Abstract
Wildfire is a dominant ecological force in Mediterranean pine forests, and post-fire silvicultural practices can substantially alter their recovery trajectories. In this study, we examined how natural regeneration and artificial plantations influence the composition, structure, and functional roles of medium and large-sized mammal communities in burned Pinus brutia forests of southwestern Türkiye. Camera trap data were combined with linear mixed-effects models, functional diversity metrics, and indicator species analysis to assess community responses. Mammalian assemblages showed marked shifts across treatments: generalist carnivores such as Vulpes vulpes and Canis aureus dominated burned areas, whereas higher-trophic specialists like Caracal caracal were restricted to unburned forests. Functional richness was consistently higher in unburned stands, while artificial plantations reduced both richness and evenness. Natural regeneration partly mitigated these declines by sustaining more balanced community structures. Indicator species analysis confirmed these patterns, with Lepus europaeus strongly associated with burned sites and C. caracal with unburned forests. Overall, findings demonstrate that post-fire silvicultural practices strongly shape mammalian community assembly and functional diversity. Natural regeneration preserves structural heterogeneity and supports functionally diverse assemblages, whereas artificial plantations promote homogenization. Effective restoration strategies should therefore integrate wildlife responses with vegetation recovery to strengthen ecosystem resilience and maintain the ecological roles of mammals.
1. Introduction
Fire is a key ecological driver in the Mediterranean Basin [1,2,3,4], and over millions of years of evolution it has become a fundamental force shaping the structure and functioning of Mediterranean ecosystems. This process is particularly evident in the eastern Mediterranean sub-basin, where forests dominated by Pinus brutia (Turkish red pine) occur alongside maquis formations.
In recent years, however, wildfires in southwestern Türkiye, especially along the coastal belt, have deviated from the natural regime and become anthropogenic environmental crises. Türkiye is among the rare regions of the world that host three of the 36 recognized global biodiversity “hotspots” [5,6,7]. Within the Mediterranean phytogeographic region, the southwestern Anatolian Muğla area is particularly known as a fire-prone landscape, where human-induced forest fires in recent years have further threatened the fragility of ecosystems. Over the last five years, intentional ignitions, waste burning, negligence from power transmission lines, and the increasing influence of climate change have led to the occurrence of megafires—large, high-intensity wildfires. These fires not only cause extensive physical damage but also disrupt community composition, ecosystem processes, and the natural regeneration capacity of forests. In the Muğla region, vast areas have been affected by wildfires since 2021. In 2021, the Marmaris İçmeler fire affected 12,289 ha, the Köyceğiz fire 10,890 ha, and the Milas fire 17,767 ha [8]. In 2022, the Marmaris Bördübet fire classified as a megafire affected 4392 ha [9]. Following these events, the Turkish General Directorate of Forestry has implemented different post-fire silvicultural practices [9,10]. These practices include artificial regeneration (afforestation/active restoration), natural regeneration (line sowing, seed dispersal, spreading cone-bearing branches/passive restoration), and unmanaged areas left for natural recovery [11,12,13].
The post-fire renewal of ecosystems is shaped not only by the silvicultural methods applied but also by the ecological adaptations that species have developed in response to fire. For example, plant species in the Mediterranean Basin are known to have evolved a variety of adaptive traits that enable them to cope with fire [1,14]. In such areas, vegetation recovery after fire occurs relatively rapidly [15,16]. However, a comprehensive evaluation of post-fire ecosystems depends not only on the recovery of plant communities but also on the ecological responses of faunal communities. Among the biotic indicators used for ecosystem monitoring, mammals—often described as climax species within faunal assemblages—are considered effective biological indicators of environmental change due to their high sensitivity to habitat conditions and their critical roles across trophic levels [17,18,19]. Large mammals, especially carnivores, occupy the upper levels of trophic hierarchies and can be defined as both biological indicator species and flagship species [20]. These species not only provide essential information for monitoring ecosystem health but also have the potential to attract public attention and generate societal support for conservation efforts [19,21,22]. Studies on post-fire changes in large mammal communities have remained relatively scarce in Mediterranean-type ecosystems, particularly compared with studies on other groups of organisms [23]. In recent years, however, interest in large mammals has increased in post-fire habitats, particularly in the Mediterranean-climate regions of California [24,25,26,27]. Similarly, research on large mammals has intensified in other fire-prone ecosystems worldwide [28,29,30,31]. In contrast, studies addressing large mammals in the Mediterranean Basin remain limited, with only a few recent contributions focusing on fire-shaped habitats [32,33,34,35]. Nevertheless, although some studies have examined the effects of different post-fire silvicultural techniques on succession through mammal species, these have generally been limited to small mammals and conducted in other forest ecosystems. However, no comparable study has focused on large mammals within Mediterranean ecosystems, particularly in Turkish red pine forests [36,37].
In this context, the present study aims to assess how different post-fire silvicultural practices affect community dynamics in Turkish red pine ecosystems of the warm Mediterranean vegetation zone. Furthermore, we discuss management approaches that promote ecological integrity, thereby enhancing ecosystem resilience and resistance under the climate crisis. Ultimately, the goal is to identify silvicultural approaches in post-fire restoration that support wildlife management.
2. Materials and Methods
2.1. Study Area
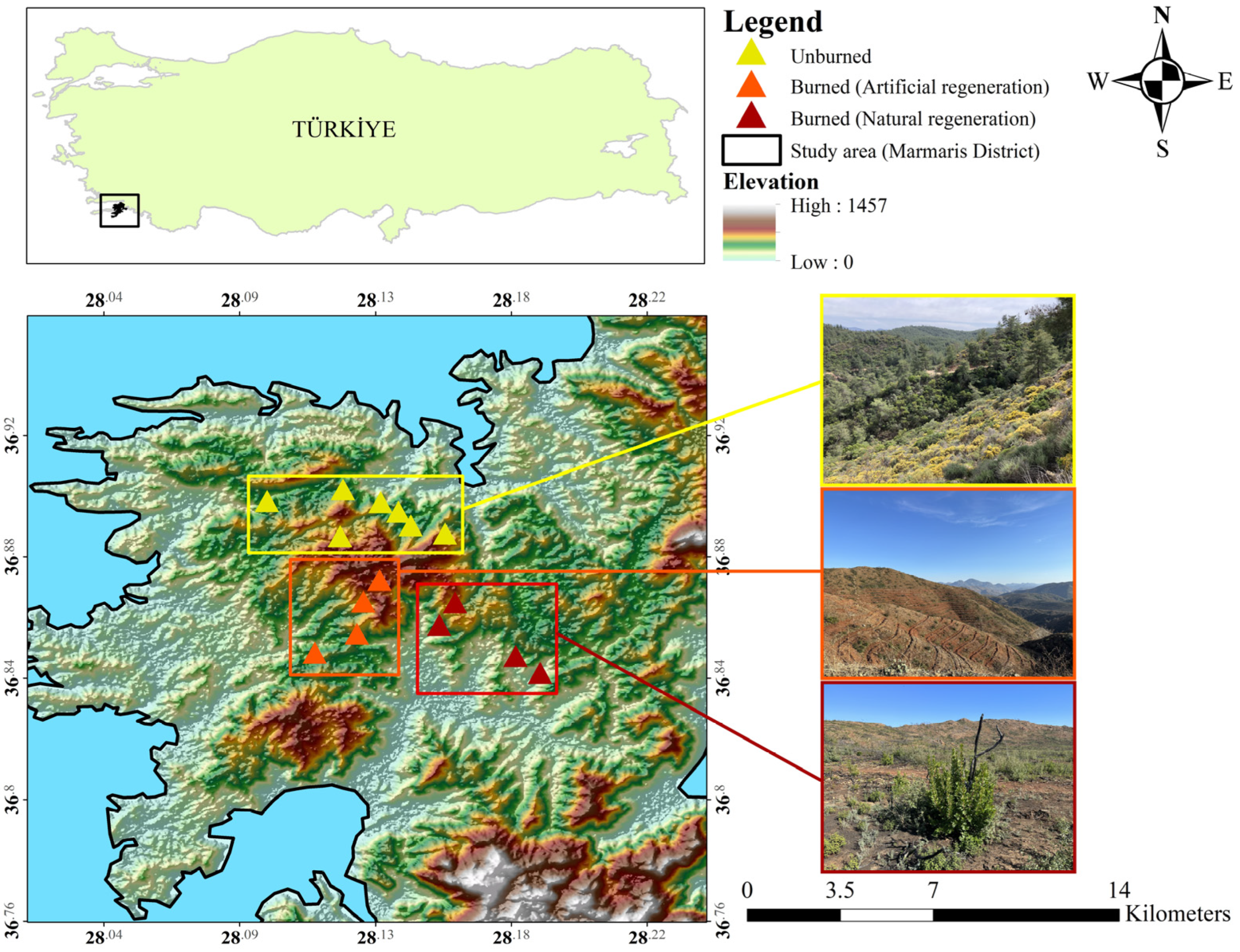
This research was conducted in the Bördübet area of Marmaris District, located in Muğla Province in southwestern Türkiye (Figure 1). The study area is notable for being one of the regions with the highest wildfire frequency in Türkiye over the past 50 years.
Figure 1.
The location of the study area and study sites.
The dominant vegetation of the study area consists of Turkish red pine forests with an understory rich in maquis and phrygana shrub species [16]. However, following the large wildfire that occurred in June 2022 in the Marmaris-Bördübet area, different types of silvicultural interventions were implemented. To the immediate north of the burned area lies an unburned control site, which represents a habitat that has regenerated from past wildfires and itself displays heterogeneous characteristics. This mosaic landscape, which includes both post-fire silvicultural treatments and a long-unburned control site, provides a suitable setting to assess the effects of fire and management practices on mammalian communities. According to previous records, the study area and its surroundings harbor numerous terrestrial mammal species, including conservation-priority taxa such as the caracal (Caracal caracal), grey wolf (Canis lupus), and brown bear (Ursus arctos) [38,39]. The study area experiences a typical Mediterranean climate, with summers that are hot and dry and winters that are mild and wet. Annual precipitation averages 1211 mm, while the mean annual temperature is 18.7 °C. These climatic conditions confer relatively high ecosystem productivity compared with similar areas in the Mediterranean Basin and facilitate rapid post-fire vegetation recovery [16,34]. Accordingly, the presence of climax species such as the brown bear can be observed in the region [39].
