Journal Description
Lipidology
Lipidology
is a peer-reviewed, open access journal on lipid sciences published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: first decisions in 19 days; acceptance to publication in 8 days (median values for MDPI journals in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Latest Articles
Statins Support the Antitumor Activity of Somatostatin Analogues in Advanced Bronchopulmonary Neuroendocrine Tumors: A Clinical and In Vitro Study
Lipidology 2026, 3(2), 12; https://doi.org/10.3390/lipidology3020012 - 11 Apr 2026
Abstract
►
Show Figures
Background/Objectives: Metabolic alterations, including dyslipidemia, may influence tumor biology and treatment outcomes in neuroendocrine tumors. However, the clinical relevance of dyslipidemia and lipid-lowering therapy in bronchopulmonary neuroendocrine tumors (BP-NETs) treated with somatostatin analogues (SSAs) remains poorly defined. This translational proof-of-concept study evaluated progression-free
[...] Read more.
Background/Objectives: Metabolic alterations, including dyslipidemia, may influence tumor biology and treatment outcomes in neuroendocrine tumors. However, the clinical relevance of dyslipidemia and lipid-lowering therapy in bronchopulmonary neuroendocrine tumors (BP-NETs) treated with somatostatin analogues (SSAs) remains poorly defined. This translational proof-of-concept study evaluated progression-free survival (PFS) in patients with advanced BP-NETs receiving SSAs according to dyslipidemia and statin therapy and explored the effects of statin-SSA combination treatment in vitro. Methods: We retrospectively analyzed 24 patients with advanced well-differentiated BP-NETs treated with SSAs as first-line therapy. Fourteen patients (58.3%) had dyslipidemia, and 11 of them were receiving statins. In parallel, NCI-H727 cells were treated with atorvastatin (10 µM), lanreotide (5 or 10 µM), or their combination for 48–72 h. Cell viability, proliferation, cell death, apoptosis, DNA damage, and ATP production were assessed. Results: Median PFS was 22.5 months overall. A trend toward longer PFS was observed in non-dyslipidemic vs. dyslipidemic patients (70 vs. 36 months, p = 0.08). Among dyslipidemic patients, statin therapy was associated with a non-significant trend toward longer PFS compared with no statin therapy (36 vs. 18 months, p = 0.30). In vitro, combined atorvastatin–lanreotide treatment reduced cell viability and proliferation, increased cell death, enhanced cleaved caspase-3 and p-γH2AX expression, and reduced ATP production. Conclusions: These findings support the potential relevance of lipid metabolism modulation as an adjunct strategy in advanced BP-NETs while highlighting the need for larger prospective studies and dedicated biochemical investigation of the underlying lipid-related pathways.
Full article
Open AccessReview
Narrative Review of Human Adiposity: From Evolutionary Energy-Thriftiness and Ancestral Wellness to the Modern Inflammatory-Related Illness. The Role of Lifestyle Transition
by
Roberto Carlos Burini
Lipidology 2026, 3(1), 11; https://doi.org/10.3390/lipidology3010011 - 18 Mar 2026
Abstract
Energy thriftiness and metabolic adaptations have had a crucial role in the emergence and spreading of the Homo lineage in the world. A higher-energy demand was required not only for the growing body mass, encephalization and human proliferation, but also for the survival
[...] Read more.
Energy thriftiness and metabolic adaptations have had a crucial role in the emergence and spreading of the Homo lineage in the world. A higher-energy demand was required not only for the growing body mass, encephalization and human proliferation, but also for the survival adaptations to the environmental stresses. Because lean body mass lacks the energy-storage capacity required to supply the body’s demands, dedicated fat-storing cells originated. To feed such fat stores, the hominid evolution developed “meat-adaptive” genes to detect, digest and metabolize higher fat diets, and body-fat stores can be affected by lifestyle through hormonal-controlled daily energy balance. In energy surplus conditions, hypertrophy and hyperplasia of adipocytes can occur, with hypertrophic adipocyte signaling both a neo-adipocyte differentiation (leading to hyperplasia) and a local macrophage density (resident + infiltrated macrophages) for fat surplus scavenging. Adiposity-induced inflammation is caused by fat-overstored (hypertrophied) adipocytes that may operate as an overactive endocrine organ secreting an array of pro-inflammatory adipokines that, in combination with resident-macrophage activity and infiltrated blood-recruited, monocyte-derived macrophages, amplify the inflammatory process by spurting pro-inflammatory cytokines into the bloodstream. From an evolutionary perspective, obese humans represent a natural selection overexpressing the “thrifty” genes evolved for efficient food collection and fat deposition intended to help in survival in prolonged periods of famine. However, genetically speaking, obesity is a polygenic multifactorial disorder. Considering the rapidity of obesity-epidemic growth worldwide, epigenetic sets forth the key assumption of the mismatch between our human genome molded over thousands of generations, coping with the unprecedented dietary and physical conditions. Consequently, obesity would be due to our evolutionary-adapted polygenic-charge expressed by a deteriorated lifestyle characterized by high energy-dense food intake coupled with a reduction in caloric expenditure stemming from new mobility-reducing technologies. As a model of lifestyle change (LiSM), our 28-year on-going longitudinal study (“Moving for Health”) has shown effectiveness in the reduction not only of obesity but especially of its comorbidities, in a (10 week to 3 year) length-dependent LiSM. However, a disappointing progressive decrease in compliance with the study has been observed and attributed to the resistance of people to change their actual “obesogenic” lifestyle, basically represented by the individuals’ demand for labor-saving technologies and convenient, affordable, palatable foods.
Full article
(This article belongs to the Special Issue Lipid Metabolism and Inflammation-Related Diseases)
►▼
Show Figures

