Abstract
This review systematizes data on the phase composition and key properties of compounds in the W–B system, including thermodynamic stability, crystal structure, and hardness. The current understanding of the binary W–B phase diagram and the stability of individual borides is discussed, alongside the influence of defects and non-stoichiometry on their properties. The main methods for synthesizing these materials and producing coatings based on them are summarized. Potential applications of tungsten borides are highlighted, particularly for high-temperature environments, cutting tools, and protective and functional coatings. Finally, key directions for future research are outlined, focusing on the refinement of phase equilibria, the scaling of production methods, and the development of W–B-based materials with tailored performance characteristics.
1. Introduction
Tungsten borides are a class of compounds that combine high hardness and wear resistance with good corrosion resistance in aggressive environments and metallic electrical conductivity [1,2,3].
According to the available data, the French chemist Henri Moissan was among the first to synthesize refractory metal borides, including tungsten borides [4]. He is known for isolating pure fluorine, inventing the high-temperature electric arc furnace, and studying boron-based refractory materials. However, the characterization methods available at the time did not allow for a reliable determination of the phase composition and properties of the borides he obtained.
The next major wave of development in this field occurred in the 1940s and 1950s, which can be seen as a period of the systematic consolidation and expansion of knowledge on tungsten borides. During these years, a series of fundamental works [5,6,7,8,9] clarified the crystal structures, properties, and synthesis routes of tungsten borides. For example, Kiessling showed that tungsten and boron not only form diborides such as WB2, but also phases like W2B and W2B5 [5], which stimulated further research on the W–B system and its potential applications.
From the 1960s onwards, the focus gradually shifted from purely fundamental studies to more application-driven research. The attractive combination of mechanical and chemical properties drew attention from the manufacturing, microelectronics, and radiation-shielding communities [10,11,12,13,14,15,16,17,18]. At the same time, this increasing interest exposed one of the central open questions of the system: the true stoichiometry and formation conditions of the highest tungsten boride, which had long been assumed to be tungsten tetraboride, WB4 [17,18].
In the early 21st century, advances in computational methods and available computing power opened new possibilities for modeling complex materials [19,20,21,22,23]. This progress renewed interest in superhard compounds with a hardness comparable to, or even exceeding, that of diamond [24,25,26,27,28,29,30]. As a result, the synthesis and characterization of the highest tungsten boride attracted significant attention again.
One of the first hypotheses regarding the superhardness of the highest tungsten boride was proposed by Brazhkin et al. [26], who discussed materials with hardness values potentially approaching 60 GPa. In 2008, Gu et al. published a paper stating that the hardness of WB4 could reach 46 GPa, effectively classifying tungsten boride as a superhard material (with hardness above 40 GPa) [31]. Later, a number of researchers conducted their own experiments [32,33], refining the synthesis parameters and testing methods, and confirmed the superhardness of WB4.
Discussions surrounding the crystal structure of the highest tungsten boride continue to this day. One of the most recent significant breakthroughs in this area was the research by A. Kvashnin et al. [34], where the crystal lattice of compounds in the W–B system was described using computational materials modeling methods. The phase corresponding to the highest tungsten boride was identified not as WB4, but as a non-stoichiometric pentaboride, WB5−. The superhardness observed in this phase is attributed to the formation of a three-dimensional framework of covalently bonded boron atoms (B–B), reinforced by W–B bonds.
Consequently, alongside the growing interest in tungsten borides and the deepening of fundamental research, synthesis methods have also evolved. While, at the beginning of research into these compounds, the primary and nearly sole method was synthesis from pure elements—sintering W and B powders at high temperatures [5]—modern practice employs a wide variety of other techniques. These include molten salt synthesis, carbothermal and metallothermic reduction, and gas-phase synthesis [35]. Certain methods, such as gas-phase synthesis, remain understudied and hold significant promise for industrial applications.
Techniques for depositing coatings based on this system are also of great interest. Methods that have proven effective for other hard tungsten compounds, such as tungsten carbide (WC) [36], do not always yield comparable results for borides. This is largely due to the properties of gaseous boron precursors and the critical difference in atomic masses between B and W, which complicates the deposition of coatings by both PVD (Physical Vapor Deposition) and CVD (Chemical Vapor Deposition).
From an application standpoint, compounds of the W–B system are only just beginning to be adopted in various industrial sectors; today, they are best described as promising rather than widely known. Their remarkable mechanical properties attract interest for use as wear-resistant coatings [37], while their high catalytic activity in acidic and alkaline environments may be valuable for hydrogen production via electrolysis [38]. Furthermore, the ability of boron structures to moderate neutrons makes this material intriguing for radiation-shielding engineering and for use in thermonuclear reactors as coatings for plasma-facing components [39,40]. New discoveries regarding the properties and formation mechanisms of higher tungsten borides undoubtedly heighten industrial interest and open new horizons for the application of this class of materials.
Despite the availability of fundamental monographs on refractory metal borides and recent highly specialized articles, the current literature lacks comprehensive studies that bridge the classical materials science of borides with recent discoveries. Existing reviews typically focus either exclusively on the theoretical aspects of superhardness or on specific synthesis methods. The present review aims to fill this gap by offering an interdisciplinary perspective on the W–B system. Its objective is to integrate updated data on phase stability (including the current consensus on the stoichiometry of the higher tungsten boride) with a critical analysis of synthesis methods for both bulk materials and coatings. Furthermore, it seeks to systematize emerging application areas, ranging from traditional metalworking to novel developments in hydrogen energy and neutron-shielding.
2. Key Properties of W–B System Compounds
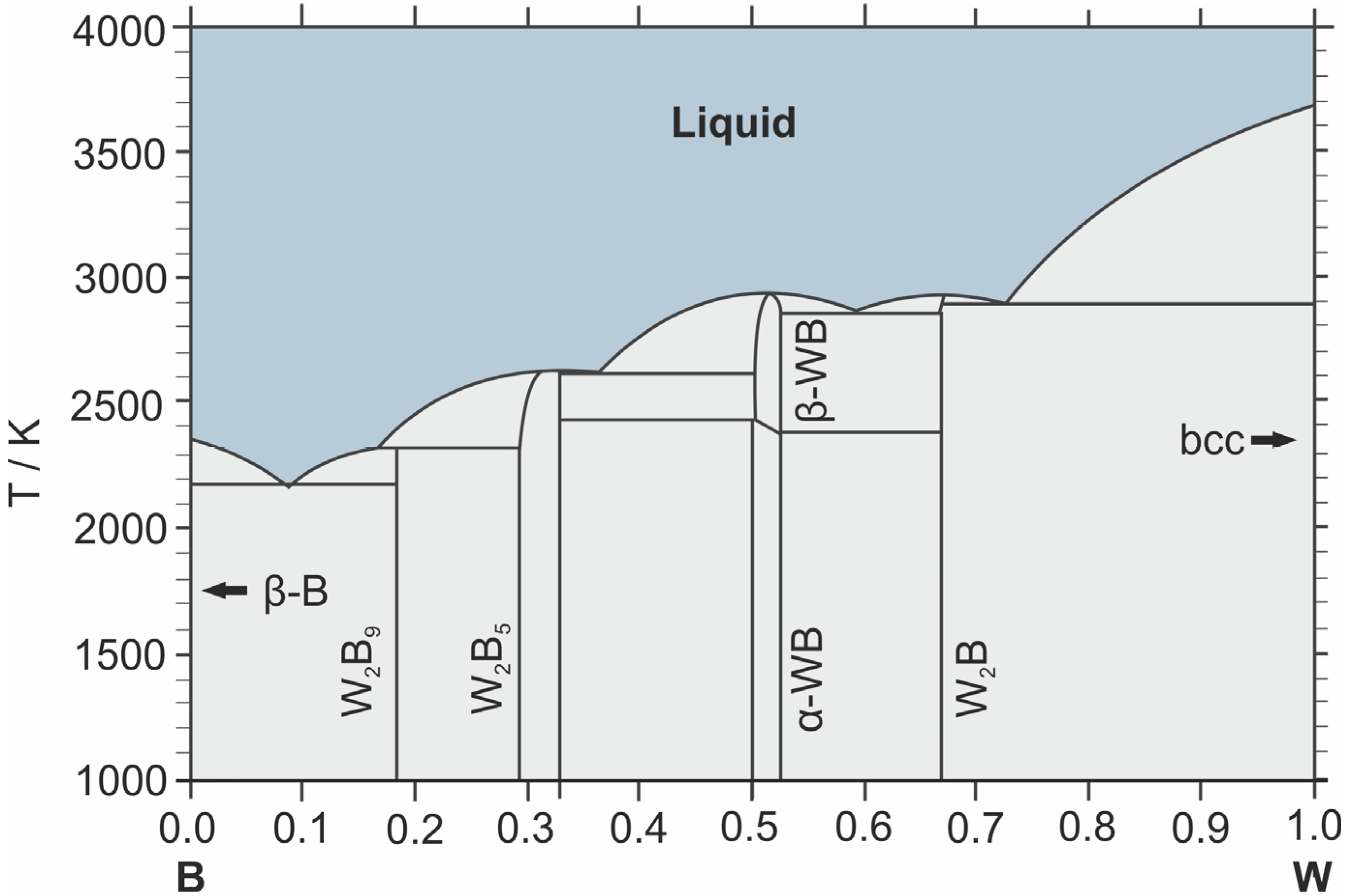
Figure 1 presents the phase diagram of the B–W system.
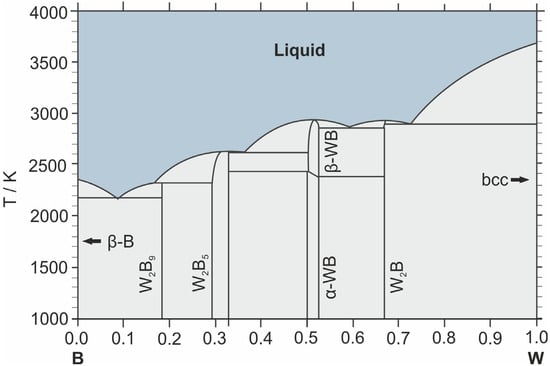
Figure 1.
Phase diagram of the W–B system [41].
This diagram was calculated using the CALPHAD method and proposed by H. Duschanek and P. Rogl [41]. It is worth noting that a comprehensive and publicly available CALPHAD thermodynamic database (e.g., in Thermo-Calc format) for the binary W–B system is still lacking. Consequently, the assessment by Duschanek and Rogl remains the most robust macroscopic thermodynamic model to date. Its distinctive feature, distinguishing it from earlier diagrams [15,42], is the inclusion of the stoichiometric W2B9 phase. In experimentally derived diagrams, this region was previously designated as WB(3+x) or WB4, which exhibited stability at low temperatures under real-world conditions but did not satisfy the computational models. By introducing the W2B9 compound into the model as a stoichiometric phase, H. Duschanek and P. Rogl achieved a close agreement between the calculated curves and experimental melting points. Nearly a quarter of a century later, new studies demonstrated that WB4 actually exists as a defect structure rather than a “pure” phase [34], confirming that the methodological approach chosen by H. Duschanek and P. Rogl to describe the W–B phase diagram was justified. This diagram has become the most widely used, entering the majority of handbooks and computational software databases as the reference standard; therefore, the subsequent description of the phase composition and properties of compounds in the W–B system will be based upon it.
The binary W–B system belongs to the class of refractory compounds and is characterized by high phase-melting temperatures and high hardness. Four key stoichiometric phases are identified within this system (Table 1): W2B, WB, W2B5, and W2B9. The properties of each phase will be discussed below, in order of increasing boron mass fraction.

Table 1.
Crystal structure and melting temperatures of various tungsten boride phases [34,41,43].
