Journal Description
Receptors
Receptors
is an international, peer-reviewed, open access journal on all aspects of receptors published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 49.9 days after submission; acceptance to publication is undertaken in 7.7 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review and reviewer names are published annually in the journal.
- Receptors is a companion journal of Biomolecules.
Latest Articles
Interleukin-17A (IL-17A): Molecular Mechanisms and Its Roles in Immune and Neuroimmune Systems
Receptors 2026, 5(2), 16; https://doi.org/10.3390/receptors5020016 - 21 May 2026
Abstract
►
Show Figures
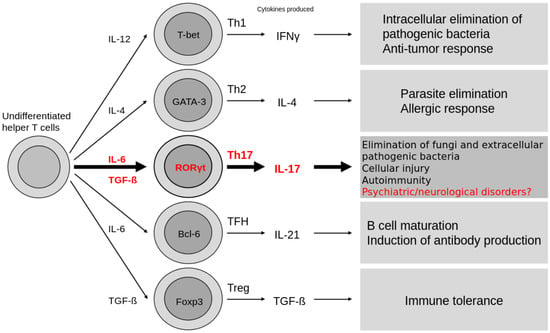
Interleukin-17A (IL-17A) is a proinflammatory cytokine that plays a pivotal role in immune responses and tissue homeostasis. Its expression is strictly regulated by transcription factors including RORγt, and it is mainly produced by Th17 cells, γδ T cells, and innate lymphoid cells. IL-17A
[...] Read more.
Interleukin-17A (IL-17A) is a proinflammatory cytokine that plays a pivotal role in immune responses and tissue homeostasis. Its expression is strictly regulated by transcription factors including RORγt, and it is mainly produced by Th17 cells, γδ T cells, and innate lymphoid cells. IL-17A signals through a heterodimeric receptor complex consisting of IL-17RA and IL-17RC, activating NF-κB, MAPK, and C/EBP pathways via the adaptor protein Act1. IL-17 signaling is counterbalanced by negative regulators including A20 and Regnase-1. Beyond its classical roles in antimicrobial defense and autoimmune inflammation, recent studies have highlighted its functions in the central nervous system, with associations to multiple sclerosis, autism spectrum disorder, and Alzheimer’s disease. The development of IL-17A inhibitors, including the dual IL-17A/F antagonist bimekizumab, has advanced markedly, with demonstrated efficacy in immune-mediated diseases such as psoriasis and psoriatic arthritis. This review provides a comprehensive overview of current knowledge of IL-17A, from its molecular characteristics to clinical applications.
Full article
Open AccessArticle
Analyzing the Molecular Effects of Endomorphin-2 Degradation on Stabilizing Interactions at the μ-Opioid Receptor
by
Celvic Coomber, Jakob J. Kresse, Surahit Chewle, Marcus Weber, Christof Schütte and Vikram Sunkara
Receptors 2026, 5(2), 15; https://doi.org/10.3390/receptors5020015 - 28 Apr 2026
Abstract
Background: Endogenous opioids, such as endomorphin-2, are key regulators of the body’s pain pathways and mediate analgesia by engaging the
Background: Endogenous opioids, such as endomorphin-2, are key regulators of the body’s pain pathways and mediate analgesia by engaging the
(This article belongs to the Collection Receptors: Exceptional Scientists and Their Expert Opinions)
►▼
Show Figures
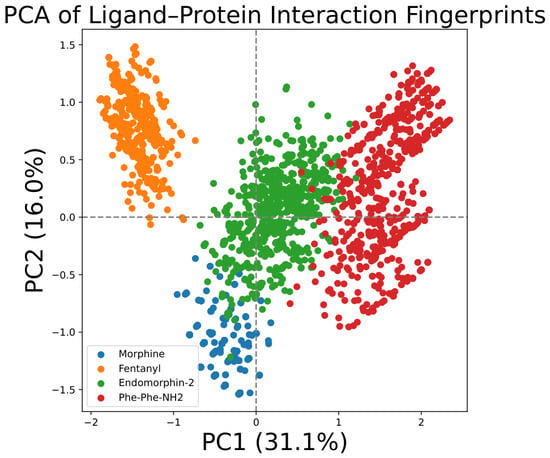
Figure 1
Open AccessReview
Classical Immune Pattern Recognition Receptors Involved in Inflammatory Trigger of Sickle Cell Anemia
by
Hershiley Oliveira Jácome, Jonatas Alencar Castro Campelo and Alexander Leonardo Silva-Junior
Receptors 2026, 5(2), 14; https://doi.org/10.3390/receptors5020014 - 21 Apr 2026
Abstract
►▼
Show Figures
Sickle cell anemia (SCA) is a genetic disorder characterized by chronic hemolysis, primarily driven by red blood cell lysis. Its pathophysiology is centered, though not exclusively, on the increased release of intracellular components, such as hemoglobin degradation products, which are known to stimulate
[...] Read more.
Sickle cell anemia (SCA) is a genetic disorder characterized by chronic hemolysis, primarily driven by red blood cell lysis. Its pathophysiology is centered, though not exclusively, on the increased release of intracellular components, such as hemoglobin degradation products, which are known to stimulate innate immune responses and promote prothrombotic states. Current therapies alleviate symptoms, yet patients remain exposed to a chronic inflammatory milieu punctuated by episodes of acute pain. The recurrence of these crises can be life-threatening due to ischemia–reperfusion injury, hypercoagulability, and respiratory complications. Central mechanisms are marked by elevated hemolysis, heightened inflammatory signaling, and increased procoagulant activity, largely driven by soluble molecules released into the plasma, such as hemoglobin, nuclear molecules and other products. These compounds are recognized from sensors on immune and endothelial cells, named Pattern Recognition Receptors (PRRs), and constitute canonical pathways for intracellular activation. Four main types have been extensively studied in the literature over recent years in both infectious and sterile inflammatory contexts; still, only a few have elucidated the mechanisms underlying acute and chronic inflammation in patients with SCA. Although Toll receptors were shown to be major in triggering immunity, other receptors were found to be important regarding this function, which suggested a multifactorial mechanism for this triggering. Therefore, here, we propose a comprehensive review of previously published findings regarding the expression, activation, and dynamics of Toll-like, NOD-like, and RIG-I–like receptors in the progression of SCA and its associated inflammatory features.
Full article

