Journal Description
Vaccines
Vaccines
is an international, peer-reviewed, open access journal on laboratory and clinical vaccine research, utilization and immunization, published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Medicine, Research and Experimental) / CiteScore - Q1 (Pharmacology (medical))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 18.1 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.4 (2024);
5-Year Impact Factor:
3.7 (2024)
Latest Articles
Correction: Watts et al. Multi-Antigen Elephant Endotheliotropic Herpesvirus (EEHV) mRNA Vaccine Induces Humoral and Cell-Mediated Responses in Mice. Vaccines 2024, 12, 1429
Vaccines 2026, 14(5), 438; https://doi.org/10.3390/vaccines14050438 (registering DOI) - 14 May 2026
Abstract
►
Show Figures
The authors would like to make the following corrections to this published paper [...]
Full article
Open AccessSystematic Review
A Possible Unmet Need: Pneumococcal Vaccination in the Workplaces—A Systematic Review of Invasive Pneumococcal Disease Among Shipyard Workers
by
Matteo Riccò, Luca Pipitò, Claudio Costantino, Silvio Tafuri, Chiara Noviello, Marco Bottazzoli, Paolo Manzoni, Daniel Fiacchini, Marco Falcone, Pasquale Gianluca Giuri, Davide Gori and Antonio Cascio
Vaccines 2026, 14(5), 437; https://doi.org/10.3390/vaccines14050437 - 13 May 2026
Abstract
Background: Workplace-related outbreaks of invasive pneumococcal disease (IPD) have been increasingly reported among shipyard workers, yet their epidemiological and clinical features remain incompletely characterized. This systematic review and meta-analysis aimed to synthesize available evidence on IPD outbreaks in shipyard settings. Methods:
[...] Read more.
Background: Workplace-related outbreaks of invasive pneumococcal disease (IPD) have been increasingly reported among shipyard workers, yet their epidemiological and clinical features remain incompletely characterized. This systematic review and meta-analysis aimed to synthesize available evidence on IPD outbreaks in shipyard settings. Methods: A systematic search of PubMed/MEDLINE, Scopus, EMBASE, and medRxiv was conducted up to March 2026. Observational studies reporting IPD outbreaks in shipyards were included. Pooled incidence rates and clinical outcomes were estimated using random-effects models, with heterogeneity assessed by I2 statistics. Risk of bias was evaluated using the Newcastle–Ottawa Scale. Results: Eight studies describing six outbreaks across four European countries (France, Norway, Northern Ireland, Finland; 2015–2025) were included, encompassing 131 cases among 35,623 workers. The pooled incidence was 368.9 cases per 100,000 workers with an attack rate of 2.36 per 1000 person-months for total cases, compared to 200.49 cases per 100,000 workers (95%CI 103.54–387.85) and 1.10 cases per 1000 person-months (95% CI 0.17–2.03) for laboratory confirmed cases, with considerable heterogeneity across studies. Most cases occurred in men (97.7%), with the median age ranging from 39 to 48 years. Hospitalizations occurred in 79.1% of cases, intensive care unit admission in 13.7%, and the case fatality ratio was 0.8%. Serotype 4 accounted for 67.2% of characterized isolates. Occupational exposures and shared accommodation may have contributed to transmission, although this could not be formally assessed. Conclusions: IPD outbreaks in shipyard settings are characterized by high incidence but relatively favorable outcomes, likely reflecting workforce demographics. However, considerable heterogeneity and methodological limitations across studies constrain the interpretation of pooled estimates. Preventive strategies, including vaccination and workplace-targeted interventions, should be considered as plausible public health measures, with a proactive role for occupational health services.
Full article
Open AccessArticle
Enhanced Anti-Lung Cancer Efficacy of Neo-BCV Combined with Cisplatin: Immune Activation and Tumor Microenvironment Remodeling
by
Quexun Cai, Qianli Yang, Kangrui Zhang, Zhengyue Fei, Ruochen Zhao, Tao Lu, Kecheng Xu, Zhenyi Wang and Peihua Lu
Vaccines 2026, 14(5), 436; https://doi.org/10.3390/vaccines14050436 - 13 May 2026
Abstract
Background: Lung cancer is the top cause of cancer-related mortality globally, and chemo-immunotherapy is a core therapeutic strategy for it. The novel bacterial composite vaccine (Neo-BCV) we developed previously can activate anti-tumor immunity. This study explored its synergistic anti-tumor effect with cisplatin (CDDP),
[...] Read more.
Background: Lung cancer is the top cause of cancer-related mortality globally, and chemo-immunotherapy is a core therapeutic strategy for it. The novel bacterial composite vaccine (Neo-BCV) we developed previously can activate anti-tumor immunity. This study explored its synergistic anti-tumor effect with cisplatin (CDDP), along with the underlying immunomodulatory mechanisms and molecular regulatory networks. Methods: A murine Lewis lung cancer (LLC) model was established to evaluate the efficacy of the combination therapy. Flow cytometry and multiplex cytokine assay were used to detect immune cell subsets and functional molecules in the spleen, serum and tumor tissues. RNA-sequencing (RNA-seq) was used to elucidate the molecular regulatory networks following the combination therapy in the tumor tissues. Body weight, blood indexes, serum biochemistry and H&E staining were monitored to verify biosafety. Results: Neo-BCV combined with CDDP achieved an 87.77% tumor growth inhibition rate, showing the most significant anti-tumor effect. The combination promoted DC maturation, enhanced effector immune cell infiltration, reduced immunosuppressive cells, upregulated Th1-type cytokines and downregulated CD8+ T cell surface PD-1. RNA-seq confirmed enrichment of multiple immune effector pathways, supporting tumor immune microenvironment remodeling. The combination alleviated CDDP-induced weight loss, had no obvious adverse effects on physiological indicators, and exhibited good biosafety. Conclusions: Neo-BCV combined with CDDP achieves enhanced anti-tumor efficacy and favorable biosafety in murine lung cancer models by regulating immune cell subsets and activating immune-related molecular pathways, providing a solid preclinical basis for its clinical translation in lung cancer treatment.
