Journal Description
Diseases
Diseases
is an international, peer-reviewed, open access, multidisciplinary journal with focus on research on human diseases and conditions, published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), PubMed, PMC, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Medicine, Research and Experimental)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 21 days after submission; acceptance to publication is undertaken in 3.5 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 8 topical sections.
Impact Factor:
3.0 (2024);
5-Year Impact Factor:
3.4 (2024)
Latest Articles
Effects of Heat Waves on Hospitalizations, Emergency Department Visits, and Outpatient Care in Frail Older Adults: A Systematic Review and Meta-Analysis
Diseases 2026, 14(5), 176; https://doi.org/10.3390/diseases14050176 (registering DOI) - 18 May 2026
Abstract
Background/Objectives: Heat waves are increasingly frequent and intense climate events with significant implications for public health, particularly among frail older adults. While most evidence has focused on mortality and morbidity, healthcare service utilization represents an additional and potentially more sensitive indicator of heat-related
[...] Read more.
Background/Objectives: Heat waves are increasingly frequent and intense climate events with significant implications for public health, particularly among frail older adults. While most evidence has focused on mortality and morbidity, healthcare service utilization represents an additional and potentially more sensitive indicator of heat-related health burden. Methods: A systematic review and meta-analysis was conducted following the PRISMA guidelines and prospectively registered in PROSPERO (CRD420251107598). PubMed/MEDLINE, Scopus, and Web of Science were searched up to August 2025. This study aimed to systematically review and quantitatively synthesize the evidence on the association between heat wave exposure and healthcare utilization—including hospitalizations, emergency department (ED) visits, and outpatient care—among frail older adults. Pooled effect estimates (RRs, IRRs, and ORs) were calculated using random-effects models. Heterogeneity was assessed using the I2 statistic, and sensitivity analyses were performed by outcome type, effect measure, and risk of bias. Results: Fifty-five studies met the inclusion criteria. Heat wave exposure was consistently associated with increased healthcare utilization. Both hospitalizations and ED visits showed significant increases during heat wave periods, with results remaining robust across sensitivity analyses. Evidence on outpatient care was limited but suggested a similar pattern. Substantial heterogeneity was observed across studies, reflecting variability in exposure definitions, populations, and study designs. Overall, the methodological quality of the included studies was acceptable, with most presenting a low-to-moderate risk of bias. Conclusions: Heat waves are associated with increased healthcare utilization among frail older adults, indicating a relevant burden on healthcare systems. Healthcare utilization may represent a sensitive indicator of heat wave impact, complementing traditional clinical outcomes.
Full article
(This article belongs to the Special Issue Frailty and Frequent Hospitalizations in Older Adults: Risk, Management, and Interventions)
Open AccessArticle
Sarcopenia Risk in Tenerife: Prevalence, Multidimensional Vulnerability, and the Socio-Economic Case for Prevention and Treatment
by
Vicente Llinares Arvelo, Carlos Enrique Martinez Alberto, David González-Martín and Serafin Corral
Diseases 2026, 14(5), 175; https://doi.org/10.3390/diseases14050175 - 18 May 2026
Abstract
Background/Objectives: Sarcopenia—the progressive loss of skeletal muscle mass and function—is a growing public health challenge in ageing populations. Island territories face compounded vulnerabilities due to distinct epidemiological and socio-economic profiles. This study examines sarcopenia risk prevalence among community-dwelling older adults in Tenerife (Canary
[...] Read more.
Background/Objectives: Sarcopenia—the progressive loss of skeletal muscle mass and function—is a growing public health challenge in ageing populations. Island territories face compounded vulnerabilities due to distinct epidemiological and socio-economic profiles. This study examines sarcopenia risk prevalence among community-dwelling older adults in Tenerife (Canary Islands, Spain) and estimates the economic burden alongside the cost-effectiveness of evidence-based interventions. Methods: A cross-sectional study was conducted among 374 community-dwelling older adults (mean age 80.4 years, SD 4.8; 51.1% female) recruited from primary care health centres across three health zones in Tenerife. Participants were stratified into a control group without established chronic disease-related functional decline (Group 1; n = 274) and a case group with multimorbidity and functional limitations (Group 3; n = 100). Sarcopenia risk was assessed using the SARC-F questionnaire (threshold ≥ 4). A comprehensive geriatric battery—including the Barthel Index, FRAIL scale, MNA-SF, Pfeiffer test, SPPB, handgrip dynamometry, and IPAQ—characterised multidimensional vulnerability. Annual direct and indirect costs were estimated using unit costs from Spanish national health accounts, and intervention cost-effectiveness was modelled using published meta-analytic data. Results: Overall sarcopenia risk prevalence was 36.4% (n = 136; SARC-F ≥ 4), rising to 83.0% in the case group versus 19.3% in controls (OR ≈ 21.5, p < 0.001). Prevalence was 42.1% in males and 30.9% in females. Diabetes was independently associated with elevated risk (44.8% vs. 29.9%; OR 1.90, 95% CI 1.23–2.92; p = 0.003). Health Zone 1 exhibited the highest prevalence (63.0%) versus Zones 2 (23.5%) and 3 (32.8%). Multidimensional vulnerability was pervasive: 28.6% of participants were frail, 75.7% had nutritional compromise, 11.5% showed moderate cognitive impairment, and 89.8% reported low or no physical activity. The estimated annual socio-economic cost of sarcopenia in Tenerife is approximately EUR 88.9 million (Spain nationally: EUR 12.1 billion). Combined exercise–nutrition interventions yield cost-per-QALY ratios of EUR 3800–7000, far below Spain’s EUR 25,000/QALY threshold. Conclusions: Sarcopenia constitutes a major, multidimensionally compounded health burden in Tenerife’s older population, concentrated among frail, diabetic, nutritionally compromised, and physically inactive individuals. The economic case for universal SARC-F screening and multicomponent intervention is compelling, exceeding cost-effectiveness thresholds by a wide margin. Territorial disparities in burden call for equity-oriented, place-based resource allocation within the Canarian health system.
Full article
Open AccessCase Report
Subcapsular Pancreatic Pseudocyst of the Right Hepatic Lobe: A Rare Case Report and Literature Review
by
Nutu Vlad, Laurentiu Budaca, Alexandra Ciubotariu, Florina-Delia Andriesi-Rusu, Mircea Florin Costache, Gigel Sandu, Andrei Cristea and Cătălin Sfarti
Diseases 2026, 14(5), 174; https://doi.org/10.3390/diseases14050174 - 15 May 2026
Abstract
The pancreatic pseudocyst is a collection of pancreatic fluid surrounded by a non-epithelialized wall comprising granulation tissue and fibrosis, occurring in approximately 10% of patients diagnosed with acute pancreatitis and in 20–38% of those with chronic pancreatitis. Most pseudocysts are situated in the
[...] Read more.
