Applications of Magnetic Nanomaterials
A topical collection in Nanomaterials (ISSN 2079-4991).
Submission Status:
Closed
|
Viewed by 92031
Share This Topical Collection
Editor
Topical Collection Information
Dear Colleagues,
Magnetic nanomaterials represent one of the most important and emerging classes of materials in nanotechnology due to a range of potential applications. These nanomaterials are used in magnetic data storage, catalysis, magnetic separation, sensing, waste water treatment, and in various biomedical applications. For example, magnetic nanoparticles have been utilized as contrast agents for magnetic resonance imaging (MRI). When exposed to an alternating magnetic field, magnetic nanoparticles can serve as powerful heat sources, destroying tumor cells, which enabled to use these nanomaterials in cancer hyperthermia therapy. Magnetic nanomaterials have also been used as drug delivery agents, which can be localized in the body at a site of interest using an external magnetic field.
This collection will be focused on prospective applications of magnetic nanomaterials in materials science, chemistry, physics, biology and medicine.
Prof. Yurii K. Gun'ko
Guest Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Nanomaterials is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript.
The Article Processing Charge (APC) for publication in this open access journal is 2400 CHF (Swiss Francs).
Submitted papers should be well formatted and use good English. Authors may use MDPI's
English editing service prior to publication or during author revisions.
Keywords
- magnetic nanoparticles
- magnetic data storage
- magnetic separation
- sensing
- catalysis
- nanomedicine
- MRI
- magnetic hyperthermia
Published Papers (15 papers)
Open AccessArticle
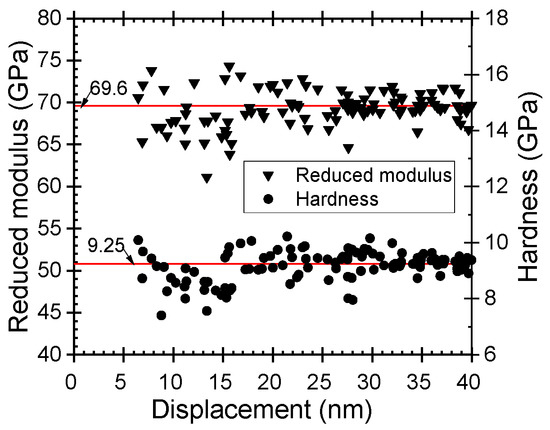
Mechanical and Magnetic Properties of Double Layered Nanostructures of Tin and Zirconium Oxides Grown by Atomic Layer Deposition
by
Aile Tamm, Helle-Mai Piirsoo, Taivo Jõgiaas, Aivar Tarre, Joosep Link, Raivo Stern and Kaupo Kukli
Cited by 4 | Viewed by 3125
Abstract
Double layered stacks of ZrO
2 and SnO
2 films, aiming at the synthesis of thin magnetic and elastic material layers, were grown by atomic layer deposition to thicknesses in the range of 20–25 nm at 300 °C from ZrCl
4, SnI
[...] Read more.
Double layered stacks of ZrO
2 and SnO
2 films, aiming at the synthesis of thin magnetic and elastic material layers, were grown by atomic layer deposition to thicknesses in the range of 20–25 nm at 300 °C from ZrCl
4, SnI
4, H
2O, and O
3 as precursors. The as-deposited nanostructures consisted of a metastable tetragonal polymorph of ZrO
2, and a stable tetragonal phase of SnO
2, with complementary minor reflections from the orthorhombic polymorph of SnO
2. The hardness and elastic modulus of the stacks depended on the order of the constituent oxide films, reaching 15 and 171 GPa, respectively, in the case of top SnO
2 layers. Nonlinear saturative magnetization could be induced in the stacks with coercive fields up to 130 Oe.
Full article
►▼
Show Figures
Open AccessArticle
Magnetic Mg-Fe/LDH Intercalated Activated Carbon Composites for Nitrate and Phosphate Removal from Wastewater: Insight into Behavior and Mechanisms
by
Omar Alagha, Mohammad Saood Manzar, Mukarram Zubair, Ismail Anil, Nuhu Dalhat Mu’azu and Aleem Qureshi
Cited by 105 | Viewed by 8404
Abstract
This experimental work focused on the synthesis, characterization, and testing of a unique, magnetically separable, and eco-friendly adsorbent composite material for the advanced treatment and efficient removal of nitrate and phosphate pollutants from wastewater. The MgAl-augmented double-layered hydroxide (Mg-Fe/LDH) intercalated with sludge-based activated
[...] Read more.
