Journal Description
Cardiogenetics
Cardiogenetics
is an international, peer-reviewed, open access journal, published quarterly online by MDPI (from Volume 10, Issue 2 - 2020).
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus, Embase, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 23.1 days after submission; acceptance to publication is undertaken in 6.2 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Impact Factor:
0.9 (2024)
Latest Articles
Beyond Wall Thickness: Clinical Predictors of Genotype Positivity in Hypertrophic Cardiomyopathy
Cardiogenetics 2026, 16(2), 10; https://doi.org/10.3390/cardiogenetics16020010 - 11 May 2026
Abstract
Background: Genetic testing in hypertrophic cardiomyopathy (HCM) yields variable positivity rates. Identifying clinical predictors of positive genetic tests could improve pre-test counseling and refine expectations about diagnostic yield. Methods: We analyzed consecutive genotyped HCM probands from a contemporary multicenter cohort across four Italian
[...] Read more.
Background: Genetic testing in hypertrophic cardiomyopathy (HCM) yields variable positivity rates. Identifying clinical predictors of positive genetic tests could improve pre-test counseling and refine expectations about diagnostic yield. Methods: We analyzed consecutive genotyped HCM probands from a contemporary multicenter cohort across four Italian tertiary centers. Genotype positivity was defined as the presence of ≥1 pathogenic or likely pathogenic variant (ACMG classes 4–5). Multivariable logistic regression identified predictors of genotype positivity. Sensitivity analyses assessed the incremental value of left atrial volume index (LAVI) ≥ 34 mL/m2 and the mode of first clinical presentation. Results: Among 274 genotyped probands (median age at diagnosis 54 years; 62% male), 86 (31%) were genotype-positive (38% MYBPC3, 29% MYH7). Age at diagnosis <40 years (OR 2.38, 95%CI 1.26–4.51, p = 0.008), family history of sudden cardiac death/major ventricular arrhythmias (OR 2.34, 95%CI 1.16–4.84, p = 0.019) and family history of non-ischemic cardiomyopathy (OR 1.92, 95%CI 1.04–3.54, p = 0.038), were independently associated with genotype positivity whereas arterial hypertension was inversely associated (OR 0.42, 95%CI 0.23–0.77). Maximal left ventricular wall thickness > 20 mm and gender were not predictive of genotype positivity. Inclusion of LAVI modestly improved the model performance (AUC 0.769, p = 0.016, ΔAUC +0.024; DeLong p = 0.016) but without leading to meaningful patient reclassification. Conclusions: Genotype positivity in HCM links to earlier onset and family history; traditional severity markers and initial presentation may not independently suggest genetic causality. These findings may help shape a personalized approach to genetic counseling in HCM.
Full article
(This article belongs to the Special Issue Contemporary and Future Approaches to Inherited Cardiomyopathies)
►
Show Figures
Open AccessArticle
Whole-Genome Sequencing in Premature Coronary Artery Disease in South Asians: A Pilot Case–Control Study
by
Iftikhar Ali Ch, Azhar Chaudhry, Fazal Jalil, Yasir Ali, Waseem Iqbal, Yusra Javed, Salman Khalid, Azeen Razzaq, Muhammad Azhar, Amna Nadeem, Tayyab Afzal, Naeem Tahirkheli, Ankur Kalra and Khurram Nasir
Cardiogenetics 2026, 16(2), 9; https://doi.org/10.3390/cardiogenetics16020009 - 29 Apr 2026
Abstract
Background/Objectives: Coronary artery disease (CAD) remains the leading cause of mortality worldwide, with South Asia bearing a disproportionately high and rising burden, particularly at younger ages. The present study aimed to investigate genetic variants associated with premature coronary artery disease (PCAD) using whole-genome
[...] Read more.
Background/Objectives: Coronary artery disease (CAD) remains the leading cause of mortality worldwide, with South Asia bearing a disproportionately high and rising burden, particularly at younger ages. The present study aimed to investigate genetic variants associated with premature coronary artery disease (PCAD) using whole-genome sequencing (WGS). Methods: WGS was conducted on 12 people (five PCAD cases, seven matched controls) to assess feasibility and methodology for future large-scale research. High-quality genomic DNA was sequenced at a minimum read depth of 10× with a quality threshold of Q30. Variant calling with stringent quality control identified single-nucleotide polymorphisms (SNPs), followed by annotation against gnomAD for allele frequencies and ClinVar for pathogenicity. Protein-coding variants were filtered, and candidate genes were prioritized for comparative analysis between cases and controls. Results: An average of over 8.8 million SNPs per individual was identified, with comparable overall variant distributions between cases and controls. Initial analyses revealed 120 SNPs exclusively present in PCAD cases. All protein-coding variants were rare (allele frequency < 0.0001), and none were previously classified as pathogenic in ClinVar. After filtration, 87 candidate genes were prioritized. Enriched or unique variants in PCAD cases are mapped to genes involved in lipid metabolism, endothelial dysfunction, inflammatory signaling, immune regulation, thrombosis, vascular remodeling, and metabolic processes. Additional variants were identified in genes related to smooth muscle proliferation, oxidative stress, and other biological pathways. Conclusions: This WGS pilot study provides an initial overview of the genomic landscape of PCAD in a South Asian cohort, highlighting rare variants across multiple biological pathways implicated in atherosclerosis that need validation in a large-scale study.
Full article
(This article belongs to the Topic Biomarkers in Cardiovascular Disease—Chances and Risks, 2nd Volume)
►▼
Show Figures

Graphical abstract
Open AccessReview
Genetic Basis of Cardiomyopathies Associated with Endocrinopathies: A Comprehensive Review
by
Antonio Concistrè, Claudia Caramazza, Marco D’Abbondanza, Rachele Santori and Giuseppe Imperoli
Cardiogenetics 2026, 16(2), 8; https://doi.org/10.3390/cardiogenetics16020008 - 7 Apr 2026
Abstract
Endocrine disorders are increasingly recognized as major contributors to secondary cardiomyopathies, leading to profound alterations in cardiac structure and function. This comprehensive review synthesizes current evidence on the genetic basis of cardiomyopathies associated with endocrine conditions, including primary aldosteronism, Cushing’s syndrome, pheochromocytoma/paraganglioma, acromegaly,
[...] Read more.
