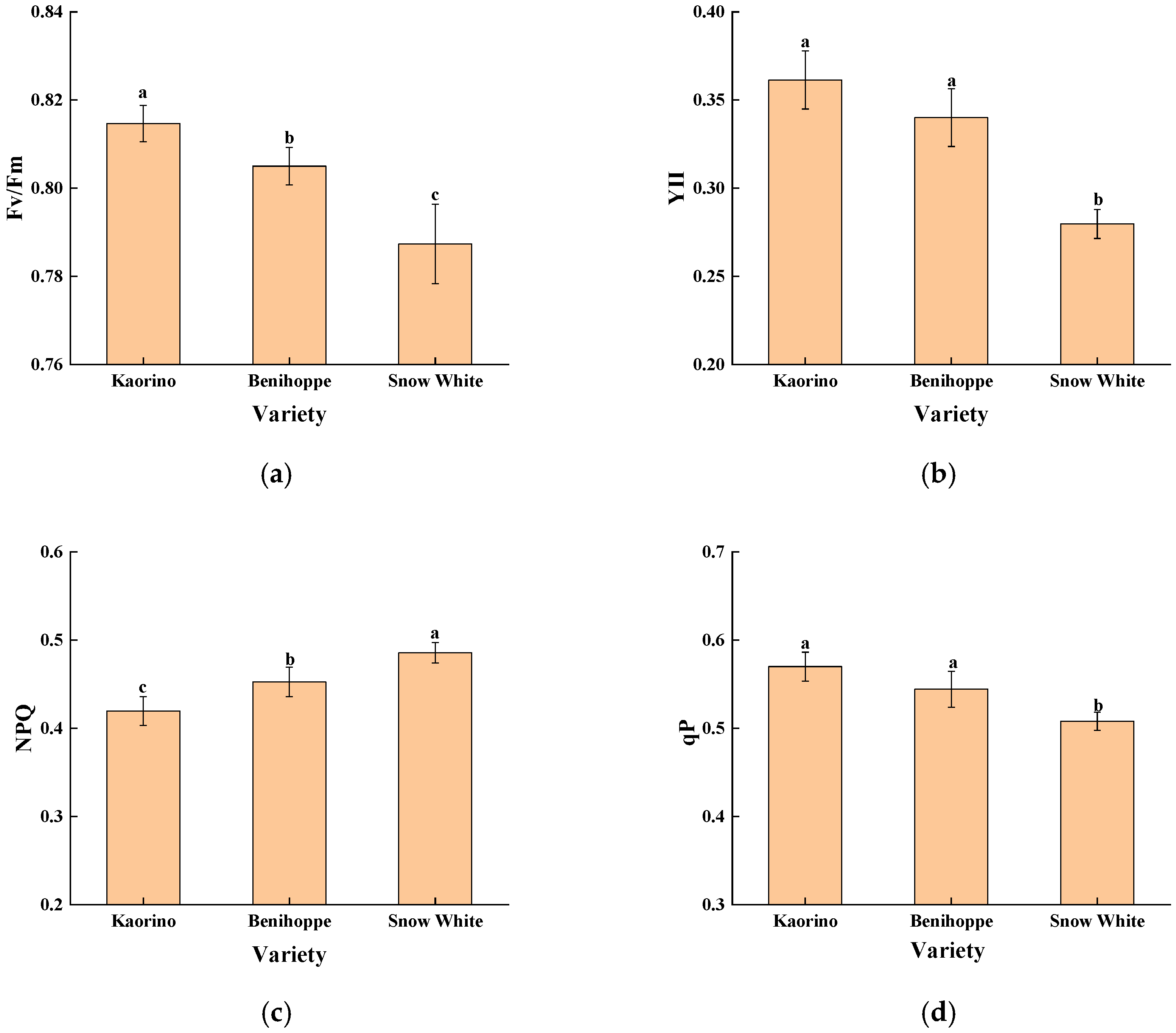Abstract
Strawberry is widely cultivated due to its short growth cycle, high yield, and significant profits. In high-latitude cold regions, the planting area of overwintering strawberry has expanded rapidly in recent years. However, although daytime temperatures inside solar greenhouses rise quickly with solar radiation, plants are frequently subjected to persistent nocturnal low-temperature stress (nocturnal temperature below 10 °C). This stress restricts photosynthesis, delays growth, and markedly reduces yield. Therefore, accurately evaluating the tolerance of strawberry varieties to low nocturnal temperatures is crucial for unheated overwintering production in cold regions. This study selected Snow White, Benihoppe, and Kaorino as experimental materials for overwintering cultivation trials in a typical cold-region solar greenhouse. We measured and analyzed growth and development, photosynthetic characteristics, phenological traits, and fruit yield. Based on photosynthetic physiology and phenotypic traits, we constructed the Photosynthesis–Fluorescence Index (PFI), the Production–Phenotype Index (PPI), and the Nocturnal Cold Tolerance Index (NCTI). The results showed that Kaorino exhibited significantly higher values in all three indices compared with Benihoppe and Snow White. After exposure to low night temperatures, Kaorino exhibited rapid photosynthetic induction, strong maintenance of PSII activity, vigorous growth, early maturation, and high yield. Moreover, all three composite indices were strongly and positively correlated with total yield (R2 > 0.97), demonstrating their effectiveness in distinguishing the nocturnal low-temperature tolerance of strawberry cultivars. These composite indices provide a scientifically robust method for selecting suitable cultivars for unheated overwinter strawberry production in high-latitude cold regions.
1. Introduction
As a perennial herb of Rosaceae, strawberry (Fragaria × ananassa Duch.) has bright red and tender fruits with a rich aroma and 98% edible parts and is well known as the Queen of Fruits due to its high nutritional and economic values [1,2]. Due to the increasing standard of living, the demand for winter strawberries is gradually increasing, and with the improved facility cultivation technology, overwintering strawberry production in solar greenhouses has become a major cultivation system in high-latitude cold regions [3,4,5]. The ripening stage of overwintering strawberry is usually from late November to May of the next year and coincides with the low production period of fruits and the peak season of fruit sale in Spring Festival. Overwintering strawberry has a high price and would achieve a profit of about CNY 60,000–100,000 per 1000 m2 and bring considerable economic benefits [6,7].
However, in high-latitude cold regions, the primary constraint on strawberry growth and yield during overwintering is not all-day low temperatures or extreme freezing events, but rather persistent nocturnal low-temperature stress [8,9]. Previous studies indicate that low nocturnal temperatures delay the next-morning recovery of PSII photochemical efficiency and inhibit temperature-sensitive enzymatic reactions of carbon fixation, leading to reduced carbon assimilation after sunrise and consequently constraining early-season yield formation; in elevated substrate-cultivation systems, when nocturnal temperatures drop below 10 °C, the strawberry yield decreases markedly [10,11,12,13,14,15]. Supplemental heating is still required under conventional cultivation practices to mitigate nocturnal cold stress. For a typical greenhouse (1000 m2), the annual heating cost typically ranges from CNY 10,000 to 20,000. With the continuous rise in energy prices, heating expenses are increasing every year, substantially elevating production costs. The escalating inputs associated with heating substantially reduce overall profitability and have become a major constraint to further expansion of the strawberry industry [16,17,18].
Breeding and selecting strawberry varieties with strong tolerance to low nocturnal temperatures is a fundamental approach for addressing the above challenges [19]. Different varieties of strawberry under nocturnal low-temperature growth conditions show significant differences in phenological indicators, growth and development performances, and photosynthetic characteristics [20,21,22,23,24]. Therefore, it is significant to screen proper strawberry varieties with good environmental adaptability, high yield, and good quality as the main varieties for the efficient production of overwintering strawberries without external heating measures.
