Abstract
Tea is one of the most widely consumed beverages globally and a significant dietary source of fluoride. This systematic review aimed to identify and evaluate the factors influencing fluoride concentration in tea infusions. A comprehensive literature search was conducted in March 2025 across PubMed, Scopus, and Web of Science databases, following PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and the PICO framework. Eligible studies investigated fluoride release in tea infusions, published in English from the year 2000 onward. Thirty articles met the inclusion criteria, and the risk of bias in the articles was assessed using the Joanna Briggs Institute (JBI) quality checklist. Fluoride concentration in tea infusions varied widely across studies, ranging from 0.008 to over 8 mg/L. Key factors influencing fluoride release included tea type (with black and green teas showing the highest values), leaf form (powdered and bagged teas released more fluoride than loose leaves), brewing time and temperature, water composition, and the presence of additives such as spices. A longer brewing time and higher temperature consistently increased fluoride extraction. Lower pH or water hardness also significantly affected fluoride availability. Regional origin of tea and production methods were additional sources of variation. Fluoride release in tea is influenced by a complex interplay of botanical, environmental, and preparation-related factors. These findings are clinically relevant, particularly for populations at risk of fluoride overexposure. Further standardized research is needed to inform safe consumption guidelines and public health recommendations.
1. Introduction
Fluoride is an element commonly found in the natural environment and is classified as a microelement. It enters the human body through the digestive and respiratory tracts and, in small amounts, through the skin. The main sources are water, food, and everyday hygiene products such as toothpastes and mouthwashes [1,2,3,4]. The recommended daily intake of fluoride varies according to age and gender. It is 0.7 mg for children and 3–4 mg for adults. These values are considered to be the adequate daily intake of fluoride. The tolerable upper intake level (UL) for adults, beyond which the risk of fluorosis increases, is 10 mg per day [2,5]. Fluoride is an element that performs important systemic and local functions in the human body [6]. It is a component of enzymes involved in the metabolism of proteins, carbohydrates, and lipids [3]. Ninety-nine percent of the fluoride absorbed by the human body is found in bones and teeth, where hydroxyapatites have been converted by the presence of fluoride into more valuable fluoroapatites and/or hydroxyfluoroapatites [1,2]. In addition, fluoride plays a very important role in dentistry as a component of an anti-caries substance [7,8]. In addition to being added to drinking water and milk, fluoride is found in everyday hygiene products such as toothpastes and mouthwashes. It is also used professionally in the form of varnishes, foams, and gels [9,10]. Its cariostatic action is based on the prevention of enamel demineralization, its remineralization (stopping caries at an early stage), and its antibacterial action. These properties have contributed significantly to the worldwide reduction in dental caries [8,9,11]. It is also worth mentioning the risks associated with overdosing on fluoride. Excess fluoride leads to enamel fluorosis, which is a disorder of the quality and/or quantity of enamel. Studies show that exceeding the daily intake of fluoride has a systemic toxic effect and, in extremely high doses, can lead to death [12,13].
Tea infusion is a beverage commonly consumed by humans. It is produced worldwide from the leaves of the Camellia sinensis tree. Apart from water and some foods such as fish and seafood, tea is the main source of fluoride in the human diet [3,14]. The amount of tea consumed varies depending on the region of the world and cultural habits. A 250 mL infusion can provide up to 1.5 mg of fluoride, which is a significant part of the recommended daily intake of this element [15]. Therefore, tea should be considered as a significant source of fluoride in the human diet, and it should be borne in mind that the accumulation of fluoride from all sources creates a risk of dental fluorosis, skeletal fluorosis, and chronic poisoning with this element [16]. Studies show that there is considerable variation in the fluoride content of teas. This is affected by many variables. The amount of fluoride depends on the type of tea. For example, black teas release the most fluoride into the infusion, followed by white and green teas [17,18]. Herbal products release the least fluoride [14,19]. The same type of tea may also have different fluoride levels depending on the region of origin [20]. The age of the plant has a major effect on the fluoride content of tea trees. Older specimens have been shown to have higher levels of fluoride due to the accumulation of fluoride over time. The pH of the soil in which the plant is grown is also important, as the lower the pH of the environment, the greater the deposition of fluoride in the stems and leaves [14,21]. In some regions of the world, frequent consumption of tea because of cultural behavior creates a situation in which a given population is exposed to exceeding the daily limit of fluoride intake [15,22]. This can result in dental and/or skeletal fluorosis and chronic poisoning leading to cardiac, neuro, and nephrotoxicity [13,15]. Finally, it should be noted that a person’s fluoride intake from tea infusions is influenced by the concentration of fluoride in the water and by factors related to the preparation of the infusion, which affect the amount of fluoride released from the leaves [13,22,23] (see Figure 1).
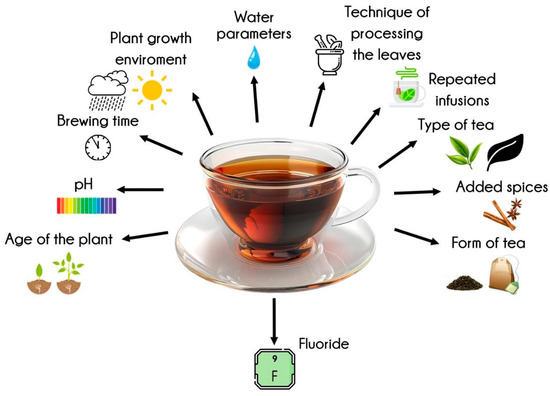
Figure 1.
Factors influencing fluoride levels in tea infusions (created with Freepik.com).
The release of fluoride from tea is influenced by a multitude of factors. These factors are diverse and related to the plant itself, including its growing environment, brewing circumstances and parameters, the form of the tea, and the technique of processing the leaves and the addition of various spices. The age of the plant itself has been demonstrated to affect the fluoride concentration. Consequently, as the plant matures, the fluoride concentration increases. This assertion is corroborated by studies undertaken by Vakdevi et al., which demonstrated a higher fluoride content in older tea leaves [21]. A closer examination of the plant itself, the soil type, or more precisely, the pH level, is clear in our considerations. It has been demonstrated that elevated levels of fluoride in the foliage are associated with acidic soil [23,24]. Consequently, the level of fluoride will be elevated. The bioavailability of ions, such as fluorine and aluminum, in soil is subject to variation and correlation with its acidity. At a pH of 5.5, aluminum ions in conjunction with fluorine ions form a stable Al-F complex, which is efficiently absorbed by tea plants [25]. The region of cultivation from which tea comes also affects the fluoride concentration [26]. The form of tea has been demonstrated to influence the quantity of fluoride released. Specifically, tea bags have been found to release a higher concentration of fluoride compared to loose-leaf tea varieties during the infusion process. This phenomenon can be attributed to the utilization of older leaves in the production of tea bags [27]. A similar trend was observed in the study by Jin et al., where it was found that the use of mature leaves resulted in a higher fluoride release during the pressing process [27]. In this instance, too, older leaves are utilized, resulting in these teas exhibiting a higher fluoride concentration [24,28].
The fluoride concentration is observed to vary depending on the type of tea. It is black tea in the infusion that—as a rule—contains the highest concentration of this element [19]. In descending order, green tea, Earl Grey, pu-erh, and white tea follow. Fluoride is present in trace amounts in herbal teas. Brick tea, as previously referenced, has been observed to contain elevated levels of fluoride, while white tea, in comparison to black, green, and oolong varieties, has been found to exhibit reduced levels [29]. Numerous studies have indicated a direct proportionality between brewing time and the fluoride concentration in the infusion. The concentration of fluoride in the infusion increases in proportion to the duration of the infusion process [30]. Furthermore, the repeated infusion of the same quantity of tea has been shown to increase the fluoride content [31]. Conversely, the infusion resulting from a single, continuous infusion contains a lower amount of fluoride. It has been demonstrated that multiple infusions of the same quantity of tea result in an increase in the amount of fluoride released. Of particular interest is the observation that the initial infusion of a given quantity of tea releases the greatest amount of fluoride. The extraction of fluoride ions into the infusion is influenced by the temperature of the water used for preparation, with boiling water being the standard [32]. The total fluoride content will also be influenced by the quality of the water itself. Endemic water may contain higher or lower concentrations of fluoride ions. Furthermore, it is imperative to acknowledge the significance of water treatment within the water supply network. Groundwater intakes are distinguished by their varying fluorine content [33]. It has been observed that the addition of spices, such as ginger or cloves, to the infusion can modify the amount of fluorine extracted [34].
