performance parameters may overlook relevant biological alterations. By incorporating genotoxic endpoints into the evaluation framework, this study provides a refined approach for assessing large-scale stem cell expansion and contributes to improving the robustness and reliability of biomanufacturing strategies for therapeutic applications.
Full article
(This article belongs to the Special Issue Adipose Tissue and Adipose Tissue-Derived Stem Cells in Multidisciplinary Research)
Open AccessArticle
Biomineral Complex with Probiotic and Detoxifying Properties for Recovery After Radiotherapy
by
Olga Ilinskaya, Konstantin Vagin, William Kurdy, Galina Yakovleva, Nazira Karamova, Pavel Zelenikhin, Alexey Kolpakov and Yuri Zuev
Int. J. Mol. Sci. 2026, 27(11), 4794; https://doi.org/10.3390/ijms27114794 - 26 May 2026
Abstract
Radiotherapy is a highly effective, safe cancer treatment, and about half of all cancer treatments involve lifesaving radiotherapy. Despite huge advances in technology that have made it safer and more effective, it is still not without side effects. They differ from patient to
[...] Read more.
Radiotherapy is a highly effective, safe cancer treatment, and about half of all cancer treatments involve lifesaving radiotherapy. Despite huge advances in technology that have made it safer and more effective, it is still not without side effects. They differ from patient to patient and can include fatigue, nausea, skin reactions, and hair loss, but dysbiosis is the most common complication associated with radiotherapy. Probiotics aimed at restoring the microbiome have found widespread use, but the problem of their rapid inactivation in the gastrointestinal tract has not yet been solved. Our study aims to confirm the effectiveness of a novel biomineral complex, based on a powdered clinoptilolite containing a rock loaded with lactobacilli for restoring the intestinal microbiome of mice exposed to radiation. Based on the 16S rRNA gene analysis, alpha-diversity and dynamics of changes in the fecal metagenome, as well as the functional potential of mice exposed to radiation, were studied, and the prospects of administering the biomineral complex to achieve positive effects were assessed. NMR analysis of the mineral carrier was carried out, and its safety was confirmed. Moreover, per os administration of the complex following irradiation led to a reduction in the level of chromosomal aberrations induced by irradiation. Thus, the biomineral complex has a microbiome-restoring effect and reduces radiation-induced clastogenesis.
Full article
(This article belongs to the Special Issue Advanced Biomaterials for Tissue Regeneration)
Open AccessArticle
Evidence for a Potential Context-Dependent Dual Role of Eb Peptide of IGF-1Eb in Cancer Cell Survival and Adaptation to UV Stress
by
Amalia Kotsifaki, Georgia Kalouda, Efthymios Karalexis, Sylvia Raftopoulou, Nektarios Alevizopoulos and Athanasios Armakolas
Int. J. Mol. Sci. 2026, 27(11), 4793; https://doi.org/10.3390/ijms27114793 - 26 May 2026
Abstract
The IGF-1Eb isoform has been proposed as a stress-responsive variant of IGF-1, yet its significance in cancer remains unclear. This investigation aims to clarify its role across breast, prostate and liver cancer cell lines and determine whether its loss or supplementation is associated
[...] Read more.
The IGF-1Eb isoform has been proposed as a stress-responsive variant of IGF-1, yet its significance in cancer remains unclear. This investigation aims to clarify its role across breast, prostate and liver cancer cell lines and determine whether its loss or supplementation is associated with alterations in cellular behavior and stress adaptation. Eb expression was modulated through targeted silencing and exogenous peptide addition. Cellular responses were evaluated under normal conditions and UV stress using proliferation, viability and rescue experiments, wound healing, immunofluorescence for Eb-knockdown confirmation, qRT-PCR, Annexin V/PI apoptosis, and PI cell-cycle evaluation. Across six cancer cell lines, Eb peptide given before UV stress was associated with partial protective effects, whereas post-UV treatment was associated with improved recovery and partial restoration of proliferative capacity. The rescue effect differed by cell type, with prostate and breast cells showing the strongest responses and liver-derived lines displaying more modest improvements. Eb knockdown revealed clear cell-type-specific dependencies. PC3 cells showed markedly reduced proliferation (p < 0.01) and sharply decreased post-UV viability (p < 0.0001). HepG2 cells maintained higher growth without UV but displayed reduced recovery following UV exposure, whereas MDA-MB-231 exhibited elevated apoptosis (p < 0.05) with limited additional UV sensitivity. Eb peptide may exert a dual, timing-dependent role, supporting protection before UV damage and facilitating recovery-associated responses afterward, with its impact differing across cell lines.
