Abstract
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) is a powerful technique for polymer identification because it can ionize intact macromolecules with large molecular weights (>100 kDa) and detect them over a theoretically unlimited mass range. However, limited mass resolution, accuracy, and sensitivity in the high-mass region restrict detailed structural characterization of polymers. Depolymerization into smaller fragments using established chemistry, such as transesterification, provides a versatile alternative approach. The resulting characteristic fragments (<3 kDa) are polymer-specific and enable both polymer identification and detailed structural analysis, benefiting from the higher mass accuracy and sensitivity in the low-mass range and allowing MS/MS-based identification. To establish general sample-preparation conditions, this study systematically investigated the effects of transesterification of biodegradable polyesters using MeO−/MeOH, EtO−/EtOH, and t-BuO−/t-BuOH on subsequent MALDI-TOF MS analysis. Among the reagents examined, EtO−/EtOH combined with THAP/THF matrix treatment was found to be the most efficient, enabling rapid polyester analysis by MALDI-TOF MS within 10 min when the reaction time and reagent concentration were carefully controlled within an optimal window.
1. Introduction
Over the past several decades, the widespread production and disposal of synthetic plastics have resulted in the accumulation of plastic waste and increasing environmental pollution [1]. Micro- and nano-plastics originating from mismanaged plastic waste, synthetic textile fibers, worn tires, and cosmetic microbeads pose serious risks to aquatic and terrestrial ecosystems, and potentially to human health [2]. Accordingly, biodegradable plastics have attracted increasing attention as potential replacements for conventional synthetic plastics in recent years [3,4]. However, if appropriate environmental conditions for degradation are not met, biodegradable plastics may also persist for long periods and fragment into microplastics during aging and partial biodegradation, similar to conventional plastics [4,5,6,7]. The environmental occurrence of biodegradable microplastics also poses potential impacts on ecosystems and human health. These issues highlight the need for analytical methods to detect and identify biodegradable plastics in environmental matrices.
Among the various analytical approaches, physical methods (such as transmission electron microscopy, scanning electron microscopy, and gravimetric analysis), respirometric tests, and thermal methods (differential scanning calorimetry and thermogravimetric analysis) can only observe physical or bulk changes in the samples. They do not provide direct information on the polymer type or on detailed chemical structures [8,9]. Gel permeation chromatography (GPC) separates polymers according to their hydrodynamic volume in solution and is widely used to determine molar mass distributions. However, it cannot resolve differences in detailed molecular structures [10].
Among spectroscopic methods, nuclear magnetic resonance spectroscopy (NMR) is a powerful tool for the structural characterization of biodegradable polymers. Whereas NMR can offer detailed and often quantitative information on polymer structure, it requires relatively large amounts of well-dissolved material and often suffers from signal overlap in complex oligomer mixtures [10,11,12,13]. Spectroscopic techniques such as Fourier transform infrared and Raman spectroscopy, often combined with microscopy, provide opportunities to characterize microplastics through vibrational fingerprinting. However, this approach offers limited reliability for polymer identification and therefore requires further development [14,15].
Among mass spectrometry (MS)-based analytical approaches, liquid chromatography (LC)–MS and gas chromatography (GC)–MS have been applied to analyze degradation products of polymers, short oligomers, and additives [16,17,18]. However, LC–MS generally requires well-soluble analytes that are compatible with the mobile phases and offers limited direct information on broader oligomer distributions [16]. Conventional GC–MS is restricted to volatile, thermally stable, low-molecular-weight compounds [17,18]. To overcome this limitation for polymer identification, pyrolysis (Py)–GC–MS is often used, in which polymers are thermally decomposed into characteristic volatile fragments before GC–MS analysis. Py–GC–MS has become a widely used technique for identifying polymer types and small degradation products, because the resulting pyrograms and electron ionization mass spectra provide characteristic fingerprints of volatile pyrolysis products derived from plastic polymers [19,20,21]. However, due to complete breakdowns of oligomeric polymer structures during Py, so Py–GC–MS mainly gives information at the level of small molecules rather than intact chains or oligomer series [21,22]. These chromatographic methods are relatively time-consuming, require careful optimization of chromatographic and ionization conditions, and are less practical for rapid screening of multiple samples or complex mixtures [16,18,21,23].
Among the available MS techniques, MALDI-TOF (matrix-assisted laser desorption/ionization time-of-flight) MS is especially attractive for polymer analysis [24,25,26,27]. It can record mass spectra over a broad mass range (>300 kDa) within a few seconds per sample and requires only sub-microgram amounts of material. It can also provide number- and weight-average molar masses (Mn and Mw), although the accuracy of such determinations strongly depends on polymer dispersity (Đ) and ionization behavior [27]. These capabilities make it well suited for studying polymer architectures, copolymer compositions, and degradation pathways [23,24,27]. Despite these advantages, MALDI-TOF MS analysis of biodegradable polyesters still has several practical limitations [25]. Like other MS platforms, MALDI-TOF MS suffers from inadequate resolving power at very high molecular masses [24,28]. Industrial biodegradable plastics are typically used at weight-average molecular weights in the 104–105 g mol−1 range, and such high-molar-mass materials are not straightforward to analyze in their intact form for detailed structural characterization by MS and MS/MS [28,29]. In this context, the application of alkaline depolymerization through well-established transesterification chemistry, which degrades biodegradable polyester polymers into smaller fragments (<3 kDa), offers a practical strategy for MALDI-TOF MS characterization [30,31,32]. These low-molecular-weight products can be analyzed in a mass range where MS provides high mass resolution and accuracy, and they are well suited for detailed structural elucidation by tandem mass spectrometry (MS/MS), such as collision-induced dissociation. Despite its potential advantages, this strategy has been explored in only a limited number of reports. For example, MeO−/MeOH-mediated transesterification has been applied to poly(ε-caprolactone) (PCL), poly(3-hydroxybutyrate) (P3HB), poly(3-hydroxybutyrate-co-3-hydroxyvalerate) [P(3HB-co-3HV)], and polyethylene terephthalate [30]. Additional studies have reported its application to polyhydroxybutyrate (PHB) and P(3HB-co-3HV) [31], as well as to poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) [P(3HB-co-3HH)] [32]. Although MeO−-assisted depolymerization has previously been applied to the MALDI characterization of biodegradable polyesters, systematic comparison with alternative alkoxide systems across structurally distinct polymers remains largely unexplored.
In this study, we examined transesterification chemistry for MALDI MS characterization of polymers using a simple workflow in which high-molar-mass biodegradable polyesters are first subjected to controlled alkaline treatment to generate soluble polyester-derived fragments, and the resulting fragments are then analyzed by MALDI-TOF MS. This study examined commercial biodegradable P3HB, poly(butylene succinate) (PBS), and PCL as model substrates. More specifically, we systematically evaluated the transesterification behavior of commonly used alkoxide reagents (MeO−/MeOH, EtO−/EtOH, and t-BuO−/t-BuOH) under varying conditions. Despite their widespread use, their relative reaction efficiency, particularly with respect to their MALDI-compatible analytical performance, have not been comprehensively understood. This work aims to provide a rational basis for developing a generalized sample-preparation strategy for rapid and reliable MALDI-TOF MS characterization of biodegradable polymers.
2. Materials and Methods
2.1. Materials
P3HB (Danimer Scientific, Bainbridge, GA, USA), PBS (Ankor Bioplastics, Wonju-si, Gangwon, Republic of Korea), and PCL (Sigma-Aldrich, Co., St. Louis, MO, USA) were commercially obtained. According to the supplier specifications, PCL had a reported Mn of approximately 80,000 g/mol. Commercial chemicals including sodium hydroxide from Duksan Pure Chemicals Co. (Ansan-si, Gyeonggi, Republic of Korea), sodium methoxide (MeO−) from Tokyo Chemical Industry, Co. (Tokyo, Japan), and sodium tert-butoxide (t-BuO−) and sodium ethoxide (EtO−) from Sigma-Aldrich, Co. were utilized. Methanol (MeOH) and ethanol (EtOH) were purchased from Samchun Chemical, Co. (Seoul, Republic of Korea), chloroform, t-butanol (t-BuOH) and deionized water were also from Sigma-Aldrich. MALDI target plates were supplied by Hudson Surface Technology, Inc. (Closter, NJ, USA). The MALDI matrix, 2,4,6-trihydroxyacetophenone (THAP), and tetrahydrofuran (THF), were also procured from Sigma-Aldrich, Co.
