Abstract
The rising demand for sustainable materials has led to a significant focus on developing resources from renewable systems, particularly through the integration of biological processes. Bacterial cellulose (BC) has emerged as a highly promising biomaterial, gaining attention across multiple industries, such as food, pharmaceuticals, materials science, and textiles, due to its renewable, biodegradable, and eco-friendly characteristics. Within the fashion industry, bacterial cellulose (BC) biofabrication presents a groundbreaking method for producing sustainable textiles and vegan leather. This systematic review emphasizes BC’s pivotal role in advancing sustainable materials, addressing challenges like low yields, strain instability, and high production costs, and exploring innovative biofabrication techniques to overcome these barriers. Current advancements aim to enhance the thickness, uniformity, and mechanical properties of BC layers by optimizing the environmental and nutritional conditions during Komagataeibacter cultivation and leveraging coculturing methods. Furthermore, recent innovations in synthetic biology and genetic engineering have opened new avenues for improving BC biosynthesis, making it a viable solution for the sustainable fashion industry. This review explores three core topics: (1) bacterial cellulose and its applications, (2) the biofabrication of BC for vegan leather, and (3) emerging innovations and patents utilizing bacterial cellulose as a sustainable industrial biomaterial.
1. Introduction
The fashion industry is a major contributor to environmental pollution, particularly through the release of microplastics from synthetic textiles [1,2]. Synthetic leather production also poses significant environmental and ethical issues [3]. Typically, synthetic leather manufactured from plastic-based materials, such as polyurethane (PU) or polyvinyl chloride (PVC) [1], are composed of long chains of synthetic polymers. These materials significantly contribute to microplastic pollution by releasing microplastic particles during use, washing, and disposal. Due to their limited biodegradability, these particles persist in the environment, leading to long-term contamination [3,4]. Utilizing, washing, and disposing of these textiles, particularly synthetic leather, have caused further environmental burdens. Studies indicate that microplastics released from these synthetic materials, along with the improper disposal of fashion items, are major contributors to this environmental impact [3].
Microplastics (MPs), defined as plastic particles smaller than 5 mm [2], represent an increasingly serious global issue due to their pervasive presence in the environment and their harmful effects on organisms. Currently, over 96% of research on MPs focuses on marine ecosystems. However, it has been confirmed that most marine MP pollution originates from land-based sources. According to various studies, both in China and internationally, microfibers from textile washing amount for approximately 35% of the microplastics detected in aquatic systems. This urgent need for greater attention to land-based sources of microplastic pollution, particularly from synthetic textiles, in efforts to mitigate their environmental impact [2,3,4,5].
Growing concerns about animal welfare and environmental sustainability have increased the demand for alternatives to animal leather [6]. This demand has driven the development of innovative materials such as vegan leather, often derived from plant-based sources and synthetic polymers [3,6]. Another option is the use of natural substances of microbial origin. One such innovative material is nanocellulose, produced by members of the Komagataeibacter genus [7]. Its unique structural properties, including a three-dimensional network and nanoscale fiber size, make BC highly absorbent and adaptable, with remarkable biocompatibility due to the absence of lignin and hemicellulose [1,8].
Unlike plant-derived cellulose, microbial cellulose holds significant potential to transform the fashion industry by providing a sustainable alternative to traditional textiles [3,9,10]. Its material properties are suitable for a wide range of applications, ranging from everyday clothing to advanced fashion technologies [3]. However, its widespread adoption is hindered by challenges related to scalability and cost-effectiveness in production [11]. Further research is required to optimize the production methods, enhance the material characteristics, and explore new applications. Moreover, consumer education and market acceptance will be crucial for the successful integration of BC into mainstream fashion [1,12].
Komagataeibacter is a well-studied microorganism for nanocellulose production, and optimizing its growth conditions can significantly improve the yield and efficiency [7,10,11]. Previous studies have examined various nutrients and environmental factors that influence the growth of K. xylinus [1,6,7,13], but comprehensive industrial-scale studies remain limited. Understanding the growth dynamics of this microorganism is essential to optimizing production processes [13,14]. Contamination during production has been a notable challenge, particularly in the nata de coco industry, where most operations in Southeast Asia are home-based and rely on conventional sterility techniques [13,15,16]. However, the use of cocultures with other microbes may provide natural nutrient supplementation, promoting the growth of K. xylinus and yielding thicker cellulose layers than monocultures [15,17]. This approach leverages microbial interactions to optimize production processes and tailor the characteristics of the cellulose sheets [11,14,15]. Bacterial cellulose has emerged as a promising biopolymer with significant potential in the textile industry, particularly as a sustainable alternative to leather [1,11]. While biofabrication is becoming a key technique for enhancing the yield and producing durable, high-quality textile materials [12], the industry currently faces numerous challenges in its transition toward more sustainable practices, including the need to improve scalability and production consistency [7,13].
BC produced by K. xylinus is a highly pure, renewable, and biodegradable material with excellent mechanical properties, including high tensile strength, flexibility, and moisture retention [12,15,18]. Unlike conventional leather, which relies on animal hides and chemical-intensive processing [3,6], BC is synthesized through a natural fermentation process, offering a low-impact and environmentally responsible option for bio-leather production [6,7]. This process involves cultivating cellulose-producing bacteria, which form a dense network of nanofibers in a gel-like structure [15,19]. This material can be processed to mimic the appearance and texture of traditional leather while offering enhanced sustainability benefits [3,12]. BC can be grown using minimal resources, such as agricultural waste, and its production does not require harmful chemicals or synthetic polymers [19,20].
Challenges remain in maintaining the homogeneity and yield of BC, particularly in industries such as fashion, where high-quality cellulose layers are essential. The thickness and durability of the cellulose layer directly impacts the quality of bio-leather produced [6,15]. Researchers have conducted numerous experiments in recent decades, concerning optimizing nutrient sources, acclimatization techniques, and bacterial propagation [1,6,7,11,18]. Recent studies have also explored by utilizing cocultures with yeast, microalgae, and other bacteria to improve fermentation processes [17,21,22]. Advances in genetic engineering are also being applied to create unique biomaterials with tailored properties, as performed by several of BC manufacture [18,21].
In this review highlights the potential for alternative methods by tailoring BC-based biofabrication as an efficient alternative resource that reduces the environmental footprint on the fashion industry, particularly on bio-leather production. Within the context of sustainable fashion, BC offers a promising alternative to conventional or synthetic leather, providing both environmental and material advantages.
2. Bacterial Cellulose and Its Application
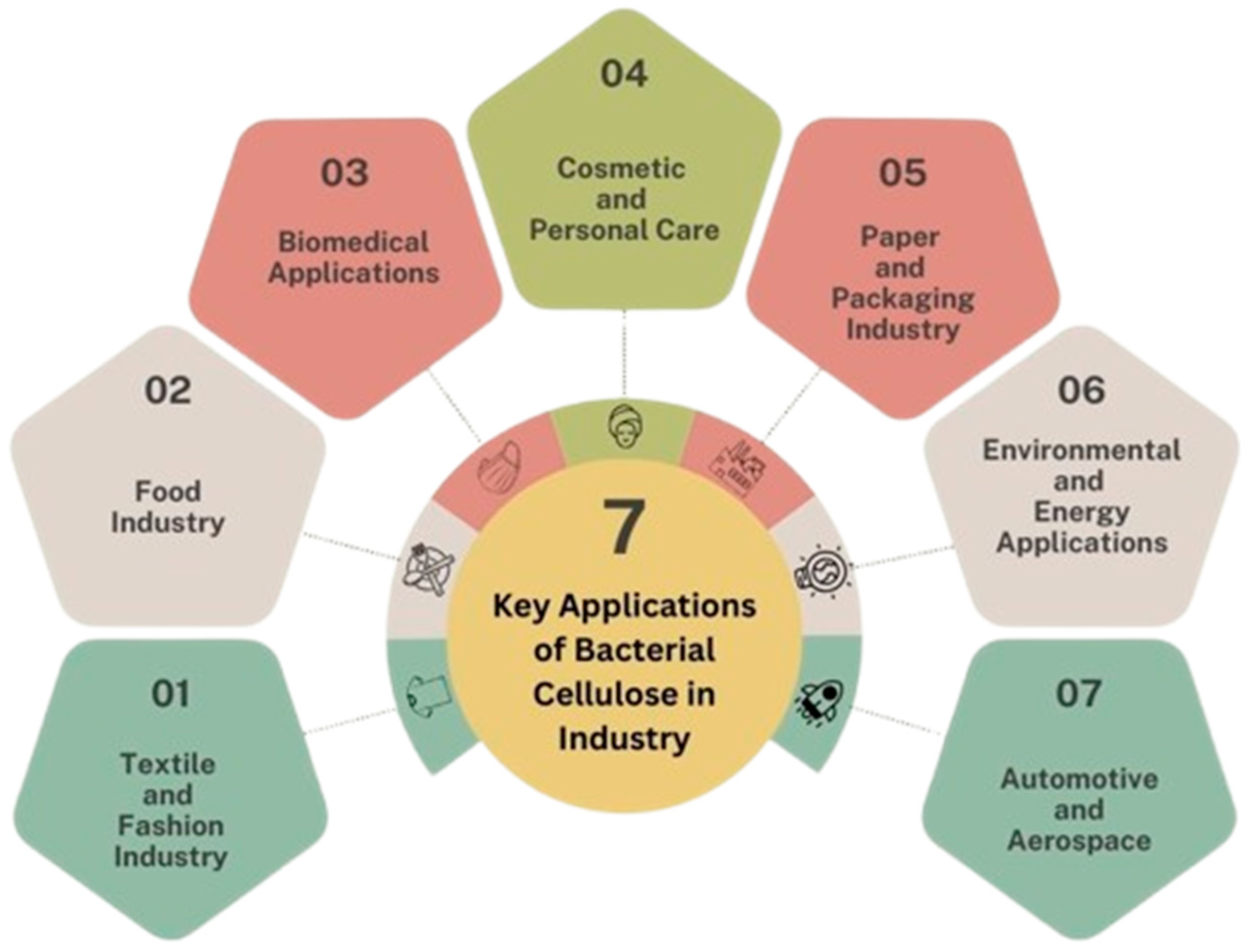
Bacterium aceti was first identified by A.J. Brown in 1886 during his experiment on acetic fermentation. Due to its distinctive fermentation yield by cellulose formation, he classified B. aceti into B. xylinum (A. xylinum) [8]. Later, Yamada and colleagues in 2012 recognized the combination phenotypic characteristic from former genera, which were Acetobacter, Gluconobacter, Asaia, Swaminathania, Saccharibacter, Neoasaia, Granulibacter, Tanticharoenia, Ameyamaea, and Neokomagataea. They proposed a new genus, Komagateibacter (gen. nov.), in honor of Dr. Kazuo Komagata, a Professor at the University of Tokyo, Bunkyo-ku, Tokyo, Japan, who made immense contributions to bacterial systematics, particularly of acetic acid bacteria [23]. In 1990, Brown and colleagues [19] successfully isolated and sequenced a gene involved in cellulose biosynthesis from A. xylinum [8,19]. This strain is widely recognized as a model organism for cellulose synthesis due to its highly crystalline structure and remarkable mechanical stability [19,24]. Moreover, the insertion of cellulose synthase genes into diverse organisms has emerged as a pioneering technique and a hallmark step in modifying cellulose formation in bacteria for tailored applications. This advancement underpins the large-scale industrial production of bacterial cellulose (BC), significantly enhancing its potential for the development of high-value, functional materials across various fields. The key industrial applications of BC are illustrated in Figure 1.