Full article
(This article belongs to the Special Issue 25th Anniversary of IJMS: Updates and Advances in Molecular Pathology, Diagnostics, and Therapeutics)
Open AccessArticle
Genome-Wide and Locus-Level Analyses Reveal Modest, Heterogeneous Genetic Sharing Between Alzheimer’s Disease and Myasthenia Gravis
by
Emmanuel O. Adewuyi, Asa Auta, Chinedu I. Ossai, Chidozie C. Anyaegbu, Thi Thu Huong Nguyen, Md Rezanur Rahman, Blossom C. M. Stephan, Gizachew A. Tessema, Dale R. Nyholt and Gavin Pereira
Int. J. Mol. Sci. 2026, 27(11), 4792; https://doi.org/10.3390/ijms27114792 - 26 May 2026
Abstract
Alzheimer’s disease (AD) is a neurodegenerative disorder, whereas myasthenia gravis (MG) is an autoimmune neuromuscular disease. Despite their distinct clinical manifestations, both disorders involve immune dysregulation and cholinergic dysfunction, and epidemiological evidence for an association remains inconclusive. Here, we investigated the genetic architecture
[...] Read more.
Alzheimer’s disease (AD) is a neurodegenerative disorder, whereas myasthenia gravis (MG) is an autoimmune neuromuscular disease. Despite their distinct clinical manifestations, both disorders involve immune dysregulation and cholinergic dysfunction, and epidemiological evidence for an association remains inconclusive. Here, we investigated the genetic architecture underlying the AD–MG relationship using large-scale European-ancestry genome-wide association study (GWAS) data, including early- and late-onset MG, within a multi-resolution analytical framework. Genome-wide analyses indicated modest polygenic overlap between AD and MG, supported by nominally significant and directionally consistent correlations across datasets, SNPeffect concordance in the primary GWAS, and robust gene-level overlap. Evidence for genome-wide correlation was weaker and non-significant across AD-MG subtypes. Local genetic correlation analyses revealed that shared AD-MG signals were largely locus-specific and heterogeneous, with regions showing both concordant and discordant effects, particularly across MG subtypes. Subtype-specific analyses indicated broader and more heterogeneous overlap for AD–late-onset MG, including both major histocompatibility complex (MHC) and non-MHC loci, whereas AD–early-onset MG showed more restricted patterns largely confined to the MHC. Cross-trait meta-analysis and colocalisation further refined these findings, identifying a limited number of loci with evidence of shared AD-MG association, while most regions were consistent with distinct causal variants. A chromosome 16 locus showed the most consistent shared cross-trait AD-MG signal across multiple analytical frameworks. Mendelian randomisation analyses provided no evidence of a causal effect of AD liability on MG and yielded only suggestive, and inconclusive evidence for the reverse direction. Gene-level and expression-informed analyses prioritised immune-related genes, as well as regulators of transcription, chromatin organisation, and synaptic processes, without implying concordant causal variants across traits. Tissue and pathway analyses suggested shared immune involvement, with differential emphasis on innate immune processes in AD and adaptive immune pathways in MG. Notably, heterogeneity of effects within the MHC and across loci suggests that overlap reflects a complex, context-dependent architecture rather than a uniform immune-driven signal. Overall, our findings indicate that the AD–MG relationship is characterised by modest genome-wide polygenic overlap, substantial locus-specific heterogeneity, and partial convergence on immune-related genetic architecture, rather than a uniformly shared mechanism.
Full article
(This article belongs to the Special Issue Genomics of Human Disease)
►▼
Show Figures

Figure 1
Open AccessArticle
Modulation of NF-κB and TLR Signaling Pathways and Complement Components in Ovine Maternal Thyroid During Early Pregnancy
by
Yaqi Zhang, Jingjing Li, Fei Yang, Chenxu Wu, Leying Zhang and Ling Yang
Int. J. Mol. Sci. 2026, 27(11), 4791; https://doi.org/10.3390/ijms27114791 - 26 May 2026
Abstract
Pregnancy modulates the function of the thyroid gland to facilitate maternal immune tolerance, and nuclear factor kappa B (NF-κB) subunits, the IκB family, and toll-like receptors (TLRs) and complement signaling pathways may be implicated in maternal thyroid immunoregulation. However, it is unclear whether
[...] Read more.
