Abstract
Passive protection is widely assumed to preserve biodiversity and ecological integrity, yet the evidence for long-term vegetation stability in protected temperate forests remains inconclusive. We resurveyed two deciduous forests in SW Poland after 30 years of strict protection to assess temporal changes in their understory vegetation, functional structure, and habitat conditions. Using paired phytosociological relevés (n = 40), collected using the Braun-Blanquet method, we compared baseline (1989–1991) and recent (2022) data with respect to species frequency, Ellenberg indicator values, basic functional traits, and functional diversity. Species composition proved highly stable: only 10% of vascular plant species exhibited significant changes in frequency in particular layers, largely reflecting the vertical redistribution of woody species rather than species turnover. Habitat conditions showed no significant temporal changes. In contrast, the functional structure of the herb layer changed markedly, with significant increases in community-weighted means of seed mass, plant height, and specific leaf area, accompanied by a significant rise in functional diversity. These shifts were partly driven by the increasing abundance of woody species and some opportunistic and invasive species. Our results demonstrate that functional traits may reveal directional ecological changes in passively protected forests even when species composition and habitat indicators remain unchanged, highlighting the importance of trait-based approaches for long-term forest surveys.
1. Introduction
The European Union (EU) biodiversity strategy for 2030 sets ambitious targets aimed at halting biodiversity loss, including the improvement of forest extent and quality and the protection of one third of the EU’s terrestrial area, out of which 10% should be placed under strict protection [1]. One of the key components of this strategy is the conservation of primary and old-growth forests [2], which currently occupy only 3.7 million ha across 32 European countries (excluding Russia) [3,4]. In many regions, such forests are rare and highly fragmented; in Poland, only 21.5 thousand ha of primary forests have been identified so far [4]. Consequently, both forests with high levels of naturalness and primary forests are expected to receive top conservation priority under the EU biodiversity strategy for 2030 [2].
Strict protection is widely regarded as an effective approach for maintaining high biodiversity levels and preserving natural forest dynamics. However, currently forests are increasingly exposed to a range of interacting threats which operate at both local and global scales [5]. Among these, rapid climate change represents a key risk factor, manifested by increasing mean global and regional temperatures [6], extreme weather events such as prolonged droughts [7] and late spring frosts [8,9], as well as enhanced evapotranspiration [10]. These pressures coincide with other large-scale drivers, including the eutrophication of forest ecosystems [11,12,13], the spread of alien plant species [14], and the introduction of novel pathogens affecting forest-forming species [15,16,17]. Together, these factors are reshaping forest species composition and functional structure at a pace unprecedented in recent natural history.
Accumulating evidence indicates that strict protection alone is often insufficient to prevent adverse structural changes in forest plant communities [18,19,20] and associated fauna [21,22]. Numerous studies report substantial transformation in forest ecosystems located within protected areas, including sites subjected to long-term strict protection, largely driven by invasive species [18,23,24,25]. As a result, the presumed role of such forests as a possible refuge for native flora [21,26,27] may, in some cases, prove illusory.
These negative trends may partly result from the limited coverage of biodiversity-rich sites by protected areas and from shortcomings in their ecological representativeness and management effectiveness [28]. Therefore, expanding the total area of protected forests with high natural value remains a critical prerequisite for long-term biodiversity conservation [28,29,30]. Equally important is the ability to quantify the rate and direction of changes occurring within forest ecosystems under strict protection [31,32] where regular forest management interventions are absent. Such assessments rely on long-term vegetation surveys, commonly based on permanent plots—the oldest of which in Europe were established as early as 1911 [33]—or on semi-permanent plots that also offer high comparative value [34,35]. Although the number of studies using resurveyed vegetation plots has grown steadily, exceeding 1300 publications by 2022 [33], only a small proportion of this body of work focuses on European forest communities.
Large-scale analyses based on extensive databases often obscure such dynamics by averaging contrasting local trends. Declines observed in some sites are frequently offset by increases elsewhere, leading to conclusions of negligible net change. For instance, Padulles et al. [36] documented a decline in the proportion of species from the Orchidaceae, Ericaceae, and Fabaceae families, which was compensated by an increase in the proportion of Rosaceae, Amaranthaceae, and Cyperaceae, which resulted in an increase in phylogenetic diversity. Moreover, species losses and gains showed no significant phylogenetic signal in response to macroclimatic change and nitrogen deposition. At broader spatial scales, forest species diversity has been reported as remaining close to zero on average [12], despite the replacement of narrow-range species by widespread taxa [37], or as exhibiting only minor net temporal changes in taxonomic, functional, and phylogenetic biodiversity across European temperate forests [38].
While this supraregional perspective reveals interesting and important patterns of slow, gradual change, it provides limited insight into dynamics at the local scale, i.e., within individual forest communities, protected areas, or smaller geographical regions. In certain locations, changes may proceed rapidly, potentially acting as early indicators for other plant communities developing in similar habitat conditions but not yet affected. In others, forest ecosystems may show symptoms of gradual change or exhibit remarkable stability over time, underscoring their high conservation value under passive protection. Addressing these contrasting trajectories requires detailed regional studies, supported by high-quality archival vegetation data.
In this study, we aimed to determine statistically significant changes in the species composition of the Carpino-Fagetea forest communities in two protected areas similar in terms of origin, geographical location, and vegetation structure. Both areas were designated as nature reserves between 1962 and 1980. Following a resurvey approach based on archival plot relocation [34,35], we established new plots as close as possible to the original sampling locations enabling paired comparisons between baseline and resurvey data.
Rather than relying solely on species richness comparisons between the baseline and resurvey data, which are highly sensitive to the random presence of sporadic species, and prone to observer and relocation errors [35], we focused on changes in species composition sensu stricto. Analyses were performed for the entire vegetation (including trees and shrubs) as well as for the herb layer alone, and were complemented by assessment of functional traits. We focused on the minimal yet informative leaf–height–seed (LHS) trait spectrum [39], which captures key ecological strategies of vascular plant species [40]. We also compared functional diversity between baseline and recent datasets [41].
Between 2012 and 2025, the terms “forest *” AND “resurvey” co-occurred in only 159 publications indexed in the Web of Science, highlighting a persistent shortage of data needed to understand temperate forest dynamics under the combined influence of climate change, eutrophication, and biological invasions. Forest communities may respond to passive protection in multiple ways, ranging from long-term stability, through gradual and evolutionary shifts, to rapid transformation which ultimately lead to a change in ecosystem type.
Because the studied forests have been subject to passive protection, we assume that they have been largely driven by natural forest dynamics. Under such conditions, changes in species composition are expected to be minimal and primarily limited to local fluctuations in the abundance of individual species. Nevertheless, in light of global trends such as species invasions, climate warming, and eutrophication, combined with the small area and isolation of the studied reserves from other ancient forests, we hypothesize that significant changes have occurred in species composition, the functional structure of herbaceous vegetation, and overall functional diversity over the past several decades. The main aim of this study was therefore to assess changes occurring in the key components of the structure of a forest ecosystem under passive protection in the context of rapidly changing climate and increasing nitrogen deposition in the 21st century, and to evaluate the effectiveness of passive forest protection.
2. Materials and Methods
2.1. Study Sites
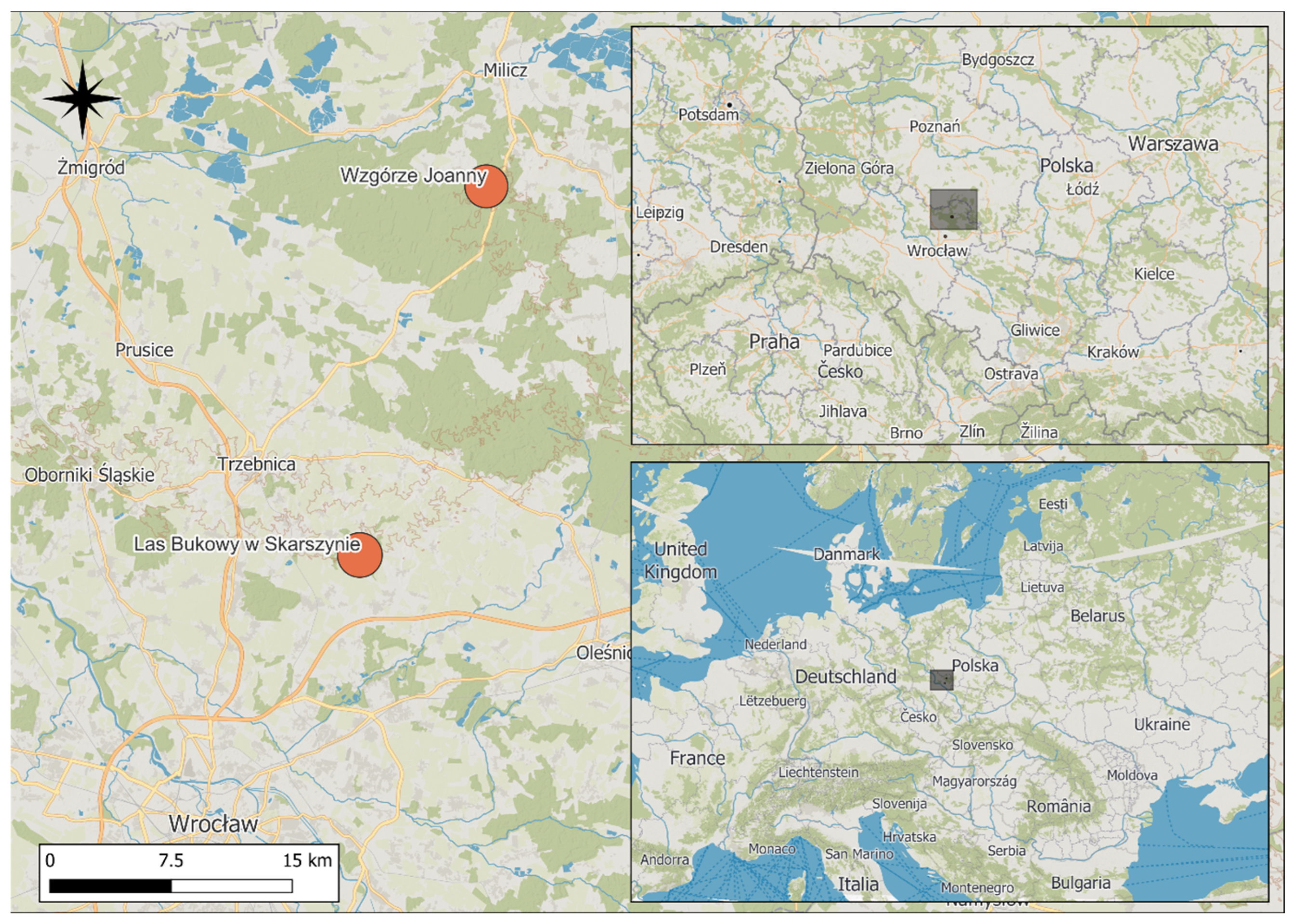
The “Las Bukowy w Skarszynie” nature reserve (23.68 ha) was established in 1980 near the village of Skarszyn (mean latitude 51.46750, mean longitude 17.25901) in the Trzebnica Hills mesoregion [42] to preserve the fragment of a near-natural beech forest with interesting and rare herbaceous species (Figure 1). The reserve is situated within Tertiary moraine formations shaped during the Warta Stage glaciation (Marine Isotope Stage 6, MIS 6), and is characterized by pronounced geomorphological heterogeneity. This is particularly evident in the northern part of the reserve, where a valley carved by the Mielnica stream is surrounded by moraine hills with steep slopes. In contrast, the relief of southern part is less diversified, with lower and gently undulating hills. The varied topography reflects the features typical of loess cover formed during the periglacial period. The reserve spans at an altitudinal range of 165–185 m a.s.l.
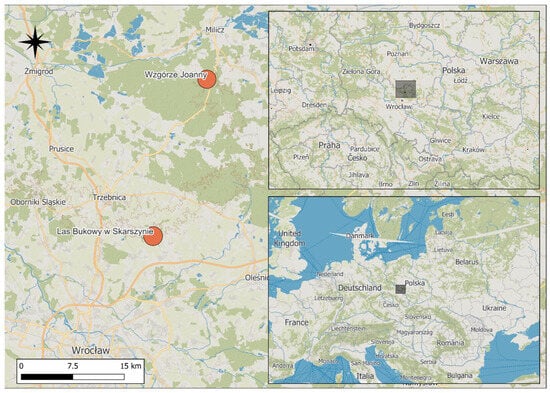
Figure 1.
Location of the study areas in SW Poland. Gray squares indicate the spatial extent of the study region; the investigated nature reserves are marked with orange circles, and forested areas are shown in green. Background from the ©MapTiler ©OpenStreetMap contributors.
Luvisols predominate, having developed from loess deposits and clayey silts overlaying sandy substrates. Almost the entire area of nature reserve is covered by acidic lowland beech forests (Deschampsio flexuosae-Fagetum Schröder 1938) and nutrient-rich oak-hornbeam forests (Stellario holosteae-Carpinetum betuli Oberdorfer 1957). The tree layer is dominated by Fagus sylvatica, with admixtures of Quercus robur and Q. petraea, Tilia cordata, Acer platanoides, and Carpinus betulus. A total of 205 species of vascular plants have been recorded within the reserve [43].
The “Wzgórze Joanny” nature reserve is located approximately 1 km southeast of the village of Postolin (mean latitude 51.25819, mean longitude 17.15377), in the Trzebnica Hills mesoregion [42]. Established in 1962, the reserve was designated for scientific and educational purposes to protect an isolated beech forest at the eastern margin of its distribution range (Figure 1). The reserve covers an area of 24.57 ha. Its landscape structure was shaped during the MIS 6, and is characterized by moraine slopes with northwestern and western exposure and a relative elevation difference of up to 30 m. Two streams flow through the reserve and merge in its northern part, forming a small marsh area.
