Journal Description
Viruses
Viruses
is a peer-reviewed, open access journal of virology, published monthly online by MDPI. The Spanish Society for Virology (SEV), Canadian Society for Virology (CSV), Italian Society for Virology (SIV-ISV), Australasian Virology Society (AVS), Brazilian Society for Virology (BSV) and Global Virus Network (GVN) are affiliated with Viruses and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, MEDLINE, PMC, Embase, PubAg, and other databases.
- Journal Rank: JCR - Q2 (Virology) / CiteScore - Q1 (Virology/Infectious Diseases)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.2 days after submission; acceptance to publication is undertaken in 2.7 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Journal Cluster of Microbiology: Acta Microbiologica Hellenica, Applied Microbiology, Bacteria, Journal of Fungi, Microorganisms, Microbiology Research, Pathogens and Viruses.
Impact Factor:
3.5 (2024);
5-Year Impact Factor:
3.7 (2024)
Latest Articles
Emerging Trends in HIV-1 Sub-Subtype A6 in Belgium: Transmission Dynamics, Drug Resistance, and Subtyping Tool Evaluation
Viruses 2026, 18(5), 554; https://doi.org/10.3390/v18050554 (registering DOI) - 12 May 2026
Abstract
The international spread of HIV-1 sub-subtype A6 raises concerns due to its association with contraindications for long-acting injectable formulations of cabotegravir (LA-CAB) and rilpivirine (LA-RPV). This study investigated its increasing proportion in Belgium, assessing transmission dynamics and potential migration links. Additionally, genotypic drug
[...] Read more.
The international spread of HIV-1 sub-subtype A6 raises concerns due to its association with contraindications for long-acting injectable formulations of cabotegravir (LA-CAB) and rilpivirine (LA-RPV). This study investigated its increasing proportion in Belgium, assessing transmission dynamics and potential migration links. Additionally, genotypic drug resistance in the Belgian HIV-1 sub-subtype A6 population were analyzed and four automatic subtyping tools were compared. A dataset of 4764 HIV-1 protease and reverse transcriptase (RT) sequences from newly diagnosed, treatment-naïve individuals in Belgium (2013–2022) was analyzed. A combination of phylogenetic analysis and online subtyping tools identified 136 sub-subtype A6 sequences. The increase in the proportion of HIV-1 sub-subtype A6 observed in Belgium since 2020 reflects changing transmission patterns, especially among Belgium-born men having sex with men, and cannot be solely linked to the recent influx of Ukrainian migrants. Of these sub-subtype A6 sequences, less than 10% showed LA-CAB + LA-RPV resistance, mainly due to E138A within RT. HIVdb and ANRS reliably assessed resistance in this therapy-naïve cohort, and HIVdb, COMET, and SmartGene® produced concordant subtyping results. While algorithm choice has little impact at low resistance prevalence, further research is necessary and HIVdb and ANRS remain more suitable for ongoing clinical and research use.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
Open AccessReview
Gut Microbiota and Probiotics in Influenza: A Narrative Review of Mechanisms and Emerging Evidence
by
Feihu Guan, Jie Zhang, Ye Tian, Bofan Fu, Ji Liu, Yafen Song, Aoyang Yan, Bing Zhang, Ling Chen, Min Zhang, Pengfei Du, Lei Wang, Xiaoyue Yang, Sifan Guo, Chenghuai Yang, Hui Zhang and Qianyi Zhang
Viruses 2026, 18(5), 553; https://doi.org/10.3390/v18050553 (registering DOI) - 12 May 2026
Abstract
The gut microbiota, often referred to as the “forgotten organ”, plays an indispensable role in maintaining host physiological metabolism, immune function, and nutrient absorption. Moreover, the gut microbiome serves as a critical biological barrier against viral infections and is increasingly recognized as a
[...] Read more.
The gut microbiota, often referred to as the “forgotten organ”, plays an indispensable role in maintaining host physiological metabolism, immune function, and nutrient absorption. Moreover, the gut microbiome serves as a critical biological barrier against viral infections and is increasingly recognized as a potential target to augment antiviral therapies. Recent studies have revealed that microbial ligands and metabolites derived from the gut microbiota are pivotal in modulating respiratory immune responses, providing compelling evidence of the complex interaction network between microorganisms and the host, particularly the signaling pathways linking the gut to distal organs such as the lungs. This review examines the communication and regulatory mechanisms between the gut microbiota and pulmonary mucosal surfaces during influenza virus infection, emphasizing how gut microbial communities and probiotics influence host immune responses, promote the production of immune-related molecules, and enhance antiviral defenses. The aim is to provide comprehensive insights into the gut–lung axis and its implications for respiratory health.
Full article
(This article belongs to the Special Issue Advances in Respiratory Viruses Research: From Basic Studies to Public Health)
Open AccessArticle
Dynamics of Paraspeckle Components in Herpes Simplex Virus 1 (HSV-1)-Infected Human Neuronal Cells
by
Carolina Filipponi, David C. Bloom, Carlo Gambotto, Callen T. Wallace, Jadranka Milosevic, Simon C. Watkins, Shane Buckley, Maribeth A. Wesesky, Vishwajit L. Nimgaonkar and Leonardo D’Aiuto
Viruses 2026, 18(5), 552; https://doi.org/10.3390/v18050552 (registering DOI) - 12 May 2026
Abstract
Paraspeckles are subnuclear ribonucleoprotein condensates that regulate host stress responses, including those triggered by viral infection. In vitro studies using non-neuronal cells have shown the involvement of specific paraspeckle components in facilitating the replication of certain viruses, including Herpes Simplex Virus 1 (HSV-1),
[...] Read more.