Full article
(This article belongs to the Section Vaccination Against Cancer and Chronic Diseases)
►▼
Show Figures
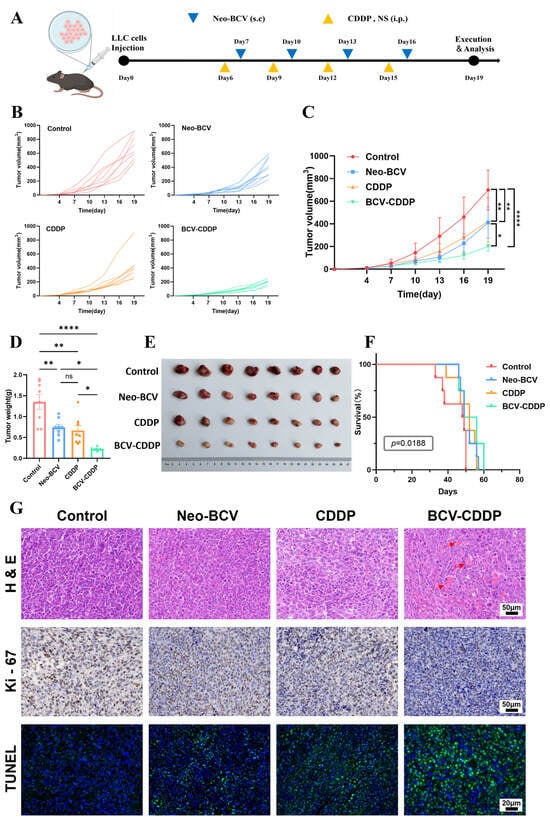
Figure 1
Open AccessReview
Immune Cell Signaling in Feline Infectious Peritonitis Virus Infection and Implications for Vaccine Design
by
Hye-Mi Lee
Vaccines 2026, 14(5), 435; https://doi.org/10.3390/vaccines14050435 - 13 May 2026
Abstract
Feline infectious peritonitis virus (FIPV) remains one of the most challenging viral diseases in veterinary medicine, largely owing to the absence of a consistently effective and safe vaccine. Despite widespread feline coronavirus infection, only a subset of infected cats progresses to feline infectious
[...] Read more.
Feline infectious peritonitis virus (FIPV) remains one of the most challenging viral diseases in veterinary medicine, largely owing to the absence of a consistently effective and safe vaccine. Despite widespread feline coronavirus infection, only a subset of infected cats progresses to feline infectious peritonitis, indicating that host immune responses are key determinants of disease outcomes. Accumulating evidence indicates that disease severity is driven not only by viral replication but also by macrophage- and monocyte-centered immune signaling, leading to excessive inflammation and systemic immunopathology in the host. Previous vaccine approaches against FIPV have failed to provide consistent protection and, in some cases, have been associated with enhanced disease. These outcomes suggest that vaccine-induced immune responses that recapitulate pathogenic signaling patterns may exacerbate disease rather than confer protection. In this review, we discuss the current knowledge of immune cell signaling pathways implicated in FIPV infection, including innate sensing through Toll-like receptors, downstream mitogen-activated protein kinases and NF-κB signaling, cytokine production profiles, Fc receptor-associated processes, and intracellular pathways such as autophagy, and how these mechanisms shape vaccine-induced immunity. By integrating insights from immune signaling kinetics, antibody functionality, adjuvant-driven pathway engagement, and platform-specific immune signatures, this review emphasizes the need to reframe FIPV vaccine development strategies that actively shape host immune responses. Rather than maximizing immunogenicity, successful vaccine design is likely to depend on limiting sustained macrophage activation and pro-inflammatory cytokine amplification while supporting antiviral immune functions, thereby reducing the risk of antibody-dependent enhancement and immunopathology. Beyond feline diseases, these considerations provide broader lessons for vaccine design in settings where immune-mediated pathology contributes to disease severity.
Full article
(This article belongs to the Section Pathogens-Host Immune Boundaries)
►▼
Show Figures
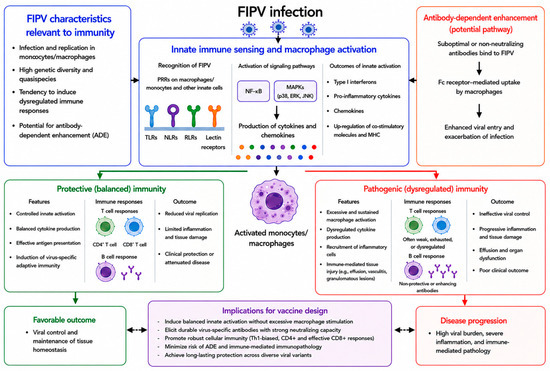
Figure 1
Open AccessEditorial
Closing Editorial—Special Issue on “Vaccines and Vaccination: HIV, Hepatitis Viruses and HPV”
by
Sonia Moretti and Ivan Schietroma
Vaccines 2026, 14(5), 434; https://doi.org/10.3390/vaccines14050434 - 12 May 2026
Abstract
We are pleased to present this closing editorial for the Special Issue “Vaccines and Vaccination: HIV, Hepatitis Viruses and HPV”, which we had the privilege of coordinating [...]
Full article
(This article belongs to the Special Issue Vaccines and Vaccination: HIV, Hepatitis Viruses, and HPV)
Open AccessCorrection
Correction: Loayza et al. Monitoring the Performance of National Immunization Programs: Innovative Methodology and Tool for Countries’ Self-Assessment. Vaccines 2026, 14, 258
by
Sergio Loayza, Bertha Capistran, Marcela Contreras, Martha Velandia and Daniel Salas
Vaccines 2026, 14(5), 433; https://doi.org/10.3390/vaccines14050433 - 12 May 2026
Abstract
The authors would like to make the following correction to this published paper [...]
Full article
(This article belongs to the Section Vaccines and Public Health)
Open AccessReview
Impact of the COVID-19 Pandemic on HPV Vaccination in Low- and Middle-Income Countries: A Scoping Review
by
Joyce Omondi, Robert Ambogo, Candy Ochieng, Marwa Farag and George Mutwiri
Vaccines 2026, 14(5), 432; https://doi.org/10.3390/vaccines14050432 - 12 May 2026
Abstract
Background: The COVID-19 pandemic caused disruptions in HPV vaccination and may have severely undermined global cervical cancer prevention, posing long-term risks to controlling cervical cancer and other HPV-related diseases. Objective: We conducted a scoping review to map and synthesize available evidence on how
[...] Read more.
Background: The COVID-19 pandemic caused disruptions in HPV vaccination and may have severely undermined global cervical cancer prevention, posing long-term risks to controlling cervical cancer and other HPV-related diseases. Objective: We conducted a scoping review to map and synthesize available evidence on how the COVID-19 pandemic has affected human papillomavirus (HPV) vaccination programs in low- and middle-income countries (LMICs) focusing on changes in vaccine delivery and coverage, determinants of uptake, economic and programmatic consequences and vaccine hesitancy. Methods: Inclusion criteria were limited to studies published in the English language between January 2020 to May 2025, and followed JBI and Arksey & O’Malley’s scoping review guidelines. The review proceeded through three stages: database searches, gray literature and citation tracking and used a PRISMA-ScR checklist to guide narrative and tabular synthesis. Results: A total of 1063 records, 57 studies were included in the final analysis, and these were spread out across 37 low- and middle-income countries (LMICs) mainly in Africa, Asia, and Latin America. Our analysis revealed that HPV vaccination coverage declined substantially during the COVID-19 pandemic, with reductions of up to 90% reported across the included studies, in the context of school closures, workforce redeployment, and supply-chain disruptions. Recovery efforts also faced major barriers including vaccine hesitancy, misinformation about COVID-19 vaccines, and travel restrictions. Strategies like digital tools, mobile clinics, and community health workers showed promise alongside integrated school- and facility-based approaches, although there is limited evidence on cost-effectiveness and long-term sustainability of these strategies. Conclusions: HPV vaccination in LMICs was significantly disrupted by the COVID-19 pandemic due to unreliable vaccine supply chains, health-worker shortages, and challenges tied to school-based vaccine delivery. Although recovery methods show potential, longer observation periods are needed to determine their full effectiveness.