2.2. Gel Permeation Chromatography
GPC instrument (Nexera GPC System, Shimadzu Co., Kyoto, Japan) was utilized for polymer size analysis, which is equipped with LF-G guard (6 μm, 4.6 × 10 mm, Shodex, Tokyo, Japan) and two LF-804 (6 μm, 8.0 × 300 mm) in series. P3HB, PCL, and PBS were dissolved in chloroform to a concentration of 20 mg/mL (1 mL total solution per polymer). Aliquots of 100 μL were injected into the GPC system in triplicate. Chloroform was used as the mobile phase at a flow rate of 1 mL/min.
2.3. Depolymerization of Biodegradable Plastics by Transesterification
Each biodegradable plastic was dissolved in chloroform to 4000 ppm. Stock solutions of sodium t-BuO−, sodium MeO−, and sodium EtO− were prepared at the required concentrations in t-BuOH, MeOH, and EtOH, respectively, and sonicated for 3 min to ensure complete dissolution. An aliquot of the polymer solution (375 μL) was transferred to a 1.5 mL Eppendorf tube, after which 125 μL of the appropriate alkoxide solution was added and the mixtures were briefly vortex-mixed. The mixtures were incubated for reaction times ranging from 0 to 30 min under ambient conditions to evaluate the effect of incubation time.
2.4. Sample Preparation for MALDI-TOF MS
For MALDI sample preparation, the sandwich method for matrix treatment was employed [33]. An amount of 0.7 μL of THAP matrix solution (20 mg/mL in THF) was first spotted onto target plate and allowed to dry completely. An aliquot (1.0 µL) of the analyte mixture was deposited onto the dried matrix layer and allowed to dry, followed by overlaying with an additional 0.7 µL of the matrix solution.
2.5. MALDI-TOF MS
MALDI mass spectra were acquired on a MALDI-TOF MS (IDSys RT instrument; ASTA, Suwon, Republic of Korea) operated in reflector mode. All analyses were performed in positive ion mode over an m/z range of 500–3000. Ionization utilized a pulsed Yb:YAG UV laser (343 nm) set to 70% power; fifty shots were accumulated at each of 100 evenly distributed positions on a spot with a 50 μm laser beam diameter. The mass resolution (FWHM) was >20,000 (M/ΔM) at m/z 2465. External mass calibration was performed with PEG300 using trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB) as matrix prior to analysis.
3. Results and Discussion
3.1. Optimization of Experimental Methods
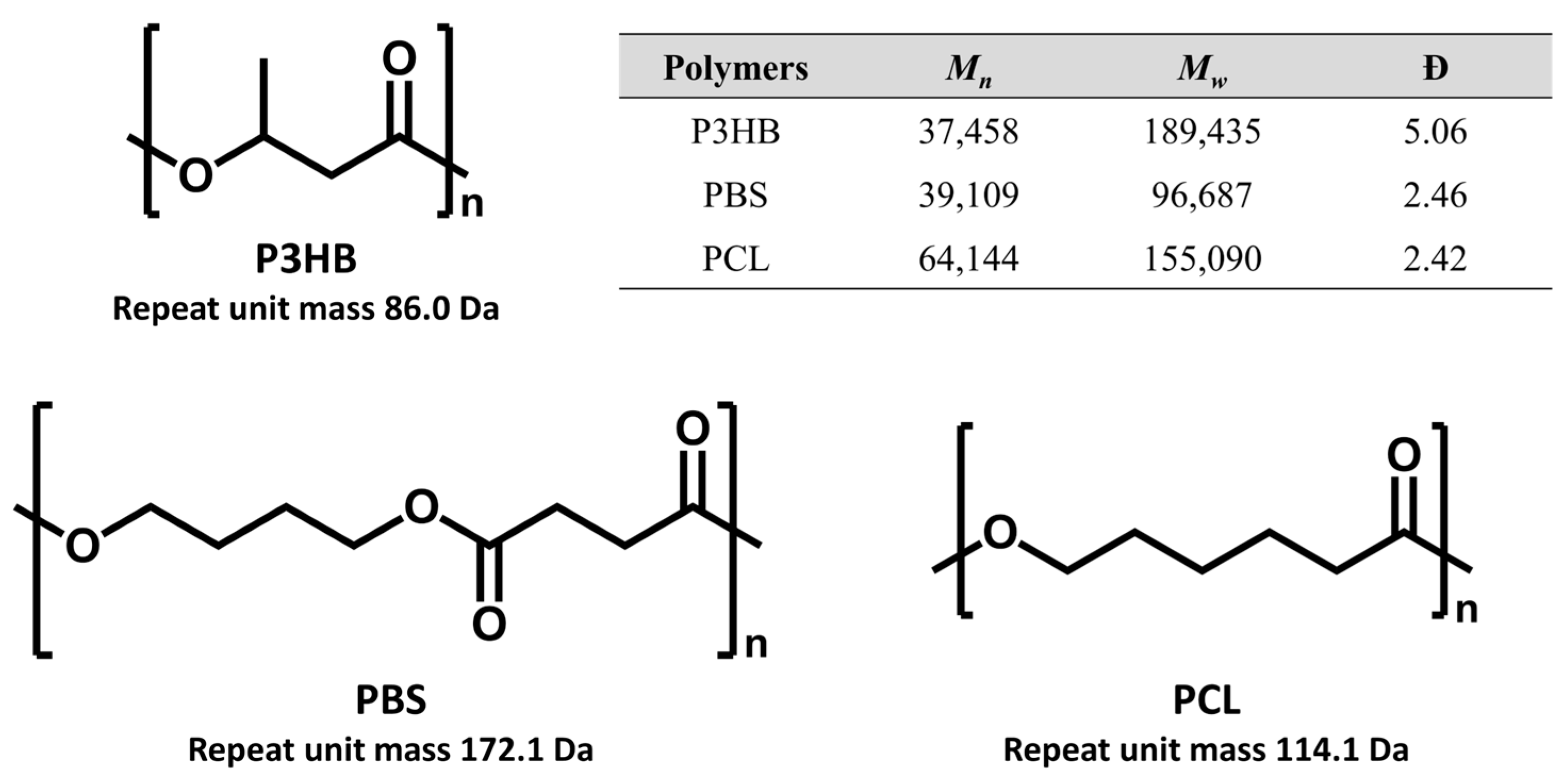
In the study, three widely used commercial biodegradable plastics were examined, including P3HB (Mn = 37,458; Mw = 189,435), PBS (39,109; 96,109) and PCL (64,144; 155,090), of which Mn and Mw, and dispersity (Đ) were obtained by GPC (Figure 1). The examined biodegradable polymers were primarily high-molecular-weight polyesters (100–200 kDa), which are amenable to alkaline depolymerization via transesterification using strong alkoxide bases [30,31]. Alkoxide treatment cleaves polymer chains into smaller oligomeric products suitable for MALDI-TOF MS analysis, producing characteristic ion series defined by repeat unit mass intervals. These low-molecular-weight oligomers further allow structural characterization by MS/MS with sufficient resolving power, which is not readily achievable for intact high-molecular-weight polymers.
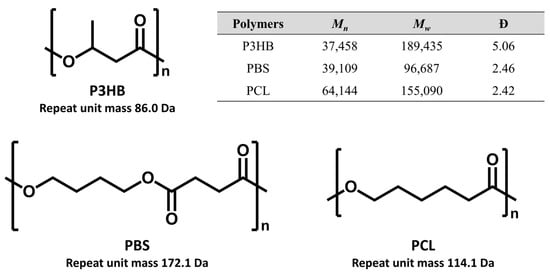
Figure 1.
Chemical structures of the biodegradable polymers, P3HB, PBS, and PCL, with a table summarizing their molecular weights, Mn and Mw, and dispersity (Đ) measured by GPC.
In the experiments, biodegradable polymers were dissolved in organic solvents, typically chloroform, and subjected to transesterification using alkoxide reagents prepared in the corresponding alcohols (RO−/ROH). The resulting products were analyzed by MALDI-TOF MS. Three representative alkoxides were evaluated for their transesterification behavior and suitability for subsequent MALDI-TOF MS analysis.
Prior to the alkoxide comparison experiments, commonly used MALDI matrices for polymer ionization—THAP, trans-indoleacrylic acid (IAA), and dithranol—were preliminarily screened in combination with THF and chloroform solvents. Among the tested conditions, THAP in THF provided the most reproducible spectra. In contrast, alternative combinations resulted in reduced peak visibility (dithranol) or elevated background peaks (IAA) under our experimental conditions. Based on these screening results, THAP in THF was chosen as the present matrix system for the comparative evaluation of transesterification reagent performance.
