Abstract
Dye-sensitized solar cells (DSSCs) represent a promising low-cost photovoltaic technology with relatively high conversion efficiency and a simple fabrication process. Natural dyes have drawn growing interest compared to ruthenium-based dyes since they are greener. However, the power conversion efficiency (PCE) of natural dyes is generally low. In this study, we investigated novel approaches to improve the PCE of DSSCs using Delonix regia extracts by polarity-based separation using preparative thin-layer chromatography (PTLC). Our study indicated that polarity-based separation can significantly enhance the PCE, with one fraction achieving a PCE of 1.13%, which is high compared to most natural dye-based DSSCs, and is also 1.85 times that of the crude methanol extract. The major compounds in the highest-efficiency layer were flavanol-based dyes. Our study demonstrates the potential antagonistic effects within Delonix regia extracts in DSSC applications, which play a critical role in improving PCE. The study is expected to support future efforts to enhance the PCE of natural compound-based DSSCs, especially those using flavanol-based natural dyes.
1. Introduction
Energy is a fundamental requirement for sustaining life in the universe. Currently, fossil fuels serve as the dominant global energy source; however, their environmental and economic drawbacks have spurred the development of cleaner, more sustainable renewable energy alternatives [1]. In recent years, renewable energy sources, particularly solar energy, have shown significant promise due to their near-limitless availability [2]. Among these, dye-sensitized solar cells (DSSCs) are a class of thin-film photovoltaics in which a nanoporous, wide-bandgap semiconductor (e.g., TiO2), sensitized with a molecular chromophore, serves as the photoanode for harvesting visible light [3]. DSSCs have attracted significant research interest due to their straightforward fabrication process [4,5], low material costs [5], achievable power conversion efficiencies, and a rapid energy payback period [6,7]. The power conversion efficiency (PCE) of DSSC has been reported as high as 15.2% under AM 1.5G illumination in a recent study [8] with newly designed co-sensitized dyes, which is the highest PCE reported at the current stage. Meanwhile, the N719 dye, di-tetrabutylammonium cis-bis(isothiocyanato)bis(2,2′-bipyridyl-4,4′-dicarboxylato)ruthenium(II), exhibits one of the best-performed PCEs in DSSCs, achieving over 13% in laboratory settings under simulated sunlight [9]. Despite its excellent performance as a dye sensitizer, N719 is expensive and not environmentally friendly [10]. Organic sensitizers with designed π-conjugated systems, particularly donor-π-acceptor structured molecules, have emerged as promising candidates to replace ruthenium complexes in DSSCs [11,12,13]. Nevertheless, their practical implementation is often hindered by challenges such as expensive starting materials, laborious synthetic protocols, and non-eco-friendly reaction byproducts [14].
As sustainable alternatives, natural pigments derived from plants and flowers, such as chlorophyll, carotenoids, and flavonoids, offer cost-effective and eco-friendly sensitizers for DSSCs [15]. These natural dyes exhibit favorable light-harvesting characteristics, including broad absorption across the visible spectrum and the potential presence of strong TiO2-anchoring groups, such as –OH and –COOH in chlorophyll, carotenoids, and flavonoids, which facilitating efficient electron injection [16,17]. A natural dye-sensitized solar cell based on chlorophyll c2 extracted from the seaweed Undaria pinnatifida (wakame) achieved a conversion efficiency of 4.6%, representing the highest efficiency reported to date for natural DSSCs [18]. Although natural dye-based DSSCs demonstrate significantly lower power conversion efficiencies compared to their synthetic counterparts, they remain promising for low-cost, greener solar applications [19,20,21]. Plant-derived pigments exhibit varying photovoltaic performance in DSSCs, with energy conversion efficiencies dependent on their molecular structure and adsorption characteristics on mesoporous TiO2 surfaces. The typically low efficiency of natural dye-sensitized cells primarily stems from the predominance of hydroxyl and carbonyl functional groups, which form weak interfacial bonds with the semiconductor oxide. This inadequate binding compromises electron injection efficiency from the photoexcited dye into the TiO2 conduction band [10]. For efficient charge transfer and improved DSSC performance, the dye molecular orbital energies must satisfy two key requirements: (1) the highest occupied molecular orbital (HOMO) level should lie sufficiently below the redox potential of the electrolyte, and (2) the lowest unoccupied molecular orbital (LUMO) level must be positioned above the TiO2 conduction band edge while remaining in close energetic proximity to it. However, the inherent complexity of natural dye compositions often leads to uncontrolled aggregation on TiO2 surfaces and enhanced charge recombination, both of which negatively impact photovoltaic performance [22,23]. These effects collectively reduce efficiency through aggregation-mediated excited-state quenching [21], suboptimal electron injection from randomly oriented dyes [24], and recombination at heterogeneous dye-TiO2 interfaces [25].
Delonix regia, commonly known as the flame tree, is cultivated in subtropical and warm temperate regions worldwide. Both its leaves and flowers exhibit medicinal properties, demonstrating hepatoprotective [26], cardioprotective [27], and antidiabetic activities in pharmacological studies [28]. These biological activities of D. regia are primarily mediated by its flavonoid constituents, particularly anthocyanins, quercetin, and kaempferol, which act as polyphenolic antioxidants, scavenging reactive oxygen species (ROS) and modulating oxidative stress pathways [29,30,31]. D. regia has been investigated as a natural sensitizer in DSSCs, with reported efficiencies varying significantly depending on extraction methods and solvents. Vijendran et al. [32] achieved an efficiency of 0.897% using ethanol and isopropyl alcohol as solvents, whereas Sanda et al. [33] reported an efficiency of 0.10% using ethanol-only extraction. Srivastava et al. [34] observed an efficiency of 0.17% using aqueous (distilled water) extraction. In our prior study, methanol-extracted D. regia flower dye demonstrated an efficiency of 0.70% [35]. These discrepancies highlight the complexity of natural sensitizer systems, in which factors such as solvent polarity, extraction methodology, and dye aggregation critically influence photovoltaic performance [36,37]. In this study, D. regia dye components were fractionated using preparative thin-layer chromatography (PTLC), yielding four distinct pigment layers. Layer 4 (the anthocyanin-enriched fraction) exhibited a 1.2 times increase in DSSC efficiency compared to the crude extract, while Layer 3 (containing quercetin and kaempferol) demonstrated a more pronounced 1.85 times enhancement. These results highlight the significant impact of flavonoid speciation on photovoltaic performance, with flavonol glycosides (Layer 3) outperforming anthocyanins (Layer 4) under identical experimental conditions.
2. Materials and Methods
2.1. General Materials
Titanium (IV) oxide (Aeroxide P-25), triton X-100, lithium iodide (99%), t-butyammonium iodide (98%), t-butylpyridine (96%), crystalline iodine (99%), and acetylacetone (99%) were purchased from Fisher Scientific (Waltham, MA, USA). N719 dye, TCO plates, Plastisol T/SP and Meltonix 1170-60 membrane were purchased from SOLARONIX S.A. (Aubonne, Switzerland). Distilled water (DI water) was obtained from an Evoqua Water Technologies deionization system (Evoqua Water Technologies LLC, Pittsburgh, PA, USA). All organic solvents, including methanol, ethanol, acetone, and hexane, were purchased from Fisher Scientific (Waltham, MA, USA). Preparative thin layer chromatography (PTLC) silica gel plates (1000 mm, 20 × 20 cm) were purchased from Fisher Scientific (Waltham, MA, USA). UV–visible electronic absorption spectra were measured by Jasco V-750 Spectrophotometer (Jasco Corporation, Tokyo, Japan). Photovoltaic measurements were performed using an LCS-100 Solar simulator and a PVIV-1A solar meter from Newport Corporation (Irvine, CA, USA). Dried samples were re-dissolved in d4-methanol, and NMR spectra were acquired through proton experiments using a JEOL 400 MHz high-resolution NMR (JEOL, Tokyo, Japan).
