Abstract
Background/Objectives: Thumb basal joint arthritis is a common degenerative condition often requiring surgery when conservative treatment fails. Dual mobility trapeziometacarpal prostheses are increasingly used, but the optimal anesthetic strategy remains debatable. This study aimed to explore whether WALANT provides intraoperative analgesia and short-term safety comparable to axillary block in dual mobility trapeziometacarpal arthroplasty. Methods: A prospective observational comparative study was carried out on 21 patients (11 WALANT, 10 axillary block) undergoing dual mobility trapeziometacarpal prosthesis for stage II–III in thumb basal joint arthritis according to Eaton–Littler classification at two hospital facilities of ASL Roma 5, from February–December 2025. Patients treated with the WALANT technique were assigned to Group A, whereas those undergoing an axillary block were assigned to Group B. Pain intensity was recorded on a 0–10 visual analogue scale at three stages: during anesthetic administration, during surgery, and 3 h after the procedure. Group A received a field infiltration with 1% mepivacaine combined with epinephrine 1:100,000 and sodium bicarbonate, while Group B underwent an ultrasound-guided brachial plexus block using 0.5–0.7% ropivacaine and a pneumatic tourniquet inflated to 250 mmHg. Results: Pain during anesthesia induction was similar between groups (Group A 3.18 ± 2.89 vs. Group B 2.20 ± 2.37, p = 0.393). Intraoperative pain did not differ significantly (Group A 2.27 ± 1.79 vs. Group B 2.00 ± 2.71, p = 0.898). At 3 h postoperative, Group B showed a trend toward lower pain levels (Group A 4.36 ± 2.54 vs. Group B 3.00 ± 3.08, p = 0.244). No anesthetic failures, no conversion to general anesthesia, and no neurological or ischemic complications occurred in either group. Conclusions: In this prospective observational comparative cohort, WALANT and axillary block provide comparable intraoperative analgesia for dual mobility trapeziometacarpal prosthesis, with comparable safety profiles. WALANT offers advantages in ease of administration, absence of tourniquet-related risks, and potential for intraoperative functional testing. Axillary block provides more prolonged postoperative analgesia in the first 3 h. The choice between techniques should be individualized based on patient-specific factors, anxiety profile, and local expertise. These results should be interpreted as preliminary and hypothesis-generating, given the exploratory design, the small sample size, and the limited statistical power of the study.
1. Introduction
Thumb basal joint arthritis is a degenerative disorder of the trapeziometacarpal joint that mainly involves women after menopause, and its frequency rises steadily with advancing age. When splints, oral medication, and image-guided injections have been tried for at least three months without meaningful relief, most patients are eventually steered toward a surgical solution. Over roughly the last twenty years, dual-mobility implants have progressively established themselves as a realistic alternative to more traditional procedures, as they tend to preserve thumb motion and pinch strength more effectively in everyday use [1,2,3,4].
For many years, these surgeries were almost always carried out under regional block or general anesthesia with an ischemic tourniquet on the arm. More recently, the WALANT approach (Wide-Awake Local Anesthesia No Tourniquet) has changed the routine in many hand units, making it possible to perform the same procedures without a tourniquet and with the patient awake, able to move the thumb and interact with the surgeon during the surgery [5,6].
The physiological basis of WALANT is relatively simple: generous field infiltration of a local anesthetic combined with a vasoconstrictor produces stable analgesia and a reasonably dry field, while avoiding both the ischemia–reperfusion phenomena and the post-tourniquet discomfort that can follow cuff inflation. Fears about epinephrine-induced digital ischemia, largely rooted in historical case reports, have been progressively scaled back by more recent series [7,8]. Despite this, experience with WALANT specifically applied to trapeziometacarpal arthroplasty is still limited, probably because the procedure involves both extensive soft-tissue work and bone preparation.
Against this background, the present study was designed as a prospective comparison between WALANT and axillary brachial plexus block in patients undergoing dual mobility trapeziometacarpal arthroplasty, with the aim of filling a gap in the current literature and offering practical guidance on anesthetic choice for this surgery.
