Journal Description
Journal of Personalized Medicine
Journal of Personalized Medicine
is an international, peer-reviewed, open access journal on personalized medicine, published monthly online by MDPI. The Inter-American Society for Minimally Invasive Spine Surgery (SICCMI), Korean Society of Brain Neuromodulation Therapy (KBNT), American Board of Precision Medicine (ABOPM) and Brazilian Society of Personalized Medicine (SBMP) are affiliated with JPM and their members receive a discount on article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, PubMed, PMC, Embase, and other databases.
- Journal Rank: CiteScore - Q1 (Medicine (miscellaneous))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 25 days after submission; acceptance to publication is undertaken in 5.8 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Latest Articles
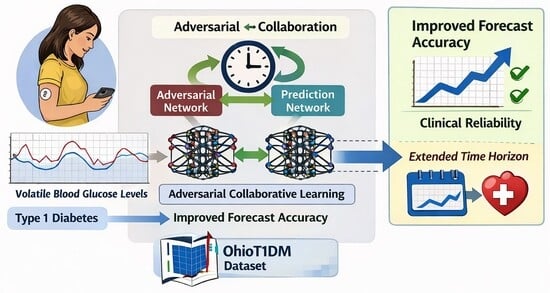
Personalised Blood Glucose Time Series Forecasting in Type 1 Diabetes: Deep Collaborative Adversarial Learning
J. Pers. Med. 2026, 16(4), 210; https://doi.org/10.3390/jpm16040210 - 8 Apr 2026
Abstract
Background/Objectives: Blood glucose prediction (BGP) for individuals with type 1 diabetes (T1D) is a clinically essential yet highly challenging task in time series forecasting (TSF) and an important problem in personalised medicine. Accurate bespoke BGP is crucial for individualised T1D management, reducing complications,
[...] Read more.
Background/Objectives: Blood glucose prediction (BGP) for individuals with type 1 diabetes (T1D) is a clinically essential yet highly challenging task in time series forecasting (TSF) and an important problem in personalised medicine. Accurate bespoke BGP is crucial for individualised T1D management, reducing complications, and supporting patient-specific glycaemic risk mitigation. However, the pronounced volatility of glycaemic fluctuations in T1D, combined with the need for mathematical rigor and clinical relevance, hampers reliable prediction. This complexity underscores the demand to explore and enhance more advanced techniques. While adversarial learning is adept at modelling intricate data variability, its potential for BGP remains largely untapped. Methods: This work presents a novel approach for BGP by addressing a key limitation in conventional adversarial learning when applied to this task. Typically, these methods optimise prediction accuracy within a set horizon by minimising adversarial loss. This focus overlooks how predictions align with longer-term patterns, which are critical for clinical relevance in BGP, thereby yielding suboptimal results. To overcome this limitation, we introduce collaborative augmented adversarial learning, designed to improve the model’s temporal awareness. Incorporating collaborative interaction optimisation, this approach enables the model to reflect extended time dependencies beyond the immediate horizon, thereby improving both the clinical reliability of predictions and overall predictive performance. We develop and evaluate four learning systems for BGP: independent learning, adversarial learning, collaborative learning, and adversarial collaborative learning. The proposed systems were evaluated for two clinically relevant prediction horizons, namely 30 min and 60 min ahead. Results: The interdependent collaboratively augmented learning frameworks, validated using the well-established Ohio T1D datasets, demonstrate statistically significant superior performance in both clinical and mathematical evaluations. Conclusions: Beyond advancing BGP accuracy and clinical reliability, the proposed approach supports personalised medicine by improving subject-specific glucose forecasting from CGM data, with potential relevance for more individualised diabetes monitoring and decision support. The proposed approach also opens new avenues for advancements in other complex TSF domains, as outlined in our future work.
Full article
(This article belongs to the Special Issue Integrating Mathematical Modeling and Data Analysis in Personalized Medical Research)
►
Show Figures
Open AccessArticle
In Silico Psycho-Oncology: Understanding Resilience Pathways in Breast Cancer—Determinants of Longitudinal Depression and Quality-of-Life Trajectories
by
Eleni Kolokotroni, Paula Poikonen-Saksela, Ruth Pat-Horenczyk, Berta Sousa, Albino J. Oliveira-Maia, Ketti Mazzocco, Haridimos Kondylakis and Georgios S. Stamatakos
J. Pers. Med. 2026, 16(4), 209; https://doi.org/10.3390/jpm16040209 - 7 Apr 2026
Abstract
Background/Objectives: Patients with breast cancer show substantial heterogeneity in terms of psychological adjustment following diagnosis. We aimed to characterize longitudinal trajectories of quality of life (QoL) and depressive symptoms during the first 18 months post-diagnosis and to identify robust clinical, psychosocial, and behavioral
[...] Read more.
Background/Objectives: Patients with breast cancer show substantial heterogeneity in terms of psychological adjustment following diagnosis. We aimed to characterize longitudinal trajectories of quality of life (QoL) and depressive symptoms during the first 18 months post-diagnosis and to identify robust clinical, psychosocial, and behavioral predictors associated with distinct adjustment pathways. Methods: Women (N = 538; mean age 55.4 years; range 40–70) with operable breast cancer (stages I–III) were drawn from the multicenter BOUNCE cohort. QoL (Global Health Status/QoL scale of the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30) and depressive symptoms (depression subscale of the Hospital Anxiety and Depression Scale) were assessed at baseline and months 3, 6, 9, 12, 15 and 18. Latent class growth analysis and growth mixture modeling identified distinct trajectory classes. Associations between early predictors and trajectory membership were examined using logistic regression combined with elastic net regularization. Results: Depression trajectories demonstrated heterogeneity, with groups characterized by persistent resilience (59.7%), stable moderate/high (25.3%), delayed onset (5.0%), and recovery (10.0%). QoL trajectories ranged from stable excellent (13.2%) and stable high (40.7%) to moderate (31.4%) and persistent low/deteriorating (6.9%), as well as a distinct recovering trajectory (7.8%). Trajectory differentiation was primarily driven by psychological resources, symptom burden, functional status, and coping processes, alongside specific contributions from clinical factors. Conclusions: Distinct subgroups of women with breast cancer follow divergent adjustment pathways. These findings highlight the multidimensional nature of resilience and support the need for tailored interventions that promote long-term well-being beyond simple risk reduction.
Full article
(This article belongs to the Special Issue Personalized Medicine for Clinical Psychology)
►▼
Show Figures