Figure 1.
Application of bacterial cellulose (BC) in industry.
2.1. Textile and Fashion Industry
Bacterial cellulose (BC) is gaining significant attention in the fashion industry as a potential alternative for conventional synthetic fibers, such as polyester and nylon, which are harmful to the environment [25]. At the same time, nanocellulose represents a step towards more sustainable and eco-conscious materials, offering extensive innovation possibilities [26].
Pioneering vegan leather companies have utilized raw materials derived from fungi as the basis for their products. Fungal bio-leathers represent an emerging class of sustainable materials produced from mycelium, the root-like network of filaments that constitutes the vegetative structure of fungi [3]. The production of fungal bio-leathers involves cultivating mycelium under controlled environmental conditions, including temperature and humidity, which influence its growth and quality [27]. Mycelium’s rapid and efficient growth process, coupled with its adaptability to various substrates, allows for scalable production methods that utilize agricultural waste, contributing to circular economy models [3,27]. Companies at the forefront of this innovation, such as MycoWorks [28], Reishi [29], MycoTech [30], and Bolt Threads [31], have pioneered the commercialization of mycelium leather, creating products such as shoes, handbags, wallets, and watch straps.
Through microbial fermentation processes, nanocellulose can be synthesized, harvested, and processed into a leather-like material [1,10,26,32]. This method requires fewer resources than traditional leather tanning or synthetic leather manufacturing [10,32]. Microbial synthesis of nanocellulose fibers, primarily by bacteria such as K. xylinus, which generates a dense and highly pure cellulose structure [10,24]. Due to its remarkable mechanical properties, BC is receiving significant attention in the fashion and material science sectors for its potential to replace traditional leather and synthetic polymers in various applications [10,26,32]. Several innovative companies are leading the charge in utilizing BC to manufacture sustainable and functional fashion products. Modern Synthesis [33], based in the UK, is pioneering the use of BC to develop next-generation bio-fabrics for footwear and apparel. This company focuses on cultivating BC composite materials and yarn-reinforced bacterial cellulose hybrid materials focusing on fashion and textile applications. Nanollose [34], an Australian biotech company, is also at the forefront of BC innovation with its creation of Nullarbor™ Tree-Free lyocell fiber, which is spun into yarn, with potential use in garments. Another notable company, ScobyTec [35] from Germany, specializes in producing Scoby leather—a 100% biodegradable and vegan leather alternative made from BC. Although no official cooperation was announced, ScobyTech showcased several sample products using their material. Similarly, the Hungarian-Estonian start-up Smobya [36] is developing NanoTwine™, a nanocellulose manufacturing platform to produce BC in a high-quality, cost-competitive way to displace animal and fossil fuel-based materials in leather-like and cosmetic applications. Figure 2 depicts the starting material of bacterial cellulose (BC) and the leather-like material produced from it.

Figure 2.
Wet BC pellicle (left side) and BC-based vegan leather (right side). Photo Credits: Smobya, Hungary [36].
2.2. Food Industry
One notable food industry project is InnoCell, [37], which emerged from the “From Peel to Peel” initiative. Originally a bachelor’s thesis exploring microbial cellulose (MC) as a sustainable packaging alternative [14,37], InnoCell now aims to increase the value of food production byproducts. It promotes the development of circular food and material systems, emphasizing upcycling and the transformation of food waste into valuable food and non-food products [14]. InnoCell’s current products include edible films and coatings, as well as biodegradable food packaging.
Bandyopadhyay et al. [38] investigated the cross-linking of BC and guar gum (GG)-based hydrogel films, modified with polyvinyl pyrrolidone–carboxymethyl cellulose (PVP-CMC), designed for fresh berry packaging. The films were evaluated for various properties including their mechanical strength, barrier capability, bioadhesion, color, gloss, rheological behavior, surface hydrophobicity, permeability, water solubility, biodegradability, and effectiveness in extending the shelf life of berries. Their findings demonstrated that incorporating GG into the PVP-CMC-BC films enhanced their elasticity, load-bearing capacity, and barrier properties. These biofilms also exhibited superior hydrophobic qualities compared to the other films tested. Importantly, all biofilms showed 80% biodegradability within 28 days in vermicompost, positioning them as a promising biodegradable alternative to traditional packaging materials.
Cazón et al. [39] tested biodegradable films made from bacterial cellulose, glycerol, and polyvinyl alcohol (PVOH) for their mechanical properties, water vapor permeability, and UV-protective characteristics. The results showed that the composite films exhibited high elongation (up to 49.89%) and good rupture resistance (13.78 MPa). The films showed a UV transmittance as low as 5.59%, 2.4%, and 0.57% in the UV-A, UV-B, and UV-C ranges, respectively. The addition of PVOH reinforced the BC matrix, while glycerol acted as a plasticizer.
Several companies and research initiatives are developing BC for food-related applications, such as Cellugy in Denmark [40] and publicly owned research institutes such as VTT Technical Research Centre in Finland [41] and Fraunhofer Society in Germany [42], all of which are actively investigating BC’s potential in food applications.
2.3. Biomedical Application
BC has also garnered significant attention in fields such as wound care, tissue and organ regeneration tailored with 3D bioprinting, bone regeneration, drug delivery for anti-cancer therapies and antibiotic delivery, and medical implants [19,32,43].
Maurer et al. [44] assessed the effectiveness of BC dressings for treating pediatric thermal injuries, analyzing clinical outcomes such as dressing changes, hospital stays, and complications. The average length of hospital stay was 6.7 days, and children underwent an average of 2.4 dressing changes under anesthesia. The wound infection rate was low, with none of the children developing an infection. Approximately 82% of children were treated exclusively with BC-based dressings, and most patients indicated good epithelialization after 10 days. Most patients exhibited favorable outcomes in terms of scar formation, with 98% showing normal pigmentation, and high rates of adequate vascularization (91%), height (92%), and pliability (92%). Recent findings by Cañas-Gutiérrez et al. [45] investigated the biomineralization of BC scaffolds designed for bone tissue engineering. The study revealed significant findings regarding the mineralization process, including the deposition of calcium phosphates (CPs) onto the BC scaffolds over five immersion cycles. These cycles involved alternating between calcium and phosphate salts. The results showed that the crystals formed on the BC surface were rod-shaped and like the calcium phosphate composition of immature bone. As the number of cycles increased, the crystals’ calcium phosphate ratio grew from 1.13 to 1.6, and their size expanded from 25.12 nm to 35.9 nm. Furthermore, in vitro results demonstrated that the scaffolds supported high cell viability (up to 95%) and promoted the osteogenic differentiation of human bone marrow mesenchymal stem cells, indicated by the expression of bone-related markers like alkaline phosphatase, osteocalcin, and osteopontin.
Notable companies and research institutes are developing biomedical applications of BC, such as BioModics (Denmark) [46], which is working on BC wound dressings suitable for advanced wound care. BC is also being explored for drug delivery systems, as demonstrated by the research on controlled antibiotic release for wound infections and implant-associated infections. In the public sector, the VTT Technical Research Centre (Finland) [41], a leading research institution, is exploring BC for 3D bioprinting, tissue engineering, and drug delivery applications in the biomedical field.
2.4. Cosmetic and Personal Care
The unique properties of bacterial cellulose (BC) have positioned it as an innovative ingredient in creams, lotions, and face masks aimed at improving skin texture and maintaining a supple, moisturized complexion [19,32,41]. Amorim et al. [47] developed a polymer-based mask designed for acne-prone skin. This BioMask incorporates BC as the primary material due to its biocompatibility, hypoallergenic properties, and high-water retention, making it particularly effective for skin hydration and inflammation reduction. The BioMask also contains a 2% propolis extract, chosen for its polyphenols and flavonoids, which have proven anti-inflammatory and healing properties. A characterization of the BC film was conducted using X-ray diffraction (XRD) and thermogravimetric analysis (TGA), confirming the stability and potential efficacy of the propolis-infused BC film as a cosmeceutical product. This mask is expected to improve skin hydration, promote faster healing, and enhance skin texture, offering a non-invasive solution for managing acne symptoms.
In another study, Fonseca et al. [48] discovered the development of microneedle patches made from BC combined with hyaluronic acid (HA). BC provides a strong, biocompatible scaffold with high mechanical strength, which makes it suitable for fabricating microneedles that can penetrate the skin. Meanwhile, hyaluronic acid, known for its hydration and biocompatibility, enhances the skin’s moisture content and is beneficial for therapeutic skin applications. In vitro evaluations of these microneedles on skin models demonstrated good skin penetration, a controlled release of bioactive compounds, and excellent biocompatibility. Then, based on in vivo evaluations, animal model studies further confirmed that the patches were safe and effective for delivering therapeutic agents. The research highlights the potential of BC-HA microneedle patches as a promising solution for minimally invasive skin treatments with enhanced efficacy and minimal side effects.
In a recent study, Kucuk et al. [49] examined the impact of a facial serum combining BC with herbal extracts and bioactive compounds on hyperpigmentation. Key ingredients, such as licorice root, white mulberry, and ascorbic acid, inhibited tyrosinase activity, thereby reducing melanin production and hyperpigmentation. The serum also promoted collagenase activity and showed no cytotoxic effects, making it suitable for cosmetic use and demonstrating effectiveness in skin-brightening applications.
As major cosmetic companies and research institutions increasingly invest in BC, its role in the industry is poised to expand. Companies like L’Oréal and Procter & Gamble are exploring BC for both product formulations and packaging, and companies like Cellugy [40] are leveraging BC for its hydrating and film-forming properties. Furthermore, research institutions, such as the University of Tokyo [50] and the Fraunhofer Society [42], are also advancing the applications of BC, paving the way for more sophisticated and sustainable personal care solutions. As the industry continues to evolve, BC stands out as a promising ingredient that combines performance with environmental consciousness, driving innovation in cosmetic and personal care products [19,32,43].
2.5. Paper and Packaging Industry
In the paper and packaging industry, BC also shows potential as a sustainable material. Chemically like plant cellulose but immensely finer in structure, BC provides strong inter-fiber hydrogen bonding, making it ideal for enhancing paper strength and durability. This improvement can increase the paper’s resistance to wear and tear [19,32,51].