2.2. Delonix regia Extraction and Separation Method
D. regia flowers were collected from the New College of Florida campus from May and July 2023, corresponding to their blooming season. The flowers were washed twice with tap water, followed by rinsing with DI water to remove dust and debris. After drying in the oven at 60 °C for 24 h, the samples were ground into a fine powder and stored in the dark until further use. Natural pigments were extracted from the powdered material using methanol at a sample to solvent ratio of 1 g per 10 mL via sonication for 15 min. Following centrifugation, the methanol was completely removed using a rotary evaporator, yielding dry pigments for subsequent experiments.
A few drops of acetone were added to the dried dye to obtain an orange pigment, which will be ready for preparative TLC. The remaining orange colored residue that was insoluble in acetone was collected, separated, and redissolved in methanol. The acetone soluble fraction was further separated into three layers by PTLC plates using the solvent system of acetone and hexane with a ratio of 3:7 (v/v). Based on increasing polarity on the TLC plates, the three layers were designated as L1 (bright yellow), L2 (orange), and L3 (dark orange/light brown). The methanol soluble fraction was labelled L4 (orange/brown).
2.3. Dye-Sensitized Solar Cells Fabrication and Measurement
The TiO2 film was coated onto a TCO glass following a previously reported method by Nazeeruddin et al. [38] with minor modifications [35]. Briefly, commercial Titanium (IV) oxide P25 (3 g) was mixed with acetylacetone (0.1 mL), deionized water (4 mL), and one drop of Triton X-100 using a mortar and pestle. The resulting paste was applied to TCO glass (17 × 25 mm) by the doctor blade technique using Scotch Magic tape (3M Center, St. Paul, MN, USA). The thickness of TiO2 films was approximately 0.055 mm. After air-drying for 30 min, the film was annealed at 450 °C for 30 min in a Muffle oven. The TiO2 coated electrode was then immersed in the dye solution (approximately 3 mg/mL) for 1 h [39] and dried at room temperature for 10 min prior to the efficiency test. The counter electrode was prepared by coating Plastisol paste onto another TCO glass, followed by annealing at 450 °C for 30 min. The electrolyte solution contained lithium iodide (0.0669 g), iodine (0.1269 g), t-butylammonium iodide (1.1081 g), and t-butylpyridine (0.366 mL) dissolved in 5 mL of acetonitrile. The DSSC was fabricated using Meltonix 1170-60 as a sealant with an active area of 0.1 cm2 (0.33 cm × 0.33 cm) for photovoltaic measurements. Each experiment was conducted in triplicate. The current–voltage (I–V) characteristics were recorded under white light irradiation (100 mW/cm2) using a Newport solar simulator (Newport, Irvine, CA, USA). The performance of the natural dye sensitizers was evaluated in terms of short-circuit current (Jsc), open-circuit voltage (Voc), fill factor (FF), and energy conversion efficiency (η%).
2.4. UV-Vis Spectroscopy
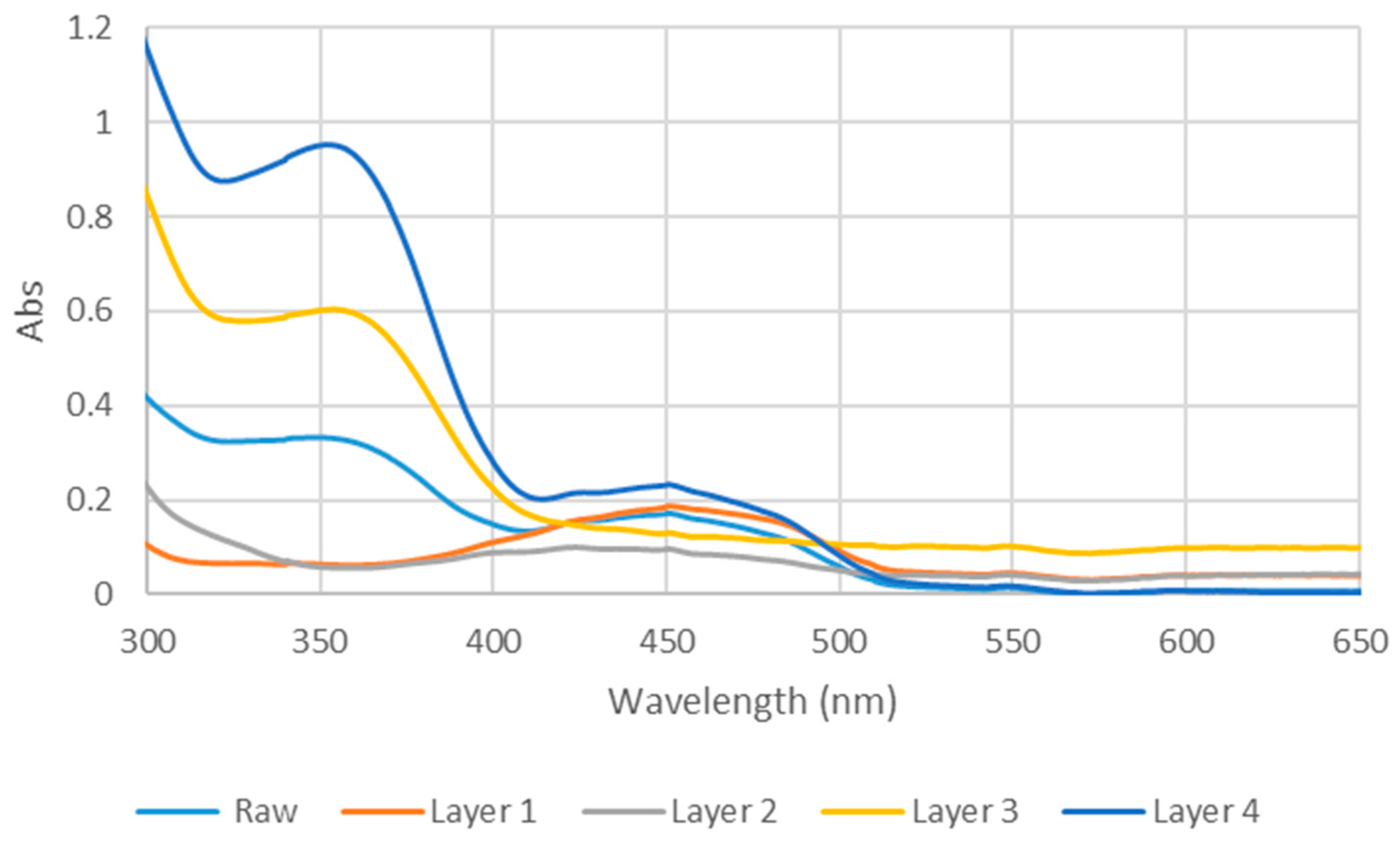
The absorption spectra of the raw D. regia extract and the four fractions (L1–L4) obtained from the separation process were recorded in methanol solution using a Jasco V-750 Spectrophotometer (Jasco Co.). Measurements were performed over the wavelength range of 300–650 nm, as none of the dyes exhibited absorption beyond 650 nm.
2.5. 1H NMR Acquisition
The crude D. regia extract and the four separated fractions were collected and dried. Each sample was then resuspended in d4-methanol in NMR tubes according to its solubility. Proton NMR spectra were acquired using a JEOL 400 MHz high-resolution NMR spectrometer.
3. Results
3.1. Delonix regia Separation
D. regia (Figure 1A) was found to be separated into four major fractions. Three fractions were obtained by preparative TLC, as shown in Figure 1B. The remaining compounds that were insoluble in acetone were dissolved in methanol and labeled as the methanol fraction L4. The top fraction, L1, exhibited a strong yellow band. L2 contained several closely migrating peaks, reflecting similar polarity on the preparative TLC plate. L3, the most polar fraction, appeared brownish orange. Fraction L4, consisting of the acetone-insoluble pigments, was a brownish layer. The solutions of D. regia and four separate layers were illustrated in Figure 1C.

Figure 1.
(A) Picture of D. regia flower. (B) Preparative TLC plate of pigments extracted from D. regia using a mobile phase with 3:7 acetone and hexane. (C) The solutions of the crude extract of D. regia and its four individual fractions separated by preparative TLC.
