Abstract
Hydrazones were obtained in 76–78% yield via condensation of (2S,5R)-2-isopropyl-5-methylcyclohexanone with 4-R-phenoxyacetic acid hydrazides in the presence of a catalytic amount of glacial acetic acid. The structure of the target compounds has been established by FTIR-ATR, Raman, 1H-NMR and 13C-NMR spectral analysis and EI/FAB/ESI mass spectrometry. Thermal properties of hydrazones 3a–3e were elucidated by differential scanning calorimetry (DSC) and their purity by HPLC coupled to mass spectrometry. Synthesized compounds were found to exist as Z/E geometrical isomers about C=N bond and cis/trans amide conformers.
1. Introduction
Currently, considerable interest in drug development is concentrated on obtaining drugs which contemporaneously affect various pharmacological targets, thus exhibiting combined action. In this context, particular interest has been focused on compounds affecting both the central and peripheral nervous system. Recently, we have embodied this idea by combination of neurotransmitter amino acids with terpenoids capable of binding to the transient receptor potential channels (TRP) [1,2]. The present work is a logical continuation and reveals a strategy for drug development containing residues of cyclic terpene l-menthone and para-substituted phenoxyacetic acids. Besides binding to TRPM8 channels resulting in pain relief, l-menthone was found to act as modulators of GABAA receptors, thereby demonstrating anticonvulsant activity [3,4]. Phenoxyacetic acid derivatives in turn also exhibit peripheral nociceptive effects and possess neuroprotective and anticonvulsant potentialities [5,6].
Based on the abovementioned, the combination of l-menthone and phenoxyacetic acid residues into one molecule is feasible for obtaining drugs which contemporaneously affect various pharmacological targets. Such a combination might be implemented by synthesis of hydrazones which are principle compounds for drug design due to their wide spectrum of pharmacological action [7]. Thus, the current paper is devoted to the synthesis and detailed structure determination of hydrazones based on (2S,5R)-2-isopropyl-5-methylcyclohexanone and para-substituted phenoxyacetic acids.
2. Results and Discussion
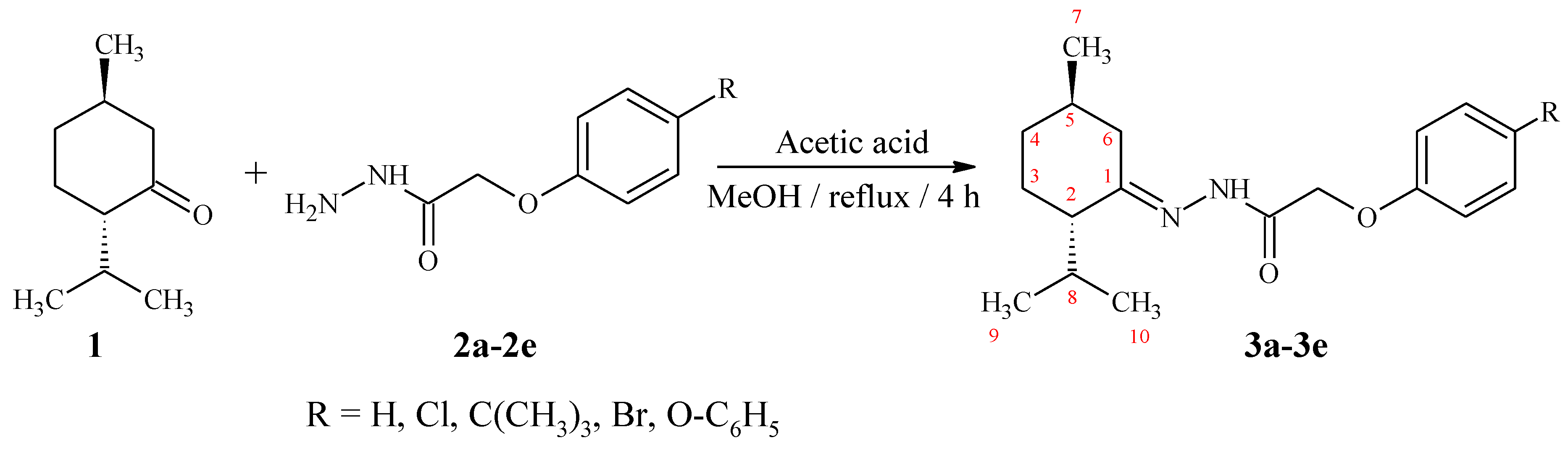
