Abstract
Chikungunya fever (CHIKF) is a re-emerging infectious disease caused by the chikungunya virus (CHIKV), transmitted primarily by Aedes mosquitoes. A significant number progress to chronic chikungunya arthritis, which shares similarities with rheumatoid arthritis (RA). Despite evidence of a link between CHIKV infection and subsequent RA development, a comprehensive analysis of the relationship between these two diseases is lacking. This study systematically analyzes the incidence of RA after CHIKV infection and its immunological mechanisms, following PRISMA guidelines with literature searches across multiple databases up to 3 September 2024. Eligible studies included retrospective and prospective designs reporting RA diagnoses after CHIKV infection. Data extraction was performed independently, and the risk of bias was assessed using appropriate tools. Sixteen studies involving 2879 patients were included, with 449 individuals diagnosed with RA following CHIKV infection, resulting in a combined incidence of 13.7% (95% CI: 6.12% to 27.87%). High heterogeneity between studies was observed (I2 = 96%), indicating variability related to diagnostic criteria and population characteristics. This review highlights the significant RA incidence after CHIKV infection, emphasizing the need for research on autoimmune mechanisms, long-term rheumatological follow-up, early diagnostic biomarkers, and CHIKV’s long-term health impacts.
1. Introduction
Chikungunya fever (CHIKF) is a mosquito-borne, viral infection that has re-emerged in global outbreaks over the past 75 years. First reported in 1952, CHIKF has since spread to over 100 countries, with over 4 million cases reported, particularly after the first case in the Americas in 2013 [1,2]. The chikungunya virus (CHIKV), a positive-sense single-stranded RNA Alphavirus virus is transmitted by Aedes mosquitoes (Ae. aegypti and Ae. albopictus) [3]. The disease presents with sudden fever, incapacitating polyarthritis, myalgia, rash, headache, and gastrointestinal symptoms, typically 2 to 6 days after infection [4]. While usually self-limiting, with recovery in 10 days, up to 40% of cases progress to a chronic phase, chronic chikungunya arthritis (CCA), characterized by persistent or deforming arthritis [5].
Following the global spread from Africa of the vectors of CHIKV, A. aegypti and A. albopictus, and the increase in international travel, there has been rapid geographic expansion of CHIKV infection. With the resultant increasing CCA cases, better characterization of the clinical features of CCA has been possible [6,7]. Within the clinical spectrum of CCA, some patients present as RA “mimics” with symmetric polyarthritis, morning stiffness, frequent hand involvement, and in some individuals, positive RA biomarkers. [8,9,10]. Not infrequently, these individuals meet ACR (American College of Rheumatology)/EULAR (European Alliance of Associations for Rheumatology) classification criteria for RA [11,12] Whether CCA mimics RA or whether CHIKV infection increases the risk of developing ongoing RA is still a subject of debate.
The similarities between CCA and RA are pathological as well as clinical [5,10,11,13]. RA is a systemic inflammatory disease of multifactorial etiology characterized by chronic and progressive synovitis [9,10,11]. RA pathogenesis involves interactions between genetic, environmental, and immunological factors, resulting in dysregulated immune responses and chronic inflammation, damaging joints and other organs [10]. Immunophenotyping of peripheral blood mononuclear cells in RA and CCA reveals a common inflammatory profile, suggesting shared immunological mechanisms. Viral persistence in joints, detection of viral genetic material in the synovium, and activation of innate and adaptive immune responses are mechanisms proposed to explain CCA pathogenesis. Autoimmunity induced by molecular mimicry or virus-induced tissue damage has also been considered [8,11].
There has long been interest in whether RA is linked to viral infection [12]. For CCA, there is a fundamental pathogenic question as to whether the arthritis phase results from persistent viral infection or a post-viral inflammatory process [13,14]. There is also a question as to whether CCA patients benefit from a disease-modifying treatment strategy, borrowed from the treatment of RA [11]. For these multiple reasons, we conducted a systematic review and meta-analysis to evaluate current evidence on the development of RA following CHIKV infection. We believe that the increasingly available data justify further investigation of pathogenic similarities between these two diseases and support CCA therapeutic strategies informed by RA treatment.
2. Materials and Methods
The study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) (protocol CRD42024585798). This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [15] guidelines to address the following question: “What are the characteristics and incidence of rheumatoid arthritis development in patients after chikungunya virus infection?”
2.1. Information Sources
We systematically searched PubMed, Cochrane Library, Scielo, LILACS, Web of Science, and Scopus for relevant literature published until 3 September 2024. We also incorporated studies found through manual searches using Google Scholar, gray literature, and reference lists. Articles in English, Spanish, or Portuguese were included.
2.2. Eligibility Criteria
Retrospective and prospective studies, randomized controlled trials, and case series reporting diagnosis of RA after CHIKV infection, with clinically and laboratory-confirmed testing for anti-CHIKV-specific immunoglobulin M (for acute cases) or immunoglobulin G (post-exposure) detected by enzyme-linked immunosorbent assay (ELISA) or RNA virus by reverse-transcriptase polymerase chain reaction were included. Studies were excluded if they did not include laboratory confirmation of CHIKV, did not follow the ACR-EULAR classification criteria, or presented insufficient data for analysis. We excluded from this systematic review: letters to the editor, editorials, review articles, commentaries, preclinical trials, and papers without relevant data.