Figure 1
Open AccessReview
Extracellular Vesicles in Osteonecrosis of the Femoral Head: An Integrated Review of Experimental and Bioinformatic Evidence
by
Elvira Immacolata Parrotta, Giorgia Lucia Benedetto, Giovanni Cuda, Umile Giuseppe Longo, Arianna Carnevale, Olimpio Galasso, Giorgio Gasparini and Michele Mercurio
J. Pers. Med. 2026, 16(4), 208; https://doi.org/10.3390/jpm16040208 - 7 Apr 2026
Abstract
Background/Objectives: Osteonecrosis of the femoral head (ONFH) is a progressive condition characterized by bone necrosis, impaired vascularization, and immune dysregulation, often resulting in femoral head collapse. Effective strategies to halt disease progression are limited. Extracellular vesicles (EVs), including exosomes and microvesicles, mediate intercellular
[...] Read more.
Background/Objectives: Osteonecrosis of the femoral head (ONFH) is a progressive condition characterized by bone necrosis, impaired vascularization, and immune dysregulation, often resulting in femoral head collapse. Effective strategies to halt disease progression are limited. Extracellular vesicles (EVs), including exosomes and microvesicles, mediate intercellular communication and influence osteogenesis, angiogenesis, and immune responses. This review summarizes current evidence on EVs in ONFH and their translational potential. Methods: A structured narrative review of PubMed, Scopus, Web of Science, and Cochrane Central databases was conducted, including in vitro, preclinical, and clinical studies on EVs in ONFH. Data on EV sources, molecular cargo, signaling pathways, functional effects, and translational implications were qualitatively synthesized. No pooled statistical analysis was performed because the extracted data were heterogeneous. Bioinformatic analyses such as Gene Ontology, KEGG enrichment, and protein–protein interaction networks were also summarized. Results: In vitro, EVs from bone marrow mesenchymal stem cells, endothelial cells, and M2 macrophages modulate osteogenic differentiation, angiogenesis, and inflammation. Preclinical studies demonstrate that EV administration reduces femoral head necrosis, improves trabecular structure, and enhances neovascularization. Clinical studies have identified EV-associated molecules (SAA1, C4A, RPS8) linked to disease stage and the risk of femoral head collapse. Bioinformatic analyses connect EV cargo to pathways regulating bone formation, vascularization, immunity, and metabolism. Conclusions: EVs appear to play key roles in ONFH pathogenesis and may represent promising candidates for diagnostic and therapeutic applications. However, current clinical evidence remains limited and requires validation in larger studies. Nonetheless, heterogeneity and limited clinical data require standardized, longitudinal studies to validate their translational relevance.
Full article
(This article belongs to the Special Issue Personalized Medicine in Orthopedics and Traumatology: Updates and Challenges)
►▼
Show Figures

Figure 1
Open AccessStudy Protocol
Rationale and Design of a Randomised Proof-of-Concept Trial to Assess the Safety of Early Discharge Using Index Microcirculatory Resistance in Patients with Acute Myocardial Infarction: SECURE Study
by
Muntaser Omari, Mohamed Ali, Luke Spray, Adam McDiarmid and Mohammad Alkhalil
J. Pers. Med. 2026, 16(4), 207; https://doi.org/10.3390/jpm16040207 - 7 Apr 2026
Abstract
Background: Current guidelines acknowledge that early discharge is not associated with late mortality and that in-hospital length of stay (LOS) of 48–72 h should be considered following successful primary percutaneous coronary intervention (PPCI) in low-risk patients. Recent studies have highlighted the safety
[...] Read more.
Background: Current guidelines acknowledge that early discharge is not associated with late mortality and that in-hospital length of stay (LOS) of 48–72 h should be considered following successful primary percutaneous coronary intervention (PPCI) in low-risk patients. Recent studies have highlighted the safety of very early discharge after PPCI in highly selected low-risk patients; however, objective tools to guide discharge timing remain limited. The Index of Microcirculatory Resistance (IMR) offers a quantitative assessment of microvascular function and may help identify patients suitable for very early discharge. We aimed to evaluate the feasibility of using IMR to guide very early discharge in patients who underwent uncomplicated PPCI. Study design and objectives: The Safety of Early Discharge Using Index Microcirculatory Resistance in Patients with Acute Myocardial Infarction (SECURE) study is designed to assess the feasibility of using IMR, measured immediately following successful PPCI, to guide early discharge from hospital within 24 h. The SECURE study is a prospective, proof-of-concept, functional non-inferiority, single-centre, randomised, open-label trial to determine if patients with low IMR can be safely discharged when compared to standard discharge policy. The SECURE study will recruit 82 patients with low IMR following successful PPCI. Participants will be 1:1 randomised to either standard discharge timing or very early discharge (within 24 h). The left ventricle ejection fraction will be assessed using cardiac magnetic resonance imaging. A telephone follow-up at 3 months will be arranged. Clinical events are collected as secondary and exploratory safety endpoints. Conclusions: The SECURE study will provide proof-of-concept data about the feasibility of using IMR to guide very early discharge following PPCI. If successful, this study will provide data to plan for a larger study to determine the safety of this personalised approach.
Full article
(This article belongs to the Special Issue New Perspectives and Current Challenges in Myocardial Infarction)
►▼
Show Figures

Figure 1
Open AccessArticle
An Assessment of GPT-3.5 and GPT-4.0 Responses to Scoliosis FAQs
by
Tu-Lan Vu-Han, Enikö Regényi, Vikram Sunkara, Paul Köhli, Friederike Schömig, Alexander P. Hughes, Michael Putzier, Matthias Pumberger and Thilo Khakzad
J. Pers. Med. 2026, 16(4), 206; https://doi.org/10.3390/jpm16040206 - 7 Apr 2026
Abstract
Background: ChatGPT is a large language model (LLM) online chatbot developed by OpenAI and launched in November 2022. Early adoption studies have shown high readiness to use this technology for health-related questions and self-diagnosis. However, the quality and clinical adequacy of health-related
[...] Read more.
Background: ChatGPT is a large language model (LLM) online chatbot developed by OpenAI and launched in November 2022. Early adoption studies have shown high readiness to use this technology for health-related questions and self-diagnosis. However, the quality and clinical adequacy of health-related responses remain incompletely characterized. This study aimed to explore responses generated by ChatGPT-3.5 and ChatGPT-4.0 to common patient questions regarding scoliosis. Methods: Ten scoliosis-related frequently asked questions (FAQs) were selected from a larger pool of over 250 patient-facing questions compiled from 17 publicly available FAQ webpages and informed by a Google Trends analysis. Questions were harmonized, grouped by theme, and then reduced by rule-based expert review to a final set intended to represent common patient concerns. Results: The median ratings of ChatGPT-3.5 and ChatGPT-4.0 responses ranged from satisfactory, requiring minimal (2) to moderate clarification (3). Across the ten matched questions, no statistically detectable difference was found between models in this study setting (W = 8.0, p = 0.59; Cliff’s δ = −0.12 [95% CI −0.58, 0.40]); however, given the small question set, unblinded rating process, and poor inter-rater reliability, this should not be interpreted as evidence of equivalence, non-inferiority, or comparable model performance. The results apply only to the 10–15 April 2024, online snapshots of ChatGPT-3.5 and ChatGPT-4.0 and should not be generalized to later model iterations. Conclusions: This study should be interpreted as a clinically oriented observational report, intended to inform physician awareness and patient-physician communication rather than validate chatbot accuracy or safety. In this 10–15 April 2024, sample, both model outputs frequently required clinician clarification. Given the small FAQ set, low inter-rater reliability, unblinded design, and single-sample outputs, the findings do not establish equivalence or superiority and apply only to the specific 10–15 April 2024, model snapshots and evaluated questions.
Full article
(This article belongs to the Special Issue AI and Precision Medicine: Innovations and Applications)
►▼
Show Figures

