Abstract
Within the Rajidae family, the blonde ray (Raja brachyura) is considered one of the less resilient species to fishing pressure and other anthropogenic pressures, primarily due to its late maturity and large maximum size, which can exceed 120 cm total length. This is the first study to provide comprehensive insights into the reproductive biology of Raja brachyura in the continental waters of Portugal, with insights into its timing, maturity, and fecundity. It was determined that egg-laying occurs from February to November, with a peak observed between April and September. Males were reproductively active throughout the year, with highest proportions of active males observed between January and May. The length at first maturity was estimated at 95.2 cm for females and 90.0 cm for males, corresponding to 85% of the maximum observed length in each sex. The potential fecundity was estimated at 115 follicles per female per year, and evidence suggests that the species has a determinate fecundity. The findings reinforce the appropriateness of current management measures in Portuguese continental waters, namely seasonal closure when overlapping with the peak of the reproductive season (May and June), and provide valuable scientific support for future conservation and management measures.
Key Contribution:
This study provides the first comprehensive characterization of the reproductive biology of Raja brachyura in Portuguese continental waters, detailing its reproductive timing, maturity, and fecundity. The findings strengthen the scientific basis for current management measures and inform future conservation actions.
1. Introduction
Blonde ray (Raja brachyura Lafont, 1871) is a coastal benthic species of the Rajidae family with a wide distribution in the Northeast Atlantic, ranging from Norway to Morocco and the Mediterranean Sea [1,2,3]. It is commonly found in shallow sandy and sand-rocky bottoms, between 20 and 150 m deep [2,4,5].
As an elasmobranch species, and in contrast to most teleost fishes, R. brachyura has a K-strategist life-history pattern, characterized by a long life cycle, slow growth rate, late sexual maturity, low fecundity, extended incubation periods, and large offspring size. These life-history traits render the species vulnerable to fishing pressure [2,4,6,7]. Furthermore, the coastal distribution of R. brachyura increases its exposure to additional anthropogenic pressures, such as pollution, habitat loss, and the impacts of coastal development. This species is classified as “near-threatened” in the global assessment of the International Union for Conservation of Nature (IUCN), primarily due to its apparent rarity in the Mediterranean Sea and declining trends observed in research surveys in Northeast Atlantic waters [8].
In Portuguese continental waters, R. brachyura accounts for approximately 23% of the total landed weight of Rajidae species, which are mainly assigned to the polyvalent fleet (multispecies, multigear fisheries) mainly operating with trammel nets and gillnets [9]. Between 2019 and 2024, this fleet accounted for around 83% of species landings in weight [9,10].
The International Council for the Exploration of the Sea (ICES) provides advice for different stocks of R. brachyura in the Northeast Atlantic. The stock distributed in Portuguese continental waters, stock unit for ICES Division 9.a (west of Galicia, Portugal, and Gulf of Cadiz), has been considered a data-limited stock [11]. Currently, the assessment methodology applied to this stock follows the Maximum Sustainable Yield (MSY) approach, which involves the application of length-based indicators and estimated life-history parameters [10], in particular growth and maturity information. Given the general lack of biological data for R. brachyura in Portuguese continental waters, prioritizing updated information on the life cycle and ecology of this species is essential for effectively monitoring population responses to fishing pressure and other anthropogenic impacts.
While studies on the biology of R. brachyura have been conducted in other geographical areas and/or on different aspects of its life-history [2,3,6,12,13,14,15,16,17,18,19,20,21,22], information regarding its reproductive strategy and life-history parameters in Portuguese continental waters remains limited. In addition, it is known that latitudinal variation in biological parameters can occur in skates [23,24], which reinforces the need to estimate parameters at a regional level. The primary objective of this study is to provide detailed insights into the reproductive biology of R. brachyura, focusing on its seasonal reproductive activity patterns, length at first maturity, and fecundity.
2. Materials and Methods
2.1. Sampling
Specimens of R. brachyura were collected from (i) commercial landings from the Portuguese polyvalent fleet, particularly from two landing ports, Matosinhos (41°11′9.3156″ N, −8°41′52.7022″ W) and Peniche (39°21′22.0926″ N, −9°22′17.493″ W) (Figure 1), under the scope of the National Data Collection Program (PNAB, European Union Data Collection Regulation–Data Collection Framework, DCF) and (ii) bottom trawl research surveys carried out along the Portuguese continental shelf held by the Instituto Português do Mar e da Atmosfera (IPMA), also under PNAB/DCF.

Figure 1.
Map of the study area. Landing ports of Matosinhos and Peniche are indicated, as well as the delimitation of ICES Division 9a.
The following measurements were registered for each sampled specimen: total length (LT) measured to the nearest cm as the distance from the tip of the snout to the end of the tail, gutted body weight (WB), gonad weight (WG), and liver weight (WL), all recorded to the nearest g. For females, additional measurements included oviducal gland width (OGW), anterior uterus width (AUW), and posterior uterus width (PUW), measured in mm. Yolked follicles (here considered follicles with a yellowish color and diameter ≥4 mm) were counted, and their diameter was measured in mm. In males, clasper length (CL) was measured in mm as the distance from the center of the cloaca to the posterior tip of the clasper.