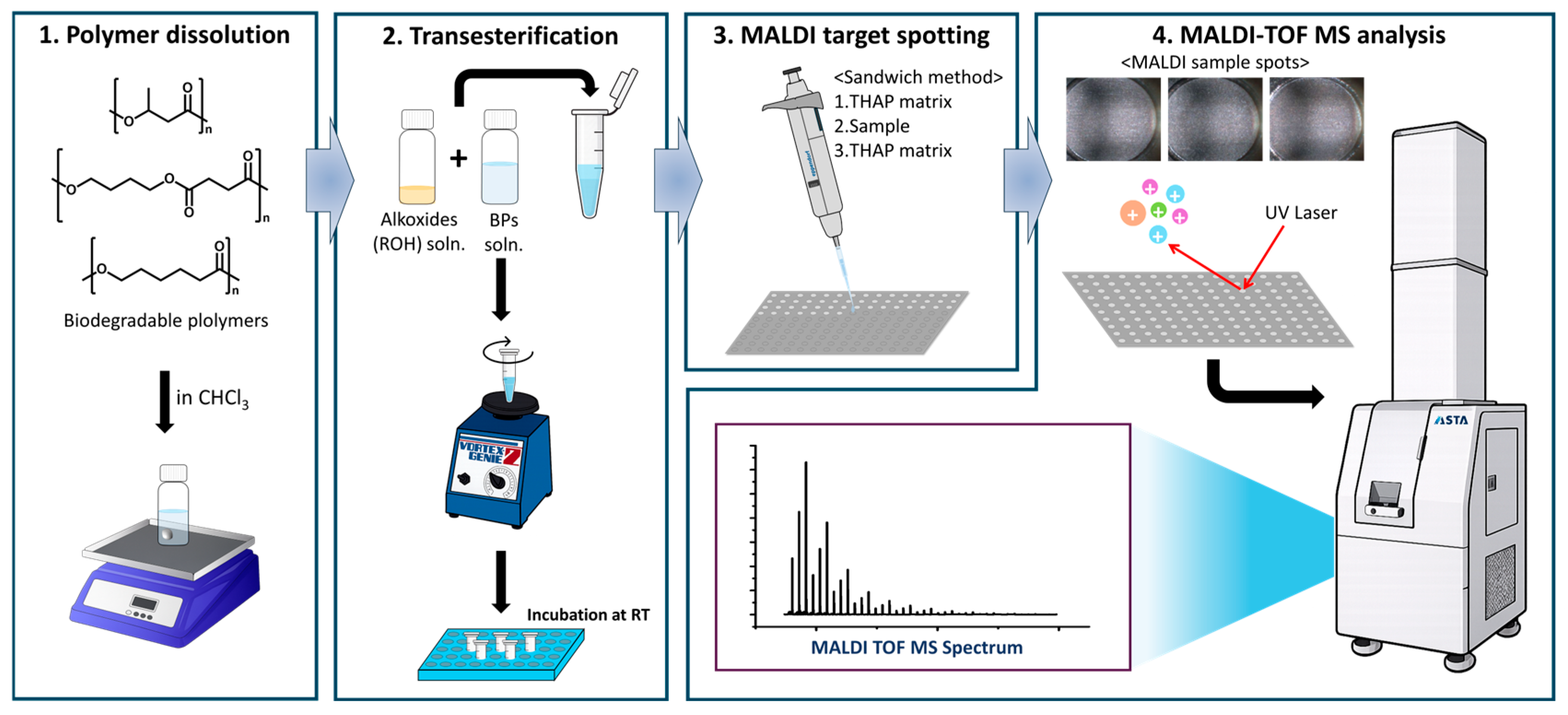
Sample-loading methods, including premixing of sample and matrix solutions, pre-deposition of a matrix layer, and the sandwich method [33], were also evaluated. The sandwich method was adopted in this study because it provided uniform sample distribution and improved signal reproducibility under the selected matrix conditions (Figures S1 and S2 in the Supplementary Materials). The overall experimental workflow is shown in Figure 2.
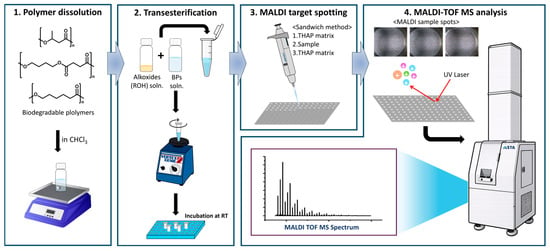
Figure 2.
Experimental workflow.
3.2. MALDI-TOF MS of Biodegradable Polymers
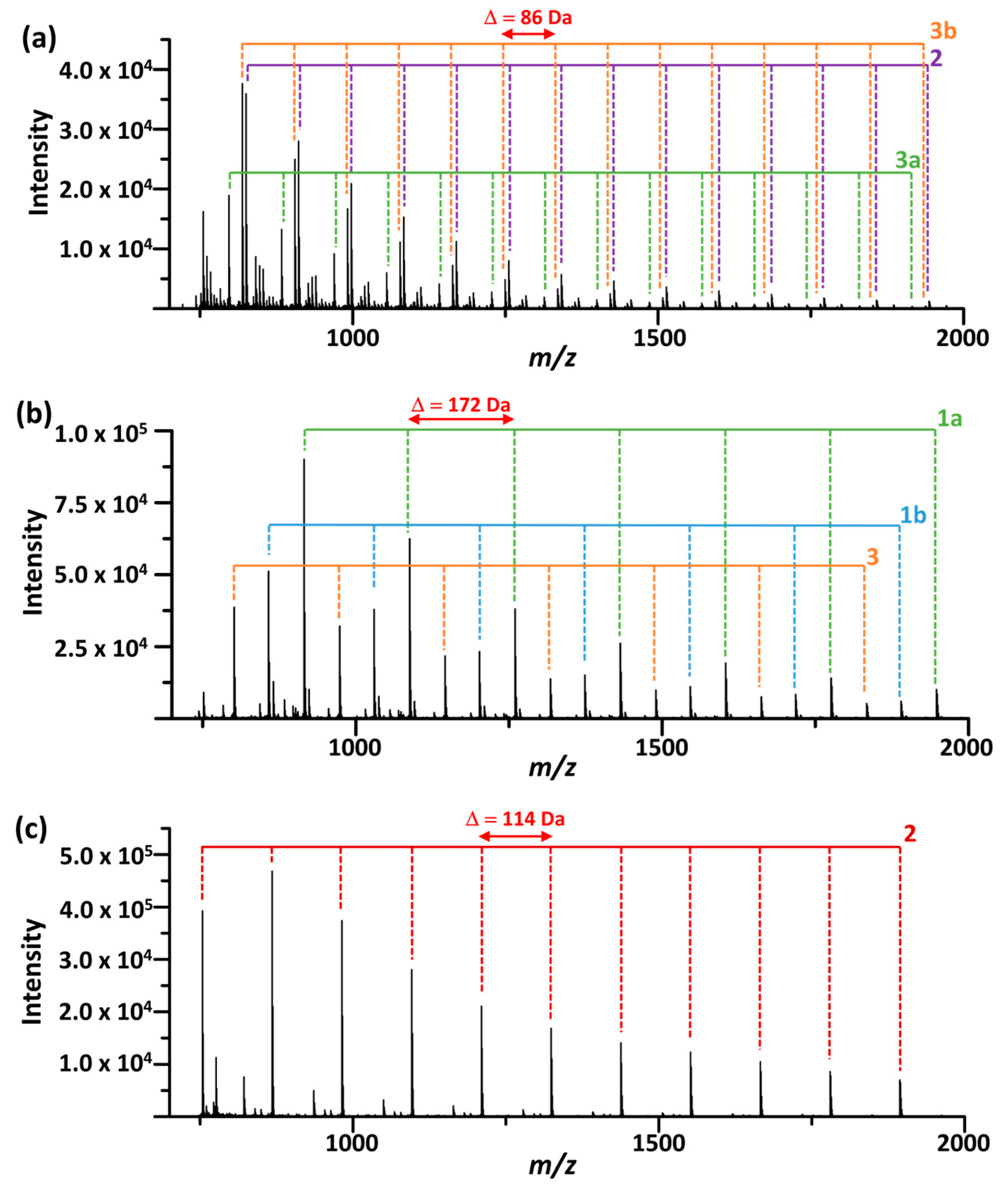
Figure 3 presents representative mass spectra of the transesterification products of the polymers. The spectra show series of oligomer ions up to m/z 3000, each exhibiting characteristic mass intervals corresponding to the repeat unit masses and differing by terminal groups. The observed product ions are summarized in Figure 4, and detailed information can be found in Table S1. The oligomer structures were further confirmed by MS/MS using collision-induced dissociation (Figure S3). These results demonstrate that the proposed MALDI-TOF MS method is suitable for the structural characterization of biodegradable polyester polymers. MALDI mass spectra of the polymers obtained without alkoxide treatment were also acquired (Figure S4). In these spectra, only trace amounts of signals corresponding to polyester oligomers were observed (intensity ~103). For PCL and PBS, the detected species were predominantly cyclic oligomers, whereas for P3HB, OH-terminated oligomers were observed. These species are attributable to residual by-products present in the industrial polymer materials.
