Journal Description
Hydrogen
Hydrogen
is an international, peer-reviewed, open access journal on all aspects of hydrogen, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus, Ei Compendex, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17 days after submission; acceptance to publication is undertaken in 4.9 days (median values for papers published in this journal in the second half of 2025).
- Journal Rank: CiteScore - Q1 (Engineering (miscellaneous))
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Journal Cluster of Energy and Fuels: Energies, Batteries, Hydrogen, Biomass, Electricity, Wind, Fuels, Gases, Solar, ESA, Bioresources and Bioproducts and Methane.
Impact Factor:
3.0 (2024);
5-Year Impact Factor:
3.5 (2024)
Latest Articles
Hydrogen’s Role in Decarbonising the Global Energy Sector: An Insightful Perspective
Hydrogen 2026, 7(2), 72; https://doi.org/10.3390/hydrogen7020072 - 28 May 2026
Abstract
►
Show Figures
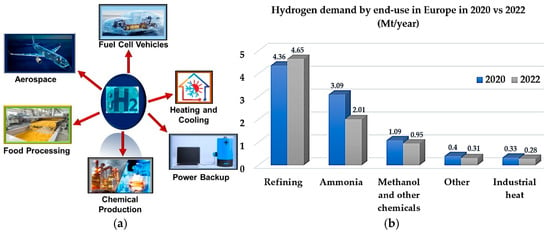
The intensifying climate problem requires substantial decarbonisation in the energy, industry, and transportation sectors, with hydrogen recognised as a crucial energy carrier. The increase in global energy consumption, driven by population growth and industrialisation, challenges the constraints of fossil fuel resources and their
[...] Read more.
The intensifying climate problem requires substantial decarbonisation in the energy, industry, and transportation sectors, with hydrogen recognised as a crucial energy carrier. The increase in global energy consumption, driven by population growth and industrialisation, challenges the constraints of fossil fuel resources and their detrimental impact on CO2 levels. Hydrogen, noted for its high energy density and versatility in generating power from both fossil and renewable sources, acts as a crucial supplement to direct electrification. Currently, worldwide hydrogen production exceeds 100 million tonnes per year, predominantly in the form of “grey hydrogen,” which significantly contributes to CO2 emissions without the use of carbon capture systems. This analysis comprehensively assesses hydrogen’s contribution to decarbonisation, encompassing the entire value chain: production methods, storage options (compressed gas, liquid hydrogen, and complex hydrides), transportation techniques (pipelines, cars, rail, and ammonia carriers), and various uses. Key performance parameters indicate trade-offs concerning energy density, storage, production expenses, and transportation alternatives. Notwithstanding advancements in hydrogen technologies, obstacles persist, encompassing energy penalties, infrastructural requirements, and safety issues. This evaluation highlights the need for coordinated policies and investment to enhance hydrogen’s adaptability, ensuring alignment with direct electrification policies to achieve net-zero emissions by 2050.
Full article
Open AccessArticle
Expert-Based Risk Mapping for Low-Carbon Hydrogen Production Under Geopolitical Constraints: A Case Study of Russia
by
Svetlana Revinova, Konstantin Gomonov, Svetlana Ratner and Artem Shaposhnikov
Hydrogen 2026, 7(2), 71; https://doi.org/10.3390/hydrogen7020071 - 25 May 2026
Abstract
The transition to low-carbon hydrogen is recognized as a priority decarbonization pathway, yet the risk profiles of hydrogen projects remain poorly characterized for non-Western, resource-rich, and geopolitically constrained economies. This study develops and applies a structured expert-based risk mapping framework for low-carbon hydrogen
[...] Read more.
The transition to low-carbon hydrogen is recognized as a priority decarbonization pathway, yet the risk profiles of hydrogen projects remain poorly characterized for non-Western, resource-rich, and geopolitically constrained economies. This study develops and applies a structured expert-based risk mapping framework for low-carbon hydrogen production in Russia. The framework integrates three procedural steps: (1) identification and classification of 21 risk factors across seven thematic groups based on systematic literature analysis; (2) construction of a directed interdependency matrix (7 × 7, ordinal scale 0–2) via structured expert elicitation (n = 10, February 2026); and (3) probability–impact prioritization using the P × S scoring heuristic (both axes on a 1–5 scale, per ISO 31000:2018). Results reveal three critical risk factors (P × S Score ≥ 20): high cost of capital and restricted access to external financing (Score = 24, P = 5, S = 5), dependence on imported electrolyzer components (Score = 20, P = 4, S = 4), and insufficient export infrastructure (Score = 20, P = 5, S = 4). The interdependency matrix identifies economic and financial risks as the primary “accumulator” of systemic influence, receiving maximum incoming impact from all other six groups. Regulatory risks occupy a medium position but exert disproportionate cascading effects on technology choice and project economics. The framework is explicitly designed for transferability to other resource-abundant, capital-constrained economies (Kazakhstan, Iran, Algeria), with structural adaptation conditions specified. Findings are relevant for policymakers, investors, and multilateral stakeholders shaping hydrogen value chains in non-Western contexts.
Full article
(This article belongs to the Special Issue Advances in Hydrogen Production, Storage, and Utilization (2nd Edition))
►▼
Show Figures

