Abstract
Background/Objectives: The prevalence of joint replacement surgeries has significantly increased over the last century, leading to a corresponding rise in complications, particularly periprosthetic joint infection (PJI). The management of a PJI involves various strategies, including debridement, antibiotic therapy, and staged revision procedures. A notable advancement in treatment is the use of calcium sulfate reabsorbable carriers, recognized for their biocompatibility, osteoconductivity, and localized antibiotic delivery. Recent reports indicate that when combined with conventional treatment regimens, calcium sulfate carriers can achieve infection eradication rates exceeding 90%. This study aims to evaluate the efficacy of calcium sulfate carriers in managing periprosthetic infections, specifically assessing their impact on healing rates in patients undergoing treatment. Study Design & Methods: A retrospective analysis was conducted at our institution, focusing on patients diagnosed with PJIs treated with 2-stage revision surgery with local application of calcium sulfate carriers with antibiotics at both stages, and systemic antibiotic therapy, and comparing results with different surgical procedures. Results: The study included 40 patients (24 males and 16 females), with a mean age of 68.7 (range 48–87) years. The affected joints included the hip (27.5%), shoulder (27.5%), and knee (45%). The findings revealed that 97% of patients achieved infection eradication at the end of the follow-up period. Conclusions: These results highlight the complexities of managing PJIs and the significant role of calcium sulfate carriers in improving outcomes, supporting their use as a standard practice in confirmed PJI cases.
1. Introduction
Prosthetic joint replacement is a highly effective procedure for the treatment of various disabling conditions, with excellent long-term outcomes. Over the past 20 years, there has been a progressive increase in the number of joint replacement procedures across all joints. This trend is attributable to a decrease in the average age at the time of first implantation and the increased functional demands of the aging population [1]. However, this rise has also led to an increase in revision procedures. One of the leading causes of prosthetic revision is periprosthetic joint infection (PJI), which represents the primary cause of revision in hip prostheses [1,2], and the second most common cause for knee and shoulder prostheses [1,2,3,4,5]. PJI rates have been reported to range between 0.3% and 2.5%, with approximately 2% consistently documented in several studies [6,7,8], a figure considered quite concerning. The mortality rate associated with these infections is estimated to range between 0.5% and 2% [9,10]. Various microorganisms can be responsible for PJIs, with Staphylococcus species—particularly Staphylococcus aureus—being among the most prevalent [6,11]. If not properly diagnosed or effectively treated, PJIs may lead to chronic infection, persistent pain and disability, hematogenous spread, and even death [12,13]. The cornerstone of effective treatment lies in timely and accurate diagnosis, and for this reason, diagnostic criteria have evolved over time [14,15,16,17,18]. Similarly, depending on the clinical presentation of the infection, different therapeutic strategies and algorithms have been developed. Key treatment options include, depending on type of PJI and surgeon’s preferences, debridement, antibiotics, and implant retention (DAIR); debridement, antibiotic pearls, and retention of the implant (DAPRI); and 1-stage, 1.5-stage, and 2-stage revision procedures, all accompanied by systemic antibiotic therapy. These strategies have become essential components in the management of PJIs, though each has specific indications and limitations [19,20]. However, because infected tissues are often poorly vascularized and biofilm formation on artificial joints limits the efficacy of systemic antibiotic therapy [21], various methods have been developed to deliver higher local concentrations of antibiotics, thus minimizing the complications associated with systemic administration. Several calcium sulfate-based carriers have been introduced into clinical practice, and they have gained attention due to their biocompatibility, osteoconductive properties, and ability to deliver antibiotics locally at high concentrations [22,23]. Unlike polymethylmethacrylate, calcium sulfate is fully resorbable and therefore does not require a subsequent surgical procedure for removal. Its gradual dissolution over time prevents bacterial colonization and eliminates the constraint of using only heat-stable antibiotics, thereby allowing a broader spectrum of antimicrobial agents to be employed. These features make calcium sulfate carriers particularly suitable for local antibiotic delivery in the setting of periprosthetic joint infection. Nonetheless, the literature on their efficacy remains heterogeneous and inconclusive [21,24,25,26,27,28,29,30,31,32]. Given the conflicting results reported in the current literature, our study aims to contribute to the ongoing debate by offering additional clinical evidence. The present case series may therefore help to broaden and refine the current understanding of this complex topic.
In this article, we present the results of a retrospective observational study conducted on patients affected by PJI, who were treated with 2-stage revision surgery and the local application of antibiotic-loaded reabsorbable carriers.
2. Materials and Methods
Study Design. A retrospective observational study was conducted at our Institution—Azienda Ospedaliera Universitaria Senese (AOUS) Policlinico Santa Maria alle Scotte. This is a nationally significant and highly specialized hospital, serving as the referral center for the territory of South-East Tuscany and functioning as the primary hospital for the city of Siena and the surrounding municipalities. The inclusion criteria were as follows: (a) patients diagnosed with PJI according to the 2021 European Bone and Joint Infection Society (EBJIS) definition criteria [17]; (b) those requiring prosthetic revision surgery (DAPRI, 1-stage, 1.5-stage, or 2-stage procedures); (c) patients managed with a standardized multidisciplinary treatment approach; and (d) patients who completed at least 12 months of follow-up. All patients were managed by a multidisciplinary team comprising orthopedic surgeons and infectious disease specialists. No exclusion criteria were applied with respect to age, sex, ethnicity, pre-existing comorbidities, previous surgeries, or prior PJI treatments. During the follow-up period, any complication was recorded, including failure of the procedure due to infection recurrence requiring further surgical intervention, the need for salvage procedures (resection arthroplasty, permanent antibiotic-loaded spacer implantation, amputation or disarticulation), and patient death. At the end of the follow-up period, patients were considered cured if the treatment was successful in eradicating the infection; otherwise, persistence of infection was diagnosed. Figure 1 provides a schematic overview of the experimental procedure.
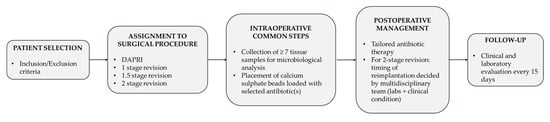
Figure 1.
