A Miniaturized QuEChERS Method Combined with Ultrahigh Liquid Chromatography Coupled to Tandem Mass Spectrometry for the Analysis of Pyrrolizidine Alkaloids in Oregano Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals, Reagents and Standard Solutions
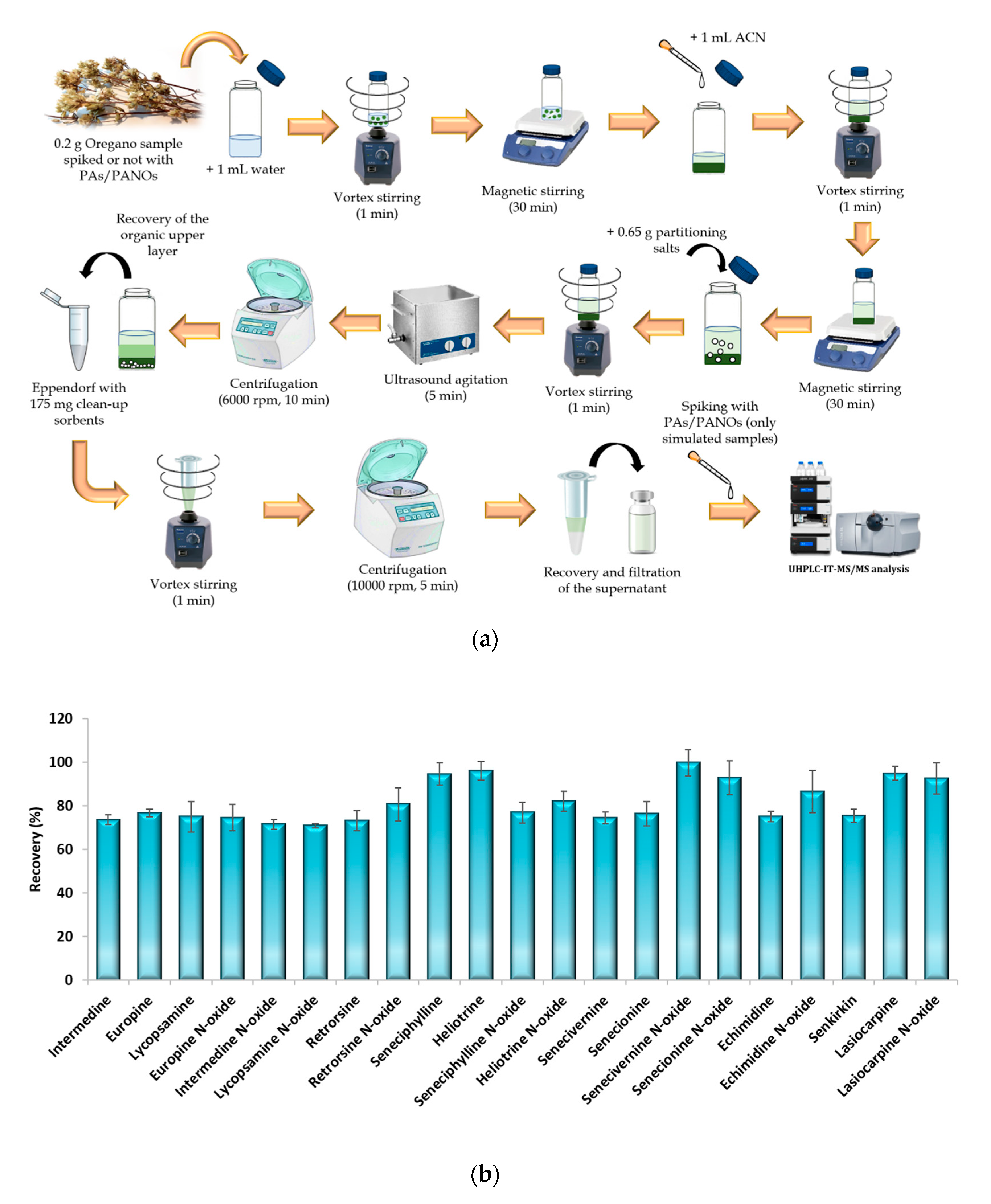
2.2. Samples and Extraction Procedure