The pancreatic pseudocyst is a collection of pancreatic fluid surrounded by a non-epithelialized wall comprising granulation tissue and fibrosis, occurring in approximately 10% of patients diagnosed with acute pancreatitis and in 20–38% of those with chronic pancreatitis. Most pseudocysts are situated in the pancreatic head and pancreatic body, but about 20% develop in extrapancreatic locations. We present the case of a 46-year-old male patient diagnosed with chronic alcohol pancreatitis with acute exacerbation, who developed a large pancreatic pseudocyst with subcapsular location in the right hepatic lobe; this was successfully treated by laparoscopic surgical drainage, with no postoperative complications and no recurrence of the pseudocyst. The computed tomography scan and postoperative biochemical analysis of the intracystic fluid played a key role in establishing the diagnosis of this rare condition. An intrahepatic pancreatic pseudocyst is a rare location for pancreatic pseudocysts, but one located in the right hepatic lobe is extremely rare. The treatment of intrahepatic pancreatic pseudocysts may be conservative, though endoscopic, percutaneous, or surgical drainage may be necessary. The presence of symptoms, signs of extrinsic compression, or complications require drainage of the pseudocyst. The “take-away” lesson learned from this case: surgical treatment for pancreatic pseudocysts located subcapsularly in the liver may be considered when they are very large, or when minimally invasive treatment has not been effective.
Full article
Open AccessReview
Lean Metabolic-Dysfunction-Associated Steatotic Liver Disease (MASLD): Pathophysiology, Diagnostic Challenges, Clinical Outcomes, and Management
by
Noor Albusta, Sara Isa and Hussain Alrahma
Diseases 2026, 14(5), 173; https://doi.org/10.3390/diseases14050173 - 15 May 2026
Abstract
Background/Objectives: Lean metabolic dysfunction-associated steatotic liver disease (lean MASLD) is an increasingly recognized phenotype occurring in individuals with normal body mass index (BMI), despite clinically important hepatic and cardiometabolic risk. This narrative review summarizes current evidence on its epidemiology, pathophysiology, diagnostic challenges, clinical
[...] Read more.
Background/Objectives: Lean metabolic dysfunction-associated steatotic liver disease (lean MASLD) is an increasingly recognized phenotype occurring in individuals with normal body mass index (BMI), despite clinically important hepatic and cardiometabolic risk. This narrative review summarizes current evidence on its epidemiology, pathophysiology, diagnostic challenges, clinical outcomes, and management. Methods: A narrative literature review was conducted using PubMed, Embase, and Cochrane Library from database inception to March 2026. Relevant studies on lean MASLD/lean NAFLD, including cohort studies, meta-analyses, clinical trials, consensus statements, and practice guidelines, were prioritized. Results: Lean MASLD reflects interactions between visceral adiposity, insulin resistance, genetic susceptibility, sarcopenia, dietary and lifestyle factors, vitamin D deficiency, and gut microbiome alterations. Diagnosis is challenging because BMI and aminotransferase levels may underestimate metabolic vulnerability, MASH, or clinically significant fibrosis. Available data suggest increased liver-related events, liver-related mortality, and all-cause mortality compared with individuals without steatotic liver disease, although comparisons with non-lean MASLD remain heterogeneous. Resmetirom and semaglutide have expanded treatment options for noncirrhotic MASH with moderate to advanced fibrosis, but lean patients are underrepresented in pivotal trials. Conclusions: Lean MASLD is an underrecognized but clinically important phenotype. Earlier recognition, fibrosis risk stratification, sarcopenia assessment, cardiometabolic optimization, and lean-specific therapeutic research are needed to improve outcomes.
Full article
Open AccessReview
Multidrug-Resistant Tuberculosis in Central and Eastern Europe: Implementation and Maturity of Whole-Genome Sequencing for Surveillance
by
Dragos Baiceanu, Laura Ioana Chivu, Roxana-Mihaela Coriu, Alexandru Stoichita, Traian-Constantin Panciu, Dragos-Cosmin Zaharia, Beatrice Mahler, Anca Matei, Elmira Ibraim and Loredana Sabina Cornelia Manolescu
Diseases 2026, 14(5), 172; https://doi.org/10.3390/diseases14050172 - 14 May 2026
Abstract
Background/Objectives: Multidrug-resistant tuberculosis (MDR-TB) remains a major public health challenge in the WHO European Region, which reports the highest global proportion of rifampicin-resistant and MDR-TB cases. Whole-genome sequencing (WGS) has emerged as a key tool for improving drug-resistance detection and supporting molecular surveillance.
[...] Read more.
Background/Objectives: Multidrug-resistant tuberculosis (MDR-TB) remains a major public health challenge in the WHO European Region, which reports the highest global proportion of rifampicin-resistant and MDR-TB cases. Whole-genome sequencing (WGS) has emerged as a key tool for improving drug-resistance detection and supporting molecular surveillance. However, the level of genomic implementation across Central and Eastern Europe (CEE) remains insufficiently characterized. This scoping review aimed to evaluate the use of WGS for MDR-TB in CEE countries and to classify implementation maturity using a predefined framework (L0–L4). Methods: A structured search of PubMed/MEDLINE and Web of Science identified original studies published in English between 2015 and 2026 reporting genomic applications in MDR-TB across 13 predefined CEE countries. Data were extracted on sequencing approaches, resistance prediction, transmission analysis, monitoring of new or repurposed drugs, bioinformatic pipelines, and programmatic integration. Countries were categorized according to a five-level maturity model based on documented capacity, scope of application, and integration into national tuberculosis programs (NTPs). Results: Twenty-eight studies were included. WGS was used in 23/28 studies (82.1%), predominantly for genomic resistance prediction (25/28). Transmission analysis was reported in 19/28 studies, with heterogeneous single nucleotide polymorphism (SNP) thresholds and clustering methodologies. Monitoring of resistance to new or repurposed drugs was described in 8/28 studies. No country achieved Level L4 (formally integrated genomic surveillance). Four countries were classified as L3 and nine as L2, while no L0 or L1 settings were identified. Conclusions: Countries in Central and Eastern Europe demonstrate increasing operational use of WGS for MDR-TB, primarily driven by clinical resistance prediction. However, the lack of formal integration into national surveillance systems highlights a persistent gap between technological adoption and structured public health implementation. Strengthening programmatic integration and methodological standardization is essential for advancing genomic surveillance of MDR-TB in the region.
Full article
(This article belongs to the Special Issue Mycobacterial Infections and Human Disease: Emerging Mechanisms and Therapeutic Strategies/Boundaries)
►▼
Show Figures

Figure 1
Open AccessArticle
Rumination as a Mediator Between Intolerance of Uncertainty and Online Health Anxiety, Moderated by Medical History
by
Mălina-Andreea Apostol, Simona Trifu, Andrei-Gabriel Zanfir and Amelia-Damiana Trifu
Diseases 2026, 14(5), 171; https://doi.org/10.3390/diseases14050171 - 13 May 2026
Abstract
Objective: We examined the psychological mechanisms underlying cyberchondria by testing whether rumination mediates the association between intolerance of uncertainty and cyberchondria and whether this indirect effect is moderated by prior medical experiences and perceived access to healthcare. Methods and Measures: A cross-sectional design
[...] Read more.
Objective: We examined the psychological mechanisms underlying cyberchondria by testing whether rumination mediates the association between intolerance of uncertainty and cyberchondria and whether this indirect effect is moderated by prior medical experiences and perceived access to healthcare. Methods and Measures: A cross-sectional design was employed with a non-clinical sample of 96 Romanian adults. Participants completed validated self-report measures of intolerance of uncertainty (IUS-12), rumination (Ruminative Responses Scale), and cyberchondria (Cyberchondria Severity Scale). Additional items assessed medical history and perceived access to healthcare. Moderated mediation analyses with bootstrapped confidence intervals were conducted, controlling for relevant sociodemographic variables. Results: Higher intolerance of uncertainty was associated with higher cyberchondria both directly and indirectly through rumination, which accounted for more than half of the total effect. The rumination–cyberchondria association, and the indirect effect of IU, were significantly stronger among individuals who had experienced a recent acute medical episode, whereas chronic illness did not significantly moderate this pathway. Cyberchondria levels were lowest among participants reporting very good access to healthcare. Conclusions: Cyberchondria appears to arise from the interaction of intolerance of uncertainty, ruminative thinking, and contextual health experiences. Targeting rumination and uncertainty tolerance may be particularly important following acute medical events.