The objective of this systematic literature review is to identify the factors influencing fluoride release from tea during infusion preparation. A comprehensive analysis of the extant literature pertaining to fluoride content measurements in infusions from diverse tea varieties, the water utilized, the spices employed, and the physical parameters of the extraction process was conducted. The findings revealed a paucity of systematic reviews addressing this subject in the literature. This constitutes a compelling rationale for the consolidation of extant scientific evidence. The present study will undoubtedly provide dietitians, medical practitioners, and, most crucially, consumers with a comprehensive framework for selecting tea as a substantial source of fluoride supplementation. A significant element of the systematic review is to ascertain the algorithms for preparing tea infusions so that the fluoride concentration is optimal. This comprehensive approach will facilitate a more profound comprehension of the entire process of fluoride extraction from tea during the preparation of the beverage.
2. Materials and Methods
2.1. Focused Question
The systematic review was conducted using the PICO framework [35], which guided the formulation of the following focused question: In the case of tea infusions (population), what are the factors (investigated condition) influencing the fluoride content (outcome) compared to infusions prepared under different conditions or with varying tea types and preparation methods (comparison condition)?
2.2. Protocol
The article selection process for this systematic review was thoroughly documented using the PRISMA flow diagram [36] (Figure 2). The review was also registered on the Open Science Framework at the following link: https://osf.io/8ayuz (accessed on 24 April 2025).
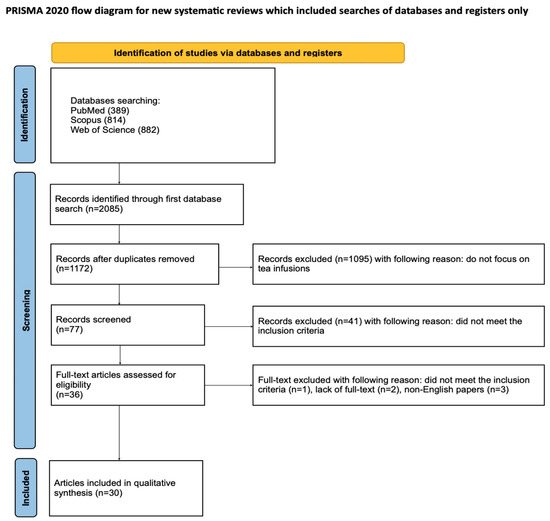
Figure 2.
The PRISMA 2020 flow diagram.
2.3. Eligibility Criteria
The researchers chose to include only those articles that met the following criteria [36,37,38,39,40,41,42,43,44]:
- Investigation of tea infusions;
- Fluoride release evaluation;
- In vitro studies;
- Studies in English;
- Full-text articles.
The exclusion criteria the reviewers agreed upon were as follows [36,37,38,39,40,41,42,43,44]:
- Not a tea infusion investigation;
- Evaluation of properties other than fluoride release;
- Non-English papers;
- Systematic review articles;
- Review articles;
- Full-text not accessible;
- Duplicated publications.
Only studies published in the year 2000 or later were considered eligible for inclusion.
2.4. Information Sources, Search Strategy, and Study Selection
In March 2025, a comprehensive literature search was conducted in the PubMed, Scopus, and Web of Science (WoS) databases to find studies that matched the established inclusion criteria. To target research on factors influencing fluoride content in tea infusions, the search was restricted to titles and abstracts containing the keywords: fluoride AND tea. All results were screened based on predefined eligibility standards, and only accessible full-text articles were considered for inclusion.
2.5. Data Collection Process and Data Items
Six independent reviewers (A.M., J.K., A.K., J.K., S.K., and M.M.) carefully selected articles that met the inclusion criteria. The extracted data included the first author, publication year, study design, article title, tea infusion, and its fluoride content. All relevant information was recorded in a standardized Excel file.
2.6. Risk of Bias and Quality Assessment
During the initial phase of study selection, each reviewer independently screened the titles and abstracts to reduce potential bias. The level of inter-reviewer agreement was assessed using Cohen’s k test. Any disagreements regarding article inclusion or exclusion were resolved through discussion among the authors [45].
2.7. Quality Assessment and Risk of Bias
The procedural quality of each study included in the article was independently assessed by two blinded reviewers (J.M. and M.D.) using the Joanna Briggs Institute (JBI) checklist designed for quasi-experimental studies (i.e., nonrandomized experimental studies) [46]. This tool includes nine specific questions:
- Is it clear in the study what is the ‘cause’ and what is the ‘effect’?
- Were the participants included in any similar comparisons?
- Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest?
- Was there a control group?
- Were there multiple measurements of the outcome both before and after the intervention/exposure?
- Was a follow-up completed, and if not, were differences between groups in terms of their follow-up adequately described and analyzed?
- Were the outcomes of participants included in any comparisons measured in the same way?
- Were the outcomes measured in a reliable way?
- Was an appropriate statistical analysis used?
Each question could be answered with “yes”, “no”, “unclear”, or “not applicable”. Any discrepancies between the reviewers’ responses were resolved through discussion until consensus was achieved. Cohen’s kappa test was performed to assess inter-rater reliability using MedCalc version 23.1.7 (MedCalc Software Ltd., Ostend, Belgium). The test results showed a kappa value of 0.87 (p < 0.001), indicating almost perfect agreement and high consistency among the reviewers’ assessments.
The quality of each study was assessed using a 0–9 point scale, where a “Yes” response earned 1 point, and all other answers received 0 points. Studies scoring 0–3 points were considered to have a high risk of bias, those with 4–6 points had a moderate risk, and scores between 7–9 indicated a low risk of bias.
3. Results
3.1. Study Selection
The initial search of PubMed, Scopus, and WoS databases yielded 2085 potentially relevant articles. After removing duplicates, 1172 articles remained, and studies not related to tea infusions were excluded. The remaining 77 articles were screened. After an initial search of titles and abstracts, 41 articles that did not meet the inclusion criteria were excluded. Of the remaining 36 studies, full text could not be accessed for 2 studies, 3 were written in a language other than English, and one did not meet the inclusion criteria after full text analysis. Ultimately, a total of 30 articles were included in the qualitative synthesis of this review. The considerable heterogeneity of the included studies prevented the conduct of a meta-analysis.
3.2. General Characteristics of the Qualified Articles
In the eligible articles, a significant proportion of authors examined more than one type of tea [19,28,31,32,34,47,48,49,50,51,52,53,54,55,56,57,58,59,60]. Black tea was the most frequently studied, appearing in 20 articles [19,28,32,34,47,49,51,52,54,55,56,57,58,60,61,62,63,64,65]. Green tea was investigated in 14 studies [19,28,31,32,47,49,51,52,54,55,56,57,59,60], followed by pu-erh tea in 7 articles [19,28,51,52,55,57,66], herbal teas in 5 articles [19,31,49,55,58], and white tea in 4 articles [28,32,47,55]. Some authors additionally assessed oolong tea [52,55].
Regarding the form of tea used, 15 studies analyzed loose leaves [19,28,31,32,34,47,50,51,52,57,58,62,63,66,67], 12 studies assessed tea in bags [19,28,31,32,49,50,51,52,54,56,58,63], and tea powder was used in 5 studies [49,54,55,65,68]. Importantly, 11 articles compared multiple tea forms [19,28,31,32,49,50,51,52,54,58,63].
Significant differences were observed in the brewing parameters. The water temperature during brewing ranged from 60 °C [68] to 100 °C [19,28,55,66], and the brewing time varied considerably, from as short as 1 min [31,56] to as long as 24 h [31]. Only three studies reported brewing according to the ISO 3103 standard [28,63,69], whereas none declared using brewing methods strictly based on manufacturer recommendations.
Fluoride concentrations were most commonly determined using a fluoride ion-selective electrode (FISE), employed in 24 studies [19,28,31,34,49,50,51,52,53,54,56,57,58,60,61,62,63,64,66,67,68,69,70]. Alternative methods included ion chromatography [32,57], molecular absorption spectrometry [10,31], ion-exclusion chromatography [57], single quadrupole ICP-MS [47], and MIP-OES techniques [59].