2.3. UHPLC-MS/MS Analysis
2.4. Statistical Analysis
3. Results and Discussion
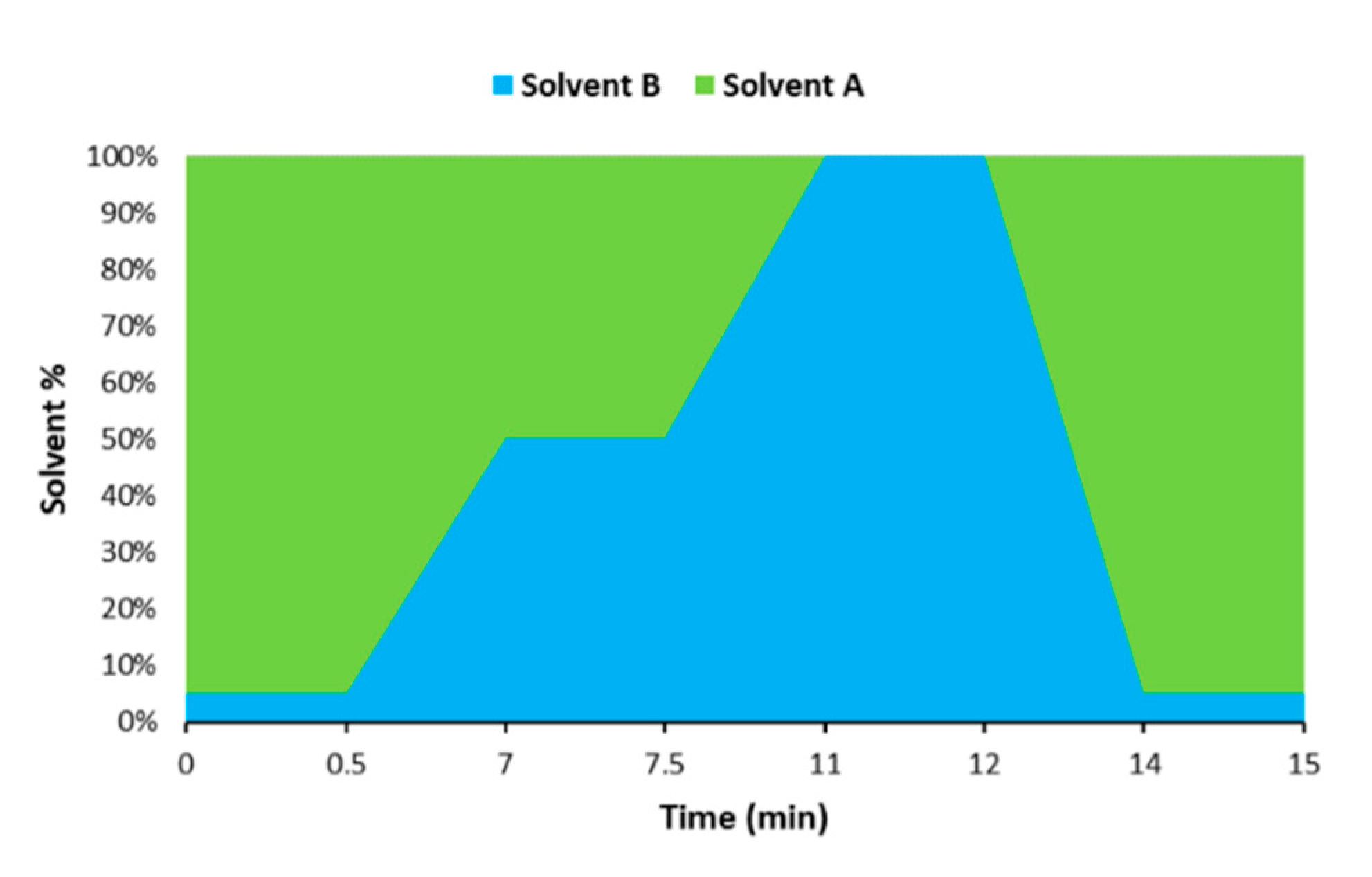
3.1. Optimization of the Chromatographic Separation
3.2. Extraction Procedure
3.3. Method Validation and Analysis of Samples
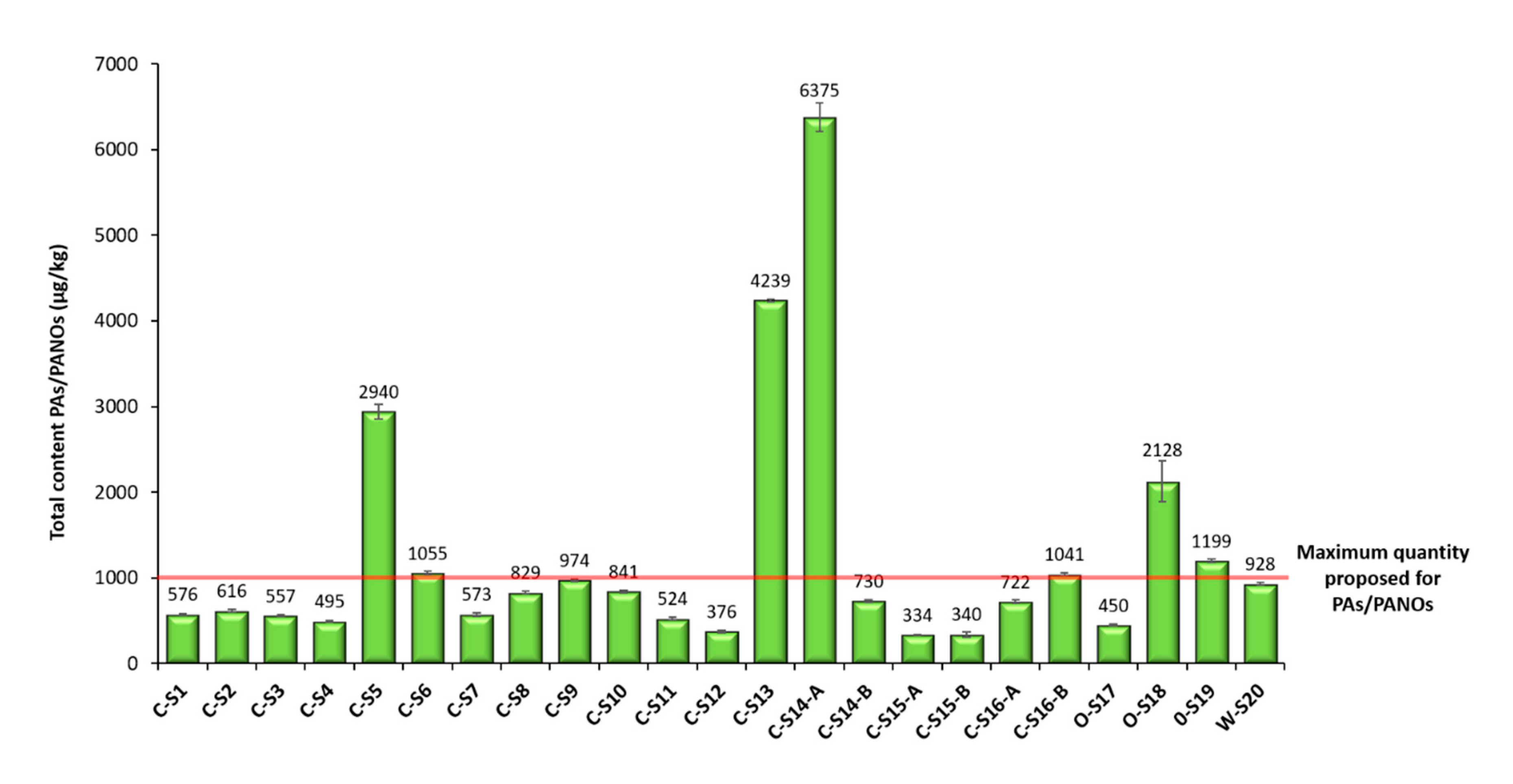
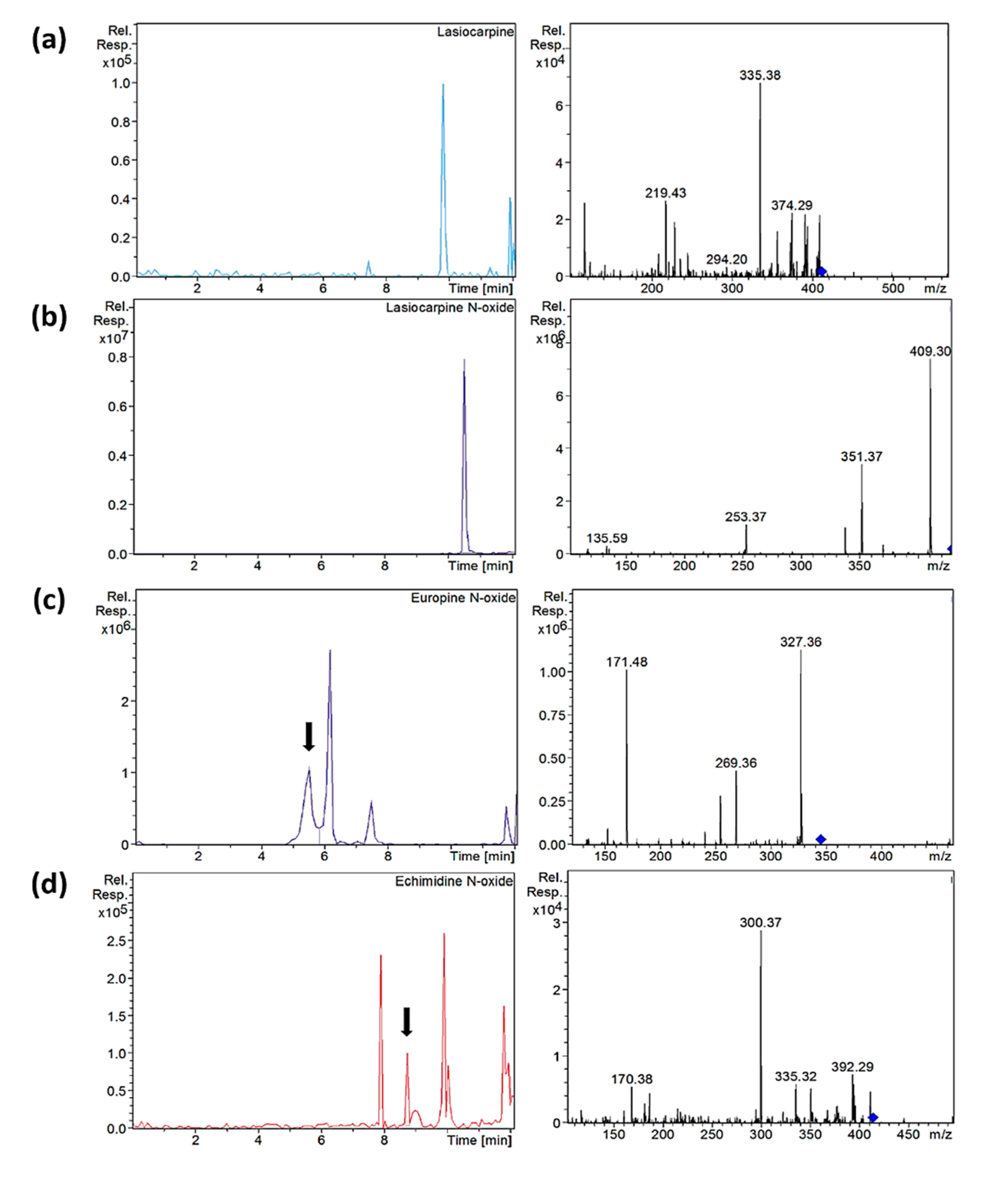
3.4. Analysis of Samples
- -
- Heliotrine-type: including europine, heliotrine, lasiocarpine and their N-oxides.
- -
- Senecionine-type: including erucifoline, jacobine, retrorsine, senecionine, seneciphylline, senecivernine, their N-oxides and senkirkin.
- -
- Lycopsamine-type: including echimidine, indicine, intermedine, lycopsamine and their N-oxides.
- -
- Monocrotaline-type: including monocrotaline, monocrotaline N-oxide and trichodesmine.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- RASFF—Food and Feed Safety Alerts. Available online: https://webgate.ec.europa.eu/rasff-window/portal/?event=SearchForm&cleanSearch=1 (accessed on 13 July 2020).