Figure 1
Open AccessReview
Lipids Meet Immunity: Metabolic Control of cGAS-STING
by
Chui Yan Mah, Xuan Yuan Aw and Ngee Kiat Chua
Lipidology 2026, 3(1), 10; https://doi.org/10.3390/lipidology3010010 - 13 Mar 2026
Abstract
►▼
Show Figures
The field of immunometabolism highlights the intricate interplay between immunity and metabolism. The cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway is a central component of innate immunity that detects double-stranded DNA (dsDNA) from a range of sources, including pathogenic and host-derived
[...] Read more.
The field of immunometabolism highlights the intricate interplay between immunity and metabolism. The cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway is a central component of innate immunity that detects double-stranded DNA (dsDNA) from a range of sources, including pathogenic and host-derived DNA. It is now recognized that the cGAS-STING pathway has broad implications in a variety of human conditions including cancer, age-related diseases, and autoimmune disorders. Given the abundance and diversity of lipids across cellular compartments serving as structural components and signaling molecules, it is unsurprising that lipid metabolism influences the regulation of cGAS-STING signaling. Lipids can directly alter signaling protein dynamics through interactions within membrane compartments, while alterations in lipid metabolism can remodel multiple cell-intrinsic signaling cascades. Here, we summarize emerging concepts and recent discoveries that have advanced our understanding of how lipid metabolism and lipids regulate the cGAS-STING pathway.
Full article

Figure 1
Open AccessArticle
Universal Newborn Screening for Familial Hypercholesterolaemia: Insights from Expectant Parents at a Tertiary Referral Centre in Queensland, Australia
by
Caroline Bachmeier, Deborah A. Johnston, Andrew Kassianos, Karam M. Kostner and Helen Tanner
Lipidology 2026, 3(1), 9; https://doi.org/10.3390/lipidology3010009 - 8 Mar 2026
Abstract
Background: Familial hypercholesterolaemia (FH) is an inherited condition that substantially increases the risk of premature atherosclerotic cardiovascular disease (ASCVD). Despite effective therapies, many affected individuals remain undiagnosed. Incorporation of FH testing into newborn screening (NBS) is a potential strategy to increase detection. This
[...] Read more.
Background: Familial hypercholesterolaemia (FH) is an inherited condition that substantially increases the risk of premature atherosclerotic cardiovascular disease (ASCVD). Despite effective therapies, many affected individuals remain undiagnosed. Incorporation of FH testing into newborn screening (NBS) is a potential strategy to increase detection. This study explores the attitudes of expectant parents toward NBS for FH. Methods: Expectant parents attending Obstetrics and/or Obstetric Medicine clinics at a tertiary referral centre were invited to participate in a semi-structured interview in person or via telephone. The interview explored views on screening children for FH in general, preferred timing of screening, opinions on integrating FH screening into routine NBS, acceptability of genetic testing in children and newborns, willingness to engage in family screening and any potential impact on participation in routine NBS programmes. Participants were also invited to discuss concerns and benefits. Results: All participants (n = 22) supported screening children for FH with the majority (n = 19, 86%) preferring screening during the newborn period, and all but one (n = 21, 95%) supporting adding FH to routine NBS. All participants (n = 22, 100%) considered genetic testing for FH acceptable for children, 21 (95%) considered it acceptable for newborns. All participants would partake in extended family screening and none indicated that including FH genetic testing would reduce their willingness to engage in routine NBS. Concerns raised by participants included administering lipid-lowering therapy to children and potential post-diagnosis stress and anxiety. All participants reported that a diagnosis would not affect parent–child bonding. Key perceived benefits raised by participants included early diagnosis and prevention of heart disease, family screening, and broader health system and community advantages. Conclusions: Expectant parents demonstrated strong support for FH NBS and indicated no negative impact on uptake of routine NBS. Further work is needed to address parental concerns and guide implementation strategies.
Full article
Open AccessArticle
The Effect of Lipoproteins on Platelet-Associated PCSK9 of Healthy Normolipidemic Volunteers: An In Vitro Study
by
Aikaterini N. Tsouka, Ioannis K. Koutsaliaris, Constantinos Tellis and Alexandros D. Tselepis
Lipidology 2026, 3(1), 8; https://doi.org/10.3390/lipidology3010008 - 25 Feb 2026
Abstract
►▼
Show Figures
Background: Proprotein convertase subtilisin/kexin type 9 (PCSK9) promotes low-density lipoprotein receptor degradation and thereby regulates plasma LDL cholesterol levels. Although PCSK9 is primarily produced by the liver, it has been detected in platelets; however, the origin and functional relevance of platelet-associated PCSK9
[...] Read more.
Background: Proprotein convertase subtilisin/kexin type 9 (PCSK9) promotes low-density lipoprotein receptor degradation and thereby regulates plasma LDL cholesterol levels. Although PCSK9 is primarily produced by the liver, it has been detected in platelets; however, the origin and functional relevance of platelet-associated PCSK9 remain unclear. Methods: Washed platelets (WPs) were isolated from normolipidemic subjects. Endogenous platelet PCSK9 content was quantified by ELISA, and PCSK9 molecular forms were assessed by immunoblotting. The WPs were incubated with recombinant PCSK9 (recPCSK9), and platelet aggregation in response to arachidonic acid (AA) or thrombin (Thr) was evaluated. The effects of LDL- or HDL-bound PCSK9 on platelet aggregation were also examined. Results: Platelets from normolipidemic subjects contained measurable amounts of PCSK9 (0.84 ± 0.27 ng/mg protein), which increased to 2.91 ± 0.53 ng/mg protein following incubation with recPCSK9. Exposure to recPCSK9 significantly enhanced AA- and Thr-induced platelet aggregation. In contrast, LDL and HDL inhibited platelet aggregation independently of their PCSK9 content. Conclusions: Human platelets contain endogenous PCSK9 and can accumulate additional PCSK9 from the extracellular environment. Exogenous PCSK9 enhances platelet aggregation, supporting a potential prothrombotic role for circulating PCSK9 even in normolipidemic individuals. These findings provide new insight into the complex interplay between PCSK9, lipoproteins, and platelet function.
Full article