3.2. Photovoltaic Performance
The photoelectrochemical parameters of the DSSCs sensitized with the natural dye fractions showed significant variations in efficiency compared to the crude flower extract (Table 1). N719 dye was used as a reference in this study. The methanol-soluble anthocyanin-rich fraction L4 exhibited a 1.2-fold higher PCE than the crude dye, while the polar flavonoid-dominant fraction L3, demonstrated superior performance with a 1.85-fold increase in PCE relative to the crude extract (Table 1).

Table 1.
The photovoltaic performance of DSSCs with different types of dyes and fractions. The detailed parameters are as follows: short circuit current (Jsc), open-circuit voltage (Voc), fill factor (FF), and energy conversion efficiency (η%). The plant names are Delonix regia. L1, L2, and L3 were separated from the preparative TLC plate. L4 was obtained from the residue of the acetone extraction and redissolved in methanol. Efficiency changes were calculated using raw D. regia as the reference. Each experiment was conducted in triplicate. The symbol ↓ indicates a decrease, while ↑ indicates an increase.
3.3. UV-Vis Absorption Spectra
The UV–visible spectra of fractions L1–L4 exhibited differences in both the 350 nm and 450 nm ranges (Figure 2). Fraction L1 showed a major absorption band at 453 nm, accompanied by two shoulders at 424 nm and 473 nm. Combined with its bright yellow color and low polarity, these spectral features are consistent with β-carotene [40,41]. Fraction L2 exhibited a single broad but weak absorption band spanning 355–510 nm, reflecting its complex composition, as indicated by multiple TLC spots and its relatively low solar cell 00efficiency (Table 1). In contrast, L3 displayed distinct peaks at 280 nm and 355 nm, consistent with the reported UV profiles of quercetin and kaempferol—flavonoids previously identified in D. regia and known for their acetone solubility [42]. Despite its light brown color, L3 adheres strongly to TiO2 surfaces, likely due to the presence of multiple hydroxyl groups in its structure. Fraction L4 exhibited absorption features similar to the crude D. regia extract, with two major bands at 355 nm and 450 nm.
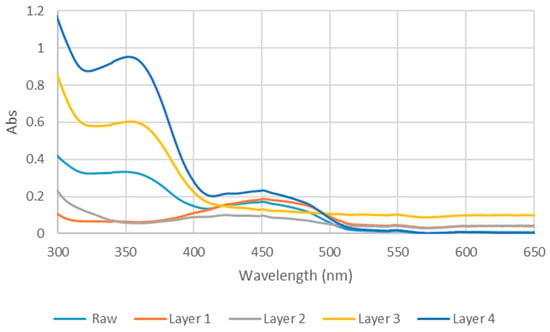
Figure 2.
UV-vis spectra of raw D. regia and the four layers in methanol.
3.4. 1H NMR Data of the Raw Extracts
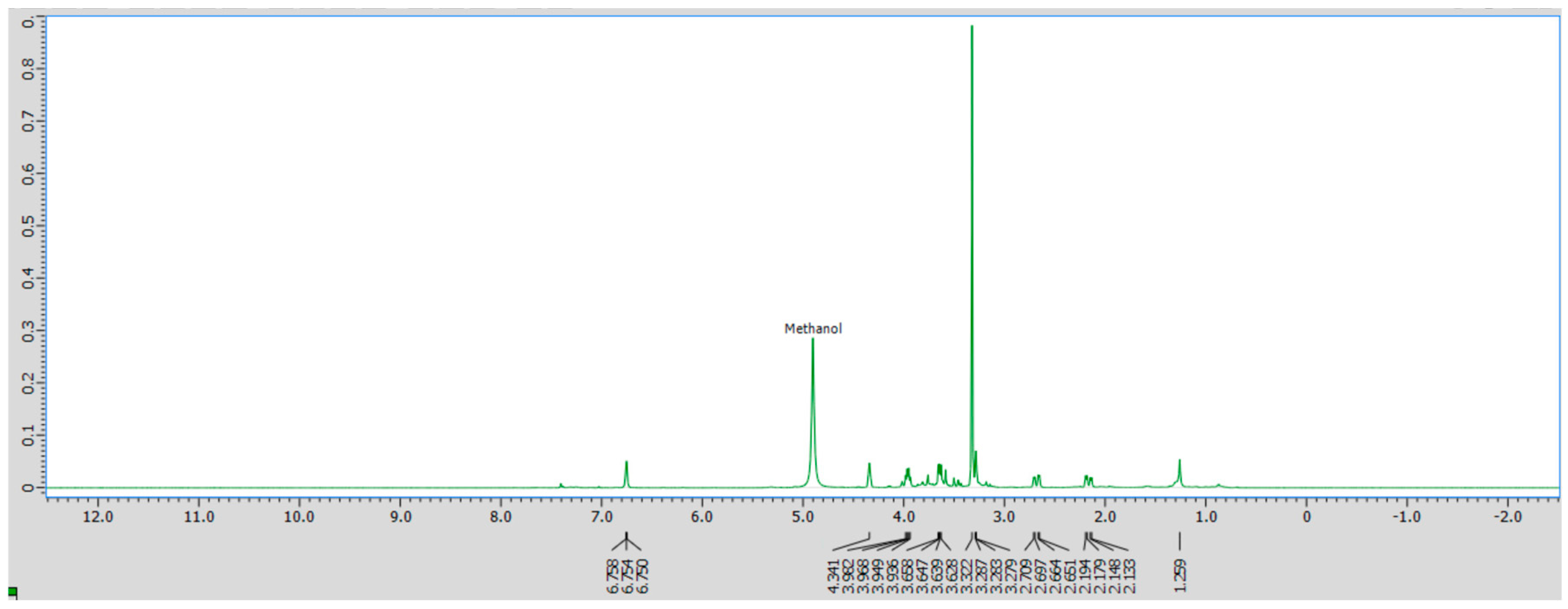
Crude D. regia extract and its individual fractions were further characterized by 1H NMR in d4-methanol to investigate the functional groups and general structures of the sample. Although precise compound identification is challenging due to the presence of multiple molecular species, 1H NMR provides insights into the general classes of functional groups present. The spectral assignments were guided by reference data for water-soluble plant metabolites in D2O reported by Sobolev et al. [43]. The 1H NMR spectrum of the crude extract in d4-methanol (Figure 3) reveals prominent signals in the 2.1–4.3 ppm region, characteristic of amino acids and carbohydrates [44]. Additionally, a distinct peak at approximately 6.8 ppm indicates the presence of phenolic compounds, consistent with the flavonoid-rich composition of this plant species [44].
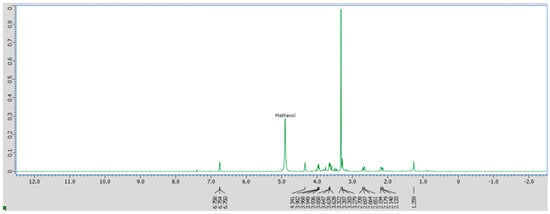
Figure 3.
1H NMR spectrum of crude D. regia in d4-methanol.
