Abstract
Integrating environmental sustainability into chemical sensor research is no longer optional and must be addressed at the laboratory scale, where material selection, fabrication strategies, and end-of-life management are defined. Although chemical sensors benefit from miniaturization and disposable architectures, their environmental footprint extends beyond the device geometry to include the electrode substrates, functional coatings and auxiliary materials. In this context, sensors based on molecularly imprinted polymers (MIPs), which are entirely synthetic and artificially engineered materials, pose specific sustainability challenges related to material choice, processing, regeneration and disposal. Addressing these aspects in a systematic and quantitative manner is therefore essential to aligning high analytical performance with sustainable sensor design. This review surveys and critically discusses the strategies currently adopted to improve the environmental sustainability of MIP-based sensors, covering key stages of the MIP sensor lifecycle, including monomer and crosslinker selection, fabrication routes, operational aspects, and end-of-life management. Representative approaches such as the use of bioderived polymerization components, low-impact solvents, cleaner analyte removal methods, and low-energy polymerization techniques are analyzed, highlighting their advantages, limitations, and cost-related trade-offs. To move beyond the qualitative assessment of greenness, sustainability is addressed through Lifecycle Assessment (LCA) and AGREE-based metrics, highlighting the importance of functional units, use phase inventories, and regeneration strategies in reducing overall environmental impacts. The review concludes by proposing actionable guidelines to support the transition of MIP-based sensors from sustainable laboratory fabrication to real-world environmental monitoring applications.
1. Introduction
Environmental sustainability is one of the most pressing challenges in contemporary research. In this context, Green Analytical Chemistry (GAC) has emerged as a guiding paradigm, setting out clear principles and priorities for the development of analytical methods and devices, spanning laboratory-scale material synthesis to portable, application-oriented analytical systems.
Sensors represent an effective link between material-oriented synthesis and real-world applications. Molecularly imprinted polymers (MIPs) are particularly attractive recognition elements, as tailored binding sites can markedly improve selectivity and sensitivity across a wide range of targets [1,2]. However, the sustainability of MIP-based sensors is strongly influenced by fabrication choices, including monomers/crosslinkers, solvents, polymerization conditions, and template removal, which may involve hazardous solvents, energy-demanding steps, and additional chemical waste. Several approaches exist to appraise the sustainability of analytical methods, including green metrics as well as whiteness and blueness models [3]; however their comparative and lifecycle-oriented application to MIP-based sensors remains limited [4].
This review addresses this gap by critically analyzing green strategies for MIP-based sensors across the entire sensor lifecycle and comparing the main quantitative decision-making tools used for sustainability assessment. The applicability, strengths, and limitations of GAC metrics and LCA approaches are discussed, with a focus on AGREE-based tools (including AGREE, AGREEprep, and the MIP-specific AGREEMIP) and the applicability-oriented BAGI. The literature was selected through a targeted screening, prioritizing recent studies on MIP sensors that explicitly, or implicitly, addressed sustainability aspects and/or quantitative assessment frameworks. These perspectives are innovatively integrated into the present review, which aims to critically discuss green strategies across the entire MIP sensor lifecycle and to compare the most common quantitative decision-making tools used in the ever-growing research field of MIPs. This combination of indices and perspectives enables the identification of sustainable trade-offs, the definition of new sustainable standards, and the limitations of systems.
2. Principles of “Green Analytical Chemistry” Applied to MIPs
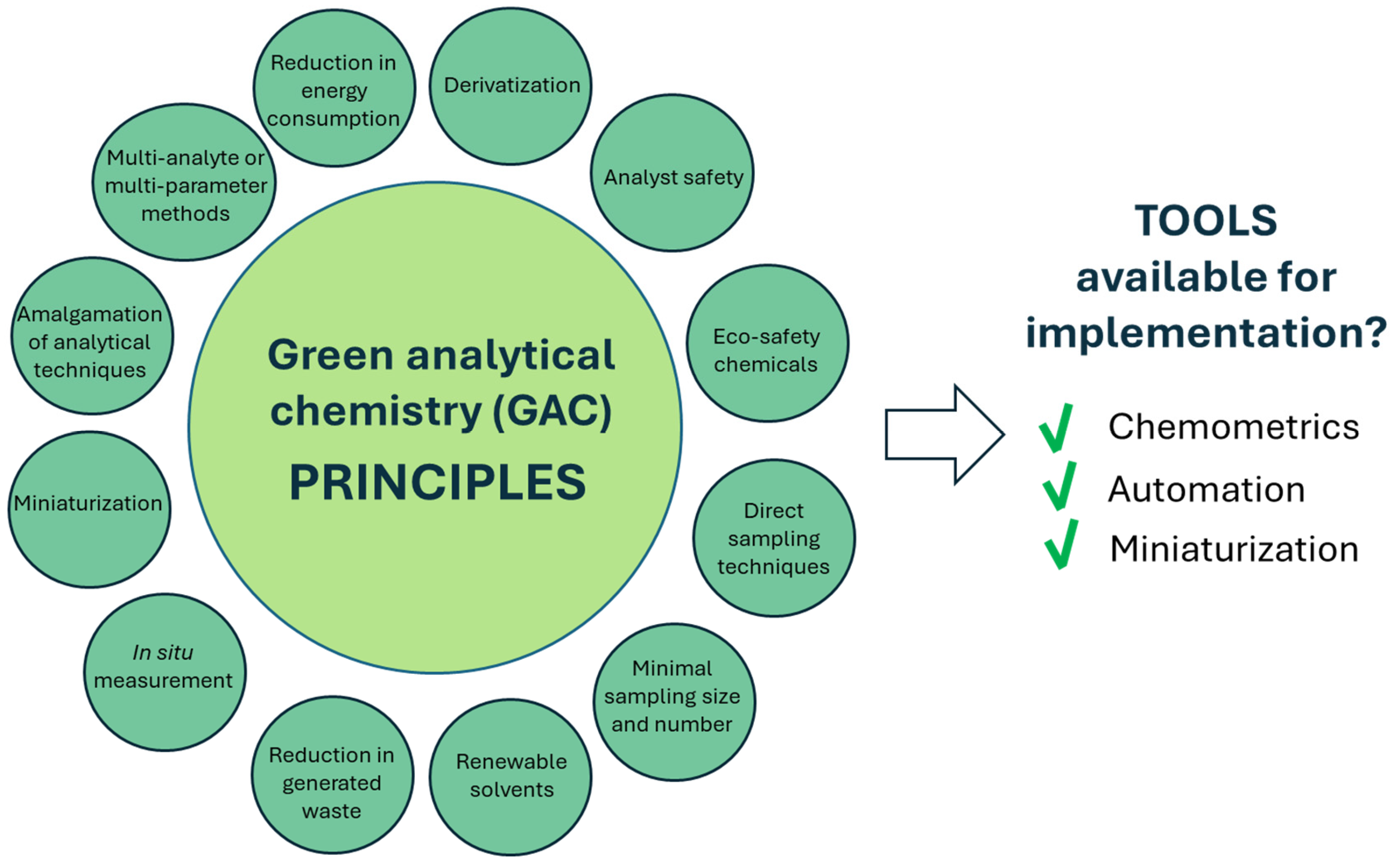
GAC was born from green chemistry in 2000 [5] as a search for practical alternatives to the off-line treatment of waste and residues, in order to replace polluting methodologies with clean ones [6]. The origins of the green branch of chemistry date back to 1987, when in Paris, during Euroanalysis VI, Professor H. Malissa discussed his ideas on changes in the paradigms of analytical chemistry [7], in accordance with the conclusions of the Pimentel report [8], on the impact of chemistry on the health of the Earth. Later, the Royal Society of the United Kingdom proposed the theme of environmental analytical chemistry as a model of analytical practices in an integrated approach to analytical chemistry, also considering environmental side effects. Soon after, green chemistry (GC) was defined as “the use of chemical techniques and methodologies that reduce or eliminate the use or generation of raw materials, products, by-products, solvents, reagents, etc. that are hazardous to human health or the environment” [9]. The main vision of GAC is depicted in Figure 1.
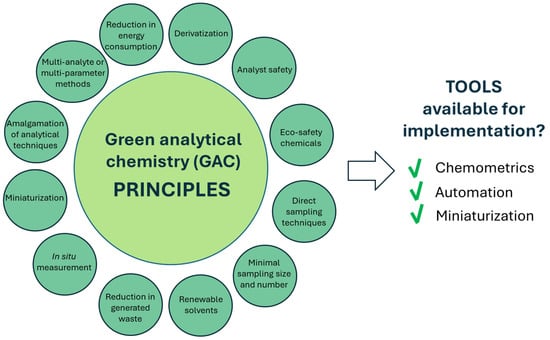
Figure 1.
Overview of GAC and tools available for its implementation. In particular, chemometric approaches enable reduction in sample and reagent volumes, thereby decreasing waste generation and energy consumption. Automation enhances reproducibility while reducing operator exposure to hazardous substances, whereas miniaturization further minimizes material consumption and environmental impact.
GAC methodologies focus on strategies aimed at minimizing or eliminating the use of toxic substances, reducing waste generation, and promoting the use of renewable raw materials. The 12 GAC principles emphasize prevention over remediation, energy efficiency, and a reduction in chemical toxicity [5] while encouraging an improvement in analytical techniques in order to enable real-time monitoring and prevent the formation of hazardous substances.
The tools available to implement the principles are as follows: (i) chemometrics, (ii) automation and (iii) miniaturization [10].
2.1. Theoretical Studies and Integrated Chemometric Problems
Design of experiments is widely adopted for optimization problems in all analytical steps from reagent selection to experimental conditions [11], which largely reduce the consumption of reagent and number of experimental trials, whereas machine learning [12] and, in general, artificial intelligence [13] are emerging tools in sensor data processing. Some of these chemometric applications to MIP sensors have been recently reviewed [14]. The usefulness of rational design strategies in the development of sustainable imprinted sensors has been confirmed by our recent work [15], which describes a voltammetric sensor based on an ionically imprinted polymer film for the determination of Co(II) in water. In this work, a Taguchi experimental design was employed to optimize electropolymerization conditions and monomer concentration, allowing for the synthesis of a high-affinity, high-selectivity polymer film with a minimum number of experimental cycles. By combining the statistical design of the experiments with the electropolymerization process, sensor fabrication required minimal reagent consumption and waste generation.
Alongside traditional DOE approaches, theoretical studies are increasingly being employed in line with GAC best practices. Recently, computational modeling has been useful for the sustainable design of MIPs, as molecular simulations can predict monomer–analyte interactions. This enables optimized component selection, reduces the number of experiments needed to achieve high-affinity and high-selectivity materials, lowers material and solvent consumption, and accelerates the development of new sensors [16].
Mohsenzadeh et al. [16] examined computationally assisted strategies for MIP design, highlighting how atomistic and quantum simulations can optimize the prepolymerization mixture and reduce the need for numerous experiments. Similarly, Kumar et al. [17] proposed a comprehensive computational strategy for the rational selection of functional monomers in the design of protein-imprinted polymers. This strategy combined molecular docking, molecular dynamic simulations, and quantum mechanical calculations. Indeed, the authors studied the interactions between the Parkinson’s disease biomarker DJ-1 and several electropentamers, including polypyrrole, polythiophene, poly(o-aminophenol), and poly(3,4-ethylenedioxythiophene), demonstrating that the latter exhibited the most stable binding configuration, supported by low RMSD and RMSF values and strong hydrogen bonding and van der Waals and electrostatic interactions. This approach reduced the need for extensive experimental screening.
Techniques such as molecular docking, molecular dynamics, and quantum chemical calculations have proven attractive for exploring monomer–template binding mechanisms and energies. Progress has been made with the work of Dhanalakshmi et al. [18] who demonstrated how a fully integrated in silico workflow, combining density functional theory (DFT) analysis and the Natural Bond Orbital 3.1 program, can be used to calculate hydrogen bonding relationships, intra- and intermolecular structural forces, and MIP active sites. Danish et al. [19] used Time-Dependent DFT to calculate the electronic transition, combined with molecular docking to study structure–property relationships and non-covalent interactions to guide rational component selection prior to polymerization. Indeed, the authors synthesized and spectroscopically validated 3-methylisoxazolo[4,5-b]pyridine in the laboratory, then extracted some factors describing the system in quantum terms and mapped the interaction sites, using docking on representative protein targets. Although their study focuses on proteins and ligands, the proposed methodology can be transferred to MIP design (monomers/crosslinkers/analyte binding), as it allows for a detailed description of the forces responsible for molecular recognition.
Another example of the practical application of computational approaches in the rational design of environmentally friendly MIP sensors is provided by Beigmoradi et al. [20], who combined molecular docking calculations and density functional theory (DFT) to aid in the selection of monomer–template interactions in the development of an electrochemical MIP sensor for malathion detection. In this study, theoretical investigations were conducted to determine the most favorable binding configurations between the pesticide and the monomers. By leveraging in silico predictions, the authors were able to simplify the experimental design, improving sensor performance while reducing the need for extensive empirical screening and the associated reagent consumption.
2.2. Automation in MIP Design
Automation is now an important base of GAC, as it reduces time, costs, errors, and resource consumption while simultaneously increasing data quality. The development of flow injection analysis (FIA) [21], sequential injection analysis (SIA) [22], and multiswitching [23] represents a starting point for automation in analytical chemistry. Indeed, these tools have the merit of having improved the method while simultaneously reducing the environmental impact. For example, in gas chromatography methods, automation is evident at multiple levels: from sample preparation to injection, all the way to post-analytical data management. While automation has already provided substantial benefits to traditional analytical techniques, it has also increasingly impacted the sensor field, enabling the development of automated platforms for continuous, high-throughput monitoring with reduced reliance on manual sampling [24]. In this respect, the use of microfluidics and lab-on-a-chip platforms, which allow for the automatic management of sample and reagent flow, greatly enhanced robotics in MIP devices [25].
