Journal Description
Catalysts
Catalysts
is a peer-reviewed open access journal of catalysts and catalyzed reactions published monthly online by MDPI. The Romanian Catalysis Society (RCS) are partners of Catalysts journal and its members receive a discount on the article processing charge.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), Inspec, CAPlus / SciFinder, CAB Abstracts, and other databases.
- Journal Rank: JCR - Q2 (Chemistry, Physical) / CiteScore - Q1 (General Environmental Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.3 days after submission; acceptance to publication is undertaken in 2.7 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.9 (2022);
5-Year Impact Factor:
4.2 (2022)
Latest Articles
Liquid–Liquid Equilibrium Behavior of Ternary Systems Comprising Biodiesel + Glycerol and Triglyceride + Methanol: Experimental Data and Modeling
Catalysts 2024, 14(5), 320; https://doi.org/10.3390/catal14050320 (registering DOI) - 12 May 2024
Abstract
Having a comprehensive knowledge of phase equilibrium is advantageous for industrial simulation and design of chemical processes. For further acquisition of primary data to facilitate the separation and purification of waste oil biodiesel systems, a liquid–liquid equilibrium (LLE) tank is deployed for the
[...] Read more.
Having a comprehensive knowledge of phase equilibrium is advantageous for industrial simulation and design of chemical processes. For further acquisition of primary data to facilitate the separation and purification of waste oil biodiesel systems, a liquid–liquid equilibrium (LLE) tank is deployed for the ternary system of waste oil biodiesel + methanol + glycerin, thereby enhancing the precision and efficiency of the process. The phase equilibrium system was constructed under the influence of atmospheric pressure at precise temperatures of 303.15 K, 313.15 K, and 323.15 K. The equilibrium components of each substance were analyzed by employing high-temperature gas chromatography, a sophisticated analytical method that enables the identification and quantification of individual components of a sample. Moreover, the ternary liquid–liquid equilibrium data were correlated by implementing the NRTL and UNIQUAC activity coefficient models. Subsequently, the binary interaction parameters of the ternary system were derived by conducting regression analysis. The experimental data demonstrated that the presence of lower methanol content in the system resulted in nearly immiscible biodiesel and glycerol phases, which ultimately facilitated the separation of biodiesel and glycerol. Conversely, with the increase in methanol content, the mutual solubility of biodiesel and glycerol was observed to increase gradually. The results showed that the calculated values of the NRTL and UNIQUAC models aligned well with the experimental values. The root-mean-square deviations of the NRTL and UNIQUAC models at 313.15 K were 2.76% and 3.56%, respectively.
Full article
(This article belongs to the Section Biomass Catalysis)
Open AccessArticle
Methane Combustion Kinetics over Palladium-Based Catalysts: Review and Modelling Guidelines
by
Roshni Sajiv Kumar, Joseph P. Mmbaga, Natalia Semagina and Robert E. Hayes
Catalysts 2024, 14(5), 319; https://doi.org/10.3390/catal14050319 (registering DOI) - 11 May 2024
Abstract
Fugitive methane emissions account for a significant proportion of greenhouse gas emissions, and their elimination by catalytic combustion is a relatively easy way to reduce global warming. New and novel reactor designs are being considered for this purpose, but their correct and efficient
[...] Read more.
Fugitive methane emissions account for a significant proportion of greenhouse gas emissions, and their elimination by catalytic combustion is a relatively easy way to reduce global warming. New and novel reactor designs are being considered for this purpose, but their correct and efficient design requires kinetic rate expressions. This paper provides a comprehensive review of the current state of the art regarding kinetic models for precious metal catalysts used for the catalytic combustion of lean methane mixtures. The primary emphasis is on relatively low-temperature operation at atmospheric pressure, conditions that are prevalent in the catalytic destruction of low concentrations of methane in emission streams. In addition to a comprehensive literature search, we illustrate a detailed example of the methodology required to determine an appropriate kinetic model and the constants therein. From the wide body of literature, it is seen that the development of a kinetic model is not necessarily a trivial matter, and it is difficult to generalize. The model, especially the dependence on the water concentration, is a function of not only the active ingredients but also the nature of the support. Kinetic modelling is performed for six catalysts, one commercial and five that were manufactured in our laboratory, for illustration purposes.
Full article
(This article belongs to the Section Computational Catalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Direct Synthesis of Calcium Lactate through the Reaction of Glycerol with Calcium Hydroxide Catalyzed by Bimetallic AuCu/SiO2 Nanocatalysts
by
Changqing Li, Xinyue Cui, Aili Wang, Hengbo Yin, Yuting Li, Qiao Lin and Junjie Guo
Catalysts 2024, 14(5), 318; https://doi.org/10.3390/catal14050318 (registering DOI) - 11 May 2024
Abstract
Bimetallic AuCu/SiO2 nanosized catalysts were prepared using the wet chemical reduction technique. From among Au0.1–1.5Cu10/SiO2 catalysts, the Au0.5Cu10/SiO2 catalyst gave the highest yield of calcium lactate of 87% at a glycerol conversion
[...] Read more.