Endocrine disorders are increasingly recognized as major contributors to secondary cardiomyopathies, leading to profound alterations in cardiac structure and function. This comprehensive review synthesizes current evidence on the genetic basis of cardiomyopathies associated with endocrine conditions, including primary aldosteronism, Cushing’s syndrome, pheochromocytoma/paraganglioma, acromegaly, thyroid disorders, hyperparathyroidism, and diabetic cardiomyopathy. We examine the contribution of somatic and germline mutations, genetic polymorphisms, shared molecular pathways transforming growth factor-β (TGF-β)/SMAD (TGF-β/SMAD signaling, the renin–angiotensin–aldosterone system, oxidative stress, and calcium handling), sarcomeric gene modifiers, ion channel variants, and epigenetic mechanisms to disease pathogenesis. We propose a conceptual framework distinguishing three major categories of genetic involvement: (i) variants causing the primary endocrinopathy; (ii) genetic modifiers of myocardial susceptibility under conditions of hormonal excess; and (iii) direct pleiotropic effects, whereby single gene variants independently cause both endocrine and cardiac phenotypes. In addition, we discuss genotype–phenotype correlations, ethnic and population differences in genetic susceptibility, the emerging role of polygenic risk scores, and precision medicine approaches. Overall, this review provides an integrated perspective on the complex genetic architecture of endocrine-related cardiomyopathies and outlines practical considerations for genetic testing aimed at improving patient management and clinical outcomes.
Full article
(This article belongs to the Section Cardiovascular Genetics in Clinical Practice)
►▼
Show Figures

Figure 1
Open AccessArticle
Report on the Post-Translational Modifications (PTMs) Prediction in Hypertrophic Cardiomyopathy-Associated Proteins MYH7, MYBPC3, TNNT2, and TNNI3, and Five Unknown PTMs in MYH7 (K129, K1451) and MYBPC3 (K14, R44, T705)
by
Natasha Trajkovska, Lenche Jovova and Done Stojanov
Cardiogenetics 2026, 16(2), 7; https://doi.org/10.3390/cardiogenetics16020007 - 2 Apr 2026
Abstract
In this study, we have performed computational PTM analysis on a panel of hypertrophic cardiomyopathy (HCM)-associated proteins: MYH7, MYBPC3, TNNT2, and TNNI3. We aimed to benchmark the prediction of PTM sites of three ML-based tools: MusiteDeep, PTMGPT2, and SiteTack, using PhosphoSitePlus as a
[...] Read more.
In this study, we have performed computational PTM analysis on a panel of hypertrophic cardiomyopathy (HCM)-associated proteins: MYH7, MYBPC3, TNNT2, and TNNI3. We aimed to benchmark the prediction of PTM sites of three ML-based tools: MusiteDeep, PTMGPT2, and SiteTack, using PhosphoSitePlus as a reference for true positives. Notably, because the highest precision tool varied by protein and PTM type, our results indicate there is no single best tool for PTM prediction. Specifically, for HCM-associated proteins, MusiteDeep had the highest precision for MYBPC3 and MYH7; PTMGPT2 was best for TNNI3, and SiteTack for TNNT2. Examining PTM type and phosphorylation in particular, MusiteDeep had the highest precision, followed by PTMGPT2 and SiteTack. However, MusiteDeep did not identify acetylation sites, where PTMGPT2 outperformed SiteTack. Beyond these benchmarking results, we also report on five high-priority candidates for experimental validation in two HCM-associated proteins: MYH7 (K1451 acetylation, K129 methylation) and MYBPC3 (T705 phosphorylation, K14 acetylation, R44 methylation).
Full article
(This article belongs to the Section Molecular Genetics)
►▼
Show Figures

Figure 1
Open AccessReview
The Genetic Architecture of Sudden Cardiac Death: A State-of-the-Art Review
by
Sabrina Montuoro, Emanuele Monda, Gaetano Diana, Emanuele Bobbio, Vera Fico, Marta Rubino, Martina Caiazza, Adelaide Fusco, Annapaola Cirillo, Federica Verrillo, Francesca Dongiglio, Giuseppe Palmiero, Federica Barra, Giulia Frisso, Maria Giovanna Russo, Paolo Calabrò and Giuseppe Limongelli
Cardiogenetics 2026, 16(1), 6; https://doi.org/10.3390/cardiogenetics16010006 - 19 Mar 2026
Abstract
Sudden cardiac death (SCD) is a major global health issue, defined as sudden natural death presumed to be of cardiac cause. While in the elderly SCD is commonly associated with coronary artery disease, in the younger population it is linked to inherited cardiomyopathies
[...] Read more.
Sudden cardiac death (SCD) is a major global health issue, defined as sudden natural death presumed to be of cardiac cause. While in the elderly SCD is commonly associated with coronary artery disease, in the younger population it is linked to inherited cardiomyopathies or channelopathies, even though SCD can remain unexplained even after a comprehensive autopsy in a substantial proportion of cases. In this context, genetic testing has gained importance, supported by the widespread availability of techniques such as next-generation and whole-exome/genome sequencing and their reduced costs. This state-of-the-art review summarizes the genetic bases of sudden cardiac death among cardiomyopathies, channelopathies and in sudden unexplained death presumed to be of arrhythmic cause. Among the structural causes, inherited cardiomyopathies such as hypertrophic, dilated, non-dilated left ventricular, arrhythmogenic right ventricular and restrictive ones represent major substrates for malignant ventricular arrhythmias mostly arising from variants in sarcomeric or desmosomal genes. Channelopathies (long or short QT syndrome, Brugada syndrome and catecholaminergic polymorphic ventricular tachycardia) are caused by variants in genes encoding cardiac ion channels and/or regulatory proteins, which equally predispose to high risk of life-threatening ventricular arrhythmias. In sudden arrhythmic death syndrome, with a structurally normal heart, post-mortem genetic testing (molecular autopsy) can uncover an underlying inherited condition. However, variants of uncertain significance are detected in more than half of the cases, underscoring the need for a multidisciplinary approach. Genetic testing also plays a key role in cascade screening of first-degree relatives. While monogenic variants drive risk in inherited cardiac disorders, emerging evidence suggests that polygenic contributions may modulate SCD susceptibility, highlighting future roles for polygenic risk scores in risk stratification.