Brown acidic soils dominate, covering approximately 93% of the reserve area. These soils developed from shallow clayey sands overlying loose glacial sands, as well as from clayey silts, medium clays, and silty clays deposited on terminal moraine tills. Almost the entire area of the reserve is occupied by nutrient-rich beech forests (Galio odorati-Fagetum Sougnez and Thill 1959) and small fragments of oak-hornbeam forests (Stellario-Carpinetum betuli). The tree layer is dominated by Fagus sylvatica, with admixtures of Quercus petraea and Carpinus betulus. A total of 236 species of vascular plants has been recorded within the reserve [44].
2.2. Baseline Material and Resurvey Sampling
The locations of the study plots were determined based on two publications describing forest communities of the “Las Bukowy w Skarszynie” reserve [43] and the “Wzgórze Joanny” reserve [44]. Using detailed descriptions provided in the archival sources and the information on tree species composition, we attempted to relocate as precisely as possible the forest patches surveyed in 1989–1991 (with a few plots originally sampled in 1975–1976). At these locations, new phytosociological relevés with a standardized area of 225 m2 were collected. The full set of location data is presented in Supplementary Material Table S1.
The resurvey comprised 18 relevés in the “Las Bukowy w Skarszynie” reserve and 22 relevés in the “Wzgórze Joanny” reserve. For consistency with the historical datasets, all vascular plant species were recorded in each plot, and their cover was estimated using exactly the same old Braun-Blanquet cover abundance scale [45] as applied in the baseline survey. Species were assigned to three vertical layers: tree layer (>7 m), shrub layer (including sapling; 1–7 m), and herb and seedling layer (<1 m).
Although some degree of plot relocation uncertainty is inherent in resurvey studies [46,47], we assume that the relocated plots adequately capture long-term changes in vegetation composition. They will accurately reflect changes that have occurred over time. This approach has been validated using extensive datasets from Central Europe and has demonstrated high effectiveness in detecting temporal changes in forest communities [35,48]. To minimize seasonal bias [49,50], resampling was conducted during months comparable to those of the baseline surveys.
All vegetation data are stored in EU-PL-006 Laura, a resurvey database of forest vegetation in southwestern Poland [51], and are also a part of forestREplot initiative https://forestreplot.ugent.be (accessed on 20 January 2026).
Resurvey fieldwork in both nature reserves was carried out with the permission of the Regional Directorate for Environmental Protection in Wrocław (decision no. WPN.6205.40.2022.AR).
2.3. Nomenclature
The nomenclature of vascular plant species is consistent with the Euro+Med PlantBase database [52]. The nomenclature of higher-rank syntaxa is consistent with the work of Mucina et al. [53], while the nomenclature of syntaxa at the community level is consistent with the studies referring to the current classification of oak-hornbeam [54] and beech [55] forests in Central Europe.
2.4. Data Analysis
2.4.1. General Patterns in Vegetation Composition
To identify species exhibiting statistically significant changes in frequency of occurrence over time, we used the JUICE software version 7.1.27 https://www.sci.muni.cz/botany/juice/NET/index.htm (accessed on 12 January 2026) [56]. The dataset was divided into two groups, i.e., baseline and resurvey, without further subdivision into individual forest communities due to the limited sample size (n = 40). For each group, species with high fidelity (φ > 0.25) [57,58,59] and a significance level of p < 0.01 in Fisher’s exact test [60] were determined separately for each vegetation layer.
Differences in ecological conditions and floristic composition were tested using multivariate analysis, including both unconstrained (detrended correspondence analysis, DCA; principal coordinates analysis, PCoA) and constrained analysis (partially canonical correspondence analysis, CCA; redundancy analysis, RDA). Species composition data exhibited a gradient length of 3.5 standard deviation (SD) units, justifying the use of unimodal ordination methods. Species abundance data were obtained from data tables prepared in JUICE software and transformed into percentage cover values. All multivariate analyses were conducted using the Canoco 5.0 software [61].
DCA [62] was applied to explore the distribution of major groups of vascular plant species and to illustrate the diversity of forest communities in relation to differences in ecological conditions inferred from the Ellenberg indicator values for Europe (EIVEs). Prior to analysis, species data were logarithmically transformed, detrended by segments, and rare species were downweighed.
PCoA [63] was used to assess changes in the species composition of the herb layer within individual plot pairs (baseline vs. resurvey). This method describes compositional changes based solely on species combinations, without explicitly incorporating environmental variables. Squared Bray–Curtis distance was applied and species scores were projected as supplementary variables.
CCA [64] was employed to test whether temporal grouping (baseline vs. resurvey) significantly influenced variation in species composition. In this analysis, time was used as explanatory (dependent) variable, allowing us to quantify the proportion of variance in species composition attributable to temporal change while accounting for shared variability among plots.
RDA [65] was finally used to describe and visualize the influence of changes in community-weighted means of functional traits on the increase in functional diversity, expressed as Rao’s quadratic entropy (FDQ), as well as the distribution of the investigated plots along gradients defined by these community-weighted means across surveys.
2.4.2. Habitat Conditions
Ecological characteristics of the studied forest communities in the baseline and resurvey periods were expressed by mean EIVEs [66]. Indicator values were calculated as unweighted by species cover [67,68]. Only species from the herb layer were included in this analysis, in accordance with the ecological scope and intended application of EIVEs (Table S1). The statistical significance of differences in ecological conditions between the baseline and resurvey was tested using the nonparametric Wilcoxon signed-rank test. All analyses were performed in PAST 5 [69].
2.4.3. Plant Functional Traits
We analyzed a set of plant functional traits widely recognized as main components of plant life-strategy [39] and commonly classified as response traits [70]. The selected traits included the following: specific leaf area (SLA, mm2·mg−1), calculated as the ratio of leaf area to its dry mass; leaf area (LA, mm2), expressed as the one-sided or projected area of an individual leaf; plant height (Height, cm), defined as plant vegetative height at maturity; leaf dry matter content (LDMC, mg g−1), calculated as the ratio of leaf dry mass to fresh mass; and seed mass (SM, mg), defined as oven-dry mass of an average seed produced by a plant of given species.
High SLA values are generally associated with rapid growth and higher photosynthetic capacity and are characteristic of short-lived species that allocate relatively little biomass to structural tissues and storage organs [71]. Such species tend to display greater plasticity in their response to spatial heterogeneity in light and soil resources [39]. In contrast, species exhibiting low SLA are typically long-lived, slow-growing, and characterized by substantial investment in structural and storage tissues, which enhances their resistance to drought and herbivory [71]. Consequently, an increase in community-level SLA may indicate intensified competitive interactions for resources, while simultaneously reflecting a reduction in environmental stress [72].
LA is associated with light interception and microclimatic conditions, and is generally higher in species adapted to shaded and humid environments. However, environmental stressors—including heat, cold, drought, or nutrient limitation—typically select for smaller leaves [73]. Plant height is closely linked to competitive ability and reflects trade-offs between strategies of avoidance and tolerance under varying environmental conditions, particularly in relation to light and nutrient supply [39,72,74]. Leaves with higher LDMC tend to decompose more slowly, are mechanically tougher, and are assumed to confer increased resistance to environmental disturbances such as drought, herbivory, and physical damage [73]. SM represents a trade-off between dispersal ability and successful establishment [39] and generally increases with habitat shadiness and vegetation height and decreases in more frequently disturbed environments [75].
Trait data were obtained from the LEDA database [76], the Seed Information Database [77], and from the FloraVeg database [78]. Mean species-level trait values were used and intraspecific trait variation was not considered [79,80]. Woody species (trees, shrubs) were excluded from this part of the data analysis. Consequently, only herbaceous plants were assigned trait values. Trait attributes were available for 149 species for SM and Height, 140 species for SLA, 130 species for LDMC, and for 66 species for LA (Table S2). Community-weighted mean (CWM) values were calculated as mean of trait values present in the community-weighted by the relative abundance of taxa bearing each value [81].
Because functional trait comparisons were conducted for the same study sites and largely involved overlapping species pools, species lacking complete trait information were retained in functional diversity analyses. This approach does not violate methodological assumptions [82] and allows for a more comprehensive representation of community-level functional structure.
Functional diversity was calculated using Rao’s quadratic entropy (FDQ), an index that integrates species relative abundances with pairwise functional differences among species [41]. FDQ captures the extent to which dominant species differ in their trait values; consequently, higher FDQ values may indicate a greater degree of niche differentiation [72,83,84]. Importantly, FDQ can be calculated for multiple traits simultaneously and is independent of species richness [41].
3. Results
3.1. Changes in Species Composition
The whole dataset, comprising both baseline and resurvey records, included a total of 177 vascular plants species (trees and shrubs included). Only 18 species (ca. 10%) showed differences in fidelity between the baseline and resurvey periods. In most cases, these differences were restricted to specific vegetation layers rather than reflecting changes across the entire vertical structure of the forest communities (Table 1).

Table 1.
Differences in species composition across individual vegetation layers of forest communities in the studied reserves between the baseline (1989–1991) and resurvey (2022) periods. Species are ordered according to their diagnostic value in a given survey. The first value denotes species frequency, while the superscript value indicates fidelity. Only species with statistically significant fidelity values (p < 0.01; Fisher’s exact test) are shown. The φ coefficient values are multiplied by 100. Vegetation layers: T—tree layer, Sh—shrub layer, Juv—juveniles (>1 m height), H—herb layer.
Baseline plots were characterized by a higher frequency and fidelity of several species typical of nutrient-rich deciduous forests. In contrast, plots resurveyed in 2022 showed an increased contribution of shrubs and understory tree species that had previously occurred mainly as seedlings and juveniles in the understory during the 1989–1991 surveys (e.g., Acer pseudoplatanus, Quercus robur, Carpinus betulus, Corylus avellana, Sambucus nigra, Quercus petraea). Notably, Acer platanoides exhibited a significantly higher frequency and fidelity in both the tree and shrub layers and was the only tree species showing this pattern consistently across both nature reserves. The resurveyed plots were also characterized by an increased occurrence of two invasive alien species, Robinia pseudoacacia and Impatiens parviflora. Apart from these changes, the species composition in both the tree and understory layers remained stable, and observed fluctuations in abundance were not significant.
When species occurrence was analyzed after merging vegetation layers [85], the results at the significance level of p < 0.01 differed slightly from those obtained in layer-specific analyses (Table 2). While the set of species with the highest fidelity in the baseline survey remained unchanged, Tilia cordata emerged as the species with the highest fidelity in the resurvey data. It co-occurred with four species identified in layer-specific analyses (Table 1) as diagnostic for the resurvey period (Sambucus nigra, Hedera helix, Robinia pseudoacacia, and Geranium robertianum).

Table 2.
Differences in species composition of forest communities in the studied reserves between the baseline (1989–1991) and resurvey (2022) periods after merging vegetation layers. Species are ordered according to their diagnostic value in a given survey. The first value denotes species frequency, while the superscript value indicates fidelity. Only species with statistically significant fidelity values (p < 0.01; Fisher’s exact test) are shown. The φ coefficient values are multiplied by 100.
This pattern indicates that tree and shrub species such as Acer platanoides, A. pseudoplatanus, Carpinus betulus, or Corylus avellana must have occurred frequently at least in the lower shrub and herb layers during the baseline survey, as their overall contribution to the communities—measured as the cumulative frequency across vegetation layers—did not change significantly over time.
All analyzed species were present in both survey periods. Therefore, observed differences in frequency may partly reflect stochastic variation or minor inaccuracies in plot relocation.
Over the 30-year period of stand development, only minor changes in the species composition of the studied communities were observed. As in the baseline survey, the upper tree layer is dominated by Fagus sylvatica, Quercus petraea, and Carpinus betulus, accompanied by Tilia cordata, Alnus glutinosa, Pinus sylvestris, Fraxinus excelsior, Acer campestre, and A. pseudoplatanus. The only species that increased its contribution to the upper stand layers during this period was Acer platanoides.