This experimental work focused on the synthesis, characterization, and testing of a unique, magnetically separable, and eco-friendly adsorbent composite material for the advanced treatment and efficient removal of nitrate and phosphate pollutants from wastewater. The MgAl-augmented double-layered hydroxide (Mg-Fe/LDH) intercalated with sludge-based activated carbon (SBAC-MgFe) composites were characterized by FT-IR, XRD, BET, VSM, SEM, and TEM techniques, revealing homogeneous and efficient dispersion of MgFe/LDH within the activated carbon (AC) matrix, a highly mesoporous structure, and superparamagnetic characteristics. The initial solution pH, adsorbent dose, contact time, and temperature parameters were optimized in order to reach the best removal performance for both pollutants. The maximum adsorption capacities of phosphate and nitrate were found to be 110 and 54.5 mg/g, respectively. The competition between phosphate and coexisting ions (Cl
−, CO
32−, and SO
42−) was studied and found to be remarkably lower in comparison with the nitrate adsorption. The adsorption mechanisms were elucidated by kinetic, isotherm, thermodynamic modeling, and post-adsorption characterizations of the composite. Modeling and mechanistic studies demonstrated that physisorption processes such as electrostatic attraction and ion exchange mainly governed the nitrate and phosphate adsorption. The composite indicated an outstanding regeneration performance even after five sequences of adsorption/desorption cycles. The fabricated composite with magnetically separable characteristics can be used as a promising adsorbent for the removal of phosphate and nitrate pollutants from wastewater.
Full article
►▼
Show Figures
Open AccessArticle
Understanding Magnetization Dynamics of a Magnetic Nanoparticle with a Disordered Shell Using Micromagnetic Simulations
by
David Aurélio and Jana Vejpravova
Cited by 14 | Viewed by 5812
Abstract
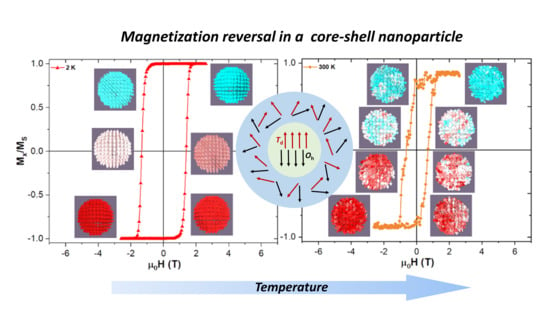
Spin disorder effects influence magnetization dynamics and equilibrium magnetic properties of real nanoparticles (NPs). In this work, we use micromagnetic simulations to try to better understand these effects, in particular, on how the magnetization reversal is projected in the character of the hysteresis
[...] Read more.
Spin disorder effects influence magnetization dynamics and equilibrium magnetic properties of real nanoparticles (NPs). In this work, we use micromagnetic simulations to try to better understand these effects, in particular, on how the magnetization reversal is projected in the character of the hysteresis loops at different temperatures. In our simulation study, we consider a prototype NP adopting a magnetic core-shell model, with magnetically coherent core and somewhat disordered shell, as it is one of the common spin architectures in real NPs. The size of the core is fixed to 5.5 nm in diameter and the shell thickness ranges from 0.5 nm to 3 nm. As a starting point in the simulations, we used typical experimental values obtained for a cobalt ferrite NP of a comparable size investigated previously. The simulations enabled us to study systematically the macrospin dynamics of the prototype NP and to address the interplay between the magnetic anisotropies of the core and the shell, respectively. We also demonstrate how the computational time step, run time, damping parameter, and thermal field influence the simulation results. In agreement with experimental studies, we observed that the direction and magnitude of the shell anisotropy influences the effective magnetic size of the core in the applied magnetic field. We conclude that micromagnetic simulations, in spite of being designed for much larger scales are a useful toolbox for understanding the magnetization processes within a single domain NP with an ordered spin structure in the core and partially disordered spins in the shell.
Full article
►▼
Show Figures
Open AccessArticle
Biophysical Characterization of (Silica-coated) Cobalt Ferrite Nanoparticles for Hyperthermia Treatment
by
Niklas Lucht, Ralf P. Friedrich, Sebastian Draack, Christoph Alexiou, Thilo Viereck, Frank Ludwig and Birgit Hankiewicz
Cited by 26 | Viewed by 6447
Abstract
Magnetic hyperthermia is a technique that describes the heating of material through an external magnetic field. Classic hyperthermia is a medical condition where the human body overheats, being usually triggered by a heat stroke, which can lead to severe damage to organs and
[...] Read more.