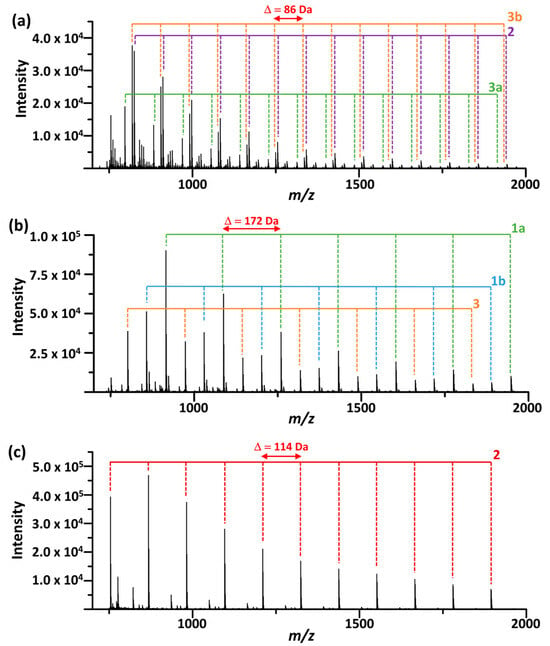
Figure 3.
MALDI mass spectra of the transesterification products of (a) P3HB and (c) PCL obtained after EtO−/EtOH treatment, and (b) PBS obtained after MeO−/MeOH treatment, using THAP as the matrix. Structural assignments of the observed oligomer series are shown in Figure 4.
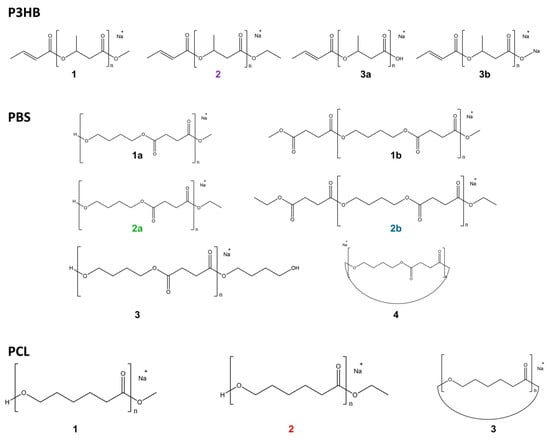
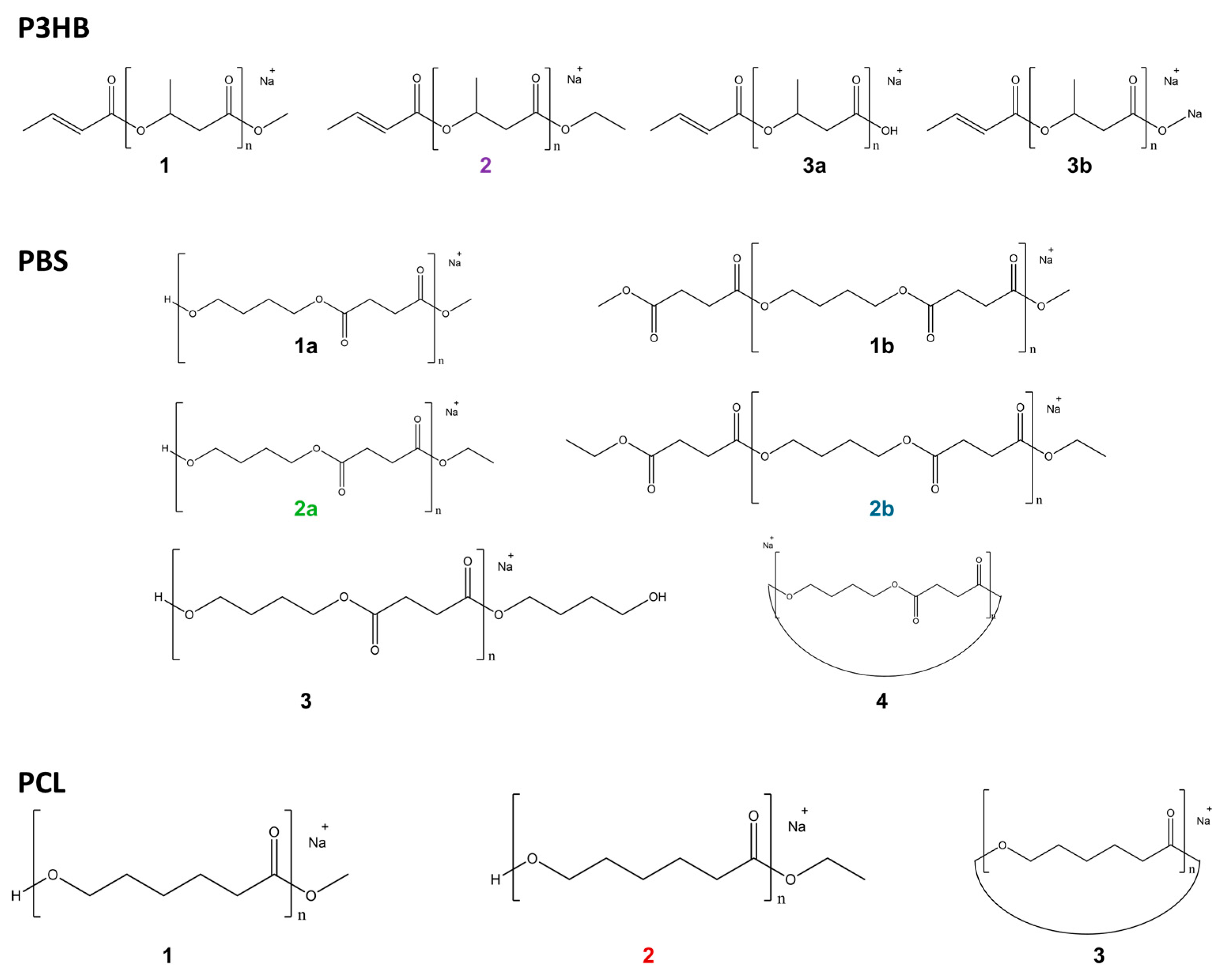
Figure 4.
Observed transesterification products of polyesters.
In the mass spectra, product ions were predominantly observed as Na+ adducts. These product ions arise from nucleophilic attack of strong alkoxide bases on the electron-deficient carbonyl carbon during well-known transesterification. Notably, double-bond formation was observed at the left terminal of P3HB oligomers, attributable to the β-elimination process [31,34]. Care should be taken when using commercially available CHCl3, as it may contain ethanol as a stabilizer; insufficient purification can lead to dominant EtO−-mediated transesterification and formation of ethyl ester terminals.