Full article
(This article belongs to the Special Issue Strategies to Increase the Uptake and Delivery of HPV Vaccination in LMICs)
►▼
Show Figures
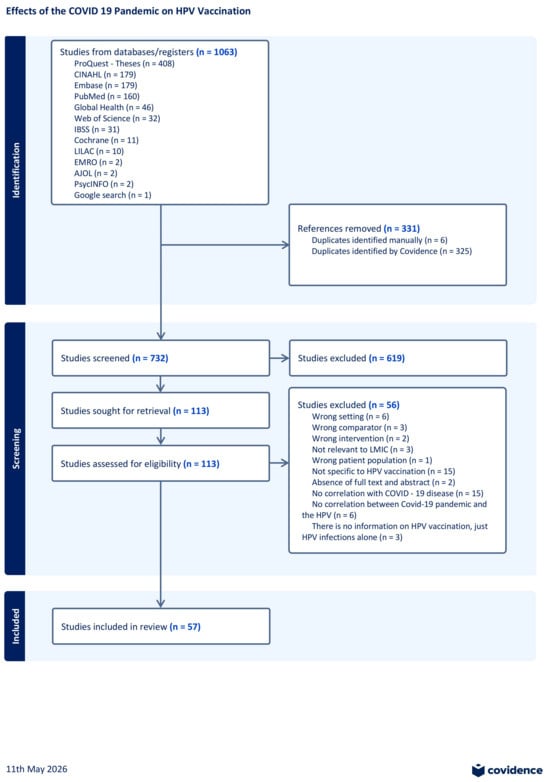
Figure 1
Open AccessArticle
Support for Tying Polio Vaccination Status to Child Tax Credit Eligibility in the United States
by
Matthew R. Boyce and Rebecca Katz
Vaccines 2026, 14(5), 431; https://doi.org/10.3390/vaccines14050431 - 12 May 2026
Abstract
Background/Objectives: In a context characterized by persistent vaccine hesitancy, shifting mandates, and stagnating immunization coverage rates, novel policy tools may be required to bolster immunization coverage in the United States. We conducted a national survey to characterize public support for a new tax
[...] Read more.
Background/Objectives: In a context characterized by persistent vaccine hesitancy, shifting mandates, and stagnating immunization coverage rates, novel policy tools may be required to bolster immunization coverage in the United States. We conducted a national survey to characterize public support for a new tax policy that would require parents to prove that their children are age-appropriately immunized against polio to be eligible for the federal child tax credit. Methods: The survey was conducted in November 2025. Respondents were asked to provide demographic information and use a Likert-scale to indicate their support for the proposed policy. Chi-squared tests and ordinal logistic regression models were used to compare support for the proposed policy across subpopulations. Results: 980 individuals were included in the analysis. 55.8% of respondents supported adding age-appropriate polio immunization to the child tax credit eligibility criteria. 20.9% of respondents opposed the policy proposal. Relative levels of support for the policy differed according to respondent gender, age, 2024 presidential election behavior, and geographic region. However, support did not differ significantly according to race, ethnicity, educational attainment, income, or partisanship. Conclusions: Results show that most survey respondents would support a policy that would add polio immunization status to the eligibility criteria for the federal child tax credit. Further, support did not differ across key demographic and political subgroups. Larger surveys should validate these findings and investigate support for adding additional vaccines to the eligibility criteria.
Full article
(This article belongs to the Special Issue New Challenges in Vaccines and Vaccination: A Public Health Perspective)
Open AccessArticle
The Yellow Fever Virus Type-Specific Epitope Recognized by Monoclonal Antibody 2D12 Neutralizes Wild Type, but Not Live Attenuated 17D or French Neurotropic Vaccine Strains
by
Clairissa A. Hansen, Shawn Rast, Jill K. Thompson, Haiping Hao, Daniel Jupiter, Stephen Higgs, Nigel Bourne and Alan D. T. Barrett
Vaccines 2026, 14(5), 430; https://doi.org/10.3390/vaccines14050430 - 12 May 2026
Abstract
Background/Objectives: The envelope (E) protein of orthoflaviviruses contains antigenic sites that are composed of one or more epitopes, which can vary in antigenic specificity, including between viral species, strains, and even substrains. Monoclonal antibodies (mAbs) that bind these epitopes vary in functionality
[...] Read more.
Background/Objectives: The envelope (E) protein of orthoflaviviruses contains antigenic sites that are composed of one or more epitopes, which can vary in antigenic specificity, including between viral species, strains, and even substrains. Monoclonal antibodies (mAbs) that bind these epitopes vary in functionality based on their specificity. This makes mAbs useful to study the differences in phenotypes between strains of viruses, such as the wild type (WT) and live attenuated vaccine strains of yellow fever virus (YFV). mAb 2D12 was raised against the 17D-204 YFV vaccine substrain virus (YF VAX®) by Schlesinger et al. in 1983. However, it only neutralizes Asibi WT virus, not the 17D-204 vaccine substrain virus. Results: We confirmed these results and demonstrated that mAb 2D12 fails to neutralize all 17D vaccine substrains (17D-204, 17DD, and 17D-213), indicating that the minor differences between these virus substrains do not affect the epitope or functionality of mAb 2D12. In addition, mAb 2D12 was found to neutralize WT strain of French viscerotropic virus (FVV), with statistically indistinguishable neutralization from the WT strain Asibi. All but one of the live attenuated French neurotropic vaccine (FNV) derivative viruses had significantly lower neutralization than WT strains Asibi and FVV. FVV, Asibi, 17D, and FNV have many amino acid differences in the membrane (M) and E proteins. It is unclear which of them contributes to this differential neutralization. However, FNV and 17D have common amino acid substitutions from WT FVV and Asibi at positions M-36 and E-331, suggesting that one or both of these residues may contribute to the 2D12 epitope. Conclusions: Overall, mAb 2D12 is a valuable tool to distinguish WT virulent strains of YFV from live attenuated vaccine strains.