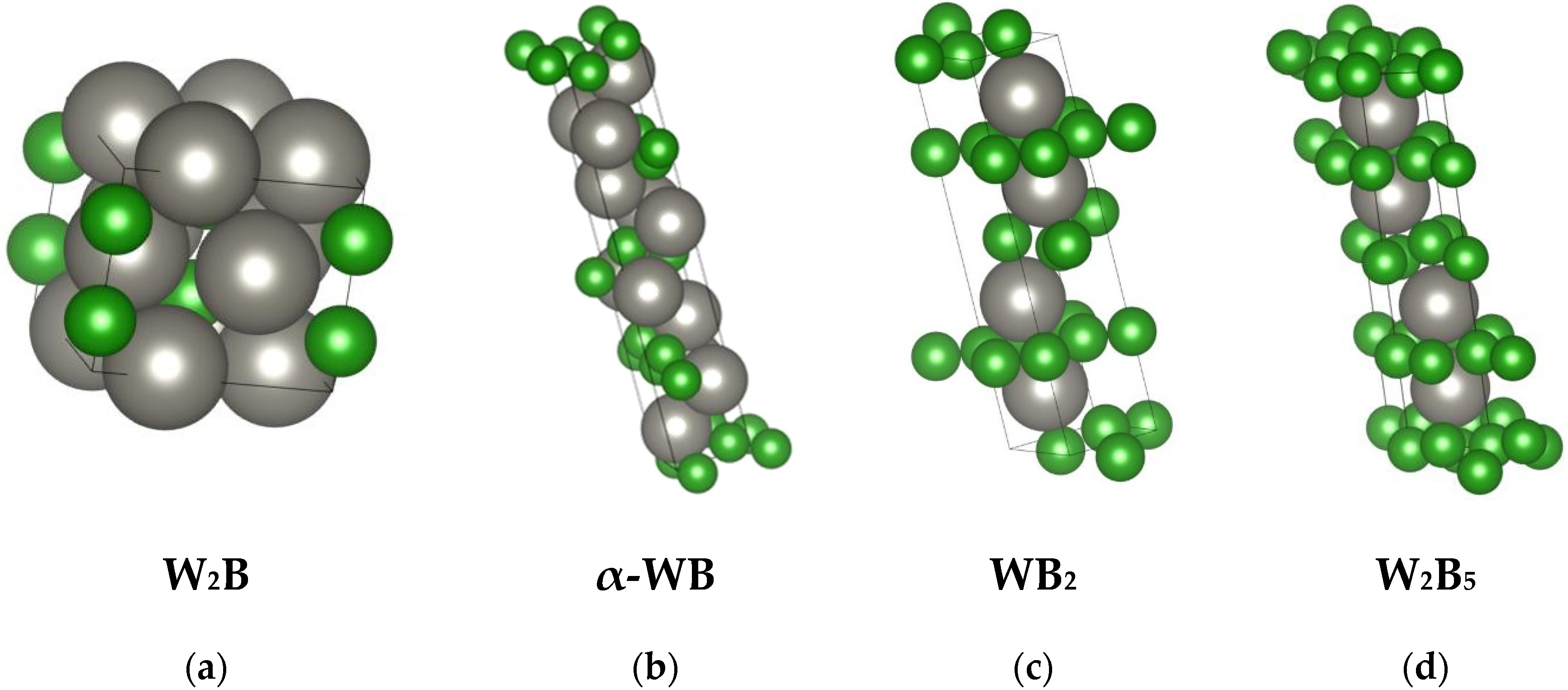
The “lowest” boride in the system is W2B, a phase that melts congruently at a temperature of 2900–2950 K. It crystallizes in the tetragonal system (Figure 2a), forming a CuAl2-type structure.
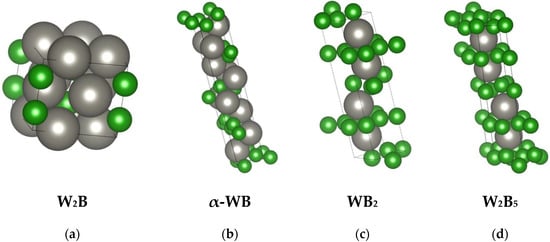
Figure 2.
Unit cells of selected tungsten borides (crystal structures [44] visualized using VESTA): W2B (a), α-WB (b), WB2 (c), and W2B5 (d). Tungsten atoms are shown in gray, and boron atoms are shown in green.
WB–tungsten monoboride is a polymorphic phase that transitions upon heating from low-temperature α-WB to high-temperature β-WB at 2380–2400 K. It forms tetragonal α-WB (Figure 2b) and orthorhombic β-WB crystals.
The W2B5 phase exhibits a wide homogeneity range (approximately 61–71 at.%) and is a congruently melting phase with a melting point of 2600 K. It forms a eutectic with β-WB at 2600 K. It crystallizes in the hexagonal system and possesses a layered structure consisting of boron atom nets (Figure 2d).
W2B9 melts via a peritectic reaction at a temperature of approximately 2310 K. At a temperature of about 2180 K and a boron content of approximately 90 at.%, the W2B9 and beta-B phases form one of the lowest-melting eutectics in the system (2100–2200 K). As mentioned earlier, in this diagram (Figure 1), the W2B9 phase is a computational assumption that provides better agreement between the experimental and model data [41]. This region of the diagram characterizes the highest tungsten boride, which, according to recent data [34], has the composition WB5−x and crystallizes in a hexagonal form with a defect lattice. Due to the sufficiently narrow temperature window and competition with the W2B5 phase, compositions from this region are the most complex and demanding to synthesize [45,46,47].
The absence of tungsten diboride (WB2) on the diagram—a characteristic phase for all transition metals and one that was previously described for tungsten borides as well [15,42]—is a consequence of the conclusions drawn by H. Duschanek and P. Rogl regarding the stability of this phase at normal pressure. They determined [41], that given the proximity of stoichiometric ratios and the similarity of the crystal lattices of the WB2 and W2B5 phases, the difference between them lies mostly in the presence of stacking faults. They concluded that W2B5 is the equilibrium phase, while what researchers had previously described as the WB2 phase was either the lower limit of the W2B5 homogeneity range or a mixture of WB/W2B5 phases. Modern DFT studies indicate that at atmospheric pressure, the formation energy of W2B5 is significantly higher than that of WB2, which theoretically makes this phase less stable [34]. Nevertheless, experimentally, it is the W2B5 phase that is frequently obtained, which is associated with the difficulty of achieving exact WB2 stoichiometry [37,48].
In the view of the present authors, the current body of experimental and computational data convincingly demonstrates that the long-standing debate surrounding the stoichiometry of the higher tungsten boride is primarily associated with the narrow homogeneity range and the highly defective nature of the most boron-rich phases, rather than the existence of several fundamentally distinct stoichiometric compounds. Describing the higher boride as a defective pentaboride, WB(5−x), currently appears to be the least contradictory approach, as it reconciles the classical CALPHAD calculations [41] of the phase diagram with the results of ab initio modeling. At the same time, ongoing reports of nominal WB4-type phases obtained under various synthesis conditions indicate that local non-equilibrium, stacking faults, and compositional gradients continue to play a critical role in evaluating experimental outcomes. Consequently, these factors must be carefully considered in future thermodynamic assessments and studies investigating structure–property relationships.
Over the last 20 years, significant progress has been made in understanding the relationship between the crystal structure, chemical composition, and macroscopic properties of tungsten borides [49,50,51,52,53]. The physico-mechanical properties of tungsten borides represent one of the key factors determining their industrial applicability.
The main difficulties in describing the physico-mechanical properties of tungsten borides are associated with the tendency of compounds in this system to form metastable phases and mixtures. Given the structural similarity of individual phases, this can complicate their identification and, consequently, the correct description of the properties of specific phases. Another important condition is the size effect, as the properties of thin films (1–20 nm) can differ significantly from those of bulk materials. Investigations of thin films deposited via physical vapor deposition (PVD), utilizing XRD, SEM, and TEM, clearly demonstrate the relationship between microstructure and properties [37,54]. For example, the formation of a nanocrystalline structure directly correlates with anomalously high hardness values [37,48,54,55]. The transition to a nanocrystalline state increases grain boundary density, which impedes dislocation motion and leads to macroscopic hardening. Furthermore, microstructural characterization data reveal that the formation of mixed nanophases (e.g., WB/WB2 composites) within a single film correlates with enhanced hardness [37,55].
Table 2 presents the hardness (HV) of the principal tungsten boride phases.

Table 2.
Hardness of the tungsten boride phases [34,56,57,58,59].
The maximum hardness (45 GPa) is exhibited by the highest tungsten boride, WB5−x, which was identified as WB4 in earlier studies. Its hardness surpasses that of the “highest” tungsten carbide, WC (22–30 GPa) [60,61], and is comparable to the hardness of cubic boron nitride, c-BN [62,63]. According to modern calculations [34], the strengthening mechanism of tungsten borides is based on three key features of compounds in this system: the high packing density of W and B atoms; the presence of various bond types (in addition to the covalent W–B bond, B–B type bonds are also present); and, in the case of the highest tungsten boride, the presence of boron framework networks in the structure.
A recent study [37] described the hardness and wear resistance of thin tungsten boride films deposited via the PVD method. The authors report that the hardness of individual layers, measured by nanoindentation, reached 50–56 GPa. The phase composition of these layers was identified as a mixture of WB and WB2 (46% WB—54% WB2 for 50–53 GPa and 70% WB—30% WB2 for 56 GPa). The reported hardness values of 50–56 GPa significantly exceed the range characteristic of the bulk phases listed in Table 2. This discrepancy is attributed to a combination of microstructural and technological factors. Firstly, PVD-deposited WB/WB2 films possess a nanocrystalline structure with a characteristic grain size on the order of tens of nanometers, sometimes existing as a mixture of nanophases. This structure enhances resistance to dislocation motion, effectively realizing the Hall–Petch effect [37,64,65]. Secondly, the presence of residual compressive stresses generated during deposition, along with the oriented texture of individual grains, further increases the local hardness measured via the nanoindentation technique employed by the authors. Finally, given the small film thickness, the influence of the substrate and the film/substrate interface can also affect the results, particularly under high indenter loads. Collectively, these factors cause the measured hardness of the thin-film system to be higher than that of the corresponding bulk phases in equilibrium. Similar studies on thin tungsten boride films conducted at various times confirm the dependence of hardness on grain size for compounds of the B–W system [54,66].
From a practical standpoint, controlling the grain size in tungsten boride films deposited via magnetron sputtering is achieved by adjusting process parameters: working gas pressure, the flow rate of B-containing species, magnetron power, substrate temperature, and deposition rate [37,55,67]. Lowering the substrate temperature and increasing the working gas pressure typically promote the formation of finer grains, whereas elevating the temperature and reducing the pressure lead to structural coarsening. An additional tool for microstructural control is alloying (e.g., with Zr or Ta) [68,69,70], which allows for the simultaneous regulation of both grain size and residual stresses.
Overall, the available data indicate that the high hardness of tungsten borides is driven by a combination of high atomic packing density, mixed covalent–metallic bonding, and the formation of extended B–B frameworks, which is particularly relevant for the most boron-rich phases. However, the hardness values of thin films and bulk particles often differ significantly. This discrepancy is attributed to grain size effects, residual stresses, and the presence of metastable or mixed phases in nanostructured coatings. From an applied perspective, this implies that the exceptionally high hardness values achieved in nanostructured coatings (50–56 GPa) cannot be directly extrapolated to bulk materials. Future research must clearly differentiate between the contribution of intrinsic lattice strengthening within the tungsten boride and extrinsic mechanisms, such as the Hall–Petch effect and multilayer coating architectures.
Combined with high hardness, tungsten borides also possess decent fracture toughness, which is crucial for materials subjected to impact loading. Most high-hardness ceramics, by contrast, are not characterized by high fracture toughness (Table 3), which creates limitations for their practical application.

Table 3.
Fracture toughness coefficients of selected hard ceramics [34,62,71,72,73].
It was shown in [66] that reducing grain size can improve fracture toughness by increasing the density of grain boundaries, which impede crack propagation. In contrast to metallic alloys, where grain refinement often improves toughness, the application of the Hall–Petch relationship to hard ceramics (including tungsten borides and carbides) has severe limitations. While reducing grain size significantly increases hardness, excessive grain refinement typically reduces fracture toughness due to the restriction of plastic strain relaxation mechanisms. A suitable solution for enhancing the fracture toughness of tungsten borides is doping with metals (Ta, Zr), the introduction of which can relieve stresses and increase fracture toughness [74,75]. Another common approach involves creating multilayer coatings consisting of hard borides and layers of other materials, which can also be applied to tungsten borides. For instance, a number of studies [76,77] have demonstrated that alternating layers of borides or nitrides with more ductile materials (e.g., Ti) allows for a significant increase in fracture toughness by restricting crack propagation at phase interfaces. For tungsten borides, this assertion requires experimental verification.
Another important property for the practical application of tungsten borides is the coefficient of thermal expansion (CTE). Compounds of the W–B system possess a relatively low CTE (Table 4). This imposes certain limitations on the selection of substrate materials for coating production. For example, during deposition onto steel, which has a higher CTE, compressive stresses arise in the W–B layers, leading to cracking and coating delamination [78]. The influence of this factor can be mitigated by applying an intermediate interlayer [79].

Table 4.
Coefficients of thermal expansion of selected tungsten borides and other materials [35,61,79,80,81,82,83,84].
Summarizing the methods for mitigating thermomechanical stresses in tungsten boride-based coatings, several primary approaches can be identified. Firstly, graded interlayers are widely employed, where the coefficient of thermal expansion (CTE) gradually transitions from that of the substrate to that of the functional layer. Such coatings facilitate stress redistribution and significantly reduce the risk of cracking and delamination [79,85,86]. Additionally, multilayered systems allow for the simultaneous regulation of CTE and crack propagation resistance by deflecting or arresting the crack growth vector at the layer interfaces [76,77]. Finally, a promising direction involves selecting substrates and structural materials with CTEs matching those of tungsten borides (e.g., W- or Ti-based substrates), as well as developing coatings with a controlled level of residual compressive stresses. Future research in this area should encompass thermocyclic stability testing of multilayered and graded W–B coatings, along with a quantitative assessment of permissible stress levels under realistic operating conditions.
The limits of thermal stability for tungsten borides under atmospheric conditions can be described by three temperature intervals. Up to 400 °C, the main mechanical properties remain practically unchanged [37]. In the 400–700 °C range, a decrease in hardness within 10–15% is observed [37]. At these temperatures, oxidation is negligible, and the phase composition remains stable. A sharp change in properties is noted above 700 °C [78], which is apparently caused by the onset of oxidative processes, the formation of WO3, and subsequent material degradation. Several studies have shown that the oxidation resistance of tungsten borides at high temperatures can be improved by additions of silicon (Si) [87] and tantalum (Ta) [78], which form a protective layer of their own oxides.
