Assessing Value in Child Health
A special issue of Children (ISSN 2227-9067). This special issue belongs to the section "Global Pediatric Health".
Deadline for manuscript submissions: closed (31 March 2021) | Viewed by 22180
Special Issue Editor
2. Institute of Health Policy, Management and Evaluation, University of Toronto, Toronto, M5T 3M6 ON, Canada
Interests: health technology assessment; child health; cost-effectiveness analysis; health-related quality of life; genomics; neurodevelopmental disorders
Special Issue Information
Dear Colleagues,
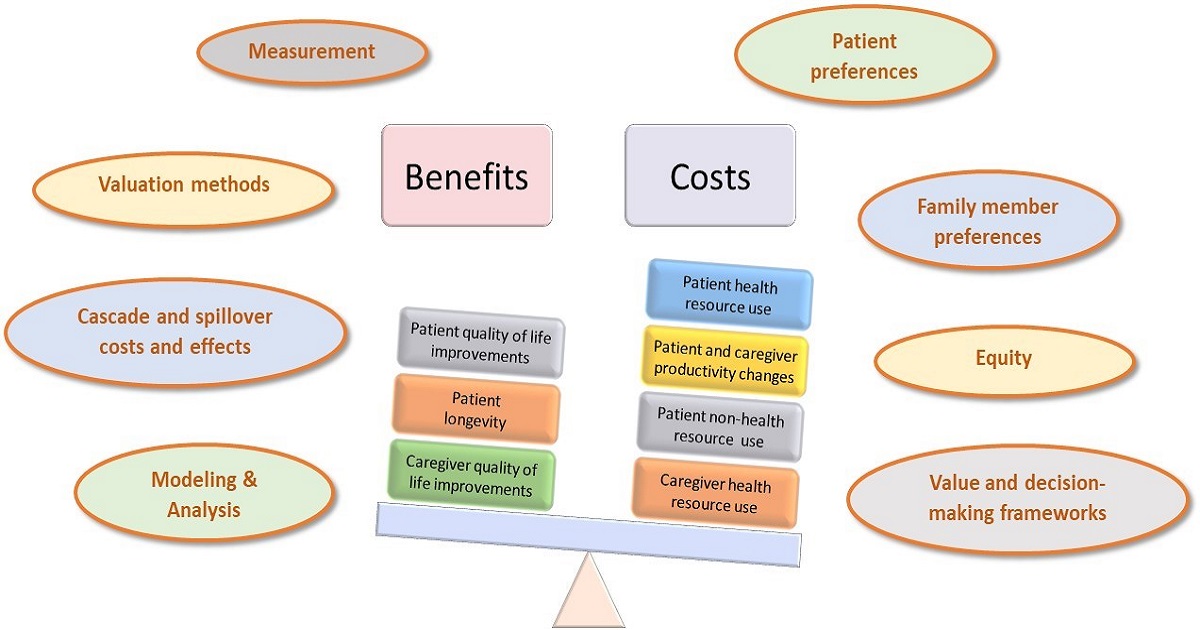
Assessing value in child health is increasingly important as health care systems face difficult choices with regard to what services and programs to offer. Researchers, practitioners, and funding and policy decision makers appreciate that value extends far beyond assessments of efficacy and safety of new treatments and interventions. Measuring cost-effectiveness, evaluating effects on quality of life for patients and their caregivers, determining preferences for treatments and services from patients, families and members of the public, and consideration of the ethical implications of new complex technologies all contribute to an understanding of value. Conducting research to measure elements of value often necessitates novel methods and approaches. Conducting such research in child health is particularly challenging given the paucity of valid pediatric patient-reported outcome (PRO) instruments, the lack of clinical trial data sources, reliance on proxies, and many other methodologic and data-related constraints. This creates exciting opportunities for methodologic research to develop and validate tools, instruments, statistical approaches, and models to measure elements of value in child health.
Alongside research to measure value and expand the methodologic toolbox for such assessments in child health, there is growing interest in the development of valid value frameworks to assist funding and policy decision makers called upon to make evidence-informed decisions based on the highest quality evidence.
This Special Issue invites submissions that pertain to the topic of assessing value in child health. Manuscripts that address the following topics are welcome:
- Health technology assessment (HTA) in child health;
- Pediatric cost-effectiveness and willingness-to-pay analysis;
- Measuring preferences (utility) of patients, family members or members of the public for pediatric health states;
- Health-related quality of life and cost spillover effects in caregivers of children with a health condition;
- Valuation of preferences for pediatric interventions and technologies by patients, family members, clinicians, and members of the public using quantitative or qualitative methods;
- Ethical analysis for valuation of complex emerging technologies in child health;
- The development of tools, instruments, statistical approaches or models to measure elements of value in child health;
- Value frameworks for funding and policy decision making for pediatric interventions and services.
Prof. Wendy J. Ungar
Guest Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the special issue website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Children is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2400 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- Health technology assessment
- Cost-effectiveness analysis
- Willingness-to-pay
- Health state preferences
- Spillover effects
- Preference ascertainment
- Ethical analysis
- Value frameworks
Benefits of Publishing in a Special Issue
- Ease of navigation: Grouping papers by topic helps scholars navigate broad scope journals more efficiently.
- Greater discoverability: Special Issues support the reach and impact of scientific research. Articles in Special Issues are more discoverable and cited more frequently.
- Expansion of research network: Special Issues facilitate connections among authors, fostering scientific collaborations.
- External promotion: Articles in Special Issues are often promoted through the journal's social media, increasing their visibility.
- Reprint: MDPI Books provides the opportunity to republish successful Special Issues in book format, both online and in print.
Further information on MDPI's Special Issue policies can be found here.






