Abstract
This study investigated the development of a sugar-free prebiotic and probiotic dark chocolate formulated with 39.76% cocoa mass, 41.856% cocoa butter, 14.8% inulin, and 1.6% stevioside. To enhance the stability of the probiotic yeast Saccharomyces boulardii, a microencapsulation technique using a cocoa powder, Na-alginate, and fructooligosaccharides (10:1:1 ratio) matrix was employed. The physicochemical properties and probiotic viability were monitored over a 120-day storage period at 25 °C. A significant increase in pH values was observed during storage (p < 0.05), while water activity (aw) values significantly decreased (p < 0.05). Encapsulation provided superior protection, with encapsulated samples retaining 2.51 log CFU/g more probiotics than unencapsulated samples after 120 days. Furthermore, in vitro gastrointestinal digestion assays conducted after one day of storage demonstrated the protective efficacy of the matrix; while unencapsulated probiotics suffered a drastic reduction of 6.42 log CFU/g under gastric conditions (pH 3, 2 h), the encapsulated probiotics showed a significantly lower reduction of only 3.99 log CFU/g. These results confirm that the Na-alginate/FOS-based encapsulation significantly improves the resilience of S. boulardii against both storage conditions and gastrointestinal stress, making this sugar-free prebiotic chocolate an effective delivery vehicle for probiotic yeasts.
1. Introduction
The growing interest in functional foods that offer significant health benefits continues to prompt the scientific community to undertake further research [1]. Consistent consumption of adequate live probiotics through food exerts positive physiological effects on the gut microbiota [2]. Reflecting this importance, the global focus on probiotics continues to increase [3]. To achieve these health benefits, the International Dairy Federation recommends maintaining a concentration of 106–107 CFU/g of live probiotics at the time of intake [4]. Consequently, there is an increasing focus within the food industry on preserving the viability of probiotics through various innovative methodologies. However, ensuring this viability remains a significant challenge [5]. Specifically, gastric acid, bile salts, and various digestive enzymes can drastically hinder the survival of probiotics during gastrointestinal transit [4]. Furthermore, the rigorous conditions encountered during the processing and storage of food products pose additional threats to probiotic survival [6]. To demonstrate their systemic efficacy, probiotics must remain viable despite these stresses [7]. Since environmental conditions during food storage often lead to a loss of viability, encapsulation has emerged as a robust method to shield probiotics from adverse surroundings [8]. Accordingly, the encapsulation method has been strategically preferred in this study.
Tolerance to acidity (pH 1.5–3.0) and bile salts (0.3% w/v) represents a fundamental selection criterion for potential probiotic yeast strains to endure the rigorous conditions of the gastrointestinal system [9]. While the probiotic potential of yeasts in traditional fermented foods has not been as extensively documented as that of bacterial probiotics, recent research has reviewed their functional possibilities, indicating that further investigation is warranted [10]. Our study contributes to the literature in this field by employing a newly isolated probiotic yeast strain. Furthermore, recent reviews exploring the health benefits, mechanisms of action, and technological applicability of S. boulardii in various food systems have further emphasized its industrial and therapeutic significance [10].
Parallel to these developments, the heightened awareness of health issues associated with excessive sucrose consumption [11] has prompted researchers to investigate “sucrose-free” alternatives in chocolate formulation. Conventional sucrose substitutes typically include a combination of various sweeteners and bulking agents. High-intensity sweeteners, such as stevia and aspartame, offer sweetness levels several hundred to a thousand times greater than sucrose; however, they lack the necessary volume-forming properties. Research has demonstrated that integrating these sweeteners with appropriate bulking agents can ensure desirable physical, rheological, and sensory characteristics in low-calorie products [12,13]. Beyond its gelling properties, inulin provides significant functional benefits as a dietary fiber [14]. Furthermore, inulin acts as a potent prebiotic, enhancing host health by selectively promoting the growth of beneficial microorganisms [15]. Stevia, a zero-calorie natural sweetener derived from the Stevia rebaudiana plant, similarly lacks volume-forming capabilities [12], necessitating its pairing with a bulking agent like inulin to effectively replicate the functional role of sucrose. Based on these considerations, a combination of inulin and stevia was utilized in this study as a sugar replacer to develop a healthier chocolate product.
Despite the global expansion of the chocolate market [16], its potential as a functional food matrix for probiotic delivery remains relatively under-researched [17]. The worldwide popularity of chocolate, coupled with the heightened health consciousness of contemporary consumers, has generated significant demand for the enrichment of various chocolate formulations with probiotics [17]. Consequently, chocolate was selected as the delivery vehicle for this study, as the encapsulation of probiotics within a chocolate matrix appears to be a highly effective strategy for preserving microbial viability against environmental stressors [18].
Encapsulation represents one of the most straightforward and effective techniques for facilitating the processing, storage, and resilience of probiotics within the gastrointestinal system [19]. The successful delivery of targeted probiotics is highly dependent on the selection of appropriate encapsulating matrices. In this context, chocolate—characterized by a complex composition of proteins, polysaccharides, and lipids—can be integrated with cocoa powder, alginate, or fructooligosaccharides (FOS) to create a robust and well-suited encapsulating mixture [20]. Freeze-drying has been identified as an optimal method for preserving probiotic integrity during encapsulation [21]. To the best of our knowledge, this is the first study to evaluate the synergistic effect of a sucrose-free matrix and a specific cocoa–alginate–prebiotic blend on yeast stability. Accordingly, the objective of this study was to develop a sucrose-free probiotic dark chocolate enriched with encapsulated S. boulardii and to evaluate its storage stability over 120 days, as well as its in vitro gastrointestinal survival, while also investigating the changes in physicochemical properties and fatty acid profiles to assess the overall quality of the product.
2. Materials and Methods
2.1. Freeze-Drying Encapsulation of Probiotics and Encapsulation Efficiency
The probiotic strain used in this study, S. boulardii, was previously isolated from traditional dairy products and molecularly identified by our research group [22]. Identification was confirmed through the sequencing of the ITS1-5.8S rRNA and partial regions of the ITS2-18S and 28S rRNA genes, showing 98% similarity with the S. boulardii reference strain MT449154.1 in the NCBI database. The strain, previously characterized for its superior probiotic properties [22], is currently maintained in our department’s culture collection for reference.