Magnetic hyperthermia is a technique that describes the heating of material through an external magnetic field. Classic hyperthermia is a medical condition where the human body overheats, being usually triggered by a heat stroke, which can lead to severe damage to organs and tissue due to the denaturation of cells. In modern medicine, hyperthermia can be deliberately induced to specified parts of the body to destroy malignant cells. Magnetic hyperthermia describes the way that this overheating is induced and it has the inherent advantage of being a minimal invasive method when compared to traditional surgery methods. This work presents a particle system that offers huge potential for hyperthermia treatments, given its good loss value, i.e., the particles dissipate a lot of heat to their surroundings when treated with an ac magnetic field. The measurements were performed in a low-cost custom hyperthermia setup. Additional toxicity assessments on Jurkat cells show a very low short-term toxicity on the particles and a moderate low toxicity after two days due to the prevalent health concerns towards nanoparticles in organisms.
Full article
►▼
Show Figures
Open AccessArticle
Dynamics of Magnetic Fluids in Crossed DC and AC Magnetic Fields
by
Alexander Pshenichnikov, Alexander Lebedev and Alexey O. Ivanov
Cited by 10 | Viewed by 3609
Abstract
In this study, we derived the equations describing the dynamics of a magnetic fluid in crossed magnetic fields (bias and alternating probe fields), considering the field dependence of the relaxation times, interparticle interactions, and demagnetizing field has been derived. For a monodisperse fluid,
[...] Read more.
In this study, we derived the equations describing the dynamics of a magnetic fluid in crossed magnetic fields (bias and alternating probe fields), considering the field dependence of the relaxation times, interparticle interactions, and demagnetizing field has been derived. For a monodisperse fluid, the dependence of the output signal on the bias field and the probe field frequency was constructed. Experimental studies were conducted in a frequency range up to 80 kHz for two samples of fluids based on magnetite nanoparticles and kerosene. The first sample had a narrow particle size distribution, low-energy magneto dipole interactions, and weak dispersion of dynamic susceptibility. The second sample had a broad particle size distribution, high-energy magneto dipole interactions, and strong dispersion of dynamic susceptibility. In the first case, the bias field led to the appearance of short chains. In the second case, we found quasi-spherical clusters with a characteristic size of 100 nm. The strong dependence of the output signal on the particle size allowed us to use the crossed field method to independently estimate the maximum diameter of the magnetic core of particles.
Full article
►▼
Show Figures
Open AccessArticle
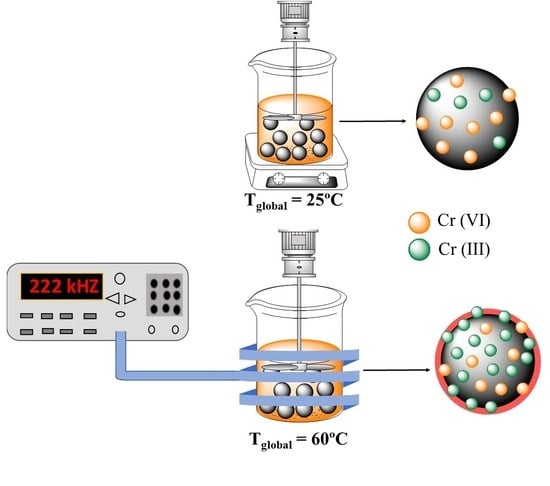
Improvement in Heavy Metal Removal from Wastewater Using an External Magnetic Inductor
by
Fernanda Lyzeth Rivera, Francisco Javier Palomares, Pilar Herrasti and Eva Mazario
Cited by 35 | Viewed by 3966
Abstract
Magnetite nanoparticles (Fe
3O
4) of 12 ± 4 nm diameter are electrochemically synthesized for the adsorption and magnetic harvesting of Cr(VI) from contaminated simulated solutions. The removal of Cr(VI) from aqueous media follows pseudo-second-order kinetics. The adsorption efficiency is evaluated
[...] Read more.
Magnetite nanoparticles (Fe
3O
4) of 12 ± 4 nm diameter are electrochemically synthesized for the adsorption and magnetic harvesting of Cr(VI) from contaminated simulated solutions. The removal of Cr(VI) from aqueous media follows pseudo-second-order kinetics. The adsorption efficiency is evaluated in three different scenarios. In standard conditions, i.e., at room temperature; in a thermal bath working at 60 °C, where the temperature could be considered homogeneous within the solution; and finally, under magnetic induction heating, while adjusting the frequency and magnetic field used to attain the same temperature as in the bath experiments. Two benefits of using a magnetic inductor are demonstrated. First, the removal efficiency is almost doubled in comparison to that of the room temperature experiments, and it is higher by 30% compared to that of the bath setup. At the same time as the adsorption occurs, a redox reaction occurs on the surface of the nanoparticles, and Cr(VI), the predominant species in the contaminated solution, is significantly reduced to Cr(III). Through X-ray photoelectron spectroscopy, it is shown that a greater reduction effect is achieved when working in induction conditions than at room temperature. This is the first time that this synergistic effect using magnetic induction heating has been demonstrated for heavy metal decontamination of wastewater.