Paraspeckles are subnuclear ribonucleoprotein condensates that regulate host stress responses, including those triggered by viral infection. In vitro studies using non-neuronal cells have shown the involvement of specific paraspeckle components in facilitating the replication of certain viruses, including Herpes Simplex Virus 1 (HSV-1), but these processes have not been investigated in human neuronal cells, which represent a relevant target of the virus. We employed human neural precursor cells (NPCs), neurons, and brain organoids derived from hiPSCs to investigate the previously unexplored dynamics of paraspeckle components in HSV-1-infected human neuronal cells. Our results reveal cell-type-specific differences in the expression of paraspeckle genes in response to HSV-1 infection. Unlike other viruses, HSV-1 orchestrates a previously unreported redistribution of paraspeckle proteins, leading to their accumulation in viral replication compartments (VRCs). Importantly, the expression of the paraspeckle proteins NONO and SFPQ correlates with HSV-1 permissiveness in human neuronal cells and may be required to establish a nuclear environment favoring viral transcription/replication. This enhances our understanding of how stress-response pathways in cells can be exploited by viruses in a cell-type-specific manner.
Full article
(This article belongs to the Special Issue 3D Models in Viral Pathogenesis)
►▼
Show Figures
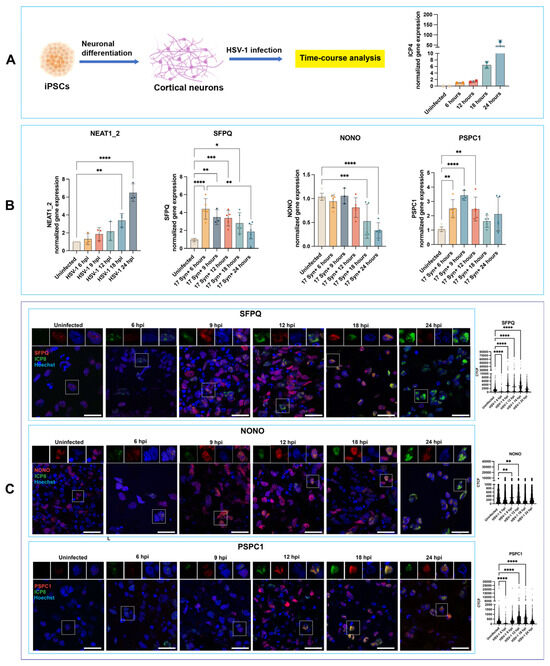
Figure 1
Open AccessEditorial
Special Issue: Microscopy Methods for Virus Research
by
Krishanu Ray
Viruses 2026, 18(5), 551; https://doi.org/10.3390/v18050551 (registering DOI) - 12 May 2026
Abstract
Significant advances in microscopy methods for biomedical applications have been made in the past two decades, and these methods are now being actively pursued for virus research [...]
Full article
(This article belongs to the Special Issue Microscopy Methods for Virus Research)
Open AccessArticle
Microglial-Derived IGF-1 Serves as a Regulator for Neuroimmune Homeostasis During Viral-Induced Demyelination
by
Vanessa M. Scarfone, Collin Pachow, Pauline U. Nguyen, Anita Lakatos, Jamie-Jean De La Torre, Alisa Xie, Kellie Fernandez, Charlene Collado, Kaitlin Murray, Roberto Tinoco, Craig M. Walsh, Trevor Owens, Agnieszka Wlodarczyk and Thomas E. Lane
Viruses 2026, 18(5), 550; https://doi.org/10.3390/v18050550 (registering DOI) - 9 May 2026
Abstract
►▼
Show Figures
This study investigated the role of microglia-derived insulin-like growth factor 1 (IGF-1) in modulating host defense and disease progression in a viral model of neuroinflammation and demyelination. Intracranial infection of susceptible mice with the glial-tropic JHM strain of mouse hepatitis virus (JHMV) induces
[...] Read more.
This study investigated the role of microglia-derived insulin-like growth factor 1 (IGF-1) in modulating host defense and disease progression in a viral model of neuroinflammation and demyelination. Intracranial infection of susceptible mice with the glial-tropic JHM strain of mouse hepatitis virus (JHMV) induces acute encephalomyelitis, followed by an immune-mediated demyelinating disease that mimics many clinical and histologic features of multiple sclerosis (MS). Utilizing an inducible fractalkine receptor (Cx3cr1) promoter-driven Cre-loxP recombinant system, we performed timed ablation of Igf1 in microglia to assess its impact on the central nervous system (CNS) response to JHMV. While the loss of microglial IGF-1 did not impair the control of viral replication, it significantly exacerbated spinal cord demyelination. CyTOF and imaging mass cytometry analysis of spinal cords indicated increased myelin damage was associated with increased accumulation of CD8+Ly6C+ effector T cells and reduced expression of TREM2 that impaired transition into a disease-associated microglia (DAM) phenotype capable of sensing and potentially mitigating myelin damage. Collectively, these findings argue that microglial IGF-1 is a non-redundant coordinator of the CNS immune responses that occur in response to CNS viral infection.
Full article
Figure 1
Open AccessArticle
Intersecting Epidemics: A Multilevel Syndemic Analysis of a Chikungunya Virus Epidemic in Colombia Through Clinical, Biological, and Socioeconomic Factors
by
Juan C. Rueda, Ana María Santos, Ignacio Angarita, Ingris Peláez-Ballesta, Alfonso Gastelum, Igor Rueda, Jaime Cortés-Ramos, Cristian Astudillo, Daniel Rincón-Sierra, Karina Guzmán, Jesús Giovanny Ballesteros, Juan Manuel Bello and John Londono
Viruses 2026, 18(5), 549; https://doi.org/10.3390/v18050549 (registering DOI) - 9 May 2026
Abstract
This study applied a syndemic framework to chikungunya virus (CHIKV) infection during the 2014–2015 Colombian epidemic, integrating biological and social determinants. Methods: A community-based cohort of 279 serologically confirmed adults from six cities was analyzed. Clinical, sociodemographic, and cytokine data were evaluated using
[...] Read more.