2.3. Search Strategy
Search terms included combinations of free-text and MeSH or Emtree terms for CHIK and RA, including (“Chikungunya virus” (MeSH) OR “Chikungunya fever” (MeSH) OR “CHIKV” OR “Chikungunya infection” OR “Chikungunya” OR “CHIK” OR “CHIKV”) AND (“Arthritis, Rheumatoid” (MeSH) OR “Rheumatoid arthritis” OR “Autoimmune arthritis” OR “RA”). Information about the databases used, search dates, search methods, and the number of studies retrieved is provided in the Supplementary Materials (Table S1).
2.4. Selection Process
Two independent reviewers (J.K.A. and R.T.S.) screened titles and abstracts based on the eligibility criteria. Studies with uncertain relevance were included to ensure all potentially relevant work was considered. Disagreements were resolved through discussion, with a third reviewer involved if necessary. The same process was applied to full-text reviews, with further evaluation of studies with unclear relevance at the abstract stage.
2.5. Data Collection Process
Independent reviewers (J.K.A. and R.T.S.) extracted data using a standardized form (Table S2). Afterward, a second reviewer independently verified the data to ensure all relevant information was included. Any discrepancies were resolved through discussion, with a third reviewer involved if necessary. The form captured details such as author, publication year, country, study design, patient demographics, type of chikungunya diagnosis, number of RA cases, comparators, evaluation measures, symptoms, clinical progression, and study results.
2.6. Data Items
Data were collected on the following outcomes: the incidence of RA, defined by the percentage of patients who developed RA after CHIKV infection, as diagnosed by the authors of the included studies using standard clinical and laboratory criteria; the clinical and demographic characteristics of patients, including mean age (±standard deviation), sex (male/female), country of origin, and type of chikungunya diagnosis (acute or post-exposure), as determined by the detection of anti-CHIKV IgM or IgG antibodies or viral RNA by PCR; the measures used to assess RA, such as clinical examinations, laboratory tests, and established diagnostic criteria; the clinical evolution of patients, including descriptions of symptoms, duration of arthritis, clinical progression of the disease, and response to specific treatments, when applicable; and other relevant outcomes, including any other relevant findings associated with the development of RA following CHIK infection, as reported in the included studies.
2.7. Study Risk of Bias Assessment
The risk of bias in the included studies was assessed using tools appropriate to each study design, focusing on those that confirmed CHIKV infection through serological tests (IgM/IgG anti-CHIKV) or other validated methods. Two reviewers independently evaluated the risk of bias using the Newcastle–Ottawa Scale (NOS) for cohort studies, the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for case series and observational studies, and the RoB 2.0 tool for randomized controlled trials [16,17,18,19].
3. Results
3.1. Study Selection
