Neuromuscular Control of Overground Walking in Transtibial Amputees: Endoskeletal vs. Exoskeletal Prostheses
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Experimental Procedures
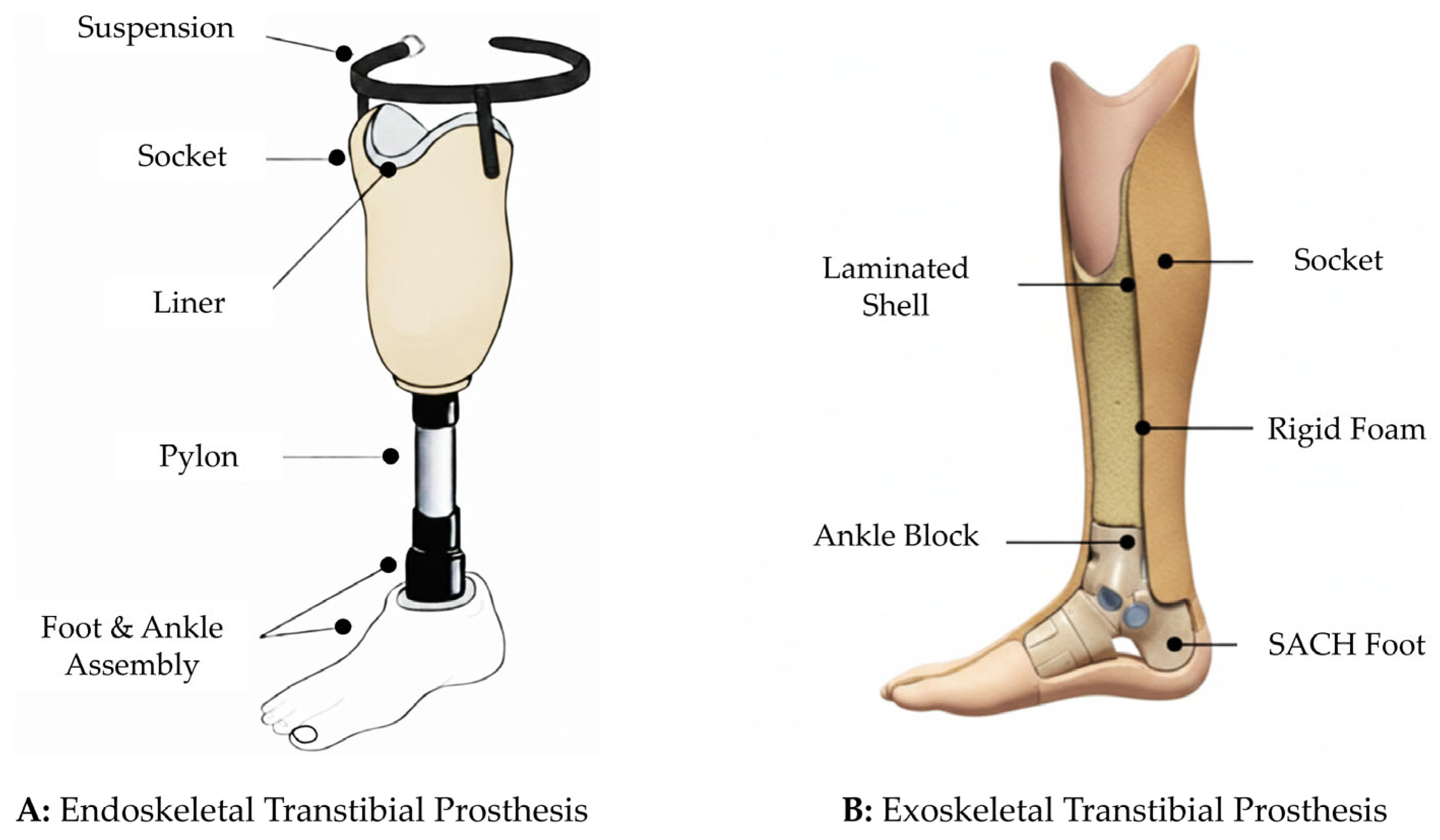
2.2. Prosthetic Characteristics
2.3. Decomposing Movement Synergies
2.4. Statistical Analysis
3. Results
3.1. Walking Movement Synergies
3.2. Structures of Walking Movement Synergies
3.3. Neuromuscular Control of Movement Synergies
3.4. Correlation Between Neuromuscular Control of Movement Synergies and Walking Speed
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lovegreen, W.; Murphy, D.P.; Stevens, P.M.; Seo, Y.I.; Webster, J.B. Lower Limb Amputation and Gait. In Braddom’s Physical Medicine and Rehabilitation; Elsevier: Amsterdam, The Netherlands, 2021; pp. 174–208.e3. [Google Scholar]
- Asif, M.; Tiwana, M.I.; Khan, U.S.; Qureshi, W.S.; Iqbal, J.; Rashid, N.; Naseer, N. Advancements, Trends and Future Prospects of Lower Limb Prosthesis. IEEE Access 2021, 9, 85956–85977. [Google Scholar] [CrossRef]
- Ismawan, A.R.; Prahasto, T.; Ariyanto, M.; Setiyana, B.; Novriansyah, R. A Review of Existing Transtibial Bionic Prosthesis: Mechanical Design, Actuators and Power Transmission. J. Biomed. Sci. Bioeng. 2022, 1, 65–72. [Google Scholar] [CrossRef]
- Green, G.V.; Short, K.; Easley, M. Transtibial Amputation: Prosthetic Use and Functional Outcome. Foot Ankle Clin. 2001, 6, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Plesec, V.; Humar, J.; Dobnik-Dubrovski, P.; Harih, G. Numerical Analysis of a Transtibial Prosthesis Socket Using 3D-Printed Bio-Based PLA. Materials 2023, 16, 1985. [Google Scholar] [CrossRef]
- Gholizadeh, H.; Abu Osman, N.A.; Eshraghi, A.; Ali, S.; Razak, N.A. Transtibial Prosthesis Suspension Systems: Systematic Review of Literature. Clin. Biomech. 2014, 29, 87–97. [Google Scholar] [CrossRef]
- LeMoyne, R. Passive Transtibial Prosthesis and Associated Prosthetic Components. In Advances for Prosthetic Technology; Springer Japan: Tokyo, Japan, 2016; pp. 59–68. [Google Scholar]
- Hafner, B.J.; Halsne, E.G.; Morgan, S.J.; Morgenroth, D.C.; Humbert, A.T. Effects of Prosthetic Feet on Metabolic Energy Expenditure in People with Transtibial Amputation: A Systematic Review and Meta-analysis. PMR 2022, 14, 1099–1115. [Google Scholar] [CrossRef]
- Senthil Selvam, P.; Sandhiya, M.; Chandrasekaran, K.; Hepzibah Rubella, D.; Karthikeyan, S. Prosthetics for Lower Limb Amputation. In Prosthetics and Orthotics; IntechOpen: London, UK, 2021. [Google Scholar]