2.3. Miniaturization and Advanced Smart Artificial Intelligence in MIP Scenarios
In MIP-based sensors, the last GAC tool, miniaturization, revealed a smart strategy not only to improve the greenness of an analytical procedure but also to simplify the production of sensor arrays. Beyond laboratory analysis, MIP-based platforms become more powerful when integrated into an Internet of Things (IoT) architecture, enabling continuous, real-time environmental monitoring with field acquisition, edge analytics, and cloud forecasting. Their integration with artificial intelligence and machine learning systems has allowed for the automation of the data processing phase. In fact, advanced algorithms can recognize complex patterns in sensory signals, paving the way for smart sensors capable of adapting to variable environmental conditions [26].
At the same time, automation enables the development of remote and in situ monitoring systems, connected to wireless networks, allowing for the collection and transmission of data in real time, for example, for water quality monitoring or the detection of emerging contaminants, eliminating the need for transport and sample preparation in the laboratory [27]. In this context, the work of Parihar et al. [28] examined the development of MIP-based electrochemical sensors integrated into the Internet of Things (IoT). Combining MIP-based recognition elements with IoT platforms enables automated data acquisition, remote monitoring, and seamless integration with wireless networks, thus minimizing the need for centralized laboratory infrastructure and the energy and reagent consumption associated with conventional analytical workflows. Deployed directly on remote devices, machine learning routines can (i) stabilize sensor baselines via drift compensation [29,30], (ii) interpret multi-analyte responses in complex matrices [31,32], and (iii) plan event-driven actions (such as scheduled measurements, interferent removal/regeneration cycles, and on-device baseline renormalization) to sustain peak performance while minimizing power and reagent consumption.
In a typical IoT-ready setup, the sensing node includes: (i) a MIP-based sensor, (ii) a portable/miniaturized potentiostat for electrochemical readouts, and (iii) an embedded controller (microcontroller or smartphone) that schedules measurements, performs basic preprocessing, and handles communication. Beduk et al. developed a portable MIP platform for bisphenol A based on laser-written graphene, combined with a home-made portable potentiostat and smartphone Bluetooth connectivity for on-site/wireless measurements [33]. Similarly, Peng et al. developed a smartphone-based MIP electrochemical system for thiamethoxam residue analysis, using a miniature electrochemical workstation with a Bluetooth module and a dedicated Android app to exchange commands and data, demonstrating a fully field-deployable connected workflow [34].
3. Green Strategies in MIP Synthesis
The adoption of green approaches in MIP synthesis represents not only a technological innovation but a paradigm for more sustainable analytics, capable of combining high performance with a strong focus on environmental impacts. Sustainable production requires the careful consideration of several factors in the design and synthesis steps, including the selection of monomers, polymerization methods, and solvents for cavity formation.
3.1. Biomass-Derived Functional Monomers and Additives During MIP Synthesis
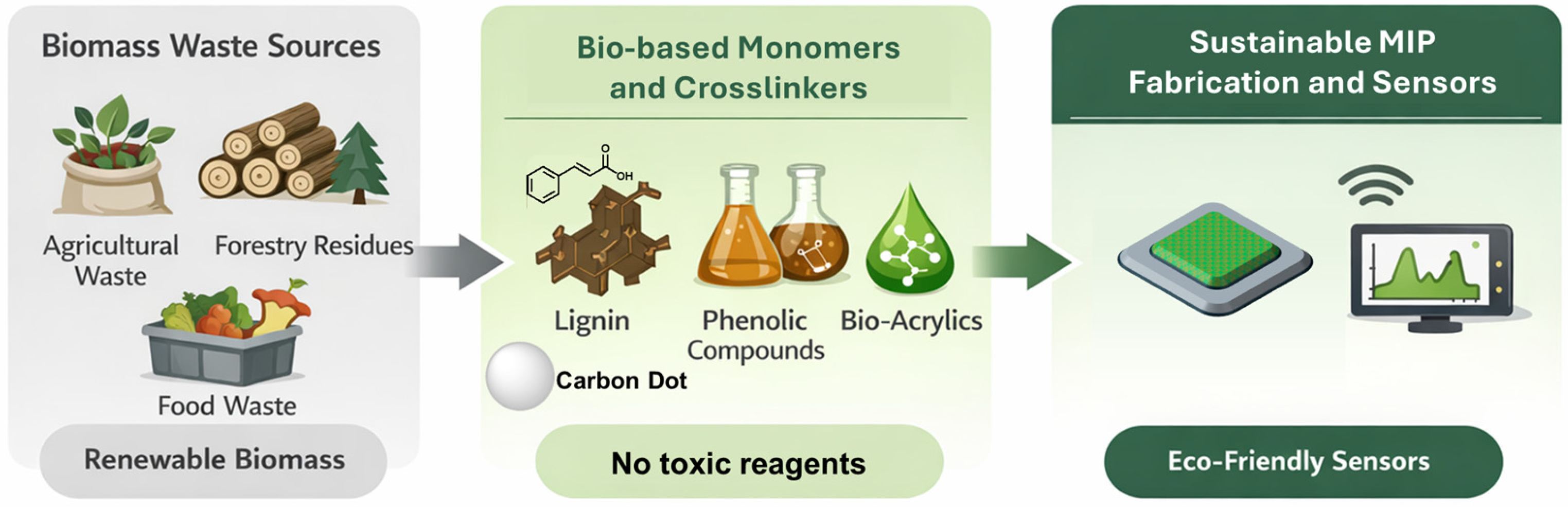
Several studies have employed crosslinkers or stabilizing agents obtained from biomass, illustrating practical approaches to incorporating renewable components into MIP fabrication [35]. Bio-based monomers, such as bio-based acrylic acid derivatives, lignin, and natural phenolic compounds, can constitute valid alternatives. The so-called greenified MIPs (GMIPs) [36] incorporate biomass-based materials, such as chitosan [37], cellulose [38], and carbon dots [39], and take advantage of the peculiar properties of these natural biomasses/materials. The former, thanks to the controlled formation of adhesive films, produce sensing material with mechanical stability and cationic sites for electrostatic interactions; cellulose can be used as a robust and porous MIP support, whereas carbon dots can serve as environmentally benign alternatives to conventional components (Figure 2).
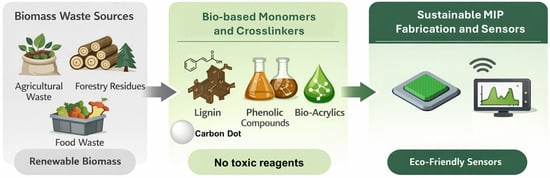
Figure 2.
A scheme for the development of sustainable sensors from renewable biomass and waste material.
Guo et al. [40] examined the use of monomers derived from natural resources in the synthesis of MIPs, giving importance to the choice of starting materials: the use of short amylose (SAM) as a bio-based functional monomer, such as λ-cyhalothrin (LC), did not compromise the recognition capacity of the resulting polymer towards λ-cyhalothrin (LC). Therefore, the use of monomers obtained from agro-industrial wastes, such as those obtained from the by-products of plant processing, offers an environmentally friendly alternative to traditional monomers of petrochemical origin [41] and is compatible with the principles of the circular economy, as it valorizes waste materials and contributes to a reduction in waste.
Hawash et al. [42], synthesized a printed polymer based on chitosan, a non-toxic, biocompatible and biodegradable monomer. Chitosan was then crosslinked with glutaraldehyde and then with Al2O3 and TiO2 nanoparticles, forming a double-crosslinked supramolecular polymer. The MIP achieved good results, demonstrating a recovery capacity of 99.03% and 99.06%, respectively, of Cd(II) and As(V) from water.
Similarly, the work of Le Goff et al. [43] demonstrates the realization of MIPs as biopesticide delivery systems of resveratrol, a natural polyphenol with antifungal activity, using plant-derived crosslinkers, such as epoxidized soybean oil acrylate (ESOA), combined with functional monomers such as 4-vinylpyridine and 1-vinylimidazole. ESOA-based MIPs showed specificity, affinity, and selectivity comparable to those prepared with conventional crosslinkers while achieving sustainability.
Interestingly, some reagents derived from natural resources, waste, or renewable resources minimize the use of toxic compounds and also promote and control the formation of MIP-based nanoparticles. In this context, Yin et al. [44] reported the synthesis of MIPs through a Pickering emulsion method, employing lignin-based dispersants as a sustainable alternative to conventional petrochemical-derived stabilizers. The authors demonstrated that the use of these lignin derivatives in the solid-phase dispersion of the matrix ensured the regular morphology and uniform particle size of the synthesized MIPs while reducing the consumption of organic solvents and shortening the analysis time.
Carbon quantum dots (CQDs) are incorporated into MIP synthesis as multifunctional nanocomponents [38,39]: they act as a nanostructured core or support around which the polymer grows (CQD@MIP) and enhance the signal generation of the final sensor, because their strong photoluminescence converts the recognition signal into a measurable fluorescence change (turn-off/turn-on) [45,46]. During synthesis, the high density of surface features on CQDs promotes the priming and stabilization of the polymer layer and helps regulate the microenvironment of the cavities formed during imprinting [39,46]. Finally, CQDs welcome some green-by-design approaches because they can be obtained via biomass derivatives, exhibit low toxicity compared to heavy metal semiconducting QDs, and are compatible with aqueous synthesis [38,39]. Kazemifard et al. [47] developed a green CQD@MIP platform by synthesizing carbon dots from rosemary leaves and subsequently stabilizing a silica (TEOS) shell on the CQDs via inverse microemulsion, before forming a silica MIP layer using APTES/TEOS around the template. Thus, the developed CQD@SiO2@MIP fluorometric sensor for thiabendazole in fruit juices combined a biomass-derived fluorophore with silica imprinting chemistry using relatively mild conditions. Similarly, Liu et al. [48] reported a simple strategy in which carbon dots were produced from sweet potato peels and then coupled with a molecularly imprinted polymer to achieve selective fluorescence quenching upon binding.
Chitosan-based MIPs have shown high selectivity and low LODs in electrochemical and photoelectrochemical systems. For example, Moreira et al. [49] developed a MIP sensor for C-reactive protein and achieved a pg mL−1 detection range. Xiao et al. [50] developed a chitosan-based MIP sensor for thiamethoxam with an LOD of 3.3 × 10−13 M. Both studies demonstrated that chitosan permits the synthesis of biocompatible polymers; however, film swelling may impact long-term stability unless the network is adequately densified, for instance, with crosslinkers [51]. Analogously, cellulose-derived hydrogels/supports provide mechanical strength and water compatibility and are attractive for flexible and wearable devices, although durability under hydration and continuous cycling conditions still determines their lifespan and requires careful reinforcement strategies [52,53]. Carbon-dot@MIP hybrids often provide strong fluorescence readouts with good selectivity for small molecules and even viral antigens [54], but photostability and matrix-dependent quenching must be engineered to avoid signal drift during prolonged use [55]. From an economic standpoint, chitosan and cellulose are low-cost and widely available biopolymers, while CQDs can be synthesized from waste streams and remain inexpensive products. Therefore, material costs are typically lower than those of specialized petroleum-derived monomers, while the final device costs are determined by the cost of the transducer or receptor [55].
3.2. Sustainable and Alternative Solvents in MIP Synthesis
The choice of solvent in MIP synthesis is critical not only for environmental sustainability but also for enhancing polymer properties and binding affinities [56]. In the past, the synthesis of MIPs was carried out mainly in aprotic organic solvents used as porogens, such as acetonitrile, toluene, and chloroform, with the aim of stabilizing the non-covalent interactions between the template and functional monomers and controlling the morphology of the material during polymerization [56,57,58] and also because water can compete for hydrogen bonds and screen ionic or dipolar interactions, reducing the stability of the prepolymerization complex and, consequently, imprinting efficiency [58]. Since many contaminants are found in aqueous environments (heavy metals, antibiotics, pesticides, perfluorates, etc.), MIPs prepared in water or water-compatible media generally exhibit improved recognition performance and adsorption capacity compared to those synthesized in organic solvents [59,60]. Polar solvents can also promote the formation of more open pores and accessible binding sites, further enhancing selectivity and functionality. For instance, in a recent study [61], an imprinted polymeric film based on 4-aminophenylacetic acid for Cd(II) detection was synthesized in a polar aqueous solution (0.5 M H2SO4), and the embedded target was subsequently removed using 0.25 M NaOH, providing an effective strategy to minimize the use of organic solvents during both polymerization and cavity formation. Another example of aqueous-phase polymerization is also reported in the study by Smolinska-Kempisty et al. [62], where they demonstrated the aqueous-phase polymerization of N-isopropylacrylamine:acrylamide:acrylic acid at various concentration ratios. This was accompanied by a molecular modeling study of the monomers, demonstrating potential binding sites between the target analyte (gentamicin) and functional monomers in water.
Another interesting approach involves the use of supercritical carbon dioxide (scCO2) as an eco-friendly, non-toxic, non-flammable, inert and recyclable solvent. The removal of both residual reagents from the polymerization mixture and template from the polymer film with scCO2 reduces chemical residues and the environmental impact of the production. Moreover, Furtado et al. [63] showed simpler scalability with reduced costs and energy consumption, and the developed dual-templated imprinted polymers (D-MIPs) were stable and resistant, with a long life, which reduces the need for frequent replacements and disposal. Nonetheless, the use of scCO2 has the disadvantage of increasing operating costs, despite the benefits of its simplified scale-up.
The use of deep eutectic solvents (DESs) and in particular the subclass Natural Deep Eutectic Solvents (NADESs), consisting of eutectic mixtures formed from naturally occurring precursors, such as amino acids, organic acids, sugars, or choline derivatives, has recently gained attention thanks to the relevant greenness. The formation of (NA)DESs is driven by a network of hydrogen bonds between a donor and an acceptor component and van der Waals and electrostatic interactions that lead to charge delocalization, resulting in changes in the chemical–physical properties compared to those of the individual components. (NA)DESs permit the modulation of their chemical properties by changing precursors and their molar ratio or adding modifiers, thus obtaining environments that influence solubilization and extraction or mass transfer processes. For this reason, DESs and NADESs are proposed as a more sustainable alternative to many ionic liquids (ILs) because they combine low toxicity and biodegradability, potential solute stabilization, sustainability and simpler synthesis and the possibility of being obtained with rapid and efficient methods [64].