Sex and maturity were assessed through visual inspection of reproductive organs, following the maturity scale proposed by the ICES Workshop on Sexual Maturity Staging of Elasmobranchs (WKMSEL2) for oviparous elasmobranchs [25] (Table 1). For analysis purposes, maturity stages were further classified into immature (immature and developing) and adult groups (capable to reproduce, egg-laying, post-laying, and regenerating individuals).

Table 1.
Maturity scale adopted from WKMSEL2.
2.2. Data Analysis
2.2.1. Reproductive Season
The identification of a reproductive season was determined based on the monthly proportion of each maturity stage within adult male and female specimens. Male and female gonodosomatic (GSI) and hepatosomatic (HSI) indexes across the year, as well as female monthly variation in OGW, were also analyzed. GSI, HSI, and OGW analyses were conducted for females capable of reproducing and egg-laying together, as these are the individuals deemed capable of contributing to reproductive potential [26]. The GSI for each sampled specimen was determined as follows:
and HSI was determined as follows:
where WB is the gutted body weight used in both GSI and HSI calculations to minimize variability associated with the weight of digestive and reproductive tracts. Box plots of the GSI and HSI indexes and OGW were constructed by month for both males and females. Differences between months were assessed using an ANOVA model.
2.2.2. Length at First Maturity
Length at first maturity (L50) for each sex, which corresponds to the length at which 50% of individuals are mature, was estimated by fitting a generalized linear model (GLM) [27] to the proportion of adult females and males by one-centimeter length intervals, considering data collected during the peak of the reproductive season. Females and males in stages 1 and 2 were considered immature, while those in stages capable of reproduction were considered mature. The model considered binomial error distribution and a logit link:
where is the proportion of adult fish at a given length-class interval; is the error term; and and are parameters. The significance of the model was tested with an ANOVA. Percentiles of 25, 50, 75, and 95% of individuals who were mature in condition and size (L25, L50, L75, and L95) were estimated as follows:
2.2.3. Reproductive Organs Development
Morphological changes and development of reproductive organs were analyzed throughout the maturation process. The relationships between LT and OGW, AUW, or PUW for females, as well as between LT and CL for males by maturity stage, were assessed. The null hypothesis of no significant differences in reproductive organ measurements across maturity stages was tested using an ANOVA.
2.2.4. Ovarian Fecundity
Differences in the total number of yolked follicles (with diameters ranging from 4 to 34 mm) across different maturity stages were tested using a post hoc Tukey test. Fecundity was characterized and compared by maturity stage by plotting the frequency distribution of the follicle’s diameter.
The size and number of follicles retained in the ovary after the end of the spawning period (referred to as storage follicles and assumed not to contribute to the reproductive potential but instead continue to develop in the subsequent reproductive cycle) were determined based on the follicle diameter distribution of females at the post-laying stage. The maximum potential fecundity (FM) was estimated in two steps. First, a generalized linear model (GLM) was fit to the individual proportion of follicles larger than the storage follicle size (14 mm) in females contributing to reproductive potential (capable to reproduce and egg-laying), considering the variable month [26]. The month in which the higher proportion was observed was set as the first month (i.e., as a reference for the model). The GLM was specified with a Gaussian error distribution and an identity link function, and is expressed as follows:
where is the individual number of follicles ≥14 mm and is the individual total number of follicles. Finally, FM was estimated as the product of the estimated maximum proportion of follicles and the observed maximum number of follicles.
Statistical analysis was carried out using R 4.4.0 [28], and the level of significance adopted for all the tests was α < 0.05.
3. Results
A total of 1064 R. brachyura specimens (604 females and 460 males) were sampled between 2003 and 2020. Total length ranged from 37 to 111 cm and 41 to 106 cm for females and males, respectively (Figure 2). Although some overlap in total lengths was observed between adjacent maturity stages, a clear increase in body size was evident from immature to reproduction stages, with a median asymptotic length reached around 100 cm in adults. Immature females ranged from 37 to 90 cm LT (n = 159), developing females ranged from 46 to 102 cm LT (n = 256), and adult females ranged (from capable to reproduce to regenerating stages) from 80 to 111 cm LT (n = 189) (Figure 3a). Immature males ranged from 41 to 83 cm LT (n = 99), developing males ranged from 46 to 99 cm TL (n = 213), and adult males (capable to reproduce and being active) ranged from 74 to 106 cm LT (n = 148) (Figure 3b).

Figure 2.
Length distribution of sampled Raja brachyura females and males in 4cm bins.

Figure 3.
Total length (LT, cm) distribution for Raja brachyura (a) females and (b) males by maturity stage: 1–immature, 2–developing, 3a–capable to reproduc, 3b–egg-laying, 4a–post-laying, and 4b–regenerating. Median, standard deviation, and range of total length are shown. Sample size per maturity stage is indicated.