3.2. Vegetation Patterns and Temporal Stability
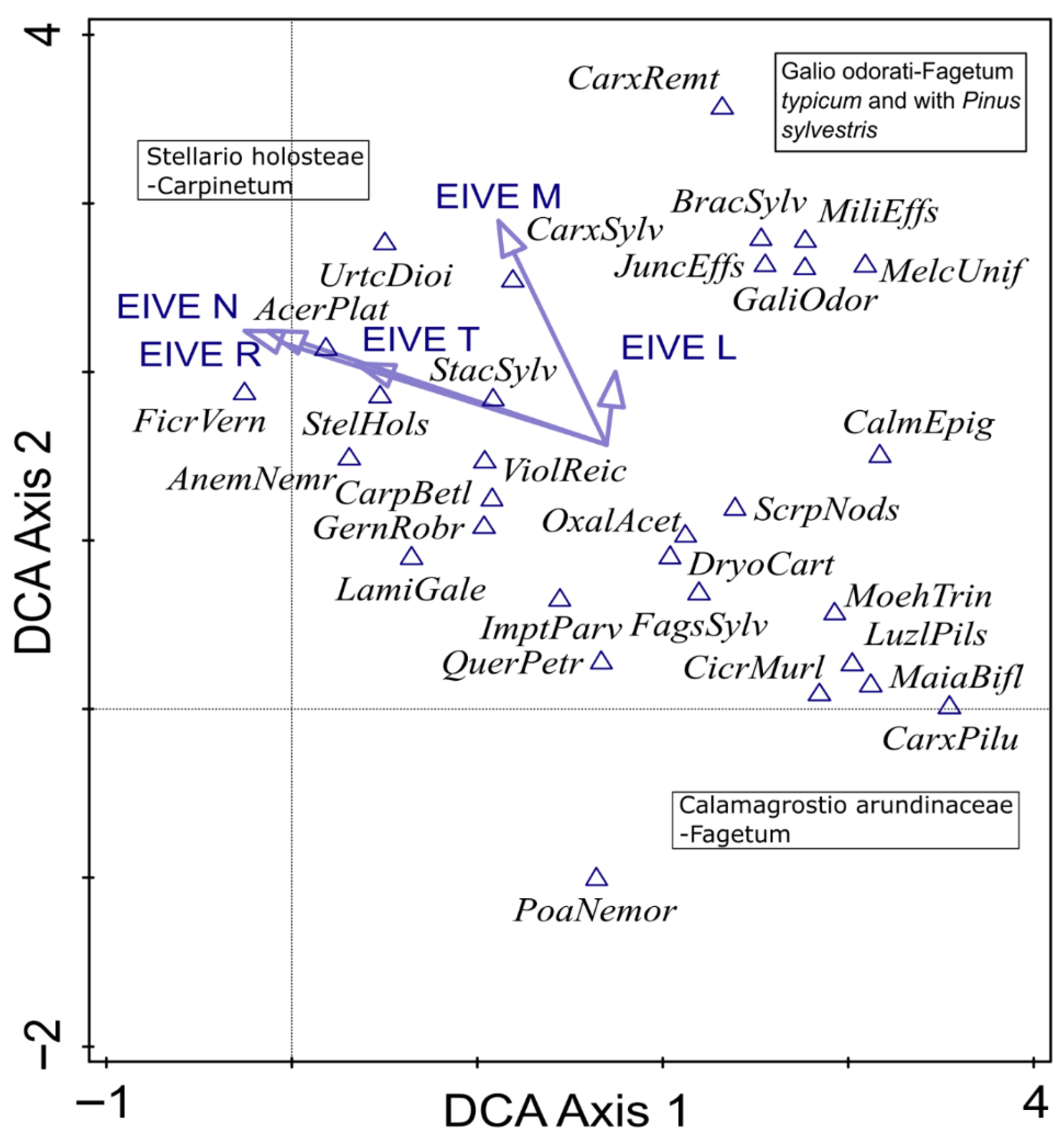
DCA revealed internal heterogeneity within the dataset along the first two ordination axes (Figure 2). The first axis appeared to be associated with light availability, which likely resulted from differences in the canopy structure and the dominance of the main tree species—from strongly shaded stands formed by Fagus sylvatica to more open oak-hornbeam forests. The second axis primarily reflected a gradient of soil nutrient availability, ranging from nutrient-poor communities corresponding to mesophilous beech forests (ass. Calamagrostio arundinaceae-Fagetum) to more nutrient-rich oak-hornbeam (ass. Stellario holosteae-Carpinetum) and beech forests (ass. Galio odorati-Fagetum). This internal differentiation is important for interpreting temporal changes observed between the baseline and resurvey periods.
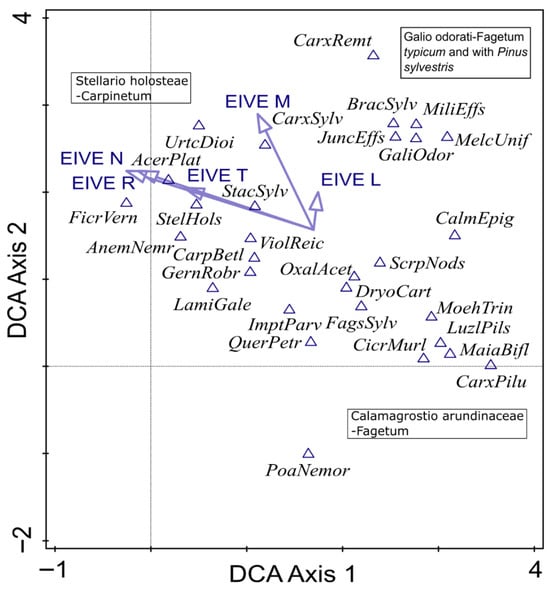
Figure 2.
Detrended correspondence analysis (DCA) diagram of the 30 most frequent understory plant species recorded in protected deciduous forests in the eastern part of the Trzebnica Hills (SW Poland). Gradient length is 3.17 SD units, and the adjusted explained variation is 18.73%. Species co-occurrence patterns reflect the differentiation of the analyzed forests into three distinct community types, indicated in the diagram. The vectors of Ellenberg indicator values for Europe (EIVEs) variables were passively projected into a diagram. Explanation: AcerPlat—Acer platanoides, AnemNemr—Anemone nemorosa, BracSylv—Brachypodium sylvaticum, CalmEpig—Calamagrostis epigejos, CarpBetl—Carpinus betulus, CarxPilu—Carex pilulifera, CarxSylv—Carex sylvatica, CicrMurl—Cicerbita muralis, DryoCart—Dryopteris carthusiana, FagsSylv—Fagus sylvatica, FicrVern—Ficaria verna, GaliOdor—Galium odoratum, GernRobr—Geranium robertianum, ImptParv—Impatiens parviflora, JuncEffs—Juncus effusus, LamiGale—Lamium galeobdolon, LuzlPils—Luzula pilosa, MajaBifl—Majanthemum bifolium, MelcUnif—Melica uniflora, MiliEffs—Millium effusum, OxalAcet—Oxalis acetosella, PoaNemor—Poa nemoralis, QuerPetr—Quercus petraea, ScrpNods—Scrophularia nodosa, StacSylv—Stachys sylvatica, StelHols—Stellaria holostea, UrtcDioi—Urtica dioica, ViolReic—Viola reichenbachiana.
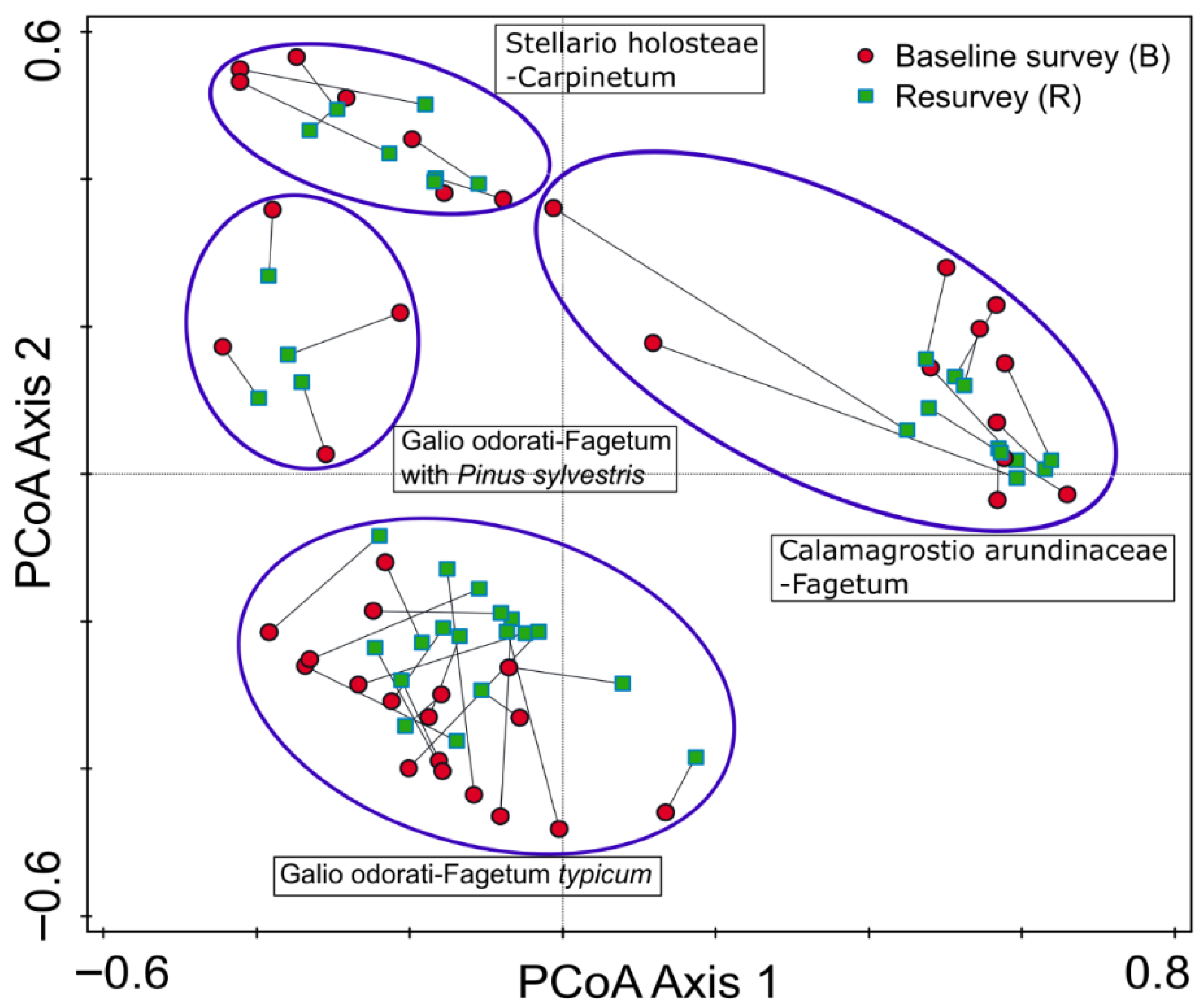
The results of the PCoA indicated that plots surveyed in 1989–1990 and resurveyed in 2022 largely overlap in ordination space (Figure 3). No consistent shifts in the position of individual plots within forest community types were detected. A noticeable change was observed in only two plots representing Calamagrostio arundinaceae-Fagetum. However, this may reflect plot relocation uncertainty rather than true compositional change.
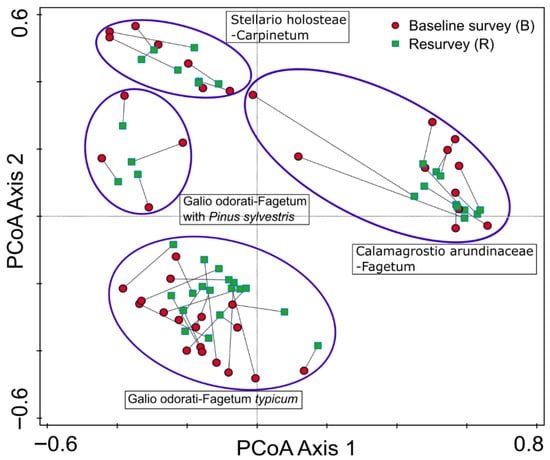
Figure 3.
Principal coordinates analysis (PCoA) ordination diagram for 80 vegetation plots in protected, near to natural deciduous forests in the eastern part of the Trzebnica Hills (SW Poland), surveyed in 1989–1991 and resurveyed in 2022. The diagram illustrates herb layer species composition. Corresponding plot pairs (baseline–resurvey) are connected by lines, and groups of plots with similar species composition and representing different forest community types are enclosed by ellipses.
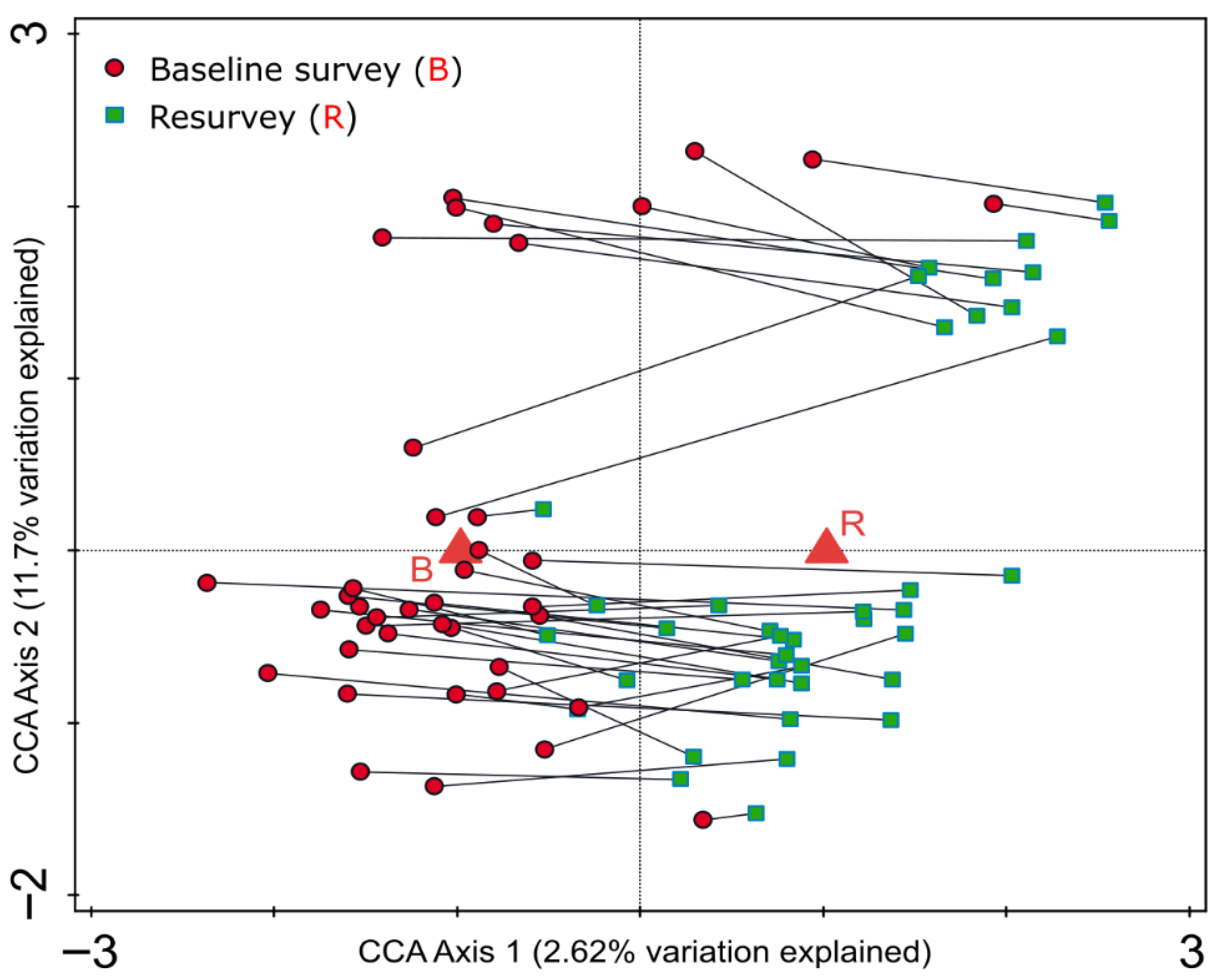
CCA revealed the presence of weak but directional changes in the species composition of the herb layer between the baseline and resurvey periods (Figure 4). Nevertheless, the temporal variable explained only 2.62% (1.38% adjusted) of the total variance in species composition, indicating that temporal changes were minor relative to overall compositional variability.