This study applied a syndemic framework to chikungunya virus (CHIKV) infection during the 2014–2015 Colombian epidemic, integrating biological and social determinants. Methods: A community-based cohort of 279 serologically confirmed adults from six cities was analyzed. Clinical, sociodemographic, and cytokine data were evaluated using multilevel and multivariate statistical approaches. Results: Among 279 patients, 141 (50. 5%) met World Health Organization (WHO) criteria for acute CHIKV infection. The cohort was predominantly female and of lower socioeconomic status (SES). The most frequent manifestations were arthralgia (91%), fatigue (58%), fever (50.5%), myalgia (45.9%), and rash (45.2%). Multivariate models identified IL-15, IL-17A, IL-12p40, MCP-1, and MIP-1α as significant correlates of fever, rash, and myalgia. Socioeconomic and ethnic factors influenced cytokine expression; Caucasian patients showed higher proinflammatory cytokine levels than Afro-American patients. Lower SES was associated with greater symptom burden. Network analyses revealed distinct immune signatures linking biological responses with clinical and demographic variables. Conclusion: Immune responses, clinical manifestations, and social disadvantages interact significantly in CHIKV infection. These findings support a syndemic model in which socioeconomic vulnerability amplifies disease impact, highlighting the need for integrated biosociological public health strategies, particularly targeting populations with low socioeconomic status.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
Open AccessArticle
Application of a Blood–Brain Barrier Organ-on-a-Chip Model for Assessment of Countermeasure Efficiency Against Eastern Equine Encephalitis Virus
by
Niloufar A. Boghdeh-Olson, Michael D. Barrera, Clayton M. Britt, David K. Schaffer, Jacquelyn A. Brown, John P. Wikswo and Aarthi Narayanan
Viruses 2026, 18(5), 548; https://doi.org/10.3390/v18050548 (registering DOI) - 9 May 2026
Abstract
Infection by neurotropic alphaviruses such as the Eastern equine encephalitis virus (EEEV) causes extensive inflammation in the central nervous system and tissue damage, including disruption of the blood–brain barrier (BBB). Neuroinflammation and BBB disruption following infection are critical pathological considerations for the development
[...] Read more.
Infection by neurotropic alphaviruses such as the Eastern equine encephalitis virus (EEEV) causes extensive inflammation in the central nervous system and tissue damage, including disruption of the blood–brain barrier (BBB). Neuroinflammation and BBB disruption following infection are critical pathological considerations for the development of robust countermeasure strategies. Encephalitic disease resulting from EEEV infection currently lacks FDA-approved therapeutic intervention strategies, thus exposing a major capability gap in the ability to address the global health burden that could result from alphavirus infections. In this manuscript, we present a gravity-flow Neurovascular Unit (gNVU) model of the human BBB that may be used for modeling EEEV-induced neuropathology and evaluating countermeasures. The data generated using this model show that EEEV infection causes a time-dependent disruption of BBB integrity and increases the inflammatory load in a manner that correlates with an increase in the viral load. The data also show that the route of introduction of the pathogen has an impact on the pathology measured, with infection through the brain side eliciting a greater inflammatory outcome than infection through the vascular route. Overall, the included data support the utility of this organ-on-a-chip (OOC) platform of the human BBB in understanding encephalitic disease caused by neurotropic viruses and evaluation of therapeutic intervention strategies.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
►▼
Show Figures

Figure 1
Open AccessArticle
Hypoxia-Enhanced N110 Glycosylation of Hemagglutinin Promotes H3N2 Influenza Virus Fitness by Modulating Receptor Binding and Immune Evasion
by
Ting Zhang, Yihui Fang, Jie Liu, Ao Guo, Bin Yuan, Yanan Zhang, Lihua Ding and Qinong Ye
Viruses 2026, 18(5), 547; https://doi.org/10.3390/v18050547 - 8 May 2026
Abstract
The hemagglutinin (HA) of influenza A/H3N2 virus evolves rapidly, with glycosylation driving immune evasion. However, how host microenvironmental cues influence this process remains poorly understood. We identified a novel N-linked glycosylation site at position 110 (N110) in contemporary H3N2 viruses (NSS genotype) that
[...] Read more.
The hemagglutinin (HA) of influenza A/H3N2 virus evolves rapidly, with glycosylation driving immune evasion. However, how host microenvironmental cues influence this process remains poorly understood. We identified a novel N-linked glycosylation site at position 110 (N110) in contemporary H3N2 viruses (NSS genotype) that enhances viral fitness by increasing receptor-binding signal, HA cleavage, and replication. Remarkably, hypoxia, which mimics the respiratory tract microenvironment, significantly augments N110 glycosylation. Mechanistically, we identified the B4GAT1-B4GALT1 complex as the key mediator of this modification. Hypoxia upregulates their expression and strengthens their interaction with HA. In ferret models, N110-glycosylated viruses exhibit heightened pathogenicity and evade ancestral antibodies. Furthermore, immunization with N110-containing HA confers broad-spectrum protection, whereas reciprocal immunization is ineffective. Our findings reveal hypoxia-driven glycosylation as a previously unrecognized mechanism of H3N2 adaptation, providing critical insights for vaccine efficacy and highlighting the importance of integrating microenvironmental factors into future antiviral strategies.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
Open AccessReview
NLR Inflammasomes in Viral Infections: From Molecular Mechanisms to Therapeutic Interventions
by
Shiyuan Hou, Xing Shen, Danni Sun, Yulin An, Yuxuan Zhou, Xing Sun, Shuhan Wang, Xinyue Liu, Mengting Zhu, Shuai Zhao, Ziyu Liu, Xingan Wu and Rongrong Liu
Viruses 2026, 18(5), 546; https://doi.org/10.3390/v18050546 - 8 May 2026
Abstract
The innate immune system serves as the primary barrier against viral invasion, utilizing pattern recognition receptors (PRRs) to orchestrate a rapid defense. Among these, the nucleotide-binding domain and leucine-rich repeat (NLR) containing proteins function as central signaling scaffolds, assembling into multiprotein complexes known
[...] Read more.