Bimetallic AuCu/SiO2 nanosized catalysts were prepared using the wet chemical reduction technique. From among Au0.1–1.5Cu10/SiO2 catalysts, the Au0.5Cu10/SiO2 catalyst gave the highest yield of calcium lactate of 87% at a glycerol conversion of 96% when the reaction of glycerol with calcium hydroxide at a mole ratio of calcium hydroxide to glycerol of 0.8:1 was conducted under an anaerobic atmosphere at 200 °C for 2 h. The interactions between metallic Au0 and Cu0 nanoparticles facilitate calcium lactate formation. The simulation of glycerol consumption rate with an empirical power-function reaction kinetics equation yielded a reaction activation energy of 44.3 kJ∙mol−1, revealing that the catalytic reaction of glycerol with calcium hydroxide to calcium lactate can be conducted by overcoming a mild energy barrier. The synthesis of calcium lactate through the catalytic reaction of glycerol with calcium hydroxide on a bimetallic AuCu/SiO2 nanosized catalyst under a safe anaerobic atmosphere is an alternative to the conventional calcium lactate production technique through the reaction of expensive lactic acid with calcium hydroxide.
Full article
(This article belongs to the Special Issue Heterogeneous Catalysis for Environmentally Compatible Reactions and Processes)
Open AccessReview
A Comprehensive Review of Fine Chemical Production Using Metal-Modified and Acidic Microporous and Mesoporous Catalytic Materials
by
Joseph Lantos, Narendra Kumar and Basudeb Saha
Catalysts 2024, 14(5), 317; https://doi.org/10.3390/catal14050317 (registering DOI) - 10 May 2024
Abstract
Fine chemicals are produced in small annual volume batch processes (often <10,000 tonnes per year), with a high associated price (usually >USD 10/kg). As a result of their usage in the production of speciality chemicals, in areas including agrochemicals, fragrances, and pharmaceuticals, the
[...] Read more.
Fine chemicals are produced in small annual volume batch processes (often <10,000 tonnes per year), with a high associated price (usually >USD 10/kg). As a result of their usage in the production of speciality chemicals, in areas including agrochemicals, fragrances, and pharmaceuticals, the need for them will remain high for the foreseeable future. This review article assesses current methods used to produce fine chemicals with heterogeneous catalysts, including both well-established and newer experimental methods. A wide range of methods, utilising microporous and mesoporous catalysts, has been explored, including their preparation and modification before use in industry. Their potential drawbacks and benefits have been analysed, with their feasibility compared to newer, recently emerging catalysts. The field of heterogeneous catalysis for fine chemical production is a dynamic and ever-changing area of research. This deeper insight into catalytic behaviour and material properties will produce more efficient, selective, and sustainable processes in the fine chemical industry. The findings from this article will provide an excellent foundation for further exploration and a critical review in the field of fine chemical production using micro- and mesoporous heterogeneous catalysts.
Full article
(This article belongs to the Special Issue Microporous and Mesoporous Materials for Catalytic Applications)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Promoter Impact on 5Ni/SAPO-5 Catalyst for H2 Production via Methane Partial Oxidation
by
Abdulaziz Al-Anazi, Omer Bellahwel, Kavitha C., Jehad Abu-Dahrieh, Ahmed A. Ibrahim, S. Santhosh, Ahmed E. Abasaeed, Anis H. Fakeeha and Ahmed S. Al-Fatesh
Catalysts 2024, 14(5), 316; https://doi.org/10.3390/catal14050316 - 10 May 2024
Abstract
Compared to steam reforming techniques, partial oxidation of methane (POM) is a promising technology to improve the efficiency of synthesizing syngas, which is a mixture of CO and H2. In this study, partial oxidation of methane (POM) was used to create
[...] Read more.
Compared to steam reforming techniques, partial oxidation of methane (POM) is a promising technology to improve the efficiency of synthesizing syngas, which is a mixture of CO and H2. In this study, partial oxidation of methane (POM) was used to create syngas, a combination of CO and H2, using the SAPO-5-supported Ni catalysts. Using the wetness impregnation process, laboratory-synthesized Ni promoted with Sr, Ce, and Cu was used to modify the SAPO-5 support. The characterization results demonstrated that Ni is appropriate for the POM due to its crystalline structure, improved metal support contact, and increased thermal stability with Sr, Ce, and Cu promoters. During POM at 600 °C, the synthesized 5Ni+1Sr/SAPO-5 catalyst sustained stability for 240 min on stream. While keeping the reactants stoichiometric ratio of (CH4:O2 = 2:1), the addition of Sr promoter and active metal Ni to the SAPO-5 increased the CH4 conversion from 41.13% to 49.11% and improved the H2/CO ratio of 3.33. SAPO-5-supported 5Ni+1Sr catalysts have great potential for industrial catalysis owing to their unique combination of several oxides. This composition not only boosts the catalyst’s activity but also promotes favorable physiochemical properties, resulting in improved production of syngas. Syngas is a valuable intermediate in various industrial processes.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures
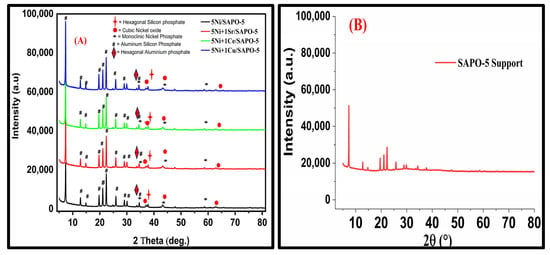
Figure 1
Open AccessArticle
Au Nanoparticles Supported on Hydrotalcite-Based MMgAlOx (M=Cu, Ni, and Co) Composite: Influence of Dopants on the Catalytic Activity for Semi-Hydrogenation of C2H2
by
Xun Sun, Wenrui Lv, Yanan Cheng, Huijuan Su, Libo Sun, Lijun Zhao, Zifan Wang and Caixia Qi
Catalysts 2024, 14(5), 315; https://doi.org/10.3390/catal14050315 - 10 May 2024
Abstract
Semi-hydrogenation of acetylene to ethylene over metal oxide-supported Au nanoparticles is an interesting topic. Here, a hydrotalcite-based MMgAlOx (M=Cu, Ni, and Co) composite oxide was exploited by introducing different Cu, Ni, and Co dopants with unique properties, and then used as support
[...] Read more.