Tungsten borides exhibit good corrosion resistance in acidic environments. They show good stability in aqueous solutions of sulfuric (H2SO4) and hydrochloric (HCl) acids [88]. The exception is nitric acid (HNO3), which causes dissolution with the formation of tungstic acid (H2WO4) and the oxidation of boron to boric acid (H3BO3) [35]. In alkaline environments, borides, like tungsten carbides, demonstrate reduced corrosion resistance, mirroring the corrosion behavior of tungsten, which dissolves to form tungstate ions [89,90,91].
Unlike classical ceramics, which generally possess poor electrical conductivity and are used as insulators, tungsten borides demonstrate the properties of a metallic conductor (Table 5). The fundamental difference between tungsten borides and traditional superhard ceramics, such as Al2O3 or c-BN, lies in their electronic structure and chemical bonding. Classical hard ceramics rely almost exclusively on strong, highly directional covalent or ionic bonds and possess a wide bandgap, inevitably making them electrical insulators. In contrast, tungsten borides are characterized by a complex, mixed bonding nature. Strong covalent W–B and B–B bonds (including extended boron frameworks in boron-rich phases) impart hardness and lattice rigidity. Simultaneously, the overlap of tungsten d-orbitals with boron p-orbitals leads to the formation of a continuous electronic spectrum where the Fermi level crosses the valence bands [92], providing a high density of delocalized electrons. It is this synergy between covalent networks, which govern mechanical strength, and metallic bonds, which facilitate electron transport, that endows tungsten borides with a rare combination of superhardness and electrical conductivity.

Table 5.
Comparative electrical conductivity and resistivity of selected tungsten borides versus other substances and compounds at room temperature [35,54,61,93,94].
The electrical resistivity of tungsten borides increases with rising temperature, which is typical for metallic conductors. For polycrystalline WB, the resistivity at room temperature is 20–30 μΩ∙cm [95], which is comparable to that of titanium and steel. Wang et al. noted that the resistivity of tungsten borides decreases with increasing grain size [54]. This is explained by the reduction in the density of grain boundaries, where charge carrier scattering occurs. At atmospheric pressure, some tungsten borides (WB, W2B5) exhibit superconductivity at temperatures of 4.3–5.4 K [96]. The combination of electrical conductivity and high hardness allows tungsten borides to be used as contacts designed to operate under extreme conditions or coatings on electrodes for electrical discharge machining (EDM). Unlike common hard ceramics (such as Al2O3 or c-BN), which are electrical insulators, the high electrical conductivity of tungsten borides makes them a promising material for electrical discharge machining (EDM). Compared to classical refractory electrodes made of pure tungsten or Cu–W pseudo-alloy, refractory metal borides—due to their superhardness and robust covalent B–B framework—exhibit significantly higher resistance to mechanical erosion and micro-chipping caused by impacts within the discharge zone, potentially increasing the lifespan of the electrode [97,98].
The radiation resistance of tungsten borides deserves special mention. Tungsten has been considered the primary candidate material for the first wall of thermonuclear reactors due to its refractory nature and its ability to effectively attenuate gamma rays and neutrons [99,100]. The introduction of boron alters the material’s interaction profile with radiation, as boron, and specifically its isotope B10, is capable of effectively capturing and absorbing neutrons [101]. The combination of these properties in tungsten borides makes them highly promising as coatings for tokamak plasma-facing components [102,103]. Heavy W nuclei effectively slow down fast neutrons to intermediate energies, after which they are thermalized (reach thermal equilibrium with the medium) and absorbed by boron nuclei. This combination of properties surpasses that of metallic tungsten [103] and may also allow for the elimination of multilayer lead-based structures [104,105] in favor of thinner, monolithic tungsten boride shields.
A study by Erdogan et al. indicates that W2B5 possesses a high atomic density, which ensures a maximum macroscopic capture cross-section [105]. A promising direction for research in this area is the verification of the radiological resistance of the highest tungsten boride, WB5−x, which possesses the highest boron-to-tungsten ratio.
The primary challenge for borides is the transmutation of B10 resulting in the formation of helium, which occurs upon neutron capture [106]. The resulting helium exhibits low solubility within the tungsten and boride lattices. Helium atoms migrate to grain boundaries and accumulate within them, exacerbating crystal lattice defects and inducing the embrittlement of the W–B layers. Studies indicate that in borides, helium bubbles tend to accumulate along grain boundaries [103,107].
To summarize the key properties of tungsten borides, it is worth highlighting their high hardness and fracture toughness, good electrical conductivity, low coefficient of thermal expansion, oxidation resistance at temperatures up to 700 °C, and corrosion resistance in acidic environments, as well as their radiation resistance. This combination of properties distinguishes tungsten borides from other high-temperature ceramics. Comparative characteristics of certain compounds of the W–B system versus selected materials are presented in Table 6.

Table 6.
Comparison of tungsten borides with other hard ceramics [32,34,35,61,62,63,94,108,109].
The combination of high hardness and fracture toughness makes tungsten borides attractive for use in the production of cutting tools and highly abrasive compounds. Their electrical conductivity and high corrosion resistance in aqueous solutions open up possibilities for their electrocatalytic applications, while their radiation resistance allows this material to be considered promising for neutron-shielding.
The primary challenges and topics for future research include improving the oxidation resistance of tungsten borides at temperatures exceeding 700 °C, enhancing the fracture toughness of coatings based on them, and conducting further studies aimed at developing applications for W–B system phases as neutron-shielding materials.
A cornerstone issue remains the synthesis of single-phase compositions and the control of stoichiometry during the production of tungsten borides. Furthermore, a critical factor is the high production cost, which currently limits their mass adoption in industrial sectors.
3. Methods for Synthesis of Tungsten Borides
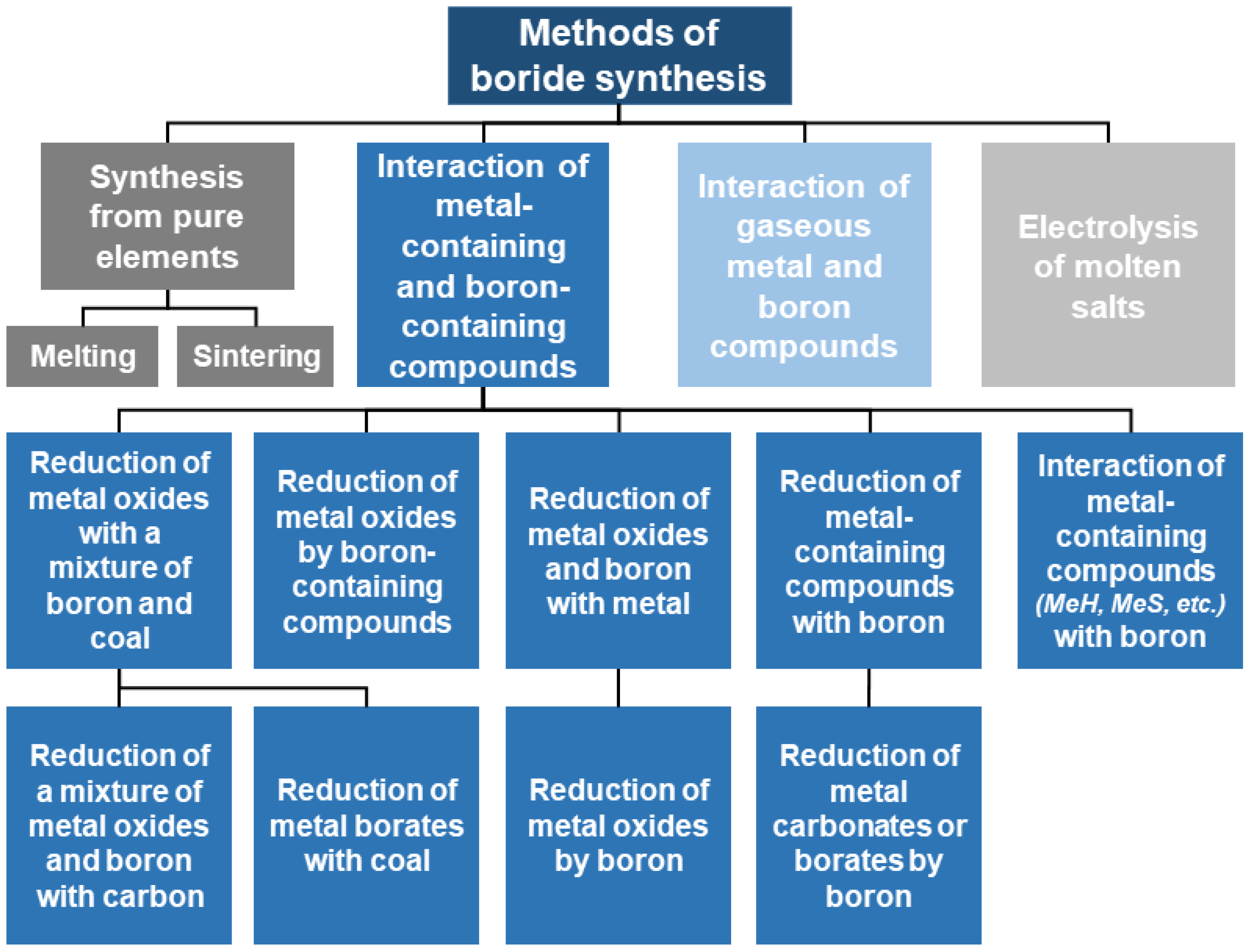
The fundamental concepts for the synthesis of refractory metal borides were outlined in the classic works of G.V. Samsonov et al. (Figure 3) [35].
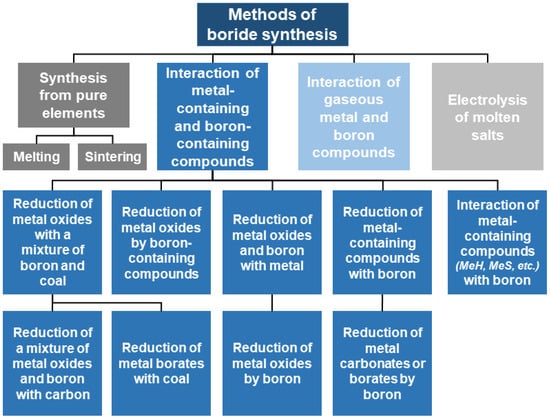
Figure 3.
Fundamental concepts for the synthesis of refractory metal borides [35].
It should be noted that not all methods presented in this classification are suitable for the synthesis of tungsten boride compounds. The following discussion will focus on those methods that are applicable for obtaining W–B system compositions and have demonstrated their effectiveness in practice.
Solid-state synthesis from powders was one of the first methods applied for the synthesis of W–B system compounds [5]. Technologically, it is quite simple: it involves holding boron and tungsten powders at high temperatures. This method allows for the production of pure tungsten borides (provided the reagents are of appropriate purity) but requires high process temperatures in the range of 1000–1600 °C [5]. Direct synthesis from pure substances is used to create materials for applications where phase composition and the absence of impurities are of critical importance.
Due to the availability of tungsten and boron oxides, synthesis methods based on reduction from oxides, as well as from carbides and other compounds containing boron and tungsten, have gained widespread use.
The reduction in tungsten oxides in the presence of pure boron or its carbide (B4C) and carbon allows for the synthesis of tungsten boride phases at atmospheric pressure and temperatures of 1000–1200 °C via Reactions (1) and (2):
4 WO3 + B4C +11 C → 4 WB + 12 CO,
WO3 + B + 3C → WB + 3 CO
However, obtaining pure material using this method is problematic, as carbon has a relatively high affinity for tungsten. Consequently, the growth of boride phases will be suppressed by the preferential formation of carbides (W2C, WC) and phases of the ternary W–C–B system [35].