NADESs were successfully used by Porfireva et al. [65] for the synthesis of epinephrine-imprinted polythionine. The resulting impedimetric sensor showed high sensitivity, with a detection limit of 50 nM, and selectivity towards structurally related neurotransmitters such as norepinephrine and serotonin. Wang et al. [66] synthesized a molecularly imprinted hydrophilic resin (HIR) in a water-and-DES-based system, eliminating the need for large amounts of hazardous organic solvents and using crosslinking agents such as hexamethylenetetramine (HMTA) instead of formaldehyde, thus reducing exposure to toxic substances. The resin was applied in aqueous environments without contamination by organic solvents to extract selected target molecules from complex biological samples, and the process was claimed to be eco-friendly, safe, low-toxic and non-volatile.
Drawbacks in employing NADESs are viscosity and mass transfer that can limit film quality, and the optimization of deposition and washing steps is often required [65]; however, economically, choline chloride/urea systems are made with inexpensive building blocks [67].
3.3. Alternative Routes in MIP Production
Additive manufacturing (AM) techniques have been recently used in the development of MIPs. Camilli et al. [68] reported the fabrication of oxytetracycline-based MIPs using Digital Light Processing (DLP) 3D printing. Their study demonstrated that complex structures with selective recognition capabilities can be achieved, opening new opportunities for environmental monitoring and water treatment. The manufacturing approach reduces material waste and enables the design of tailored geometries with increased surface area, thus improving binding efficiency, highlighting how the integration of 3D printing technologies with molecular imprinting could pave the way for eco-friendlier and more versatile MIP-based devices. Despite the promising results, several limitations of 3D-printed MIPs need to be acknowledged. The formulations employed in the reported study relied on conventional monomers and solvents, such as methacrylic acid and dimethyl sulfoxide, which are not considered green reagents. Furthermore, the overall oxytetracycline removal efficiency (32.5%) was lower than that typically achieved with traditional MIPs. Nonspecific interactions also contributed to reduced imprinting factors, particularly at low analyte concentrations, while repeated binding and washing cycles led to structural stress and the limited reusability of the printed devices. Additive manufacturing can reduce waste and unlock new architectures that improve sensor performance. However, for these promises to truly materialize in eco-friendly MIP platforms, DLP must be combined with bio-/renewable monomers and other environmentally friendly elements. The limitations of these printed structures are scalability and mechanical robustness along with difficulties with thermolabile models/molecules.
The reduction in energy consumption is another GAC principle that can also be pursued with microwave (MW) polymerization [69], photopolymerization [70] or polymerization at room temperature. Microwaves (MWs) are energy-efficient, so their use should be encouraged when temperatures greater than RT are required. Chen et al. [71] prepared florfenicol-imprinted polymers in just one hour, achieving uniform spherical morphology and significantly reducing the consumption of organic solvents thanks to MW-assisted emulsion polymerization. Their study demonstrated that MW heating accelerated the polymerization process compared to conventional thermal or UV initiation and improved the reproducibility and adsorption capacity of the resulting polymers. The benefit of this type of polymerization is the reduction in energy consumption compared to traditional methods.
Astuti et al. [72] compared MW irradiation and conventional stirring methods in the preparation of enoxaparin-imprinted polymers. The microwave-assisted approach produced polymers with higher adsorption capacity (43.47 ± 0.40 mg g−1 vs. 40.77 ± 0.75 mg g−1) and a higher imprinting factor, selectivity, and recovery rates while reducing synthesis time and energy consumption. However, this technique may not always be suitable for thermolabile analytes, as the heating associated with microwave irradiation can compromise their structural integrity.
When the target is a high-molecular-weight analyte, such as proteins or synthetic polymers, the most sustainable routes to specificity are those that avoid harsh organic media and aggressive treatments while keeping the recognition sites accessible [73,74]. Therefore, this pushes protein–MIP design towards approaches such as thin films or surface imprinting, which typically require lower reagent volumes, reduce device material throughput, and facilitate faster template removal and rebinding [73,75]. A particularly green strategy is epitope imprinting, in which a short peptide fragment replaces the entire protein because it supports greener manufacturing because peptides are chemically robust, can be reproducibly prepared, and allow for imprinting under softer conditions than those often required to handle fragile full-length protein templates [73,76]. Similarly, solid-phase nanoMIP synthesis can contribute to sustainability by increasing specificity, as the template is covalently immobilized on a reusable solid support, and the workflow includes the affinity-type selection of high-affinity ligands that reduces the loss of recognition elements and enables template reuse, which is a concrete lever for reducing waste and costs, especially for expensive protein targets [73,77]. Finally, maintaining protein specificity often requires gentle aqueous polymerization conditions and the careful, clean removal of the template. Therefore, the enzymatic digestion of protein templates has been demonstrated as an effective alternative to aggressive chemical elution while preserving cavity integrity [73,78]. Meng et al. [78], for instance, developed an imprinted β-lactoglobulin electrochemical sensor for the detection of adulterants in camel milk, using trypsin as the cleavage reagent. Unlike conventional chemical treatments (e.g., NaOH or methanol/acetic acid), enzymatic hydrolysis allowed for the complete digestion of the template into peptides without affecting the cavities, thus preserving recognition sites and reducing the use of toxic chemicals, demonstrating that enzymatic template removal may represent a sustainable alternative to traditional methods. The MIPs were combined with gold nanoparticles and reduced graphene oxide that enhanced electron transfer, and the resulting sensor showed high sensitivity (LOD 3.58 ng/mL) and selectivity in the analysis of β-lactoglobulin in real samples. Although enzymatic template removal reduces the use of aggressive solvents and does not cause damage to the imprinted sites, the cost of analytical-grade proteases [79] makes the strategy more expensive than acidic or basic treatments, also considering that the template removal step is quite often the least cost-effective step in MIP development [80].
4. Sustainability Assessment Tools for MIP Production
Lifecycle Assessment (LCA) emerged as a suitable tool for quantifying the overall environmental impact associated with an analytical process, from raw material extraction to reagent and instrument disposal. Unlike traditional assessments that focus solely on the “greenness” of chemical reagents or operational steps, LCA enables a comprehensive and quantitative comparison between different methods by integrating factors such as energy consumption, global warming potential, resource depletion, ecotoxicity, and waste generation within a standardized framework [81].
From a sustainability perspective, the fabrication of MIP-based sensors has increasingly incorporated LCA metrics to translate theoretical principles of sustainable synthesis into real-world applications. Accordingly, the adoption of LCA in the MIP field has been proposed to identify the most environmentally impactful stages, including polymer synthesis, template removal, device fabrication, and operational energy consumption, thus providing guidance for improving the sustainability of MIP-based sensors.
4.1. The ReCiPe Method
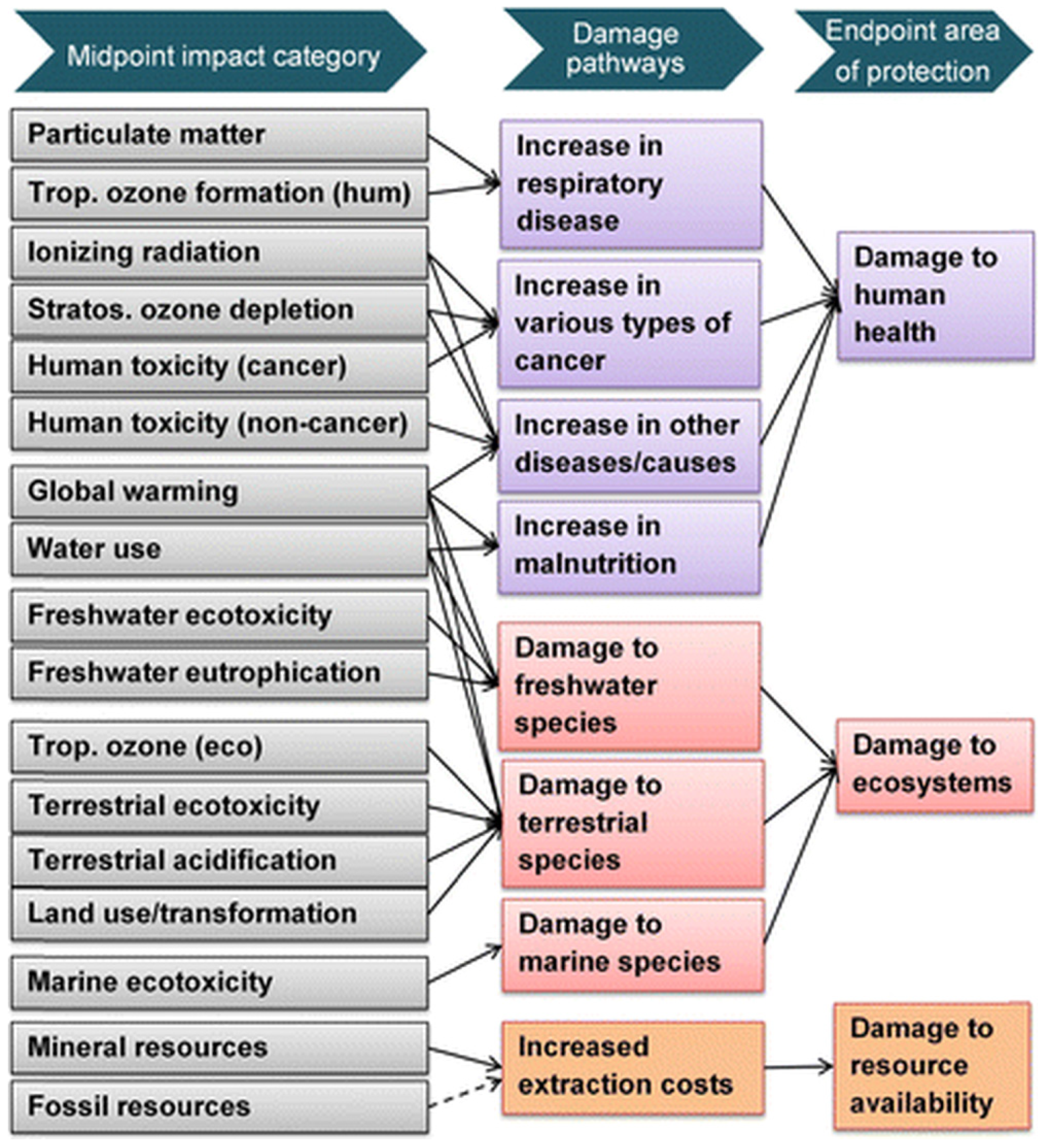
ReCiPe is a well-established and widely adopted Lifecycle Impact Assessment (LCIA) methodology within the LCA framework. The acronym represents the initials of the institutions that contributed the most to the development of this method: RIVM and Radboud University, CML and PRé Consultants. It was specifically developed to comprehensively evaluate the environmental impacts associated with products, services, and organizations, encompassing a broad range of impact categories, including climate change, acidification, human toxicity, and resource consumption. This metric is structured around two complementary assessment levels. The first is the midpoint level, which includes 17 impact categories, such as climate change, acidification, eutrophication, particulate matter formation, toxicity, water use, land use, and resource scarcity, each quantified through characterization factors expressed in equivalent units. The second is the endpoint level, which aggregates the midpoint impacts into three areas of protection, human health, ecosystem quality, and resource scarcity, using category-specific mid-to-endpoint conversion factors [82], as the scheme in Figure 3 shows.
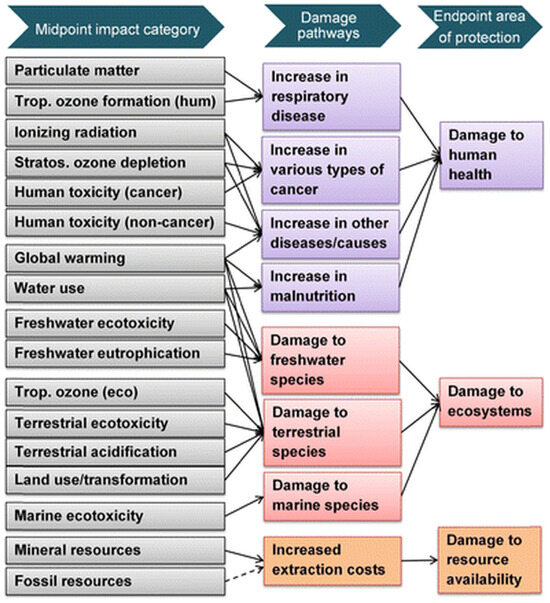
Figure 3.
Impact categories included in ReCiPe2016 method and their relationship with protection areas. From [82].
