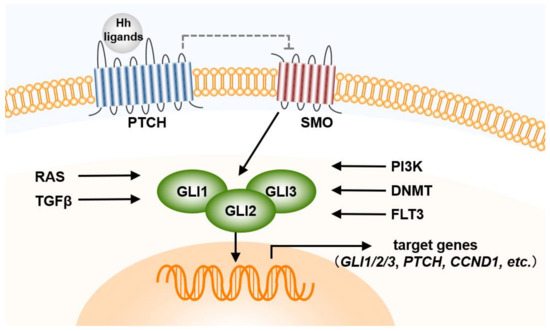
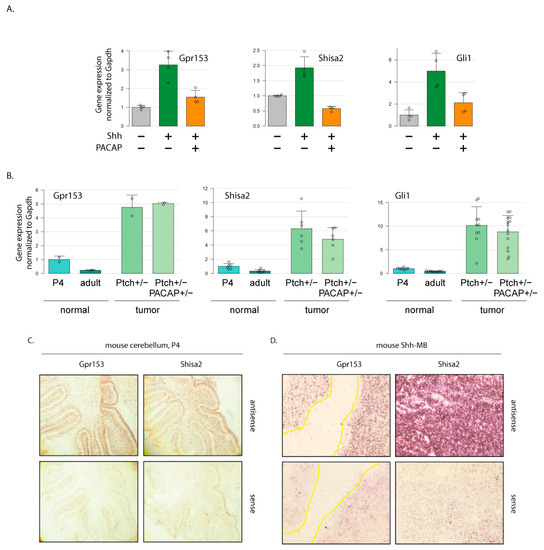
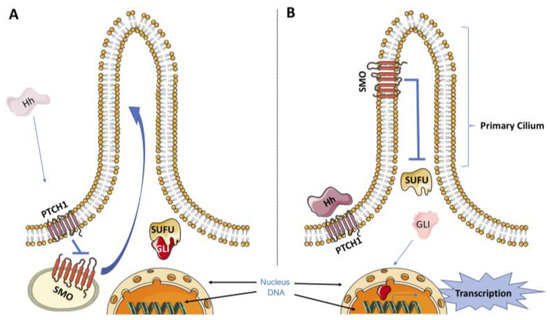
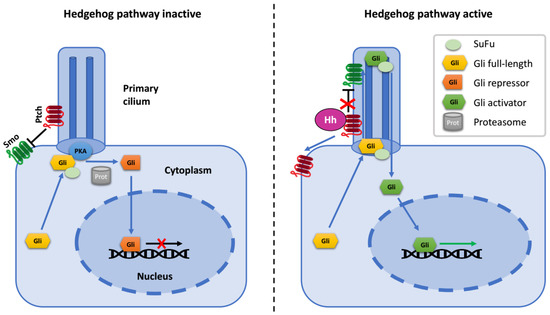
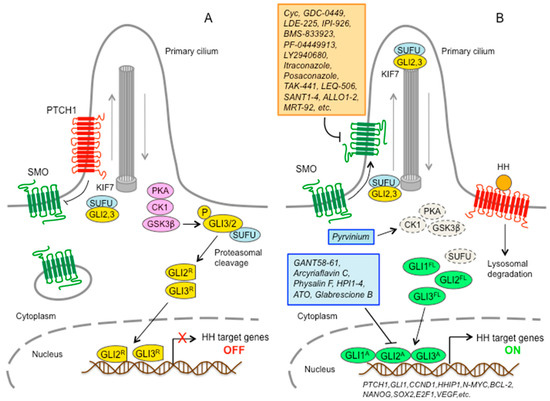
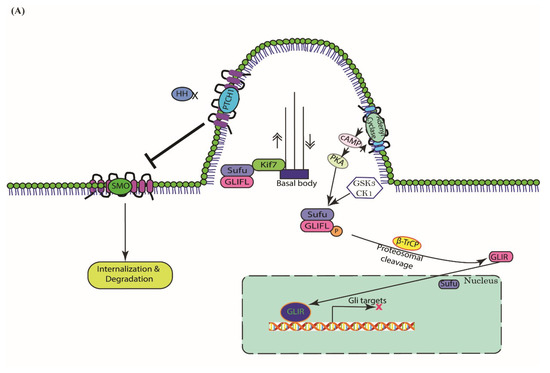
Hedgehog Signal Transduction in Physiology and Disease
A topical collection in Cells (ISSN 2073-4409). This collection belongs to the section "Cell Signaling".