The reduction in a mixture of boron oxide (B2O3) and tungsten oxide (WO3) in the presence of aluminum or magnesium is known as the aluminothermic and magnesiothermic method or self-propagating high-temperature synthesis (SHS), which utilizes the high exothermicity of Reactions (3) and (4) as the primary heat source:
2 WO3 + B2O3 + 9 Mg → 2 WB + 9 MgO
2 WO3 + 6 B2O3 + 2 Al → 2 WB + 3 Al2O3
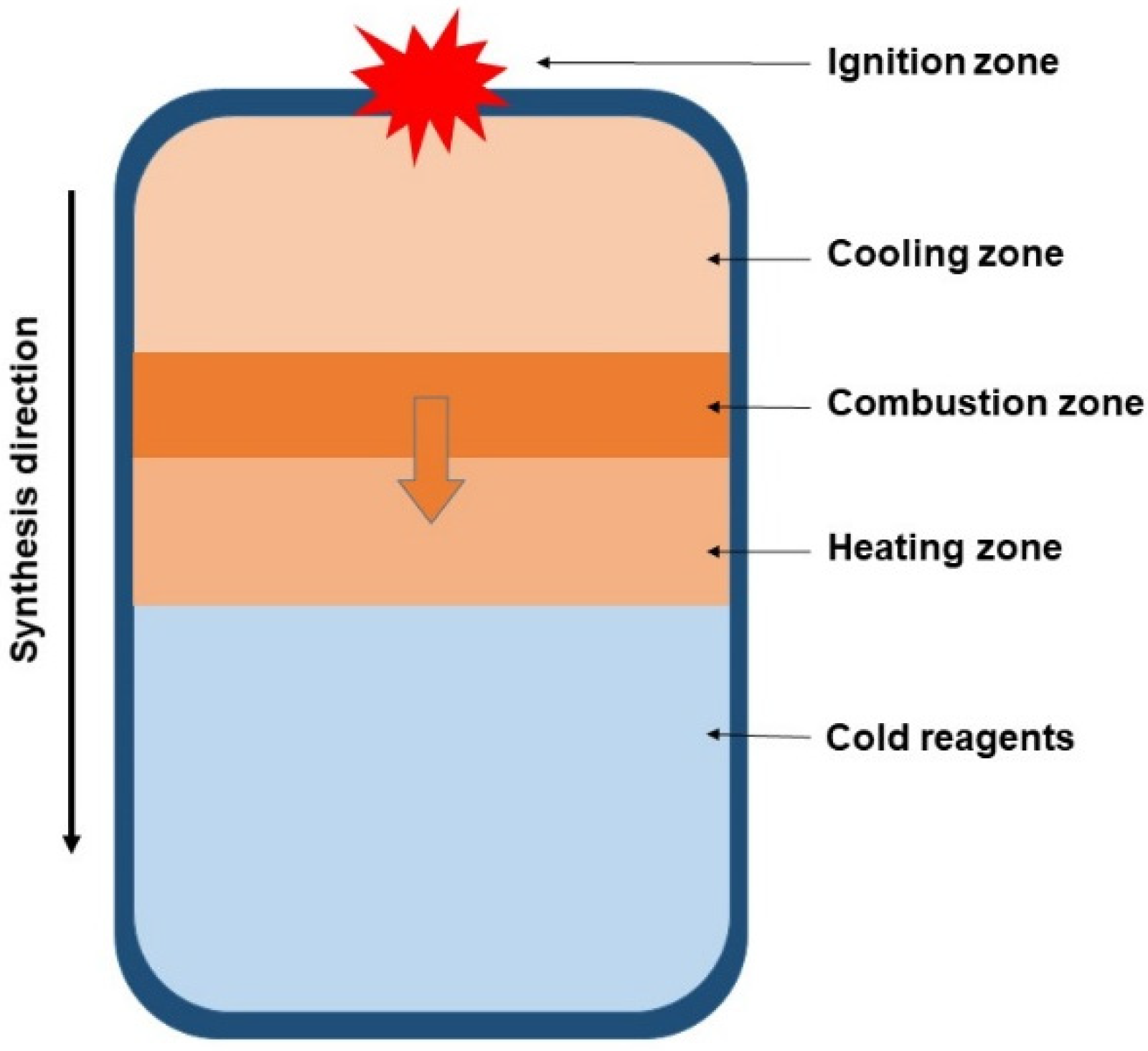
Like direct synthesis, this method is technologically quite simple (Figure 4). However, the synthesis product will inevitably be contaminated with oxides of the reducing metal, requiring additional purification steps [110,111]. In the case of magnesiothermic reduction, the tungsten borides are encapsulated within an MgO matrix, and acceptable purity can be achieved by leaching the MgO with hydrochloric acid [111]. Aluminothermic reduction has not found widespread application because removing Al2O3 impurities is difficult due to its resistance to most strong acids and alkalis. Dissolving Al2O3 requires exposure to heated hydrofluoric acid (HF) under elevated pressure [112,113], which is far more complex than the simple leaching of MgO with hydrochloric acid. Furthermore, aluminum is capable of incorporating into the tungsten boride lattice and forming ternary W–Al–B compounds [114]. Additionally, a significant drawback of the SHS method is the poor predictability of the phase composition of the obtained borides.
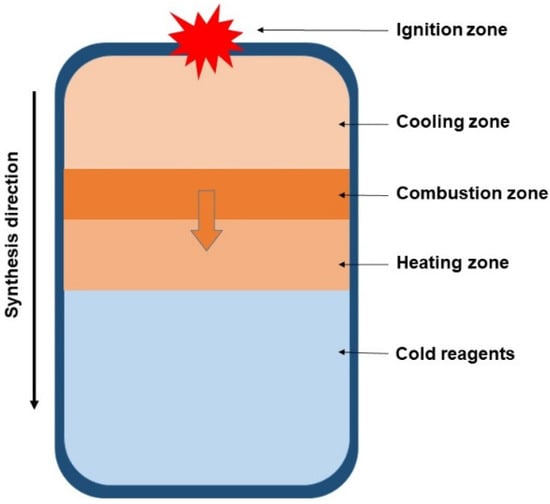
Figure 4.
Principal scheme of SHS process [35]. Arrows indicate the direction of the combustion wave propagation.
A comparative analysis of synthesis methods for bulk tungsten boride particles reveals that the choice of a specific technique is primarily dictated by the final application of the material, as each method entails a compromise among phase purity, process temperature, and scalability. Direct solid-state synthesis from pure powders remains the preferred route for obtaining specific tungsten boride phases; however, the required high temperatures and prolonged holding times limit its scalability and industrial appeal. In contrast, carbothermic reduction and self-propagating high-temperature synthesis (SHS) require a lower overall energy input and offer better scalability, but they inevitably lead to the formation of secondary carbide or oxide phases, complicating precise stoichiometric control. It appears that a promising direction for the synthesis of bulk materials lies in hybrid approaches and stepwise reduction schemes, which provide additional capabilities for controlling the phase composition and purifying the final product [111,115,116]
A distinct challenge lies not only in the synthesis but also in the production of coatings based on tungsten borides. Given the prospects for practical application, the chosen coating method must ensure good adhesion to the substrate, the required layer thickness, and cross-sectional uniformity. For certain applications, porosity (low or high) may also be important, as well as the tendency of deposited layers to crack in both cold and hot states.
The molten salt electrolysis method involves the electrochemical reduction from melts containing boron and tungsten ions (e.g., LiCl–KCl–Na2WO4–Na2B4O7), which are deposited onto the cathode under the influence of an electric current. The typical temperature range for the process is 700–1000 °C [38]. This method allows for the growth of large tungsten boride single crystals or their deposition onto conductive substrates. From the perspective of depositing W–B layers directly onto a substrate, the main limitations of this method are the difficulty in controlling the phase composition and the relatively low deposition rates [117,118].
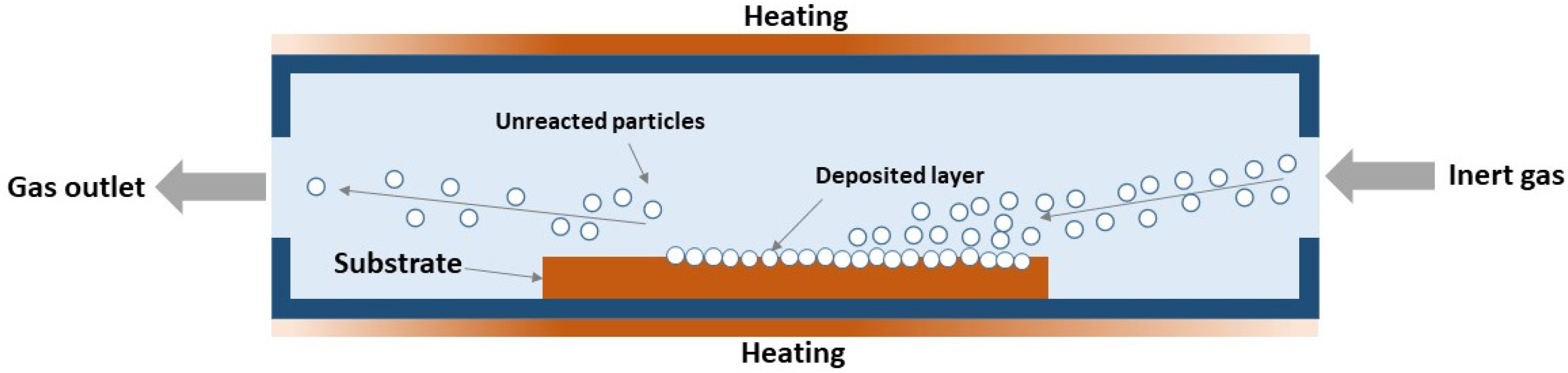
The interaction of gas-phase metal and boron compounds, or chemical vapor deposition (CVD), involves the simultaneous co-deposition of gaseous precursors, typically in the presence of a reducing gas. This method allows for a controlled phase composition due to the possibility of finer control over the synthesis process, where an excess or shortage of any precursor can be regulated by controlling its feed volume into the reaction zone. To date, this method, as applied to the W–B system, remains relatively poorly studied. There are studies in which tungsten borides were obtained by depositing gaseous precursors (typically tungsten and boron chlorides, WCl6 or BCl3) onto a boron or tungsten filament heated to 1500–2000 °C [35,119], but such a process has rather low practical value due to the low yields. The key advantage of the CVD method is specifically the ability to obtain not just a compound, but a coating deposited directly onto a part or specimen acting as a substrate. Generally, CVD coatings exhibit low porosity and good adhesion to the substrate, and can be synthesized at temperatures reaching 20–30% of the melting points of the source materials [120,121,122], which is particularly valuable for coatings based on refractory compounds, such as those of the W–B system. A schematic of CVD synthesis in a hot-wall reactor is presented in Figure 5.
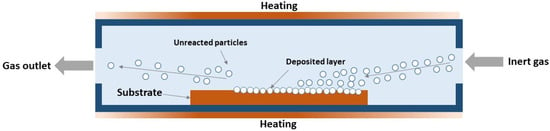
Figure 5.
Principal scheme of CVD synthesis in hot-wall reactor [35].
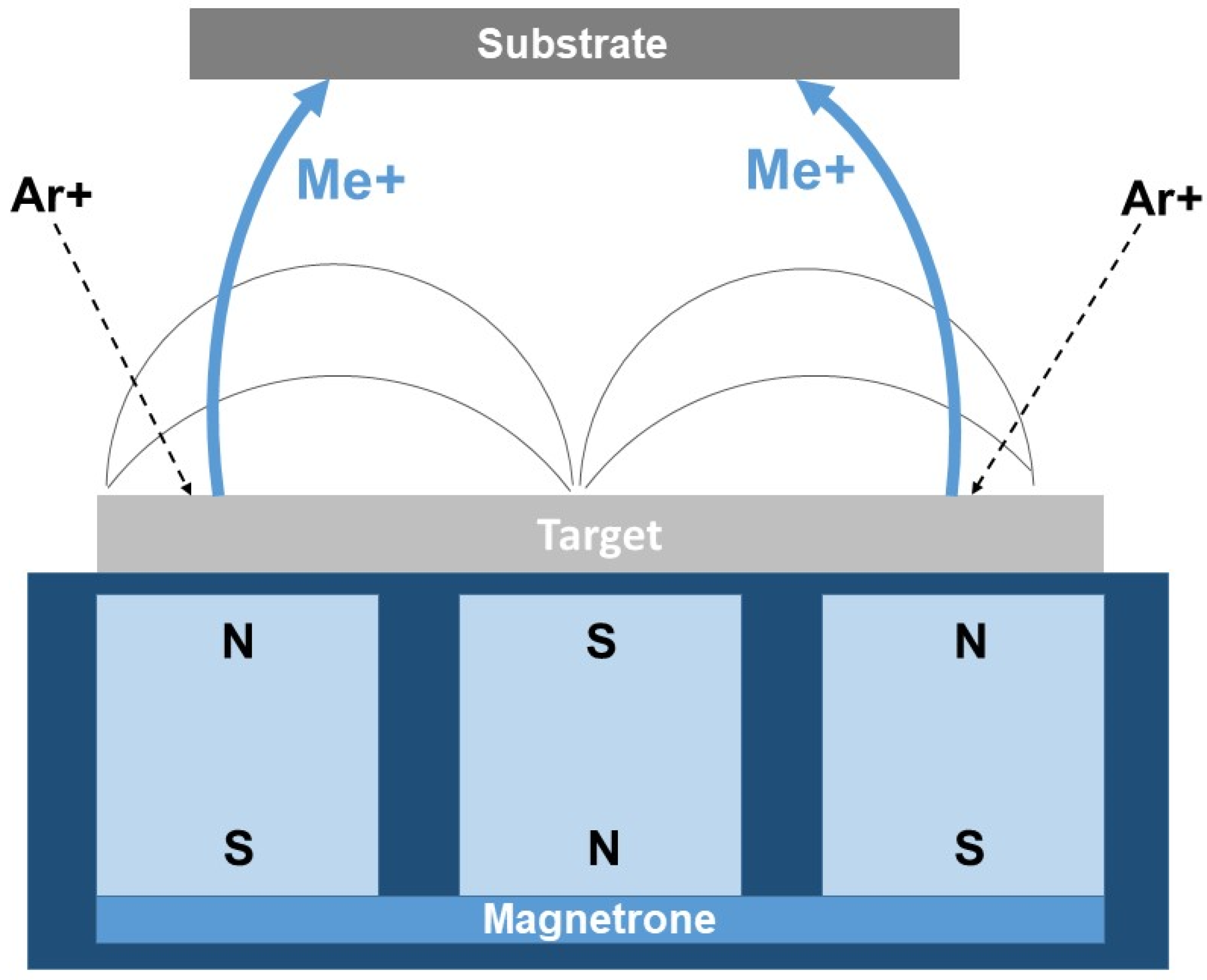
In the practice of producing tungsten boride-based coatings, the magnetron sputtering (PVD) method has proven highly effective. This process is based on bombarding targets consisting of the compound to be deposited with inert gas ions (most commonly argon), which are accelerated by an electric field generated by a magnetron [122]. The accelerated particles from the W–B target are deposited onto a heated substrate, crystallize, and form a coating (Figure 6).