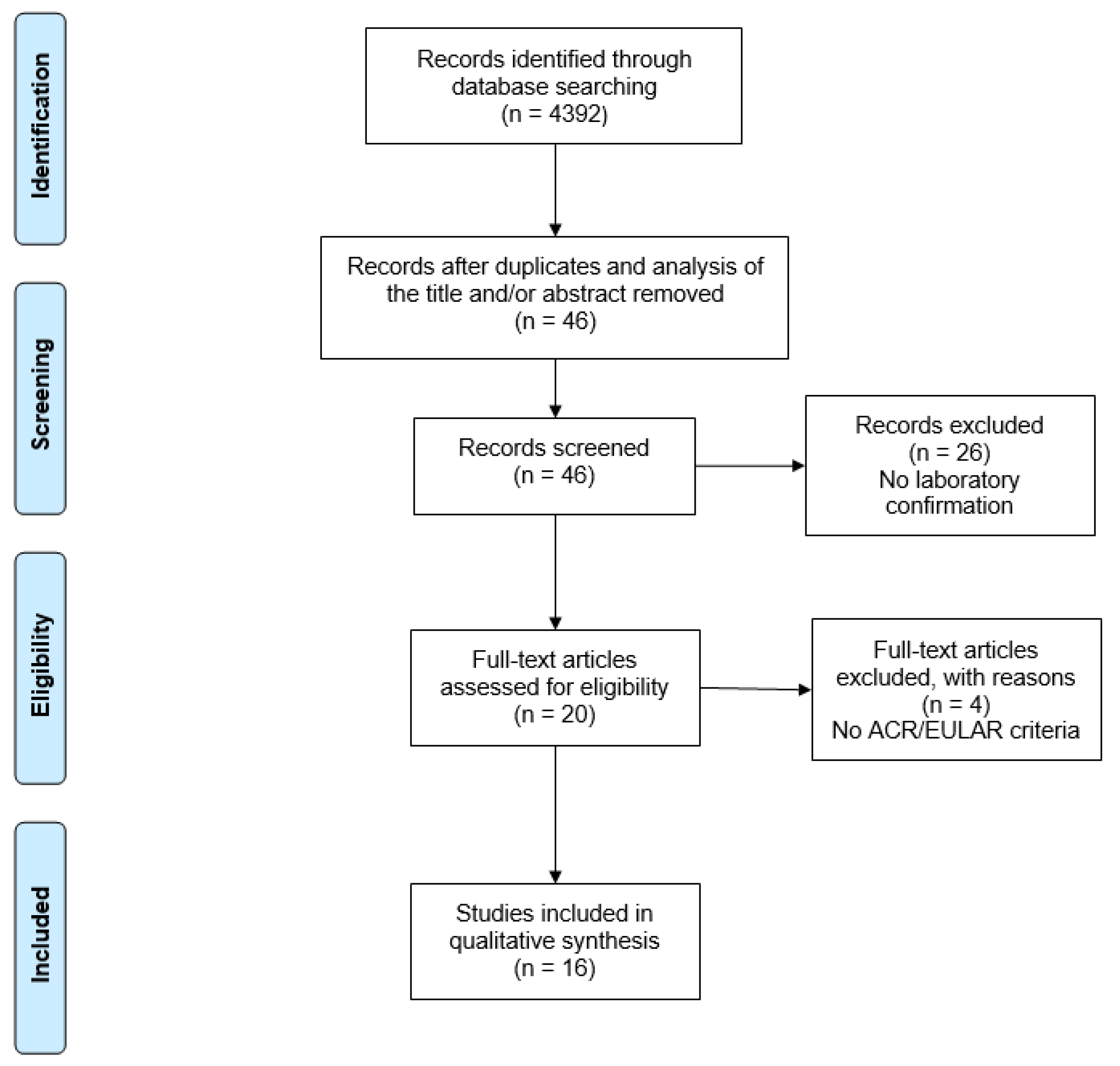
A total of 4392 studies were retrieved from the database searches. After exclusion by reading the titles and/or abstracts and duplicates, only 46 articles were selected for full reading. Sixteen studies met the eligibility criteria and were included in the systematic review (Figure 1).
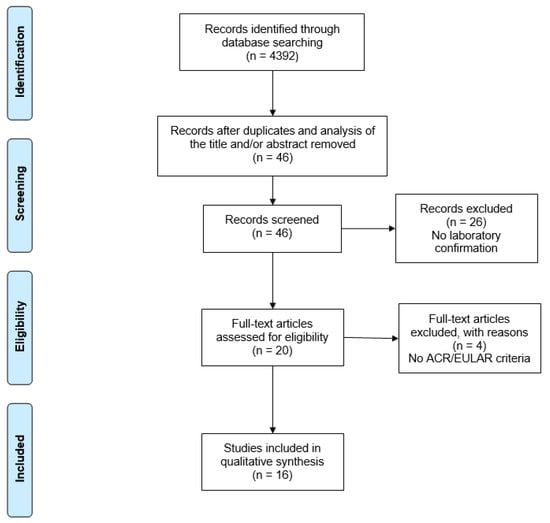
Figure 1.
PRISMA flow diagram of search results. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
3.2. Study Characteristics
The studies included in this systematic review were conducted in diverse geographical and epidemiological settings, reflecting the wide geographic distribution of CHIKV infection and its rheumatological consequences. Two cross-sectional studies were conducted in Brazil [20] and Colombia [21]. Nine cohort studies included research from Bangladesh [22], the United States [23], France [24], India [25,26,27], Colombia [28,29], and Reunion Island [30]. Four case series studies were conducted in the United States [31], France [32], Reunion Island [33], and Brazil [34]. The randomized controlled trial was conducted in India [35].
3.3. Risk of Bias in Studies
3.3.1. Cross-Sectional and Case Series Studies
Among the cross-sectional studies, both Hayd et al. [20] and Tritsch et al. [21] were rated as having a moderate risk of bias. These studies showed limitations in controlling for potential confounding factors that could affect the validity of their findings. Similarly, among the case series studies, there was variable bias risk. While some case series, such as Javelle et al. [33], were rated with low risk of bias due to their clear inclusion criteria and consistent measurement of outcomes, others, including Miner et al. [31], Bouquillard and Combe [32], and Amaral et al. [34], were assessed as having a moderate risk of bias due to concerns related to participant selection and the handling of incomplete data.
3.3.2. Cohort Studies
A significant proportion of the cohort studies (n = 7 of 9) were rated with low risk of bias. These studies, such as Pollett et al. [23], Manimunda et al. [25], Segura-Charry et al. [28], Rodriguez-Morales et al. [29], Paul et al. [26], Mathew et al. [27], and Guillot et al. [30], adhered to rigorous methodological standards, including valid and reliable measurement of exposures and outcomes, identification and control of confounding factors, and sufficient follow-up duration to capture relevant outcomes.
Two cohort studies were rated with a moderate risk of bias. Hossain et al. [22] and Bouquillard et al. [24] had incomplete follow-up and inadequate handling of confounding factors that could introduce bias into their findings.
3.3.3. Randomized Controlled Trials
The randomized controlled trial by Ravindran and Alias [35] was rated with some bias concerns using the RoB 2.0 tool. The study was an open-label design, raising potential bias in the measurement of subjective outcomes. Although the trial demonstrated strength in areas like randomization and handling of missing data, the open-label nature could have influenced participant behavior or reporting, leading to “some concerns” about the overall risk of bias.