Full article
(This article belongs to the Special Issue Genetic Insights into Sudden Cardiac Death: From Risk Stratification to Precision Prevention)
►▼
Show Figures

Figure 1
Open AccessReview
Influence of Genetic and Epigenetic Factors in Takotsubo Syndrome: Insights and Gaps of an Incompletely Understood Disease
by
Giulio La Rosa, Gemma Pelargonio, Francesco Santoro, Sergio Conti, Francesco Campo and Giuseppe Sgarito
Cardiogenetics 2026, 16(1), 5; https://doi.org/10.3390/cardiogenetics16010005 - 12 Mar 2026
Abstract
Takotsubo syndrome (TTS) is a temporary and reversible form of cardiomyopathy that clinically mimics acute coronary syndrome, typically triggered by intense physical or emotional stress. It mainly affects postmenopausal women and exhibits significant variation among individuals regarding its onset, progression, and outcomes. Although
[...] Read more.
Takotsubo syndrome (TTS) is a temporary and reversible form of cardiomyopathy that clinically mimics acute coronary syndrome, typically triggered by intense physical or emotional stress. It mainly affects postmenopausal women and exhibits significant variation among individuals regarding its onset, progression, and outcomes. Although significant advances have been made since its initial description in 1990, the underlying pathophysiological mechanisms remain incompletely understood, limiting the development of effective prevention and targeted treatment strategies. A potential genetic predisposition has been suggested, supported by reports of familial clustering; however, a systematic and updated characterization of genetic and epigenetic factors associated with TTS is still lacking. This systematic and critical review aims to offer a comprehensive overview of current evidence on genetic susceptibility and epigenetic biomarkers potentially involved in the pathogenesis of TTS. Due to the heterogeneity and inconsistency of available findings, particular attention is also given to the methodological limitations of existing genetic studies. Finally, the review examines emerging multimodal approaches that may offer new perspectives for understanding the complex biological foundations of this syndrome.
Full article
(This article belongs to the Section Biomarkers)
►▼
Show Figures

Figure 1
Open AccessReview
Exploring the Genetic Architecture of Myocarditis and Inherited Cardiomyopathies
by
Sukruth Pradeep Kundur, Ali Malik, Rasi Mizori and Sanjay Sivalokanathan
Cardiogenetics 2026, 16(1), 4; https://doi.org/10.3390/cardiogenetics16010004 - 10 Mar 2026
Abstract
Myocarditis is a complex inflammatory myocardial disease. Although traditionally regarded as exclusively immune-mediated, recent evidence highlights the significant role of underlying genetics on susceptibility, phenotypic variability, and long-term prognosis. This narrative review examines the evolving genetic architecture of myocarditis and its relationship to
[...] Read more.
Myocarditis is a complex inflammatory myocardial disease. Although traditionally regarded as exclusively immune-mediated, recent evidence highlights the significant role of underlying genetics on susceptibility, phenotypic variability, and long-term prognosis. This narrative review examines the evolving genetic architecture of myocarditis and its relationship to inherited cardiomyopathies, integrating mechanistic insights from molecular, imaging, and clinical studies. Variants in desmosomal genes such as desmoplakin (DSP) and plakophilin-2 (PKP2) are increasingly linked to recurrent myocarditis that may evolve into arrhythmogenic cardiomyopathy, supporting the concept of a genetically predisposed myocardium in which inflammatory stressors can act as triggers. Truncating variants in titin (TTN) and Filamin C (FLNC) are associated with fulminant or dilated phenotypes. Conversely, mutations in Lamin A/C (LMNA), Desmin (DES), and BCL2-Associated Athanogene 3 (BAG3) contribute to inflammatory myocardial remodeling and other forms of inherited cardiomyopathies. These findings collectively have the potential to redefine myocarditis as an inflammatory disorder influenced by genetic factors. Furthermore, advancements in genetic testing and multi-omics approaches show promise in enhancing diagnostic accuracy and informing management strategies.
Full article
(This article belongs to the Section Molecular Genetics)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
Genetic Variants as a Potentially Arrhythmogenic Substrate in Mitral Annular Disjunction: Case Report and a Systematic Review of the Literature
by
Lorenzo Bianchi, Marialaura Buscemi, Domenico Coviello, Massimiliano Cecconi, Andrea Minghini, Stefano Cornara, Matteo Astuti, Francesco Pentimalli, Pietro Bellone, Emmanuel Androulakis and Alberto Somaschini
Cardiogenetics 2026, 16(1), 3; https://doi.org/10.3390/cardiogenetics16010003 - 26 Feb 2026
Abstract
Mitral annular disjunction (MAD) is associated with an increased risk of ventricular arrhythmias and sudden cardiac death, yet its genetic background remains poorly defined. We report the case of a 50-year-old man with MAD who survived cardiac arrest and carries three variants of
[...] Read more.