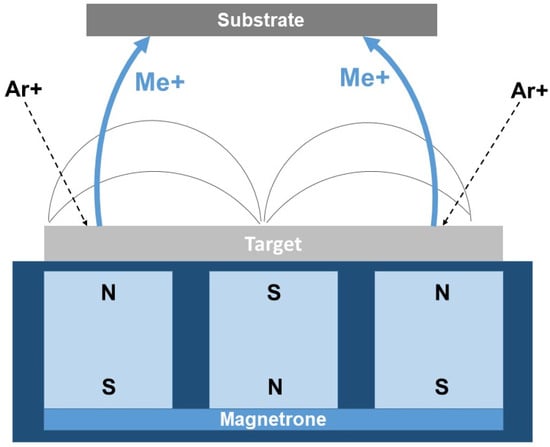
Figure 6.
Principal scheme of magnetron sputtering (PVD) [109].
There are various types of magnetrons that are suitable for working with specific targets. Direct current (DC) magnetrons are suitable only for conductive targets [123], including tungsten borides. They are characterized by a simple electrical circuit and relatively high deposition rates. If working with non-conductive materials, radio frequency (RF) magnetrons exist, which rely on the application of alternating fields [124]. Recently, pulsed magnetrons, which operate by delivering short, high-power pulses (up to 10–12 kW) [125], have also gained popularity. The HiPIMS (High-Power Impulse Magnetron Sputtering) process has lower deposition rates than RF sputtering but allows for the production of denser and more predictable (in terms of phase composition) coatings at lower substrate temperatures due to the higher ionization of sputtered atoms [126,127,128].
Substrate temperature is one of the critically important parameters determining the phase composition and structure of deposited layers in the W–B system. J. Chrzanowska-Giżyńska et al. [37] investigated the deposition process of boride coatings using RF sputtering; an analysis of the results showed that at 300 °C, amorphous structures with low hardness (15–25 GPa) are formed, whereas at 300–400 °C, the crystalline WB2 phase was obtained, increasing hardness to 35–40 GPa. At 500–600 °C and an argon pressure of 0.8 Pa, maximum crystallization of the W2B phase and traces of W2B5 were achieved, with the hardness of the deposited layers reaching 45–50 GPa. One of the primary challenges when depositing tungsten borides onto steel or titanium substrates is ensuring good adhesion. To improve the bonding of the coating to the substrate, preliminary surface bombardment with inert gas ions is performed, as well as the application of an underlayer [129,130].
Another well-known method for producing W–B system coatings is the diffusion boriding of tungsten (powder-pack process) at 900–1300 °C [35,131]. This method involves heating the workpieces to be coated in a boriding agent medium; the agent diffuses into the tungsten crystal lattice and forms boride compounds. Typically, boron carbide or amorphous boron powders serve as the boriding agent. Since boron diffusion rates into tungsten are extremely low at temperatures below 1800 °C [47,131], an activator (e.g., KBF4) is added to the mixture. This promotes diffusion by facilitating a chemical transport mechanism of boron particles to the interface between the solid tungsten phase and the boron powder.
Materials obtained through SHS or other processes producing bulk particles can be applied to substrates using High-Velocity Oxy-Fuel (HVOF) spraying. HVOF relies on accelerating (approx. 600 m/s), heating, and depositing powder particles within a hot gas stream. Using this method, WC-WB-Co composite coatings can be produced [132,133,134]. The primary limitation for producing pure tungsten boride coatings is the onset of active oxidation of tungsten borides at temperatures exceeding 700 °C [78]. The resulting layers exhibit good substrate adhesion, high density, and wear resistance. Structural analysis of the deposited layers revealed that the hardness advantage over WC-Co HVOF coatings is achieved through the formation of nanostructured compounds within the ternary W-B-Co system [134].
Summary data describing methods for producing coatings based on W–B system compounds are presented in Table 7.

Table 7.
Methods of obtaining tungsten boride coatings, comparative characteristics.
4. Practical Applications of Tungsten Borides and Promising Research Directions
To date, one of the most developed areas of application for tungsten borides is protective coatings on cutting tools. The combination of high hardness and moderate toughness ensures resistance to abrasive wear and minimizes the risk of brittle fracture of the tool’s cutting edge. Tungsten boride coatings are capable of significantly extending the service life of tools operating at high cutting speeds, which is particularly important for industries requiring maximum continuity of the production process due to the high cost of maintenance downtime. Experimental data show a significant increase in tool service life compared to unstrengthened variants. Specifically, quantitative wear resistance tests indicate that the application of high-hardness boron-containing coatings can increase tool life by a factor of 2 to 3 compared to uncoated tools when turning heavy tungsten alloys at cutting speeds exceeding 50 m/min [135]. In the future, due to their hardness and thermal stability, protective coatings based on W–B system compounds may carve out a niche among classic strengthening coatings based on metal carbides and nitrides (TiN, TiC, TiCN, Al2O3, WC).
A promising direction is the application of tungsten borides as coatings in functional component pairs experiencing mutual friction under elevated temperature conditions. When heated to 500–600 °C, tungsten borides form a thin oxide film that serves as a natural dry lubricant and significantly reduces frictional wear [136]. Furthermore, under high-temperature friction conditions (at 500–600 °C), the formation of a boron oxide (B2O3) tribofilm reduces the coefficient of friction (COF) of boride coatings to values between 0.2 and 0.3 while simultaneously decreasing the wear rate by an order of magnitude compared to untextured surfaces [137]. Such a set of characteristics could be beneficial in sliding bearings for high-temperature machinery and tool guides (under dry cutting conditions), as well as in the aerospace industry, where numerous components undergo friction at elevated temperatures.
Over the last 2–3 years, there has been a significant breakthrough in the understanding of the electrocatalytic properties of tungsten borides. While researchers previously focused on molybdenum-containing materials, a series of recent studies have demonstrated that tungsten borides exhibit remarkable properties and can serve as an alternative to expensive platinum in catalysts for hydrogen production via electrolysis for hydrogen evolution reaction (HER) [38,52,115]. From the perspective of the hydrogen evolution reaction (HER) mechanism, tungsten borides represent an intriguing class of non-noble metal catalysts combining several advantageous factors. For instance, electronic structure calculations reveal that in certain WB2-type phases and higher borides (including two-dimensional WB4 lattices), flat boron layers exist with a hydrogen adsorption energy (Δ) close to zero. This makes such surfaces catalytically active, exhibiting a performance comparable to that of platinum [138,139]. In turn, the metallic conductivity of the W–B framework ensures efficient charge transfer, while its chemical stability in acidic environments enables operation without rapid electrode degradation. Additionally, nanocrystalline forms of WB2 exhibit low overpotentials, typically in the range of 120–150 mV at a current density of 10 mA/cm2, in both acidic and mildly alkaline electrolytes [38,140]. For transition metal-based boride HER catalysts (including MoB2/WB2 systems), the characteristic range for Tafel slopes is approximately 80–90 mV/dec. [139]. This brings the performance of tungsten borides closer to that of the best non-noble metal HER catalysts (such as molybdenum sulfides and hybrid Co/Ni catalysts), making these systems a potential, more cost-effective alternative to Pt-containing materials. Further progress in this area is expected through the fine-tuning of boride compositions (for instance, by developing variants with boron-rich layers), an approach that has already been demonstrated in various Mo/W borides as an effective strategy for enhancing HER activity [141,142].
Studies on the radiological properties of tungsten borides are only just gaining popularity; nevertheless, their remarkable properties could be utilized in radiation protection engineering as a replacement for lead-based multilayer coatings [104,105]. Another promising research direction is the potential use of tungsten borides as a material for the first wall of spherical tokamaks, which is in direct contact with plasma [103]. Since, in addition to its neutron-shielding properties, boron has a low atomic number (Z = 5), boron particles resulting from inevitable wall erosion would contribute far less to total bremsstrahlung radiation and plasma cooling than heavy tungsten particles [100,143,144].
Summarizing the spectrum of applications for tungsten borides, it can be stated that their utility is determined by the same set of properties that distinguishes them from classical ceramics: high hardness combined with moderate fracture toughness, metallic-type electrical conductivity, and a pronounced neutron absorption capacity. For cutting and friction assemblies, these materials prove to be most promising in scenarios where traditional carbides and nitrides reach their limits due to wear at elevated temperatures or rapid oxidative degradation. In the field of electrocatalysis, recent studies demonstrate that WB2- and WB4/WB5−x-based catalysts can serve as highly efficient catalysts for the hydrogen evolution reaction (HER) while remaining more accessible than noble metal-based catalysts, making them intriguing candidates for large-scale hydrogen production. For applications in radiation-shielding and thermonuclear energy, W–B phases offer a unique opportunity to combine the attenuation of gamma and neutron radiation by heavy W atoms with the efficient capture of thermal neutrons by boron nuclei within a single material. This potentially simplifies the design of shielding systems compared to classical multilayered solutions.
In terms of promising research directions, the following are worth noting: the synthesis of high-entropy borides, which are positioned as next-generation materials [145,146]; the creation of multilayer composite coatings to improve adhesion and increase fracture toughness; the study of catalytic properties in HER reaction; the study of hardening phenomena in nanostructured layers; the development of the CVD method to enable the production of coatings with a predictable phase composition; and the deepening of research using computational modeling to predict material properties and structural features.
5. Conclusions
The unique combination of physico-mechanical and physico-chemical properties makes tungsten borides a highly promising class of materials. The main conclusions characterizing the current state of this field are as follows:
- Structural Consensus and Superhardness: Recent fundamental breakthroughs, particularly the identification of the defective WB5−x phase, have resolved long-standing debates regarding the structure of the higher tungsten boride. This has confirmed its superhardness (>40 GPa), which is attributed to the presence of a three-dimensional covalent boron framework.
- Multifunctionality: Tungsten borides combine metallic electrical conductivity with high fracture toughness and radiation resistance. This makes them a unique choice for the most demanding applications, ranging from electrical discharge machining (EDM) tools to electrocatalysis (HER) and protective coatings for the plasma-facing first wall in thermonuclear reactors.
- Synthesis Scalability: While traditional solid-state methods and molten salt synthesis enable the production of high-purity bulk materials, thin-film technologies—primarily HiPIMS and chemical vapor deposition (CVD)—are emerging as the most promising routes for industrial scale-up. They enable the formation of dense, highly adherent coatings with a controlled phase composition on components with complex geometric shapes.
When discussing future prospects, it is important to note that the broad industrial implementation of tungsten borides depends directly on resolving several critical research challenges. The primary barrier remains the improvement in their oxidation resistance at temperatures above 700 °C. Addressing this issue will likely require transitioning to the concept of high-entropy borides or complex transition metal alloying (e.g., with Ta, Cr, or Si). From a technological standpoint, the precise prediction and control of phase stoichiometry in PVD and CVD processes necessitate the development of more advanced and comprehensive thermodynamic databases, as well as the active implementation of coupled multi-physics simulations. Furthermore, ensuring the consistency of measurement methods for fundamental physico-mechanical properties remains a pressing issue. It is essential to clearly distinguish between the strengthening effect in nanoscale coatings and the intrinsic hardness of specific tungsten boride phases, which is governed by their crystal lattice structure.
Author Contributions
Conceptualization, A.V.P., E.A.R., K.M.O., A.A.S. and V.V.D.; validation, V.V.D., E.A.R. and A.A.S.; writing—original draft preparation, A.V.P.; writing—review and editing, V.V.D.; visualization, K.M.O. All authors have read and agreed to the published version of the manuscript.
Funding
The work was supported by the Ministry of Science and Higher Education of the Russian Federation (project number 125012200581-1).