The details are reported in the Supplementary Materials.
3.4. Results of Individual Studies
A total of 2879 patients infected by CHIKV were found in the included studies, with 449 diagnosed with RA. The studies differed by location and methods but consistently identified patients meeting RA diagnostic criteria after infection. Common symptoms included persistent joint pain, morning stiffness, polyarthralgia, joint swelling, and auto-antibodies such as rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP). Severe cases showed bone erosions, synovitis, and required advanced therapies like tumor necrosis factor (TNF) blockers and other biologics.
For example, Segura-Charry et al. [28] reported that 87 of 410 patients developed RA, with many requiring biologics therapy due to disease severity. In Guillot et al. [30], 40 of 159 patients had persistent symptoms up to 13 years post infection, highlighting the chronic nature of RA after CHIKV. Bouquillard and Combe [32] and Javelle et al. [33] demonstrated the effectiveness of methotrexate (MTX) in alleviating symptoms and slowing disease progression. Ravindran and Alias found combination therapy with MTX, sulfasalazine, and hydroxychloroquine (HCQ) more effective than HCQ monotherapy in reducing RA activity after CHIKV [35].
See Table 1 for details on each study, including diagnosed cases and clinical characteristics.

Table 1.
Characteristics and outcomes of studies evaluating rheumatoid arthritis following chikungunya virus infection.
3.5. Quality Assessment
Most of the studies included in the systematic review demonstrated good methodological quality, with low or moderate risk of bias. The rigorous application of the NOS, JBI, and RoB 2.0 bias assessment tools allowed for a clear identification of the strengths and limitations of each study.
3.6. Meta-Analysis
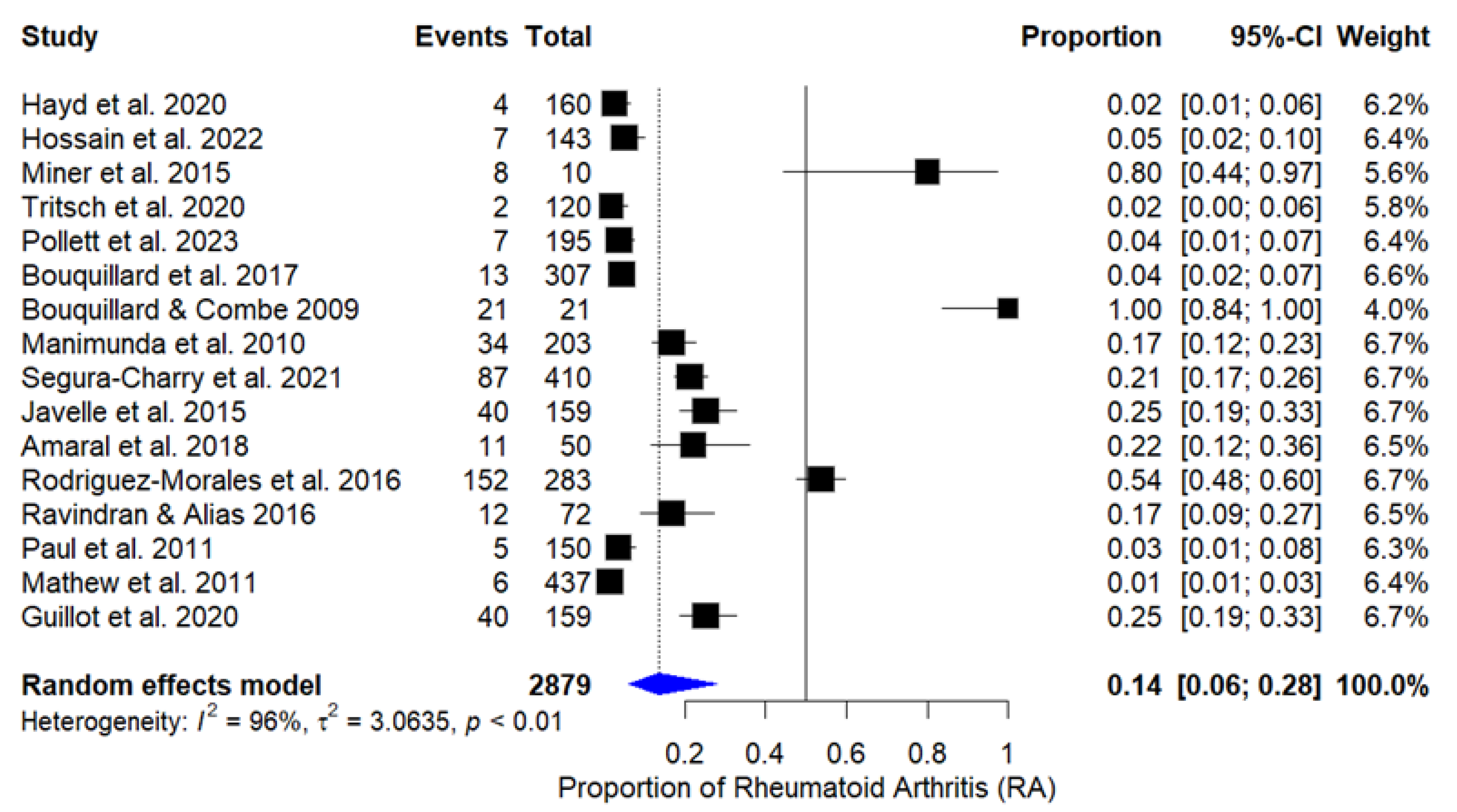
The combined proportion of patients who developed RA after CHIKV infection was 13.7% (IC 95%: 6.12% to 27.87%), based on the random effects model, due to high heterogeneity among studies (I2 = 96%, Q = 373.00, p < 0.0001). The graph in Figure 2 presents the individual estimates of RA proportion for each study, as well as the combined estimate obtained through the random effects model. After excluding cross-sectional studies and case series from the meta-analysis, heterogeneity reduced from 96% to 76.8% (Q = 33.4969, p < 0.0001), indicating better homogeneity among the remaining studies.