In another interesting experiment, Soares da Silva et al. [52] explored the combination of BC with polyhydroxybutyrate-valerate (PHBV) to enhance its barrier properties and maintain its eco-friendliness. The composite was plasticized using glycerol or polyethylene glycol to improve its flexibility, then coated with PHBV to reduce water vapor permeability and increase hydrophobicity. This combination improved the mechanical strength of BC with poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV), aiming to produce a biodegradable food packaging that requires moisture protection, such as for meat products. The resulting material exhibited a significant reduction in water vapor transmission, from 0.990 to 0.032 g/m2·day·Pa, offering a biodegradable alternative to conventional plastics used in food packaging. Companies like Ecovative Design [53] and Cellugy [40] are at the forefront of integrating BC into packaging solutions, driving eco-friendly alternatives that contribute to a circular economy.
In other research related to the enhancement of the quality of paper, Tabarsa et al. [54] investigated whether BC could improve the tensile strength and flexibility of paper made from softwood pulp. The study also examined the optimal BC content, including reinforcement (60%). BC improved the tensile strength and flexibility of the paper with an increment of water retention (20%), without significantly adding to its weight. Furthermore, Lourenço and colleagues [55] investigated the role of BC in improving the properties of fine paper, both as an internal additive and a coating agent. BC, due to its strength, high surface area, and film-forming capabilities, was found to enhance paper quality, particularly its printability. The application of a 50:50 BC coating to uncoated office paper significantly improved color reproduction, as indicated by a more than 25% increase in the color gamut area, compared to untreated paper. These findings suggest that BC could be a valuable material for producing high-quality, eco-friendly fine papers for printing and other applications requiring a high-resolution output.
Table 1 highlights the advantages of BC over plant-based cellulose, particularly in terms of its finer structure, durability, and adaptability to innovative, eco-friendly applications.

Table 1.
Comparison features in between BC and plant-based cellulose.
2.6. Environmental and Energy Sector
BC-based materials have shown potential for use in energy storage devices such as advanced batteries and supercapacitors. Lokhande et al. [56] and Ma et al. [57] performed extensive research related to BC for separators, binders, and electrode materials (in its carbonized form) in supercapacitors and lithium-ion batteries. Through tuning BC into various fiber, layered, or porous forms, subsequent studies could enhance ion transport and storage capacity by up to 20–30%. The formation of micropores and mesopore structures in electrodes also increases electrode–electrolyte contact, boosting overall conductivity and energy density. BC-based materials are thus able to increase the energy storage capacity and improve device stability. Other environmentally friendly applications have been developed by Ecovative Design (Green Island, USA) [53], which focuses on using BC for creating eco-friendly alternatives to conventional materials, including energy-efficient insulation and filtration systems.
2.7. Automotive and Aerospace Sector
Aisyah et al. [58] and Nurazzi et al. [59] reviewed the use of nanocellulose composites to reinforce polymeric matrices, improving their mechanical and thermal properties. BC composites, when combined with synthetic nanofillers like carbon nanotubes or graphene, exhibit enhanced stiffness, strength, and thermal stability, all of which are crucial for reducing vehicle weight and improving fuel efficiency in the automotive industry. These composites also offer excellent thermal stability, flexibility, and low thermal expansion, making them suitable for use in aerospace applications where high-temperature resistance is essential. BC composites are also being explored for their corrosion resistance, and, in automotive applications, BC is being used in components such as bumpers and foam pads, contributing to energy absorption and crash resistance.
Several companies, including Toyota Motor Corporation (Toyota City, Japan) [60], Boeing (Everett, WA, USA), and Airbus (Toulouse, France) [61], are integrating nanocellulose into automotive and aerospace technologies. These companies, alongside research institutions like the Georgia Institute of Technology [62], are driving forward the adoption of BC in automotive and aerospace applications, highlighting its transformative potential [61,62].
However, challenges related to the hydrophilic nature of nanocellulose and its dispersion in polymer matrices remain, but advances in processing technologies are expected to overcome these issues and further expand the use of nanocellulose composites.
3. Biofabrication of BC on Vegan Leather Industry
The biofabrication of BC in vegan leather synthesis represents an innovative and sustainable approach. It utilizes biodegradable waste materials as carbon sources for BC cultivation, allowing for natural decomposition by microorganisms after disposal. This process collaborates with eco-friendly tanning methods, reducing the impact of chemical waste pollution [10,12,63,64].
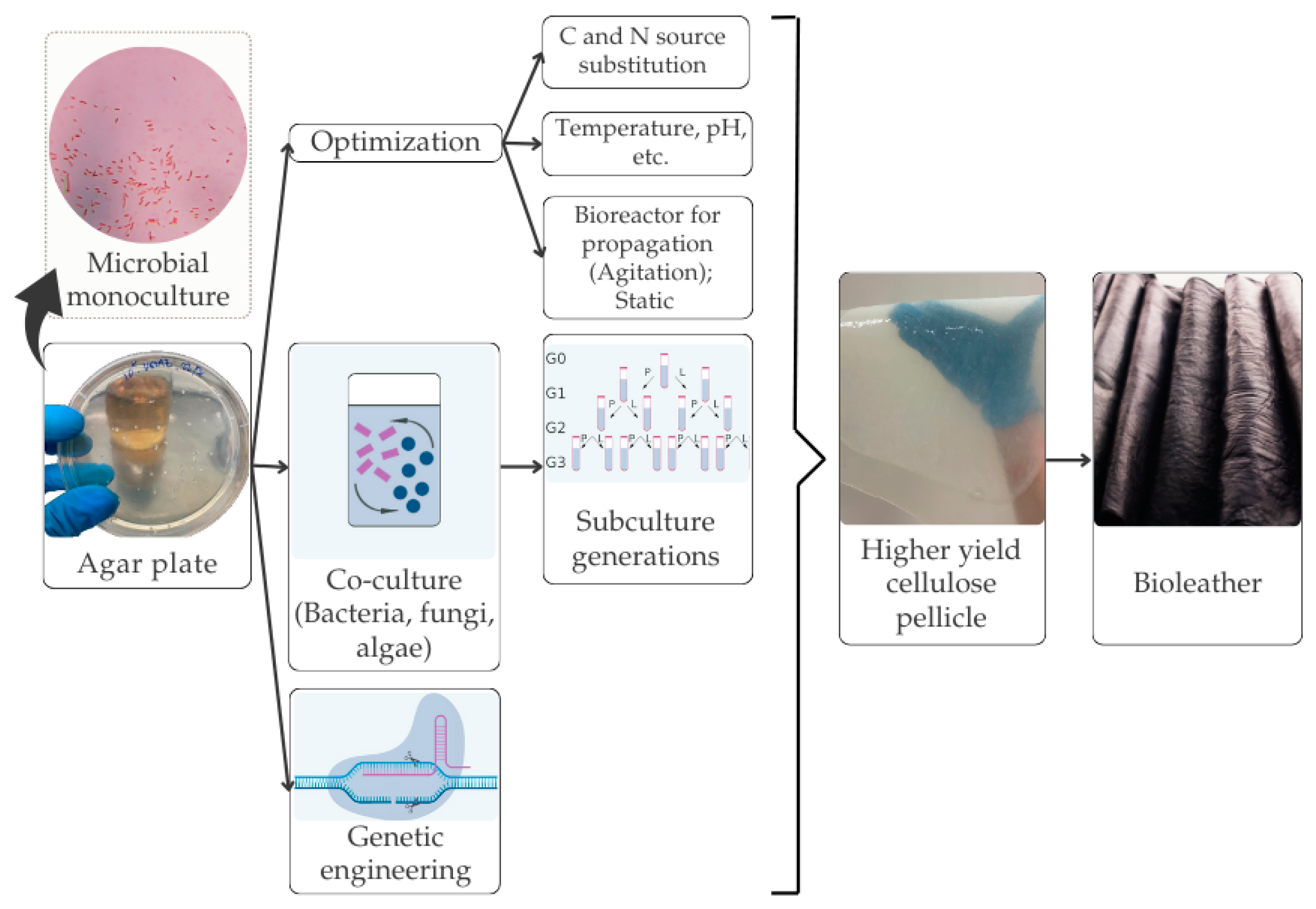
In a previous study, Yang et al. [63] employed waste coconut water and natural dyes from sources such as indigo, carrot peel, grape skin, and coffee grounds to create vegan leather. The production cost for these vegan leather shoes was approximately $97.92/m2 (excluding labor and operational costs), while the raw material cost was around $7.52/m2, while the commercial cowhide cost ranges from $39.70 to $220.80/m2. Additionally, BC vegan leather crosslinked with oxidized edible oil demonstrated a remarkable tensile strength of 82.14 MPa, surpassing that of traditional cow leather for shoe uppers by 2.08 times. When plasticized with glycerol, the tensile strength reached 56.19 MPa, with an elongation at break of 36%. These properties make BC vegan leather an attractive eco-friendly alternative to conventional leather manufacturing. The workflow for the biofabrication of BC, from cultivation to the production of vegan leather, is schematically depicted in Figure 3.
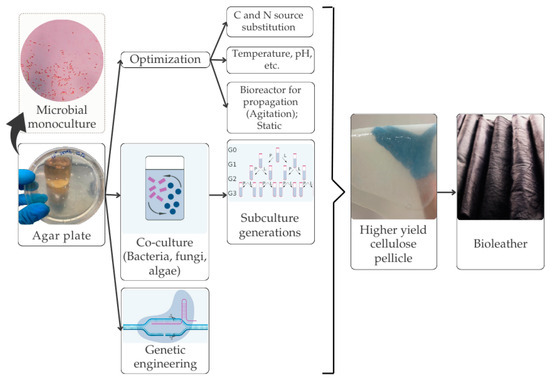
Figure 3.
Biofabrication of BC for vegan leather products.
The biofabrication of BC involves cultivating cellulose-producing bacteria in nutrient-rich media, typically containing glucose or other sugars as carbon sources [65]. Bacteria from the Komagataeibacter genus could synthesize cellulose from a wide range of carbon sources, including monosaccharides, disaccharides, oligosaccharides, and sugar alcohols [10,65,66]. Under both static and agitated conditions, these bacteria produce cellulose fibers that form a hydrogel-like matrix at the air–liquid interface. This nanocellulose can then be harvested, purified, and processed into various forms, such as films, sheets, or fibers, for multiple applications [67]. Extensive optimization efforts have focused on areas such as inoculum density, temperature, pH, agitation, and types of carbon and nitrogen sources to cultivate the bacteria effectively [65,66,68,69]. Additional factors, such as light, humidity, incubation period, and BC treatment method (static or agitated), significantly impact the cellulose growth and yield [68]. Studies have examined the effects of various carbon and nitrogen sources, as well as the initial pH, on the BC yield and its properties [10,68,69]. Glucose remains the most used carbon source for cultivating cellulose-producing bacteria [66,70]. Maintaining an optimal pH range is essential to prevent gluconic acid accumulation, which can inhibit cellulose production [66]. Optimizing the fermentation process is critical to improving the BC yield and reducing production costs [70]. Generally, fermentation processes have been optimized by varying one independent factor at a time, while holding others constant [65,68]. However, at an industrial scale, the complexity of interacting variables requires more sophisticated optimization approaches, as conventional methods often fail to capture intricate relationships affecting BC production [11]. Consequently, this limits the effectiveness of scaling production rates and achieving high cellulose yields.