This investigation was conceived as a prospective observational comparative exploratory study, aimed at describing and comparing intraoperative analgesia and early postoperative pain between two routinely used anesthetic techniques in clinical practice (WALANT and axillary brachial plexus block) for dual mobility trapeziometacarpal arthroplasty. The working hypothesis was that WALANT would provide clinically comparable intraoperative pain control and a similar short-term safety profile to axillary block in this setting.
2. Materials and Methods
2.1. Study Design
A prospective observational comparative study of 21 patients undergoing dual mobility trapeziometacarpal prostheses was carried out at two hospitals of ASL Roma 5 (Ospedale Coniugi Bernardini di Palestrina, Ospedale SS. Gonfalone di Monterotondo, Rome, Italy) from February–December 2025. Assignment to the two anesthetic groups (WALANT n = 11, block n = 10) occurred in a non-randomized manner based on patient-specific preferences and clinical considerations of the anesthetic and surgical team.
Given the non-randomized allocation and small sample size, the study was designed as an exploratory, hypothesis-generating comparison rather than a formal non-inferiority trial.
Patients receiving WALANT anesthesia were designated as Group A. Patients receiving axillary block anesthesia were designated as Group B.
2.2. Inclusion and Exclusion Criteria
2.2.1. Inclusion Criteria
- -
- Thumb basal joint arthritis at stage II or III according to Eaton–Littler radiographic classification
- -
- History of pain at the base of the thumb with functional limitation in daily activities
- -
- No response to at least 3 months of conservative treatment (oral NSAID therapy and local corticosteroid infiltrations)
- -
- Written informed consent
2.2.2. Exclusion Criteria
- -
- Medical contraindications to either of the anesthetic techniques (allergies, serious comorbidities, hemodynamic instability)
- -
- Previous surgical interventions on the ipsilateral hand at the same location
- -
- Inability to understand the study protocol
- -
- Preexisting peripheral neuropathy interfering with pain assessment
- -
- Pantrapezoidal arthritis at stage IV according to Eaton–Littler, especially with involvement of the scaphotrapeziotrapezoid (STT) joint
- -
- Positive history of trapezium fractures, poor bone stock of trapezium, or insufficient trapezium height (<8 mm)
2.3. Population Characteristics
The study included 21 patients (19 females, 2 males), mean age 61.4 years (range 49–75). Group A comprised 11 patients (10 females, 1 male; mean age 63.5 ± 8.0 years), and Group B comprised 10 patients (9 females, 1 male; mean age 59.7 ± 7.5 years). No statistically significant differences were present in sex and age distribution between the groups.
2.4. Anesthetic Protocols
All surgical procedures were performed according to one of two standardized anesthetic protocols, with non-random assignment made by the patient and anesthetic/surgical team according to the surgeons’ and anesthesiologists’ preferences.
Group A (n = 11): Infiltration of mepivacaine 1% (20 mg/mL) with epinephrine 1:100,000 and sodium bicarbonate 8.4% (0.5%), total volume 15–20 mL, infiltrated around the TMC joint and surrounding soft tissues. Standardized waiting time: 30 min. No patient received sedatives, additional analgesics, or tourniquet. All patients remained conscious and cooperative.
Group B (n = 10): Premedication with midazolam 1 mg IV and fentanyl 50 mcg IV. Axillary brachial plexus block under ultrasound guidance with ropivacaine 0.5–0.7% (20–25 mL). Pneumatic tourniquet at 250 mmHg applied at the root of the upper extremity to reduce operative bleeding.
Mepivacaine 1% with epinephrine was chosen for WALANT because it represents the standard local anesthetic solution for wide-awake hand procedures in our institutions, can be safely combined with epinephrine, and is compatible with sodium bicarbonate buffering without visible precipitation, in line with current WALANT practice recommendations.