Figure 1
Open AccessArticle
Personalized Prediction of Postoperative Recurrence in Lung Squamous Cell Carcinoma: Integrating AI-Based Nuclear Morphometry and Clinical Data
by
Tomokazu Omori, Akira Saito, Yoshihisa Shimada, Yujin Kudo, Jun Matsubayashi, Toshitaka Nagao, Masahiko Kuroda and Norihiko Ikeda
J. Pers. Med. 2026, 16(4), 205; https://doi.org/10.3390/jpm16040205 - 6 Apr 2026
Abstract
►▼
Show Figures
Background: This study employed artificial intelligence (AI) to analyze quantitative nuclear morphological features obtained from digital pathology images to predict postoperative recurrence in patients with lung squamous cell carcinoma (LSQCC). We aimed to develop a prediction model that contributes to the realization of
[...] Read more.
Background: This study employed artificial intelligence (AI) to analyze quantitative nuclear morphological features obtained from digital pathology images to predict postoperative recurrence in patients with lung squamous cell carcinoma (LSQCC). We aimed to develop a prediction model that contributes to the realization of ‘personalized postoperative management’ tailored to individual tumor biology by integrating AI-extracted morphological features with clinical information. Methods: A total of 185 of the 253 surgically resected LSQCC cases were included; 136 were randomly assigned to the training set and 49 to the test set. Nuclear features from manually selected regions of interest were extracted and used to build AI-based prediction models. Three recurrence models were developed: recurrence within 2 years, within 5 years, and a three-category model (≤2 years, 3–5 years, >5 years or no recurrence). Support vector machine (SVM) and random forest (RF) algorithms were applied to each, yielding six predictive models. An ensemble approach was used to calculate AI-based risk scores, and a “total risk score” was developed by integrating these with the pathologic stage. Results: All six AI models demonstrated stable predictive performance, with AUC values ranging from 0.76 to 0.91. Kaplan–Meier analysis showed that the total risk score provided the most precise risk stratification (p < 0.005), with clearer separation between risk groups than the AI-based risk score alone. Conclusions: The integration of AI-based nuclear morphology analysis and clinical data provides an objective and practical tool for personalized postoperative management in LSQCC. This approach enables tailored clinical decision-making by identifying patients at high risk for early recurrence and customizing postoperative treatment plans to meet the specific needs of each individual.
Full article

Graphical abstract
Open AccessArticle
Does Provider Identity at Triage Improve Machine Learning Prediction of Hospital Admission? A Comparative Analysis of Ten Supervised Classifiers with SHAP Explainability
by
Adam E. Brown, Chance W. Marostica and Wayne A. Martini
J. Pers. Med. 2026, 16(4), 204; https://doi.org/10.3390/jpm16040204 - 5 Apr 2026
Abstract
Background/Objectives: Machine learning (ML) models can predict hospital admission from emergency department (ED) triage data with areas under the receiver operating characteristic curve (AUC) exceeding 0.85. Whether incorporating the assigned provider’s identity—as a proxy for unmeasured practice variation—improves prediction has not been systematically
[...] Read more.
Background/Objectives: Machine learning (ML) models can predict hospital admission from emergency department (ED) triage data with areas under the receiver operating characteristic curve (AUC) exceeding 0.85. Whether incorporating the assigned provider’s identity—as a proxy for unmeasured practice variation—improves prediction has not been systematically studied. We aimed to compare 10 supervised ML classifiers for predicting hospital admission at ED triage, with and without provider identity, and to characterize model reasoning using SHapley Additive exPlanations (SHAP). Methods: We conducted a retrospective cohort study of 186,094 ED visits (2020–2023, training) and 58,151 visits (2024, temporal holdout test) at one academic tertiary-care ED. Ten classifiers spanning linear, distance-based, tree-based, ensemble, probabilistic, and neural network families were each trained in two conditions: baseline (23 triage features) and with provider identity appended. SHAP TreeExplainer was applied to the top-performing models (CatBoost and XGBoost). Results: The admission rate was 31.3% (training) and 31.7% (test). CatBoost achieved the highest baseline AUC of 0.8906 (0.8878–0.8933). Adding provider identity produced negligible AUC changes across all models (ΔAUC range: −0.0029 to +0.0015; all DeLong p > 0.05). SHAP analysis identified ESI level, respiratory rate, temperature, complaint category, and age as the dominant predictors, with clinically intuitive directionality. Conclusions: Provider identity does not meaningfully improve ML prediction of hospital admission beyond standard triage variables. The observed 28-percentage-point variation in provider admission rates is explained by patient case-mix differences than with independent practice pattern effects on prediction. SHAP provides transparent, clinically interpretable explanations suitable for bedside decision support.
Full article
(This article belongs to the Special Issue AI and Precision Medicine: Innovations and Applications)
►▼
Show Figures