(2S,5R)-2-Isopropyl-5-methylcyclohexanone hydrazones 3a–3e were synthesized via condensation of l-menthone 1 with 4-R-phenoxyacetic acid hydrazides 2a–2e in the presence of a catalytic amount of glacial acetic acid, as shown in Scheme 1. Synthesized hydrazones were isolated in 76–78% yield as white solid well soluble in organic solvents (chloroform, acetonitrile, benzene, ethyl acetate) and fully characterized by 13C-NMR, 1H-NMR, FTIR-ATR, Raman-spectroscopy and FAB-, EI-, ESI-mass spectrometry. Thermal behavior of compounds 3a–3e was performed by differential scanning calorimetry (DSC). Additionally, HPLC analysis was carried out to determine the purity of title compounds. For this purpose, the reversed-phase HPLC method with isocratic elution of acetonitrile: 0.01% formic acid aqueous solution (70:30) was applied. HPLC analysis of recrystallized compounds 3a–3e revealed two chromatographic peaks due to the existence of Z/E geometrical isomers around the C=N bond that was corroborated by mass spectrometry. The observed retention time for these peaks ranges from 8.283 min to 23.162 min (major peak) and from 6.139 to 14.180 (minor peak) depending on compounds’ lipophilicity which is increased in order: 3a–3b–3d–3e–3c.

Scheme 1.
Synthesis of (2S,5R)-2-isopropyl-5-methylcyclohexanone hydrazones.
The formation of titled compounds was reliably confirmed by mass spectrometry using different types of ionization methods, which were fast-atom bombardment (FAB), electron impact (EI) and electrospray ionization (ESI). Molecular ion peaks for all synthesized compounds 3a–3e are in agreement with their molecular formulas. The FTIR spectra obtained using the ATR technique exhibits absorption bands of N–H bonds (3184–3206 cm−1), C=O groups (1672–1697 cm−1), aromatic and alkyl C–H. Since C=N and C=O vibrations in FTIR-ATR spectra appear in the same region, the formation of the imine (C=N) group has been proven by Raman spectroscopy to be the intense peak at 1639–1641 cm−1. The 1H-NMR and 13C-NMR spectral data of hydrazones 3a–3e contain resonance signals described by their chemical shift, integration and multiplicity that are in full agreement with the presented molecular formulas. Signals of axial and equatorial ring protons are also presented in 1H-NMR spectrum, their position and multiplicity correspond to similar signals in the spectrum of l-menthone.