3.5. 1H NMR Analysis of Different Layers
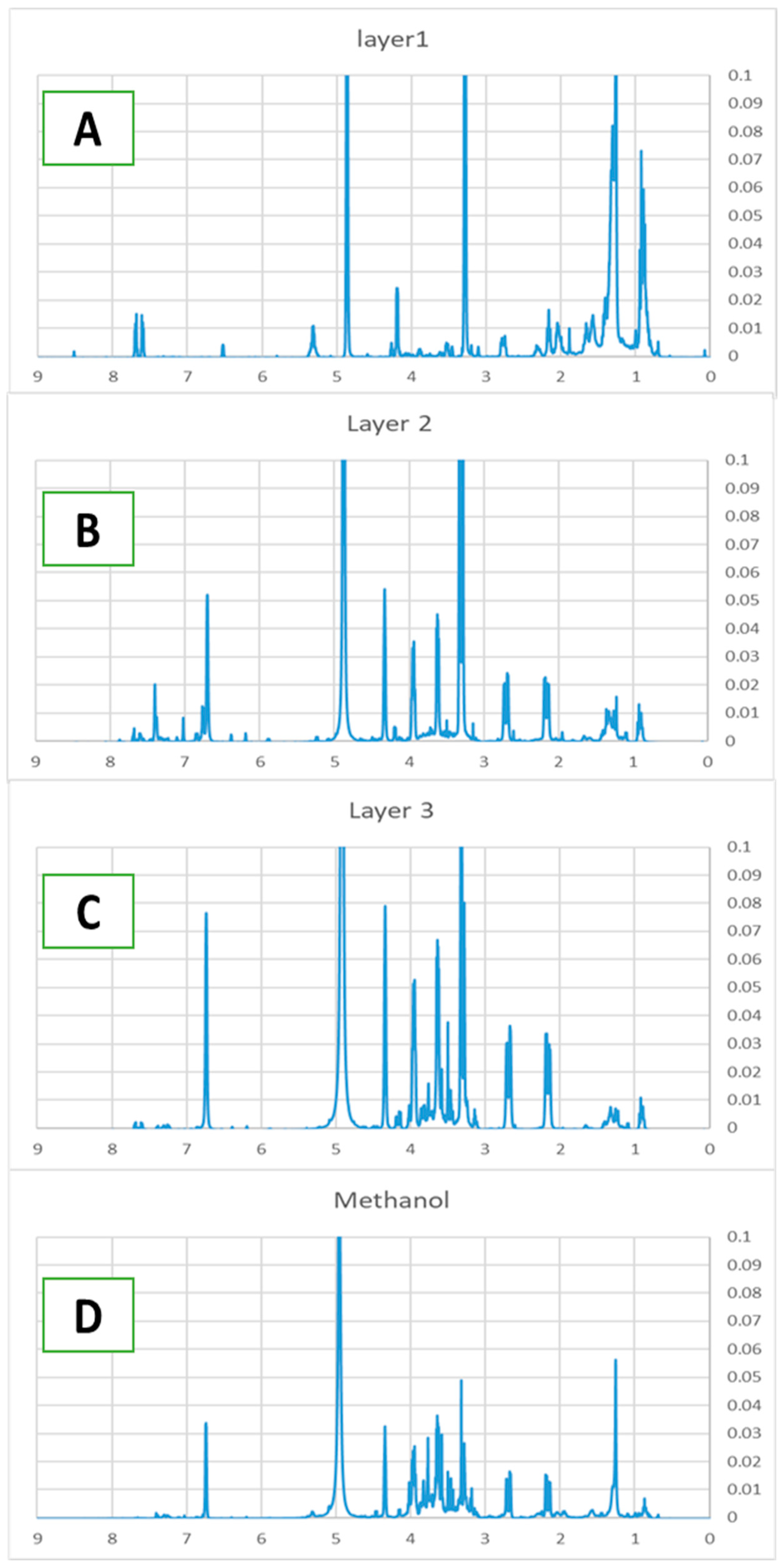
One major component in L1 was identified as β-carotenoid based on its UV-vis spectrum. In the 1H NMR spectrum (Figure 4A), many peaks in the 3–4 ppm region present in the crude extract are no longer observed, indicating the removal of carbohydrate related components. The 1H NMR spectrum of L2 (Figure 4B) exhibits prominent signals in the 2.1–3.9 ppm range, characteristic of carbohydrates that were absent in the top fraction. Additionally, a peak near 6.8 ppm suggests the presence of a phenolic ring, which may serve as an anchoring group for TiO2 binding. A minor peak observed at approximately 7.4 ppm further indicates the presence of an aromatic ring, consistent with flavonoid derivatives or other conjugated systems.
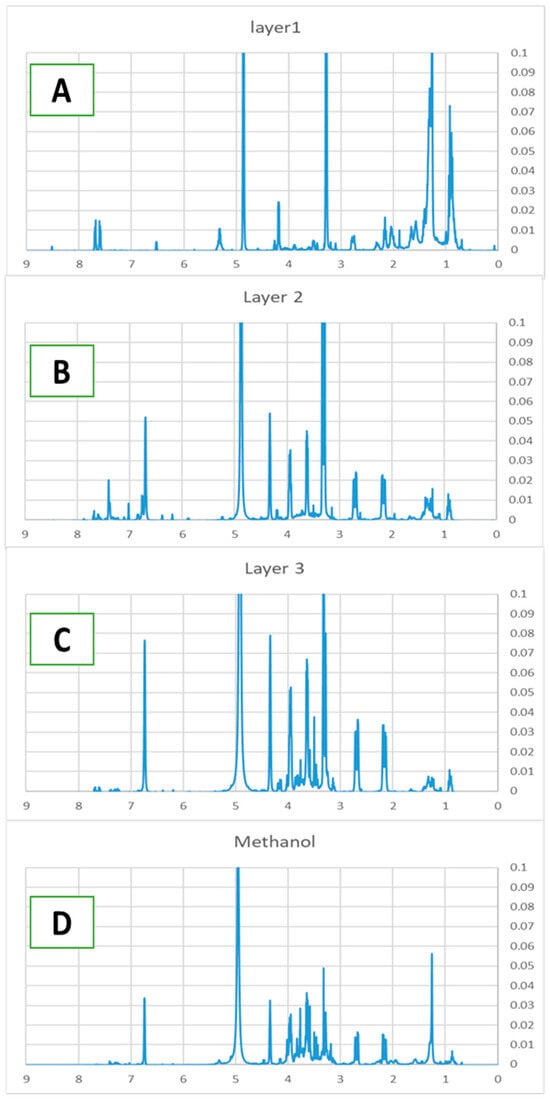
Figure 4.
1H NMR of L1 (A), L2 (B), L3 (C), methanol layer L4 (D) of D. regia.
The 1H NMR spectrum of L3 (Figure 4C) exhibits its highest proton density in the 2.1–4.3 ppm region, displaying significantly more peaks than the middle fraction. The aromatic region (6.5–8 ppm) shows well-resolved signals that correspond closely to reference spectra of quercetin and kaempferol, supporting the UV-vis identification of these flavonoid compounds.
The methanol fraction (L4) of D. regia exhibits a distinct 1H NMR profile that differs markedly from the other fractions (Figure 4D). Aromatic signals in the 6.5–8.5 ppm region indicate a higher concentration of phenolic compounds. Prominent peaks in the 3.0–4.5 ppm range are characteristic of carbohydrates and amino acid moieties, while a strong signal at approximately 1.2 ppm corresponds to methylene groups in aliphatic carbon chains.