- European Food Safety Authority. Scientific Opinion on Pyrrolizidine alkaloids in food and feed. EFSA J. 2011, 9, 1–134. [Google Scholar] [CrossRef]
- Dusemund, B.; Nowak, N.; Sommerfeld, C.; Lindtner, O.; Schäfer, B.; Lampen, A. Risk assessment of pyrrolizidine alkaloids in food of plant and animal origin. Food Chem. Toxicol. 2018, 115, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Liu, Y.; Zhu, L.; Ji, H.; Song, X.; Guo, H.; Yi, T. Determination and regulation of hepatotoxic pyrrolizidine alkaloids in food: A critical review of recent research. Food Chem. Toxicol. 2018, 119, 50–60. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Opinion of the Panel on contaminants in the food chain [CONTAM] related to pyrrolizidine alkaloids as undesirable substances in animal feed. EFSA J. 2007, 447, 1–51. [Google Scholar] [CrossRef]
- Mulder, P.P.; Sánchez, P.L.; These, A.; Preiss-Weigert, A.; Castellari, M. Occurrence of pyrrolizidine alkaloids in food. EFSA Support. Publ. 2015, 12, EN-859. [Google Scholar] [CrossRef]
- European Food Safety Authority. Dietary exposure assessment to pyrrolizidine alkaloids in the European population. EFSA J. 2016, 14, 4572. [Google Scholar] [CrossRef]
- European Food Safety Authority. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements. EFSA J. 2017, 15, 4908. [Google Scholar] [CrossRef]
- Picron, J.F.; Herman, M.; Van Hoeck, E.; Goscinny, S. Analytical strategies for the determination of pyrrolizidine alkaloids in plant based food and examination of the transfer rate during the infusion process. Food Chem. 2018, 266, 514–523. [Google Scholar] [CrossRef]
- Cramer, L.; Schiebel, H.M.; Ernst, L.; Beuerle, T. Pyrrolizidine alkaloids in the food chain: Development, validation, and application of a new HPLC-ESI-MS/MS sum parameter method. J. Agric. Food Chem. 2013, 61, 11382–11391. [Google Scholar] [CrossRef]
- Kapp, T. Pyrrolizidine Alkaloids in Culinary Herbs—Take Caution with Borage-Containing Herbal Mixes; Report Published on 22 May 2017. CVUA Stuttgart, 2017. Available online: https://www.ua-bw.de/pub/beitrag.asp?subid=1&Thema_ID=2&ID=2485&lang=EN&Pdf=No (accessed on 13 July 2020).
- Kaltner, F.; Rychlik, M.; Gareis, M.; Gottschalk, C. Occurrence and risk assessment of pyrrolizidine alkaloids in spices and culinary herbs from various geographical origins. Toxins 2020, 12, 155. [Google Scholar] [CrossRef]
- Kaltner, F.; Stiglbauer, B.; Rychlik, M.; Gareis, M.; Gottschalk, C. Development of a sensitive analytical method for determining 44 pyrrolizidine alkaloids in teas and herbal teas via LC-ESI-MS/MS. Anal. Bioanal. Chem. 2019, 411, 7233–7249. [Google Scholar] [CrossRef] [PubMed]
- Dübecke, A.; Beckh, G.; Lüllmann, C. Pyrrolizidine alkaloids in honey and bee pollen. Food Addit. Contam. A 2011, 28, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Griffin, C.T.; Danaher, M.; Elliott, C.T.; Kennedy, D.G.; Furey, A. Detection of pyrrolizidine alkaloids in commercial honey using liquid chromatography–ion trap mass spectrometry. Food Chem. 2013, 136, 1577–1583. [Google Scholar] [CrossRef] [PubMed]
- Vaclavik, L.; Krynitsky, A.J.; Rader, J.I. Targeted analysis of multiple pharmaceuticals, plant toxins and other secondary metabolites in herbal dietary supplements by ultra-high performance liquid chromatography–quadrupole-orbital ion trap mass spectrometry. Anal. Chim. Acta 2014, 810, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Cristofoli, C.; Gallina, A.; Mutinelli, F. Easy and rapid method for the quantitative determination of pyrrolizidine alkaloids in honey by ultra performance liquid chromatography-mass spectrometry: An evaluation in commercial honey. Food Control 2014, 37, 146–152. [Google Scholar] [CrossRef]
- Valese, A.C.; Molognoni, L.; de Sá Ploêncio, L.A.; de Lima, F.G.; Gonzaga, L.V.; Górniak, S.L.; Daguer, H.; Barreto, F.; Costa, A.C.O. A fast and simple LC-ESI-MS/MS method for detecting pyrrolizidine alkaloids in honey with full validation and measurement uncertainty. Food Control 2016, 67, 183–191. [Google Scholar] [CrossRef]
- Wang, T.; Frandsen, H.L.; Christiansson, N.R.; Rosendal, S.E.; Pedersen, M.; Smedsgaard, J. Pyrrolizidine alkaloids in honey: Quantification with and without standards. Food Control 2019, 98, 227–237. [Google Scholar] [CrossRef]