Full article
(This article belongs to the Special Issue Arboviral Infections (Dengue, Chikungunya, Zika, and Other Mosquito-Borne Diseases): Immune Response and Vaccine Development)
►▼
Show Figures
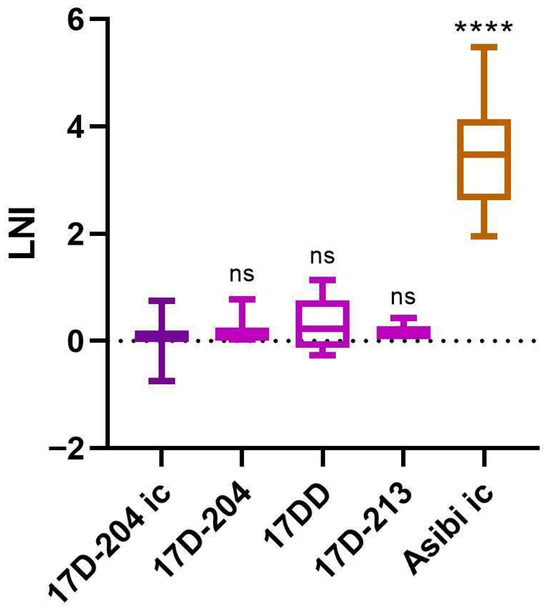
Figure 1
Open AccessArticle
Determinants and Dynamics of COVID-19 Vaccine Hesitancy in University Students: A Machine Learning Analysis
by
Daliana Lobo Torres and Zahid Ahmad Butt
Vaccines 2026, 14(5), 429; https://doi.org/10.3390/vaccines14050429 - 11 May 2026
Abstract
Background: Booster vaccine hesitancy poses a challenge to sustained COVID-19 immunization even among individuals who accepted primary vaccination. This study examined associated factors and patterns of change in vaccine attitudes among university students in Ontario, Canada. Methods: A cross-sectional survey dataset was analyzed
[...] Read more.
Background: Booster vaccine hesitancy poses a challenge to sustained COVID-19 immunization even among individuals who accepted primary vaccination. This study examined associated factors and patterns of change in vaccine attitudes among university students in Ontario, Canada. Methods: A cross-sectional survey dataset was analyzed using validated psychometric scales to measure hesitancy toward primary and booster COVID-19 vaccination. Changes in hesitancy were operationalized as the continuous difference between booster and primary scores (
(This article belongs to the Special Issue Acceptance and Hesitancy in Vaccine Uptake: 3rd Edition)
►▼
Show Figures
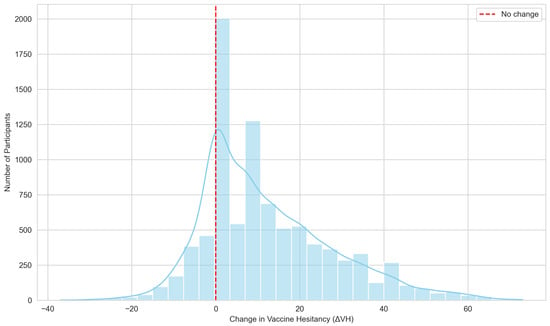
Figure 1
Open AccessArticle
Mpox Vaccination Willingness, Concern Profiles, and Associated Factors Among Men Who Have Sex with Men in Changsha, China
by
Yingying Zhou, Wenqiang Wang, Yun Kuang, Qiang Hu, Lin Shen, Qiangming Xie and Zhi Xie
Vaccines 2026, 14(5), 428; https://doi.org/10.3390/vaccines14050428 - 10 May 2026
Abstract
Background: Mpox vaccination is an important prevention strategy for men who have sex with men (MSM), yet responses to vaccination may not be adequately captured by a simple willing-versus-unwilling framing. We examined correlates of vaccination willingness and heterogeneity within the delayed/refused responses
[...] Read more.
Background: Mpox vaccination is an important prevention strategy for men who have sex with men (MSM), yet responses to vaccination may not be adequately captured by a simple willing-versus-unwilling framing. We examined correlates of vaccination willingness and heterogeneity within the delayed/refused responses subgroup in Changsha, China. Methods: A cross-sectional survey was conducted using respondent-driven sampling (RDS). Vaccination willingness was classified as immediate willingness versus delayed/refused responses. Analyses included cluster-robust logistic regression, RDS-weighted regression, generalized estimating equations, and a recruiter-linked network-lag model. Among respondents with delayed/refused responses, concern profiles were explored using unsupervised clustering of standardized concern items. Results: Among 405 recruited MSM without a self-reported mpox infection history, immediate willingness and delayed/refused responses were nearly equally distributed, indicating that lack of immediate willingness was common. Across primary models, ever use of pre-exposure prophylaxis (PrEP) and higher mpox-related information exposure frequency were the most consistent correlates of immediate willingness versus delayed/refused responses, whereas basic sociodemographic variables showed little evidence of independent association. Within the delayed/refused group, three partially overlapping patterns emerged: broadly elevated cross-domain concern, low-concern delay with few strongly endorsed barriers, and more selective safety- and burden-related concerns. These findings suggest that a lack of immediate willingness may arise through different psychosocial pathways rather than a single common mechanism. Conclusions: Mpox vaccination willingness among MSM in Changsha appeared to be shaped more by prevention-related behaviors and psychosocial factors than by basic sociodemographic profiles alone. Vaccination strategies may benefit from cross-topic sexual-health communication, integrated prevention efforts, and subgroup-sensitive approaches to delayed or refused willingness.
Full article
(This article belongs to the Special Issue Epidemiology and Vaccinations in Infectious Diseases)
►▼
Show Figures
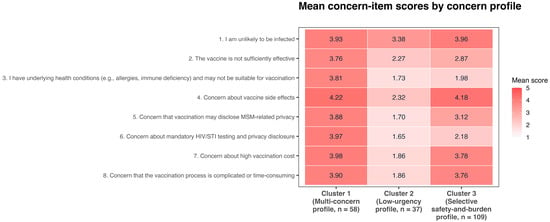
Figure 1
Open AccessArticle
Comparison Between a Minimally Invasive Capillary Blood Sampling Technique and Venipuncture for Core Vaccine Antibody Titration in Dogs
by
Paola Dall‘Ara, Lucia Tamanza, Federico Alghisi, Davide Raccagni, Alice Garegnani and Joel Filipe
Vaccines 2026, 14(5), 427; https://doi.org/10.3390/vaccines14050427 - 10 May 2026
Abstract
►▼
Show Figures
Background/Objectives: International guidelines advocate for personalized vaccination protocols using point-of-care (POC) antibody titration to identify dogs requiring boosters for CPV-2, CDV, and CAdV-1. As traditional venipuncture can be challenging in specific patients, this study evaluate the clinical agreement of a novel minimally
[...] Read more.
