- Article
Effect of Partial Co and Fe Substitution on LaFeO3@C, LaCoO3@C Catalysts in the Oxidation of Furfural
- Diego Diaz,
- Dana Arias and
- Catherine Sepulveda
- + 6 authors
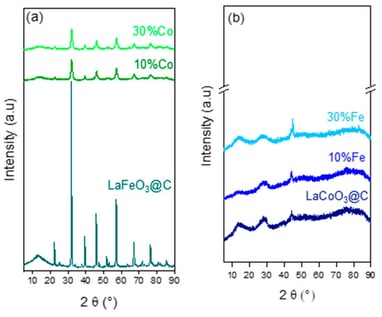
Pure LaFeO3@C and LaCoO3@C and substituted LaFe1-xCoxO3 and LaCo1-xFexO3 perovskites (x = 0.10; 0.30) were used as catalysts for the liquid-phase oxidation of furfural at 150 °C and 30 bar of O2 pressure. The perovskites were characterized by XRD, H2-TPR, N2 physisorption, TPR-MeOH, and XPS. The carbon in situ incorporation (@C) increases the surface area, favoring oxygen mobility leading to LaFeO3@C stabilizing the redox pair Fe3+/Fe2+. In contrast, no evidence of the formation of a LaCoO3@C perovskite structure through @C incorporation was observed. The gradual substitution of Fe with Co (10 and 30%) in LaFeO3@C decreases the crystallinity, redox and basic properties, and surface area. For LaCoO3@C, after the substitution of Co with 10 and 30% of Fe, only metal (La, Fe, Co) oxides as segregated phases were observed. The highest catalytic activity and selectivity to maleic acid of LaFeO3@C is attributed to the higher surface area, crystalline structure, and surface-reducible Fe3+ species, favoring oxygen mobility and promoting their more oxidizing capacity. The lower catalytic activity of LaCoO3@C, the Co- and Fe-substituted LaFeO3@C and LaCoO3@C catalysts, is attributed to the smaller surface area, and the similar selectivity towards maleic acid, 5-hydroxy-2(5H) and furanone indicates that the active site type is not modified in comparison to LaFeO3@C.
9 April 2026