Three main vegetation types can be identified in the study area. The first is Turkish red pine (Pinus brutia) forests at different stages of post-fire succession. These forests are intermingled with tall shrub formations (maquis), largely characterized by evergreen species in shrub form. Dominant species in these areas include Arbutus andrachne and Arbutus unedo, while other typical maquis elements comprise Quercus spp., Erica spp., and Phillyrea latifolia. In addition, xerophytic shrublands (phrygana) covered by low shrubs are also present. Characteristic species of this formation include Thymus spp., Cistus spp., Genista acanthoclada, and Lavandula stoechas [35,38].
2.2. Site Selection and Sampling
A comprehensive survey was conducted to compare the structure of mammal communities in burned areas subjected to different silvicultural treatments and in unburned sites. The wildfire that affected the region occurred in June 2022. In the aftermath of the fire, the Turkish General Directorate of Forestry implemented a series of post-fire silvicultural operations across approximately 4000 ha, including salvage logging, site preparation, planting, and afforestation. These interventions were completed by December 2023. To avoid potential disturbance-related bias, field sampling was initiated only after all operations had ceased, meaning that the monitoring period reflects post-treatment habitat conditions rather than the transition phase during active management.
Sampling stations were established in each habitat type, and field surveys were carried out. The sampling design was structured to enable comparisons of the effects of silvicultural interventions and habitat conditions on mammal species. A total of eight sampling stations were established in the burned areas and were further classified according to the post-fire regeneration strategy: four in naturally regenerated stands and four in artificially regenerated stands. In addition, seven stations were placed in unburned habitats, resulting in a total of fifteen sampling stations overall (Figure 1). Camera traps equipped with motion sensors were deployed at all stations using a passive monitoring approach, with a spacing of 1–2 km to ensure spatial independence. No baiting was applied during the sampling period, thereby allowing natural movement patterns to be recorded. In total, 3195 camera-trap days of sampling effort were accumulated across fifteen stations.
2.3. Functional Trait Selection and Functional Diversity Indices
In this study, a set of biological and ecological traits was selected to assess the functional roles of mammal species within ecosystems. These included key life-history attributes such as body size (BS), activity period (AP), dispersal ability (DA), and habitat preference (HP), as well as reproductive and tolerance-related traits, including the average number of offspring per year (AO) and anthropogenic tolerance (AT). In addition, trophic level (TL) and a series of functional role descriptors were incorporated to characterize species’ positions and ecological contributions within ecosystems, namely seed disperser (SD), soil digger (DG), gallery-type nesting in the soil (GA), top predator (TP), and important prey (IP). These categorical traits and their coding scheme were compiled from the published literature to represent species’ ecological functions as comprehensively as possible. For functional diversity analyses (FD), a trait matrix containing these attributes and a community matrix containing abundance data were first constructed. Categorical traits entered as text (e.g., carnivore, herbivore) were converted to numeric form to enable calculation of functional diversity metrics.
To evaluate the effects of post-fire silvicultural practices on the functional structure of mammal communities, four functional diversity metrics were calculated. The first, Functional Richness (FRic), represents the total functional space occupied by the species in a community, based on their traits [40]. It is an indicator of species’ functional diversity and ecological niche differentiation and is particularly critical for maintaining habitat complexity and ecosystem functioning. Functional Evenness (FEve) measures how evenly species are distributed within functional space [40,41]. High FEve values indicate that functional traits are represented more homogeneously within the community, while low values suggest the dominance of certain traits. Functional Divergence (FDiv) reflects the degree to which species deviate from the mean position in functional space [41]. High FDiv values indicate that species with distinct functional strategies are dominant, signaling greater niche differentiation. Rao’s Quadratic Entropy (RaoQ) [42] provides a general measure of diversity at the community level by simultaneously accounting for both species abundances and functional distances among species [43]. This metric integrates trait similarity with abundance patterns, offering a more holistic view of functional differences among communities. Finally, Community Weighted Mean (CWM) values were calculated to interpret functional traits at the community level [44]. CWM represents the abundance-weighted mean of each trait across species, thereby indicating the extent to which the traits of dominant species shape the overall functional composition of the community. Each of these metrics was applied to evaluate how post-fire silvicultural processes are reflected in community structure through species’ functional traits. In this way, potential functional differences between burned and unburned areas were analyzed from a comprehensive perspective.
2.4. Statistical Analysis
In the statistical analysis, we first examined differences between burned and unburned areas in terms of functional trait metrics and community weighted means (CWM). Subsequently, to assess both the differences between burned and unburned sites and the effects of seasonal variation, linear mixed-effects models (LMMs) were employed. For this purpose, abundance data were collected across four seasons—winter, spring, summer, and autumn—and seasonal replicates were generated for each station. In the models, Treatment (burned/unburned) and Season (winter, spring, summer, autumn) were included as fixed effects, while Station was included as a random effect to account for repeated measures. The model structure can be summarized as follows:
Linear mixed-effects models were used to test both the overall differences between burned and unburned areas, the variation among seasons, and the interaction between these two factors. Estimated marginal means (EMMs) were calculated using the emmeans package, and Tukey-adjusted multiple comparisons were performed to assess burned–unburned differences within each season and seasonal differences within each treatment. Results were presented graphically as EMM ± SE based on the season × treatment interaction [45].
Finally, an Indicator Species Analysis was conducted to identify species that were functionally decisive between burned sites (with natural and artificial regeneration) and unburned areas. Through this method, the specificity and fidelity of each species to a certain group (e.g., burned areas) were statistically tested, and it was aimed to reveal potential indicator species [46,47,48]. All statistical analyses were performed in R software (version 4.3.0). Functional diversity metrics were calculated using the FD package [40,49]. Linear mixed models were fitted with lme4 [50] and emmeans [51], and visualizations were generated using ggplot2 [52,53]. Indicator species analyses were carried out using the multipatt function in the indicspecies package [54].
3. Results
A total of eight mammal species were recorded in this study (Table 1). Among them were species of conservation priority for Türkiye and Mediterranean ecosystems, including the caracal (Caracal caracal) and the brown bear (Ursus arctos) (Figure 2).

Table 1.
Recorded mammal species and their conservation status according to the IUCN (2025–1). The ecological traits used in the functional analysis are listed for each species (see main text for trait definitions). X represent the presence of a specific categorical (binary) functional trait for a given species.


Figure 2.
Some record samples of mammal species from study area: (a) Brown bear; (b) Caracal; (c) Eurasian badger; (d) Wild boar.
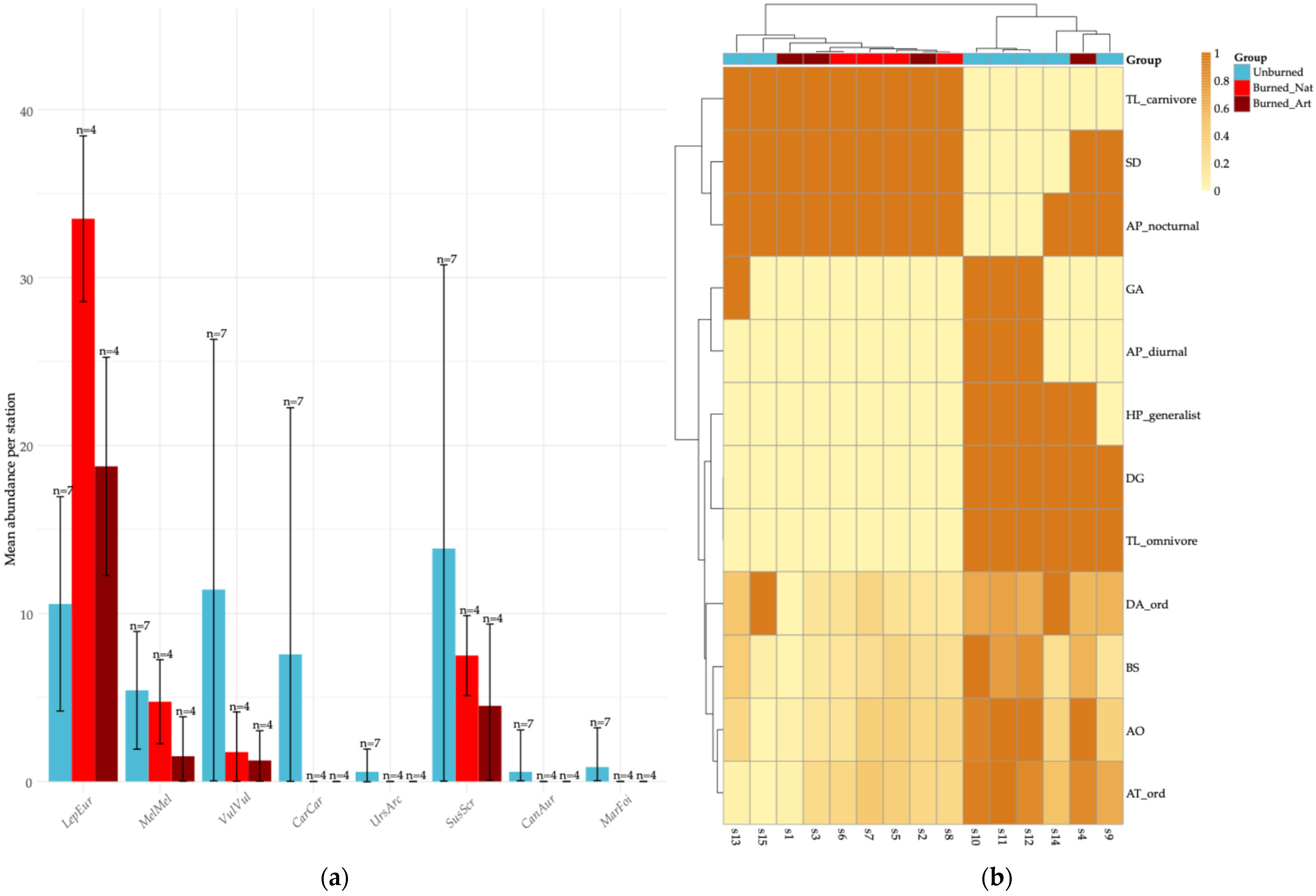
Results of the functional diversity metrics and community-weighted means (CWM) revealed clear treatment-level contrasts while also indicating substantial variability among stations (Figure 3). These patterns are based on station-level values pooled across seasons, as our objective was to capture overall treatment contrasts rather than seasonal fluctuations. Burned sites, particularly those under natural regeneration, tended to exhibit higher CWM scores for traits associated with carnivory, nocturnal activity, and greater dispersal capacity. However, these tendencies were not consistent across all stations, reflecting fine-scale structural heterogeneity within burned stands. In contrast, several unburned stations showed higher contributions of traits associated with larger body size, higher reproductive output, and tolerance of moist, structurally complex habitats. The relatively wide SD ranges in species abundances and CWM values suggest substantial among-station heterogeneity within treatments, consistent with a mosaic-like functional structure rather than a single, homogeneous community pattern.