Mitral annular disjunction (MAD) is associated with an increased risk of ventricular arrhythmias and sudden cardiac death, yet its genetic background remains poorly defined. We report the case of a 50-year-old man with MAD who survived cardiac arrest and carries three variants of unknown significance (VUS) in genes involved in cardiomyopathy pathogenesis. To explore the genetic basis of non-syndromic MAD, we performed a systematic review of the literature, identifying five case reports and one retrospective cohort study. The case reports described patients with MAD harboring four pathogenic variants and ten VUS. Two pathogenic variants were linked to cardiomyopathies, involving proteins of the nuclear envelope and cytoskeleton, while two were associated with channelopathies. The retrospective cohort study identified a recurrent variant in a gene involved in intercellular adhesion segregating within a family affected by MAD. Overall, available evidence suggests that genetic factors may hypothetically modulate susceptibility to MAD, not only in connective tissue disorders but also in isolated mitral valve disease. Variants associated with arrhythmogenic cardiomyopathies and channelopathies appear to cluster in families with non-syndromic MAD and arrhythmic phenotypes, suggesting a role in the arrhythmic substrate. However, in absence of definitive functional, segregation, or longitudinal data, the contribution of genetic variants to MAD should be interpreted with caution. Further genomic studies are needed to clarify their genetic contribution and prognostic implications.
Full article
(This article belongs to the Section Cardiovascular Genetics in Clinical Practice)
►▼
Show Figures

Figure 1
Open AccessReview
Non-Lysosomal Glycogen Storage Cardiomyopathy with Hypertrophic Phenotype Due to PRKAG2 c.905G>A (p.Arg302Gln): Case Report and Narrative Review
by
Pasquale Crea, Alice Moncada, Francesco Catanzariti, Graziella Agnelli, Michela Navarra, Claudia Rubino, Irene Scimè, Lucio Teresi, Maurizio Cusmà Piccione, Luigi Colarusso, Roberto Licordari, Giuseppe Dattilo and Gianluca Di Bella
Cardiogenetics 2026, 16(1), 2; https://doi.org/10.3390/cardiogenetics16010002 - 21 Feb 2026
Abstract
Background: PRKAG2 cardiac syndrome is a rare autosomal dominant glycogen-storage cardiomyopathy that mimics sarcomeric hypertrophic cardiomyopathy (HCM) but features ventricular pre-excitation, progressive conduction disease and concentric hypertrophy due to intracellular glycogen accumulation. The c.905G>A (p.Arg302Gln) variant is one of the most frequently reported
[...] Read more.
Background: PRKAG2 cardiac syndrome is a rare autosomal dominant glycogen-storage cardiomyopathy that mimics sarcomeric hypertrophic cardiomyopathy (HCM) but features ventricular pre-excitation, progressive conduction disease and concentric hypertrophy due to intracellular glycogen accumulation. The c.905G>A (p.Arg302Gln) variant is one of the most frequently reported pathogenic substitutions. Case summary: We describe a three-generation family carrying the heterozygous PRKAG2 p.Arg302Gln variant. The proband, a 41-year-old man, presented with paroxysmal atrial fibrillation, short PR interval and abnormal intraventricular conduction associated with concentric left ventricular hypertrophy and preserved ejection fraction. Holter monitoring disclosed episodes of high-grade atrioventricular block, prompting implantation of a primary-prevention dual-chamber ICD. Two gene-positive brothers exhibited milder hypertrophy but shared sinus bradycardia, ventricular pre-excitation and supraventricular arrhythmias; one underwent catheter ablation of a posteroseptal accessory pathway. The affected mother displayed a hypertrophic phenotype complicated by sick sinus syndrome and permanent atypical atrial flutter requiring pacemaker implantation. No relevant extracardiac involvement was detected in any family member. Review and novelty: Using this family as a starting point, we provide a concise narrative review of PRKAG2 syndrome with emphasis on the Arg302Gln genotype, molecular mechanisms and emerging treatment strategies. We highlight key multimodality imaging and tissue-characterization features that help distinguish diffuse, concentric glycogen-storage hypertrophy from the often-asymmetric pattern of sarcomeric HCM. Integration of our findings with published Arg302Gln cohorts illustrates the broad phenotypic variability in conduction disease, pre-excitation and atrial arrhythmias. Conclusions: PRKAG2 p.Arg302Gln-related cardiomyopathy should be suspected in patients with otherwise unexplained left ventricular hypertrophy associated with short PR interval, pre-excitation or early brady–tachy arrhythmias. Early recognition of red-flag features, systematic genetic testing, family screening and tailored arrhythmia/device management are crucial, while emerging gene- and pathway-targeted therapies may offer future disease-modifying potential.
Full article
(This article belongs to the Section Rare Disease-Genetic Syndromes)
►▼
Show Figures

Figure 1
Open AccessCase Report
Digenic Contribution of Heterozygous ALPK3 and TRIM63 Variants to End-Stage Hypertrophic Cardiomyopathy in a Young Adult
by
Olga S. Chumakova, Natalia V. Milovanova, Elena A. Mershina, Sergey I. Kutsev and Ekaterina Y. Zakharova
Cardiogenetics 2026, 16(1), 1; https://doi.org/10.3390/cardiogenetics16010001 - 1 Jan 2026
Abstract
Hypertrophic cardiomyopathy (HCM), the most common inherited cardiac disorder, is usually caused by pathogenic variants in sarcomeric genes and is inherited in an autosomal dominant manner. Around 5% of cases are caused by variants in non-sarcomeric genes, which may involve alternative modes of
[...] Read more.
Hypertrophic cardiomyopathy (HCM), the most common inherited cardiac disorder, is usually caused by pathogenic variants in sarcomeric genes and is inherited in an autosomal dominant manner. Around 5% of cases are caused by variants in non-sarcomeric genes, which may involve alternative modes of inheritance. This study presents the first reported case of HCM associated with digenic contribution of heterozygous variants in two non-sarcomeric genes: ALPK3 and TRIM63. The patient was incidentally diagnosed with non-obstructive HCM in childhood and developed extreme myocardial hypertrophy with moderate heart failure at the age of 18. Rapid progressive left ventricular dysfunction promptly resulted in death at the age of 26. Genetic testing with an extended HCM panel identified no sarcomeric variants but revealed two truncating variants in the ALPK3 and TRIM63 genes. Whole-genome sequencing excluded any other causes of the disease. Heterozygous ALPK3 variants are typically associated with late-onset HCM, whereas TRIM63 variants are only considered pathogenic in a recessive state. This case, therefore, suggests a synergistic contribution of both variants to the development of a severe phenotype. The potential mechanisms of interaction between the protein products of ALPK3 and TRIM63 within the M-band of the sarcomere are discussed.