Background/Objectives: International guidelines advocate for personalized vaccination protocols using point-of-care (POC) antibody titration to identify dogs requiring boosters for CPV-2, CDV, and CAdV-1. As traditional venipuncture can be challenging in specific patients, this study evaluate the clinical agreement of a novel minimally invasive capillary blood sampling technique (ear-prick) for core vaccine antibody titration. Methods: Paired blood samples were collected from 55 healthy dogs using venipuncture and an ear-prick technique with a portable lancet. Antibody titers were determined using a semi-quantitative POC kit (VacciCheck® Canine). The procedure was optimized comparing 28G and 21G lancets, with the latter used in 43 dogs to ensure adequate blood flow. Comprehensive statistical methods evaluated the correlation and agreement between the two sampling techniques. Results: Statistical analysis showed no significant differences between sampling methods (p > 0.05). In the optimized group (21G lancet), full match rates reached 81.4% for CPV-2, 76.8% for CDV, and 74.4% for CAdV-1. Clinical concordance was exceptionally high: 95.3% for CPV-2, 90.7% for CDV, and 100% for CAdV-1. Statistical analysis confirmed perfect agreement (1.00) for CPV-2 and CAdV-1, and moderate agreement (0.48) for CDV. Conclusions: The ear-prick technique using a 21G lancet is a reliable, minimally invasive alternative to venipuncture for antibody titration. This method simplifies clinical procedures and facilitates personalized immunization monitoring. Given the minimal blood volume required, it represents a versatile approach for evaluating immune status and protection levels to core vaccines in diverse settings, including pediatric and shelter medicine.
Full article

Figure 1
Open AccessEditorial
Vaccination in Immune-Mediated Intestinal Diseases: Efficacy, Safety, and Future Directions
by
Adi Lahat and Kassem Sharif
Vaccines 2026, 14(5), 426; https://doi.org/10.3390/vaccines14050426 - 9 May 2026
Abstract
Vaccination remains one of the most effective public health interventions for preventing infectious diseases, reducing morbidity, mortality, and healthcare burden worldwide [...]
Full article
(This article belongs to the Special Issue Vaccine Efficacy and Safety in Patients with Immune-Mediated Intestinal Diseases)
Open AccessArticle
A Human Mobility-Based Modeling Study of Influenza Vaccination Strategies Across Socioeconomically Disparate Regions in China
by
Lei Zhou, Yunkang Zhao, Hongjie Wei, Kang Fang, Huimin Qu, Yanshu Ke, Jia Rui, Dayan Wang, Tianmu Chen and Liming Li
Vaccines 2026, 14(5), 425; https://doi.org/10.3390/vaccines14050425 - 9 May 2026
Abstract
Background: China‘s influenza vaccination coverage remains at a low rate, with significant regional socioeconomic disparities, lacking targeted distribution strategies and achievable coverage targets. This study aims to provide scientific evidence for formulating differentiated and feasible vaccination strategies across Chinese provinces based on regional
[...] Read more.
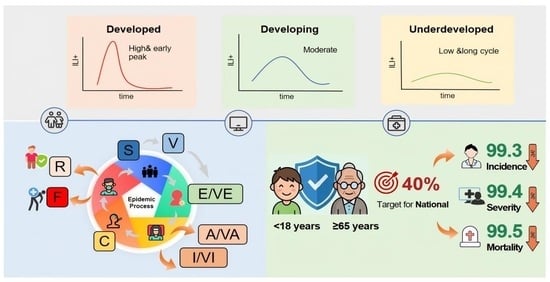
Background: China‘s influenza vaccination coverage remains at a low rate, with significant regional socioeconomic disparities, lacking targeted distribution strategies and achievable coverage targets. This study aims to provide scientific evidence for formulating differentiated and feasible vaccination strategies across Chinese provinces based on regional economic gradients. Methods: We employed the Susceptible-Vaccinated-Exposed-Asymptomatic-Infectious-Critical-Fatal-Recovered/Removed (SVEAICFR) model to simulate various vaccination strategies, analyzing the reduction in disease burden and vaccine dose requirements across underdeveloped, developing, and developed regions. The optimal strategy and achievable coverage targets were subsequently determined. Results: The 31 provinces were clustered into three categories based on economic levels, showing significant spatiotemporal differences in epidemics (Kruskal–Wallis test, all p < 0.001). Developed regions showed the earliest onset and highest peaks (influenza-like illness positive (ILI+) index ≈ 12–13, Baidu Influenza Search Index (BISI) ≈ 310,000). Developing regions exhibited moderate lagging by 1–2 weeks, while underdeveloped regions had the lowest peaks (ILI+ 3–4) and longer epidemic cycles. During the 2023–2024 influenza season, the national predicted vaccination rate was only 2.89% with marked regional disparities. Baseline incidence, severity, and mortality rates were 13,374.93, 49.52, and 8.37 cases per 100,000 population, respectively. Modeling indicates that increasing influenza vaccination coverage rates for populations aged <18 and ≥65 to a theoretical threshold (39.73% of the total population) before the season could reduce incidence, severity, and mortality rate by 99.26%,99.42%, and 99.46%, respectively. Conclusions: Influenza prevalence in China exhibits significant regional heterogeneity, necessitating differentiated measures based on regional economic gradients. Regional support mechanisms should be implemented to promote equitable vaccine distribution. Priority vaccination for high-risk populations (aged <18 and ≥65), to reach a 40% theoretical national coverage target, is recommended via realistic implementation pathways to minimize the disease burden of influenza.
Full article
(This article belongs to the Special Issue New Technology for Vaccines and Vaccine-Preventable Diseases)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Incidence of COVID-19 and Influenza-Related Outcomes and Vaccinations in the United States, October 2022 Through December 2024
by
Heather R. Hensler, Tianyi Lu, Yoonyoung Park, Machaon Bonafede, Isabelle Winer, Christopher Adams, Keya Joshi and Amanda Wilson
Vaccines 2026, 14(5), 424; https://doi.org/10.3390/vaccines14050424 - 8 May 2026
Abstract
Background/Objectives: We still do not clearly know whether COVID-19 continues to impose a greater clinical burden than influenza in the “post-pandemic” era. Our study quantified and compared monthly COVID-19 and influenza hospitalization incidence among adult subgroups from October 2022 through December 2024. We
[...] Read more.