Full article
(This article belongs to the Special Issue The Future of Mental Health: Bridging the Translational Gap Between Mechanism, Practice, and Ethics)
►▼
Show Figures

Figure 1
Open AccessArticle
Cancer Patterns and Barriers to Care Among Socioeconomically Vulnerable Populations in Tripoli: A Descriptive Study from a Local NGO
by
Mouhamad J. Darwich, Dalal Ksair, Zein Adra, Rafaela-Yomn Naji, Bushra Sayed, Rihab Nasr and Zeina Dassouki
Diseases 2026, 14(5), 170; https://doi.org/10.3390/diseases14050170 - 12 May 2026
Abstract
Background/Objectives: Cancer patterns in low-resource and crisis-affected settings are poorly characterized, particularly among socioeconomically vulnerable populations. This study aimed to describe cancer distribution, age at diagnosis, and barriers to care among patients presenting to a non-governmental organization (NGO) in Tripoli, Lebanon. Methods: We
[...] Read more.
Background/Objectives: Cancer patterns in low-resource and crisis-affected settings are poorly characterized, particularly among socioeconomically vulnerable populations. This study aimed to describe cancer distribution, age at diagnosis, and barriers to care among patients presenting to a non-governmental organization (NGO) in Tripoli, Lebanon. Methods: We conducted a retrospective analysis of patients with histopathologically confirmed cancers presenting to a single NGO. Sociodemographic, clinical, and behavioral data were extracted from medical records. Socioeconomic status (SES) was assessed using a validated composite scale. Age-standardized proportions (ASPs) were calculated using GLOBOCAN and WHO standard weights. Barriers to care were categorized into financial, geographic, system-level, and sociocultural domains. Associations were assessed using chi-square tests and regression models. Results: Breast cancer was the most common malignancy (32.0%), followed by colorectal (CRC: 9.8%). A total of 440 patients were included. Colorectal cancer (CRC) was the second-most common malignancy, with 37% of cases occurring before age 50. Breast cancer accounted for nearly half of female cancers. Smoking-related malignancies, particularly bladder and lung cancers, were prominent. Sex differences were cancer-specific, with male sex associated with bladder cancer but not overall cancer distribution. Barriers to care were highly prevalent: 97.3% reported at least one financial barrier, 95.4% system-level barriers, and 72.4% geographic barriers. Low SES was significantly associated with geographic barriers (p < 0.001). Conclusions: Cancer patterns in this vulnerable population are characterized by early-onset disease, a high burden of smoking-related cancers, and pervasive barriers to care. These findings highlight the importance of integrating SES and access-related variables into cancer surveillance systems and support the development of targeted, equity-focused interventions.
Full article
(This article belongs to the Topic Multidimensional Disparities in Cancer Care and Outcomes)
►▼
Show Figures

Figure 1
Open AccessArticle
Predicting Diagnostic Success and Procedural Efficiency in Robotic Bronchoscopy Using Machine Learning
by
Juliana Guarize, Claudia Bardoni, Cristina Diotti, Stefano Maria Donghi and Luca Bertolaccini
Diseases 2026, 14(5), 169; https://doi.org/10.3390/diseases14050169 - 11 May 2026
Abstract
►▼
Show Figures
Background. Robotic-assisted bronchoscopy with the ION™ Endoluminal System facilitates precise access to peripheral pulmonary lesions. However, procedural duration and diagnostic performance remain influenced by patient and lesion-specific factors. To investigate the impact of lesion diameter, radiological appearance, and presence of bronchial signs on
[...] Read more.
Background. Robotic-assisted bronchoscopy with the ION™ Endoluminal System facilitates precise access to peripheral pulmonary lesions. However, procedural duration and diagnostic performance remain influenced by patient and lesion-specific factors. To investigate the impact of lesion diameter, radiological appearance, and presence of bronchial signs on procedural duration and diagnostic yield using conventional regression and gradient boosting machine learning models. Methods. In this single-center retrospective cohort study, 189 ION™ Endoluminal System procedures (November 2024–June 2025) were analyzed. Procedural duration and diagnostic yield served as primary outcomes. Predictive modeling included multivariable regression and gradient boosting. Feature importance metrics were extracted. Results. The median lesion diameter was 12.3 mm, with a “strict” diagnostic yield of 87.3%. Gradient boosting regression identified lesion diameter as the primary predictor of procedural time (89.2% importance; test MSE = 865.6). Diagnostic classification achieved an ROC-AUC of 0.68, with lesion diameter (85.8%) and bronchial sign (14.2%) as key predictors. Conclusions. Lesion diameter emerged as the most consistent predictor of procedural efficiency and was associated with diagnostic performance, albeit within the limitations of the dataset. Broader datasets are needed for external validation and generalizability.
Full article
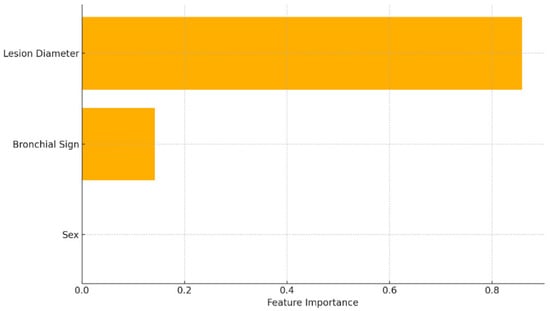
Figure 1
Open AccessCase Report
Seven Years of 700 Cholesterol Without Coronary Atherosclerosis: A Lean Mass Hyper-Responder Case Report
by
Nicholas G. Norwitz, David Feldman and Adrian Soto-Mota
Diseases 2026, 14(5), 168; https://doi.org/10.3390/diseases14050168 - 11 May 2026
Abstract
Background: While reducing LDL cholesterol (LDL-C) remains central focuses of conventional preventive cardiology, substantial heterogeneity exists in the cardiovascular risk associated with even extreme LDL-C elevations, likely depending heavily on the broader metabolic context. Specifically, the lean mass hyper-responder (LMHR) phenotype—characterized by markedly
[...] Read more.
Background: While reducing LDL cholesterol (LDL-C) remains central focuses of conventional preventive cardiology, substantial heterogeneity exists in the cardiovascular risk associated with even extreme LDL-C elevations, likely depending heavily on the broader metabolic context. Specifically, the lean mass hyper-responder (LMHR) phenotype—characterized by markedly elevated LDL-C with elevated high-density lipoprotein cholesterol (HDL-C) and low triglycerides in the setting of a ketogenic diet—has recently been described, though its long-term risk profile remains poorly defined. Case Presentation: We describe a male in his 30s without any congenital dyslipidemia who adopted a ketogenic diet for the management of ulcerative colitis and who subsequently exhibited a sixfold increase in LDL-C from a baseline of 95 mg/dL to 574 mg/dL, with total cholesterol of up to 705 mg/dL, HDL-C at 124 mg/dL, and triglycerides at 34 mg/dL. Despite maintaining these extreme lipid levels for nearly seven years, he demonstrated no coronary plaque or stenosis on coronary computed tomography angiography (CCTA; CAD-RADS = 0). Additionally, quantification of coronary plaque as assessed by AI-guided quantified analysis by Heartflow® identified 0 mm3 plaque in any vessels, placing him in the lowest percentile for atherosclerotic plaque. Conclusions: This case represents an extreme and extensively characterized example of the LMHR phenotype and highlights the limitations of extrapolating cardiovascular risk from LDL-C levels alone without consideration of broader patient context and the etiology of hypercholesterolemia. While a single case cannot redefine clinical practice, this well-characterized case is consistent with emergent literature on LMHR, and careful study of such individuals may provide valuable insights into lipid metabolism, atherosclerosis biology, and precision cardiovascular risk assessment.