The innate immune system serves as the primary barrier against viral invasion, utilizing pattern recognition receptors (PRRs) to orchestrate a rapid defense. Among these, the nucleotide-binding domain and leucine-rich repeat (NLR) containing proteins function as central signaling scaffolds, assembling into multiprotein complexes known as inflammasomes. These complexes drive the maturation of pro-inflammatory cytokines IL-1β and IL-18, and initiate gasdermin D (GSDMD)-mediated pyroptosis, a lytic cell death pathway that eliminates intracellular replication niches. This comprehensive review synthesizes the diversified landscape of inflammasome activation during viral infections, extending beyond the canonical NLRP3 inflammasome to include specialized sensors such as NLRP6, NLRP9, NLRP1, NLRP12, and NLRC4. We critically evaluate the evolutionary “arms race” between host defenses and viral pathogens, detailing the sophisticated immune evasion strategies employed by viruses—ranging from the expression of decoy proteins and direct proteolytic cleavage of immune sensors to the manipulation of post-translational modifications (PTMs). Furthermore, we discuss the dual nature of inflammasome activation, which balances protective viral clearance against pathological hyperinflammation, and provide an exhaustive analysis of novel therapeutic strategies, including direct NLR inhibitors and downstream cytokine blockers, currently navigating clinical transition.
Full article
(This article belongs to the Special Issue Viral Mechanisms of Immune Evasion)
►▼
Show Figures

Figure 1
Open AccessArticle
Identification of Cyclin L1 as a Host Factor Regulating Hepatitis B Virus Replication
by
Collins Oduor Owino, Balakrishnan Chakrapani Narmada, Gian Yi Lin, Pauline Poh Kim Aw, Nivrithi Ganesh, Jovi Tan Siying, Marie-Laure Plissonnier, Thangavelu Thangavelu Matan, Niranjan Shirgaonkar, Pablo Bifani, Massimo Levrero, Giridharan Periyasamy, Seng Gee Lim and Ramanuj DasGupta
Viruses 2026, 18(5), 545; https://doi.org/10.3390/v18050545 - 8 May 2026
Abstract
Background and Aims: Understanding regulatory interactions between hepatitis B virus (HBV) and host factors is essential for the development of next generation host-directed antiviral therapies and the achievement of a functional HBV cure. Here, we investigated HBV-induced alterations in host gene expression in
[...] Read more.
Background and Aims: Understanding regulatory interactions between hepatitis B virus (HBV) and host factors is essential for the development of next generation host-directed antiviral therapies and the achievement of a functional HBV cure. Here, we investigated HBV-induced alterations in host gene expression in primary human hepatocytes (PHH) to identify host factors exploited by the virus for replication and persistence. Whole-transcriptome sequencing (WTS) of HBV-infected PHH identified host pathways with potential roles in the HBV life cycle. RNA interference-based functional screening of dysregulated candidate genes identified cyclin L1 (CCNL1) as a key host factor. RNAi-mediated knockdown of CCNL1 reduced HBV gene expression, including hepatitis B surface antigen (HBsAg). Mechanistically, CCNL1 regulates phosphorylation of the C-terminal domain (CTD) of RNA polymerase II (RNAPII) at serine 2 (S2), consistent with a role in transcriptional regulation. CCNL1 knockdown further reduced the binding of total and phospho- (Ser2/Ser5) RNAPII, pan-acetylated histone H3 (H3ac), and H3K27ac to HBV covalently closed circular DNA (cccDNA), indicating impaired cccDNA-dependent transcription. In addition, CCNL1 expression was elevated in chronic hepatitis B patients compared with those with resolved infection. Collectively, these data demonstrate that CCNL1 promotes HBV transcription and replication through modulation of RNAPII phosphorylation and chromatin-associated transcriptional activity, identifying CCNL1 as a potential host susceptibility factor for HBV. Importance: Hepatitis B virus infection remains a major threat to human health in areas with high prevalence. There is need to fully understand the complex interactions between the virus and human host factors/processes to support ongoing efforts to develop anti-HBV therapies that can be used with existing therapies to achieve a better cure. HBV relies on host cellular factors and biological processes to establish and maintain efficient infection, making host–virus interactions attractive targets for therapeutic intervention. Thus, identifying host factors that support and/or restrict HBV infection is essential for understanding the molecular basis of chronic HBV infection and for developing host-targeting anti-HBV drugs. This study identifies cyclin L1 (CCNL1) as a host susceptibility factor that promotes HBV transcription and replication through regulation of RNA polymerase II activity and or post-transcriptional mechanisms.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
►▼
Show Figures
Figure 1
Open AccessSystematic Review
Current Challenges and Potential Strategies to Enhance Efficacy of Oral Phage Therapy in Food Animals: A Systematic Review with Quantitative Analysis
by
Md Ashiqur Rahman, Rebecca Abraham, David J. Hampson, Sam Abraham and Jasim M. Uddin
Viruses 2026, 18(5), 544; https://doi.org/10.3390/v18050544 (registering DOI) - 8 May 2026
Abstract
Phage therapy has enormous potential in combating bacterial resistance in food animals. However, its application via the oral route remains limited due to challenges associated with the gastrointestinal tract (GIT) environment and a lack of rigorous clinical trial evidence. Therefore, we systematically searched
[...] Read more.