Figure 1
Open AccessArticle
Coupled Quantification of Physical and Chemical Inhibition Effects of N2 and CO2 on NH3/H2/air Explosions
by
Di Hu, Guohua Chen, Qiming Xu, Wei Zhou and Xiyue Wang
Hydrogen 2026, 7(2), 70; https://doi.org/10.3390/hydrogen7020070 - 15 May 2026
Abstract
This study investigates the explosion suppression of N2 and CO2 on NH3/H2/air mixtures (NH3:H2 = 4:6, φ = 0.6–1.5) in a 5 L constant volume chamber. Results show that CO2 exhibits superior efficacy
[...] Read more.
This study investigates the explosion suppression of N2 and CO2 on NH3/H2/air mixtures (NH3:H2 = 4:6, φ = 0.6–1.5) in a 5 L constant volume chamber. Results show that CO2 exhibits superior efficacy compared to N2. At φ = 1.0, the addition of 25% CO2 reduces the maximum explosion overpressure and maximum pressure rise rate by 59.28% and 91.00%, respectively, whereas the corresponding reductions caused by 25% N2 are 29.28% and 83.85%. The laminar burning velocity decreases continuously with increasing inhibitor concentration under all investigated conditions, and the reduction caused by CO2 is markedly greater than that caused by N2. Kinetic analysis indicates that the stronger suppression effect of CO2 is mainly associated with its stronger dilution and heat-absorption effects, together with its stronger influence on radical evolution, as reflected by the more pronounced reduction in key radical concentrations. To compare the suppression behaviours of N2 and CO2 under different conditions, three indices—the experimental maximum explosion overpressure suppression efficiency (ST), the theoretical maximum explosion overpressure suppression efficiency (SE), and the operating-condition suppression efficiency (η)—were introduced as comparative parameters. These results provide useful insight into the different suppression behaviours of N2 and CO2 in NH3/H2/air explosions.
Full article
(This article belongs to the Special Issue Innovations in Hydrogen Combustion and Safety)
►▼
Show Figures

Figure 1
Open AccessArticle
Long-Term Exposure in Liquid Hydrogen: Mechanical Properties and Microstructural Investigation of 304 Austenitic Steel After 30 Years of Service
by
Camelia Schulz, Monzer Maarouf, Zahra Abbasi, Elvina Gaisina, Astrid Pundt and Klaus-Peter Weiss
Hydrogen 2026, 7(2), 69; https://doi.org/10.3390/hydrogen7020069 - 14 May 2026
Abstract
Although austenitic steels have been implemented in direct liquid hydrogen (LH2) contact for decades, detailed microstructural and mechanical studies are still rare at a temperature of 20 K and inexistent for long-term exposure in LH2. Therefore, austenitic stainless-steel parts,
[...] Read more.
Although austenitic steels have been implemented in direct liquid hydrogen (LH2) contact for decades, detailed microstructural and mechanical studies are still rare at a temperature of 20 K and inexistent for long-term exposure in LH2. Therefore, austenitic stainless-steel parts, which were in direct contact with LH2, from a container for LH2 transport from the company Linde GmbH that has been in service for over 30 years, was chosen as a material model system for this investigation. In the present work, the possible influence of cryogenic gaseous and liquid H2 (GH2 and LH2) on the micro- and macroscopic as well as mechanical properties of the container was investigated. Monitoring the properties after long-term GH2 and LH2-exposed material assesses the durability and the failure characteristics of these austenitic steels. A mean content of 2.5 ppm H was detected in the container walls after the long-term exposure. The microhardness of the long-term GH2 and LH2 are similar to an H2 non-exposed sample. Based on the SEM investigations, no microstructural change could be detected in the material after long-term H2 exposure and the residual tensile properties are still similar to those of ‘fresh’ non-exposed material. The hydrogen embrittlement (HE) occurred in the container material only after additional thermal H-charging, where the ductility reduced to about 50% at 200 K.
Full article
(This article belongs to the Special Issue Engineering the Global Hydrogen Transition: Materials, Processes, Infrastructure, and Deployment)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Dynamic Control of a PV/T Electrolysis System for Hydrogen and Hot-Water Production: Multi-Regional Analysis with Machine Learning
by
Mohamed Hamdi and Souheil Elalimi
Hydrogen 2026, 7(2), 68; https://doi.org/10.3390/hydrogen7020068 - 13 May 2026
Abstract
This study explores a photovoltaic/thermal (PV/T)-based electrolysis system designed for dual production of hydrogen fuel and domestic hot water (DHW), providing a sustainable energy solution amid rising global emissions. A dynamic rule-based control mechanism with hysteresis thresholds on hydrogen-storage state of charge (SoC)
[...] Read more.
This study explores a photovoltaic/thermal (PV/T)-based electrolysis system designed for dual production of hydrogen fuel and domestic hot water (DHW), providing a sustainable energy solution amid rising global emissions. A dynamic rule-based control mechanism with hysteresis thresholds on hydrogen-storage state of charge (SoC) is implemented to balance electrolyzer operation with intermittent solar availability, maintaining PV/T power outputs while preventing storage overfilling and minimizing start–stop cycling. The system is assessed across 27 geographically diverse cities spanning a wide range of solar irradiation and energy price structures. Annual hydrogen yields range from 20 kg/yr in high-latitude locations (Helsinki, Stockholm) to 33.5 kg/yr in high-irradiation regions (Riyadh, Abu Dhabi), while the levelized cost of hydrogen (LCOH) spans from 6.47 USD/kg (Riyadh) to 22.86 USD/kg (Helsinki). Economically, the system achieves its strongest performance in solar-rich, high-energy-cost environments: Rome records the highest net annual cash flow (858.9 USD/yr) and shortest payback period (2.47 years), followed by Davos, Madrid, Brasília, and Canberra. In contrast, locations with subsidized energy tariffs—such as Algiers, Kyiv, and Tehran—yield low or negative net cash flows, rendering the system economically unviable without policy support. Environmental analysis reveals annual CO2 avoidance ranging from 0.33 ton/yr (Stockholm) to 2.97 ton/yr (Riyadh), with a global mean of 1.095 ton/yr and a combined total of approximately 29.6 tons/yr across all examined sites. A machine learning model is developed to generalize performance predictions across unseen locations, achieving leave-one-out (LOO) R2 values of 0.953 (net cash flow), 0.935 (LCOH), and 0.947 (LCO-DHW), with mean absolute errors below ±1 USD/kg and ±0.03 USD/kWh. The findings confirm that, under fixed capital cost assumptions, local electricity price and solar irradiation are the dominant drivers of economic viability, while grid carbon intensity and solar resource jointly govern environmental performance, with markets offering irradiation above 1500 kWh/m2·yr and electricity prices exceeding 0.2 USD/kWh representing the most promising deployment targets.
Full article
(This article belongs to the Special Issue Hydrogen for a Clean Energy Future)
►▼
Show Figures