Because stress tolerance and productivity are complex traits, composite or multi-parameter indices are widely used to integrate physiological and agronomic traits for rapid screening under stress [25,26,27]. In this study, three major strawberry varieties that are widely grown in northern China (Snow White, Benihoppe, and Kaorino), which exhibit pronounced differences in low-temperature tolerance, were used as tested materials. Overwintering cultivation trials were conducted in a typical cold-region solar greenhouse, during which plant growth, photosynthetic characteristics, phenology, and fruit yield were systematically measured. The objective was to develop a composite, multi-parameter index for strawberry tolerance to low nocturnal temperatures and to provide a scientific basis and methodological support for the rapid screening of cold-tolerant cultivars and the breeding of new cultivars that are suitable for cold regions.
2. Materials and Methods
2.1. Materials
The tested materials were the three main strawberry varieties cultivated in northern China: Fragaria × ananassa cv. Snow White, Fragaria × ananassa cv. Benihoppe, and Fragaria × ananassa cv. Kaorino.
2.2. Methods
The experiment was performed in a solar greenhouse at the Institute of Vegetables and Flowers of Jilin Province (125°23′37.1″ E, 43°49′51.9″ N) from August 2022 to May 2023. The strawberry plants were settled in three-dimensional cultivation tanks in the solar greenhouse on 20 August 2022. The cultivation tanks had the dimensions of 6 m × 0.3 m × 0.3 m (length × width × height) and were filled with mixed substrates of peat soil, vermiculite, and perlite (at a volume ratio of 2:1:1). Two rows of strawberries were cultivated in the tanks, with a row spacing of 20 cm and a plant spacing of 18 cm, and 300 plants were cultivated for each variety. After strawberries were planted, water and fertilizer were supplied once every 3 days at a dosage of 230 mL per plant, and the nutrient solution was prepared with the Japanese Yamazaki strawberry solution formula [28]. After the rejuvenation period, we randomly selected ten plants from each variety for measurement, ensuring that they were taken from the center of the solar greenhouse with consistent temperature and light conditions. The phenological period, growth and development status, fruit yield, and photosynthetic and physiological indicators of each variety were measured and compared during the whole growth cycle. Each trait was investigated according to Description Specifications and Data Standard for Strawberry Germplasm Resources [29], and the phenological period was determined according to an existing method [30].
2.3. Investigation Items and Methods
2.3.1. Growth and Development Indicators
After 25 September 2022, ten strawberry plants of each variety were selected for the measurements of growth and development indicators such as stem thickness, crown width, number of leaves, and number of inflorescences every other month. In addition, the starting points of budding stage, initial flowering stage, blooming stage, and initial ripening stage of fruit were observed to determine the phenological period of strawberries.
2.3.2. Yield
After 13 November 2022, mature strawberries were harvested, and the total yield was measured.
2.3.3. Chlorophyll Content
Leaf samples were selected from the corresponding parts of the middle of both sides of the main veins of the 3rd functional leaf. Then, 0.1 g of leaf samples was weighed, cut into thin stripes, and carefully transferred into a 10 mL centrifuge tube. After 10 mL of 80% acetone was added, the samples were soaked at 4 °C for 48 h. Then, the chlorophyll extracts were determined by UV/VIS spectrophotometer (UV5, Mettler-Toledo Ltd., Greifensee, Zurich, Switzerland) at 663, 645, and 470 nm wavelengths, and the chlorophyll content was calculated with Arnon’s equation [31]. Measurements were conducted in late December 2022, late November 2022, and late December 2022 and were repeated three times for accuracy.
2.3.4. Photosynthetic Indicators
The net photosynthetic rate of leaves was continuously measured with a continuous photosynthesis meter (YZQ-100, Yi Zong Qi Technology Co., Ltd., Beijing, China). For each variety, three leaves were selected for measurements: the 3rd to 5th fully developed leaves starting from the leaflet. Each continuous measurement period was 24 h, with an interval of 8 min. The measurement was performed on sunny days in late October 2022, late November 2022, and late December 2022.
2.3.5. Chlorophyll Fluorescence Imaging
The determination of chlorophyll fluorescence parameters was conducted using a chlorophyll fluorescence imaging system (IMAGIN-PAM, Heinz Waltz, Effeltrich, Bavaria, Germany). After 30 min of dark adaptation at room temperature, the third fully expanded functional leaf was cut with scissors, the protective cover was opened, and it was placed on the measurement platform of the fluorometer and secured with a string to keep the leaf flat. Three regions of interest (AOI; user-defined region of interest used to extract mean pixel values from fluorescence image) were selected on the leaf using IMAGIN-WIN (v2.56zn). Firstly, the measuring light was turned on (with an intensity of 0.1 µmol·m−2s−1) to obtain the initial fluorescence yield (Fo), and then the maximum fluorescence yield (Fm) was measured using saturating-pulse light (with an intensity of 2700 µmol·m−2s−1). Next, the actinic light (with an intensity of 300 µmol·m−2s−1) was turned on to measure the actual fluorescence yield (F′), the maximum fluorescence yield under light-adapted conditions (Fm′), and the minimum fluorescence yield under light-adapted conditions (Fo′). The maximum photochemical efficiency was calculated using the following formula: Fv/Fm = (Fm − Fo)/Fm. The actual photosynthetic efficiency of PSII was calculated as Y(II) = (Fm′ − F)/Fm′. The electron transport rate was calculated as ETR = Y(II) × PAR × Abs. × 0.5, where PAR is the photosynthetically active radiation (400–700 nm) of the actinic light, and Abs. is the leaf absorptance. The photochemical quenching coefficient was calculated as qP = (Fm′ − F)/(Fm′ − Fo′). The non-photochemical quenching coefficient was calculated as NPQ = (Fm − Fm′)/Fm′. Chlorophyll fluorescence images and parameters were processed and analyzed using the proprietary software IMAGIN-WIN (v2.56zn). Each treatment was replicated three times, and average pixel values from the images were employed for parameter calculation. All measurements were conducted in late December 2022, with fluorescence parameters being measured consistently in the early morning.
To further investigate the activation characteristics of the photosynthetic apparatus around sunrise in different varieties, we continuously monitored the temporal changes in Y(II), qP, and ETR after uncovering the thermal insulating quilt. The first measurement was taken in the early morning just before the thermal insulating quilt was uncovered; thereafter, the thermal insulating quilt was uncovered, and measurements were performed every 10 min for a total of six time points. Measurements were conducted in late December 2022 and were repeated three times to calculate statistical significance.
2.4. Construction of Composite Indices
To achieve quantitative and comparable characterization of strawberry varieties’ tolerance to low nocturnal temperatures, three composite indices were constructed based on photosynthetic physiological parameters, chlorophyll fluorescence responses, and production traits: the Photosynthesis–Fluorescence Index (PFI), the Production–Phenotype Index (PPI), and the Nocturnal Cold Tolerance Index (NCTI). Through data standardization and weighted integration, these indices form a composite evaluation model that simultaneously reflects both physiological freezing tolerance and production performance.
Due to significant differences in the dimensions and value ranges of the measured indicators, z-score standardization was first applied to the raw data for dimensionless processing and to ensure comparability:
where is the original value of the i-th indicator, is the mean of that indicator, and s is the standard deviation. Standardized variables were used for subsequent composite parameter calculations.
The Photosynthesis–Fluorescence Index (PFI) was defined as follows:
where
PFI = z(Fv/Fm_12) − z(YII_T50) + z(YII_slope20) + z(YII_AUC50) + z(Pmax_Dec)
Fv/Fm_12: maximum quantum efficiency of PSII measured in December.
YII_T50: time (min) required for Y(II) to recover to 50% of its maximum value after uncovering the thermal insulating quilt (a shorter time indicates faster recovery and stronger cold tolerance, hence the negative sign).
YII_slope20: slope of the increase in Y(II) over the first 20 min after uncovering the thermal insulating quilt (linear regression slope).
YII_AUC50: area under the Y(II) curve from 0 to 50 min after uncovering the thermal insulating quilt.