Viewed by 119195
Share This Topical Collection
Editor
 Dr. Natalia Riobo-Del Galdo
Dr. Natalia Riobo-Del Galdo
 Dr. Natalia Riobo-Del Galdo
Dr. Natalia Riobo-Del Galdo
E-Mail
Website
Collection Editor
School of Molecular and Cellular Biology and Leeds Institute of Medical Research, University of Leeds, Leeds LS2 9JT, UK
Interests: signal transduction; cancer biology; Hedgehog pathway
Topical Collection Information
Dear Colleagues,
Since its discovery in 1998, the Hedgehog proteins and the signals they stimulate in different cell types and organisms have been the subject of intense investigation in relation to their roles in embryonic development and some disease conditions such as cancer and fibrosis. Mutations in any of the genes encoding the central components lead to congenital defects and/or cancer development. Regulation of key cellular processes by Hedgehog signaling includes proliferation, differentiation, apoptosis, autophagy, and cell-type specific functions, such as cytotoxic T cell killing and excitable cell action potential tuning. This Topic Collection is open to submissions of mechanistic aspects of canonical and non-canonical Hedgehog signaling in normal cells and tissues, as well as its dysregulation in disease conditions, development and/or characterization of small molecule modulators of the pathway, crosstalk with other signaling pathways, identification of key transcriptional outputs, and development of novel animal and cell-based models.
We look forward to your valuable contributions.
Dr. Natalia Riobo-Del Galdo
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cells is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript.
The Article Processing Charge (APC) for publication in this open access journal is 2700 CHF (Swiss Francs).
Submitted papers should be well formatted and use good English. Authors may use MDPI's
English editing service prior to publication or during author revisions.
Keywords
- Hedgehog
- Cancer
- Smoothened inhibitors
- Non-canonical activation of Gli
- Efficacy and safety
- Mechanisms of treatment resistance
Published Papers (15 papers)
Open AccessArticle
Inhibition of Hedgehog Signaling in Fibroblasts, Pancreatic, and Lung Tumor Cells by Oxy186, an Oxysterol Analogue with Drug-Like Properties
by
Feng Wang, Frank Stappenbeck and Farhad Parhami
Cited by 22 | Viewed by 6831
Abstract
The widespread involvement of the Hedgehog (Hh) signaling pathway in human malignancies has motivated the clinical development of Smoothened (Smo) antagonists, such as vismodegib and sonidegib. However, Smo antagonists have failed to benefit patients suffering from Hh pathway-dependent solid tumors, such as pancreatic,
[...] Read more.
The widespread involvement of the Hedgehog (Hh) signaling pathway in human malignancies has motivated the clinical development of Smoothened (Smo) antagonists, such as vismodegib and sonidegib. However, Smo antagonists have failed to benefit patients suffering from Hh pathway-dependent solid tumors, such as pancreatic, colorectal, or ovarian cancer. Hh-dependent cancers are often driven by activating mutations that occur downstream of Smo and directly activate the transcription factors known as glioma-associated oncogenes (
Gli1-3). Hence, the direct targeting of Gli could be a more effective strategy for achieving disease modification compared to Smo antagonism. In this study, we report on the biological and pharmacological evaluation of Oxy186, a semisynthetic oxysterol analogue, as a novel inhibitor of Hh signaling acting downstream of Smo, with encouraging drug-like properties. Oxy186 exhibits strong inhibition of ligand-induced Hh signaling in NIH3T3-E1 fibroblasts, as well as in constitutively activated Hh signaling in Suppressor of Fused (Sufu) null mouse embryonic fibroblast (MEF) cells. Oxy186 also inhibits Gli1 transcriptional activity in NIH3T3-E1 cells expressing exogenous Gli1 and Gli-dependent reporter constructs. Furthermore, Oxy186 suppresses Hh signaling in PANC-1 cells, a human pancreatic ductal adenocarcinoma (PDAC) tumor cell line, as well as PANC-1 cell proliferation in vitro, and in human lung cancer cell lines, A549 and H2039.