Full article
►▼
Show Figures
Open AccessArticle
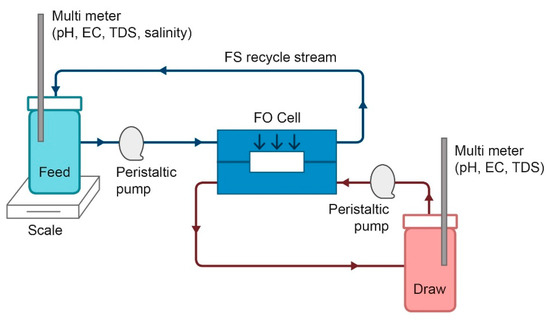
Synthesis of Poly-Sodium-Acrylate (PSA)-Coated Magnetic Nanoparticles for Use in Forward Osmosis Draw Solutions
by
Irena Ban, Sabina Markuš, Sašo Gyergyek, Miha Drofenik, Jasmina Korenak, Claus Helix-Nielsen and Irena Petrinić
Cited by 38 | Viewed by 4899
Abstract
The synthesis of magnetic nanoparticles (MNPs) coated with hydrophilic poly-sodium-acrylate (PSA) ligands was studied to assess PSA-MNP complexes as draw solution (DS) solutes in forward osmosis (FO). For MNP-based DS, the surface modification and the size of the MNPs are two crucial factors
[...] Read more.
The synthesis of magnetic nanoparticles (MNPs) coated with hydrophilic poly-sodium-acrylate (PSA) ligands was studied to assess PSA-MNP complexes as draw solution (DS) solutes in forward osmosis (FO). For MNP-based DS, the surface modification and the size of the MNPs are two crucial factors to achieve a high osmolality. Superparamagnetic nanoparticles (NP) with functional groups attached may represent the ideal DS where chemical modifications of the NPs can be used in optimizing the DS osmolality and the magnetic properties allows for efficient recovery (DS re-concentration) using an external magnetic field. In this study MNPs with diameters of 4 nm have been prepared by controlled chemical co-precipitation of magnetite phase from aqueous solutions containing suitable salts of Fe
2+ and Fe
3+ under inert atmosphere and a pure magnetite phase could be verified by X-ray diffraction. Magnetic colloid suspensions containing PSA-coated MNPs with three different molar ratios of PSA:MNP = 1:1, 1:2 and 1:3 were prepared and assessed in terms of osmotic pressure, aggregation propensity and magnetization. Fourier Transform Infrared Spectroscopy (FTIR) confirmed the presence of PSA on coated MNPs and pristine PSA-MNPs with a molar ratio PSA:MNP = 1:1 exhibited an osmotic pressure of 30 bar. Molar ratios of PSA:MNP = 1:2 and 1:3 lead to the formation of less stabile magnetic colloid solutions, which led to the formation of aggregates with larger average hydrodynamic sizes and modest osmotic pressures (5.5 bar and 0.2 bar, respectively). After purification with ultrafiltration, the 1:1 nanoparticles exhibited an osmotic pressure of 9 bar with no aggregation and a sufficient magnetization of 25 emu/g to allow for DS regeneration using an external magnetic field. However, it was observed that the amount of PSA molecules attached to the MNPs decreased during DS recycling steps, leaving only strong chelate-bonded core-shell PSA as coating on the MNPs. This demonstrates the crucial role of MNP coating robustness in designing an efficient MNP-based DS for FO.