Figure 1
Open AccessReview
Environmental Factors and Lipid Metabolism in Atherosclerosis Development
by
Mikhail V. Caga-Anan, Nirodhi N. Dasanayaka and Anusha N. Seneviratne
Lipidology 2026, 3(1), 7; https://doi.org/10.3390/lipidology3010007 - 19 Feb 2026
Abstract
►▼
Show Figures
Atherosclerosis is a progressive cardiovascular disease characterized by lipid accumulation, oxidative stress, and inflammation within the arterial walls. Environmental pollutants, including particulate matter (PM), diesel exhaust particles (DEPs), and heavy metals, contribute directly to the initiation and progression of arterial plaques by promoting
[...] Read more.
Atherosclerosis is a progressive cardiovascular disease characterized by lipid accumulation, oxidative stress, and inflammation within the arterial walls. Environmental pollutants, including particulate matter (PM), diesel exhaust particles (DEPs), and heavy metals, contribute directly to the initiation and progression of arterial plaques by promoting LDL oxidation, endothelial dysfunction, foam cell formation, and vascular inflammation, whilst high-density lipoprotein (HDL) provides protective effects. This review examines the mechanistic links between environmental exposures, lipid dysregulation, and plaque formation, highlighting how both gaseous and particulate pollutants and toxic and essential metals, as well as endocrine disrupting chemicals, influence atherosclerotic risk. Potential antioxidant and lifestyle interventions to mitigate these pollutant-driven effects are also discussed.
Full article

Figure 1
Open AccessReview
Lipids and Their Role in Aging and Neurodegenerative Decline
by
Smita Lata, Sumira Malik, Sagar Mondal, Jutishna Bora, Swati Priya, Dinusha T Veettil and Perinthottathil Sreejith
Lipidology 2026, 3(1), 6; https://doi.org/10.3390/lipidology3010006 - 12 Feb 2026
Abstract
►▼
Show Figures
Lipids are a diverse group of hydrophobic molecules including fats, oils, phospholipids, and steroids that are vital for numerous biological functions including energy storage, cellular structure, and signaling whose composition and metabolism undergo profound transformations with age. These age-related shifts due to increased
[...] Read more.
Lipids are a diverse group of hydrophobic molecules including fats, oils, phospholipids, and steroids that are vital for numerous biological functions including energy storage, cellular structure, and signaling whose composition and metabolism undergo profound transformations with age. These age-related shifts due to increased lipid peroxidation, disrupted cholesterol homeostasis, and altered membrane phospholipid content, actively contribute to progressive loss in cellular homeostasis and pathogenesis of major age-related diseases. This review explores the critical role of lipids: as master regulators of cellular signaling pathways, and as key drivers of chronic inflammation and metabolic dysfunction. Dysregulated lipid metabolism is central to cardiovascular disease which is driven by altered myocardial energy substrate utilization and lipoprotein dynamics. In neurodegenerative disorders like Alzheimer’s and Parkinson’s disease, disruptions in ceramide, cholesterol, and specialized pro-resolving lipid mediators fuel neuroinflammation and protein aggregation. Furthermore, we explore the dual role of dietary lipids, which can either exacerbate or mitigate age-related decline, highlighting the potential of personalized nutritional approaches and lipid-targeting therapeutics. By integrating the mechanisms of lipid signaling, inflammation, and metabolic regulation, this analysis highlights that lipids are not merely passive structural components but active drivers of the aging process, positioning lipid metabolism as a promising frontier for interventions aimed at promoting health span and combating age-related disease.
Full article