3.1. Reproductive Season
The proportion of females capable to reproduce was highest from January to March, with a peak in March at 76%. Egg-laying females were observed from February to November, with the highest proportion occurring between April and September, when over 50% of the adult specimens were laying eggs (Figure 4a). In the remaining months, the percentage of egg-laying females was below 30%. Post-laying females were most common between August and November, while regenerating females showed higher proportions from November to January. Active males were observed year-round, with the highest proportions recorded between January and May, and again in October, when more than 60% of adult males were active (Figure 4b).

Figure 4.
Monthly proportion of maturity stages in adult Raja brachyura: (a) females (n = 189) and (b) males (n = 148), classified as capable to reproduce, egg-laying (females) or active (males), post-laying or regenerating. Sample size per month is indicated.
Median GSI values of capable to reproduce and egg-laying females peaked in May and gradually decreased until December. In January, GSI values began to gradually increase, reaching higher levels in April. Significant differences in GSI values were observed between months (ANOVA, F = 6.603, d.f. = 11, p < 0.05) (Figure 5a). In contrast, the median GSI values for males showed no significant variation throughout the year (ANOVA, F = 0.931, d.f. = 11, p > 0.05) (Figure 5b).

Figure 5.
Gonadosomatic index (GSI) monthly variation in Raja brachyura, for capable to reproduce and egg-laying/active individuals combined: (a) females (n = 131) and (b) males (n = 142). Range and interquartile range, as well as the median values of GSI, are shown. Sample size per month is indicated.
Median HSI values of capable to reproduce and egg-laying females combined showed a steady decline from January to June, when the lowest values were reached (mean around 4), before a marked increase from September to December (Figure 6a). HSI values differed significantly between months of the year (ANOVA, F = 10.46, d.f. = 11, p < 0.05). For males, median HSI values of capable to reproduce and active individuals were highest between October and March and were lowest from April to September, showing a significant monthly variation (ANOVA, F = 2.59, d.f. = 11, p < 0.05) (Figure 6b).

Figure 6.
Monthly variation in hepatosomatic index (HSI) in Raja brachyura, capable to reproduce and egg-laying/active combined: (a) females (n = 137) and (b) males (n = 147). Range and interquartile range, as well as the median values of HSI, are shown. Sample size per month is indicated.
Median OGW values for capable to reproduce and egg-laying females combined increased from January, attaining their highest values between April and August (Figure 7). After August, median values decreased until December. OGW values showed significant variation across months (ANOVA, F = 2.944, d.f. = 11, p < 0.05).

Figure 7.
Monthly variation in the oviducal gland width (OGW) in Raja brachyura, for capable to reproduce and egg-laying females combined (n = 138). Range and interquartile range, as well as the median values in OGW, are shown. Sample size per month is indicated.
3.2. Length at First Maturity
The estimated L50 for females was 95.2 ± 6.59 cm (ANOVA, F = 386.6, d.f. = 1, p < 0.01, R2 = 0.817, n = 291) (Figure 8). The interquartile length at maturity (L25–L75) was estimated at 93.0–97.4 cm LT, and L95 was estimated at 101.3 cm LT. The largest immature female measured 102 cm LT, while the smallest and largest adult females measured 80 cm LT and 111 cm LT, respectively.

Figure 8.
Raja brachyura maturity ogives for males and females in 1 cm bins. The observed proportion of adult males by LT is represented by (•), while that for females is represented by (○).
The estimated L50 for males was 90.0 ± 8.49 cm (ANOVA, F = 156.6, d.f. = 1, p < 0.01, R2 = 0.55, n = 217) (Figure 8). The interquartile length at maturity (L25–L75) was estimated at 85.9–94.0 cm LT, and L95 was estimated at 100.9 cm LT. The largest immature male measured 99 cm LT, while the smallest and largest mature males measured 76 cm LT and 106 cm LT, respectively.
For both females and males, the estimated L50 corresponded to 85% of the maximum observed LT.
3.3. Reproductive Organs Development
The reproductive structures showed a clear increase in size for both total body length and maturity stage, with the inflection point observed at the estimated L50 (Figure 9). In females with LT < 85 cm, the AUW, PUW, and OGW grew slowly with increasing LT, but accelerated as females approached maturity, particularly around 95.2 cm LT (Figure 9a–d). In smaller males (<80 cm LT), CL increased gradually with LT, before increasing at a faster rate as maturity was reached, around 90.0 cm LT (Figure 9d). Statistically significant differences were observed across female maturity stages in AUW (ANOVA, F = 272.7, d.f. = 2, p < 0.05), PUW (ANOVA, F = 732.9, d.f. = 2, p < 0.05), and OGW (ANOVA, F = 563.7, d.f. = 2, p < 0.05). Similarly, males exhibited statistically significant differences in CL across maturity stages (ANOVA, F = 613.9, d.f. = 2, p < 0.05).

Figure 9.