Full article
►▼
Show Figures
Open AccessArticle
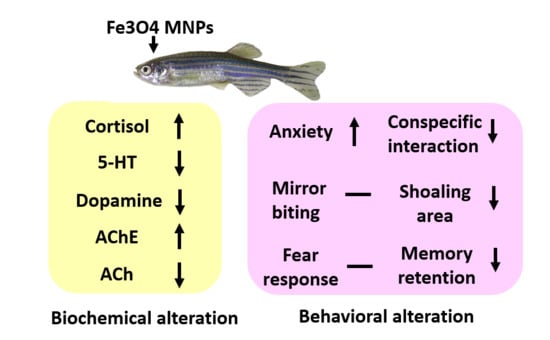
Ecotoxicity Assessment of Fe3O4 Magnetic Nanoparticle Exposure in Adult Zebrafish at an Environmental Pertinent Concentration by Behavioral and Biochemical Testing
by
Nemi Malhotra, Jung-Ren Chen, Sreeja Sarasamma, Gilbert Audira, Petrus Siregar, Sung-Tzu Liang, Yu-Heng Lai, Geng-Ming Lin, Tzong-Rong Ger and Chung-Der Hsiao
Cited by 47 | Viewed by 7297
Abstract
Magnetic Nanoparticles (MNPs) are widely being investigated as novel promising multifunctional agents, specifically in the fields of development for theranostics, electronics, waste water treatment, cosmetics, and energy storage devices. Unique, superior, and indispensable properties of magnetization, heat transfer, and melting temperature make MNPs
[...] Read more.
Magnetic Nanoparticles (MNPs) are widely being investigated as novel promising multifunctional agents, specifically in the fields of development for theranostics, electronics, waste water treatment, cosmetics, and energy storage devices. Unique, superior, and indispensable properties of magnetization, heat transfer, and melting temperature make MNPs emerge in the field of therapeutics in future healthcare industries. However, MNPs ecotoxicity as well as behavioral toxicity is still unexplored. Ecotoxicity analysis may assist investigate MNPs uptake mechanism and its influence on bioavailability under a given set of environmental factors, which can be followed to investigate the biomagnification of MNPs in the environment and health risk possessed by them in an ecological food chain. In this study, we attempted to determine the behavioral changes in zebrafishes at low (1 ppm) or high (10 ppm) concentration levels of Fe
3O
4 MNPs. The synthesized Fe
3O
4 MNPs sized at 15 nm were characterized by the transmission electron microscope (TEM), the superconducting quantum interference device (SQUID) magnetometer, and the multiple behavior tests for novel tank, mirror biting, conspecific social interaction, shoaling, circadian rhythm, and short-term memory of zebrafish under MNPs chronic exposure were demonstrated. Low concentration MNP exposure did not trigger alteration for majority behavioral and biochemical tests in adult zebrafish. However, tight shoal groups were observed at a high concentration of MNPs exposure along with a modest reduction in fish exploratory behavior and a significant reduction in conspecific social interaction behavior. By using enzyme-linked immunosorbent assays (ELISA), we found a high dose of MNPs exposure significantly elevated cortisol, acetylcholine, and catalase levels while reducing serotonin, acetylcholine esterase, and dopamine levels in the brain. Our data demonstrates chronic MNPs exposure at an environmentally-relevant dose is relatively safe by supporting evidence from an array of behavioral and biochemical tests. This combinational approach using behavioral and biochemical tests would be helpful for understanding the MNPs association with anticipated colloids and particles effecting bioavailability and uptake into cells and organisms.
Full article
►▼
Show Figures
Open AccessArticle
A Novel Coloration of Polyester Fabric through Green Silver Nanoparticles (G-AgNPs@PET)
by
K. M. Faridul Hasan, Md. Nahid Pervez, Md. Eman Talukder, Mst. Zakia Sultana, Sakil Mahmud, Md. Mostakim Meraz, Vipul Bansal and Cao Genyang
Cited by 99 | Viewed by 7560
Abstract
This paper reports a novel route for the coloration of polyester fabric with green synthesized silver nanoparticles (G-AgNPs@PET) using chitosan as a natural eco-friendly reductant. The formation of AgNPs was confirmed by UV-visible spectroscopy. The morphologies and average particles size of G-AgNPs was
[...] Read more.
This paper reports a novel route for the coloration of polyester fabric with green synthesized silver nanoparticles (G-AgNPs@PET) using chitosan as a natural eco-friendly reductant. The formation of AgNPs was confirmed by UV-visible spectroscopy. The morphologies and average particles size of G-AgNPs was investigated by transmission electron microscope (TEM) analysis. The uniform deposition of G-AgNPs on the PET fabric surface was confirmed with scanning electron microscopy (SEM) and Fourier transform infrared (FT-IR) spectroscopy. The thermal properties were investigated using a thermogravimetric analyzer (TGA). The coloration and fastness properties of fabric were found to be significantly improved, a result related to the surface plasmon resonance of G-AgNPs. The antibacterial properties of fabric were also found to be excellent as more than 80% bacterial reduction was noticed even after 10 washing cycles. Overall, the proposed coating process using green nanoparticles can contribute to low-cost production of sustainable textiles.