Graphical abstract
Open AccessReview
Dysregulated Skeletal Muscle Lipid Handling Drives Myocardial Mitochondrial Dysfunction Through ASK-1 and PPARγ Signaling
by
Preyangsee Dutta and Dwaipayan Saha
Lipidology 2026, 3(1), 5; https://doi.org/10.3390/lipidology3010005 - 31 Jan 2026
Abstract
Cardiovascular disease is the leading cause of mortality in insulin-resistant individuals, with metabolic cardiomyopathy preceding overt heart failure in a substantial proportion of patients with diabetes. Skeletal muscle accounts for approximately 40% of body mass and nearly 80% of insulin-stimulated glucose disposal, positioning
[...] Read more.
Cardiovascular disease is the leading cause of mortality in insulin-resistant individuals, with metabolic cardiomyopathy preceding overt heart failure in a substantial proportion of patients with diabetes. Skeletal muscle accounts for approximately 40% of body mass and nearly 80% of insulin-stimulated glucose disposal, positioning it as a major determinant of systemic lipid flux. Dysregulation of lipid droplet dynamics, lipolysis, and fatty acid trafficking in skeletal muscle alters circulating lipid availability and promotes ectopic lipid deposition and mitochondrial stress in the myocardium. Intramyocellular lipid handling is governed by coordinated actions of lipid droplets, perilipin proteins (PLIN2 and PLIN3), adipose triglyceride lipase (ATGL), and diacylglycerol acyltransferases (DGAT1/2), which together regulate the rate and composition of fatty acid release into the circulation. Impaired coupling between intramyocellular lipid droplet turnover and mitochondrial oxidation in insulin-resistant muscle increases circulating free fatty acids, reducing cardiac oxidative capacity. In response, the myocardium undergoes mitochondrial lipid remodeling, including alterations in cardiolipin composition that impair cristae structure and electron transport chain efficiency. Excess lipid exposure activates apoptosis signal-regulating kinase-1 (ASK-1), promoting cardiomyocyte apoptosis and inflammatory signaling, while peroxisome proliferator-activated receptor gamma (PPARγ) modulates lipid uptake, storage, and mitochondrial oxidation in a context-dependent manner. This review integrates skeletal muscle–cardiac lipid crosstalk with ASK-1 and PPARγ signaling to define mechanisms linking peripheral insulin resistance to early myocardial dysfunction and to identify targets for intervention before irreversible cardiac remodeling develops.
Full article
(This article belongs to the Special Issue Lipid Metabolism and Inflammation-Related Diseases)
►▼
Show Figures
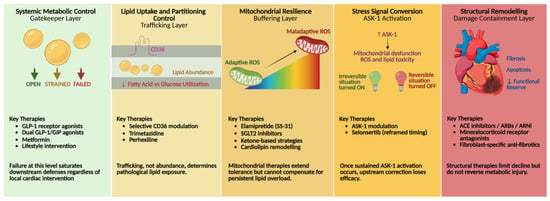
Figure 1
Open AccessArticle
Evaluating the Effects of Full-Fat Yogurt Consumption on Circulating Inflammatory Biomarkers and Ex Vivo Peripheral Blood Mononuclear Cell Inflammatory Responses in a Randomized-Controlled Crossover Trial
by
Victoria M. Taormina, Simonne Eisenhardt, Matthew P. Gilbert, C. Lawrence Kien, Matthew E. Poynter and Jana Kraft
Lipidology 2026, 3(1), 4; https://doi.org/10.3390/lipidology3010004 - 15 Jan 2026
Abstract
Chronic, low-grade inflammation is a characteristic of metabolic diseases like type 2 diabetes. Despite recommendations to select low- or non-fat dairy foods over full-fat dairy foods for metabolic health, recent research suggests potential anti-inflammatory benefits of dairy fat consumption. We aimed to compare
[...] Read more.
Chronic, low-grade inflammation is a characteristic of metabolic diseases like type 2 diabetes. Despite recommendations to select low- or non-fat dairy foods over full-fat dairy foods for metabolic health, recent research suggests potential anti-inflammatory benefits of dairy fat consumption. We aimed to compare the systemic inflammatory tone (i.e., circulating inflammatory biomarker concentrations and ex vivo peripheral blood mononuclear cell inflammatory responses) of individuals with prediabetes after consuming diets with full-fat (3.25%) or non-fat yogurt. We hypothesized that short-term consumption of three daily full-fat yogurt servings beneficially affects inflammatory tone. Thirteen participants aged 45–75 years completed an eight-week randomized, double-masked, controlled crossover study. The two, three-week experimental diets comprised three daily servings of full-fat or non-fat yogurt and were each preceded by a one-week run-in diet. Following each diet, circulating inflammatory biomarkers and cytokine concentrations in the supernatants of peripheral blood mononuclear cells under control or lipopolysaccharide-stimulated conditions were measured. Compared with non-fat yogurt intake, circulating immature granulocyte concentrations were lower following full-fat yogurt intake, but there were no other differences in leukocyte concentrations. Circulating concentrations of cytokines or other inflammatory markers did not differ by diet. Cell supernatant interleukin-1β concentrations were lower following the full-fat yogurt diet under unstimulated conditions but were not different between diets under stimulated conditions. There were no differences by diet in supernatant concentrations of other cytokines under unstimulated or stimulated conditions. Together, minimal differences in inflammatory tone were observed following the short-term consumption of three daily servings of full-fat or non-fat yogurt in individuals with prediabetes.
Full article
(This article belongs to the Special Issue Lipid Metabolism and Inflammation-Related Diseases)
►▼
Show Figures

Graphical abstract
Open AccessReview
Lipid Analysis by Thin-Layer Chromatography—Detection, Staining and Derivatization
by
Johanna W. Schubarth, Jenny Leopold, Kathrin M. Engel and Jürgen Schiller
Lipidology 2026, 3(1), 3; https://doi.org/10.3390/lipidology3010003 - 13 Jan 2026
Abstract
►▼
Show Figures
Thin-layer chromatography (TLC) remains a widely used, cost-effective and convenient method to separate small molecules, particularly in the field of natural products and (phospho)lipids. Despite advances in chromatographic methods such as high-performance liquid chromatography (HPLC), TLC retains several advantages, including simplicity and accessibility.
[...] Read more.
Thin-layer chromatography (TLC) remains a widely used, cost-effective and convenient method to separate small molecules, particularly in the field of natural products and (phospho)lipids. Despite advances in chromatographic methods such as high-performance liquid chromatography (HPLC), TLC retains several advantages, including simplicity and accessibility. However, a critical step is the visualization of the separated lipids on the TLC plate. Although the majority of the regularly used methods were established decades ago, there are still a number of potential pitfalls and widely unknown aspects. This review provides a concise overview about commonly used stationary phases and the solvent systems in TLC analysis of lipids. The main focus is on visualization techniques, spanning from non-specific, destructive (charring by semi-concentrated acids) to specific, non-destructive approaches (e.g., exposition to iodine to monitor unsaturated lipids). The advantages and disadvantages of the different methods will be critically discussed and frequently occurring problems highlighted. Furthermore, the combination of TLC with mass spectrometry (MS) detection will be introduced, covering both extraction-based electrospray ionization MS techniques as well as desorption techniques such as matrix-assisted laser desorption/ionization MS. MS detection, while generally more sensitive and offering molecular specificity, introduces higher technical and financial requirements compared to conventional staining. Nonetheless, the combination of TLC with MS holds significant potential for enhancing lipidomic workflows, particularly in complex biological samples.
Full article