4. Discussion
4.1. The Analysis of L3 Performance
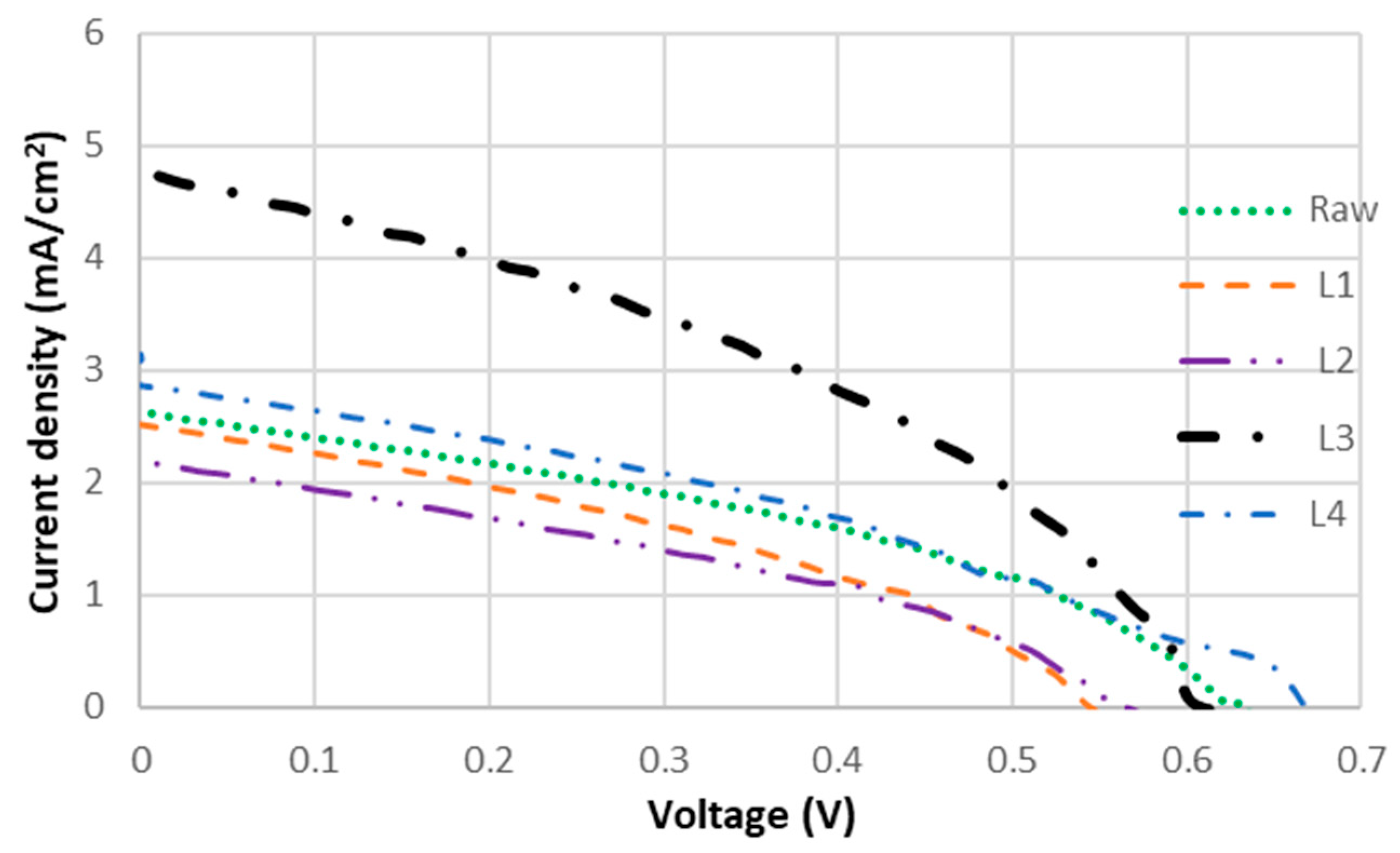
J-V curves of DSSCs sensitized with the crude extract and individual fractions are summarized in Figure 5. Fractionation led to notable variations in PCE, with L3 exhibiting the highest performance and a significant increase in PCE. The UV–vis spectra showed that L3 exhibited a higher absorption intensity across a comparable wavelength range relative to the crude extract, which is consistent with the observed increase in Jsc. The acetone-insoluble fraction L4 also showed improved performance compared to the crude extract. Both L3 and L4 exhibited absorption around 350 nm and achieved higher PCE than the crude dye, indicating that absorption in this region is closely associated with improved Jsc. Since the crude extract contains all the dyes present in L3 and L4, the higher Jsc observed in the fractions is unlikely to result solely from improved light-harvesting efficiency.
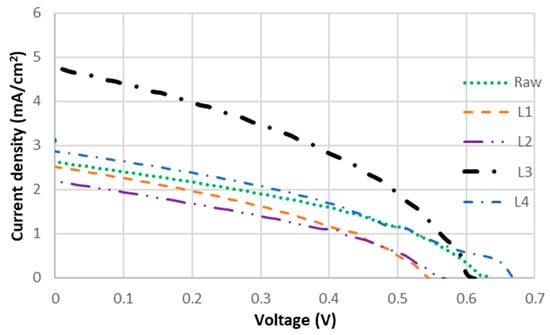
Figure 5.
J-V curves of DSSCs sensitized with Layer 1, 2, 3, 4 and raw D. regia extract.
As the most polar fraction isolated by TLC, L3 exhibits an NMR profile indicative of an abundance of functional groups suitable for surface anchoring, including multiple phenolic hydroxyls (as evidenced by the aromatic proton signals) and other polar moieties contributing to the complex 2.1–4.3 ppm region. These structural features likely enhance its binding affinity to TiO2 surfaces in photovoltaic applications. Moreover, A quantitative dye adsorption experiment by measuring the dye loss (adsorbed) from the solution before and after 1 h of adsorption was conducted (Table S1). The calculated adsorption percentages were 26.6% for L1, 40.8% for L2, 74.2% for L3, and 33.2% for L4, which indicated that L3 exhibits substantially higher dye adsorption than the other fractions. The results supported that the improved performance of L3 is potentially attributed to enhanced dye adsorption and dye-TiO2 interactions.
Improved Jsc has been reported to correlate with high adsorption affinity to TiO2 via multiple anchoring groups, which facilitate efficient electron injection and extend the electron lifetime (τn), thereby reducing charge recombination [45]. In the absence of such anchoring groups, dyes cannot bind effectively to TiO2 on the photoelectrode, preventing the initiation of the electrical circuit [46]. The superior performance of L3 is likely due to the higher adsorption affinity of flavonoids to TiO2. The flavanol-rich fraction L3 demonstrated enhanced light-harvesting capability, as evidenced by its significantly higher short-circuit current density (Jsc = 4.64 mA/cm2) compared to the crude dye extract and other isolated fractions. This increased photocurrent generation directly contributes to L3’s improved PCE (1.85-fold higher than the crude extract), indicating that the flavanol components (quercetin and kaempferol) not only provide effective TiO2 binding through multiple anchoring groups but also maintain favorable molecular orientation for efficient light absorption and charge injection.
In addition, the stability of the L3-based solar cells was evaluated after 1 h and 2 h of continuous solar illumination, as well as after 24 h, 48 h, and 72 h of storage under room light (Table S2). Our results indicated that the PCE remained stable after 1 and 2 h of solar illumination, ranging from 1.25 to 1.29% (Table S2). However, significant electrolyte evaporation was observed after 2 h, and the PCE was measured after refilling the electrolyte. Continuous solar illumination may damage the solar cell after 2 h due to electrolyte evaporation, which is the main reason that the 24 h, 48 h, and 72 h data were collected under room light. The stability under room light also showed stable efficiency (1.25–1.30%) up to 72 h. Therefore, the L3 dye demonstrated good stability under 2 h of continuous illumination and 72 h of room light storage.