Data Availability Statement
All data and figures used in this work have been appropriately referenced.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Matkovich, V.I. Boron and Refractory Borides; Springer: Berlin/Heidelberg, Germany, 1977. [Google Scholar]
- Samsonov, G.V.; Vinickij, I.M. Handbook of Refractory Compounds; Springer: New York, NY, USA, 1980. [Google Scholar]
- Brewer, L.; Haraldsen, H. The Thermodynamic Stability of Refractory Borides. J. Electrochem. Soc. 1955, 102, 399–406. [Google Scholar] [CrossRef]
- Samsonov, G.V.; Obolonchik, V.A. Frederic Henri Moissan, on the 120th Anniversary of His Birth. Powder Metall. Met. Ceram. 1972, 11, 766–768. [Google Scholar] [CrossRef]
- Kiessling, R.; Wetterholm, A.; Sillén, L.G.; Linnasalmi, A.; Laukkanen, P. The Crystal Structures of Molybdenum and Tungsten Borides. Acta Chem. Scand. 1947, 1, 893–916. [Google Scholar] [CrossRef]
- Post, B.; Glaser, F.W.; Moskowitz, D. Transition Metal Diborides. Acta Metall. 1954, 2, 20–25. [Google Scholar] [CrossRef]
- Post, B.; Glaser, F.W. Borides of Some Transition Metals. J. Chem. Phys. 1952, 20, 1050–1051. [Google Scholar] [CrossRef]
- Meerson, G.A.; Samsonov, G.V. Preparation of Borides of High-Melting Metals by the Vacuum-Thermal Process. Zhurnal Prikl. Khimii 1954, 27, 1115–1120. [Google Scholar]
- Woods, H.P.; Wawner, F.E.; Fox, B.G. Tungsten Diboride: Preparation and Structure. Science 1966, 151, 75. [Google Scholar] [CrossRef]
- Peshev, P.; Bliznakov, G.; Leyarovska, L. On the Preparation of Some Chromium, Molybdenum and Tungsten Bo-Rides. J. Less Common Met. 1967, 13, 241–247. [Google Scholar] [CrossRef]
- Leitnaker, J.M.; Bowman, M.G.; Gilles, P.W. Thermodynamic Properties of the Tantalum and Tungsten Borides. J. Electrochem. Soc. 1962, 109, 441. [Google Scholar] [CrossRef]
- Bodrova, L.G.; Koval’chenko, M.S.; Serebryakova, T.I. Preparation of Tungsten Tetraboride. Powder Metall. Met. Ceram. 1974, 13, 1–3. [Google Scholar] [CrossRef]
- Itoh, H.; Matsudaira, T.; Naka, S.; Hamamoto, H.; Obayashi, M. Formation Process of Tungsten Borides by Solid State Reaction between Tungsten and Amorphous Boron. J. Mater. Sci. 1987, 22, 2811–2815. [Google Scholar] [CrossRef]
- Kuz’ma, Y.B.; Serebryakova, T.I.; Plakhina, A.M. The Polymorphic Transformation of Tungsten Borides—A New Phase in the W-B System. Zhurnal Neorg. Khimii 1967, 12, 559–560. [Google Scholar]
- Lundström, T. The Structure of Ru2B3 and WB2.0 as Determined by Single-Crystal Diffractometry, and Some Notes on the W-B System. Ark. Kemi 1968, 30, 115. [Google Scholar]
- Romans, P.A.; Krug, M.P. Composition and Crystallographic Data for the Highest Boride of Tungsten. Acta Cryst. 1966, 20, 313–315. [Google Scholar] [CrossRef]
- Chretien, A.; Helgorsky, J. Borides of Molybdenum and Tungsten, MoB4 and WB4; New Compounds. Compt. Rendus 1961, 252, 742. [Google Scholar]
- Nowotny, H.; Haschke, H.; Benesovsky, F. Bor-reiche Wolframboride. Monatshefte Chem. 1967, 98, 547–554. [Google Scholar] [CrossRef]
- Curtarolo, S.; Hart, G.L.W.; Nardelli, M.B.; Mingo, N.; Sanvito, S.; Levy, O. The High-Throughput Highway to Com-putational Materials Design. Nat. Mater. 2013, 12, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Greeley, J.; Jaramillo, T.F.; Bonde, J.; Chorkendorff, I.; Nørskov, J.K. Computational High-Throughput Screening of Electrocatalytic Materials for Hydrogen Evolution. Nat. Mater. 2006, 5, 909–913. [Google Scholar] [CrossRef]
- Agrawal, A.; Choudhary, A. Perspective: Materials Informatics and Big Data: Realization of the “Fourth Paradigm” of Science in Materials Science. APL Mater. 2016, 4, 053208. [Google Scholar] [CrossRef]
- Neugebauer, J.; Hickel, T. Density Functional Theory in Materials Science. WIREs Comput. Mol. Sci. 2013, 3, 438–448. [Google Scholar] [CrossRef]
- Hafner, J. Ab-initio Simulations of Materials Using VASP: Density-functional Theory and Beyond. J. Comput. Chem. 2008, 29, 2044–2078. [Google Scholar] [CrossRef]
- Vepřek, S.; Prilliman, S.G.; Clark, S.M. Elastic Moduli of Nc-TiN/a-Si3N4 Nanocomposites: Compressible, yet Su-Perhard. J. Phys. Chem. Solids 2010, 71, 1175–1178. [Google Scholar] [CrossRef]
- Solozhenko, V.L.; Andrault, D.; Fiquet, G.; Mezouar, M.; Rubie, D.C. Synthesis of Superhard Cubic BC2N. Appl. Phys. Lett. 2001, 78, 1385–1387. [Google Scholar] [CrossRef]
- Brazhkin, V.V.; Lyapin, A.G.; Hemley, R.J. Harder than Diamond: Dreams and Reality. Philos. Mag. A 2002, 82, 231–253. [Google Scholar] [CrossRef]
- Kaner, R.B.; Gilman, J.J.; Tolbert, S.H. Designing Superhard Materials. Science 2005, 308, 1268–1269. [Google Scholar] [CrossRef]
- Oganov, A.R.; Glass, C.W. Crystal Structure Prediction Using Ab Initio Evolutionary Techniques: Principles and Ap-Plications. J. Chem. Phys. 2006, 124, 244704. [Google Scholar] [CrossRef] [PubMed]
- Cumberland, R.W.; Weinberger, M.B.; Gilman, J.J.; Clark, S.M.; Tolbert, S.H.; Kaner, R.B. Osmium Diboride, An Ultra-Incompressible, Hard Material. J. Am. Chem. Soc. 2005, 127, 7264–7265. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xu, B.; Yu, D.; Ma, Y.; Wang, Y.; Jiang, Y.; Hu, W.; Tang, C.; Gao, Y.; Luo, K.; et al. Ultrahard Nanotwinned Cubic Boron Nitride. Nature 2013, 493, 385–388. [Google Scholar] [CrossRef]
- Gu, Q.; Krauss, G.; Steurer, W. Transition Metal Borides: Superhard versus Ultra-incompressible. Adv. Mater. 2008, 20, 3620–3626. [Google Scholar] [CrossRef]
- Mohammadi, R.; Lech, A.T.; Xie, M.; Weaver, B.E.; Yeung, M.T.; Tolbert, S.H.; Kaner, R.B. Tungsten Tetraboride, an Inexpensive Superhard Material. Proc. Natl. Acad. Sci. USA 2011, 108, 10958–10962. [Google Scholar] [CrossRef] [PubMed]
- Kvashnin, A.G.; Allahyari, Z.; Oganov, A.R. Computational Discovery of Hard and Superhard Materials. J. Appl. Phys. 2019, 126, 040901. [Google Scholar] [CrossRef]
- Kvashnin, A.G.; Zakaryan, H.A.; Zhao, C.; Duan, Y.; Kvashnina, Y.A.; Xie, C.; Dong, H.; Oganov, A.R. New Tung-Sten Borides, Their Stability and Outstanding Mechanical Properties. J. Phys. Chem. Lett. 2018, 9, 3470–3477. [Google Scholar] [CrossRef]
- Samsonov, G.V.; Serebryakova, T.I.; Neronov, V.A. Boridy; Atomizdat: Moscow, Russia, 1975; p. 376. [Google Scholar]
- Dushik, V.V.; Rozhanskii, N.V.; Lifshits, V.O.; Rybkina, T.V.; Kuzmin, V.P. The Formation of Tungsten and Tung-Sten Carbides by CVD Synthesis and the Proposed Mechanism of Chemical Transformations and Crystallization Pro-Cesses. Mater. Lett. 2018, 228, 164–167. [Google Scholar] [CrossRef]
- Chrzanowska-Giżyńska, J.; Denis, P.; Woźniacka, S.; Kurpaska, Ł. Mechanical Properties and Thermal Stability of Tungsten Boride Films Deposited by Radio Frequency Magnetron Sputtering. Ceram. Int. 2018, 44, 19603–19611. [Google Scholar] [CrossRef]
- Liu, X.; Cao, M.; Wen, J.; Gong, Y. Facile Synthesis of Tungsten Diboride (WB2 -191) in Molten Salt and Its Hydrogen Evolution Performance in Acidic and Alkaline Media. ACS Sustain. Chem. Eng. 2024, 12, 17460–17467. [Google Scholar] [CrossRef]
- Humphry-Baker, S.A.; Smith, G.D.W. Shielding Materials in the Compact Spherical Tokamak. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2019, 377, 20170443. [Google Scholar] [CrossRef]
- Dolmatov, O.; Kuznecov, M.S.; Semenov, A.O.; Shamanin, I.V.; Verhoturova, V.V. Obosnovanie vozmozhnosti primeneniya borsoderzhashchih materialov, poluchennyh v rezhime samorasprostranyayushchegosya vysokotemper-aturnogo sinteza. Vopr. Teor. Prakt. Radiatsionnoy Zashchity At. Energii 2021, 131, 8–11. [Google Scholar]
- Duschanek, H.; Rogl, P. Critical assessment and thermodynamic calculation of the binary system boron-tungsten (B-W). J. Phase Equilibria Diffus. 1995, 16, 150–161. [Google Scholar] [CrossRef]
- Rudy, E.; Chang, A.Y.; Windisch, S. Ternary Phase Equilibria in Transition Metal-Boron-Carbon-Silicon Systems. Part 1. Related Binary Systems; Mo-C System; Defense Technical Information Center: Fort Belvoir, VA, USA, 1965; Volume 1. [Google Scholar] [CrossRef]
- Kvashnin, A.G.; Rybkovskiy, D.V.; Filonenko, V.P.; Bugakov, V.I.; Zibrov, I.P.; Brazhkin, V.V.; Oganov, A.R.; Osiptsov, A.A.; Zakirov, A.Y. WB5−X: Synthesis, Properties, and Crystal Structure—New Insights into the Long-Debated Compound. Adv. Sci. 2020, 7, 2000775. [Google Scholar] [CrossRef]
- Springer Nature Switzerland AG. SpringerMaterials. Available online: https://materials.springer.com (accessed on 19 January 2026).