Raja brachyura relationship between total length (LT) and (a) anterior uterus width (AUW), (b) posterior uterus width (PUW), and (c) oviducal gland width (OGW) for females and between (d) clasper length (CL) for males by maturity stage. Symbols represent the different maturity stages: (○) immature; (+) developing; and (•) adults (capable to reproduce, egg-laying/active, regressing and regenerating combined). The dashed grey line corresponds to the estimated L50 for females (95.2 cm) and males (90.0 cm).
3.4. Ovarian Fecundity
The total number of follicles among females contributing to reproductive potential (i.e., capable to reproduce and egg-laying) and those in other stages showed significant differences (Tukey’s HSD, p < 0.05). Based on these results, two groups were defined: (1) developing, post-laying, and regenerating females, and (2) capable to reproduce and egg-laying females.
The follicle frequency distribution in developing (pre-laying), post-laying (immediately after egg-laying season) and regenerating (preparing for the next egg-laying season) females was dominated by follicles of 6 to 14 mm in diameter (Figure 10); therefore, a diameter of 14 mm was considered to represent a threshold that distinguishes these maturity stages from those contributing to the reproductive potential of the population during the egg-laying season. Post-laying females still presented a small number of follicles 34 mm entering atresia. Additionally, follicles <14 mm that were not released during the spawning season could be considered storage follicles, which will continue to develop in the subsequent reproductive cycle. The maximum number of storage follicles observed in regressing females was 36.

Figure 10.
Follicle frequency distribution by diameter class (in mm) from Raja brachyura females at different maturity stages: (a) developing (n = 7), (b) post-laying (n = 5), and (c) regenerating (n = 23).
As no significant differences were found in the follicle distribution between capable to reproduce and egg-laying females, the analysis of fecundity was performed by combining observations from both maturity stages. The follicle size distribution in these females followed a similar overall pattern (Figure 11). However, when comparing the proportion of small (<14 mm) and large (14 mm) follicles between the two maturity stages, significant differences were found (ANOVA: F = 22.14, d.f. = 1, p < 0.05). The mean proportion of small diameter classes was higher in capable to reproduce females, while the mean proportion of large diameter classes was higher in egg-laying females.

Figure 11.
Frequency of follicle distribution for Raja brachyura females by diameter class in (a) capable to reproduce (n = 52) and (b) egg-laying (n = 60) stages.
In females considered to be contributing to reproductive potential (females capable to reproduce and egg-laying), the number and the monthly variation in the proportion of follicles <14 mm and 14 mm in relation to the total number of follicles showed opposite patterns throughout the year (Figure 12). The proportion of follicles <14 mm was higher from August to March, while that for follicles 14 mm was higher in April and May, followed by a gradual decrease until September (Figure 12). In April, both follicle groups corresponded to around 50% of the follicles in the ovaries. Figure 13 illustrates these findings, showcasing the follicle distribution in three contexts: (1) a female at the beginning of the reproductive season (April, 100 cm LT); (2) a female in the middle of the reproductive season (June, 105 cm LT); and (3) a female nearing the end of the reproductive season (August, 103 cm LT). Globally, the number of follicles <14 mm decreased throughout the reproductive season, reflecting their development and subsequent transition into the pool of follicles 14 mm, which are released during the actual egg-laying season.

Figure 12.
Monthly variation in the proportion and number of follicles (top row) <14 mm and (bottom row) >14 mm in Raja brachyura females capable to reproduce and egg-laying.
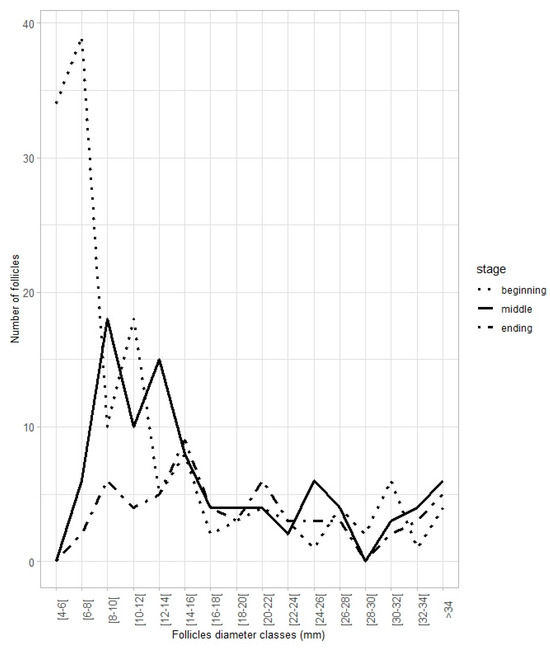
Figure 13.
Follicle frequency distribution for egg-laying Raja brachyura females in different phases of the spawning season: beginning, middle, and ending.