4.2. The Analysis of L4 Performance
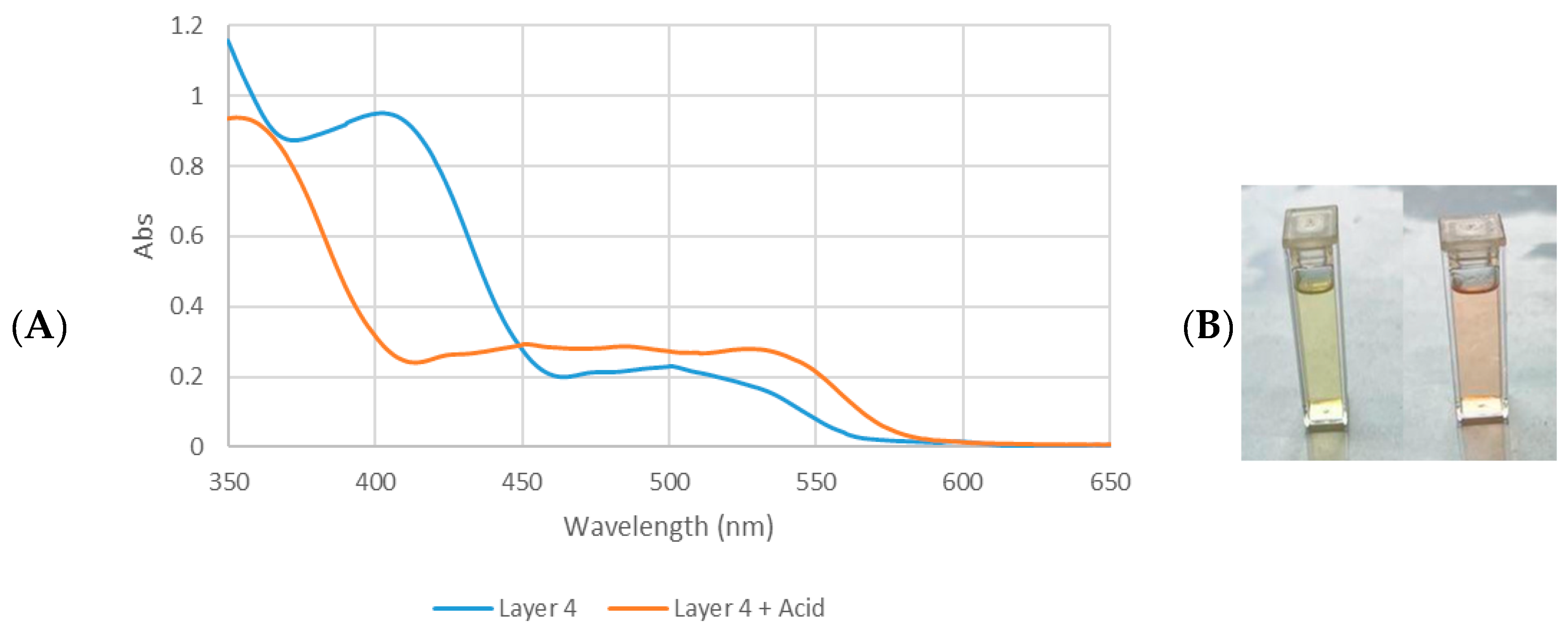
One of the major pigments in D. regia flowers has been characterized as anthocyanins [47]. Their presence was further confirmed using a pH-dependent colorimetric assay. Anthocyanins undergo a reversible bathochromic shift under acidic conditions (red at pH 1–2) and producing an additional peak near 600 nm [48]. To test this behavior, all fractions were acidified with 0.1 M HCl (3 drops; pH ≈ 1–2, estimated via pH strips due to methanol interference). Only L4 and the crud extract turned from yellow to red (Figure 6B), whereas the TLC-derived fractions (L1–L3) remained unchanged. These observations are consistent with the UV-vis spectra of L4 before and after acid treatment (Figure 6A), confirming the presence of anthocyanins in this fraction.
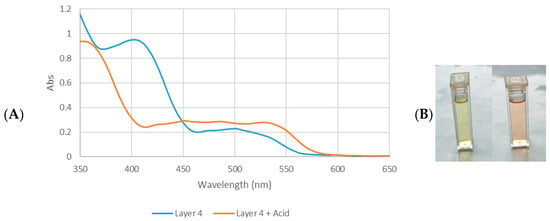
Figure 6.
(A) UV-vis absorption spectra of D. regia Layer 4 and Layer 4 + HCl tuned to a pH of between 1 and 2. (B) Color of Layer 4 (left) and color of L4 + HCl (right).
Although anthocyanins in L4 are limited by pH-dependent stability [49], its unique spectral profile indicates that this fraction contains a complex mixture of compounds, including glycosylated flavonoids (as evidenced by combined aromatic and carbohydrate signals), anthocyanin derivatives (confirmed by UV-vis) and hydrophobic domains (aliphatic proton resonance). The coexistence of polar (phenolic/carbohydrate) and nonpolar (aliphatic) features likely contributes to both solvent solubility and surface-binding property. In contrast, the favorable combination of electronic and structural properties in L3 accounts for its outstanding photovoltaic performance relative to the anthocyanin-containing L4 fraction. These findings highlight that selective fractionation of plant pigments can significantly enhance DSSC performance, with flavonol-rich fractions offering particular promise due to their efficient light harvesting, appropriate molecular complexity, and strong surface binding properties.
4.3. The Analysis Low-Performance Layers L1 and L2
Fraction L1 exhibits very few peaks in the 3–4 ppm region present in the crude sample, suggesting a substantial absence of potential anchoring groups for TiO2 binding. Instead, the spectrum exhibits intense signals in the 0.88–1.3 ppm range, indicative of abundant sp3-hybridized alkyl chains, along with resonances corresponding to highly conjugated alkene protons (5–6 ppm). These features are consistent with the expected structure of β-carotene, which contains a long polyene chain and conjugated system but lacks carboxylates or phenolic moieties. The spectral profile matches previously reported 1H NMR data for β-carotene, corroborating its identification in L1 [50].
According to PTLC analysis, L2 is composed of multiple layers, indicating a highly complex mixture. The abundance of amino acids, carbohydrates, and phenolic hydroxyl groups implies favorable anchoring moieties for binding to TiO2 surfaces, which is critical for dye-sensitized solar cell (DSSC) applications. However, the complexity of such natural extracts may also promote excessive dye aggregation, potentially reducing photovoltaic performance [51]. Molecular polarity has been reported to play a critical role in aggregation-mediated quenching, as low-polarity species tend to aggregate easily through intermolecular interactions [52]. The fractionation process could reduce the dye aggregation on the TiO2 surface by separating the low-polarity components. As a result, L3 contains more high-polarity compounds, which likely mitigate aggregation-induced quenching compared to the crude extract and the L1–L2 fractions.
5. Conclusions
This study demonstrates that fractionation of D. regia pigments on polarity can significantly enhance photovoltaic performance in DSSCs. A simple separation and purification process using PTLC can alter the composition of natural compounds, thereby affecting both light-harvesting efficiency and TiO2-binding affinity. In our study, the flavanol-rich fraction (L3) exhibited superior light-harvesting properties, achieving the highest Jsc (4.64 mA/cm2) and an 85.25% increase (1.85-fold) in power conversion efficiency over the crude dye. The NMR and UV-vis data indicate that this enhancement arises from improved TiO2-binding affinity, more efficient electron injection, and reduced charge recombination. The anthocyanin-containing methanol fraction (L4) also showed 1.2-fold increased performance (19.67% higher efficiency than crude dye), although its applications are limited by pH-dependent stability and dye aggregation. These findings highlight the critical role of selective pigment isolation and molecular structure optimization for natural dye-sensitizers studies in solar energy applications. Future studies should explore the synergistic combination of natural compounds within these dyes to further enhance their photovoltaic performance for practical applications, which can potentially improve efficiency and lower costs.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/colorants5010008/s1, Table S1: Dye adsorption percentage (A%) of different layers on 100 mg of TiO2 after immersion in a 3 mg/mL dye solution for 1 h. The absorption rate was calculated through the equation [(Iinitial–I1h)/Iinitial] x 100% using λmax; Table S2: Photoelectrochemical parameters of L3-based DSSCs over a 3-day stability test. * The 1 and 2 h measurements were carried out under continuous solar illumination, and the 24 h and after were carried out after storage under room light.