Figure 1
Open AccessArticle
Soluble Isoforms of PD-1 and PD-L1 in Non-Small Cell Lung Cancer: Correlation with Tumor Stage, Longitudinal Analysis and Prognostic Implications
by
Konstantinos Vachlas, Dimitra Grapsa, Stylianos Gaitanakis, Anna Papadopoulou, Paraskevi Moutsatsou, Nikolaos Syrigos and Ioannis P. Trontzas
J. Pers. Med. 2026, 16(4), 203; https://doi.org/10.3390/jpm16040203 - 4 Apr 2026
Abstract
Background: Soluble immune checkpoint molecules, including soluble PD-1 (sPD-1) and soluble PD-L1 (sPD-L1), have emerged as potential minimally invasive biomarkers in non-small cell lung cancer (NSCLC). However, their diagnostic, kinetic, and prognostic significance across different disease settings remains unclear. This prospective study evaluated
[...] Read more.
Background: Soluble immune checkpoint molecules, including soluble PD-1 (sPD-1) and soluble PD-L1 (sPD-L1), have emerged as potential minimally invasive biomarkers in non-small cell lung cancer (NSCLC). However, their diagnostic, kinetic, and prognostic significance across different disease settings remains unclear. This prospective study evaluated baseline levels, longitudinal fluctuations, and clinical associations of sPD-1 and sPD-L1 in early- and advanced-stage NSCLC. Methods: Three cohorts were prospectively enrolled: early-stage NSCLC patients undergoing curative surgery (n = 25), advanced-stage NSCLC patients receiving pembrolizumab-based immunotherapy (n = 55), and non-oncological controls (n = 16). Serum sPD-1 and sPD-L1 were measured by ELISA at baseline and at four months post-surgery (early stage) or six months post-treatment (advanced stage). Baseline comparisons, longitudinal changes, correlation with tumor PD-L1 expression (TPS), and associations with recurrence (early stage) or 6-month objective response (advanced stage) were assessed. Results: Baseline sPD-1 and sPD-L1 levels did not differ significantly among controls, early-stage, and advanced-stage cohorts. In early-stage patients, sPD-L1 increased post-operatively (p = 0.006) while sPD-1 decreased (p < 0.001). In advanced-stage disease, sPD-1 declined during immunotherapy (p < 0.001), whereas sPD-L1 remained unchanged (p = 0.37). Baseline levels and continuous percent changes were not predictive of most outcomes. However, a ≥20% postoperative increase in sPD-L1 was strongly associated with recurrence in early-stage NSCLC (OR = 10.29; 95% CI: 1.40–215.20; p = 0.019). No sPD-1/PD-L1 metric predicted response in advanced disease. Baseline sPD-L1 showed no correlation with tumor PD-L1 expression (ρ = −0.09, p = 0.53) in the advanced-stage cohort. Conclusions: sPD-1 and sPD-L1 demonstrate distinct kinetic patterns across NSCLC settings. A postoperative >20% surge in sPD-L1 may identify early-stage patients at elevated risk of recurrence, whereas soluble checkpoints were not predictive of treatment response in advanced disease. These findings support further investigation of soluble checkpoint dynamics as complementary biomarkers in NSCLC management in larger cohorts.
Full article
(This article belongs to the Special Issue From Biomarkers to Breakthroughs: Advancing Lung Cancer Research)
►▼
Show Figures

Figure 1
Open AccessReview
Integrating AI Segmentation, Simulated Digital Twins, and Extended Reality into Medical Education: A Narrative Technical Review and Proof-of-Concept Case Study
by
Parhesh Kumar, Ingharan Siddarthan, Catharine Kelsh Keim, Daniel K. Cho, John E. Rubin, Robert S. White and Rohan Jotwani
J. Pers. Med. 2026, 16(4), 202; https://doi.org/10.3390/jpm16040202 - 3 Apr 2026
Abstract
Background/Objectives: Simulation digital twins (DT) models that integrate patient-specific imaging with artificial intelligence (AI)-based segmentation and extended reality (XR) technologies are rapidly increasing in relevance in personalized medicine. While their clinical applications are expanding, their role as reusable educational tools and the
[...] Read more.
Background/Objectives: Simulation digital twins (DT) models that integrate patient-specific imaging with artificial intelligence (AI)-based segmentation and extended reality (XR) technologies are rapidly increasing in relevance in personalized medicine. While their clinical applications are expanding, their role as reusable educational tools and the technical pipeline utilized for their development remain incompletely characterized. This narrative review examines current approaches to digital twin creation and XR integration, illustrated by a scoliosis-specific proof-of-concept educational case study. Methods: A narrative technical review was conducted by identifying relevant search keywords within the fields of AI-based image segmentation, extended reality in medicine, and medical education based on the authors’ expertise and familiarity with the subject. PubMed, Google Scholar, and Scopus were searched for English-language studies published primarily between 2015 and 2025 addressing patient-specific three-dimensional modeling, AI-driven segmentation, and XR applications in spine, orthopedic, anesthesiology, and interventional care. A de-identified case of scoliosis is used to present a proof-of-concept example of this process of creating a simulated digital twin for the purpose of medical education in a recorded XR format. Results: Prior studies demonstrated benefits of patient-specific 3D models for anatomical understanding and procedural planning, while highlighting limitations in segmentation accuracy and workflow integration. Nevertheless, while DTs have traditionally served clinical roles in surgical planning or pre-procedural rehearsal, their pedagogical potential remains under-explored. In the proof-of-concept case study, AI-assisted segmentation enabled rapid creation of an anatomically detailed scoliosis digital twin that was incorporated into XR and used to produce a reusable, spatially anchored instructional experience focused on neuraxial access. Conclusions: AI-enabled digital twin models integrated with XR represent a promising approach for personalized, anatomy-driven medical education. Further evaluation is needed to assess educational outcomes, scalability, and integration into clinical training workflows.
Full article
(This article belongs to the Special Issue Artificial Intelligence for Personalized Medicine: Bridging Innovative Technologies and Patient-Centric Care)
►▼
Show Figures

Figure 1
Open AccessArticle
Role of Cardiovascular Magnetic Resonance in Post-Heart Transplant Surveillance: Integrating Evidence with Prospective Cohort Data
by
Ricardo Carvalheiro, Vera Vaz Ferreira, Ana Raquel Santos, Isabel Cardoso, António Valentim Gonçalves, Rita Ilhão Moreira, Tiago Pereira da Silva, Sílvia Aguiar Rosa and Rui Cruz Ferreira
J. Pers. Med. 2026, 16(4), 201; https://doi.org/10.3390/jpm16040201 - 3 Apr 2026
Abstract
Background: Heart transplantation remains the definitive therapy for selected patients with end-stage heart failure, but outcomes are limited by acute rejection, chronic allograft injury, and cardiac allograft vasculopathy. Endomyocardial biopsy (EMB) remains the reference standard for rejection surveillance but is invasive and
[...] Read more.
Background: Heart transplantation remains the definitive therapy for selected patients with end-stage heart failure, but outcomes are limited by acute rejection, chronic allograft injury, and cardiac allograft vasculopathy. Endomyocardial biopsy (EMB) remains the reference standard for rejection surveillance but is invasive and imperfectly captures diffuse myocardial injury. Cardiovascular magnetic resonance (CMR) offers noninvasive, multiparametric assessment of graft structure, function, tissue composition, and perfusion. We aimed to review current evidence supporting CMR in post-heart transplant surveillance and to evaluate the performance of serial CMR for acute cellular rejection in a prospective cohort. Methods: We performed a focused narrative review of the literature on CMR for detection of acute rejection, assessment of chronic allograft injury and prognosis, and evaluation of cardiac allograft vasculopathy and microvascular disease. In parallel, we conducted a prospective observational study of adult heart transplant recipients undergoing early post-transplant CMR (CMR1) and follow-up CMR (CMR2) with temporally matched EMB. Multiparametric CMR included cine imaging, native T1 and T2 mapping, extracellular volume fraction (ECV), and late gadolinium enhancement (LGE). Clinically significant acute cellular rejection was defined as ISHLT grade ≥ 2R. Results: Eighteen recipients were included (median 53 days to CMR1 and 192 days to CMR2). Baseline CMR parameters correlated with invasive hemodynamic and biomarkers. Two patients had biopsy-proven ≥2R rejection at follow-up. T2 values at CMR2 were significantly higher in rejection versus non-rejection patients (59.0 ± 1.4 ms vs. 51.1 ± 1.9 ms; p = 0.015), with greater LGE burden in rejection (p = 0.029). In longitudinal analyses, rejection was associated with divergent patterns of cardiac remodelling and tissue characterization, including increases in indexed ventricular volumes and T2 over time, whereas non-rejection patients demonstrated stable ventricular volumes and a decline in T2. Conclusions: Multiparametric CMR, anchored by T2 mapping, provides clinically meaningful, non-invasive information for acute rejection surveillance after heart transplantation and complements EMB within a personalized, risk-adapted follow-up framework. Establishing individualized baseline CMR phenotypes and monitoring longitudinal changes may support more personalized, less invasive graft surveillance strategies. Larger multicentre prospective studies are needed to define standardized implementation pathways.
Full article
(This article belongs to the Special Issue Personalized Treatment for Heart Failure)
►▼
Show Figures