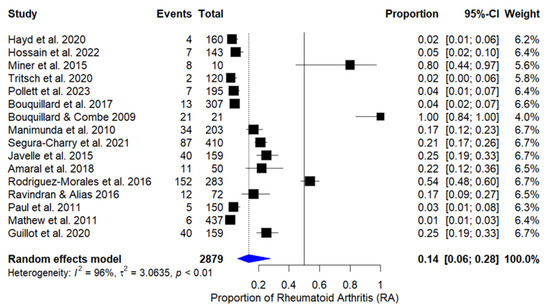
Figure 2.
Forest plot of proportion of rheumatoid arthritis in patients after chikungunya infection [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35].
The analysis only with cohort studies classified as having low risk of bias resulted in an incidence of rheumatoid arthritis of 17.7% (95% CI: 4.2–31.3%). Although heterogeneity decreased compared to the full meta-analysis, it still remained high (I2 = 81.07%).
The certainty of the evidence was assessed using the GRADE approach, considering risk of bias, inconsistency, imprecision, publication bias, and applicability of the results. For the incidence of RA post CHIKV, the certainty was considered moderate, downgraded due to inconsistency (I2 = 96%, IC 95%: 6.12–27.87%).
A forest plot showing the proportions of patients who developed rheumatoid arthritis after chikungunya infection in each study is included in the meta-analysis. The diamond at the bottom represents the combined proportion with a 95% confidence interval. The random effects model was used due to the high heterogeneity observed.
4. Discussion
This systematic review is, to our knowledge, the first to explore the relationship between CHIKV infection and the subsequent development of RA. Despite the heterogeneity of RA incidence among these studies, the meta-analysis suggests that a significant proportion of patients develop RA after CHIKV infection. However, the observed variability indicates that factors such as population characteristics, diagnostic methods, and follow-up duration may influence results. Our systematic review was restricted to studies that assessed RA as recognized in ACR-EULAR classification criteria [11]. Rodríguez-Morales et al. conducted a more general review in which they classified RA as part of a group called “post-CHIK chronic arthritis” that also included post-viral or nonspecific arthritis and seronegative spondylitis, finding a prevalence of 13.66%. However, they did not specifically evaluate RA separately [36].
Despite the variation in follow-up time among the studies in this review, we suggest that a longer period of time may be necessary to identify RA patients after CHIKV infection, as demonstrated in the study by Guillot et al. [30]. We acknowledge that the absence of a standardized observation period among the studies may have influenced the variability of the estimates, representing a limitation of this analysis.
Many CCA patients develop RA symptoms that clinically resemble classic RA, such as persistent joint pain, morning stiffness, polyarthralgia, and joint swelling [37,38,39]. But is this RA or an RA “mimic”? Many of these patients had severe disease, with joint erosions and synovitis, suggesting that CHIKV may cause the same inflammatory processes associated with RA, as described in another review [5]. The presence of auto-antibodies, such as RF and anti-CCP, also indicates that CHIKV may trigger autoimmunity, clinically indistinguishable from RA in non-CHIKV-infected individuals [8].
These clinical features are consistent with the known pathogenesis of post-CHIKV infection RA [14]. Viral persistence and molecular mimicry are possible mechanisms for the development of RA after CHIKV infection. Bouquillard et al. demonstrated that viral components can remain in synovial tissue long after acute infection, leading to chronic inflammation [32]. Additionally, Miner et al. [31] and Guillot et al. [30] report that CHIKV-induced arthritis shares immunological characteristics with idiopathic RA.
The efficacy of treatments for RA, such as MTX and biologic therapy in patients with CHIKV-induced arthritis has been documented in studies by Bouquillard and Combe [32] and Javelle et al. [33]. In these cohorts, patients with more severe manifestations required more aggressive treatment, such as TNF inhibitors, reflecting the chronic and refractory nature of post-CHIKV RA. These treatments significantly improved outcomes, reducing pain and disease activity. However, a recent clinical trial review did not demonstrate additional benefits of these drugs compared to anti-inflammatories or placebo [40].
Our review and meta-analysis included mostly observational studies, which may represent an important limitation because this type of study is more susceptible to selection bias and residual confounding. This may contribute to the high heterogeneity observed in the results (reduction of I2 from 96% to 76.8%), and the incidence estimate, which went from 16.2% to 12.4%. However, due to the nature of the topic investigated, there is a limited availability of randomized controlled trials that could reduce these biases. Another limitation that should be mentioned is the variation in sample sizes (between 10 and 437 patients), which may influence the precision of the pooled estimates.