- Gini, G.; Godlimpi, Z.; Pandelani, T. A Scoping Review of Advances in Active Below-Knee Prosthetics: Integrating Biomechanical Design, Energy Efficiency, and Neuromuscular Adaptation. Prosthesis 2025, 7, 165. [Google Scholar] [CrossRef]
- Alluhydan, K.; Siddiqui, M.I.H.; Elkanani, H. Functionality and Comfort Design of Lower-Limb Prosthetics: A Review. J. Disabil. Res. 2023, 2, 10–23. [Google Scholar] [CrossRef]
- van der Linde, H.; Hofstad, C.J.; Geurts, A.C.H.; Postema, K.; Geertzen, J.H.B.; van Limbeek, J. A Systematic Literature Review of the Effect of Different Prosthetic Components on Human Functioning with a Lower-Limb Prosthesis. J. Rehabil. Res. Dev. 2004, 41, 555. [Google Scholar] [CrossRef]
- Malchow, C.; Fiedler, G. Effect of Observation on Lower Limb Prosthesis Gait Biomechanics. Prosthet. Orthot. Int. 2016, 40, 739–743. [Google Scholar] [CrossRef]
- Rusaw, D.; Ramstrand, N. Motion-Analysis Studies of Transtibial Prosthesis Users. Prosthet. Orthot. Int. 2011, 35, 8–19. [Google Scholar] [CrossRef]
- Cherni, Y.; Laurendeau, S.; Robert, M.; Turcot, K. The Influence of Transtibial Prosthesis Type on Lower-Body Gait Adaptation: A Case Study. Int. J. Environ. Res. Public Health 2023, 20, 439. [Google Scholar] [CrossRef]
- Latash, M.L.; Scholz, J.P.; Schöner, G. Toward a New Theory of Motor Synergies. Motor Control 2007, 11, 276–308. [Google Scholar] [CrossRef] [PubMed]
- Herdiman, L.; Adiputra, I.N.; Tirtayasa, K.; Manuaba, I.B.A. Improvement in Walking Efficiency of Transtibial Amputee Using Prosthetic Leg with Multi-Axis Joint and Energy Store Return Ankle. In Proceedings of the Joint International Conference on Electric Vehicular Technology and Industrial, Mechanical, Electrical and Chemical Engineering (ICEVT & IMECE), Surakarta, Indonesia, 4–5 November 2015; IEEE: New York, NY, USA, 2016; pp. 148–152. [Google Scholar]
- De Marchis, C.; Ranaldi, S.; Varrecchia, T.; Serrao, M.; Castiglia, S.F.; Tatarelli, A.; Ranavolo, A.; Draicchio, F.; Lacquaniti, F.; Conforto, S. Characterizing the Gait of People With Different Types of Amputation and Prosthetic Components Through Multimodal Measurements: A Methodological Perspective. Front. Rehabil. Sci. 2022, 3, 804746. [Google Scholar] [CrossRef] [PubMed]