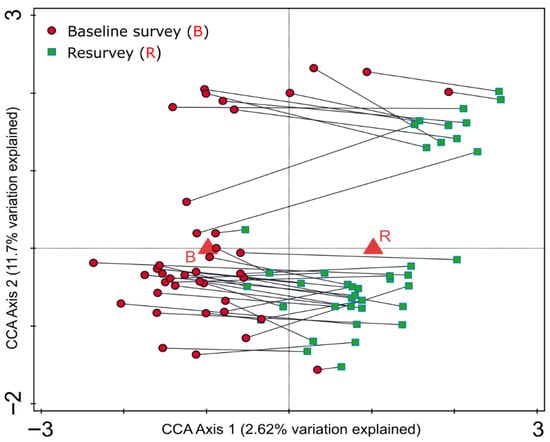
Figure 4.
Canonical correspondence analysis (CCA) ordination diagram for 80 vegetation plots in protected, near to natural deciduous forests in the eastern part of the Trzebnica Hills (SW Poland), surveyed in 1989–1991 and resurveyed in 2022. The diagram illustrates changes in herb layer species composition over time. The diagram shows the herbaceous vegetation pattern within plots during this period. Corresponding plot pairs (baseline–resurvey) are connected by lines. The explanatory temporal variable (B—baseline vs. R—resurvey) accounts for 2.62% (adjusted explained variation is 1.38%) of the total variation.
3.3. Changes in Ecological Indicator Values
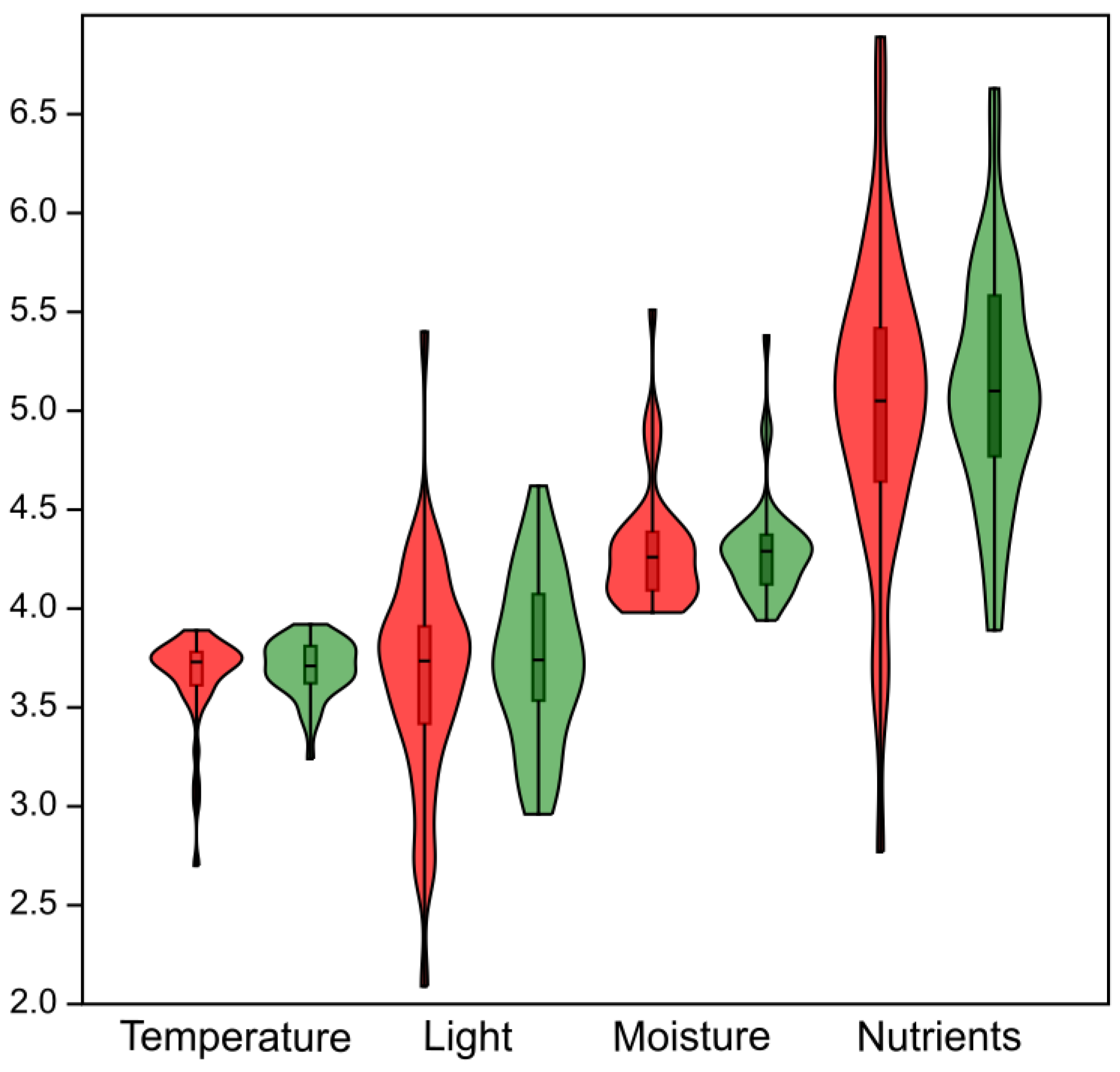
Results of the Wilcoxon signed-rank test indicate that no statistically significant differences in habitat conditions occurred between the baseline and resurvey periods (Figure 5). Specifically, no significant changes were detected for EIVEs related to temperature (p = 0.45), light (p = 0.7), humidity (p = 0.67), and nutrient availability (p = 0.53).
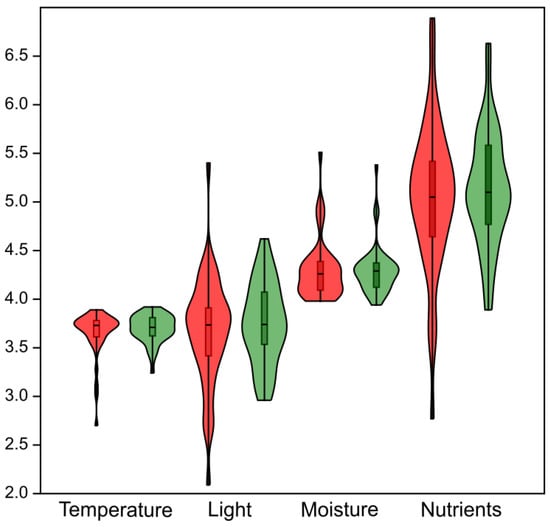
Figure 5.
Violin plots summarizing changes in ecological conditions of protected deciduous forests in the Trzebnica Hills (SW Poland) expressed using Ellenberg indicator values for Europe (EIVEs). All analyzed indicators, calculated from the herb layer species composition, show no statistically significant differences between the baseline and resurvey periods. Red—baseline survey; green—resurvey.
3.4. Changes in Functional Structure
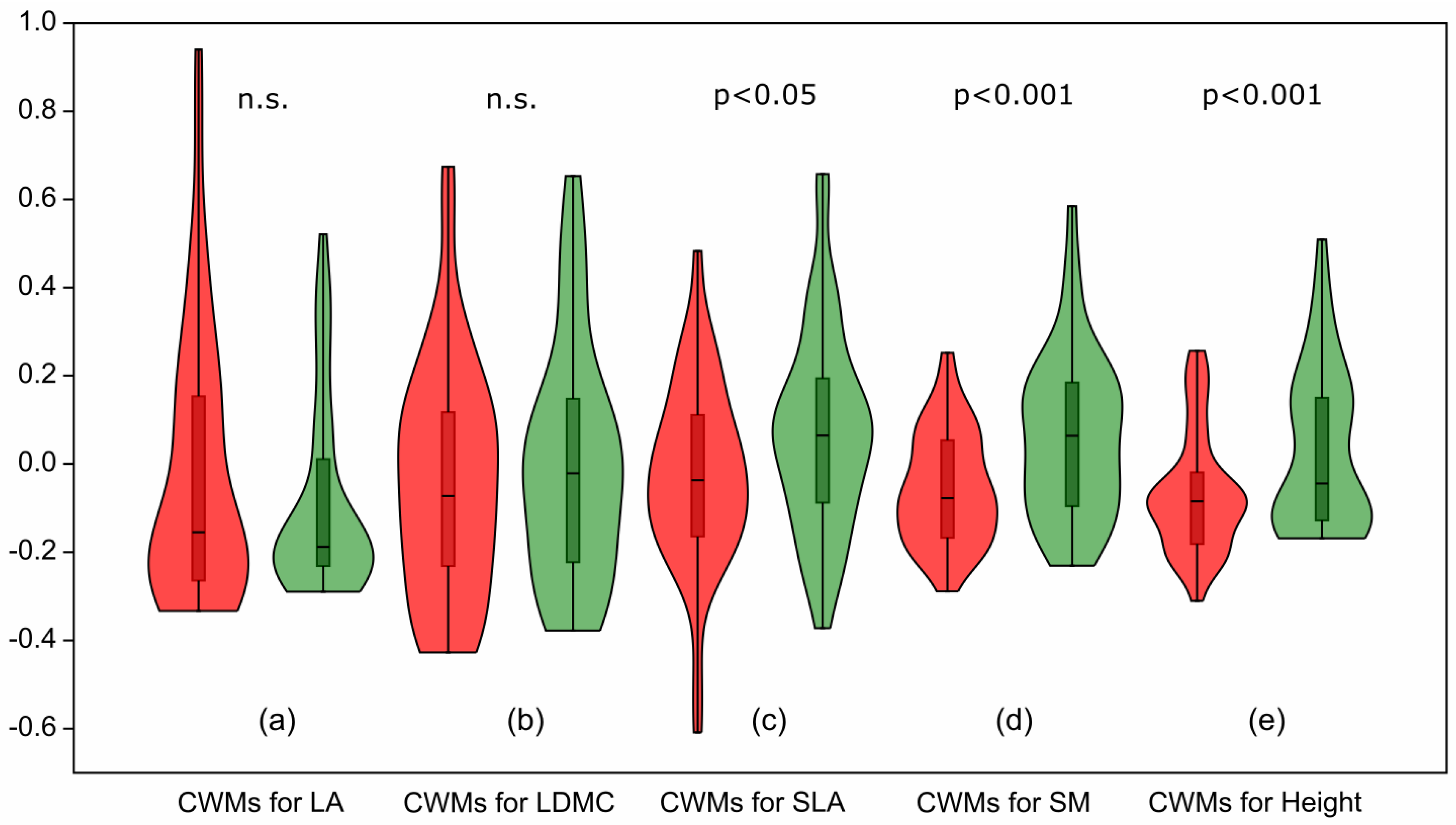
Analysis of the CWMs of functional traits revealed that some traits in the herb layer remained unchanged between the baseline and resurvey periods. No statistically significant differences were found for LA (p = 0.69; Figure 6a) and LDMC (p = 0.53; Figure 6b).
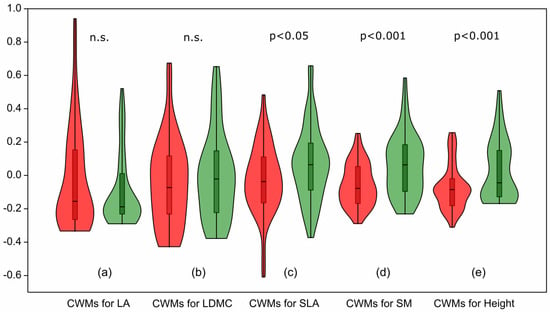
Figure 6.
Violin plots illustrating changes in community-weighted means (CWMs) of functional traits of herb layer species between the baseline and resurvey periods in deciduous forests of the Trzebnica Hills (SW Poland). Leaf area (LA) and leaf dry matter content (LDMC) show no significant differences, whereas specific leaf area (SLA), seed mass (SM), and plant height (Height) differ significantly between survey periods. Red—baseline survey, green—resurvey.
In contrast, three other functional traits showed statistically significant changes. The CWM for SLA slightly increased with marginal statistical significance (p = 0.042; Figure 6c). Much stronger and highly significant changes (p < 0.001) were observed for CWMs for SM (Figure 6d) and Height (Figure 6e).