Full article
►▼
Show Figures
Open AccessArticle
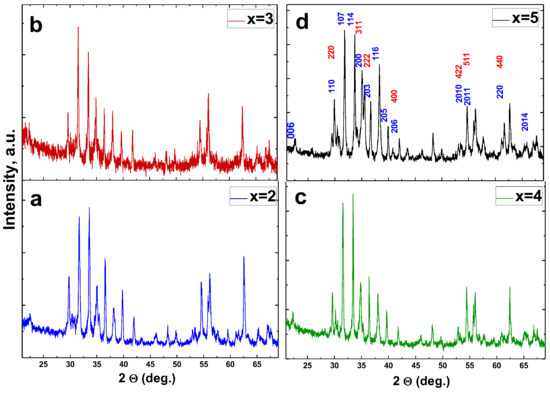
Correlation Between Composition and Electrodynamics Properties in Nanocomposites Based on Hard/Soft Ferrimagnetics with Strong Exchange Coupling
by
Munirah Abdullah Almessiere, Alex V. Trukhanov, Yassine Slimani, K.Y. You, Sergei V. Trukhanov, Ekaterina L. Trukhanova, F. Esa, A. Sadaqat, K. Chaudhary, Maxim Zdorovets and Abdulhadi Baykal
Cited by 247 | Viewed by 8193
Abstract
In this work, Sr
0.3Ba
0.4Pb
0.3Fe
12O
19/(CuFe
2O
4)
x (x = 2, 3, 4, and 5) as strongly exchange-coupled nanosized ferrites were fabricated using a one-pot sol–gel combustion method (citrate sol-gel method). The
[...] Read more.
In this work, Sr
0.3Ba
0.4Pb
0.3Fe
12O
19/(CuFe
2O
4)
x (x = 2, 3, 4, and 5) as strongly exchange-coupled nanosized ferrites were fabricated using a one-pot sol–gel combustion method (citrate sol-gel method). The X-ray diffraction (XRD) powder patterns of the products confirmed the occurrence of pure, exchange-coupled ferrites. Frequency dependencies of the microwave characteristics (MW) were investigated using a co-axial method. The non-linear behavior of the MW with the composition transformation may be due to different degrees of Fe ion oxidation on the spinel/hexaferrite grain boundaries and strong exchange coupling during the hard and soft phases.
Full article
►▼
Show Figures
Open AccessArticle
Manganese/Yttrium Codoped Strontium Nanohexaferrites: Evaluation of Magnetic Susceptibility and Mossbauer Spectra
by
Munirah Abdullah Almessiere, Yassine Slimani, Hakan Güngüneş, Abdulhadi Baykal, S.V. Trukhanov and A.V. Trukhanov
Cited by 123 | Viewed by 5910
|
Correction
Abstract
Manganese (Mn)- and yttrium (Y)-substituted Sr-nanohexaferrites (MYSNHFs) of composition Sr
1−xMn
xFe
12−xY
xO
19 (with 0.0 ≤ x ≤ 0.5) were prepared by citrate sol-gel autocombustion method. As-prepared MYSNHFs were characterized via diverse analytical techniques to determine the
[...] Read more.
Manganese (Mn)- and yttrium (Y)-substituted Sr-nanohexaferrites (MYSNHFs) of composition Sr
1−xMn
xFe
12−xY
xO
19 (with 0.0 ≤ x ≤ 0.5) were prepared by citrate sol-gel autocombustion method. As-prepared MYSNHFs were characterized via diverse analytical techniques to determine the influence of Mn and Y cosubstitution on their microstructures and magnetic properties.
57Fe Mössbauer spectra of the MYSNHFs were used to evaluate the variation in the line width, isomer shift, quadrupole splitting, and hyperfine magnetic field values. It was shown that the dopant ions could preferentially occupy the 12k, 4f
2, and 2b sites. Furthermore, the observed shift in the blocking temperatures of the studied MYSNHFs towards lower values with rising Mn
2+ and Y
3+ contents was attributed to the overall particles size reduction. Meanwhile, the AC susceptibility of the proposed MYSNHFs revealed that the magnetic interactions were weakened with the increase in dopant contents which was ascribed to the replacement of both Sr
2+ and Fe
3+ ions by the Mn
2+ and Y
3+ dopants.