Pmax_Dec: maximum net photosynthetic rate measured in December.
The Production–Phenotype Index (PPI) was defined as follows:
where
PPI = z(EY%) + z(S) + z(Chl_ratio)
EY% (early yield percentage) = (cumulative yield from November to January/total yield) × 100%;
S (composite growth index) = z (crown width) + z (stem thickness) + z (number of inflorescences);
Chl_ratio (chlorophyll retention ratio) = total chlorophyll content in December/total chlorophyll content in October.
To comprehensively evaluate the nocturnal low-temperature adaptability and economic value of each cultivar, the Nocturnal Cold Tolerance Index (NCTI) was further constructed as follows:
NCTI = 0.6 × PFI + 0.4 × PPI
2.5. Data Processing Method
The processing and analysis of experimental data were completed with Microsoft Excel 2013 and SPSS 22.0. Duncan’s method was used to make multiple comparisons at a significance level of 0.05 when using data statistical methods for significance and statistical analysis. All the pictures were obtained using the Origin 2018 software.
3. Results
3.1. Temperature in the Solar Greenhouse
In the overwintering production of strawberries, the temperature and light intensity in the solar greenhouse play decisive roles [32]. Figure 1 shows the highest daytime temperature and the lowest nocturnal temperature recorded by temperature sensors in the solar greenhouse without heating measures from October 2022 to January 2023. As shown in the figure, daytime temperatures on sunny days can exceed 30 °C, whereas the minimum nocturnal temperatures frequently remain below 10 °C, with the lowest value reaching 5.1 °C. This pattern is characteristic of the persistently low nocturnal temperatures that are typical of unheated greenhouses in high-latitude cold regions. Although these temperatures do not reach levels that cause acute freezing injury, previous studies have demonstrated that nocturnal temperatures below 10 °C can suppress photosynthetic induction, reduce PSII recovery capacity, hinder floral bud differentiation, and ultimately decrease early-season yield in strawberries [15,33]. Therefore, such low nocturnal temperatures, while not constituting extreme cold damage, represent a critical environmental stress factor influencing winter greenhouse production.

Figure 1.
Temperature variation in the solar greenhouse from October 2022 to January 2023. Dates are expressed in the month/day format (MM/DD).
3.2. Growth and Development
The indicators of biological characteristics of strawberry varieties are commonly used to judge the nutritional growth status of plants and directly indicate the growth vigor of strawberry plants [34]. The stem thickness of Kaorino was up to 2.21 cm, which was significantly thicker than those of Benihoppe and Snow White (Figure 2a). The stem thicknesses of Benihoppe and Snow White, respectively, reached 85.1% and 68.3% of that of Kaorino. In addition, the crown width of Kaorino was 44.5 cm, which was 19.6% larger than that of Benihoppe and 42.6% larger than that of Snow White (Figure 2b). In terms of the number of leaves (Figure 2c), in the early and middle planting stage, the effective number of leaves of Benihoppe was the highest and reached 13, followed by Kaorino. The number of leaves of Snow White showed an overall increasing trend. The number of leaves of Snow White was the highest in the middle and late development stages and reached 14. The numbers of inflorescences of the three varieties of strawberries were significantly different and decreased in the following order: Kaorino > Benihoppe > Snow White (Figure 2d). In general, Kaorino exhibited the more advantageous growth vigor and nutritional growth, followed by Benihoppe and Snow White.


Figure 2.
Growth and development states of strawberry plants of different varieties: (a) stem thickness; (b) crown width; (c) number of leaves; (d) number of inflorescences. Different letters in the same column indicate significant differences (p < 0.05) based on Duncan’s multiple range test. ns represents no significant difference at the 5% level.
3.3. Chlorophyll Content
Chlorophyll content directly influences the efficiency of photosynthesis in plants with regard to light energy absorption. Previous studies have established a positive correlation between chlorophyll content and the rate of photosynthesis [35]. Exposure to nocturnal low-temperature stress can reduce the concentration of photosynthetic pigments in plant leaves. This reduction affects the plants’ normal light energy absorption and disrupts the subsequent transmission, dissipation, and distribution of light energy. Consequently, it leads to alterations in chlorophyll fluorescence parameters, disrupting the normal course of photosynthesis [36]. In October, the chlorophyll content in Kaorino was significantly higher than in other varieties at 2.11 mg/g (Table 1), while Snow White had the lowest chlorophyll content at 1.93 mg/g. From October to December, as temperatures dropped sharply, the chlorophyll content of Snow White also declined rapidly and had decreased to only 1.66 mg/g by December. This reduction may be attributed to either decreased chlorophyll synthesis under nocturnal low-temperature stress or a loss of chloroplast activity due to external factors, or possibly a combination of both.

Table 1.
Chlorophyll contents of different strawberry varieties in late December.
3.4. Net Photosynthetic Rate
Photosynthesis is a complex metabolic pathway that converts light energy into chemical energy and provides the primary carbon source for dry matter accumulation in plants. Therefore, leaf photosynthetic capacity is a key determinant of fruit yield [37]. The daily curves of photosynthetic rates of all three varieties of strawberry in October show a single peak (Figure 3a). After sunrise at 6:00, the photosynthetic rates of all three varieties of strawberry leaves gradually increased, peaked at 12:00 and, respectively, reached 21.48 μmol·m−2s−1, 16.08 μmol·m−2s−1, and 12.03 μmol·m−2s−1. After that, the net photosynthetic rate of strawberry leaves gradually decreased and became negative at night due to respiration. The overall net photosynthetic rates of the leaves of three varieties decreased in the following order: Kaorino > Benihoppe > Snow White.

Figure 3.
Diurnal variation in photosynthetic rate of different strawberry varieties. (a) October; (b) November; (c) December. Black line on X axis represents dark period.
The daily photosynthetic rate curves of the three varieties of strawberry in November show a single peak (Figure 3b). The photosynthetic rate of the three varieties gradually increased after sunrise at 7:00 and peaked at 12:00. The photosynthetic rate of Kaorino reached 20.25 μmol·m−2s−1 and was the highest, followed by Benihoppe and Snow White. The photosynthetic rates of Benihoppe and Snow White were, respectively, 79.9% and 55.2% of that of Kaorino. After that, the net photosynthetic rate of strawberry leaves gradually decreased and was negative at night due to respiration. The overall net photosynthetic rate decreased in the following order: Kaorino > Benihoppe > Snow White.
The daily photosynthetic rate curves of three varieties of strawberry in December show a single peak (Figure 3c). The photosynthetic rate of the three varieties gradually increased after sunrise at 8:00 and peaked at 12:00. The photosynthetic rate of Kaorino was the highest and reached 19.21 μmol·m−2s−1, 16.91 μmol·m−2s−1, and 11.55 μmol·m−2s−1. After that, the net photosynthetic rate of strawberry leaves gradually decreased. The overall net photosynthetic rate decreased in the following order: Kaorino > Benihoppe > Snow White.
CO2 assimilation is the absorbed CO2 minus released CO2 by plants in photosynthesis [38]. The CO2 assimilation rate of Kaorino in October, November, and December was the highest, reaching 1209 mmol·m−2d−1, 945 mmol·m−2d−1, and 722 mmol·m−2d−1, respectively, followed by Benihoppe (Figure 4). The CO2 assimilation rate of Benihoppe in October, November, and December, respectively, reached 58.1%, 68.5%, and 86.2% of the corresponding values for Kaorino. The CO2 assimilation rate of Snow White in October, November, and December was the lowest, reaching, respectively, 50.6%, 53.9%, and 77.7% of the corresponding values for Kaorino. The CO2 assimilation rate gradually decreased from October to December due to the influence of various environmental factors such as the gradually decreasing temperature and daytime.

Figure 4.
Variations in CO2 assimilation rates of different varieties of strawberry in different months. Only three leaves of each variety of strawberry were analyzed in the study, so an analysis of significant differences was not performed.