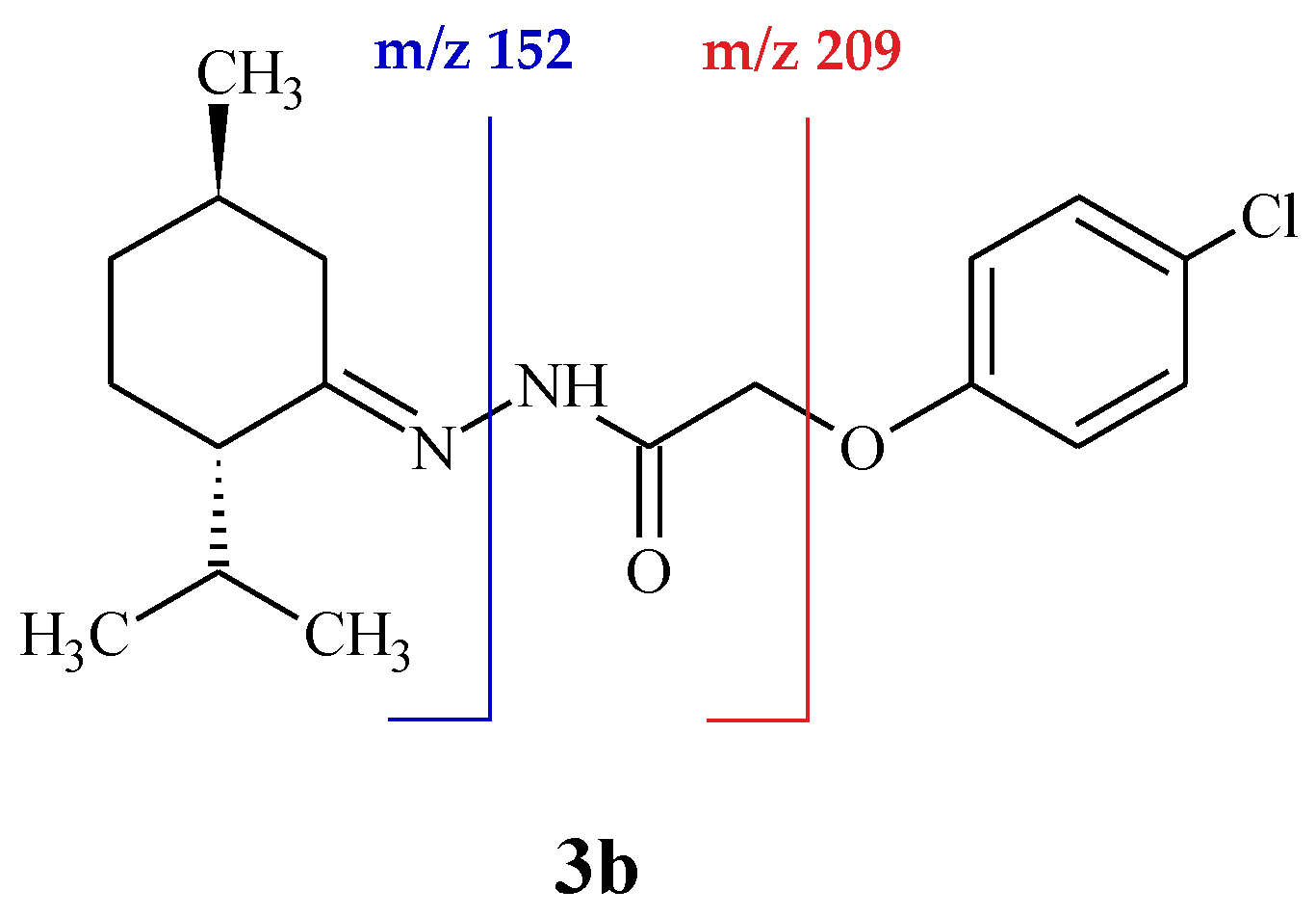
In order to distinguish Z/E geometrical isomers, ion mobility-tandem mass spectrometry (IM-MS/MS) analysis has been applied. Compared to E forms, Z isomers have less mobility, due to steric hindrance of bulky groups, that decrease structure compactness (see Supplementary Materials). Based on MS/MS spectra of Z/E forms we may conclude that these isomers have different fragmentation patterns caused by their stability. For example, in MS/MS spectrum of hydrazone 3b (Z-isomer) the most abundant fragment signal is observed at m/z 152 (cleavage of N–N bond) while for the E form of this compound it is observed at m/z 209 (cleavage of C–O bond). Since Z isomer is less stable, the heavy side chain is leaving easily in order to reduce the stress on the molecule. In contrast, E isomer is more relaxed and the small side chain is left from the molecule forming, thus, the ion fragments at m/z 209 (Figure 1).

Figure 1.