Ropivacaine 0.5–0.7% was used for the axillary brachial plexus block as it is the routine long-acting local anesthetic for brachial plexus blocks in our centers, offering a prolonged sensory block and extended postoperative analgesia.
2.5. Surgical Technique
All procedures utilized the same standardized technique with TOUCH® dual mobility prosthesis. The approach was dorsolateral, 3 cm, centered on the TMC joint. The procedure included: removal of osteophytes, metacarpal base osteotomy, trapezial reaming, and prosthetic cup placement. All surgeries were performed by the same surgical team, consistently composed of two hand surgeons who alternated as first and second operator, both with more than 5 years of specific experience in dual-mobility trapeziometacarpal arthroplasty.
2.6. Outcomes Assessed
- -
- Primary outcome: pain assessed via the Visual Analogue Scale (VAS 0–10) at three timepoints: during anesthesia induction, intraoperatively, and 3 h postoperatively.
- -
- Secondary outcomes: anesthetic and surgical complications; anesthetic failure/conversion to general anesthesia.
2.7. Statistical Analysis
Between-group comparisons for VAS were performed using Mann–Whitney U test (primary test, given the violation of normality in Group B for intraoperative VAS, Shapiro–Wilk p = 0.014) and Welch’s t-test (validation). Effect size was calculated with Cohen’s d. Significance level was α = 0.05. Statistical power was estimated ~0.25–0.30 to detect a medium effect (d = 0.43) with n = 10–11 per group.
Given the small sample size and low statistical power, the study was planned as an exploratory observational comparison. It was not powered or structured as a formal non-inferiority or equivalence trial, and an a priori non-inferiority margin was not defined.
2.8. Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki, and all patients provided written informed consent. According to the institutional policy, this non-interventional observational study of routine clinical practice did not require formal ethics committee approval (see Institutional Review Board Statement).
3. Results
3.1. Pain During Anesthesia Induction
Group A: 3.18 ± 2.89 (median 3.0; range 0–8)
Group B: 2.20 ± 2.37 (median 1.5; range 0–6)
- -
- Welch’s t-test: p = 0.393
- -
- Mann–Whitney U: p = 0.387
- -
- Effect size (Cohen’s d): 0.37 (small effect)
These results indicate that discomfort related to the anesthetic procedure is comparable between the WALANT and axillary block in the studied sample.
3.2. Intraoperative Pain
Group A: 2.27 ± 1.79 (median 2.0; range 0–5)
Group B: 2.00 ± 2.71 (median 1.0; range 0–8)
- -
- Welch’s t-test: p = 0.906
- -
- Mann–Whitney U: p = 0.898
- -
- Cohen’s d: 0.10 (negligible effect)
No significant differences were observed in intraoperative pain control between the WALANT and axillary block; both techniques provided adequate analgesia during the procedure.
3.3. Pain at 3 h Postoperative
Group A: 4.36 ± 2.54 (median 5.0; range 2–10)
Group B: 3.00 ± 3.08 (median 2.0; range 0–8)
- -
- Welch’s t-test: p = 0.244
- -
- Mann–Whitney U: p = 0.190
- -
- Cohen’s d: 0.43 (small-to-medium effect)
3.4. Temporal Pain Profile
Group A: 3.18 → 2.27 → 4.36 (28.6% reduction intraoperatively, 92.1% increase postoperatively)
Group B: 2.20 → 2.00 → 3.00 (9.1% reduction intraoperatively, 50% increase postoperatively)
Both techniques provided good intraoperative pain control, but the plexic block showed a more prolonged analgesia profile in the first postoperative hours compared to WALANT. Figure A2.
3.5. Anesthesia/Surgery Duration
Group A: anesthesia 33.5 ± 8.2 min; surgery 59.4 ± 24.4 min
Group B: anesthesia 24.1 ± 5.3 min; surgery 66.3 ± 12.2 min
No statistical significant differences.
3.6. Complications
There was no anesthetic failures, no conversions to general anesthesia, and no neurological or ischemic complications in either group. One patient in Group B underwent concurrent tenosynovitis release for de Quervain’s disease without showing any differences in pain profile.
