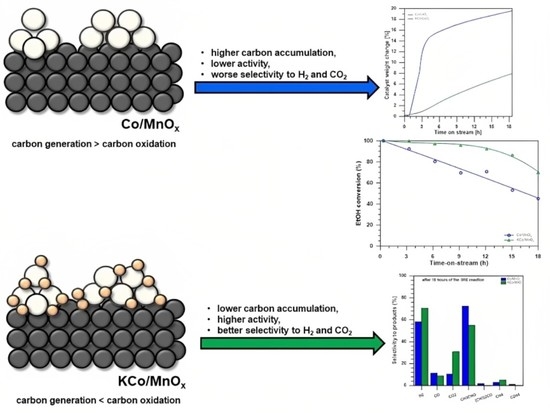
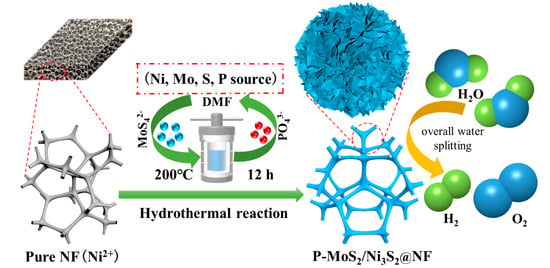
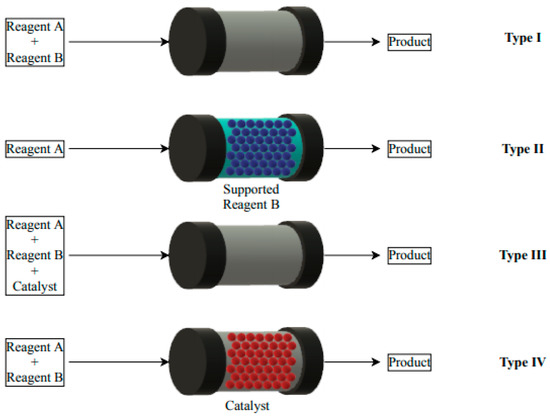
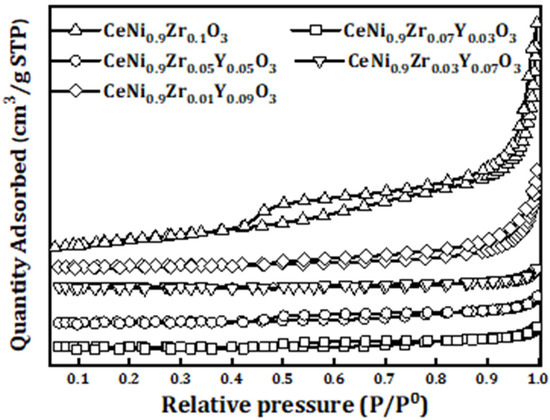
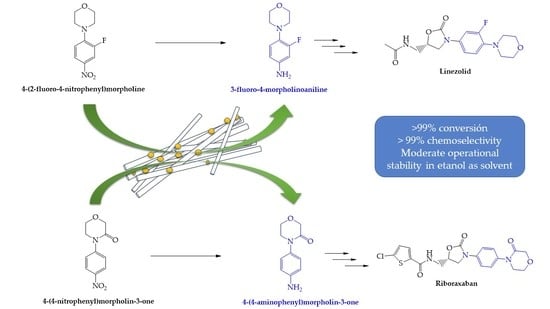
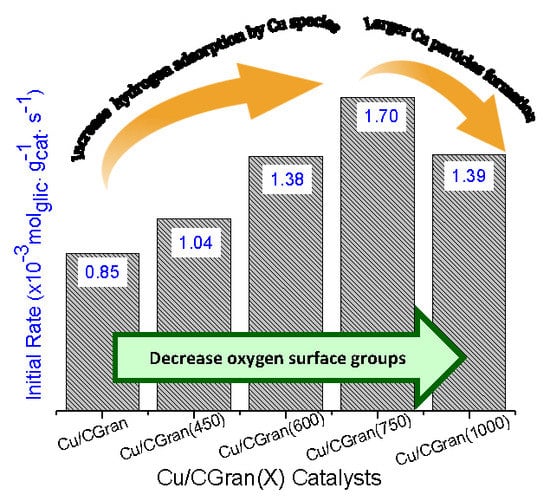
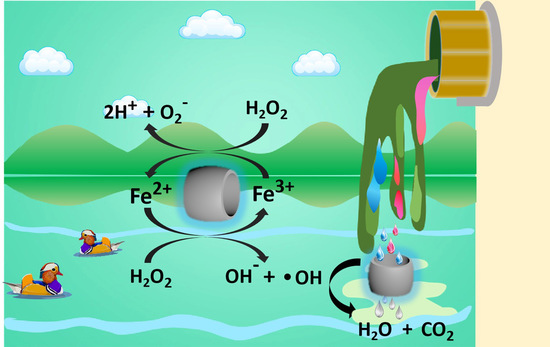
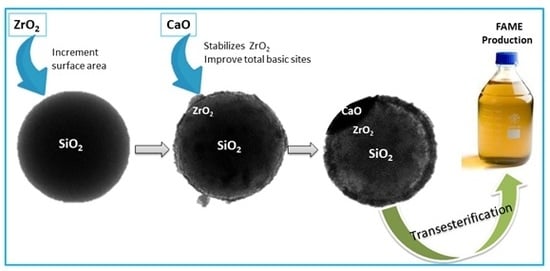
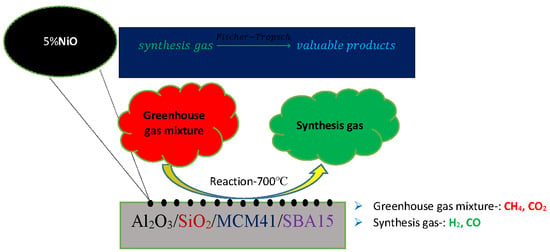
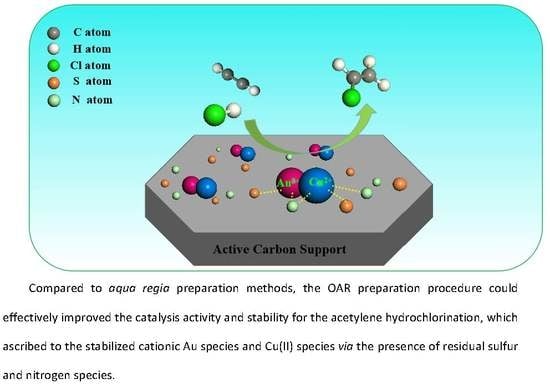
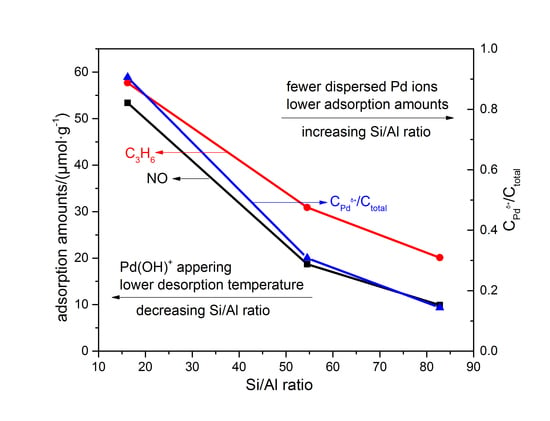
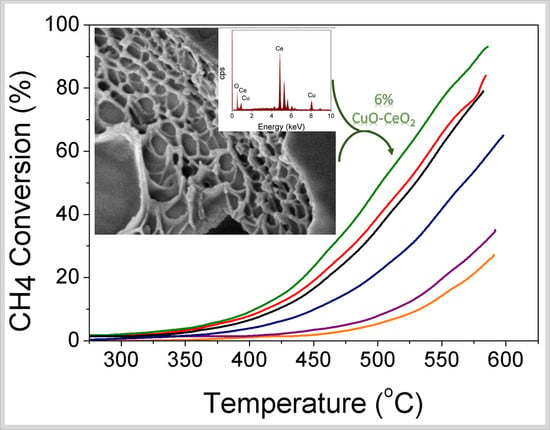
Catalysts: Preparation, Catalytic Performance and Catalytic Reaction
A topical collection in Materials (ISSN 1996-1944). This collection belongs to the section "Catalytic Materials".
Viewed by 117893Editors
Interests: heterogeneous catalysts; materials chemistry; catalysis
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
Catalysts influence our daily lives and play a pivotal role in environmental protection. To be useful, catalysts must have a large activity (turnover frequency), selectivity (to a desired product), and durability (operational stability), properties not possible to find as a lucky circumstance. Therefore, to design a new catalyst, it is necessary to carry out a careful preparation and a deep characterization to understand catalytic performance, one of the earliest prioritie to be used in a given catalytic reaction.
The aim of this Special Issue is to cover the promising recent research and novel trends in the fields of the preparation of catalysts that reach the thermodynamic equilibrium faster with the lowest energy supply to be used in catalytic processes with academic and industrial interest. Full papers, short communications, and reviews in the field are welcome. Mini-reviews with an overview on the state-of-the-art with future perspectives and trends in this domain will be also considered.
The editors welcome contributions increasing the role of catalysts in environmental protection without any restrictions regarding the nature of the catalyst and the preparation method.
Prof. Gina Pecchi
Prof. Cristian H. Campos
Guest Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Materials is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2600 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- catalysis
- catalytic performance
- catalytic reaction
- characterization
- synthesis