3.3. Comparison of Alkoxides for MALDI-TOF MS of Polyester Polymers
3.3.1. Concentration Dependence
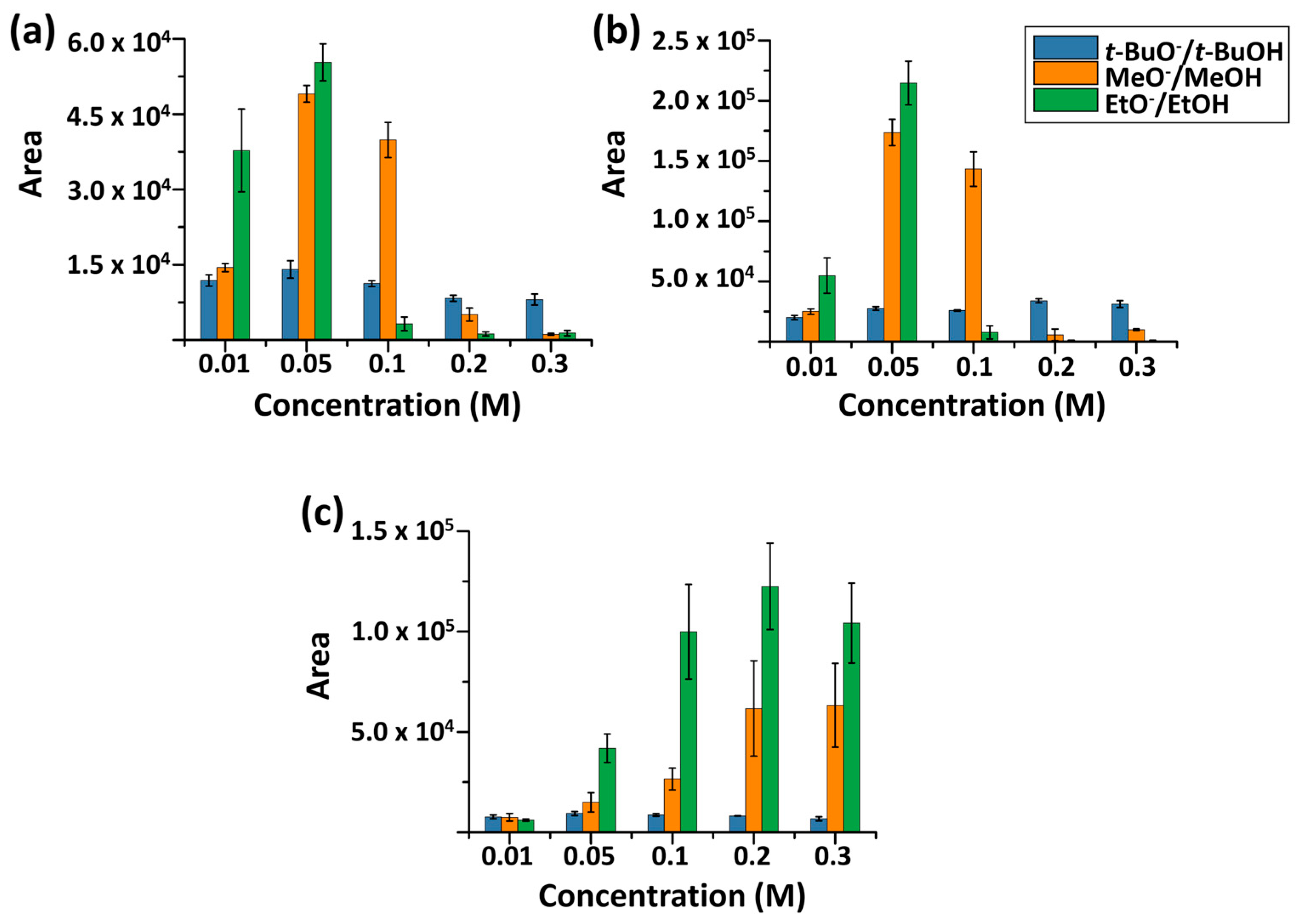
Using the optimized method, depolymerization with MeO−/MeOH, EtO−/EtOH, and t-BuO−/t-BuOH was systematically examined for MALDI-TOF MS analysis of biodegradable polymers. First, to assess the efficiency of alkoxide-mediated transesterification, the concentration dependence of reaction efficiency and MALDI compatibility was examined. For comparison, all reactions were conducted for 8 min, and other parameters like polymer concentration and MALDI-TOF MS instrument settings were kept constant. Signal intensity was evaluated by summing the peak areas of the five most intense ions in each mass spectrum. Experiments were performed in triplicate (Figure 5).
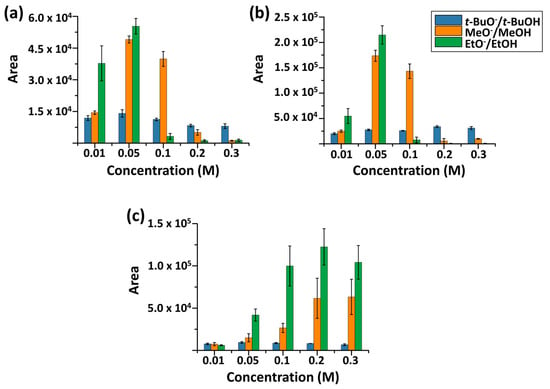
Figure 5.
Alkoxide concentration dependence of the product ions for (a) P3HB, (b) PBS, and (c) PCL over the range of 0.01–0.3 M. Error bars represent standard deviations (N = 3).
As shown in Figure 5, detection efficiency varied depending on the polymer. Comparable detection efficiencies were observed for PBS and PCL, whereas a slightly lower efficiency was observed for P3HB. Notably, detection efficiency also differed markedly among alkoxides.
Under all conditions examined, EtO−/EtOH resulted in the highest detection efficiency, followed by MeO−/MeOH. Although t-BuO− is the strongest base among the three alkoxides (pKa of t-BuOH ~17), compared to EtO− (EtOH ~16) and MeO− (MeOH ~15–16), its detection efficiency was substantially lower. This behavior is largely attributable to the bulky t-butyl group, which limits effective nucleophilic attack on carbonyl carbon, despite its strongest basicity. Consistent with this interpretation, products formed using t-BuO−/t-BuOH were predominantly cyclic species, e.g., PBS (4) and PCL (3a), in this concentration dependence study.
The concentration window of 0.05–0.1 M was identified for achieving balanced transesterification efficiency and MALDI-TOF MS detection across the examined polymers. Although the optimal concentration slightly differed among individual systems—particularly for PCL—this range provided reproducible signal intensity and adequate oligomer distribution for comparative analysis. At concentrations above this range, product-ion detection decreased, likely due to increased sodium salt content suppressing MALDI ionization, despite the comparatively high salt tolerance of MALDI relative to other ionization methods.
Overall, EtO−-mediated transesterification and the concentration range around 0.05–0.1 M produced analytically useful MALDI mass spectra for polyester identification, whereas t-BuO− showed limited effectiveness under the tested conditions.
3.3.2. Reaction Time Dependence
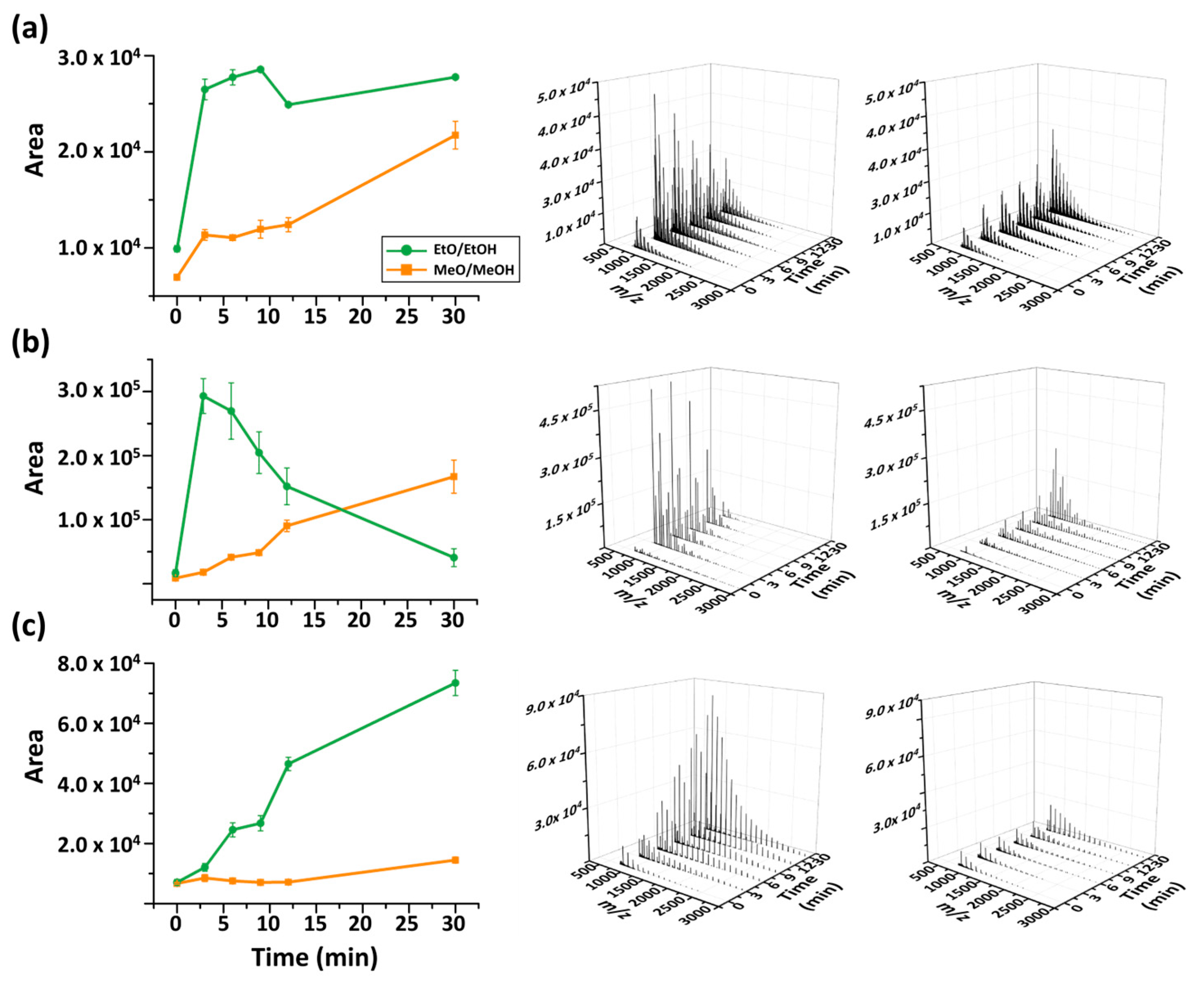
The effect of reaction time on transesterification was further investigated. In the study, the alkoxide concentration was fixed at 0.05 M and the effect was explored for a time period up to 30 min. All measurements were performed in triplicate, and the averages and standard deviations of the summed areas of five strongest peaks are presented in Figure 6.