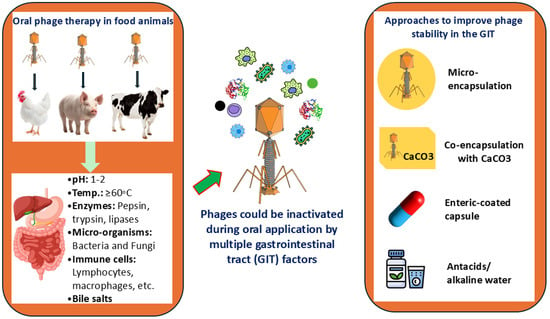
Phage therapy has enormous potential in combating bacterial resistance in food animals. However, its application via the oral route remains limited due to challenges associated with the gastrointestinal tract (GIT) environment and a lack of rigorous clinical trial evidence. Therefore, we systematically searched in Google Scholar, PubMed, Scopus, and Web of Science databases following PRISMA guidelines and finally identified 111 articles on oral phage therapy in food animals from where we summarized the key physiological and chemical factors of the gut environment hindering the effectiveness of oral phage therapy (OPT), examined the methods used to evaluate phage stability in the GI environment, and highlighted potential strategies to mitigate these challenges. In addition, we performed quantitative analysis to visualize in vitro pH and thermal stability patterns of phages targeting bacteria isolated from food animals and variability in buffer and incubation period across stability studies. The GIT consists of several anatomically and functionally distinct segments, where complex interactions occur among digestive enzymes, gastric acids, electrolytes, commensal microbiota, and mucosal immune components. The acidic pH of the stomach is a major barrier to successful oral phage delivery. According to our analysis of pH stability testing data from the reviewed studies, most phages targeting antimicrobial-resistant bacteria in food animals remained stable at pH 5–9 and inactivated under highly acidic (pH ≤ 2) or highly alkaline (pH ≥ 11) conditions. In addition, phages are susceptible to high temperatures (above 60 °C), digestive enzymes (e.g., pepsin, trypsin, lipases), bile salts, and host immune responses. Several in vitro laboratory techniques are available to assess phage stability under simulated GI conditions, but variations occur in the assessment protocols. Microencapsulation using alginate and chitosan has been used to protect phages from the adverse GI environment. Additionally, enteric-coated capsules, antacids, co-encapsulation with acid-neutralizing agents, consumption of alkaline water, and daily phage administration are suggested to improve phage survival and efficacy. For the successful clinical implementation of OPT in food animals, future research should focus on elucidating the molecular and physicochemical determinants of phage stability, understanding the humoral immune response to OPT, standardizing laboratory protocol for assessing phage viability, improving the scalability of encapsulation methods, and exploring other potential delivery techniques.
Full article
(This article belongs to the Section Bacterial Viruses)
►▼
Show Figures
Graphical abstract
Open AccessReview
Decoding the Structural Complexity of Viral RNAs with SHAPE to Guide Antiviral Therapeutics
by
Laura Broglia, Camilla Canale, Andrea Vandelli, Gian Gaetano Tartaglia and Riccardo Delli Ponti
Viruses 2026, 18(5), 543; https://doi.org/10.3390/v18050543 - 8 May 2026
Abstract
RNA viruses encode multiple layers of regulatory information within their genomes, extending beyond their protein-coding sequences. Through local secondary structures and long-range RNA–RNA interactions, viral RNAs control essential steps of the viral life cycle, including translation, replication, genome cyclization, packaging, and evasion of
[...] Read more.
RNA viruses encode multiple layers of regulatory information within their genomes, extending beyond their protein-coding sequences. Through local secondary structures and long-range RNA–RNA interactions, viral RNAs control essential steps of the viral life cycle, including translation, replication, genome cyclization, packaging, and evasion of host defenses. Over the last two decades, chemical probing approaches—particularly Selective 2′-Hydroxyl Acylation analyzed by a primer extension (SHAPE) and its high-throughput derivatives—have transformed our ability to investigate these structures at a single nucleotide resolution and on a genome-wide scale. These technologies have revealed that viral genomes are highly structured and contain numerous functional RNA elements within untranslated regions as well as coding sequences. In this review, we summarize the main experimental strategies used to profile viral RNA architecture, with a focus on SHAPE-based methodologies and complementary approaches. We then discuss the major classes of functional RNA structures identified across diverse viral families, focusing on elements involved in translation and replication, such as internal ribosome entry sites (IRES) and cyclization elements, as well as other functional structures, including XRN1-resistant and frameshifting elements. Finally, we examine how structure-guided analyses are opening new avenues for antiviral intervention, including antisense oligonucleotides, small molecules, and RNA-degrading chimeras. Together, these advances highlight the viral RNA structure as both a key determinant of virus biology and a promising target for therapeutic innovation.
Full article
(This article belongs to the Special Issue Functional Structures in RNA Viruses)
Open AccessReview
Mucosal Dynamics Contributing to Innate Immune Responses to HIV in the Human Female Genital Tract
by
Genna E. Moldovan, Gabriel P. Faber and Marta Rodriguez-Garcia
Viruses 2026, 18(5), 542; https://doi.org/10.3390/v18050542 - 8 May 2026
Abstract
HIV is primarily acquired in women at the female genital mucosa through heterosexual contact. Mucosal immune cells reside adjacent to, within, below, and distant from the epithelium that lines the surface of the female genital tract (FGT) mucosa. Innate immune cells play dual
[...] Read more.
HIV is primarily acquired in women at the female genital mucosa through heterosexual contact. Mucosal immune cells reside adjacent to, within, below, and distant from the epithelium that lines the surface of the female genital tract (FGT) mucosa. Innate immune cells play dual roles in HIV acquisition, both poised to rapidly recognize and respond to HIV, but are also capable of promoting HIV infection locally and distantly in the lymph nodes. In this review we emphasize recent human research on the roles of specific innate immune cells in HIV pathogenesis in the FGT, including dendritic cells, macrophages, neutrophils and innate lymphoid cells. We review how FGT mucosal dynamics, including anatomical compartmentalization, menstrual cycle regulation, reproductive history, menopause and chronological aging contribute to tissue conditioning of these cells and changes in HIV susceptibility in women throughout their lives.