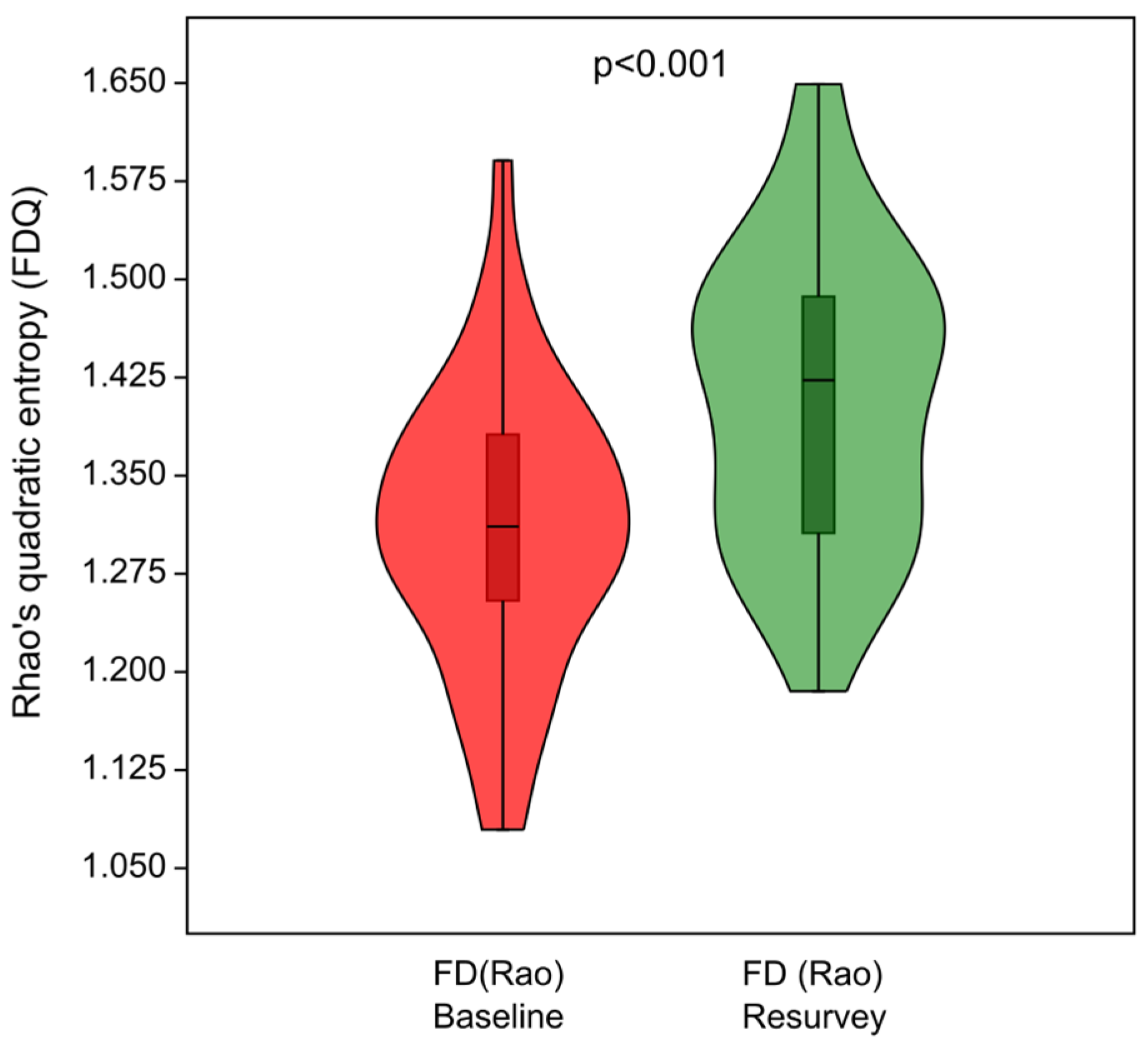
The functional diversity of the herb layer, expressed as Rao’s quadratic entropy (FDQ), increased over time (Figure 7), and the differences in FDQ values between the baseline and resurvey were highly significant (p < 0.001). The observed increase in the functional diversity of the herb layer was associated with the increase in CWMs for SLA, SM, and plant height (Figure 6 and Figure 8a). Figure 8b demonstrates the distribution of selected plots resurveyed in 2022 along axes representing changes in functional traits, particularly SM, corresponding to the direction of change in FDQ values.
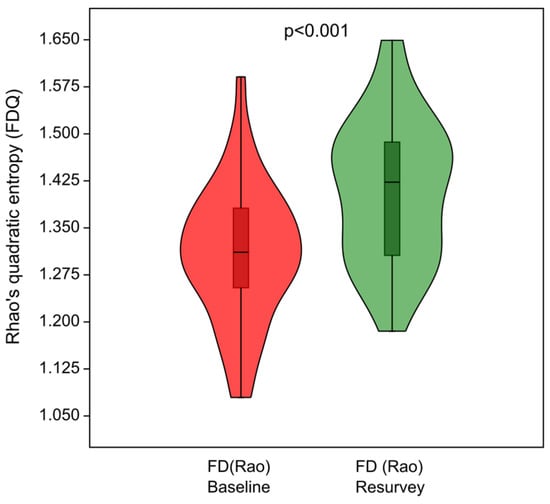
Figure 7.
Violin plots summarizing changes in functional diversity of the herb layer expressed as Rao’s quadratic entropy (FDQ) between the baseline and resurvey studies in the deciduous, protected forests of the Trzebnica Hills (SW Poland).
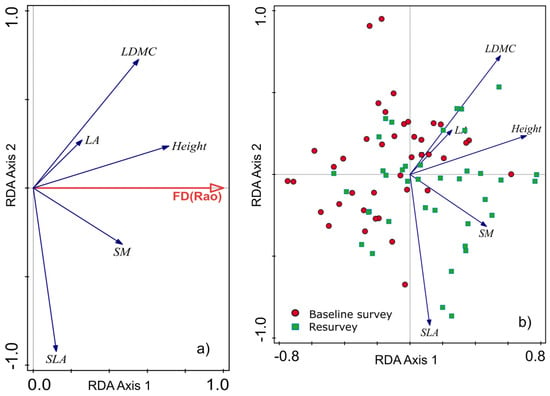
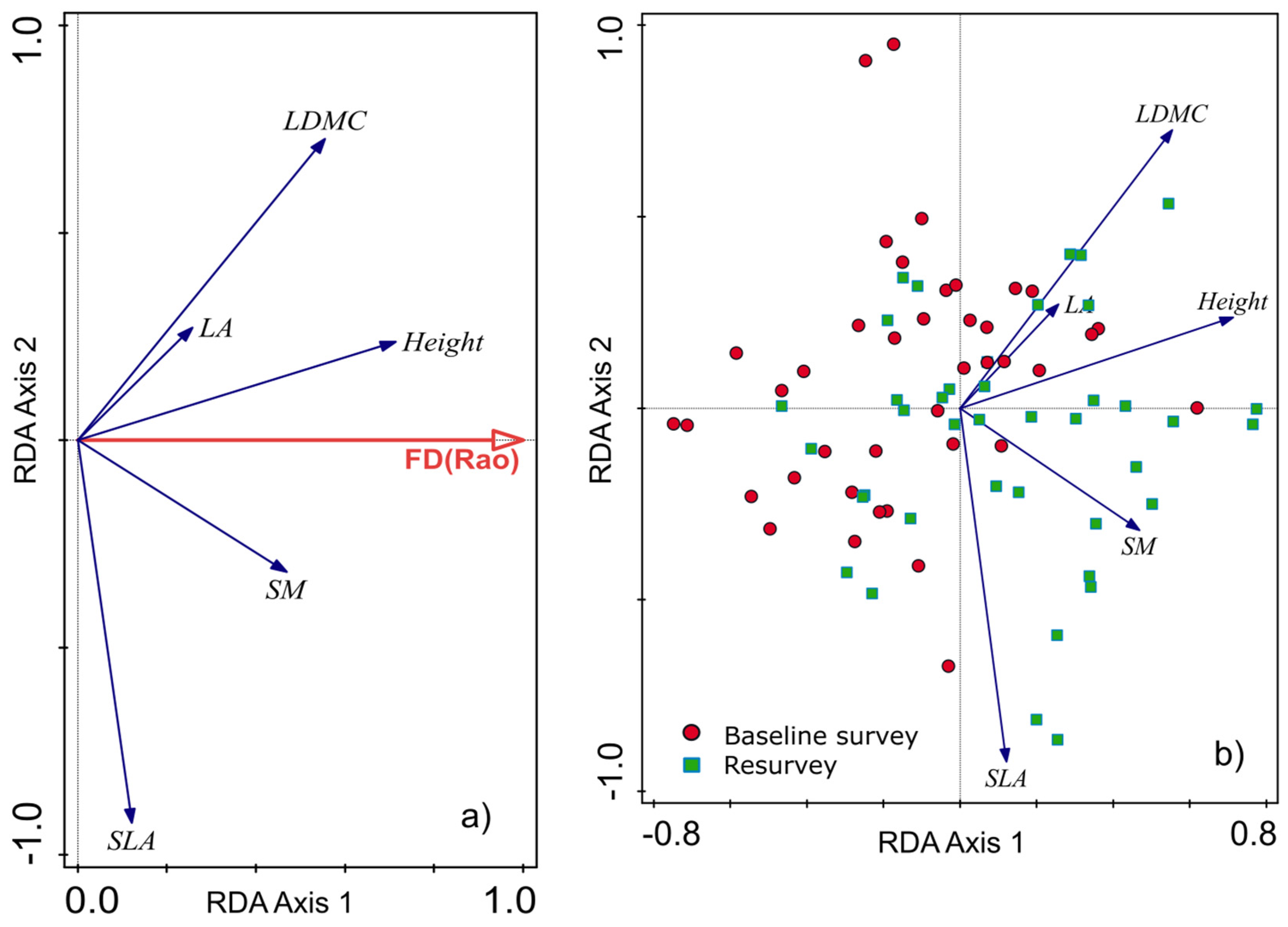
Figure 8.
Redundancy analysis (RDA) diagrams demonstrating the relationship between changes in community-weighted means (CWMs) of functional plant traits and functional diversity expressed as Rao’s quadratic entropy (FDQ). (a) Diagram shows the influence of changes in CWMs of plant traits on FDQ. (b) Diagram presents the distribution of investigated plots in relation to CWMs within the baseline and resurvey surveys conducted in two protected forests in the Trzebnica Hills (SW Poland).
The results obtained in this study revealed parallel trends whose direction may appear contradictory at first glance. Statistically significant changes were detected in several functional traits (seed mass, plant height, and—to a lesser extent—specific leaf area), suggesting the functional reorganization of the studied forest communities over the three decades separating the baseline and resurvey periods. At the same time, compositional changes concern only a small fraction of the recorded vascular plants (10 out of 177 species). The observed shifts mainly reflect the transition of some woody species from the herb layer into shrub and tree layers (Table 1 and Table 2), while the majority of plots retained species combinations typical of their respective phytosociological units (Figure 3). This is further supported by the CCA results, which show that temporal differences explain only 2.6% of the total variation in species composition (Figure 4). Moreover, habitat conditions expressed by the Ellenberg indicator values (EIVEs) remained statistically unchanged, indicating a high stability of habitat conditions over time.
4. Discussion
This study was designed to test whether long-term passive protection effectively stabilizes forest understory vegetation, or whether global change drivers such as climate warming, nitrogen deposition, and biological invasions override this stabilizing effect.
We hypothesized that, despite the absence of direct management, significant changes would have occurred over the past three decades in both the species composition and functional structure of the herb layer of the studied forest communities. Our results provide a nuanced picture: while species composition and ecological indicator values remained remarkably stable, substantial and directional changes were observed in the functional traits and functional diversity, indicating ongoing ecosystem reorganization under seemingly stable environmental conditions.
4.1. Stability of Species Composition in the Context of Long-Term Studies in European Forests
Many recent studies conducted across Europe report pronounced changes in forests under passive protection, affecting both species composition and functional traits. For example, Kermavnar and Kutnar [86] documented marked shifts in overstory composition (i.e., a decrease and an increase in frequency of Quercus robur and Carpinus betulus, respectively), high species turnover (61%), a significant decline in species richness, and progressing taxonomic homogenization in Slovenian oak-hornbeam forests. Moreover, the analysis of EIVEs suggested forest community reorganization driven by increasing shade and cooler conditions promoting perennial herbs which tolerate closed canopies. Similarly, Lanta et al. [13], in a 60-year study of riparian forests in Czechia, observed declines in species richness and phylogenetic diversity associated with the loss of light-demanding species and the spread of tall, woody generalists characterized by conservative strategies, including high LDMC and heavier seeds. Comparable patterns of species turnover (64%), accompanied by a 24% decline in herb layer cover, were reported by Kotrík et al. [87] in Quercus-dominated forests of the Western Carpathians, which the authors attributed to increasing eutrophication and canopy closure. A decline in herb layer diversity associated with stand development and progressive shading has likewise been documented in beech forests in Germany [88,89]. Pronounced changes, mainly related to water availability and climate change, were also observed in forests of the Drawieński National Park in Poland, affecting both the functional structure and habitat conditions; however, fertile broadleaved forest communities proved to be the most stable [90]. Even stronger transformations of the herb layer vegetation have been reported in former coppice and coppice-with-standards forests, where the abandonment of coppicing has led to a rapid loss of light-demanding species following canopy closure [89,91,92,93,94,95,96,97,98,99,100]. Holeštová et al. [101] go so far as to argue that even the natural riparian forests examined by the authors should be managed using traditional, low-intensity agroforestry practices, due to the ongoing decline in highly specialized forest species.
On the one hand, many of the available studies identify the increasing canopy closure and associated light limitation as the primary drivers of declining herb layer diversity in forests under passive protection, a conclusion supported by experimental studies and supraregional synthesis [13,102,103,104,105]. On the other hand, this general pattern is not universal.
Naaf and Wulf [106], analyzing the functional traits of declining species in northwestern Germany, found no evidence that canopy closure, drainage, or eutrophication were responsible for herb layer changes; instead, loosing species tended to have higher SLA, were sensitive to deer browsing, and showed performance optima at higher soil pH. Likewise, Reczyńska and Świerkosz [107] observed increasing species richness in protected oak coppice forests, while Strubelt et al. [108] reported a rise in mean species richness, particularly in nutrient-poor sites, despite a largely unchanged total species pool—a pattern similar to that presented in this study.