Figure 1
Open AccessArticle
Hydrogen Isotope Permeation, Retention, and Embrittlement Response of 310S Austenitic Stainless Steel Under High-Temperature Gaseous Deuterium Charging
by
Yulong Jiang, Zhifu Wang, Jian Li and Zhonghao Heng
Hydrogen 2026, 7(2), 67; https://doi.org/10.3390/hydrogen7020067 - 12 May 2026
Abstract
High-temperature gaseous deuterium charging was used to investigate hydrogen isotope permeation, retention, microstructural stability, and fracture response in 310S austenitic stainless steel. Gas-driven permeation, thermal desorption spectroscopy, two-dimensional diffusion simulation, XRD/EBSD characterization, tensile testing, and fractographic analysis were combined to correlate isotope transport
[...] Read more.
High-temperature gaseous deuterium charging was used to investigate hydrogen isotope permeation, retention, microstructural stability, and fracture response in 310S austenitic stainless steel. Gas-driven permeation, thermal desorption spectroscopy, two-dimensional diffusion simulation, XRD/EBSD characterization, tensile testing, and fractographic analysis were combined to correlate isotope transport with mechanical and fracture behavior. The deuterium permeability and diffusion coefficient followed an Arrhenius relationship, and the diffusion coefficient extrapolated at 673 K was 1.11 × 10−11 m2/s. With increasing charging time, the deuterium distribution evolved from a surface-enriched unsaturated state to an overall near-saturated state with higher retention. Although deuterium charging had little influence on yield strength, ultimate tensile strength, and elongation under the present room-temperature tensile condition, local quasi-cleavage-like facets, secondary cracks, and serrated fracture edges became more evident after charging. These results indicate that the embrittlement response of 310S stainless steel was mainly characterized by localized hydrogen-assisted damage rather than dominant brittle fracture.
Full article
(This article belongs to the Special Issue Advances in Hydrogen Production, Storage, and Utilization (2nd Edition))
►▼
Show Figures

Figure 1
Open AccessReview
Review of the Thermodynamics of Hydrogen Charging in Hydrogen Storage and Transportation Devices
by
Jianhua Yang, Fangyi Han, Wenbin Cheng, Yaqiang Yang, Chaoming Shen, Fushan Li and Meiliang Zhong
Hydrogen 2026, 7(2), 66; https://doi.org/10.3390/hydrogen7020066 - 9 May 2026
Abstract
►▼
Show Figures
High-pressure gaseous hydrogen storage is widely adopted in the hydrogen energy industry chain due to its simplicity, reliability, and economic viability. However, when these systems are subjected to rapid filling, a series of complex thermodynamic behaviors are induced. These have been identified as
[...] Read more.
High-pressure gaseous hydrogen storage is widely adopted in the hydrogen energy industry chain due to its simplicity, reliability, and economic viability. However, when these systems are subjected to rapid filling, a series of complex thermodynamic behaviors are induced. These have been identified as a bottleneck restricting the safety and service life of hydrogen storage and transportation equipment. In this paper, a detailed review is conducted on the recent domestic and international research progress regarding the thermodynamic issues encountered during the charging process of storage devices. Research achievements related to the thermodynamics of the process are systematically classified, summarized and discussed. These achievements are analyzed from four aspects: thermodynamic theoretical models, numerical simulation analysis, experimental testing, and thermal management strategies. The thermodynamic mechanism of the charging process is revealed, and the variation laws of thermodynamic responses during charging are sorted out. Key factors affecting the thermodynamic behaviors of charging are clarified, and the implementation effects of different thermal management strategies are elaborated. Finally, based on the future development trend and prominent potential challenges in high-pressure hydrogen storage, the future development directions of the thermodynamics of hydrogen fueling in storage and transportation devices are explored and prospected.
Full article