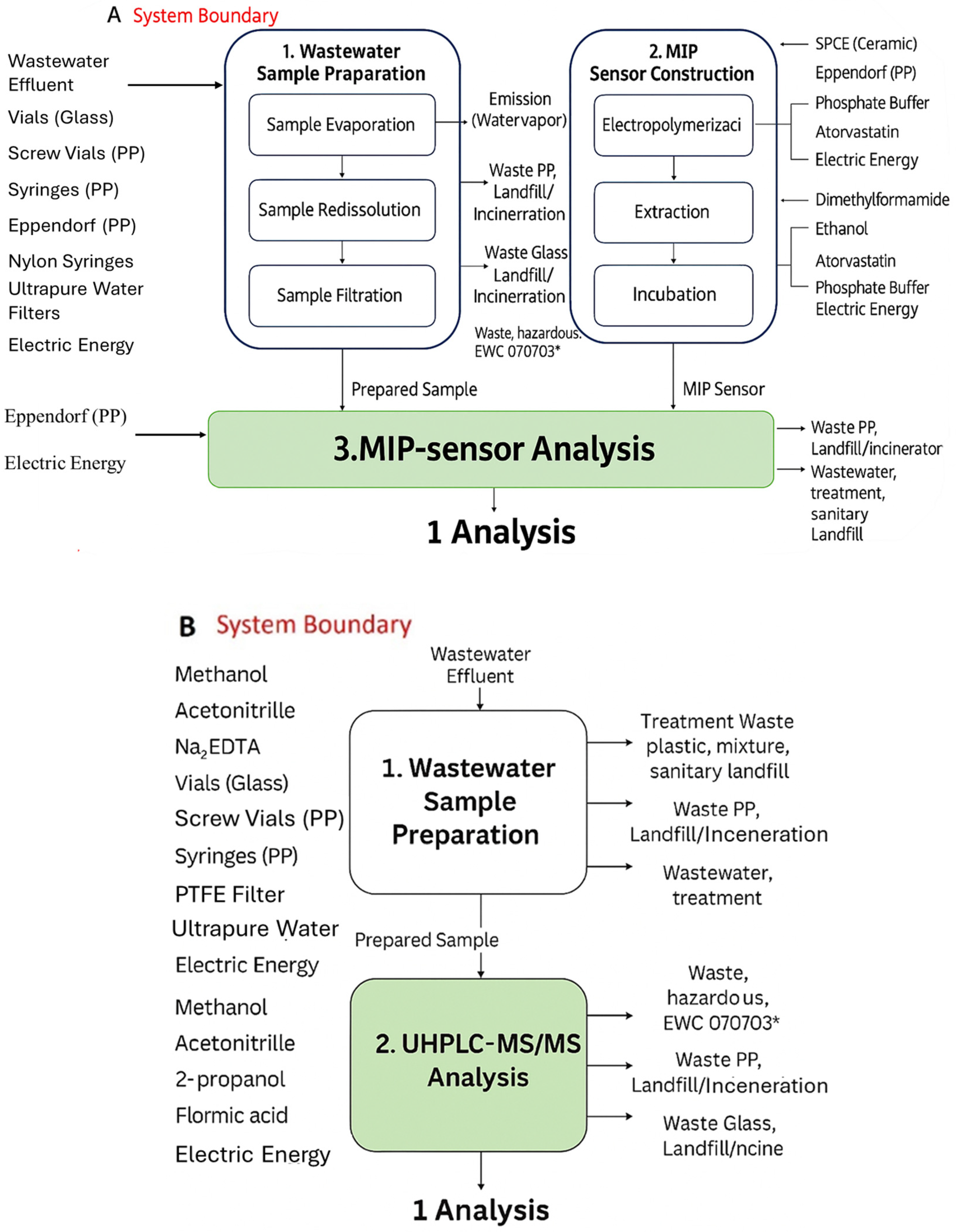
The applicability and reliability of LCA methodologies in the field of MIP-based sensors have been effectively demonstrated in recent studies, showing their suitability for benchmarking sensor-based approaches against established analytical techniques. In this context, Rebelo et al. [83] developed a MIP-based electrochemical sensor for the detection of atorvastatin (ATV) in wastewater and surface water effluents and benchmarked its analytical performance against ultra-high-performance liquid chromatography coupled with tandem mass spectrometry (UHPLC–MS/MS), as shown in Figure 4. To evaluate the environmental implications of both approaches, the authors performed a LCA using the ReCiPe Midpoint method [84]. The results demonstrated that the MIP-based sensor exhibits a significantly lower environmental impact than the chromatographic technique, highlighting its potential as a more sustainable alternative for pharmaceutical monitoring in aquatic environments. This reduced footprint was mainly attributed to the use of less impactful materials and milder processes in the fabrication and operation of the MIP sensor compared with conventional UHPLC–MS/MS methodologies. Despite the successful application of ReCiPe in the MIP sensor context, some limitations inherent to the methodology and to lifecycle inventory remain. In particular, the increasing use of environmentally friendly solvents, monomers, and crosslinkers, often not yet included in LCA databases, may introduce uncertainties or methodological failure in impact assessment. Moreover, because sensors are typically designed for on-site operation, it is essential to harmonize the different operational scenarios and to account for device durability and reuse, reporting the environmental impacts as a function of the repetitive measurement cycles.
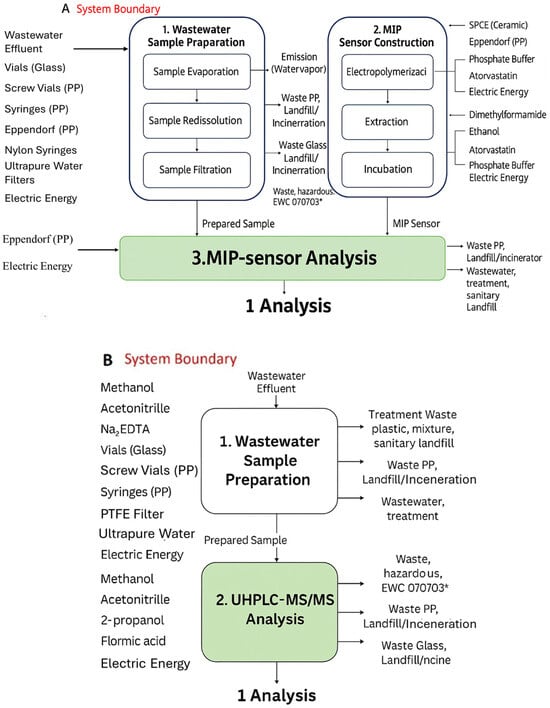
Figure 4.
Flowchart of analysis of atorvastatin (ATV) using two methods described in paper by Rebelo et al.: (A) electrochemical MIP sensor and (B) UHPLC-MS/MS. Adapted from [83].
An important factor in the LCA performance of MIP-based sensors is the reusability enabled by regeneration. Each sensor regeneration reduces the amount of consumables, solvent use, energy, and material throughput per analysis. Altintas et al. [85] demonstrated that nanoMIP-based sensors immobilized on SPR chips for the detection of diclofenac could be regenerated and reused for subsequent analyses while maintaining the same performance. Imprinted nanoparticles were synthetized by solid-phase synthesis and using UV light as an initiator in an organic environment. Sensor regeneration was performed by washing the nanoMIP-functionalized SPR chip with an acidic glycine–HCl solution. The pH affects hydrogen bonding and electrostatic interactions between the analyte and the recognition element sites, allowing for target release and baseline recovery for subsequent measurement cycles.
4.2. AGREE Methods
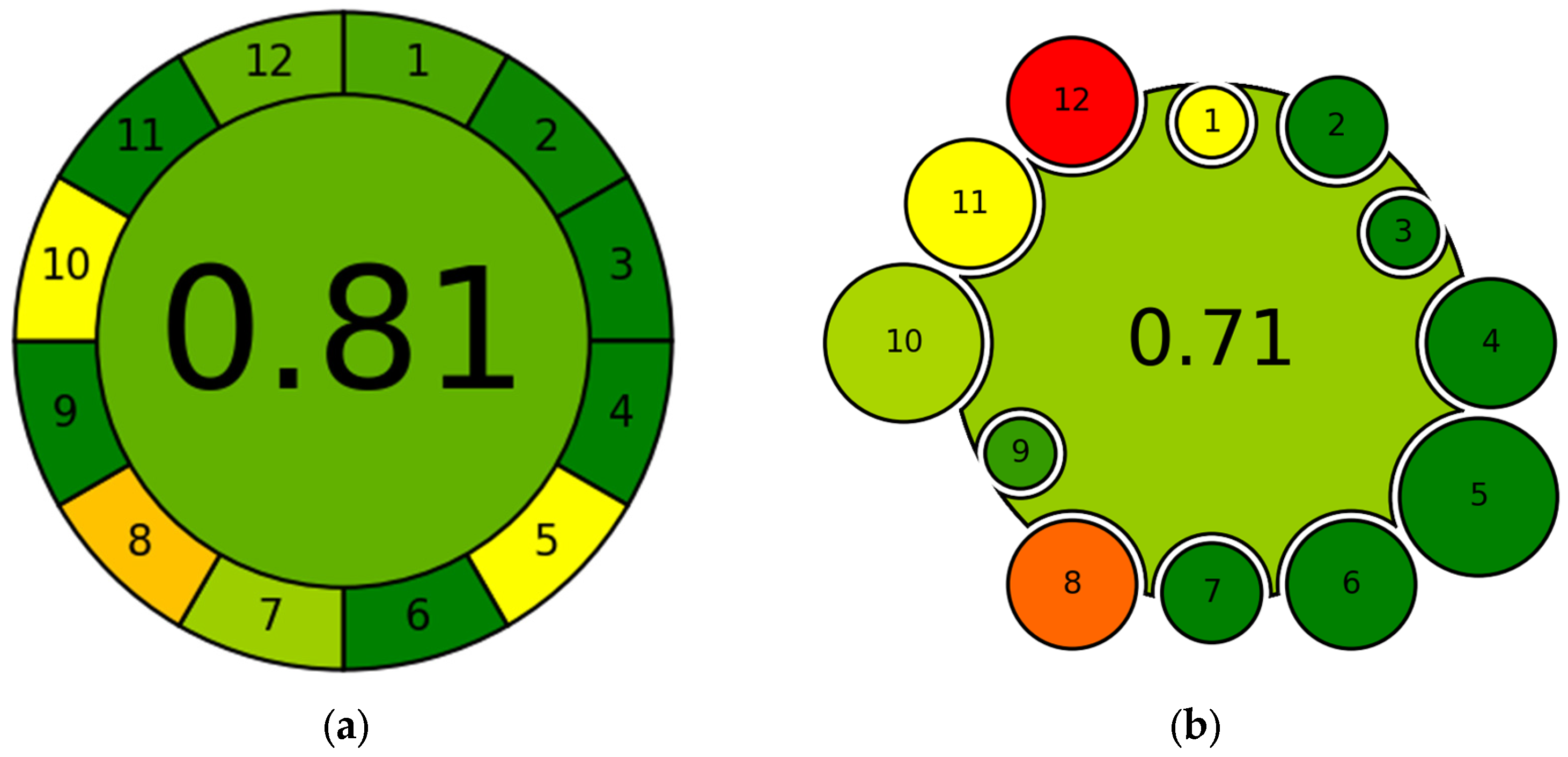
The AGREE method is an evaluation tool based on the principles of GAC, providing a score between 0 and 1, where values close to 1, represented in green for immediate interpretation (Figure 5a), indicate a highly environmentally friendly approach, while values close to 0, represented in red, indicate nonenvironmentally friendly practices and therefore a significant environmental impact [86]. The criteria for assigning this index are shown in Table A1.
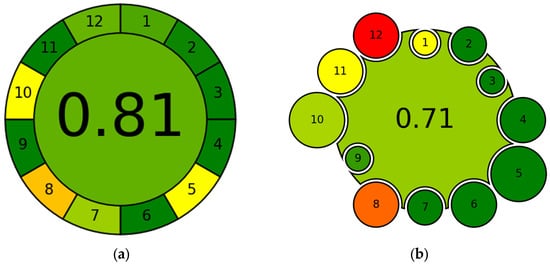
Figure 5.
Example of graphical representation of (a) AGREE and (b) AGREEMIP. The numbers displayed within each sector identify the criterion assessed by the tool. Each sector is colored based on the score (0–1) assigned to that criterion: red = slightly green approach; orange/yellow = intermediate; green = green approach.
AGREEprep is a tool that evaluates environmental sustainability at the sample preparation phase with higher levels of accuracy and specificity, providing a score from 0 to 1 [87]. It is based on 10 criteria addressing various aspects, such as solvent and reagent safety, waste and material minimization, energy efficiency, automation and integration of steps, use of sustainable and reusable materials, and operator safety.
The AGREEMIP tool [4] represents a key advancement in MIP-based sensors, providing one of the first frameworks specifically designed for systematic sustainability assessment in MIP fabrication. This tool assigns a quantitative “greenness” score to MIP synthesis by analyzing key parameters, such as the type and amount of monomers and crosslinkers, the nature and volume of solvents, energy consumption, waste generation, and the recyclability or final disposal of the material. This approach not only allows for the comparison of different synthesis protocols but also for the identification of critical points requiring intervention to improve overall sustainability and for easily assessing the degree of eco-sustainability of MIP synthesis. Marc et al. [4] state that the tool can provide useful information on which aspects of synthesis need to be improved to make the process more environmentally friendly and that the assigned score should not be considered an absolute value but rather a benchmark from which to start to improve the degree of eco-sustainability. An example of a graphical representation of AGREEMIP is shown in Figure 6b with the respective criteria of evaluation in Table A2.
