- Article
Radiosurgery in Upper Tract Urothelial Carcinoma (UTUC)
- Michael Staehler,
- Iulia Blajan and
- Alexander Muacevic
- + 10 authors
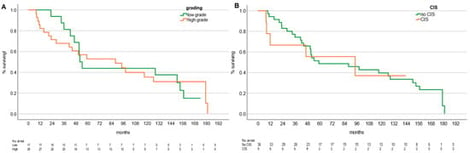
Objectives Radiosurgery (RS) using high-dose hypo-fractionated radiation is a novel treatment for renal cell carcinoma. This study investigates long-term oncological outcomes of RS for upper tract urothelial carcinoma (UTUC) in patients unfit for surgery or with a solitary kidney. Methods We performed a retrospective analysis of three prospective institutional databases from tertiary referral centers, including patients with UTUC treated with RS between 2008 and 2024. Results The study included 45 patients with a median age of 71.9 years (range 47.8–89.1). Of these, 28 had high-grade disease, and 9 carcinoma in situ (CIS). Twenty-eight patients had a solitary kidney. The median RS dose per fraction was 25 Gy (range 10–25). The median number of fractions was 1 (range 1–4 fractions). The median follow-up was 27.6 months (range 3.1–150.3), and the median overall survival (OS) was 91.7 months (95% CI 72.0–111.5). OS did not correlate with tumor grade or CIS. The median OS for low-grade patients was 52.06 months, while for high-grade patients, it was 87.90 months. Treatment response included complete response in 37 patients (82.2%), partial response in 2 (4.4%), stable disease in 2 (4.4%), and progression in 2 (4.4%). The median baseline and follow-up CKD-EPI were 64.2 and 53.5 mL/min/1.73 m2, respectively. Three patients (6.7%) required hemodialysis. Conclusions RS for UTUC is an effective local ablation therapy with durable long-term control and should be considered in patients not suitable for surgical approaches. It should be evaluated as a potential new standard of care in prospective studies in conjunction with peri-interventional systemic therapy.
19 March 2026