Full article
►▼
Show Figures
Open AccessArticle
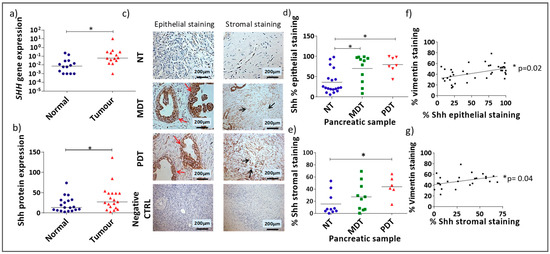
Sonic Hedgehog Ligand: A Role in Formation of a Mesenchymal Niche in Human Pancreatic Ductal Adenocarcinoma
by
Francesca Saini, Richard H. Argent and Anna M. Grabowska
Cited by 13 | Viewed by 4222
Abstract
Pancreatic ductal adenocarcinoma (PDAC) is characterised by desmoplasia, thought to support progression and chemotherapeutic resistance. The Hedgehog pathway is known to play an important role in this cancer. While the upregulation of Sonic hedgehog (Shh) in the epithelium of PDAC is known, we
[...] Read more.
Pancreatic ductal adenocarcinoma (PDAC) is characterised by desmoplasia, thought to support progression and chemotherapeutic resistance. The Hedgehog pathway is known to play an important role in this cancer. While the upregulation of Sonic hedgehog (Shh) in the epithelium of PDAC is known, we investigated its expression in the tumour microenvironment in order to find new targets for new chemotherapeutical approaches. Immunohistochemistry was used for the investigation of Shh and Vimentin in primary human pancreatic tissues. Gene (qRT-PCR) and protein (immunofluorescence) expression of Shh, αSMA (a marker of the mesenchymal phenotype) and periostin (a marker of mesenchymal cells within a mixed population) were investigated in in vitro cell models. Shh expression was significantly upregulated in the stromal and epithelial compartments of poorly-differentiated PDAC samples, with a strong correlation with the amount of stroma present. Characterisation of stromal cells showed that there was expression of Shh ligand in a mixed population comprising αSMA
+ myofibroblasts and αSMA
− mesenchymal stem cells. Moreover, we demonstrated the interaction between these cell lines by showing a higher rate of mesenchymal cell proliferation and the upregulation of periostin. Therefore, targeting stromal Shh could affect the equilibrium of the tumour microenvironment and its contribution to tumour growth.
Full article
►▼
Show Figures
Open AccessReview
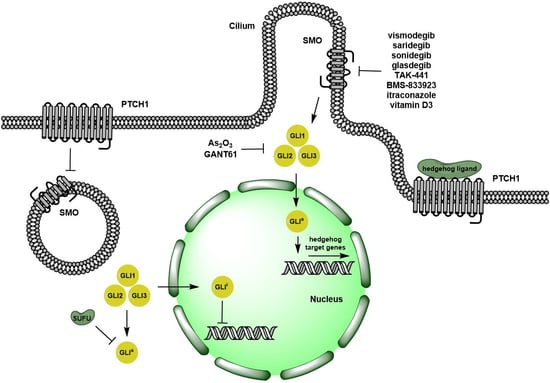
Recent Advances in the Clinical Targeting of Hedgehog/GLI Signaling in Cancer
by
Hao Xie, Brooke D. Paradise, Wen Wee Ma and Martin E. Fernandez-Zapico
Cited by 76 | Viewed by 6878
Abstract
The Hedgehog/GLI signaling pathway plays an important role in normal embryonic tissue development and has been implicated in the pathogenesis of various human cancers. In this review article, we summarize pre-clinical evidence supporting the suitability of targeting this signaling pathway in cancers. We
[...] Read more.
The Hedgehog/GLI signaling pathway plays an important role in normal embryonic tissue development and has been implicated in the pathogenesis of various human cancers. In this review article, we summarize pre-clinical evidence supporting the suitability of targeting this signaling pathway in cancers. We review agents blocking both the ligand-dependent and ligand-independent cascades, and discuss the clinical evidence, which has led to the FDA approval of Hedgehog receptor Smoothened inhibitors, vismodegib, and sonidegib, in different malignancies. Finally, we provide an overview of published and ongoing clinical trial data on single agent or combination therapeutic strategies, targeting Hedgehog/GLI signaling pathway, in both advanced solid tumors and hematologic malignancies.