Full article
(This article belongs to the Section Molecular Genetics)
►▼
Show Figures

Figure 1
Open AccessReview
The Hidden Face of Danon Disease: Unique Challenges for Female Patients
by
Laura Torlai Triglia, Federico Barocelli, Enrico Ambrosini, Alberto Bettella, Filippo Luca Gurgoglione, Michele Bianconcini, Angela Guidorossi, Francesca Russo, Antonio Percesepe and Giampaolo Niccoli
Cardiogenetics 2025, 15(4), 32; https://doi.org/10.3390/cardiogenetics15040032 - 4 Dec 2025
Abstract
Danon Disease (DD) is a rare X-linked autophagic vacuolar myopathy caused by pathogenic variants in the lysosome-associated membrane protein 2 (LAMP-2) gene. Alternative splicing of the terminal exon 9 leads to the creation of three different isoforms, each with essential roles in regulating
[...] Read more.
Danon Disease (DD) is a rare X-linked autophagic vacuolar myopathy caused by pathogenic variants in the lysosome-associated membrane protein 2 (LAMP-2) gene. Alternative splicing of the terminal exon 9 leads to the creation of three different isoforms, each with essential roles in regulating autophagy. DD is characterized by cardiomyopathy, skeletal myopathy, cognitive impairment, and retinal disorders, with cardiac involvement being the primary cause of morbidity and mortality. Muscle biopsy may reveal signs of vacuolar myopathy, but the diagnosis is typically confirmed through sequencing and deletion/duplication analysis of the LAMP-2 gene using peripheral blood. Although few genotype–phenotype correlations have been described, with most being limited to isoform 2B of exon 9, the most significant prognostic indicator remains sex. The disease manifests earlier and with a more severe systemic presentation in males due to their hemizygous status, whereas in females, the typical presentation is late-onset hypertrophic or dilated cardiomyopathy, generally without extracardiac involvement. Cases of severely affected women have been described, potentially due to non-random or defective X-inactivation. The less typical and delayed clinical presentation in females can result in incorrect or missed diagnoses. The aim of this narrative review is to summarize the natural history, diagnostic criteria, management strategies, and recent advancements in the understanding of DD in women.
Full article
(This article belongs to the Section Rare Disease-Genetic Syndromes)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Sinus Bradycardia and Long QT Syndrome: Double Heterozygosity for Variants in KCNH2 and HCN4
by
Jaël S. Copier, Fenna Tuijnenburg, Karolina Andrzejczyk, Alex V. Postma, Saskia N. van der Crabben, Oussama Najih, Caroline Pham, Leander Beekman, Arie O. Verkerk, Ahmad S. Amin and Elisabeth M. Lodder
Cardiogenetics 2025, 15(4), 31; https://doi.org/10.3390/cardiogenetics15040031 - 13 Nov 2025
Abstract
Introduction: Clinical variability within families harbouring disease-causing genetic variants hampers clinical care and risk stratification. We studied a multigenerational family presenting with sinus bradycardia and long QT syndrome type 2 (LQTS2). The family harboured a pathogenic variant in KCNH2, which co-segregated
[...] Read more.
Introduction: Clinical variability within families harbouring disease-causing genetic variants hampers clinical care and risk stratification. We studied a multigenerational family presenting with sinus bradycardia and long QT syndrome type 2 (LQTS2). The family harboured a pathogenic variant in KCNH2, which co-segregated with the observed LQTS2. We studied the genetic cause of the high occurrence of sinus bradycardia in this family. Methods: Clinical data was collected, including heart rate, QT-interval, symptoms, and echocardiographic parameters. QTc was calculated using the Bazett and the Fridericia formula. Sanger sequencing of HCN4 was performed, followed by segregation analysis of the identified variant with sinus bradycardia. The biophysiological consequences of two variants, KCNH2-p.L69P (c.206T>C) and HCN4-p.R666W (c.1996C>T), were assessed by patch-clamp experiments. Therefore, a heterologous model was generated by transfection of HEK293A or CHO-k1 cells, respectively. Results: Sanger sequencing of HCN4 identified HCN4-p.R666W (c.1996C>T), which has a stronger segregation with the observed sinus bradycardia than KCNH2-p.L69P. Patch-clamp experiments revealed that KCNH2-p.L69P and HCN4-p.R666W lead to a decrease in the corresponding current densities, which explains the LQTS and sinus bradycardia observed in the patients. Carriers of both genetic variants have a more severe LQTS2 phenotype, reflected in longer QT and higher incidence of syncope. Conclusions: We identified two (likely) pathogenic variants, KCNH2-p.L69P and HCN4-p.R666W, co-segregating with LQTS2 and sinus bradycardia, respectively. Patients carrying both variants showed a more severe phenotype. These findings highlight the importance of additional genetic testing when discordant features are present, thereby enabling more accurate diagnosis, risk prediction, and management.
Full article
(This article belongs to the Section Molecular Genetics)
►▼
Show Figures

Figure 1
Open AccessCase Report
Integrating Genetic, Clinical, and Histopathological Data for Definitive Diagnosis of PRKAG2-Related Disease
by
Martina Caiazza, Emanuele Monda, Francesco Loffredo, Rossana Bussani, Vera Fico, Emanuele Bobbio, Chiara Cirillo, Anna Murredda, Immacolata Viscovo, Alessandra Scatteia, Santo Dellegrottaglie, Diego Colonna, Berardo Sarubbi, Maria Giovanna Russo, Paolo Golino, Gianfranco Sinagra and Giuseppe Limongelli
Cardiogenetics 2025, 15(4), 30; https://doi.org/10.3390/cardiogenetics15040030 - 4 Nov 2025
Cited by 2
Abstract
Background: PRKAG2-related disease is an autosomal dominant disorder caused by pathogenic variants in the PRKAG2 gene, leading to glycogen accumulation in cardiomyocytes. It is characterized by left ventricular hypertrophy (LVH), ventricular pre-excitation, and conduction disease. Due to the rarity of the condition and
[...] Read more.