Full article
(This article belongs to the Special Issue Viruses in the Reproductive Tract)
►▼
Show Figures
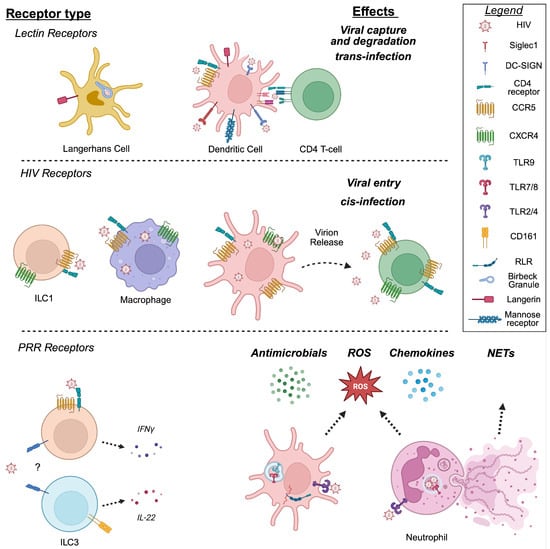
Figure 1
Open AccessReview
Evaluating the Role of Anopheles Mosquitoes in the Global Spread of Arboviruses: A Review of Laboratory-Confirmed Viral Competence
by
Rosheen Sungeni Mthawanji, Matthew Baylis, Maya Wardeh and Marcus S. C. Blagrove
Viruses 2026, 18(5), 541; https://doi.org/10.3390/v18050541 - 8 May 2026
Abstract
Mosquito-borne diseases are a major global health concern, infecting up to 700 million people annually and causing over one million deaths. Of the several genera of biting mosquitoes, species of Anopheles are mostly studied for their ability (vector competence) to transmit Plasmodium protozoan
[...] Read more.
Mosquito-borne diseases are a major global health concern, infecting up to 700 million people annually and causing over one million deaths. Of the several genera of biting mosquitoes, species of Anopheles are mostly studied for their ability (vector competence) to transmit Plasmodium protozoan parasites, some species of which cause malaria. More than 480 species of Anopheles have been described worldwide, and about 70 of these are responsible for Plasmodium spp. transmission. However, the focus on Anopheles as vectors of Plasmodium has led to a relative lack of study about the ability of Anopheles to transmit viruses. Some Anopheles species have been experimentally confirmed as competent for various arboviruses. In most cases, they are secondary vectors, with relatively low competence, contributing to overall transmission while other species of mosquito or other vectors are responsible for sustained transmission. Although secondary vectors may contribute less to transmission, they may play important epidemiological roles by extending transmission seasons and/or providing a means of overwintering viruses. Here, we conducted a review of scientific repositories to build a database of known Anopheles competence for arboviruses. After exclusions, we retained 427 laboratory-confirmed studies from 7343 papers reviewed. Our analysis suggests some Anopheles spp. could contribute to arbovirus overwintering in temperate regions.
Full article
(This article belongs to the Special Issue Current Trends in Arbovirus Outbreaks and Research)
►▼
Show Figures
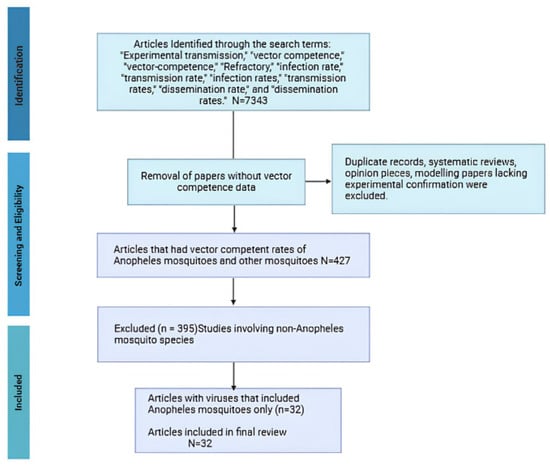
Figure 1
Open AccessReview
Temporal Evolution of Drug Resistance to HIV Integrase Inhibitors
by
Indrani Choudhuri, Jocelyn G. Olvera, Avik Biswas, Allan Haldane, Ronald M. Levy and Dmitry Lyumkis
Viruses 2026, 18(5), 540; https://doi.org/10.3390/v18050540 - 8 May 2026
Abstract
HIV-1 integrase (IN) strand transfer inhibitors (INSTIs) are central to modern antiretroviral therapy (ART) because of their high potency and durable effect on viral suppression. However, drug resistance mutations (DRMs) within HIV-1 IN emerge, which can compromise long-term treatment efficacy. Many distinct DRMs
[...] Read more.
HIV-1 integrase (IN) strand transfer inhibitors (INSTIs) are central to modern antiretroviral therapy (ART) because of their high potency and durable effect on viral suppression. However, drug resistance mutations (DRMs) within HIV-1 IN emerge, which can compromise long-term treatment efficacy. Many distinct DRMs that arise under INSTI therapy have been extensively tabulated in public repositories and literature. However, the timelines over which they emerge, accumulate, and consolidate in patients have not been systematically integrated across clinical and experimental studies. In this review, we synthesize current evidence on the temporal evolution of DRMs within HIV-1 IN by examining mutational kinetic data from viruses derived from people living with HIV/AIDS (PLWH) and from in vitro selection experiments. We compare experimental timelines to recent computational predictions derived from Potts-based fitness landscapes coupled with kinetic Monte Carlo simulations and identify reproducible kinetic classes that distinguish fast-, intermediate-, and slow-emerging DRMs. Rapidly emerging DRMs such as E92Q and N155H typically appear early under drug pressure and often represent low-barrier adaptive responses, whereas the most clinically consequential mutations, such as Q148H/K/R, G140A/S, and E138K, arise only after extended therapy and generally require compensatory mutational backgrounds to persist. Although absolute emergence times vary substantially between in vivo and in vitro systems, consistent temporal trends across datasets support the existence of underlying epistatic constraints that shape drug resistance evolution. Understanding DRM timelines is clinically relevant because it provides a framework for interpreting resistance detected at virological failure, informs optimal timing of resistance testing, and may enable earlier identification of high-risk evolutionary trajectories before durable resistance is established.
Full article
(This article belongs to the Special Issue 15-Year Anniversary of Viruses)
►▼
Show Figures
Figure 1
Open AccessArticle
Factors Associated with Antiretroviral Therapy Re-Engagement Among Men Who Have Sex with Men in South Africa: A Multi-District Analysis
by
Betty Sebati and Anthony Brown
Viruses 2026, 18(5), 539; https://doi.org/10.3390/v18050539 - 7 May 2026
Abstract
Men who have sex with men (MSM) are among of the key population groups that have been disproportionately affected by the HIV epidemic globally. Hence, MSM living with HIV may experience unique challenges leading to their disengagement from and re-engagement with care. This
[...] Read more.