Schematic flowchart of the study design and patient management protocol. Following patient selection, subjects were assigned to one of four surgical procedures (DAPRI, 1-stage, 1.5-stage, or 2-stage revision). Regardless of the procedure type, three core intraoperative steps were performed in all cases: (1) collection of no less than seven tissue samples for microbiological analysis; (2) placement of calcium sulfate beads loaded with antibiotic(s); and (3) administration of tranexamic acid when not contraindicated. Postoperatively, systemic antibiotic therapy was tailored by the infectious disease specialist according to culture results or broad-spectrum regimens if needed. In 2-stage revisions, the timing of reimplantation was based on normalization of laboratory markers and clinical assessment by the multidisciplinary team. Follow-up comprised clinical and laboratory evaluations every 15 days.
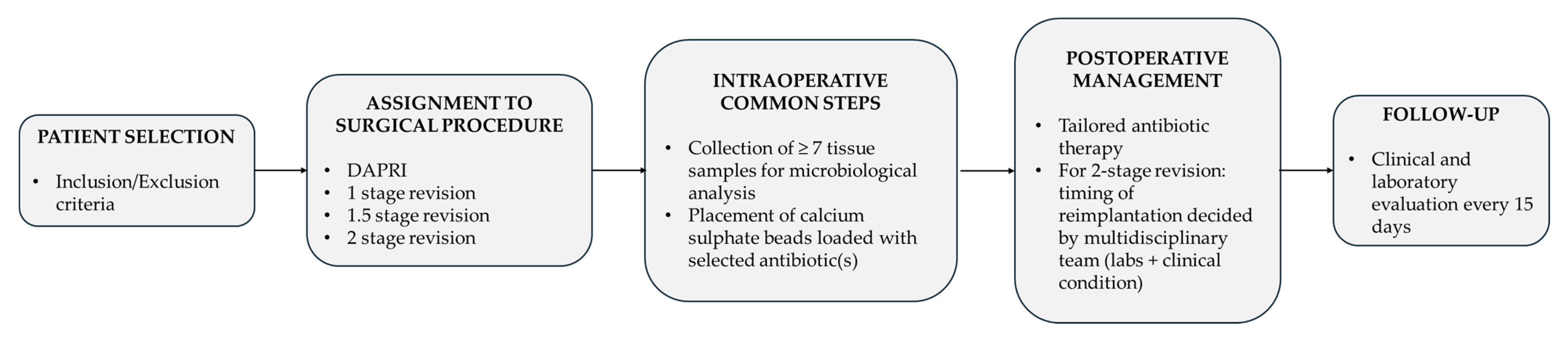
Surgical Technique. The choice of surgical procedure (DAPRI, 1-stage, 1.5-stage, or 2-stage revision) needed was based on patient-specific factors, time since the onset of symptoms and previous surgery, and the identification of an etiological agent. The DAPRI surgical procedure was performed according to the technique described by Indelli et al. [33] for the knee and adapted to the joint in question. The original technique involves the injection of a methylene blue solution into the joint to highlight contaminated areas, followed by aggressive debridement and a combined thermal (argon plasma coagulator, APC) and mechanical (brush impregnated with 4% chlorhexidine gluconate) removal of biofilm from the metallic surfaces, along with the replacement of all mobile components of the prosthesis (Figure 2).
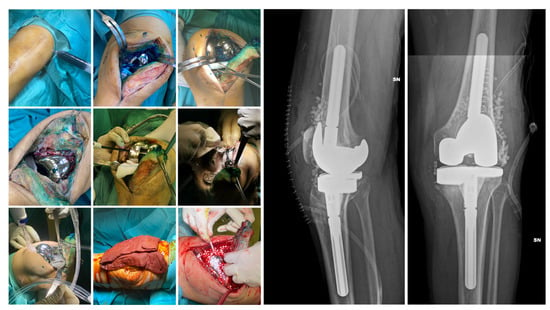
Figure 2.
DAPRI procedure for the knee. Methylene blue solution is injected into the joint before skin incision is made and distributed with flexion–extension cycles. The polyethylene insert is removed, and a tumor-like debridement of the stained portions is performed. Thermal debridement of the prosthetic surfaces is performed with an APC, followed by mechanical debridement with a sponge soaked in 4% chlorhexidine gluconate and irrigation with normal saline and povidone–iodine solution. Finally, the insert is replaced, and calcium sulfate beads loaded with the targeted antibiotic(s) are positioned. In the postoperative X-rays the beads are visible.
In all cases, mobile components were removed, including the insert and head for hip prostheses; the polyethylene insert for knee prostheses; and the glenosphere, insert, and adapter tray for shoulder prostheses. Particular care was taken to remove all tissues stained with methylene blue, especially in hard-to-reach areas such as the posterior capsular portion of the knee. Additionally, attention was paid to the precise use of an APC on all metallic surfaces. Surgical procedures adhered to the following protocol. Patients underwent meticulous debridement, and at least 7 tissue samples were collected from different areas for microbiological analysis, alongside the explanted prosthetic components or antibiotic-loaded spacers. In all surgeries, calcium sulfate carriers (Stimulan®, Biocomposite, Keele, UK) were used, impregnated with the most appropriate antibiotic/antibiotics based on antibiogram results, or with broad-spectrum antibiotics if the pathogen was unknown. Stimulan® was selected due to its therapeutic flexibility, as it is not preloaded with a specific antibiotic. This feature allows customization of the antibiotic choice according to the patient’s profile and the type of infection, enabling the use of a wide range of antibiotics and tailoring therapy to individual clinical needs. Calcium sulfate carrier was used in all cases in the form of beads, which were placed within the joint space and, in 2-stage revision procedures, also within the medullary canals at the time of spacer implantation. Since the antibiotic used was not the same in all procedures—being either tailored to the antibiogram or selected as a broad-spectrum agent—the preparation method was modified accordingly, based on the manufacturer’s instructions. At the end of the procedure, to control bleeding and in the absence of contraindications, tranexamic acid was administered both intravenously and topically, as previously described [34].