Graphical abstract
Open AccessReview
Omega-3 Fatty Acids and Mood Disorders: A Critical Narrative Review
by
Mara Lastretti, Lorenzo Campedelli, Tiziano Scarparo, Sabina Spagna, Andrea Cicoli, Gavino Faa and Ettore D’Aleo
Lipidology 2026, 3(1), 2; https://doi.org/10.3390/lipidology3010002 - 3 Jan 2026
Abstract
Background: In recent decades, interest has grown in the link between lipid metabolism and mental health. Omega-3 fatty acids (EPA and DHA) play a crucial role in brain function, neuroplasticity, and emotional regulation. Deficiency or an imbalanced omega-6/omega-3 ratio, typical of Western diets,
[...] Read more.
Background: In recent decades, interest has grown in the link between lipid metabolism and mental health. Omega-3 fatty acids (EPA and DHA) play a crucial role in brain function, neuroplasticity, and emotional regulation. Deficiency or an imbalanced omega-6/omega-3 ratio, typical of Western diets, has been associated with an increased risk of mood disorders, such as major depression and bipolar disorder. Objective: This review critically synthesizes neurobiological and clinical evidence on the role of omega-3s in mood disorders, assessing mechanisms, randomized controlled trials, and meta-analyses, with a focus on inflammatory biomarkers and their integration into a personalized psychiatry framework. Methods: A narrative review of epidemiological studies, randomized controlled trials (RCTs), meta-analyses, and experimental research was conducted, evaluating mechanistic pathways (synaptic, inflammatory, neuroplastic, and redox) and clinical moderators (particularly inflammatory markers and dietary lipid profiles). Results: Consistent evidence indicates that EPA-enriched formulations (≥60%) exert antidepressant effects, particularly in patients with elevated inflammatory markers (CRP, IL-6, TNF-α). In contrast, DHA-only preparations show limited efficacy, and outcomes vary depending on dietary omega-6/omega-3 balance. However, the literature remains heterogeneous in design, sample size, and biomarker stratification, which limits generalizability and contributes to conflicting findings across studies. Conclusions: While omega-3 fatty acids represent promising adjunctive interventions for mood disorders, methodological weaknesses and inconsistent formulations hinder definitive conclusions. Future research should prioritize biomarker-guided, precision-based approaches to clarify therapeutic efficacy and optimize clinical use.
Full article
Open AccessArticle
Fatty Acids Differentially Induce Lipid Droplet Formation in HeLa Cells
by
Jacob J. Adler
Lipidology 2026, 3(1), 1; https://doi.org/10.3390/lipidology3010001 - 30 Dec 2025
Abstract
►▼
Show Figures
Background/Objectives: Long-chain fatty acids induce lipid droplet formation in several cell types including cancer cells. These lipid droplets have been shown to accumulate in various cancers and are dysregulated in many pathologies. Thus, this study was designed to examine the many unique
[...] Read more.
Background/Objectives: Long-chain fatty acids induce lipid droplet formation in several cell types including cancer cells. These lipid droplets have been shown to accumulate in various cancers and are dysregulated in many pathologies. Thus, this study was designed to examine the many unique long-chain fatty acids and their abilities to induce lipid droplet formation in cancer cells. Methods: HeLa human cervical cancer cells were incubated with individual fatty acids and live-stained for lipid droplets. This study analyzed four saturated, four monounsaturated, and nine polyunsaturated (4 omega-3, 4 omega-6, and 1 omega-9) fatty acids. This diversity of fatty acids was chosen to highlight any important non-uniform differences in the regulation of lipid droplet formation by unsaturated fatty acids. The area of the lipid droplets and the number of lipid droplets per cell were measured and compared between the different fatty acid conditions. Results: Unsaturated fatty acids induced lipid droplets differently compared to saturated fatty acids. Further, an inverse relationship was established between average area of lipid droplets and the average number of lipid droplets per cell. Finally, two perilipin genes (PLIN1/2) involved in lipid droplet formation were shown to have significantly higher expression with the two polyunsaturated fatty acids (alpha- and gamma-linolenic acid) versus the saturated fatty acid (stearic acid) condition. Conclusions: Together, different fatty acids produce structurally different lipid droplets. It will be important to further investigate the biochemistry and mechanistic differences in the formation of these lipid droplets under these specific long-chain fatty acid conditions.
Full article