No patient in either group required suspension of the procedure or conversion to general anesthesia due to pain or excessive bleeding.
4. Discussion
4.1. Pain Perception During Anesthetic Administration
This study evaluated differences in pain perception between two groups during anesthetic administration. In Group A, patients remained conscious and awake during WALANT infiltration around the trapeziometacarpal joint, fully perceiving the injection and local discomfort. In contrast, Group B patients received premedication with midazolam 1 mg IV and fentanyl 50 mcg IV administered 10 min before nerve block execution. These drugs are used for their sedative effects and may cause mild anterograde amnesia, significantly reducing awareness and pain perception during axillary block execution [9,10].
Consequently, although VAS pain during anesthesia did not differ statistically between groups (WALANT 3.18 vs. block 2.20; p = 0.393), this equivalence was influenced by premedication: Group A patients fully perceived injection discomfort while fully conscious, whereas Group B patients had reduced awareness and memory of block execution due to sedation.
Despite this difference, VAS scores during anesthetic induction were similar between groups, suggesting that WALANT can provide adequate analgesia during local infiltration. However, this apparent equivalence must be interpreted with caution, as premedication (midazolam and fentanyl) in the block group represents a major confounder that likely reduced both awareness and recall of injection-related discomfort, thereby biasing VAS ratings in favor of the axillary block.
4.2. Comparable Intraoperative Analgesia
Study data demonstrated comparable analgesia between Group A and Group B (Group A 2.27 vs. Group B 2.00; p = 0.898, negligible effect size). This finding agrees with recent randomized studies. Faraz et al. (2025), in a randomized study of 130 patients undergoing endoscopic carpal tunnel release, reported similar intraoperative pain and comfort between WALANT (100% anesthetic success) and ultrasound-guided axillary block (89% success), although WALANT was numerically superior in the absence of failures [11].
This finding contradicts the misconception that lidocaine or mepivacaine anesthesia does not provide sufficient analgesia during surgical interventions requiring invasive bone procedures. Local anesthetic appropriately infiltrated around the periosteum provides complete analgesia—when local anesthetic directly contacts the periosteum, it blocks nociceptive stimuli at their source in periosteal nociceptors, providing clinically comparable analgesia to regional nerve block, with the advantage that the patient remains conscious and can communicate any discomfort [12].
4.3. Postoperative Pain: Pharmacological Divergence, Not Technical
In the present study, Group B showed a trend toward lower pain at 3 h postoperative (WALANT 4.36 vs. block 3.00; p = 0.244). However, this divergence does not reflect intrinsic WALANT inferiority but rather the choice of local anesthetic used.
Group A received mepivacaine 1%, with duration of action approximately 30–60 min. At 3 h postoperatively, the anesthetic effect had worn off, explaining the higher VAS (4.36).
Group B received ropivacaine 0.5–0.7%, with duration of action 120–180 min. At 3 h, the anesthetic effect was still present, reducing the pain (VAS 3.00).
In our series, the trend toward lower pain at 3 h in the axillary block group is more plausibly explained by the use of a long-acting local anesthetic (ropivacaine) in Group B versus an intermediate-acting agent (mepivacaine) in the WALANT group, rather than by an intrinsic limitation of the WALANT technique itself. Ropivacaine provides a significantly longer sensory block than intermediate-acting agents, but, unlike lidocaine or mepivacaine, it is poorly compatible with the buffered mixtures commonly used in WALANT—several in-vitro studies and a recent WALANT-specific experimental work have shown that ropivacaine tends to precipitate when alkalinized with sodium bicarbonate to near-physiological pH, making its routine use in bicarbonate-buffered WALANT solutions impractical [13].
Consequently, in our protocol, mepivacaine was selected for WALANT because it can be safely combined with epinephrine and buffered with sodium bicarbonate without visible precipitate, whereas ropivacaine was reserved for the axillary block where no bicarbonate buffering is required.