In contrast to most of the above mentioned studies, the forests investigated here did not exhibit strong declines in species richness or clear signals of taxonomic homogenization. This discrepancy likely reflects differences in the initial forest structure, developmental stage, and environmental context at the time when the baseline survey was conducted.
4.2. The Role of Forest Developmental Stage and Initial Conditions
Based on the obtained results, as well as other studies conducted in European forest communities, we suggest that the direction and magnitude of changes in forest understory communities may depend strongly on the developmental stage of the forest at the onset of protection and on the initial site conditions, such as canopy composition and structure [89,109], soil chemistry (pH, nitrogen content) [110,111,112], and water conditions [13,90].
The forests examined here are over 160 years old, meaning that the baseline survey was conducted when stands were already approaching maturity, with closed canopies and few gaps. Light-demanding species such as Lathyrus niger, Hylotelephium maximum, or Campanula persicifolia were already rare at that time and occurred only sporadically. Although some moderately thermophilus species disappeared, others achieved low frequencies during the resurvey (e.g., Fragaria moschata and Prunus spinosa), resulting in no statistically significant change in species composition (Wilcoxon paired signed-rank test, p = 0.22). The lack of change in EIVEs further supports the interpretation that the studied communities were already documented in a phase of relatively stable, closed-canopy forest during the baseline survey.
This contrasts with forests where passive protection was introduced at earlier successional stages or following the abandonment of coppicing, where rapid canopy closure often leads to pronounced declines in herb layer diversity [93,95,97,98]. Our results therefore emphasize the importance of the forest history, initial canopy structure, and site conditions in shaping long-term vegetation patterns under passive protection.
4.3. Functional Reorganization Without Strong Species Turnover
The observed shifts in the CWMs of functional traits most likely reflect ongoing forest development towards a more stabilized, near-natural state. Increases in SM and plant height are typical of ecosystems dominated by conservative life-history strategies, characteristic of mature forests [75]. Similar trends have been documented in other eutrophilous deciduous forests elsewhere, and in many cases they are accompanied by increases in LDMC, often linked to reduced water availability [13,90]. In the present study, these functional changes coincide with an increased contribution of tree and shrub regeneration (Table 1), a phenomenon widely reported in unmanaged forests [13,86,113].
The marginally significant increase in SLA (p < 0.04) suggests a growing contribution of pioneer and nitrophilous species. In the Trzebnica Hills forests, this group includes the invasive Impatiens parviflora as well as native forest species such as Geum urbanum, Chrysosplenium alternifolium, and Geranium robertianum. Similar patterns were reported from riparian forests protected in nature reserves along the Oder River, where increases in functional diversity were largely driven by Impatiens parviflora and associated opportunistic species with high SLA [114]. Notably, Impatiens parviflora and Robinia pseudoacacia were the only neophytes recorded in the studied forests, indicating relatively high resistance to invasion compared to managed forests [27,30,115]. However, the contribution of Robinia pseudoacacia to the observed changes in functional traits cannot be assessed, as this species was excluded from the functional trait analyses due to its tree growth form. Moreover, the increase in the abundance of climbers, particularly Hedera helix, observed in the studied forests aligns with reports of the widespread “lianification” of temperate forests across Europe [116].
4.4. Implications for Passive Forest Protection and Ecosystem Dynamics
In summary, our results call for caution in formulating general rules regarding vegetation dynamics in temperate forests under passive protection. While many studies report declining understory diversity and functional homogenization, our findings demonstrate that alternative trajectories are possible, particularly in mature forests with stable site conditions.
Passive protection does not imply ecological stability. Instead, it allows for gradual, directional changes in the functional structure that may result in new equilibria. In such scenarios, many structural and functional features of near-natural forests are maintained, and functional diversity may increase rather than decline. These changes may involve shifts in traditional species combinations, including greater contributions of nitrophilous and invasive species [100], a process that appears difficult to prevent entirely [14,19,117]; however, it can be substantially slowed down [30,118] and restricted to a limited number of species.
5. Conclusions
Changes in the species composition of the studied forest communities are detectable but proceed very slowly. Over the 30-year study period, their effects are difficult to capture statistically: neither temporal differences in species composition nor habitat conditions explain a substantial proportion of the observed variability. In contrast, statistically significant changes were detected in the functional structure of the herb layer, with increasing contributions of species characterized by heavier seeds, greater height, and higher specific leaf area. These changes were accompanied by a significant increase in functional diversity.
The stand remains dominated by Fagus sylvatica, Quercus petraea, and Carpinus betulus; the only species that has increased its contribution to this layer is Acer platanoides.
The observed patterns suggest two parallel processes: on the one hand, a gradual increase in species with conservative life-history strategies typical of mature forest ecosystems, including woody species and perennial climbers; on the other hand, the establishment of functionally labile, shade-tolerant species with short life cycles, such as Impatiens parviflora and native nitrophilous herbs. It may therefore indicate that, in passively protected deciduous forests, we are witnessing the formation of forest ecosystems that are primarily native in character, yet increasingly incorporate non-forest species (including geographically alien taxa), which may give rise to new—and potentially stable—species assemblages under changing environmental conditions.
The results of our study are directly applicable and may be cautiously extrapolated to similar forest ecosystems, namely temperate beech and oak-hornbeam forests under passive protection. Moreover, they indicate that assessments of vegetation dynamics based solely on species composition may underestimate the ongoing changes in protected forest ecosystems. Therefore, integrating functional trait-based approaches into long-term vegetation surveys is essential for understanding forest ecosystem responses to global drivers and passive protection, which may differ among developmental stages.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/f17020178/s1, Table S1: Basic data of phytosociological relevés used in the study; Table S2: List of species with plant functional traits used in the study.
Author Contributions
Conceptualization, K.R. and K.Ś.; methodology, K.R. and K.Ś.; formal analysis, K.R. and K.Ś.; investigation, S.C., K.R. and K.Ś.; data curation, K.Ś.; writing—original draft preparation, K.R. and K.Ś.; writing—review and editing, K.R., K.Ś. and S.C.; writing—preparation for printing in accordance with the requirements of the publishing house, preparing references, K.R. and K.Ś.; visualization, K.Ś. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data are stored in the forestREplot database and in the EU-PL-006 Laura database [51].
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| DCA | Detrended correspondence analysis |
| EIVEs | Ellenberg indicator values for Europe |
| RDA | Redundancy analysis |
| PCoA | Principal coordinates analysis |
| CCA | Canonical correspondence analysis |
| SM | Seed mass |
| LDMC | Leaf dry matter content |
| SLA | Specific leaf area |
| LA | Leaf area |
| CWMs | Community-weighted means |
| FD | Functional diversity |
References
- European Union. EU Biodiversity Strategy for 2030: Bringing Nature Back into Our Lives; Publications Office of the European Union: Luxembourg, 2021. [Google Scholar]
- European Commission. Criteria and Guidance for Protected Areas Designations; European Commission: Brussels, Belgium, 2022. [Google Scholar]
- Sabatini, F.M.; Burrascano, S.; Keeton, W.S.; Levers, C.; Lindner, M.; Pötzschner, F.; Verkerk, P.J.; Bauhus, J.; Buchwald, E.; Chaskovsky, O.; et al. Where Are Europe’s Last Primary Forests? Divers. Distrib. 2018, 24, 1426–1439. [Google Scholar] [CrossRef]
- Sabatini, F.M.; Bluhm, H.; Kun, Z.; Aksenov, D.; Atauri, J.A.; Buchwald, E.; Burrascano, S.; Cateau, E.; Diku, A.; Duarte, I.M.; et al. European Primary Forest Database v2.0. Sci. Data 2021, 8, 220. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate Change Impacts, Adaptive Capacity, and Vulnerability of European Forest Ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Climate Change: Global Temperature. Available online: https://www.climate.gov/news-features/understanding-climate/climate-change-global-temperature (accessed on 4 December 2025).
- Guastello, P.; Smith, K.H.; Knutson, C.; Svoboda, M. Drought Hotspots Around the World 2023–2025; Tsegai, D., Diallo, B.L., Eds.; United Nations Convention to Combat Desertification: Bonn, Germany, 2025. [Google Scholar]
- Qiu, H.; Yan, Q.; Yang, Y.; Huang, X.; Wang, J.; Luo, J.; Peng, L.; Bai, G.; Zhang, L.; Zhang, R.; et al. Flowering in the Northern Hemisphere Is Delayed by Frost after Leaf-out. Nat. Commun. 2024, 15, 9123. [Google Scholar] [CrossRef]
- Vitasse, Y.; Spafford, L.; Reim, J.; Baumgarten, F.; Martínez-Sancho, E. Emerging Trend of Increasing Spring Frost Damage for Beech at Higher Elevations in the Jura Mountains: Evidence from Tree-Ring Data. New Phytol. 2025, 248, 600–614. [Google Scholar] [CrossRef]
- Swain, D.L.; Prein, A.F.; Abatzoglou, J.T.; Albano, C.M.; Brunner, M.; Diffenbaugh, N.S.; Singh, D.; Skinner, C.B.; Touma, D. Hydroclimate Volatility on a Warming Earth. Nat. Rev. Earth Environ. 2025, 6, 35–50. [Google Scholar] [CrossRef]
- Perring, M.P.; Diekmann, M.; Midolo, G.; Schellenberger Costa, D.; Bernhardt-Römermann, M.; Otto, J.C.J.; Gilliam, F.S.; Hedwall, P.-O.; Nordin, A.; Dirnböck, T.; et al. Understanding Context Dependency in the Response of Forest Understorey Plant Communities to Nitrogen Deposition. Environ. Pollut. 2018, 242, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Staude, I.R.; Pereira, H.M.; Daskalova, G.N.; Bernhardt-Römermann, M.; Diekmann, M.; Pauli, H.; Van Calster, H.; Vellend, M.; Bjorkman, A.D.; Brunet, J.; et al. Directional Turnover towards Larger-Ranged Plants over Time and across Habitats. Ecol. Lett. 2022, 25, 466–482. [Google Scholar] [CrossRef]
- Lanta, V.; Wiatzková, A.; Altman, J.; Bartoš, M.; Černý, T.; Chlumská, Z.; Dvorský, M.; Hauck, D.; Hummel, J.; Karlík, P.; et al. Changes in Plant Diversity of European Lowland Forests: Increased Homogenization and Expansion of Shade-Tolerant Trees. Biol. Conserv. 2024, 296, 110719. [Google Scholar] [CrossRef]
- Wagner, V.; Chytrý, M.; Jiménez-Alfaro, B.; Pergl, J.; Hennekens, S.; Biurrun, I.; Knollová, I.; Berg, C.; Vassilev, K.; Rodwell, J.S.; et al. Alien Plant Invasions in European Woodlands. Divers. Distrib. 2017, 23, 969–981. [Google Scholar] [CrossRef]
- Oliva, J.; Colinas, C. Decline of Silver Fir (Abies alba Mill.) Stands in the Spanish Pyrenees: Role of Management, Historic Dynamics and Pathogens. For. Ecol. Manag. 2007, 252, 84–97. [Google Scholar] [CrossRef]
- George, J.-P.; Sanders, T.G.M.; Timmermann, V.; Potočić, N.; Lang, M. European-Wide Forest Monitoring Substantiate the Neccessity for a Joint Conservation Strategy to Rescue European Ash Species (Fraxinus spp.). Sci. Rep. 2022, 12, 4764. [Google Scholar] [CrossRef] [PubMed]
- Havrdová, A.; Douda, J.; Boublík, K.; Doudová, J.; Král, P.; Černý, K.; Pecka, Š.; Zahradník, D.; Strnadová, V.; Sucharová, J.; et al. Decadal Decline in Herbaceous Species Richness in Wetland Forests: Effects of an Introduced Pathogen and Environmental Change. For. Ecol. Manag. 2025, 583, 122569. [Google Scholar] [CrossRef]
- Foxcroft, L.C.; Pysek, P.; Richardson, D.M.; Genovesi, P. (Eds.) Plant Invasions in Protected Areas: Patterns, Problems and Challenges, 2013th ed.; Invading Nature-Springer Series in Invasion Ecology; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Lapin, K.; Oettel, J.; Steiner, H.; Langmaier, M.; Sustic, D.; Starlinger, F.; Kindermann, G.; Frank, G. Invasive Alien Plant Species in Unmanaged Forest reserves, Austria. NeoBiota 2019, 48, 71–96. [Google Scholar] [CrossRef]