Coculture-based biofabrication of BC is another innovative approach, enhancing cellulose production by combining bacterial strains or microbial communities. Coculturing promotes synergistic interactions, improving the yield, structure, and functional properties of nanocellulose [15,22,71,72]. For example, coculturing Komagataeibacter with other cellulose- or non-cellulose-producing microbes can increase the cellulose output or produce bio-leather that mimics traditional leather in appearance, texture, and durability [73]. A previous study demonstrated that coculturing K. hansenii, a well-known BC producer, with A. pullulans, a producer of the polysaccharide pullulan, significantly improved the mechanical properties of BC. Optimized conditions led to a 22.4% increase in BC production and a 4.5- to 6-fold improvement in elastic modulus compared to baseline conditions in both shake-flask and bioreactor systems [22,74]. Similarly, another study reported an increase in BC production from 2.5 to 13.8 g/L by coculturing K. nataicola with L. fermentum [15]. The coculture approaches summarized in Table 2 highlight significant advancements in bacterial cellulose (BC) production through various strain consortia, substrate utilization, and additive applications. Among these pairings, the most effective coculture was K. nataicola Q2 and L. fermentum SR, yielding both the highest BC production and improved crystallinity. Lignocellulosic substrates and pullulan additives demonstrated their value in enhancing sustainability and yield, respectively. These findings underscore the importance of strain compatibility, substrate selection, and additive usage in maximizing BC production. Coculture fermentation holds potential for (a) increased product yields, (b) the synthesis of novel compounds, and (c) metabolic symbiosis [74,75]. However, maintaining microbial community stability poses challenges, as cocultivated microorganisms often require distinct growth conditions and media.

Table 2.
Studies on coculture approaches to improve bacterial cellulose production in the last 5 years.
Research into sub-cultured generations examines the consistency of BC production across multiple sub-cultured generations, focusing on whether repeated culturing and transfer to fresh media impact cellulose quality, fiber structure, or yield [74]. This research emphasized the importance of preserving bacterial strains capable of producing nanofibers with desirable properties, in terms of thickness, length, and strength, which are critical for evaluating the scalability of BC production in the fashion industry.
The bioindustry of BC is often constrained by low production yields, microbial strain instability, and high culture media costs, all of which hinder its scalability for large-scale manufacturing [12,32]. Previous studies have shown that engineered K. xylinus strains can produce thicker BC films than wild-type strains, with engineered strains achieving film thicknesses between 6.2 and 10.2 μm compared to 5.1 μm in wild-type strains [76]. Recently, Jang et al. [77] reported that the heterologous overexpression of the pgi gene in the K. xylinus 2325 strain led to an 115% increase in the BC yield, reaching approximately 3.15 g/L, compared to the 1.46 g/L yield of the wild-type strain. However, further research is critical to achieving the technological advancements required for large-scale production and commercialization. As shown in Table 3, improvements in BC production—such as enhanced thickness and crystallinity—have been achieved through gene overexpression, the inhibition of gluconic acid (a byproduct that can limit cellulose yield), and optimized oxygen availability to maintain essential oxygen levels for large-scale BC production. Recent developments in synthetic biology and CRISPR interference technologies have enabled the precise modulation of BC biosynthesis, resulting in BC-based materials with improved characteristics [78,79]. Additionally, genetic toolkits and CRISPR-based approaches, which have shown success in other microorganisms, hold potential for overcoming current challenges in BC production, including unrevealed gene functions.

Table 3.
Genetic modifications of cellulose-producing bacteria.
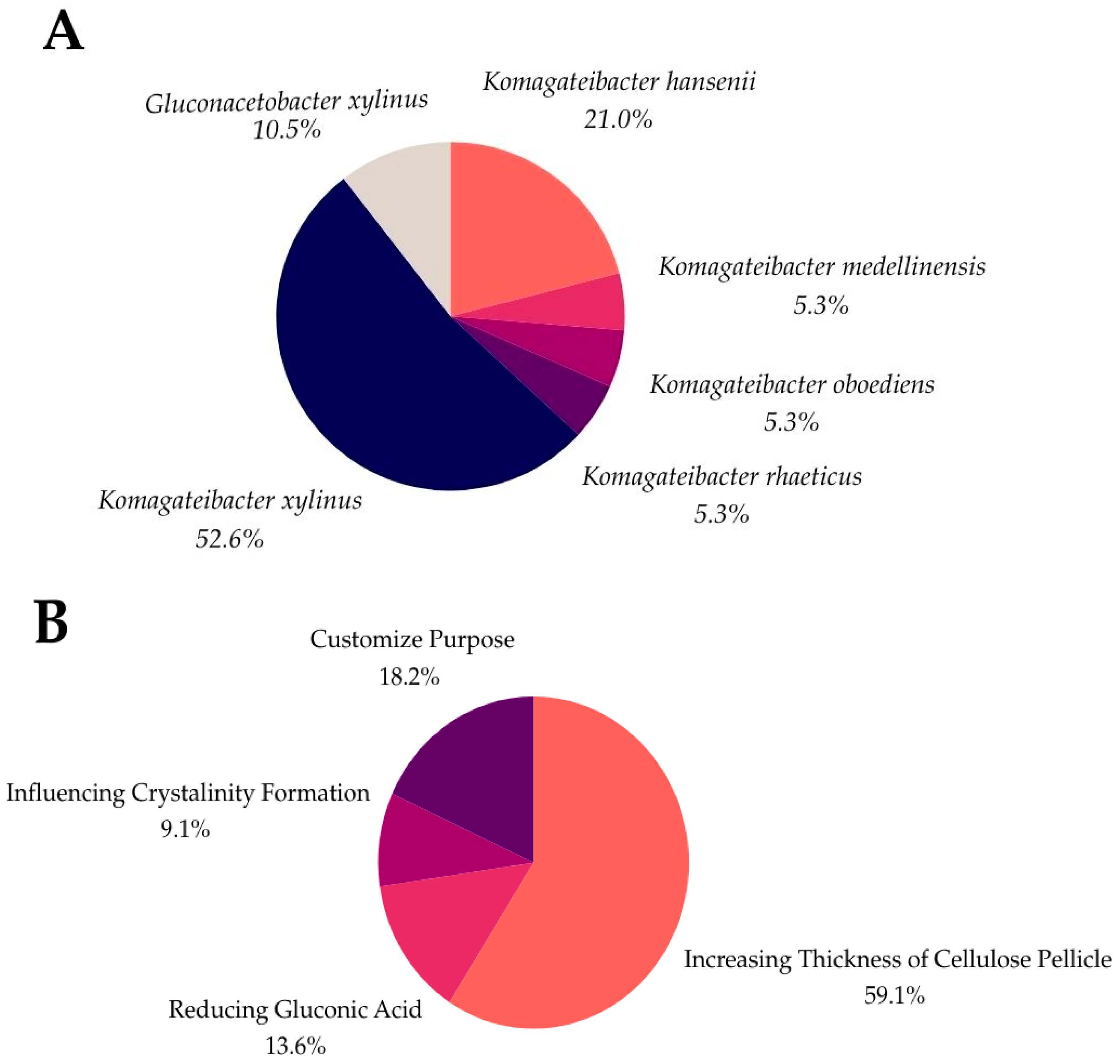
As shown in Figure 4, over 50% of genetic modifications are concerned with enhancing the thickness of the cellulose pellicle, addressing a critical industry challenge in meeting consumer demands. Additionally, 13.6% of these genetic modifications aim to reduce gluconic acid formation, leading to increased BC production. Other modifications target improvements in the crystallinity, which influences the durability and robustness of the cellulose. Another biofabrication strategy involves the integration of the tyrosinase enzyme, which allows for in situ dyeing by catalyzing the oxidation of phenolic compounds to generate color [94]. Although this self-dyeing process presents technical challenges, such as regulating the color intensity and ensuring a uniform dye distribution, ongoing research is working to overcome these obstacles for broader applications.
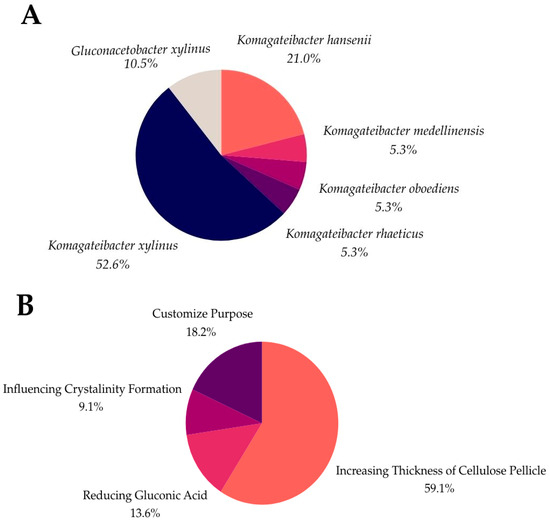
Figure 4.
Strains of BC (A) and genetic modification purposes in BC (B).
Further advancements in BC biofabrication highlight its versatility for sustainable design. The integration of materials such as mesh or woolen yarn during growth enables the creation of complex textures and hybrid forms. Post-cultivation treatments, including dyeing and laser cutting, further enhance functionality. Optimized conditions, such as a six-day growth period with a 15 g/L tea concentration, allow for the efficient production of 2D sheets and seamless 3D forms, minimizing waste and exemplifying sustainable innovation in modern fashion [95].
Another innovative project, Ava, demonstrates the fusion of traditional craftsmanship and modern technology. By leveraging origami-inspired folds, it overcomes BC’s rigidity and incorporates LEDs and conductive threads for interactive designs. This approach addresses limitations such as water resistance and rigidity, while also showcasing BC’s potential for avant-garde fashion and sculptural accessories. The emphasis on adaptability, minimal environmental impact, and artistic expression positions microbial 3D fiber art as a pioneering field in sustainable and futuristic design [96].
BC, a biopolymer produced through microbial fermentation, offers high tensile strength, biocompatibility, and customizable properties, making it highly suitable for integration into 3D printing processes. In a study on 3D biofabrication, Polewka et al. [97] explored the use of 3D printing technologies with biomaterials as sustainable alternatives for textile and fashion production. Their research highlights how additive manufacturing (AM) techniques, such as fused filament fabrication (FFF), utilize bio-based materials like polylactic acid (PLA), cellulose, and lignin, which are renewable and biodegradable resources. These materials can be employed to produce textile-like structures, garments, and prototypes with minimal waste, contributing significantly to the eco-friendliness of fashion manufacturing.