Semi-hydrogenation of acetylene to ethylene over metal oxide-supported Au nanoparticles is an interesting topic. Here, a hydrotalcite-based MMgAlOx (M=Cu, Ni, and Co) composite oxide was exploited by introducing different Cu, Ni, and Co dopants with unique properties, and then used as support to obtain Au/MMgAlOx catalysts via a modified deposition–precipitation method. XRD, BET, ICP-OES, TEM, Raman, XPS, and TPD were employed to investigate their physic-chemical properties and catalytic performances for the semi-hydrogenation of acetylene to ethylene. Generally, the catalytic activity of the Cu-modified Au/CuMgAlOx catalyst was higher than that of the other modified catalysts. The TOR for Au/CuMgAlOx was 0.0598 h−1, which was 30 times higher than that of Au/MgAl2O4. The SEM and XRD results showed no significant difference in structure or morphology after introducing the dopants. These dopants had an unfavorable effect on the Au particle size, as confirmed by the TEM studies. Accordingly, the effects on catalytic performance of the M dopant of the obtained Au/MMgAlOx catalyst were improved. Results of Raman, NH3-TPD, and CO2-TPD confirmed that the Au/CuMgAlOx catalyst had more basic sites, which is beneficial for less coking on the catalyst surface after the reaction. XPS analysis showed that gold nanoparticles exhibited a partially oxidized state at the edges and surfaces of CuMgAlOx. Besides an increased proportion of basic sites on Au/CuMgAlOx catalysts, the charge transfer from nanogold to the Cu-doped matrix support probably played a positive role in the selective hydrogenation of acetylene. The stability and deactivation of Au/CuMgAlOx catalysts were also discussed and a possible reaction mechanism was proposed.
Full article
(This article belongs to the Special Issue Nanomaterials in Catalysis: Design, Characterization and Applications)
►▼
Show Figures

Figure 1
Open AccessArticle
Metal–Organic Framework Fe-BTC as Heterogeneous Catalyst for Electro-Fenton Treatment of Tetracycline
by
Taylor Mackenzie Fisher, Alexsandro J. dos Santos and Sergi Garcia-Segura
Catalysts 2024, 14(5), 314; https://doi.org/10.3390/catal14050314 - 10 May 2024
Abstract
This study explores the use of the iron-containing metal–organic framework (MOF), Basolite®F300, as a heterogeneous catalyst for electrochemically-driven Fenton processes. Electrochemical advanced oxidation processes (EAOPs) have shown promise on the abatement of recalcitrant organic pollutants such as pharmaceuticals. Tetracyclines (TC) are
[...] Read more.
This study explores the use of the iron-containing metal–organic framework (MOF), Basolite®F300, as a heterogeneous catalyst for electrochemically-driven Fenton processes. Electrochemical advanced oxidation processes (EAOPs) have shown promise on the abatement of recalcitrant organic pollutants such as pharmaceuticals. Tetracyclines (TC) are a frequently used class of antibiotics that are now polluting surface water and groundwater sources worldwide. Acknowledging the fast capability of EAOPs to treat persistent pharmaceutical pollutants, we propose an electrochemical Fenton treatment process that is catalyzed by the use of a commercially available MOF material to degrade TC. The efficiency of H2O2 generation in the IrO2/carbon felt setup is highlighted. However, electrochemical oxidation with H2O2 production (ECO-H2O2) alone is not enough to achieve complete TC removal, attributed to the formation of weak oxidant species. Incorporating Basolite®F300 in the heterogeneous electro-Fenton (HEF) process results in complete TC removal within 40 min, showcasing its efficacy. Additionally, this study explores the effect of varying MOF concentrations, indicating optimal removal rates at 100 mg L−1 due to a balance of kinetics and limitation of active sites of the catalysts. Furthermore, the impact of the applied current on TC removal is investigated, revealing a proportional relationship between current and removal rates. The analysis of energy efficiency emphasizes 50 mA as the optimal current, however, balancing removal efficiency with electrical energy consumption. This work highlights the potential of Basolite®F300 as an effective catalyst in the HEF process for pollutant abatement, providing valuable insights into optimizing electrified water treatment applications with MOF nanomaterials to treat organic pollutants.