Several studies also examined the influence of external factors on fluoride release, such as water hardness [32], the pH of the infusion [31,55,67], or the fluoride concentration in the brewing water [54,56,65]. Brewing time and water temperature consistently appeared as significant factors affecting fluoride levels [19,28,32,49,51,52,54,55,56,58,60,63,64,65,68]. In some cases, additives like milk or spices were also considered [34,50]. The general information about the included studies is demonstrated in Table 1.

Table 1.
General characteristics of the included studies.
3.3. Main Study Outcomes
3.3.1. Variability in Fluoride Concentration Across Samples
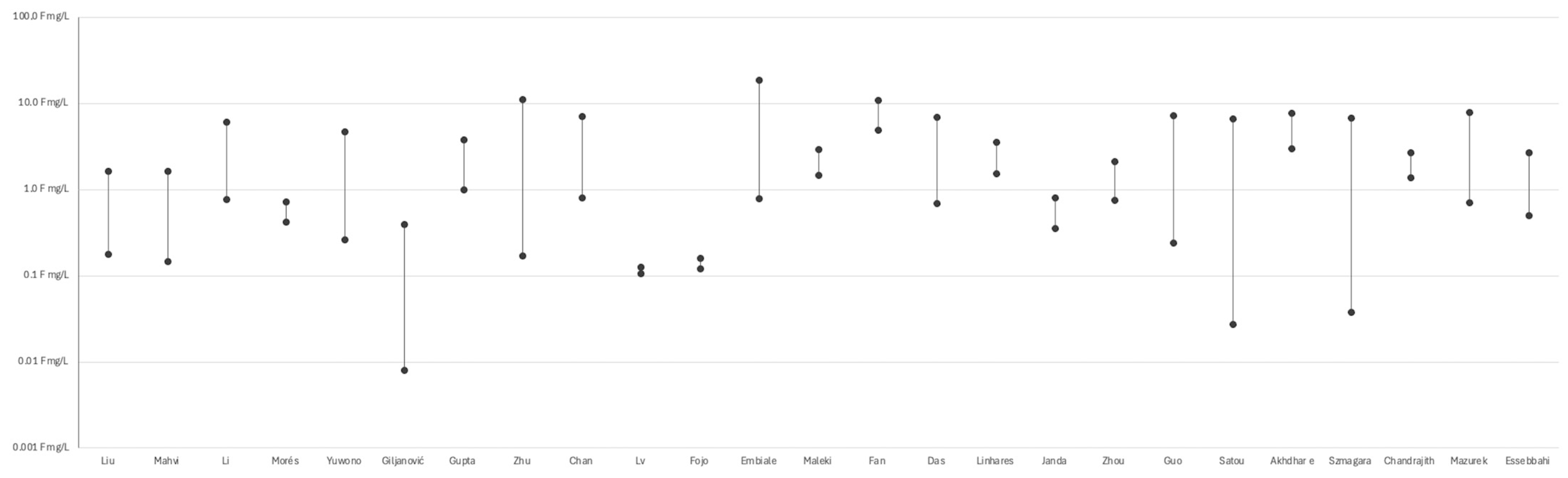
Numerous studies demonstrated substantial variability in fluoride concentrations across different tea samples, with values spanning over three orders of magnitude—even under comparable brewing conditions (see Figure 2). For instance, Szmagara et al. [19] reported values ranging from 0.04 to 6.76 mg/L, while Satou et al. [58] observed a similar spread among black and green teas (approximately 0.06 to 5.99 mg/L). Zhu et al. [51] and Guo et al. [47] documented particularly high levels in Chinese teas, reaching up to 11.05 mg/L and 7.25 mg/L, respectively. Das et al. [55], Chan et al. [52], and Linhares et al. [56] highlighted the impact of tea type (e.g., black, green, herbal), form (powder, leaves, bags), and geographical origin on fluoride release. Fan et al. [70] reported high fluoride exposure from brick tea, especially among Tibetan populations. Embiale et al. [54] demonstrated that using highly fluoridated water during brewing can significantly increase final fluoride content in the infusion. Beyond composition and origin, technological and chemical factors also influence fluoride levels. Chandrajith et al. [63] found that fermentation increases fluoride content in black tea, while Mazurek et al. [28] reported higher levels in granulated and bagged teas compared to loose-leaf forms. In addition, Janda et al. [34] observed an inverse correlation between fluoride concentration and antioxidant potential, suggesting a trade-off between fluoride exposure and potential health benefits. Essebbahi et al. [60] and Morés et al. [48] further emphasized the role of tea matrix composition, reinforcing the multifactorial nature of fluoride variability across samples. (see Figure 3) A detailed description of the included studies is presented in Supplementary Table S1.
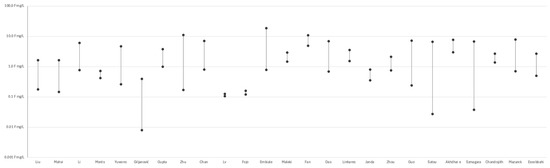
Figure 3.
Variability in fluoride levels among tea samples reported in selected studies (Liu et al. [68], Mahvi et al. [62], Li et al. [22], Morés et al. [48], Yuwono [49], Giljanović et al. [31], Gupta et al. [50], Zhu et al [51], Chan et al. [52], Lv et al. [66], Fojo et al. [53] Embiale et al. [54], Maleki et al [32], Fan et al. [70], Das et al. [55], Linhares et al. [56], Janda et al. [34], Zhou et al. [57], Akhdhar et al. [59], Szmagara et al. [19], Chandrajith et al. [63], Mazurek et al. [28], Essebbahi et al. [60]).
3.3.2. Fluoride Content by Tea Type and Geographic Origin
The included studies demonstrate substantial variability in fluoride concentrations across tea infusions, even when the same plant species and similar brewing conditions are used. This variability appears to be primarily influenced by the type of tea, country of origin, processing methods, and brand. For example, Satou et al. [58] reported fluoride levels in black tea ranging from 1.057 mg/L (Sri Lanka) to 6.68 mg/L (India), while Szmagara et al. [19] also identified black tea as having the highest fluoride concentrations among tested varieties. Guo et al. [47] observed concentrations as high as 7.25 mg/L in Chinese green teas, consistent with findings by Zhu et al. [51] for black tea bags.
Although average values often fall within safe consumption limits [19,20,28], Chan et al. [52] highlighted that consuming 1 L of certain teas could provide up to 150% of the recommended daily fluoride intake. Fan et al. [70] reported excessive fluoride exposure among Tibetans consuming up to 3.2 L of tea daily. Other studies [22,55] confirmed that chronic tea consumption can contribute to dental or skeletal fluorosis, particularly among vulnerable populations. Yuwono et al. [49] even recommended that children already affected by fluorosis should avoid black and green teas.