- Reczyńska, K.; Świerkosz, K. Does Protection Really Matter? A Case Study from Central European Oak Forests. Diversity 2019, 12, 6. [Google Scholar] [CrossRef]
- Geldmann, J.; Barnes, M.; Coad, L.; Craigie, I.D.; Hockings, M.; Burgess, N.D. Effectiveness of Terrestrial Protected Areas in Reducing Habitat Loss and Population Declines. Biol. Conserv. 2013, 161, 230–238. [Google Scholar] [CrossRef]
- Rija, A.A.; Critchlow, R.; Thomas, C.D.; Beale, C.M. Global Extent and Drivers of Mammal Population Declines in Protected Areas under Illegal Hunting Pressure. PLoS ONE 2020, 15, e0227163. [Google Scholar] [CrossRef] [PubMed]
- Asner, G.P.; Hughes, R.F.; Vitousek, P.M.; Knapp, D.E.; Kennedy-Bowdoin, T.; Boardman, J.; Martin, R.E.; Eastwood, M.; Green, R.O. Invasive Plants Transform the Three-Dimensional Structure of Rain Forests. Proc. Natl. Acad. Sci. USA 2008, 105, 4519–4523. [Google Scholar] [CrossRef] [PubMed]
- Pyšek, P.; Hulme, P.E.; Simberloff, D.; Bacher, S.; Blackburn, T.M.; Carlton, J.T.; Dawson, W.; Essl, F.; Foxcroft, L.C.; Genovesi, P.; et al. Scientists’ Warning on Invasive Alien Species. Biol. Rev. Camb. Philos. Soc. 2020, 95, 1511–1534. [Google Scholar] [CrossRef]
- Florens, F.B.V.; Baider, C.; Martin, G.M.N.; Seegoolam, N.B.; Zmanay, Z.; Strasberg, D. Invasive Alien Plants Progress to Dominate Protected and Best-Preserved Wet Forests of an Oceanic Island. J. Nat. Conserv. 2016, 34, 93–100. [Google Scholar] [CrossRef]
- Levine, J.M.; Adler, P.B.; Yelenik, S.G. A Meta-analysis of Biotic Resistance to Exotic Plant Invasions. Ecol. Lett. 2004, 7, 975–989. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C.; González-Moreno, P.; Pergl, J.; Pizarro, M.; Pyšek, P.; Thuiller, W.; Yesson, C.; Vilà, M. Protected Areas Offer Refuge from Invasive Species Spreading under Climate Change. Glob. Change Biol. 2017, 23, 5331–5343. [Google Scholar] [CrossRef]
- Brondízio, E.S.; Settele, J.; Díaz, S.; Ngo, H.T. The Global Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; PBES Secretariat: Bonn, Germany, 2019. [Google Scholar]
- Pysek, P.; Jarosik, V.; Kucera, T. Patterns of Invasion in Temperate Nature Reserves. Biol. Conserv. 2002, 104, 13–24. [Google Scholar] [CrossRef]
- Pyšek, P.; Jarošík, V.; Kučera, T. Inclusion of Native and Alien Species in Temperate Nature Reserves: An Historical Study from Central Europe. Conserv. Biol. 2003, 17, 1414–1424. [Google Scholar] [CrossRef]
- Le Saout, S.; Hoffmann, M.; Shi, Y.; Hughes, A.; Bernard, C.; Brooks, T.M.; Bertzky, B.; Butchart, S.H.M.; Stuart, S.N.; Badman, T.; et al. Protected Areas and Effective Biodiversity Conservation. Science 2013, 342, 803–805. [Google Scholar] [CrossRef]
- Vačkář, D.; Harmáčková, Z.V.; Kaňková, H.; Stupková, K. Human Transformation of Ecosystems: Comparing Protected and Unprotected Areas with Natural Baselines. Ecol. Indic. 2016, 66, 321–328. [Google Scholar] [CrossRef]
- Knollová, I.; Chytrý, M.; Bruelheide, H.; Dullinger, S.; Jandt, U.; Bernhardt-Römermann, M.; Biurrun, I.; de Bello, F.; Glaser, M.; Hennekens, S.; et al. ReSurveyEurope: A Database of Resurveyed Vegetation Plots in Europe. J. Veg. Sci. 2024, 35, e13235. [Google Scholar] [CrossRef]
- Verheyen, K.; De Frenne, P.; Baeten, L.; Waller, D.M.; Hédl, R.; Perring, M.P.; Blondeel, H.; Brunet, J.; Chudomelova, M.; Decocq, G.; et al. Combining Community Resurvey Data to Advance Global Change Research. Bioscience 2017, 67, 73–83. [Google Scholar] [CrossRef]
- Verheyen, K.; Bažány, M.; Chećko, E.; Chudomelová, M.; Closset-Kopp, D.; Czortek, P.; Decocq, G.; De Frenne, P.; De Keersmaeker, L.; Enríquez García, C.; et al. Observer and Relocation Errors Matter in Resurveys of Historical Vegetation Plots. J. Veg. Sci. 2018, 29, 812–823. [Google Scholar] [CrossRef]
- Padullés Cubino, J.; Lenoir, J.; Li, D.; Montaño-Centellas, F.A.; Retana, J.; Baeten, L.; Bernhardt-Römermann, M.; Chudomelová, M.; Closset, D.; Decocq, G.; et al. Evaluating Plant Lineage Losses and Gains in Temperate Forest Understories: A Phylogenetic Perspective on Climate Change and Nitrogen Deposition. New Phytol. 2023, 241, 2287–2299. [Google Scholar] [CrossRef]
- Staude, I.R.; Waller, D.M.; Bernhardt-Römermann, M.; Bjorkman, A.D.; Brunet, J.; De Frenne, P.; Hédl, R.; Jandt, U.; Lenoir, J.; Máliš, F.; et al. Replacements of Small- by Large-Ranged Species Scale up to Diversity Loss in Europe’s Temperate Forest Biome. Nat. Ecol. Evol. 2020, 4, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; De Lombaerde, E.; Vanneste, T.; Sanczuk, P.; Baeten, L.; Lenoir, J.; De Pauw, K.; Verheyen, K.; Bernhardt-Römermann, M.; Becker-Scarpitta, A.; et al. Small Net Local Temporal Changes in Taxonomic, Functional, and Phylogenetic Biodiversity across European Temperate Forests. New Phytol. 2025, 248, 2128–2142. [Google Scholar] [CrossRef] [PubMed]
- Westoby, M. A Leaf-Height-Seed (LHS) Plant Ecology Strategy Scheme. Plant Soil 1998, 199, 213–227. [Google Scholar] [CrossRef]
- Diaz, S.; Hodgson, J.G.; Thompson, K.; Cabido, M.; Cornelissen, J.H.C.; Jalili, A.; Montserrat-Martí, G.; Grime, J.P.; Zarrinkamar, F.; Asri, Y.; et al. The Plant Traits That Drive Ecosystems: Evidence from Three Continents. J. Veg. Sci. 2004, 15, 295–304. [Google Scholar] [CrossRef]
- Botta-Dukát, Z. Rao’s Quadratic Entropy as a Measure of Functional Diversity Based on Multiple Traits. J. Veg. Sci. 2005, 16, 533–540. [Google Scholar] [CrossRef]
- Solon, J.; Borzyszkowski, J.; Bidłasik, M.; Richling, A.; Badora, K.; Balon, J.; Brzezińska-Wójcik, T.; Chabudziński, Ł.; Dobrowolski, R.; Grzegorczyk, I.; et al. Physico-Geographical Mesoregions of Poland: Verification and Adjustment of Boundaries on the Basis of Contemporary Spatial Data. Geogr. Pol. 2018, 91, 143–170. [Google Scholar] [CrossRef]
- Pender, K.; Rybałtowska, Z. Szata Roślinna Rezerwatu “Las Bukowy w Skarszynie”. Acta Univ. Wratislav. Pr. Bot. 1995, 62, 79–100. [Google Scholar]
- Macicka-Pawlik, T.; Wilczyńska, W. Flora i Zbiorowiska Roślinne Rezerwatu “Wzgórze Joanny” Koło Postolina w Świetle Zmian Antropogenicznych. Acta Univ. Wratislav. Pr. Bot. 1995, 62, 251–276. [Google Scholar]
- Mueller-Dombois, D.; Ellenberg, H. Aims and Methods in Vegetation Ecology; The Blackburn Press: Caldwell, NJ, USA, 2002; p. 547. [Google Scholar]
- Hédl, R.; Bernhardt-Römermann, M.; Grytnes, J.-A.; Jurasinski, G.; Ewald, J. Resurvey of Historical Vegetation Plots: A Tool for Understanding Long-Term Dynamics of Plant Communities. Appl. Veg. Sci. 2017, 20, 161–163. [Google Scholar] [CrossRef]
- Douda, J.; Doudová, J.; Holeštová, A.; Chudomelová, M.; Vild, O.; Boublík, K.; Černá, M.; Havrdová, A.; Petřík, P.; Pychová, N.; et al. Historical Sampling Error: A Neglected Factor in Long-Term Biodiversity Change Research. Biol. Conserv. 2023, 286, 110317. [Google Scholar] [CrossRef]
- Kopecký, M.; Macek, M. Vegetation Resurvey Is Robust to Plot Location Uncertainty. Divers. Distrib. 2015, 21, 322–330. [Google Scholar] [CrossRef]
- Hédl, R.; Chudomelová, M. Understanding the Dynamics of Forest Understorey: Combination of Monitoring and Legacy Data Reveals Patterns across Temporal Scales. J. Veg. Sci. 2020, 31, 733–743. [Google Scholar] [CrossRef]
- Fischer, F.M.; Chytrý, K.; Chytrá, H.; Chytrý, M.; Těšitel, J. Seasonal Beta-diversity of Dry Grassland Vegetation: Divergent Peaks of Above-ground Biomass and Species Richness. J. Veg. Sci. 2023, 34, e13182. [Google Scholar] [CrossRef]
- Świerkosz, K.; Wójcicka-Rosińska, A.; Kuras-Hilares, I.; Pech, P.; Reczyńska, K. LAURA: A Resurvey Database of Forest Vegetation in Southwestern Poland. Veg. Classif. Surv. 2025, 6, 131–132. [Google Scholar] [CrossRef]
- Euro+Med PlantBase. Available online: https://europlusmed.org/ (accessed on 14 October 2023).
- Mucina, L.; Bültmann, H.; Dierßen, K.; Theurillat, J.-P.; Raus, T.; Arni, A.Č.; Umberová, K.Š.; Willner, W.; Dengler, J.; Gavilán García, R.; et al. Vegetation of Europe: Hierarchical Floristic Classification System of Vascular Plant, Bryophyte, Lichen, and Algal Communities. Appl. Veg. Sci. 2016, 19, 3–294. [Google Scholar] [CrossRef]
- Novák, P.; Willner, W.; Zukal, D.; Kollár, J.; Roleček, J.; Świerkosz, K.; Ewald, J.; Wohlgemuth, T.; Csiky, J.; Onyshchenko, V.; et al. Oak-Hornbeam Forests of Central Europe: A Formalized Classification and Syntaxonomic Revision. Preslia 2020, 92, 1–34. [Google Scholar] [CrossRef]
- Świerkosz, K.; Reczyńska, K.; Pech, P.; Kuras, I.; Hédl, R. Syntaxonomy and Ecology of Beech Forest Vegetation in Southwestern Poland. Phytocoenologia 2018, 48, 297–320. [Google Scholar] [CrossRef]
- Tichý, L. JUICE, Software for Vegetation Classification. J. Veg. Sci. 2002, 13, 451–453. [Google Scholar] [CrossRef]
- Chytrý, M.; Tichý, L.; Holt, J.; Botta-Dukát, Z. Determination of Diagnostic Species with Statistical Fidelity Measures. J. Veg. Sci. 2002, 13, 79–90. [Google Scholar] [CrossRef]
- Willner, W.; Tichý, L.; Chytrý, M. Effects of Different Fidelity Measures and Contexts on the Determination of Diagnostic Species. J. Veg. Sci. 2009, 20, 130–137. [Google Scholar] [CrossRef]
- Lötter, M.; Mucina, L.; Witkowski, E. The Classification Conundrum: Species Fidelity as Leading Criterion in Search of a Rigorous Method to Classify a Complex Forest Data Set. Community Ecol. 2013, 14, 121–132. [Google Scholar] [CrossRef]
- Fisher, R.A. On the Interpretation of χ2 from Contingency Tables, and the Calculation of P. J. R. Stat. Soc. 1922, 85, 87–94. [Google Scholar] [CrossRef]
- ter Braak, C.; Šmilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination, Version 5.0; Microcomputer Power: Ithaca, NY, USA, 2012. [Google Scholar]
- Hill, M.O.; Gauch, H.G. Detrended Correspondence Analysis: An Improved Ordination Technique. Vegetatio 1980, 42, 47–58. [Google Scholar] [CrossRef]
- Gower, J.C. Some Distance Properties of Latent Root and Vector Methods Used in Multivariate Analysis. Biometrika 1966, 53, 325–338. [Google Scholar] [CrossRef]
- Legendre, P. Studying Beta Diversity: Ecological Variation Partitioning by Multiple Regression and Canonical Analysis. J. Plant Ecol. 2007, 1, 3–8. [Google Scholar] [CrossRef]
- van den Wollenberg, A.L. Redundancy Analysis an Alternative for Canonical Correlation Analysis. Psychometrika 1977, 42, 207–219. [Google Scholar] [CrossRef]
- Dengler, J.; Jansen, F.; Chusova, O.; Hüllbusch, E.; Nobis, M.P.; Van Meerbeek, K.; Axmanová, I.; Bruun, H.H.; Chytrý, M.; Guarino, R.; et al. Ecological Indicator Values for Europe (EIVE) 1.0. Veg. Classif. Surv. 2023, 4, 7–29. [Google Scholar] [CrossRef]
- Midolo, G.; Herben, T.; Axmanová, I.; Marcenò, C.; Pätsch, R.; Bruelheide, H.; Karger, D.N.; Aćić, S.; Bergamini, A.; Bergmeier, E.; et al. Disturbance Indicator Values for European Plants. Glob. Ecol. Biogeogr. 2023, 32, 24–34. [Google Scholar] [CrossRef]
- Ostrowski, G.; Aicher, S.; Mankiewicz, A.; Chusova, O.; Dembicz, I.; Widmer, S.; Dengler, J. Mean Ecological Indicator Values: Use EIVE but No Cover-Weighting. Veg. Classif. Surv. 2025, 6, 57–67. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Lavorel, S.; Garnier, E. Predicting Changes in Community Composition and Ecosystem Functioning from Plant Traits: Revisiting the Holy Grail. Funct. Ecol. 2002, 16, 545–556. [Google Scholar] [CrossRef]
- Diaz, S.; Cabido, M. Plant Functional Types and Ecosystem Function in Relation to Global Change. J. Veg. Sci. 1997, 8, 463–474. [Google Scholar] [CrossRef]
- Bello, F.d.; Lavorel, S.; Lavergne, S.; Albert, C.H.; Boulangeat, I.; Mazel, F.; Thuiller, W. Hierarchical Effects of Environmental Filters on the Functional Structure of Plant Communities: A Case Study in the French Alps. Ecography 2013, 36, 393–402. [Google Scholar] [CrossRef]
- Pérez-Harguindeguy, N.; Díaz, S.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Bret-Harte, M.S.; Cornwell, W.K.; Craine, J.M.; Gurvich, D.E.; et al. New Handbook for Standardised Measurement of Plant Functional Traits Worldwide. Aust. J. Bot. 2013, 61, 167–234. [Google Scholar] [CrossRef]
- Moles, A.T.; Warton, D.I.; Warman, L.; Swenson, N.G.; Laffan, S.W.; Zanne, A.E.; Pitman, A.; Hemmings, F.A.; Leishman, M.R. Global Patterns in Plant Height. J. Ecol. 2009, 97, 923–932. [Google Scholar] [CrossRef]
- Pakeman, R.J.; Garnier, E.; Lavorel, S.; Ansquer, P.; Castro, H.; Cruz, P.; Doležal, J.; Eriksson, O.; Freitas, H.; Golodets, C.; et al. Impact of Abundance Weighting on the Response of Seed Traits to Climate and Land Use. J. Ecol. 2008, 96, 355–366. [Google Scholar] [CrossRef]
- Kleyer, M.; Bekker, R.M.; Knevel, I.C.; Bakker, J.P.; Thompson, K.; Sonnenschein, M.; Poschlod, P.; van Groenendael, J.M.; Klimeš, L.; Klimešová, J.; et al. The LEDA Traitbase: A Database of Life-History Traits of the Northwest European Flora. J. Ecol. 2008, 96, 1266–1274. [Google Scholar] [CrossRef]
- Society for Ecological Restoration, International Network for Seed Based Restoration and Royal Botanic Gardens Kew. Available online: https://ser-sid.org/about (accessed on 2 September 2025).