- Lech, A.T.; Turner, C.L.; Mohammadi, R.; Tolbert, S.H.; Kaner, R.B. Structure of Superhard Tungsten Tetraboride: A Missing Link between MB2 and MB12 Higher Borides. Proc. Natl. Acad. Sci. USA 2015, 112, 3223–3228. [Google Scholar] [CrossRef]
- Bykova, E.; Ovsyannikov, S.V.; Bykov, M.; Yin, Y.; Fedotenko, T.; Holz, H.; Gabel, S.; Merle, B.; Chariton, S.; Prakapenka, V.B.; et al. Crystal Structure, and Properties of Stoichiometric Hard Tungsten Tetraboride. WB4 J. Mater. Chem. A 2022, 10, 20111–20120. [Google Scholar] [CrossRef]
- Yang, J.; Yang, Z.; Lei, X.; Huang, J.; Chen, S.; Ye, Z.; Zhao, Y. Behavior and Mechanism for Boron Atom Diffusing across Tungsten Grain Boundary in the Preparation of WB Coating: A First-Principles Calculation. Appl. Surf. Sci. 2021, 543, 148778. [Google Scholar] [CrossRef]
- Fuger, C.; Hahn, R.; Zauner, L.; Wojcik, T.; Weiss, M.; Limbeck, A.; Hunold, O.; Polcik, P.; Riedl, H. Anisotropic Su-per-Hardness of Hexagonal WB2±z Thin Films. Mater. Res. Lett. 2022, 10, 70–77. [Google Scholar] [CrossRef]
- Razumov, N.; Makhmutov, T.; Kim, A.; Popovich, A. Structure and Properties of High Entropy Boride Ceramics Syn-Thesized by Mechanical Alloying and Spark Plasma Sintering. Materials 2023, 16, 6744. [Google Scholar] [CrossRef] [PubMed]
- Haponova, O.; Tarelnyk, V.; Mościcki, T.; Zielińska, K.; Myslyvchenko, O.; Bochenek, K.; Garbiec, D.; Laponog, G.; Jasinski, J.J. Improving the Wear Resistance of Steel-Cutting Tools for Nuclear Power Facilities by Electrospark Alloying with Hard Transition Metal Borides. Materials 2025, 18, 5005. [Google Scholar] [CrossRef]
- Ingole, S.; Liang, H.; Usta, M.; Bindal, C.; Ucisik, A.H. Multi-Scale Wear of a Boride Coating on Tungsten. Wear 2005, 259, 849–860. [Google Scholar] [CrossRef]
- Hossain, M.A.; Delgado, L.; Joshi, R.; Rangan, S.; Akopov, G. Probing Layered Y(TM)B4 (TM = Cr, Mo and W) Bo-Rides as Efficient Hydrogen Evolution Reaction Electrocatalysts. Chem. Commun. 2026, 62, 1497–1500. [Google Scholar] [CrossRef] [PubMed]
- Hatipoglu, E.; Peighambardoust, N.S.; Sadeghi, E.; Aydemir, U. Design of Metal-substituted Tungsten Diboride as an Efficient Bifunctional Electrocatalyst for Hydrogen and Oxygen Evolution. Int. J. Energy Res. 2022, 46, 17540–17555. [Google Scholar] [CrossRef]
- Wang, C.; Song, L.; Xie, Y. Mechanical and Electrical Characteristics of WB2 Synthesized at High Pressure and High Temperature. Materials 2020, 13, 1212. [Google Scholar] [CrossRef]
- Chrzanowska-Giżyńska, J.; Denis, P.; Giżyński, M.; Kurpaska, Ł.; Mihailescu, I.; Ristoscu, C.; Szymański, Z.; Mościcki, T. Thin WBx and WyTi1−yBx Films Deposited by Combined Magnetron Sputtering and Pulsed Laser Deposi-Tion Technique. Appl. Surf. Sci. 2019, 478, 505–513. [Google Scholar] [CrossRef]
- Yang, X.-H.; Wang, K.-F.; Zhang, G.-H. One-Step In Situ Spark Plasma Sintering (SPS) Synthesis of Dense W2B5−x Composites with High Strength and Toughness. JOM 2025. [Google Scholar] [CrossRef]
- Qin, Z.; Gong, W.; Song, X.; Wang, M.; Wang, H.; Li, Q. Effect of Pressure on the Structural, Electronic and Mechanical Properties of Ultraincompressible W2 B. RSC Adv. 2018, 8, 35664–35671. [Google Scholar] [CrossRef]
- Chen, Y.; He, D.; Qin, J.; Kou, Z.; Bi, Y. Ultrasonic and Hardness Measurements for Ultrahigh Pressure Prepared WB Ceramics. Int. J. Refract. Met. Hard Mater. 2011, 29, 329–331. [Google Scholar] [CrossRef]
- Dai, B.; Ding, X.; Deng, X.; Zhu, J.; Ran, S. Synthesis of W2B5 Powders by the Reaction between WO3 and Amorphous B in NaCl/KCl Flux. Ceram. Int. 2020, 46, 14469–14473. [Google Scholar] [CrossRef]
- Nino, A.; Sekine, T.; Sugiyama, S. Synthesis and Mechanical Properties of Silicon Nitride–Tungsten Carbide Compo-Site Ceramics. J. Jpn. Soc. Powder Powder Metall. 2022, 69, 78–86. [Google Scholar] [CrossRef]
- Kurlov, A.S.; Gusev, A.I. Tungsten Carbides: Structure, Properties and Application in Hardmetals; Springer Series in Mate-Rials Science; Springer International Publishing: Cham, Switzerland, 2013; Volume 184. [Google Scholar] [CrossRef]
- Solozhenko, V.L.; Bushlya, V.; Zhou, J. Mechanical Properties of Ultra-Hard Nanocrystalline Cubic Boron Nitride. J. Appl. Phys. 2019, 126, 075107. [Google Scholar] [CrossRef]
- Dubrovinskaia, N.; Solozhenko, V.L.; Miyajima, N.; Dmitriev, V.; Kurakevych, O.O.; Dubrovinsky, L. Superhard Nanocomposite of Dense Polymorphs of Boron Nitride: Noncarbon Material Has Reached Diamond Hardness. Appl. Phys. Lett. 2007, 90, 101912. [Google Scholar] [CrossRef]
- Hall, E.O. The Deformation and Ageing of Mild Steel: III Discussion of Results. Proc. Phys. Soc. B 1951, 64, 747–753. [Google Scholar] [CrossRef]
- Petch, N.J. The Cleavage Strength of Polycrystals. J. Iron Steel Inst. 1953, 174, 25–28. [Google Scholar]
- Yang, L.; Zhang, K.; Wen, M.; Hou, Z.; Gong, C.; Liu, X.; Hu, C.; Cui, X.; Zheng, W. Highly Hard yet Toughened Bcc-W Coating by Doping Unexpectedly Low B Content. Sci. Rep. 2017, 7, 9353. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-M.; Li, T.; Liu, F.; Pei, Z.-L. Thermal Stability of WB2 and W–B–N Films Deposited by Magnetron Sputtering. Acta Metall. Sin. (Engl. Lett.) 2019, 32, 136–144. [Google Scholar] [CrossRef]
- Mościcki, T.; Chrzanowska-Giżyńska, J.; Psiuk, R.; Denis, P.; Mulewska, K.; Kurpaska, Ł.; Chmielewski, M.; Wiśniewska, M.; Garbiec, D. Thermal and Mechanical Properties of (W,Zr)B2-z Coatings Deposited by RF Magnetron Sputtering Method. Int. J. Refract. Met. Hard Mater. 2022, 105, 105811. [Google Scholar] [CrossRef]
- Psiuk, R.; Milczarek, M.; Jenczyk, P.; Denis, P.; Jarząbek, D.M.; Bazarnik, P.; Pisarek, M.; Mościcki, T. Improved Me-Chanical Properties of W-Zr-B Coatings Deposited by Hybrid RF Magnetron—PLD Method. Appl. Surf. Sci. 2021, 570, 151239. [Google Scholar] [CrossRef]
- Psiuk, R.; Mościcki, T.; Chrzanowska-Giżyńska, J.; Kurpaska, Ł.; Radziejewska, J.; Denis, P.; Garbiec, D.; Chmielew-ski, M. Mechanical and Thermal Properties of W-Ta-B Coatings Deposited by High-Power Impulse Magnetron Sputter-Ing (HiPIMS). Materials 2023, 16, 664. [Google Scholar] [CrossRef]
- Swab, J.J.; Tice, J.; Wereszczak, A.A.; Kraft, R.H. Fracture Toughness of Advanced Structural Ceramics: Applying ASTM C1421. J. Am. Ceram. Soc. 2015, 98, 607–615. [Google Scholar] [CrossRef]
- Hevorkian, E.; Samociuk, W.; Rucki, M.; Krzysiak, Z.; Pieniak, D.; Nerubatskyi, V.; Chyshkala, V.; Lytovchenko, S.; Chalko, L.; Morozow, D.; et al. Microstructure and Properties of Binderless μWC Obtained Using the Elec-Troconsolidation Method. Materials 2025, 18, 4646. [Google Scholar] [CrossRef] [PubMed]
- Gludovatz, B.; Wurster, S.; Hoffmann, A.; Pippan, R. Fracture Toughness of Polycrystalline Tungsten Alloys. Int. J. Refract. Met. Hard Mater. 2010, 28, 674–678. [Google Scholar] [CrossRef]
- Maździarz, M.; Psiuk, R.; Krawczyńska, A.; Lewandowska, M.; Mościcki, T. Effect of Zirconium Doping on the Me-Chanical Properties of W1-xZrxB2 on the Basis of First-Principles Calculations and Magnetron Sputtered Films. Arch. Civ. Mech. Eng. 2022, 22, 193. [Google Scholar] [CrossRef]
- Matsokin, N.A.; Eremin, R.A.; Kuznetsova, A.A.; Humonen, I.S.; Krautsou, A.V.; Lazarev, V.D.; Vassilyeva, Y.Z.; Pak, A.Y.; Budennyy, S.A.; Kvashnin, A.G.; et al. Discovery of Chemically Modified Higher Tungsten Boride by Means of Hybrid GNN/DFT Approach. npj Comput. Mater. 2025, 11, 163. [Google Scholar] [CrossRef]
- Hahn, R.; Bartosik, M.; Soler, R.; Kirchlechner, C.; Dehm, G.; Mayrhofer, P.H. Superlattice Effect for Enhanced Fracture Toughness of Hard Coatings. Scr. Mater. 2016, 124, 67–70. [Google Scholar] [CrossRef]
- Kumar, D.D.; Kumar, N.; Kalaiselvam, S.; Dash, S.; Jayavel, R. Wear Resistant Super-Hard Multilayer Transition Metal-Nitride Coatings. Surf. Interfaces 2017, 7, 74–82. [Google Scholar] [CrossRef]
- Fuger, C.; Schwartz, B.; Wojcik, T.; Moraes, V.; Weiss, M.; Limbeck, A.; Macauley, C.A.; Hunold, O.; Polcik, P.; Primetzhofer, D.; et al. Influence of Ta on the Oxidation Resistance of WB2−z Coatings. J. Alloys Compd. 2021, 864, 158121. [Google Scholar] [CrossRef]
- Wu, Z.; Ye, R.; Bakhit, B.; Petrov, I.; Hultman, L.; Greczynski, G. Improving Oxidation and Wear Resistance of TiB2 Films by Nano-Multilayering with Cr. Surf. Coat. Technol. 2022, 436, 128337. [Google Scholar] [CrossRef]
- Jiang, C.L.; Pei, Z.L.; Liu, Y.M.; Lei, H.; Gong, J.; Sun, C. Determination of the Thermal Properties of AlB2-Type WB2. Appl. Surf. Sci. 2014, 288, 324–330. [Google Scholar] [CrossRef]
- Boyer, H.E.; Gall, T.L. Metals Handbook, Desk ed.; American Society for Metals: Metals Park, OH, USA, 1985. [Google Scholar]
- Hatch, J.E. Aluminum: Properties and Physical Metallurgy; Aluminum Association, American Society for Metals, Eds.; American Society for Metals: Metals Park, OH, USA, 1984. [Google Scholar]
- Donachie, M.J. Titanium: A Technical Guide; ASM International: Materials Park, OH, USA, 2000. [Google Scholar]
- White, G.K.; Minges, M.L. Thermophysical Properties of Some Key Solids: An Update. Int. J. Thermophys. 1997, 18, 1269–1327. [Google Scholar] [CrossRef]
- Emmerich, T.; Qu, D.; Vaßen, R.; Aktaa, J. Development of W-Coating with Functionally Graded W/EUROFER-Layers for Protection of First-Wall Materials. Fusion Eng. Des. 2018, 128, 58–67. [Google Scholar] [CrossRef]
- Yu, M.H.; Zhou, B.; Bi, D.B.; Shaw, D. Preparation of Graded Multilayer Materials and Evaluation of Residual Stresses. Mater. Des. 1980, 31, 2478–2482. [Google Scholar] [CrossRef]
- Glechner, T.; Oemer, H.G.; Wojcik, T.; Weiss, M.; Limbeck, A.; Ramm, J.; Polcik, P.; Riedl, H. Influence of Si on the Oxidation Behavior of TM-Si-B2±z Coatings (TM = Ti, Cr, Hf, Ta, W). Surf. Coat. Technol. 2022, 434, 128178. [Google Scholar] [CrossRef]
- Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions; Association for Materials Protection and Performance (AMPP): Houston, TX, USA, 1974. [Google Scholar]
- Dushik, V.V.; Redkina, G.V.; Rozhanskii, N.V.; Rybkina, T.V.; Shaporenkov, A.A.; Maschenko, V.E. Corrosion and Electrochemical Behavior of β-W CVD Coatings in NaCl Solution. Prot. Met. Phys. Chem. Surfaces 2020, 56, 1321–1324. [Google Scholar] [CrossRef]
- Anik, M. Anodic Behavior of Tungsten in H3PO4-K2SO4-H2SO4/KOH Solutions. Turk. J. Chem. 2002, 26, 915–924. [Google Scholar]
- Lillard, R.S.; Kanner, G.S.; Butt, D.P. The Nature of Oxide Films on Tungsten in Acidic and Alkaline Solutions. J. Electrochem. Soc. 1998, 145, 2718–2725. [Google Scholar] [CrossRef]
- Stadler, S.; Winarski, R.P.; MacLaren, J.M.; Ederer, D.L.; vanEk, J.; Moewes, A.; Grush, M.M.; Callcott, T.A.; Perera, R.C.C. Electronic Structures of the Tungsten Borides WB, W2B and W2B5. J. Electron Spectrosc. Relat. Phenom. 2000, 110–111, 75–86. [Google Scholar] [CrossRef]
- Properties and Selection: Nonferrous Alloys and Special-Purpose Materials. In ASM Handbook; ASM International: Materials Park, OH, USA, 2007.