Figure 1
Open AccessReview
Liposomes as “Trojan Horses” in Cancer Treatment: Design, Development, and Clinical Applications
by
Juan Sabín, Andrea Santisteban-Veiga, Alba Costa-Santos, Óscar Abelenda and Vicente Domínguez-Arca
Lipidology 2025, 2(4), 25; https://doi.org/10.3390/lipidology2040025 - 8 Dec 2025
Abstract
►▼
Show Figures
Liposomes started to be studied for drug delivery in 1970s, taking advantage of their ability to encapsulate hydrophilic and hydrophobic drugs using biodegradable and biocompatible molecules. Nowadays, they remain one of the most promising strategies for drug delivery not only in cancer treatment
[...] Read more.
Liposomes started to be studied for drug delivery in 1970s, taking advantage of their ability to encapsulate hydrophilic and hydrophobic drugs using biodegradable and biocompatible molecules. Nowadays, they remain one of the most promising strategies for drug delivery not only in cancer treatment but also in gene therapies and vaccines. The design and development of liposomal systems have evolved significantly over the past decades, moving from conventional formulations to advanced, stimulus-responsive, and multifunctional nanocarriers. Analogous to the myth of the Trojan Horse, liposomes must mislead the host immune system to reach the interior of cancer cells in order to deliver the therapeutic payload. There are many barriers that liposomes have to overcome to circulate through the bloodstream and specifically target cancer cells without damaging other tissues. Crucial parameters such as lipid composition, particle size, zeta potential, and PEGylation have been systematically optimized to enhance pharmacokinetics and biodistribution and to improve delivery efficiency. Furthermore, conjugation with antibodies, peptides, or small molecules has enabled active targeting, while stimuli such as pH, temperature, and enzymatic activity have been exploited for controlled drug release within the tumor microenvironment. Such innovations have laid the groundwork for translating liposomal formulations from the bench to clinical applications. In this paper, we evaluate the physicochemical features of liposomal design that underpin their suitability and efficacy for anticancer drug delivery. We aimed to focus on two main aspects: conducting an exhaustive review of the physicochemical parameters of liposomal drugs that have already been approved by regulatory agencies, while maintaining a pedagogical approach when explaining the key design parameters for the optimal design of liposomes in oncology in detail.
Full article
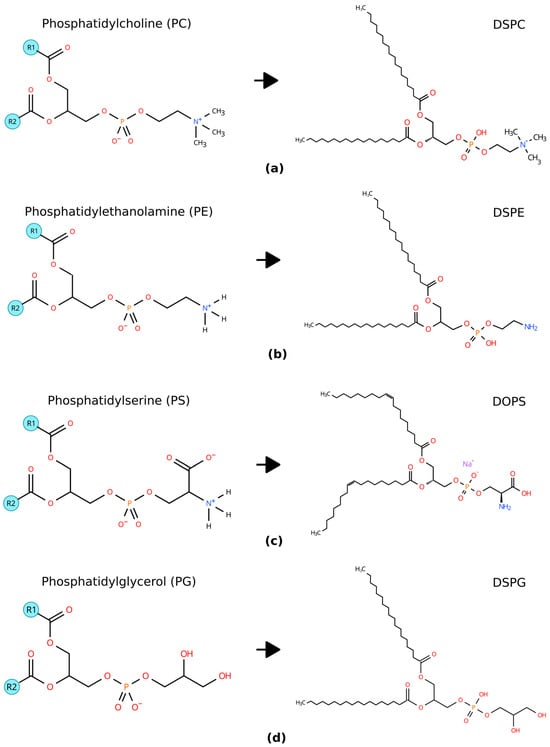
Figure 1
Open AccessArticle
Milk Fatty Acid Profiling as a Tool for Estimating Methane Emissions in Conventionally Fed Dairy Cows
by
Emily C. Youngmark and Jana Kraft
Lipidology 2025, 2(4), 24; https://doi.org/10.3390/lipidology2040024 - 2 Dec 2025
Abstract
►▼
Show Figures
Milk fatty acid (FA) synthesis and enteric methanogenesis share common biochemical pathways related to rumen fermentation patterns and microbial volatile FA production. The FA profile of milk is known to correlate with methane (CH4) emissions; thus, FA profiling has been proposed
[...] Read more.
Milk fatty acid (FA) synthesis and enteric methanogenesis share common biochemical pathways related to rumen fermentation patterns and microbial volatile FA production. The FA profile of milk is known to correlate with methane (CH4) emissions; thus, FA profiling has been proposed as an indirect method to predict CH4 emissions from dairy cattle. This study aimed to (1) investigate the milk FA profiles of Holstein cows to identify candidate biomarkers for predicting CH4 output (g/d), CH4 yield (g/kg dry matter intake), and CH4 intensity (g/kg energy-corrected milk), and (2) develop and compare regression models predicting CH4 emissions. Forty-eight cows, fed industry standard diets, were enrolled in an exploratory trial. Milk samples and CH4 measurements were collected thrice per day, and intake was recorded daily. Milk lipids were extracted, transesterified, and subsequently analyzed via gas–liquid chromatography. Three penalized regression models were compared for predicting CH4 emission metrics using milk FAs and management variables. Methane emission metrics corelated positively with short- and medium-chain FAs, polyunsaturated FAs, and branched-chain FAs, while monounsaturated FAs correlated negatively. Notably, this study observed novel correlations between 11-cyclohexyl-11:0; and 20:3 c5,c8,c11 and CH4 metrics (|r| = 0.58–0.79). Across all CH4 metrics, the models demonstrated high predictive accuracy (R2 = 0.71–0.87; concordance correlation coefficient = 0.83–0.93). The findings of this study indicate that milk FA profiling may be an effective method to detect CH4 emissions from cows fed industry standard diets and highlight the need for further refinement of prediction models.
Full article

Graphical abstract
Open AccessCommunication
Fatty Acid Composition and Lipid Oxidation in Plant-Based Meat Analogue Chicken Schnitzels Under Different Cooking Conditions
by
Owen Miller, Christopher J. Scarlett and Taiwo O. Akanbi
Lipidology 2025, 2(4), 23; https://doi.org/10.3390/lipidology2040023 - 25 Nov 2025
Abstract
►▼
Show Figures
Background/objectives: Plant-based meat analogues (PBMAs) are designed to mimic meat products and to be cooked under similar conditions by consumers. There have been few studies into the lipid stability of PBMAs, and no published studies have investigated the effect of cooking on the
[...] Read more.
Background/objectives: Plant-based meat analogues (PBMAs) are designed to mimic meat products and to be cooked under similar conditions by consumers. There have been few studies into the lipid stability of PBMAs, and no published studies have investigated the effect of cooking on the lipid stability of PBMAs. Methods: This study analysed the effect of recommended cooking conditions on the lipid oxidation of three commercial chicken schnitzel PBMAs with differing fatty acid composition. Fatty acids and lipid classes were analysed using gas chromatography (GC) and capillary chromatography (Iatroscan) with flame ionisation detectors, respectively. Lipid oxidation was analysed using multiple tests, including peroxide value (POV), p-Anisidine value, acid value, and thiobarbituric acid reactive substance (TBARS) tests, which then allowed for the total oxidation (TOTOX) to be calculated. Results: Fatty acid analysis by GC showed different levels of saturated and unsaturated fatty acid contents in all PBMAs, with oleic acid (C18:1) being the most abundant (product A = 52%; product B = 62%; product C = 37%). Meanwhile, lipid class analyses by Iatroscan revealed that the oils used in the PBMAs were composed of triacylglycerol (TAG), which remained intact after cooking. Lipid oxidation tests showed no major increases between the raw and cooked PBMA. Also, the TOTOX values for each product did not increase significantly (p < 0.05) due to cooking (TOTOX values for raw/cooked product A = 9.36/9.99; product B = 5.88/6.19; product C = 11.31/11.92), suggesting a broad stability of the lipids. Conclusions: Therefore, if the on-package cooking instructions are followed for these three PBMA products, their lipid oxidation levels remain within safe limits.
Full article
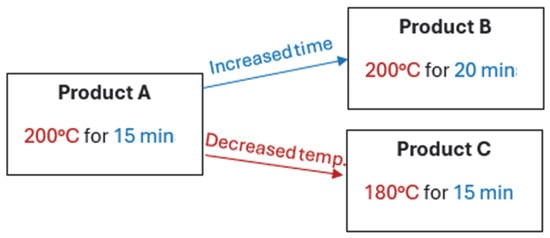
Figure 1
Open AccessArticle
Glucose-6-Phosphate Dehydrogenase Deficiency and Cardiovascular Risk in Familial Hypercholesterolemia: A Retrospective Cohort Study
by
Alessandra Errigo, Maria Pina Dore, Elettra Merola and Giovanni Mario Pes
Lipidology 2025, 2(4), 22; https://doi.org/10.3390/lipidology2040022 - 11 Nov 2025
Abstract
►▼
Show Figures
Background: Familial hypercholesterolemia (FH) is a monogenic disorder causing markedly elevated low-density lipoprotein cholesterol (LDL-C) and premature atherosclerosis. Glucose-6-phosphate dehydrogenase (G6PD) is a key enzyme in antioxidant defense via NADPH production. G6PD deficiency, an X-linked disorder impairing redox homeostasis, may contribute to cardiovascular
[...] Read more.
Background: Familial hypercholesterolemia (FH) is a monogenic disorder causing markedly elevated low-density lipoprotein cholesterol (LDL-C) and premature atherosclerosis. Glucose-6-phosphate dehydrogenase (G6PD) is a key enzyme in antioxidant defense via NADPH production. G6PD deficiency, an X-linked disorder impairing redox homeostasis, may contribute to cardiovascular disease (CVD) risk. This study examined whether G6PD deficiency increases CVD risk in FH patients. Methods: We retrospectively analyzed 217 FH patients. Clinical data included demographics, lipid profiles, G6PD status, and atherosclerotic CVD outcomes (coronary, cerebrovascular, or peripheral arterial disease). In a subset, FH was confirmed by LDLR gene sequencing, and G6PD Mediterranean and Seattle variants were genotyped. Cumulative CVD prevalence was compared between G6PD-deficient and G6PD-normal FH patients. Multivariable logistic regression was adjusted for age, sex, body mass index, high blood pressure, and smoking. Results: Participants (mean age 47 years, 60% female) had markedly elevated LDL-C (mean 292 mg/dL at diagnosis). Atherosclerotic CVD was present in 119 (55%) patients. G6PD-deficient FH patients had a significantly higher CVD prevalence than those with normal G6PD activity (77.4% vs. 39.8%, p < 0.0001). LDL-C levels were higher in the G6PD-deficient group than in the non-deficient group, and this difference reached statistical significance in the univariate analysis. In the multivariable analysis, G6PD deficiency remained an independent CVD predictor (adjusted OR 3.57, 95% CI 1.30–9.83) after controlling for conventional risk factors. Conclusions: In FH, hereditary G6PD deficiency is associated with a markedly increased risk of atherosclerotic CVD. A pro-oxidative state in G6PD-deficient FH patients may play a role in premature atherogenesis. G6PD status may represent a cardiovascular risk modifier in FH, warranting further research into underlying mechanisms and targeted management.
Full article