The encapsulating matrix was formulated based on optimized combinations from literature, specifically utilizing a cocoa powder: Na-alginate (Sigma Aldrich, Steinheim, Germany): FOS (Sigma Aldrich, Germany) ratio of 10:1:1 (w/w/w) [23]. The probiotic yeast was aerobically cultivated at 27 °C for 72 h in a selective Tryptic Soy Broth (Merck, Darmstadt, Germany) supplemented with 0.5% (w/v) yeast extract to enhance nitrogen availability and promote optimal biomass yield. Biomass was harvested via centrifugation at 5000× g for 15 min at 4 °C, followed by two consecutive washes with 0.85% (w/v) physiological saline. The encapsulation process was adapted from the methodology described by Hossain et al. [23].
The encapsulation components were dissolved in Milli-Q water and homogenized at 10,000 rpm for 15 min (IKA T25 digital Ultraturrax, Staufen, Germany). The mixture was allowed to hydrate at room temperature for 2 h before being pasteurized in a water bath at 75 °C for 30 min and subsequently cooled to 42 °C [24]. The resulting yeast pellets (yielding approximately 12 log CFU/g) were integrated into the matrix and homogenized at 4000 rpm for 5 min. Following a 1 h stabilization period at room temperature to facilitate biopolymer-probiotic interactions [25], the mixture was aliquoted into 50 mL sterile Falcon tubes and frozen at −20 °C. Lyophilization was performed at −40 °C for 48 h using a freeze-dryer (Labconco Freezone 6, Kansas City, MO, USA), under a vacuum pressure of 0.050 mbar. The lyophilization process was monitored until a constant weight was achieved. The effectiveness of the process was verified by the initial water activity values (aw) of the chocolates (0.367–0.387), ensuring a suitable environment for probiotic stability. The encapsulation efficiency (EE%) was determined using the following equation [25]:
where N represents the number of viable cells after encapsulation (log CFU/g) and N0 is the number of viable cells before encapsulation (log CFU/g).
% EE = (N/N0) × 100
2.2. Chocolate Preparation
The chocolate was formulated with 39.76% cocoa mass, 41.856% cocoa butter, 14.8% inulin, 1.6% stevioside, and 0.15% soy lecithin [26,27]. Due to the high total fat content of the formulation, the ingredients were combined and homogenized using a high-shear laboratory mixer at 50 °C for 60 min, ensuring rapid and uniform integration of the solid phase into the lipid phase without the need for traditional long-term conching. To maintain probiotic viability, the encapsulated yeast was incorporated into the chocolate matrix at a concentration of 1.84% (w/w) once the temperature reached 45 °C during the tempering stage. The incorporation was specifically performed during the cooling phase of the tempering process. While the chocolate was initially melted and held at 45 °C, the temperature was gradually lowered, and the encapsulated yeast was introduced and thoroughly mixed when the mass reached 31 °C. This temperature was maintained to prevent thermal degradation of the probiotic cells while ensuring proper lipid crystallization. The final formulation was designed to ensure a minimum probiotic load of 107 CFU/g [28]. Both probiotic-enriched chocolate (containing encapsulated yeast) and control samples (containing non-encapsulated yeast) were stored under aseptic conditions at 25 °C for 120 days to monitor stability [29].
2.3. Moisture, pH, and Water Activity (aw) Analysis
Moisture content was determined according to the AOAC 931.04 method [30] before and after the 120-day storage period. Samples were analyzed in triplicate to ensure accuracy during long-term monitoring.
2.4. Textural Analysis
Chocolate hardness was assessed before and after storage using a Brookfield, Middleboro, MA, USA CT3 Texture Analyzer, following the protocol described by Biswas et al. [31].
2.5. Bloom Formation and Surface Morphology
Visual and microscopic evaluation of fat/sugar bloom was conducted biweekly. Surface changes and crystal formations were recorded using a stereomicroscope according to the methods of Kinta and Hartel [32].
2.6. pH and Water Activity (aw) Measurement
pH changes in the chocolate samples were measured using a Mettler-Toledo, Columbus, OH, USA pH meter. To avoid fat interference, 5 g of chocolate was homogenized in 45 mL of distilled water (40 °C), and the pH was measured from the aqueous phase after phase separation. Water activity (aw) was determined using a dew-point water activity meter (Aqualab Series 3TE, METER Group, Pullman, WA, USA). Analyses of these two parameters were conducted at 30-day intervals over the entire 120-day storage period, as they can directly impact the viability and metabolic performance of the probiotics [33].
2.7. Microstructural Analysis via Scanning Electron Microscopy (SEM)
Scanning Electron Microscopy (SEM) was employed to characterize the microstructure of freeze-dried encapsulated S. boulardii and its integration within the chocolate matrix. The lyophilized powder and chocolate samples were mounted onto aluminum stubs using double-sided adhesive carbon tape. Excess particles were removed with a dry air jet. Samples were coated with platinum using a sputter coater for 60 s at 18 mA. Morphological observations were conducted at an acceleration voltage of 10 kV at various magnifications to visualize the surface topography of the encapsulated yeast before and after incorporation into the chocolate [34].
2.8. Probiotic Viability During Storage
The viability of S. boulardii was monitored at 30-day intervals throughout the 120-day storage period at 25 ˚C. Chocolate samples (25 g) were homogenized with 225 mL of sterile 0.1% (w/v) peptone water. The mixture was subjected to vigorous mechanical homogenization in a stomacher for 2 min to ensure the maximum possible release of yeast cells from the lipid matrix into the aqueous phase. Serial dilutions were performed, and the yeast count was determined by plating onto Potato Dextrose Agar (PDA, Merck, Germany) followed by aerobic incubation at 27 °C for 72 h. Results were expressed as log CFU/g [23].
2.9. In Vitro Gastrointestinal Digestion Simulation
The resilience of encapsulated and non-encapsulated S. boulardii during gastrointestinal transit was evaluated according to the standardized in vitro INFOGEST protocol [35]. The simulation consisted of three sequential phases:
Oral Phase: Chocolate samples (2.5 g) were mixed with 1.75 mL of simulated salivary fluid (SSF), 0.25 mL of salivary α-amylase (1500 U/mL), 12.5 μL of 0.3 M CaCl2, and 487.5 μL of Milli-Q water. The mixture was vortexed for 2 min to simulate mastication.
Gastric Phase: The oral bolus was combined with 3.75 mL of simulated gastric fluid (SGF), 0.8 mL of pepsin (3200–4500 U/mg), and 2.5 μL of 0.3 M CaCl2. The pH was adjusted to 3.0 using 1 M HCl, and the mixture was incubated in a shaking incubator at 37 °C for 2 h under anaerobic conditions.