Postoperative Protocol. The choice of antibiotic and the duration of systemic postoperative therapy, as well as the type of antibiotic added to the carrier intraoperatively, were determined by the infectious disease specialist based on preoperative and/or intraoperative culture results. For 2-stage procedures, the timing of reimplantation was determined by a multidisciplinary team based on normalization of inflammatory markers and the patient’s clinical condition. Each patient underwent clinical and laboratory evaluations every 15 days (including complete blood count with differential, C-reactive protein, erythrocyte sedimentation rate, and procalcitonin). Clinical evaluation focused on detecting swelling, discharge, or other signs of infection.
Local Ethical Committee approval was obtained (Protocol No. 29764), and all patients provided informed consent for anonymous data collection and its use for educational and research purposes.
3. Results
Study population. A consecutive series of 40 patients (63 surgical procedures) operated on at our institution between November 2020 and April 2024 met the inclusion criteria: 24 males (60%) and 16 females (40%) with a mean age (± standard deviation, SD) at the time of surgery of 68.7 ± 11.3 (range 48–87) years and a mean follow-up of 26 ± 15 (range 12–58) months. Four patients did not complete the 12-month follow-up: one patient was lost to follow-up, and three had died. One death was related to septic complications of the PJI, while two patients died due to causes unrelated to PJI before reimplantation of the definitive prosthesis could be performed. The joints involved were the hip in 27.5% of cases (n = 11), the knee in 45% (n = 18), and the shoulder in 27.5% (n = 11). The surgical procedures performed were the DAPRI procedure in 19% (12 cases), 1-stage revisions in 1.6% (1 case), 1.5-stage revisions in 3.2% (2 cases), 2-stage revisions in 71.4% (of which 60% included prosthesis removal and antibiotic spacer implantation), and resection arthroplasty in 3.2% (2 cases) of the cases. Four patients underwent multiple revision surgeries due to infection recurrence: three males and a female with a mean age of 68.5 ± 15 (range 51–88) years. In all these cases, the patient had a history of multiple surgeries to the site of the infection. As regards to complications and outcomes, procedure failure occurred in 12.5% of cases (25% in DAPRI procedures and 7.7% in 2-stage revisions), necessitating additional surgery (2-stage revision or salvage procedure). Three patients (7.5%) died, with only one death attributed to PJI-related complications. In 20% of cases, patients underwent salvage procedures for PJI management: resection arthroplasty (5%), permanent spacer implantation (12.5%), and amputation (2.5%). Resection arthroplasty was used exclusively for hip PJIs, while permanent spacers were applied only in shoulder PJIs. Among the 36 patients who completed the 12-month follow-up, 97% (n = 35) achieved infection eradication. One patient showed persistent infection, managed with suppressive antibiotic therapy. Overall demographic characteristics are summarized in Table 1.

Table 1.
Demographic characteristics of the study population. Some patients underwent more than one surgical procedure.
Pathogen identification. Culture results were positive in 84% of surgical procedures. Notably, in one-third of theoretically “non-infected” procedures (i.e., second-stage of the 2-stage procedures), cultures were positive. Pathogens isolated were Staphylococcus species in 78% of cases: Methicillin-sensitive Staphylococcus aureus (MSSA): 43%; Methicillin-sensitive Staphylococcus epidermidis (MSSE): 23%; Methicillin-resistant Staphylococcus epidermidis (MRSE): 8.6%; Methicillin-resistant Staphylococcus aureus (MRSA): 3%; and other species: 29%. Other microorganisms identified included Pseudomonas aeruginosa (6%), Candida species (4%), and Cutibacterium acnes (7%). Polymicrobial infections were observed in 13% of cases. Differences in pathogen prevalence were noted across the three joint types. MSSA was the most frequently isolated organism in hip and knee PJIs, found in 40% and 38% of cases respectively. Cutibacterium acnes was exclusively isolated in shoulder PJIs (33%). The prevalence of polymicrobial infections also varied: 6% in hips, 9.5% in knees, and 33% in shoulders. The average volume of carrier used was 12 ± 5.2 (range 5–30) cm3. Systemic antibiotic therapy lasted between 6 and 8 weeks. Differences in microorganism prevalence are presented in Table 2.

Table 2.
Microbiology results.
4. Discussion
PJIs are an increasingly recognized complication due to their potentially devastating impact on patients’ lives [30]. Accurate diagnosis and identification of the causative organism are of paramount importance. Every opportunity should be utilized to improve diagnostic accuracy, including submitting intraoperative specimens and explanted prosthetic components for microbiological analysis. The therapeutic algorithm is highly complex and requires the collaboration of multiple specialists, including the orthopedic surgeon, the infectious disease specialist, and the microbiologist.
In our patient cohort, although there was wide variability in age, this factor did not appear to represent a risk for infection recurrence or persistence. However, due to the small sample size, it was not possible to identify a statistically significant difference or to apply formal statistical tests. A similar observation applies to sex. Nevertheless, a potential risk factor for infection persistence and recurrence was identified: a history of previous surgical procedures. In fact, these complications occurred exclusively in patients who had undergone multiple surgeries at the site affected by the PJI, such as fracture fixation or previous prosthetic replacements.