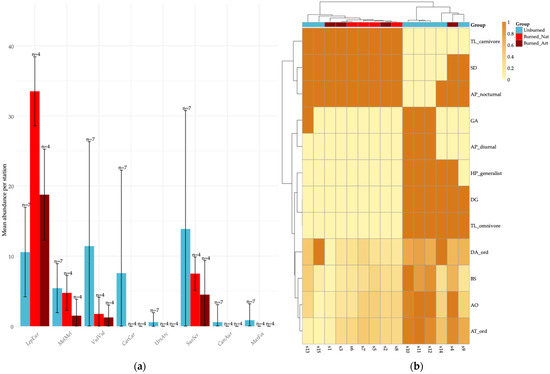
Figure 3.
Mammalian community responses to fire in terms of species abundance and functional trait composition: (a) Mean abundance of mammal species across stations, with error bars representing standard deviation (SD) and labels indicating sample size (n) per treatment. Burned sites are presented as two distinct post-fire management categories—naturally regenerated (Burned_Nat) and artificially regenerated (Burned_Art)—while Unburned sites represent undisturbed reference habitats. Consistent with these contrasts, Lepus europaeus shows higher abundance in burned areas, whereas Caracal caracal occurs more frequently in unburned sites; (b) Heatmap of community-weighted means (CWM) of functional traits (scaled 0–1) at the station level, with hierarchical clustering illustrating among-station differences in functional composition. The colored annotation bar indicates treatment identity and clarifies the grouping of stations, whereas the internal color scale reflects CWM values only and does not correspond to treatment colors.
Differences in overall species composition and functional trait distributions between unburned and burned areas reveal a fire-driven restructuring of community organization. However, the functional structure of mammal communities is shaped not only by fire itself but also by the silvicultural treatments applied in burned sites and by seasonal variation throughout the year. Accordingly, functional diversity metrics (FRic, FEve, FDiv, RaoQ) were analyzed using mixed models, allowing the assessment of community-level differences in relation to burning and silvicultural treatment groups as well as seasonal patterns.
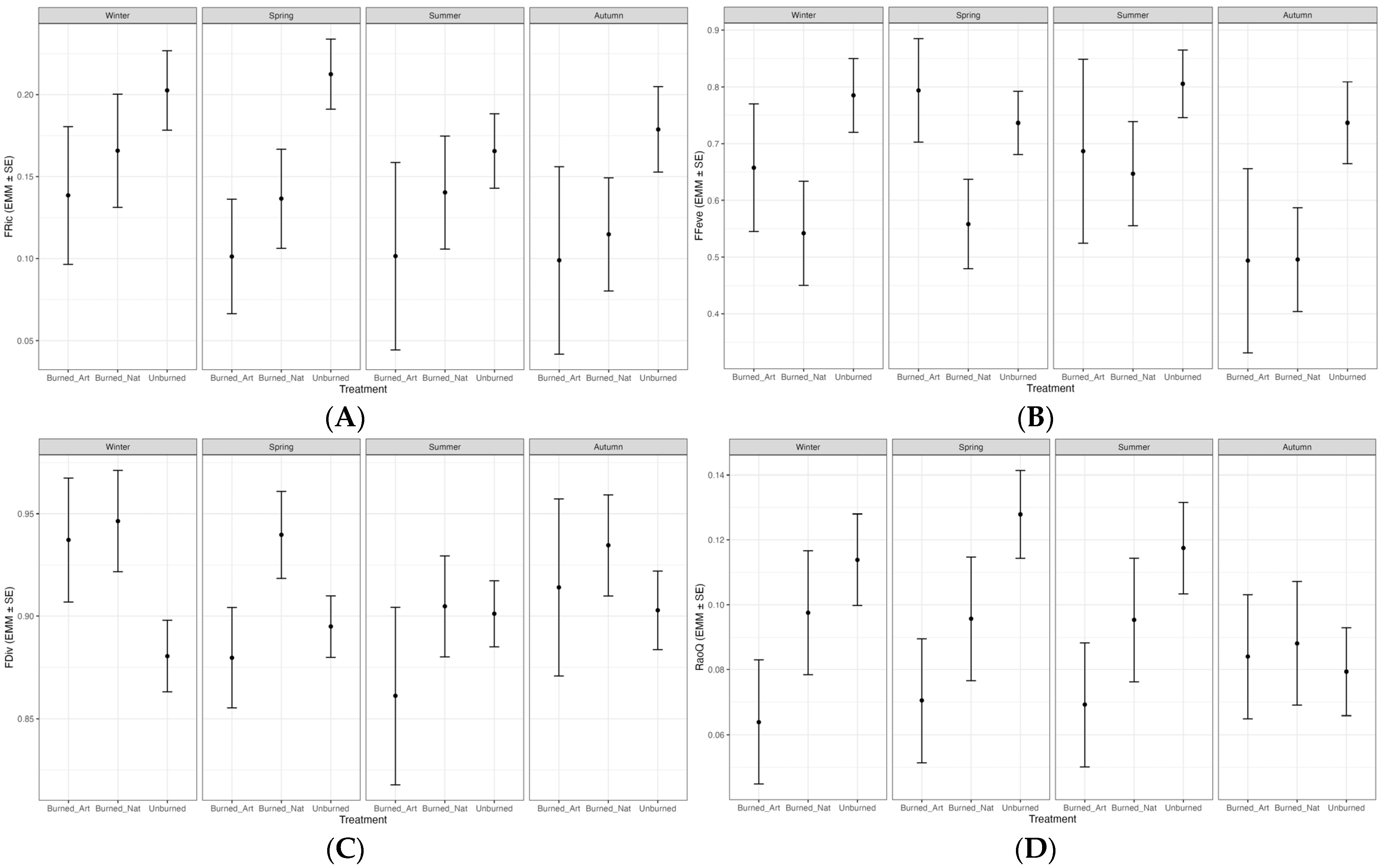
Mixed model analyses showed that functional diversity metrics were influenced by burning, silvicultural treatments, and seasonal variation (Table 2). In particular, Treatment had a significant effect on Functional Richness (FRic) (p = 0.046). FRic values were generally higher in unburned areas, while within burned areas, sites under natural regeneration had higher values than those under artificial regeneration. This difference became especially pronounced in spring and summer (Figure 4A). Detailed station × season results of functional diversity metrics are presented in Appendix A Table A1.

Table 2.
Type-III ANOVA results from linear mixed-effects models testing the effects of fire history, post-fire silvicultural treatment, and season on functional diversity metrics (FRic, FEve, FDiv, RaoQ).
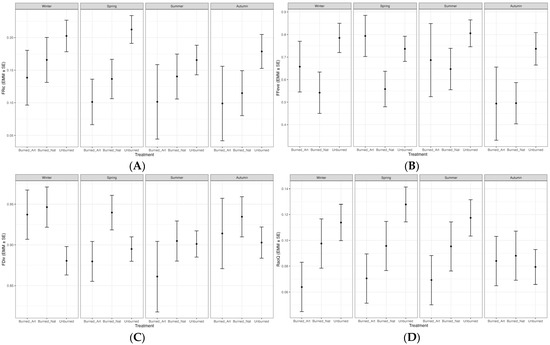
Figure 4.
Estimated marginal means (±SE) of functional diversity metrics across treatments and seasons derived from linear mixed-effects models: (A) functional richness (FRic); (B) functional evenness (FEve); (C) functional divergence (FDiv); (D) Rao’s quadratic entropy (RaoQ). Treatments include artificially regenerated burned sites (Burned_Art), naturally regenerated burned sites (Burned_Nat), and unburned reference sites (Unburned).
Regarding Functional Evenness (FEve), Treatment had a strongly significant effect (p = 0.002), whereas neither Season (p = 0.413) nor the interaction (p = 0.803) was significant. FEve values were higher in unburned areas but dropped markedly in burned sites, particularly under artificial regeneration (Figure 4B).
For Functional Divergence (FDiv), no significant effects were detected for Treatment (p = 0.150), Season (p = 0.448), or their interaction (p = 0.440). However, mean values indicated higher FDiv in burned areas, especially those subjected to artificial regeneration (Figure 4C).
In the case of Rao’s Quadratic Entropy (RaoQ), none of the factors were statistically significant. Nevertheless, values tended to be higher in unburned areas, particularly during summer (Figure 4D).
The effect of Season was significant (p = 0.041) for Functional Evenness (FEve), whereas Treatment had no statistically significant effect. FEve values were lowest in winter and peaked in spring (Figure 4B). Functional Divergence (FDiv) was significantly affected by Treatment (p = 0.021), with the highest values recorded in burned areas, particularly in sites under artificial regeneration. By contrast, the main effect of Season was not significant, although the Treatment × Season interaction suggested a partially supportive trend (Figure 4C). Rao’s Quadratic Entropy (RaoQ) showed no significant effects of either Treatment or Season, yet values tended to be higher in unburned areas, especially during summer (Figure 4D).