The combination of BC and 3D printing represents a transformative approach in fashion, blending biofabrication with cutting-edge technology to create eco-friendly, customizable garments and textile products. Wang et al. [98] investigated cellulose nanofibrils (CNFs)-reinforced polylactic acid (PLA) biocomposite filaments for fused deposition modeling (FDM) 3D printing. CNFs, prepared through enzymatic hydrolysis and high-pressure homogenization, were integrated into PLA to improve the composite’s mechanical, thermal, and water absorption properties. Similar to CNFs, BC could provide analogous benefits while potentially expanding functionality. Its hydrophilic nature supports post-processing, such as dyeing, and its ability to integrate into biodegradable matrices aligns with the emphasis on sustainability and environmental consciousness in consumer products. Both materials represent promising paths for eco-friendly solutions in 3D biofabrication, particularly for textiles and apparel, where customization, functionality, and sustainability are paramount.
Data that were collected from previous studies indicate that K. xylinus is the most used strain for genetic modification (52.6%), followed by K. hansenii (21.0%) and G. xylinus (10.5%). Less commonly used strains for genetic engineering are K. rhaeticus, K. oboediens, and K. medellinensis at 5.3%, respectively.
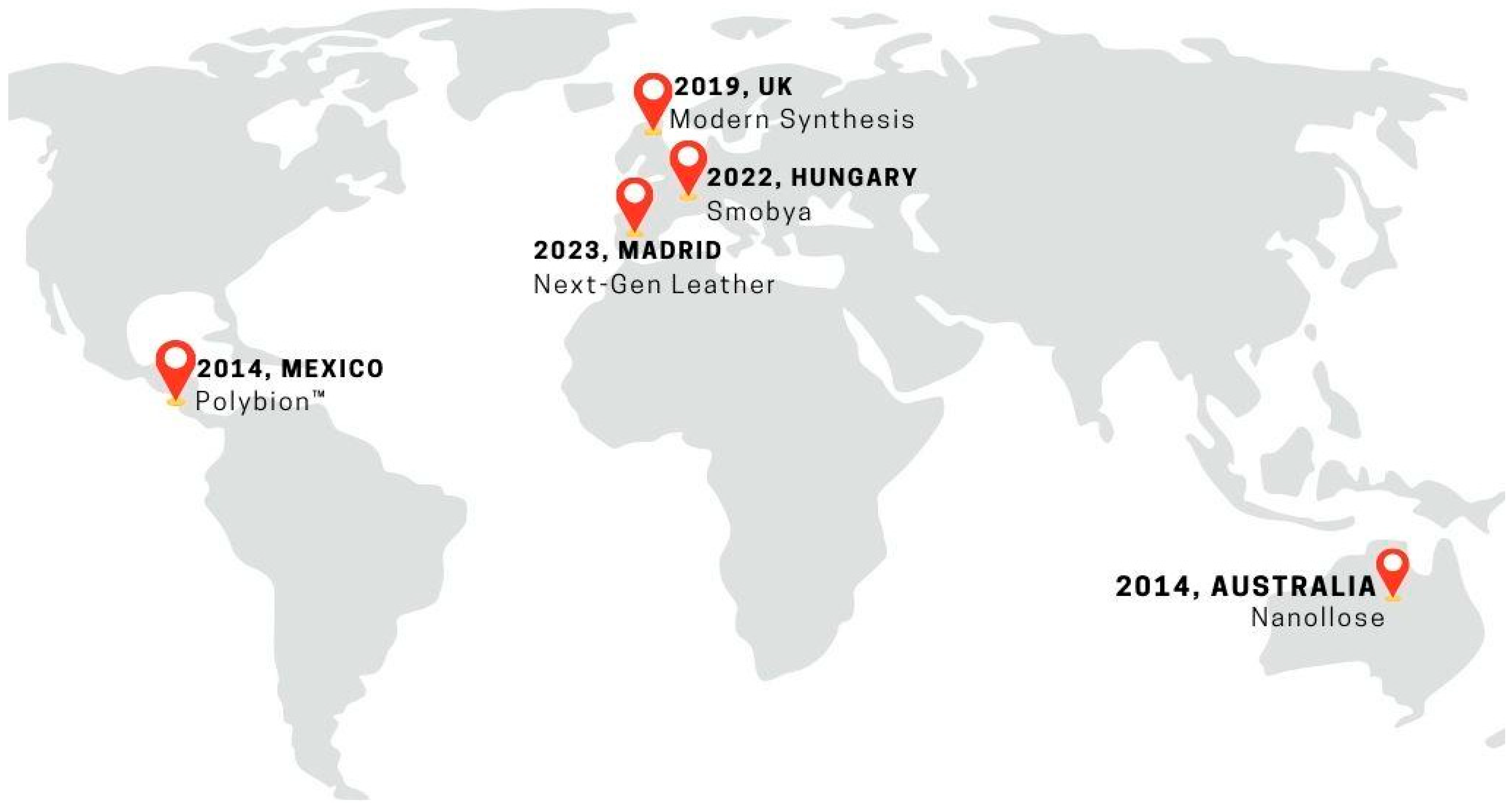
These companies are leading the advancement of sustainable fashion by using Komagataeibacter strains, renowned for their high cellulose yield, in the production of vegan leather. This innovative approach emphasizes the potential of BC as a viable and eco-friendly alternative to traditional leather materials within the fashion industry (Figure 5).
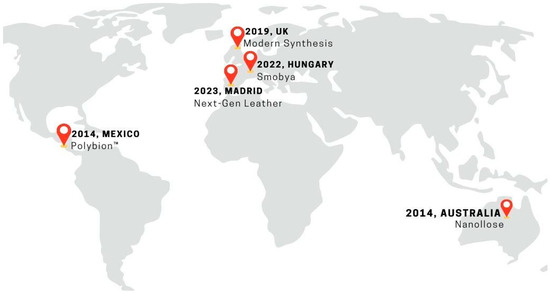
Figure 5.
Forefront of biotechnology companies utilizing biofabrication of bacterial nanocellulose for the vegan leather industry.
4. Patent Search
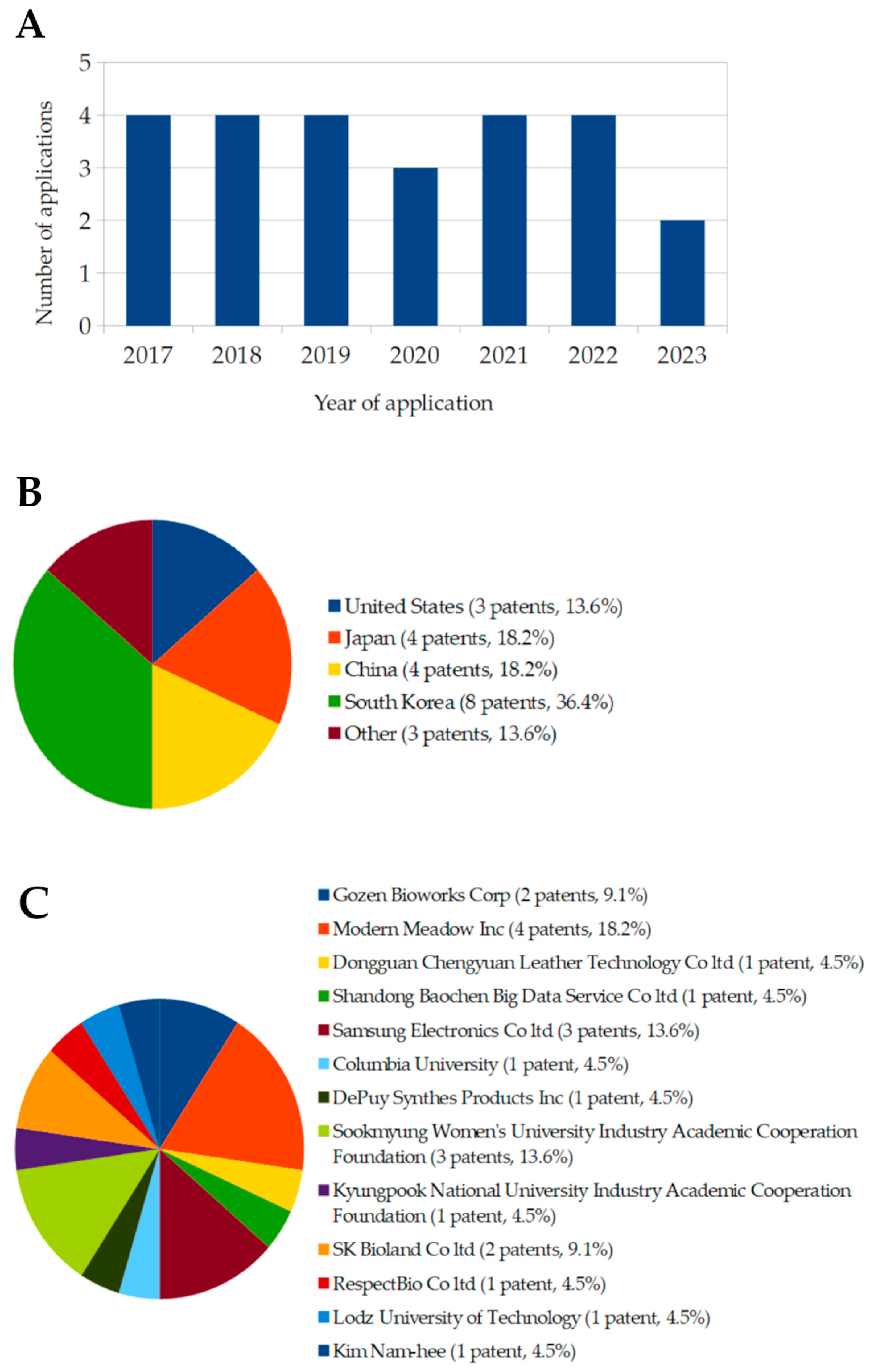
A patent search was conducted using the European Espacenet database, with further verification through Google Patents. The search employed keywords such as “Komagataibacter”, “bacterial cellulose”, “leather”, “sustainable textile”, and “biofabrication”, resulting in 23 published patents from 2017 to 2023 (Table 4). Patents in this domain are geographically diverse, although the number of applications has declined since 2017 (Figure 6A). South Korea holds the largest share of patents (36.4%), followed by China and Japan (18.2% each), with additional contributions from the United States, Poland, and Turkey (Figure 6B). Modern Meadow Inc. leads in patent publications, holding 18.2% of the patents, followed by Samsung Electronics Co., and Sookmyung Women’s University Industry-Academic Cooperation Foundation (13.6% each). Other notable contributors include Gozen Bioworks Corp. and SK Bioland Co. (9.1% each), along with Dongguan Chengyuan Leather Technology Co., Ltd., Shandong Baochen Big Data Services Co., Ltd., Columbia University, DePuy Synthes Product Inc., RespectBio Co., Ltd., Kyungpook National University Industry-Academic Cooperation Foundation, and Song Ji-eun et al. (4.5% each) (Figure 6C). Figure 6 illustrates the fluctuation in the number of registrations and publications of applications utilizing bacterial cellulose (BC) for biofabrication purposes between 2017 and 2023. While the data initially show a consistent high trend from 2017 to 2019, with four applications each year, there was a noticeable decline starting in 2020, where the number dropped to three applications. This downward trend continued in 2022 and 2023. Based on this observed pattern, it is projected that this decreasing trend may persist in subsequent years, potentially reflecting challenges in the development and commercialization of BC-based biofabrication technologies.