Pregnancy modulates the function of the thyroid gland to facilitate maternal immune tolerance, and nuclear factor kappa B (NF-κB) subunits, the IκB family, and toll-like receptors (TLRs) and complement signaling pathways may be implicated in maternal thyroid immunoregulation. However, it is unclear whether early pregnancy modulates the expression of NF-κB subunits, the IκB family, TLRs, and complement components in the maternal thyroid. The objective of this study was to analyze the effects of early pregnancy on the expression of genes and proteins of these signaling pathways in the maternal thyroid in ewes. In this study, ovine thyroids (n = 6 for each group) were sampled on day 16 of the estrous cycle (N16) and on days 13, 16, and 25 of pregnancy (P13, P16, and P25) with one conceptus. The ewes had an average weight of 41 kg and a body condition score of 3. The mRNA and protein expression of the NF-κB subunits and IκB family were analyzed by RT-qPCR, western blot, and immunohistochemistry. The results showed that the expression of all NF-κB subunits, the IκB family, and TLRs, as well as C1q, C1r, C2, C4a, C5b, and C9, peaked at P16 among these four stages (p < 0.05). In addition, C1s expression was greater at N16 and P16 than at P13 and P25 (p < 0.05), and C3 expression was stronger at P16 and P25 compared to N16 and P13 (p < 0.05). In conclusion, early pregnancy modulates the expression of NF-κB subunits, the IκB family, TLRs, and complement components in the ovine thyroid at both the mRNA and protein levels, which may be essential for maternal thyroid adaptation to pregnancy and beneficial for the prevention of pregnancy-related thyroid diseases in ewes.
Full article
(This article belongs to the Special Issue Molecular Research on Reproductive Physiology and Endocrinology)
Open AccessArticle
Lichen Extracts Containing Volatile Compounds Induce Oxidative Stress and Modulate the Growth of Microcystis aeruginosa and Chlorella sorokiniana
by
Yasser Essadki, El Mehdi Darrag, Soukaina El Amrani Zerrifi, Mohamed Haida, Aafaf Krimech, Rosario Martins, Alexandre Campos, Vitor Vasconcelos, Noureddine Bouaïcha, Abdelaziz Baçaoui, Abdelilah Meddich, Brahim Oudra, Zakaria Tazart and Fatima El Khalloufi
Int. J. Mol. Sci. 2026, 27(11), 4790; https://doi.org/10.3390/ijms27114790 - 26 May 2026
Abstract
This study evaluates volatile extracts (HE1 and HE2) from the lichen Pseudevernia furfuracea as eco-friendly agents to control algal proliferation, specifically targeting the cyanobacterium Microcystis aeruginosa and the green microalga Chlorella sorokiniana. Both extracts exhibited potent anti-microalgal activity against the two species
[...] Read more.
This study evaluates volatile extracts (HE1 and HE2) from the lichen Pseudevernia furfuracea as eco-friendly agents to control algal proliferation, specifically targeting the cyanobacterium Microcystis aeruginosa and the green microalga Chlorella sorokiniana. Both extracts exhibited potent anti-microalgal activity against the two species with a minimum inhibitory concentration (MIC) ranging from 375 to 750 µg/mL. Furthermore, both extracts reduced cell density by more than 98% after eight days of treatment. Chlorophyll a and protein levels decreased significantly (>80%) in both species, indicating suppression of pigment synthesis. However, their physiological responses were distinct: M. aeruginosa underwent early acute oxidative stress and severe membrane damage, while C. sorokiniana exhibited delayed oxidative activation and a negative growth rate, suggesting non-lytic metabolic inhibition. An in silico study by molecular docking of the most abundant compounds identified in these volatile extracts, such as terpenoids (abietatriene, δ-cadinene) and a phenolic compound (atraric acid), showed that these compounds interact with vital cellular targets in M. aeruginosa and C. sorokiniana and likely contribute to the effects observed in these two species. Predictive toxicity by applying the ADMET framework confirmed the favorable bioavailability and low acute toxicity of these volatile compounds. Therefore, P. furfuracea volatiles are promising, species-specific, and environmentally safe candidates for mitigating aquatic algal proliferation through targeted oxidative and metabolic interference.