Post hoc comparisons supported these patterns, revealing significant differences in FRic during spring between unburned areas and burned areas under artificial regeneration (Δ = −0.111 ± 0.041, p = 0.031), and in FEve during winter between burned areas under natural regeneration and unburned areas (Δ = −0.243 ± 0.112, p = 0.042) (Table 3). All other pairwise comparisons were not significant, with detailed results provided in Appendix A Table A2.

Table 3.
Significant post hoc pairwise comparisons (emmeans contrasts) between treatments within seasons for functional richness (FRic) and functional evenness (FEve). Only significant contrasts (p < 0.05) are shown here; the complete set of comparisons for all functional diversity metrics is provided in Appendix A Table A2.
Indicator species analysis revealed that certain species exhibited specificity to different habitat groups. In unburned areas, V. vulpes (IndVal = 0.586, p = 0.001) and C. caracal (IndVal = 0.510, p = 0.001) were identified as significant indicator species. In burned areas, L. europaeus emerged as a strong indicator species (IndVal = 0.853, p = 0.001). Moreover, M. meles was associated with both unburned areas and burned areas under natural regeneration (IndVal = 0.563, p = 0.004), serving as a shared indicator of these habitats (Table 4).

Table 4.
Indicator species analysis results for mammal assemblages across treatments *.
4. Discussion
Fire in forest ecosystems acts not only as a driver of species richness but also as a force reshaping the functional structure of communities [1,63,64,65,66]. The findings of this study suggest that mammal communities undergo a marked functional reorganization after fire, particularly in burned areas, where carnivorous species become more prominent and functional-trait diversity declines, reflecting strong environmental filtering. Although our study did not directly quantify small-mammal abundance, previous research from the same region (Pinus brutia forests) reports that post-fire environments can promote increases in opportunistic and pyrophilous invertebrates during the early stages of succession [8,67,68]. These studies further indicate that insect taxa from the orders Hemiptera, Homoptera, and Lepidoptera frequently exploit the foliage of resprouting plants, and that the associated rise in invertebrate abundance can facilitate the occurrence of Soricidae as potential bioindicators of post-fire regeneration dynamics [69,70]. Within this ecological context, the presence of small mammals in burned habitats may, in turn, create conditions that facilitate the succession of opportunistic carnivores. In line with this interpretation, our results suggest that the red fox (Vulpes vulpes) is more frequently detected in burned areas during the early post-fire stage. Such transformations are not limited to changes in species presence or absence; rather, they reshape biological organization through the functional roles of species [71].
Beyond carnivores, our findings also highlight the functional relevance of wild boar (Sus scrofa) within post-fire Mediterranean forests. This species is among the most influential ecosystem engineers in the region [72]. Through intensive rooting and soil disturbance, S. scrofa modifies soil structure, nutrient cycling, and seed-bank dynamics, thereby directly influencing post-fire vegetation recovery trajectories [73]. By increasing micro-habitat heterogeneity, these disturbances may facilitate germination in both herbaceous and woody plants and indirectly shape prey availability for higher trophic levels. Accordingly, the ecological importance of S. scrofa is likely underestimated when evaluated solely through abundance-based indicators, underscoring the relevance of trait-based frameworks in interpreting post-fire community responses [74,75].
The relatively low detection frequency of some species in our camera-trap dataset should therefore not be construed as evidence of low ecological relevance. Several taxa recorded at low frequency—including golden jackal (Canis aureus), brown bear (Ursus arctos), and stone marten (Martes foina)—are characterized by behavioral flexibility, large home-range requirements, or habitat associations that inherently reduce detectability. For instance, U. arctos typically occurs at low population densities and shows strong affinity to structurally complex, late-successional habitats, which is consistent with its occurrence exclusively in unburned stands in our study [76]. C. aureus often shifts habitat use seasonally in response to resource availability, leading to transient use of burned areas rather than persistent site fidelity during early post-fire stages, which may reduce repeated detections [77]. Likewise, M. foina is closely associated with rocky refugia and dense shrub cover, conditions that can constrain camera visibility and detection probability despite continued functional presence in the system [78].
Taken together, these patterns indicate that camera-trap detections reflect both ecological activity and methodological constraints, and that function-based interpretations provide a more reliable lens for understanding how species contribute to post-fire community reorganization.
The functional diversity metrics indicated a clear contraction in mammal communities after fire. In unburned areas, significantly higher FRic values pointed to the maintenance of a broad functional niche spectrum, whereas burned habitats were marked by the opposite pattern: depressed FRic combined with elevated FDiv. This configuration suggests that species with specific adaptations rise to prominence. They increasingly dominate resource-use differentiation within the community. These dynamics imply that, during the early stages of post-fire succession, species with strong dispersal ability and generalist feeding strategies are more likely to become dominant, thereby promoting functional homogenization [41,79].
Silvicultural interventions following fire further shaped the trajectory of functional assembly. Artificially regenerated stands exhibited markedly lower FRic values compared with the partial recovery observed under natural regeneration. This pattern resonates with findings from small-mammal studies: Torre et al. [37] noted that habitat homogenization under artificial regeneration compressed community structure, while natural regeneration enhanced structural diversity and facilitated recolonization. Raybuck et al. [36] likewise demonstrated that oak regeneration strategies had a substantial influence on the early dynamics of mammal communities. Taken together, these findings show that functional diversity metrics provide a sensitive lens for detecting the effects of silvicultural practices and seasonal processes on community organization.
The seasonal decline of FEve in winter followed by an increase in spring suggests that communities exhibit flexible responses to shifting environmental conditions. Elevated FDiv values, particularly in burned areas subjected to artificial regeneration, indicate that species with specific strategies become relatively more abundant, thereby amplifying niche differentiation. Although RaoQ did not reach statistical significance, the consistently higher values observed in unburned habitats indicate reduced functional similarity and the emergence of more heterogeneous communities. Taken together, these patterns underscore that functional diversity metrics are shaped not only by silvicultural practices but also by strong seasonal dynamics.
Seasonal patterns indeed proved decisive in the reorganization of communities. The pronounced differences in FRic during spring and summer indicate that resource abundance and habitat availability directly influence functional diversity. In contrast, the convergence observed in autumn and winter suggests that under conditions of resource scarcity, species tend toward more homogeneous structures. Comparable patterns have been reported across other ecosystems as well [80]. This finding aligns closely with our results, emphasizing that the structural diversity and resource richness maintained in unburned areas facilitate the expression of interspecific functional differences.
Functional diversity analyses further reveal that unburned habitats provide structurally complex conditions that allow the coexistence of species with diverse ecological strategies. This is particularly relevant to carnivores such as C. caracal, whose habitat use is tightly linked to both vegetation cover and prey availability. This species requires not only shrubs, rocky areas, and multilayered vegetation that facilitate concealment and hunting, but also the diverse prey base supported by such structures. Vegetation diversity offers more than physical shelter; it also fosters the formation of fine-scale habitat mosaics that sustain a broader spectrum of food resources [38,81]. Such conditions enable the persistence of species with broad trophic niches, such as C. caracal, thereby enhancing functional heterogeneity at the community level [39,82,83]. In this context, the high FRic values observed in unburned areas indicate that these ecosystems provide more suitable and sustainable environments for upper trophic components. These results highlight the importance of post-fire silvicultural practices that enhance habitat heterogeneity, particularly natural regeneration techniques. The occurrence of caracal in naturally regenerated stands in our study further supports the ecological significance of these stands.
The shifts in species composition observed after fire are not confined to the direct effects on animal communities but are also closely intertwined with transformations in vegetation structure [84,85]. In the early post-fire phase, vegetation typically evolves toward a more homogeneous state, with structural changes such as increased shrub encroachment, the dominance of herbaceous species, or the spread of pioneer taxa [84]. These alterations reshape fundamental life strategies-sheltering, foraging, and mobility-across a broad range of taxa, from top predators to herbivorous mammals. For instance, a decline in vegetation heterogeneity can simultaneously constrain the concealment opportunities of prey species and affect the hunting success of predators. Such dynamics may, in turn, induce substantial shifts in the spatial distribution of mammal species and in the very nature of reciprocal biotic interactions, including predator-prey balances and competitive relationships.
The structural simplification of vegetation induced by fire also brings about microclimatic alterations, imposing additional ecological stress on thermally sensitive species [86]. This process increases the risk of local extinctions for habitat specialists, while creating favorable conditions for generalist and stress-tolerant species to thrive. The functional homogenization detected in this study thus emerges as a direct reflection of the novel environmental and biotic framework shaped by fire. Notably, the prevalence of ecologically flexible generalists such as V. vulpes [87] and apex predators such as C. caracal [88] in unburned areas underscores the pivotal role of habitat filtering at the functional level. At the same time, the greater dispersal and occurrence of the red fox in naturally regenerated habitats, relative to the caracal, can likewise be interpreted within this framework, given its higher tolerance to environmental stress.
The indicator species analysis further corroborates these patterns. Both V. vulpes and C. caracal showed statistically significant specificity and fidelity to unburned areas, underscoring how high vegetation heterogeneity and prey abundance in these habitats sustain predator presence. Particularly for C. caracal, whose wide-ranging movements and apex position in the trophic hierarchy demand structurally complex environments, the results indicate that a sustainable ecological niche can only be maintained where such habitat complexity is preserved [34,39,46]. Conversely, our findings highlight L. europaeus as a strong indicator of burned areas, revealing how species adapted to open and simplified habitats gain a competitive advantage in post-fire landscapes. This study further demonstrates that natural regeneration practices facilitate the colonization of hare populations in burned areas. Indeed, under natural successional dynamics, hare abundance typically peaks during the second post-fire year [33]. Here, however, our results show that the expected second-year abundances can be reached as early as the first year under natural regeneration. This suggests that natural silvicultural interventions can accelerate successional processes, acting as a rapid “remedy” for ecological degradation and thereby shortening the path to ecosystem recovery.