Table 4.
List of patents related to BC-based application and patent ownership from 2017 to 2023.
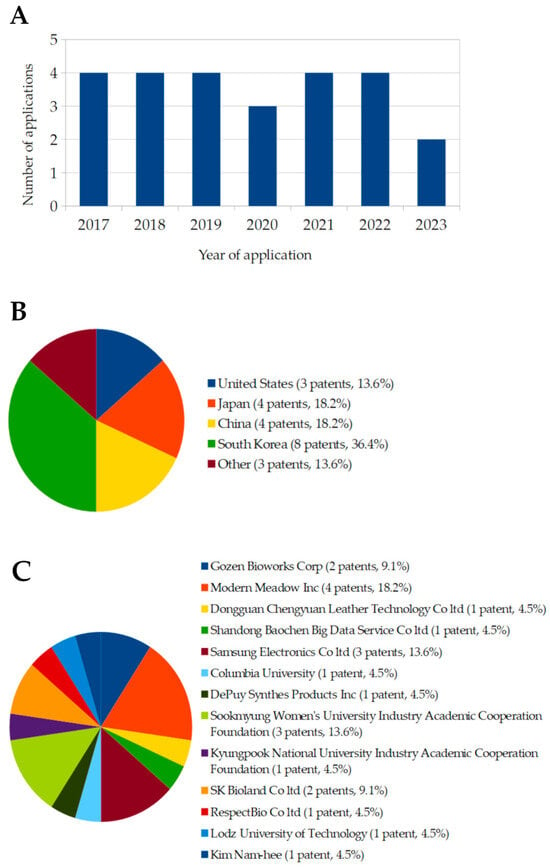
Figure 6.
Number of patents (A), publication locations (B), and patent ownership (C) related to BC-based technologies and associated fields between 2017 and 2023. The data were obtained through searches conducted on the European database Espacenet and Google Patents, accessed on 15 September 2024.
5. Conclusions and Future Perspectives
The pursuit of biomaterials as a reliable solution to mitigate the ecological impact of microplastic pollution and facilitate the recycling of agricultural waste has gained considerable attention. The biofabrication of BC offers transformative potential for the fashion industry, providing a sustainable, versatile, and high-performing alternative to traditional textiles. This aligns with the growing consumer demand for environmentally conscious fashion products. Over the past two decades, biofabrication technologies utilizing microorganisms, such as fungi and bacteria grown on agricultural residues, have been developed to generate natural cellulose. This review underscores the potential of BC across various industries, focusing on alternative biofabrication processes, the strains used, and the promising outcomes related to scalability and innovation in sustainable textiles. BC presents key advantages over conventional synthetic materials, including cost-effectiveness, safety, biodegradability, and a reduced environmental footprint. Nevertheless, challenges remain in scaling production to industrial levels, while ensuring the stability and uniformity of the final product. Future research should prioritize the optimization of substrate parameters to better understand their interactions and enhance the quality of cellulose pellicles. Moreover, relevant patent trends of BC have also been summarized. Collaborative efforts combining optimization, metabolic and genetic engineering, and autonomous technologies will be essential in advancing the bio-industry from lab-scale to commercial production, and are particularly important for the development of bio-leather as a sustainable alternative in the fashion industry.
Author Contributions
Conceptualization, D.A. and C.V. (Csaba Vágvölgyi); methodology, D.A., M.P., and C.V. (Csaba Vágvölgyi); software, C.V. (Csilla Veres); validation, C.V. (Csaba Vágvölgyi); formal analysis, D.A. and C.V. (Csilla Veres); investigation, D.A. and C.V. (Csilla Veres); resources, D.A. and C.V. (Csaba Vágvölgyi); data, M.P. and C.V. (Csilla Veres); funding acquisition, M.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors sincerely thank the Stipendium Hungaricum program for providing financial support in the form of a grant to Dheanda Absharina.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Provin, A.P.; Cubas, A.L.V.; Dutra, A.R. Textile Industry and Environment: Can the Use of Bacterial Cellulose in the Manufacture of Biotextiles Contribute to the Sector? Clean Technol. Environ. Policy 2021, 23, 2813–2825. [Google Scholar] [CrossRef]
- Xu, C.; Zhanga, B.; Gua, C.; Shena, C.; Yin, S.; Aamir, M.; Li, F. Are We Underestimating the Sources of Microplastic Pollution in Terrestrial Environment? J. Hazard. Mater. 2022, 400, 123228. [Google Scholar] [CrossRef]
- Muthu, S.S.; Ramchandani, M. Vegan Alternatives for Leather; Sustainable Textiles: Production, Processing, Manufacturing & Chemistry Series; Springer: Heidelberg, Germany, 2024; Available online: https://www.springer.com/series/16490 (accessed on 13 September 2024).
- Boucher, J.; Friot, D. Primary Microplastics in the Oceans: A Global Evaluation of Sources; IUCN: Gland, Switzerland, 2017; Available online: https://www.teraz-srodowisko.pl/media/pdf/aktualnosci/3144-primary-microplastics-in-the-oceans.pdf (accessed on 13 September 2024).
- Lu, K.; Qiao, R.; An, H.; Zhang, Y. Influence of Microplastics on the Accumulation and Chronic Toxic Effects of Cadmium in Zebrafish (Danio rerio). Chemosphere 2018, 202, 514–520. [Google Scholar] [CrossRef]
- Kefale, G.Y.; Kebede, Z.T.; Birlie, A.A. A Systematic Review on Potential Bio Leather Substitute for Natural Leather. J. Eng. 2023, 1, 1629174. [Google Scholar] [CrossRef]
- Martínez, E.; Posada, L.; Botero, J.C.; Rios-Arango, J.A.; Zapata-Benabithe, Z.; López, S.; Molina-Ramírez, C.; Osario, M.A.; Castro, C.I. Nata de fique: A Cost-Effective Alternative for the Large-Scale Production of Bacterial Nanocellulose. J. Ind Crops Prod. 2023, 192, 116015. [Google Scholar] [CrossRef]
- Brown, A.J. XLIII. On an Acetic Ferment which Forms Cellulose. J. Chem. Soc. Trans. 1886, 49, 432–439. [Google Scholar] [CrossRef]
- Mazotto, A.M.; de Ramos Silva, J.; de Brito, L.A.; Rocha, N.U.; de Souza Soares, A. How Can Microbiology Help to Improve Sustainability in the Fashion Industry? J. Environ. Technol. Innov. 2021, 23, 101760. [Google Scholar] [CrossRef]
- Mehrotra, R.; Sharma, S.; Shree, N.; Kaur, K. Bacterial Cellulose: An Ecological Alternative as a Biotextile. Biosci. Biotechnol. Res. Asia. 2023, 20, 449–463. [Google Scholar] [CrossRef]
- Mcmeeking, A.; Dieckmann, E.; Cheeseman, C. Production Methods for Bacterial Biomaterials: A Review. J. Mater. Today Sustain. 2023, 25, 100623. [Google Scholar] [CrossRef]
- Hildebrandt, J.; Thrän, D.; Bezama, A. The Circularity of Potential Bio-Textile Production Routes: Comparing Life Cycle Impacts of Bio-Based Materials Used Within the Manufacturing of Selected Leather Substitutes. J. Clean. Prod. 2021, 287, 125470. [Google Scholar] [CrossRef]
- Blanco Parte, F.G.; Santoso, S.P.; Chou, C.C.; Verma, V.; Wang, H.T.; Ismadji, S.; Cheng, K.C. Current Progress on the Production, Modification, and Applications of Bacterial Cellulose. Crit. Rev. Biotechnol. 2020, 40, 397–414. [Google Scholar] [CrossRef]
- Cohen, N.; Sicher, E.; Merino, I.; Uğur Yavuz, S. Designing with Microbial Cellulose to Feed New Biological Cycles1. Int. J. Food Des. 2020, 4, 155–171. [Google Scholar] [CrossRef]
- Jiang, H.; Song, Z.; Hao, Y.; Hu, X.; Lin, X.; Liu, S.; Li, C. Effect of Co-culture of Komagataeibacter nataicola and Selected Lactobacillus fermentum on the Production and Characterization of Bacterial Cellulose. LWT 2023, 173, 114224. [Google Scholar] [CrossRef]
- Yuan, Y.; Kang, J.; Fei, S.; Qin, X.; Lin, X.; Wang, X.; Zhao, B.; He, C.; Liu, S.; Li, C. Lactiplantibacillus plantarum Causes the Abnormal Fermentation of Bacterial Cellulose by Komagataeibacter nataicola During Nata de coco Production. Food Biosci. 2024, 61, 104603. [Google Scholar] [CrossRef]
- Devanthi, P.V.P.; Pratama, F.; Kho, K.; Taherzadeh, M.J.; Aslanzadeh, S. The Effect of Dekkera bruxellensis Concentration and Inoculation Time on Biochemical Changes and Cellulose Biosynthesis by Komagataeibacter intermedius. J. Fungi 2022, 8, 1206. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhu, X.; Chen, Y.; Wang, J. Enhanced Bacterial Cellulose Production in Gluconacetobacter xylinus by Overexpression of Two Genes (bscC and bcsD) and a Modified Static Culture. Int. J. Biol. Macromol. 2024, 260, 129552. [Google Scholar] [CrossRef]
- Brown, R.M., Jr. Cellulose Structure and Biosynthesis: What is in Store for the 21st Century? J. Polym. Sci. Part A Polym. Chem. 2004, 42, 487–495. [Google Scholar] [CrossRef]
- Quintana-Quirino, M.; Vigueras-Ramírez, G.; Alonso-Segura, D.; Shirai, K. Microbial Cellulose: Biosynthesis and Textile Applications. In Fundamentals of Natural Fibres and Textiles; Woodhead Publishing: Sawston, UK, 2021; pp. 65–85. [Google Scholar]
- Jin, K.; Jin, C.; Wu, Y. Synthetic Biology-Powered Microbial Co-culture Strategy and Application of Bacterial Cellulose-Based Composite Materials. Carbohydr. Polym. 2022, 283, 119171. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Catchmark, J.M.; Demirci, A. Co-culture Fermentation on the Production of Bacterial Cellulose Nanocomposite Produced by Komagataeibacter hansenii. Carbohydr. Polym. Technol. Appl. 2021, 2, 100028. [Google Scholar] [CrossRef]
- Yamada, Y.; Yukphan, P.; Vu, H.T.L.; Muramatsu, Y.; Ochaikul, D.; Tanasupawat, S.; Nakagawa, Y. Description of Komagataeibacter gen. nov., with Proposals of New Combinations (Acetobacteraceae). J. Gen. Appl. Microbiol. 2012, 58, 397–404. [Google Scholar] [CrossRef]
- Srivastava, S.; Mathur, G. Bacterial Cellulose: A Multipurpose Biomaterial for Manmade World. Curr. Appl. Sci. Technol. 2023, 23, 10–55003. [Google Scholar] [CrossRef]
- Provin, A.P.; de Aguiar Dutra, A.R.; Gouveia, I.C.A.S.S.; Cubas, A.L.V. Circular Economy for the Fashion Industry: Use of Waste from the Food Industry for the Production of Biotextiles. Technol. Forecast. Soc. Chang. 2021, 169, 120858. [Google Scholar] [CrossRef]
- Nayak, R.; Cleveland, D.; Tran, G.; Joseph, F. Potential of Bacterial Cellulose for Sustainable Fashion and Textile Applications: Review. J. Mater. Sci. 2024, 59, 6685–6710. [Google Scholar] [CrossRef]
- Ariyani, N.R.; Wijayanti, S.P.; Putra, N.G.; Kusumawati, M.B.; Setiawan, A.A.; Isharyadi, F.; Widyastuti, N.; Djarot, I.N.; Handayani, T. Mycelium-based Leather Potential for Sustainability: A review. AIP Conf. Proc. 2024, 3001, 030012. [Google Scholar]
- MycoWorks. Available online: https://www.mycoworks.com (accessed on 15 September 2024).