Graphical abstract
Open AccessReview
Edible Oils from Health to Sustainability: Influence of the Production Processes in the Quality, Consumption Benefits and Risks
by
Viviane de Souza Silva, Luna Valentina Angulo Arias, Franciane Colares Souza Usberti, Rafael Augustus de Oliveira and Farayde Matta Fakhouri
Lipidology 2025, 2(4), 21; https://doi.org/10.3390/lipidology2040021 - 10 Nov 2025
Cited by 1
Abstract
►▼
Show Figures
This systematic review examines the impact of vegetable oil consumption, such as extra virgin olive, olive, soybean, palm olein, corn, and camellia seed oils, on both human and animal health, alongside factors influencing oil quality and safety. A variety of studies were included,
[...] Read more.
This systematic review examines the impact of vegetable oil consumption, such as extra virgin olive, olive, soybean, palm olein, corn, and camellia seed oils, on both human and animal health, alongside factors influencing oil quality and safety. A variety of studies were included, such as clinical trials, cohort studies, animal experiments, and reports on production methods and contamination (polycyclic aromatic hydrocarbons (PAHs) and heavy metals). The literature search was performed in scientific databases indexed up to September 2025, and risk of bias was assessed using specific tools appropriate for each study design. The findings suggest that, among the oils studied, extra virgin olive oil showed the most consistent benefits, including improvements in endothelial function, lipid profiles, weight management, and reduced mortality in humans. Animal studies corroborate hepatoprotective effects and weight regulation benefits. Oil quality is influenced by genetic, environmental, and technological factors, including cultivar selection, pollination, post-harvest handling, and extraction techniques (cold, hot, DIC, MFA, encapsulation) and refining processes, which can affect both nutritional benefits and contaminant levels. Although contaminants such as PAHs and heavy metals (Cd, Pb, As) are generally below regulatory thresholds, some contexts may present health risks. High-quality vegetable oils confer cardiovascular, metabolic, and hepatic benefits. However, their contribution to public health relies on strict production practices, continuous monitoring of contaminants, and the implementation of technological innovations to ensure both safety and optimal health outcomes.
Full article

Figure 1
Open AccessReview
ATP Citrate Lyase in Lipid Metabolism: Comparative Insights Across Eukaryotes with Emphasis on Yarrowia lipolytica
by
Stylianos Fakas and Ayodeji Odunsi
Lipidology 2025, 2(4), 20; https://doi.org/10.3390/lipidology2040020 - 24 Oct 2025
Cited by 1
Abstract
►▼
Show Figures
ATP citrate lyase (ACL) is a highly conserved enzyme across eukaryotes that catalyzes the generation of cytosolic acetyl-CoA from citrate—a pivotal step linking central carbon metabolism to lipid biosynthesis. In the oleaginous yeast Yarrowia lipolytica, ACL is encoded by two genes, ACL1
[...] Read more.
ATP citrate lyase (ACL) is a highly conserved enzyme across eukaryotes that catalyzes the generation of cytosolic acetyl-CoA from citrate—a pivotal step linking central carbon metabolism to lipid biosynthesis. In the oleaginous yeast Yarrowia lipolytica, ACL is encoded by two genes, ACL1 and ACL2, forming a heteromeric complex that mirrors the multidomain architecture of the single-chain ACL enzymes found in mammals and plants. This conservation of catalytic architecture reflects a shared catalytic strategy across kingdoms, underscoring ACL’s fundamental role in metabolic integration. In Y. lipolytica, ACL is essential for directing mitochondrial citrate toward acetyl-CoA production and subsequent lipid accumulation. Yet, in contrast to well-characterized ACLs in animals and plants, the functional mechanisms and regulation of yeast ACL remain incompletely understood. A deeper understanding of ACL in Y. lipolytica offers not only evolutionary insights but also potential avenues for engineering lipid overproduction in microbial systems.
Full article