- Chen, L.; Mulder, P.P.; Peijnenburg, A.; Rietjens, I.M. Risk assessment of intake of pyrrolizidine alkaloids from herbal teas and medicines following realistic exposure scenarios. Food Chem. Toxicol. 2019, 130, 142. [Google Scholar] [CrossRef]
- Picron, J.F.; Herman, M.; Van Hoeck, E.; Goscinny, S. Monitoring of pyrrolizidine alkaloids in beehive products and derivatives on the Belgian market. Environ. Sci. Pollut. Res. 2020, 27, 5693–5708. [Google Scholar] [CrossRef]
- Black, C.; Haughey, S.A.; Chevallier, O.P.; Galvin-King, P.; Elliott, C.T. A comprehensive strategy to detect the fraudulent adulteration of herbs: The oregano approach. Food Chem. 2016, 210, 551–557. [Google Scholar] [CrossRef]
- Moreda-Piñeiro, J.; Moreda-Piñeiro, A. Recent advances in combining microextraction techniques for sample pre-treatment. TrAC 2015, 71, 265–274. [Google Scholar] [CrossRef]
- Płotka-Wasylka, J.; Szczepańska, N.; Owczarek, K.; Namieśnik, J. Miniaturized Solid Phase Extraction. In Green Extraction Techniques: Principles, Advances and Applications; Ibañez, E., Cifuentes, A., Eds.; Comprehensive Analytical Chemistry Elsevier: Amsterdam, The Netherlands, 2017; Volume 76, pp. 279–318. [Google Scholar]
- Filippou, O.; Bitas, D.; Samanidou, V. Green approaches in sample preparation of bioanalytical samples prior to chromatographic analysis. J. Chromatogr. B 2017, 1043, 44–62. [Google Scholar] [CrossRef] [PubMed]
- Casado, N.; Gañán, J.; Morante-Zarcero, S.; Sierra, I. New advanced materials and sorbent-based microextraction techniques as strategies in sample preparation to improve the determination of natural toxins in food samples. Molecules 2020, 25, 702. [Google Scholar] [CrossRef] [PubMed]
- Anastassiades, M.; Lehotay, S.J.; Štajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef] [PubMed]
- European Standards CSN EN 15662:2008. Foods of Plant Origin—Determination of Pesticide Residues Using GC-MS and/or LC-MS/MS Following Acetonitrile Extraction/Partitioning and Clean-Up by Dispersive SPE—QuEChERS-Method. Available online: https://www.aenor.com/normas-y-libros/buscador-de-normas/bsi?c=000000000030162467 (accessed on 17 September 2020).
- Avula, B.; Sagi, S.; Wang, Y.H.; Zweigenbaum, J.; Wang, M.; Khan, I.A. Characterization and screening of pyrrolizidine alkaloids and N-oxides from botanicals and dietary supplements using UHPLC-high resolution mass spectrometry. Food Chem. 2015, 178, 136–148. [Google Scholar] [CrossRef]
- Chmit, M.S.; Wahrig, B.; Beuerle, T. Quantitative and qualitative analysis of pyrrolizidine alkaloids in liqueurs, elixirs and herbal juices. Fitoterapia 2019, 136, 104172. [Google Scholar] [CrossRef]
- Griffin, C.T.; Gosetto, F.; Danaher, M.; Sabatini, S.; Furey, A. Investigation of targeted pyrrolizidine alkaloids in traditional Chinese medicines and selected herbal teas sourced in Ireland using LC-ESI-MS/MS. Food Addit. Contam. A 2014, 31, 940–961. [Google Scholar] [CrossRef]
- Griffin, C.T.; Mitrovic, S.M.; Danaher, M.; Furey, A. Development of a fast isocratic LC-MS/MS method for the high-throughput analysis of pyrrolizidine alkaloids in Australian honey. Food Addit. Contam. A 2015, 32, 214–228. [Google Scholar] [CrossRef]
- QuEChERS CVUA Stuttgart. A Mini-Multiresidue Method for the Analysis of Pesticide Residues in Low-Fat Products. Available online: https://www.quechers.com/pdf/reality.pdf (accessed on 13 July 2020).
- European Commission SANTE/11813/2017. Guidance Document on Analytical Quality Control and Method Validation Procedures for Pesticide Residues and Analysis in Food and Feed. Available online: https://www.eurl-pesticides.eu/userfiles/file/EurlALL/AqcGuidance_SANTE_2019_12682.pdf (accessed on 13 July 2020).
- Commission Regulation (EC) No 401/2006. Laying Down the Methods of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006R0401&from=ES (accessed on 13 July 2020).
- Kapp, T.; Hägele, F.; Plate, E.M. Oregano—An Aromatic but Loaded Culinary Herb; Part I: Pyrrolizidine Alkaloids; Report Published on 15 August 2019. CVUA Stuttgart, 2019. Available online: https://www.cvuas.de/pesticides/beitrag_en.asp?subid=1&Thema_ID=5&ID=3024&lang=EN&Pdf=No (accessed on 13 July 2020).