Figure 1
Open AccessOpinion
Does Biochemical Life Exist for a Receptor Agonist Outside Its Receptor? The Case of Melatonin
by
Jean A. Boutin and Jérôme Leprince
Receptors 2026, 5(2), 13; https://doi.org/10.3390/receptors5020013 - 15 Apr 2026
Abstract
Melatonin is reported to exert two types of actions: those based on its interaction with cognate receptors (characterized by very high affinities—1 nM and below), and those mediated by unknown targets (characterized by high concentrations—100 µM and above). Whereas receptor-mediated activities are known
[...] Read more.
Melatonin is reported to exert two types of actions: those based on its interaction with cognate receptors (characterized by very high affinities—1 nM and below), and those mediated by unknown targets (characterized by high concentrations—100 µM and above). Whereas receptor-mediated activities are known to regulate circadian rhythm, the high-dose effects are reported to be independent of these receptors and to produce literally dozens of beneficial effects in almost all human diseases, including cancer, neurodegenerative disorders, viral infections, obesity and many others. In the present opinion paper, we discuss this extensive set of claims and place them in perspective with a sum of evidence that collectively challenges the validity of these alleged beneficial effects.
Full article
Open AccessReview
From the Plate to the Nucleus: Dietary Control of Nuclear Receptors in the Development and Prevention of Metabolic Diseases
by
Ivan Torre-Villalvazo, Claudia Tovar-Palacio, Andrea Díaz-Villaseñor and Berenice Palacios-González
Receptors 2026, 5(2), 12; https://doi.org/10.3390/receptors5020012 - 9 Apr 2026
Abstract
►▼
Show Figures
Nutrient-sensing nuclear receptors (NSNRs), including PPARs, FXR, LXRs, RAR/RXR, VDR, and related orphan receptors, integrate a molecular interface that allows diet to communicate directly with the genome. By binding fatty acids, bile acids, sterols, vitamins, polyphenols, and other food-derived metabolites, NSNRs translate qualitative
[...] Read more.
Nutrient-sensing nuclear receptors (NSNRs), including PPARs, FXR, LXRs, RAR/RXR, VDR, and related orphan receptors, integrate a molecular interface that allows diet to communicate directly with the genome. By binding fatty acids, bile acids, sterols, vitamins, polyphenols, and other food-derived metabolites, NSNRs translate qualitative and quantitative features of the diet into coordinated transcriptional programmes across metabolically active organs. This ligand-dependent signalling network integrates dietary information to orchestrate inter-organ lipid and glucose metabolism, mitochondrial function, thermogenesis, and immune response, thereby enabling the organism to adapt dynamically to fasting–feeding cycles. In this review, we synthesise current evidence on the integrated roles of major NSNRs in the liver, skeletal muscle, white and brown adipose tissue, and kidney, emphasising how receptor networks within and between metabolic organs collectively govern energy expenditure, substrate partitioning, and systemic metabolic flexibility. We propose a conceptual framework in which diet functions as an “external endocrine organ”, acting as the primary source of chemically diverse NSNR ligands, while metabolic tissues serve as secondary signal amplifiers and integrators. Through circulating lipid species, bile acids, oxysterols, and other metabolites, these organs engage in continuous bidirectional communication that reprograms NSNR activity across tissues. We then examine how the global shift from minimally processed, nutrient-rich foods to nutrient-poor, energy-dense ultra-processed diets leads to a reduction in NSNR ligand diversity, promoting hepatic steatosis, muscle metabolic inflexibility, adipose tissue dysfunction, renal lipotoxicity, and chronic low-grade inflammation, ultimately causing obesity, type 2 diabetes, and cardiometabolic disease. Finally, we explore strategies to restore NSNR function, including Mediterranean and plant-based dietary patterns, as well as diets enriched with ω-3 polyunsaturated fatty acids, monounsaturated fats, and polyphenols. By integrating molecular, physiological, and clinical evidence, this review aims to clarify how NSNR networks translate dietary cues into coordinated inter-organ metabolism and how nutrient-poor diets lead to metabolic diseases trough a loss of metabolic information, rather than merely by energy excess. This framework supports a paradigm shift from calorie-centred nutrition to diet quality as the main therapeutic target for preventing metabolic diseases and promoting health.
Full article