Men who have sex with men (MSM) are among of the key population groups that have been disproportionately affected by the HIV epidemic globally. Hence, MSM living with HIV may experience unique challenges leading to their disengagement from and re-engagement with care. This study aimed to identify factors associated with ART re-engagement among MSM in selected districts of South African provinces. A retrospective observational study design was followed, utilising MSM routine programme data from 1 January 2018 to 31 December 2022. The programme enrolled 3337 MSM aged 16 years or older who resided in the selected provinces/districts. Descriptive statistics characterised participants’ baseline profiles. Binary logistic regression identified factors associated with re-engagement with ART. Data analysis was done using SPSS version 31.0.1.0; p < 0.05 was considered statistically significant. The district was the only statistically significant predictor of re-engagement among MSM, wherein eThekwini district had lower odds of re-engagement (aOR = 0.248, 95% CI: 0.144–0.428, p < 0.001). This represented a 75% reduction in the likelihood of re-engagement compared to the City of Tshwane. There was no association between being re-initiated on ART and HIV testing modality (aOR = 7.299, 95% CI: 0.567–94.037, p = 0.127). Future studies should longitudinally and qualitatively investigate specific programme and contextual factors driving district-level variation in re-engagement, while incorporating individual-level factors.
Full article
(This article belongs to the Special Issue Advances in Research on HIV Drug Resistance and Other Determinants of Treatment Success: 3rd Edition)
Open AccessArticle
A Prolonged Norovirus Infection and the Molecular Evolution of Human Norovirus Within-Host in a Child with Burkitt Lymphoma
by
Liping Jia, Ri De, Zeng Li, Zhenzhi Han, Liying Liu, Huijin Dong, Shunqiao Feng, Rong Liu and Linqing Zhao
Viruses 2026, 18(5), 538; https://doi.org/10.3390/v18050538 - 7 May 2026
Abstract
It has been reported that chronic infection of human norovirus (HuNoV) may potentially serve as a reservoir for viral variants with the possibility to evade population immunity or alter the binding sites of HBGA receptors. In this study, a child diagnosed with Burkitt
[...] Read more.
It has been reported that chronic infection of human norovirus (HuNoV) may potentially serve as a reservoir for viral variants with the possibility to evade population immunity or alter the binding sites of HBGA receptors. In this study, a child diagnosed with Burkitt lymphoma and positive for HuNoV determined by real-time PCR (qPCR) firstly in 15 August 2016, was followed up until 20 March 2018, and 26 fecal specimens and one vomitus were collected to trace the evolutionary characteristics of HuNoV by phylogenetic analysis, meta-genomics next-generation sequencing (mNGS), and temporal evolutionary analysis of VP1 among 23 specimens positive for HuNoV. There were 15 specimens with partial RdRp gene sequences forming an independent cluster with sequences of GII.P31, 14 with the region C sequences and 11 with P domain sequences of VP1 gene clustered together with HuNoV GII.4 Sydney_2012. All these sequences showed that mutations accumulated nearly in a time order, and more mutations were shown in the key epitopes A–E or near the binding sites for HBGA in subdomain P2 with higher evolutionary rates. Analysis of NGS data identified intra-host viral quasi-species, and two genome sequences of the same length from mNGS were assembled from N705, with mutations located in the region of subdomain P2 (1171 nt–1202 nt) which led to five amino acid mutations. In conclusion, the accumulated mutations of HuNoV, especially in subdomain P2, were explored in a child with Burkitt lymphoma, and the sequencing of HuNoV from immunocompromised individuals was proven critical for monitoring intra-host quasi-species evolution and potential variant emergence, providing basic data for clinical infection control.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
►▼
Show Figures
Figure 1
Open AccessArticle
Evaluation of Antigen Productivity and Inactivation Kinetics of a Recombinant Foot-and-Mouth Disease SAT1 Vaccine Strain
by
Jae Young Kim, Sun Young Park, Gyeongmin Lee, Seung-A Hwangbo, Giyoun Cho, Jong-Hyeon Park and Young-Joon Ko
Viruses 2026, 18(5), 537; https://doi.org/10.3390/v18050537 - 6 May 2026
Abstract
The Republic of Korea has implemented routine vaccination against foot-and-mouth disease virus (FMDV) in livestock using a bivalent vaccine comprising serotypes O and A following the massive FMD outbreak in 2010, while antigens for the remaining serotypes are maintained in overseas antigen banks.
[...] Read more.
The Republic of Korea has implemented routine vaccination against foot-and-mouth disease virus (FMDV) in livestock using a bivalent vaccine comprising serotypes O and A following the massive FMD outbreak in 2010, while antigens for the remaining serotypes are maintained in overseas antigen banks. The recent geographic expansion of FMDV Southern African Territories 1 (SAT1) beyond Africa underscores the need for enhanced preparedness in previously unaffected regions. In this study, we evaluated the SAT1 BOT-R strain as a candidate vaccine seed for potential domestic vaccine production by optimizing antigen production conditions, assessing scalability, determining virus inactivation parameters, and examining immunogenicity in pigs. Optimal antigen yield was achieved at 20 h−24 h post infection with a multiplicity of infection of 0.005−0.01, with production remaining stable under mildly alkaline conditions. Antigen productivity was consistently maintained during scale-up from shake flasks to a bioreactor, yielding up to 9.5 μg/mL. Complete virus inactivation was achieved using binary ethylenimine at 2 mM for 24 h at 26 °C. Vaccines formulated from both flask- and bioreactor-derived antigens elicited comparable neutralizing antibody responses in pigs, reaching a median titer of 1:500 following booster immunization. Collectively, these findings demonstrate that the SAT1 BOT-R strain is a viable and scalable candidate for SAT1 antigen banking and future domestic vaccine production, providing a practical framework for strengthening national preparedness against potential incursions of FMDV SAT1.