Full article
►▼
Show Figures
Open AccessFeature PaperEditor’s ChoiceReview
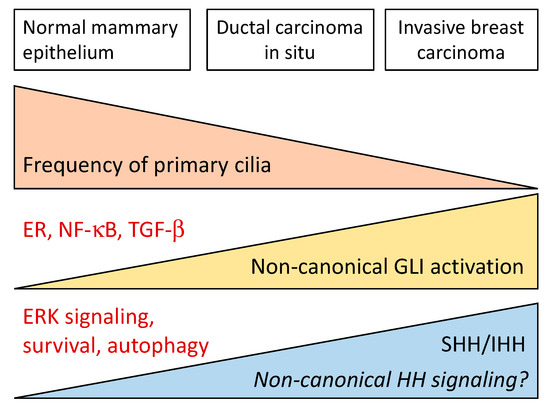
Role of Hedgehog Signaling in Breast Cancer: Pathogenesis and Therapeutics
by
Natalia A. Riobo-Del Galdo, Ángela Lara Montero and Eva V. Wertheimer
Cited by 130 | Viewed by 10733
Abstract
Breast cancer (BC) is the leading cause of cancer-related mortality in women, only followed by lung cancer. Given the importance of BC in public health, it is essential to identify biomarkers to predict prognosis, predetermine drug resistance and provide treatment guidelines that include
[...] Read more.
Breast cancer (BC) is the leading cause of cancer-related mortality in women, only followed by lung cancer. Given the importance of BC in public health, it is essential to identify biomarkers to predict prognosis, predetermine drug resistance and provide treatment guidelines that include personalized targeted therapies. The Hedgehog (Hh) signaling pathway plays an essential role in embryonic development, tissue regeneration, and stem cell renewal. Several lines of evidence endorse the important role of canonical and non-canonical Hh signaling in BC. In this comprehensive review we discuss the role of Hh signaling in breast development and homeostasis and its contribution to tumorigenesis and progression of different subtypes of BC. We also examine the efficacy of agents targeting different components of the Hh pathway both in preclinical models and in clinical trials. The contribution of the Hh pathway in BC tumorigenesis and progression, its prognostic role, and its value as a therapeutic target vary according to the molecular, clinical, and histopathological characteristics of the BC patients. The evidence presented here highlights the relevance of the Hh signaling in BC, and suggest that this pathway is key for BC progression and metastasis.
Full article
►▼
Show Figures
Open AccessReview
Targeting Hedgehog (Hh) Pathway for the Acute Myeloid Leukemia Treatment
by
Toshiki Terao and Yosuke Minami
Cited by 47 | Viewed by 8604
Abstract
The Hedgehog (Hh) pathway, containing the Patched (PTCH) and Smoothened (SMO) multitransmembrane proteins, is the main regulator of vertebrate embryonic development. A non-canonical Hh pathway was recently observed in numerous types of solid cancers and hematological malignancies. Although acute myeloid leukemia (AML) is
[...] Read more.
The Hedgehog (Hh) pathway, containing the Patched (PTCH) and Smoothened (SMO) multitransmembrane proteins, is the main regulator of vertebrate embryonic development. A non-canonical Hh pathway was recently observed in numerous types of solid cancers and hematological malignancies. Although acute myeloid leukemia (AML) is a common and lethal myeloid malignancy, the chemotherapy for AML has not changed in the last three decades. The Hh pathway and other intracellular signaling pathways are important for the tumor cells’ cycle or therapeutic resistance of AML cells. In this article, we will review the current trends in Hh pathway inhibitors for treating AML.
Full article
►▼
Show Figures
Open AccessArticle
Differential Expression of Mitochondrial Biogenesis Markers in Mouse and Human SHH-Subtype Medulloblastoma
by
Maria Łastowska, Agnieszka Karkucińska-Więckowska, James A. Waschek and Paweł Niewiadomski
Cited by 4 | Viewed by 4563
Abstract
Medulloblastoma is a brain tumor that arises predominantly in infants and children. It is the most common pediatric brain malignancy. Around 25% of medulloblastomas are driven by constitutive activation of the Hedgehog signaling pathway. Hedgehog-driven medulloblastoma is often studied in the laboratory using
[...] Read more.
Medulloblastoma is a brain tumor that arises predominantly in infants and children. It is the most common pediatric brain malignancy. Around 25% of medulloblastomas are driven by constitutive activation of the Hedgehog signaling pathway. Hedgehog-driven medulloblastoma is often studied in the laboratory using genetic mouse models with overactive Hedgehog signaling, which recapitulate many of the pathological features of human Hedgehog-dependent tumors. However, we show here that on a molecular level the human and mouse HH-dependent MB are quite distinct, with human, but not mouse, tumors characterized by the presence of markers of increased oxidative phosphorylation and mitochondrial biogenesis. The latter suggests that, unlike for many other types of tumors, a switch to glycolytic metabolism might not be co-opted by human SHH-MB to perpetuate their survival and growth. This needs to be taken into consideration and could potentially be exploited in the design of therapies.