Background/Objectives: We still do not clearly know whether COVID-19 continues to impose a greater clinical burden than influenza in the “post-pandemic” era. Our study quantified and compared monthly COVID-19 and influenza hospitalization incidence among adult subgroups from October 2022 through December 2024. We assessed vaccine coverage trends and examined vaccination status among those hospitalized. Methods: Using the Veradigm linked claims and electronic health record dataset, we conducted a non-interventional, retrospective cohort study; three monthly cohorts included individuals aged 65+, high-risk (HR) adults (defined as adults 18+ with HR conditions and/or aged 65+), and adults aged 50–64 years who were enrolled with both medical and pharmacy coverage. We estimated monthly cumulative incidence of COVID-19 and influenza-related hospitalizations, vaccination coverage rates, and the proportion of hospitalized individuals who had received yearly updated vaccines. Results: COVID-19 hospitalizations consistently exceeded those of influenza across months and populations. Among adults aged 65+, COVID-19 hospitalization rates were 2–3 times higher than influenza in winter and 20–30 times higher during off-season months, with similar trends observed in high risk adults. COVID-19 incidence surged in summer, while influenza remained seasonally confined. Vaccination coverage for influenza peaked near 50% annually; COVID-19 coverage was lower, peaking at ~26% by December each year. Most hospitalizations occurred among unvaccinated individuals, particularly for COVID-19. Conclusions: COVID-19 continues to impose a substantial, year-round burden, particularly in older and high-risk adults, exceeding that of influenza. The high proportion of unvaccinated hospitalizations highlight a critical gap in prevention efforts and underscore the need for improved public health messaging and vaccine adoption.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
►▼
Show Figures
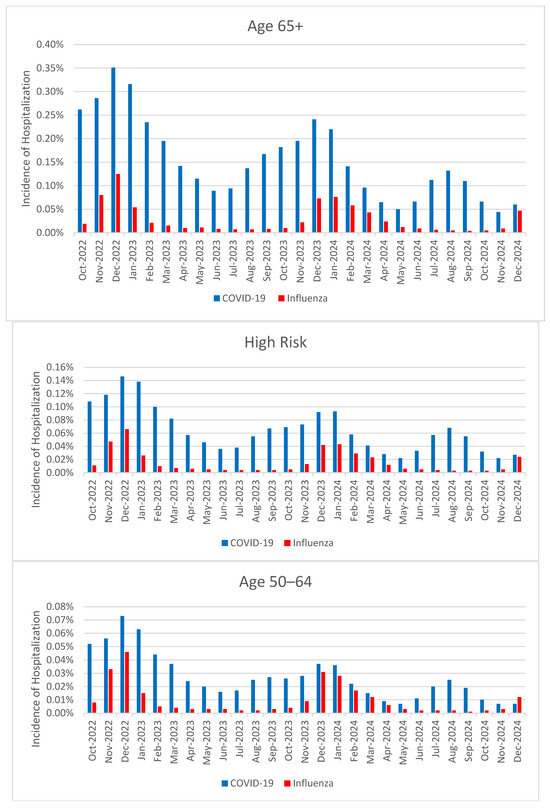
Figure 1
Open AccessArticle
Cellular Immune Response and T Cell Epitope Mapping of Plasmodium falciparum Chimeric Vaccine Candidate GMZ2.6c and Its Components (MSP-3, GLURP and Pfs48/45) in Individuals Naturally Exposed to Malaria in Brazilian Amazon
by
Barbara de Oliveira Baptista, Isabela Ferreira Soares, Hugo Amorim dos Santos de Souza, Jenifer Peixoto de Barros, Evelyn Kety Pratt Riccio, Rodrigo Medeiros Martorano, Rodrigo Nunes Rodrigues-da-Silva, Linda Eva Amoah, Susheel Kumar Singh, Michael Theisen, Josué da Costa Lima-Junior, Paulo Renato Rivas Totino, Cláudio Tadeu Daniel-Ribeiro and Lilian Rose Pratt-Riccio
Vaccines 2026, 14(5), 423; https://doi.org/10.3390/vaccines14050423 - 8 May 2026
Abstract
Background/Objectives: The GMZ2.6c malaria vaccine candidate is a multi-stage P. falciparum chimeric protein that contains a fragment of the sexual stage Pfs48/45-6c protein genetically fused to GMZ2, which is an asexual stage vaccine construct consisting of conserved domains of Glutamate-Rich Protein (GLURP) and
[...] Read more.
Background/Objectives: The GMZ2.6c malaria vaccine candidate is a multi-stage P. falciparum chimeric protein that contains a fragment of the sexual stage Pfs48/45-6c protein genetically fused to GMZ2, which is an asexual stage vaccine construct consisting of conserved domains of Glutamate-Rich Protein (GLURP) and Merozoite Surface Protein-3 (MSP-3). Previous studies showed that GMZ2.6c is widely recognized by antibodies from individuals living in endemic areas of Brazil and that levels of anti-GMZ2.6c increase with malaria exposure and may contribute to immunity against the parasite. As cell-mediated responses are crucial for parasite control and protection, identifying antigens that elicit antigen-specific T cell recall in naturally exposed populations is the key to vaccine development. This study aimed to evaluate the cellular immune response against GMZ2.6c and its components (MSP-3, GLURP, and Pfs48/45) and to identify promiscuous T cell epitopes in individuals exposed to malaria in the Brazilian Amazon, considering the impact of active P. falciparum infection on antigen-specific T cell recall. Methods: This study was carried out using peripheral blood mononuclear cells (PBMCs) from individuals with active P. falciparum infection (PFI) and non-infected individuals exposed to malaria (NI) from Cruzeiro do Sul and Mâncio Lima, Acre State, and Guajará, Amazonas State. The PBMCs were stimulated with GMZ2.6c and its components, and cellular activation, CD4+ and CD8+ memory T cell subsets, and cytokine production were evaluated by flow cytometry. IFN-γ-secreting T cells were quantified by ELISpot using predicted T cell epitopes. Results: The individuals infected by P. falciparum displayed more CD8+ T cell activation in response to MSP-3 and Pfs48/45 and an increase in CD4+ TCM cells and a reduction in CD4+ TEM cells following stimulation with Pfs48/45 and GMZ2.6c. The PBMCs from both groups showed elevated production of IL-6 and TNF after stimulation with GMZ2.6c, MSP-3, and Pfs48/45, but only the non-infected individuals had high levels of IL-10. T cell epitope prediction identified sequences within MSP-3, GLURP, and Pfs48/45 that elicited IFN-γ responses in both the non-infected and P. falciparum-infected individuals. Conclusions: Individuals exhibit cellular immune responses to MSP-3 and Pfs48/45 that are recalled following GMZ2.6c stimulation. P. falciparum infection may modulate immune response, inducing a prominent pro-inflammatory response. Conversely, in the absence of the parasite, the individuals displayed balanced Th1/Th2 cytokine production. Several promiscuous T cell epitopes were able to recall IFN-γ responses. Further studies are needed to fully ascertain the potential of GMZ2.6c as a protective candidate vaccine against malaria.