Full article
(This article belongs to the Section Environmental Catalysis)
►▼
Show Figures
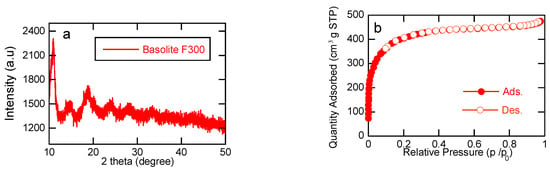
Figure 1
Open AccessArticle
Perovskite Oxide Catalysts for Enhanced CO2 Reduction: Embroidering Surface Decoration with Ni and Cu Nanoparticles
by
Andrea Osti, Lorenzo Rizzato, Jonathan Cavazzani, Ambra Meneghello and Antonella Glisenti
Catalysts 2024, 14(5), 313; https://doi.org/10.3390/catal14050313 - 10 May 2024
Abstract
The imperative reduction of carbon dioxide into valuable fuels stands as a crucial step in the transition towards a more sustainable energy system. Perovskite oxides, with their high compositional and property adjustability, emerge as promising catalysts for this purpose, whether employed independently or
[...] Read more.
The imperative reduction of carbon dioxide into valuable fuels stands as a crucial step in the transition towards a more sustainable energy system. Perovskite oxides, with their high compositional and property adjustability, emerge as promising catalysts for this purpose, whether employed independently or as a supporting matrix for other active metals. In this study, an A-site-deficient La0.9FeO3 perovskite underwent surface decoration with Ni, Cu or Ni + Cu via a citric acid-templated wet impregnation method. Following extensive characterization through XRD, N2 physisorption, H2-TPR, SEM-EDX, HAADF STEM-EDX mapping, CO2-TPD and XPS, the prepared powders underwent reduction under diluted H2 to yield metallic nanoparticles (NPs). The prepared catalysts were then evaluated for CO2 reduction in a CO2/H2 = 1/4 mixture. The deposition of Ni or Cu NPs on the perovskite support significantly enhanced the conversion of CO2, achieving a 50% conversion rate at 500 °C, albeit resulting in only CO as the final product. Notably, the catalyst featuring Ni-Cu co-deposition outperformed in the intermediate temperature range, exhibiting high selectivity for CH4 production around 350 °C. For this latter catalyst, a synergistic effect of the metal–support interaction was evidenced by H2-TPR and CO2-TPD experiments as well as a better nanoparticle dispersion. A remarkable stability in a 20 h time-span was also demonstrated for all catalysts, especially the one with Ni-Cu co-deposition.
Full article
(This article belongs to the Special Issue Advanced Research of Perovskite Materials as Catalysts)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Highly Efficient PtSn/Al2O3 and PtSnZnCa/Al2O3 Catalysts for Ethane Dehydrogenation: Influence of Catalyst Pretreatment Atmosphere
by
Seetharamulu Podila, Abdulrahim A. Al-Zahrani, Muhammad A. Daous and Hesham Alhumade
Catalysts 2024, 14(5), 312; https://doi.org/10.3390/catal14050312 - 9 May 2024
Abstract
Increased demand for ethylene has motivated direct ethane dehydrogenation over Pt-based catalysts. PtSn/γ-Al2O3 and PtSnZnCa/γ-Al2O3 catalysts were investigated with the aim of understanding the effect of the pretreatment environment on the state of dispersed Pt for ethane
[...] Read more.
Increased demand for ethylene has motivated direct ethane dehydrogenation over Pt-based catalysts. PtSn/γ-Al2O3 and PtSnZnCa/γ-Al2O3 catalysts were investigated with the aim of understanding the effect of the pretreatment environment on the state of dispersed Pt for ethane dehydrogenation. The catalysts were prepared by the impregnation method and pretreated in different environments like static air (SA), flowing air (FA), and nitrogen (N2) atmospheres. A comprehensive characterization of the catalysts was performed using Brunauer–Emmett–Teller (BET), X-ray diffraction (XRD), Temperature-Programmed Reduction (TPR), NH3 Temperature-Programmed Desorption (NH3-TPD), X-ray photoelectron spectroscopy (XPS), and Transmission Electron Microscopy (TEM) techniques. The results reveal that the PtSn on Al2O3 catalyst pretreated in the static air environment (PtSn-SA) exhibits 21% ethylene yield with 95% selectivity at 625 °C. XPS analysis found more platinum and tin on the catalyst surface after static air treatment. The overall acidity of the catalysts decreased after thermal treatment in static air. Elemental mapping demonstrated that Pt agglomeration was pronounced in catalysts calcined under flowing air and nitrogen. These factors are responsible for the enhanced activity of the PtSn-SA catalyst compared to the other catalysts. The addition of Zn and Ca to the PtSn catalysts increases the yield of the catalyst calcined in static air (PtSnZnCa-SA). The PtSnZnCa-SA catalyst showed the highest ethylene yield of 27% with 99% selectivity and highly stable activity at 625 °C for 10 h.
Full article
(This article belongs to the Special Issue Sustainable Catalytic Routes for the Production of Green Synthetic Fuels and Other Value-Added Products)
►▼
Show Figures

Figure 1
Open AccessArticle
Remediation of Polycyclic Aromatic Hydrocarbon-Contaminated Soil by Using Activated Persulfate with Carbonylated Activated Carbon Supported Nanoscale Zero-Valent Iron
by
Changzhao Chen, Zhe Yuan, Shenshen Sun, Jiacai Xie, Kunfeng Zhang, Yuanzheng Zhai, Rui Zuo, Erping Bi, Yufang Tao and Quanwei Song
Catalysts 2024, 14(5), 311; https://doi.org/10.3390/catal14050311 - 8 May 2024
Abstract
Soil contamination by polycyclic aromatic hydrocarbons (PAHs) has been an environmental issue worldwide, which aggravates the ecological risks faced by animals, plants, and humans. In this work, the composites of nanoscale zero-valent iron supported on carbonylated activated carbon (nZVI-CAC) were prepared and applied
[...] Read more.