Mechanism of hydrazone 3b fragmentation: Z-isomer (blue line), E-isomer (red line).
Synthesized hydrazones, along with Z/E geometrical isomers around the C=N bond may exist in solution as a mixture of cis/trans conformers due to hindered rotation on the C–N amide bond [8]. 1H-NMR spectra of compounds 3a–3e in DMSO-d6 solution display two sets of singlets related to methylene (CH2) and imine (NH) protons indicating the presence of cis/trans conformers. According to the literature, in polar aprotic solvents (DMSO-d6) both cis and trans amide conformers are observed, however, the cis form is prevalent because of dimers’ formation stabilized by C=O···H–N hydrogen bonds [8,9]. In the 1H-NMR spectra the upfield peak of CH2 group belongs to trans conformer whereas downfield peak–to cis form [8]. It ought to be pointed out that a similar pattern was observed in the 1H-NMR spectra of menthone derivatives 3a–3e: two singlets for CH2 protons (4.58–4.62 ppm and 4.92–4.97 ppm) as well as two singlets for NH protons (10.34–10.41 ppm and 10.51–10.63 ppm). Thus, we may conclude that the obtained hydrazones 3a–3e exist in DMSO-d6 solution as a mixture of cis/trans conformers.
3. Materials and Methods
3.1. General Information
The following chemicals were used as obtained from their commercial suppliers: l-menthone, phenol, 4-bromophenol, 4-chlorophenol, 4-tert-butylphenol, 4-phenoxyphenol (TCI, Philadelphia, PA, USA). 4-R-phenoxyacetic acid hydrazides 2a–2e were not obtained commercially and had been synthesized according to the procedure found in the literature [10]. The structure of the compound obtained was established by 1H-NMR spectroscopy on an AVANCE DRX 500 (500 MHz) instrument (Bruker, Davis, CA, USA) and by 13C-NMR spectroscopy on Varian-Mercury 400 spectrometer (Varian Inc., Palo Alto, CA, USA) using DMSO-d6 as a solvent and TMS as an internal standard. FAB mass spectra were obtained on a VG 70-70EQ mass spectrometer (VG Analytical Ltd., Manchester, UK) equipped with Xe ion gun (8 kV); the sample was mixed with m-nitrobenzyl-alcohol matrix. EI mass spectra were recorded on mass spectrometer MX-1321 (ionization voltage 70 eV, temperature of the ionization chamber is 200 °C). Mass spectrometry (MS) was performed on a 6530 Accurate Mass quadrupole time of flight (Q-TOF) spectrometer (Agilent, Santa Clara, CA, USA) using electrospray ionization (ESI) coupled to an Agilent 1260 Infinity HPLC system. The IR spectrum was measured with a FT-IR-8400S spectrometer (Shimadzu, Columbia, MD, USA) using the ATR technique. Ion mobility-tandem mass spectrometry (IM-MS/MS) analysis was carried out using a trapped ion mobility spectrometry–time-of-flight (TIMS–TOF) mass spectrometer equipped with an ESI source (Bruker Daltonics, Bremen, Germany). Sample concentration was 0.003 mg/mL prepared in a 7:3 (v/v) mixture of acetonitrile and water containing 0.1% formic acid. The sample solution was directly infused into the ESI source at a flow rate of 3.0 µL/min. IM-MS/MS analysis was performed using the following ESI source settings: 4500 V capillary voltage, −500 V end plate offset, 2000 V charging voltage, 0.3 bar nebulizer pressure, 3.0 L/min dry gas, 200 °C dry heater temperature. The TIMS parameter settings were as follows: IMS ramp start: 0.40 V·s/cm2, ramp end 1.80 V·s/cm2; ramp time 73.0 ms; ΔV ramp start and accumulation exit: 100 V. Nitrogen was used as a bath gas at ca. 300 K. Collision-induced dissociation was applied following TIMS separation to isomer ions having m/z 337.16 with identical collision energy of approximately 31 eV in the collision cell. Acquired data were processed using DataAnalysis 5.0 software provided by Bruker. Raman spectra were measured using a DXR Raman Microscope (Thermo Fisher Scientific, Madison, WI, USA). DSC curves were recorded in a Q2000 Differential Scanning Calorimeter (TA Instruments, New Castle, DE, USA) using aluminum crucibles containing approximately 2 mg of samples, in a dynamic nitrogen atmosphere and with a heating rate of 5 °C min−1 in the temperature range of 20 to 200 °C.