- Selmar, D.; Radwan, A.; Nowak, M. Horizontal natural product transfer: A so far unconsidered source of contamination of plant-derived commodities. J. Environ. Anal. Toxicol. 2015, 5, 1000287. [Google Scholar] [CrossRef]
- Selmar, D.; Wittke, C.; Beck-vonWolersdor, I.; Klier, B.; Lewerenz, L.; Kleinwächter, M.; Nowak, M. Transfer of pyrrolizidine alkaloids between living plants: A disregarded source of contaminations. Environ. Pollut. 2019, 248, 456–461. [Google Scholar] [CrossRef]
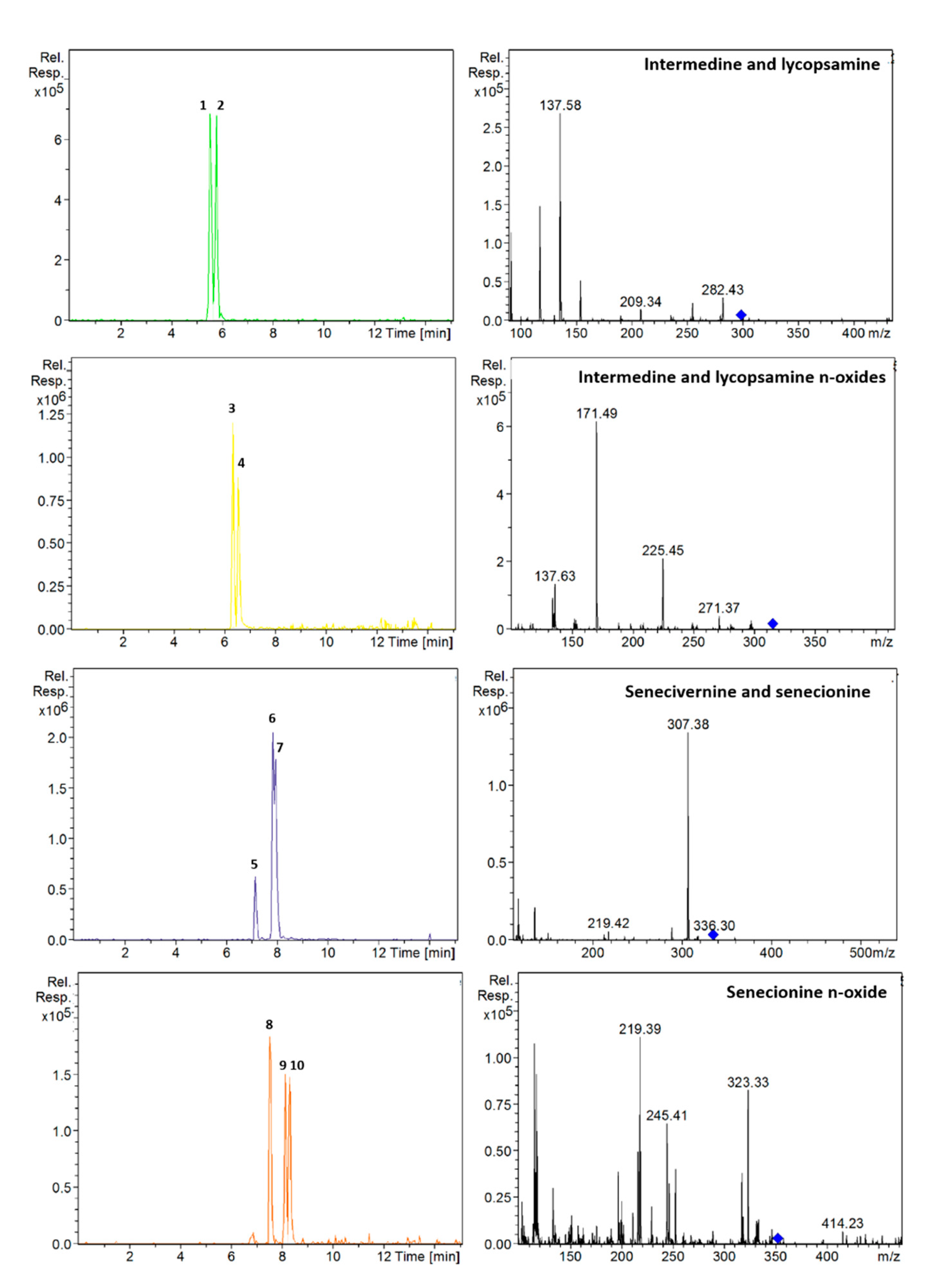
| Analyte | Retention Time (min) | Ionization Mode | Precursor Ion (m/z) | Fragmentation Amplitude | MS2. Product Ions a (m/z) |
|---|---|---|---|---|---|
| Intermedine | 5.6 | ESI (+) | 299 | 0.70 | 138 *, 120 |
| Europine | 5.7 | ESI (+) | 329 | 0.80 | 253 *, 138 |
| Lycopsamine | 5.8 | ESI (+) | 299 | 0.70 | 138 *, 120 |
| Europine N-oxide | 6.2 | ESI (+) | 345 | 0.80 | 327 *, 171.5 |
| Intermedine N-oxide | 6.4 | ESI (+) | 315 | 0.80 | 225, 171.5 * |
| Lycopsamine N-oxide | 6.5 | ESI (+) | 315 | 0.80 | 171.5 *, 138 |
| Retrorsine | 6.8 | ESI (+) | 351 | 0.80 | 323 *, 275 |
| Retrorsine N-oxide | 7.0 | ESI (+) | 367 | 0.90 | 339 *, 245 |
| Seneciphylline | 7.2 | ESI (+) | 333 | 0.80 | 305 *, 120 |
| Heliotrine | 7.2 | ESI (+) | 313.5 | 0.70 | 138 *, 120 |
| Seneciphylline N-oxide | 7.5 | ESI (+) | 350 | 0.80 | 321 *, 118 |
| Heliotrine N-oxide | 7.6 | ESI (+) | 329 | 1.00 | 171 *, 136 |
| Senecivernine | 7.9 | ESI (+) | 335 | 0.80 | 307 *, 120 |
| Senecionine | 7.9 | ESI (+) | 335 | 0.80 | 307 *, 120 |