In our series, approximately one-third of the cases initially classified as “theoretically” non-infected demonstrated positive microbiological cultures, thereby refining the diagnosis and guiding appropriate treatment. Postoperatively, patients were treated with targeted parenteral antibiotic therapy. In our cohort, the predominance of Staphylococcus species as the leading pathogen aligns with the existing literature, which consistently identifies these microorganisms as major contributors to PJIs across joint types. Specifically, Staphylococcus species accounted for 78% of the identified organisms, in line with previous reports indicating that Staphylococcus aureus and coagulase-negative staphylococci are responsible for approximately 50–60% of all PJIs [35,36,37]. As regards shoulder PJIs, Cutibacterium acnes emerged as a notable pathogen contributing to approximately 33% of cases. This finding is consistent with studies highlighting Cutibacterium acnes as a significant pathogen in shoulder infections, primarily due to its biofilm-forming ability and indolent behavior, which often complicates diagnosis and management [38,39,40]. Another important feature of shoulder PJIs observed in our cohort is that approximately one-third of infections were polymicrobial, most commonly involving both Cutibacterium acnes and Staphylococcus epidermidis. Considering hip PJIs, Staphylococcus aureus remains the predominant pathogen with reported rates as high as 50% [41,42,43]. Staphylococcus epidermidis is also frequently isolated, accounting for approximately 20–30% of cases [44]. Other organisms, such as Enterococcus faecalis and Pseudomonas aeruginosa, are implicated in 5–10% of cases, especially in patients with comorbidities like diabetes or those who have undergone prolonged hospital stays [45,46]. Knee PJIs present a particularly complex microbiological profile. In our study, 9.5% of knee PJIs were polymicrobial, which supports existing evidence describing the multifactorial nature of these infections. Knee PJIs commonly involve both Gram-positive and Gram-negative bacteria, and occasionally fungi, posing substantial treatment challenges [47,48,49]. Among Gram-positive bacteria, Staphylococcus aureus and Staphylococcus epidermidis are the most frequently isolated pathogens. The latter, a common skin commensal, is known for biofilm formation on prosthetic surfaces, contributing to persistent infections. The literature reports that Staphylococcus epidermidis may be responsible for 30–40% of knee PJIs, often co-occurring with Staphylococcus aureus, which is found in 20–30% of cases [50,51]. In addition to Gram-positive organisms, Gram-negative pathogens such as Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae are also relevant, particularly in acute infections and in patients with risk factors such as diabetes or immunosuppression. Pseudomonas aeruginosa has been reported in 5–10% of cases and is associated with more severe clinical presentations and poorer outcomes [52,53]. The emergence of multidrug-resistant Gram-negative strains adds a significant challenge, necessitating more aggressive and individualized antibiotic strategies [54]. Although less frequent, fungal infections add further complexity to the management of knee PJIs. Candida species have been identified in 2–5% of cases, particularly in immunocompromised patients or those with prolonged antibiotic exposure [55,56]. Coinfection with fungal and bacterial organisms requires combined antifungal and antibiotic therapy, complicating treatment protocols [43].
The complexity of managing PJIs necessitates a multidisciplinary approach, particularly in cases involving resistant organisms or complicated infections. The involvement of infectious disease specialists is critical to optimize antibiotic therapy and guide surgical decision-making, ultimately improving patient outcomes. This collaborative strategy enhances care quality and supports a deeper understanding of the multifactorial nature of PJIs, enabling more effective diagnostic and therapeutic approaches [10,57].
Due to the poor vascularization of infected tissues and the presence of biofilms on prosthetic surfaces, systemic antibiotic therapy alone is often insufficient [21]. As a result, bioabsorbable antibiotic-loaded carriers have gained popularity, though their efficacy remains controversial in the treatment of PJIs. They have proven effective in the treatment of osteomyelitis [58,59], in the prevention of PJI in patients with non-modifiable risk factors [60], and in the management of septic arthritis [61]. These carriers have been described as facilitating localized antibiotic release, promoting osteoconductivity and gradual resorption, thereby supporting bone healing at the infection site [8,35,62,63]. The ability of calcium sulfate to deliver high local concentrations of antibiotics is particularly advantageous in the treatment of resistant organisms, which are becoming increasingly prevalent in PJIs [64,65]. In our view, the integration of calcium sulfate carriers into the treatment algorithm represents a major advancement in PJI management. The high cure rate of 97% observed in this study supports the recent literature advocating their use as a standard component in treating confirmed PJIs [26,27,28,29,30,66,67,68,69,70].
The ongoing debate between 1-stage and 2-stage revision surgery persists among orthopedic surgeons, influenced by institutional protocols and surgeon preference. However, the favorable outcomes observed in this study, particularly the high cure rate associated with the 2-stage revision approach, reinforce the current literature supporting this method as the standard of care for confirmed PJIs [70]. The Infectious Diseases Society of America has issued guidelines to assist surgeons in selecting the most appropriate surgical strategy, emphasizing the need for individualized patient management. Preoperative antibiotic administration is another key factor in the management of PJIs, as seen in our cohort where 70% of patients received antibiotics before surgery. This approach may influence microbial culture results and subsequent treatment planning. Timely and appropriate antibiotic therapy is essential for treatment success and minimizing complications [71,72]. Recent studies warn that inappropriate antibiotic use may promote the development of resistant strains, further complicating subsequent management.
5. Conclusions
Periprosthetic joint infections remain a major challenge for orthopedic surgeons. Our study underlines the importance of a multidisciplinary approach, including infectious disease specialists and microbiologists, for their effective management. The use of calcium sulfate carriers loaded with specific antibiotics showed potential as an adjuvant strategy for infection eradication, suggesting a possible role in routine PJI management. A key finding of our work is that the strongest risk factor for recurrence was represented by previous surgical procedures on the same prosthetic site. Furthermore, in one-third of the cases classified as theoretically non-infected, microbiological analyses identified pathogenic organisms, which directly influenced the therapeutic strategy. This highlights the crucial role of systematic intraoperative sampling and microbiological assessment in guiding treatment decisions. It is likely that this integrated approach—including multidisciplinary patient management, targeted intraoperative diagnostics, and the use of calcium sulfate carriers—contributed to the remarkably high healing rate observed in our cohort (97%). Despite these advances, PJIs remain complex, and further research is warranted to refine strategies for prevention, early diagnosis, and optimized treatment pathways.