April was set as the first month (i.e., as a reference for the model) for the GLM to estimate the maximum potential fecundity, since this was the month in which the maximum proportion of follicles 14 mm (0.94) was observed in a female. Results from the model indicate that the highest mean estimate of the proportion of follicles 14 mm corresponded to 0.76 ± 0.10, estimated for the month of May. Considering the maximum number of yolked follicles (152) observed in an egg-laying female (106 cm LT) (in March, the month immediately preceding the egg-laying peak), this leads to an estimated maximum potential fecundity of 115 follicles per female per year (Akaike information criterion, AIC = −105.9, R2adj = 0.65). The remaining proportion (0.24, 36 follicles) is assumed to correspond to storage follicles.
4. Discussion
The present study provides the first insight into the reproductive biology of Raja brachyura in Portuguese continental waters, highlighting key aspects of its reproductive timing, maturity, and fecundity. Although this species has a wide distribution across the Northeast Atlantic and Mediterranean Sea, such information was limited to the Irish Sea [17], British Isles [12,22], and parts of the Mediterranean Sea [3,20]. Given its high susceptibility to fisheries and to other anthropogenic pressures, understanding the reproductive biology of R. brachyura is crucial for assessing population status and developing effective management and conservation strategies, both in Portugal and across its distributional range.
Results suggest that, in Portugal, R. brachyura reproduces from February to November, exhibiting a prolonged but seasonal reproductive cycle, with a clear peak during spring and summer. The absence of egg-laying females in the winter months suggests the existence of a resting period, rather than continuous year-round reproduction. Within the reproductive season, not all adult females are reproductively active simultaneously, indicating individual-level asynchrony in the reproductive cycle at the population level.
The monthly variability in GSI, HSI, and OGW further supports this hypothesis, indicating the coexistence of females at different reproductive stages and highlighting the existence of individual biological rhythms. Similar patterns have been observed for other Rajidae species inhabiting Portuguese continental waters, such as the thornback ray Raja clavata [29], cuckoo ray Leucoraja naevus [30], and undulate ray Raja undulata [27], for which long spawning periods with a peak were observed. It is important to note that sampling was opportunistic and carried on for a long period of time (2003–2020), which means that interannual variation was not considered. Despite some level of asynchrony in the reproductive timing at the population level, over 60% of the sampled adult females were egg-laying between April and September, indicating the existence of a reproductive peak (also supported by the monthly variation in the proportion of post-laying and regenerating females). Notably, the highest proportion of capable to reproduce females was observed in March, the month immediately preceding the egg-laying peak. Higher GSI values in May and their progressive decrease during the following months illustrate the release of follicles during the reproductive season. The HSI exhibited a temporal pattern that decreased from January to June, reflecting the conditioning of energy to produce gametes, vitellogenesis, and egg capsule formation [31]. Monthly variation in the OGW also corroborated the existence of an egg-laying peak, with higher values being registered between April and August, when egg capsules were produced by a higher proportion of females. For R. brachyura, the existence of an egg-laying peak during the spring and summer months was also observed in other geographic areas, such as the Southwest of England [12] and in the Mediterranean Sea [3,20]. Holden et al. [13] suggested that temperature serves as a crucial environmental cue influencing reproductive timing and that warmer waters occurring during spring and summer seem to stimulate egg deposition. In the center of Portugal, in particular in the vicinity of Peniche landing port, where the great majority of our sampled specimens came from, average monthly water surface temperature during the study period started to increase in April, reaching the highest values in August when the average temperature was 18 °C, and progressively decreasing afterwards to less than 14 °C during autumn and winter months (EU Copernicus Marine Service information-https://marine.copernicus.eu, accessed on 20 January 2025).
The estimated L50 for R. brachyura in Portuguese continental waters was 95.2 cm for females and 90.0 for males, which is higher than the estimates available for other geographic regions. In the Northeast Atlantic, first maturity was estimated at 83.6 for females and 81.9 cm for males in the Irish Sea [17] and as 83.4 for females and 78 cm for males in British waters [22]. In the Sardinian Seas, the L50 was estimated at 87.2 and 80.8 cm for females and males, respectively [3]. In the present study, the largest immature female measured 102 cm, contrasting with previous studies, with reported lengths of around 92 cm [3,22]. These differences may arise from several factors, including environmental variations, influence of water temperature, species behavior, and different levels of fishing pressure. High levels of fishing exploitation can impact population densities and lead to changes in life-history traits, such as length at first maturity [32]. For example, increased growth rates and decreased length at first maturity due to rising fishing mortality have been reported for R. clavata in the central and northwestern North Sea [16]. Similar trends have been noted for other elasmobranch species, such as, for example, the lesser spotted dogfish Scyliorhinus canicula in the Mediterranean Sea [32] or the yellownose skate Dipturus chilensis in the southwestern Atlantic Ocean [33].