Full article
(This article belongs to the Special Issue Metabolic Disorders: Insights into Pathogenesis and Novel Therapeutic Strategies (2nd Edition))
►▼
Show Figures
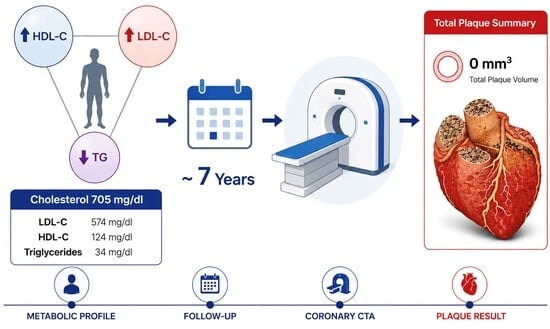
Graphical abstract
Open AccessSystematic Review
Vitamin D and Vitamin B12 in Psychiatric Disorders: An Exploratory Systematic Review and Meta-Analysis of Nutrient-Specific Status and Supplementation Evidence
by
Lavinia-Alexandra Moroianu, Cecilia Curis, Valeriu Ardeleanu, Roxana Elena Bogdan-Goroftei, Simona-Dana Mitincu-Caramfil, Marius Moroianu and Alina Pleșea-Condratovici
Diseases 2026, 14(5), 167; https://doi.org/10.3390/diseases14050167 - 10 May 2026
Abstract
Background/Objectives: Evidence linking vitamins D and B12 to psychiatric outcomes remains heterogeneous across designs, populations, phenotypes, exposures, and outcome formats. Methods: We conducted a PRISMA 2020 systematic review and exploratory meta-analysis of nutrient-specific status and supplementation evidence. PubMed/MEDLINE, APA PsycInfo, Cochrane Library,
[...] Read more.
Background/Objectives: Evidence linking vitamins D and B12 to psychiatric outcomes remains heterogeneous across designs, populations, phenotypes, exposures, and outcome formats. Methods: We conducted a PRISMA 2020 systematic review and exploratory meta-analysis of nutrient-specific status and supplementation evidence. PubMed/MEDLINE, APA PsycInfo, Cochrane Library, Google Scholar, ClinicalTrials.gov, and ProQuest were searched for human studies published in 2016–2025, with a final update on 1 March 2026. Forty-six studies were included (24 randomized controlled trials, 22 observational studies; N = 69,902), and 44 contributed quantitative data. Effects were harmonized to odds ratios (ORs) for cross-family comparability and pooled using Hartung–Knapp random-effects models; supplementation evidence was additionally interpreted on the standardized mean difference (SMD) scale. Results: Across the main evidence families, pooled estimates showed substantial heterogeneity and limited generalizability. Vitamin D supplementation showed an initial inverse estimate on the secondary harmonized OR scale (OR = 0.439, 95% CI 0.272–0.710) and a clinically interpretable SMD of −0.454 (95% CI −0.718 to −0.189), but heterogeneity was high (I2 = 84.2%) and trim-and-fill attenuated the OR estimate to the null (OR = 0.88, 95% CI 0.48–1.63). Vitamin D status showed a similar pattern (primary OR = 0.615, 95% CI 0.424–0.890; trim-and-fill OR = 0.90, 95% CI 0.54–1.49). Vitamin B12 status was inversely associated with outcomes (OR = 0.310, 95% CI 0.115–0.834), but heterogeneity was extreme (I2 = 94.8%). B12 supplementation evidence was sparse and null. Conclusions: The evidence supports targeted deficiency assessment, not routine supplementation.
Full article
(This article belongs to the Special Issue Mental Health—Management and Care, Multidisciplinary Approaches and Perspectives)
Open AccessReview
RHOBTB2-Associated Neurological Phenotypes and Underlying Mechanisms: Alternating Hemiplegia of Childhood Beyond ATP1A3
by
Ruzica Kravljanac, Kristel Klaassen, Vladimir Oparnica, Biljana Vucetic Tadic, Marina Andjelkovic, Anita Skakic, Sara Stankovic and Maja Stojiljkovic
Diseases 2026, 14(5), 166; https://doi.org/10.3390/diseases14050166 - 9 May 2026
Abstract
Alternating hemiplegia of childhood (AHC) represents a severe and complex pediatric neurodevelopmental disorder, predominantly characterized by the occurrence of paroxysmal episodes of transient unilateral or bilateral paresis prior to 18 months of age. It belongs to a group of ultra-rare neurological disorders with
[...] Read more.
Alternating hemiplegia of childhood (AHC) represents a severe and complex pediatric neurodevelopmental disorder, predominantly characterized by the occurrence of paroxysmal episodes of transient unilateral or bilateral paresis prior to 18 months of age. It belongs to a group of ultra-rare neurological disorders with a prevalence from 1:100,000 to 1:1,000,000. Even though the majority of AHC patients harbor pathogenic variants in the ATP1A3 gene, recent studies have pinpointed to other causative genes in ATP1A3-negative patients, including RHOBTB2. In this review, we report the case of a patient with a severe phenotype of AHC associated with developmental delay, aphasia and epilepsy, caused by a pathogenic de novo variant in the RHOBTB2 gene. Furthermore, we contribute a literature review on ATP1A3-negative AHC with a special focus on RHOBTB2-related AHC phenotypes, along with an overview of the pathophysiological mechanism of variants affecting residues in the BTB domain of the RHOBTB2 protein. The results of our study indicate that RHOBTB2-related AHC might have a more severe clinical presentation compared to ATP1A3-related AHC. Variants in the RHOBTB2 gene should be considered as disease-causing in patients with early-onset seizures, delayed psychomotor development and alternating hemiplegia of childhood.