Author Contributions
Conceptualization, N.M. and S.G.; data curation, S.A. and E.T.; writing—original draft preparation, S.A., C.R.C., C.L., T.G. and E.T.; writing—review and editing, E.T. and N.M.; supervision, N.M. and S.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Regional Ethics Committee for Clinical Trials of the Tuscany Region—South East Area (Protocol No. 29764, 26 February 2026).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hauer, G.; Rasic, L.; Klim, S.; Leitner, L.; Leithner, A.; Sadoghi, P. Septic complications are on the rise and aseptic loosening has decreased in total joint arthroplasty: An updated complication based analysis using worldwide arthroplasty registers. Arch. Orthop. Trauma. Surg. 2024, 144, 5199–5204. [Google Scholar] [CrossRef] [PubMed]
- Longo, U.G.; Papalia, R.; Salvatore, G.; Tecce, S.M.; Jedrzejczak, A.; Marcozzi, M.; Piergentili, I.; Denaro, V. Epidemiology of revision hip replacement in Italy: A 15-year study. BMC Surg. 2022, 22, 355. [Google Scholar] [CrossRef] [PubMed]
- Boileau, P. Complications and revision of reverse total shoulder arthroplasty. Orthop. Traumatol. Surg. Res. 2016, 102, S33–S43. [Google Scholar] [CrossRef] [PubMed]
- Chae, J.; Siljander, M.; Wiater, J.M. Instability in Reverse Total Shoulder Arthorplasty. J. Am. Acad. Orthop. Surg. 2018, 26, 587–596. [Google Scholar] [CrossRef]
- Markes, A.R.; Cheung, E.; Ma, C.B.; Cheung, E. Failed Reverse Shoulder Arthroplasty and Recommendations for Revision. Curr. Rev. Musculoskelet. Med. 2020, 13, 1–10. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Amorese, V.; Corda, M.; Donadu, M.; Usai, D.; Pisanu, F.; Milia, F.; Marras, F.; Sanna, A.; Delogu, D.; Mazzarello, V.; et al. Total hip prosthesis complication, periprosthetic infection with external fistulizing due to Enterobacter cloacae complex multiple drugs resistance: A clinical case report. Int. J. Surg. Case Rep. 2017, 36, 90–93. [Google Scholar] [CrossRef]
- Luo, H.; Chen, Z.; Pan, Q.; Mei, H.; Chen, W.; Zhu, Z. The application of topical antibiotics for the prevention of infections in primary joint arthroplasty. An umbrella review of systematic reviews and meta-analysis. Int. Wound J. 2024, 21, e14726. [Google Scholar] [CrossRef]
- Romano, V.; Di Gennaro, D.; Sacco, A.M.; Festa, E.; Roscetto, E.; Basso, M.A.; Ascione, T.; Balato, G. Cell Toxicity Study of Antiseptic Solutions Containing Povidone–Iodine and Hydrogen Peroxide. Diagnostics 2022, 12, 2021. [Google Scholar] [CrossRef]
- Savvidou, O.D.; Kaspiris, A.; Trikoupis, I.; Kakouratos, G.; Goumenos, S.; Melissaridou, D.; Papagelopoulos, P.J. Efficacy of antimicrobial coated orthopaedic implants on the prevention of periprosthetic infections: A systematic review and meta-analysis. J. Bone Jt. Infect. 2020, 5, 212–222. [Google Scholar] [CrossRef]
- Reisener, M.; Perka, C. Do Culture-Negative Periprosthetic Joint Infections Have a Worse Outcome Than Culture-Positive Periprosthetic Joint Infections? A Systematic Review and Meta-Analysis. Biomed. Res. Int. 2018, 2018, 6278012. [Google Scholar] [CrossRef]
- Peng, H.M.; Wang, L.C.; Cheng, J.Y.; Zhou, Y.X.; Tian, H.; Lin, J.H.; Guo, W.S.; Lin, Y.; Qu, T.B.; Guo, A.; et al. Rates of periprosthetic infection and surgical revision in Beijing (China) between 2014 and 2016: A retrospective multicenter cross-sectional study. J. Orthop. Surg. Res. 2019, 14, 463. [Google Scholar] [CrossRef]
- Gundtoft, P.H.; Overgaard, S.; Schonheyder, H.C.; Moller, J.K.; Kjærsgaard-Andersen, P.; Pedersen, A.B. The “true” incidence of surgically treated deep prosthetic joint infection after 32,896 primary total hip arthroplasties. Acta Orthop. 2015, 86, 326–334. [Google Scholar] [CrossRef]
- Ren, X.; Ling, L.; Qi, L.; Liu, Z.; Zhang, W.; Yang, Z.; Wang, W.; Tu, C.; Li, Z. Patients’ risk factors for periprosthetic joint infection in primary total hip arthroplasty: A meta-analysis of 40 studies. BMC Musculoskelet. Disord. 2021, 22, 776. [Google Scholar] [CrossRef]
- Parvizi, J.; Zmistowski, B.; Berbari, E.F.; Bauer, T.W.; Springer, B.D.; Della Valle, C.J.; Garvin, K.L.; Mont, M.A.; Wongworawat, M.D.; Zalavras, C.G. New Definition for Periprosthetic Joint Infection: From the Workgroup of the Musculoskeletal Infection Society. Clin. Orthop. Relat. Res. 2011, 469, 2992. [Google Scholar] [CrossRef] [PubMed]
- Osmon, D.R.; Berbari, E.F.; Berendt, A.R.; Lew, D.; Zimmerli, W.; Steckelberg, J.M.; Rao, N.; Hanssen, A.; Wilson, W.R. Diagnosis and management of prosthetic joint infection: Clinical practice guidelines by the infectious diseases Society of America. Clin. Infect. Dis. 2013, 56, e1–e25. [Google Scholar] [CrossRef] [PubMed]
- Parvizi, J.; Tan, T.L.; Goswami, K.; Higuera, C.; Della Valle, C.; Chen, A.F.; Shohat, N. The 2018 Definition of Periprosthetic Hip and Knee Infection: An Evidence-Based and Validated Criteria. J. Arthroplast. 2018, 33, 1309–1314.e2. [Google Scholar] [CrossRef] [PubMed]