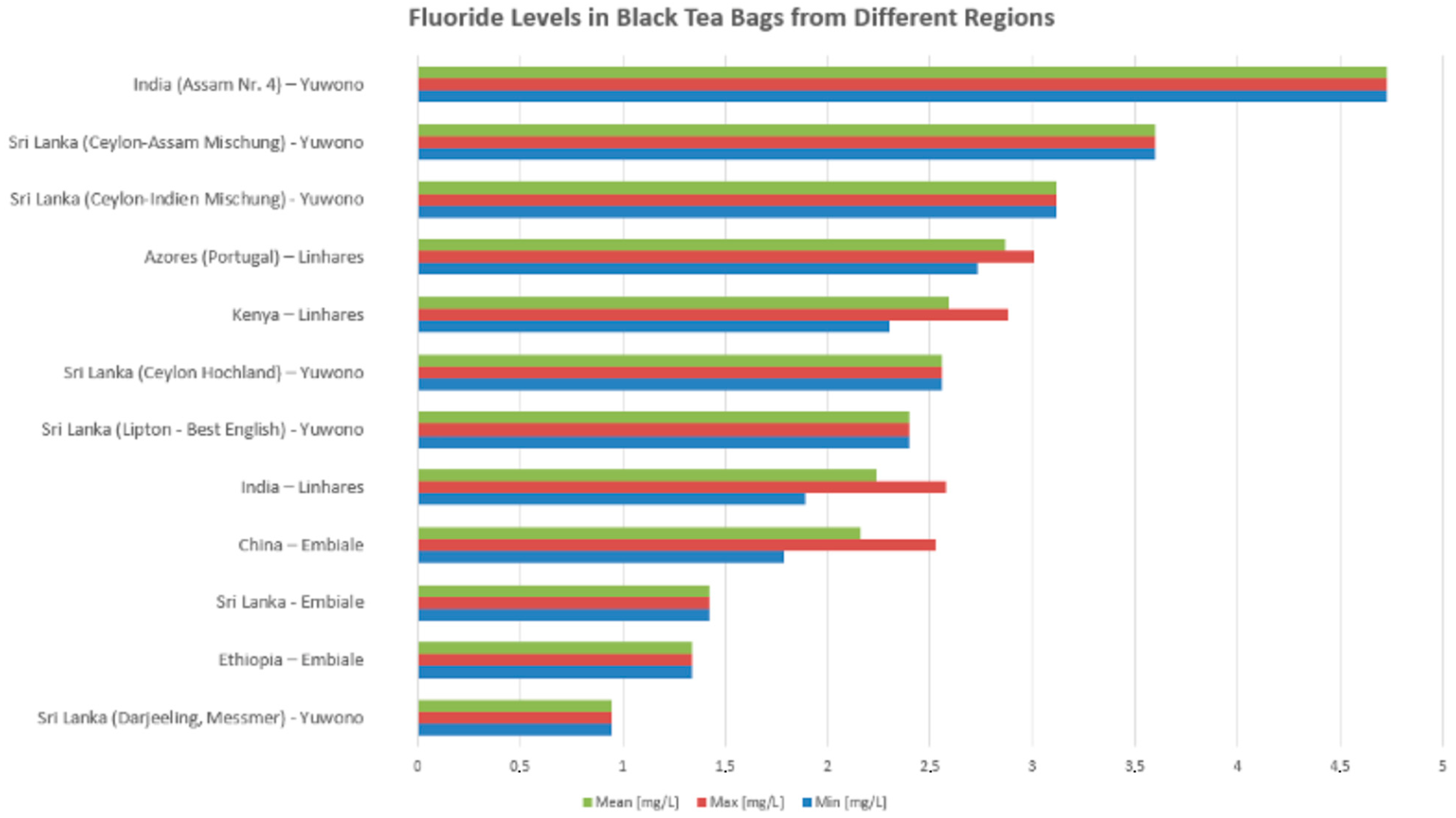
For studies on black tea in bag form brewed for 3–5 min, fluoride concentrations vary significantly across tea samples of different geographic origins. The highest fluoride content was observed in India (Assam Nr. 4) with a maximum level nearing 4.8 mg/L, followed by Sri Lankan blends such as Ceylon-Assam Mischung and Ceylon-Indien Mischung, both exceeding 3 mg/L in mean values. Teas from the Azores (Portugal) and Kenya also exhibited relatively high fluoride levels, with mean concentrations above 2.5 mg/L. Moderate fluoride levels were noted in samples from Sri Lanka (Ceylon Hochland), Lipton—Best English (Sri Lanka), and India (general samples), typically around 2.3–2.5 mg/L. Lower fluoride concentrations were recorded in teas from China, Sri Lanka (Embiale), and Ethiopia, ranging between 1.3 and 1.9 mg/L, while the lowest levels were found in the Sri Lanka (Darjeeling, Messmer) sample, which remained under 1 mg/L across all measures (min, mean, and max) (see Figure 4).
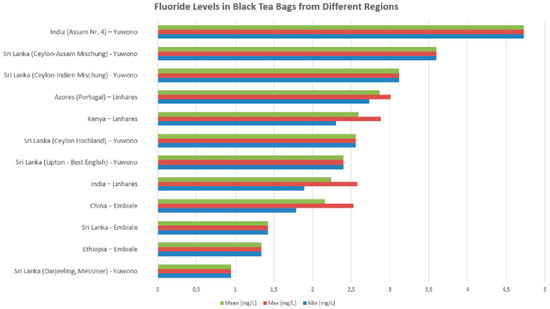
Figure 4.
Variability in fluoride levels among tea samples from different regions reported in selected studies (Yuwono [49], Linhares et al. [56], Embiale et al. [54]).
3.3.3. Effect of Brewing Time on Fluoride Release
There is strong agreement across studies that fluoride release increases with longer brewing times [19,31,32,51,52,54,56,61,63,64,68]. Aktug et al. [61] demonstrated that after approximately 20 min, fluoride concentrations tend to reach a plateau—a finding corroborated by Liu et al. [68] and Linhares et al. [56]. This supports the view that extended infusion times enhance fluoride extraction, though the marginal gain diminishes over time.
3.3.4. Influence of Tea Mass and Leaf Fragmentation
The degree of leaf fragmentation is another important factor influencing fluoride content. Teas in bagged or powdered form—characterized by finer particle size and greater surface area—release significantly more fluoride than loose leaf teas [19,28,31,32,49,50,51,52,54,58,63]. Mazurek et al. [28] reported particularly high fluoride levels in infusions made from granulated or bagged black teas, highlighting the role of processing in fluoride release. (see Table 2)

Table 2.
Influence of Tea Mass and Leaf Fragmentation on Fluoride Content.
3.3.5. Impact of Brewing Temperature and Water Composition
Several physicochemical parameters of the brewing process also affect fluoride concentrations. Lower pH values have been associated with increased fluoride release [22,55,56,58,67], although Zhu et al. [51] did not find a consistent pH effect, suggesting that its influence may depend on other variables.
Repeated brewing of the same tea leaves typically results in decreased fluoride release with each successive infusion [56,63,68,69]. Furthermore, the fluoride content of the brewing water itself plays a critical role: Li et al. [22], Chandrajith et al. [63], and Rajiv et al. [65] demonstrated a positive correlation between fluoride concentrations in water and tea infusion. Interestingly, Embiale et al. [54] observed that tea leaves can adsorb fluoride from high-fluoride water, occasionally resulting in lower concentrations in the infusion than in the original water.
Water hardness and ion content further modulate fluoride extraction. High calcium carbonate concentrations significantly reduced fluoride release [32,48]. Likewise, elevated levels of aluminum and iron (III) ions were found to bind fluoride and reduce its availability in the infusion [67]. (see Table 3).

Table 3.
Impact of Temperature and Water Composition on Fluoride Release; ↑- increase, ↓-decrease.
3.4. Quality Assessment and Risk of Bias of the Included Studies
For all of the 9 questions, 8 papers received a positive answer to 8 of them [31,32,48,50,52,54,64,67], 10 papers received a positive answer to 7 of them [22,34,49,51,53,56,62,66,68,70], 9 papers received a positive answer to 6 of them [19,28,47,55,58,59,63,65,69], and 3 remaining papers received a positive answer to 6 of them [57,60,61] (see Supplementary Table S2.)
Among the thirty studies included in this systematic review, thirteen were classified as having a low risk of bias, with total scores ranging from 7 to 9 points [22,28,31,32,34,48,50,51,52,53,54,64,67]. The remaining seventeen studies were rated as having a moderate risk of bias, scoring 5 or 6 points [19,47,49,55,56,57,58,59,60,61,62,63,65,66,68,69,70]. None of the studies were classified as high risk, as no score fell below the threshold of five points (see Figure 5).

Figure 5.
Risk of bias across the included studies, assessed using the JBI checklist for quasi-experimental studies. Green indicates low risk, red indicates high risk.