Future studies should prioritize the standardization of diagnostic criteria and the long-term follow-up of specific subgroups in order to more accurately clarify the risk factors for the development of RA after CHIKV infection. Moreover, research focused on early interventions is essential to reduce the impact of this disease. Given the chronic and debilitating nature of CHIKV-associated RA, it is crucial to explore the mechanisms of autoimmunity triggered by the virus. For this, longitudinal studies with well-defined cohorts are fundamental, allowing not only for a better understanding of disease progression but also for the identification of biomarkers that facilitate early diagnosis. Finally, clinical trials are indispensable to assess the long-term efficacy and safety of biological therapies and combined treatments, offering new perspectives for therapeutic management.
5. Conclusions
This systematic review and meta-analysis revealed that a significant proportion of patients infected with CHIKV develop RA, with a combined estimate of 13.7% (95% CI: 6.12% to 27.87%). However, the high heterogeneity between the studies (I2 = 96%) indicates considerable variability in the findings, suggesting that factors such as differences in diagnostic criteria, study populations, and follow-up durations may have influenced the results.
These findings reinforce the need for long-term rheumatological follow-up in patients who have survived CHIKV infection, especially in endemic regions. The identification of autoimmune mechanisms associated with viral infection and the development of biomarkers for early diagnosis are important areas for future investigations.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/tropicalmed10020054/s1, Table S1: Details of risk of bias assessments based on the Newcastle–Ottawa Scale (NOS) for cohort studies; Table S2: Details of risk of bias assessments based on The Joanna Briggs Institute Critical Appraisal tool for cross-sectional analysis studies; Table S3: Details of risk of bias assessments based on The Joanna Briggs Institute Critical Appraisal tool for cohort studies; Table S4: Details of risk of bias assessments based on The Joanna Briggs Institute Critical Appraisal tool for case series; Table S5: Details of risk of bias assessments based on RoB 2.0 tool for randomized trials.
Author Contributions
Conceptualization, J.K.A.; methodology, J.K.A.; software, J.K.A.; validation, J.K.A., R.T.S., M.E.W. and E.L.C.; formal analysis, J.K.A.; investigation, J.K.A. and R.T.S.; resources, J.K.A. and M.E.W.; data curation, J.K.A.; writing—original draft preparation, J.K.A. and R.T.S.; writing—review and editing, J.K.A., R.T.S., M.E.W. and E.L.C.; visualization, J.K.A.; supervision, R.T.S. and E.L.C.; project administration, J.K.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article and Supplementary Materials. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Costa, L.B.; Barreto, F.K.A.; Barreto, M.C.A.; dos Santos, T.H.P.; Andrade, M.d.M.O.d.; Farias, L.A.B.G.; de Freitas, A.R.R.; Martinez, M.J.; Cavalcanti, L.P.d.G. Epidemiology and Economic Burden of Chikungunya: A Systematic Literature Review. Trop. Med. Infect. Dis. 2023, 8, 301. [Google Scholar] [CrossRef]
- Ferreira de Almeida, I.; Codeço, C.T.; Lana, R.M.; Bastos, L.S.; Oliveira, S.d.S.; Ferreira, D.A.d.C.; Godinho, V.B.; Riback, T.I.S.; Cruz, O.G.; Coelho, F.C. The Expansion of Chikungunya in Brazil. Lancet Reg. Health Am. 2023, 25, 100571. [Google Scholar] [CrossRef]
- Bartholomeeusen, K.; Daniel, M.; LaBeaud, D.A.; Gasque, P.; Peeling, R.W.; Stephenson, K.E.; Ng, L.F.P.; Ariën, K.K. Chikungunya Fever. Nat. Rev. Dis. Primers 2023, 9, 17, Erratum in Nat. Rev. Dis. Primers 2023, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Mourad, O.; Makhani, L.; Chen, L.H. Chikungunya: An Emerging Public Health Concern. Curr. Infect. Dis. Rep. 2022, 24, 217–228. [Google Scholar] [CrossRef]
- Amaral, J.K.; Taylor, P.C.; Schoen, R.T. Bone Erosions and Joint Damage Caused by Chikungunya Virus: A Systematic Review. Rev. Soc. Bras. Med. Trop. 2024, 57, e00404. [Google Scholar] [CrossRef] [PubMed]
- Wahid, B.; Ali, A.; Rafique, S.; Idrees, M. Global Expansion of Chikungunya Virus: Mapping the 64-Year History. Int. J. Infect. Dis. 2017, 58, 69–76. [Google Scholar] [CrossRef]