- Reishi. Available online: http://www.madewithreishi.com (accessed on 12 September 2024).
- MyCoTech. Available online: http://www.mycl.bio/mylea/ (accessed on 12 September 2024).
- Bolt Threads. Available online: https://boltthreads.com/ (accessed on 14 September 2024).
- Choi, S.M.; Rao, K.M.; Zo, S.M.; Shin, E.J.; Han, S.S. Bacterial Cellulose and Its Applications. Polymers 2022, 14, 1080. [Google Scholar] [CrossRef] [PubMed]
- Modern Synthesis. Available online: https://www.modernsynthesis.com/ (accessed on 28 September 2024).
- Nanollose. Available online: https://nanollose.com/ (accessed on 15 September 2024).
- ScobyTec. Available online: https://www.scobytec.com (accessed on 16 September 2024).
- Smobya. Available online: https://smobya.com (accessed on 15 September 2024).
- Design Friction Lab Innocell Materials. Available online: https://designfrictionlab.com/project/innocell-materials/ (accessed on 29 September 2024).
- Bandyopadhyay, S.; Saha, N.; Brodnjak, U.V.; Sáha, P. Bacterial Cellulose and Guar Gum based Modified PVP-CMC Hydrogel Films: Characterized for Packaging Fresh Berries. Food Packag. Shelf Life 2019, 22, 100402. [Google Scholar] [CrossRef]
- Cazón, P.; Velazquez, G.; Vazquez, M. Characterization of Mechanical and Barrier Properties of Bacterial Cellulose, Glycerol and Polyvinyl Alcohol (PVOH) Composite Films with Eco-Friendly UV-Protective Properties. Food Hydrocoll. 2020, 99, 105323. [Google Scholar] [CrossRef]
- Cellugy. Available online: https://www.cellugy.com/ (accessed on 12 September 2024).
- VTT Technical Research Centre—Our Research. Available online: https://www.vttresearch.com/en/ourservices (accessed on 29 September 2024).
- Fraunhofer Institute for Process Engineering and Packaging IVV—Our Research. Available online: https://www.iap.fraunhofer.de/en/research.html (accessed on 28 September 2024).
- Bielecki, S.; Kalinowska, H.; Krystynowicz, A.; Kubiak, K.; Kołodziejczyk, M.; De Groeve, M. Wound Dressings and Cosmetic Materials from Bacterial Nanocellulose. In Bacterial Nanocellulose; CRC Press: Boca Raton, FL, USA, 2012; pp. 157–174. [Google Scholar]
- Maurer, K.; Renkert, M.; Duis, M.; Weiss, C.; Wessel, L.M.; Lange, B. Application of Bacterial Nanocellulose-based Wound Dressings in the Management of Thermal Injuries: Experience in 92 Children. Burns 2022, 48, 608–614. [Google Scholar] [CrossRef]
- Cañas-Gutiérrez, A.; Toro, L.; Fornaguera, C.; Borrós, S.; Osorio, M.; Castro-Herazo, C.; Arboleda-Toro, D. Biomineralization in Three-Dimensional Scaffolds Based on Bacterial Nanocellulose for Bone Tissue Engineering: Feature Characterization and Stem Cell Differentiation. Polymers 2023, 15, 2012. [Google Scholar] [CrossRef] [PubMed]
- Biomodics. Available online: https://www.biomodics.com (accessed on 13 September 2024).
- Amorim, J.D.; Galdino, C.J.S.; Costa, A.F.; Nascimento, H.A.; Vinhas, G.M.; Sarubbo, L.A. BioMask, a Polymer Blend for Treatment and Healing of Skin Prone to Acne. Chem. Eng. Trans. 2020, 79, 205–210. [Google Scholar]
- Fonseca, D.F.; Vilela, C.; Pinto, R.J.; Bastos, V.; Oliveira, H.; Catarino, J.; Faisca, P.; Rosado, C.; Silvestre, A.J.; Freire, C.S. Bacterial Nanocellulose-Hyaluronic Acid Microneedle Patches for Skin Applications: In Vitro and In Vivo Evaluation. Mater. Sci. Eng. C. Mater. Biol. Appl. 2021, 118, 111350. [Google Scholar] [CrossRef]
- Kucuk, S.D.; Groso, A.; Collet, G.; Daniellou, R.; Caliskan, U.K. Effect of Bacterial Nanocellulose and Plant-Containing Facial Serum on Hyperpigmentation in In-vitro Conditions. BioResources 2024, 19, 3208. [Google Scholar] [CrossRef]
- Laboratory of Bionanomaterials and Cellulose Sciences, Department of Biomaterial Sciences, Graduate School of Agricultural and Life Sciences, The University of Tokyo. Available online: https://psl.fp.a.u-tokyo.ac.jp/research-e.html (accessed on 28 September 2024).
- Skočaj, M. Bacterial Nanocellulose in Papermaking. Cellulose 2019, 26, 6477–6488. [Google Scholar] [CrossRef]
- AG Soares da Silva, F.; Matos, M.; Dourado, F.; AM Reis, M.; Branco, P.C.; Poças, F.; Gama, M. Development of a Layered Bacterial Nanocellulose-PHBV Composite for Food Packaging. J. Sci. Food Agric. 2023, 103, 1077–1087. [Google Scholar] [CrossRef] [PubMed]
- Ecovative. Available online: https://ecovative.github.io/EB-Web/ (accessed on 29 September 2024).
- Tabarsa, T.; Sheykhnazari, S.; Ashori, A.; Mashkour, M.; Khazaeian, A. Preparation and Characterization of Reinforced Papers using Nano Bacterial Cellulose. Int. J. Biol. Macromol. 2017, 101, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, A.F.; Martins, D.; Dourado, F.; Sarmento, P.; Ferreira, P.J.; Gamelas, J.A. Impact of Bacterial Cellulose on the Physical Properties and Printing Quality of Fine Papers. J. Carbohydr. Polym. 2023, 314, 120915. [Google Scholar] [CrossRef] [PubMed]
- Lokhande, P.E.; Singh, P.P.; Vo, D.V.N.; Kumar, D.; Balasubramanian, K.; Mubayi, A.; Srivastava, A.; Sharma, A. Bacterial Nanocellulose: Green Polymer Materials for High Performance Energy Storage Applications. J. Environ. Chem. Eng. 2022, 10, 108176. [Google Scholar] [CrossRef]
- Ma, L.; Bi, Z.; Xue, Y.; Zhang, W.; Huang, Q.; Zhang, L.; Huang, Y. Bacterial Cellulose: An Encouraging Eco-Friendly Nano-Candidate for Energy Storage and Energy Conversion. J. Mater. Chem. A. 2020, 8, 5812–5842. [Google Scholar] [CrossRef]
- Aisyah, H.A.; Zainudin, E.S.; Bakar, B.F.A.; Ilyas, R.A. Nanofillers in Automotive and Aerospace Industry. In Nanofillers for Sustainable Applications, 1st ed.; Nurazzi, N.M., Bayraktar, E., Norrrahim, M.N.F., Aisyah, H.A., Abdullah, N., Asyraf, M.R.M., Eds.; CRC Press: Boca Raton, FL, USA, 2023; pp. 420–434. [Google Scholar]
- Nurazzi, N.M.; Jenol, M.A.; Kamarudin, S.H.; Aisyah, H.A.; Hao, L.C.; Yusuff, S.M.; Amira, M.N.; Harussani, M.M.; Norrrahim, M.N.F.; Ilyas, R.A.; et al. 19—Nanocellulose Composites in the Automotive Industry. In Industrial Applications of Nanocellulose and Its Nanocomposites; Sapuan, S.M., Norrrahim, M.N.F., Ilyas, R.A., Soutis, C., Eds.; Woodhead Publishing: Sawston, UK, 2022; pp. 439–467. [Google Scholar]
- The Government of Japan—Plant-Derived Material Will Change the Future of Automobiles. Available online: https://www.japan.go.jp/kizuna/2021/02/plant-derived_material.html (accessed on 29 September 2024).