4.4. Excellent Safety: Tourniquet vs. Epinephrine
A critical issue underlying this study concerns the distinct safety profile of the two techniques.
Tourniquet (Group B): In our study, zero complications were observed, with mean duration well below the critical threshold of 120 min. However, Chang et al. (2022), in a systematic review, reported 64 cases of tourniquet-related nerve injury, although 89.7% fully recovered [14]. Oragui et al. (2010) documented that occlusion periods > 180 min induce significant cellular damage, while periods < 120 min are biologically safe [15].
Epinephrine (Group A): Historically, there was concern about ischemic risk related to epinephrine injection. Denkler (2001), analyzing 48 cases of digital gangrene reported over 120 years of literature, found that no cases were associated with commercial lidocaine with epinephrine use [7,16]. Mantilla-Rivas et al. (2019), evaluating 402 polydactyly excision procedures in neonates (population with critical vascularization) with lidocaine + epinephrine, reported zero cases of digital ischemia [17].
In our study, no ischemic complications were observed in Group A. The concentration of epinephrine 1:100,000 confirms safety when properly administered, supported by a meta-analysis of 15,595 WALANT-treated patients, in which only 1.7% experienced complications and no patient digital ischemia [18].
4.5. WALANT: Not an Operator-Dependent Technique
A fundamental difference between the two techniques concerns ease of execution and operator dependence.
Axillary block: This is an operator-dependent technique in terms of anesthetic success. Chan et al. (2007) reported success rates varying from 62.9% (nerve stimulation alone) to 82.8% (ultrasound alone) depending on technique [19]. Sites et al. (2006) documented a 29% conversion rate to general anesthesia with the transarterial technique, reduced to 0% with ultrasound-guided technique [20]. In our study, 100% success in Group B reflects operator expertise; however, an intrinsic failure risk remains when the anesthetist lacks adequate training.
WALANT: This is less vulnerable to failure related to poor operator competence. The technique’s learning curve is brief, being a local infiltration. Lawand et al. (2024), in a systematic meta-analysis of 79 studies and 15,595 WALANT patients, reported an overall complication rate of only 1.7%, without documented anesthetic failures [18]. In the present study, 100% anesthetic success was also achieved in Group A, consistent with the technique’s intrinsic stability. No differences in analgesia were noted depending on operator experience in anesthetic administration.
4.6. Clinical Implications
In the context of TMC arthroplasty, WALANT is safe and effective, comparable to the axillary block for intraoperative analgesia, with the added advantage of permitting intraoperative functional testing (assessment of range of motion and joint stability with conscious patient) and, consequently, more targeted prosthesis selection. Axillary block remains a valid technique, particularly for patients with high preoperative anxiety or when WALANT is not feasible, offering more prolonged postoperative analgesia.
4.7. Limitations
The present study has several limitations: (1) small sample size (n = 21) with limited statistical power (0.25–0.30); (2) non-randomized group assignment; (3) brief follow-up (3 h), insufficient to assess pain profile at 24 h or beyond; (4) only two centers within the same ASL, with limited generalizability; (5) absence of systematic documentation of intraoperative functional testing; (6) absence of short and long-term functional outcomes (ROM, grip strength, QuickDASH).
In addition, the low statistical power of the present study (approximately 0.25–0.30 for detecting a medium effect size) implies a substantial risk of type II error. Therefore, non-significant between-group differences in VAS scores should not be interpreted as proof of equivalence or formal non-inferiority. Finally, the use of sedation in the axillary block group and the choice of local anesthetics with different duration of action (mepivacaine vs. ropivacaine) represent major confounders that may have influenced both pain perception during anesthetic administration and early postoperative pain and must be taken into account when interpreting our findings.
5. Conclusions
This prospective observational comparative study represents, to our knowledge, the first comparison between WALANT and axillary block specifically for dual mobility trapeziometacarpal prostheses. Both techniques provided low VAS scores during anesthetic administration and intraoperatively, with an excellent short-term safety profile. It is important to underline that this comparability was observed despite the use of different anesthetic pharmacological protocols: in the axillary block group, sedative premedication with midazolam and fentanyl, which reduces awareness and recall of injection-related pain, was combined with long-acting ropivacaine, whereas in the WALANT group no sedation was used and an intermediate-acting mepivacaine solution was administered.