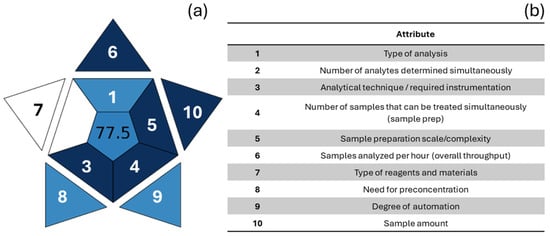
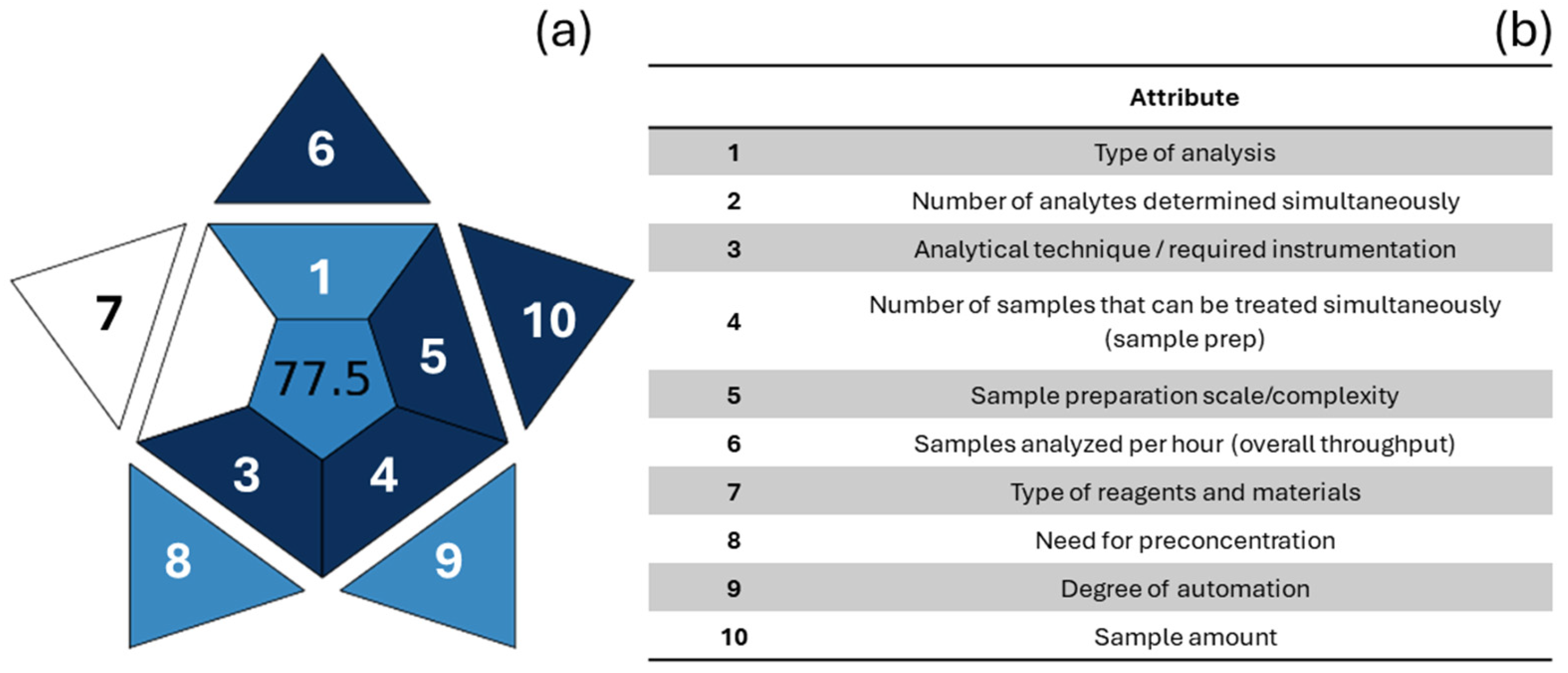
Figure 6.
Graphical representation of BAGI (a). Attributes are listed in (b), and each has corresponding shape in graph (a). Each sector is colored based on the score (0–100) assigned to the relative criterion according to the BAGI scale: dark blue = high compliance/practicality; light blue = intermediate compliance; white = low or no compliance. The overall BAGI score (0–100), which is placed in the center, summarizes the applicability/practicality of the method.
As summarized in Table 1, various green strategies have been adopted in the synthesis and testing of MIP-based sensors, although they do not always address all aspects of sustainability. Many studies emphasize the synthesis stage, using low-impact supports and media (e.g., biodegradable paper or PBS), low-energy polymerizations (electropolymerization, UV for short time), and eco-friendly crosslinkers/monomers, which often results in high AGREEMIP scores. In contrast, the sensor preparation step (AGREEPREP) is less frequently documented and generally shows room for improvement. Table A3 shows the criteria for AGREEprep.

Table 1.
Summary of green metrics for recent studies on MIP-based sensor development.
4.3. BAGI Method
To provide a more comprehensive overview of environmental sensitivity, Manousi et al. [102] proposed a new index for assessing the practicality and applicability of an analytical method, the Blue Applicability Grade Index (BAGI). This new index (Figure 6) complements the previously mentioned tools, but unlike the others, it focuses on practical aspects, considering ten criteria to produce a score describing the applicability and functionality of an analytical method. The criteria are: the type of analysis, the number of analytes being simultaneously determined, the analytical technique and required analytical instrumentation, the number of samples that can be simultaneously treated, the sample preparation, the number of samples that can be analyzed per hour, the type of reagents and materials used in the analytical method, the preconcentration requirement, the degree of automation, and the sample size (Figure 6b, Table A4). The resulting pictogram is a blue color scale, from dark blue to light blue to white, indicating, respectively, the high, medium, low, and no compliance of the method with the established criteria. The authors recommend a total score above 60 so that the analytical method can be considered “practical.” The main advantage of the BAGI is its ease of use and application, as it is accessible simply via a website (bagi-index.anvil.app, accessed on 31 December 2025).
Shehabeldeen et al. [103] present automated MIP-based dispersive microsolid-phase extraction for the determination of ketoprofen in environmental waters as a proof-of-concept performance feasibility study. The presented method is environmentally friendly because it favors aqueous environments, microvolumes, and a simplified workflow, free of impactful reactions and energy-intensive steps. These choices, according to the AGREE metric, have a positive impact on reagent/solvent use, waste, energy, miniaturization, and automation, with a score of 0.78. Finally, regarding practical transferability (common instrumentation, number of samples/hours, degree of automation, materials used), the BAGI provides a complementary applicability index that helps balance sustainability and the practical usability of the protocol, with a score of 75.0 thanks to the use of common instruments such as a compact potentiostat and SPE.
4.4. Complementarity and Conflicts Between Lifecycle Metrics
The tools adopted in this work for the greenness assessment of a MIP-based sensor cover different phases of a sensor’s lifecycle and should be interpreted in a complementary manner. In summary, AGREEMIP focuses on synthesis (monomers/crosslinkers, solvents/porogens, priming, elution, reuse), while AGREE and AGREEprep weigh the overall analytical method and sample preparation, respectively. The BAGI describes the practicality/transferability of the procedure, and LCA (ReCiPe) offers a quantitative estimate of environmental impacts across the entire lifecycle, when the inventory is available. Table A5 summarizes the metrics analyzed in this work, highlighting any conflicts.
The data in Table 1 may reveal conflicts for studies that measured at least two indicators. For the baricitinib [93] and scopolamine [94] sensor, a high AGREEMIP score coexists with a lower AGREE score, highlighting that green synthesis does not automatically guarantee a low-impact analytical process, indicating that the environmental impact is often concentrated in downstream processes. Conversely, for the glyphosate sensor [91], balanced scores on AGREEprep and AGREEMIP indicate consistent green choices throughout most of the analytical process, from synthesis to use. In the case of the abacavir [96] sensor, in which all three metrics were measured, it scored higher on the AGREEMIP index, achieving good scores also in the sample preparation phase and throughout the analytical process in general. Sun et al. [99] presented a green strategy for an electrochemical MIP sensor for the rapid quantification of Polygonatum sibiricum polysaccharides (PSPs). The device was built on a carbonaceous glass electrode modified with a carboxylated activated carbon/polyaniline/chitosan composite and was optimized using a Box–Behnken experimental design, which reduced impedance, increased active area, and improved performance, achieving excellent results in terms of selectivity, reproducibility, and stability. The sustainability elements are varied, including the miniaturization of the analytical process; use of chitosan as a biopolymer component of the functional film; use of DoE to maximize information and minimize evidence; and, above all, the systematic evaluation of “greenness” through AGREE, AGREEprep, and AGREEMIP, achieving 0.77, 0.7, and 0.9, respectively.
Birari et al. [97] admit that, despite achieving a score of 0.72 in AGREEMIP, the visual output highlights weaknesses, especially in criterion 10 (template elution solvent), aiming to plan adjustments to achieve a fully sustainable and environmentally friendly model.
Finally, LCA can sometimes diverge from the “green” scores if the use phase, consumables, or end-of-life phase dominate the impact, and it can suffer from uncertainties when green reagents are not yet included in databases. Therefore, it is a useful tool if used as in Rebelo et al. [83] as a quantitative check for method vs. method comparisons, such as sensor vs. instrumental technique, as shown for MIP vs. UHPLC–MS/MS in the detection of atorvastatin.
These examples clearly highlight the importance of calculating and displaying one or more indices because they offer a basis for implementing the process with greener alternatives with the aim of having the lowest possible impact.
5. Conclusions and Future Outlooks
The transition to sustainable and real-world MIP devices requires an integrated approach, where molecular design, manufacturing, and environmental impact assessment are addressed across the entire sensor lifecycle. The adoption of bioderived monomers/crosslinkers, the use of deep eutectic solvents (DESs/NADESs), and low-energy processes, combined with cleaner template removal strategies, form the basis for reducing consumption, waste, and risks. Different tools, AGREE/AGREEprep/AGREEMIP/BAGI, have been designed to prove that “green” claims are actually feasible and can be used to identify critical stages (synthesis/polymerization, template removal, use/regeneration and reusability, end-of-life). Regarding reusability, regeneration must be designed from the start, using regenerable materials, low-toxicity elution protocols, and safe decommissioning strategies, not only selectivity or imprinting factors. In terms of innovation, multiscale computational design and DoE reduce trial and error and power consumption, while miniaturization, microfluidics, IoT integration, and artificial intelligence and machine learning (AI/ML) enable real-time monitoring. Short-term priorities therefore include (i) regeneration and cycle counting standards, (ii) developing “green by design” formulations (aqueous systems), and (iii) the use of LCA tools to identify critical steps and assess the quality of a MIP-based sensor. Looking to the future, scale-up plans require the combination of nanotechnology, AI/ML-driven modeling and computational chemistry to pave the way for sustainable and innovative analytical chemistry, capable of overcoming the challenges that have so far slowed the application of MIP-based sensor technologies in real-world settings.
Author Contributions
Conceptualization, M.C., S.D.M. and G.E.D.B.; writing—original draft preparation, M.C.; writing—review and editing, S.D.M. and G.E.D.B.; supervision, G.E.D.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AGREE | Analytical GREEnness metric approach and software |
| AGREEMIP | Analytical greenness assessment tool for molecularly imprinted polymer synthesis |
| AGREEprep | Analytical greenness metric for sample preparation |
| AI/ML | Artificial intelligence/machine learning |
| AM | Additive manufacturing |
| AMVI | Analytical method volume intensity |
| APTES | (3-aminopropyl)triethoxysilane |
| ATV | Atorvastatin |
| Au NPs | Gold nanoparticles |
| BAGI | Blue applicability grade index |
| BBD | Box–Behnken design |
| CC | Creative Commons |
| CC BY | Creative Commons Attribution license |
| CCD | Central composite design |
| CML | Centrum voor Milieukunde (Institute of Environmental Sciences, Leiden University) |
| CQD(s) | Carbon quantum dot(s) |
| DA | Dopamine |
| DES(s) | Deep eutectic solvent(s) |
| DFT | Density functional theory |
| DJ-1 | Parkinson’s disease protein 7 (DJ-1 biomarker) |
| DLP | Digital light processing |
| DOE | Design of experiments |
| ED | Emodin |
| ESOA | Epoxidized soybean oil acrylate |
| FIA | Flow injection analysis |
| GAC | Green analytical chemistry |
| GC | Green chemistry |
| GCE | Glassy carbon electrode |
| HIR | Hydrophilic imprinted resin |
| HMTA | Hexamethylenetetramine |
| IoT | Internet of things |
| JAK | Janus kinase |
| LCA | Lifecycle assessment |
| LCIA | Lifecycle impact assessment |
| LOD | Limit of detection |
| LSTM | Long short-term memory (neural network) |
| MD | Molecular docking |
| MIP(s) | Molecularly imprinted polymer(s) |
| MIR | Molecularly imprinted resin (as used in PDA@MIR) |
| MW | Microwave |
| NADES(s) | Natural deep eutectic solvent(s) |
| NANOTEC | CNR NANOTEC—Institute of Nanotechnology |
| NBO | Natural bond orbital |
| NCI | Non-covalent interaction |
| OFAT | One factor at a time |
| PBS | Phosphate-buffered saline |
| PDA | Polydopamine |
| PDA@MIR | Polydopamine molecularly imprinted resin |
| PEGDA | Poly(ethylene glycol) diacrylate |
| PLoS | Public Library of Science |
| PSPs | Polygonatum sibiricum polysaccharides |
| ReCiPe | ReCiPe lifecycle impact assessment method |
| rGO | Reduced graphene oxide |
| RIVM | National Institute for Public Health and the Environment (Netherlands) |
| RMSD | Root mean square deviation |
| RMSF | Root mean square fluctuation |
| RT | Room temperature |
| SAM | Short amylose |
| scCO2 | Supercritical carbon dioxide |
| SI | Solvent intensity |
| SIA | Sequential injection analysis |
| SPE | Solid-phase extraction |
| SPE–GC/MS | Solid-phase extraction–gas chromatography/mass spectrometry |
| SPR | Surface plasmon resonance |
| SVM | Support vector machine |
| TD-DFT | Time-dependent density functional theory |
| TEOS | Tetraethyl orthosilicate (tetraethoxysilane) |
| UHPLC–MS/MS | Ultra-high-performance liquid chromatography–tandem mass spectrometry |
| UV | Ultraviolet |
| WCP | Whatman Grade 1 chromatography paper |
| WFP | Whatman Grade 1 filter paper |
Appendix A

Table A1.
Criteria for AGREE.
Table A1.
Criteria for AGREE.
| Criteria | |
|---|---|
| 1 | Direct analytical techniques should be applied to avoid sample treatment. |
| 2 | Minimal sample size and minimal number of samples are goals. |
| 3 | If possible, measurements should be performed in situ. |
| 4 | Integration of analytical processes and operations saves energy and reduces the use of reagents. |
| 5 | Automated and miniaturized methods should be selected. |
| 6 | Derivatization should be avoided. |
| 7 | Generation of a large volume of analytical waste should be avoided, and proper management of analytical waste should be provided. |
| 8 | Multi-analyte or multi-parameter methods are preferred versus methods using one analyte at a time. |
| 9 | The use of energy should be minimized. |
| 10 | Reagents obtained from renewable sources should be preferred. |
| 11 | Toxic reagents should be eliminated or replaced. |
| 12 | Operator’s safety should be increased. |

Table A2.