- de Cerqueira Soares, A.S.O.; Yamaguti, E.Y.; Mochizuki, L.; Amadio, A.C.; Serrão, J.C. Biomechanical Parameters of Gait among Transtibial Amputees: A Review. Sao Paulo Med. J. 2009, 127, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Latash, M.L. Human Movements: Synergies, Stability, and Agility. In Springer Tracts in Advanced Robotics; Springer: Berlin/Heidelberg, Germany, 2019; Volume 124, pp. 135–154. [Google Scholar] [CrossRef]
- Latash, M.L.; Gorniak, S.; Zatsiorsky, V.M. Hierarchies of Synergies in Human Movements. Kinesiology 2008, 40, 29. [Google Scholar]
- Promsri, A.; Federolf, P. Effects of On- and Off-Medication Periods on Walking Performance in Parkinson’s Disease: Insights from Movement Synergies. Digit. Biomark. 2025, 9, 104–112. [Google Scholar] [CrossRef]
- Federolf, P.; Tecante, K.; Nigg, B. A Holistic Approach to Study the Temporal Variability in Gait. J. Biomech. 2012, 45, 1127–1132. [Google Scholar] [CrossRef]
- Promsri, A.; Werner, I.; Federolf, P. Gait Recovery after Total Hip Arthroplasty: Insights from Principal Component Analysis of Pre- and Post-Surgical Walking Performance. J. Biomech. 2026, 197, 113173. [Google Scholar] [CrossRef]
- Collins, D.M.; Karmarkar, A.; Relich, R.; Pasquina, P.F.; Cooper, R.A. Review of Research on Prosthetic Devices for Lower Extremity Amputation. Crit. Rev. Biomed. Eng. 2006, 34, 379–438. [Google Scholar] [CrossRef]
- Samala, M.; Rattanakoch, J.; Guerra, G.; Tharawadeepimuk, K.; Nanbancha, A.; Niamsang, W.; Kerdsomnuek, P.; Suwanmana, S.; Limroongreungrat, W. A Dataset of Optical Camera and IMU Sensor Derived Kinematics of Thirty Transtibial Prosthesis Wearers. Sci. Data 2024, 11, 922. [Google Scholar] [CrossRef]
- Barre, A.; Armand, S. Biomechanical ToolKit: Open-Source Framework to Visualize and Process Biomechanical Data. Comput. Methods Programs Biomed. 2014, 114, 80–87. [Google Scholar] [CrossRef]
- Haid, T.H.; Zago, M.; Promsri, A.; Doix, A.-C.M.; Federolf, P.A. PManalyzer: A Software Facilitating the Study of Sensorimotor Control of Whole-Body Movements. Front. Neuroinform. 2019, 13, 24. [Google Scholar] [CrossRef]
- Promsri, A.; Mohr, M.; Federolf, P. Principal Postural Acceleration and Myoelectric Activity: Interrelationship and Relevance for Characterizing Neuromuscular Function in Postural Control. Hum. Mov. Sci. 2021, 77, 102792. [Google Scholar] [CrossRef]
- Ó’Reilly, D.; Federolf, P. Identifying Differences in Gait Adaptability across Various Speeds Using Movement Synergy Analysis. PLoS ONE 2021, 16, e0244582. [Google Scholar] [CrossRef]
- Latash, M.L. Synergic Control of Movement: From Single Muscles to the Whole Body. In Progress in Motor Control: From Neuroscience to Patient Outcomes; Academic Press: Cambridge, MA, USA, 2024; pp. 25–47. [Google Scholar] [CrossRef]