Full article
(This article belongs to the Special Issue Recent Advances in Algal Stress Physiology)
Open AccessReview
The Gut–Pancreas Axis in Type 1 Diabetes: Emerging Insights into Microbiota and Immune Interactions
by
Rahul Mittal, Priyanka Sinha, Jhanvi Doshi, Rebecca Goldmann, Mannat Mittal, Naisha Chaudhary, Vibha Ravindra and Khemraj Hirani
Int. J. Mol. Sci. 2026, 27(11), 4789; https://doi.org/10.3390/ijms27114789 - 26 May 2026
Abstract
The gut microbiota is increasingly recognized as an important factor in the pathogenesis of type 1 diabetes (T1D), although its exact role in disease initiation and progression remains uncertain. Earlier interpretations considered alterations in intestinal microbial composition as secondary effects of immune dysregulation
[...] Read more.
The gut microbiota is increasingly recognized as an important factor in the pathogenesis of type 1 diabetes (T1D), although its exact role in disease initiation and progression remains uncertain. Earlier interpretations considered alterations in intestinal microbial composition as secondary effects of immune dysregulation or metabolic disturbance. Recent longitudinal studies, however, suggest that specific microbial changes occur before the onset of islet autoimmunity, indicating a potential contributory role in the early phases of disease development. In this narrative review article, the gut–pancreas axis (GPA) is described as a dynamic and reciprocal system in which microbial, metabolic, and immune processes influence each other to shape β-cell outcomes. Evidence from human cohorts and experimental models links early life reductions in microbial diversity, impaired intestinal barrier function, and decreased production of short-chain fatty acids (SCFAs) to altered immune activation and β-cell damage. Microbiota transferred from individuals at risk for T1D has been shown to accelerate disease in animal models, supporting a possible causal relationship. Although experimental models support mechanistic links between microbiota alterations and autoimmune diabetes, current human evidence remains largely associative. Together, these findings suggest that microbial and immune networks interact in a feedback manner that can sustain immune tolerance or promote autoimmunity depending on environmental and host factors. Understanding T1D as a state of disrupted microbial and immune integration provides a basis for restoring gut–pancreas communication and preserving β-cell integrity.
Full article
(This article belongs to the Section Molecular Microbiology)
►▼
Show Figures

Figure 1
Open AccessReview
From Lysosomal Storage to Neurodegeneration: Sphingolipid Signaling as a Driver of CNS Pathology and Biomarker Strategy in Neuronopathic Gaucher Disease
by
Krista Casazza, Reena V. Kartha and Jeanine R. Jarnes
Int. J. Mol. Sci. 2026, 27(11), 4788; https://doi.org/10.3390/ijms27114788 - 26 May 2026
Abstract
Gaucher disease is a prototypical lysosomal sphingolipid storage disorder caused by pathogenic variants in GBA1, resulting in glucocerebrosidase deficiency and accumulation of bioactive lipids, including glucosylceramide and glucosylsphingosine (lyso-Gb1). While non-neuronopathic Gaucher disease is effectively managed with enzyme replacement and substrate reduction
[...] Read more.