Criteria for AGREEMIP.
Table A2.
Criteria for AGREEMIP.
| Criterion | Weight | |
|---|---|---|
| 1. | Removal of polymerization inhibitors | 1 |
| Waste generation | ||
| 2. | Functional monomer | 2 |
| Total mass of the functional monomer | ||
| 3. | Template | 1 |
| Total mass of the template | ||
| 4. | Crosslinking agent | 3 |
| Total mass of the crosslinking agent | ||
| 5. | Porogen/solvent | 4 |
| Total mass of the porogen/solvent | ||
| 6. | Other reagents, adjuvants, or carriers | 3 |
| Total mass of other reagents, adjuvants, or carriers | ||
| 7. | Core/particle preparation and surface modification | 2 |
| Mass of reagents used in core/particle preparation, surface modification, or both | ||
| 8. | Polymerization initiation | 3 |
| Initiator | ||
| 9. | Size of polymer particles | 1 |
| Grain size range | ||
| 10. | Template elution solvent | 4 |
| No. of distinct hazards | ||
| 11. | Template elution technique | 3 |
| Elution technique | ||
| 12. | Final product reusability | 3 |
| Times the end products can be reused |

Table A3.
Criteria for AGREEprep.
Table A3.
Criteria for AGREEprep.
| Criterion | Description | Score | |
|---|---|---|---|
| 1 | Favor in situ sample preparation | Prioritize in-line/on-line in situ operations to minimize transport, storage and additional materials/energy. | In-line/In situ = 1.00; On-line/In situ = 0.66; On site = 0.33; Ex situ (lab after transport) = 0.00. |
| 2 | Use safer solvents and reagents | Prefer solvent-free/reagent-less; otherwise select low-toxicity, non-persistent/non-bioaccumulative chemicals. | Score 1.00 for solvent/reagent-free; 0.00 if >50 mL/g of hazardous solvents/reagents; intermediate values computed via a logarithmic function of hazardous amount. |
| 3 | Target sustainable, reusable, and renewable materials | Favor bio-based/renewable and reusable materials; promote regeneration/reuse over disposables. | Only sustainable and reusable materials used several times = 1.00; >75% sustainable/renewable = 0.75; 50–75% sustainable (single-use) = 0.50; non-sustainable but reusable = 0.50; 25–50% sustainable = 0.25; <25% sustainable (single-use) = 0.00. |
| 4 | Minimize waste | Reduce total mass/volume of waste (solvents, reagents, consumables; include sample when contaminated). | Score computed via logarithmic function of total generated waste; <1 g of waste typically yields > 0.5. |
| 5 | Minimize sample, chemical and material amounts | Smaller sample sizes and minimal chemicals/materials reduce time, energy, costs; ensure representativeness. | Score computed via logarithmic function of total amounts (sample + chemicals + materials); passive sampling can yield score 1.00. |
| 6 | Maximize sample throughput | Increase speed and/or parallelization (e.g., multi-well formats) to prepare more samples per hour. | Score computed via logarithmic function of number of samples prepared per hour (series or parallel). |
| 7 | Integrate steps and promote automation | Simplify workflows by reducing steps and increasing automation to lower reagents, energy and exposure. | Two sub-scores: steps (≤2 = 1.00; 3 = 0.75; 4 = 0.50; 5 = 0.25; ≥6 = 0.00) × automation (fully = 1.00; semi = 0.50; manual = 0.25); final score = product. |
| 8 | Minimize energy consumption | Lower total energetic requirement expressed as Wh per sample (normalize by parallel processing). | Score increases as Wh per sample decreases; e.g., <10 Wh/sample ≈ 1.00; intermediate values calculated via function; divide device Wh by samples processed simultaneously. |
| 9 | Choose the greenest post-sample preparation configuration for analysis | Prefer analytical configurations that avoid additional high-impact steps and are compatible with low-impact detection. | Score reflects the downstream analytical setup’s greenness (for example, avoids hazardous derivatization, uses low-energy detection). |
| 10 | Ensure safe procedures for the operator | Minimize operator exposure; adopt PPE and designs that reduce contact with hazardous substances. | Score derived from hazard assessment (e.g., MSDS pictograms) and procedure safety; fewer hazards and better protection give higher score. |

Table A4.
Explanation, possible options and scores for each attributable criterion according to BAGI.
Table A4.
Explanation, possible options and scores for each attributable criterion according to BAGI.
| Attribute | Options | Purpose | Score | |
|---|---|---|---|---|
| 1 | Type of analysis | Qualitative | Identifies a substance based on chemical/biological/physical properties, without quantifying it. | 2.5 |
| Screening | Detects the presence of a substance/class at the level of interest; designed to avoid false “compliant” results. | 5.0 | ||
| Quantitative | Determines the amount or mass fraction as a numerical value with appropriate units. | 7.5 | ||
| Quantitative + confirmatory | Quantifies and unequivocally identifies the analyte (structural information; aligned with EC 657/2002). | 10.0 | ||
| 2 | Number of analytes determined simultaneously | >15 analytes | Rewards multi-analyte methods: more analytes per run = higher practicality/applicability. | 10.0 |
| 6–15 (same class) or 2–15 (different classes) | 7.5 | |||
| 2–5 (same class) | 5.0 | |||
| 1 analyte | 2.5 | |||
| 3 | Analytical technique/required instrumentation | Portable, easy-to-operate instrumentation | Assesses accessibility/realism of instrumentation for routine labs (portability, widespread availability vs. rare/advanced equipment). | 10.0 |
| “Common” instrumentation (available in most labs) | 7.5 | |||
| Sophisticated instrumentation (e.g., MS, home-made interfaces/advanced systems) | 5.0 | |||
| Instrumentation not commonly available | 2.5 | |||
| 4 | Number of samples that can be treated simultaneously (sample prep) | >96 | Evaluates the ability to parallelize sample preparation (high-throughput), reducing the sample prep bottleneck. | 10.0 |
| 13–95 | 7.5 | |||
| 2–12 | 5.0 | |||
| 1 | 2.5 | |||
| 5 | Sample preparation scale/complexity | On-site sample prep or no sample preparation required | Captures time/cost/complexity of sample preparation, favoring simple or no-prep approaches. | 10.0 |
| Simple, low-cost preparation | 7.5 | |||
| Miniaturized extraction | 5.0 | |||
| Conventional multi-step preparation | 2.5 | |||
| 6 | Samples analyzed per hour (overall throughput) | >10 samples/hour | Measures real productivity including all steps (prep → determination). | 10.0 |
| 5–10 samples/hour | 7.5 | |||
| 2–4 samples/hour | 5.0 | |||
| ≥1 h per single sample | 2.5 | |||
| 7 | Type of reagents and materials | Common, readily purchasable (e.g., MeOH, ACN, HNO3, common gases…) | Assesses practicality of sourcing/using reagents and materials (relevant for accredited and routine labs). | 10.0 |
| Commercial but “uncommon” in QC labs (derivatization reagents, SPE cartridges, SPME fibers…) | 7.5 | |||
| Must be synthesized in-house (simple procedure, common lab equipment) | 5.0 | |||
| Must be synthesized in-house (requires advanced know-how/equipment, e.g., MOFs…) | 2.5 | |||
| 8 | Need for preconcentration | No preconcentration | Evaluates fit-for-purpose without extra steps; if needed, integrated one-step preconcentration is preferred. | 10.0 |
| Preconcentration in one step | 7.5 | |||
| Multi-stage preconcentration (e.g., extraction + evaporation + reconstitution) | 2.5 | |||
| 9 | Degree of automation | Fully automated | Rewards reduced human intervention (fewer errors, lower analyst exposure), distinguishing standard vs. custom automation. | 10.0 |
| Semi-automated with common devices (e.g., HPLC autosampler) | 7.5 | |||
| Semi-automated with special/home-made systems | 5.0 | |||
| Manual | 2.5 | |||
| 10 | Sample amount | Very low (bio: <100 µL or <100 mg; food/env: <10 mL or <10 g) | Considers sample availability and impact on sensitivity/waste; distinguishes bioanalytical matrices from food/environmental matrices. For “hybrid” matrices (e.g., cosmetics, plastic leachates), classify case by case based on availability. | 10.0 |
| Low–medium (bio: 101–500 µL; food/env: 10.1–50 mL or g) | 7.5 | |||
| Medium–high (bio: 501–1000 µL; food/env: 51–100 mL or g) | 5.0 | |||
| High (bio: >1000 µL; food/env: >100 mL or g) | 2.5 |

Table A5.
Overview of each tool focused on its information and limitations and typical conflicts.
Table A5.
Overview of each tool focused on its information and limitations and typical conflicts.
| Tool | LCA (ReCiPe 2016) | AGREE | AGREEprep | AGREEMIP | BAGI |
|---|---|---|---|---|---|
| Output | Midpoint (17 categories) and endpoint; normalized/weighted results | 0–1 score on the 12 GAC principles (radar plot) | 0–1 score on 10 criteria dedicated to sample preparation | 0–1 score on 12 criteria specific to MIP synthesis (monomers, solvents, energy, elution, reuse) | Applicability/practicality (score 0–100; recommended threshold > 60) across 10 criteria |
| System boundary | Cradle-to-grave (if inventory is complete): materials, energy, use phase, waste | Analytical method as a whole (measurement and workflow) | Sample preparation only | Synthesis of the MIP receptor | Analytical method (transferability to routine) |
| Informative at lifecycle stage | Entire lifecycle; particularly useful for use/regeneration and consumables | Use phase and method organization (in situ, automation, miniaturization) | Sample preparation (solvents, waste, energy, automation) | Synthesis stage (monomers/crosslinkers, solvents, template removal, reusability) | Transferability and routine use (instrumentation, throughput, automation) |
| Strengths | Quantitative comparison across methods; highlights hotspots | Fast, visual; guides operational choices | Fills AGREE gaps in sample preparation | First MIP-specific metric; highlights actionable levers | Easy to use; complementary to greenness |
| Limitations | Relies on databases; green reagents sometimes not covered; requires accurate inventories | No quantitative environmental impacts; does not detail receptor synthesis | Does not consider MIP synthesis; no LCA quantification | Does not cover sample prep and use; depends on lab data not standardized | Not an environmental metric; may reward practical solutions that are not green |
| Example of typical conflicts | May contradict “green” scores if the use phase dominates impacts [104] | Can diverge from AGREEMIP: method not green even if synthesis is green | Can be low even when AGREEMIP score is high (green synthesis, onerous prep) | Typical conflict with AGREE/AGREEprep (good synthesis, other method less green) | Trade-off with AGREE: very practical method may have higher impact or vice versa |
References
- Ramajayam, K.; Ganesan, S.; Ramesh, P.; Beena, M.; Kokulnathan, T.; Palaniappan, A.J.B. Molecularly imprinted polymer-based biomimetic systems for sensing environmental contaminants, biomarkers, and bioimaging applications. Biomimetics 2023, 8, 245. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, A.M.; Barton, S.J.; Wren, S.P.; Barker, J. Review on molecularly imprinted polymers with a focus on their application to the analysis of protein biomarkers. TrAC Trends Anal. Chem. 2021, 144, 116431. [Google Scholar] [CrossRef]
- Hammad, S.F.; Hamid, M.A.A.; Adly, L.; Elagamy, S.H. Comprehensive review of greenness, whiteness, and blueness assessments of analytical methods. Green Anal. Chem. 2025, 12, 100209. [Google Scholar] [CrossRef]
- Marć, M.; Wojnowski, W.; Pena-Pereira, F.; Tobiszewski, M.; Martín-Esteban, A. AGREEMIP: The Analytical greenness assessment tool for molecularly imprinted polymers synthesis. ACS Sustain. Chem. Eng. 2024, 12, 12516–12524. [Google Scholar] [CrossRef]
- Rao, T.R.; Srilaxmi, B. Green Analytical Chemistry: A Comprehensive Review of Eco-Scale, Greenness Metrics, and Sustaina-bility Approaches. J. Drug Deliv. Ther. 2025, 15, 179–182. [Google Scholar]
- Armenta, S.G.S.; de la Guardia, M. Green Analytical Chemistry. Trends Anal. Chem. 2008, 27, 497–511. [Google Scholar] [CrossRef]
- Malissa, H.; Roth, E. Euroanalysis VI: Reviews on Analytical Chemistry; RothLes, E., Ed.; Editions de Physique: Les Ules, France, 1987. [Google Scholar]