Figure 1
Open AccessArticle
Design and Evaluation of High-Safety Differential Pressure Power Generation Technologies for Hydrogen and Ammonia Gas
by
Guohui Song, Xiang Wang, Haiming Gu, Sheng Wang, Jingxin Xu, Cai Liang, Hao Zhao and Lirong Wang
Hydrogen 2026, 7(2), 65; https://doi.org/10.3390/hydrogen7020065 - 8 May 2026
Abstract
The use of differential pressure energy for green hydrogen and ammonia comes with significant safety challenges. Two zero-emission technical schemes—one based on magnetic coupling transmission and another based on dual magnetic fluid seals—were proposed and designed. The energy performance of both schemes was
[...] Read more.
The use of differential pressure energy for green hydrogen and ammonia comes with significant safety challenges. Two zero-emission technical schemes—one based on magnetic coupling transmission and another based on dual magnetic fluid seals—were proposed and designed. The energy performance of both schemes was first analyzed for a DN200 pipe using the DWSIM software (Version 8.6.6). Subsequently, the levelized cost of electricity and the dynamic payback period were evaluated and compared. The results show that the magnetic coupling transmission scheme exhibits relatively low energy efficiency (54.9–61.7%), whereas the scheme based on dual magnetic fluid seals is more complex yet achieves higher energy efficiency (65.8–67.1%). The levelized electricity cost of both schemes under a differential pressure of 0.5 MPa is estimated to be lower than the feed-in tariff of coal-fired power plants in China, and the dynamic payback period is estimated to be less than 5.5 years. Overall, both schemes provide benefits in energy savings and profitability. These schemes warrant further experimental investigation and pilot testing.
Full article
(This article belongs to the Special Issue Hydrogen Energy and Fuel Cell Technology)
►▼
Show Figures

Figure 1
Open AccessHypothesis
Hydrogen-Enriched Saline for Redox Modulation During Hydrosurgical Debridement: A Hypothesis for Promoting Wound Healing
by
Ryosuke Shinkai and Takashi Tomita
Hydrogen 2026, 7(2), 64; https://doi.org/10.3390/hydrogen7020064 - 7 May 2026
Abstract
►▼
Show Figures
Pressure ulcers are chronic wounds characterized by repeated ischemia–reperfusion injury, persistent inflammation, and redox imbalance, in which excessive production of reactive oxygen species (ROS) contributes to delayed healing. Thus, debridement is an essential therapeutic procedure for removing necrotic tissue and biofilm, thereby reconstructing
[...] Read more.
Pressure ulcers are chronic wounds characterized by repeated ischemia–reperfusion injury, persistent inflammation, and redox imbalance, in which excessive production of reactive oxygen species (ROS) contributes to delayed healing. Thus, debridement is an essential therapeutic procedure for removing necrotic tissue and biofilm, thereby reconstructing the wound microenvironment. Recent experimental studies suggest that molecular hydrogen may improve wound healing through attenuation of oxidative stress and modulation of inflammatory responses, while debridement represents a dynamic intervention phase in which redox imbalance may transiently develop. Here, we propose the hypothesis that the use of hydrogen-enriched saline as an irrigation solution during hydrosurgical debridement may attenuate excessive redox imbalance and stabilize the wound microenvironment during this dynamic intervention phase. Such intra-procedural modulation may facilitate the transition from inflammation to the proliferative phase of wound healing, thereby promoting tissue repair. This approach is expected to attenuate the transient oxidative burst following debridement, as reflected by reductions in redox-related biomarkers in the wound environment, including ROS levels and oxidative damage markers such as 8-hydroxy-2′-deoxyguanosine and lipid peroxidation products, with relative decreases in these biomarkers compared with conventional debridement, potentially consistent with reductions observed in preclinical oxidative stress models. These findings are consistent with findings from previous experimental studies demonstrating attenuation of oxidative stress markers following hydrogen administration. This hypothesis introduces a novel therapeutic concept, redox modulation during the debridement process, offering a practical strategy for integrating hydrogen-based therapy into existing wound management without altering current surgical techniques.
Full article

Graphical abstract
Open AccessArticle
An Integrated Approach to Controlling the Al/H2O Reaction in Hydrogen Generation
by
Olga Morozova and Olga Kudryashova
Hydrogen 2026, 7(2), 63; https://doi.org/10.3390/hydrogen7020063 - 6 May 2026
Abstract
The reaction of aluminum with water is a promising method for producing hydrogen on demand for autonomous energy systems. However, its practical implementation faces the challenge of process control due to high exothermicity, leading to particle sintering and thermal instability, especially when using
[...] Read more.
The reaction of aluminum with water is a promising method for producing hydrogen on demand for autonomous energy systems. However, its practical implementation faces the challenge of process control due to high exothermicity, leading to particle sintering and thermal instability, especially when using highly reactive nanopowders. The goal of this study is to implement an integrated approach to controlling this reaction, aimed at minimizing these risks. The approach is based on the principle of spatial and temporal distribution of reactants to ensure uniform heat release. Two process management methods were investigated: electrostatic application of aluminum powder to the reactor walls with its gradual release and pre-treatment of a nanopowder-ice mixture. Using a macrokinetic mathematical model, calculations of the conversion kinetics and heat release were performed and compared with experimental data. The results showed that both methods prevent slurry self-heating and achieve uniform hydrogen generation at a constant rate. In particular, the use of a pre-frozen mixture ensured stable hydrogen production over a long period of time without additional heating or stirring. The proposed approaches can be used in the design of safe and efficient hydrogen generators for autonomous power plants.
Full article
(This article belongs to the Special Issue Hydrogen Energy and Fuel Cell Technology)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
Initial Cyclic Stability Tests of a First-Generation Rechargeable Metal Hydride–Air Battery Prototype
by
Borislav Abrashev, Valentin Terziev and Tony Spassov
Hydrogen 2026, 7(2), 62; https://doi.org/10.3390/hydrogen7020062 - 4 May 2026
Abstract
►▼
Show Figures
The main goal of this study was to develop and validate a laboratory-scale prototype of a rechargeable metal hydride (MH)–air battery integrating gas diffusion electrodes (GDEs) and MH electrodes with stable performance over extended operation (>500 h) and repeated charge–discharge cycling (>100 cycles).
[...] Read more.
The main goal of this study was to develop and validate a laboratory-scale prototype of a rechargeable metal hydride (MH)–air battery integrating gas diffusion electrodes (GDEs) and MH electrodes with stable performance over extended operation (>500 h) and repeated charge–discharge cycling (>100 cycles). This work addresses the critical transition from optimized electrode materials to a functioning system by investigating its operation under deep-discharge conditions, a key but still insufficiently explored regime in the context of stationary renewable energy storage. In this respect, this study explicitly targets the practical applicability of the developed system rather than focusing solely on material-level performance. The most efficient electrode materials, previously optimized, were successfully integrated into a single-cell configuration and systematically evaluated under various operating conditions. By determining the limiting current density for stable GDE operation, an appropriate operating window was defined, enabling maximum capacity utilization without compromising electrode integrity. At a current density of 10 mA, the maximum depth of discharge was achieved at a cell voltage of 575 mV, ensuring operation in a regime that limits GDE degradation while maintaining high energy efficiency. In addition, the electrode retains its mechanical stability after operation is interrupted, indicating good structural robustness. Furthermore, the performance of two identical cells connected in series was investigated to assess system scalability. The cells were operated under near-limit conditions and exhibited stable behavior. Overall, the present results confirm that the developed MH–air battery system extends beyond laboratory-scale validation and shows strong potential for implementation in stationary energy storage applications.
Full article