Background: PRKAG2-related disease is an autosomal dominant disorder caused by pathogenic variants in the PRKAG2 gene, leading to glycogen accumulation in cardiomyocytes. It is characterized by left ventricular hypertrophy (LVH), ventricular pre-excitation, and conduction disease. Due to the rarity of the condition and the frequent occurrence of private variants, functional or pathological testing is required for definitive pathogenicity classification. Case Presentation: We describe a 22-year-old male referred for evaluation after experiencing exertional dyspnea and a syncopal episode. Family history revealed sudden cardiac deaths and conduction disease requiring pacemaker implantation. The patient exhibited mild LVH on imaging, conduction abnormalities on electrophysiological study, and a heterozygous PRKAG2 variant (c.1643C>T; p.Ser548Leu), classified as likely pathogenic according to ACMG guidelines. Cascade screening identified the variant in three family members, one of whom exhibited a positive phenotype. Endomyocardial biopsy revealed glycogen accumulation, providing histopathological confirmation of PRKAG2-related disease. Conclusions: This case underscores the importance of integrating genetic, clinical, and histopathological data in variant interpretation. Endomyocardial biopsy can provide definitive evidence to reclassify a PRKAG2 variant as pathogenic, thereby guiding management and family screening.
Full article
(This article belongs to the Section Rare Disease-Genetic Syndromes)
►▼
Show Figures

Figure 1
Open AccessReview
From Genetics to Phenotype: Understanding the Diverse Manifestations of Cardiovascular Genetic Diseases in Pediatric Populations
by
Jule Leonie Gutmann, Alina Spister and Lara Baticic
Cardiogenetics 2025, 15(4), 29; https://doi.org/10.3390/cardiogenetics15040029 - 11 Oct 2025
Abstract
Congenital genetic heart defects are major contributors to pediatric morbidity and mortality, underscoring the importance of early detection and individualized therapeutic strategies. This review aimed to summarize current knowledge on a spectrum of inherited cardiovascular disorders, with a focus on their genetic etiology,
[...] Read more.
Congenital genetic heart defects are major contributors to pediatric morbidity and mortality, underscoring the importance of early detection and individualized therapeutic strategies. This review aimed to summarize current knowledge on a spectrum of inherited cardiovascular disorders, with a focus on their genetic etiology, molecular pathogenesis, and phenotypic presentation in children. Conditions discussed include Marfan syndrome, Noonan syndrome, various cardiomyopathies, Duchenne muscular dystrophy, DiGeorge syndrome, and the tetralogy of Fallot. These six conditions were selected to represent the spectrum of pediatric cardiovascular genetic diseases, encompassing connective tissue disorders, multisystem syndromes, primary myocardial diseases, neuromuscular cardiac involvement, and structural congenital defects, thereby illustrating how distinct genotypes lead to diverse phenotypes. For each disorder, the underlying genetic mutations, associated molecular pathways, cardiovascular involvement, clinical features, and approaches to diagnosis and management are examined. Emphasis is placed on the role of timely diagnosis, genetic counseling, and personalized treatment in improving patient outcomes. The review concludes by highlighting emerging research directions and novel therapeutic interventions aimed at enhancing care for these complex pediatric conditions.
Full article
(This article belongs to the Section Inherited Heart Disease-Children)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
MicroRNA and DNA Methylation Adaptation Mechanism to Endurance Training in Cardiovascular Disease: A Systematic Review
by
Jil Delhez, Jeanne Ougier, Francisco Xavier de Araujo, Raphael Martins de Abreu and Camilo Corbellini
Cardiogenetics 2025, 15(4), 28; https://doi.org/10.3390/cardiogenetics15040028 - 11 Oct 2025
Cited by 1
Abstract
Background: Regular endurance training induces physiological changes in cardiac structure and function. The precise epigenetic mechanisms by which cardiovascular adaptations are mediated are still unclear. This review seeks to clarify the role of epigenetic regulation in exercise-induced cardiovascular adaptation. Methods: This systematic review
[...] Read more.
Background: Regular endurance training induces physiological changes in cardiac structure and function. The precise epigenetic mechanisms by which cardiovascular adaptations are mediated are still unclear. This review seeks to clarify the role of epigenetic regulation in exercise-induced cardiovascular adaptation. Methods: This systematic review was conducted in accordance with the PRISMA guidelines up to 30 April 2025, using the databases PubMed, VHL, and LILACS Plus. Studies were included if they focused on microRNA expression and DNA methylation in individuals with cardiovascular disease who underwent endurance training. Results: Six articles, including 384 participants with heart failure, coronary artery disease, and hypertension, were included in the final analysis. Changes in DNA methylation and microRNA expression of specific genes involved in cardiovascular structural and functional adaptation were observed. Significant improvements were found in body composition, VO2peak, systolic and diastolic blood pressure, and left ventricular function and structure. Conclusions: Endurance training has a positive impact on epigenetic mechanisms related to cardiovascular structural and functional adaptation. A clear causal link between epigenetic modifications and clinical outcomes remains to be established.