Full article
(This article belongs to the Special Issue Immune Responses After Malaria Infection and Next Generation Malaria Vaccine)
►▼
Show Figures
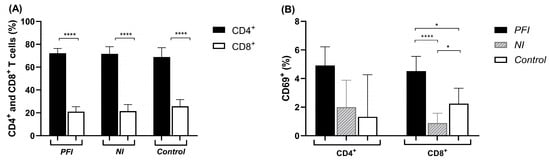
Figure 1
Open AccessArticle
Parents’ Preferred Age (9–12) for HPV Vaccination: Decision-Making and Rationale
by
Holly B. Fontenot, Siobhan Coad, Erica J. Liebermann, Erika L. Thompson, Emma Collo, Tiffannie Chang, Melanie Kornides and Gregory Zimet
Vaccines 2026, 14(5), 422; https://doi.org/10.3390/vaccines14050422 - 7 May 2026
Abstract
Background/Objective: The objective of this study was to explore parental preferences for the age of HPV vaccination (9–12) and the rationales for these preferences. Methods: Four online text-based focus groups were conducted with a national sample of 43 parents who have
[...] Read more.
Background/Objective: The objective of this study was to explore parental preferences for the age of HPV vaccination (9–12) and the rationales for these preferences. Methods: Four online text-based focus groups were conducted with a national sample of 43 parents who have at least one child aged 9–10 years. Participants discussed preferred age for HPV vaccination and how it relates to the routine adolescent vaccine schedule in the United States (US). Content analysis was utilized to identify emergent themes. Results: Three themes surrounding parents’ motivating factors related to HPV-vaccination schedule preferences emerged from the analysis of the focus group discussions: (1) a belief that age 9 is too young versus a belief in early protection, (2) the number of shots administered per visit (a desire to spread shots out or group together), and (3) the parent follows their health care provider’s recommendations. Conclusions: This qualitative study of parental preferences regarding HPV vaccination age and how it relates to the routine adolescent vaccine schedule reveals mixed parental decision-making and rationales for vaccine acceptance and at which age. Given the dynamic vaccine policy landscape in the US, it is essential for providers to understand parental perspectives and motivating factors related to vaccine decision-making and integrate these drivers into clinical practice to best support families and public health at large.
Full article
Open AccessArticle
Mucosal Delivery of Recombinant SARS-CoV-2 Spike Receptor-Binding Domain Antigen Containing Immune-Stimulating Peptides Induces Protective Immune Responses Against Viral Infection in huACE2 Mice
by
Byeol-Hee Cho, Ju Kim and Yong-Suk Jang
Vaccines 2026, 14(5), 421; https://doi.org/10.3390/vaccines14050421 - 7 May 2026
Abstract
Background: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infects host cells through the interaction between the spike protein receptor-binding domain (RBD) and the human angiotensin-converting enzyme 2 (hACE2) receptor, which is expressed on epithelial cells in various tissues, including the respiratory tract.
[...] Read more.
Background: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infects host cells through the interaction between the spike protein receptor-binding domain (RBD) and the human angiotensin-converting enzyme 2 (hACE2) receptor, which is expressed on epithelial cells in various tissues, including the respiratory tract. Therefore, mucosal immunity in the respiratory tract plays a key role in protection against viral infection. Previously, we demonstrated that intranasal administration of antigens (Ags) conjugated with the M cell-targeting peptide Co4B enhances both mucosal and systemic immune responses. That conjugation with human β-defensin 2 (HBD2) increases neutralizing antibody (Ab) responses. Methods: A recombinant antigen conjugate incorporating both Co4B and HBD2 was designed to enhance immunogenicity. Its immunogenicity was evaluated in mice following intranasal immunization. Antigen-specific antibody responses were measured in serum and bronchoalveolar lavage fluid. T-cell responses were evaluated in lungs and spleens. Protective efficacy was assessed using SARS-CoV-2-susceptible hACE2 knock-in mice. Results: Ag-specific Ab levels increased in both serum and bronchoalveolar lavage fluid of mice immunized intranasally with the conjugate. Especially, T-cell responses were significantly enhanced in the lungs and spleens of immunized hACE2 knock-in mice. In challenge experiments, intranasal administration of the conjugate reduced viral load. Moreover, Siglec F was identified as a potential receptor for Co4B, a previously uncharacterized M cell-targeting ligand. Conclusions: A recombinant viral Ag containing Co4B and HBD2 induces virus-specific humoral and cellular immune responses. Although further optimization of the vaccine formulation and administration strategy is needed, this conjugate shows potential as a platform for improving mucosal and systemic immunity.
Full article
(This article belongs to the Special Issue Mucosal Immunity and Vaccine)
►▼
Show Figures
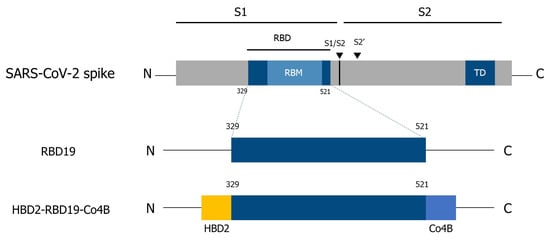
Figure 1
Open AccessReview
Perspectives on the Development of Immune Memory Associated with Vaccination
by
Pu He, Yuhe Zhou, Yu Feng, Ting Zhou, Fei Li, Youjun Mi, Wenhua Zhang and Bingdong Zhu
Vaccines 2026, 14(5), 420; https://doi.org/10.3390/vaccines14050420 - 7 May 2026
Abstract
The goal of vaccination is to induce long-term immune memory. Traditionally, immune memory has been thought to be mediated by memory T cells and B cells. In recent years, trained immunity mediated by the innate immune cells (e.g., NK cells, neutrophils, and monocytes/macrophages)
[...] Read more.
The goal of vaccination is to induce long-term immune memory. Traditionally, immune memory has been thought to be mediated by memory T cells and B cells. In recent years, trained immunity mediated by the innate immune cells (e.g., NK cells, neutrophils, and monocytes/macrophages) has garnered increasing attention. Trained immunity exhibits an antigen-nonspecific immune memory that provides broader protection against heterologous infections. This article reviews the mechanisms involved in the development of trained immunity, memory T cells, and B cells with a particular focus on metabolic reprogramming and epigenetic modifications. Moreover, the effects of aging on immune memory and the factors involved in regulating the vaccine-induced immune memory in older people are discussed. By understanding immune memory and its regulatory mechanisms, we can better design vaccines and optimize vaccination strategies to induce long-lasting immune memory.