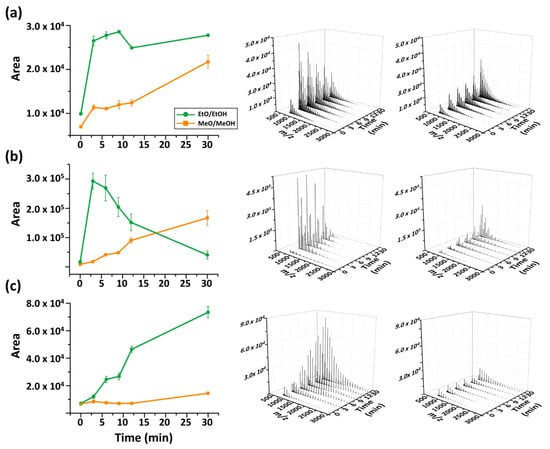
Figure 6.
Reaction time dependence of MALDI spectra up to 30 min. (a) P3HB, (b) PBS, (c) PCL.
In most cases, extended reaction times increased overall signal intensity. EtO−/EtOH consistently provided the highest cumulative signal intensity (based on the five strongest peaks) in MALDI-TOF MS analysis. However, its time dependence differed depending on the polymer. For example, depolymerization of P3HB and PCL using EtO−/EtOH ion intensities increased with longer incubation periods, whereas for PBS the efficiency reached a maximum at 3 min and decreased sharply thereafter. The behavior of PBS case suggests that prolonged transesterification may promote secondary reactions or changes in ionization efficiency, leading to reduced detectable product signals. Overall, EtO−-mediated transesterification provided the most consistently high signal response across the examined polymers when the incubation time was maintained at approximately 3–9 min.
3.3.3. Alkoxide-Dependent Product Distribution and End-Group Analysis
To elucidate how alkoxides influence depolymerization behavior under our MALDI conditions, the structures of dominant oligomer species were analyzed. The complete list of observed product ions is provided in Table S1, in which the most intense peaks that were used for evaluation are highlighted.
As for P3HB, depolymerization predominantly yielded oligomers bearing crotonate-type end groups, following the well-known β-elimination pathway under alkaline conditions [31,34]. Upon t-BuO−/BuOH treatment, the most intense species corresponded primarily to oligomers terminated with OH or ONa group (3a, 3b in Figure 4). Similarly, OH− and ONa-terminated species were the dominant products at early reaction times for MeO−/MeOH and EtO−/EtOH treatments. However, with increasing reaction time, alkyl-terminated products became predominant; methyl-terminated oligomers (1) became predominant after approximately 9 min under MeO−/MeOH, and ethyl-terminated oligomers (2) after approximately 3 min under EtO−/EtOH. Notably, cyclic products were not significantly detected under the examined conditions, consistent with preferential β-elimination limiting intramolecular backbiting reactions.
In contrast, PBS exhibited distinct alkoxide-dependent product distributions. t-BuO−/t-BuOH treatment primarily generated cyclic oligomers together with OH-terminated species (3, 4). Under methoxide treatment, cyclic species were initially observed; however, after approximately 3 min, mono-methyl-terminated oligomers (1a) appeared, and then after 6 min, di-methyl-terminated products (1b) gradually increased in relative intensity. For ethoxide treatment, mono-ethyl-terminated oligomers (2a) appeared from early reaction times alongside cyclic species, whereas di-ethyl-terminated products (2b) became detectable after approximately 3 min and increased over time. These results indicate that PBS readily forms both cyclic and dialkyl-terminated oligomers, with product distributions strongly influenced by alkoxide type and reaction time. This behavior is consistent with the structure of PBS, which contains two ester functionalities per repeating unit, allowing alkoxide substitution at both chain ends.
As for PCL, t-BuO−/t-BuOH treatment predominantly produced cyclic oligomers (3), with comparatively low depolymerization efficiency. MeO−/MeOH treatment resulted in the appearance of methyl-terminated products (1) after approximately 9 min, whereas EtO−/EtOH treatment produced ethyl-terminated oligomers (2) from approximately 3 min onward. Cyclic oligomers were already detectable in the untreated polymer spectrum (Figure S4), indicating that they are intrinsic components of the original material; however, their signal intensity increased markedly following alkaline treatment, from approximately 103 to >104 in relative intensity units. Taken together, the presence and relative abundance of cyclic species require careful interpretation. Cyclic oligomers in PBS and PCL cannot be attributed exclusively to alkoxide-mediated transesterification, as they are detectable in untreated materials. Nevertheless, their increase in relative intensity after alkoxide treatment suggests that the treatment promotes the formation of cyclic products.