Graphical abstract
Open AccessReview
FAM3 Cytokine-like Proteins, Their Putative Receptors and Signaling Pathways in Metabolic Diseases and Cancers
by
José E. Belizário, Izabela D. S. Caldeira, Bruna Moreira, João Marcelo Occhiucci, Brant Burkhardt and Humberto M. Garay-Malpartida
Receptors 2026, 5(2), 11; https://doi.org/10.3390/receptors5020011 - 30 Mar 2026
Abstract
►▼
Show Figures
FAM3A, FAM3B, FAM3C and FAM3D are members of the “family with sequence similarity 3” (FAM3) gene family, an emerging class of cytokine-like proteins with a unique structural globular β-β-α fold and distinct biological functions. With widespread expression in tissue, organs and in many
[...] Read more.
FAM3A, FAM3B, FAM3C and FAM3D are members of the “family with sequence similarity 3” (FAM3) gene family, an emerging class of cytokine-like proteins with a unique structural globular β-β-α fold and distinct biological functions. With widespread expression in tissue, organs and in many cell types, their specific roles in human diseases have been the focus of much research. FAM3A acts as a positive regulator of metabolic health, typically activating canonical pro-survival and metabolic pathways. FAM3B, also called PANDER (PANcreatic DERived Factor), exerts critical physiological functions in the regulation of glycemic levels via promotion of hepatic glucose production and pancreatic β-cell insulin secretion. FAM3C, also named ILEI (Interleukin-like EMT inducer), is involved as an inducer of epithelial–mesenchymal transition (EMT) and cancer metastasis, as well as osteoblast differentiation and bone mineralization. FAM3D is a gut-secreted protein and potential regulator of gastrointestinal homeostasis and microbiota-induced inflammation. Here we provide an overview of previous studies supporting that FAM3 proteins act through putative membrane receptors and co-partners, including fibroblast growth factor receptor (FGFR), leukemia inhibitory factor receptor (LIFR), formyl peptide receptor (FPR1/2), to activate diverse downstream signaling pathways on different cellular contexts. Basic and clinical studies suggest that the FAM3 family influences both obesity, diabetes, and other metabolic disorders; thus, its expression may have diagnostic potential. The differential and often cancer-specific expression patterns make members of the FAM3 family promising candidates for biomarkers and therapeutic targets of some types of neoplasia.
Full article

Figure 1
Open AccessReview
Ion Channels as Targets of the Vitamin D Receptor: A Long Journey with a Promising Future
by
Verna Cázares-Ordoñez, Ramiro José González-Duarte, Michiyasu Ishizawa, Luis A. Pardo and Makoto Makishima
Receptors 2026, 5(2), 10; https://doi.org/10.3390/receptors5020010 - 26 Mar 2026
Cited by 1
Abstract
►▼
Show Figures
The vitamin D receptor (VDR) acts as both a nuclear transcription factor and a non-genomic mediator that regulates a broad spectrum of physiological processes beyond calcium and phosphate homeostasis. VDR plays an important role in the modulation of ion channels across multiple tissues,
[...] Read more.
The vitamin D receptor (VDR) acts as both a nuclear transcription factor and a non-genomic mediator that regulates a broad spectrum of physiological processes beyond calcium and phosphate homeostasis. VDR plays an important role in the modulation of ion channels across multiple tissues, including osteoblasts, renal and intestinal epithelial cells, neurons, and vascular smooth muscle. These regulatory mechanisms encompass genomic actions through vitamin D response elements in target genes—such as TRPV5, TRPV6, KCNK3, and KCNH1—as well as rapid, non-genomic actions at the plasma membrane involving protein disulfide isomerase A3 and associated signaling cascades. VDR-mediated transcriptional control of calcium, potassium, and chloride channels contributes to the fine-tuning of cellular excitability, calcium transport, and mitochondrial function. Evidence also implicates VDR–ion channel crosstalk in various pathological contexts, including renal cell carcinoma, breast and cervical cancers, pulmonary arterial hypertension, and osteoporosis. Understanding the molecular interplay between VDR and ion channels provides new perspectives on the pleiotropic effects of vitamin D and offers promising therapeutic opportunities in oncology, cardiovascular disease, and skeletal disorders. This review synthesizes previous and current evidence on the genomic and non-genomic mechanisms underlying VDR–ion channel regulation and highlights novel frontiers in vitamin D signaling relevant to human health and disease.
Full article