Full article
(This article belongs to the Special Issue Diseases: From Molecular to the Clinical Perspectives)
►▼
Show Figures
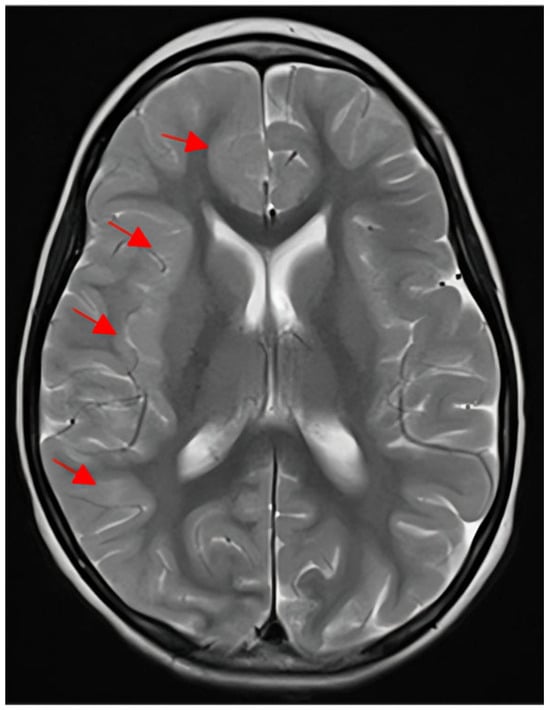
Figure 1
Open AccessArticle
A Unified Histopathological Framework of Liver Fibrogenesis in Chronic Viral Hepatitis B, C and Coinfection
by
Alina Dumitrache (Păunescu), Nicoleta Anca Ionescu (Șuțan), Liliana Cristina Soare, Maria Cristina Ponepal, Ana Cătălina Țânțu, Monica Marilena Țânțu, Ileana Monica Baniță and Cătălina Gabriela Pisoschi
Diseases 2026, 14(5), 165; https://doi.org/10.3390/diseases14050165 - 8 May 2026
Abstract
Background: Chronic hepatitis B and C remain major causes of progressive liver disease, while HBV–HCV coinfection is associated with accelerated fibrosis and hepatocellular injury. Methods: This study evaluated integrated biochemical, histopathological, and immunohistochemical features in patients with chronic hepatitis B (CHB, n =
[...] Read more.
Background: Chronic hepatitis B and C remain major causes of progressive liver disease, while HBV–HCV coinfection is associated with accelerated fibrosis and hepatocellular injury. Methods: This study evaluated integrated biochemical, histopathological, and immunohistochemical features in patients with chronic hepatitis B (CHB, n = 29), chronic hepatitis C (CHC, n = 15), and CHB+C coinfection (CHB+C, n = 10). Liver biopsies were assessed using Ishak and METAVIR scoring systems, alongside immunohistochemical analysis of α-smooth muscle actin (α-SMA), transforming growth factor-β1 (TGF-β1), CD5L, and glial fibrillary acidic protein (GFAP), quantified by H-score. These findings were correlated with biochemical, hematological, and prognostic parameters. Results: Coinfected patients exhibited significantly higher serum ALT, AST, and GGT levels (p ≤ 0.011) and increased CD5L expression (median H-score 197.5 vs. 135 in CHB, p = 0.009), indicating enhanced macrophage-associated inflammatory activity. Although fibrosis stages were comparable across groups, median H-scores for α-SMA, TGF-β1, and GFAP showed a consistent upward trend in CHB+C, suggesting intensified profibrogenic signaling. Principal Component Analysis identified distinct biochemical clusters related to hepatocellular injury, hepatic functional impairment (synthetic and excretory axis), and lipid metabolism. Conclusions: These findings highlight a multidimensional pattern of liver injury in chronic viral hepatitis, with CHB+C coinfection amplifying profibrogenic and hepatocellular markers, both biochemically and histologically.
Full article
(This article belongs to the Special Issue Viral Hepatitis: Diagnosis, Treatment and Management—2nd Edition)
Open AccessArticle
Diagnostic Performance and Discordance of Kato–Katz Method, POC-CCA Test, and PCR in Detecting Schistosoma mansoni in a Low-Prevalence South African Setting
by
Maryline Vere, Wilma ten Ham-Baloyi, Lucy Ochola, Opeoluwa Oyedele, Lindsey Beyleveld, Siphokazi Tili, Takafira Mduluza and Paula Melariri
Diseases 2026, 14(5), 164; https://doi.org/10.3390/diseases14050164 - 8 May 2026
Abstract
►▼
Show Figures
Background/Objectives: Intestinal schistosomiasis caused by Schistosoma mansoni is often underestimated in low-transmission settings due to the limited sensitivity of traditional stool microscopy. More sensitive approaches, including antigen detection and molecular diagnostics, are required to detect infections where egg excretion is low or intermittent.
[...] Read more.
Background/Objectives: Intestinal schistosomiasis caused by Schistosoma mansoni is often underestimated in low-transmission settings due to the limited sensitivity of traditional stool microscopy. More sensitive approaches, including antigen detection and molecular diagnostics, are required to detect infections where egg excretion is low or intermittent. This study aimed to determine the prevalence of S. mansoni infection among school-going children in Nelson Mandela Bay (NMB) using a multi-modal diagnostic approach. Methods: This cross-sectional study included 759 schoolchildren aged 5–14 years from 15 primary schools in NMB. Stool samples were analyzed using the Kato–Katz technique to detect S. mansoni eggs, while urine samples were tested using the point-of-care circulating cathodic antigen (POC-CCA) assay for antigen detection. A subset of stool samples from POC-CCA-positive participants (n = 28) was further analyzed using conventional PCR (cPCR), targeting the S. mansoni cox1 gene, for molecular confirmation. Only a single stool specimen was collected per participant. Results: Among the 759 participants (58% male, 42% female), no egg-positive cases were detected. However, POC-CCA testing identified S. mansoni antigen in 3.2% of participants. Of the 28 POC-CCA-positive samples analyzed by cPCR, 9 (32.1%) were PCR-positive, representing molecular confirmation within the antigen-positive subset rather than overall prevalence. Conclusions: Traditional microscopy underestimated S. mansoni prevalence in this low-prevalence setting. Antigen detection combined with molecular diagnostics improved case identification and highlighted ongoing transmission. These findings support the integration of sensitive diagnostic tools into schistosomiasis surveillance and control strategies in South Africa.
Full article

Figure 1
Open AccessArticle
Association Between Lifestyle Factors and the Prevalence of Non-Communicable Diseases in Saudi Adults Across Different Age Groups: A Cross-Sectional Study
by
Somia A. Nassar
Diseases 2026, 14(5), 163; https://doi.org/10.3390/diseases14050163 - 7 May 2026
Abstract
►▼
Show Figures
Objectives: This cross-sectional study examined associations of lifestyle factors (physical activity (PA), diet, obesity, and smoking), age groups, and sex with the prevalence of non-communicable diseases (NCDs) in Saudi adults, including the World Health Organization (WHO) core NCDs (type 2 diabetes (T2DM), hypertension
[...] Read more.
Objectives: This cross-sectional study examined associations of lifestyle factors (physical activity (PA), diet, obesity, and smoking), age groups, and sex with the prevalence of non-communicable diseases (NCDs) in Saudi adults, including the World Health Organization (WHO) core NCDs (type 2 diabetes (T2DM), hypertension (HTN), chronic obstructive pulmonary disease (COPD) and NCD-associated conditions (osteoporosis (OP), chronic kidney disease (CKD)). Methods: A cross-sectional study was conducted across Saudi Arabia involving 2877 participants aged ≥30 years. Data were collected via an electronic survey using a standardized questionnaire. PA was assessed using the International PA Questionnaire (IPAQ-SF), diet using the Alternative Healthy Eating Index (AHEI), and smoking using the WHO Global Adult Tobacco Survey. Results: Prevalence estimates were: OP 22%, diabetes 21.8%, HTN 13.4%, COPD 4.3%, and CKD 5.1%. All conditions were more prevalent among inactive vs. active individuals (e.g., diabetes: 23.5% vs. 18.8%). An unhealthy diet was associated with higher prevalence (e.g., HTN: 16.3% vs. 10.8%). Obesity showed the strongest association with diabetes (37.1% in obese vs. 14.9% in normal-weight). Smoking was associated with higher prevalence (e.g., COPD: 7.9% vs. 3.7%). Women had higher prevalence than men for most conditions (e.g., OP: 23.4% vs. 19.7%), except COPD (5.1% in men vs. 3.8% in women). Prevalence increased with age (e.g., HTN: 7.2% at age 30–40 vs. 17.3% at age > 60). All comparisons were tested using chi-square tests (p < 0.05). Conclusions: The findings underscore an urgent need for targeted public health interventions to promote PA, improve nutrition, combat obesity, and reduce smoking.