Soil contamination by polycyclic aromatic hydrocarbons (PAHs) has been an environmental issue worldwide, which aggravates the ecological risks faced by animals, plants, and humans. In this work, the composites of nanoscale zero-valent iron supported on carbonylated activated carbon (nZVI-CAC) were prepared and applied to activate persulfate (PS) for the degradation of PAHs in contaminated soil. The prepared nZVI-CAC catalyst was characterized by scanning electron microscopy (SEM), X-ray diffractometer (XRD), Fourier transform infrared spectroscopy (FTIR), and X-ray photoelectron spectroscopy (XPS). It was found that the PS/nZVI-CAC system was superior for phenanthrene (PHE) oxidation than other processes using different oxidants (PS/nZVI-CAC > PMS/nZVI-CAC > H2O2/nZVI-CAC) and it was also efficient for the degradation of other six PAHs with different structures and molar weights. Under optimal conditions, the lowest and highest degradation efficiencies for the selected PAHs were 60.8% and 90.7%, respectively. Active SO4−• and HO• were found to be generated on the surface of the catalysts, and SO4−• was dominant for PHE oxidation through quenching experiments. The results demonstrated that the heterogeneous process using activated PS with nZVI-CAC was effective for PAH degradation, which could provide a theoretical basis for the remediation of PAH-polluted soil.
Full article
(This article belongs to the Special Issue Recent Catalytic Progresses for Environmental Remediation and Pollutant Degradation)
►▼
Show Figures

Figure 1
Open AccessCorrection
Correction: Zhou et al. Removal of Emerging Organic Pollutants by Zeolite Mineral (Clinoptilolite) Composite Photocatalysts in Drinking Water and Watershed Water. Catalysts 2024, 14, 216
by
Pengfei Zhou, Fei Wang, Yanbai Shen, Xinhui Duan, Sikai Zhao, Xiangxiang Chen and Jinsheng Liang
Catalysts 2024, 14(5), 310; https://doi.org/10.3390/catal14050310 - 8 May 2024
Abstract
There was an error in the original publication [...]
Full article
Open AccessFeature PaperArticle
Strong and Hierarchical Ni(OH)2/Ni/rGO Composites as Multifunctional Catalysts for Excellent Water Splitting
by
Lixin Wang, Ailing Song, Yue Lu, Manman Duanmu, Zhipeng Ma, Xiujuan Qin and Guangjie Shao
Catalysts 2024, 14(5), 309; https://doi.org/10.3390/catal14050309 - 7 May 2024
Abstract
The lack of efficient and non-precious metal catalysts poses a challenge for electrochemical water splitting in hydrogen and oxygen evolution reactions. Here, we report on the preparation of growing Ni(OH)2 nanosheets in situ on a Ni and graphene hybrid using supergravity electrodeposition
[...] Read more.
The lack of efficient and non-precious metal catalysts poses a challenge for electrochemical water splitting in hydrogen and oxygen evolution reactions. Here, we report on the preparation of growing Ni(OH)2 nanosheets in situ on a Ni and graphene hybrid using supergravity electrodeposition and the hydrothermal method. The obtained catalyst displays outstanding performance with small overpotentials of 161.7 and 41 mV to acquire current densities of 100 and 10 mA cm−2 on hydrogen evolution reaction, overpotentials of 407 and 331 mV to afford 100 and 50 mA cm−2 on oxygen evolution reaction, and 10 mA·cm−2 at a cell voltage of 1.43 V for water splitting in 1 M KOH. The electrochemical activity of the catalyst is higher than most of the earth-abundant materials reported to date, which is mainly due to its special hierarchical structure, large surface area, and good electrical conductivity. This study provides new tactics for enhancing the catalytic performance of water electrolysis.
Full article
(This article belongs to the Special Issue Hierarchically Catalysts for Water Splitting and Selective Hydrogenation)
►▼
Show Figures

Graphical abstract
Open AccessFeature PaperReview
Dehydration of Methanol to Dimethyl Ether—Current State and Perspectives
by
Lucjan Chmielarz
Catalysts 2024, 14(5), 308; https://doi.org/10.3390/catal14050308 - 7 May 2024
Abstract
The main groups of catalytic materials used in the conversion of methanol to dimethyl ether (the MTD process) were presented with respect to their advantages, disadvantages, and the methods of their modifications, resulting in catalysts with improved activity, selectivity, and stability. In particular,
[...] Read more.
The main groups of catalytic materials used in the conversion of methanol to dimethyl ether (the MTD process) were presented with respect to their advantages, disadvantages, and the methods of their modifications, resulting in catalysts with improved activity, selectivity, and stability. In particular, the effects of strength, surface concentration, and the type of acid sites, the porous structure and morphology of the catalytic materials, the role of catalyst activators, and others, were considered. The prosed mechanisms of the MTD process over various types of catalysts are presented. Moreover, the advantages of membrane reactors for the MTD process are presented and analysed. The perspectives in the development of effective catalysts for the dehydration of methanol to dimethyl ether are presented and discussed.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Influence of Power Fluctuation on Ni-Based Electrode Degradation and Hydrogen Evolution Reaction Performance in Alkaline Water Splitting: Probing the Effect of Renewable Energy on Water Electrolysis
by
Congying Liu, Bing Lin, Hailong Zhang, Yingying Wang, Hangzhou Wang, Junlei Tang and Caineng Zou
Catalysts 2024, 14(5), 307; https://doi.org/10.3390/catal14050307 - 6 May 2024
Abstract
The combination of water electrolysis and renewable energy to produce hydrogen is a promising way to solve the climate and energy crisis. However, the fluctuating characteristics of renewable energy not only present a significant challenge to the use of water electrolysis electrodes, but
[...] Read more.