Full article
(This article belongs to the Section Viral Immunology, Vaccines, and Antivirals)
►▼
Show Figures
Figure 1
Open AccessArticle
Efficient Plasmid-Based Rescue of T7 RNA Polymerase-Driven Calicivirus Reverse Genetics Systems in Mammalian Cells Using Vaccinia Virus RNA-Capping Enzymes
by
Frazer J. T. Buchanan, Markella Loi, Charlotte Chim, ShuXian Zhou, Rebekah Penrice-Randal, Leandro X. Neves, Maximilian Erdmann and Edward Emmott
Viruses 2026, 18(5), 536; https://doi.org/10.3390/v18050536 - 4 May 2026
Abstract
The caliciviruses include important human and animal pathogens such as norovirus, sapovirus and feline calicivirus. Viral reverse genetics is performed to understand the fundamental biology of these viruses, as well as a potential route to generate live-attenuated vaccines. Calicivirus reverse genetics systems have
[...] Read more.
The caliciviruses include important human and animal pathogens such as norovirus, sapovirus and feline calicivirus. Viral reverse genetics is performed to understand the fundamental biology of these viruses, as well as a potential route to generate live-attenuated vaccines. Calicivirus reverse genetics systems have typically relied on either the production of in vitro-transcribed RNA or plasmid-based rescue, either from a mammalian promoter or through supplementing with helper enzymes through means of a helper virus. Here, we present a novel system integrating vaccinia capping enzymes D1R and D12L encoded on plasmids as part of a system for murine norovirus (MNV) reverse genetics. The addition of D1R, D12L and T7 RNA polymerase-expressing plasmids increases the viral titres of rescued MNV in both BSR-T7 cells and transgenic BSR-T7 cells expressing murine CD300LF (BSR-T7CD300LF), and viral protein abundance. When the murine norovirus receptor is expressed in BSR-T7CD300LF, viral titres increased 100–1000-fold compared to standard BSR-T7 cells. This system offers a robust, increased throughput means of assessing viral mutants over parallel in vitro transcription and capping reactions for multiple mutants, without requiring a helper virus.
Full article
(This article belongs to the Section General Virology)
►▼
Show Figures
Figure 1
Open AccessReview
Bat-Borne Viruses and Pandemic Risk: Could Europe Be an Emergence Hotspot?
by
Krzysztof Skowron, Justyna Bauza-Kaszewska, Anna Budzyńska, Natalia Wiktorczyk-Kapischke, Julia Czuba, Ewa Wałecka-Zacharska, Kacper Wnuk, Mariusz Zapadka, Krzysztof Kasprzyk and Katarzyna Grudlewska-Buda
Viruses 2026, 18(5), 535; https://doi.org/10.3390/v18050535 - 2 May 2026
Abstract
The recent SARS-CoV-2 pandemic—which had significant worldwide health, economic, and other effects—indicated the need to monitor zoonotic viruses with pandemic potential. The aim of this review is to assess bat-borne viruses as a potential pandemic risk, with a particular focus on Europe. The
[...] Read more.
The recent SARS-CoV-2 pandemic—which had significant worldwide health, economic, and other effects—indicated the need to monitor zoonotic viruses with pandemic potential. The aim of this review is to assess bat-borne viruses as a potential pandemic risk, with a particular focus on Europe. The presence and activity of bats, as well as diseases emerging in humans in various regions of the world, point to their importance in the context of a possible outbreak of future epidemics. The rate of genetic change observed among viruses requires constant scrutiny on all continents, including Europe. Bats are a considerable source of many zoonotic viruses, including coronaviruses, filoviruses and paramyxoviruses. Among viruses associated with bats, RNA viruses are the dominant ones, characterized by high pathogenicity and often leading to interspecies transmission. The majority (about 80%) of RNA viruses were identified in bats from three families: Vespertilionidae, Rhinolophidae and Pteropodidae. Understanding how viruses are transmitted in the environment and the role of reservoir organisms and intermediate hosts is crucial to determining the level of epidemic risk. This review discuses viruses identified in bats globally, with a special focus on Europe, and evaluates their potential to cause epidemics.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Viruses Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cancers, IJMS, Vaccines, Viruses, JMP
Advances in HPV-Driven Head and Neck Cancer: Pathogenesis, Treatment, and Prevention
Topic Editors: Shilpi Gupta, Bhudev Chandra Das, Prabhat KumarDeadline: 20 April 2027
Topic in
Antibiotics, Diseases, Vaccines, Viruses, Epidemiologia
HIV Prevention and Treatment: A Guide to Health and Safety
Topic Editors: Olanrewaju Oladimeji, Martha ChadyiwaDeadline: 31 December 2027

Conferences
Special Issues
Special Issue in
Viruses
Opportunistic Viral Infections, 3rd Edition
Guest Editor: Carlo ContiniDeadline: 15 May 2026
Special Issue in
Viruses
Virus Infections and Host Metabolism 2026
Guest Editors: Kuan Rong Chan, Yaw Shin Ooi, Liang CuiDeadline: 15 May 2026
Special Issue in
Viruses
Viral RNA and Its Interaction with the Host
Guest Editors: Redmond Smyth, Neva CaliskanDeadline: 15 May 2026
Special Issue in
Viruses
Multiscale Modeling and Forecasting of COVID-19 and Respiratory Virus Dynamics
Guest Editors: Gerardo Chowell, Sunmi LeeDeadline: 15 May 2026
Topical Collections
Topical Collection in
Viruses
Mathematical Modeling of Viral Infection
Collection Editors: Amber M. Smith, Ruian Ke
Topical Collection in
Viruses
Efficacy and Safety of Antiviral Therapy
Collection Editors: Giordano Madeddu, Andrea De Vito, Agnese Colpani
Topical Collection in
Viruses
Phage Therapy
Collection Editors: Nina Chanishvili, Jean-Paul Pirnay, Mikael Skurnik