Figure 1
Open AccessArticle
Personalized Venetoclax Dose Adjustment in Unfit Acute Myeloid Leukemia Patients: A Real-Life Case Series Study
by
Serena Luponio, Bianca Serio, Idalucia Ferrara, Andrea Gigantiello, Anna Maria Della Corte, Denise Morini, Italia Conversano, Francesco Verdesca, Francesca Velino, Anna Maria Sessa, Simona Caruso, Rossella Marcucci, Martina De Leucio, Valentina Giudice, Maddalena Langella and Carmine Selleri
J. Pers. Med. 2026, 16(4), 200; https://doi.org/10.3390/jpm16040200 - 2 Apr 2026
Abstract
Background/Objectives: Minimal residual disease (MRD) negativity is associated with improved outcomes in acute myeloid leukemia (AML) patients. In this retrospective observational real-life case series study, we investigated the efficacy and safety of venetoclax dose adjustment in unfit AML patients and the role
[...] Read more.
Background/Objectives: Minimal residual disease (MRD) negativity is associated with improved outcomes in acute myeloid leukemia (AML) patients. In this retrospective observational real-life case series study, we investigated the efficacy and safety of venetoclax dose adjustment in unfit AML patients and the role of WT1 expression levels as a surrogate marker of MRD monitoring. Methods: A total of 24 consecutive unfit AML patients treated with azacytidine and venetoclax were enrolled in this study, and MRD monitoring was performed by flow cytometry as per international guidelines and by WT1 expression levels assessed by RT-qPCR. Dose adjustment of venetoclax was decided based on MRD status and the onset of grade > 2 neutropenia. Results: The overall response rate was 87.5%, and 16 patients achieved a response already at the first re-evaluation (66.7%). No statistically significant differences were observed between patients who received the standard dose and those with venetoclax dose adjustment in terms of overall survival (19.6 months vs. 30.1 months, respectively; p = 0.9428) and progression-free survival (not reached vs. 22.1 months, respectively; p = 0.3865), although a numerical trend toward lower relapse rates was observed in subjects with late (33.3%) or early and late dose reduction (37.5%) compared to those who had dose adjustment only at the first re-evaluation (75%) (p = 0.3014). The toxicity rate was 33.3% in patients who had early and late dose adjustments, which was lower than that observed with early adjustment (58.3%) and than that reported in the VIALE-A study (84%). Conclusions: Reduced-dose venetoclax regimens (from 28 to 21 days per cycle) in unfit AML patients do not affect response rates or survival and are associated with comparable rates of neutropenia and infectious events, supporting flexible dosing strategies based on patient response and side effects. In addition, WT1 expression could serve as a reliable marker for MRD monitoring.
Full article
(This article belongs to the Special Issue Acute Myeloid Leukemia: Current Progress and Future Directions)
►▼
Show Figures

Figure 1
Open AccessReview
Integrating Intestinal Ultrasound in the Personalized Management of IBD
by
Cristina Lanzotti, Mariangela Allocca, Alessandra Zilli, Ferdinando D’Amico, Virginia Solitano, Sara Massironi, Silvio Danese and Federica Furfaro
J. Pers. Med. 2026, 16(4), 199; https://doi.org/10.3390/jpm16040199 - 1 Apr 2026
Abstract
Personalized medicine is increasingly shaping the management of inflammatory bowel disease (IBD), with the goal of tailoring diagnostic and therapeutic strategies to individual patients. Intestinal ultrasound (IUS) has emerged as a pivotal, non-invasive, and repeatable tool for assessing disease activity, treatment response, and
[...] Read more.
Personalized medicine is increasingly shaping the management of inflammatory bowel disease (IBD), with the goal of tailoring diagnostic and therapeutic strategies to individual patients. Intestinal ultrasound (IUS) has emerged as a pivotal, non-invasive, and repeatable tool for assessing disease activity, treatment response, and complications in both Crohn’s disease and ulcerative colitis. Beyond its role in routine monitoring, IUS enables real-time decision-making and facilitates tight control strategies, aligning with the principles of precision medicine. By combining morphological assessment with advanced techniques, such as contrast-enhanced ultrasound and elastography, IUS offers unique opportunities for risk stratification and individualized treatment planning. Moreover, its accessibility, safety, and patient acceptability make IUS particularly suited for longitudinal follow-up and early detection of therapeutic failure, thereby reducing the need for invasive procedures. This review discusses the integration of IUS into personalized IBD care pathways, highlighting current evidence, clinical applications, and future perspectives.
Full article
(This article belongs to the Special Issue Inflammatory Bowel Disease (IBD): Diagnosis and Personalized Treatment)
►▼
Show Figures

Figure 1
Open AccessArticle
Risk Stratification in Primary Gastric Malignancies in Children, Adolescents, and Young Adults: A SEER-Based Analysis
by
Stavros P. Papadakos, Ioannis Katsaros, Stamatina Vogli, Alexandra Argyrou, Ioannis Karniadakis, Ioannis A. Ziogas, Paraskevas Gkolfakis, Stamatios Theocharis and Dimitrios Schizas
J. Pers. Med. 2026, 16(4), 198; https://doi.org/10.3390/jpm16040198 - 1 Apr 2026
Abstract
Backgrounds: Gastric malignancies are exceptionally rare among children and young adults, and their clinicopathological characteristics and outcomes remain poorly defined. This study aimed to evaluate the demographic, clinical, and survival features of gastric cancer in patients aged ≤24 years using population-based data. Methods:
[...] Read more.
Backgrounds: Gastric malignancies are exceptionally rare among children and young adults, and their clinicopathological characteristics and outcomes remain poorly defined. This study aimed to evaluate the demographic, clinical, and survival features of gastric cancer in patients aged ≤24 years using population-based data. Methods: Data were extracted from the Surveillance, Epidemiology, and End Results (SEER) database (2004–2016) for individuals ≤ 24 years with primary gastric malignancies. Cox proportional hazards models were used to identify predictors of overall survival (OS). Results: A total of 324 patients were included (mean age: 20 years; 50% female). Adenocarcinoma was the most common histologic subtype (52%), nearly half of which were signet ring cell carcinomas (46%). The majority of patients (59%) were diagnosed at stage IV. Median OS was 18 months, and 5-year OS was 49%. Children and adolescents (≤18 years) had significantly better survival than young adults (median OS: 34 vs. 15 months, p < 0.01). In multivariable analysis, advanced AJCC stage and higher lymph node ratio were independently associated with worse overall survival, while lymphoma histology and radiotherapy were linked to more favourable outcomes. Conclusions: Gastric cancers in patients ≤ 24 years often present at advanced stages and are dominated by aggressive histology. Prognosis is primarily determined by tumour stage, histological subtype, and lymph nodal burden. These population-based risk estimates support risk stratification and prognostic assessment in young patients by highlighting histology- and nodal-burden-driven prognostic strata.
Full article
(This article belongs to the Special Issue Personalized Medicine for Gastrointestinal Diseases)
►▼
Show Figures