Graphical abstract
Open AccessReview
Function and Modulation of Sphingosine-1-Phosphate Receptors in the Central Nervous System
by
Elizabeth Gulliksen, Sriya Darsi, Ladan Haidarbaigi, Lucas J. Codispoti, Devam Purohit, Ashley Jung, Aishwarya Chilamula and Jason Newton
Receptors 2026, 5(1), 9; https://doi.org/10.3390/receptors5010009 - 17 Mar 2026
Abstract
►▼
Show Figures
Sphingolipids, first discovered in 1874 by Johann Thudicum, are among the eight recognized classes of lipids and are present in essentially all plants, animals, and fungi, as well as some viruses and prokaryotes. In mammals, sphingolipids are enriched in the central nervous system
[...] Read more.
Sphingolipids, first discovered in 1874 by Johann Thudicum, are among the eight recognized classes of lipids and are present in essentially all plants, animals, and fungi, as well as some viruses and prokaryotes. In mammals, sphingolipids are enriched in the central nervous system (CNS), where they play vital roles in tissue development; membrane structure; cell adhesion and recognition; and, importantly, signaling. A subset of sphingolipids including ceramide, glucosylceramide, and sphingosine has been shown to have bioactive properties, but two sphingolipids in particular (ceramide-1-phosphate and sphingosine-1-phosphate) have been shown to exert their effects at least in part due to the activation of cell surface-expressed G protein-coupled receptors. In the CNS, sphingosine-1-phosphate signaling has specifically emerged as a productive therapeutic target for the treatment of neurodegenerative disease, with the first small molecule targeting sphingosine-1-phosphate receptors approved roughly 15 years ago for the treatment of multiple sclerosis. As more specific activators and inhibitors of these receptors have been developed and entered the clinical trial pipeline, now is an appropriate time to examine the current state of our knowledge of the role that these receptors play in the CNS and highlight the current landscape of available modulators targeting these pathways.
Full article

Figure 1
Open AccessReview
The Role of Progesterone in the Reproductive Physiology of Females of Viviparous Squamata
by
Norma Berenice Cruz-Cano, Uriel Ángel Sánchez-Rivera, Carmen Álvarez-Rodríguez, Hibraim Adán Pérez-Mendoza and Martín Martínez-Torres
Receptors 2026, 5(1), 8; https://doi.org/10.3390/receptors5010008 - 27 Feb 2026
Abstract
►▼
Show Figures
Progesterone (P4) regulates diverse reproductive processes across vertebrates through nuclear receptors; however, its mechanisms in squamate reptiles—particularly in viviparous species—remain poorly understood. In Squamata, P4 primarily acts through progesterone receptor (PR) isoforms A and B, although relatively few reptilian PR
[...] Read more.
Progesterone (P4) regulates diverse reproductive processes across vertebrates through nuclear receptors; however, its mechanisms in squamate reptiles—particularly in viviparous species—remain poorly understood. In Squamata, P4 primarily acts through progesterone receptor (PR) isoforms A and B, although relatively few reptilian PR sequences have been characterized to date. Squamate PR exhibits ~50% overall sequence divergence from mammalian homologs yet retains striking conservation in both the ligand and DNA-binding domain across vertebrates. Despite the broadly conserved physiological roles of P4 (folliculogenesis, ovulation, courtship behavior, pregnancy maintenance, and parturition/oviposition), P4 dynamics in viviparous squamates remain unresolved due to heterogeneous circulating hormone concentrations and limited PR phylogeny and structure studies. While mammalian models dominate P4 research due to their biomedical relevance, squamates offer unique evolutionary insights: as the only reptile order exhibiting both oviparity and viviparity within the same clade, squamates represent an ideal model for investigating transitions in parity mode. Elucidating P4 mechanisms in squamates will help bridge this critical evolutionary gap, with important implications for reproductive biology and conservation.
Full article

Figure 1
Open AccessReview
Coevolution Between Three-Finger Toxins and Target Receptors
by
Jéssica Lopes de Oliveira and Henrique Roman-Ramos
Receptors 2026, 5(1), 7; https://doi.org/10.3390/receptors5010007 - 14 Feb 2026
Abstract
►▼
Show Figures
Background: Three-finger toxins (3FTxs) are a major axis of functional diversification in advanced snake venoms, with canonical paralytic activity mediated through muscle-type nicotinic acetylcholine receptors (nAChRs) and a broader set of non-nicotinic targets. This review integrates evidence bearing on coevolution between 3FTxs
[...] Read more.
Background: Three-finger toxins (3FTxs) are a major axis of functional diversification in advanced snake venoms, with canonical paralytic activity mediated through muscle-type nicotinic acetylcholine receptors (nAChRs) and a broader set of non-nicotinic targets. This review integrates evidence bearing on coevolution between 3FTxs and target receptors, spanning toxin origin, diversification, receptor evolution, and ecological context. Methods: The synthesis draws on comparative genomic and transcriptomic studies of 3FTx gene-family evolution, codon-model analyses of selection, structural characterisation of toxin–receptor interfaces, and functional assays (including receptor-mimicking peptide binding) that link sequence variation to binding and toxicity. Results: Across lineages, 3FTx diversification is repeatedly structured by strong constraint on the disulphide-rich scaffold with accelerated change concentrated in solvent-exposed loops, alongside birth–death dynamics and exon/segment-level innovation that expand binding specificity. On the receptor side, resistance-associated variation is most intensively characterised for the nAChR α1 orthosteric site and includes convergent, mechanistically distinct solutions such as electrostatic repulsion and glycosylation-mediated steric interference. Within the predominantly elapid systems currently examined, integrative datasets indicate that prey-selective binding and geographically variable susceptibility can arise from modest substitutions at toxin–receptor interfaces, but they also reveal substantial taxonomic and target-specific biases. Conclusions: Current evidence supports adaptive diversification in both toxins and receptors, while broader evolutionary interpretations are limited by uneven sampling and the frequent lack of matched toxin and receptor variants analysed within a common evolutionary framework. Development of predictive models will require joint pipelines linking genomics, structure-informed evolutionary inference, scalable functional assays, and explicit ecological network context.
Full article