Graphical abstract
Open AccessArticle
Analysis of Hydrogen Storage Methods for Decarbonizing Maritime Transport: A Multi-Criteria Decision Analysis Tool
by
Rocio Maceiras, Victor Alfonsin, Miguel A. Alvarez-Feijoo, Jorge Feijoo and Adrian Lopez-Granados
Hydrogen 2026, 7(2), 61; https://doi.org/10.3390/hydrogen7020061 - 2 May 2026
Abstract
Decarbonizing maritime transport requires hydrogen storage technologies that are efficient, safe, and compatible with fuel cell systems. This study evaluates three hydrogen storage technologies (compressed hydrogen (CH2), liquid hydrogen (LH2), and metal hydrides (MH)) based on five key criteria:
[...] Read more.
Decarbonizing maritime transport requires hydrogen storage technologies that are efficient, safe, and compatible with fuel cell systems. This study evaluates three hydrogen storage technologies (compressed hydrogen (CH2), liquid hydrogen (LH2), and metal hydrides (MH)) based on five key criteria: safety, autonomy, environmental impact, cost, and implementation feasibility. Applying two multi-criteria decision-making (MCDM) methods, Analytic Hierarchy Process (AHP) and Technique for Order Preference by Similarity to Ideal Solution (TOPSIS), the alternatives are systematically ranked to identify the most suitable option. Both methods consistently highlight compressed hydrogen as the most viable storage solution, offering a good balance of safety, infrastructure maturity, and economic performance. Liquid hydrogen, despite its superior autonomy, is limited by high energy and infrastructure costs. Metal hydrides, although safer and more compact in terms of volumetric density, are limited by low gravimetric efficiency at the system level due to the additional weight of the storage material and associated components.
Full article
(This article belongs to the Special Issue Women’s Special Issue Series: Hydrogen)
►▼
Show Figures
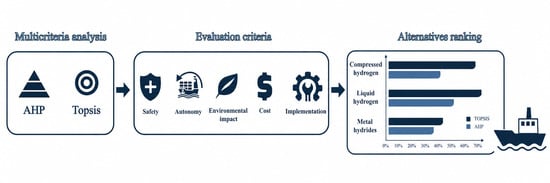
Graphical abstract
Open AccessReview
A Review on the Hydrogen-Based Molten Reduction of Iron Oxides
by
Xuejun Zhou, Jianliang Zhang, Yaozu Wang, Ben Feng, Shaofeng Lu and Zhengjian Liu
Hydrogen 2026, 7(2), 60; https://doi.org/10.3390/hydrogen7020060 - 2 May 2026
Abstract
In the context of global carbon neutrality goals, substituting hydrogen for carbon as a reductant represents a critical pathway for mitigating emissions in the iron and steel industry. Hydrogen-based molten reduction technology, characterized by its rapid reaction kinetics and high feedstock flexibility, has
[...] Read more.
In the context of global carbon neutrality goals, substituting hydrogen for carbon as a reductant represents a critical pathway for mitigating emissions in the iron and steel industry. Hydrogen-based molten reduction technology, characterized by its rapid reaction kinetics and high feedstock flexibility, has emerged as a pivotal direction for the industry’s low-carbon transition. This article systematically reviews research progress on the hydrogen-based reduction of molten iron oxides. The thermodynamic behavior of molten systems is discussed, confirming the feasibility of reducing molten FeO with hydrogen at elevated temperatures. Furthermore, discrepancies and nonlinear characteristics within current mainstream thermodynamic databases regarding the high-temperature molten region are identified. Kinetic studies demonstrate that reduction rates in the molten state significantly exceed those in the solid state. The rate-limiting step is shown to vary with reaction conditions, primarily shifting between interfacial chemical reaction and liquid-phase mass transfer control. Additionally, the influence mechanisms of key parameters—including temperature, reaction time, gas flow rate, gas composition, and slag composition—on the reduction process are comprehensively reviewed. By synthesizing existing methodologies and theoretical advancements, this review aims to provide a theoretical reference for optimizing hydrogen-based molten reduction processes for iron oxides.
Full article
(This article belongs to the Special Issue Engineering the Global Hydrogen Transition: Materials, Processes, Infrastructure, and Deployment)
►▼
Show Figures