Figure 1
Open AccessReview
Personalized Breast Reconstruction After Breast-Conserving Therapy: Risk-Informed Approaches to Technique Selection and Timing
by
Thomas J. Sorenson, Carter J. Boyd, Rebecca Lisk and Nolan S. Karp
J. Pers. Med. 2026, 16(4), 197; https://doi.org/10.3390/jpm16040197 - 1 Apr 2026
Abstract
Breast-conserving therapy (BCT), consisting of lumpectomy followed by adjuvant radiation, provides oncologic outcomes equivalent to mastectomy for many patients with breast cancer. As survivorship increases, the demand for aesthetic restoration after BCT has grown; however, reconstructive strategies in this setting remain less standardized
[...] Read more.
Breast-conserving therapy (BCT), consisting of lumpectomy followed by adjuvant radiation, provides oncologic outcomes equivalent to mastectomy for many patients with breast cancer. As survivorship increases, the demand for aesthetic restoration after BCT has grown; however, reconstructive strategies in this setting remain less standardized than those following mastectomy. Reconstruction after BCT presents distinct challenges due to partial tissue loss, nonuniform radiation injury, progressive fibrosis, and wide variability in patient expectations and tolerance for revision surgery. Consequently, mastectomy-based reconstructive algorithms are often insufficient for guiding care in this population. This review synthesizes contemporary reconstructive options following BCT through a personalized medicine framework, emphasizing patient-specific risk factors that influence technique selection, timing, and long-term outcomes. Key determinants include radiation exposure, breast morphology, comorbid conditions, prior breast surgery, and psychosocial preferences. Oncoplastic volume displacement, implant-based augmentation, fat grafting, and autologous reconstruction each demonstrate distinct risk profiles in the post-BCT tissue environment and require individualized application. Timing of reconstruction and willingness to undergo staged procedures play a central role in outcome durability and patient satisfaction. Across reconstructive strategies, revision burden emerges as a clinically meaningful, patient-centered outcome that is not adequately captured by traditional short-term complication metrics. A risk-informed approach that integrates individualized risk assessment with transparent counseling and shared decision-making may improve alignment between reconstructive planning and patient goals. Personalized reconstruction after BCT requires moving beyond technique-driven paradigms toward flexible, longitudinal care pathways. Future efforts should focus on developing BCT-specific predictive models and incorporating patient-reported outcomes to advance personalized reconstructive care.
Full article
(This article belongs to the Section Personalized Therapy in Clinical Medicine)
►▼
Show Figures

Figure 1
Open AccessArticle
Emerging Insights into Hereditary Alpha-Tryptasemia in the Context of Mast Cell Disorders: A Greek Case Series
by
Fotios Koliofotis, Natalia Katrachoura, Niki Papapostolou, Styliani Taka, Maria Martinou, Anthi Bouchla, Sotirios G. Papageorgiou and Michael Makris
J. Pers. Med. 2026, 16(4), 196; https://doi.org/10.3390/jpm16040196 - 1 Apr 2026
Abstract
Background/Objectives: Hereditary alpha-tryptasemia (HαT) is increasingly recognized as a genetic modifier of mast cell-mediated disease severity and has been associated with heightened mediator-related symptoms and an elevated risk of anaphylaxis. This study aimed to describe the clinical characteristics, multisystem manifestations, and treatment
[...] Read more.
Background/Objectives: Hereditary alpha-tryptasemia (HαT) is increasingly recognized as a genetic modifier of mast cell-mediated disease severity and has been associated with heightened mediator-related symptoms and an elevated risk of anaphylaxis. This study aimed to describe the clinical characteristics, multisystem manifestations, and treatment responses of eight patients with HαT and concomitant mast cell disorders. Methods: In this single-center retrospective study, eight adults with confirmed TPSAB1 copy number gain and a diagnosis of systemic mastocytosis (SM), cutaneous mastocytosis (CM), or mast cell activation syndrome (MCAS) were evaluated. Baseline assessments included demographics, clinical history, basal serum tryptase (BST), TPSAB1 genotyping, KIT D816V testing, and bone marrow examination when indicated. Symptom burden was quantified at baseline and week 8 using the Mastocytosis Activity Score (MAS). All patients received mediator-targeted therapy; omalizumab was administered in selected high-risk cases. Results: Eight patients (62.5% male, mean age 53.9 ± 12.0 years) carried TPSAB1 duplication. The median BST was 16.2 ng/mL (range, 14.3–51.2). Severe anaphylaxis occurred in 75% of patients, predominantly drug-induced, while Hymenoptera venom triggered the remaining cases. Gastroesophageal reflux (87.5%), cutaneous symptoms (62.5%), neuropsychiatric features (62.5%), and autonomic dysfunction (37.5%) were common. The mean MAS decreased significantly from 27.25 ± 7.40 to 18.25 ± 6.48 after 8 weeks of high-dose antihistamines, with omalizumab providing marked additional benefit in selected patients. Conclusions: In this cohort, patients with HαT and coexisting mast cell disorders exhibited a high burden of mediator-related symptoms and a notable frequency of anaphylaxis. TPSAB1 genotyping may provide additional genetic information that aids in contextualizing clinical heterogeneity and mediator-related symptom burden in patients with mast cell disorders. Incorporation of HαT testing into routine evaluation may optimize individualized management.
Full article
(This article belongs to the Section Personalized Therapy in Clinical Medicine)
►▼
Show Figures