Intestinal Phase: The gastric chyme was mixed with 5.5 mL of simulated intestinal fluid (SIF), 2.5 mL of pancreatin (800 U/mL), 1.25 mL of fresh bile (160 mM), and 20 μL of 0.3 M CaCl2. The pH was adjusted to 7.0 using 1 M NaOH, followed by incubation at 37 °C for 2 h.
Viability was assessed on the 1st and 120th days of storage for samples kept at 25 °C to determine the protective effect of encapsulation over time.
2.10. Analysis of Fatty Acids in Samples Using GC-FID
Prior to analysis, fatty acid methyl esters (FAMEs) were prepared using a base-catalyzed transesterification procedure. Briefly, 0.1 g of the extracted lipid phase was dissolved in 2 mL of n-heptane, followed by the addition of 0.2 mL of 2 N methanolic potassium hydroxide (KOH). The mixture was vortexed for 30 s and allowed to settle at room temperature for 5 min. The upper heptane layer containing the FAMEs was then collected and filtered through a 0.45 µm membrane filter. Fatty acid composition was analyzed using an Agilent 7890B Gas Chromatography system equipped with a Flame Ionization Detector (GC-FID) (Agilent Technologies, Santa Clara, CA, USA). Separation was achieved on a Wax fame column (30 m × 0.25 mm × 0.25 µm). The injector was operated in split mode (1:50) at 250 °C with Hydrogen as the carrier gas (1 mL/min). The oven temperature program was as follows: initial temperature of 120 °C, increased to 150 °C at 5 °C/min (held for 7.5 min), then increased to 250 °C at 7 °C/min (held for 7.21 min) [36]. Fatty acid profiles were compared between encapsulated and control samples at Day 0 and Day 120 of storage at 25 °C.
2.11. Statistical Analysis
All experiments were performed in duplicate with at least two parallel measurements per replicate. Data are presented as mean values ± standard deviation. Statistical significance was evaluated using one-way Analysis of Variance (ANOVA) followed by Student’s t-test where applicable. Differences were considered statistically significant at p ≤ 0.05. Statistical analyses were performed using SPSS Statistics software (v.19.0; IBM Corp., Armonk, NY, USA).
3. Results and Discussion
3.1. Encapsulation Efficiency (EE) of S. boulardii
The encapsulation of S. boulardii using a cocoa powder, sodium alginate, and FOS (10:1:1) matrix yielded a high encapsulation efficiency (EE) of 93.57 ± 1.02%. This result indicates that the chosen biopolymer blend and the freeze-drying process effectively protected the yeast cells during the immobilization process. According to Xu et al. [37], an EE exceeding 85% is considered successful for probiotic delivery systems, as it ensures a sufficient microbial load for therapeutic effects. The high encapsulation efficiency (93.57%) can be attributed to the strong electrostatic interactions between the carboxyl groups of sodium alginate and the probiotic cell wall components.
The EE reported in this study is notably higher than the 85.49% reported by Yasmin et al. [38], who utilized whey protein concentrate and pectin. This superiority can be attributed to the synergistic effect of the matrix components: sodium alginate provides a robust gel network, while the dietary fibers in cocoa powder and the prebiotic nature of FOS (fructooligosaccharides) likely acted as filler agents and cryoprotectants, reinforcing the structural integrity of the capsules during the sublimation phase of freeze-drying.
In conclusion, the combination of cocoa powder, sodium alginate, and FOS appears to be a highly effective matrix for encapsulating S. boulardii, offering enhanced protection and potential for integration into functional food supplements.
3.2. Moisture Determination
The moisture content and hardness values of the dark chocolate samples during the 120-day storage period are presented in Table 1. Statistical analysis indicated that there was no significant change (p ≥ 0.05) in either parameter for both encapsulated and non-encapsulated samples when stored at 25 °C.

Table 1.
Moisture Content (%) and Hardness (N) of Chocolate Samples on Day 0 and Day 120.
In traditional dark chocolates, highly hygroscopic components like sucrose can lead to the formation of “sandy” lumps and increased moisture on particle surfaces, which subsequently raises friction and viscosity [39]. In the current sugar-free formulation, the moisture content remained stable, which is crucial for preventing alterations in the chocolate’s microscopic structure [40]. This stability is also a key factor in inhibiting the migration of moisture that typically contributes to the formation of bloom [41], thereby maintaining both textural and storage stability.
The literature suggests that while sucrose-containing chocolates possess a complex structural matrix, the type of sucrose substitute used—in this case, inulin and stevioside—significantly influences the final hardness through particle-particle interactions [42,43,44]. The molecular structure and particle size of these substitutes are determinant factors; for instance, smaller crystal structures typically increase hardness. However, Aidoo et al. [45] observed that large inulin crystals could limit particle interactions, leading to a decrease in hardness. In our study, the consistent hardness values throughout storage suggest that the inulin-based matrix provided a stable network that resisted the softening effects often associated with the hygroscopic nature of volume-increasing agents in sugar-free formulations.
3.3. Bloom Formation
Bloom formation, characterized by the development of a white or grayish film on the chocolate surface, negatively impacts visual quality and consumer perception. In this study, no bloom was observed in any of the chocolate samples stored at 25 °C throughout the 120-day storage period, as confirmed by stereomicroscopic examination.
These results are consistent with the findings of Rodriguez Furlan et al., [40], who suggested that chocolates formulated with sweeteners possessing higher melting enthalpies are less prone to surface bloom development. The stability of the cocoa butter crystal network, combined with the absence of significant moisture migration (as discussed in Section 3.3), likely prevented the recrystallization processes that lead to both fat and sugar bloom.
3.4. Measurement of pH and Water Activity (aw)
The changes in pH and water activity (aw) of the probiotic chocolates during storage at 25 °C are summarized in Table 2. A gradual and significant increase in pH values was observed for both encapsulated and non-encapsulated groups over the 120-day period (p < 0.05). Conversely, aw values exhibited a significant decline during the same period (p < 0.05).
The results suggest that the increase in pH during storage was not directly related to the metabolic activity of the added S. boulardii. This is supported by similar findings in the literature; for instance, a study on probiotic-enriched chocolates concluded that pH elevations were independent of the probiotic presence [29]. Such changes may instead be attributed to lipolytic activity or fat oxidation occurring within the chocolate matrix during storage. Kobus-Cisowska et al. [33] also reported a rise in pH for chocolates containing Bacillus coagulans, linking it to the inherent chemical dynamics of the dark chocolate components.