M. meles was associated with both unburned and naturally regenerated areas, a pattern that likely reflects its reliance on intermediate levels of habitat complexity and its avoidance of competition with carnivores of similar size [89,90]. Indeed, in this study the species emerged as an indicator in both burned and unburned sites. Its lack of direct competition with the red fox in burned habitats and with the caracal in unburned habitats helps explain this dual association. Another key factor is its fossorial behavior: the badger’s digging ability facilitates its persistence in both environments [56]. Colonization of burned landscapes is behaviorally straightforward for this species, as individuals can readily reoccupy pre-fire burrows after disturbance. Functioning almost as an “ecosystem engineer,” badgers create favorable conditions for post-fire recovery by reshaping the soil environment. Through digging, they generate microhabitats that can facilitate the arrival of new plant and animal species into burned areas [91,92]. In our study, badgers were more frequently recorded in naturally regenerated areas than in artificially regenerated stands. This pattern is plausibly explained by the lower degree of disturbance under natural regeneration, where soils are not deeply tilled by heavy machinery. The preservation of intact burrow systems reduces habitat disruption and encourages the settlement of new individuals. In addition, badgers can alter the characteristic acidic topsoil profile of Turkish red pine forests: by excavating, they bring deeper soil material to the surface, reducing soil acidity and increasing the availability of other elements [92]. Taken together, these findings suggest that natural regeneration is an effective restoration approach. It promotes both floristic and faunistic diversity by increasing habitat heterogeneity in Mediterranean ecosystems. This reinforces the notion that environmental filtering processes shape not only functional traits but also habitat preferences and life-history strategies of species.
In conclusion, this study demonstrates that the post-fire restructuring of mammal communities is not confined to the direct effects of fire alone; rather, it reflects a multilayered process shaped by silvicultural practices, habitat complexity, and seasonal dynamics. The contraction in functional diversity underscores the dominance of environmental filtering mechanisms, while also revealing that habitat management practices can either mitigate or exacerbate these patterns. Accordingly, post-fire ecosystem management should move beyond passive protection. It should also incorporate restoration strategies that actively restore habitat mosaics and vegetation diversity.
Our findings further highlight that focusing exclusively on vegetation recovery is insufficient for post-fire ecosystem management; the dynamics of wildlife must also be explicitly integrated into decision-making. The roles played by species across multiple trophic levels—from large mammals to smaller faunal groups—are critical in steering successional trajectories and safeguarding ecosystem integrity. Therefore, post-fire restoration policies should not only enhance habitat mosaics and structural diversity but also be urgently reinforced through holistic approaches that place wildlife management at their core. Such integrated strategies will not only strengthen ecological resilience but also ensure the sustainable persistence of conservation-priority species.
Author Contributions
Conceptualization, Y.İ., S.Ö. and O.Ü.; methodology, Y.İ. and S.Ö.; formal analysis, S.Ö.; investigation, Y.İ. and O.Ü.; data curation, Y.İ., S.Ö. and O.Ü.; visualization, S.Ö.; supervision, Y.İ., S.Ö. and O.Ü.; project administration, Y.İ. and O.Ü.; funding acquisition, Y.İ. and O.Ü.; writing—original draft, Y.İ., S.Ö. and O.Ü.; writing—review and editing, Y.İ., S.Ö. and O.Ü. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. The data and figures that support the findings of this study are openly available in Zenodo at https://zenodo.org/records/18097187 (Accessed date 30 December 2025). Further inquiries can be directed to the corresponding author.
Acknowledgments
The camera trap data in this study were obtained as part of the “Don’t Let the Caracals Get Hurt” Project supported by WWF-Turkey, and the “Last Wild Cats of Mediterranean Habitats” Project, led by the Support Foundation for Civil Society and the Turkish Mozaik Foundation and supported by the Turkish Coast Environment Fund both carried out by the NATURA Association for the Conservation of Nature and Culture. We would like to thank the officials of the Muğla Regional Directorate of Forestry, Marmaris Forest Management Directorate, and Marmaris National Parks Directorate for their official permissions and logistic support during the field work.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| FD | Functional Diversity |
| FRic | Functional Richness |
| FEve | Functional Evenness |
| RaoQ | Rao’s Quadratic Entropy |
| CWM | Community Weighted Mean |
| EMM | Estimated Marginal Mean |
| LMM | Linear Mixed-Effects Model |
| IUCN | International Union for Conservation of Nature |
| AO | Average Number of Offspring per Year |
| AP | Activity Period |
| AT | Anthropogenic Tolerance |
| BS | Body Size |
| DA | Dispersal Ability |
| DG | Soil Digger |
| GA | Gallery Type Nesting in the Soil |
| HP | Habitat Preference |
| IP | Important Prey |
| SD | Seed Disperser |
| TL | Trophic Level |
| TP | Top Predator |
| Burned_Art | Burned stands where post-fire artificial silvicultural practices |
| Burned_Nat | Burned stands where no artificial practices |
| Unburned | Representing areas unaffected by wildfire |
Appendix A

Table A1.
Station × season values of functional diversity metrics (FRic, FEve, FDiv, RaoQ). NA values indicate cases where indices could not be calculated due to insufficient species richness or inadequate trait dimensionality to construct a valid functional trait space.
Table A1.
Station × season values of functional diversity metrics (FRic, FEve, FDiv, RaoQ). NA values indicate cases where indices could not be calculated due to insufficient species richness or inadequate trait dimensionality to construct a valid functional trait space.
| nbsp * | sing.sp ** | FRic | FEve | FDiv | RaoQ | |
|---|---|---|---|---|---|---|
| s1_Winter | 1 | 0 | NA | NA | NA | 0 |
| s1_Spring | 1 | 0 | NA | NA | NA | 0 |
| s1_Summer | 1 | 0 | NA | NA | NA | 0 |
| s1_Autumn | 1 | 0 | NA | NA | NA | 0 |
| s2_Winter | 2 | 1 | NA | NA | NA | 4.214 |
| s2_Spring | 3 | 2 | 0.298 | 0.690 | 0.895 | 9.967 |
| s2_Summer | 2 | 2 | NA | NA | NA | 8.563 |
| s2_Autumn | 2 | 0 | NA | NA | NA | 6.660 |
| s3_Winter | 4 | 3 | 0.508 | 0.608 | 0.973 | 5.751 |
| s3_Spring | 3 | 1 | 0.310 | 0.935 | 0.907 | 6.135 |
| s3_Summer | 2 | 1 | NA | NA | NA | 7.225 |
| s3_Autumn | 3 | 2 | 0.298 | 0.520 | 0.925 | 8.056 |
| s4_Winter | 3 | 1 | 0.210 | 0.905 | 0.915 | 9.546 |
| s4_Spring | 3 | 1 | 0.210 | 0.735 | 0.899 | 8.850 |
| s4_Summer | 3 | 2 | 0.210 | 0.681 | 0.869 | 8.059 |
| s4_Autumn | 2 | 0 | NA | NA | NA | 9.436 |
| s5_Winter | 3 | 1 | 0.298 | 0.397 | 0.973 | 6.225 |
| s5_Spring | 3 | 1 | 0.298 | 0.794 | 0.955 | 10.014 |
| s5_Summer | 3 | 2 | 0.298 | 0.690 | 0.895 | 9.967 |
| s5_Autumn | 3 | 0 | 0.298 | 0.610 | 0.929 | 9.004 |
| s6_Winter | 4 | 0 | 0.508 | 0.709 | 0.953 | 9.377 |
| s6_Spring | 3 | 1 | 0.298 | 0.446 | 0.959 | 6.952 |
| s6_Summer | 4 | 3 | 0.508 | 0.782 | 0.935 | 10.288 |
| s6_Autumn | 3 | 2 | 0.298 | 0.262 | 0.965 | 4.342 |
| s7_Winter | 4 | 1 | 0.508 | 0.695 | 0.948 | 8.601 |
| s7_Spring | 4 | 1 | 0.508 | 0.673 | 0.955 | 7.838 |
| s7_Summer | 3 | 2 | 0.298 | 0.520 | 0.925 | 8.056 |
| s7_Autumn | 3 | 1 | 0.298 | 0.690 | 0.934 | 9.660 |
| s8_Winter | 2 | 0 | NA | NA | NA | 7.643 |
| s8_Spring | 3 | 1 | 0.298 | 0.417 | 0.919 | 6.698 |
| s8_Summer | 2 | 1 | NA | NA | NA | 4.214 |
| s8_Autumn | 2 | 1 | NA | NA | NA | 4.718 |
| s9_Winter | 3 | 2 | 0.210 | 0.585 | 0.891 | 7.273 |
| s9_Spring | 4 | 1 | 0.508 | 0.823 | 0.843 | 9.902 |
| s9_Summer | 3 | 0 | 0.310 | 0.999 | 0.964 | 10.297 |
| s9_Autumn | 1 | 0 | NA | NA | NA | 0.000 |
| s10_Winter | 2 | 1 | NA | NA | NA | 6.842 |
| s10_Spring | 5 | 3 | 0.736 | 0.733 | 0.766 | 11.575 |
| s10_Summer | 4 | 4 | 0.508 | 0.935 | 0.900 | 11.323 |
| s10_Autumn | 2 | 1 | NA | NA | NA | 4.284 |
| s11_Winter | 5 | 1 | 0.661 | 0.852 | 0.871 | 11.599 |
| s11_Spring | 5 | 2 | 0.661 | 0.535 | 0.941 | 9.570 |
| s11_Summer | 4 | 2 | 0.508 | 0.574 | 0.937 | 9.879 |
| s11_Autumn | 4 | 2 | 0.508 | 0.778 | 0.965 | 9.525 |
| s12_Winter | 6 | 2 | 0.670 | 0.752 | 0.912 | 10.627 |
| s12_Spring | 6 | 4 | 0.816 | 0.525 | 0.928 | 10.034 |
| s12_Summer | 3 | 0 | 0.298 | 0.798 | 0.886 | 10.906 |
| s12_Autumn | 6 | 3 | 0.934 | 0.595 | 0.915 | 11.804 |
| s13_Winter | NA | NA | NA | NA | NA | NA |
| s13_Spring | 3 | 3 | 0.236 | 0.825 | 0.783 | 11.344 |
| s13_Summer | NA | NA | NA | NA | NA | NA |
| s13_Autumn | 1 | 1 | NA | NA | NA | 0.000 |
| s14_Winter | 7 | 0 | 0.727 | 0.733 | 0.899 | 11.026 |
| s14_Spring | 6 | 1 | 0.670 | 0.597 | 0.918 | 11.849 |
| s14_Summer | 5 | 1 | 0.661 | 0.930 | 0.884 | 12.110 |
| s14_Autumn | 4 | 0 | 0.463 | 0.684 | 0.863 | 9.808 |
| s15_Winter | 4 | 0 | 0.463 | 0.777 | 0.833 | 9.728 |
| s15_Spring | 3 | 0 | 0.138 | 0.874 | 0.871 | 9.614 |
| s15_Summer | 4 | 2 | 0.463 | 0.933 | 0.946 | 10.816 |
| s15_Autumn | 3 | 0 | 0.138 | 0.834 | 0.728 | 8.140 |
* nb.sp: Number of species (species richness); the total number of species recorded in each station–season combination. ** sing.sp: Number of singleton species; species occurring with an abundance of 1 within the corresponding station–season community.