WALANT offers additional practical advantages, including independence from the risk of block failure, the absence of tourniquet-related risks, and the possibility of performing intraoperative functional testing (assessment of range of motion and implant stability with a conscious patient), which may help the surgeon select the most appropriate prosthetic components to implant.
Axillary block remains a valid option, particularly for patients with high preoperative anxiety or when WALANT is not feasible, and it provides a longer early postoperative analgesic effect in our protocol, largely due to the use of long-acting ropivacaine compared with intermediate-acting mepivacaine in the WALANT group.
Surgeons did not perceive any clinically problematic bleeding in either group, and no case required interruption of the procedure, conversion to another anesthetic technique, or, in the WALANT group, tourniquet inflation because of bleeding (Video S1). However, intraoperative blood loss was not systematically quantified, and the operating surgeons were not blinded to the anesthetic technique, so these observations must be considered descriptive and do not allow robust comparative conclusions regarding bleeding between WALANT and the axillary block.
This study has several important limitations: the small sample size (n = 21) with low statistical power (approximately 0.25–0.30), the non-randomized allocation to groups, the very short follow-up (3 h) limited to early pain assessment, and the use of different local anesthetics and sedation protocols between groups.
Within these constraints, our findings should be interpreted as preliminary, hypothesis-generating data suggesting that WALANT is a feasible and safe anesthetic option for dual mobility trapeziometacarpal arthroplasty, providing short-term intraoperative analgesia that appears comparable to axillary block in routine practice. The choice between techniques should remain individualized based on patient-specific factors, anxiety profile, comorbidities, and local expertise. Future larger randomized studies and extended follow-up, including functional outcomes (ROM, grip strength, QuickDASH), are needed to confirm these preliminary observations and to formally evaluate non-inferiority and long-term recovery profiles for both techniques.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/surgeries7010031/s1, Supplementary Video S1: WALANT TMC arthroplasty showing intraoperative implant testing without tourniquet.
Author Contributions
Conceptualization: E.B.; Methodology: E.B.; Investigation: E.B.; Formal Analysis: E.B.; Writing—Original Draft Preparation: A.R.; Writing—Review and Editing: G.K. and G.F.; Supervision: M.G. All authors have read and agreed to the published version of the manuscript.
Funding
This study received no external funding.
Institutional Review Board Statement
This study was conducted in accordance with the principles of the Declaration of Helsinki. According to the policy of the Ethics Committee of ASL Roma 5 (Comitato Etico di ASL Roma 5), formal ethical approval was not required because the project is a non-interventional observational study of standard clinical practice, with no deviation from routinely adopted diagnostic or therapeutic pathways and with anonymized data collection. This classification is consistent with the definition of non-interventional clinical studies provided by Regulation (EU) No. 536/2014, Article 2(2)(4), and with the Italian national framework on observational studies (AIFA Guideline for the classification and conduct of observational studies, Determina n. 425/2024), which distinguish non-interventional observational studies in which the therapeutic strategy follows routine clinical practice and is not determined by the study protocol.
Informed Consent Statement
Written informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data presented in this study are available upon request from the corresponding author for purposes of scientific research and compliance with ethical requirements.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CMC-1 | Carpometacarpal joint of the thumb (thumb basal joint) |
| WALANT | Wide-Awake Local Anesthesia No Tourniquet |
| TMC arthroplasty | Trapeziometacarpal arthroplasty |
| TMC | Trapeziometacarpal (joint) |
| ASL | Azienda Sanitaria Locale |
| IV | Intravenous |
| NSAID | Non-Steroidal Anti-Inflammatory Drug |
| STT | Scaphotrapeziotrapezoid (joint) |
| ROM | Range of Motion |
| QuickDASH | Quick Disabilities of the Arm, Shoulder and Hand questionnaire |
| IVRA | Intravenous Regional Anesthesia |
| VAS | Visual Analogue Scale |
| α | Alpha (statistical significance level) |
| SD | Standard Deviation |
| n° | Sample size |
| p | p-value |
| d | Cohen’s d (effect size) |
Appendix A

Table A1.