3.3.4. Application of the Optimized Workflow to Polymer Mixtures
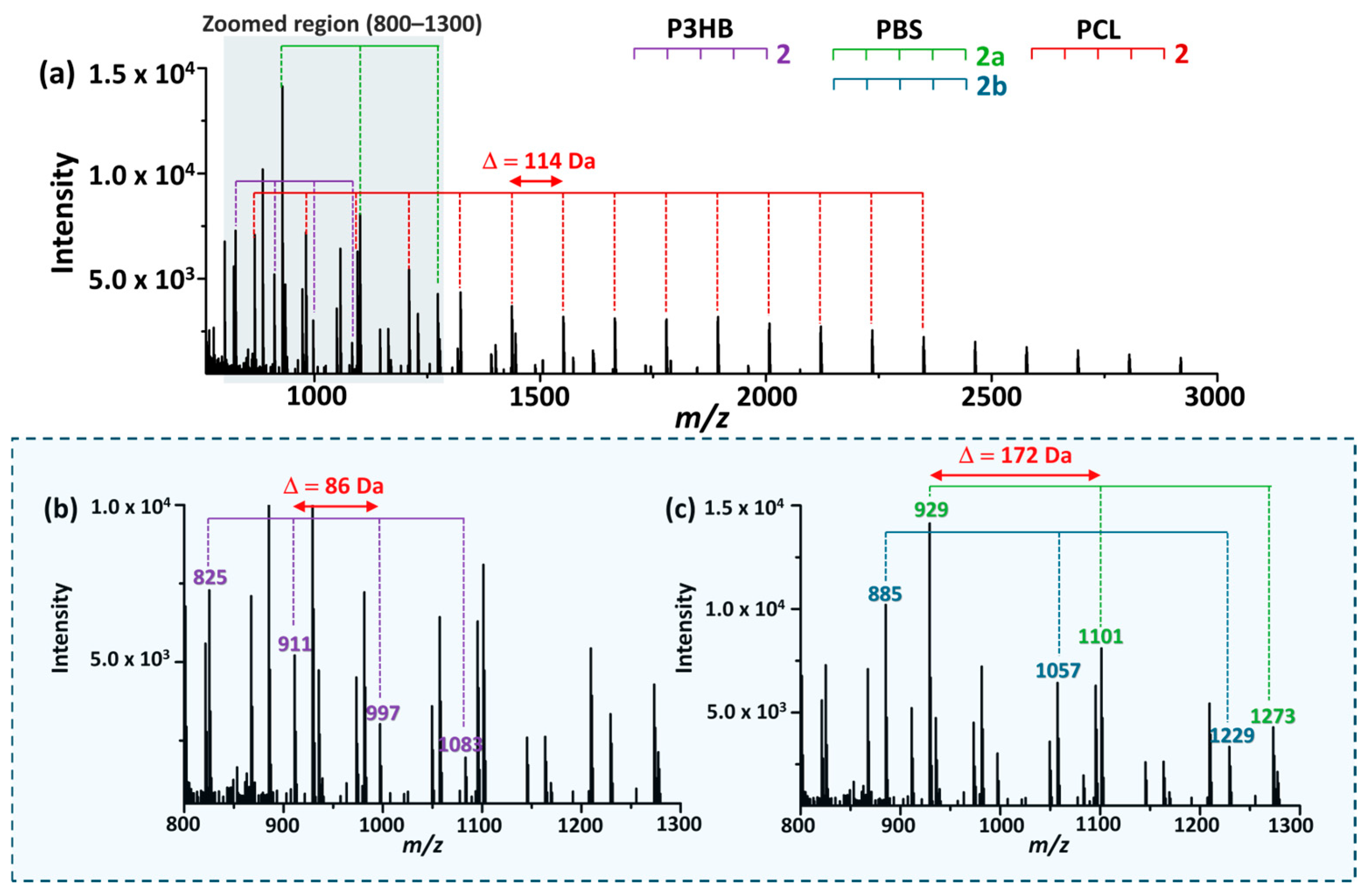
The MALDI-TOF mass spectrum of the mixed polymer sample obtained under an optimized reaction conditions (0.05 M EtO−/EtOH, 6 min) is shown in Figure 7. Despite partial overlap in the mass range, each polymer could be distinguished based on its characteristic repeat unit spacing and end-group patterns.
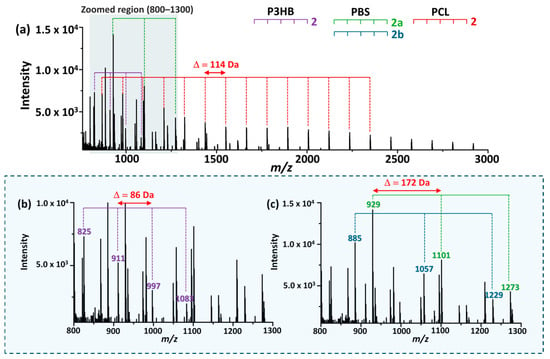
Figure 7.
(a) MALDI-TOF mass spectrum of a mixed sample containing P3HB, PBS, and PCL obtained under the optimized reaction conditions. (b,c) show magnified views of panel (a) over the same m/z range of 800–1300. Polymer-specific oligomer series are indicated, exhibiting distinct repeat unit mass spacings (Δm = 86 for P3HB, 172 for PBS, and 114 for PCL), enabling simultaneous differentiation of the three polyesters within a single spectrum.
For P3HB, a series corresponding to crotonate–ethyl-terminated oligomers (2) were observed with a repeat unit spacing of 86 Da (Figure 7b), consistent with the product distribution identified during optimization. For PBS, multiple product series were detectable, including OH-terminated oligomers (3), mono-ethyl-terminated oligomers (2a), and di-ethyl-terminated oligomers (2b), all exhibiting a characteristic repeat unit spacing of 172 Da. For clarity, only the ethyl-terminated series is indicated in Figure 7c. In the case of PCL, ethyl-terminated oligomers (2) displayed the broadest distribution and a distinct repeat-unit spacing of 114 Da, enabling clear differentiation from P3HB and PBS. Cyclic products (3) were also observed; however, only the ethyl-terminated series is highlighted in the figure for simplicity.
These findings show that polymer-specific oligomer series identified during individual optimization experiments remain distinguishable in mixed samples under the selected reaction conditions.
4. Conclusions
In conclusion, this study demonstrates that alkoxide-mediated transesterification enables rapid MALDI-TOF MS identification and structural analysis of high-molecular-weight biodegradable polyesters. Among the tested systems, EtO−/EtOH combined with THAP/THF in a sandwich deposition configuration provided the most consistent generation of polymer-specific oligomer series suitable for MALDI-TOF MS analysis. Although reaction behavior and signal response were polymer-dependent, a practical concentration window centered around 0.05 M and reaction times of approximately 3–9 min allowed reliable oligomer detection across all examined systems. This protocol enabled simultaneous identification of P3HB, PBS, and PCL within a single MALDI-TOF mass spectrum, facilitating rapid characterization of mixed biodegradable polymer samples within 10 min.
The comparative evaluation of alkoxide systems presented here may provide practical guidance for optimizing MALDI-compatible depolymerization conditions in mixed polyester analysis and support the broader application of MALDI-TOF MS in biodegradable polymer characterization.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/analytica7010022/s1, Figure S1: Photos of MALDI sample spots using THAP matrix prepared by sandwich method; Figure S2: Comparison of MALDI sample-loading methods; Figure S3: MS/MS spectra of transesterification products of PBS polymer; Figure S4: MALDI MS spectra of untreated polymers; Table S1: List of observed transesterification product ions in MALDI MS spectra.
Author Contributions
Conceptualization, S.Y.H.; methodology, investigation, and writing—original draft preparation, M.K.; writing—review and editing, M.K. and S.Y.H.; supervision, funding acquisition, S.Y.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2024-00454586). This work was supported by Korea Basic Science Institute (NFEC) grant funded by the Ministry of Education (2020R1A6C101A184). This work was supported by the Gachon University Research Fund (GL-202301120001).
Data Availability Statement
The original data presented in this study are available in the article and in the Supplementary Materials.