The analysis of the reproductive organs across maturity stages demonstrates quick development close to the length at first maturity (L50), supporting the established profile of maturation processes in elasmobranchs [23,34,35]. Unlike most teleost species, elasmobranchs tend to mature late in their life cycle, reaching maturity at approximately 75 to 90% of their maximum LT, which is characteristic of K-strategist species [34]. In Portuguese continental waters, R. brachyura reaches sexual maturity at approximately 85% of the maximum LT sampled (111 and 106 cm for females and males, respectively), showing an extended immature phase and minimal growth post-maturity. The estimated L50 for R. brachyura in Portuguese waters differed between sexes, which is consistent with findings from other geographic regions [3,6,22]. Such sexual dimorphism is common among elasmobranch species [34]. The larger size and age at maturity of female rajids likely relate to differing reproductive strategies between sexes [36]. For females, a larger size provides more abdominal space for carrying yolked follicles [3,29], while males’ faster growth rates enable them to reach sexual maturity sooner. These traits may optimize reproductive success in these species [2].
Holden et al. [13] observed, in captivity, the release of 90 eggs per female over a 77-day period. This is comparable to the results of the present study (115 follicles per female per year) when considering the full spawning season in Portuguese continental waters, which lasts approximately six months (about 180 days). In the Mediterranean Sea, Porcu et al. [3] reported an ovarian fecundity of 44 yolked follicles based on observations of 18 mature females. However, this estimate does not account for the continuous recruitment of small developing follicles into the ovulatory oocyte pool throughout the spawning season. In the present study, differences in follicle frequency distribution, particularly in small follicles, between females at the beginning and at the end of the spawning season illustrate this continuous follicular development throughout the reproductive cycle. The persistence/permanence of small developing follicles in females that reached the end of the reproductive season indicates that vitellogenesis is a continuous process occurring year-round once individuals reach maturity. Nevertheless, differences in biological parameters between the Northeast Atlantic and Mediterranean waters are not unexpected and have been reported for several elasmobranch species [37,38].
In females contributing to reproductive potential, the proportion of small follicles was higher in the first quarter of the year, immediately preceding the reproductive season. As the reproductive peak approached, these follicles underwent vitellogenesis, developing into larger yolked follicles ready for ovulation and fertilization. By the end of April, the proportion of large follicles peaked, followed by a gradual decline throughout the egg-laying season as egg capsules were released onto the sea floor. By the end of the reproductive season, the few large follicles that remained in the ovaries were likely to undergo atresia, while the remaining small follicles were retained, continuing to slowly develop, and contribute to the next egg-laying season. The observed decrease in small follicles along the egg-laying season, together with the absence of large follicles at its end, supports the hypothesis that R. brachyura exhibits determinate fecundity.
5. Conclusions
This study further supports the measures currently in place in Portuguese continental waters for the management and conservation of R. brachyura. In particular, a fishing closure period applied to all fisheries capturing Rajidae is implemented during May and June (Portaria nº 47/2016). This seasonal closure overlaps with the reproductive peak identified in this study, reinforcing its relevance as an important conservation measure aimed at protecting reproductive females. Additionally, a minimum landing size of 52 cm TL was established in 2014 for all Rajidae species (Portaria no 170/2014), which was later updated to 60 cm TL (Portaria nº 255/2022). While this may provide some degree of protection to immature R. brachyura, our findings indicate that this management measure is not sufficient to protect adult individuals, since it is far lower than the estimated length at first maturity for both females and males. To support effective conservation and sustainable exploitation of R. brachyura, further studies on growth, habitat use, and species resilience are essential. These will enhance understanding of the species’ population dynamics, productivity, and susceptibility to environmental and human pressures, and inform the proposal of additional or refined protection measures as needed.
Author Contributions
Conceptualization, C.M., I.F. (Ivone Figueiredo) and B.S.-P.; Methodology, C.M., B.S.-P. and T.M.; Software, C.M. and B.S.-P.; Validation, T.M. and B.S.-P.; Formal analysis, C.M., T.M. and B.S.-P.; Biological sampling, N.L., C.M. and I.F. (Inês Farias); Data curation, N.L.; Writing—original draft preparation, C.M.; Writing—review and editing, C.M., T.M., I.F. (Inês Farias), B.S.-P., I.F. (Ivone Figueiredo) and N.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the IPMA Portuguese National Programme for Biological Sampling (PNAB), integrated in the EU Data Collection Framework (DCF).
Institutional Review Board Statement
Ethical review and approval were waived for this study because all specimens consisted of animals that were already deceased and commercially acquired from auction markets at landing ports. No animals were killed, handled, or subjected to experimental procedures for the purposes of this research.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data of this research are available upon request.