Author Contributions
L.J.: Conceptualization (lead); formal analysis (equal); funding acquisition (lead); investigation (equal); methodology (lead); project administration (lead); writing—original draft (lead); writing—review and editing (equal). K.H.: Data curation (equal); visualization (equal). B.W.: Data curation (equal); investigation (equal); validation (equal); visualization (equal); writing—review and editing (equal). All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Acknowledgments
This project was supported by the New College of Florida Student Research and Travel Grant program. LJ would like to thank the New College of Florida for the financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Aghazada, S.; Nazeeruddin, M.K. Ruthenium complexes as sensitizers in dye-sensitized solar cells. Inorganics 2018, 6, 52. [Google Scholar] [CrossRef]
- Dhorkule, M.; Lamrood, P.; Ralegankar, S.; Patole, S.P.; Wagh, S.S.; Pathan, H.M. Unveiling the efficiency of dye-sensitized solar cells: A journey from synthetic to natural dyes. ES Food Agrofor. 2024, 16, 1086. [Google Scholar] [CrossRef]
- Richhariya, G.; Meikap, B.C.; Kumar, A. Review on fabrication methodologies and its impacts on performance of dye-sensitized solar cells. Environ. Sci. Pollut. Res. 2022, 29, 15233–15251. [Google Scholar] [CrossRef] [PubMed]
- Leyrer, J.; Hunter, R.; Rubilar, M.; Pavez, B.; Morales, E.; Torres, S. Development of dye-sensitized solar cells based on naturally extracted dye from the maqui berry (Aristotelia chilensis). Opt. Mater. 2016, 60, 411–417. [Google Scholar] [CrossRef]
- Ghann, W.; Kang, H.; Sheikh, T.; Yadav, S.; Chavez-Gil, T.; Nesbitt, F.; Uddin, J. Fabrication, optimization and characterization of natural dye sensitized solar cell. Sci. Rep. 2017, 7, 41470. [Google Scholar] [CrossRef]
- Song, W.; Gong, Y.; Tian, J.; Cao, G.; Zhao, H.; Sun, C. Novel photoanode for dye-sensitized solar cells with enhanced light-harvesting and electron-collection efficiency. ACS Appl. Mater. Interfaces 2016, 8, 13418–13425. [Google Scholar] [CrossRef]
- Wei, L.; Wang, P.; Yang, Y.; Fan, R.; Yang, Y.; Qiu, Y. Construction of efficient photoanodes for dye sensitized solar cells: TiO2 films with a gradient content of graphene. Sustain. Energy Fuels 2017, 1, 1112–1122. [Google Scholar] [CrossRef]
- Ren, Y.; Zhang, D.; Suo, J.; Cao, Y.; Eickemeyer, F.T.; Vlachopoulos, N.; Zakeeruddin, S.M.; Hagfeldt, A.; Grätzel, M. Hydroxamic acid pre-adsorption raises the efficiency of cosensitized solar cells. Nature 2023, 613, 60–65. [Google Scholar] [CrossRef]
- Zhang, D.; Stojanovic, M.; Ren, Y.; Cao, Y.; Eickemeyer, F.T.; Socie, E.; Vlachopoulos, N.; Moser, J.-E.; Zakeeruddin, S.M.; Hagfeldt, A. A molecular photosensitizer achieves a Voc of 1.24 V enabling highly efficient and stable dye-sensitized solar cells with copper (II/I)-based electrolyte. Nat. Commun. 2021, 12, 1777. [Google Scholar] [CrossRef]
- Maurya, I.C.; Singh, S.; Srivastava, P.; Maiti, B.; Bahadur, L. Natural dye extract from Cassia fistula and its application in dye-sensitized solar cell: Experimental and density functional theory studies. Opt. Mater. 2019, 90, 273–280. [Google Scholar] [CrossRef]
- Deshpande, R.; Wang, B.; Dai, L.; Jiang, L.; Hartley, C.S.; Zou, S.; Wang, H.; Kerr, L. Opp-Dibenzoporphyrins as a Light-Harvester for Dye-Sensitized Solar Cells. Chem.–Asian J. 2012, 7, 2662–2669. [Google Scholar] [CrossRef]
- Hemavathi, B.; Jayadev, V.; Ramamurthy, P.C.; Pai, R.K.; KN, N.U.; Ahipa, T.; Soman, S.; Balakrishna, R.G. Variation of the donor and acceptor in D–A–π–A based cyanopyridine dyes and its effect on dye sensitized solar cells. New J. Chem. 2019, 43, 15673–15680. [Google Scholar] [CrossRef]
- Hemavathi, B.; Jayadev, V.; Pradhan, S.C.; Gokul, G.; Jagadish, K.; Chandrashekara, G.; Ramamurthy, P.C.; Pai, R.K.; Unni, K.N.; Ahipa, T. Aggregation induced light harvesting of molecularly engineered DA-π-A carbazole dyes for dye-sensitized solar cells. Sol. Energy 2018, 174, 1085–1096. [Google Scholar] [CrossRef]
- Baby, R.; Nixon, P.D.; Kumar, N.M.; Subathra, M.; Ananthi, N. A comprehensive review of dye-sensitized solar cell optimal fabrication conditions, natural dye selection, and application-based future perspectives. Environ. Sci. Pollut. Res. 2021, 29, 371–404. [Google Scholar] [CrossRef]
- Triyanto, A.; Ali, N.A.; Salleh, H.; Setiawan, J.; Yatim, N.I. Development of natural dye photosensitizers for dye-sensitized solar cells: A review. Environ. Sci. Pollut. Res. 2024, 31, 31679–31690. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, U.; Prajapat, K.; Dhonde, M.; Sahu, K.; Shirage, P.M. Natural dyes for dye-sensitized solar cells (DSSCs): An overview of extraction, characterization and performance. Nano-Struct. Nano-Objects 2024, 37, 101111. [Google Scholar] [CrossRef]
- Malhotra, S.S.; Ahmed, M.; Gupta, M.K.; Ansari, A. Metal-free and natural dye sensitized solar cells: Recent advancement and future perspectives. Sustain. Energy Fuels 2024, 8, 4127–4163. [Google Scholar] [CrossRef]
- Wang, X.-F.; Zhan, C.-H.; Maoka, T.; Wada, Y.; Koyama, Y. Fabrication of dye-sensitized solar cells using chlorophylls c1 and c2 and their oxidized forms c1’ and c2’ from Undaria pinnatifida (Wakame). Chem. Phys. Lett. 2007, 447, 79–85. [Google Scholar] [CrossRef]
- Iman, R.N.; Harrabi, K.; Younas, M.; Mekki, A. Fabrication of efficient natural dye-sensitized Solar Cells using Mediterranean olive leaves as natural dye sensitizer. J. Photochem. Photobiol. A Chem. 2024, 450, 115477. [Google Scholar] [CrossRef]
- Hussain, M.; Jalali, T.; Maftoon-Azad, L.; Osfouri, S. Performance evaluation of natural dye-sensitized solar cells: A comparison of density functional theory and experimental data on chlorophyll, anthocyanin, and cocktail dyes as sensitizers. ACS Appl. Electron. Mater. 2024, 6, 1693–1709. [Google Scholar] [CrossRef]
- Minicante, S.A.; Ambrosi, E.; Back, M.; Barichello, J.; Cattaruzza, E.; Gonella, F.; Scantamburlo, E.; Trave, E. Development of an eco-protocol for seaweed chlorophylls extraction and possible applications in dye sensitized solar cells. J. Phys. D Appl. Phys. 2016, 49, 295601. [Google Scholar] [CrossRef]
- Neale, N.R.; Kopidakis, N.; Van De Lagemaat, J.; Grätzel, M.; Frank, A.J. Effect of a coadsorbent on the performance of dye-sensitized TiO2 solar cells: Shielding versus band-edge movement. J. Phys. Chem. B 2005, 109, 23183–23189. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barichello, J.; Mariani, P.; Vesce, L.; Spadaro, D.; Citro, I.; Matteocci, F.; Bartolotta, A.; Di Carlo, A.; Calogero, G. Bifacial dye-sensitized solar cells for indoor and outdoor renewable energy-based application. J. Mater. Chem. C 2024, 12, 2317–2349. [Google Scholar] [CrossRef]