Full article
►▼
Show Figures
Open AccessReview
Targeting the Hedgehog Pathway in Cancer: Current Evidence and Future Perspectives
by
Daniel Girardi, Adriana Barrichello, Gustavo Fernandes and Allan Pereira
Cited by 54 | Viewed by 6133
Abstract
The Hedgehog pathway (HhP) plays an important role in normal embryonic development and its abnormal function has been linked to a variety of neoplasms. Recently, the complex mechanisms involved in this pathway have been deciphered and the cross talks with other important pathways
[...] Read more.
The Hedgehog pathway (HhP) plays an important role in normal embryonic development and its abnormal function has been linked to a variety of neoplasms. Recently, the complex mechanisms involved in this pathway have been deciphered and the cross talks with other important pathways involved in carcinogenesis have been characterized. This knowledge has led to the development of targeted therapies against key components of HhP, which culminated in the approval of vismodegib for the treatment of advanced basal cell carcinoma in 2012. Since then, other compounds have been developed and evaluated in preclinical and clinical studies with interesting results. Today, several medications against components of the HhP have demonstrated clinical activity as monotherapies and in combination with cytotoxic treatment or other targeted therapies against mitogenic pathways that are linked to the HhP. This review aims to clarify the mechanism of the HhP and the complex crosstalk with others pathways involved in carcinogenesis and to discuss both the evidence associated with the growing number of medications and combined therapies addressing this pathway and future perspectives.
Full article
►▼
Show Figures
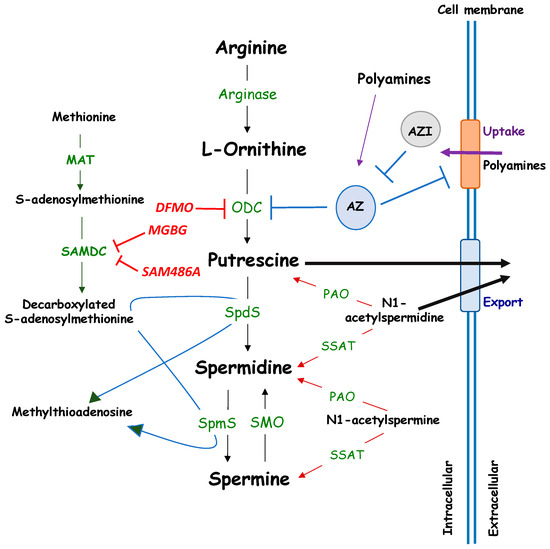
Open AccessReview
Polyamine Metabolism as a Therapeutic Target in Hedgehog-Driven Basal Cell Carcinoma and Medulloblastoma
by
Sonia Coni, Laura Di Magno, Silvia Maria Serrao, Yuta Kanamori, Enzo Agostinelli and Gianluca Canettieri
Cited by 20 | Viewed by 6308
Abstract
Hedgehog (Hh) signaling is a critical developmental regulator and its aberrant activation, due to somatic or germline mutations of genes encoding pathway components, causes Basal Cell Carcinoma (BCC) and medulloblastoma (MB). A growing effort has been devoted at the identification of druggable vulnerabilities
[...] Read more.
Hedgehog (Hh) signaling is a critical developmental regulator and its aberrant activation, due to somatic or germline mutations of genes encoding pathway components, causes Basal Cell Carcinoma (BCC) and medulloblastoma (MB). A growing effort has been devoted at the identification of druggable vulnerabilities of the Hedgehog signaling, leading to the identification of various compounds with variable efficacy and/or safety. Emerging evidence shows that an aberrant polyamine metabolism is a hallmark of Hh-dependent tumors and that its pharmacological inhibition elicits relevant therapeutic effects in clinical or preclinical models of BCC and MB. We discuss here the current knowledge of polyamine metabolism, its role in cancer and the available targeting strategies. We review the literature about the connection between polyamines and the Hedgehog signaling, and the potential therapeutic benefit of targeting polyamine metabolism in two malignancies where Hh pathways play a well-established role: BCC and MB.
