Development of Oncolytic Viruses as Platforms for Drug Delivery to Cancers
A special issue of Pharmaceutics (ISSN 1999-4923). This special issue belongs to the section "Gene and Cell Therapy".
Deadline for manuscript submissions: 31 August 2026 | Viewed by 852
Special Issue Editors
Interests: oncolytic viruses; herpes simplex virus type 1; gene therapy; tumor immunology
Interests: oncolytic viruses; immunotherapy; hepatocellular carcinoma; pancreatic cancer
Special Issues, Collections and Topics in MDPI journals
Special Issue Information
Dear Colleagues,
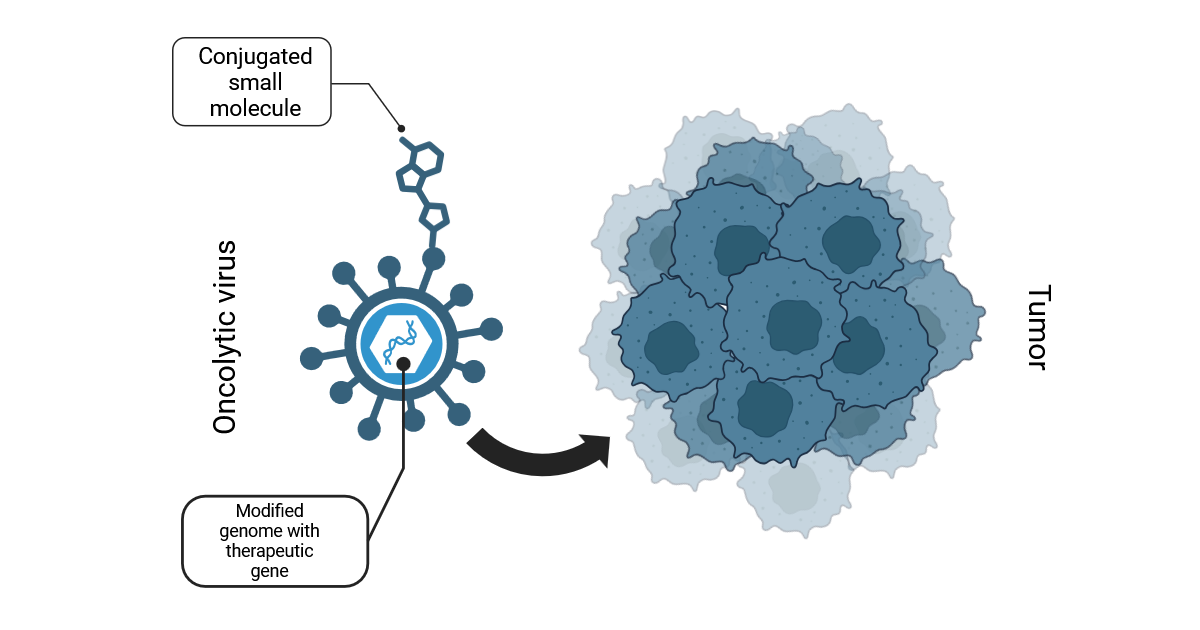
Oncolytic viruses (OVs) are a promising and emerging class of antitumoral therapeutics that selectively target cancer cells by exploiting defects in intrinsic antiviral pathways or the presence of specific receptors. While only one OV has been clinically approved so far by the FDA and the EMA, numerous trials are ongoing, and it is therefore conceivable that over the next years there will be more OVs available in clinical practice as new weapons against difficult-to-treat tumors. One of the most interesting features of these vectors is their versatility, which allows OVs to combine direct cytotoxicity, an immunotherapeutic mechanism of action, and the delivery of therapeutic genes and/or drugs to cancers. Most OVs can be engineered to directly encode therapeutic genes, encompassing biological drugs such as immune checkpoint inhibitors. Therefore, a very active area of research in oncolytic virotherapy is the identification and optimization of the best drugs and therapeutic genes to maximize the treatment efficacy. This Special Issue of Pharmaceutics will be focused on the development of oncolytic viruses as platforms for the targeted delivery of drugs to tumors, including both therapeutic genes encoded by the viral genome and proteins or small molecules that can be attached to viral particles before administration.
Dr. Alberto Reale
Dr. Jennifer Altomonte
Guest Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the special issue website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Pharmaceutics is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- oncolytic viruses
- cancer
- targeted drug delivery
- immunotherapy
- gene therapy
- tumor microenvironment
Benefits of Publishing in a Special Issue
- Ease of navigation: Grouping papers by topic helps scholars navigate broad scope journals more efficiently.
- Greater discoverability: Special Issues support the reach and impact of scientific research. Articles in Special Issues are more discoverable and cited more frequently.
- Expansion of research network: Special Issues facilitate connections among authors, fostering scientific collaborations.
- External promotion: Articles in Special Issues are often promoted through the journal's social media, increasing their visibility.
- Reprint: MDPI Books provides the opportunity to republish successful Special Issues in book format, both online and in print.
Further information on MDPI's Special Issue policies can be found here.