- McNally, M.; Sousa, R.; Wouthuyzen-Bakker, M.; Chen, A.F.; Soriano, A.; Vogely, H.C.; Clauss, M.; Higuera, C.A.; Trebše, R. The EBJIS definition of periprosthetic joint infection: A practical guide for clinicians. Bone Jt. J. 2021, 103-B, 18. [Google Scholar] [CrossRef]
- Parvizi, J.; Gehrke, T. Definition of periprosthetic joint infection. J. Arthroplast. 2014, 29, 1331. [Google Scholar] [CrossRef]
- Sax, F.H.; Fink, B. Total Knee Arthroplasty in Unrecognized Septic Arthritis-A Descriptive Case Series Study. Antibiotics 2023, 12, 1153. [Google Scholar] [CrossRef]
- Otto-Lambertz, C.; Yagdiran, A.; Schmidt-Hellerau, K.; Meyer-Schwickerath, C.; Eysel, P.; Jung, N. Establishment of an interdisciplinary board for bone and joint infections. Infection 2021, 49, 1213–1220. [Google Scholar] [CrossRef]
- Abosala, A.; Ali, M. The Use of Calcium Sulphate beads in Periprosthetic Joint Infection, a systematic review. J. Bone Jt. Infect. 2020, 5, 43–49. [Google Scholar] [CrossRef]
- Lei, X.; Xiang, J.; Yang, H.; Bao, H.; Zhu, Z.; Luo, H. Intraosseous regional prophylactic antibiotics decrease the risk of infection in total knee arthroplasty compared with intravenous antibiotics: A systematic review and meta-analysis. EFORT Open Rev. 2023, 8, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Marmor, S.; Bauer, T.; Desplaces, N.; Heym, B.; Roux, A.L.; Sol, O.; Rogé, J.; Mahé, F.; Désiré, L.; Aegerter, P.; et al. Multiplex antibody detection for noninvasive genus-level diagnosis of prosthetic joint infection. J. Clin. Microbiol. 2016, 54, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Popa, M.; Cretu, B.; Iacobescu, G.L.; Iordache, S.; Cursaru, A.; Serban, B.; Cirstoiu, C. Enhancing Outcomes in Prosthetic Joint Infections: The Significance of the Periprosthetic Joint Infection Tumor, Node, and Metastasis (PJI-TNM) Classification and Biodegradable Antibiotic Beads. Cureus 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Bourget-Murray, J.; Azad, M.; Gofton, W.; Abdelbary, H.; Garceau, S.; Grammatopoulos, G. Is the routine use of local antibiotics in the management of periprosthetic joint infections justified? HIP Int. 2023, 33, 4–16. [Google Scholar] [CrossRef]
- Sun, X.; Tan, J.; Zhan, L.; Sheng, M.; Tang, Z.; Wu, L.; Xu, J.; Ma, H. Short-term follow-up of antibiotic-loaded calcium sulfate in treating chronic periprosthetic joint infection during two-stage revision. Front. Bioeng. Biotechnol. 2025, 13, 1352895. [Google Scholar] [CrossRef]
- McPherson, E.J.; Chowdhry, M.; Dipane, M.V.; Marahrens, B.; Dela Pena, D.; Stavrakis, A.I. Antibiotic-Loaded Calcium Sulphate Beads for Treatment of Acute Periprosthetic Joint Infection in Total Knee Arthroplasty: Results Based on Risk Stratification. J. Clin. Med. 2025, 14, 1531. [Google Scholar] [CrossRef]
- Shah, N.A.; Shah, R.V.; Patel, V.D.; Patel, D.V. Clinical Experience of Dissolvable Calcium Sulfate (Stimulan) Carrier for Antibiotic Delivery in Orthopedic Surgery: A Study of 143 Patients. J. Long. Term. Eff. Med. Implants. 2025, 35, 31–44. [Google Scholar] [CrossRef]
- Dimofte, F.; Dimofte, C.; Ungurianu, S.; Serban, C.; Țocu, G.; Cârneciu, N.; Filip, I.; Bezman, L.; Ciuntu, B.M.; Abdulan, I.M.; et al. The Efficacy of Antibiotic-Loaded Calcium Sulfate Beads (Stimulan) in Patients with Hip Arthroplasty Infections. J. Clin. Med. 2024, 13, 4004. [Google Scholar] [CrossRef]
- Iorio, R.; Iannotti, F.; Previ, L.; Viglietta, E.; Gugliotta, Y.; Corsetti, F.; Fenucci, S.; De Carli, A.; Indelli, P.F.; Redler, A. A Modified Technique for Two-Stage Revision in Knee PJI Treatment. J. Clin. Med. 2023, 12, 7323. [Google Scholar] [CrossRef]
- Piovan, G.; Farinelli, L.; Screpis, D.; Marocco, S.; Motta, L.; Palazzolo, G.; Natali, S.; Zorzi, C. The role of antibiotic calcium sulfate beads in acute periprosthetic knee infection: A retrospective cohort study. Arthroplasty 2022, 4, 42. [Google Scholar] [CrossRef]
- Tarity, T.D.; Xiang, W.; Jones, C.W.; Gkiatas, I.; Nocon, A.; Selemon, N.A.; Carli, A.; Sculco, P.K. Do Antibiotic-Loaded Calcium Sulfate Beads Improve Outcomes After Debridement, Antibiotics, and Implant Retention? A Matched Cohort Study. Arthroplast. Today 2022, 14, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Calanna, F.; Chen, F.; Risitano, S.; Vorhies, J.S.; Franceschini, M.; Giori, N.J.; Indelli, P.F. Debridement, antibiotic pearls, and retention of the implant (DAPRI): A modified technique for implant retention in total knee arthroplasty PJI treatment. J. Orthop. Surg. 2019, 27, 2309499019874413. [Google Scholar] [CrossRef] [PubMed]
- De Falco, L.; Troiano, E.; Cesari, M.; Aiuto, P.; Peri, G.; Nuvoli, N.; Fortina, M.; Mondanelli, N.; Giannotti, S. Intra-operative local plus systemic tranexamic acid significantly decreases post-operative bleeding and the need for allogeneic blood transfusion in total knee arthroplasty. Med. Glas. 2021, 18, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Darwich, A.; Dally, F.J.; Bdeir, M.; Kehr, K.; Miethke, T.; Hetjens, S.; Gravius, S.; Assaf, E.; Mohs, E. Delayed rifampin administration in the antibiotic treatment of periprosthetic joint infections significantly reduces the emergence of rifampin resistance. Antibiotics 2021, 10, 1139. [Google Scholar] [CrossRef]