4. Discussion
This systematic review sought to answer the research question: What factors influence the fluoride content in tea infusions? The findings consistently demonstrate that fluoride release is not uniform but rather modulated by multiple interacting variables, including tea type, form of tea, brewing parameters, and external modifiers such as the water’s properties or the addition of spices. While wide variations in fluoride concentrations were observed, common trends were identified. Black and green teas tended to release the highest amounts of fluoride, particularly when processed into bagged or powdered forms. A particular threat in the context of fluorosis risk is the consumption of economy tea, which is a blend of teas from China, Sri Lanka, and India. Chan et al. [52] proved that after 2 min, the infusion contains almost twice as much fluoride as black tea, which, according to most studies, is considered the greatest source of fluoride among all types of tea. Consumption of 1 L of economy tea, which is about 4 cups, can lead to up to 150% of the daily recommended fluoride intake. This dietary exposure is further supported by systemic data. In addition to the variability in fluoride levels found in tea infusions, some studies have also explored systemic fluoride exposure resulting from high tea consumption. For example, Fan et al. [70] examined fluoride levels in urine samples from more than 1700 individuals in Tibet and found that excessive intake of brick tea (up to 3.2 L/day) was associated with elevated fluoride concentrations in the body and a high prevalence of dental and skeletal fluorosis. These findings highlight the value of integrating biomarker-based assessments—such as fluoride levels in urine or blood—into future research to better evaluate the health risks linked to chronic tea consumption. Longer brewing times and higher water temperatures consistently increased fluoride release, although a plateau was often observed beyond a certain point. Moreover, the mineral composition and pH of the water used for infusion emerged as additional significant factors. These results highlight the multifactorial nature of fluoride release during tea preparation and suggest that both intrinsic properties of the tea and extrinsic brewing conditions must be considered when evaluating potential fluoride exposure from tea consumption. Some publications draw attention to the significant effect of drinking tea infusion on the concentration of fluoride in the blood [71,72,73,74]. It has been proven that drinking significant amounts of brewed tea, in the amount of several cups or 30 glasses of iced tea per day, is equivalent to 13–14 mg of fluoride [71]. Such amounts lead to reaching a toxic level of fluoride in the blood, i.e., >15 umol/L, which leads to osteosclerosis, pain, bone fractures, kidney disorders, and digestive system disorders [72]. It is worth mentioning that the above symptoms are influenced by the sum of fluoride from water and tea, as well as from drugs such as flucoxentine, and the accumulation of fluoride can be intensified by kidney diseases [73]. Disorders related to excessive tea consumption are usually caused by drinking habits practiced for even 30 years, and in some cases, restoring the correct level of fluoride in the plasma (0–4 umol/L) is impossible despite radical treatment lasting even more than 10 years [71,74]. Future studies should also focus on the above issue to accurately assess what amounts of tea are safe to consume in people for whom it is a significant component of the diet.
Among the factors analyzed, tea type emerged as one of the most significant determinants of fluoride release. Across the majority of included studies, black teas exhibited the highest fluoride concentrations, followed closely by green teas [19,28,32,47,51,52,55]. This pattern is likely linked to the maturity of the tea leaves utilized; older leaves, commonly used for black and green teas, accumulate fluoride over time due to prolonged environmental exposure. Moreover, the regional origin of the tea plants strongly influenced fluoride accumulation, as areas with acidic soils and high environmental fluoride levels contributed to higher internal fluoride content [22,53,58]. Interestingly, some studies, such as that by Satou et al. [58], found green tea to release more fluoride than black tea, suggesting that local agricultural practices and specific cultivars could shift the typical trend. In contrast, white, pu-erh, and herbal teas consistently demonstrated lower fluoride levels. Herbal teas, in particular, released minimal fluoride, confirming that non-Camellia sinensis infusions are generally a safer choice for individuals needing to limit fluoride intake [19,31,49,57]. Nonetheless, variability among tea types also reflects different processing techniques; for instance, extensive fermentation processes, typical for pu-erh teas, may alter fluoride availability [19,66]. Overall, tea type remains a crucial, but not sole, predictor of fluoride content in the infusion.
The form of tea and degree of leaf fragmentation significantly influenced fluoride release. Fragmented tea, such as powders and tea bags, consistently yielded higher fluoride concentrations than loose-leaf teas [19,28,31,32,49,50,51,52,54,58,63]. The greater surface area provided by fragmented tea particles facilitates more efficient ion extraction into the water. Studies by Mazurek et al. [28] and Giljanović et al. [31] directly confirmed that tea bags and instant tea products released substantially higher fluoride concentrations compared to whole-leaf forms. Additionally, industrial processing methods, including mechanical pressing and cutting of leaves during tea bag production, contribute to increased cellular disruption and subsequent fluoride liberation. Gupta et al. [50] highlighted that granulated teas contained fluoride concentrations comparable to or exceeding those of bagged teas, emphasizing the role of mechanical processing. These findings are critical from a public health perspective, as tea bags and powdered teas are often preferred for their convenience, potentially exposing frequent consumers to higher fluoride levels unknowingly. Thus, the form of tea preparation must be considered when assessing individual fluoride intake from tea beverages.
Brewing conditions—particularly time, temperature, and water composition—further modulated fluoride release. A robust relationship was observed between longer brewing times and increased fluoride extraction [19,31,32,51,52,54,56,61,63,64,68]. Aktug et al. [61] and others showed that fluoride concentrations rose steeply during the first 20 min of brewing, after which the rate of release plateaued. This suggests that most available fluoride is solubilized early in the brewing process, and prolonged brewing does not proportionally increase fluoride concentrations. Similarly, higher water temperatures enhanced fluoride extraction due to increased solubility and diffusion rates [51,54,68]. Beyond brewing parameters, the chemical composition of the brewing water played a vital role. Studies revealed that using water with higher baseline fluoride levels led to increased fluoride content in the resulting tea infusion [22,63,65]. However, Embiale et al. [54] observed that in water with extremely high fluoride content, tea leaves could absorb part of the fluoride, thus slightly reducing the infusion’s fluoride concentration compared to the original water. Moreover, water hardness negatively correlated with fluoride extraction: higher concentrations of calcium carbonate precipitated fluoride ions, thereby lowering their availability in the infusion [32,48]. These findings emphasize that not only tea characteristics but also water quality must be accounted for in fluoride exposure assessments. Furthermore, one of the included studies—Gupta et al. [50]—examined different methods of tea preparation: plain tea, tea with milk, and tea with milk and sugar, and found that the addition of milk slightly decreased the concentration of fluoride in the infusion. This reduction is likely due to the binding of fluoride ions with calcium and other minerals present in milk.
Despite the strengths of this systematic review, several limitations must be acknowledged. The included studies displayed considerable methodological heterogeneity regarding tea types, brewing protocols, measurement techniques, and reporting standards. Differences in the use of fluoride ion-selective electrodes versus more sophisticated methods such as ion chromatography or ICP-MS [28,47,59] likely contributed to variability between reported fluoride concentrations. Furthermore, many studies lacked detailed information about the specific origin, cultivar, or processing methods of the tea samples, limiting the possibility of fully disentangling botanical from environmental influences. The absence of standardized brewing protocols (e.g., differences in leaf-to-water ratios, brewing vessels, agitation methods) further complicates comparisons. Future research should prioritize standardized methodologies, particularly in experimental design and fluoride quantification, to allow more robust comparisons and meta-analyses. In addition, more studies should explore the effects of additives (e.g., milk, spices) and consider the impact of habitual versus occasional tea consumption patterns in different populations. In medicine, serum fluoride levels are often used as an indicator of exposure to skeletal fluorosis. Further studies by using this marker could therefore confirm whether consuming a certain amount of fluoride from tea poses an actual risk of fluorosis or chronic poisoning. Understanding cumulative fluoride exposure from multiple sources remains critical for refining public health recommendations, particularly in regions where tea consumption is culturally significant.
5. Conclusions
This systematic review of thirty studies demonstrated that the fluoride content in tea infusions is influenced by a wide range of factors. A consistent finding across the reviewed studies was that fluoride concentration increases with longer brewing times, regardless of the temperature used. Additionally, a positive correlation between higher brewing temperatures and elevated fluoride concentrations was frequently observed. The presence of other ions, such as aluminum, iron, calcium, and fluoride itself, in the brewing water significantly affected the amount of free fluoride released into the infusion, highlighting the importance of considering both water hardness and fluoride content when preparing tea. It is also noteworthy that repeated brewing of the same tea leaves results in progressively lower fluoride concentrations in subsequent infusions.
The global diversity and variety of teas available on the market contribute to the observed variability in fluoride concentrations among tea infusions. Nevertheless, irrespective of the country of origin or specific tea variety, black tea infusions consistently exhibited the highest fluoride levels. Tea bags and loose tea leaves were the most frequently studied forms, with tea bag infusions consistently releasing higher fluoride concentrations than loose-leaf counterparts. Regarding analytical techniques, fluoride determination was most commonly performed using potentiometry with an ion-selective electrode. However, more precise and reliable measurements can be achieved using methods such as ion chromatography.