- Pimentel, G.C.; Coonrod, J.A.J. Opportunities in Chemistry: Today and Tomorrow; National Academy Press: Washington, DC, USA, 1987. [Google Scholar]
- Anastas, P.T. Green chemistry and the role of analytical methodology development. Crit. Rev. Anal. Chem. 1999, 29, 167–175. [Google Scholar] [CrossRef]
- Garrigues, S. The concept of green analytical chemistry. In Handbook of Green Analytical Chemistry; Wiley: Hoboken, NJ, USA, 2012; pp. 1–16. [Google Scholar]
- Tamandani, M.; Hashemi, S.H. Central composite design (CCD) and Box-Behnken design (BBD) for the optimization of a molecularly imprinted polymer (MIP) based pipette tip micro-solid phase extraction (SPE) for the spectrophotometric determination of chlorpyrifos in food and juice. Anal. Lett. 2022, 55, 2394–2408. [Google Scholar] [CrossRef]
- Wang, M.; Cetó, X.; del Valle, M. A sensor array based on molecularly imprinted polymers and machine learning for the analysis of fluoroquinolone antibiotics. ACS Sens. 2022, 7, 3318–3325. [Google Scholar] [CrossRef]
- Geng, L.; Wang, H.; Li, Z.; Huang, J.; Sun, X.; Guo, Y.; Du, F. Research progress on molecularly imprinted polymers (MIPs)-based sensors for the detection of organophosphorus pesticides. Food Chem. 2025, 490, 145137. [Google Scholar] [CrossRef]
- Di Masi, S.; De Benedetto, G.E.; Malitesta, C. Optimisation of electrochemical sensors based on molecularly imprinted polymers: From OFAT to machine learning. Anal. Bioanal. Chem. 2024, 416, 2261–2275. [Google Scholar] [CrossRef]
- Di Masi, S.; Rodriguez, N.A.M.; Costa, M.; De Benedetto, G.E.; Malitesta, C. Rational design of a portable chemometric-assisted voltammetric sensor based on ion-imprinted polymeric film for Co(II) determination in water. Nanomaterials 2024, 14, 536. [Google Scholar] [CrossRef]
- Mohsenzadeh, E.; Ratautaite, V.; Brazys, E.; Ramanavicius, S.; Zukauskas, S.; Plausinaitis, D.; Ramanavicius, A. Design of molecularly imprinted polymers (MIP) using computational methods: A review of strategies and approaches. WIREs Comput. Mol. Sci. 2024, 14, e1713. [Google Scholar] [CrossRef]
- Kumar, M.D.; Karthikeyan, M.; Kaniraja, G.; Muthukumar, K.; Muneeswaran, G.; Karunakaran, C. Computational modelling and optimization studies of electropentamer for molecular imprinting of DJ-1. J. Mol. Graph. Model. 2024, 128, 108715. [Google Scholar] [CrossRef]
- Dhanalakshmi, E.; Rajesh, P.; Suresh, S.; Priyadharshini, M.; Prabhaharan, M. Green synthesis, spectroscopic investigation, quantum chemical and molecular docking studies of 3-methylisoxazolo [4,5-b]pyridine. J. Mol. Struct. 2024, 1298, 136964. [Google Scholar] [CrossRef]
- Isravel, A.D.; Jeyaraj, J.K.; Thangasamy, S.; John, W.J. DFT, NBO, HOMO-LUMO, NCI, stability, Fukui function and hole–Electron analyses of tolcapone. Comput. Theor. Chem. 2021, 1202, 113296. [Google Scholar] [CrossRef]
- Beigmoradi, F.; Moghadam, M.R.; Bazmandegan-Shamili, A. Molecular Docking and Density Functional Studies of Selective Molecularly Imprinted Polymers Combined with an Electrochemical Sensor for the Detection of Malathion. J. Anal. Chem. 2024, 79, 1143–1158. [Google Scholar] [CrossRef]
- Růžička, J.; Hansen, E.H. Flow Injection Analysis; Wiley-Interscience: Hoboken, NJ, USA, 1988. [Google Scholar]
- Ruzicka, J.; Marshall, G.D. Sequential injection: A new concept for chemical sensors, process analysis and laboratory assays. Anal. Chim. Acta 1990, 237, 329–343. [Google Scholar] [CrossRef]
- Reis, B.F.; Giné, M.F.; Zagatto, E.A.; Lima, J.L.F.; Lapa, R.A. Multicommutation in flow analysis. Part Binary sampling: Concepts, instrumentation and spectrophotometric determination of iron in plant digests. Anal. Chim. Acta 1994, 293, 129–138. [Google Scholar] [CrossRef]
- Lerch, O.; Temme, O.; Daldrup, T. Comprehensive automation of the solid phase extraction gas chromatographic mass spectrometric analysis (SPE-GC/MS) of opioids, cocaine, and metabolites from serum and other matrices. Anal. Bioanal. Chem. 2014, 406, 4443–4451. [Google Scholar] [CrossRef]
- Grieshaber, D.; MacKenzie, R.; Vörös, J.; Reimhult, E. Electrochemical biosensors-sensor principles and architectures. Sensors 2008, 8, 1400–1458. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, Y.; Jiang, N.; Yetisen, A.K. Wearable artificial intelligence biosensor networks. Biosens. Bioelectron. 2022, 219, 114825. [Google Scholar] [CrossRef]
- Sohrabi, H.; Hemmati, A.; Majidi, M.R.; Eyvazi, S.; Jahanban-Esfahlan, A.; Baradaran, B.; Adlpour-Azar, R.; Mokhtarzadeh, A.; de la Guardia, M. Recent advances on portable sensing and biosensing assays applied for detection of main chemical and biological pollutant agents in water samples: A critical review. TrAC Trends Anal. Chem. 2021, 143, 116344. [Google Scholar] [CrossRef]
- Parihar, A.; Sharma, P.; Choudhary, N.K.; Khan, R.; Mostafavi, E. Internet-of-things-integrated molecularly imprinted polymer-based electrochemical nano-sensors for pesticide detection in the environment and food products. Environ. Pollut. 2024, 351, 124029. [Google Scholar] [CrossRef]
- Zhao, X.; Li, P.; Xiao, K.; Meng, X.; Han, L.; Yu, C. Sensor Drift Compensation Based on the Improved LSTM and SVM Multi-Class Ensemble Learning Models. Sensors 2019, 19, 3844. [Google Scholar] [CrossRef]
- Badawi, D.; Agambayev, A.; Ozev, S.; Cetin, A.E. Real-time low-cost drift compensation for chemical sensors using a deep neural network with hadamard transform and additive layers. IEEE Sens. J. 2021, 21, 17984–17994. [Google Scholar] [CrossRef]
- Giordano, G.F.; Ferreira, L.F.; Bezerra, Í.R.S.; Barbosa, J.A.; Costa, J.N.Y.; Pimentel, G.J.C.; Lima, R.S. Machine learning toward high-performance electrochemical sensors. Anal. Bioanal. Chem. 2023, 415, 3683–3692. [Google Scholar] [CrossRef]
- Cancelliere, R.; Molinara, M.; Licheri, A.; Maffucci, A.; Micheli, L. Artificial intelligence-assisted electrochemical sensors for qualitative and semi-quantitative multiplexed analyses. Digit. Discov. 2025, 4, 338–342. [Google Scholar] [CrossRef]
- Beduk, T.; Gomes, M.; Filho, J.I.D.O.; Shetty, S.S.; Khushaim, W.; Garcia-Ramirez, R.; Durmus, C.; Lahcen, A.A.; Salama, K.N. A portable molecularly imprinted sensor for on-site and wireless environmental bisphenol a monitoring. Front. Chem. 2022, 10, 833899. [Google Scholar] [CrossRef]
- Peng, S.; Wang, A.; Lian, Y.; Zhang, X.; Zeng, B.; Chen, Q.; Yang, H.; Li, J.; Li, L.; Dan, J.; et al. Smartphone-based molecularly imprinted sensors for rapid detection of thiamethoxam residues and applications. PLoS ONE 2021, 16, e0258508. [Google Scholar] [CrossRef]
- Fu, L.; Zhang, X.; Zhao, X.; Wang, L.; Fu, Y. Advances in biomass-based molecularly imprinted polymers: From sustainable design to diverse applications. Green Chem. 2025, 27, 9055–9093. [Google Scholar] [CrossRef]
- Del Sole, R.; Mele, G.; Bloise, E.; Mergola, L. Green aspects in molecularly imprinted polymers by biomass waste utilization. Polymers 2021, 13, 2430. [Google Scholar] [CrossRef]
- Sarnaghi, S.P.; Ayazi, Z. Synthesis of Chitosan-Based Molecularly Imprinted Biopolymer for Thin-Film Solid-Phase Microextraction of Tramadol Followed by Its On-Site Detection Using Digital Image Colorimetry. Anal. Chim. Acta 2025, 1369, 344335. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Tang, N.; Huang, H.; Zhang, X.; Lu, R.; Li, Y. Green and eco-friendly porous imprinted hydrogels with cellulose nanocrystals-based precise-designed hyperbranched sites for high-accuracy separation and purification of naringin. Chem. Eng. J. 2024, 493, 152821. [Google Scholar] [CrossRef]
- Keçili, R.; Hussain, C.G.; Hussain, C.M. Emerging trends in green carbon dots coated with molecularly imprinted polymers for sensor platforms. Trends Anal. Chem. 2023, 166, 117205. [Google Scholar] [CrossRef]
- Guo, X.; Yang, D.; Su, Y.; Chen, Y.; Ding, J.; Ding, L.; Song, D. High selectivity molecularly imprinted polymer based on short amylose as bio-based functional monomers for selective extraction of λ-cyhalothrin. Int. J. Biol. Macromol. 2024, 271, 132566. [Google Scholar] [CrossRef]
- Ortega, F.; Versino, F.; López, O.V.; García, M.A. Biobased composites from agro-industrial wastes and by-products. Emergent Mater. 2022, 5, 873–921. [Google Scholar] [CrossRef]
- Hawash, H.B.; Hagar, M.; Elkady, M.F.; Moneer, A.A.; El-Qelish, M.; El-Tahawy, M.M.; Kassem, T.S. Microwave-assisted supramolecular double crosslinked chitosan-based molecularly imprinted polymer for synergistic recognition and selective recovery of Cd(II) and As(V) from water: Performance and mechanistic insights. Int. J. Biol. Macromol. 2024, 281, 136263. [Google Scholar] [CrossRef]
- Le Goff, N.; Fomba, I.; Prost, E.; Merlier, F.; Haupt, K.; Duma, L.; Fayeulle, A.; Falcimaigne-Cordin, A. Renewable plant oil-based molecularly imprinted polymers as biopesticide delivery systems. ACS Sustain. Chem. Eng. 2020, 8, 15927–15935. [Google Scholar] [CrossRef]
- Yin, R.; Chen, L.; Ma, L. Extraction of matrine from soil with matrix solid-phase dispersion by molecularly imprinted polymers derived from lignin-based Pickering emulsions. J. Sep. Sci. 2019, 42, 3563–3570. [Google Scholar] [CrossRef]
- Díaz-Álvarez, M.; Martín-Esteban, A. Molecularly imprinted polymer-quantum dot materials in optical sensors: An overview of their synthesis and applications. Biosensors 2021, 11, 79. [Google Scholar] [CrossRef]
- Wang, Z.; Zhou, C.; Wu, S.; Sun, C. Ion-imprinted polymer modified with carbon quantum dots as a highly sensitive copper(II) ion probe. Polymers 2021, 13, 1376. [Google Scholar] [CrossRef]
- Kazemifard, N.; Ensafi, A.A.; Rezaei, B. Green synthesized carbon dots embedded in silica molecularly imprinted polymers, characterization and application as a rapid and selective fluorimetric sensor for determination of thiabendazole in juices. Food Chem. 2020, 310, 125812. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ding, L.; Chen, L.; Chen, Y.; Zhou, T.; Li, H.; Xu, Y.; Zhao, L.; Huang, N. A facile, green synthesis of biomass carbon dots coupled with molecularly imprinted polymers for highly selective detection of oxytetracycline. J. Ind. Eng. Chem. 2019, 69, 455–463. [Google Scholar] [CrossRef]
- Moreira, B.; Calvet, S.; Marques, A.; Martins, G.V. Chitosan-based molecularly imprinted polymers for point-of-care C-reactive protein detection. Talanta 2026, 296, 128465. [Google Scholar] [CrossRef]
- Xiao, W.; Wang, L.; Wei, X.; Li, J. Chitosan-based molecularly imprinted photoelectric sensor with ZnO/Bi2O3/Bi2S3 sensing layer for thiamethoxam determination. Microchim. Acta 2022, 189, 247. [Google Scholar] [CrossRef]
- Zouaoui, F.; Bourouina-Bacha, S.; Bourouina, M.; Jaffrezic-Renault, N.; Zine, N.; Errachid, A. Electrochemical sensors based on molecularly imprinted chitosan: A review. TrAC Trends Anal. Chem. 2020, 130, 115982. [Google Scholar] [CrossRef]
- Yao, X.; Zhang, S.; Wei, N.; Qian, L.; Coseri, S. Cellulose-based conductive hydrogels for emerging intelligent sensors. Adv. Fiber Mater. 2024, 6, 1256–1305. [Google Scholar] [CrossRef]
- Yang, L.; Yuan, Q.-Y.; Lou, C.-W.; Li, T.-T.; Lin, J.-H. Modified nanocellulose hydrogels and applications in sensing fields. Gels 2025, 11, 140. [Google Scholar] [CrossRef]
- El-Husseini, D.M.; Elmasry, D.M.A.; Hatab, E.M.A.; Kassem, S. Development of a paper-based fluorescent carbon quantum dots MIPs sensor for selective detection of lumpy skin disease virus. RSC Adv. 2024, 14, 27438–27448. [Google Scholar] [CrossRef]
- Kamal, A.; Hong, S.; Ju, H. Carbon quantum dots: Synthesis, characteristics, and quenching as biocompatible fluorescent probes. Biosensors 2025, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- van Wissen, G.; Lowdon, J.W.; Cleij, T.J.; Eersels, K.; van Grinsven, B. Porogenic solvents in molecularly imprinted polymer synthesis: A comprehensive review of current practices and emerging trends. Polymers 2025, 17, 1057. [Google Scholar] [CrossRef] [PubMed]
- Sellergren, B. Molecularly Imprinted Polymers: Man-Made Mimics of Antibodies and Their Application in Analytical Chemistry; Elsevier: Amsterdam, The Netherlands, 2000. [Google Scholar]
- Mosbach, K.; Ramström, O. The emerging technique of molecular imprinting and its future impact on biotechnology. Nat. Biotechnol. 1996, 14, 163–170. [Google Scholar] [CrossRef]