Regarding water activity, the observed decrease may be linked to moisture migration or potential micro-cracking of the outer layers during storage, which facilitates water loss [33]. The observed decrease in water activity, despite constant moisture content, suggests a higher degree of water binding within the inulin-cocoa matrix, which may further contribute to the inhibition of microbial spoilage and structural stability. Furthermore, while a high cocoa mass can facilitate water binding and potentially increase aw initially [29], long-term storage of high-fat dark chocolate often involves complex oxidative processes and moisture equilibration that can result in the recorded aw reduction. The low water activity (aw < 0.4) of our dark chocolate was a critical factor in maintaining the metabolic dormancy of S. boulardii, thereby extending its shelf-life without significant loss of viability.

Table 2.
pH and Water Activity (aw) Values of Probiotic Chocolates During Storage at 25 °C.
Table 2.
pH and Water Activity (aw) Values of Probiotic Chocolates During Storage at 25 °C.
| pH | |||||
|---|---|---|---|---|---|
| Day 0 | Day 30 | Day 60 | Day 90 | Day 120 | |
| Encapsulated Chocolate | 5.77 ± 0.17 Da | 5.74 ± 0.10 Da | 6.28 ± 0.00 Ba | 6.37 ± 0.02 Aa | 6.08 ± 0.13 Ca |
| Non-encapsulated Chocolate | 5.25 ± 0.07 Cb | 5.30 ± 0.00 CBb | 5.73 ± 0.20 ABb | 5.73 ± 0.15 ABb | 5.87 ± 0.06 Ab |
| aw | |||||
| Encapsulated Chocolate | 0.387 ± 0.06 Aa | 0.373 ± 0.20 ABa | 0.373 ± 0.20 ABa | 0.330 ± 0.00 BCa | 0.325 ± 0.07 Ca |
| Non-encapsulated Chocolate | 0.367 ± 0.14 Aa | 0.367 ± 0.14 Aa | 0.340 ± 0.00 ABb | 0.330 ± 0.00 BCa | 0.304 ± 0.03 Cb |
a, b: Different superscript letters indicate significant differences between samples (p < 0.05). A, B, C, D: Different superscript letters indicate significant differences among rows (p < 0.05).
3.5. Examination of Encapsulated Probiotics Before and After Mixing with Chocolate Using Scanning Electron Microscopy
Scanning electron microscopy (SEM) was employed to visualize the structural integration and morphological integrity of S. boulardii within the different matrices. The SEM images revealed the distinct characteristics of the encapsulated probiotic cells alone (Figure 1), the encapsulated cells incorporated within the chocolate matrix (Figure 2), and the unencapsulated (free) cells within the chocolate samples (Figure 3).

Figure 1.
SEM Images of Encapsulated S. boulardii.

Figure 2.
SEM Images of Encapsulated S. boulardii in Chocolate Samples.

Figure 3.
SEM Images of Non-encapsulated S. boulardii in Chocolate Samples.
The analysis confirmed that the encapsulated yeast was effectively trapped and protected within the chocolate network. The spherical yeast cells exhibited a homogeneous distribution, and their characteristic cellular morphology remained consistent across all samples. Notably, the SEM images at consistent magnification levels indicate that the size and shape of the S. boulardii cells were not adversely affected by the freeze-drying process or the subsequent mixing with the chocolate ingredients.
This observation aligns with previous findings by de Araujo et al. [25], suggesting that the chosen encapsulating materials provide a stable microenvironment. Furthermore, a comparison between Figure 2 and Figure 3 clearly illustrates the protective role of the encapsulation matrix; while encapsulated cells were shielded by the biopolymer-cocoa layer, free cells (Figure 3) showed direct exposure to the chocolate components. These results further validate the suitability of cocoa powder as a functional encapsulating agent; its particulate nature likely provides an additional physical barrier that enhances the protective properties of the carrier system, ensuring the probiotics remain structurally intact within the final product.
3.6. Probiotic Viability in Chocolates
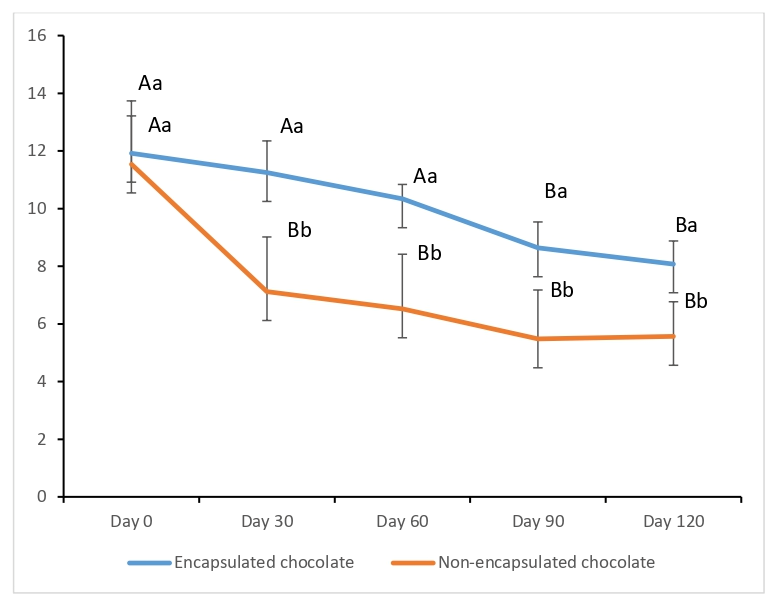
The viability of encapsulated and free S. boulardii within the chocolate samples was monitored monthly over a 120-day storage period at 25 °C (Figure 4). On the initial day of storage (Day 0), all chocolate formulations exhibited high and comparable probiotic loads (>log CFU/g), indicating that the manufacturing process and ingredients did not cause immediate lethal stress to the yeast cells.
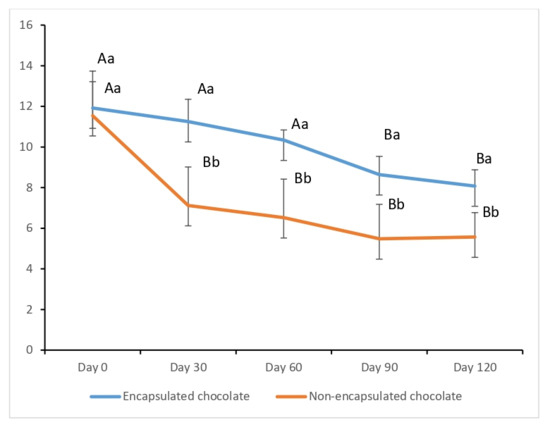
Figure 4.