The findings of this systematic review demonstrate that the fluoride content in tea infusions ranges from 0.008 to over 8 mg/L, while the recommended daily intake for adults is 3–4 mg. Based on these results, regular consumption of teas with high fluoride content (particularly black and green teas in bagged or powdered form) could potentially contribute significantly to exceeding the daily fluoride requirements for adults, especially when prepared with longer brewing times and higher water temperatures. Several studies, including Chan et al. [52], indicated that drinking 1 L of tea could provide up to 150% of the daily fluoride requirement, while Fan et al. [70] reported that populations consuming up to 3.2 L of tea daily were at risk of excessive fluoride intake. Conversely, herbal teas, white teas, and loose-leaf preparations generally contain lower fluoride levels and are less likely to cause overconsumption. Based on these findings, the recommendation are for the tea consumers, particularly those who drink multiple cups daily, to choose herbal teas, white teas, or loose-leaf preparations, which generally contain lower fluoride levels; limit consumption of bagged or powdered black and green teas, which consistently show the highest fluoride concentrations; consider shorter brewing times (under 5 min) to reduce fluoride extraction; and be aware that tea brewing in areas with naturally high-fluoride water compounds the total exposure.
Given the widespread consumption of tea worldwide, understanding these factors is critical for assessing potential fluoride exposure through diet. Further standardized research is necessary to refine safe consumption guidelines, especially for populations at higher risk of fluoride overexposure.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/app15115974/s1, Table S1: Detailed characteristics of the included studies; Table S2: Quality assessment—JBI checklist for quasi-experimental studies (nonrandomized experimental studies).
Author Contributions
Conceptualization, A.M., J.M. and M.D.; methodology, A.M. and J.M.; software, A.M. and M.J.; validation, J.M. and M.D.; formal analysis, M.D. and J.M.; investigation, A.M., A.K., J.K. (Julia Kensy), J.K. (Jan Kiryk), S.K. and M.M.; resources, A.M., A.K., M.M., J.K. (Julia Kensy), J.K. (Jan Kiryk) and S.K.; data curation, J.M. and M.D.; writing—original draft preparation, A.M., A.K., J.K. (Julia Kensy), J.K. (Jan Kiryk) and S.K; writing—review and editing, A.M., J.K (Julia Kensy), M.J. and J.M.; visualization, J.K. (Jan Kiryk); supervision, M.D. and J.M.; project administration, J.M. and M.D.; funding acquisition, J.M. and M.D. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financed by a subsidy from Wroclaw Medical University, number SUBZ.B180.25.091.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ahmad, S.; Singh, R.; Arfin, T.; Neeti, K. Fluoride Contamination, Consequences and Removal Techniques in Water: A Review. Environ. Sci. Adv. 2022, 1, 620–661. [Google Scholar] [CrossRef]
- Aoun, A.; Darwiche, F.; Al Hayek, S.; Doumit, J. The Fluoride Debate: The Pros and Cons of Fluoridation. Prev. Nutr. Food Sci. 2018, 23, 171–180. [Google Scholar] [CrossRef]
- Lubojanski, A.; Piesiak-Panczyszyn, D.; Zakrzewski, W.; Dobrzynski, W.; Szymonowicz, M.; Rybak, Z.; Mielan, B.; Wiglusz, R.J.; Watras, A.; Dobrzynski, M. The Safety of Fluoride Compounds and Their Effect on the Human Body—A Narrative Review. Materials 2023, 16, 1242. [Google Scholar] [CrossRef] [PubMed]
- Peckham, S.; Awofeso, N. Water Fluoridation: A Critical Review of the Physiological Effects of Ingested Fluoride as a Public Health Intervention. Sci. World J. 2014, 2014, 293019. [Google Scholar] [CrossRef] [PubMed]
- Olczak-Kowalczyk, D.; Mielczarek, A.; Jackowska, T.; Mielnik-Błaszczak, M.; Turska-Szybka, A.; Opydo-Szymaczek, J.; Jurczak, A.; Kaczmarek, U. Fluoride agents in the prevention and treatment of dental caries and erosion in children, adolescents and adults—Recommendations of Polish Experts. Update of recommendations: Individual fluoride prevention in children and adolescents—Recommendations of Polish Experts. Nowa Stomatol. 2022, 27, 35–59. [Google Scholar] [CrossRef]
- Herman, K.; Czajczyńska-Waszkiewicz, A.; Kowalczyk-Zając, M.; Dobrzyński, M. Assessment of the influence of vegetarian diet on the occurrence of erosive and abrasive cavities in hard tooth tissues. Postepy Hig. Med. Dosw. (Online) 2011, 65, 764–769. [Google Scholar] [CrossRef]
- Zohoori, F.V.; Buzalaf, M. Fluoride. Adv. Nutr. 2022, 13, 2679–2680. [Google Scholar] [CrossRef]
- Nassar, Y.; Brizuela, M. The Role of Fluoride on Caries Prevention. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Mankar, N.; Kumbhare, S.; Nikhade, P.; Mahapatra, J.; Agrawal, P. Role of Fluoride in Dentistry: A Narrative Review. Cureus 2023, 15, e50884. [Google Scholar] [CrossRef]
- Piszko, P.J.; Piszko, A.; Kiryk, S.; Kiryk, J.; Kensy, J.; Michalak, M.; Matys, J.; Dobrzyński, M. Fluoride Release from Two Commercially Available Dental Fluoride Gels—In Vitro Study. Gels 2025, 11, 135. [Google Scholar] [CrossRef]
- Jullien, S. Prophylaxis of Caries with Fluoride for Children under Five Years. BMC Pediatr. 2021, 21, 351. [Google Scholar] [CrossRef]
- Vasisth, D.; Mehra, P.; Yadav, L.; Kumari, V.; Bhatia, U.; Garg, R. Fluoride and Its Implications on Oral Health: A Review. J. Pharm. Bioallied Sci. 2024, 16, S49–S52. [Google Scholar] [CrossRef]
- Guth, S.; Hüser, S.; Roth, A.; Degen, G.; Diel, P.; Edlund, K.; Eisenbrand, G.; Engel, K.-H.; Epe, B.; Grune, T.; et al. Toxicity of Fluoride: Critical Evaluation of Evidence for Human Developmental Neurotoxicity in Epidemiological Studies, Animal Experiments and in Vitro Analyses. Arch. Toxicol. 2020, 94, 1375–1415. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, E.; Inkielewicz, I.; Czarnowski, W.; Szefer, P. Assessment of Fluoride Concentration and Daily Intake by Human from Tea and Herbal Infusions. Food Chem. Toxicol. 2008, 46, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.T.; Potter, W.; Limeback, H.; Godfrey, M. Risk Assessment of Fluoride Intake from Tea in the Republic of Ireland and Its Implications for Public Health and Water Fluoridation. Int. J. Environ. Res. Public. Health 2016, 13, 259. [Google Scholar] [CrossRef]
- Rankin, S.J.; Levy, S.M.; Warren, J.J.; Gilmore, J.E.; Broffitt, B. Fluoride content of solid foods impacts daily intake. J. Public Health Dent. 2012, 72, 128–134. [Google Scholar] [CrossRef]
- Peng, C.; Cai, H.; Zhu, X.; Li, D.; Yang, Y.; Hou, R.; Wan, X. Analysis of Naturally Occurring Fluoride in Commercial Teas and Estimation of Its Daily Intake through Tea Consumption. J. Food Sci. 2016, 81, H235–H239. [Google Scholar] [CrossRef] [PubMed]
- Esfehani, M.; Ghasemzadeh, S.; Mirzadeh, M. Comparison of Fluoride Ion Concentration in Black, Green and White Tea. Int. J. Ayurvedic Med. 2018, 9, 263–265. [Google Scholar] [CrossRef]