Table A2.
Full results of post hoc pairwise comparisons.
Table A2.
Full results of post hoc pairwise comparisons.
| Fric | Months | Contrast | Season | Estimate | SE | df | t.ratio | p |
| 1 | Burned_Art − Burned_Nat | Winter | −0.027 | 0.054 | 29.162 | −0.502 | 0.871 | |
| 2 | Burned_Art − Unburned | Winter | −0.064 | 0.049 | 29.458 | −1.321 | 0.395 | |
| 3 | Burned_Nat − Unburned | Winter | −0.037 | 0.042 | 28.597 | −0.874 | 0.661 | |
| 4 | Burned_Art − Burned_Nat | Spring | −0.035 | 0.046 | 24.822 | −0.764 | 0.728 | |
| 5 | Burned_Art − Unburned | Spring | −0.111 | 0.041 | 24.822 | −2.718 | 0.031 | |
| 6 | Burned_Nat − Unburned | Spring | −0.076 | 0.037 | 24.822 | −2.052 | 0.121 | |
| 7 | Burned_Art − Burned_Nat | Summer | −0.039 | 0.067 | 33.319 | −0.581 | 0.831 | |
| 8 | Burned_Art − Unburned | Summer | −0.064 | 0.061 | 33.658 | −1.042 | 0.556 | |
| 9 | Burned_Nat − Unburned | Summer | −0.025 | 0.041 | 27.892 | −0.612 | 0.815 | |
| 10 | Burned_Art − Burned_Nat | Autumn | −0.016 | 0.067 | 33.319 | −0.237 | 0.969 | |
| 11 | Burned_Art − Unburned | Autumn | −0.080 | 0.063 | 33.797 | −1.272 | 0.420 | |
| 12 | Burned_Nat − Unburned | Autumn | −0.064 | 0.043 | 29.538 | −1.482 | 0.314 | |
| Feve | Months | Contrast | Season | Estimate | SE | df | t.ratio | p |
| 1 | Burned_Art − Burned_Nat | Winter | 0.116 | 0.145 | 34.000 | 0.797 | 0.708 | |
| 2 | Burned_Art − Unburned | Winter | −0.128 | 0.130 | 34.000 | −0.982 | 0.593 | |
| 3 | Burned_Nat − Unburned | Winter | −0.243 | 0.112 | 34.000 | −2.164 | 0.042 | |
| 4 | Burned_Art − Burned_Nat | Spring | 0.236 | 0.120 | 34.000 | 1.956 | 0.139 | |
| 5 | Burned_Art − Unburned | Spring | 0.057 | 0.107 | 34.000 | 0.537 | 0.854 | |
| 6 | Burned_Nat − Unburned | Spring | −0.178 | 0.097 | 34.000 | −1.846 | 0.170 | |
| 7 | Burned_Art − Burned_Nat | Summer | 0.040 | 0.186 | 34.000 | 0.214 | 0.975 | |
| 8 | Burned_Art − Unburned | Summer | −0.119 | 0.173 | 34.000 | −0.687 | 0.773 | |
| 9 | Burned_Nat − Unburned | Summer | −0.158 | 0.109 | 34.000 | −1.449 | 0.328 | |
| 10 | Burned_Art − Burned_Nat | Autumn | −0.002 | 0.186 | 34.000 | −0.010 | 1.000 | |
| 11 | Burned_Art − Unburned | Autumn | −0.243 | 0.177 | 34.000 | −1.370 | 0.368 | |
| 12 | Burned_Nat − Unburned | Autumn | −0.241 | 0.116 | 34.000 | −2.070 | 0.111 | |
| Fdiv | Months | Contrast | Season | Estimate | SE | df | t.ratio | p |
| 1 | Burned_Art − Burned_Nat | Winter | −0.009 | 0.039 | 33.124 | −0.236 | 0.970 | |
| 2 | Burned_Art − Unburned | Winter | 0.057 | 0.035 | 33.209 | 1.622 | 0.251 | |
| 3 | Burned_Nat − Unburned | Winter | 0.066 | 0.030 | 33.073 | 2.180 | 0.090 | |
| 4 | Burned_Art − Burned_Nat | Spring | −0.060 | 0.033 | 32.247 | −1.848 | 0.170 | |
| 5 | Burned_Art − Unburned | Spring | −0.015 | 0.029 | 32.247 | −0.529 | 0.858 | |
| 6 | Burned_Nat − Unburned | Spring | 0.045 | 0.026 | 32.247 | 1.721 | 0.213 | |
| 7 | Burned_Art − Burned_Nat | Summer | −0.044 | 0.050 | 33.936 | −0.878 | 0.657 | |
| 8 | Burned_Art − Unburned | Summer | −0.040 | 0.046 | 33.978 | −0.869 | 0.663 | |
| 9 | Burned_Nat − Unburned | Summer | 0.004 | 0.030 | 32.895 | 0.123 | 0.992 | |
| 10 | Burned_Art − Burned_Nat | Autumn | −0.021 | 0.050 | 33.936 | −0.412 | 0.911 | |
| 11 | Burned_Art − Unburned | Autumn | 0.011 | 0.047 | 33.994 | 0.236 | 0.970 | |
| 12 | Burned_Nat − Unburned | Autumn | 0.032 | 0.031 | 33.334 | 1.014 | 0.574 | |
| RaoQ | Months | Contrast | Season | Estimate | SE | df | t.ratio | p |
| 1 | Burned_Art − Burned_Nat | Winter | −0.034 | 0.027 | 26.863 | −1.245 | 0.438 | |
| 2 | Burned_Art − Unburned | Winter | −0.050 | 0.024 | 27.874 | −2.106 | 0.107 | |
| 3 | Burned_Nat − Unburned | Winter | −0.016 | 0.024 | 27.874 | −0.689 | 0.772 | |
| 4 | Burned_Art − Burned_Nat | Spring | −0.025 | 0.027 | 26.863 | −0.934 | 0.624 | |
| 5 | Burned_Art − Unburned | Spring | −0.057 | 0.023 | 26.863 | −2.456 | 0.053 | |
| 6 | Burned_Nat − Unburned | Spring | −0.032 | 0.023 | 26.863 | −1.378 | 0.366 | |
| 7 | Burned_Art − Burned_Nat | Summer | −0.026 | 0.027 | 26.863 | −0.968 | 0.603 | |
| 8 | Burned_Art − Unburned | Summer | −0.048 | 0.024 | 27.874 | −2.035 | 0.123 | |
| 9 | Burned_Nat − Unburned | Summer | −0.022 | 0.024 | 27.874 | −0.934 | 0.624 | |
| 10 | Burned_Art − Burned_Nat | Autumn | −0.004 | 0.027 | 26.863 | −0.151 | 0.988 | |
| 11 | Burned_Art − Unburned | Autumn | 0.005 | 0.023 | 26.863 | 0.199 | 0.978 | |
| 12 | Burned_Nat − Unburned | Autumn | 0.009 | 0.023 | 26.863 | 0.373 | 0.926 |
References
- Keeley, J.E.; Pausas, J.G.; Rundel, P.W.; Bond, W.J.; Bradstock, R.A. Fire as an evolutionary pressure shaping plant traits. Trends Plant Sci. 2011, 16, 406–411. [Google Scholar] [CrossRef]
- Kelly, L.T.; Brotons, L. Using fire to promote biodiversity. Science 2017, 355, 1264–1265. [Google Scholar] [CrossRef]
- Rundel, P.W.; Arroyo, M.T.; Cowling, R.M.; Keeley, J.E.; Lamont, B.B.; Pausas, J.G.; Vargas, P. Fire and plant diversification in Mediterranean-climate regions. Front. Plant Sci. 2018, 9, 851. [Google Scholar] [CrossRef]
- Güney, C.O.; Mert, A.; Gülsoy, S. Assessing fire severity in Turkey’s forest ecosystems using spectral indices from satellite images. J. For. Res. 2023, 34, 1747–1761. [Google Scholar] [CrossRef]
- Mittermeier, R.A.; Myers, N.; Mittermeier, C.G.; Robles Gil, P. Hotspots: Earth’s Biologically Richest and Most Endangered Terrestrial Ecoregions; CEMEX: Mexico City, Mexico, 1999. [Google Scholar]
- Hrdina, A.; Romportl, D. Evaluating global biodiversity hotspots—Very rich and even more endangered. J. Landsc. Ecol. 2017, 10, 108–115. [Google Scholar] [CrossRef]
- Uyar, Ç.; Özdemir, S.; Perkumienė, D.; Aleinikovas, M.; Šilinskas, B.; Škėma, M. Spatiotemporal patterns of avian species richness across climatic regions. Diversity 2025, 17, 557. [Google Scholar] [CrossRef]
- Kalem, S.; Ürker, O.; İlemin, Y.; Tavşanoğlu, Ç.; Günlü, A.; Yenisey Kaynaş, B.; Köşk, U.C. Ecological and Socio-Economic Effects of Mega Forest Fires in the Mediterranean Region; Technical Report; PrintWorld Publishing: Ankara, Turkey, 2022. [Google Scholar]
- Ürker, O.; İlemin, Y.; Ergan, G.; Kemer, N. Guide for Post-Fire Ecological Restoration in Protected Areas; The Publications of NATURA—The Society for the Conservation of Nature and Culture: Marmaris, Turkey, 2024. [Google Scholar]
- Kavgacı, A.; Başaran, M.A. (Eds.) Forest Fires; The Publication of The Forester’s Association of Türkiye: Ankara, Turkey, 2023. [Google Scholar]
- Odabaşı, T.; Çalışkan, A.; Bozkış, F. Orman Bakımı; İstanbul Üniversitesi Orman Fakültesi Yayın No. 4458: İstanbul, Turkey, 2004. [Google Scholar]
- Puettmann, K.J.; Coates, K.D.; Messier, C.C. A Critique of Silviculture: Managing for Complexity; Island Press: Washington, DC, USA, 2012. [Google Scholar]
- Genç, M. Silvikültür Tekniği; Süleyman Demirel Üniversitesi Orman Fakültesi Yayın No. 46: Isparta, Turkey, 2013. [Google Scholar]
- Tavşanoğlu, Ç.; Pausas, J.G. A functional trait database for Mediterranean Basin plants. Sci. Data 2018, 5, 180135. [Google Scholar] [CrossRef]
- Kavgacı, A.; Tavşanoğlu, Ç. Akdeniz tipi ekosistemlerde yangın sonrası vejetasyon dinamiği. Turk. J. For. 2010, 11, 149–166. [Google Scholar]