Full article
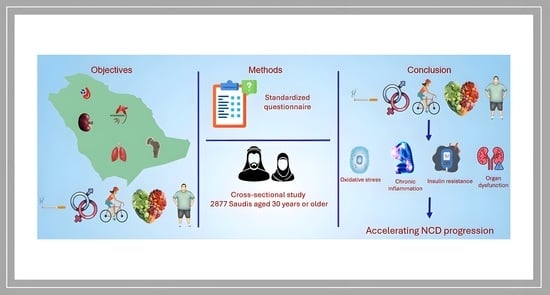
Graphical abstract
Open AccessArticle
Post-Vaccination Surveillance of Invasive Pneumococcal Disease in Ghana
by
Fleischer C. N. Kotey, Reuben E. Arhin, Nicholas T. K. D. Dayie, Emmanuel O. Ampah, Abass Abdul-Karim, Deric A. Baah, Ruth M. Afful, Georgina Tetteh-Ocloo, Roland T. Kom-Zuta, Francis K. M. Tetteh, Mary-Magdalene Osei, Yvonne N. A. Brew, Mame Y. Nyarko, Karikari Asafo-Adjei, Patience B. Tetteh-Quarcoo, Edem M. A. Tette and Eric S. Donkor
Diseases 2026, 14(5), 162; https://doi.org/10.3390/diseases14050162 - 7 May 2026
Abstract
Background: Streptococcus pneumoniae, also referred to as pneumococcus, is of immense public health significance. In particular, it causes severe invasive diseases among children. This has led to the recommendation of anti-pneumococcal prophylaxis, including the administration of penicillin and pneumococcal conjugate vaccines (PCVs),
[...] Read more.
Background: Streptococcus pneumoniae, also referred to as pneumococcus, is of immense public health significance. In particular, it causes severe invasive diseases among children. This has led to the recommendation of anti-pneumococcal prophylaxis, including the administration of penicillin and pneumococcal conjugate vaccines (PCVs), which have become available in about 90% of the countries in sub-Saharan Africa. Nonetheless, breakthrough disease still occurs. Also, PCVs can cause a shift in the distribution of pneumococcal serotypes, usually towards non-vaccine types. However, in many sub-Saharan African countries where PCVs have been introduced, there are hardly any comprehensive post-vaccination surveillance data on pneumococcus. Aim: To describe the post-vaccination epidemiology of invasive pneumococcal disease (IPD) in Ghana, including the prevalence, serotype distribution and antibiotic resistance. Methods: The study was cross-sectional and involved 14,597 patients recruited at the Korle Bu Teaching Hospital, Greater Accra Regional Hospital, Princess Marie Louise Children’s Hospital, Ho Regional Hospital, Eastern Regional Hospital, and Zonal Public Health and Reference Laboratory, Tamale. Specimens of cerebrospinal fluid (obtained by lumbar puncture) and blood were collected routinely from meningitis patients, while blood specimens were taken from pneumonia patients. These were cultured for S. pneumoniae following standard microbiological methods and subjected to antimicrobial susceptibility testing. The isolates were serotyped by the pneumotest latex agglutination kit, and the results confirmed by Quellung reaction, using serotype-specific antisera. Results: The overall prevalence of IPD was 0.66% (n = 97), varying across syndromes: bloodstream infections (0.53%, n = 38), meningitis (2.45%, n = 43), and pneumonia (0.28%, n = 16). The majority of the cases (56.70%, n = 55) occurred in the 11–20-year-old group. Ten pneumococcal serotypes were identified, with Serotype 1 being predominant (58.76%), followed by Serotypes 23B (11.34%), 33F (9.28%), and 12F (8.24%). Vaccine serotypes accounted for 81.44% of the isolates, while 18.56% were non-vaccine serotypes (23A, 23B, and 38). Antimicrobial resistance was highest against sulphamethoxazole-trimethoprim (52%), ampicillin (51%), and penicillin (46%). No resistance was observed against ciprofloxacin, levofloxacin, and vancomycin. The multidrug resistance proportion was 42.3% (n = 41). Conclusions: Even in the post-vaccination era, vaccine-type IPD remains a significant public health issue in Ghana. The observed serotype distribution and antimicrobial resistance patterns warrant sustained surveillance, more adaptive vaccination policies, and rigorous antibiotic stewardship to effectively mitigate IPD burden.
Full article
(This article belongs to the Section Infectious Disease)
►▼
Show Figures
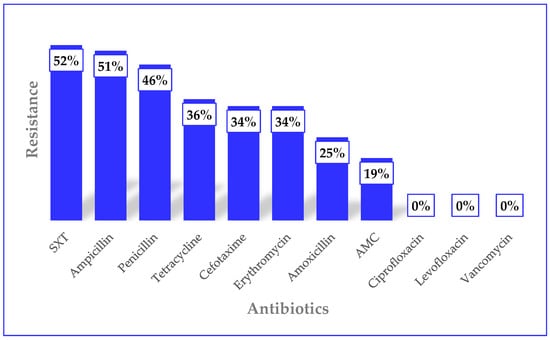
Figure 1
Open AccessArticle
Galectin-3 Mediates Heme-Induced Multi-Organ Dysfunction by Modulating the Splenic Immune Microenvironment
by
Mirjana Milinkovic, Marija Milovanovic and Jelena Milovanovic
Diseases 2026, 14(5), 161; https://doi.org/10.3390/diseases14050161 - 6 May 2026
Abstract
►▼
Show Figures
Background/Objectives: Acute intravascular hemolysis is associated with the release of labile heme, which contributes to systemic inflammation and organ dysfunction. Galectin-3 (Gal-3) is a known modulator of inflammatory responses. However, its specific role in heme-induced organ injury remains to be fully elucidated. Methods:
[...] Read more.
Background/Objectives: Acute intravascular hemolysis is associated with the release of labile heme, which contributes to systemic inflammation and organ dysfunction. Galectin-3 (Gal-3) is a known modulator of inflammatory responses. However, its specific role in heme-induced organ injury remains to be fully elucidated. Methods: We used a phenylhydrazine (PHZ)-induced model of acute hemolysis in wild-type (WT) and Gal-3 knockout (KO) mice to investigate the influence of Gal-3 on tissue alterations and the inflammatory response. Results: Despite equivalent levels of hemolysis and anemia in both genotypes, Gal-3 deficiency was associated with reduced injury in the liver, kidneys, and pancreas. In WT mice, Gal-3 was associated with a pro-inflammatory splenic microenvironment. Conversely, Gal-3 KO mice exhibited a shift toward an immunoregulatory phenotype, characterized by an increased frequency of CD4 + CD25 + FoxP3+ regulatory T cells and IL-10+ macrophages. This shift correlated with preserved organ architecture and a more controlled inflammatory profile. Conclusions: Our findings suggest that Gal-3 may act as a mediator of heme-induced systemic inflammation. By influencing the splenic immune microenvironment and promoting a regulatory phenotype, the absence of Gal-3 appears to alleviate multi-organ stress, suggesting its potential as a target for modulating complications during acute hemolytic crises.