Gaucher disease is a prototypical lysosomal sphingolipid storage disorder caused by pathogenic variants in GBA1, resulting in glucocerebrosidase deficiency and accumulation of bioactive lipids, including glucosylceramide and glucosylsphingosine (lyso-Gb1). While non-neuronopathic Gaucher disease is effectively managed with enzyme replacement and substrate reduction therapies, neuronopathic forms remain largely refractory to treatment due to progressive central nervous system (CNS) involvement and limited penetration of current therapies across the blood–brain barrier. Disease pathobiology extends beyond lysosomal substrate accumulation to encompass dysregulated sphingolipid signaling, particularly sphingosine-1-phosphate (S1P)-mediated “inside-out” signaling, alongside neuroinflammation, oxidative stress, and glial activation, which collectively drive neurodegeneration. In this review, we synthesize current knowledge on sphingolipid metabolism and signaling in neuronopathic Gaucher disease and integrate these mechanisms into a three-tier, CNS-focused biomarker framework. The first tier comprises substrate-proximal markers of lysosomal burden (lyso-Gb1), which reflect GCase deficiency and correlate with systemic disease severity but incompletely capture CNS pathology. The second tier comprises markers of glial activation and neuroinflammation (glial fibrillary acidic protein [GFAP], glycoprotein non-metastatic melanoma protein B [GPNMB]), which reflect the downstream neuroimmune response to sphingolipid accumulation. The third tier comprises markers of neuroaxonal injury (neurofilament light chain [NfL]), which index irreversible neuronal damage as the terminal consequence of uncontrolled CNS disease. Together, these tiers map distinct but mechanistically interconnected stages of disease progression, from lysosomal dysfunction through glial activation to neuroaxonal loss, enabling stage-specific interpretation of biomarker signals that single-analyte approaches cannot provide. We further examine how S1P-mediated inside-out signaling links intracellular lipid dysregulation to extracellular neuroimmune and neurovascular responses and how the blood–brain barrier shapes compartment-dependent biomarker behavior across cerebrospinal fluid and blood. By grounding biomarker selection in this mechanistic cascade, the framework provides explicit criteria for pairing analytes across tiers, interpreting discordance between peripheral and CNS compartments, and designing multi-modal endpoints for clinical trials of CNS-penetrant therapies. Despite these advances, significant challenges remain, including limited longitudinal datasets, variability in assay methodologies, and incomplete validation of biomarkers as surrogates of CNS disease progression. Addressing these gaps will require harmonized, multi-modal approaches integrating biochemical, functional, and imaging measures. By positioning neuronopathic Gaucher disease as a model of sphingolipid-driven neurodegeneration, this review highlights opportunities for biomarker-guided therapeutic development relevant to Gaucher disease and the broader spectrum of sphingolipid-associated neurological disorders.
Full article
(This article belongs to the Special Issue Sphingolipids: Health and Disease)
►▼
Show Figures

Figure 1
Open AccessReview
When Fertilization Is Not Enough: Maternal-Zygotic Transition as a Determinant of Embryo Competence in IVF
by
Charalampos Voros, Fotios Chatzinikolaou, Georgios Papadimas, Ali Can Gunes, Aristotelis-Marios Koulakmanidis, Ioannis Papapanagiotou, Athanasios Karpouzos, Diamantis Athanasiou, Kyriakos Bananis, Antonia Athanasiou, Aikaterini Athanasiou, Charalampos Tsimpoukelis, Maria Anastasia Daskalaki, Christina-Maria Trakatelli, Nikolaos Thomakos, Panagiotis Antsaklis, Dimitrios Loutradis and Georgios Daskalakis
Int. J. Mol. Sci. 2026, 27(11), 4787; https://doi.org/10.3390/ijms27114787 - 26 May 2026
Abstract
A significant concern with IVF is that many embryos fail to develop despite proper fertilization. This gap indicates that factors outside sperm-oocyte fusion influence developmental competency. The maternal-zygotic transition (MZT) is a crucial developmental phase during which control shifts from maternally inherited transcripts
[...] Read more.
A significant concern with IVF is that many embryos fail to develop despite proper fertilization. This gap indicates that factors outside sperm-oocyte fusion influence developmental competency. The maternal-zygotic transition (MZT) is a crucial developmental phase during which control shifts from maternally inherited transcripts to activation of the embryonic genome. During the early stages following fertilization, the embryo depends only on maternal mRNA and proteins amassed throughout oogenesis. For successful development, these transcripts must be expeditiously removed with the concurrent genome activation. Any disruption, whether due to inadequate maternal mRNA degradation, aberrant translational control, or delayed genome activation, has been associated with premature developmental stoppage and diminished blastocyst formation. Principal regulators, such as BTG4, CPEB1, DAZL, and components of the translational machinery, govern this modification and seem to be affected by the quality of the oocyte and the age of the mother. Increasing evidence suggests that disruption of MZT may account for instances of suboptimal embryo development that conventional assessment techniques cannot elucidate. MZT offers a biological framework for assessing embryo competency beyond simple appearance. If scientists had a deeper understanding of this process, they might identify molecular markers and enhance the selection of embryos in IVF.