The combination of water electrolysis and renewable energy to produce hydrogen is a promising way to solve the climate and energy crisis. However, the fluctuating characteristics of renewable energy not only present a significant challenge to the use of water electrolysis electrodes, but also limit the development of the hydrogen production industry. In this study, the effects of three different types of waveforms (square, step, and triangle, which were used to simulate the power input of renewable energy) on the electrochemical catalysis behavior of Ni plate cathodes for HER was investigated. During the test, the HER performance of the Ni cathode increased at first and then slightly decreased. The fluctuating power led to the degradation of the Ni cathode surface, which enhanced the catalysis effect by increasing the catalytic area and the active sites. However, prolonged operation under power fluctuations could have damaged the morphology of the electrode surface and the substances comprising this surface, potentially resulting in a decline in catalytic efficiency. In addition, the electrochemical catalysis behavior of the prepared FeNiMo-LDH@NiMo/SS cathode when subjected to square-wave potential with different fluctuation amplitudes was also extensively studied. A larger amplitude of fluctuating power led to a change in the overpotential and stability of the LDH electrode, which accelerated the degradation of the cathode. This research provides a technological basis for the coupling of water electrolysis and fluctuating renewable energy and thus offers assistance to the development of the “green hydrogen” industry.
Full article
(This article belongs to the Section Electrocatalysis)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Study on NH3-SCR Activity and HCl/H2O Tolerance of Titanate-Nanotube-Supported MnOx-CeO2 Catalyst at Low Temperature
by
Qiulin Wang, Feng Liu, Zhihao Wu, Jing Jin, Xiaoqing Lin, Shengyong Lu and Juan Qiu
Catalysts 2024, 14(5), 306; https://doi.org/10.3390/catal14050306 - 5 May 2024
Abstract
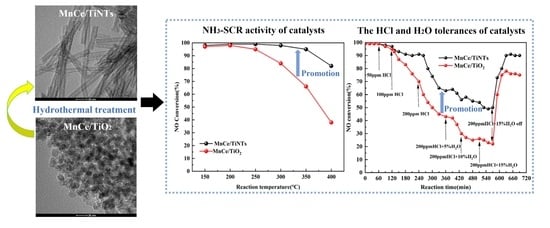
Manganese oxide-cerium oxide supported on titanate nanotubes (i.e., MnCe/TiNTs) were prepared and their catalytic activities towards NH3-SCR of NO were tested. The results indicated that the MnCe/TiNT catalyst can achieve a high NO removal efficiency above 95% within the temperature range
[...] Read more.
Manganese oxide-cerium oxide supported on titanate nanotubes (i.e., MnCe/TiNTs) were prepared and their catalytic activities towards NH3-SCR of NO were tested. The results indicated that the MnCe/TiNT catalyst can achieve a high NO removal efficiency above 95% within the temperature range of 150–350 °C. Even after exposure to a HCl-containing atmosphere for 2 h, the NO removal efficiency of the MnCe/TiNT catalyst maintains at approximately 90% at 150 °C. This is attributed to the large specific surface area as well as the unique hollow tubular structure of TiNTs that exposes more Ce atoms, which preferentially react with HCl and thus protect the active Mn atoms. Moreover, the abundant OH groups on TiNTs serve as Brønsted acid sites and provide H protons to expel Cl atom from the catalyst surface. The irreversible deactivation caused by HCl can be alleviated by H2O. That is because the dissociated adsorption of H2O on TiNTs forms additional OH groups and relieves HCl poisoning.
Full article
(This article belongs to the Special Issue NOx, VOCs (Volatile Organic Compounds) and Soot Emission Control in Catalysis, 2nd Edition)
►▼
Show Figures
Graphical abstract
Open AccessFeature PaperArticle
Binuclear Dioxomolybdenum(VI) Complex Based on Bis(2-pyridinecarboxamide) Ligand as Effective Catalyst for Fuel Desulfurization
by
Fátima Mirante, Catarina N. Dias, André Silva, Sandra Gago and Salete S. Balula
Catalysts 2024, 14(5), 305; https://doi.org/10.3390/catal14050305 - 4 May 2024
Abstract
A binuclear dioxomolybdenum catalyst [(MoO2Cl2)2(L)] (1) (with L (1S,2S)-N,N′-bis(2-pyridinecarboxamide)-1,2-cyclohexane) was prepared and used as catalyst for the desulfurization of a multicomponent model fuel containing the most refractory
[...] Read more.