Graphical abstract
Open AccessArticle
Essential Envelope Spike Motifs for Cell Entry of Transmissible Gastroenteritis Virus and Its Evolution in Coronavirus
by
Gaurav Mudgal, Fernando Almazán, Tadeo Moreno-Chicano, Alberto Martínez-Colom, Cesar Santiago, Luis Enjuanes and José M. Casasnovas
Receptors 2026, 5(1), 6; https://doi.org/10.3390/receptors5010006 - 30 Jan 2026
Abstract
►▼
Show Figures
Background: Transmissible gastroenteritis virus (TGEV), a coronavirus (CoV) infecting pigs, uses its spike (S) glycoprotein to bind porcine aminopeptidase N (pAPN) for cell entry. Although structural studies have identified receptor-binding motifs (RBMs) within the receptor-binding domain (RBD) of the S protein, the
[...] Read more.
Background: Transmissible gastroenteritis virus (TGEV), a coronavirus (CoV) infecting pigs, uses its spike (S) glycoprotein to bind porcine aminopeptidase N (pAPN) for cell entry. Although structural studies have identified receptor-binding motifs (RBMs) within the receptor-binding domain (RBD) of the S protein, the functional relevance of individual residues for TGEV receptor recognition, cell entry, and infection remain unclear. Methods: In this study, we performed structure-guided mutagenesis of the TGEV RBD to evaluate the contribution of specific residues to receptor binding and viral infectivity. Results: Using soluble RBD proteins, we found that most of the RBD residues within the pAPN-binding interface contribute to the binding interaction. Nonetheless, TGEV reverse genetics experiments revealed that just three RBD residues (Gly527, Tyr528, and Trp571) were indispensable for viral cell entry. Mutations at these positions, which are conserved among group 1 alpha-CoVs abolished infectivity, highlighting their central role in the virus–receptor interface. Conclusions: Our findings provide a detailed functional map of the TGEV RBD and offer insights into the evolution of receptor recognition across CoV.
Full article

Graphical abstract
Open AccessReview
Exploring Nuclear Receptor Functions in Multipotent Mesenchymal Stromal Stem Cell Differentiation
by
Alivia Dougherty and Bruce Blumberg
Receptors 2026, 5(1), 5; https://doi.org/10.3390/receptors5010005 - 19 Jan 2026
Abstract
►▼
Show Figures
Multipotent mesenchymal stromal stem cells have captivated the scientific community in recent years due to their ability to differentiate into multiple adult cell types. Central to this potential are many members of the nuclear hormone receptor superfamily, comprising 48 ligand-modulated transcription factors involved
[...] Read more.
Multipotent mesenchymal stromal stem cells have captivated the scientific community in recent years due to their ability to differentiate into multiple adult cell types. Central to this potential are many members of the nuclear hormone receptor superfamily, comprising 48 ligand-modulated transcription factors involved in key biological processes such as metabolism, physiology, embryonic development, and reproduction. These transcription factors influence cellular fate by regulating gene expression networks critical for MSC specification, commitment, and differentiation. This review explores the role of nuclear receptors in MSC development, focusing on interactions with chromatin structure, co-regulatory complexes, and responsiveness to extracellular stimuli such as hormones, metabolic cues, and endocrine-disrupting chemicals. We conclude with a discussion of the dangers posed by exogenous and aberrant signaling through nuclear receptors.
Full article

Figure 1
Open AccessReview
The GLI3–Androgen Receptor Axis: A Feedback Circuit Sustaining Shh Signaling in Prostate Cancer
by
Stephanie I. Nuñez-Olvera, Enoc Mariano Cortés-Malagón, Isela Montúfar-Robles, José Javier Flores-Estrada, María Elizbeth Alvarez-Sánchez and Jonathan Puente-Rivera
Receptors 2026, 5(1), 4; https://doi.org/10.3390/receptors5010004 - 19 Jan 2026
Abstract
►▼
Show Figures
The Hedgehog (Hh) signaling pathway regulates key cellular processes, such as proliferation, differentiation, and morphogenesis. Although its canonical activation involves ligand binding to PTCH1, which activates Smoothened (SMO), noncanonical features of the pathway significantly contribute to cancer progression, particularly in prostate cancer (PCa).
[...] Read more.
The Hedgehog (Hh) signaling pathway regulates key cellular processes, such as proliferation, differentiation, and morphogenesis. Although its canonical activation involves ligand binding to PTCH1, which activates Smoothened (SMO), noncanonical features of the pathway significantly contribute to cancer progression, particularly in prostate cancer (PCa). GLI3, a central transcription factor in the Hh pathway, can act as a repressor or activator depending on posttranslational modifications. In androgen-deprived PCa, GLI3 plays a critical role in driving castration-resistant phenotypes by interacting with the androgen receptor (AR), particularly the AR-V7 variant. This interaction enhances tumor survival and growth even under androgen deprivation therapy (ADT). Aberrant GLI3 activity is further driven by mutations in upstream regulators such as SPOP and MED12, which contribute to the progression of both prostate and other malignancies. Preclinical studies have shown promise in reducing tumor cell proliferation and migration, and in inducing apoptosis, by pharmacologically inhibiting the GLI3 pathway with SMO antagonists or GSK3β inhibitors. Recent evidence also highlights reciprocal interactions between Sonic Hedgehog (Shh) signaling and the AR that sustain tumor growth under ADT. GLI3 engagement with AR reinforces AR-dependent transcription, supporting tumor progression through noncanonical pathways. These findings suggest that targeting GLI3, particularly in combination with AR inhibition, could effectively overcome castration resistance and improve outcomes in patients with castration-resistant prostate cancer (CRPC). This review explores the role of GLI3 in both canonical and noncanonical Hh signaling, its potential as a therapeutic target, and future directions for overcoming resistance in Hh-driven cancers.
Full article