Full article
►▼
Show Figures
Open AccessReview
Gli Proteins: Regulation in Development and Cancer
by
Paweł Niewiadomski, Sylwia M. Niedziółka, Łukasz Markiewicz, Tomasz Uśpieński, Brygida Baran and Katarzyna Chojnowska
Cited by 161 | Viewed by 17138
Abstract
Gli proteins are transcriptional effectors of the Hedgehog signaling pathway. They play key roles in the development of many organs and tissues, and are deregulated in birth defects and cancer. We review the molecular mechanisms of Gli protein regulation in mammals, with special
[...] Read more.
Gli proteins are transcriptional effectors of the Hedgehog signaling pathway. They play key roles in the development of many organs and tissues, and are deregulated in birth defects and cancer. We review the molecular mechanisms of Gli protein regulation in mammals, with special emphasis on posttranslational modifications and intracellular transport. We also discuss how Gli proteins interact with co-activators and co-repressors to fine-tune the expression of Hedgehog target genes. Finally, we provide an overview of the regulation of developmental processes and tissue regeneration by Gli proteins and discuss how these proteins are involved in cancer progression, both through canonical regulation via the Hedgehog pathway and through cross-talk with other signaling pathways.
Full article
►▼
Show Figures
Open AccessFeature PaperArticle
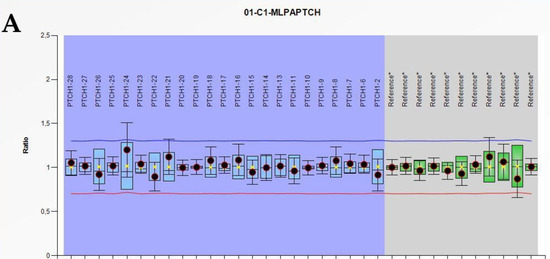
Nevoid Basal Cell Carcinoma Syndrome: PTCH1 Mutation Profile and Expression of Genes Involved in the Hedgehog Pathway in Argentinian Patients
by
Maria Florencia Martinez, Maria Vanesa Romano, Alfredo Pedro Martinez, Abel González, Carolina Muchnik, Fernando Miguel Stengel, Luis Daniel Mazzuoccolo and Pablo Javier Azurmendi
Cited by 20 | Viewed by 5591
Abstract
Nevoid basal cell carcinoma syndrome (NBCCS) is an autosomal dominant disorder characterized by multiple basal cell carcinomas (BCC), mainly caused by
PTCH1 gene mutations. Our current study aimed to establish (1)
PTCH1 germinal and somatic mutational status, (2) component and Hedgehog (HH) pathway
[...] Read more.
Nevoid basal cell carcinoma syndrome (NBCCS) is an autosomal dominant disorder characterized by multiple basal cell carcinomas (BCC), mainly caused by
PTCH1 gene mutations. Our current study aimed to establish (1)
PTCH1 germinal and somatic mutational status, (2) component and Hedgehog (HH) pathway targets gene expression patterns, and (3) profile variations according to the genetic background in BCC and normal surrounding skin (NSS). We collected 23 blood and 20 BCC patient samples and analyzed the
PTCH1 gene using bidirectional sequencing and multiplex ligation-dependent probe amplification. Quantitative PCR was used to determine the mRNA expression levels of
PTCH1,
SMO,
GLI3, and
CCND1 in paired samples of BCC and NSS from 20 patients and four non-NBCCS skin controls (C). Our analyses identified 12 germline and five somatic sequence variants in
PTCH1. mRNA levels of
PTCH1,
SMO, and
GLI3 were higher in NSS compared to C samples, reaching maximum values in BCC samples (
p < 0.05). NSS with
PTCH1 germline mutations had modified
SMO, PTCH1, and
GLI3 mRNA levels compared to samples without mutation (
p < 0.01). Two
PTCH1 mutations in BCC led to an increase in
PTCH1,
SMO, and
GLI3, and a decrease in
CCND1 mRNA levels (
p < 0.01 vs. BCC with germline mutation only). These results indicate that besides
PTCH1, other genes are responsible for NBCCS and BCC development in a population exposed to high UV radiation. Additionally, the mutational events caused increased expression of HH-related genes, even in phenotypically normal skin.