Acknowledgments
We are grateful to ASTA Corp. (Suwon, Republic of Korea) for providing access to MALDI-TOF MS instrumentation and for technical assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| MALDI | Matrix-Assisted Laser Desorption/Ionization |
| TOF | Time-Of-Flight |
| MS | Mass Spectrometry |
| P3HB | Poly(3-hydroxybutyrate) |
| PBS | Poly(butylene succinate) |
| PCL | Poly(ε-caprolactone) |
| THAP | 2,4,6-trihydroxyacetophenone |
References
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tao, L.; Wang, Q.; Wang, F.; Li, G.; Song, M. Potential Health Impact of Microplastics: A Review of Environmental Distribution, Human Exposure, and Toxic Effects. Environ. Health 2023, 1, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Haider, T.P.; Völker, C.; Kramm, J.; Landfester, K.; Wurm, F.R. Plastics of the Future? The Impact of Biodegradable Polymers on the Environment and on Society. Angew. Chem. Int. Ed. 2019, 58, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Haq, F.; Kiran, M.; Khan, I.A.; Mehmood, S.; Aziz, T.; Haroon, M. Exploring the Pathways to Sustainability: A Comprehensive Review of Biodegradable Plastics in the Circular Economy. Mater. Today Sustain. 2025, 29, 101067. [Google Scholar] [CrossRef]
- Payanthoth, N.S.; Mut, N.N.N.; Samanta, P.; Li, G.; Jung, J. A Review of Biodegradation and Formation of Biodegradable Microplastics in Soil and Freshwater Environments. Appl. Biol. Chem. 2024, 67, 110. [Google Scholar] [CrossRef]
- Hu, L.; Zhou, Y.; Chen, Z.; Zhang, D.; Pan, X. Oligomers and Monomers from Biodegradable Plastics: An Important but Neglected Threat to Ecosystems. Environ. Sci. Technol. 2023, 57, 9895–9897. [Google Scholar] [CrossRef]
- Withana, P.A.; Yuan, X.; Im, D.; Choi, Y.; Bank, M.S.; Lin, C.S.K.; Hwang, S.Y.; Ok, Y.S. Biodegradable Plastics in Soils: Sources, Degradation, and Effects. Environ. Sci. Process. Impacts 2025, 27, 3321–3343. [Google Scholar] [CrossRef]
- Baidurah, S. Methods of Analyses for Biodegradable Polymers: A Review. Polymers 2022, 14, 4928. [Google Scholar] [CrossRef]
- Mndlovu, H.; Kumar, P.; du Toit, L.C.; Choonara, Y.E. A Review of Biomaterial Degradation Assessment Approaches Employed in the Biomedical Field. npj Mater. Degrad. 2024, 8, 66. [Google Scholar] [CrossRef]
- Striegel, A.M. Size-Exclusion Chromatography: A Twenty-First Century Perspective. Chromatographia 2022, 85, 307–313. [Google Scholar] [CrossRef]
- Kulkarni, S.O.; Kanekar, P.P.; Nilegaonkar, S.S.; Sarnaik, S.S.; Jog, J.P. Production and Characterization of a Biodegradable Poly (Hydroxybutyrate-co-Hydroxyvalerate) (PHB-co-PHV) Copolymer by Moderately Haloalkalitolerant Halomonas campisalis MCM B-1027 Isolated from Lonar Lake, India. Bioresour. Technol. 2010, 101, 9765–9771. [Google Scholar] [CrossRef]
- Spiess, H.W. 50th Anniversary Perspective: The Importance of NMR Spectroscopy to Macromolecular Science. Macromolecules 2017, 50, 1761–1777. [Google Scholar] [CrossRef]
- Zhang, R.; Miyoshi, T.; Sun, P. (Eds.) NMR Methods for Characterization of Synthetic and Natural Polymers; New Developments in NMR; Royal Society of Chemistry: Cambridge, UK, 2019; Volume 20. [Google Scholar] [CrossRef]
- Ciornii, D.; Hodoroaba, V.D.; Benismail, N.; Maltseva, A.; Ferrer, J.F.; Wang, J.; Parra, R.; Jézéquel, R.; Receveur, J.; Gabriel, D.; et al. Interlaboratory comparison reveals state of the art in microplastic detection and quantification methods. Anal. Chem. 2025, 97, 8719–8728. [Google Scholar] [CrossRef]
- Brandt, J.; Mattsson, K.; Hassellov, M. Deep learning for reconstructing low-quality FTIR and Raman Spectra—A case study in microplastic analyses. Anal. Chem. 2021, 93, 16360–16368. [Google Scholar] [CrossRef]
- Borrowman, C.K.; Bücking, M.; Göckener, B.; Adhikari, R.; Saito, K.; Patti, A.F. LC-MS Analysis of the Degradation Products of a Sprayable, Biodegradable Poly(ester-urethane-urea). Polym. Degrad. Stab. 2020, 178, 109218. [Google Scholar] [CrossRef]
- Han, Y.; Simmons, B.A.; Singh, S. Perspective on Oligomeric Products from Lignin Depolymerization: Their Generation, Identification, and Further Valorization. Ind. Chem. Mater. 2023, 1, 207–223. [Google Scholar] [CrossRef]
- Cho, J.Y.; Kim, S.H.; Cho, D.H.; Jung, H.J.; Kim, B.C.; Bhatia, S.K.; Gurav, R.; Lee, J.; Park, S.-H.; Park, K.; et al. Simultaneous Monitoring of Each Component on Degradation of Blended Bioplastic Using Gas Chromatography-Mass Spectrometry. Anal. Biochem. 2022, 655, 114832. [Google Scholar] [CrossRef] [PubMed]
- Seeley, M.E.; Lynch, J.M. Previous Successes and Untapped Potential of Pyrolysis–GC/MS for the Analysis of Plastic Pollution. Anal. Bioanal. Chem. 2023, 415, 2873–2890. [Google Scholar] [CrossRef] [PubMed]
- Dumont, G.; Rodrigues, A.; Velimirovic, M.; Lievens, S.; Jordens, J.; Focant, J.-F.; Stefanuto, P.H. Microplastic and Nanoplastic Analysis: From Pyrolysis Gas Chromatography-Mass Spectrometry to Pyrolysis Two-Dimensional Gas Chromatography-Mass Spectrometry: A Critical Review. J. Sep. Sci. 2025, 48, e70287. [Google Scholar] [CrossRef] [PubMed]
- De Falco, F.; Nacci, T.; Durndell, L.; Thompson, R.C.; Degano, I.; Modugno, F. A Thermoanalytical Insight into the Composition of Biodegradable Polymers and Commercial Products by EGA-MS and Py-GC-MS. J. Anal. Appl. Pyrolysis 2023, 171, 105937. [Google Scholar] [CrossRef]
- Santos, L.H.; Insa, S.; Arxé, M.; Buttiglieri, G.; Rodríguez-Mozaz, S.; Barceló, D. Analysis of Microplastics in the Environment: Identification and Quantification of Trace Levels of Common Types of Plastic Polymers Using Pyrolysis-GC/MS. MethodsX 2023, 10, 102143. [Google Scholar] [CrossRef]
- Ivleva, N.P. Chemical Analysis of Microplastics and Nanoplastics: Challenges, Advanced Methods, and Perspectives. Chem. Rev. 2021, 121, 11886–11936. [Google Scholar] [CrossRef]
- Wesdemiotis, C.; Williams-Pavlantos, K.N.; Keating, A.R.; McGee, A.S.; Bochenek, C. Mass Spectrometry of Polymers: A Tutorial Review. Mass Spectrom. Rev. 2024, 43, 427–476. [Google Scholar] [CrossRef]
- Rizzarelli, P.; Carroccio, S. Modern Mass Spectrometry in the Characterization and Degradation of Biodegradable Polymers. Anal. Chim. Acta 2014, 808, 18–43. [Google Scholar] [CrossRef]
- Rizzarelli, P.; Leanza, M.; Rapisarda, M. Investigations into the Characterization, Degradation, and Applications of Biodegradable Polymers by Mass Spectrometry. Mass Spectrom. Rev. 2025, 44, 947–988. [Google Scholar] [CrossRef]
- Montaudo, G.; Samperi, F.; Montaudo, M.S. Characterization of Synthetic Polymers by MALDI-MS. Prog. Polym. Sci. 2006, 31, 277–357. [Google Scholar] [CrossRef]
- Byrd, H.M.; McEwen, C.N. The Limitations of MALDI-TOF Mass Spectrometry in the Analysis of Wide Polydisperse Polymers. Anal. Chem. 2000, 72, 4568–4576. [Google Scholar] [CrossRef]
- Ntrivala, M.A.; Pitsavas, A.C.; Lazaridou, K.; Baziakou, Z.; Karavasili, D.; Papadimitriou, M.; Ntagkopoulou, C.; Balla, E.; Bikiaris, D.N. Polycaprolactone (PCL): The biodegradable polyester shaping the future of materials—A review on synthesis, properties, biodegradation, applications and future perspectives. Eur. Polym. J. 2025, 234, 114033. [Google Scholar] [CrossRef]
- Nakamura, S.; Fouquet, T.; Sato, H. Molecular Characterization of High Molecular Weight Polyesters by Matrix-Assisted Laser Desorption/Ionization High-Resolution Time-of-Flight Mass Spectrometry Combined with On-Plate Alkaline Degradation and Mass Defect Analysis. J. Am. Soc. Mass Spectrom. 2019, 30, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Saeed, K.A.; Ayorinde, F.O.; Eribo, B.E.; Gordon, M.; Collier, L. Characterization of Partially Transesterified Poly(β-hydroxyalkanoate)s Using Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Rapid Commun. Mass Spectrom. 1999, 13, 1951–1957. [Google Scholar] [CrossRef]
- Kanayama, N.; Nakamura, S.; Sano, S.; Tanaka, M.; Takahara, N.; Sato, H.; Hagihara, H. Comonomer distribution analysis of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate)s using high-resolution MALDI-TOF mass spectrometry. Anal. Methods 2025, 17, 7618–7627. [Google Scholar] [CrossRef] [PubMed]
- Kemptner, J.; Marchetti-Deschmann, M.; Mach, R.; Druzhinina, I.S.; Kubicek, C.P.; Allmaier, G. Evaluation of Matrix-Assisted Laser Desorption/Ionization (MALDI) Preparation Techniques for Surface Characterization of Intact Fusarium Spores by MALDI Linear Time-of-Flight Mass Spectrometry. Rapid Commun. Mass Spectrom. 2009, 23, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Kopinke, F.D.; Remmler, M.; Mackenzie, K. Thermal Decomposition of Biodegradable Polyesters-I: Poly(β-hydroxybutyric acid). Polym. Degrad. Stab. 1996, 52, 25–38. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.