Full article
(This article belongs to the Collection Advances in Cell and Molecular Biology)

Journal Menu
► ▼ Journal Menu-
- IJMS Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal Browser-
arrow_forward_ios
Forthcoming issue
arrow_forward_ios Current issue - Vol. 27 (2026)
- Vol. 26 (2025)
- Vol. 25 (2024)
- Vol. 24 (2023)
- Vol. 23 (2022)
- Vol. 22 (2021)
- Vol. 21 (2020)
- Vol. 20 (2019)
- Vol. 19 (2018)
- Vol. 18 (2017)
- Vol. 17 (2016)
- Vol. 16 (2015)
- Vol. 15 (2014)
- Vol. 14 (2013)
- Vol. 13 (2012)
- Vol. 12 (2011)
- Vol. 11 (2010)
- Vol. 10 (2009)
- Vol. 9 (2008)
- Vol. 8 (2007)
- Vol. 7 (2006)
- Vol. 6 (2005)
- Vol. 5 (2004)
- Vol. 4 (2003)
- Vol. 3 (2002)
- Vol. 2 (2001)
- Vol. 1 (2000)
Highly Accessed Articles
Latest Books
E-Mail Alert
News
27 January 2026
Meet Us at the 5th Molecules Medicinal Chemistry Symposium, 14–17 May 2026, Beijing, China
Meet Us at the 5th Molecules Medicinal Chemistry Symposium, 14–17 May 2026, Beijing, China

22 January 2026
“Do Not Be Afraid of New Things”: Prof. Michele Parrinello on Scientific Curiosity and the Importance of Fundamental Research
“Do Not Be Afraid of New Things”: Prof. Michele Parrinello on Scientific Curiosity and the Importance of Fundamental Research
Topics
Topic in
Cancers, IJMS, Pharmaceuticals, Pharmaceutics, Sci. Pharm., Current Oncology, Molecules
Recent Advances in Anticancer Strategies, 2nd Edition
Topic Editors: Hassan Bousbaa, Zhiwei HuDeadline: 31 May 2026
Topic in
Biomedicines, IJMS, JCM, Medicina, Neurology International
Advances in Exercise-Induced Neurogenesis, Neuronal and Functional Adaptations in Neurorehabilitation
Topic Editors: Carlos Bernal-Utrera, Cleofas Rodriguez-Blanco, Maria Livia Carrascal MorenoDeadline: 29 June 2026
Topic in
Dentistry Journal, IJMS, JCM, Medicina, Applied Sciences
Oral Health Management and Disease Treatment
Topic Editors: Christos Rahiotis, Felice Lorusso, Sergio Rexhep TariDeadline: 31 July 2026
Topic in
Biomedicines, Biomolecules, Cancers, Cells, Hematology Reports, IJMS
Advances in Molecular Pathogenesis and Targeted Therapies for Multiple Myeloma
Topic Editors: Chung Hoow Kok, Cindy H. S. Lee, Claudio CerchioneDeadline: 3 August 2026

Conferences
Special Issues
Special Issue in
IJMS
Novel Therapeutic Strategies Targeting Cancer Stem Cells
Guest Editor: Luciano VellónDeadline: 28 May 2026
Special Issue in
IJMS
Beneficial Plant–Microbe Interactions Under a Climate Change Scenario
Guest Editor: Juan Manuel Ruiz LozanoDeadline: 28 May 2026
Special Issue in
IJMS
Heavy Metal and Fluoride Stress: Toxicity and Tolerance Mechanisms in Plants
Guest Editor: Hatem RouachedDeadline: 30 May 2026
Special Issue in
IJMS
Cytokines and Inflammatory Diseases
Guest Editor: Pio ContiDeadline: 30 May 2026
Topical Collections
Topical Collection in
IJMS
Latest Review Papers in Molecular Pathology, Diagnostics, and Therapeutics
Collection Editor: Abdelkrim Hmadcha
Topical Collection in
IJMS
Feature Papers Collection in Biochemistry
Collection Editor: Clemente Capasso
Topical Collection in
IJMS
Molecular Research on Hematologic Diseases
Collection Editor: Kazuya Sato
Topical Collection in
IJMS
State-of-the-Art Materials Science in China
Collection Editors: Yujiang Fan, Yin Wang