- Tavşanoğlu, Ç.; Gürkan, B. Long-term post-fire dynamics of co-occurring woody species in Pinus brutia forests: The role of regeneration mode. Plant Ecol. 2014, 215, 355–365. [Google Scholar] [CrossRef]
- Caro, T.M. Species richness and abundance of small mammals inside and outside an African national park. Biol. Conserv. 2001, 98, 251–257. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; McCarthy, M.A.; Parris, K.M.; Pope, M.L. Habitat fragmentation, landscape context, and mammalian assemblages in south-eastern Australia. J. Mammal. 2000, 81, 787–797. [Google Scholar] [CrossRef]
- Sergio, F.; Caro, T.; Brown, D.; Clucas, B.; Hunter, J.; Ketchum, J.; McHugh, K.; Hiraldo, F. Top predators as conservation tools: Ecological rationale, assumptions, and efficacy. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 1–19. [Google Scholar] [CrossRef]
- Lazarus, M.; Sekovanić, A.; Orct, T.; Reljić, S.; Kusak, J.; Jurasović, J.; Huber, Đ. Apex predatory mammals as bioindicator species in environmental monitoring of elements in Dinaric Alps (Croatia). Environ. Sci. Pollut. Res. 2017, 24, 23977–23991. [Google Scholar] [CrossRef]
- Caro, T.M.; O’Doherty, G. On the use of surrogate species in conservation biology. Conserv. Biol. 1999, 13, 805–814. [Google Scholar] [CrossRef]
- Leader-Williams, N.; Dublin, H.T. Charismatic megafauna as flagship species. In Conservation Biology Series; Cambridge University Press: Cambridge, UK, 2000; pp. 53–84. [Google Scholar]
- Quinn, R.D. Animals, fire, and vertebrate herbivory in Californian chaparral and other Mediterranean-type ecosystems. In The Role of Fire in Mediterranean-type Ecosystems; Moreno, J.M., Oechel, W.C., Eds.; Springer: New York, NY, USA, 1994; pp. 46–78. [Google Scholar]
- Turschak, G.; Rochester, C.J.; Hathaway, S.A.; Stokes, D.C.; Haas, C.; Fisher, R.N. Effects of Large-Scale Wildfire on Carnivores in San Diego County, California; US Geological Survey, Western Ecological Center: Sacramento, CA, USA, 2010.
- Borchert, M.I. Mammalian carnivore use of a high-severity burn in conifer forests in the San Bernardino Mountains of southern California, USA. Hystrix 2012, 23, 51. [Google Scholar]
- Schuette, P.A.; Diffendorfer, J.E.; Deutschman, D.H.; Tremor, S.; Spencer, W. Carnivore distributions across chaparral habitats exposed to wildfire and rural housing in southern California. Int. J. Wildl. Fire 2014, 23, 591–600. [Google Scholar] [CrossRef]
- van Mantgem, P.J.; Caprio, A.C.; Stephenson, N.L.; Das, A.J. Does prescribed fire promote resistance to drought in low elevation forests of the Sierra Nevada, California, USA? Fire Ecol. 2016, 12, 13. [Google Scholar] [CrossRef]
- Green, D.S.; Roloff, G.J.; Heath, B.R.; Holekamp, K.E. Temporal dynamics of the responses by African mammals to prescribed fire. J. Wildl. Manag. 2015, 79, 235–242. [Google Scholar] [CrossRef]
- Chia, E.K.; Bassett, M.; Leonard, S.W.; Holland, G.J.; Ritchie, E.G.; Clarke, M.F.; Bennett, A.F. Effects of the fire regime on mammal occurrence after wildfire: Site effects vs landscape context in fire-prone forests. For. Ecol. Manag. 2016, 363, 130–139. [Google Scholar] [CrossRef]
- McGregor, H.W.; Legge, S.; Jones, M.E.; Johnson, C.N. Extraterritorial hunting expeditions to intense fire scars by feral cats. Sci. Rep. 2016, 6, 22559. [Google Scholar] [CrossRef]
- Geary, W.L.; Doherty, T.S.; Nimmo, D.G.; Tulloch, A.I.; Ritchie, E.G. Predator responses to fire: A global systematic review and meta-analysis. J. Anim. Ecol. 2020, 89, 955–971. [Google Scholar] [CrossRef]
- Birtsas, P.; Sokos, C.; Exadactylos, S. Carnivores in burned and adjacent unburned areas in a Mediterranean ecosystem. Mammalia 2012, 76, 4. [Google Scholar] [CrossRef]
- Sokos, C.; Birtsas, P.; Papaspyropoulos, K.G.; Tsachalidis, E.; Giannakopoulos, A.; Milis, C.; Milis, C.; Spyrou, V.; Manolakou, K.; Valiakos, G. Mammals and habitat disturbance: The case of brown hare and wildfire. Curr. Zool. 2016, 62, 421–430. [Google Scholar] [CrossRef]
- Soyumert, A.; Ertürk, A.; Tavşanoğlu, Ç. Fire-created habitats support large mammal community in a Mediterranean landscape. Mammal Res. 2020, 65, 323–330. [Google Scholar] [CrossRef]
- İlemin, Y. Mortality record of caracal and habitat loss for wildcat, depending on catastrophic wildfires of 2021 in south-western Turkey. Biol. Bull. Russ. Acad. Sci. 2021, 48, 92–95. [Google Scholar] [CrossRef]
- Raybuck, A.L.; Wang, G.; Keyser, P.D. Short-term response of small mammals following oak regeneration silviculture treatments. For. Ecol. Manag. 2012, 270, 201–209. [Google Scholar] [CrossRef]
- Torre, I.; Ribas, A.; Puig-Gironès, R. Effects of post-fire management on a Mediterranean small mammal community. Fire 2023, 6, 34. [Google Scholar] [CrossRef]
- İlemin, Y.; Gürkan, B. Status and activity patterns of the caracal, Caracal caracal (Schreber, 1776), in Datça and Bozburun Peninsulas, south-western Turkey. Zool. Middle East 2010, 50, 3–10. [Google Scholar] [CrossRef]
- İlemin, Y.; Koç, H.; Kaynaş, B.Y. Ecological characteristics of caracal (Caracal caracal) in Türkiye: An isolated feline population. J. Wildl. Biodivers. 2023, 7, 199–213. [Google Scholar]
- Laliberté, E.; Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 2010, 91, 299–305. [Google Scholar] [CrossRef]
- Villéger, S.; Mason, N.W.; Mouillot, D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 2008, 89, 2290–2301. [Google Scholar] [CrossRef]
- Rao, C.R. Diversity and dissimilarity coefficients: A unified approach. Theor. Popul. Biol. 1982, 21, 24–43. [Google Scholar] [CrossRef]
- Botta-Dukát, Z. Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. J. Veg. Sci. 2005, 16, 533–540. [Google Scholar] [CrossRef]
- Bricca, A.; Conti, L.; Tardella, M.F.; Catorci, A.; Iocchi, M.; Theurillat, J.P.; Cutini, M. Community assembly processes along a sub-Mediterranean elevation gradient: Analysing the interdependence of trait community weighted mean and functional diversity. Plant Ecol. 2019, 220, 1139–1151. [Google Scholar] [CrossRef]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef]
- Dufrêne, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Özdemir, S.; Çınar, T. Determining indicator plant species of Pinus brutia Ten. site index classes using interspecific correlation analysis in Antalya (Turkey). Cerne 2023, 29, e-103188. [Google Scholar] [CrossRef]
- Özdemir, S. Effects of environmental variables on taxonomic diversity and grouping of plant communities in the Mediterranean region (Antalya). Eur. J. For. Res. 2024, 143, 1903–1914. [Google Scholar] [CrossRef]
- Laliberté, E.; Legendre, P.; Shipley, B. FD, R package version 1.0–12; Measuring Functional Diversity from Multiple Traits, and Other Tools for Functional Ecology; CRAN: Vienna, Austria, 2014.
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Lenth, R.V. emmeans, R Package Version 1.7.0; Estimated Marginal Means, aka Least-Squares Means; CRAN: Vienna, Austria, 2021. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 7 September 2025).
- Wickham, H. ggplot2. Wiley Interdiscip. Rev. Comput. Stat. 2011, 3, 180–185. [Google Scholar] [CrossRef]
- Kassambara, A. ggpubr, R package version 2.0; ‘ggplot2’-Based Publication-Ready Plots; CRAN: Vienna, Austria, 2018.