Full article
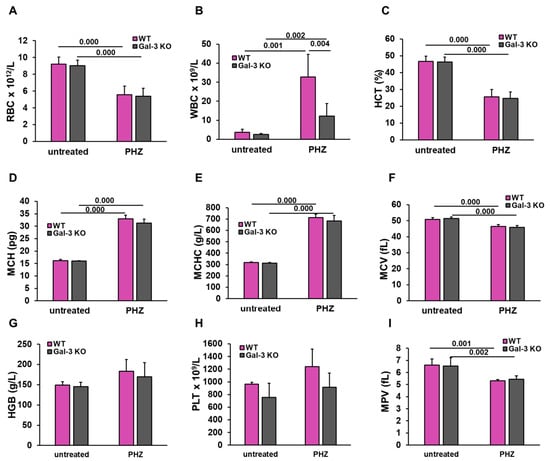
Figure 1
Open AccessArticle
Association Between Abscess Size, Inflammatory Markers, and the Need for Drainage in Renal Abscesses
by
Dragoș Puia, Marius Ivănuță, Ovidiu Daniel Bîcă, Nicolae Stoican, Mihaela Corlade-Andrei, Bogdan Doroftei and Cătălin Pricop
Diseases 2026, 14(5), 160; https://doi.org/10.3390/diseases14050160 - 30 Apr 2026
Abstract
►▼
Show Figures
Background: Renal abscesses represent a serious complication of urinary tract infections, with management decisions often being guided by abscess size and clinical parameters. However, there is no universally accepted size threshold for intervention, and the role of inflammatory markers such as white blood
[...] Read more.
Background: Renal abscesses represent a serious complication of urinary tract infections, with management decisions often being guided by abscess size and clinical parameters. However, there is no universally accepted size threshold for intervention, and the role of inflammatory markers such as white blood cell count (WBC) and C-reactive protein (CRP) in guiding treatment remains uncertain. We aimed to evaluate the relationship between abscess size, inflammatory markers, and the need for drainage in patients with renal abscesses treated in a tertiary urology clinic. Methods: A retrospective analysis was conducted on 103 adult patients diagnosed with renal abscesses between 2020 and 2025. Patients were categorized into two groups based on abscess size: Group A (<50 mm) and Group B (50 mm). Results: The cohort included 59 females and 44 males, with a mean age of 60.5 years. Computed tomography was used for diagnosis in 55.3% of cases. The most common comorbidities were hypertension (46.6%) and diabetes mellitus (40.8%). Microbiological cultures most frequently identified Escherichia coli (38.3%) and Klebsiella spp. (21.7%). Antibiotic resistance was highest to ampicillin (79.5%), while amikacin (5.8%) and piperacillin/tazobactam (6.2%) showed the lowest resistance rates. Conservative antibiotic therapy was effective in 43 patients (42.7%), whereas 60 patients (58.3%) required percutaneous drainage. Abscess size was associated with invasive intervention, with 88.1% of drained abscesses measuring ≥50 mm compared to 9.1% in the conservatively managed group (p < 0.001). Patients with larger abscesses had significantly lower haemoglobin levels (p = 0.003), while no significant differences were observed in WBC or CRP levels. Conclusions: Abscess size was associated with the need for drainage, supporting its role in clinical decision-making. In contrast, inflammatory markers such as WBC and CRP showed limited value in predicting the need for intervention in this cohort. These findings should be interpreted in the context of the retrospective design.
Full article

Figure 1
Open AccessArticle
Locked-Window EQ-5D-5L (Index and VAS) Benchmarking in Sarcoma Care: Rule-Based Traffic-Light Classification Across Two Institutions
by
Isabel Gloor, Beatrice Meier, Jehona Rexhai, Philip Heesen, Georg Schelling, Bettina Vogel, Gabriela Studer, Bruno Fuchs and on behalf of the Swiss Sarcoma Network
Diseases 2026, 14(5), 159; https://doi.org/10.3390/diseases14050159 - 30 Apr 2026
Abstract
►▼
Show Figures
Background: Value-based sarcoma care requires outcome measures that reflect the patient perspective; however, many sarcoma episodes begin with near-normal function and undergo necessary morbidity for oncologic control, making simple “improvement” an unreliable proxy of value. In routine care, patient-reported outcome data are often
[...] Read more.
Background: Value-based sarcoma care requires outcome measures that reflect the patient perspective; however, many sarcoma episodes begin with near-normal function and undergo necessary morbidity for oncologic control, making simple “improvement” an unreliable proxy of value. In routine care, patient-reported outcome data are often irregular and incomplete, limiting benchmarking and learning across institutions. We therefore developed a rule-based EQ-5D-5L (index and VAS) traffic-light framework and evaluated its feasibility and benchmarking signal in two institutions. Methods: We performed a retrospective, two-institution cohort analysis of 729 malignant and intermediate episodes, defined using a prespecified histology behavior mapping. PROM evaluation was anchored to a hierarchical T0 (index surgery date; if unavailable, radiotherapy start date; if unavailable, systemic therapy start date where a valid and interpretable start date was available). EQ-5D-5L index and EQ-VAS were assigned to prespecified locked windows: baseline (−90 to +14 days preferred; +15 to +90 days fallback), 12 months (180–365 days; target 270), and 24 months (660–820 days; target 730). A rule-based traffic-light classification was applied at 12 and 24 months (RED if index < 0.75 or VAS < 50; GREEN if index ≥ 0.85 and VAS ≥ 70; otherwise YELLOW). PROM evaluability was defined as the availability of at least one valid EQ-5D-5L index and/or EQ-VAS value within each window. Results: PROM evaluability in locked windows was feasible but incomplete. Baseline PROMs were available for 107/729 episodes (14.7%), 12-month PROMs for 119/729 (16.3%), and 24-month PROMs for 84/729 (11.5%). At 12 months, evaluable episodes included 75 from Institution A and 44 from Institution B; at 24 months, 56 and 28, respectively. Traffic-light outputs showed heterogeneity at both timepoints and clearer cross-institution difference at 24 months than at 12 months. At 12 months, the distribution was predominantly GREEN in both institutions (Institution A: 73.3% GREEN, 9.3% YELLOW, 17.3% RED; Institution B: 65.9% GREEN, 18.2% YELLOW, 15.9% RED; p = 0.373). At 24 months, Institution A maintained a high GREEN proportion with a low RED fraction (76.8% GREEN, 17.9% YELLOW, 5.4% RED), whereas Institution B showed a lower GREEN proportion and higher YELLOW/RED fractions (50.0% GREEN, 25.0% YELLOW, 25.0% RED; p = 0.014). Absolute EQ-5D-5L medians remained high overall, but the follow-up distributions showed a broader lower tail in Institution B. Conclusions: A prespecified EQ-5D-5L (index and VAS) traffic-light framework anchored by hierarchical T0 and evaluated in locked windows yields interpretable patient-perspective benchmarking signals in real-world sarcoma care. The approach was operationally feasible within the evaluable subset and appeared more discriminative at 24 months than at 12 months, while incomplete PROM capture remains a major implementation limitation for representative and reliable network-scale benchmarking and learning.
Full article

Graphical abstract
Open AccessSystematic Review
Physiotherapy Intervention for Diabetic Foot Ulcers: A Scoping Review
by
Shinsuke Imaoka, Shohei Minata, Taisuke Teroh, Kotaro Matsuki and Ryotaro Hiramatsu
Diseases 2026, 14(5), 158; https://doi.org/10.3390/diseases14050158 - 29 Apr 2026
Abstract
►▼
Show Figures
Background/Objectives: Understanding the available interventions and circumstances under which physical therapy is administered to patients with diabetic foot ulcers is important to provide more evidence regarding physical therapy and associated outcomes in this population. This study aimed to investigate the scope, nature, and
[...] Read more.