3.5. Chlorophyll Fluorescence Imaging
Chlorophyll fluorescence imaging allows for the acquisition of fluorescence intensity data from different regions of plant leaves, facilitating the analysis of spatial distribution and heterogeneity in leaf photosynthesis [39,40]. Low-temperature stress typically inhibits the efficiency of PSII photochemical reactions in strawberry leaves [41,42]. This leads to a diminished proportion of absorbed light energy being allocated to PSII photochemical reactions, increasing the unregulated energy dissipation in PSII and causing PSII photoinhibition [43,44]. In the fluorescence images (Figure 5), the blue color that is visible along the veins does not indicate high photochemical activity but rather autofluorescence from cyclic phenolic compounds and lignin-like substances that are present in vascular tissues, which emit fluorescence across a broad spectral range. Therefore, the blue signal in the veins originates from non-photosynthetic vascular tissues, whereas the surrounding mesophyll represents the actual photosynthetic area.

Figure 5.
Chlorophyll fluorescence images of different strawberry varieties in late December. Fv/Fm, maximum quantum efficiency of PSII in the dark-adapted state; Y(II), effective quantum yield of PSII under actinic light; NPQ, non-photochemical quenching coefficient reflecting regulated thermal energy dissipation; qP, photochemical quenching coefficient indicating the fraction of open PSII reaction centers. The color scale represents the relative intensity of fluorescence parameters; red color indicates low values, while yellow, green, and blue colors represent progressively higher values.
In the Fv/Fm images, Kaorino appears dark blue, indicating higher maximum photochemical efficiency, whereas Snow White shows a blue color that is interspersed with green and yellow spots, signifying lower maximum photochemical efficiency. This suggests that after sufficient dark adaptation, Kaorino leaves have higher photochemical efficiency at low temperatures and are more resilient to nocturnal low-temperature stress. Additionally, the green color at the petiole of Snow White could imply that low nocturnal temperatures have adversely affected the photosystem II cells there.
In the Y(II) images, the larger dark blue area in Kaorino suggests significantly higher photochemical efficiency compared to Benihoppe and Snow White under illuminated conditions. This implies that the actual quantum efficiency of the PSII reaction center in Kaorino leaves is higher in the presence of action light. A visible spatial heterogeneity within the Y(II) levels in Kanno leaves is also observed, with stronger activity in the central leaf region and weaker activity at the margins.
For NPQ images, the extensive dark blue region in Snow White indicates strong non-photochemical quenching abilities, suggesting protection for photosystem II from excessive light energy. In contrast, Kaorino predominantly appears green, implying lower non-photochemical quenching activity at low temperatures. Snow White shows a distinct purple patch at the leaf edges, indicating higher non-photochemical quenching activity in that region.
In qP images, the larger blue area in Kaorino suggests that its reaction centers are more open, utilizing light energy for photochemical reactions more efficiently. There is also noticeable spatial heterogeneity within Kaorino leaves, with a blue color in the central region and a green color at the edges, indicating less efficient energy conversion in the leaf margins.
The quantitative analysis of chlorophyll fluorescence parameters further confirmed these visual observations (Figure 6), revealing that Kaorino exhibited the highest Fv/Fm and Y(II) values, whereas Snow White showed elevated NPQ and lower qP, indicating stronger photochemical efficiency and better PSII stability in Kaorino under nocturnal low-temperature stress.

Figure 6.
Chlorophyll fluorescence parameters of different strawberry varieties in late December. (a) Fv/Fm; (b) YII; (c) NPQ; (d) qP. Different lowercase letters above the bars indicate significant differences among varieties (p < 0.05).
To further investigate the activation characteristics of the photosynthetic apparatus around sunrise in different varieties, we continuously monitored the temporal changes in Y(II), qP, and ETR after uncovering the thermal insulating quilt.
Y(II) dynamic images showed that Kaorino reached a relatively high level within 20 min after light exposure (Figure 7), with black areas (low activity) rapidly decreasing, indicating that its PSII could quickly activate and enter an efficient photochemical state, possessing rapid photosynthetic induction and efficient light energy conversion. Benihoppe started with lower Y(II), activating more slowly but showing a steadily increasing trend. Snow White recovered the slowest, with significant low-activity areas still being present after 50 min, reflecting weak recovery capacity of its photosynthetic apparatus after low nocturnal temperatures.

Figure 7.
Changes in Y(II) among different strawberry varieties.
The dynamic changes in qP further supported these patterns (Figure 8). In Kaorino, qP increased significantly within 10 min of illumination, indicating that PSII reaction centers quickly entered the open state and initiated electron transport. In Benihoppe, qP increased more gradually, whereas Snow White showed a clear lag, with qP beginning to rise only after about 30 min. This suggests that Snow White has poorer PSII activation and slower initiation of the electron transport chain, with a lower proportion of open reaction centers after low nocturnal temperatures.

Figure 8.
Changes in qP among different strawberry varieties.
The electron transport rate (ETR) is a parameter reflecting the PSII electron transport rate and an important indicator of photosynthetic activity. Its dynamic changes further confirmed the above trends (Figure 9). In Kaorino, the ETR increased rapidly after light exposure, indicating a strong capacity to quickly re-establish photosynthetic electron flow. In Benihoppe, the ETR increased steadily, whereas in Snow White, the increase was slow and limited in magnitude, indicating overall low photosynthetic electron transport efficiency.

Figure 9.
Changes in ETR among different strawberry varieties. Different letters in the same column indicate significant differences (p < 0.05) based on Duncan’s multiple range test, whereas the same letter indicates no significant difference.
3.6. Phenological Period
The phenological period of strawberry indicates various stages of growth and development [45]. Among the three varieties of strawberries, planted at the same time, the budding stage of Kaorino started on 24 September and was 10 days earlier than that of Benihoppe and 52 days earlier than that of Snow White (Table 2). From 4 October, the three varieties entered the initial flowering stage. The initial flowering stage of Kaorino was the earliest, followed by Benihoppe. The initial flowering stage of Snow White, on 27 November, was the latest. The ripening stage is one of the main factors influencing the price of strawberries. The earlier the ripening stage is, the more advantageous the price of strawberries is. Kaorino, Benihoppe, and Snow White could be harvested, respectively, at 84, 100, and 141 days after planting. The ripening stage of Kaorino was shorter, followed by Benihoppe. The ripening stage of Snow White was longer, indicating that Kaorino was suitable for cultivation in high-latitude cold regions.

Table 2.
Phenological periods of different strawberry varieties.
3.7. Yield
The yields of different varieties of strawberry were different (Table 3). From November 2022 to January 2023, the yields of all three varieties of strawberry were relatively low. Snow White had no yield in November or December due to its late ripening stage, and its yield in January was only 20.31 g/plant, which was lower than those of the other two varieties. Benihoppe had a medium yield, and the yield of Kaorino was the highest. The yields of Kaorino from November to January were, respectively, 26.22 g/plant, 45.94 g/plant, and 62.89 g/plant, which were significantly higher than those of the other two varieties.

Table 3.
Yields of different strawberry varieties.
From February to April, the yields of the three varieties were quite high, accounting for more than 70% of the total yield. Especially, the yield of Snow White in these three months accounted for 90.5% of the total yield. The total yield of Kaorino was 472.08 g/plant, which was significantly higher than that of the other two varieties. The total yield of Benihoppe was 117.11 g/plant less than that of Kaorino. The total yield of Snow White was 214.79 g/plant, which was the lowest and only 45.5% of that of Kaorino. The total yield of Snow White was the lowest. In general, the yield of Kaorino was the highest, displaying the advantage of abundant production, followed by Benihoppe and Snow White.
3.8. Comprehensive Evaluation of Strawberry Nocturnal Cold Tolerance Based on Composite Indices
In this study, the Photosynthesis–Fluorescence Index (PFI), the Production–Phenotype Index (PPI), and the Nocturnal Cold Tolerance Index (NCTI) were constructed to evaluate the overall performance of Kaorino, Benihoppe, and Snow White under low-temperature conditions.