- Chytrý, M.; Řezníčková, M.; Novotný, P.; Holubová, D.; Preislerová, Z.; Attorre, F.; Biurrun, I.; Blažek, P.; Bonari, G.; Borovyk, D.; et al. FloraVeg.EU—An Online Database of European Vegetation, Habitats and Flora. Appl. Veg. Sci. 2024, 27, e12798. [Google Scholar] [CrossRef]
- Violle, C.; Navas, M.-L.; Vile, D.; Kazakou, E.; Fortunell, C.; Hummel, I.; Garner, E. Let the Concept of Trait Be Functional! Oikos 2007, 116, 882–892. [Google Scholar] [CrossRef]
- Paź-Dyderska, S.; Dyderski, M.K.; Szwaczka, P.; Brzezicha, M.; Bigos, K.; Jagodziński, A.M. Leaf Traits and Aboveground Biomass Variability of Forest Understory Herbaceous Plant Species. Ecosystems 2020, 23, 555–569. [Google Scholar] [CrossRef]
- Lavorel, S.; Grigulis, K.; McIntyre, S.; Williams, N.S.G.; Garden, D.; Dorrough, J.; Berman, S.; Quétier, F.; Thébault, A.; Bonis, A. Assessing Functional Diversity in the Field–Methodology Matters! Funct. Ecol. 2008, 22, 134–147. [Google Scholar]
- Pakeman, R.J. Functional Trait Metrics Are Sensitive to the Completeness of the Species’ Trait Data? Methods Ecol. Evol. 2014, 5, 9–15. [Google Scholar] [CrossRef]
- Mason, N.W.H.; Mouillot, D.; Lee, W.G.; Wilson, J.B. Functional Richness, Functional Evenness and Functional Divergence: The Primary Components of Functional Diversity. Oikos 2005, 111, 112–118. [Google Scholar] [CrossRef]
- Mason, N.W.H.; de Bello, F.; Mouillot, D.; Pavoine, S.; Dray, S. A Guide for Using Functional Diversity Indices to Reveal Changes in Assembly Processes along Ecological Gradients. J. Veg. Sci. 2013, 24, 794–806. [Google Scholar] [CrossRef]
- Fischer, H.S. On the Combination of Species Cover Values from Different Vegetation Layers. Appl. Veg. Sci. 2015, 18, 169–170. [Google Scholar] [CrossRef]
- Kermavnar, J.; Kutnar, L. Plant Diversity Decrease and Directional Species Turnover Induced by Shifting Overstory Dominance in the Oak-Hornbeam Forest Reserve over 50 Years. Flora 2025, 327, 152742. [Google Scholar] [CrossRef]
- Kotrík, M.; Bažány, M.; Čiliak, M.; Knopp, V.; Máliš, F.; Ujházyová, M.; Vaško, Ľ.; Vladovič, J.; Ujházy, K. Half a Century of Herb Layer Changes in Quercus-Dominated Forests of the Western Carpathians. For. Ecol. Manag. 2023, 544, 121151. [Google Scholar] [CrossRef]
- Mölder, A.; Streit, M.; Schmidt, W. When Beech Strikes Back: How Strict Nature Conservation Reduces Herb-Layer Diversity and Productivity in Central European Deciduous Forests. For. Ecol. Manag. 2014, 319, 51–61. [Google Scholar] [CrossRef]
- Heinrichs, S.; Schmidt, W. Biotic Homogenization of Herb Layer Composition between Two Contrasting Beech Forest Communities on Limestone over 50 Years. Appl. Veg. Sci. 2017, 20, 271–281. [Google Scholar] [CrossRef]
- Wrońska-Pilarek, D.; Rymszewicz, S.; Jagodziński, A.M.; Gawryś, R.; Dyderski, M.K. Temperate Forest Understory Vegetation Shifts after 40 Years of Conservation. Sci. Total Environ. 2023, 895, 165164. [Google Scholar] [CrossRef]
- Jakubowska-Gabara, J. Decline of Potentillo albae-Quercetum Libb. 1933 Phytocoenoses in Poland. Vegetatio 1996, 124, 45–59. [Google Scholar] [CrossRef]
- Van Calster, H.; Baeten, L.; De Schrijver, A.; De Keersmaeker, L.; Rogister, J.E.; Verheyen, K.; Hermy, M. Management Driven Changes (1967–2005) in Soil Acidity and the Understorey Plant Community Following Conversion of a Coppice-with-Standards Forest. For. Ecol. Manag. 2007, 241, 258–271. [Google Scholar] [CrossRef]
- Hédl, R.; Kopecký, M.; Komárek, J. Half a Century of Succession in a Temperate Oakwood: From Species-Rich Community to Mesic Forest. Divers. Distrib. 2010, 16, 267–276. [Google Scholar] [CrossRef]
- Altman, J.; Hédl, R.; Szabó, P.; Mazůrek, P.; Riedl, V.; Müllerová, J.; Kopecký, M.; Doležal, J. Tree-Rings Mirror Management Legacy: Dramatic Response of Standard Oaks to Past Coppicing in Central Europe. PLoS ONE 2013, 8, e55770. [Google Scholar] [CrossRef]
- Kopecký, M.; Hédl, R.; Szabó, P. Non-Random Extinctions Dominate Plant Community Changes in Abandoned Coppices. J. Appl. Ecol. 2013, 50, 79–87. [Google Scholar] [CrossRef]
- Vild, O.; Roleček, J.; Hédl, R.; Kopecký, M.; Utinek, D. Experimental Restoration of Coppice-with-Standards: Response of Understorey Vegetation from the Conservation Perspective. For. Ecol. Manag. 2013, 310, 234–241. [Google Scholar] [CrossRef]
- Müllerová, J.; Hédl, R.; Szabó, P. Coppice Abandonment and Its Implications for Species Diversity in Forest Vegetation. For. Ecol. Manag. 2015, 343, 88–100. [Google Scholar] [CrossRef]
- Becker, T.; Spanka, J.; Schröder, L.; Leuschner, C. Forty Years of Vegetation Change in Former Coppice-with-Standards Woodlands as a Result of Management Change and N Deposition. Appl. Veg. Sci. 2017, 20, 304–313. [Google Scholar] [CrossRef]
- Roleček, J.; Vild, O.; Sladký, J.; Řepka, R. Habitat Requirements of Endangered Species in a Former Coppice of High Conservation Value. Folia Geobot. 2017, 52, 59–69. [Google Scholar] [CrossRef]
- Vojík, M.; Boublík, K. Fear of the Dark: Decline in Plant Diversity and Invasion of Alien Species Due to Increased Tree Canopy Density and Eutrophication in Lowland Woodlands. Plant Ecol. 2018, 219, 749–758. [Google Scholar] [CrossRef]
- Holeštová, A.; Havrdová, A.; Černá, M.; Doudová, J.; Boublík, K.; Douda, J. From Past Diversity to Present Decline: Age Heterogeneity and Plant Composition Shifts in Central European Floodplain Forests. For. Ecol. Manag. 2025, 587, 122732. [Google Scholar] [CrossRef]
- Bernhardt-Römermann, M.; Baeten, L.; Craven, D.; De Frenne, P.; Hédl, R.; Lenoir, J.; Bert, D.; Brunet, J.; Chudomelová, M.; Decocq, G.; et al. Drivers of Temporal Changes in Temperate Forest Plant Diversity Vary across Spatial Scales. Glob. Change Biol. 2015, 21, 3726–3737. [Google Scholar] [CrossRef]
- Maes, S.L.; Perring, M.P.; Vanhellemont, M.; Depauw, L.; Van den Bulcke, J.; Brūmelis, G.; Brunet, J.; Decocq, G.; den Ouden, J.; Härdtle, W.; et al. Environmental Drivers Interactively Affect Individual Tree Growth across Temperate European Forests. Glob. Change Biol. 2019, 25, 201–217. [Google Scholar] [CrossRef]
- Blondeel, H.; Perring, M.P.; Depauw, L.; De Lombaerde, E.; Landuyt, D.; De Frenne, P.; Verheyen, K. Light and Warming Drive Forest Understorey Community Development in Different Environments. Glob. Change Biol. 2020, 26, 1681–1696. [Google Scholar] [CrossRef] [PubMed]
- Depauw, L.; Perring, M.P.; Landuyt, D.; Maes, S.L.; Blondeel, H.; De Lombaerde, E.; Brūmelis, G.; Brunet, J.; Closset-Kopp, D.; Czerepko, J.; et al. Light Availability and Land-use History Drive Biodiversity and Functional Changes in Forest Herb Layer Communities. J. Ecol. 2020, 108, 1411–1425. [Google Scholar] [CrossRef]
- Naaf, T.; Wulf, M. Traits of Winner and Loser Species Indicate Drivers of Herb Layer Changes over Two Decades in Forests of NW Germany. J. Veg. Sci. 2011, 22, 516–527. [Google Scholar] [CrossRef]
- Reczyńska, K.; Świerkosz, K. Compositional Changes in Thermophilous Oak Forests in Poland over Time: Do They Correspond to European Trends? Appl. Veg. Sci. 2017, 20, 293–303. [Google Scholar] [CrossRef]
- Strubelt, I.; Diekmann, M.; Zacharias, D. Changes in Species Composition and Richness in an Alluvial Hardwood Forest over 52 Yrs. J. Veg. Sci. 2017, 28, 401–412. [Google Scholar] [CrossRef]
- Amatangelo, K.L.; Fulton, M.R.; Rogers, D.A.; Waller, D.M. Converging Forest Community Composition along an Edaphic Gradient Threatens Landscape-level Diversity. Divers. Distrib. 2011, 17, 201–213. [Google Scholar] [CrossRef]
- Verstraeten, G.; Baeten, L.; Van den Broeck, T.; De Frenne, P.; Demey, A.; Tack, W.; Muys, B.; Verheyen, K. Temporal Changes in Forest Plant Communities at Different Site Types. Appl. Veg. Sci. 2013, 16, 237–247. [Google Scholar] [CrossRef]
- Reinecke, J.; Klemm, G.; Heinken, T. Vegetation Change and Homogenization of Species Composition in Temperate Nutrient Deficient Scots Pine Forests after 45 Yr. J. Veg. Sci. 2014, 25, 113–121. [Google Scholar] [CrossRef]
- Naaf, T.; Kolk, J. Initial Site Conditions and Interactions between Multiple Drivers Determine Herb-Layer Changes over Five Decades in Temperate Forests. For. Ecol. Manag. 2016, 366, 153–165. [Google Scholar] [CrossRef]
- Kermavnar, J.; Kutnar, L. Three Decades of Understorey Vegetation Change in Quercus-dominated Forests as a Result of Increasing Canopy Mortality and Global Change Symptoms. J. Veg. Sci. 2024, 35, e13317. [Google Scholar] [CrossRef]
- Reczyńska, K.; Orczewska, A.; Yurchenko, V.; Wójcicka-Rosińska, A.; Świerkosz, K. Changes in Species and Functional Diversity of the Herb Layer of Riparian Forest despite Six Decades of Strict Protection. Forests 2022, 13, 747. [Google Scholar] [CrossRef]
- Foxcroft, L.C.; Jarošík, V.; Pyšek, P.; Richardson, D.M.; Rouget, M. Protected-Area Boundaries as Filters of Plant Invasions: Park Boundaries and Invasive Plants. Conserv. Biol. 2011, 25, 400–405. [Google Scholar] [CrossRef]
- Perring, M.P.; De Frenne, P.; Hertzog, L.R.; Blondeel, H.; Depauw, L.; Maes, S.L.; Wasof, S.; Verbeeck, H.; Verheyen, K.; forestREplot; et al. Increasing Liana Frequency in Temperate European Forest Understories Is Driven by Ivy. Front. Ecol. Environ. 2020, 18, 550–557. [Google Scholar] [CrossRef]
- Wagner, V.; Večeřa, M.; Jiménez-Alfaro, B.; Pergl, J.; Lenoir, J.; Svenning, J.; Pyšek, P.; Agrillo, E.; Biurrun, I.; Campos, J.A.; et al. Alien Plant Invasion Hotspots and Invasion Debt in European Woodlands. J. Veg. Sci. 2021, 32, e13014. [Google Scholar] [CrossRef]
- Moustakas, A.; Voutsela, A.; Katsanevakis, S. Sampling Alien Species inside and Outside Protected Areas: Does It Matter? Sci. Total Environ. 2018, 625, 194–198. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.