Yeast Counts of Probiotic Chocolates During Storage at 25 °C. a, b: Different superscript letters indicate significant differences between samples (p < 0.05). A, B: Different superscript letters indicate significant differences among rows (p < 0.05).
However, a significant divergence in stability emerged during the storage period. In chocolates enriched with encapsulated probiotics, the counts remained remarkably stable, staying above 8.08 log CFU/g throughout the 120 days. In stark contrast, the non-encapsulated (free) samples experienced a rapid and drastic 4 log reduction within the first 30 days. This accelerated decline in free yeast cells suggests that without protection, they were highly susceptible to the intrinsic chemical environment of the chocolate matrix and oxidative stress. By the end of 120 days, the encapsulated samples maintained a count of 2.51 log units higher than the non-encapsulated ones, clearly demonstrating the robust protective efficacy of the cocoa–alginate–FOS encapsulating matrix.
According to international standards, probiotic-enriched foods must maintain a minimum therapeutic value of 107 CFU/g at the time of consumption to confer health benefits [28]. Our results show that while the encapsulated chocolate remained well above this threshold throughout the storage, reaching 8.08 log CFU/g at the end of 120 days, the non-encapsulated version lost its therapeutic functionality significantly earlier, dropping to 5.57 log CFU/g by the end of the study.
The importance of the matrix composition is further supported by the literature. Pandiyan et al. [46] utilized a combination of L. acidophilus, S. boulardii, and fructooligosaccharides (FOS) in whey protein-based ice cream. While they also noted that the presence of FOS significantly enhanced viability (p ≤ 0.01) by providing a favorable microenvironment, they recorded a steady decrease in counts during storage. In our study, the integration of cocoa powder into the encapsulating matrix, alongside sodium alginate and FOS, appeared to provide a superior barrier. This synergy not only protected the probiotics during storage but, as suggested by the SEM results, created a dense structural shield that potentially enhances the preservation of S. boulardii in functional food applications.
3.7. Testing the Survival of Probiotics During In Vitro Gastrointestinal Digestion of Probiotic Chocolate
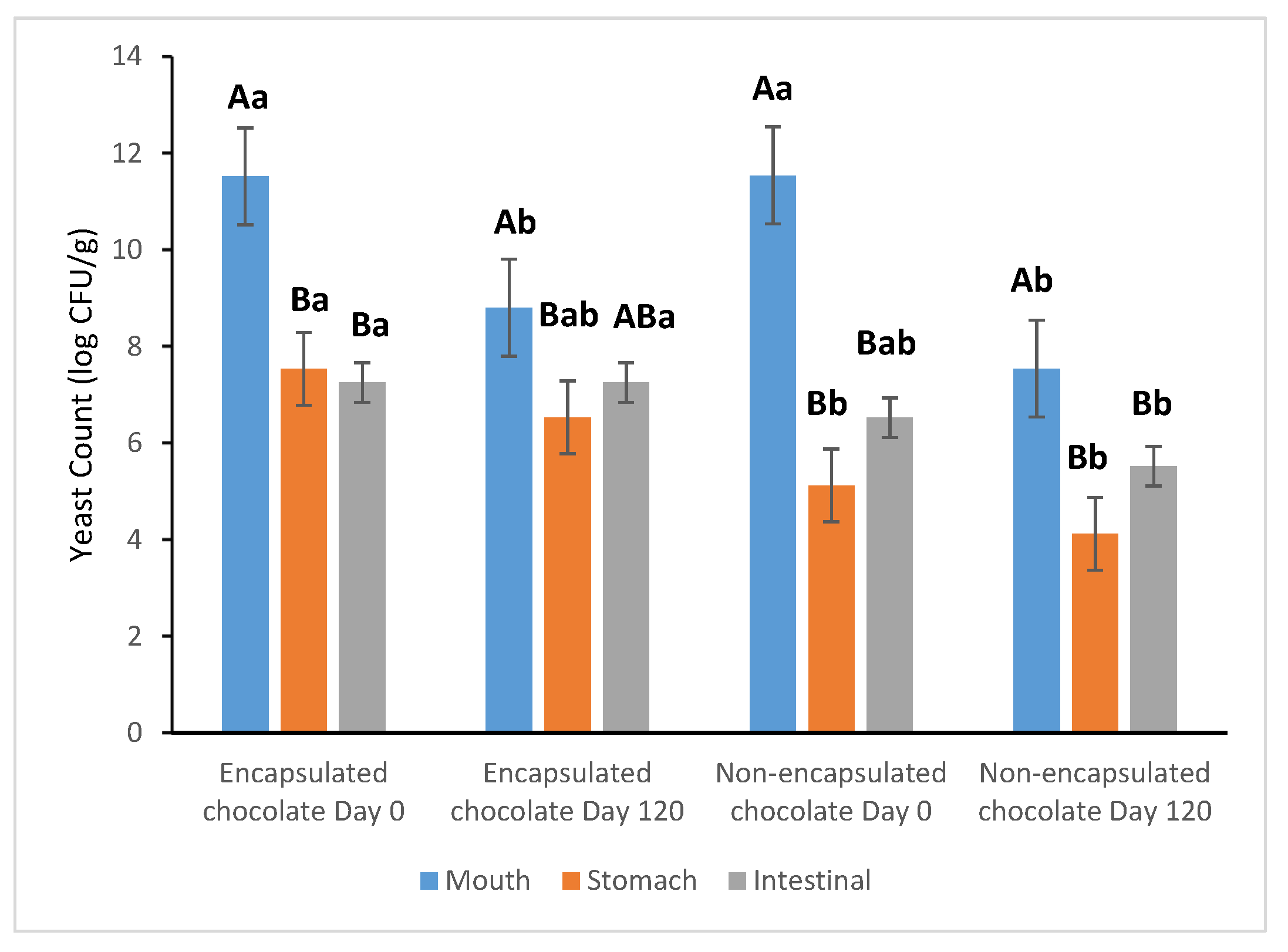
The survival of S. boulardii during simulated gastrointestinal transit was assessed on the 1st and 120th days of storage at 25 °C (Figure 5). The in vitro digestion assay revealed a significant disparity in the resilience of encapsulated versus non-encapsulated (free) yeast cells when exposed to harsh gastric conditions.