Acknowledgments
The authors would like to thank to the PNAB team for their help collecting samples for this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Stehmann, M.; Bürkel, D.L. Rajidae. In Fishes of the North-Eastern Atlantic and Mediterranean; Whitehead, P.J.P., Bauchot, M.-L., Hureau, J.-C., Nielsen, J., Tortonese, E., Eds.; UNESCO: Paris, France, 1984; Volume 1, pp. 163–196. [Google Scholar]
- Catalano, B.; Dalù, M.; Scacco, U.; Vacchi, M. New biological data on Raja brachyura (Chondrichthyes, Rajidae) from around Asinara Island (NW Sardinia, Western Mediterranean). Ital. J. Zool. 2007, 74, 55–61. [Google Scholar] [CrossRef]
- Porcu, C.; Bellodi, A.; Cannas, R.; Marongui, M.F.; Mulas, A.; Follesa, M.C. Life-history traits of the commercial blonde ray, Raja brachyura, from the central-western Mediterranean Sea. Mediterr. Mar. Sci. 2015, 16, 90–102. [Google Scholar] [CrossRef]
- Ellis, J.R.; Cruz-Martínez, A.; Rackham, B.D.; Rogers, S.I. The distribution of chondrichthyan fishes around the British Isles and implications for conservation. J. Northwest Atl. Fish. Sci. 2005, 35, 195–213. [Google Scholar] [CrossRef]
- Serra-Pereira, B.; Erzini, K.; Maia, C.; Figueiredo, I. Identification of potential essential fish habitats for skates based on Fishers’ knowledge. Environ. Manag. 2014, 53, 985–998. [Google Scholar] [CrossRef]
- Thys, K.J.; Lemey, L.; Van Bogaert, N. Blondes do it better? A comparative study on the morphometry and life-history traits of commercially important skates blonde ray Raja brachyura, thornback ray Raja clavata, and spotted ray Raja montagui, with management implications. Fish. Res. 2023, 263, 106679. [Google Scholar] [CrossRef]
- Elliott, S.A.M.; Carpentier, A.; Feunteun, E.; Trancart, T. Distribution and life history trait models indicate vulnerability of skates. Prog. Oceanogr. 2020, 181, 102256. [Google Scholar] [CrossRef]
- IUCN Red List of Threatened Species. Available online: www.iucnredlist.org (accessed on 21 May 2024).
- Figueiredo, I.; Maia, C.; Lagarto, N.; Serra-Pereira, B. Bycatch estimation of Rajiformes in multispecies and multigear fisheries. Fish. Res. 2020, 232, 105727. [Google Scholar] [CrossRef]
- ICES. Report of the Working Group on Elasmobranch Fishes (WGEF). ICES Sci. Rep. 2024, 6, 994. [Google Scholar]
- ICES. Working Group on Elasmobranch Fishes (WGEF). ICES Sci. Rep. 2021, 3, 822. [Google Scholar]
- Clark, R.S. Rays and skates (Raiae). N. 1. Eggs capsulae and young. J. Mar. Biol. Assoc. UK 1922, 12, 577–643. [Google Scholar] [CrossRef]
- Holden, M.J.; Rout, D.W.; Humphreys, C.N. The rate of egg laying by three species of ray. ICES J. Mar. Sci. 1971, 33, 335–339. [Google Scholar] [CrossRef]
- Ryland, J.S.; Ajayi, T.O. Growth and population dynamics of three Raja species (batoidea) in Carmarthen Bay, British Isles. J. Cons. Int. Explor. Mer. 1984, 41, 111–120. [Google Scholar] [CrossRef]
- Ellis, J.R.; Pawson, M.G.; Shackley, S.E. The comparative feeding ecology of six species of shark and four species of ray (Elasmobranchii) in the North-East Atlantic. J. Mar. Biol. Assoc. UK 1996, 76, 89–106. [Google Scholar] [CrossRef]
- Walker, P.A.; Hislop, J.R.G. Sensitive skates or resilient rays? Spatial and temporal shifts in ray species composition in the central and north-western North Sea between 1930 and the present day. ICES J. Mar. Sci. 1998, 55, 392–402. [Google Scholar] [CrossRef]
- Gallagher, M.J.; Nolan, C.P.; Jeal, F. Age, growth and maturity of the commercial ray species from the Irish Sea. J. Northwest Atl. Fish. Sci. 2005, 35, 47–66. [Google Scholar] [CrossRef]
- Farias, I.; Figueiredo, I.; Moura, T.; Gordo, L.S.; Neves, A.; Serra-Pereira, B. Diet comparison of four ray species (Raja clavata, Raja brachyura, Raja montagui and Leucoraja naevus) caught along the Portuguese continental shelf. Aquat. Living Resour. 2006, 19, 105–114. [Google Scholar] [CrossRef]
- Follesa, M.C.; Mulas, A.; Cabidou, S.; Porcu, C.; Deiana, A.M.; Cau, A. Diet and feeding habits of two ray species, Raja brachyura and Raja miraletus (Chondrichthyes, Rajidae) in Sardinian waters (central-western Mediterranean). Ital. J. Zool. 2010, 77, 53–60. [Google Scholar] [CrossRef]
- Porcu, C.; Mulas, A.; Cabiddu, S.; Gastoni, A.; Follesa, M.C. Reproductive biology of the blonde ray Raja brachyura (Chondrichthyes: Rajidae) in Sardinian Seas. Biol. Mar. Mediterr. 2010, 17, 256–257. [Google Scholar]