A binuclear dioxomolybdenum catalyst [(MoO2Cl2)2(L)] (1) (with L (1S,2S)-N,N′-bis(2-pyridinecarboxamide)-1,2-cyclohexane) was prepared and used as catalyst for the desulfurization of a multicomponent model fuel containing the most refractory sulfur compounds in real fuels. This complex was shown to have a high efficiency to oxidize the aromatic benzothiophene derivative compounds present in fuels, mainly using a biphasic 1:1 model fuel/MeOH system. This process conciliates catalytic oxidative and extractive desulfurization, resulting in the oxidation of the sulfur compounds in the polar organic solvent. The oxidative catalytic performance of (1) was shown to be influenced by the presence of water in the system. Using 50% aq. H2O2, it was possible to reuse the catalyst and the extraction solvent, MeOH, during ten consecutive cycles without loss of desulfurization efficiency.
Full article
(This article belongs to the Special Issue Advances in Green Catalysis for Sustainable Organic Synthesis, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
Functionalized Chitosan and Alginate Composite Hydrogel-Immobilized Laccase with Sustainable Biocatalysts for the Effective Removal of Organic Pollutant Bisphenol A
by
Hong Zhang, Xin Zhang, Lei Wang, Bo Wang, Xu Zeng and Bo Ren
Catalysts 2024, 14(5), 304; https://doi.org/10.3390/catal14050304 - 3 May 2024
Abstract
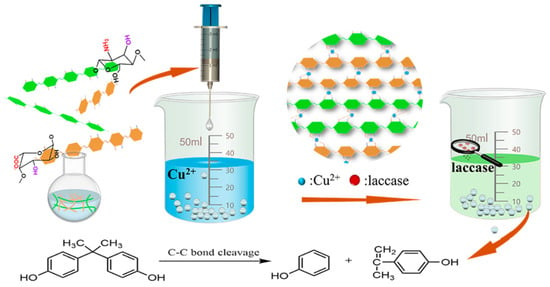
The immobilization of enzymes is an important strategy to improve their stability and reusability. Enzyme immobilization technology has broad application prospects in biotechnology, biochemistry, environmental remediation, and other fields. In this study, composites of chitosan (CS) and sodium alginate (SA) with Cu2+
[...] Read more.
The immobilization of enzymes is an important strategy to improve their stability and reusability. Enzyme immobilization technology has broad application prospects in biotechnology, biochemistry, environmental remediation, and other fields. In this study, composites of chitosan (CS) and sodium alginate (SA) with Cu2+ forming a double-network crosslinked structure of hydrogels were prepared and used for the immobilization of laccase. Fourier infrared spectroscopy, scanning electron microscopy, and X-ray photoelectron spectroscopy tests revealed that laccase molecules were immobilized on the composite hydrogel surface by a covalent bonding method. Compared to free laccase, the pH, temperature, and storage stability of the immobilized laccase were markedly improved. In addition, the immobilized laccase could be easily separated from the reaction system and reused, and it maintained 81.6% of its initial viability after six cycles of use. Bisphenol A (BPA) in polluted water was efficiently degraded using immobilized laccase, and the factors affecting the degradation efficiency were analyzed. Under the optimal conditions, the BPA removal was greater than 82%, and the addition of a small amount of ABTS had a significant effect on BPA degradation, with a removal rate of up to 99.1%. Experimental results indicated that immobilized laccases had enormous potential in actual industrial applications.
Full article
(This article belongs to the Section Biocatalysis)
►▼
Show Figures
Graphical abstract
Open AccessFeature PaperReview
Recent Advances in the Development of Nanocarbon-Based Electrocatalytic/Electrode Materials for Proton Exchange Membrane Fuel Cells: A Review
by
Adelina A. Zasypkina, Nataliya A. Ivanova, Dmitry D. Spasov, Ruslan M. Mensharapov, Matvey V. Sinyakov and Sergey A. Grigoriev
Catalysts 2024, 14(5), 303; https://doi.org/10.3390/catal14050303 - 3 May 2024
Abstract
The global issue for proton exchange membrane fuel cell market development is a reduction in the device cost through an increase in efficiency of the oxygen reduction reaction occurring at the cathode and an extension of the service life of the electrochemical device.
[...] Read more.
The global issue for proton exchange membrane fuel cell market development is a reduction in the device cost through an increase in efficiency of the oxygen reduction reaction occurring at the cathode and an extension of the service life of the electrochemical device. Losses in the fuel cell performance are due to various degradation mechanisms in the catalytic layers taking place under conditions of high electric potential, temperature, and humidity. This review is devoted to recent advances in the field of increasing the efficiency and durability of electrocatalysts and other electrode materials by introducing structured carbon components into their composition. The main synthesis methods, physicochemical and electrochemical properties of materials, and performance of devices on their basis are presented. The main correlations between the composition and properties of structured carbon electrode materials, which can provide successful solutions to the highlighted issues, are revealed.