Full article
►▼
Show Figures
Open AccessArticle
Regulation of Survivin Isoform Expression by GLI Proteins in Ovarian Cancer
by
Diana Trnski, Maja Gregorić, Sonja Levanat, Petar Ozretić, Nikolina Rinčić, Tajana Majić Vidaković, Držislav Kalafatić, Ivana Maurac, Slavko Orešković, Maja Sabol and Vesna Musani
Cited by 14 | Viewed by 6262
Abstract
Ovarian cancer (OC) is the most lethal female gynecological malignancy, mostly due to diagnosis in late stages when treatment options are limited. Hedgehog-GLI (HH-GLI) signaling is a major developmental pathway involved in organogenesis and stem cell maintenance, and is activated in OC. One
[...] Read more.
Ovarian cancer (OC) is the most lethal female gynecological malignancy, mostly due to diagnosis in late stages when treatment options are limited. Hedgehog-GLI (HH-GLI) signaling is a major developmental pathway involved in organogenesis and stem cell maintenance, and is activated in OC. One of its targets is survivin (
BIRC5), an inhibitor of apoptosis protein (IAP) that plays a role in multiple processes, including proliferation and cell survival. We wanted to investigate the role of different GLI proteins in the regulation of survivin isoform expression (WT, 2α, 2B, 3B, and Δex3) in the SKOV-3 OC cell line. We demonstrated that survivin isoforms are downregulated in GLI1 and GLI2 knock-out cell lines, but not in the GLI3 knock-out. Treatment of GLI1 knock-out cells with GANT-61 shows an additional inhibitory effect on several isoforms. Additionally, we examined the expression of survivin isoforms in OC samples and the potential role of
BIRC5 polymorphisms in isoform expression. Clinical samples showed the same pattern of survivin isoform expression as in the cell line, and several
BIRC5 polymorphisms showed the correlation with isoform expression. Our results showed that survivin isoforms are regulated both by different GLI proteins and
BIRC5 polymorphisms in OC.
Full article
►▼
Show Figures
Open AccessReview
Targeting Hedgehog Signalling through the Ubiquitylation Process: The Multiple Roles of the HECT-E3 Ligase Itch
by
Paola Infante, Ludovica Lospinoso Severini, Flavia Bernardi, Francesca Bufalieri and Lucia Di Marcotullio
Cited by 24 | Viewed by 6280
Abstract
Hedgehog signalling (Hh) is a developmental conserved pathway strongly involved in cancers when deregulated. This important pathway is orchestrated by numerous regulators, transduces through distinct routes and is finely tuned at multiple levels. In this regard, ubiquitylation processes stand as essential for controlling
[...] Read more.
Hedgehog signalling (Hh) is a developmental conserved pathway strongly involved in cancers when deregulated. This important pathway is orchestrated by numerous regulators, transduces through distinct routes and is finely tuned at multiple levels. In this regard, ubiquitylation processes stand as essential for controlling Hh pathway output. Although this post-translational modification governs proteins turnover, it is also implicated in non-proteolytic events, thereby regulating the most important cellular functions. The HECT E3 ligase Itch, well known to control immune response, is emerging to have a pivotal role in tumorigenesis. By illustrating Itch specificities on Hh signalling key components, here we review the role of this HECT E3 ubiquitin ligase in suppressing Hh-dependent tumours and explore its potential as promising target for innovative therapeutic approaches.
Full article
►▼
Show Figures
Open AccessReview
Targeting the Oncoprotein Smoothened by Small Molecules: Focus on Novel Acylguanidine Derivatives as Potent Smoothened Inhibitors
by
Silvia Pietrobono and Barbara Stecca
Cited by 43 | Viewed by 8951
Abstract
Hedgehog-GLI (HH) signaling was originally identified as a critical morphogenetic pathway in embryonic development. Since its discovery, a multitude of studies have reported that HH signaling also plays key roles in a variety of cancer types and in maintaining tumor-initiating cells. Smoothened (SMO)
[...] Read more.
Hedgehog-GLI (HH) signaling was originally identified as a critical morphogenetic pathway in embryonic development. Since its discovery, a multitude of studies have reported that HH signaling also plays key roles in a variety of cancer types and in maintaining tumor-initiating cells. Smoothened (SMO) is the main transducer of HH signaling, and in the last few years, it has emerged as a promising therapeutic target for anticancer therapy. Although vismodegib and sonidegib have demonstrated effectiveness for the treatment of basal cell carcinoma (BCC), their clinical use has been hampered by severe side effects, low selectivity against cancer stem cells, and the onset of mutation-driven drug resistance. Moreover, SMO antagonists are not effective in cancers where HH activation is due to mutations of pathway components downstream of SMO, or in the case of noncanonical, SMO-independent activation of the GLI transcription factors, the final mediators of HH signaling. Here, we review the current and rapidly expanding field of SMO small-molecule inhibitors in experimental and clinical settings, focusing on a class of acylguanidine derivatives. We also discuss various aspects of SMO, including mechanisms of resistance to SMO antagonists.