3.2. Synthesis of (2S,5R)-2-Isopropyl-5-methylcyclohexanone hydrazones (3a–3e)
To solution of l-menthone (1.0 g, 6.5 mmol) in methanol (25 mL), the equimolar amount (6.5 mmol) of appropriate 4-R-phenoxyacetic acid hydrazides and 2 drops of glacial acetic acid were added. The reaction mixture was refluxed for 4 h, cooled and then was poured into the ice. The solid mass was filtered, dried and purified by recrystallization from methanol:water (1:1) system.
2-Phenoxy-N′-[(2S,5R)-5-methyl-2-(propan-2-yl)cyclohexylidene]acetohydrazide (3a). 1H-NMR (500 MHz, DMSO-d6) δ: 0.89 (d, J = 6.28 Hz, 3H, CH3-10), 0.92 (d, J = 5.52 Hz, 6H, СН3-7 + CH3-9), 1.14–1.24 (m, 2Н, Н-4а + H-4e), 1.48–1.53 (m, 2Н, Н-3а + H-3e), 1.76–1.79 (m, 1Н, H-5), 1.86 (m, 1Н, (CH3)2CH), 1.90–1.92 (m, 2Н, Н-6a + Н-6е), 2.16–2.19 (m, 1Н, H-2), 4.96 (s, 2Н, CH2), 6.84 (d, J = 1.4 Hz, 2Н, Ar-H), 6.93 (t, 1Н, Ar-H), 7.27 (t, 2Н, Ar-H), 10.61 (s, 1H, NH). 13C-NMR (100 MHz, DMSO-d6) δ: 169.9 (C=O), 158.7 (C-1), 157.62 (Ar-C), 129.8 (Ar-C), 121.1 (Ar-C), 114.7 (Ar-C), 65.3 (CH2), 50.1 (C-2), 34.7 (C-4), 33.6 (C-6), 28.0 (C-5), 26.7 (C-3), 22.0 (C-10), 21.9 (C-7), 19.2 (C-9). FTIR-ATR (νmax, cm−1): 3207 (N-H), 3093 (C-H, Ar), 2955–2839 (C-H), 1691 (C=O), 1597–1586 (C-C, Ar), 883–688 (C-H, Ar). Raman (νmax, cm−1): 3200 (N-H), 3070 (C-H, Ar), 2957–2871 (C-H), 1683 (C=O), 1641 (C=N). MS (EI) m/z: 302 [M]+. MS (ESI-TOF) calculated for C18H26N2O2 [M + H]+ 303.42, found 303.26. HPLC: tr = 8.283 min (E-isomer), 6.139 (Z-isomer). M.p. (DSC) onset: 109.61 °C, peak max: 111.01 °C.