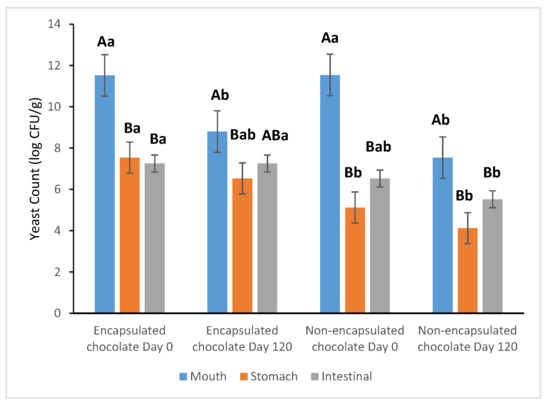
Figure 5.
The survival rate of S. boulardii in chocolate was subjected to in vitro digestion after storage at 25 °C for 1 day and 120 days. a, b: Different superscript letters indicate significant differences between samples (p < 0.05). A, B: Different superscript letters indicate significant differences among rows (p < 0.05).
During the analysis conducted on the first day of storage, the non-encapsulated probiotics suffered a drastic and significant (p < 0.05) reduction of 6.42 ± 0.0 log after 2 h of exposure to simulated gastric fluid (pH 3.0). In contrast, the encapsulated probiotics exhibited significantly higher resilience, with a notably lower reduction of 3.99 ± 0.2 log. This protective effect underscores that the cocoa–alginate–FOS encapsulation matrix, combined with the lipid-rich chocolate carrier, effectively buffers the yeast cells against the detrimental effects of low pH and enzymatic activity.
Upon transition to simulated intestinal fluid (pH 7.0), which provides a more favorable environment for probiotic growth, the encapsulated samples maintained their viability more effectively than the free cells. These results demonstrate that the encapsulated S. boulardii formulation is capable of maintaining the minimum therapeutic levels required for health benefits, even after 120 days of storage.
Furthermore, the complex nutritional profile of the chocolate matrix—including carbohydrates and lipids—acts as a secondary protective layer and a nutrient source, facilitating the survival and potential colonization of probiotics within the gastrointestinal tract. This synergistic protection ensures that a sufficient microbial load reaches the intestines, confirming the viability of probiotic-enriched dark chocolate as a functional delivery system.
3.8. Fatty Acid Composition
The fatty acid profiles of the probiotic dark chocolate samples were analyzed to evaluate chemical stability over the 120-day storage period (Table 3). In the encapsulated probiotic chocolates, the predominant fatty acids—Caproic (C6:0), Palmitic (C16:0), Stearic (C18:0), Oleic (C18:1n9c), Linoleic (C18:2n6c), Arachidic (C20:0), and Nervonic acid (C24:1n9)—showed no significant fluctuations (p > 0.05). Notably, Lauric acid (C12:0) was detected at a rate of 1.35% only on the 120th day, suggesting minor late-stage lipid dynamics.

Table 3.
Fatty acid values (%) of encapsulated and non-encapsulated chocolates on days 0 and 120.
In the non-encapsulated samples, the core fatty acid profile remained similarly stable, with Behenic acid (C22:0) appearing at 0.95% by day 120. The primary saturated fatty acids, C16:0 (25.31%) and C18:0 (33.68%), aligned closely with the findings of Melo et al. [47], who identified these as the major constituents in functional chocolates. These results are also consistent with the standard composition of cocoa butter, where Palmitic, Stearic, Oleic, and Linoleic acids typically constitute over 98% of the total lipid fraction [48].
Beyond chemical stability, the fatty acid profile plays a decisive role in the physical quality of the final product. Inter-particle interactions and the formation of crystalline network structures dictate the compressibility and fracture properties of chocolate, which are perceived as texture [49]. The concentration and chain length of the fatty acids used in the formulation directly influence these particle interactions; for instance, the high proportion of stearic and palmitic acids in our samples likely contributed to the stable hardness values observed in Section 3.3 by facilitating a robust fat crystal network.
In food processing technology, the quality and type of fats (derived from cocoa butter, milk powder, or vegetable oils) are critical, as they impact both the sensory attributes and the nutritional profile of the chocolate. The stability of the fatty acid composition in this study confirms that the incorporation of encapsulated S. boulardii does not compromise the structural integrity or the health-promoting lipid profile of the dark chocolate, ensuring a high-quality functional product for the consumer. The detection of trace amounts of Lauric and Behenic acids at day 120 did not lead to any observable rancidity or off-flavors, suggesting that lipid oxidation remained within acceptable limits for premium dark chocolate.
4. Conclusions
The significantly lower log reduction observed in encapsulated samples highlights the dual protective role of the cocoa–alginate–FOS matrix and the chocolate lipid phase during in vitro digestion. While the harsh acidic conditions of the stomach (pH 3.0) typically compromise microbial integrity, the subsequent transition to the intestinal environment (pH 7.0) demonstrated that the encapsulated probiotics could effectively recover and maintain counts above the minimum therapeutic threshold.
These findings suggest that the complex chemical composition of dark chocolate—comprising essential fatty acids, polyphenols, and dietary fibers—not only acts as a physical shield but also serves as a supportive nutrient source for S. boulardii within the gastrointestinal tract.
Based on the comprehensive findings of this study, dark chocolate enriched with encapsulated S. boulardii represents a highly stable and effective functional snack. The synergistic combination of cocoa powder, sodium alginate, and FOS as an encapsulating agent successfully preserved the yeast’s viability, structural integrity, and therapeutic potential throughout a 120-day storage period and simulated digestion.
Crucially, the results indicate that this probiotic chocolate can be safely stored at room temperature without compromising its functional properties, offering a significant advantage for logistics and consumer accessibility. In conclusion, this formulation serves as both a nutritional delivery system and a potential prebiotic source, marking a significant advancement in the development of non-dairy probiotic functional foods.