- Paziuk, T.; Cox, R.M.; Gutman, M.J.; Rondon, A.J.; Nicholson, T.; Belden, K.; Namdari, S. Periprosthetic joint infections of the shoulder: A 10-year retrospective analysis outlining the heterogeneity among these patients. Shoulder Elb. 2022, 14, 598–605. [Google Scholar] [CrossRef]
- Cooper, M.E.; Trivedi, N.N.; Sivasundaram, L.; Karns, M.R.; Voos, J.E.; Gillespie, R.J. Diagnosis and management of periprosthetic joint infection after shoulder arthroplasty. JBJS Rev. 2019, 7, e3. [Google Scholar] [CrossRef]
- Santoso, A.; Yoon, T.R.; Park, K.S.; Anwar, I.B.; Utomo, P.; Soetjahjo, B.; Sibarani, T. The results of two-stage revision for methicillin-resistant periprosthetic joint infection (PJI) of the hip. Malays. Orthop. J. 2020, 14, 18–23. [Google Scholar] [CrossRef]
- Warren, S.I.; Charville, G.W.; Manasherob, R.; Amanatullah, D.F. Immune checkpoint upregulation in periprosthetic joint infection. J. Orthop. Res. 2022, 40, 2663–2669. [Google Scholar] [CrossRef]
- Chen, S.; Jiang, Y.; Wang, W.; Chen, J.; Zhu, J. The effect and mechanism of iodophors on the adhesion and virulence of Staphylococcus aureus biofilms attached to artificial joint materials. J. Orthop. Surg. Res. 2023, 18, 756. [Google Scholar] [CrossRef]
- Dusane, D.H.; Brooks, J.R.; Sindeldecker, D.; Peters, C.W.; Li, A.; Farrar, N.R.; Diamond, S.M.; Knecht, C.S.; Plaut, R.D.; Delury, C.; et al. Complete killing of agar lawn biofilms by systematic spacing of antibiotic-loaded calcium sulfate beads. Materials 2019, 12, 4052. [Google Scholar] [CrossRef] [PubMed]
- Koch, J.A.; Pust, T.M.; Cappellini, A.J.; Mandell, J.B.; Ma, D.; Shah, N.B.; Brothers, K.M.; Urish, K.L. Staphylococcus epidermidis biofilms have a high tolerance to antibiotics in periprosthetic joint infection. Life 2020, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Mandell, J.B.; Orr, S.; Koch, J.; Nourie, B.; Ma, D.; Bonar, D.D.; Shah, N.; Urish, K.L. Large variations in clinical antibiotic activity against Staphylococcus aureus biofilms of periprosthetic joint infection isolates. J. Orthop. Res. 2019, 37, 1604–1609. [Google Scholar] [CrossRef] [PubMed]
- Fokkema, A.T.; Kampschreur, L.M.; Pirii, L.E.; Zijlstra, W.P. Rat bite fever in a total knee arthroplasty: An unusual case of periprosthetic joint infection. Arthroplasty 2022, 4, 13. [Google Scholar] [CrossRef]
- Tabliago, N.R.A.; Maish, D.; Martinez, R.M.; Shelly, M.A.; Yablonski, B.; Kwon, T. Acremonium (Sarocladium) periprosthetic joint infection: Case report, literature review, and proposed antifungal regimen. BMC Infect. Dis. 2022, 22, 964. [Google Scholar] [CrossRef]
- Lamret, F.; Colin, M.; Mongaret, C.; Gangloff, S.C.; Reffuveille, F. Antibiotic tolerance of staphylococcus aureus biofilm in periprosthetic joint infections and antibiofilm strategies. Antibiotics 2020, 9, 547. [Google Scholar] [CrossRef]
- Escolà-Vergé, L.; Rodríguez-Pardo, D.; Corona, P.S.; Pigrau, C. Candida periprosthetic joint infection: Is it curable? Antibiotics 2021, 10, 458. [Google Scholar] [CrossRef]
- Jami, S.A.; Shi, J.; Zhou, Z.; Liu, C. The necessity of treating asymptomatic bacteriuria with antibiotics in the perioperative period of joint arthroplasty: A metaanalysis. Turkish J. Med. Sci. 2021, 51, 464–472. [Google Scholar] [CrossRef]
- Brückner, C.; Straube, E.; Petersen, I.; Sachse, S.; Keller, P.; Layher, F.; Matziolis, G.; Spiegl, U.; Zajonz, D.; Edel, M.; et al. Low-grade infections as a possible cause of arthrofibrosis after total knee arthroplasty. Patient Saf. Surg. 2019, 13, 1. [Google Scholar] [CrossRef]
- Bezuidenhout, M.B.; Booysen, E.; Van Staden, A.D.; Uheida, E.H.; Hugo, P.A.; Oosthuizen, G.A.; Dimitrov, D.M.; Dicks, L.M.T. Selective Laser Melting of Integrated Ti6Al4V ELI Permeable Walls for Controlled Drug Delivery of Vancomycin. ACS Biomater. Sci. Eng. 2018, 4, 4412–4424. [Google Scholar] [CrossRef]
- Li, C.; Margaryan, D.; Perka, C.; Trampuz, A. The role of biopsy in diagnosing infection after hip and knee arthroplasty: A meta-analysis. Arch. Orthop. Trauma Surg. 2023, 143, 1779–1792. [Google Scholar] [CrossRef] [PubMed]
- Pangaud, C.; Ollivier, M.; Argenson, J.N. Outcome of single-stage versus two-stage exchange for revision knee arthroplasty for chronic periprosthetic infection. EFORT Open Rev. 2019, 4, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Tone, S.; Naito, Y.; Wakabayashi, H.; Sudo, A. Use of antibiotic-impregnated hydroxyapatite for infection following total knee arthroplasty. Mod. Rheumatol. 2021, 31, 1073–1077. [Google Scholar] [CrossRef] [PubMed]
- Balkhair, A.; Al Maskari, S.; Ibrahim, S.; Al Busaidi, I.; Al Amin, M.; Ba Taher, H. Brucella Periprosthetic Joint Infection Involving Bilateral Knees with Negative Synovial Fluid Alpha-Defensin. Case Rep. Infect. Dis. 2019, 2019, 1–3. [Google Scholar] [CrossRef]