The PFI primarily characterizes the photosynthetic rate and PSII activity maintenance capacity after low nocturnal temperatures. A higher PFI value indicates rapid photosynthetic initiation, lower photoinhibition, and stronger PSII functional maintenance in the cold. The PFI of Kaorino was significantly higher than the other varieties (Figure 10), indicating rapid photosynthetic induction, high PSII activity, and efficient photochemistry after nocturnal low-temperature conditions. In contrast, Snow White had the lowest PFI, suggesting that its photosynthetic apparatus was severely impaired by low nocturnal temperatures.

Figure 10.
Radar plots and score heatmaps of cold tolerance indices for three strawberry varieties.
The PPI reflects growth vigor and early yield performance during the low-temperature period. Varieties with high PPI values possess strong growth vitality, early maturity potential, and economic competitiveness under cold conditions. Kaorino again showed the highest PPI, indicating strong growth vigor, good earliness, and high early yield under low temperatures. Snow White had the lowest PPI, which was consistent with its slow growth and low yield.
To comprehensively assess both low-temperature adaptability and economic performance, the NCTI was constructed as a weighted integration of PFI and PPI. Kaorino exhibited the highest NCTI value (4.7), which was clearly superior to those of Benihoppe (0.24) and Snow White (−4.9). This indicates that Kaorino holds advantages in both physiological cold tolerance and production efficiency, thus leading to higher productivity.
To further verify the practical utility of the composite indices, we analyzed the correlations between the PFI, PPI, NCTI, and total yield (Figure 11). The results showed that the PFI, PPI, and NCTI were all strongly and positively correlated with total yield (R2 = 0.97, 0.98, and 0.98, respectively), indicating that these composite indices can effectively predict cultivar yield performance under nocturnal low-temperature conditions. Kaorino achieved the highest values for all three indices, as well as for total yield, further confirming its superior cold tolerance and high-yield potential.

Figure 11.
Correlations between PFI, PPI, NCTI, and total yield. (a) PFI; (b) PPI; (c) NCTI.
4. Discussion
4.1. Effects of Cultivar Differences on Overwintering Production of Strawberry
In this study, the three strawberry varieties exhibited pronounced differences in phenology, growth, photosynthetic performance, and yield, which directly determined their suitability and economic value in overwinter production.
With respect to phenology, the cultivar differences were particularly striking. Kaorino reached its first fruit harvest in mid-November, enabling it to capitalize on the seasonal market gap for winter fruits. In contrast, the first fruit of Snow White ripened nearly two months later than that of Kaorino, thereby missing the optimal price period and markedly reducing its economic return. Flower bud differentiation is the consequence of the accumulation and coordination of nutrients, hormones, and genetic material in the cell cluster at the growing point, and the bud differentiation time is affected by many factors, such as light, temperature, nutritional state, and endogenous hormones [46,47]. Due to differences in genetic background, varieties vary intrinsically in their responsiveness to low-temperature induction. The early flowering and early ripening characteristics of Kaorino suggest that it requires a relatively lower level of chilling accumulation or a lower photoperiod threshold to trigger flower bud differentiation, or that it possesses higher sensitivity to inductive environmental cues. As a result, it can initiate reproductive growth rapidly once external conditions become favorable. In contrast, Snow White remained in vegetative growth for an extended period after transplanting and even showed growth stagnation under low temperatures. This indicates that it responds more slowly to the low-temperature and short-day conditions of winter, requiring a longer period of chilling exposure to complete flower bud differentiation.
Differences among varieties were also reflected in the low-temperature response and recovery capacity of the photosynthetic apparatus [48]. Under low nocturnal temperatures, the photosynthetic machinery (including temperature-sensitive enzymatic processes and thylakoid membrane-associated regulation) becomes functionally constrained, which leads to a reduced electron transport flux and photochemical efficiency, thereby limiting ATP production and carbon assimilation pathways and constraining photosynthetic recovery at dawn. Consequently, after sunrise, plants must rapidly activate PSII and re-establish the electron transport chain under physiologically unfavorable conditions. Chlorophyll fluorescence kinetics and imaging analyses showed that Kaorino not only maintained a high maximum photochemical efficiency (Fv/Fm) under cold stress but also exhibited a rapid photosynthetic induction after uncovering the thermal insulating quilt in the early morning, with both Y(II) and qP increasing sharply upon light exposure. This indicates that the photosynthetic machinery of Kaorino retained strong structural integrity and functional flexibility after low nocturnal temperatures, enabling rapid activation of photochemical reactions and prompt initiation of the electron transport chain, thereby efficiently utilizing transient light energy for carbon assimilation. In contrast, the PSII reaction centers of ‘Snow White’ showed signs of photoinhibition, and its photosynthetic induction was markedly delayed, suggesting that its photosynthetic apparatus is more vulnerable to low-temperature damage and possesses weaker recovery capacity. These physiological differences ultimately manifested in dry matter accumulation, which directly influenced vegetative growth vigor and fruit yield. Overall, the differences in cold tolerance among the varieties reflect the integrated expression of genetically determined physiological mechanisms under low-temperature stress and fundamentally determine their adaptability and economic performance in overwinter production.
4.2. Conventional Screening Methods for Cold-Tolerant Strawberry Varieties
Selecting cold-tolerant varieties is a fundamental approach to overcoming the challenges of overwinter strawberry production in high-latitude cold regions. A key requirement is the establishment of a scientific and efficient screening system. Conventional approaches for evaluating cold tolerance in strawberry mainly rely on morphological traits, physiological and biochemical indicators, and phenological observations [49,50]. Although these methods offer useful guidance, they also present clear limitations.
The growth performance of plants under low-temperature conditions is the most intuitive basis for selection. In this study, we measured morphological traits such as stem diameter, canopy width, leaf number, and inflorescence number. The results showed that Kaorino exhibited significantly greater stem thickness, larger canopy spread, and more inflorescences than Benihoppe and Snow White, reflecting its stronger vegetative vigor and reproductive potential—traits that are generally associated with environmental adaptability and stress resistance. However, despite producing the greatest number of leaves in the mid-to-late growth stages, Snow White did not exhibit correspondingly higher cold tolerance or greater yield. This suggests that single or isolated morphological traits may be misleading and are insufficient to fully reflect a plant’s cold tolerance or photosynthetic productivity. Although morphological indicators are simple and practical for preliminary field assessment, they represent the final expression of stress outcomes rather than the underlying physiological mechanisms that are responsible for these outcomes.
Physiological and biochemical indicators are commonly used to assess membrane stability, metabolic balance, and photosynthetic capacity [51,52,53]. In this study, the chlorophyll content of all varieties declined from October to December as temperatures dropped. Kaorino exhibited the smallest reduction and maintained the highest chlorophyll level in December (2.02 mg g−1). In contrast, Snow White showed the most pronounced decline, falling from 1.93 mg g−1 to 1.66 mg g−1. This indicates that chlorophyll content serves as an indicator of leaf senescence and photodamage under cold stress. Low temperatures disrupt chloroplast structure and inhibit chlorophyll synthesis, leading to declines in chlorophyll content [22]. However, the chlorophyll content only reflects pigment abundance and does not reveal whether absorbed light energy is effectively utilized for electron transport. Even with similar chlorophyll levels, varieties may differ greatly in their light-use efficiency or speed of photosynthetic induction [54]—critical aspects that conventional physiological indicators fail to capture.
Phenology links varietal characteristics with economic outcomes [55]. In this study, Kaorino displayed distinct early-maturing habits, with earlier budding, flowering, and fruit ripening than the other varieties. In overwinter production systems, early-ripening varieties gain a clear advantage by entering the high-price winter market, significantly enhancing profitability. Therefore, phenology serves as a practical indicator for cultivar selection. However, phenology represents cumulative responses to prior environmental conditions and does not reveal the physiological status during the low-temperature period. Even if a plant eventually flowers and fruits, severe impairment or slow recovery of its photosynthetic system during cold periods can reduce overall growth and yield.