Author Contributions
Conceptualization, S.Ö.Y. and E.K.K.; methodology, E.K.K.; writing—original draft preparation, E.K.K.; writing—review and editing, E.K.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Scientific and Technological Research Council of Turkey (TÜBİTAK, grant number 123O288) and Sakarya University Scientific Research Projects Unit (Project number: 2019-7-25-290).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hossain, M.N.; Ranadheera, C.S.; Fang, Z.; Ajlouni, S. Healthy chocolate enriched with probiotics: A review. Food Sci. Technol. 2021, 41, 531–543. [Google Scholar] [CrossRef]
- Wasilewski, A.; Zielińska, M.; Storr, M.; Fichna, J. Beneficial effects of probiotics, prebiotics, synbiotics, and psychobiotics in inflammatory bowel disease. Inflamm. Bowel Dis. 2015, 21, 1674–1682. [Google Scholar] [CrossRef]
- Probiotics Market Outlook. Development Trends, Market Demands, İndustry Analysis & Forecast by 2024; Probiotics Market Outlook: Maharashtra, India, 2018. [Google Scholar]
- Frakolaki, G.; Giannou, V.; Kekos, D.; Tzia, C. A review of the microencapsulation techniques for the incorporation of probiotic bacteria in functional foods. Crit. Rev. Food Sci. Nutr. 2021, 61, 1515–1536. [Google Scholar]
- Granato, D.; Barba, F.J.; Kovačević, D.B.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional foods: Product development, technological trends, efficacy testing, and safety. Annu. Rev. Food Sci. Technol. 2020, 11, 93–118. [Google Scholar] [CrossRef]
- Mani-López, E.; Palou, E.; López-Malo, A. Probiotic viability and storage stability of yogurts and fermented milks prepared with several mixtures of lactic acid bacteria. J. Dairy Sci. 2014, 97, 2578–2590. [Google Scholar] [CrossRef] [PubMed]
- Monteagudo-Mera, A.; Rastall, R.A.; Gibson, G.R.; Charalampopoulos, D.; Chatzifragkou, A. Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Appl. Microbiol. Biotechnol. 2019, 103, 6463–6472. [Google Scholar] [CrossRef] [PubMed]
- de Souza Simões, L.; Madalena, D.A.; Pinheiro, A.C.; Teixeira, J.A.; Vicente, A.A.; Ramos, Ó.L. Micro-and nano bio-based delivery systems for food applications: In vitro behavior. Adv. Colloid Interface Sci. 2017, 243, 23–45. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, F.; Sooklim, C.; Songdech, P.; Duangpakdee, O.; Soontorngun, N. Selection of potential yeast probiotics and a cell factory for xylitol or acid production from honeybee samples. Metabolites 2021, 11, 312. [Google Scholar] [CrossRef]
- Staniszewski, A.; Kordowska-Wiater, M. Probiotic and potentially probiotic yeasts—Characteristics and food application. Foods 2021, 10, 1306. [Google Scholar] [CrossRef]
- Van de Walle, D.; Heymans, R.; Dewettinck, K. Linking the physicochemical properties of bulking agents to the sensory characteristics of fat-based suspensions. J. Food Eng. 2018, 234, 82–89. [Google Scholar] [CrossRef]
- Aidoo, R.P.; Depypere, F.; Afoakwa, E.O.; Dewettinck, K. Industrial manufacture of sugar-free chocolates—Applicability of alternative sweeteners and carbohydrate polymers as raw materials in product development. Trends Food Sci. Technol. 2013, 32, 84–96. [Google Scholar] [CrossRef]
- Meyer, D.; Bayarri, S.; Tarrega, A.; Costell, E. Inulin as texture modifier in dairy products. Food Hydrocoll. 2011, 25, 1881–1890. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Introducing inulin-type fructans. Br. J. Nutr. 2005, 93, S13–S25. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Rashid, S. Functional and therapeutic potential of inulin: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1–13. [Google Scholar] [CrossRef]
- ICCO, International Cocoa Organization. Supply-Demand QBCS XLVII, No. 1 (Online); ICCO: Abidjan, Côte d’Ivoire, 2021. [Google Scholar]
- Min, M.; Bunt, C.R.; Mason, S.L.; Hussain, M.A. Non-dairy probiotic food products: An emerging group of functional foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 2626–2641. [Google Scholar] [CrossRef]
- Silva, M.P.; Tulini, F.L.; Marinho, J.F.; Mazzocato, M.C.; De Martinis, E.C.; Luccas, V.; Favaro-Trindade, C. Semisweet chocolate as a vehicle for the probiotics Lactobacillus acidophilus LA3 and Bifidobacterium animalis subsp. lactis BLC1: Evaluation of chocolate stability and probiotic survival under in vitro simulated gastrointestinal conditions. LWT—Food Sci. Technol. 2017, 75, 640–647. [Google Scholar] [CrossRef]
- Afzaal, M.; Saeed, F.; Arshad, M.U.; Nadeem, M.T.; Saeed, M.; Tufail, T. The effect of encapsulation on the stability of probiotic bacteria in ice cream and simulated gastrointestinal conditions. Probiot. Antimicrob. Proteins 2019, 11, 1348–1354. [Google Scholar] [CrossRef]
- Oracz, J.; Nebesny, E.; Zyzelewicz, D.; Budryn, G.; Luzak, B. Bioavailability and metabolism of selected cocoa bioactive compounds: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2020, 60, 1947–1985. [Google Scholar] [CrossRef]
- Chavez, B.; Ledeboer, A. Drying of probiotics: Optimization of formulation and process to enhance storage survival. Dry. Technol. 2007, 25, 1193–1201. [Google Scholar] [CrossRef]
- Kanak, E.K.; Yılmaz, S.Ö. Determination of the Probiotic and Functional Properties of Yeasts Isolated from Different Dairy Products. Fermentation 2025, 11, 104. [Google Scholar] [CrossRef]
- Hossain, M.N.; Ranadheera, C.S.; Fang, Z.; Hutchinson, G.; Ajlouni, S. Protecting the viability of encapsulated Lactobacillus rhamnosus LGG using chocolate as a carrier. Emir. J. Food Agric. 2021, 33, 647–656. [Google Scholar] [CrossRef]
- Gebara, C.; Chaves, K.S.; Ribeiro, M.C.E.; Souza, F.N.; Grosso, C.R.; Gigante, M.L. Viability of Lactobacillus acidophilus La5 in pectin–whey protein microparticles during exposure to simulated gastrointestinal conditions. Food Res. Int. 2013, 51, 872–878. [Google Scholar] [CrossRef]