Results.
Table A1.
Results.
| Timepoint | WALANT Mean ± SD | BLOCK Mean ± SD | Difference | t-Test p-Value | Mann–Whitney p-Value | Cohen’s d | Sig. |
|---|---|---|---|---|---|---|---|
| VAS During Anesthesia | 3.18 ± 2.89 | 2.20 ± 2.37 | 0.98 | 0.393 | 0.387 | 0.37 | ns |
| VAS During Surgery | 2.27 ± 1.79 | 2.00 ± 2.71 | 0.27 | 0.906 | 0.898 | 0.10 | ns |
| VAS 3 h Post-op | 4.36 ± 2.54 | 3.00 ± 3.08 | 1.36 | 0.244 | 0.190 | 0.43 | ns |
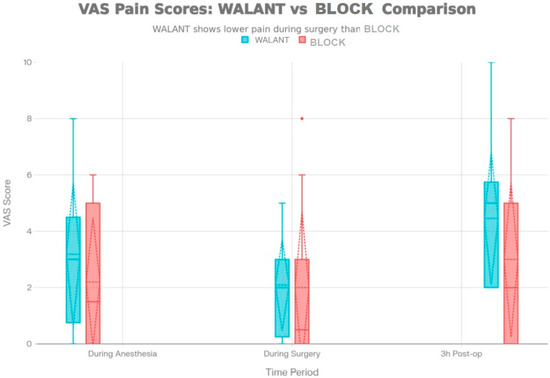
Figure A1.
VAS pain score.
Figure A1.
VAS pain score.
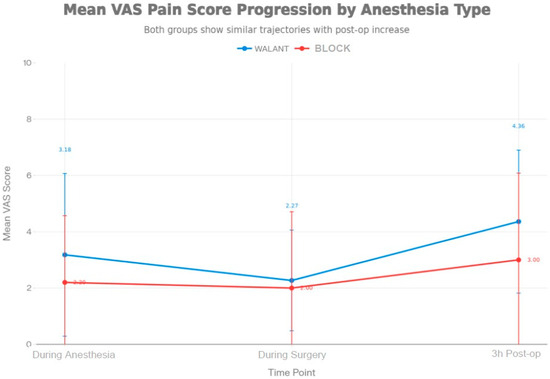
Figure A2.
Pain score progression by anesthesia type.
Figure A2.
Pain score progression by anesthesia type.
References
- Eaton, R.G.; Littler, J.W. Ligament Reconstruction for the Painful Thumb Carpometacarpal Joint. J. Bone Jt. Surg. 1973, 55, 1655–1666. [Google Scholar] [CrossRef]
- Tomaino, M.M.; Pellegrini, V.D., Jr.; Burton, R.I. Arthroplasty of the basal joint of the thumb. Hand Clin. 2001, 17, 203–215. [Google Scholar]
- Frittella, G.; Arioli, L.; Guzzini, M. Outcomes of dual mobility arthroplasty in thumb basal joint arthritis: A clinical and radiographic study of one hundred and fifty prostheses with four-years follow-up. Int. Orthop. 2025, 49, 2867–2873. [Google Scholar] [CrossRef] [PubMed]
- Smeraglia, F.; Carità, E.; Frittella, G.; Tamborini, F.; Diaz, L.; Donadelli, A.; Guzzini, M. Intra-patient comparison of trapeziectomy with ligament reconstruction and tendon interposition versus dual mobility prosthesis for trapeziometacarpal osteoarthritis: A multicenter observational study. Eur. J. Orthop. Surg. Traumatol. 2025, 35, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Lalonde, D.H. Reconstruction of the Hand with Wide Awake Surgery. Clin. Plast. Surg. 2011, 38, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Lalonde, D.H. Wide awake hand surgery. Plast. Surg. 2014, 22, 8–15. [Google Scholar]
- Denkler, K. A Comprehensive Review of epinephrine in the finger: To do or not to do. Plast. Reconstr. Surg. 2001, 108, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Finsen, V. Local anaesthetic with adrenaline in digits is safe. Acta Orthop. 2013, 84, 321–323. [Google Scholar]
- Dundee, J.W.; Halliday, N.J.; Harper, K.W.; Brogden, R.N. Midazolam a review of its pharmacological properties and therapeutic use. Drugs 1984, 28, 519–543. [Google Scholar] [CrossRef] [PubMed]
- Reves, J.G.; Fragen, R.J.; Vinik, H.R.; Greenblatt, D.J. Midazolam: Pharmacology and uses. Anesthesiology 1985, 62, 310–324. [Google Scholar] [PubMed]