- Charrel, R.N.; de Lamballerie, X.; Raoult, D. Chikungunya Outbreaks—The Globalization of Vector-Borne Diseases. N. Engl. J. Med. 2007, 356, 769–771. [Google Scholar] [CrossRef]
- Bezerra, L.A.; da Silva Bastos, Y.C.; Gonçales, J.P.; Silva Júnior, J.V.J.; De Lorena, V.M.B.; Duarte, A.L.B.P.; Marques, C.D.L.; Coêlho, M.R.C.D. Synergism between Chikungunya Virus Infection and Rheumatoid Arthritis on Cytokine Levels: Clinical Implications? Braz. J. Microbiol. 2023, 54, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Alivernini, S.; Firestein, G.S.; McInnes, I.B. The Pathogenesis of Rheumatoid Arthritis. Immunity 2022, 55, 2255–2270. [Google Scholar] [CrossRef] [PubMed]
- Scherer, H.U.; Häupl, T.; Burmester, G.R. The Etiology of Rheumatoid Arthritis. J. Autoimmun. 2020, 110, 102400. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid Arthritis Classification Criteria: An American College of Rheumatology/European League Against Rheumatism Collaborative Initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Amaral, J.K.; Bingham, C.O., 3rd; Taylor, P.C.; Vilá, L.M.; Weinblatt, M.E.; Schoen, R.T. Pathogenesis of Chronic Chikungunya Arthritis: Resemblances and Links with Rheumatoid Arthritis. Travel Med. Infect. Dis. 2023, 52, 102534. [Google Scholar] [CrossRef] [PubMed]
- Kudaeva, F.M.; Speechley, M.R.; Pope, J.E. A Systematic Review of Viral Exposures as a Risk for Rheumatoid Arthritis. Semin. Arthritis Rheum. 2019, 48, 587–596. [Google Scholar] [CrossRef]
- Kril, V.; Aïqui-Reboul-Paviet, O.; Briant, L.; Amara, A. New Insights into Chikungunya Virus Infection and Pathogenesis. Annu. Rev. Virol. 2021, 8, 327–347. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.A.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P.; The Ottawa Hospital Research Institute. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2009. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 14 September 2024).
- Munn, Z.; Barker, T.H.; Moola, S.; Tufanaru, C.; Stern, C.; McArthur, A.; Stephenson, M.; Aromataris, E. Methodological Quality of Case Series Studies: An Introduction to the JBI Critical Appraisal Tool. JBI Evid. Synth. 2020, 18, 2127–2133. [Google Scholar] [CrossRef]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetcu, R.; Currie, M.; Qureshi, R.; Mattis, P.; Lisy, K.; et al. Chapter 7: Systematic Reviews of Etiology and Risk. In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020; Available online: https://synthesismanual.jbi.global (accessed on 14 September 2024).
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A Revised Tool for Assessing Risk of Bias in Randomised Trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Hayd, R.L.N.; Moreno, M.R.; Naveca, F.; Amdur, R.; Suchowiecki, K.; Watson, H.; Firestein, G.S.; Simon, G.; Chang, A.Y. Persistent Chikungunya Arthritis in Roraima, Brazil. Clin. Rheumatol. 2020, 39, 2781–2787. [Google Scholar] [CrossRef] [PubMed]
- Tritsch, S.R.; Encinales, L.; Pacheco, N.; Cadena, A.; Cure, C.; McMahon, E.; Watson, H.; Ramirez, A.P.; Mendoza, A.R.; Li, G.; et al. Chronic Joint Pain 3 Years After Chikungunya Virus Infection Largely Characterized by Relapsing-Remitting Symptoms. J. Rheumatol. 2020, 47, 1267–1274. [Google Scholar] [CrossRef]
- Hossain, S.; Choudhury, M.R.; Islam, M.A.; Hassan, M.; Yeasmin, S.; Hossain, F.; Zaman, M.M. Post-Chikungunya Arthritis: A Longitudinal Study in a Tertiary Care Hospital in Bangladesh. Trop. Med. Health 2022, 50, 21. [Google Scholar] [CrossRef] [PubMed]
- Pollett, S.; Hsieh, H.C.; Lu, D.; Grance, M.; Richard, S.; Nowak, G.; Lanteri, C.; Tribble, D.; Burgess, T. The Risk and Risk Factors of Chikungunya Virus Infection and Rheumatological Sequelae in a Cohort of U.S. Military Health System Beneficiaries: Implications for the Vaccine Era. PLoS Negl. Trop. Dis. 2024, 18, e0011810. [Google Scholar] [CrossRef]
- Bouquillard, E.; Fianu, A.; Bangil, M.; Charlette, N.; Ribéra, A.; Michault, A.; Favier, F.; Simon, F.; Flipo, R.-M. Rheumatic Manifestations Associated with Chikungunya Virus Infection: A Study of 307 Patients with 32-Month Follow-Up (RHUMATOCHIK Study). Jt. Bone Spine 2018, 85, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Manimunda, S.P.; Vijayachari, P.; Uppoor, R.; Sugunan, A.P.; Singh, S.S.; Rai, S.K.; Sudeep, A.B.; Muruganandam, N.; Chaitanya, I.K.; Guruprasad, D.R. Clinical Progression of Chikungunya Fever During Acute and Chronic Arthritic Stages and the Changes in Joint Morphology as Revealed by Imaging. Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.J.; Pannarkady, G.; Moni, S.P.; Thachil, E.J. Clinical Profile and Long-Term Sequelae of Chikungunya Fever. Indian J. Rheumatol. 2011, 6, 12–19. [Google Scholar] [CrossRef]