- de Araújo Etchepare, M.; Nunes, G.L.; Nicoloso, B.R.; Barin, J.S.; Moraes Flores, E.M.; de Oliveira Mello, R.; Ragagnin de Menezes, C. Improvement of the viability of encapsulated probiotics using whey proteins. LWT—Food Sci. Technol. 2020, 117, 108601. [Google Scholar] [CrossRef]
- Lim, P.Y.; Wong, K.Y.; Thoo, Y.Y.; Siow, L.F. Effect of inulin, fructo-oligosaccharide, trehalose or maltodextrin (M10 and M30) on the physicochemical and sensory properties of dark compound chocolate. LWT—Food Sci. Technol. 2021, 149, 111964. [Google Scholar] [CrossRef]
- Hinneh, M.; Van de Walle, D.; Haeck, J.; Abotsi, E.E.; De Winne, A.; Saputro, A.D.; Messens, K.; Van Durme, J.; Afoakwa, E.O.; De Cooman, L.; et al. Applicability of the melanger for chocolate refining and Stephan mixer for conching as small-scale alternative chocolate production techniques. J. Food Eng. 2019, 253, 59–71. [Google Scholar] [CrossRef]
- Dong, Q.Y.; Chen, M.Y.; Xin, Y.; Qin, X.Y.; Cheng, Z.; Shi, L.E.; Tang, Z.X. Alginate-based and protein-based materials for probiotics encapsulation: A review. Int. J. Food Sci. Technol. 2013, 48, 1339–1351. [Google Scholar] [CrossRef]
- Hossain, M.N.; Ranadheera, C.S.; Fang, Z.; Masum, A.K.M.; Ajlouni, S. Viability of Lactobacillus delbrueckii in chocolates during storage and in-vitro bioaccessibility of polyphenols and SCFAs. Curr. Res. Food Sci. 2022, 5, 1266–1275. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis of AOAC International; AOAC International: Arlington, VA, USA, 2000. [Google Scholar]
- Biswas, N.; Cheow, Y.L.; Tan, C.P.; Siow, L.F. Physical, rheological and sensorial properties, and bloom formation of dark chocolate made with cocoa butter substitute (CBS). LWT—Food Sci. Technol. 2017, 82, 420–428. [Google Scholar] [CrossRef]
- Kinta, Y.; Hartel, R.W. Bloom formation on poorly-tempered chocolate and effects of seed addition. J. Am. Oil Chem. Soc. 2010, 87, 19–27. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Szymanowska, D.; Maciejewska, P.; Szczepaniak, O.; Kmiecik, D.; Gramza-Michałowska, A.; Kulczyński, B.; Cielecka-Piontek, J. Enriching novel dark chocolate with Bacillus coagulans as a way to provide beneficial nutrients. Food Funct. 2019, 10, 997–1006. [Google Scholar] [CrossRef]
- Masum, A.; Chandrapala, J.; Adhikari, B.; Huppertz, T.; Zisu, B. Effect of lactose-to-maltodextrin ratio on emulsion stability and physicochemical properties of spray-dried infant milk formula powders. J. Food Eng. 2019, 254, 34–41. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carriere, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, T.; Sugitate, K.; Nakai, T.; Jikumaru, Y.; Ishihara, G. Rapid profiling method for mammalian feces short chain fatty acids by GC-MS. Anal. Biochem. 2018, 543, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Gagné-Bourque, F.; Dumont, M.J.; Jabaji, S. Encapsulation of Lactobacillus casei ATCC 393 cells and evaluation of their survival after freeze-drying, storage and under gastrointestinal conditions. J. Food Eng. 2016, 168, 52–59. [Google Scholar] [CrossRef]
- Yasmin, I.; Saeed, M.; Pasha, I.; Zia, M.A. Development of whey protein concentrate-pectin-alginate based delivery system to improve survival of B. longum BL-05 in simulated gastrointestinal conditions. Probiot. Antimicrob. Proteins 2019, 11, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Afoakwa, E.O.; Paterson, A.; Fowler, M. Factors influencing rheological and textural qualities in chocolate—A review. Trends Food Sci. Technol. 2007, 18, 290–298. [Google Scholar] [CrossRef]
- Rodriguez Furlan, L.T.; Baracco, Y.; Lecot, J.; Zaritzky, N.; Campderrós, M.E. Effect of sweetener combination and storage temperature on physicochemical properties of sucrose free white chocolate. Food Chem. 2017, 229, 610–620. [Google Scholar] [CrossRef]
- Stortz, T.A.; Marangoni, A.G. Heat resistant chocolate. Trends Food Sci. Technol. 2011, 22, 201–214. [Google Scholar] [CrossRef]
- Farzanmehr, H.; Abbasi, S. Effects of inulin and bulking agents on some physicochemical, textural and sensory properties of milk chocolate. J. Texture Stud. 2009, 40, 536–553. [Google Scholar] [CrossRef]
- Hadnađev, M.; Dokic, L.; Hadnađev, T.D.; Pajin, B.; Krstonosic, V. The impact of maltodextrin-based fat mimetics on rheological and textural characteristics of edible vegetable fat. J. Texture Stud. 2011, 42, 404–411. [Google Scholar] [CrossRef]
- Shah, A.B.; Jones, G.P.; Vasiljevic, T. Sucrose-free chocolate sweetened with Stevia rebaudiana extract and containing different bulking agents—Effects on physicochemical and sensory properties. Int. J. Food Sci. Technol. 2010, 45, 1426–1435. [Google Scholar] [CrossRef]
- Aidoo, R.P.; Afoakwa, E.O.; Dewettinck, K. Optimization of inulin and polydextrose mixtures as sucrose replacers during sugar-free chocolate manufacture—Rheological, microstructure and physical quality characteristics. J. Food Eng. 2014, 126, 35–42. [Google Scholar] [CrossRef]
- Pandiyan, C.; Annal Villi, R.; Kumaresan, G.; Murugan, B.; Gopalakrishnamurthy, T.R. Development of synbiotic ice cream incorporating Lactobacillus acidophilus and Saccharomyces boulardii. Int. Food Res. J. 2012, 19, 1177–1181. [Google Scholar]
- Melo, C.W.B.D.; Bandeira, M.D.J.; Maciel, L.F.; Bispo, E.D.S.; Souza, C.O.D.; Soares, S.E. Chemical composition and fatty acids profile of chocolates produced with different cocoa (Theobroma cacao L.) cultivars. Food Sci. Technol. 2020, 40, 326–333. [Google Scholar] [CrossRef]
- Shekarchizadeh, H.; Kadivar, M.; Ghaziaskar, H.S.; Rezayat, M. Optimization of enzymatic synthesis of cocoa butter analog from camel hump fat in supercritical carbon dioxide by response surface method (RSM). J. Supercrit. Fluids 2009, 49, 209–215. [Google Scholar] [CrossRef]
- Do, T.A.; Hargreaves, J.M.; Wolf, B.; Hort, J.; Mitchell, J.R. Impact of particle size distribution on rheological and textural properties of chocolate models with reduced fat content. J. Food Sci. 2007, 72, E541–E552. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.