Background/Objectives: Understanding the available interventions and circumstances under which physical therapy is administered to patients with diabetic foot ulcers is important to provide more evidence regarding physical therapy and associated outcomes in this population. This study aimed to investigate the scope, nature, and extent of literature on physical therapy interventions for adults with diabetic foot ulcers. Methods: Articles on physiotherapy interventions for adults with diabetic foot ulcers published up to 30 June 2024 were included. Relevant articles were identified through searches of PubMed, Scopus, MEDLINE, and the Cochrane Library databases. Opinion articles, study protocols, meeting abstracts, and articles that did not describe physical therapy interventions were excluded. Results: The systematic search identified 13 articles that met the inclusion criteria. Eleven of the 13 articles were specifically related to outpatient physical therapy. Outpatient physiotherapy included unloading gait instruction, ankle stretching instruction, progressive resistance training, and aerobic exercise. In two other cases, exercise instructions were practiced in the early postoperative period of the wound during the hospitalization period. A multidisciplinary approach aimed at improving postoperative activities of daily living was included. The main efficacy indices were the wound reduction rate, plantar pressure reduction, hemodynamics, ankle joint range of motion, walking ability, and other physical function-related parameters. Conclusions: Physiotherapy during outpatient follow-up may contribute to preventing wound deterioration and maintaining physical function in patients with stable DFUs. However, standardized protocols regarding intervention timing, exercise intensity, and wound severity remain unestablished, and interventions should be applied cautiously based on individual clinical conditions.
Full article
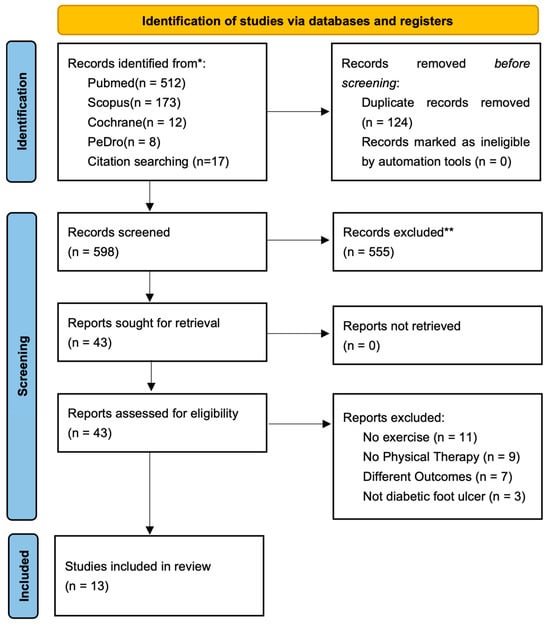
Figure 1
Open AccessArticle
Depressive Symptoms in Pediatric Tuberculosis: A Retrospective Two-Time-Point Observational Study
by
Oana Mariana Mihailov, Loredana Stavăr Matei, George Țocu, Valerii Luțenco, Cosmin George Popovici and Raul Mihailov
Diseases 2026, 14(5), 157; https://doi.org/10.3390/diseases14050157 - 29 Apr 2026
Abstract
Background: Tuberculosis (TB) in children is associated not only with infectious burden but also with potential psychological distress, which remains insufficiently explored. The aim of this study was to evaluate the pattern and evolution of depressive symptoms in pediatric TB patients during treatment
[...] Read more.
Background: Tuberculosis (TB) in children is associated not only with infectious burden but also with potential psychological distress, which remains insufficiently explored. The aim of this study was to evaluate the pattern and evolution of depressive symptoms in pediatric TB patients during treatment using a structured screening approach. Methods: We conducted a retrospective observational study including 190 pediatric patients aged 7–18 years diagnosed with tuberculosis between 2019 and 2021. Depressive symptoms were assessed at two time points, namely at diagnosis (T0) and at first follow-up (T1), using a 10-item structured clinical screening tool routinely applied in practice. A threshold of ≥50% affirmative responses was used to identify patients with suspected depressive symptoms. The Children’s Depression Inventory (CDI) was administered to patients with positive screening results, according to standard clinical protocols. Descriptive and comparative analyses were performed to evaluate changes over time. Results: A high proportion of patients screened positive for depressive symptoms at baseline (T0). At follow-up (T1), a reduction in the proportion of patients with suspected depressive symptoms was observed; however, a substantial number of patients continued to report symptoms suggestive of emotional distress. Most symptom changes between T0 and T1 were not statistically significant, with the exception of decreased appetite, which showed a modest improvement. The overall pattern suggests persistence of symptoms in a subset of patients over time. Conclusions: These findings suggest that symptoms indicative of psychological distress are common among pediatric TB patients and may persist during treatment. However, given the use of a non-validated screening tool and the retrospective design, the results should be interpreted with caution. The study highlights the potential value of systematic psychological assessment in this population and supports the need for further research using validated instruments.
Full article
(This article belongs to the Special Issue Mental Health—Management and Care, Multidisciplinary Approaches and Perspectives)
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Cancers, Current Oncology, Diseases, Onco
Genomic Signature of Ocular Tumors
Topic Editors: Bita Esmaeli, Natalie WolkowDeadline: 20 October 2026
Topic in
Cancers, Diagnostics, Diseases, IJMS, JPM
Advances in Genetics and Precision Medicine in Human Diseases: 2nd Edition
Topic Editors: Shun-Fa Yang, Shih-Chi SuDeadline: 14 November 2026
Topic in
Biomedicines, Diseases, JCM, JPM, Uro, Reports
Clinical, Translational, and Basic Research and Novel Therapy on Functional Bladder Diseases and Lower Urinary Tract Dysfunctions
Topic Editors: Hann-Chorng Kuo, Yao-Chi Chuang, Chun-Hou LiaoDeadline: 31 December 2026
Topic in
Cancers, Cells, Diseases, Genes, IJMS, ncRNA, Sci
Alternative Splicing in RNA Metabolism, Tissue Homeostasis and Human Disease: From Mechanisms to Therapeutics and Clinical Applications
Topic Editors: Christos K. Kontos, Dongyu JiaDeadline: 15 January 2027

Conferences
Special Issues
Special Issue in
Diseases
Advances in Melanoma: From Basic Research to Clinical Management, and Future Horizons Exploitation
Guest Editor: Laura AtzoriDeadline: 31 May 2026
Special Issue in
Diseases
Digital Pathology in Precision Oncology: Emerging Tools for Diagnosis and Treatment Guidance
Guest Editors: Daniela Hartmann, Maximilian DeussingDeadline: 25 June 2026
Special Issue in
Diseases
Functional Foods and Supplements: Special Focus on Mechanisms of Action and Potential Clinical Application
Guest Editors: Emilio Jirillo, Omar Cauli, Luigi SantacroceDeadline: 30 June 2026
Special Issue in
Diseases
Advances in Head and Neck Surgery: Precision Tools and Smart Technologies for Improved Surgical Outcomes
Guest Editors: Stefano Settimi, Claudio ParrillaDeadline: 31 July 2026
Topical Collections
Topical Collection in
Diseases
Lysosomal Storage Diseases
Collection Editors: Jose Sanchez-Alcazar, Luis Jiménez Jiménez