Full article
►▼
Show Figures
Open AccessArticle
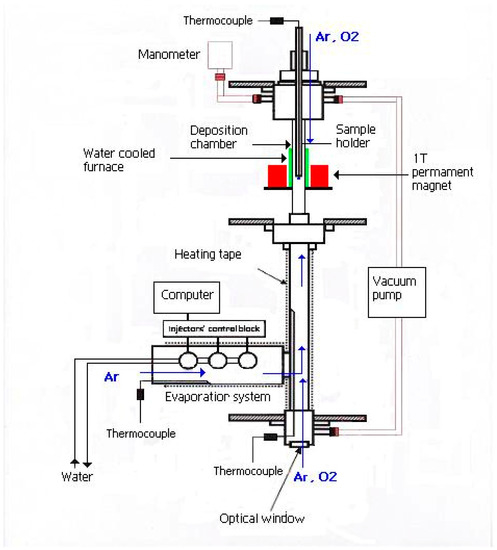
Deposition of Magnetite Nanofilms by Pulsed Injection MOCVD in a Magnetic Field
by
Anna Zukova, Arunas Teiserskis, Yuliya Rohava, Alexander V. Baranov, Sebastiaan Van Dijken and Yurii K. Gun’ko
Cited by 5 | Viewed by 4878
Abstract
This report is on the growth of Fe
3O
4 nanofilms on Al
2O
3(0001) and MgO(001) substrates with and without the presence of an external magnetic field using a pulsed injection metallorganic chemical vapour deposition (PI MOCVD) technique. The
[...] Read more.
This report is on the growth of Fe
3O
4 nanofilms on Al
2O
3(0001) and MgO(001) substrates with and without the presence of an external magnetic field using a pulsed injection metallorganic chemical vapour deposition (PI MOCVD) technique. The effects of growing magnetic oxide nanofilms in a 1 T field have been examined using various instrumental methods. It was found that the application of a magnetic field during PI MOCVD does not drastically alter the crystalline texture, surface morphology, and film thickness, but it significantly modifies the Fe
3O
4 film magnetisation and coercive field. Moreover, it was shown that the application of a 1 T field during the cooling of the sample also improves the magnetic properties. We believe that the large external field orients the magnetic spin structure at high temperatures (during growth or the initial stages of cool down) and that cooling through local magnetic ordering temperatures at Fe
3O
4 defect sites subsequently favours a ferromagnetic spin alignment. The control of magnetic properties of magnetite nanofilms by the application of magnetic fields during growth opens up new routes towards the fabrication and application of magnetic thin film devices.
Full article
►▼
Show Figures
Open AccessArticle
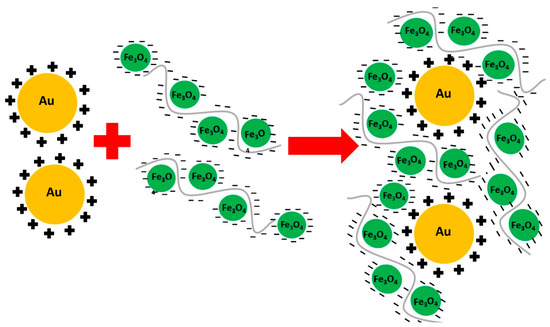
Polyelectrolyte-Stabilised Magnetic-Plasmonic Nanocomposites
by
Shelley Stafford, Coralie Garnier and Yurii K. Gun’ko
Cited by 3 | Viewed by 4938
Abstract
In this work, new magnetic-plasmonic nanocomposites have been developed through the use of two complementary polyelectrolytes–polystyrene sulfonate (PSS) and poly(allylamine hydrochloride) (PAH). PSS, a negatively charged polyelectrolyte, was utilized as a stabiliser for magnetite nanoparticles, and PAH, a positively charged polyelectrolyte, was used
[...] Read more.
In this work, new magnetic-plasmonic nanocomposites have been developed through the use of two complementary polyelectrolytes–polystyrene sulfonate (PSS) and poly(allylamine hydrochloride) (PAH). PSS, a negatively charged polyelectrolyte, was utilized as a stabiliser for magnetite nanoparticles, and PAH, a positively charged polyelectrolyte, was used to stabilize gold nanoparticles. The combination of these two entities resulted in a magnetic-plasmonic nanocomposite that is highly reproducible and scalable. This approach was found to work for a variety of PSS concentrations. The produced magnetic-plasmonic nanomaterials have been characterized by vibrational sample magnetometry (VSM), transmission electron microscopy (TEM) and UV-Vis spectroscopy. These nanocomposite materials have the potential to be used in a variety of biological applications including bioseparation and biosensing.