- Serra-Pereira, B.; Farias, I.; Moura, T.; Gordo, L.S.; Santos, M.; Figueiredo, I. Morphometric ratios of six commercially landed species of skate from the Portuguese continental shelf, and their utility for identification. ICES J. Mar. Sci. 2010, 67, 1596–1603. [Google Scholar] [CrossRef]
- McCully, S.R.; Scott, F.; Ellis, J.R. Lengths at maturity and conversion factors for skates (Rajidae) around the British Isles, with an analysis of data in the literature. ICES J. Mar. Sci. 2012, 69, 1812–1822. [Google Scholar] [CrossRef]
- Frisk, M.G.; Miller, T.J. Age, growth, and latitudinal patterns of two Rajidae species in the northwestern Atlantic: Little skate (Leucoraja erinacea) and winter skate (Leucoraja ocellata). Can. J. Fish. Aquat. Sci. 2006, 63, 1078–1091. [Google Scholar] [CrossRef]
- Licandeo, R.; Cerna, F.T. Geographic variation in life-history traits of the endemic kite skate Dipturus chilensis (Batoidea: Rajidae), along its distribution in the fjords and channels of southern Chile. J. Fish. Biol. 2007, 71, 421–440. [Google Scholar] [CrossRef]
- ICES. Report of the Workshop on Sexual Maturity Staging of Elasmobranchs (WKMSEL2); ICES: Lisbon, Portugal, 2013; p. 113. [Google Scholar]
- Serra-Pereira, B.; Erzini, K.; Figueiredo, I. Using biological variables and reproductive strategy of the undulate ray Raja undulata to evaluate productivity and susceptibility to exploitation. J. Fish. Biol. 2015, 86, 1471–1490. [Google Scholar] [CrossRef] [PubMed]
- McCullagh, P.; Nelder, J.A. Generalized Linear Models, 2nd ed.; Chapman & Hall: London, UK, 1989; p. 532. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. In R Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2024; Available online: https://www.R-project.org/ (accessed on 10 April 2024).
- Serra-Pereira, B.; Figueiredo, I.; Gordo, L.S. Maturation of the gonads and reproductive tracts of the thornback ray Raja clavata, with comments on the development of a standardized reproductive terminology for oviparous elasmobranchs. Mar. Coast. Fish. 2011, 3, 160–175. [Google Scholar] [CrossRef]
- Maia, C.; Erzini, K.; Serra-Pereira, B.; Figueiredo, I. Reproductive biology of cuckoo ray Leucoraja naevus. J. Fish Biol. 2012, 81, 1285–1296. [Google Scholar] [CrossRef] [PubMed]
- Oddone, M.C.; Velasco, G. Relationship between liver weight, body size and reproductive activity in Atlantoraja cyclophora (Elasmobranchii: Rajidae: Arhynchobatinae) in oceanic waters off Rio Grande do Sul, Brazil. Neotrop. Biol. Conserv. 2006, 1, 12–16. [Google Scholar]
- Ramírez-Amaro, S.; Ordines, F.; Esteban, A.; García, C.; Guijarro, B.; Salmerón, F.; Terrasa, B.; Massutí, E. The diversity of recent trends for chondrichthyans in the Mediterranean reflects fishing exploitation and a potential evolutionary pressure towards early maturation. Sci. Rep. 2020, 10, 547. [Google Scholar] [CrossRef]
- Paesch, L.; Oddone, M.C. Change in size-at-maturity of the yellownose skate Dipturus chilensis (Guichenot, 1848) (Elasmobranchii: Rajidae) in the SW Atlantic. Neotrop. Ichthyol. 2008, 6, 223–230. [Google Scholar] [CrossRef]
- Ebert, D.A. Reproductive biology of skates, Bathyraja (Ishiyama), along the eastern Bering Sea continental slope. J. Fish Biol. 2005, 66, 618–649. [Google Scholar] [CrossRef]
- Oddone, M.C.; Vooren, C.M. Reproductive biology of Atlantoraja cyclophora (Regan 1903) (Elasmobranchii: Rajidae) off southern Brazil. ICES J. Mar. Sci. 2005, 62, 1095–1103. [Google Scholar] [CrossRef]
- Gayford, J.H.; Sternes, P.C. The origins and drivers of sexual dimorphism in sharks. Ecol. Evol. 2025, 15, e71858. [Google Scholar] [CrossRef] [PubMed]
- Villagra, D.; Van Bogaert, N.; Ampe, B.; Walker, P.; Uhlmann, S.S. Life-history traits of batoids (Superorder Batoidea) in the Northeast Atlantic and the Mediterranean. Rev. Fish Biol. Fish. 2022, 32, 473–495. [Google Scholar] [CrossRef]
- Gubili, C.; Sims, D.W.; Veríssimo, A.; Domenici, P.; Ellis, J.; Grigoriou, P.; Johnson, A.F.; McHugh, M.; Neat, F.; Satta, A.; et al. A tale of two seas: Contrasting patterns of population structure in the small-spotted catshark across Europe. R. Soc. Open Sci. 2014, 1, 140175. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.