Figure 1
Open AccessReview
NR4A Receptors in Immunity: Bridging Neuroendocrine and Inflammatory Pathways
by
Simone Lemes Ferreira and Natalia Santucci
Receptors 2026, 5(1), 3; https://doi.org/10.3390/receptors5010003 - 25 Dec 2025
Abstract
►▼
Show Figures
Nuclear receptors (NRs) are ligand-activated transcription factors that mediate diverse cellular processes, including signalling, survival, proliferation, immune response and metabolism, through both genomic and non-genomic mechanisms in response to hormones and metabolic ligands. Given their central role in inter-organ, tissue, and cellular communication,
[...] Read more.
Nuclear receptors (NRs) are ligand-activated transcription factors that mediate diverse cellular processes, including signalling, survival, proliferation, immune response and metabolism, through both genomic and non-genomic mechanisms in response to hormones and metabolic ligands. Given their central role in inter-organ, tissue, and cellular communication, NRs are critical for maintaining homeostasis and have become a major focus in biomedical research and drug discovery due to their association with numerous diseases. Among NRs, the NR4A subfamily (NR4A1/Nur77, NR4A2/Nurr1, and NR4A3/Nor1) responds to various stimuli—such as insulin, growth factors, inflammatory cytokines, and β-adrenergic signals—though their endogenous ligands remain unidentified. Their expression is tissue-dependent, particularly in energy-demanding tissues, where they modulate leukocyte function and promote an anti-inflammatory profile. Like other NRs, NR4As regulate acute and chronic inflammation by suppressing pro-inflammatory transcription factors (e.g., NF-κB) or enhancing their inhibitors, thereby polarising macrophages toward an anti-inflammatory phenotype. This review summarises current knowledge on the role of NR4A receptors in immune responses. Given their well-documented involvement in autoimmune diseases, inflammatory conditions, and cancer, elucidating their contributions to neuro–immune–endocrine crosstalk may uncover their therapeutic potential for immunopathological disorders.
Full article

Figure 1
Open AccessReview
Astrocytic Receptor Systems of the Basal Ganglia
by
Aleksandar Tushevski, Linus Happe, Elena Stocco, Raffaele De Caro, Veronica Macchi, Andrea Porzionato and Aron Emmi
Receptors 2026, 5(1), 2; https://doi.org/10.3390/receptors5010002 - 23 Dec 2025
Abstract
►▼
Show Figures
Astrocytes are increasingly recognized as active participants of synaptic communication, yet their role in the basal ganglia circuitry remains poorly defined. Emerging evidence indicates that astrocytes in this region express a diverse array of neurotransmitter receptors thought to regulate intracellular calcium signaling, gliotransmitter
[...] Read more.
Astrocytes are increasingly recognized as active participants of synaptic communication, yet their role in the basal ganglia circuitry remains poorly defined. Emerging evidence indicates that astrocytes in this region express a diverse array of neurotransmitter receptors thought to regulate intracellular calcium signaling, gliotransmitter release, synaptic plasticity, and neuroimmune responses. However, the literature is limited by methodological variability and a pronounced focus on the striatum, with comparatively little data on other basal ganglia nuclei. This review aims to organize the current literature on astrocytic receptor systems within the basal ganglia, including dopaminergic (D1–D5), glutamatergic (AMPA, NMDA, mGluRs), GABAergic (GABA-A, GABA-B), purinergic (P1, P2), and adrenergic (α, β) receptors. By organizing receptor-specific findings across basal ganglia structures, this review provides a foundation for future investigations into astrocytic function in this complex neural network.
Full article