Full article
►▼
Show Figures
Open AccessArticle
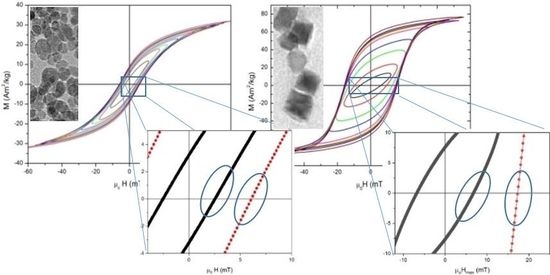
High Frequency Hysteresis Losses on γ-Fe2O3 and Fe3O4: Susceptibility as a Magnetic Stamp for Chain Formation
by
Irene Morales, Rocio Costo, Nicolas Mille, Gustavo B. Da Silva, Julian Carrey, Antonio Hernando and Patricia De la Presa
Cited by 60 | Viewed by 7754
Abstract
In order to understand the properties involved in the heating performance of magnetic nanoparticles during hyperthermia treatments, a systematic study of different γ-Fe
2O
3 and Fe
3O
4 nanoparticles has been done. High-frequency hysteresis loops at 50 kHz carried out
[...] Read more.
In order to understand the properties involved in the heating performance of magnetic nanoparticles during hyperthermia treatments, a systematic study of different γ-Fe
2O
3 and Fe
3O
4 nanoparticles has been done. High-frequency hysteresis loops at 50 kHz carried out on particles with sizes ranging from 6 to 350 nm show susceptibility χ increases from 9 to 40 for large particles and it is almost field independent for the smaller ones. This suggests that the applied field induces chain ordering in large particles but not in the smaller ones due to the competition between thermal and dipolar energy. The specific absorption rate (SAR) calculated from hysteresis losses at 60 mT and 50 kHz ranges from 30 to 360 W/g
Fe, depending on particle size, and the highest values correspond to particles ordered in chains. This enhanced heating efficiency is not a consequence of the intrinsic properties like saturation magnetization or anisotropy field but to the spatial arrangement of the particles.
Full article
►▼
Show Figures
Open AccessArticle
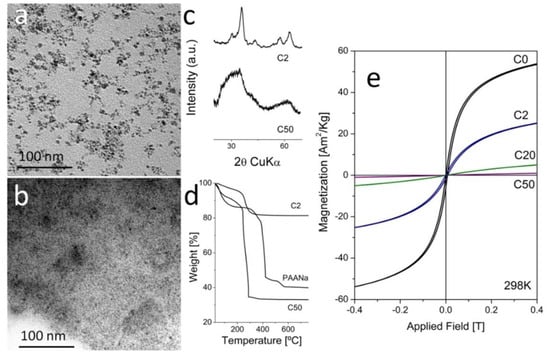
Effect of the Sodium Polyacrylate on the Magnetite Nanoparticles Produced by Green Chemistry Routes: Applicability in Forward Osmosis
by
Juan Zufía-Rivas, Puerto Morales and Sabino Veintemillas-Verdaguer
Cited by 17 | Viewed by 5693
Abstract
Aqueous dispersions of magnetic nanocomposites have been proposed as draw electrolytes in forward osmosis. One possible approach for the production of nanocomposites based on magnetite nanoparticles and sodium polyacrylate is the synthesis of the magnetic iron oxide by coprecipitation or oxidative precipitation in
[...] Read more.
Aqueous dispersions of magnetic nanocomposites have been proposed as draw electrolytes in forward osmosis. One possible approach for the production of nanocomposites based on magnetite nanoparticles and sodium polyacrylate is the synthesis of the magnetic iron oxide by coprecipitation or oxidative precipitation in the presence of an excess of the polymer. In this work, we explored the effect of the polymer proportion on the nanomaterials produced by these procedures. The materials obtained were compared with those obtained by the coating of magnetite nanocrystals produced beforehand with the same polymer. The samples were characterized by chemical analysis, photon correlation spectroscopy, thermogravimetry, X-ray diffraction, infrared spectroscopy, transmission electron microscopy, and magnetometry. The general trend observed is that the polymers heavily modify the texture of the magnetic material during the synthesis, with a drastic reduction of the particle size and magnetic response. The polycrystalline texture that is generated permits the incorporation of the polymer both on the external surface and in the intergranular space. The aqueous dispersions of the nanocomposites were highly stable, with a hydrodynamic size that was roughly independent of the polymer/magnetite ratio. Such dispersions show an osmotic pressure that is proportional to the concentration of the polymer. Interestingly, the proportionality constant was similar to that of the free polymer only in the case of the samples prepared by oxidative precipitation, being lower in the case of the samples prepared by coprecipitation. Finally, the possibilities of using these materials as draw electrolytes in forward osmosis will be briefly discussed.
Full article
►▼
Show Figures