- Article
Enhancing the Detection of Long-Chain Aldehydes by Peptide-Based Biosensors Through Counter-Ion Exchange
- Tomasz Wasilewski,
- Damian Neubauer and
- Marek Wojciechowski
- + 5 authors
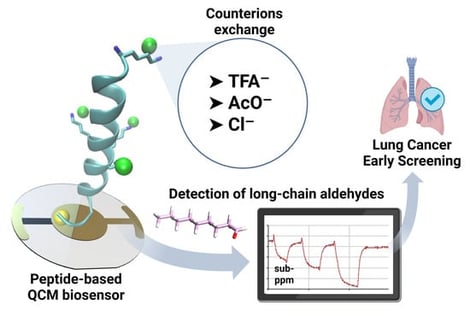
Long-chain aldehydes, particularly nonanal, are recognized as potential volatile biomarkers of lung cancer in exhaled breath. This study investigates the influence of peptide counter-ions on the performance of QCM-based biosensors using two odorant-binding protein-derived peptides (OBPP4 and OBPP4 GSGSGS) for the selective gas-phase detection of these aldehydes. Exchanging the counter-ion from trifluoroacetate to chloride improves biosensor sensitivity and lowers the limit of detection within the set of biosensors investigated in this study. The OBPP4 GSGSGS with chloride exhibited the highest sensitivity to nonanal (0.153 Hz/ppm) and the lowest LOD (9.8 ppm), with excellent selectivity over other groups of volatiles. The novelty of this work lies in demonstrating, for the first time, that simple counter-ion exchange in synthetic peptides can significantly enhance the gas-phase binding of volatile aldehydes, classified as lung cancer biomarkers, without altering the peptide sequence, offering a straightforward and effective optimization strategy for peptide-based piezoelectric biosensors.
13 March 2026



![Schematic illustration of target-induced SDA to activate DNA nanotube for cleaving the immobilized probe (a) and machine learning analysis for AD diagnosis (b). Reprinted with permission from ref. [62]. Copyright 2025 Elsevier B.V.](https://mdpi-res.com/cdn-cgi/image/w=281,h=192/https://mdpi-res.com/biosensors/biosensors-16-00161/article_deploy/html/images/biosensors-16-00161-g001-550.jpg)