Figure 1
Open AccessArticle
Distribution and Levels of Insulin-like Growth Factor 2 Receptor Across Mouse Brain Cell Types
by
Jessica R. Gaunt, Gokul Manoj and Cristina M. Alberini
Receptors 2026, 5(1), 1; https://doi.org/10.3390/receptors5010001 - 23 Dec 2025
Cited by 1
Abstract
►▼
Show Figures
Background: The insulin-like growth factor 2 receptor (IGF-2R), also known as the cation-independent mannose 6-phosphate receptor (CI-M6PR), is emerging as a critical receptor for brain function and disease. IGF-2R, in fact, plays a key role in long-term memory, and its activation by several
[...] Read more.
Background: The insulin-like growth factor 2 receptor (IGF-2R), also known as the cation-independent mannose 6-phosphate receptor (CI-M6PR), is emerging as a critical receptor for brain function and disease. IGF-2R, in fact, plays a key role in long-term memory, and its activation by several ligands shows beneficial effects in multiple neurodevelopmental and neurodegenerative disease models. Thus, its targeting is very promising for neuropsychiatric therapeutic interventions. IGF-2R’s main known functions are transport of lysosomal enzymes and regulation of developmental tissue growth, but in the brain, it also controls learning-dependent protein synthesis underlying long-term memory. However, little is known about this receptor in brain cells, including its cell-type-specific and subcellular expression. Methods: We conducted a comprehensive investigation to comparatively assess IGF-2R protein levels in different brain cell types across various brain regions in adult male C57BL/6J mice using dual and multiplex immunofluorescent staining with cell-type-specific markers. The IGF-2R protein distribution was also compared with Igf2r mRNA expression in publicly available single-cell RNA sequencing databases. Results: A ranking of IGF-2R levels in the soma of various cell types in the hippocampus and cortical regions revealed that the highest enrichment is, by far, in excitatory and inhibitory neurons, followed by vascular mural cells and subpopulations of oligodendrocyte lineage cells, with low to undetectable levels in astrocytes, microglia, vascular endothelial cells, and perivascular fibroblasts. High levels of IGF-2R were also found in ependymal cells, choroid plexus epithelial cells, and a subpopulation of meningeal fibroblast-like cells. IGF-2R was found in dendritic and putative axonal compartments throughout the brain, with particularly high levels in the stratum lucidum. The receptor’s protein distribution aligned with that of the mRNA in mouse brain databases. Conclusions: These results suggest that IGF-2R-mediated functions in the brain vary across different cell types and subcellular compartments, with the most active roles in specific subpopulations of neurons, mural cells, ependymal cells, meningeal cells, and cells of the oligodendrocyte lineage. This study advances our understanding of IGF-2R’s distribution in the brain, which is essential for formulating new hypotheses about its functions and therapeutic targeting.
Full article

Figure 1
Open AccessReview
Role of Transient Receptor Potential Vanilloid Channels in Gastrointestinal Physiology and Pathology
by
Omar Tluli, Ahmed Arabi, Humam Emad Rajha, Lana Abugharbieh, Faissal Al Zeir, Maryam Hamdan, Ayeda Abuqaba and Ammar Boudaka
Receptors 2025, 4(4), 24; https://doi.org/10.3390/receptors4040024 - 10 Nov 2025
Abstract
►▼
Show Figures
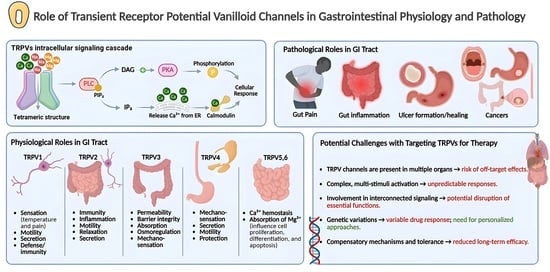
Transient Receptor Potential Vanilloid (TRPV) channels represent one of the seven subfamilies of TRP receptors and are widely expressed throughout the human body where they play pivotal roles in various physiological processes. In the gastrointestinal (GI) system, TRPV channels regulate critical functions such
[...] Read more.
Transient Receptor Potential Vanilloid (TRPV) channels represent one of the seven subfamilies of TRP receptors and are widely expressed throughout the human body where they play pivotal roles in various physiological processes. In the gastrointestinal (GI) system, TRPV channels regulate critical functions such as nutrient absorption, motility, and secretions. Beyond maintaining cellular homeostasis, these channels are involved in pain and inflammation, contributing to diverse pathologies. Their central role in the pathophysiology of different digestive system disorders has made TRPV channels a significant focus of research. Moreover, the involvement of TRPV channels in numerous GI cancers has further heightened research interest in the role of these channels. Accordingly, this review elucidates the structural components and intricate signaling pathways of TRPV channels, focusing on the unique characteristics of each family member (TRPV1–6) in GI physiology. Furthermore, we explore the therapeutic potential of targeting these channels to modulate their physiological and pathological roles, highlighting their promise in treating GI disorders. Additionally, we address the challenges associated with their therapeutic application, considering their interactions in different systems, inherent biochemical characteristics, and the alterations required for effective design.
Full article
Graphical abstract
Open AccessReview
Neurokinin-1 Receptor Regulation of Fibroblast Phenotype and Function
by
Scott P. Levick
Receptors 2025, 4(4), 23; https://doi.org/10.3390/receptors4040023 - 6 Nov 2025
Abstract
Injury to tissue induces the normal wound healing process to repair damage. This is a normal and critical response developed by the body to maintain short-term organ function, and therefore, survival. Should this process become aberrant, then fibrosis can develop. Fibrosis is the
[...] Read more.
Injury to tissue induces the normal wound healing process to repair damage. This is a normal and critical response developed by the body to maintain short-term organ function, and therefore, survival. Should this process become aberrant, then fibrosis can develop. Fibrosis is the excess accumulation of extracellular matrix proteins. Unlike normal wound healing that is designed to maintain organ/tissue function, fibrosis interferes with the normal architecture of the organ and has long-term functional implications. Fibroblasts are the cells responsible for producing extracellular matrix in both wound healing and fibrosis. Substance P is the cognate ligand for the neurokinin-1 receptor, and both substance P and the neurokinin-1 receptor have been demonstrated to be involved in organ remodeling; this includes regulation of fibroblast function. In this review we will focus on substance P/neurokinin-1 receptor regulation of fibroblast function in the setting of both wound healing and fibrosis. This review describes actions of substance P and the neurokinin-1 receptor on fibroblasts from multiple organs, thus identifying central actions common to all fibroblasts studied. This review also identifies gaps in the literature and future directions needed to improve understanding of substance P and the neurokinin-1 receptor regulation of fibroblast phenotype.
Full article
(This article belongs to the Special Issue Biological and Pharmacological Aspects of the Neurokinin-1 Receptor)
►▼
Show Figures