- Shackelford, J.F.; Han, Y.-H.; Kim, S.; Kwonm, S.-H. (Eds.) CRC Materials Science and Engineering Handbook, 4th ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Boca Raton, FL, USA, 2016. [Google Scholar]
- Samsonov, G.V. Plenum Press Handbooks of High-Temperature Materials: No 2 Properties Index; Plenum Press: New York, NY, USA, 1964. [Google Scholar]
- Kayhan, M.; Hildebrandt, E.; Frotscher, M.; Senyshyn, A.; Hofmann, K.; Alff, L.; Albert, B. Neutron Diffraction and Observation of Superconductivity for Tungsten Borides, WB and W2B4. Solid State Sci. 2012, 14, 1656–1659. [Google Scholar] [CrossRef]
- Khanra, A.K.; Sarkar, B.R.; Bhattacharya, B.; Pathak, L.C.; Godkhindi, M.M. Performance of ZrB2–Cu Composite as an EDM Electrode. J. Mater. Process. Technol. 2007, 183, 122–126. [Google Scholar] [CrossRef]
- Mathan Kumar, P.; Sivakumar, K.; Vinu Kumar, S.M. Experimental Investigation of Electrical Dis-Charge Machining of Inconel 718 Using a TiB2-Cu Sintered Composite Electrode. Mater. Tehnol. 2022, 56, 131–138. [Google Scholar] [CrossRef]
- Luo, C.; Xu, L.; Zong, L.; Shen, H.; Wei, S. Research Status of Tungsten-Based Plasma-Facing Materials: A Review. Fusion Eng. Des. 2023, 190, 113487. [Google Scholar] [CrossRef]
- Philipps, V. Tungsten as Material for Plasma-Facing Components in Fusion Devices. J. Nucl. Mater. 2011, 415, 2–9. [Google Scholar] [CrossRef]
- Safford, G.J.; Taylor, T.I.; Rustad, B.M.; Havens, W.W. Precision Determination of the Slow Neutron Absorption Cross Section of B 10. Phys. Rev. 1960, 119, 1291–1294. [Google Scholar] [CrossRef]
- Yang, L.; Wirth, B.D. First-Principles Study of Diffusion of Intrinsic Point Defects in Tungsten Borides. J. Nuclear Mater. 2024, 591, 154931. [Google Scholar] [CrossRef]
- Windsor, C.G.; Astbury, J.O.; Davidson, J.J.; McFadzean, C.J.R.; Morgan, J.G.; Wilson, C.L.; Humphry-Baker, S.A. Tungsten Boride Shields in a Spherical Tokamak Fusion Power Plant. Nucl. Fusion 2021, 61, 086018. [Google Scholar] [CrossRef]
- Avcıoğlu, C.; Avcıoğlu, S. Transition Metal Borides for All-in-One Radiation Shielding. Materials 2023, 16, 6496. [Google Scholar] [CrossRef]
- Erdogan, F.; Bermudez, S.; Mohammadi, R.; Rojas, J.V. Enhanced Radiation Shielding Performance of Tungsten Bo-Rides-Epoxy Composites. Compos. Sci. Technol. 2025, 269, 111233. [Google Scholar] [CrossRef]
- Jung, Y.; Li, J. Boron-10 Stimulated Helium Production and Accelerated Radiation Displacements for Rapid Devel-Opment of Fusion Structural Materials. J. Mater. 2024, 10, 377–385. [Google Scholar] [CrossRef]
- Li, Y.-P.; Ran, G.; Liu, X.-Y.; Qiu, X.; Han, Q.; Li, W.-J.; Guo, Y.-J. In-Situ TEM Observation of the Evolution of Helium Bubbles in Mo during He+ Irradiation and Post-Irradiation Annealing. Chin. Phys. B 2021, 30, 086109. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Lu, Y.; Wang, X.; Duan, Y.; Peng, M. Theoretical Insights to Elastic and Thermal Properties of WB4 Tetraborides: A First-Principles Calculation. Vacuum 2022, 196, 110731. [Google Scholar] [CrossRef]
- Hirama, K.; Taniyasu, Y.; Yamamoto, H.; Kumakura, K. Control of N-Type Electrical Conductivity for Cubic Boron Nitride (c-BN) Epitaxial Layers by Si Doping. Appl. Phys. Lett. 2020, 116, 162104. [Google Scholar] [CrossRef]
- Ghorbantabar Omran, J.; Sharifitabar, M.; Shafiee Afarani, M. On the Self-Propagating High-Temperature Synthesis of Tungsten Boride Containing Composite Powders from WO3–B2O3–Mg System. Ceram. Int. 2018, 44, 14355–14362. [Google Scholar] [CrossRef]
- Yazici, S.; Derin, B. Production of Tungsten Boride from CaWO4 by Self-Propagating High-Temperature Synthesis Followed by HCl Leaching. Int. J. Refract. Met. Hard Mater. 2011, 29, 90–95. [Google Scholar] [CrossRef]
- Cano, A.M.; Marquardt, A.E.; DuMont, J.W.; George, S.M. Effect of HF Pressure on Thermal Al2 O3 Atomic Layer Etch Rates and Al2O3 Fluorination. J. Phys. Chem. C 2019, 123, 10346–10355. [Google Scholar] [CrossRef]
- Ćurković, L.; Jelača, M.F.; Kurajica, S. Corrosion Behavior of Alumina Ceramics in Aqueous HCl and H2SO4 Solu-Tions. Corros. Sci. 2008, 50, 872–878. [Google Scholar] [CrossRef]
- Yeh, C.L.; Wang, H.J. Preparation of Tungsten Borides by Combustion Synthesis Involving Borothermic Reduction of WO3. Ceram. Int. 2011, 37, 2597–2601. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.-B.; Wang, K.-F.; Zhang, G.-H. Preparation of Monophasic Tungsten Boride Powder from Tungsten and Boron Carbide. Ceram. Int. 2021, 47, 9543–9550. [Google Scholar] [CrossRef]
- Woo, K.E.; Kong, S.; Chen, W.; Chang, T.H.; Viswanathan, G.; Díez, A.M.; Sousa, V.; Kolen’ko, Y.V.; Lebedev, O.I.; Costa Figueiredo, M.; et al. Topotactic BI3 -Assisted Borodization: Synthesis and Electrocatalysis Applications of Transition Metal Borides. J. Mater. Chem. A 2022, 10, 21738–21749. [Google Scholar] [CrossRef]
- Malyshev, V.V. High-Temperature Electrochemical Synthesis of Tungsten and Molybdenum Boride Phases in Chloride—Cryolite—Oxide Melts. ChemInform 1998, 29, 199836034. [Google Scholar] [CrossRef]
- Matiašovský, K.; Chrenková-Paučírová, M.; Fellner, P.; Makyta, M. Electrochemical and Thermochemical Boriding in Molten Salts. Surf. Coat. Technol. 1988, 35, 133–149. [Google Scholar] [CrossRef]
- Powell, C.F.; Blocher, J.M.; Oxley, J.H. Vapor Deposition; Electrochemical Society Series; Powell, C.F., Oxley, J.H., John, M., Blocher, E., Jr., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 1966. [Google Scholar]
- Choy, K. Chemical Vapour Deposition of Coatings. Progress Mater. Sci. 2003, 48, 57–170. [Google Scholar] [CrossRef]
- Pierson, H.O. Handbook of Chemical Vapor Deposition (CVD): Principles, Technology, and Applications; Noyes Publications: Park Ridge, NJ, USA, 1992. [Google Scholar]
- Martin, P.M. Handbook of Deposition Technologies for Films and Coatings: Science, Applications and Technology, 3rd ed.; William Andrew: Norwich, NY, USA, 2009. [Google Scholar]
- Handbook of Physical Vapor Deposition (PVD) Processing, 2nd ed.; Mattox, D.M., Ed.; William Andrew: Oxford, UK, 2010. [Google Scholar]
- Chapman, B. Glow Discharge Processes: Sputtering and Plasma Etching; A Wiley Interscience Publication; Wiley: New York, NY, USA, 1980. [Google Scholar]
- Gudmundsson, J.T.; Brenning, N.; Lundin, D.; Helmersson, U. High Power Impulse Magnetron Sputtering Dis-Charge. J. Vac. Sci. Technol. A Vac. Surf. Film. 2012, 30, 030801. [Google Scholar] [CrossRef]
- Samuelsson, M.; Lundin, D.; Jensen, J.; Raadu, M.A.; Gudmundsson, J.T.; Helmersson, U. On the Film Density Us-Ing High Power Impulse Magnetron Sputtering. Surf. Coat. Technol. 2010, 205, 591–596. [Google Scholar] [CrossRef]
- Alami, J.; Bolz, S.; Sarakinos, K. High Power Pulsed Magnetron Sputtering: Fundamentals and Applications. J. Alloys Compd. 2009, 483, 530–534. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, G.; Wang, Z.; Yuan, J.; Ke, P.; Wang, A. HiPIMS Induced High-Purity Ti3AlC2 MAX Phase Coating at Low-Temperature of 700 °C. J. Eur. Ceram. Soc. 2023, 43, 4673–4683. [Google Scholar] [CrossRef]
- Xia, M.; Ding, H.; Zhou, G.; Zhang, Y. Improvement of Adhesion Properties of TiB2 Films on 316L Stainless Steel by Ti Interlayer Films. Trans. Nonferrous Met. Soc. China 2013, 23, 2957–2961. [Google Scholar] [CrossRef]
- Pan, Z.; Wu, Z.; Li, F.; Li, H.; Wang, Q. Improving Toughness and Oxidation Resistance of TiB2 Films by Introduc-Ing W Interlayer. Surf. Coat. Technol. 2025, 512, 132438. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, X.; Lai, C.; Ma, J.; Meng, X.; Zhang, L.; Xu, G.; Lu, Y.; Li, H.; Wang, J.; et al. Boriding of Tungsten by the Powder-Pack Process: Phase Formation, Growth Kinetics and Enhanced Neutron Shielding. Int. J. Refract. Met. Hard Mater. 2023, 110, 106049. [Google Scholar] [CrossRef]
- Jones, M.; Horlock, A.J.; Shipway, P.H.; McCartney, D.G.; Wood, J.V. A Comparison of the Abrasive Wear Behav-Iour of HVOF Sprayed Titanium Carbide- and Titanium Boride-Based Cermet Coatings. Wear 2001, 251, 1009–1016. [Google Scholar] [CrossRef]
- Bolelli, G.; Lyphout, C.; Berger, L.-M.; Testa, V.; Myalska-Głowacka, H.; Puddu, P.; Sassatelli, P.; Lusvarghi, L. Wear Resistance of HVOF- and HVAF-Sprayed (Ti,Mo)(C,N)–Ni Coatings from an Agglomerated and Sintered Powder. Wear 2023, 512–513, 204550. [Google Scholar] [CrossRef]
- Brezinová, J.; Guzanová, A.; Draganovská, D.; Brezina, J. Characterization of Selected Properties of WC–WB–Co and WC–FeCrAl Coatings Applied by HVOF Technology. Koroze A Ochr. Mater. 2019, 63, 167–173. [Google Scholar] [CrossRef]
- Wang, Q.; Jin, Z.; Zhao, Y.; Niu, L.; Guo, J. A Comparative Study on Tool Life and Wear of Uncoated and Coated Cutting Tools in Turning of Tungsten Heavy Alloys. Wear 2021, 482–483, 203929. [Google Scholar] [CrossRef]
- Sun, S.; Wang, H.; Liu, X.; Liu, C.; Lu, H.; Nie, Z.; Song, X. Outstanding Anti-Oxidation Performance of Boride Coat-Ing under High-Temperature Friction. Corros. Sci. 2021, 179, 109133. [Google Scholar] [CrossRef]
- Debnárová, S.; Souček, P.; Vašina, P.; Zábranský, L.; Buršíková, V.; Mirzaei, S.; Pei, Y.T. The Tribological Properties of Short Range Ordered W-B-C Protective Coatings Prepared by Pulsed Magnetron Sputtering. Surf. Coat. Technol. 2019, 357, 364–371. [Google Scholar] [CrossRef]
- Wang, A.; Shen, L.; Zhao, M.; Wang, J.; Zhou, W.; Li, W.; Feng, Y.; Liu, H. Tungsten Boride: A 2D Multiple Dirac Semimetal for the Hydrogen Evolution Reaction. J. Mater. Chem. C 2019, 7, 8868–8873. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, Z.; Jiao, L. Development Strategies in Transition Metal Borides for Electrochemical Water Splitting. Energy Environ. Mater. 2022, 5, 470–485. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Y.; Long, Y.; Lin, H.-T. Electrocatalytic Activity of WB2 Electrodes for Hydrogen Evolution Reac-Tion Prepared by Different Methods. J. Eur. Ceram. Soc. 2025, 45, 117185. [Google Scholar] [CrossRef]
- Wang, H.; Yang, X.; Bao, L.; Zong, Y.; Gao, Y.; Miao, Q.; Zhang, M.; Ma, R.; Zhao, J. Nanocrystalline Transition Metal Tetraborides as Efficient Electrocatalysts for Hydrogen Evolution Reaction at the Large Current Density. J. Colloid Interface Sci. 2025, 677, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Zhang, Y.; Lee, E.; Shankhari, P.; Fokwa, B.P.T. High-Current-Density HER Electrocatalysts: Gra-Phene-like Boron Layer and Tungsten as Key Ingredients in Metal Diborides. ChemSusChem 2019, 12, 3726–3731. [Google Scholar] [CrossRef]
- Bolt, H.; Barabash, V.; Federici, G.; Linke, J.; Loarte, A.; Roth, J.; Sato, K. Plasma Facing and High Heat Flux Materials—Needs for ITER and Beyond. J. Nucl. Mater. 2002, 307–311, 43–52. [Google Scholar] [CrossRef]
- Dibon, M.; Rohde, V.; Stelzer, F.; Hegele, K.; Uhlmann, M. New Boronization System at ASDEX Upgrade. Fusion Eng. Des. 2021, 165, 112233. [Google Scholar] [CrossRef]
- Gild, J.; Zhang, Y.; Harrington, T.; Jiang, S.; Hu, T.; Quinn, M.C.; Mellor, W.M.; Zhou, N.; Vecchio, K.; Luo, J. High-Entropy Metal Diborides: A New Class of High-Entropy Materials and a New Type of Ultrahigh Temperature Ce-Ramics. Sci. Rep. 2016, 6, 37946. [Google Scholar] [CrossRef]
- Xu, H.; Jiang, L.; Chen, K.; Huang, Q.; Zhou, X. High-Entropy Rare-Earth Diborodicarbide: A Novel Class of High-Entropy (Y0.25Yb0.25Dy0.25Er0.25)B2C2 Ceramics. J. Adv. Ceram. 2023, 12, 1430–1440. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.