- Mathew, A.J.; Goyal, V.; George, E.; Thekkemuriyil, D.V.; Jayakumar, B.; Chopra, A.; Trivandrum COPCORD Study Group. Rheumatic-Musculoskeletal Pain and Disorders in a Naïve Group of Individuals 15 Months Following a Chikungunya Viral Epidemic in South India: A Population-Based Observational Study. Int. J. Clin. Pract. 2011, 65, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Segura-Charry, J.S.; Parada-Martinez, M.A.; Segura-Puello, H.R.; Muñoz-Forero, D.M.; Nieto-Mosquera, D.L.; Villamil-Ballesteros, A.C.; Cortés-Muñoz, A.J. Musculoskeletal Disorders Due to Chikungunya Virus: A Real Experience in a Rheumatology Department in Neiva, Huila. Reumatol. Clin. 2021, 17, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Morales, A.J.; Gil-Restrepo, A.F.; Ramírez-Jaramillo, V.; Montoya-Arias, C.P.; Acevedo-Mendoza, W.F.; Bedoya-Arias, J.E.; Chica-Quintero, L.A.; Murillo-García, D.R.; García-Robledo, J.E.; Castrillón-Spitia, J.D.; et al. Post-Chikungunya Chronic Inflammatory Rheumatism: Results from a Retrospective Follow-Up Study of 283 Adult and Child Cases in La Virginia, Risaralda, Colombia. F1000Res. 2016, 5, 360. [Google Scholar] [CrossRef]
- Guillot, X.; Ribera, A.; Gasque, P. Chikungunya-Induced Arthritis in Reunion Island: A Long-Term Observational Follow-Up Study Showing Frequently Persistent Joint Symptoms, Some Cases of Persistent Chikungunya Immunoglobulin M Positivity, and no Anticyclic Citrullinated Peptide Seroconversion After 13 Years. J. Infect. Dis. 2020, 222, 1740–1744. [Google Scholar] [CrossRef] [PubMed]
- Miner, J.J.; Aw-Yeang, H.X.; Fox, J.M.; Taffner, S.; Malkova, O.N.; Oh, S.T.; Kim, A.H.J.; Diamond, M.S.; Lenschow, D.J.; Yokoyama, W.M. Chikungunya Viral Arthritis in the United States: A Mimic of Seronegative Rheumatoid Arthritis. Arthritis Rheumatol. 2015, 67, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Bouquillard, E.; Combe, B. A Report of 21 Cases of Rheumatoid Arthritis Following Chikungunya Fever: A Mean Follow-Up of Two Years. Jt. Bone Spine 2009, 76, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Javelle, E.; Ribera, A.; Degasne, I.; Gaüzère, B.A.; Marimoutou, C.; Simon, F. Specific Management of Post-Chikungunya Rheumatic Disorders: A Retrospective Study of 159 Cases in Reunion Island from 2006–2012. PLoS Negl. Trop. Dis. 2015, 9, e0003603. [Google Scholar] [CrossRef]
- Amaral, J.K.; Bingham, C.O., 3rd; Schoen, R.T. Successful Methotrexate Treatment of Chronic Chikungunya Arthritis. J. Clin. Rheumatol. 2020, 26, 119–124. [Google Scholar] [CrossRef]
- Ravindran, V.; Alias, G. Efficacy of Combination DMARD Therapy vs. Hydroxychloroquine Monotherapy in Chronic Persistent Chikungunya Arthritis: A 24-Week Randomized Controlled Open Label Study. Clin. Rheumatol. 2017, 36, 1335–1340. [Google Scholar] [CrossRef]
- Rodríguez-Morales, A.J.; Cardona-Ospina, J.A.; Urbano-Garzón, S.F.; Hurtado-Zapata, S. Prevalence of Post-Chikungunya Infection Chronic Inflammatory Arthritis: A Systematic Review and Meta-Analysis. Arthritis Care Res. 2016, 68, 1849–1858. [Google Scholar] [CrossRef]
- Edington, F.; Varjão, D.; Melo, P. Incidence of Articular Pain and Arthritis After Chikungunya Fever in the Americas: A Systematic Review of the Literature and Meta-Analysis. Joint Bone Spine 2018, 85, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Rama, K.; de Roo, A.M.; Louwsma, T.; Hofstra, H.S.; Amaral, G.S.G.D.; Vondeling, G.T.; Postma, M.J.; Freriks, R.D. Clinical Outcomes of Chikungunya: A Systematic Literature Review and Meta-Analysis. PLoS Negl. Trop. Dis. 2024, 18, e0012254. [Google Scholar] [CrossRef] [PubMed]
- Paixão, E.S.; Rodrigues, L.C.; Costa, M.D.C.N.; Itaparica, M.; Barreto, F.; Gérardin, P.; Teixeira, M.G. Chikungunya Chronic Disease: A Systematic Review and Meta-Analysis. Trans. R. Soc. Trop. Med. Hyg. 2018, 112, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, C.; Herath, T.; Wickramarachchi, U.; Fernando, D.; Rajapakse, S. Treatment of Chikungunya-Associated Joint Pain: A Systematic Review of Controlled Clinical Trials. Trans. R. Soc. Trop. Med. Hyg. 2022, 116, 889–899. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).