Figure 1
Open AccessArticle
Design of Hydrogen Separation from Depleted Gas Field Mixtures Under Variable Operating Conditions
by
Maroš Križan, Ivan Červeňanský and Roman Zavada
Hydrogen 2026, 7(2), 59; https://doi.org/10.3390/hydrogen7020059 - 1 May 2026
Abstract
Large-scale and long-term hydrogen storage is one of the main obstacles to the wider use of hydrogen as a possible substitute for natural gas. A solution could be depleted natural gas fields, which have proven capacity and are already geologically prospected. However, part
[...] Read more.
Large-scale and long-term hydrogen storage is one of the main obstacles to the wider use of hydrogen as a possible substitute for natural gas. A solution could be depleted natural gas fields, which have proven capacity and are already geologically prospected. However, part of this field remains occupied by residual natural gas, meaning that hydrogen is mixed with natural gas during storage and purification after extraction is therefore necessary. The aim of this study was to design and evaluate a hydrogen purification process for separating hydrogen from natural gas after extraction from a depleted natural gas field while maintaining the required hydrogen purity and recovery. Input data provided by Nafta a.s. were used for the mathematical simulation of hydrogen separation throughout a 150-day extraction period. A mathematical model of membrane separation and pressure swing adsorption (PSA) was developed. A single membrane stage was only able to operate effectively during the first 50 days of withdrawal while maintaining at least 80% hydrogen recovery. A two-stage membrane configuration achieved hydrogen purity above 98% with final recoveries above 80–85%, while the hybrid membrane–PSA system enabled hydrogen purity of 99.8% and total recovery of 82.5% on the last day of extraction.
Full article
(This article belongs to the Special Issue Green and Low-Emission Hydrogen: Pathways to a Sustainable Future)
►▼
Show Figures

Figure 1
Open AccessReview
Recent Advancements in Electrode Materials for Hydrogen Production via Hydrogen Sulfide (H2S) Electrolysis
by
Ivelina Tsacheva, Mehmet Suha Yazici, Cenk Turutoglu, Gergana Raikova, Konstantin Petrov and Dzhamal Uzun
Hydrogen 2026, 7(2), 58; https://doi.org/10.3390/hydrogen7020058 - 30 Apr 2026
Abstract
►▼
Show Figures
The production of green hydrogen via aqueous electrolysis of hydrogen sulfide (H2S) holds significant potential to address challenges related to sustainable energy generation and environmental protection. The electrocatalytic splitting of water polluted with highly toxic H2S is attractive for
[...] Read more.
The production of green hydrogen via aqueous electrolysis of hydrogen sulfide (H2S) holds significant potential to address challenges related to sustainable energy generation and environmental protection. The electrocatalytic splitting of water polluted with highly toxic H2S is attractive for industrial applications because the process: (i) is less power-consuming than direct thermal H2S decomposition; (ii) achieves high Faradaic efficiencies for hydrogen production; and (iii) yields elemental sulfur as an added-value by-product. This review covers a brief discussion on sulfide-containing water sources and electrochemical methods for hydrogen production from H2S, specifically Direct, Indirect, and Electrochemical Membrane Reactor (EMR) systems. To become commercially and economically attractive, these approaches require improvements in electrolysis efficiency through the development of low-cost electrode materials that are resistant to sulfur poisoning and corrosion, while possessing high catalytic activity, enhanced stability, and durability. Early research focused on carbon-based materials combined with noble metal oxides, transition metal compounds, and related materials. Since their practical performance is limited, investigations have shifted toward nanostructured electrocatalysts with unique crystal structures and designs, which show significantly improved efficiency for H2S electrolysis. This review highlights the potential of H2S electrolysis for hydrogen production, giving special attention to recent advancements in electrode materials.
Full article

Figure 1
Open AccessArticle
An Empirical Model of the Kinetics of Hydrogen-Induced Cracking in API 5L Steel: Part 1
by
Diego Israel Rivas-López, Manuel Alejandro Beltrán-Zúñiga, Jorge Luis González-Velázquez, Gabriel Sepúlveda-Cervantes, Héctor Javier Dorantes-Rosales, Darío Alberto Sigala-García and Suset Santana-Hernández
Hydrogen 2026, 7(2), 57; https://doi.org/10.3390/hydrogen7020057 - 27 Apr 2026
Abstract
►▼
Show Figures
An empirical model of the kinetics of Hydrogen-Induced Cracking (HIC) in API 5L steels was derived using the best-fit equation for experimental data obtained from cathodic charging tests. The model represents the growth of both individual and interconnecting cracks, using a double exponential
[...] Read more.
An empirical model of the kinetics of Hydrogen-Induced Cracking (HIC) in API 5L steels was derived using the best-fit equation for experimental data obtained from cathodic charging tests. The model represents the growth of both individual and interconnecting cracks, using a double exponential equation known as the Gumbel distribution. Current density was the main independent input variable, as it is related to the hydrogen influx during the cathodic charging experiment. The results indicated that in the initial hours of cathodic charging most of the available HIC nucleation sites are activated, the growth of these individual cracks being the main contribution to the overall kinetics. Further crack growth is due to the interconnection of individual cracks, decreasing the growth rate until it becomes nearly zero. The proposed model is used in a simulation algorithm that accurately describes the complete HIC kinetics, for both short- and long-term hydrogen charging exposure, reproducing the effects of applied current density on the total cracked area and growth rates. Finally, the simulation algorithm adequately predicts the spatial distribution of HIC in a bidimensional plane that emulates the detection of HIC by C-scan ultrasonic inspection.
Full article