Full article
►▼
Show Figures
Open AccessReview
Hedgehog Signaling in Cancer: A Prospective Therapeutic Target for Eradicating Cancer Stem Cells
by
Ita Novita Sari, Lan Thi Hanh Phi, Nayoung Jun, Yoseph Toni Wijaya, Sanghyun Lee and Hyog Young Kwon
Cited by 191 | Viewed by 10643
Abstract
The Hedgehog (Hh) pathway is a signaling cascade that plays a crucial role in many fundamental processes, including embryonic development and tissue homeostasis. Moreover, emerging evidence has suggested that aberrant activation of Hh is associated with neoplastic transformations, malignant tumors, and drug resistance
[...] Read more.
The Hedgehog (Hh) pathway is a signaling cascade that plays a crucial role in many fundamental processes, including embryonic development and tissue homeostasis. Moreover, emerging evidence has suggested that aberrant activation of Hh is associated with neoplastic transformations, malignant tumors, and drug resistance of a multitude of cancers. At the molecular level, it has been shown that Hh signaling drives the progression of cancers by regulating cancer cell proliferation, malignancy, metastasis, and the expansion of cancer stem cells (CSCs). Thus, a comprehensive understanding of Hh signaling during tumorigenesis and development of chemoresistance is necessary in order to identify potential therapeutic strategies to target various human cancers and their relapse. In this review, we discuss the molecular basis of the Hh signaling pathway and its abnormal activation in several types of human cancers. We also highlight the clinical development of Hh signaling inhibitors for cancer therapy as well as CSC-targeted therapy.
Full article
►▼
Show Figures
Open AccessReview
Targeting the Multidrug Transporter Ptch1 Potentiates Chemotherapy Efficiency
by
Anida Hasanovic and Isabelle Mus-Veteau
Cited by 36 | Viewed by 7557
Abstract
One of the crucial challenges in the clinical management of cancer is resistance to chemotherapeutics. Multidrug resistance (MDR) has been intensively studied, and one of the most prominent mechanisms underlying MDR is overexpression of adenosine triphosphate (ATP)-binding cassette (ABC) transporters. Despite research efforts
[...] Read more.
One of the crucial challenges in the clinical management of cancer is resistance to chemotherapeutics. Multidrug resistance (MDR) has been intensively studied, and one of the most prominent mechanisms underlying MDR is overexpression of adenosine triphosphate (ATP)-binding cassette (ABC) transporters. Despite research efforts to develop compounds that inhibit the efflux activity of ABC transporters and thereby increase classical chemotherapy efficacy, to date, the Food and Drug Administration (FDA) has not approved the use of any ABC transporter inhibitors due to toxicity issues. Hedgehog signaling is aberrantly activated in many cancers, and has been shown to be involved in chemotherapy resistance. Recent studies showed that the Hedgehog receptor Ptch1, which is over-expressed in many recurrent and metastatic cancers, is a multidrug transporter and it contributes to the efflux of chemotherapeutic agents such as doxorubicin, and to chemotherapy resistance. Remarkably, Ptch1 uses the proton motive force to efflux drugs, in contrast to ABC transporters, which use ATP hydrolysis. Indeed, the “reversed pH gradient” that characterizes cancer cells, allows Ptch1 to function as an efflux pump specifically in cancer cells. This makes Ptch1 a particularly attractive therapeutic target for cancers expressing Ptch1, such as lung, breast, prostate, ovary, colon, brain, adrenocortical carcinoma, and melanoma. Screening of chemical libraries have identified several molecules that are able to enhance the cytotoxic effect of different chemotherapeutic agents by inhibiting Ptch1 drug efflux activity in different cancer cell lines that endogenously over-express Ptch1. In vivo proof of concept has been performed in mice where combining one of these compounds with doxorubicin prevented the development of xenografted adrenocortical carcinoma tumors more efficiently than doxorubicin alone, and without obvious undesirable side effects. Therefore, the use of a Ptch1 drug efflux inhibitor in combination with classical or targeted therapy could be a promising therapeutic option for Ptch1-expressing cancers.
Full article
►▼
Show Figures