Full article
(This article belongs to the Special Issue Memory T Cells in Vaccine-Induced Immunity for Infectious Diseases)
►▼
Show Figures
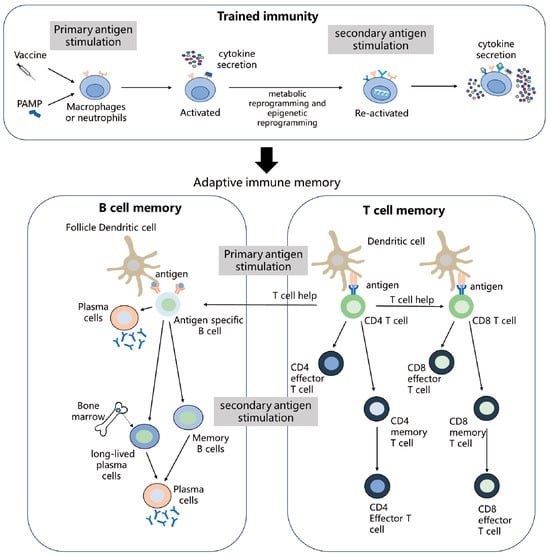
Figure 1
Open AccessSystematic Review
Vaccine-Induced Immunity in Children and Adolescents After Chemotherapy for Acute Lymphoblastic Leukemia: A Systematic Review
by
Cláudia F. C. Valente, Heloisa Ihle Garcia Giamberardino, Tânia Cristina de Mattos Barros Petraglia, Cristiane Feitosa Salviano, Priscilla Lemos Gomes, Roberia Mendonça de Pontes, Maria Eduarda Canellas de Castro, Lis R. V. Antonelli, Olindo Assis Martins-Filho, Cleandro Pires de Albuquerque and Licia Maria Henrique da Mota
Vaccines 2026, 14(5), 419; https://doi.org/10.3390/vaccines14050419 - 7 May 2026
Abstract
Background: Acute lymphoblastic leukemia is the most prevalent childhood cancer and the leading cause of cancer mortality before the age of 20. Although therapeutic advances have significantly improved survival, children and adolescents treated for acute lymphoblastic leukemia remain vulnerable to infections, largely preventable
[...] Read more.
Background: Acute lymphoblastic leukemia is the most prevalent childhood cancer and the leading cause of cancer mortality before the age of 20. Although therapeutic advances have significantly improved survival, children and adolescents treated for acute lymphoblastic leukemia remain vulnerable to infections, largely preventable by vaccination, due to humoral and cellular immune dysfunction induced by disease and treatment. Materials and Methods: This systematic review, based on electronic databases, aims to evaluate antibody levels associated with potential protective immunity against vaccine antigens for diphtheria, pertussis, tetanus, poliomyelitis, Haemophilus influenzae type b, measles, mumps, rubella, influenza, varicella-zoster virus, yellow fever, pneumococcal, and meningococcal diseases in children and adolescents treated for acute lymphoblastic leukemia after completion of chemotherapy. Results: A total of twenty-four studies published between 1981 and 2023 were included, comprising 1110 children and adolescents. Protective antibody levels ranged from 11% to 97% for diphtheria, 0% to 90% for pertussis, 20% to 100% for tetanus, and 11% to 95% for poliomyelitis. Haemophilus influenzae type b, protection ranged from 16.7% to 100%. Viral vaccines also showed heterogeneous responses, with protection rates of 25–79% for mumps, 16–86% for measles, 35–98% for rubella, and 23–75% for varicella-zoster virus. Antibody responses to pneumococcal and meningococcal vaccines were consistently low, with protection rates of 5–38% for pneumococcal studies and 12% in a single meningococcal study. Conclusions: This review found a consistent and clinically relevant loss of vaccine-induced immunity in children and adolescents treated for acute lymphoblastic leukemia. The recommendation of vaccine booster doses for this vulnerable population, irrespective of serological status, may represent a more practical approach to ensuring adequate post-chemotherapy treatment protection.
Full article
(This article belongs to the Section Epidemiology and Vaccination)
►▼
Show Figures
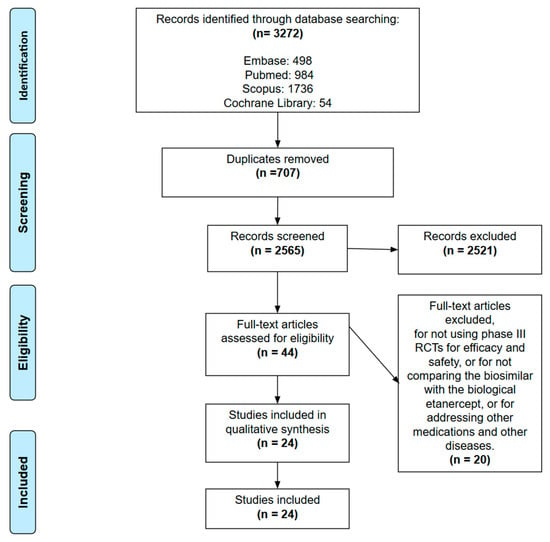
Figure 1

Journal Menu
► ▼ Journal Menu-
- Vaccines Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomolecules, Cells, IJMS, Pathogens, Vaccines, Biologics, Microorganisms, Biomedicines
Advances in Vaccines and Antimicrobial Therapy—2nd Edition
Topic Editors: Raffaele D’Amelio, Roberto PaganelliDeadline: 30 September 2026
Topic in
Animals, Arthropoda, Insects, Vaccines, Veterinary Sciences, Pathogens
Ticks and Tick-Borne Pathogens: 2nd Edition
Topic Editors: Alina Rodriguez-Mallon, Alejandro Cabezas-CruzDeadline: 31 March 2027
Topic in
Biomedicines, Cancers, IJMS, Vaccines, Viruses, JMP
Advances in HPV-Driven Head and Neck Cancer: Pathogenesis, Treatment, and Prevention
Topic Editors: Shilpi Gupta, Bhudev Chandra Das, Prabhat KumarDeadline: 20 April 2027
Topic in
Animals, Aquaculture Journal, Biology, Fishes, Pathogens, Vaccines, IJMS
Novel Insights and Advanced Research in Aquatic Animal Diseases and Immunology
Topic Editors: Erlong Wang, Zihao YuanDeadline: 30 July 2027

Conferences
Special Issues
Special Issue in
Vaccines
Swine Vaccines and Vaccination
Guest Editor: Fanzhi KongDeadline: 15 May 2026
Special Issue in
Vaccines
Vaccination and Treatments Against Viral Hepatitis: Achievements, Challenges and Perspectives, 2nd Edition
Guest Editors: Bárbara Vieira Do Lago, Vinicius MelloDeadline: 20 May 2026
Special Issue in
Vaccines
Innovative Vaccines and Technologies to Address Tropical Parasitic Diseases
Guest Editors: Cecilia Maria Perez Brandan, Iván Marcipar, Fernando Sanchez-ValdezDeadline: 30 May 2026
Special Issue in
Vaccines
Clinical Strategies to Improve Efficacy, Effectiveness, and Safety of Vaccination in Humans
Guest Editor: Kay Choong SeeDeadline: 31 May 2026
Topical Collections
Topical Collection in
Vaccines
Research on Monoclonal Antibodies and Antibody Engineering
Collection Editor: Tatsuya Yamazaki
Topical Collection in
Vaccines
Factors Associated with Vaccine Hesitancy
Collection Editor: Brian D. Poole
Topical Collection in
Vaccines
The Safety and Immunogenicity of the Bivalent Omicron-Containing mRNA-1273.214 Booster Vaccine
Collection Editors: Lalit Batra, Shailendra Kumar Verma