- Karczewski, D.; Winkler, T.; Perka, C.; Müller, M. The Preoperative Microbial Detection is No Prerequisite for the Indication of Septic Revision in Cases of Suspected Periprosthetic Joint Infection. Biomed. Res. Int. 2018, 2018, 1729605. [Google Scholar] [CrossRef]
- Xu, Z.; Cai, Y.; Xinyu, F.; Huang, Z.; Zhang, C.; Li, W.; Zhang, W. Short- and medium-term clinical outcomes of culture-negative and culture-positive periprosthetic joint infection. Res. Sq. 2019, preprint. [Google Scholar] [CrossRef]
- Rajaee, S.S.; Kavolus, J.J.; Hayden, B.L.; Estok, D.M. National Decline in Knee Fusions Performed for Salvage of Chronic Periprosthetic Total Knee Infections. J. Knee Surg. 2022, 35, 971–977. [Google Scholar] [CrossRef]
- Mereddy, P.; Nallamilli, S.R.; Gowda, V.P.; Kasha, S.; Godey, S.K.; Nallamilli, R.R.; Gprk, R.; Meda, V.G.R. The use of Stimulan in bone and joint infections. Bone Jt. Open 2023, 4, 516–522. [Google Scholar] [CrossRef]
- Thahir, A.; Lim, J.A.; West, C.; Krkovic, M. The Use of Calcium Sulphate Beads in the Management of Osteomyelitis of Femur and Tibia: A Systematic Review. Arch. Bone Jt. Surg. 2022, 10, 320. [Google Scholar] [CrossRef]
- de Lachica, J.C.V.; Reyes, S.S.S.; Ureña, J.A.P.; Fragoso, M.A.R. Decrease in acute periprosthetic joint infections incidence with vancomycin-loaded calcium sulfate beads in patients with non-modifiable risk factors. A randomized clinical trial. J. ISAKOS 2022, 7, 201–205. [Google Scholar] [CrossRef]
- Alongi, S.; Troiano, E.; Latino, C.; Colasanti, G.B.; Greco, T.; Perisano, C.; Mosca, M.; Giannotti, S.; Mondanelli, N. Arthroscopic Debridement Enhanced by Intra-Articular Antibiotic-Loaded Calcium Sulphate Beads for Septic Arthritis of a Native Knee Following Iatrogenic Joint Injection: A Case Report. Medicina 2024, 60, 1636. [Google Scholar] [CrossRef] [PubMed]
- Eckers, F.; Laux, C.J.; Schaller, S.; Berli, M.; Achermann, Y.; Fucentese, S.F. Risk factor analysis for above-knee amputation in patients with periprosthetic joint infection of the knee: A case-control study. BMC Musculoskelet. Disord. 2021, 22, 884. [Google Scholar]
- Rosas, S.; Ong, A.C.; Buller, L.T.; Sabeh, K.G.; Law, T.Y.; Roche, M.W.; Hernandez, V.H. Season of the year influences infection rates following total hip arthroplasty. World J. Orthop. 2017, 8, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Wier, J.; Liu, K.C.; Richardson, M.K.; Gettleman, B.S.; Kistler, N.M.; Heckmann, N.D.; Lieberman, J.R. Higher Blood Glucose Levels on the Day of Surgery Are Associated with an Increased Risk of Periprosthetic Joint Infection after Total Hip Arthroplasty. J. Bone Jt. Surg. 2024, 106, 276–287. [Google Scholar] [CrossRef]
- Sreckovic, S.; Ladjevic, N.; Jokanovic, M.; Vracevic, D.; Milovanovic, D.; Simic, M.; Korica, S.; Kadija, M. Rifampicin-induced fever during treatment against staphylococcal biofilm in a patient with periprosthetic joint infection. Br. J. Clin. Pharmacol. 2022, 88, 3887–3890. [Google Scholar] [CrossRef]
- Herman, B.V.; Nyland, M.; Somerville, L.; Macdonald, S.J.; Lanting, B.A.; Howard, J.L. Functional outcomes of infected hip arthroplasty: A comparison of different surgical treatment options. HIP Int. 2017, 27, 245–250. [Google Scholar] [CrossRef]
- Lucenti, L.; Testa, G.; Caldaci, A.; Sammartino, F.; Cicio, C.; Ilardo, M.; Sapienza, M.; Pavone, V. Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature. Healthcare 2024, 12, 666. [Google Scholar] [CrossRef]
- Catellani, F.; Coscione, A.; Usai, L.; Roscitano, C.; Fiorentino, G. Treatment of Proximal Femoral Fragility Fractures in Patients with COVID-19 During the SARS-CoV-2 Outbreak in Northern Italy. J. Bone Joint Surg. Am. 2020, 102, e58. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wouthuyzen-Bakker, M.; Sebillotte, M.; Lomas, J.; Kendrick, B.; Palomares, E.B.; Murillo, O.; Parvizi, J.; Shohat, N.; Reinoso, J.C.; Sánchez, R.E.; et al. Timing of implant-removal in late acute periprosthetic joint infection: A multicenter observational study. J. Infect. 2019, 79, 199–205. [Google Scholar] [CrossRef]
- Chang, Y.; Li, Y.; Fan, T.; Jiang, K.; Lv, J.; Huang, J. Pathogenic bacteria characteristics and drug resistance in acute, delayed, and chronic periprosthetic joint infection: A retrospective analysis of 202 patients. Int. Wound J. 2023, 20, 3315–3323. [Google Scholar] [CrossRef]
- Schmitt, J.W.; Benden, C.; Dora, C.; Werner, C.M.L. Is total hip arthroplasty safely performed in lung transplant patients? Current experience from a retrospective study of the Zurich lung transplant cohort. Patient Saf. Surg. 2016, 10, 17. [Google Scholar] [CrossRef]
- Grondin, J.; Menu, P.; Métayer, B.; Crenn, V.; Dauty, M.; Fouasson-Chailloux, A. Intra-articular injections prior to total knee arthroplasty do not increase the risk of periprosthetic joint infection: A prospective cohort study. Antibiotics 2021, 10, 330. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.