- Pan, G.; Ma, Y.; Zhang, Y.; Guo, X.; Li, C.; Zhang, H. Controlled synthesis of water-compatible molecularly imprinted polymer microspheres with ultrathin hydrophilic polymer shells via surface-initiated reversible addition-fragmentation chain transfer polymerization. Soft Matter 2011, 7, 8428–8439. [Google Scholar] [CrossRef]
- Surapong, N.; Santaladchaiyakit, Y.; Burakham, R. A water-compatible magnetic dual-template molecularly imprinted polymer fabricated from a ternary biobased deep eutectic solvent for the selective enrichment of organophosphorus in fruits and vegetables. Food Chem. 2022, 384, 132475. [Google Scholar] [CrossRef]
- Costa, M.; Di Masi, S.; Garcia-Cruz, A.; Piletsky, S.A.; Malitesta, C. Disposable electrochemical sensor based on ion imprinted polymeric receptor for Cd(II) ion monitoring in waters. Sens. Actuators B Chem. 2023, 383, 133559. [Google Scholar] [CrossRef]
- Smolinska-Kempisty, K.; Cowen, T.; Duda, J.; Bryjak, M. Environmentally friendly molecularly imprinted polymers as an insert for SPE type columns in the gentamicin monitoring process. Talanta 2025, 282, 126966. [Google Scholar] [CrossRef]
- Furtado, A.I.; Lobato, D.; Bonifácio, V.D.; Viveiros, R.; Casimiro, T. Sustainable development of dual–templating molecularly imprinted polymers for biopurification. Mater. Today Chem. 2025, 44, 102586. [Google Scholar] [CrossRef]
- Santana, A.P.; Mora-Vargas, J.A.; Guimarães, T.G.; Amaral, C.D.; Oliveira, A.; Gonzalez, M.H. Sustainable synthesis of natural deep eutectic solvents (NADES) by different methods. J. Mol. Liq. 2019, 293, 111452. [Google Scholar] [CrossRef]
- Porfireva, A.; Goida, A.; Evtugyn, V.; Evtugyn, G. Impedimetric sensor based on molecularly imprinted polythionine from deep eutectic solvent for epinephrine determination. Green Anal. Chem. 2024, 9, 100113. [Google Scholar] [CrossRef]
- Wang, M.; Zhou, L.; Qiao, F.; Yan, H. Facile and green synthesis of a hydrophilic imprinted resin in a deep eutectic solvent–water medium for the specific molecular recognition of tumor biomarkers in complex biological matrices. Green Chem. 2024, 26, 2000–2010. [Google Scholar] [CrossRef]
- González-Laredo, R.F.; Sayago-Monreal, V.I.; Moreno-Jiménez, M.R.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Landeros-Macías, L.F.; Rosales-Castro, M. Natural deep eutectic solvents (NaDES) as an emerging technology for the valorisation of natural products and agro-food residues: A review. Int. J. Food Sci. Technol. 2023, 58, 6660–6673. [Google Scholar] [CrossRef]
- Camilli, E.; Bertana, V.; Frascella, F.; Cocuzza, M.; Marasso, S.L.; Roppolo, I. Digital light processing 3D printing of molecularly imprinted polymers for antibiotic removal. React. Funct. Polym. 2025, 208, 106164. [Google Scholar] [CrossRef]
- Yıldız, Ü.Y.; Hussain, C.G.; Keçili, R.; Hussain, C.M. Green approaches for the preparation of molecularly imprinted polymers. In Green Imprinted Materials; Elsevier: Amsterdam, The Netherlands, 2024; pp. 69–94. [Google Scholar]
- Saidi, K.; Elfadil, D.; Amine, A. Towards the Mass Production of Molecularly Imprinted Polymers via Cost-Effective Photopolymerization Synthesis and Colorimetric Detection via Smartphone. Chemosensors 2024, 12, 232. [Google Scholar] [CrossRef]
- Chen, H.; Son, S.; Zhang, F.; Yan, J.; Li, Y.; Ding, H.; Ding, L. Rapid preparation of molecularly imprinted polymers by microwave-assisted emulsion polymerization for the extraction of florfenicol in milk. J. Chromatogr. B 2015, 983–984, 32–38. [Google Scholar] [CrossRef]
- Astuti, E.J.; Ibrahim, S.; Zulfikar, M.A.; Damayanti, S. Preparation and evaluation of enoxaparin-imprinted polymer via precipitation polymerization using microwave irradiation and conventional stirring methods. React. Funct. Polym. 2025, 216, 106444. [Google Scholar] [CrossRef]
- Teixeira, S.P.B.; Reis, R.L.; Peppas, N.A.; Gomes, M.E.; Domingues, R.M.A. Epitope-imprinted polymers: Design principles of synthetic binding partners for natural biomacromolecules. Sci. Adv. 2021, 7, abi9884. [Google Scholar] [CrossRef]
- Erdőssy, J.; Kassa, E.; Farkas, A.; Horváth, V. Enzymatic digestion as a tool for removing proteinaceous templates from molecularly imprinted polymers. Anal. Methods 2017, 9, 4496–4503. [Google Scholar] [CrossRef]
- Akgönüllü, S.; Kılıç, S.; Esen, C.; Denizli, A. Molecularly imprinted polymer-based sensors for protein detection. Polymers 2023, 15, 629. [Google Scholar] [CrossRef]
- Pasquardini, L.; Bossi, A.M. Molecularly imprinted polymers by epitope imprinting: A journey from molecular interactions to the available bioinformatics resources to scout for epitope templates. Anal. Bioanal. Chem. 2021, 413, 6101–6115. [Google Scholar] [CrossRef]
- Canfarotta, F.; Poma, A.; Guerreiro, A.; Piletsky, S. Solid-phase synthesis of molecularly imprinted nanoparticles. Nat. Protoc. 2016, 11, 443–455. [Google Scholar] [CrossRef]
- Meng, F.; Duan, M.; Wu, W.; Shao, S.; Qin, Y.; Zhang, M. Enzymatic construction Au NPs-rGO based MIP electrochemical sensor for adulteration detection of bovine-derived allergen in camel milk. Food Chem. 2024, 436, 137638. [Google Scholar] [CrossRef] [PubMed]
- Heissel, S.; Frederiksen, S.J.; Bunkenborg, J.; Højrup, P. Enhanced trypsin on a budget: Stabilization, purification and high-temperature application of inexpensive commercial trypsin for proteomics applications. PLoS ONE 2019, 14, e0218374. [Google Scholar] [CrossRef] [PubMed]
- Lamaoui, A.; Mani, V.; Durmus, C.; Salama, K.N.; Amine, A. Molecularly imprinted polymers: A closer look at the template removal and analyte binding. Biosens. Bioelectron. 2024, 243, 115774. [Google Scholar] [CrossRef] [PubMed]
- Martín-Esteban, A. Green molecularly imprinted polymers for sustainable sample preparation. J. Sep. Sci. 2022, 45, 233–245. [Google Scholar] [CrossRef]
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.; Zijp, M.; Hollander, A.; van Zelm, R. ReCiPe2016: A harmonised life cycle impact assessment method at midpoint and endpoint level. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]
- Rebelo, P.; Seguro, I.; Surra, E.; Paíga, P.; Pacheco, J.G.; Delerue-Matos, C. Analysis of atorvastatin in environmental waters: Validation of an electrochemical molecularly imprinted polymer sensor with application of life cycle assessment. Sci. Total Environ. 2024, 921, 171169. [Google Scholar] [CrossRef]
- Goedkoop, M.; Heijungs, R.; Huijbregts, M.; De Schryver, A.; Struijs, J.; Van Zelm, R. ReCiPE 2008: A Life Cycle Impact as-Sessment Method Which Comprises Harmonised Category Indicators at the Midpoint and the Endpoint Level; First Edition Report I: Characterisation; Dutch Ministry of Housing, Spatial Planning and Environment (VROM): Den Haag, The Netherlands, 2009; Volume 1, pp. 1–126. [Google Scholar]
- Altintas, Z.; Guerreiro, A.; Piletsky, S.A.; Tothill, I.E. NanoMIP based optical sensor for pharmaceuticals monitoring. Sens. Actuators B Chem. 2015, 213, 305–313. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Wojnowski, W.; Tobiszewski, M. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar] [CrossRef]
- Wojnowski, W.; Tobiszewski, M.; Pena-Pereira, F.; Psillakis, E. AGREEprep—Analytical greenness metric for sample preparation. TrAC Trends Anal. Chem. 2022, 149, 116553. [Google Scholar] [CrossRef]
- Njoku, D.I.; Dai, W.; Chen, J.; Gao, Y.; Sun, H.; Mao, G.; Peng, Y.-K.; Chen, J.L. Fluorescence-Enabled Portable Droplet Microfluidic Paper-based Analytical Device for Rapid and Sensitive Biotoxicity Assay. J. Environ. Chem. Eng. 2025, 13, 116784. [Google Scholar] [CrossRef]
- Feng, H.; Xu, J.; Xu, J.; Zou, H. Green β-cyclodextrin-based molecularly imprinted polymer for selective adsorption of sulfa-methoxazole. Environ. Res. 2025, 287, 123135. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chen, K.; Tian, M.; Yang, X.; Gong, J.; Liang, Q.; Xie, X.; Li, C.; Zhao, C. A boronate-affinity thermoresponsive molecularly imprinted resin as a green solid-phase extraction filler for highly selective adsorption of psoralen. Chem. Eng. J. 2025, 517, 164219. [Google Scholar] [CrossRef]
- Feliciano, C.d.R.; Marques, T.; Coelho, Y.L.; da Silva, L.H.M.; Gorup, L.F.; de Moura, A.R.; Virtuoso, L.S.; Santos, M.G. A novel fluorescent and magnetic molecularly imprinted sensor for the determination of glyphosate in environmental and potable water samples. Chem. Eng. J. 2025, 507, 160340. [Google Scholar] [CrossRef]
- Purwaningsih, D.R.; Patikorn, C.; Hasan, F.; Karuwan, C.; Kraiya, C.; Ozkan, S.A.; Alahmad, W. A green, portable, and highly selective MIP-based graphene electrochemical sensor for rapid screening and detection of ethylene glycol contamination in medicine products. Microchem. J. 2025, 216, 114723. [Google Scholar] [CrossRef]
- Isa, A.; Banevičiūtė, E.; Piskin, E.; Cetinkaya, A.; Atici, E.B.; Ramanaviciene, A.; Ozkan, S.A. Design of an electrochemical sensor based on molecularly imprinted polymers for sensitive and selective detection of the JAK inhibitor baricitinib. J. Pharm. Biomed. Anal. 2025, 267, 117154. [Google Scholar] [CrossRef]
- Abdulsalam, M.A.A.; Karcioglu, N.; Cetinkaya, A.; Caglayan, M.G.; Ozkan, S.A. A highly selective and sensitive detection of scopolamine using a functionalized gold nanoclusters-assisted molecularly imprinted polymer-based electrochemical sensor. Microchem. J. 2025, 209, 112773. [Google Scholar] [CrossRef]
- Li, C.; Gong, J.; Tian, M.; Ge, H.; Xie, X.; Liang, Q.; Yang, Y.; Zhao, C.; Liu, Y. A green and sustainable synthesis of polydopamine molecularly imprinted resin for selective recognition emodin in Rumex acetosa L.: Experiments and simulation. Food Biosci. 2025, 68, 106674. [Google Scholar] [CrossRef]
- Samanci, S.N.; Kaya, S.I.; Unal, M.A.; Ozkan, S.A. Eco-friendly nanoscale recognition: A MIP-based electrochemical sensor for selective determination of abacavir in pharmaceutical and serum samples. Anal. Bioanal. Chem. 2025, 1–17. [Google Scholar] [CrossRef]
- Birari, K.A.; Patil, P.O.; Taleuzzaman, M.; Alam, S.; Wahab, S.; Khalid, M.; Khan, Z.G. Design and optimization of a novel fluorescent molecularly imprinted polymer for selective niclosamide sensing in real samples: Greenness assessment of MIP synthesis using AGREEMIP. Microchim. Acta 2025, 192, 242. [Google Scholar] [CrossRef]
- de Paula, D.D.M.; da Silva, R.C.S.; Dinali, L.A.F.; Silva, C.F.; de Oliveira, M.A.L.; Pereira, A.C.; Borges, K.B. Magnetic solid phase extraction based on restricted access molecularly imprinted polymer with dually coated for enantioselective determina-tion of tramadol in human plasma by capillary electrophoresis with ultraviolet detection. Talanta 2025, 288, 127676. [Google Scholar] [CrossRef]
- Sun, B.; Wang, X.; Sun, Y.; Qin, C.; Da, X.; Zhou, M.; He, H.; Kang, J.; Wu, Y.; Li, H.; et al. A green molecularly imprinted electrochemical sensor strategy: Toward rapid detection of Polygonatum sibiricum polysaccharides. Microchem. J. 2025, 216, 114834. [Google Scholar] [CrossRef]
- Chansud, N.; Bunkoed, O. A magnetic molecularly imprinted polymer enhanced by incorporating nitrogen-doped graphene quantum dots and mesoporous silica for the selective extraction of triazine herbicides. Talanta 2025, 297, 128748. [Google Scholar] [CrossRef]
- Mokbel, S.A.; Abdel-Halim, M.; Dinc, M.; Mizaikoff, B.; El Gohary, N.A. Development of a magnetic molecularly imprinted polymer for the removal of dexamethasone from river nile water. Sci. Rep. 2025, 15, 45661. [Google Scholar] [CrossRef]
- Manousi, N.; Wojnowski, W.; Płotka-Wasylka, J.; Samanidou, V. Blue applicability grade index (BAGI) and software: A new tool for the evaluation of method practicality. Green Chem. 2023, 25, 7598–7604. [Google Scholar] [CrossRef]
- Shehabeldeen, M.T.; Mansour, F.R.; El-Malla, S.F.; Abdella, A.A. Automated MIP-based In-tip Dispersive Micro-solid Phase Extraction for Determination of Ketoprofen in Environmental Water: A Proof of Concept and Performance Evaluation. Talanta Open 2025, 12, 100522. [Google Scholar] [CrossRef]
- van Zelm, R.; Hennequin, T.; Huijbregts, M.A.J. Performing life cycle impact assessment with the midpoint and endpoint method ReCiPe. Nat. Protoc. 2025, 20, 3400–3411. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.