Although these traditional methods are widely used in strawberry cold tolerance evaluation, they have several limitations. The indicators are often independent of one another and lack unified quantitative criteria, making it difficult to form an integrated evaluation system. Moreover, most indicators capture static or end-point measurements and cannot reflect the dynamic physiological responses and recovery processes under cold stress. In addition, traditional evaluations are often poorly correlated with final economic yield, which may result in the selection of varieties that perform poorly in actual production. Therefore, there is a critical need to establish a comprehensive evaluation framework that integrates dynamic physiological responses with agronomic and production-related traits to more accurately assess cold tolerance in strawberry varieties.
4.3. Construction and Application Value of the Composite Cold Tolerance Index System
Cold tolerance in plants is a complex trait involving multiple physiological and agronomic dimensions, including the stability of the photosynthetic apparatus, energy metabolism balance, coordinated vegetative and reproductive development, and final economic yield [56]. Based on a systematic analysis of the photosynthetic responses and production performance of strawberry varieties under nocturnal low-temperature stress, this study innovatively developed a composite evaluation system comprising the Photosynthesis–Fluorescence Index (PFI), the Production–Phenotype Index (PPI), and the Nocturnal Cold Tolerance Index (NCTI). This framework integrates both physiological mechanisms and agronomic performance, enabling a multidimensional assessment that links cold-response physiology with actual economic outcomes.
The PFI is designed to characterize the initiation speed, operational efficiency, and photochemical stability of the photosynthetic system after low nocturnal temperatures. Fv/Fm12, representing the maximum photochemical efficiency after dark adaptation, serves as a fundamental indicator of PSII structural integrity, and declines in this parameter often signify photoinhibition or irreversible damage. YII_t50 and YII_slope20 together describe the recovery dynamics from the suppressed photosynthetic state induced by low nocturnal temperatures, with the former reflecting recovery duration and the latter representing the initial induction rate. Together, they reveal the plant’s capacity to capture and utilize transient morning light under sharp diurnal temperature fluctuations. YII_AUC50 quantifies cumulative photochemical efficiency over time and represents the sustained operational capacity of PSII. Pmax_Dec indicates the peak carbon assimilation rate during the coldest period. After Z-score normalization, these five indicators were linearly integrated to form a single PFI value reflecting the cultivar’s ability to maintain and rapidly initiate photosynthesis under low nocturnal temperatures.
The PPI evaluates cold tolerance from the perspective of agronomic performance and economic productivity. This index emphasizes sustained growth, earliness, and preservation of photosynthetic function during the cold period, directly linking physiological traits to production efficiency. The early yield percentage (EY%) is a key indicator that translates physiological cold tolerance into economic value. A high EY% indicates strong production capacity during the high-price winter and Spring Festival markets. The composite growth index (S), combining crown width, stem thickness, and number of inflorescences, reflects both vegetative and reproductive development under low temperatures, with strong vigor providing structural support for high yield. The chlorophyll retention ratio (Chl_ratio) reflects the stability of photosynthetic function under low temperatures, with declines indicating functional degradation and early senescence. Varieties with high PPI values therefore demonstrate robust low-temperature growth vigor, early maturity, and strong economic competitiveness.
As the final integrative indicator, the NCTI combines physiological mechanisms and production performance into a single composite score. The weighting coefficients were assigned according to the relative contribution of physiological stability and agronomic performance to cold tolerance. Because photosynthesis is the sole source of energy for plant growth and yield formation [57], maintaining the functionality of the photosynthetic system (PFI) is fundamental to cold tolerance and was therefore assigned a weight of 60%. As agricultural production ultimately aims to maximize economic return, production-related performance (PPI) accounted for the remaining 40%.
More broadly, the rationale of our composite indices aligns with integrative evaluation strategies that have been extensively used in other crops, where chlorophyll fluorescence and stress-related traits are synthesized into a unified metric to facilitate ranking. In Brassica vegetables, the performance index on an absorption basis (PIabs), derived from OJIP chlorophyll fluorescence kinetics, has been used for evaluation of cold tolerance [25]. Likewise, multi-trait stress tolerance indices have been used to compare performance under combined heat–drought stress in wheat [26], and fluorescence-based comprehensive evaluation has been used to rank peanut genotypes for seedling-stage cold resistance [27]. These studies collectively support the value of integrating multiple physiological signals into concise indices. In this study, we further extend this logic by explicitly linking cold-response physiology to yield-related outcomes, thereby improving interpretability and practical utility for cultivar screening under nocturnal low-temperature conditions.
Correlation analyses revealed that the PFI, PPI, and NCTI were all highly and positively correlated with total yield (R2 > 0.97). This demonstrates that the composite index system is not only conceptually coherent but also highly predictive of actual production performance. The NCTI framework thus provides a reliable quantitative tool for rapidly and accurately identifying varieties that are suitable for overwinter production in high-latitude cold regions.
5. Conclusions
This study established a composite evaluation model for assessing strawberry varieties’ tolerance to low nocturnal temperatures in unheated solar greenhouses in high-latitude cold regions, integrating the Photosynthesis–Fluorescence Index (PFI), the Production–Phenotype Index (PPI), and the Nocturnal Cold Tolerance Index (NCTI). Among the three varieties, Kaorino exhibited the strongest nocturnal low-temperature adaptability, confirming its suitability as the preferred cultivar for overwinter production in unheated solar greenhouses in high-latitude cold regions. Benihoppe exhibited moderate tolerance to nocturnal low-temperature stress and may serve as an alternative cultivar. Snow White displayed low photosynthetic efficiency, slow growth, and poor yield under low nocturnal temperatures, making it unsuitable for overwinter production.
Although the proposed evaluation system effectively differentiated cultivar responses, the current study is preliminary due to the limited sample size and reliance on single-season data. Future work will include multi-year validation, expansion of cultivar diversity, and incorporation of biochemical and molecular indicators to enhance the predictive capacity and robustness of the evaluation framework.