Figure 1
Open AccessReview
Antibody–Drug Conjugates in Gastrointestinal Oncology: Clinical Efficacy and Inpatient Toxicity Management
by
Ashish Sharma, Harendra Kumar, Ruchir Paladiya, Rajvardhan Sisodia, Hareesha Rishab Bharadwaj, Islam Mohamed, Saqr Alsakarneh, Umar Hayat, Sneh Sonaiya, Hema Sameera Pinnam, Hassam Ali and Dushyant Singh Dahiya
J. Pers. Med. 2026, 16(4), 195; https://doi.org/10.3390/jpm16040195 - 1 Apr 2026
Abstract
Antibody–drug conjugates (ADCs) are reshaping the therapeutic approach to advanced gastrointestinal cancers by integrating tumor-specific monoclonal antibodies with potent cytotoxic payloads to improve targeted tumor cell destruction while minimizing systemic exposure. Compared to traditional chemotherapy, trastuzumab deruxtecan has significantly improved objective response rates
[...] Read more.
Antibody–drug conjugates (ADCs) are reshaping the therapeutic approach to advanced gastrointestinal cancers by integrating tumor-specific monoclonal antibodies with potent cytotoxic payloads to improve targeted tumor cell destruction while minimizing systemic exposure. Compared to traditional chemotherapy, trastuzumab deruxtecan has significantly improved objective response rates and overall survival in HER2-positive gastric and gastroesophageal junction tumors after trastuzumab-based therapy. This supports its role as an important second-line or later treatment option. The ongoing advancement of ADCs targeting CLDN18.2, TROP2, and CEACAM5 indicates that this therapeutic category will continue to expand across gastrointestinal neoplasms. Nonetheless, these advancements are accompanied by a specific and clinically significant toxicity profile. Hematologic suppression, gastrointestinal side effects, hepatotoxicity, and notably interstitial lung disease (ILD) are essential consequences that may need inpatient assessment and care. Interstitial lung disease (ILD), although uncommon, may be severe or lethal if not identified immediately and treated swiftly with medication cessation and corticosteroids. In hospitalized patients, distinguishing ADC-related toxicity from infection or disease progression is often difficult owing to overlapping clinical manifestations, requiring meticulous evaluation and interdisciplinary cooperation. As ADCs are integrated into earlier treatment lines and across a broader patient population, hospital systems must evolve to ensure prompt identification, consistent management protocols, and efficient collaboration between oncology and inpatient teams. This study analyzes the mechanisms, clinical effectiveness, and safety profile of ADCs in gastrointestinal oncology, pointing out the importance of institutional preparedness to safely incorporate these medicines into standard clinical practice. These features also align ADC therapy with personalized medicine by emphasizing biomarker-guided patient selection and individualized toxicity monitoring.
Full article
(This article belongs to the Special Issue Personalized Oncology in Gastrointestinal Cancer: Emerging Therapies and Challenges)
►▼
Show Figures

Figure 1
Open AccessArticle
In Vivo Long Head of the Biceps Tendon Stiffness Varies with Forearm Position During Active Contraction: Implications for Personalized Rehabilitation After SLAP Lesions
by
Zade Pederson and Hugo Giambini
J. Pers. Med. 2026, 16(4), 194; https://doi.org/10.3390/jpm16040194 - 1 Apr 2026
Abstract
Background/Objectives: Type II superior labrum anterior–posterior (SLAP) lesions of the long head of the biceps (LHB) tendon are associated with excessive tendon loading and are commonly treated surgically using SLAP repair, tenotomy, or tenodesis. These procedures alter musculotendinous length and loading and
[...] Read more.
Background/Objectives: Type II superior labrum anterior–posterior (SLAP) lesions of the long head of the biceps (LHB) tendon are associated with excessive tendon loading and are commonly treated surgically using SLAP repair, tenotomy, or tenodesis. These procedures alter musculotendinous length and loading and may affect functional outcomes, including forearm supination strength. Appropriate restoration of tendon tension is critical for favorable muscle adaptation and recovery. Shear wave elastography (SWE) is a non-invasive imaging technique capable of quantifying tissue stiffness as a surrogate for in vivo musculotendinous tension. This study aimed to characterize LHB tendon tension across forearm positions and loading conditions to improve the understanding of functional tendon loading relevant to postoperative activation and rehabilitation. Methods: In this controlled laboratory study, thirteen healthy female volunteers without shoulder pathology were assessed using SWE with the elbow positioned at 90° flexion. LHB tendon tension was measured in forearm pronation and supination under passive, active (unresisted), and weighted conditions. Paired t-tests were used to compare forearm positions within each loading condition. Results: LHB tendon tension was significantly greater during active and weighted conditions compared with passive loading in the pronated position (p < 0.05). During active contraction, tendon tension was significantly lower in supination than pronation (p < 0.05), whereas no positional differences were observed under passive or weighted conditions. Relative to passive loading, tendon tension increased by approximately 18.2% and 89.2% in supination, and 67.0% and 97.9% in pronation during active and weighted conditions, respectively. Conclusions: Forearm position selectively influences LHB tendon tension during active, unresisted contraction. Forearm orientation affected LHB tendon stiffness primarily during active, unweighted contraction, where pronation resulted in higher stiffness than supination. On the other hand, stiffness outcomes measured during passive and weighted positions were comparable between forearm orientations, indicating that positional effects are most evident when tendon loading is primarily muscle-driven. These findings highlight the relevance of forearm positioning during early postoperative activation and provide normative in vivo reference data to inform personalized rehabilitation strategies and future investigations of postoperative tendon loading following SLAP lesion treatment.
Full article
(This article belongs to the Special Issue Personalized Diagnosis and Treatment in Sports Medicine)
►▼
Show Figures

Figure 1
Open AccessArticle
Acid-Suppressive Therapy Choice and Risk of Treatment Escalation in Inflammatory Bowel Disease: A Real-World Comparative Retrospective Study to Inform Personalized Treatment
by
Yan Sun, Donovan Veccia, Gengqing Song and Nisheet Waghray
J. Pers. Med. 2026, 16(4), 193; https://doi.org/10.3390/jpm16040193 - 1 Apr 2026
Abstract
Background/Objectives: Proton pump inhibitors (PPIs) are known to alter gut microbiota composition; however, their association with disease courses and outcomes in patients with inflammatory bowel disease (IBD) remains uncertain. Our aims were to evaluate the association between PPI use and treatment escalation, Clostridioides
[...] Read more.
Background/Objectives: Proton pump inhibitors (PPIs) are known to alter gut microbiota composition; however, their association with disease courses and outcomes in patients with inflammatory bowel disease (IBD) remains uncertain. Our aims were to evaluate the association between PPI use and treatment escalation, Clostridioides difficile infection, and healthcare utilization in IBD. Methods: We conducted a retrospective cohort study on the TriNetX platform. IBD patients with PPIs or histamine-2 receptor antagonists (H2RAs) were matched one-to-one using propensity scores. Outcomes included initiation of corticosteroids, biologic therapy, Clostridioides difficile (C. difficile) infection, and healthcare utilization. Outcomes were assessed during the 0–12-month and 3–12-month follow-up windows. Associations were estimated using odds ratios (ORs) and hazard ratios (HRs) with 95% confidence intervals. Results: After matching, 12,808 patients were included in each group. During 0–12 months of follow-up, PPI use was associated with higher odds of systemic corticosteroid exposure (OR 1.56, 1.35–1.79), biologic therapy initiation (OR 1.99, 1.72–2.29), C difficile infection (OR 1.42, 1.18–1.70), and healthcare utilization (OR 1.18, 1.03–1.36) compared with H2RA use. Time-to-event analyses showed persistent associations with systemic corticosteroid exposure (HR 1.50, 1.31–1.72) and biologic therapy initiation (HR 1.91, 1.66–2.19), with attenuation of associations for infection and healthcare utilization in 3–12-month lag-time analyses. Similar patterns were observed in ulcerative colitis and Crohn’s disease subgroups. Conclusions: PPI was associated with higher risks of treatment escalation and C. difficile compared with H2RA in IBD. These findings highlight the importance of individualized selection and periodic reassessment of acid suppression therapy as part of personalized management strategies in IBD.
Full article
(This article belongs to the Special Issue Inflammatory Bowel Disease (IBD): Diagnosis and Personalized Treatment)
►▼
Show Figures