- Szmagara, A.; Krzyszczak, A.; Stefaniak, E.A. Determination of Fluoride Content in Teas and Herbal Products Popular in Poland. J. Environ. Health Sci. Eng. 2022, 20, 717–727. [Google Scholar] [CrossRef]
- Emekli-Alturfan, E.; Yarat, A.; Akyuz, S. Fluoride Levels in Various Black Tea, Herbal and Fruit Infusions Consumed in Turkey. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2009, 47, 1495–1498. [Google Scholar] [CrossRef]
- Validandi, V.; Viswanathan, G.; Khandare, A.L. Comparison of Fluoride Levels (Total and Extracted) in Young, Old Tea Leaves and Market Tea Samples along with Impact of Tea Infusion on Dental Fluorosis in Fluoride Endemic Villages of Nalgonda District, India. Adv. Dent. Oral Health 2019, 10, 1–8. [Google Scholar] [CrossRef]
- Li, H.; Liu, Q.; Wang, W.; Yang, L.; Li, Y.; Feng, F.; Zhao, X.; Hou, K.; Wang, G. Fluoride in Drinking Water, Brick Tea Infusion and Human Urine in Two Counties in Inner Mongolia, China. J. Hazard. Mater. 2009, 167, 892–895. [Google Scholar] [CrossRef] [PubMed]
- Fung, K.F.; Zhang, Z.Q.; Wong, J.W.C.; Wong, M.H. Fluoride Contents in Tea and Soil from Tea Plantations and the Release of Fluoride into Tea Liquor during Infusion. Environ. Pollut. 1999, 104, 197–205. [Google Scholar] [CrossRef]
- Cai, H.; Zhu, X.; Peng, C.; Xu, W.; Li, D.; Wang, Y.; Fang, S.; Li, Y.; Hu, S.; Wan, X. Critical Factors Determining Fluoride Concentration in Tea Leaves Produced from Anhui Province, China. Ecotoxicol. Environ. Saf. 2016, 131, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, C.; Li, J.; Zhang, Y.; Zhu, C.; Gu, D.; Zeng, L. Critical Review of Fluoride in Tea Plants (Camellia sinensis): Absorption, Transportation, Tolerance Mechanisms, and Defluorination Measures. Beverage Plant Res. 2024, 4, e019. [Google Scholar] [CrossRef]
- Zhang, D.; Xu, X.; Wu, X.; Lin, Y.; Li, B.; Chen, Y.; Li, X.; Shen, J.; Xiao, L.; Lu, S. Monitoring Fluorine Levels in Tea Leaves from Major Producing Areas in China and the Relative Health Risk. J. Food Compos. Anal. 2023, 118, 105205. [Google Scholar] [CrossRef]
- Cao, J.; Zhao, Y.; Li, Y.; Deng, H.J.; Yi, J.; Liu, J.W. Fluoride Levels in Various Black Tea Commodities: Measurement and Safety Evaluation. Food Chem. Toxicol. 2006, 44, 1131–1137. [Google Scholar] [CrossRef]
- Mazurek, A.; Kowalska, G.; Włodarczyk-Stasiak, M.; Wyrostek, J.; Kowalski, R. The Influence of the Preparation of Tea Infusion on the Content of Fluoride and the Assessment of Health Risk for the Consumer in Poland. Appl. Sci. 2023, 13, 5075. [Google Scholar] [CrossRef]
- Jin, C.; Yan, Z.; Jianwei, L. Processing Procedures of Brick Tea and Their Influence on Fluorine Content. Food Chem. Toxicol. 2001, 39, 959–962. [Google Scholar] [CrossRef]
- Amorello, D.; Barreca, S.; Pensato, F.; Orecchio, S. Potentiometric Analysis of Fluoride in Commonly Consumed Beverages: Method Development, Evaluation, and Risk Assessment. J. Food Compos. Anal. 2025, 137, 106836. [Google Scholar] [CrossRef]
- Giljanović, J.; Prkić, A.; Bralić, M.; Brkljača, M. Determination of Fluoride Content in Tea Infusion by Using Fluoride Ion-Selective Electrode. Int. J. Electrochem. Sci. 2012, 7, 2918–2927. [Google Scholar] [CrossRef]
- Maleki, A.; Abulmohammadi, P.; Teymouri, P.; Zand, S.; Daraei, H.; Mahvi, A.; Shahsawari, S. Effect of Brewing Time and Water Hardness on Fluoride Release from Different Iranian Teas. Fluoride 2016, 49, 263–273. [Google Scholar]
- Lu, Y.; Guo, W.-F.; Yang, X.-Q. Fluoride Content in Tea and Its Relationship with Tea Quality. J. Agric. Food Chem. 2004, 52, 4472–4476. [Google Scholar] [CrossRef] [PubMed]
- Gutowska, I.; Gutowska, I.; Janda, K.; Jakubczyk, K.; Woźniak, K.; Siwiec, E.; Wolska, J. Does the Addition of Spices Change the Content of Fluoride and Antioxidants in Black Tea Infusions? J. Elem. 2018, 23, 599–609. [Google Scholar] [CrossRef]
- Huang, X.; Lin, J.; Demner-Fushman, D. Evaluation of PICO as a Knowledge Representation for Clinical Questions. AMIA. Annu. Symp. Proc. 2006, 2006, 359–363. [Google Scholar]
- Homa, K.; Zakrzewski, W.; Dobrzyński, W.; Piszko, P.J.; Piszko, A.; Matys, J.; Wiglusz, R.J.; Dobrzyński, M. Surface Functionalization of Titanium-Based Implants with a Nanohydroxyapatite Layer and Its Impact on Osteoblasts: A Systematic Review. J. Funct. Biomater. 2024, 15, 45. [Google Scholar] [CrossRef]
- Kensy, J.; Dobrzyński, M.; Wiench, R.; Grzech-Leśniak, K.; Matys, J. Fibroblasts Adhesion to Laser-Modified Titanium Surfaces—A Systematic Review. Materials 2021, 14, 7305. [Google Scholar] [CrossRef]
- Kiryk, J.; Kiryk, S.; Kensy, J.; Świenc, W.; Palka, B.; Zimoląg-Dydak, M.; Dobrzyński, W.; Matys, J.; Dobrzyński, M. Effectiveness of Laser-Assisted Teeth Bleaching: A Systematic Review. Appl. Sci. 2024, 14, 9219. [Google Scholar] [CrossRef]
- Matys, J.; Kensy, J.; Gedrange, T.; Zawiślak, I.; Grzech-Leśniak, K.; Dobrzyński, M. A Molecular Approach for Detecting Bacteria and Fungi in Healthcare Environment Aerosols: A Systematic Review. Int. J. Mol. Sci. 2024, 25, 4154. [Google Scholar] [CrossRef]
- Piszko, P.J.; Piszko, A.; Kiryk, J.; Lubojański, A.; Dobrzyński, W.; Wiglusz, R.J.; Matys, J.; Dobrzyński, M. The Influence of Fluoride Gels on the Physicochemical Properties of Tooth Tissues and Dental Materials—A Systematic Review. Gels 2024, 10, 98. [Google Scholar] [CrossRef]
- Oleniacz-Trawińska, M.; Kotela, A.; Kensy, J.; Kiryk, S.; Dobrzyński, W.; Kiryk, J.; Gerber, H.; Fast, M.; Matys, J.; Dobrzyński, M. Evaluation of Factors Affecting Fluoride Release from Compomer Restorative Materials: A Systematic Review. Materials 2025, 18, 1627. [Google Scholar] [CrossRef]
- Tokarczuk, D.; Tokarczuk, O.; Kiryk, J.; Kensy, J.; Szablińska, M.; Dyl, T.; Dobrzyński, W.; Matys, J.; Dobrzyński, M. Fluoride Release by Restorative Materials after the Application of Surface Coating Agents: A Systematic Review. Appl. Sci. 2024, 14, 4956. [Google Scholar] [CrossRef]
- Morawska-Wilk, A.; Kensy, J.; Kiryk, S.; Kotela, A.; Kiryk, J.; Michalak, M.; Grychowska, N.; Fast, M.; Matys, J.; Dobrzyński, M. Evaluation of Factors Influencing Fluoride Release from Dental Nanocomposite Materials: A Systematic Review. Nanomaterials 2025, 15, 651. [Google Scholar] [CrossRef]
- Struzik, N.; Kensy, J.; Piszko, P.J.; Kiryk, J.; Wiśniewska, K.; Kiryk, S.; Korjat, Ł.; Horodniczy, T.; Sobierajska, P.; Matys, J.; et al. Contamination in Bone Substitute Materials: A Systematic Review. Appl. Sci. 2024, 14, 8266. [Google Scholar] [CrossRef]
- Watson, P.F.; Petrie, A. Method Agreement Analysis: A Review of Correct Methodology. Theriogenology 2010, 73, 1167–1179. [Google Scholar] [CrossRef]
- Chapter 3: Systematic Reviews of Effectiveness. In JBI Manual for Evidence Synthesis; JBI: Adelide, Australia, 2020; ISBN 978-0-6488488-0-6.