2-(4-Chlorophenoxy)-N′-[(2S,5R)-5-methyl-2-(propan-2-yl)cyclohexylidene]acetohydrazide (3b). 1H-NMR (500 MHz, DMSO-d6) δ: 0.88 (d, J = 6.02 Hz, 3H, CH3-10), 0.91 (d, J = 6.52 Hz, 6H, СН3-7 + CH3-9), 1.13–1.23 (m, 2Н, Н-4а + H-4e), 1.47–1.53 (m, 2Н, Н-3а + H-3e), 1.76–1.78 (m, 1Н, H-5), 1.86–1.89 (m, 3Н, (CH3)2CH + Н-6a + Н-6е), 2.16–2.17 (m, 1Н, H-2), 4.97 (s, 2Н, CH2), 6.87 (d, J = 2.5 Hz, 2Н, Ar-H), 7.31 (d, J = 1.7 Hz, 2Н, Ar-H), 10.63 (s, 1H, NH). 13C-NMR (100 MHz, DMSO-d6) δ: 169.6 (C=O), 157.7 (C-1), 129.5 (Ar-C), 124.7 (Ar-C), 116.5 (Ar-C), 65.7 (CH2), 50.1 (C-2), 34.7 (C-4), 33.6 (C-6), 28.0 (C-5), 26.7 (C-3), 22.0 (C-10), 21.9 (C-7), 19.2 (C-9). FTIR-ATR (νmax, cm−1): 3191 (N-H), 3130 (C-H, Ar), 2951–2871 (C-H), 1697 (C=O), 1597–1582 (C-C, Ar), 821–663 (C-H, Ar). Raman (νmax, cm−1): 3071 (C-H, Ar), 2958–2848 (C-H), 1679 (C=O), 1641 (C=N). MS (EI) m/z: 336 [M]+. MS (ESI-TOF) calculated for C18H25ClN2O2 [M + H]+ 337.85, found 337.39. HPLC: tr = 11.663 min (E-isomer), 7.375 (Z-isomer). M.p. (DSC) onset: 120.60 °C, peak max: 122.98 °C (first peak); onset: 125.51 °C, peak max: 127.47 °C (second peak).
2-(4-tert-Butylphenoxy)-N′-[(2S,5R)-5-methyl-2-(propan-2-yl)cyclohexylidene]acetohydrazide (3c). 1H-NMR (500 MHz, DMSO-d6) δ: 0.89 (d, J = 6.52 Hz, 3H, CH3-10), 0.92 (d, J = 6.52 Hz, 6H, СН3-7 + CH3-9), 1.13–1.19 (m, 2Н, Н-4а + H-4e), 1.24 (s, 9H, C(CH3)3), 1.47–1.53 (m, 2Н, Н-3а + H-3e), 1.75–1.78 (m, 1Н, H-5), 1.86–1.89 (m, 3Н, (CH3)2CH + Н-6a + Н-6е), 2.15–2.18 (m, 1Н, H-2), 4.92 (s, 2Н, CH2), 6.76 (d, J = 1.1 Hz, 2Н, Ar-H), 7.27 (d, J = 1.1 Hz, 2Н, Ar-H), 10.51 (s, 1H, NH). 13C-NMR (100 MHz, DMSO-d6) δ: 170.0 (C=O), 157.5 (C-1), 156.4 (Ar-C), 143.2 (Ar-C), 126.4 (Ar-C), 114.1 (Ar-C), 65.4 (CH2), 50.1 (C-2), 34.8 (C-4), 34.2 (C-6), 31.8 ((CH3)3)28.1 (C-5), 26.7 (C-3), 22.1 (C-10), 21.9 (C-7), 19.2 (C-9). FTIR-ATR (νmax, cm−1): 3190 (N-H), 3107 (C-H, Ar), 2949–2863 (C-H), 1691 (C=O), 1584 (C-C, Ar), 841–669 (C-H, Ar). Raman (νmax, cm−1): 3072 (C-H, Ar), 2962–2866 (C-H), 1672 (C=O), 1641 (C=N). MS (FAB) m/z: 359 [M + H]+. MS (ESI-TOF) calculated for C22H34N2O2 [M + H]+ 359.51, found 359.63. HPLC: tr = 23.162 min (E-isomer), 14.180 (Z-isomer). M.p. (DSC) onset: 146.73 °C, peak max: 148.23 °C.