Figure 1
Open AccessReview
Artificial Intelligence in Cardiovascular Medicine: A Giant Step in Personalized Medicine?
by
Stanislovas S. Jankauskas, Fahimeh Varzideh, Urna Kansakar and Gaetano Santulli
J. Pers. Med. 2026, 16(4), 192; https://doi.org/10.3390/jpm16040192 - 1 Apr 2026
Abstract
Artificial intelligence (AI) is rapidly reshaping cardiovascular (CV) medicine, driving a paradigm shift toward truly personalized and data-driven care. This comprehensive review examines the conceptual foundations, clinical applications, and future implications of AI across the CV continuum, spanning prevention, diagnosis, risk stratification, and
[...] Read more.
Artificial intelligence (AI) is rapidly reshaping cardiovascular (CV) medicine, driving a paradigm shift toward truly personalized and data-driven care. This comprehensive review examines the conceptual foundations, clinical applications, and future implications of AI across the CV continuum, spanning prevention, diagnosis, risk stratification, and therapy. Core AI methodologies (including machine learning, deep learning, natural language processing, and computer vision) are discussed in the context of cardiology’s uniquely data-rich environment, encompassing imaging, electrocardiography, electronic health records, wearable devices, and multi-omics data. This systematic review highlights major clinical domains where AI has demonstrated a substantial impact, including CV imaging, ECG interpretation, hypertension and heart failure management, coronary artery disease, acute coronary syndromes, interventional cardiology, and cardiac surgery. AI-driven predictive analytics enable early detection of subclinical disease, improved prognostication, and individualized prevention strategies, while wearable technologies and remote monitoring platforms facilitate continuous, real-world patient surveillance. Emerging applications in pharmacotherapy, drug repurposing, and genomics further reinforce AI’s role in advancing precision cardiology. Equally emphasized are the ethical, legal, and social challenges accompanying AI adoption, such as algorithmic bias, data privacy, cybersecurity, interpretability, and regulatory oversight. Our review underscores the necessity of rigorous clinical validation, transparent model design, and seamless integration into clinical workflows to ensure safety, equity, and physician trust. Ultimately, AI is best positioned as an augmentative tool that complements (but does not replace!) clinical expertise. By fostering hybrid intelligence that integrates human judgment with computational power, AI has the potential to redefine CV care delivery, improve outcomes, and support a more proactive, patient-centered healthcare model.
Full article
(This article belongs to the Special Issue Personalized Medicine in Cardiovascular and Metabolic Diseases)
Open AccessReview
A Conceptual Framework for Understanding Patient Expectations in Individualised Anaesthesia and Analgesia: A Narrative Review and Future Directions
by
Krister Mogianos and Anna K. M. Persson
J. Pers. Med. 2026, 16(4), 191; https://doi.org/10.3390/jpm16040191 - 1 Apr 2026
Abstract
Acute postoperative pain remains a major clinical challenge, affecting both recovery and resource utilisation. Beyond nociceptive input, pain is shaped by cognitive and emotional factors, including patient expectations. This narrative review examines the role of expectations in perioperative pain modulation, framed within predictive
[...] Read more.
Acute postoperative pain remains a major clinical challenge, affecting both recovery and resource utilisation. Beyond nociceptive input, pain is shaped by cognitive and emotional factors, including patient expectations. This narrative review examines the role of expectations in perioperative pain modulation, framed within predictive coding and Bayesian inference models. These models conceptualise pain as a probabilistic process that integrates sensory input with prior expectations, weighted by precision. In theory, positive expectations may enhance analgesic efficacy, whereas negative expectations may amplify pain via nocebo mechanisms. Control modifies expectations and may reduce perceived pain, while uncertainty diminishes these benefits. Evidence from observational studies links preoperative pain self-efficacy and anticipated pain scores to postoperative outcomes, yet interventional trials remain scarce. In this narrative review, we propose that expectation-sensitive strategies, including structured communication and computational modelling, may inform individualised anaesthesia and analgesia. Future research should validate these frameworks in clinical trials, optimise preoperative expectation management, and explore synergistic approaches that combine pharmacology with cognitive modulation. Understanding and leveraging expectations may offer a promising conceptual direction for more individualised perioperative care, although this approach remains hypothesis-generating at present.
Full article
(This article belongs to the Special Issue New Insights into Personalized Medicine for Anesthesia and Pain)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- JPM Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Healthcare, JCM, JPM, Oral
Advances in Dental Health, 2nd Edition
Topic Editors: Sabina Saccomanno, Gianni GallusiDeadline: 25 July 2026
Topic in
JPM, Mathematics, Applied Sciences, Stats, Healthcare
Application of Biostatistics in Medical Sciences and Global Health
Topic Editors: Bogdan Oancea, Adrian Pană, Cǎtǎlina Liliana AndreiDeadline: 31 October 2026
Topic in
Cancers, Diagnostics, Diseases, IJMS, JPM
Advances in Genetics and Precision Medicine in Human Diseases: 2nd Edition
Topic Editors: Shun-Fa Yang, Shih-Chi SuDeadline: 14 November 2026
Topic in
JCM, JPM, Medicina, Healthcare
New Advances in Physical Therapy and Occupational Therapy, 2nd Edition
Topic Editors: Hye-Rim Suh, Suk-Chan Hahm, Hwi-Young Cho, Ki-Hun ChoDeadline: 15 December 2026

Conferences
Special Issues
Special Issue in
JPM
Personalized Diagnosis and Treatment of Urological Diseases
Guest Editors: Alexandru Ciudin, Lluís PeriDeadline: 10 April 2026
Special Issue in
JPM
Emergency Medicine: Clinical Advances and Challenges in Diagnosis and Treatment, 3rd Edition
Guest Editor: Ovidiu Alexandru MederleDeadline: 15 April 2026
Special Issue in
JPM
Mental Health: Clinical Advances in Personalized Medicine
Guest Editors: Hong Xie, Jony SheyninDeadline: 15 April 2026
Special Issue in
JPM
Personalized Medicine in Gastroenterology and Hepatology
Guest Editor: Shreyak SharmaDeadline: 15 April 2026