- Article
Verifying Quantum Network Nonlocality Based on the Extended Mermin Inequality
- Xinyue Li,
- Yan-Han Yang and
- Ming-Xing Luo
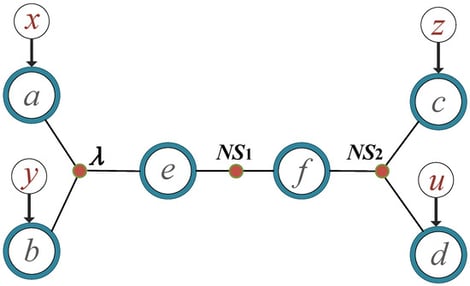
This work proposes an extended Mermin inequality based on a hybrid classical model that involves only one classical source, with the remaining sources being post-quantum. In a chain-structured quantum network consisting of hybrid Einstein–Podolsky–Rosen (EPR) pairs and Greenberger–Horne–Zeilinger (GHZ) states, joint measurements are performed at the central node, while local measurements are conducted at the peripheral nodes. This setup shows that the obtained quantum correlations can violate the proposed inequality with fewer measurement settings, thereby verifying network nonlocality. Furthermore, we extend this method to chain networks of arbitrary length n and show that the proposed inequality remains effective in verifying network nonlocality.
1 May 2026