- Bernstein, N.A. The Co-Ordination and Regulation of Movements: Conclusions towards the Study of Motor Co-Ordination. In Biodynamics of Locomotion; Pergamon Press: Oxford, UK, 1967; pp. 104–113. [Google Scholar]
- Winter, D.A. Biomechanics and Motor Control of Human Movement, 4th ed.; Wiley: Hoboken, NJ, USA, 2009; ISBN 9780470398180. [Google Scholar]
- Promsri, A. Assessing Walking Stability Based on Whole-Body Movement Derived from a Depth-Sensing Camera. Sensors 2022, 22, 7542. [Google Scholar] [CrossRef] [PubMed]
- Holm, S. A Simple Sequentially Rejective Multiple Test Procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- Wu, A.R.; Kuo, A.D. Determinants of Preferred Ground Clearance during Swing Phase of Human Walking. J. Exp. Biol. 2016, 219, 3106–3113. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.-J.; Wang, M.-J.J. The Change of Gait Parameters during Walking at Different Percentage of Preferred Walking Speed for Healthy Adults Aged 20–60 Years. Gait Posture 2010, 31, 131–135. [Google Scholar] [CrossRef]
- Promsri, A. Changes in Walking Stability at Different Percentages of Preferred Walking Speed in Healthy Young and Older Adults: Insights From Movement Component Analysis. Sci. World J. 2025, 2025, 971520. [Google Scholar] [CrossRef]
- Wong, C.K.; Ehrlich, J.E.; Ersing, J.C.; Maroldi, N.J.; Stevenson, C.E.; Varca, M.J. Exercise Programs to Improve Gait Performance in People with Lower Limb Amputation. Prosthet. Orthot. Int. 2016, 40, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Sinitski, E.H.; Lemaire, E.D.; Baddour, N.; Besemann, M.; Dudek, N.; Hebert, J.S. Maintaining Stable Transtibial Amputee Gait on Level and Simulated Uneven Conditions in a Virtual Environment. Disabil. Rehabil. Assist. Technol. 2021, 16, 40–48. [Google Scholar] [CrossRef]
- Garnier, Y.M.; Lepers, R.; Dubau, Q.; Pageaux, B.; Paizis, C. Neuromuscular and Perceptual Responses to Moderate-Intensity Incline, Level and Decline Treadmill Exercise. Eur. J. Appl. Physiol. 2018, 118, 2039–2053. [Google Scholar] [CrossRef]
- Harrison, K.; Holmes, H.H.; Finley, E.B.; Guzman, K.S.; Kimbrough, K.C.; Roper, J.A. Incline and Decline Running Alters Joint Moment Contributions but Not Peak Support Moments in Individuals with an Anterior Cruciate Ligament Reconstruction and Controls. Front. Sports Act. Living 2023, 5, 1217783. [Google Scholar] [CrossRef]