Full article
(This article belongs to the Section Cardiovascular Genetics in Clinical Practice)
►▼
Show Figures
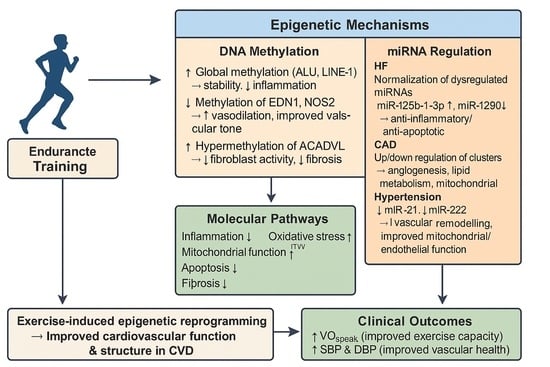
Graphical abstract
Open AccessReview
Polygenic Risk Scores and Coronary Artery Disease
by
Salman Ansari, Suvasini Lakshmanan and Matthew J. Budoff
Cardiogenetics 2025, 15(4), 27; https://doi.org/10.3390/cardiogenetics15040027 - 26 Sep 2025
Cited by 1
Abstract
Background: Polygenic risk scores (PRSs) aggregate the effects of many common genetic variants and are being investigated as tools to refine coronary artery disease (CAD) risk prediction beyond traditional clinical models. Methods and Results: We review the development of PRS from early unweighted
[...] Read more.
Background: Polygenic risk scores (PRSs) aggregate the effects of many common genetic variants and are being investigated as tools to refine coronary artery disease (CAD) risk prediction beyond traditional clinical models. Methods and Results: We review the development of PRS from early unweighted scores to contemporary genome-wide models and summarize evidence from major studies. We identified key studies through PubMed searches using the terms “polygenic risk score,” “genetic risk prediction,” and “coronary artery disease,” supplemented by citation chaining of highly cited articles and recent reviews. Large cohorts, such as the UK Biobank, show that individuals in the highest PRS percentiles have a 3–5-fold higher risk of CAD, and may gain the greatest benefit from statin therapy. PRS can also reclassify younger adults at borderline or intermediate risk and may complement coronary artery calcium (CAC) scoring. Conclusions: PRSs hold promise for lifetime risk stratification and targeted prevention in CAD but are limited by ancestry bias in GWAS, underrepresentation of diverse populations, inconsistency in individual estimates, and lack of standardized reporting. Future research should focus on expanding multi-ancestry databases, standardizing methods, prospective validation, and effective communication strategies to support equitable and evidence-based clinical use.
Full article
(This article belongs to the Section Cardiovascular Genetics in Clinical Practice)
►▼
Show Figures

Figure 1
Open AccessReview
Ethical Considerations Regarding Advanced Heart Failure Therapies in Patients Affected by Dystrophinopathies
by
Marco Spagnolin, Luca Fazzini, Amedeo Terzi, Attilio Iacovoni, Raffaele Abete, Ottavio Zucchetti, Michele Senni and Mauro Gori
Cardiogenetics 2025, 15(3), 26; https://doi.org/10.3390/cardiogenetics15030026 - 22 Sep 2025
Abstract
Dystrophinopathies, including Duchenne and Becker muscular dystrophies (DMD and BMD), are inherited neuromuscular disorders frequently complicated by progressive cardiac involvement, ultimately leading to advanced heart failure. While heart transplantation and long-term left ventricular assist device (LVAD) therapy represent potential therapeutic options, their application
[...] Read more.
Dystrophinopathies, including Duchenne and Becker muscular dystrophies (DMD and BMD), are inherited neuromuscular disorders frequently complicated by progressive cardiac involvement, ultimately leading to advanced heart failure. While heart transplantation and long-term left ventricular assist device (LVAD) therapy represent potential therapeutic options, their application in this population raises significant ethical challenges. This review explores the ethical implications surrounding the allocation of scarce medical resources, particularly in patients with limited life expectancy and multisystem disease, as in DMD. Decisions regarding eligibility for heart transplantation must balance individual benefit, considering the impact of excluding other potential recipients. LVAD therapy, although more accessible, still demands careful patient selection due to high perioperative risk and postoperative complications. The review emphasizes the need for transparent, multidisciplinary decision-making processes that respect patient autonomy while ensuring equitable and rational distribution of healthcare resources. Ultimately, while advanced therapies may be feasible in selected cases, particularly in BMD, ethical deliberation remains central to determining their appropriateness in the context of dystrophinopathies.
Full article
(This article belongs to the Section Rare Disease-Neuromuscular Diseases)
►▼
Show Figures

Figure 1
Open AccessArticle
Genetic Profile of Pediatric-Onset Cardiac Channelopathies
by
Sara Giovani, Adelaide Ballerini, Alessia Gozzini, Michele Di Lorenzo, Davide Mei, Silvia Passantino, Mattia Zampieri, Alessia Tomberli, Alberto Marchi, Giovanni Battista Calabri, Gaia Spaziani, Giulio Porcedda, Elena Bennati, Silvia Favilli, Iacopo Olivotto and Francesca Girolami
Cardiogenetics 2025, 15(3), 25; https://doi.org/10.3390/cardiogenetics15030025 - 12 Sep 2025
Abstract
This study investigates the genetic background of pediatric-onset cardiac channelopathies, a rare group of genetic disorders causing arrhythmias and sometimes sudden death, whose genetic background remains partially unknown. The research analyzed 59 pediatric patients (<18 years of age) diagnosed with different channelopathies (LQTS,
[...] Read more.
This study investigates the genetic background of pediatric-onset cardiac channelopathies, a rare group of genetic disorders causing arrhythmias and sometimes sudden death, whose genetic background remains partially unknown. The research analyzed 59 pediatric patients (<18 years of age) diagnosed with different channelopathies (LQTS, BrS, CPVT, SQTS, and conduction disorders), along with 40 of their family members, using Next-Generation Sequencing (NGS) after genetic counseling. A causative genetic variant was found in 47% of cases, mainly in the KCNQ1 (42%), RYR2 (16%), CACNA1C (10%), and SCN5A (10%) genes. Notably, a de novo large deletion in KCNH2 was detected in an LQTS patient, and a pathogenic CALM1 variant was identified in a child. A compound heterozygous KCNQ1 was consistent with Jervell and Lange-Nielsen syndrome. In light of these data, genetic testing is crucial for diagnosis, prognosis, and treatment planning; cascade screening allowed early risk identification and preventive interventions for family members. Expanding NGS technologies and research on new candidate genes may enhance personalized therapies in the future.