- Thavasi, V.; Renugopalakrishnan, V.; Jose, R.; Ramakrishna, S. Controlled electron injection and transport at materials interfaces in dye sensitized solar cells. Mater. Sci. Eng. R Rep. 2009, 63, 81–99. [Google Scholar] [CrossRef]
- Khataee, A.; Mansoori, G.A. Nanostructured Titanium Dioxide Materials: Properties, Preparation and Applications; World Scientific: Hackensack, NJ, USA, 2011. [Google Scholar]
- El-Sayed, A.M.; Ezzat, S.M.; Salama, M.M.; Sleem, A.A. Hepatoprotective and cytotoxic activities of Delonix regia flower extracts. Pharmacogn. J. 2011, 3, 49–56. [Google Scholar] [CrossRef]
- Wang, L.-S.; Lee, C.-T.; Su, W.-L.; Huang, S.-C.; Wang, S.-C. Delonix regia leaf extract (DRLE): A potential therapeutic agent for cardioprotection. PLoS ONE 2016, 11, e0167768. [Google Scholar] [CrossRef]
- Modi, A.; Mishra, V.; Bhatt, A.; Jain, A.; Mansoori, M.H.; Gurnany, E.; Kumar, V. Delonix regia: Historic perspectives and modern phytochemical and pharmacological researches. Chin. J. Nat. Med. 2016, 14, 31–39. [Google Scholar]
- Khongkaew, P.; Wattanaarsakit, P.; Papadopoulos, K.I.; Chaemsawang, W. Antioxidant effects and in vitro cytotoxicity on human cancer cell lines of flavonoid-rich flamboyant (Delonix regia (Bojer) Raf.) flower extract. Curr. Pharm. Biotechnol. 2021, 22, 1821–1831. [Google Scholar] [CrossRef]
- Harshitha, D.; Kumar, A.; Mahesh, H.; Renuka, C. Anthocyanins of Delonix regia floral petals: A novel approach on fluorescence enhancement, forster resonance energy transfer mechanism and photostability studies for optoelectronic applications. J. Fluoresc. 2024, 35, 3195–3226. [Google Scholar] [CrossRef] [PubMed]
- Azab, S.S.; Abdel-Daim, M.; Eldahshan, O.A. Phytochemical, cytotoxic, hepatoprotective and antioxidant properties of Delonix regia leaves extract. Med. Chem. Res. 2013, 22, 4269–4277. [Google Scholar] [CrossRef]
- Vijendran, V.; Ramalingam, S.; Balasundaram, J.; Kumar, A.; Samdani, M.S.; Priya, R.; Saravanakumar, M. Exciton generation using sensitizers derived from floral and foliar sources for dye sensitized solar cells. Opt. Quantum Electron. 2025, 57, 263. [Google Scholar] [CrossRef]
- Sanda, M.D.; Badu, M.; Awudza, J.; Boadi, N. Development of TiO2-based dye-sensitized solar cells using natural dyes extracted from some plant-based materials. Chem. Int. 2021, 7, 9–20. [Google Scholar]
- Srivastava, A.; Chauhan, B.S.; Yadav, S.C.; Tiwari, M.K.; Satrughna, J.A.K.; Kanwade, A.; Bala, K.; Shirage, P.M. Performance of dye-sensitized solar cells by utilizing Codiaeum Variegatum Leaf and Delonix regia Flower as natural sensitizers. Chem. Phys. Lett. 2022, 807, 140087. [Google Scholar] [CrossRef]
- Sullivan, H.; Wang, B.; Jiang, L. Investigation of tropical plant-based natural dyes combination and adsorption optimization for natural dye-sensitized solar cell. Environ. Prog. Sustain. Energy 2022, 41, e13809. [Google Scholar] [CrossRef]
- Al-horaibi, S.A.; Al-Odayni, A.-B.; Alezzy, A.; ALSaeedy, M.; Al-Adhreai, A.; Saeed, W.S.; Hasan, A. Novel Squaraine dyes for high-performance in dye-sensitized solar cells: Photophysical properties and adsorption behavior on TiO2 with different anchoring groups. J. Mol. Struct. 2023, 1290, 135943. [Google Scholar] [CrossRef]
- Al-Alwani, M.A.; Mohamad, A.B.; Kadhum, A.A.H.; Ludin, N.A. Effect of solvents on the extraction of natural pigments and adsorption onto TiO2 for dye-sensitized solar cell applications. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 138, 130–137. [Google Scholar] [CrossRef]
- Nazeeruddin, M.K.; Kay, A.; Rodicio, I.; Humphry-Baker, R.; Müller, E.; Liska, P.; Vlachopoulos, N.; Grätzel, M. Conversion of light to electricity by cis-X2bis (2, 2′-bipyridyl-4, 4′-dicarboxylate) ruthenium (II) charge-transfer sensitizers (X = Cl-, Br-, I-, CN-, and SCN-) on nanocrystalline titanium dioxide electrodes. J. Am. Chem. Soc. 1993, 115, 6382–6390. [Google Scholar] [CrossRef]
- Krawczak, E.; Zdyb, A. The influence of the dye adsorption time on the DSSC performance. In E3S Web of Conferences; EDP Sciences: Les Ulis, France, 2019; Volume 100, p. 00040. [Google Scholar]
- Cvetković, D.; Marković, D. Stability of carotenoids toward UV-irradiation in hexane solution. J. Serbian Chem. Soc. 2008, 73, 15–27. [Google Scholar] [CrossRef]
- Meinhardt-Wollweber, M.; Suhr, C.; Kniggendorf, A.-K.; Roth, B. Absorption and resonance Raman characteristics of β-carotene in water-ethanol mixtures, emulsion and hydrogel. AIP Adv. 2018, 8, 055320. [Google Scholar] [CrossRef]
- Adjé, F.A.; Lozano, Y.F.; Le Gernevé, C.; Lozano, P.R.; Meudec, E.; Adima, A.A.; Gaydou, E.M. Phenolic acid and flavonol water extracts of Delonix regia red flowers. Ind. Crops Prod. 2012, 37, 303–310. [Google Scholar] [CrossRef]
- Sobolev, A.P.; Brosio, E.; Gianferri, R.; Segre, A.L. Metabolic profile of lettuce leaves by high-field NMR spectra. Magn. Reson. Chem. 2005, 43, 625–638. [Google Scholar] [CrossRef]
- Loum, J.; Byamukama, R.; Wanyama, P. UV–Vis Spectrometry for Quantitative Study of Tannin and Flavonoid Rich Dyes from Plant Sources. Chem. Afr. 2020, 3, 449–455. [Google Scholar] [CrossRef]
- Kumara, N.; Petrović, M.; Peiris, D.; Marie, Y.A.; Vijila, C.; Petra, M.I.; Chandrakanthi, R.; Lim, C.M.; Hobley, J.; Ekanayake, P. Efficiency enhancement of Ixora floral dye sensitized solar cell by diminishing the pigments interactions. Sol. Energy 2015, 117, 36–45. [Google Scholar] [CrossRef]
- Zhang, L.; Cole, J.M. Anchoring groups for dye-sensitized solar cells. ACS Appl. Mater. Interfaces 2015, 7, 3427–3455. [Google Scholar] [CrossRef]
- Adjé, F.; Lozano, Y.F.; Lozano, P.; Adima, A.; Chemat, F.; Gaydou, E.M. Optimization of anthocyanin, flavonol and phenolic acid extractions from Delonix regia tree flowers using ultrasound-assisted water extraction. Ind. Crops Prod. 2010, 32, 439–444. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Acylated anthocyanins from edible sources and their applications in food systems. Biochem. Eng. J. 2003, 14, 217–225. [Google Scholar] [CrossRef]
- Mejica, G.F.C.; Unpaprom, Y.; Balakrishnan, D.; Dussadee, N.; Buochareon, S.; Ramaraj, R. Anthocyanin pigment-based dye-sensitized solar cells with improved pH-dependent photovoltaic properties. Sustain. Energy Technol. Assess. 2022, 51, 101971. [Google Scholar] [CrossRef]
- Gurkok, S. A novel carotenoid from Metabacillus idriensis LipT27: Production, extraction, partial characterization, biological activities and use in textile dyeing. Arch. Microbiol. 2022, 204, 296. [Google Scholar] [CrossRef]
- Xu, F.; Testoff, T.T.; Wang, L.; Zhou, X. Cause, regulation and utilization of dye aggregation in dye-sensitized solar cells. Molecules 2020, 25, 4478. [Google Scholar] [CrossRef]
- Zhang, L.; Cole, J.M. Dye aggregation in dye-sensitized solar cells. J. Mater. Chem. A 2017, 5, 19541–19559. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.