- Federolf, P.; Roos, L.; Nigg, B.M. Analysis of the Multi-Segmental Postural Movement Strategies Utilized in Bipedal, Tandem and One-Leg Stance as Quantified by a Principal Component Decomposition of Marker Coordinates. J. Biomech. 2013, 46, 2626–2633. [Google Scholar] [CrossRef]
- Gholizadeh, H.; Lemaire, E.D.; Sinitski, E.H.; Nielen, D.; Lebel, P. Transtibial Amputee Gait with the Unity Suspension System. Disabil. Rehabil. Assist. Technol. 2020, 15, 350–356. [Google Scholar] [CrossRef]
- Comon, P. Independent Component Analysis, A New Concept? Signal Process. 1994, 36, 287–314. [Google Scholar] [CrossRef]
- Hyvärinen, A.; Oja, E. Independent Component Analysis: Algorithms and Applications. Neural Netw. 2000, 13, 411–430. [Google Scholar] [CrossRef] [PubMed]




| Variable | Endoskeletal (n = 10) | Exoskeletal (n = 10) | p-Value |
|---|---|---|---|
| Age (yrs) | 54.7 ± 6.1 | 57.9 ± 8.7 | 0.425 |
| Body mass (kg) | 70.4 ± 13.9 | 64.8 ± 12.3 | 0.466 |
| Height (m) | 1.65 ± 0.1 | 1.65 ± 0.1 | 0.825 |
| Body mass index (kg/m2) | 25.7 ± 3.8 | 23.7 ± 3.1 | 0.339 |
| Amputated side (Left/Right) | 5/5 | 3/7 | - |
| Preferred walking speed (m/s) | 1.7 ± 0.2 | 1.4 ± 0.2 | 0.020 * |
| Prosthetic characteristics | |||
| 7/1/2/0 | 2/2/3/3 | - |
| 10/0 | 7/2/1 | - |
| 4/6/0 | 5/4/1 | - |
| 10/0/0 | 8/1/1 | - |
| Variable | A: RMS | B: N | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PA1 | PA2 | PA3 | PA4 | PA5 | PA1 | PA2 | PA3 | PA4 | PA5 | ||
| Walking speed | ρ | −0.033 | 0.639 | −0.626 | 0.310 | −0.208 | −0.940 | −0.955 | −0.952 | −0.944 | −0.890 |
| p | 0.890 | 0.002 * | 0.003 * | 0.184 | 0.380 | <0.001 ** | <0.001 ** | <0.001 ** | <0.001 ** | <0.001 ** | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Promsri, A. Neuromuscular Control of Overground Walking in Transtibial Amputees: Endoskeletal vs. Exoskeletal Prostheses. Prosthesis 2026, 8, 21. https://doi.org/10.3390/prosthesis8020021
Promsri A. Neuromuscular Control of Overground Walking in Transtibial Amputees: Endoskeletal vs. Exoskeletal Prostheses. Prosthesis. 2026; 8(2):21. https://doi.org/10.3390/prosthesis8020021
Chicago/Turabian StylePromsri, Arunee. 2026. "Neuromuscular Control of Overground Walking in Transtibial Amputees: Endoskeletal vs. Exoskeletal Prostheses" Prosthesis 8, no. 2: 21. https://doi.org/10.3390/prosthesis8020021
APA StylePromsri, A. (2026). Neuromuscular Control of Overground Walking in Transtibial Amputees: Endoskeletal vs. Exoskeletal Prostheses. Prosthesis, 8(2), 21. https://doi.org/10.3390/prosthesis8020021