- Virtos, M.; Chassery, C.; Marty, P.; Basset, B.; Casalprim, J.; Vuillaume, C.; De Lussy, A.; Atthar, V.; Naudin, C.; Joshi, G.; et al. Wide awake local anesthesia no tourniquet (WALANT) versus ultrasound-guided axillary block in carpal tunnel release: A non-inferiority randomized controlled trial. Reg. Anesth. Pain Med. 2025, 50, 107152. [Google Scholar] [CrossRef]
- Joshi, G.P.; Machi, A. Surgical site infiltration: A neuroanatomical approach. Best. Pract. Res. Clin. Anaesthesiol. 2019, 33, 317–324. [Google Scholar] [CrossRef] [PubMed]
- AlShammari, A.; Harrison, J.M.; Greene, B.; Kwofie, M.K.M.; Lalonde, D.H.M.; Krauss, E.M.M. A practical guide to local anesthetic options for wide-awake hand surgery (WALANT): A laboratory-based experimental study. Plast. Reconstr. Surg. Glob. Open 2025, 13, e6730. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Bhandari, L.; Messana, J.; Alkabbaa, S.; Hamidian Jahromi, A.; Konofaos, P. Management of tourniquet-related nerve injury (TRNI): A systematic review. Cureus 2022, 14, e27685. [Google Scholar] [CrossRef] [PubMed]
- Oragui, E.; Parsons, A.; White, T.; Longo, U.G.; Khan, W.S. Tourniquet use in upper limb surgery. Hand 2011, 6, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Mantilla-Rivas, E.; Tan, P.; Zajac, J.; Tilt, A.; Rogers, G.F.; Oh, A.K. Is epinephrine safe for infant digit excision? A retrospective review of 402 polydactyly excisions in patients younger than 6 months. Plast. Reconstr. Surg. 2019, 144, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Lawand, M.; Hantouly, A.; Bouri, F.; Muneer, M.; Farooq, A.; Hagert, E. Complications and side effects of wide-awake local anaesthesia no tourniquet (WALANT) in upper limb surgery: A systematic review and meta-analysis. Int. Orthop. 2024, 48, 1257–1269. [Google Scholar] [CrossRef] [PubMed]
- Chan, V.W.S.; Perlas, A.; McCartney, C.J.L.; Brull, R.; Xu, D.; Abbas, S. Ultrasound guidance improves success rate of axillary brachial plexus block. Can. J. Anaesth. 2007, 54, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Sites, B.D.; Gallagher, J.D.; Cravero, J.; Lundberg, J.; Blike, G. The learning curve associated with a simulated ultrasound-guided interventional task by inexperienced anesthesia residents. Reg. Anesth. Pain Med. 2004, 29, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Guzzini, M.; Arioli, L.; Biondi, E.; Romano, A. Clinical and radiological evaluation after dual mobility cup prostheses for the treatment of trapeziometacarpal osteoarthritis: A 2-year follow-up study. Riv. Chir. Mano 2021, 58, 45–53. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