- Guo, W.; Lin, X.; Jin, L.; Hu, S. Single Quadrupole Inductively Coupled Plasma-Mass Spectrometry for the Measurement of Fluorine in Tea Infusions and Its Health Risk Assessment. J. Food Compos. Anal. 2020, 86, 103378. [Google Scholar] [CrossRef]
- Morés, S.; Monteiro, G.C.; Santos, F. da S.; Carasek, E.; Welz, B. Determination of Fluorine in Tea Using High-Resolution Molecular Absorption Spectrometry with Electrothermal Vaporization of the Calcium Mono-Fluoride CaF. Talanta 2011, 85, 2681–2685. [Google Scholar] [CrossRef]
- Yuwono, M. Determination of Fluoride in Black, Green and Herbal Teas by Ionselective Electrode Using a Standard-Addition Method. Dent. J. Maj. Kedokt. Gigi 2005, 38, 91. [Google Scholar] [CrossRef]
- Gupta, P.; Sandesh, N. Estimation of Fluoride Concentration in Tea Infusions, Prepared from Different Forms of Tea, Commercially Available in Mathura City. J. Int. Soc. Prev. Community Dent. 2012, 2, 64–68. [Google Scholar] [CrossRef]
- Zhu, J.J.; Ath, T.; Matinlinna, J.P.; Jkh, T.; Hägg, U. Potentiometric Determination of Fluoride Release from Three Types of Tea Leaves. Int. J. Electrochem. Sci. 2013, 8, 11142–11150. [Google Scholar] [CrossRef]
- Chan, L.; Mehra, A.; Saikat, S.; Lynch, P. Human Exposure Assessment of Fluoride from Tea (Camellia Sinensis L.): A UK Based Issue? Food Res. Int. 2013, 51, 564–570. [Google Scholar] [CrossRef]
- Fojo, C.; Figueira, M.E.; Almeida, C.M.M. Fluoride Content of Soft Drinks, Nectars, Juices, Juice Drinks, Concentrates, Teas and Infusions Marketed in Portugal. Food Addit. Contam. Part Chem. Anal. Control Expo. Risk Assess. 2013, 30, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Embiale, A.; Chandravanshi, B.S.; Zewge, F. Levels of Fluoride in the Ethiopian and Imported Black Tea (Camellia sinensis) Infusions Prepared in Tap and Fluoride-Rich Natural Waters. Int. J. Food Eng. 2014, 10, 447–455. [Google Scholar] [CrossRef]
- Das, S.; de Oliveira, L.M.; da Silva, E.; Liu, Y.; Ma, L.Q. Fluoride Concentrations in Traditional and Herbal Teas: Health Risk Assessment. Environ. Pollut. Barking Essex 1987 2017, 231, 779–784. [Google Scholar] [CrossRef] [PubMed]
- Linhares, D.P.S.; Garcia, P.V.; Amaral, L.; Ferreira, T.; Dos Santos Rodrigues, A. Safety Evaluation of Fluoride Content in Tea Infusions Consumed in the Azores—A Volcanic Region with Water Springs Naturally Enriched in Fluoride. Biol. Trace Elem. Res. 2017, 179, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Li, H.; Ye, S.; Tan, H. A New Method for Determination of Fluoride Ion in Commodity Tea by Ion-Exclusion Chromatography. CyTA-J. Food 2018, 16, 637–641. [Google Scholar] [CrossRef]
- Satou, R.; Oka, S.; Sugihara, N. Risk Assessment of Fluoride Daily Intake from Preference Beverage. J. Dent. Sci. 2021, 16, 220–228. [Google Scholar] [CrossRef]
- Akhdhar, A.; Schneider, M.; Hellmann, S.; Orme, A.; Carasek, E.; Krupp, E.M.; Feldmann, J. The Use of Microwave-Induced Plasma Optical Emission Spectrometry for Fluorine Determination and Its Application to Tea Infusions. Talanta 2021, 227, 122190. [Google Scholar] [CrossRef]
- Essebbahi, I.; Ouazzani, C.; Moustaghfir, A.; Er-ramly, A.; El Baroudi, Y.; Dami, A.; Balouch, L. Analysis of the Fluoride Levels of Well Water and Tea Consumed by the Moroccan Population in Different Rural Areas. Mater. Today Proc. 2023, 72, 3347–3350. [Google Scholar] [CrossRef]
- Aktuğ, Ü.; Duydu, Y. The Effects of Color and Brightness of Brewed Black Tea on Its Fluoride Concentration in Türkiye. Ank. Univ. Eczaci. Fak. Derg. 2023, 47, 10. [Google Scholar] [CrossRef]
- Mahvi, A.H.; Zazoli, M.A.; Younecian, M.; Esfandiari, Y. Fluoride Content of Iranian Black Tea and Tea Liquor. Fluoride 2006, 39, 266. [Google Scholar]
- Chandrajith, R.; Bhagya, S.; Diyabalanage, S.; Wimalasiri, S.; Ranatunga, M.A.B.; Barth, J.A.C. Exposure Assessment of Fluoride Intake Through Commercially Available Black Tea (Camellia sinensis L.) from Areas with High Incidences of Chronic Kidney Disease with Undetermined Origin (CKDu) in Sri Lanka. Biol. Trace Elem. Res. 2022, 200, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Edussuriya, R.; Hettithanthri, O.; Rajapaksha, A.U.; Jayasinghe, C.; Vithanage, M. Intake of Fluoride and Other Hofmeister Ions from Black Tea Consumption in CKDu Prevalent Areas, Sri Lanka. Environ. Sci. Pollut. Res. 2023, 30, 41900–41909. [Google Scholar] [CrossRef]
- Rajiv, D.; Prem, D.; Ramesh, M.; Jacob, M.; Indrapriyadharshini, K. Fluoride Content in Various Types of Tea Used by Tea Stalls in Salem District—An in Vitro Cross Sectional Study. J. Oral Maxillofac. Pathol. JOMFP 2023, 27, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.-P.; Lin, Z.; Tan, J.-F.; Guo, L. Contents of Fluoride, Lead, Copper, Chromium, Arsenic and Cadmium in Chinese Pu-Erh Tea. Food Res. Int. 2013, 53, 938–944. [Google Scholar] [CrossRef]
- Erdemoglu, S.B.; Türkdemir, H.; Gücer, S. Determination of Total and Fluoride Bound Aluminium in Tea Infusions by Ion Selective Electrode and Flame Atomic Absorption Spectrometry. Anal. Lett. 2000, 33, 1513–1529. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, B.; Wang, B.; Wang, M. The Characteristics of Dissolution of Fluoride and Aluminum from Brick Tea. Chin. J. Geochem. 2005, 24, 382–385. [Google Scholar] [CrossRef]
- Kanrar, B.; Kundu, S.; Sengupta, S.; Yeasin, M.; Paul, R.K.; Karak, T. Assessment and Health Risk of Fluoride from Northeast Indian Tea (Camellia sinensis L.): Fixing up the Maximum Residue Level of Fluoride in Tea. J. Food Compos. Anal. 2024, 127, 105928. [Google Scholar] [CrossRef]
- Fan, Z.; Gao, Y.; Wang, W.; Gong, H.; Guo, M.; Zhao, S.; Liu, X.; Yu, B.; Sun, D. Prevalence of Brick Tea-Type Fluorosis in the Tibet Autonomous Region. J. Epidemiol. 2016, 26, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Jasim, S.; Wenger, D.; Wermers, R.A. Skeletal fluorosis related to habitual tea consumption: Long-term follow-up after reduction and discontinuation of tea. AACE Clin. Case Rep. 2018, 4, 98–103. [Google Scholar] [CrossRef]
- Johnson, J.E.H.; Kearns, A.E.; Doran, P.M.; Khoo, T.K.; Wermers, R.A. Fluoride-related bone disease associated with habitual tea consumption. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 2007; pp. 719–724. [Google Scholar] [CrossRef]
- Whyte, M.P.; Totty, W.G.; Lim, V.T.; Whitford, G.M. Skeletal fluorosis from instant tea. J. Bone Miner. Res. 2008, 23, 759–769. [Google Scholar] [CrossRef]
- Izuora, K.; Twombly, J.G.; Whitford, G.M.; Demertzis, J.; Pacifici, R.; Whyte, M.P. Skeletal fluorosis from brewed tea. J. Clin. Endocrinol. Metab. 2011, 96, 2318–2324. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).