Full article
(This article belongs to the Special Issue Feature Review Papers in Electrocatalysis)
►▼
Show Figures
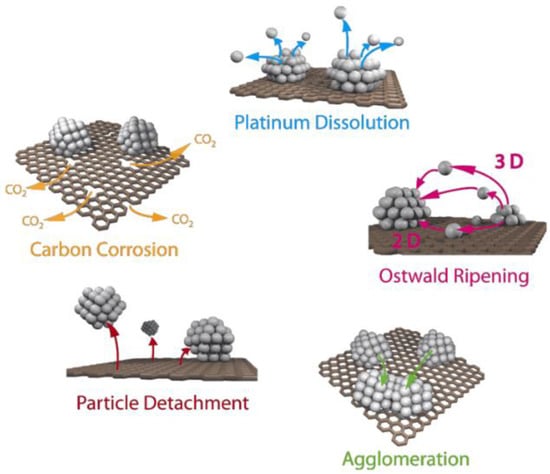
Figure 1
Open AccessArticle
Electrochemical Properties of NiCo2O4/WO3/Activated Carbon Wheat Husk Nano-Electrocatalyst for Methanol and Ethanol Oxidation
by
Mohammad Bagher Askari, Parisa Salarizadeh, Seyed Rouhollah Samareh Hashemi, Mohsen Shojaeifar and Sadegh Azizi
Catalysts 2024, 14(5), 302; https://doi.org/10.3390/catal14050302 - 2 May 2024
Abstract
It is common to use efficient catalysts in the anodes and cathodes of methanol and ethanol fuel cells, such as platinum and ruthenium. However, due to their expansivity and rarity, finding a suitable alternative is important. In this work, multi-component catalysts consisting of
[...] Read more.
It is common to use efficient catalysts in the anodes and cathodes of methanol and ethanol fuel cells, such as platinum and ruthenium. However, due to their expansivity and rarity, finding a suitable alternative is important. In this work, multi-component catalysts consisting of tungsten oxide, nickel cobaltite, and activated carbon were synthesized through the hydrothermal method. The performance of catalysts in the processes of methanol and ethanol oxidation reactions (MOR and EOR) were investigated. The addition of activated carbon obtained from wheat husk, with an excellent active surface and acceptable electrical conductivity, to the matrix of the catalyst significantly facilitated the oxidation process of alcohols and enhanced the efficiency of the catalyst. The physical and electrochemical characterization of the NiCo2O4/WO3 hybridized with the wheat husk-derived activated carbon (ACWH) catalyst indicated its successful synthesis and good performance in the alcohol oxidation process. NiCo2O4/WO3/ACWH with an oxidation current density of 63.39 mA/cm2 at the peak potential of 0.58 V (1.59 vs. RHE), a cyclic stability of 98.6% in the methanol oxidation reaction (MOR) and 27.98 mA/cm2 at the peak potential of 0.67 V (1.68 vs. RHE), and a cyclic stability of 95.7% in the ethanol oxidation reaction (EOR) process can be an interesting option for application in the anodes of alcohol fuel cells.
Full article
(This article belongs to the Section Catalysis for Sustainable Energy)
►▼
Show Figures

Graphical abstract
Open AccessEditorial
New Trends in the Use of Catalysts for Biofuel and Bioproduct Generation
by
José María Encinar Martín and Sergio Nogales-Delgado
Catalysts 2024, 14(5), 301; https://doi.org/10.3390/catal14050301 - 2 May 2024
Abstract
Green technologies are gaining a vital role in the energy and industrial fields, as society faces challenges such as geopolitical conflicts and pollution related to the exploitation of petroleum resources [...]
Full article
(This article belongs to the Special Issue New Trends in the Use of Catalysts for Biofuel and Bioproduct Generation)
►▼
Show Figures
Figure 1

Journal Menu
► ▼ Journal Menu-
- Catalysts Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Batteries, Catalysts, Energies, Hydrogen, Sustainability
Preparation, Storage, and Transportation of Green Hydrogen and Multi-Scenario Application Technologies
Topic Editors: Weihua Cai, Chao Xu, Zhonghao Rao, Fuqiang Wang, Ming GaoDeadline: 30 June 2024
Topic in
Catalysts, Coatings, Crystals, Energies, Materials, Nanomaterials
Interfacial Bonding Design and Applications in Structural and Functional Materials
Topic Editors: Junlei Qi, Pengcheng Wang, Yaotian YanDeadline: 20 July 2024
Topic in
Energies, Catalysts, Fermentation, Processes, Waste
Valorizing Waste through Thermal and Biological Processes for Sustainable Energy Production
Topic Editors: Margarida Gonçalves, Cândida VilarinhoDeadline: 31 August 2024
Topic in
Cancers, Catalysts, Current Oncology, Plasma, Sci
Advances in Low-Temperature Plasma Cancer Therapy
Topic Editors: Michael Keidar, Li Lin, Dayun YanDeadline: 20 September 2024

Conferences
Special Issues
Special Issue in
Catalysts
Heterogeneous Catalysis for Selective Hydrogenation
Guest Editors: Shihui Zou, Juanjuan LiuDeadline: 15 May 2024
Special Issue in
Catalysts
Advances in Photocatalytic and Photothermal Catalytic CO2 Reduction
Guest Editor: Fernando FresnoDeadline: 6 June 2024
Special Issue in
Catalysts
Modern Catalytic Reactor: From Active Center to Application Tests, 2nd Edition
Guest Editors: Anna Gancarczyk, Agnieszka CiemięgaDeadline: 15 June 2024
Special Issue in
Catalysts
Visible Light Photocatalysis: Mechanisms and Applications
Guest Editors: Kunlei Wang, Zhishun Wei, Zuzanna BielanDeadline: 26 June 2024
Topical Collections
Topical Collection in
Catalysts
Photocatalytic Water Splitting
Collection Editors: Weilong Shi, Guigao Liu
Topical Collection in
Catalysts
Catalytic Conversion of Biomass to Bioenergy
Collection Editors: Sergio Nogales Delgado, Juan Félix González, Simona M. Coman