Graphical abstract
Open AccessReview
The Crosstalk Between Non-Coding RNAs and Lipid Metabolism in Chronic Disease Progression
by
Zoofa Zayani, Arash Matinahmadi, Alireza Tavakolpournegari, Seyedeh Safoora Moosavi and Seyed Hesamoddin Bidooki
Lipidology 2025, 2(4), 19; https://doi.org/10.3390/lipidology2040019 - 21 Oct 2025
Abstract
►▼
Show Figures
In the last twenty years, an increasing volume of research has characterized lipids as dynamic signaling molecules that play essential roles in various physiological and pathological processes, especially concerning chronic diseases such as cardiovascular disorders, diabetes, liver disease, neurodegeneration, cancer, obesity, diabetic and
[...] Read more.
In the last twenty years, an increasing volume of research has characterized lipids as dynamic signaling molecules that play essential roles in various physiological and pathological processes, especially concerning chronic diseases such as cardiovascular disorders, diabetes, liver disease, neurodegeneration, cancer, obesity, diabetic and chronic kidney diseases and atherosclerosis. Dysregulation of lipid synthesis and storage, lipolysis, fatty acid oxidation, lipid signaling pathways, and organelle-specific lipid modifications, including mitochondrial phospholipid remodeling and endoplasmic reticulum stress induced by saturated fatty acids, are recognized as contributors to the initiation and progression of this pathogenesis. Concurrently with the increasing comprehension of lipid metabolism, the last decade has seen progress in the understanding of genome control, especially with non-coding RNAs (ncRNAs). MicroRNAs, long non-coding RNAs, and circular RNAs, as ncRNAs, are essential modulators of gene expression at the epigenetic, transcriptional, and post-transcriptional levels that affect a number of lipid metabolism-related processes, such as fatty acid synthesis and oxidation, cholesterol homeostasis, and lipid droplet dynamics. Therapeutically, ncRNAs hold considerable promise owing to their tissue specificity and modularity, with antisense oligonucleotides and CRISPR-based editing currently under preclinical evaluation. In this context, we review recent studies exploring the interplay between ncRNAs and the regulatory networks governing lipid metabolism, and how disruptions in these networks contribute to chronic disease. This emerging paradigm underscores the role of ncRNA–lipid metabolism interactions as central nodes in metabolic and inflammatory pathways, highlighting the need for a holistic approach to therapeutic targeting.
Full article

Figure 1
Open AccessArticle
Human Monocyte-Derived Macrophages Acquire an Inflammatory Phenotype Relative to Risk Factors Typical of Atherogenic Dyslipidaemia
by
Corinne D. Mack, Lily D. Quagliata, Rana Baraz, Sravanthi Naralashetty, Suat Dervish, Helen Williams, Stephen C. H. Li and Heather J. Medbury
Lipidology 2025, 2(4), 18; https://doi.org/10.3390/lipidology2040018 - 17 Oct 2025
Abstract
Background: Dyslipidaemia promotes atherosclerotic plaque formation. Plaques that are vulnerable to rupture have a higher proportion of inflammatory (M1:CD86) macrophages in their cap. Many plaque macrophages are derived from blood monocytes which have been exposed to elevated blood lipid levels. Here, we explored
[...] Read more.
Background: Dyslipidaemia promotes atherosclerotic plaque formation. Plaques that are vulnerable to rupture have a higher proportion of inflammatory (M1:CD86) macrophages in their cap. Many plaque macrophages are derived from blood monocytes which have been exposed to elevated blood lipid levels. Here, we explored whether the inflammatory state of monocyte-derived macrophages is associated with blood lipid levels and assessed whether oxidised low-density lipoprotein (oxLDL) directly induces some of the observed changes. Method: Blood was collected from 20 individuals. Lipid profiles were measured, and monocytes differentiated into macrophages. Macrophage inflammatory state was assessed by flow cytometry for phenotypic markers (e.g., CD86 and CD163) and cytokine production: TNF, IL-1β, and IL-6. Furthermore, monocytes were isolated from 6 normo-lipidaemic individuals and cultured with oxLDL, followed by stimulation with LPS/IFNγ and assessment of the cytokine response. Results: The inflammatory phenotype acquired by macrophages (ex vivo) was related to levels of in vivo circulating lipids. Correlations for CD86/CD163 were found with CVD risk markers; most strongly with triglycerides (TG) and TG/HDL-C, but also with cholesterol/HDL-C and ApoB/ApoA1 and inversely with LDL particle size. Functionally, macrophage production of inflammatory cytokines (TNF and IL-1β) correlated with oxLDL levels and inversely with ApoA1. Macrophages differentiated from monocytes cultured with oxLDL produced significantly higher IL-1β but lower IL-10 (in response to LPS/IFNγ), compared to control cells. Conclusions: Monocyte-derived macrophages adopt an inflammatory phenotype relative to the levels of circulating lipid factors that are characteristic of atherogenic dyslipidaemia (such as high TG, TG/HDL-C and low LDL particle size), but not LDL-C.
Full article
(This article belongs to the Special Issue Lipid Metabolism and Inflammation-Related Diseases)
►▼
Show Figures

Graphical abstract
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
BioChem, Biomedicines, Foods, Metabolites, Lipidology, CIMB, Sci
Metabolomics in Biomedicine and Food Science
Topic Editors: Leonardo Tenori, Stefano CacciatoreDeadline: 15 June 2027

Conferences
Special Issues
Special Issue in
Lipidology
Lipid Metabolism and Inflammation-Related Diseases
Guest Editors: Maria Pia Adorni, Bianca PapottiDeadline: 31 May 2026