- De Cáceres, M.; Jansen, F.; Dell, N. indicspecies, R package version 1.7.14; Relationship Between Species and Groups of Sites; CRAN: Vienna, Austria, 2025. Available online: https://CRAN.R-project.org/package=indicspecies (accessed on 29 September 2025).
- Hackländer, K.; Schai-Braun, S. Lepus europaeus. In The IUCN Red List of Threatened Species 2019; e.T41280A45187424; IUCN: Gland, Switzerland, 2016. [Google Scholar]
- Kranz, A.; Abramov, A.V.; Herrero, J.; Maran, T. Meles meles. In The IUCN Red List of Threatened Species 2016; e.T29673A45203002; IUCN: Gland, Switzerland, 2016. [Google Scholar]
- Hoffmann, M.; Sillero-Zubiri, C. Vulpes vulpes (amended version of 2016 assessment). In The IUCN Red List of Threatened Species 2021; e.T23062A193903628; IUCN: Gland, Switzerland, 2021. [Google Scholar]
- Avgan, B.; Henschel, P.; Ghoddousi, A. Caracal caracal. In The IUCN Red List of Threatened Species 2016; e.T3847A102424310; IUCN: Gland, Switzerland, 2016. [Google Scholar]
- Huber, D. Ursus arctos (Europe assessment). In The IUCN Red List of Threatened Species 2025; e.T41688A216873475; IUCN: Gland, Switzerland, 2025. [Google Scholar]
- Keuling, O.; Leus, K. Sus scrofa. In The IUCN Red List of Threatened Species 2019; e.T41775A44141833; IUCN: Gland, Switzerland, 2019. [Google Scholar]
- Hoffmann, M.; Arnold, J.; Duckworth, J.W.; Jhala, Y.; Kamler, J.F.; Krofel, M. Canis aureus (errata version published in 2020). In The IUCN Red List of Threatened Species 2018; IUCN: Gland, Switzerland, 2018. [Google Scholar]
- Abramov, A.V.; Kranz, A.; Herrero, J.; Choudhury, A.U.; Maran, T. Martes foina (amended version of 2016 assessment). In The IUCN Red List of Threatened Species 2024; e.T29672A259348828; IUCN: Gland, Switzerland, 2024. [Google Scholar]
- Pausas, J.G.; Keeley, J.E. Evolutionary ecology of resprouting and seeding in fire-prone ecosystems. New Phytol. 2014, 204, 55–65. [Google Scholar] [CrossRef]
- Güney, C.; Özkan, K.; Şentürk, Ö. Modelling of spatial prediction of fire ignition risk in the Antalya-Manavgat district. J. Fac. For. Istanbul Univ. 2016, 66, 459–470. [Google Scholar]
- Oncel Cekim, H.; Güney, C.O.; Şentürk, Ö.; Özel, G.; Özkan, K. A novel approach for predicting burned forest area. Nat. Hazards 2021, 105, 2187–2201. [Google Scholar] [CrossRef]
- González, T.M.; González-Trujillo, J.D.; Muñoz, A.; Armenteras, D. Effects of fire history on animal communities: A systematic review. Ecol. Process. 2022, 11, 11. [Google Scholar] [CrossRef]
- Wikars, L.O.; Schimmel, J. Immediate effects of fire severity on soil invertebrates in cut and uncut pine forests. For. Ecol. Manag. 2001, 141, 189–200. [Google Scholar] [CrossRef]
- Wikars, L.O. Dependence on fire in wood-living insects: An experiment with burned and unburned spruce and birch logs. J. Insect Conserv. 2002, 6, 1–12. [Google Scholar] [CrossRef]
- Haim, A.; Rozenfeld, A.; Izhaki, I. Post-fire response of shrews (Crocidura suaveolens) on Mount Carmel, Israel. Mammalia 1997, 61, 527–536. [Google Scholar] [CrossRef]
- Soyumert, A.; Tavşanoğlu, Ç.; Macar, O.; Kaynaş, B.Y.; Gürkan, B. Presence of large and medium-sized mammals in a burned pine forest in southwestern Turkey. Hystrix 2010, 21, 1–6. [Google Scholar]
- Wilson, L.J.; Fulton, C.J.; Hogg, A.M.; Joyce, K.E.; Radford, B.T.; Fraser, C.I. Climate-driven changes to ocean circulation and their inferred impacts on marine dispersal patterns. Glob. Ecol. Biogeogr. 2016, 25, 923–939. [Google Scholar] [CrossRef]
- Ünal, Y.; Pekin, B.K.; Oğurlu, İ.; Süel, H.; Koca, A. Human, Domestic Animal, Caracal (Caracal caracal), and Other Wildlife Species Interactions in a Mediterranean Forest Landscape. Eur. J. Wildl. Res. 2020, 66, 5. [Google Scholar] [CrossRef]
- Don, A.; Hagen, C.; Grüneberg, E.; Vos, C. Simulated Wild Boar Bioturbation Increases the Stability of Forest Soil Carbon. Biogeosciences 2019, 16, 4145–4155. [Google Scholar] [CrossRef]
- Tuinder, Q.A.; van Leeuwen, B.O.; Vergeer, P.; Scherer, G.; Fartmann, T.; Jansen, P.A. Impact of Wild Boar (Sus scrofa) Rooting Succession on Grasshoppers (Orthoptera) in Abandoned Calcareous Grasslands. J. Insect Conserv. 2025, 29, 78. [Google Scholar] [CrossRef]
- Sandom, C.J.; Hughes, J.; Macdonald, D.W. Rewilding the Scottish Highlands: Do Wild Boar, Sus scrofa, Use a Suitable Foraging Strategy to Be Effective Ecosystem Engineers? Restor. Ecol. 2013, 21, 336–343. [Google Scholar] [CrossRef]
- Zyśk-Gorczyńska, E.; Jakubiec, Z.; Wuczyński, A. Brown Bears (Ursus arctos) as Ecological Engineers: The Prospective Role of Trees Damaged by Bears in Forest Ecosystems. Can. J. Zool. 2015, 93, 133–141. [Google Scholar] [CrossRef]
- Negi, T. Review on Current Worldwide Status, Distribution, Ecology and Dietary Habits of Golden Jackal, Canis aureus. Octa J. Environ. Res. 2014, 2, 338–359. [Google Scholar]
- Goszczyński, J.; Posłuszny, M.; Pilot, M.; Gralak, B. Patterns of Winter Locomotion and Foraging in Two Sympatric Marten Species: Martes martes and Martes foina. Can. J. Zool. 2007, 85, 239–249. [Google Scholar] [CrossRef]
- Mouillot, D.; Graham, N.A.; Villéger, S.; Mason, N.W.; Bellwood, D.R. A functional approach reveals community responses to disturbances. Trends Ecol. Evol. 2013, 28, 167–177. [Google Scholar] [CrossRef]
- Martín-Regalado, M.; Pérez-Flores, J.; Bonilla-Valencia, M. Spatial patterns of taxonomic, functional, and phylogenetic diversity of mammals. Ecol. Evol. 2025, 15, e12345. [Google Scholar] [CrossRef]
- Tomassini, O.; Massolo, A. From fire to recovery: Temporal-shift of predator–prey interactions among mammals in Mediterranean ecosystems. Mamm. Biol. 2024, 104, 583–600. [Google Scholar] [CrossRef]
- Herfindal, I.; Linnell, J.D.; Odden, J.; Nilsen, E.B.; Andersen, R. Prey density, environmental productivity and home-range size in the Eurasian lynx (Lynx lynx). J. Zool. 2005, 265, 63–71. [Google Scholar] [CrossRef]
- Hayward, M.W.; O’Brien, J.; Kerley, G.I. Carrying capacity of large African predators: Predictions and tests. Biol. Conserv. 2007, 139, 219–229. [Google Scholar] [CrossRef]
- Pausas, J.G.; Keeley, J.E. A burning story: The role of fire in the history of life. BioScience 2009, 59, 593–601. [Google Scholar] [CrossRef]
- Ürker, O.; Tavşanoğlu, Ç.; Gürkan, B. Post-fire recovery of the plant community in Pinus brutia forests: Active vs. indirect restoration techniques after salvage logging. iForest 2018, 11, 635–642. [Google Scholar] [CrossRef]
- Grau-Andrés, R.; Moreira, B.; Pausas, J.G. Global plant responses to intensified fire regimes. Glob. Ecol. Biogeogr. 2024, 33, e13858. [Google Scholar] [CrossRef]
- Gil-Fernández, M.; Harcourt, R.; Newsome, T.; Towerton, A.; Carthey, A. Adaptations of the red fox (Vulpes vulpes) to urban environments in Sydney, Australia. J. Urban Ecol. 2020, 6, juaa009. [Google Scholar] [CrossRef]
- Leighton, G.R.; Froneman, W.; Serieys, L.E.; Bishop, J.M. Trophic downgrading of an adaptable carnivore in an urbanising landscape. Sci. Rep. 2023, 13, 21582. [Google Scholar] [CrossRef]
- Konstantinov, Y.; Spassov, N.; Acosta-Pankov, I. First records of golden jackal and European badger non-antagonistic interaction at the Lower Danube (Bulgaria). NW J. Zool. 2022, 18, 100–103. [Google Scholar]
- Pérez-Vigo, I.; Ferreras, P.; Finat, R.; Villafuerte, R. Tolerance and intraguild commensalism: The case of the European badger and the Iberian lynx. Eur. J. Wildl. Res. 2025, 71, 15. [Google Scholar] [CrossRef]
- Beca, G.; Valentine, L.E.; Galetti, M.; Hobbs, R.J. Ecosystem roles and conservation status of bioturbator mammals. Mammal Rev. 2022, 52, 192–207. [Google Scholar] [CrossRef]
- Kurek, P.; Kapusta, P.; Holeksa, J. Burrowing by badgers (Meles meles) and foxes (Vulpes vulpes) changes soil conditions and vegetation in a European temperate forest. Ecol. Res. 2014, 29, 1–11. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.