2-(4-Bromophenoxy)-N′-[(2S,5R)-5-methyl-2-(propan-2-yl)cyclohexylidene]acetohydrazide (3d). 1H-NMR (500 MHz, DMSO-d6) δ: 0.88 (d, J = 6.02 Hz, 3H, CH3-10), 0.91 (d, J = 6.52 Hz, 6H, СН3-7 + CH3-9), 1.13–1.23 (m, 2Н, Н-4а + H-4e), 1.47–1.53 (m, 2Н, Н-3а + H-3e), 1.76–1.78 (m, 1Н, H-5), 1.86–1.89 (m, 3Н, (CH3)2CH + Н-6a + Н-6е), 2.16–2.17 (m, 1Н, H-2), 4.97 (s, 2Н, CH2), 6.87 (d, J = 2.7 Hz, 2Н, Ar-H), 7.31 (d, J = 1.6 Hz, 2Н, Ar-H), 10.63 (s, 1H, NH). 13C-NMR (100 MHz, DMSO-d6) δ: 169.6 (C=O), 157.7 (C-1), 129.5 (Ar-C), 124.7 (Ar-C), 116.5 (Ar-C), 65.7 (CH2), 50.1 (C-2), 34.7 (C-4), 33.6 (C-6), 28.0 (C-5), 26.7 (C-3), 22.0 (C-10), 21.9 (C-7), 19.2 (C-9). FTIR-ATR (νmax, cm−1): 3190 (N-H), 3089 (C-H, Ar), 2954–2869 (C-H), 1692 (C=O), 1596–1577 (C-C, Ar), 889–698 (C-H, Ar). Raman (νmax, cm−1): 3071 (C-H, Ar), 2958–2839 (C-H), 1683 (C=O), 1641 (C=N). MS (FAB) m/z: 381 [M]+. MS (ESI-TOF) calculated for C18H25BrN2O2 [M + H]+ 383.31, found 383.51. HPLC: tr = 12.832 min (E-isomer), 8.073 (Z-isomer). M.p. (DSC) onset: 134.01 °C, peak max: 135.50 °C.
2-(4-Phenoxyphenoxy)-N′-[(2S,5R)-5-methyl-2-(propan-2-yl)cyclohexylidene]acetohydrazide (3e). 1H-NMR (500 MHz, DMSO-d6) δ: 0.88 (d, J = 5.78 Hz, 3H, CH3-10), 0.91 (d, J = 6.77 Hz, 6H, СН3-7 + CH3-9), 1.13–1.23 (m, 2Н, Н-4а + H-4e), 1.47–1.52 (m, 2Н, Н-3а + H-3e), 1.75–1.77 (m, 1Н, H-5), 1.86–1.91 (m, 3Н, (CH3)2CH + Н-6a + Н-6е), 2.15–2.18 (m, 1Н, H-2), 4.95 (s, 2Н, CH2), 6.87-6.92 (m, 4H, Ar-H), 6.97 (d, J = 2.7 Hz, 2Н, Ar-H), 7.05 (t, 1H, Ar-H), 7.33 (t, 2Н, Ar-H), 10.62 (s, 1H, NH). 13C-NMR (100 MHz, DMSO-d6) δ: 169.9 (C=O), 158.4 (C-1), 157.6 (Ar-C), 149.9 (Ar-C), 129.5 (Ar-C), 130.3 (Ar-C), 123.0 (Ar-C), 121.0 (Ar-C), 117.7 (Ar-C), 116.0 (Ar-C), 65.9 (CH2), 50.1 (C-2), 34.7 (C-4), 33.6 (C-6), 28.0 (C-5), 26.7 (C-3), 22.0 (C-10), 21.9 (C-7), 19.2 (C-9). FTIR-ATR (νmax, cm−1): 3184 (N-H), 3081 (C-H, Ar), 2950–2843 (C-H), 1689 (C=O), 1597–1575 (C-C, Ar), 837-668 (C-H, Ar). Raman (νmax, cm−1): 3071 (C-H, Ar), 2958–2839 (C-H), 1683 (C=O), 1641 (C=N). MS (FAB) m/z: 395 [M + H]+. MS (ESI-TOF) calculated for C24H30N2O3 [M + H]+ 395.51, found 395.64. HPLC: tr = 17.018 min (E-isomer), 11.821 (Z-isomer). M.p. (DSC) onset: 111.16 °C, peak max: 116.43 °C.