Full article
(This article belongs to the Section Molecular Genetics)
►▼
Show Figures

Figure 1
Open AccessCase Report
TNNC1 Gene Mutation in Ebstein’s Anomaly and Left Ventricular Hypertrabeculation: A Case Report of a New Causative Mutation?
by
Irene Raso, Claudia Chillemi, Giorgia Prontera, Arianna Laoreti, Elisa Cattaneo, Valeria Calcaterra, Gian Vincenzo Zuccotti and Savina Mannarino
Cardiogenetics 2025, 15(3), 24; https://doi.org/10.3390/cardiogenetics15030024 - 26 Aug 2025
Cited by 1
Abstract
Background: Ebstein’s anomaly (EA) is a rare congenital heart defect characterized by failure of tricuspid valve delamination during embryogenesis. Left ventricular (LV) hypertrabeculation results from incomplete myocardial compaction during fetal development. EA is associated with LV hypertrabeculation in 0.14% of cases, and EA
[...] Read more.
Background: Ebstein’s anomaly (EA) is a rare congenital heart defect characterized by failure of tricuspid valve delamination during embryogenesis. Left ventricular (LV) hypertrabeculation results from incomplete myocardial compaction during fetal development. EA is associated with LV hypertrabeculation in 0.14% of cases, and EA is the most common congenital heart disease in LV hypertrabeculation (up to 29%), suggesting a shared embryogenetic pathway. Case Report: We describe a female patient prenatally diagnosed with EA and a large ventricular septal defect. Postnatal echocardiography confirmed EA with moderate regurgitation and revealed previously unnoticed left ventricular excessive trabeculations. Whole exome sequencing revealed a heterozygous never-described variant of unknown significance in the TNNC1 gene. Discussion: The genetic link between EA and LV hypertrabeculation remains unclear, though variants in sarcomeric or cytoskeletal genes like MYH7, TPM1, and NKX2.5—essential for cardiac development—have been implicated. A developmental hypothesis suggests that aberrant contraction during endocardial-to-mesenchymal and epicardial-to-mesenchymal transformation (5th–8th gestational weeks) may affect valve delamination and ventricular compaction via parallel signaling pathways. TNNC1 encodes troponin C1, a subunit of the troponin complex involved in muscle contraction. Its mutations are known to alter calcium sensitivity and impair cardiac contractility. Conclusions: EA and LV hypertrabeculation patients diagnosed in infancy have a greater risk of negative outcomes. Early, especially prenatal, diagnosis is crucial. Genetic analysis can provide fundamental insight into cardiac development. This new and rare variant of TNNC1 gene supports the hypothesis that early cardiomyocytes dysfunction disrupts both valve delamination and left ventricular compaction and that the two diseases share a common genetic pathway related to cardiomyocyte contraction.
Full article
(This article belongs to the Section Inherited Heart Disease-Children)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
Association Between Angiotensinogen Gene M235T and Renin–Angiotensin System Insertion/Deletion Variants and Risk of Cardiovascular Disease in North African and Middle Eastern Populations: A Systematic Review and Meta-Analysis
by
Rajaa El Mansouri, Hind Dehbi and Rachida Habbal
Cardiogenetics 2025, 15(3), 23; https://doi.org/10.3390/cardiogenetics15030023 - 8 Aug 2025
Abstract
►▼
Show Figures
Background: The renin–angiotensin system (RAS) is pivotal in regulating cardiovascular function, while cardio-genomics offers insights into genetic factors influencing cardiovascular disease (CVD) susceptibility. Aim: This study investigates the relationship between the angiotensin-converting enzyme insertion/deletion variant (ACE I/D) and the angiotensinogen gene M235T variant
[...] Read more.
Background: The renin–angiotensin system (RAS) is pivotal in regulating cardiovascular function, while cardio-genomics offers insights into genetic factors influencing cardiovascular disease (CVD) susceptibility. Aim: This study investigates the relationship between the angiotensin-converting enzyme insertion/deletion variant (ACE I/D) and the angiotensinogen gene M235T variant (AGT M235T) in Mediterranean, North African, and Middle Eastern populations. Methods: A systematic review and meta-analysis, encompassing studies until December 2023, were conducted utilizing the PubMed and Scopus databases. The study followed the PICO checklist to enroll in the review process. The meta-analysis results were obtained using CMA software V2. Results: An analysis of 12 studies (2984 participants) for ACE I/D and 7 studies (2275 participants) for AGT M235T revealed significant associations between these gene variants and increased CVD risk in Mediterranean and North African populations. Conclusions: These findings underscore the utility of cardio-genomics in delineating CVD susceptibility among these groups, emphasizing targeted interventions and personalized treatment strategies
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Cardiogenetics, Hearts, JCDD, JCM
Adult Congenital Heart Disease: Advances in Diagnosis, Surgery, and Lifelong Care
Topic Editors: Satoshi Akagi, Jin Young SongDeadline: 30 June 2026
Topic in
Biomedicines, Biomolecules, Cardiogenetics, JCDD, JCRM, JMP, Metabolites, JPM
Advanced Imaging, Physiology, and Biomarkers in Ischemic Heart Disease
Topic Editors: Krishnaraj Sinhji Rathod, Alexandru CorlateanuDeadline: 10 March 2027

Conferences
Special Issues
Special Issue in
Cardiogenetics
Genetic Insights into Sudden Cardiac Death: From Risk Stratification to Precision Prevention
Guest Editors: Giuseppe Limongelli, Lia Crotti, Emanuele Bobbio, Saskia Nanette Van Der CrabbenDeadline: 31 August 2026
Special Issue in
Cardiogenetics
Contemporary and Future Approaches to Inherited Cardiomyopathies
Guest Editors: Michele Ciabatti, Alessia ArgiroDeadline: 20 December 2026
Special Issue in
Cardiogenetics
Genetics in Valvular Heart Diseases
Guest Editor: Giancarlo TrimarchiDeadline: 20 April 2027