Author Contributions
Conceptualization, H.C. and J.W.; methodology, J.W.; software, J.L. and Q.H.; validation, Y.L.; formal analysis, Q.H.; investigation, H.C.; resources, J.W.; data curation, Q.Z.; writing—original draft preparation, H.C.; writing—review and editing, H.C. and J.W.; visualization, H.C.; supervision, J.W. and H.Z.; project administration, Q.Z., J.W. and H.Z.; funding acquisition, H.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the China Agricultural Research System of MOF and MARA, grant number CARS-24-G-06; the Jilin Provincial Science and Technology Development Plan, grant number 20240303036NC; and the Jilin Agricultural Research System, grant number JLARS-2025-120307.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Li, Q.; Cao, B.; Wang, X.; Wu, J.J. Systematic Water-saving Management for Strawberry in Basic Greenhouses Based on the Internet of Things. Appl. Eng. Agric. 2021, 37, 205–217. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Temocico, G.; Fierascu, I.; Ortan, A.; Babeanu, N.E. Fragaria Genus: Chemical Composition and Biological Activities. Molecules 2020, 25, 498. [Google Scholar] [CrossRef] [PubMed]
- Symington, H.A.; Glover, B.J. Strawberry Varieties Differ in Pollinator-relevant Floral Traits. Ecol. Evol. 2024, 14, e10914. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.B.; Han, Q.Y.; Liu, Y.N.; Lu, S.K.; Zhang, Z.J.; Yang, M.L.; Wang, J.F. Effects of Laser Supplementary Lighting on Growth, Photosynthetic Characteristics, and Yield of Strawberry in Solar Greenhouse. J. Jilin Agric. Univ. 2025, 47, 663–671. [Google Scholar] [CrossRef]
- Chou, Z.; Lei, C.; Cai, X.; Li, Y.; Zeng, D.; Gong, S.; Wang, J.; Gong, Z. Strawberry Performance and Rhizospheric Health Were Efficiently Improved After Long-Term Sheep Manure Organic Fertilizer Application. Horticulturae 2025, 11, 1000. [Google Scholar] [CrossRef]
- Jiao, Y.; Wang, Y.K.; Miao, B.C. Ecological and Efficient Cultivation Techniques of Protected Strawberry with Multi-layer Mulching. Henan Agric. 2019, 1, 11. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Z.M.; Jiang, X.M. Overwintering Greenhouse Strawberry Cultivation Techniques in Heilongjiang Province. Bot. Res. 2019, 8, 218–221. [Google Scholar] [CrossRef]
- Hoffmann, R.; Muttarak, R.; Peisker, J.; Stanig, P. Climate Change Experiences Raise Environmental Concerns and Promote Green Voting. Nat. Clim. Change 2022, 12, 148–155. [Google Scholar] [CrossRef]
- Simpson, D. The Economic Importance of Strawberry Crops. In The Genomes of Rosaceous Berries and Their Wild Relatives; Springer: Cham, Switzerland, 2018; pp. 1–7. [Google Scholar] [CrossRef]
- Redondo-Gómez, S.; García-López, J.V.; Mesa-Marín, J.; Pajuelo, E.; Rodriguez-Llorente, I.D.; Mateos-Naranjo, E. Synergistic Effect of Plant-Growth-Promoting Rhizobacteria Improves Strawberry Growth and Flowering with Soil Salinization and Increased Atmospheric CO2 Levels and Temperature Conditions. Agronomy 2022, 12, 2082. [Google Scholar] [CrossRef]
- Cui, M.Y.; Minh, D.P.; Hwang, H.; Chun, C. Flower Development and Fruit Malformation in Strawberries after Short-term Exposure to High or Low Temperature. Sci. Hortic. 2021, 288, 110308. [Google Scholar] [CrossRef]
- Moreira, A.F.P.; Resende, J.T.V.; Shimizu, G.D.; Hata, F.T.; Nascimento, D.d.; Oliveira, L.V.B.; Zanin, D.S.; Mariguele, K.H. Characterization of Strawberry Genotypes with Low Chilling Requirement for Cultivation in Tropical Regions. Sci. Hortic. 2022, 292, 110629. [Google Scholar] [CrossRef]
- Hayat, F.; Sun, Z.; Ni, Z.; Iqbal, S.; Xu, W.; Gao, Z.; Qiao, Y.; Tufail, M.A.; Jahan, M.S.; Khan, U.; et al. Exogenous Melatonin Improves Cold Tolerance of Strawberry (Fragaria × ananassa Duch.) through Modulation of DREB/CBF-COR Pathway and Antioxidant Defense System. Horticulturae 2022, 8, 194. [Google Scholar] [CrossRef]
- Huang, C.; Qin, N.; Sun, L.; Yu, M.; Hu, W.; Qi, Z. Selenium Improves Physiological Parameters and Alleviates Oxidative Stress in Strawberry Seedlings under Low-temperature Stress. Int. J. Mol. Sci. 2018, 19, 1913. [Google Scholar] [CrossRef]
- Khammayom, N.; Maruyama, N.; Chaichana, C. The Effect of Climatic Parameters on Strawberry Production in a Small Walk-In Greenhouse. AgriEngineering 2022, 4, 104–121. [Google Scholar] [CrossRef]
- Paris, B.; Vandorou, F.; Balafoutis, A.T.; Vaiopoulos, K.; Kyriakarakos, G.; Manolakos, D.; Papadakis, G. Energy Use in Greenhouses in the EU: A Review Recommending Energy Efficiency Measures and Renewable Energy Sources Adoption. Appl. Sci. 2022, 12, 5150. [Google Scholar] [CrossRef]
- Huang, Y.; Abbas, Q.; Sharif, M. Addressing Agricultural Energy Poverty to Enhance Farmers’ Profitability and Productivity: Policy Interventions for Global Food Security Challenges. Energy Policy 2025, 206, 114696. [Google Scholar] [CrossRef]
- Islam, S. Agriculture, Food Security, and Sustainability: A Review. Explor. Foods Foodom. 2025, 3, 101082. [Google Scholar] [CrossRef]
- Luo, G.J.; Xue, L.; Xu, W.J.; Zhao, J.; Wang, J.; Ding, Y.; Luan, K.; Lei, J.J. Breeding Decaploid Strawberry with Improved Cold Resistance and Fruit Quality. Sci. Hortic. 2019, 251, 1–8. [Google Scholar] [CrossRef]
- Paparozzi, E.T.; Meyer, G.E.; Schlegel, V.; Blankenship, E.E.; Adams, S.A.; Conley, M.E.; Loseke, B.; Read, P.E. Strawberry Cultivars Vary in Productivity, Sugars and Phytonutrient Content when Grown in a Greenhouse during the Winter. Sci. Hortic. 2018, 227, 1–9. [Google Scholar] [CrossRef]
- Weber, C.A. Performance of Strawberry Varieties Developed for Perennial Matted-Row Production in Annual Plasticulture in a Cold Climate Region. Agronomy 2021, 11, 1407. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Ashraf, M. Proline Alleviates Abiotic Stress Induced Oxidative Stress in Plants. J. Plant Growth Regul. 2023, 42, 4629–4651. [Google Scholar] [CrossRef]
- Koehler, G.; Wilson, R.C.; Goodpaster, J.V.; Sønsteby, A.; Lai, X.; Witzmann, F.A.; You, J.-S.; Rohloff, J.; Randall, S.K.; Alsheikh, M. Proteomic Study of Low-Temperature Responses in Strawberry Cultivars (Fragaria × ananassa) That Differ in Cold Tolerance. Plant Physiol. 2012, 159, 1787–1805. [Google Scholar] [CrossRef]
- Jiang, N.; Yang, Z.; Zhang, H.; Xu, J.; Li, C. Effect of Low Temperature on Photosynthetic Physiological Activity of Different Photoperiod Types of Strawberry Seedlings and Stress Diagnosis. Agronomy 2023, 13, 1321. [Google Scholar] [CrossRef]
- Dai, Y.; Yuan, L.; Zhang, S.; Wang, J.; Xie, S.; Zhao, M.; Chen, G.; Sun, R.; Wang, C. Comprehensive Evaluation for Cold Tolerance in Wucai (Brassica campestris L.) by the Performance Index on an Absorption Basis (PIabs). Agronomy 2019, 9, 61. [Google Scholar] [CrossRef]
- Bhandari, R.; Paudel, H.; Alharbi, S.A.; Ansari, M.J.; Poudel, M.R.; Neupane, M.P.; Solanki, P.; Kushwaha, U.K.S. Evaluating stress tolerance indices for their comparative validity to assess terminal heat stress and heat-drought tolerance of winter wheat (Triticum aestivum L.) genotypes. J. Agric. Food Res. 2024, 18, 101506. [Google Scholar] [CrossRef]