Figure 1
Open AccessArticle
Kinetic Uncertainty in Hydrogen Jet Flames Using Lagrangian Particle Statistics
by
Shuzhi Zhang, Vansh Sharma and Venkat Raman
Hydrogen 2026, 7(2), 56; https://doi.org/10.3390/hydrogen7020056 - 22 Apr 2026
Abstract
►▼
Show Figures
Hydrogen-enriched fuel injection in staged gas-turbine combustors is commonly achieved through jet-in-crossflow (JICF) configurations, where flame stabilization is governed by a local balance between flow-induced strain/mixing and chemical reaction rates. This work investigates turbulent reacting JICF relevant to staged combustion conditions using high-fidelity
[...] Read more.
Hydrogen-enriched fuel injection in staged gas-turbine combustors is commonly achieved through jet-in-crossflow (JICF) configurations, where flame stabilization is governed by a local balance between flow-induced strain/mixing and chemical reaction rates. This work investigates turbulent reacting JICF relevant to staged combustion conditions using high-fidelity simulations with adaptive mesh refinement (AMR) and differential-diffusion effects together with Lagrangian particle statistics. Chemistry model uncertainties are incorporated by using a projection method that maps uncertainty estimates from detailed mechanisms into the model used in this work. Results show that the macroscopic flame topology remains in a stable two-branch regime (lee-stabilized and lifted) and is primarily controlled by the jet momentum–flux ratio J. Visualization of the normalized scalar dissipation rate reveals that the flame front resides on the low-dissipation side of intense mixing layers, occupying an intermediate region between over-strained and under-mixed regions. While hydrogen content does not significantly change the global stabilization mode for the cases studied, uncertainty analysis reveals composition-dependent differences that are not apparent in the mean behavior alone. In particular, visualization in Eulerian (χ, T) state-space analysis and particle statistics conditioned on the stoichiometric surface demonstrate that higher-hydrogen cases observe a lower scalar dissipation rate and exhibit substantially reduced variability in OH production under kinetic-parameter perturbations, whereas lower-hydrogen blends experience higher dissipation and amplified chemical sensitivity. These findings highlight that, even in globally similar JICF regimes, the hydrogen content can modify the local response of the flame to kinetic-parameter uncertainty, motivating uncertainty-aware interpretation and design for hydrogen-fueled staging systems.
Full article

Figure 1
Open AccessArticle
A Simple Study of Hydrogen Production from Recycled Aluminum Microparticles in Alkaline Media
by
Sergio Martínez-Vargas, José-Enrique Flores-Chan, Humberto-Julián Mandujano-Ramírez, Salatiel Pérez-Montejo, Damián Calan-Canche and Cristobal Patino-Carachure
Hydrogen 2026, 7(2), 55; https://doi.org/10.3390/hydrogen7020055 - 22 Apr 2026
Abstract
Hydrogen (H2) was produced from recycled aluminum microparticles (180–250, 300–425, and 425–500 μm) via alkaline hydrolysis using a 1.0 M NaOH solution to enhance oxide layer removal and aluminum dissolution. Maximum hydrogen flow rates of approximately 13, 15, and 19 mL·min
[...] Read more.
Hydrogen (H2) was produced from recycled aluminum microparticles (180–250, 300–425, and 425–500 μm) via alkaline hydrolysis using a 1.0 M NaOH solution to enhance oxide layer removal and aluminum dissolution. Maximum hydrogen flow rates of approximately 13, 15, and 19 mL·min−1 were obtained, confirming that smaller particle sizes promote faster reaction rates due to increased specific surface area. The hydrogen evolution exhibited two-stage kinetic behavior: an initial stage characterized by rapid aluminum dissolution and increasing H2 production, followed by a gradual decline associated with the formation of a passivating Al(OH)3 layer. Despite the higher reaction rates observed for smaller particles, the maximum cumulative hydrogen production was obtained for the intermediate particle size (363 µm, 132 mL), compared to 106 mL and 102 mL for 215 µm and 463 µm, respectively, indicating a trade-off between surface area and passivation effects. Kinetic analysis based on the shrinking core model showed excellent agreement (R2 = 99.94–99.97%), with rate constants of 0.137, 0.064, and 0.050 min−1. The relationship k ∝ d−n (n ≈ 1.4) suggests a mixed kinetic regime involving both surface reaction and diffusion through the Al(OH)3 layer. These findings indicate that hydrogen generation can be modulated by particle size; however, the relatively low flow rates and yields limit its immediate practical applicability.
Full article
(This article belongs to the Special Issue Women’s Special Issue Series: Hydrogen)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Optimal Efficiency Control of Photovoltaic–Energy Storage–Hydrogen Production System Considering Proton Exchange Membrane Electrolyzer Efficiency
by
Chao Fu, Zeyu Chen, Hanqing Liu, Long Ma and Yuwei Sun
Hydrogen 2026, 7(2), 54; https://doi.org/10.3390/hydrogen7020054 - 22 Apr 2026
Abstract
Hydrogen is a clean energy carrier with broad application potential. This study focuses on improving hydrogen production efficiency in a proton exchange membrane (PEM) electrolyzer system that integrates a photovoltaic (PV) array, a battery energy storage system, and the electrolyzer. The PV array
[...] Read more.
Hydrogen is a clean energy carrier with broad application potential. This study focuses on improving hydrogen production efficiency in a proton exchange membrane (PEM) electrolyzer system that integrates a photovoltaic (PV) array, a battery energy storage system, and the electrolyzer. The PV array is interfaced with the electrolyzer through a buck converter using a maximum power point tracking (MPPT) algorithm to ensure maximum energy harvesting. A key contribution of this work is the integration of a battery system through a dual-active-bridge (DAB) converter. The DAB converter employs a multilayer perceptron (MLP) model to dynamically regulate the electrolyzer current and maintain optimal operating efficiency. An adaptive energy management strategy is further proposed to address solar irradiance fluctuations and enhance long-term operational stability. The MLP model is developed in Python and embedded into a PLECS simulation environment. The simulation results verify the effectiveness of the proposed control approach and efficiency optimization scheme. Throughout the simulation period, the PEM electrolyzer sustains an optimal efficiency of 69.9% under maximum PV power output. A limitation of this study is that the efficiency model is derived from the literature and does not yet consider all operational factors, indicating the need for refinement in future work.
Full article
(This article belongs to the Special Issue Hydrogen Energy and Fuel Cell Technology)
►▼
Show Figures