Graphical abstract
Open AccessReview
Neuroprotective Role of Cannabinoids in Retinal Disease
by
George Ayoub
Receptors 2025, 4(4), 22; https://doi.org/10.3390/receptors4040022 - 4 Nov 2025
Abstract
Cannabinoids, compounds that interact with the endocannabinoid system, have shown promising neuroprotective effects in various neurodegenerative diseases, including those affecting the retina. This review evaluates evidence for the presence and action of cannabinoids in the retina, their function in protecting against oxidative stress
[...] Read more.
Cannabinoids, compounds that interact with the endocannabinoid system, have shown promising neuroprotective effects in various neurodegenerative diseases, including those affecting the retina. This review evaluates evidence for the presence and action of cannabinoids in the retina, their function in protecting against oxidative stress and modulating neuroinflammation, and the outcomes observed in animal models of retinal diseases such as glaucoma and age-related macular degeneration (AMD), the most common causes of vision loss. Cannabinoids have proven effective in reducing the neurodegeneration seen in these eye diseases, acting via the CB1 and CB2 cannabinoid receptors. The cannabinoid neuroprotective effect is often of a similar magnitude to the other proven therapy of medical dosage of vitamins, though it confers a greater risk due to neurotoxicity with high THC:CBD ratios, making the vitamin therapy of greater efficacy when time is available. Given the increased ratio of THC:CBD in commercial cannabis strains, rising from 10:1 at the beginning of this century to 100:1 now, the risk of neurotoxicity has increased, reducing the neuroprotective benefit. The proven safety and efficacy of vitamin therapy may be a more viable neuroprotective method than cannabinoid use for chronic conditions, with cannabinoids proving their utility in more acute conditions. This review evaluates both the method of action of cannabinoids and the receptor pathway utilized and compares the suggested therapeutic applicability of cannabinoids with proven vitamin therapy.
Full article
(This article belongs to the Special Issue Understanding Cannabinoid Receptor Signaling Complexity: Keys for Improved Therapeutic Drug Development)
►▼
Show Figures

Graphical abstract
Open AccessReview
Impaired Hemostasis and the Role of Oxidative Stress-Induced Signaling in Thrombotic Disorders—Where We Stand Today
by
Sofia K. Georgiou-Siafis and Paraskevi Kotsi
Receptors 2025, 4(4), 21; https://doi.org/10.3390/receptors4040021 - 31 Oct 2025
Abstract
►▼
Show Figures
Both primary and secondary hemostasis consist of finely regulated pathways, forming a blood clot to stop bleeding. These orchestrated mechanisms involve multiple plasma- and platelet/endothelial-derived receptors, factors, enzymes, and proteins, such as the von Willebrand factor (vWF), fibrinogen, and thrombin. Over-activation or improper
[...] Read more.
Both primary and secondary hemostasis consist of finely regulated pathways, forming a blood clot to stop bleeding. These orchestrated mechanisms involve multiple plasma- and platelet/endothelial-derived receptors, factors, enzymes, and proteins, such as the von Willebrand factor (vWF), fibrinogen, and thrombin. Over-activation or improper resolution of the coagulation cascade leads to severe pathological disorders, arterial and venous. Despite the fact that the genetic etiology of thrombophilia has gained the main research interest, there is growing evidence that the disturbed redox network of key hemostatic pathways signals thrombus formation. Oxidized LDL in dyslipidemias and many endogenous and exogenous compounds act as pro-oxidant stimuli that lead to post-translational modifications of proteins, such as sulfenylation, nitrosation, disulfide formation, glutathionylation, etc. Oxidation of cysteine and methionine residues of vWF, fibrinogen, and thrombomodulin has been detected at thrombotic episodes. Increased homocysteine levels due to, but not restricted to, methylenetetrahydrofolate reductase gene (MTHFR) mutations have been incriminated as a causative factor for oxidative stress, leading to a pro-thrombotic phenotype. Alterations in the vascular architecture, impaired vascular relaxation through decreased bioavailability of NO, accumulation of Nε-homocysteinylated proteins, ER stress, and endothelial cells’ apoptosis are among the pro-oxidant mechanisms of homocysteine. This review article focuses on describing key concepts on the oxidant-based molecular pathways that contribute to thrombotic episodes, with emphasis on the endogenous compound, homocysteine, aiming to promote further molecular, clinical, and pharmacological research in this field.
Full article

Graphical abstract
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Receptors
Biological and Pharmacological Aspects of the Neurokinin-1 Receptor
Guest Editors: Rafael Coveñas Rodríguez, Miguel MuñozDeadline: 30 June 2026
Special Issue in
Receptors
Understanding Cannabinoid Receptor Signaling Complexity: Keys for Improved Therapeutic Drug Development
Guest Editor: Paul L. PratherDeadline: 30 June 2026
Topical Collections
Topical Collection in
Receptors
Receptors: Exceptional Scientists and Their Expert Opinions
Collection Editor: Stephen H. Safe