- Ye, L.; Wang, T.; Wu, R.; Zheng, C.; Zhan, L.; Chen, J.; Guo, S.; Chen, Y. Evaluation of Cold Resistance at Seedling Stage for 70 Peanut Genotypes Based on Photosynthetic Fluorescence Characteristics. Agronomy 2024, 14, 1699. [Google Scholar] [CrossRef]
- Yamazaki, K. Nutrient Solution Culture; Pak-kyo Co.: Tokyo, Japan, 1982; p. 41. [Google Scholar]
- Zhao, M.Z. Strawberry Germplasm Description Specifications and Data Standards; China Agricultural Press: Beijing, China, 2006. [Google Scholar]
- Tang, L.N.; Yang, X.Y. New Technology of High Quality and High Yield of Strawberry; Golden Shield Press: Beijing, China, 2013. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts: Polyphenoloxidase in Beta Vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Takei, M. The Cultivation of Strawberry in Japan; Farm Extension Officer: Nagano, Japan, 2010. [Google Scholar]
- Kadir, S.; Sidhu, G.; Al-Khatib, K. Strawberry (Fragaria × ananassa Duch.) Growth and Productivity as Affected by Temperature. HortScience 2006, 41, 1423–1430. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, L.J.; Du, M.Q.; Lian, P. Differences in Main Characters of Different Strawberry Varieties. Hubei Agric. Sci. 2018, 57, 79–83. [Google Scholar] [CrossRef]
- Yang, F.X.; He, H.; Tang, L.; Li, K.Y.; Liu, F.; Chen, J.J. Effects of Suboptimal Low Temperature on Leaf Structure and Photosynthetic Fluorescence Characteristics during the Reproductive Stage of Strawberry. J. China Agric. Sci. Technol. 2025, 27, 51–62. [Google Scholar] [CrossRef]
- Li, C.S.; Wang, M.M.; Chen, X.D.; Yang, C.K. Effects of Low Temperature on Physiological Characteristics of Strawberry. Chin. J. Mid-Low Latit. Mt. Meteorol. 2021, 45, 84–89. [Google Scholar]
- Yang, Y.F.; Yang, M.Y.; Xu, L.Y.; Song, Y.Q.; Tang, Y.W.; Chen, Y.R.; Yu, Q.F.; Xu, J.T. Effects of Different Light Qualities on Growth and Light Utilization Efficiency of Strawberry Grown in Elevated Substrate Systems in Protected Cultivation. Acta Agric. Boreali-Occident. Sin. 2025, 34, 2045–2054. [Google Scholar]
- Gao, B.W.; Ding, S.H.; Chen, X.J.; Wen, X.G.; Tian, L.J.; Lu, Q.T. Strategies for Optimizing Photosynthesis to Improve Agricultural Production Efficiency. Biotechnol. Bull. 2025, 41, 54–63. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll Fluorescence—A Practical Guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Allahverdiyeva, Y.; Suorsa, M.; Rossi, F.; Pavesi, A.; Kater, M.M.; Antonacci, A.; Tadini, L.; Pribil, M.; Schneider, A.; Wanner, G.; et al. Arabidopsis Plants Lacking PsbQ and PsbR Subunits of the Oxygen-evolving Complex Show Altered PSII Supercomplex Organization and Short-term Adaptive Mechanisms. Plant J. 2013, 75, 671–684. [Google Scholar] [CrossRef]
- Sharma, A.; Kumar, V.; Shahzad, B.; Muthusamy, R. Photosynthetic Response of Plants under Different Abiotic Stresses: A Review. J. Plant Growth Regul. 2020, 39, 509–531. [Google Scholar] [CrossRef]
- Popson, D.; D-Silva, S.; Wheeless, K.; Morgan-Kiss, R. Permanent Stress Adaptation and Unexpected High-light Tolerance in the Shade-adapted Chlamydomonas Priscuii. Plants 2024, 13, 2254. [Google Scholar] [CrossRef]
- Xie, P.; Zhao, Y.; Zhao, X.; Xu, L.B.; Wang, K.; Jia, R.D.; Kou, Y.P.; Ge, H.; Wang, W.J.; Yang, S.H. Divergent Photosynthetic Strategies of Lupinus Polyphyllus and Helleborus Viridis during Cold Acclimation and Freezing–thaw recovery. Plants 2025, 14, 607. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Wang, M.T.; Yang, Z.Q.; Zheng, Q.T. Low Temperature and Low Irradiation Induced Irreversible Damage of Strawberry Seedlings. Photosynthetica 2020, 58, 156–164. [Google Scholar] [CrossRef]
- Jin, Z.; Lu, S.; Jiang, J.H.; Li, S.R.; Zhang, N.; Jiang, X.Y.; Wu, F. Advances in Factors and Mechanisms Influencing Flower Bud Differentiation in Horticultural Plants. Acta Hortic. Sin. 2023, 50, 1151–1164. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Y.Q.; Li, J.L.; Xu, H.; Li, W.H.; Li, T. Effects of LED Supplementary Lighting on Production and Leaf Physiological Characteristics of Strawberry Grown in Substrate in Solar Greenhouse. Sci. Agric. Sin. 2025, 58, 975–990. [Google Scholar]
- Symons, G.M.; Chua, Y.J.; Ross, J.J.; Quittenden, L.J.; Davies, N.W.; Reid, J.B. Hormonal Changes during Non-climacteric Ripening in Strawberry. J. Exp. Bot. 2012, 63, 4741–4750. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A. Effect of Low Temperature Stress on Photosynthesis and Allied Traits: A Review. In Physiological Processes in Plants Under Low Temperature Stress; Springer: Singapore, 2022. [Google Scholar] [CrossRef]
- Yang, F.X.; Jin, F.; Yan, X. Comprehensive Evaluation of Cold Tolerance in Different Strawberry Cultivars. J. Fruit Sci. 2010, 27, 368–372. [Google Scholar] [CrossRef]
- Wang, Y.M.; Chen, J.M.; Ju, C.C.; Zheng, Q.L.; Cheng, J.S.; Zhang, K.; Liu, B.F. Physiological Mechanisms of Cold Tolerance in Wild Diploid Fragaria Vesca from China. Mol. Plant Breed. 2025, 1–9. Available online: https://link.cnki.net/urlid/46.1068.S.20250124.1050.002 (accessed on 1 November 2025).
- Wilson, K.E.; Ivanov, A.G.; Öquist, G.; Grodzinski, B.; Sarhan, F.; Huner, N.P.A. Energy Balance, Organellar Redox Status, and Acclimation to Environmental Stress. Botany 2006, 84, 1355–1370. [Google Scholar] [CrossRef]
- Akbari, A.; Barzegar, T.; Rabiei, V.; Nicola, S. Phenylalanine, Cysteine, and Sodium Selenate Alleviate Chilling Injury in Cape Gooseberry (Physalis peruviana L.) Seedlings by Enhancing Antioxidant Activities and Membrane Stability. Horticulturae 2024, 10, 978. [Google Scholar] [CrossRef]
- Jurczyk, B.; Grzesiak, M.; Pociecha, E.; Wlazło, M.; Rapacz, M. Diverse Stomatal Behaviors Mediating Photosynthetic Acclimation to Low Temperatures in Hordeum Vulgare. Front. Plant Sci. 2019, 9, 1963. [Google Scholar] [CrossRef]
- Li, B.D.; Li, Z.C.; Zhu, G.H.; Peng, X.X.; Zhang, Z.S. Effects of Overexpression of Glycolate Oxidase in Chloroplasts on Photosynthesis and Growth of Rice. Biotechnol. Bull. 2025, 41, 87–97. [Google Scholar] [CrossRef]
- Gong, Y.F.; Zhang, Z.P.; Wu, L.L.; Chen, F.; Chang, Y.; Wei, Y.J.; Su, Y.J. Analysis of Agronomic Traits and Filament Yield of Safflower in the Hexi Corridor. Smart Agric. Guide 2025, 5, 45–49. [Google Scholar] [CrossRef]
- Ren, D.Q.; Zhang, S.; Li, Y.J.; Chen, C.L. Physiological Responses and Cold-resistance Evaluation of Dendrobium Officinale from Different Regions under Low-temperature Stress. Guizhou Agric. Sci. 2025, 53, 16–23. [Google Scholar]
- Niinemets, Ü. Within-Canopy Variations in Functional Leaf Traits: Structural, Chemical and Ecological Controls and Diversity of Responses. In Canopy Photosynthesis: From Basics to Applications; Hikosaka, K., Niinemets, Ü., Anten, N., Eds.; Advances in Photosynthesis and Respiration; Springer: Dordrecht, The Netherlands, 2016; Volume 42. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.