4. Conclusions
Condensation of (2S,5R)-2-isopropyl-5-methylcyclohexanone with 4-R-phenoxyacetic acid hydrazides in the presence of a catalytic amount of glacial acetic acid was successfully applied to synthesize the title compounds followed by structure confirmation via FTIR-ATR, Raman, 1H-NMR and 13C-NMR spectral analysis and mass spectrometry.
Supplementary Materials
Copies of the 1H-NMR 13C-NMR, FTIR-ATR, Raman, EI/FAB/ESI mass spectra, DSC thermograms and HPLC chromatograms are available online.
Author Contributions
I.K. conceived and designed the experiments; M.N. and I.Z. performed the synthesis and analyzed NMR spectral data; D.B. performed the analysis of FTIR-ATR, Raman, DSC and HPLC experiments; I.R. carried out FAB and EI characterization of the compound; B.S. and M.A. carried out IM-MS/MS analysis; all authors contributed in manuscript writing.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Nesterkina, M.; Shishkina, S.; Maltsev, G.; Rakipov, I.; Kravchenko, I. (1R,2S,5R)-2-Isopropyl-5-methylcyclohexyl 4-aminobutyrate hydrochloride. Molbank 2017, 2017, M956. [Google Scholar] [CrossRef]
- Nesterkina, M.; Kravchenko, I. Synthesis and pharmacological properties of novel esters based on monocyclic terpenes and GABA. Pharmaceuticals 2016, 9, 32. [Google Scholar] [CrossRef] [PubMed]
- Vriens, J.; Nilius, B.; Vennekens, R. Herbal compounds and toxins modulating TRP channels. Curr. Neuropharmacol. 2008, 6, 79–96. [Google Scholar] [PubMed]
- Sánchez-Borzone, M.E.; Marin, L.D.; García, D.A. Effects of insecticidal ketones present in mint plants on GABAA receptor from mammalian neurons. Pharmacogn. Mag. 2017, 13, 114–117. [Google Scholar] [PubMed]
- Turan-Zitouni, G.; Yurttaş, L.; Kaplancıklı, Z.A.; Can, Ö.D.; Özkay, Ü.D. Synthesis and anti-nociceptive, anti-inflammatory activities of new aroyl propionic acid derivatives including N-acylhydrazone motif. Med. Chem. Res. 2015, 24, 2406–2416. [Google Scholar] [CrossRef]
- Pages, N.; Maurois, P.; Bac, P.; Eynde, J.J.V.; Tamariz, J.; Labarrios, F.; Chamorro, G.; Vamecq, J. The α-asarone/clofibrate hybrid compound, 2-methoxy-4-(2-propenyl)phenoxyacetic acid (MPPA), is endowed with neuroprotective and anticonvulsant potentialities. Biomed. Aging Pathol. 2011, 1, 210–215. [Google Scholar] [CrossRef]
- Rollas, S.; Küçükgüzel, S.G. Biological activities of hydrazone derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar] [CrossRef] [PubMed]
- Palla, G.; Predieri, G.; Domiano, P.; Vignali, C.; Turner, W. Conformational behaviour and E/Z isomerization of N-acyl and N-aroylhydrazones. Tetrahedron 1986, 42, 3649–3654. [Google Scholar] [CrossRef]
- Johansson, A.; Kollman, P.; Rothenberg, S.; McKelvey, J. Hydrogen bonding ability of the amide group. J. Am. Chem. Soc. 1974, 96, 3794–3800. [Google Scholar] [CrossRef]
- Kaur, P.; Kaur, R.; Goswami, M. A review on methods of synthesis of 1,2,4-triazole derivatives. IRJP 2018, 9, 1–35. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).