Figure 1
Open AccessArticle
Agent-Based Modeling of Green Hydrogen Industry Scale-Up in Russia: Critical Thresholds, Phase Dynamics, and Investment Requirements
by
Konstantin Gomonov, Svetlana Ratner, Arsen A. Petrosyan and Svetlana Revinova
Hydrogen 2026, 7(2), 53; https://doi.org/10.3390/hydrogen7020053 - 20 Apr 2026
Abstract
The development of a green hydrogen industry is a strategic priority for Russia’s energy transition, yet the dynamics of scaling up this nascent sector remain poorly understood. This study uses agent-based modeling (ABM) to simulate the co-evolution of Russia’s electricity, hydrogen, and electrolyzer
[...] Read more.
The development of a green hydrogen industry is a strategic priority for Russia’s energy transition, yet the dynamics of scaling up this nascent sector remain poorly understood. This study uses agent-based modeling (ABM) to simulate the co-evolution of Russia’s electricity, hydrogen, and electrolyzer sectors over 2024–2050. The model incorporates three types of heterogeneous agents (power producers, hydrogen producers, and electrolyzer manufacturers) operating under bounded rationality. Four scenarios are examined across 50 Monte Carlo runs each, varying the electrolyzer learning rate (10–25%), willingness to pay for green hydrogen (2–6 $/kg), and government support intensity. The results reveal an endogenous three-phase development pattern: Phase I (2024–2028) dominated by renewable capacity build-up reaching ~30 GW; Phase II (2029–2040) characterized by rapid electrolyzer deployment scaling to 14.5 GW; and Phase III (2041–2050) marked by stabilization at approximately 30 GW producing 1.12 Mt/year at 3.1 $/kg. Two critical thresholds are identified: renewable capacity exceeding 30–38 GW and low-cost electricity above 4–7 TWh/year. The electrolyzer learning rate emerges as the most influential parameter, while the pessimistic scenario confirms market failure without a green premium (WTP < 2 $/kg). Strategic investment losses of 2–6 billion USD are necessary catalysts for industry emergence. Russia’s 2030 production target (0.55 Mt) is found structurally infeasible under all scenarios.
Full article
(This article belongs to the Special Issue Green Hydrogen Production)
►▼
Show Figures

Graphical abstract
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Energies, Fuels, Hydrogen, Nanoenergy Advances
Hydrogen Energy Technologies, 3rd Edition
Topic Editors: Bahman Shabani, Mahesh SuryawanshiDeadline: 1 November 2026
Topic in
Energies, Processes, Batteries, Hydrogen, Fuels
Mechanical Impacts and Multiphysics Interactions in PEM Fuel Cells: Modelling, Characterization and Design from Components to Stacks
Topic Editors: Denis Candusso, Dominique Chamoret, Yann MeyerDeadline: 30 November 2026
Topic in
Energies, Materials, Sustainability, Catalysts, C, Hydrogen, Solids
Functional Carbon-Based Materials and Systems for Energy and Environmental Applications
Topic Editors: Nikolaos Kostoglou, Claus RebholzDeadline: 31 December 2026
Topic in
Catalysts, Energies, Hydrogen, Processes, Sustainability
Advances in Hydrogen Energy
Topic Editors: Samuel Simon Araya, Vincenzo LisoDeadline: 31 March 2027

Special Issues
Special Issue in
Hydrogen
Production of Hydrogen from Biomass and Organic Waste
Guest Editor: Nadia CeroneDeadline: 31 May 2026
Special Issue in
Hydrogen
Advances in Hydrogen Storage Materials: Integrating Theory, Computation and Experimental Insights
Guest Editors: Lusheng Wang, Yuxin WangDeadline: 20 June 2026
Special Issue in
Hydrogen
Hydrogen for a Clean Energy Future
Guest Editor: Rajender BoddulaDeadline: 30 June 2026
Special Issue in
Hydrogen
Women’s Special Issue Series: Hydrogen
Guest Editors: Chiara Milanese, Rocio Maceíras CastroDeadline: 30 July 2026