- Marsh, G. Airbus Takes on Boeing with Reinforced Plastic A350 XWB. Reinf. Plast. 2007, 51, 26–29. [Google Scholar] [CrossRef]
- Pervaiz, M.; Panthapulakkal, S.; KC, B.; Sain, M.; Tjong, J. Emerging Trends in Automotive Lightweighting through Novel Composite Materials. Mater. Sci. Appl. 2016, 7, 26. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Z.; Yuan, J.; Xu, J.; Ji, Q.; Bai, Y. Eco-friendly Production of Leather-like Material from Bacterial Cellulose and Waste Resources. J. Clean. Prod. 2024, 476, 143700. [Google Scholar] [CrossRef]
- Pavani, C.; Rao, P.A.; Vishnu, P.; Raja, H.; Sriram; Sirisha, N. Vegan Leather from Agricultural Waste: Exploring Sustainable and Cruelty-Free Alternatives. In From Waste to Wealth; Arya, R.K., Verros, G.D., Verma, O.P., Hussain, C.M., Eds.; Springer: Singapore, 2024; pp. 951–964. [Google Scholar]
- El-Naggar, N.E.A.; El-Malkey, S.E.; Abu-Saied, M.A.; Mohammed, A.B.A. Exploration of a Novel and Efficient Source for Production of Bacterial Nanocellulose, Bioprocess Optimization, and Characterization. Sci. Rep. 2022, 12, 18533. [Google Scholar] [CrossRef]
- Aswini, K.; Gopal, N.O.; Uthandi, S. Optimized Culture Conditions for Bacterial Cellulose Production by Acetobacter senegalensis MA1. BMC Biotechnol. 2020, 20, 46. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.; Sicher, E.; Merino, I.; Yavuz, S.U. An Open-Source Bioreactor Enhancing Microbial Cellulose Production and Novel Sustainable Substances. In Sustainable Design and Manufacturing Smart Innovation, Systems and Technologies; Scholz, S.G., Howlett, R.J., Setchi, R., Eds.; Springer: Singapore, 2021; pp. 77–86. [Google Scholar]
- Núñez, D.; Oyarzún, P.; González, S.; Martínez, I. Toward Biomanufacturing of Next-Generation Bacterial Nanocellulose (BNC)-Based Materials with Tailored Properties: A Review on Genetic Engineering Approaches. Biotechnol. Adv. 2024, 74, 108390. [Google Scholar] [CrossRef] [PubMed]
- Reshmy, R.; Philip, E.; Thliuomas, D.; Madhavan, A.; Sindhu, R.; Binod, P.; Varjani, S.; Awasthi, M.K.; Pandey, A. Bacterial Nanocellulose: Engineering, Production, and Applications. J. Bioeng. 2021, 12, 11463–11483. [Google Scholar]
- Mouro, C.; Gomes, A.P.; Gouveia, I.C. Microbial Exopolysaccharides: Structure, Diversity, Applications, and Future Frontiers in Sustainable Functional Materials. Polysaccharides 2024, 5, 241–287. [Google Scholar] [CrossRef]
- Cubas, A.L.V.; Provin, A.P.; Dutra, A.R.A.; Mouro, C.; Gouveia, I.C. Advances in the Production of Biomaterials through Kombucha Using Food Waste: Concepts, Challenges, and Potential. Polymers 2023, 15, 1701. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Catchmark, J.M.; Demirci, A. Effects of Pullulan Additive and Co-culture of Aureobasidium cereans on Bacterial Cellulose Produced by Komagataeibacter hansenii. Bioprocess Biosyst. Eng. 2022, 45, 573–587. [Google Scholar] [CrossRef]
- Li, W.; Huang, X.; Liu, H.; Lian, H.; Xu, B.; Zhhanseang, W.; Sun, X.; Wang, W.; Jia, S.; Zhong, C. Improvement in Bacterial Cellulose Production by Co-culturing Bacillus cereus and Komagataeibacter xylinus. Carbohydr. Polym. 2023, 313, 120892. [Google Scholar] [CrossRef]
- Wood, J.; van der Gast, C.; Rivett, D.; Verran, J.; Redfern, J. Reproducibility of Bacterial Cellulose Nanofibers Over Sub-Cultured Generations for the Development of Novel Textiles. Front. Bioeng. Biotechnol. 2022, 10, 876822. [Google Scholar] [CrossRef]
- Huang, Y.C.; Khumsupan, D.; Lin, S.P.; Santoso, S.P.; Hsu, H.Y.; Cheng, K.C. Production of Bacterial Cellulose (BC)/Nisin Composite with Enhanced Antibacterial and Mechanical Properties through Co-cultivation of Komagataeibacter xylinum and Lactococcus lactis subsp. lactis. Int. J. Biol. Macromol. 2024, 258, 128977. [Google Scholar] [CrossRef]
- Mangayil, R.; Rajala, S.; Pammo, A.; Sarlin, E.; Luo, J.; Santala, V.; Karp, M.; Tuukkanen, S. Engineering and Characterization of Bacterial Nanocellulose Films as Low Cost and Flexible Sensor Material. ACS Appl. Mater. Interfaces 2017, 9, 19048–19056. [Google Scholar] [CrossRef]
- Jang, W.D.; Kim, T.Y.; Kim, H.U.; Shim, W.Y.; Ryu, J.Y.; Park, J.H.; Lee, S.Y. Genomic and Metabolic Analysis of Komagataeibacter xylinus DSM 2325 Producing Bacterial Cellulose Nanofiber. Biotechnol. Bioeng. 2019, 116, 3372–3381. [Google Scholar] [CrossRef]
- Huang, L.H.; Liu, Q.J.; Sun, X.W.; Li, X.J.; Liu, M.; Jia, S.R.; Xie, Y.Y.; Zhong, C. Tailoring Bacterial Cellulose Structure through CRISPR Interference-Mediated Downregulation of galU in Komagataeibacter xylinus CGMCC 2955. J. Biotechnol. Bioeng. 2020, 117, 2165–2176. [Google Scholar] [CrossRef] [PubMed]
- Mangayil, R.; Sarlin, E.; Ellis, T.; Santala, V. Modulating Bacterial Nanocellulose Crystallinity through Post-Transcriptional Repression in Komagataeibacter xylinus. bioRxiv 2024. [Google Scholar] [CrossRef]
- Al-Hagar, O.E.A.; Abol-Fotouh, D. A Turning Point in the Bacterial Nanocellulose Production Employing Low Doses of Gamma Radiation. Sci. Rep. 2022, 12, 7012. [Google Scholar] [CrossRef] [PubMed]
- Jacek, P.; Ryngajłło, M.; Bielecki, S. Structural Changes of Bacterial Nanocellulose Pellicles Induced by Genetic Modification of Komagataeibacter hansenii ATCC 23769. Appl. Microbiol. Biotechnol. 2019, 103, 5339–5353. [Google Scholar] [CrossRef] [PubMed]
- Jacek, P.; Kubiak, K.; Ryngajłło, M.; Rytczak, P.; Paluch, P.; Bielecki, S. Modification of Bacterial Nanocellulose Properties through Mutation of Motility Related Genes in Komagataeibacter hansenii ATCC 53582. New Biotechnol. 2019, 52, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Taweecheep, P.; Naloka, K.; Matsutani, M.; Yakushi, T.; Matsushita, K.; Theeragool, G. Superfine Bacterial Nanocellulose Produced by Reverse Mutations in the bcsC Gene During Adaptive Breeding of Komagataeibacter oboediens. Carbohydr. Polym. 2019, 226, 115243. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Arriaga, A.M.; Del Cerro, C.; Urbina, L.; Eceiza, A.; Corcuera, M.A.; Retegi, A.; Auxiliadora Prieto, M. Genome Sequence and Characterization of the Bcs Clusters for the Production of Nanocellulose from the Low pH Resistant Strain Komagataeibacter medellinensis ID 13488. Microb. Biotechnol. 2019, 12, 620–632. [Google Scholar] [CrossRef]
- Fang, J.; Kawano, S.; Tajima, K.; Kondo, T. In Vivo Curdlan/Cellulose Bionanocomposite Synthesis by Genetically Modified Gluconacetobacter xylinus. J. Biol. Macromol. 2015, 16, 3154–3160. [Google Scholar] [CrossRef]
- Buldum, G.; Bismarck, A.; Mantalaris, A. Recombinant Biosynthesis of Bacterial Cellulose in Genetically Modified Escherichia coli. Bioprocess Biosyst. Eng. 2018, 41, 265–279. [Google Scholar] [CrossRef]
- Peng, Z.; Lv, Z.; Liu, J.; Wang, Y.; Zhang, T.; Xie, Y.; Jia, S.; Xin, B.; Zhong, C. Engineering PTS-based Glucose Metabolism for Efficient Biosynthesis of Bacterial Cellulose by Komagataeibacter xylinus. Carbohydr. Polym. 2024, 343, 122459. [Google Scholar] [CrossRef]
- Kawano, S.; Tajima, K.; Kono, H.; Erata, T.; Munekata, M.; Takai, M. Effects of Endogenous Endo-Beta-1,4-Glucanase on Cellulose Biosynthesis in Acetobacter xylinum ATCC23769. J. Biosci. Bioeng. 2002, 94, 275–281. [Google Scholar]
- Hur, D.H. Enhanced Production of Cellulose in Komagataeibacter xylinus by Preventing Insertion of IS Element into Cellulose Synthesis Gene. Biochem. Eng. J. 2020, 156, 107527. [Google Scholar] [CrossRef]
- Lasagni, F.; Cassanelli, S.; Gullo, M. How Carbon Sources Drive Cellulose Synthesis in Two Komagataeibacter xylinus Strains. Sci. Rep. 2024, 14, 20494. [Google Scholar] [CrossRef]
- Kuo, C.H.; Teng, H.Y.; Lee, C.K. Knock-out of Glucose Dehydrogenase Gene in Gluconacetobacter xylinus for Bacterial Cellulose Production Enhancement. Biotechnol. Bioprocess Eng. 2015, 20, 18–25. [Google Scholar] [CrossRef]
- Chien, L.J.; Chen, H.T.; Yang, P.F.; Lee, C.K. Enhancement of Cellulose Pellicle Production by Constitutively Expressing Vitreoscilla Hemoglobin in Acetobacter xylinum. Biotechnol. Prog. 2006, 22, 1598–1603. [Google Scholar] [CrossRef]
- Yang, F.; Cao, Z.; Li, C.; Chen, L.; Wu, G.; Zhou, X.; Hong, F.F. A Recombinant Strain of Komagataeibacter xylinus ATCC 23770 for Production of Bacterial Cellulose from Mannose-Rich Resources. New Biotechnol. 2023, 76, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Walker, K.T.; Keane, J.; Goosens, V.J.; Song, W.; Lee, K.Y.; Ellis, T. Self-Dyeing Textiles Grown from Cellulose-Producing Bacteria with Engineered Tyrosinase Expression. bioRxiv. 2023. [Google Scholar] [CrossRef]
- Ng, F.M.; Wang, P.W. Natural Self-Grown Fashion from Bacterial Cellulose: A Paradigm Shift Design Approach in Fashion Creation. Des. J. 2016, 19, 837–855. [Google Scholar] [CrossRef]
- Ng, A. Grown Microbial 3D Fiber Art, Ava: Fusion of Traditional Art with Technology. In Proceedings of the 2017 ACM International Symposium on Wearable Computers, Maui, HI, USA, 11–15 September 2017; pp. 209–214. [Google Scholar]
- Polewka, M.; Enz, F.; Jennißen, M.; Wirth, E.; Sabantina, L. 3D Printing with Biomaterials—The New Sustainable Future of Textiles? Eng. Proc. 2023, 37, 59. [Google Scholar] [CrossRef]
- Wang, Q.; Ji, C.; Sun, L.; Sun, J.; Liu, J. Cellulose Nanofibrils Filled Poly (Lactic Acid) Biocomposite Filament for FDM 3D Printing. Molecules 2020, 25, 2319. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).