| Senecivernine N-oxide | 8.1 | ESI (+) | 351 | 0.80 | 323 *, 219.5 |
| Senecionine N-oxide | 8.3 | ESI (+) | 352 | 1.00 | 220, 118 * |
| Echimidine | 8.7 | ESI (+) | 398 | 0.60 | 220, 120 * |
| Echimidine N-oxide | 8.7 | ESI (+) | 413 | 0.70 | 395 *, 351 |
| Senkirkin | 9.1 | ESI (+) | 365 | 0.80 | 167.5 *, 150 |
| Lasiocarpine | 9.8 | ESI (+) | 411 | 0.70 | 335 *, 219.5 |
| Lasiocarpine N-oxide | 10.4 | ESI (+) | 428 | 0.80 | 409 *, 352 |
| Analytes | Linear Range (µg/kg) | Matrix-Matched Calibration R2/cm | Accuracy | Precision | MDL (µg/kg) | MQL (µg/kg) | ME (%) | ||
|---|---|---|---|---|---|---|---|---|---|
| Recovery (% ± sd) | Mean Recovery (% ± sd) | Intra-Day Precision (RSD%) | Inter-Day Precision (RSD%) | ||||||
| Intermedine | 25.0–500.0 | y = 2035x + 5771 0.999/93 | 78 ± 7 a | 77 ± 3 | 5 a | 6 a | 7.5 | 25.0 | 23 |
| 74 ± 2 b | 7 b | 10 b | |||||||
| 80 ± 6 c | 3 c | 3 c | |||||||
| Europine | 10.0–500.0 | y = 4482x + 58,782 0.999/98 | 83 ± 4 a | 83 ± 7 | 4 a | 7 a | 3.0 | 10.0 | 23 |
| 77 ± 2 b | 5 b | 6 b | |||||||
| 90 ± 11 c | 4 c | 5 c | |||||||
| Lycopsamine | 22.0–500.0 | y = 1559x + 8916 0.999/99 | 95 ± 7 a | 90 ± 13 | 7 a | 12 a | 6.7 | 22.0 | 18 |
| 75 ± 7 b | 5 b | 8 b | |||||||
| 99 ± 7 c | 1 c | 4 c | |||||||
| Europine N-oxide | 7.5–500.0 | y = 6816x + 43,628 0.999/100 | 100 ± 5 a | 88 ± 13 | 6 a | 8 a | 2.2 | 7.5 | 16 |
| 75 ± 6 b | 5 b | 9 b | |||||||
| 90 ± 5 c | 3 c | 5 c | |||||||
| Intermedine N-oxide | 7.5–500.0 | y = 2111x + 75,308 0.999/93 | 78 ± 3 a | 80 ± 11 | 8 a | 12 a | 2.2 | 7.5 | 17 |
| 71 ± 2 b | 7 b | 11 b | |||||||
| 92 ± 4 c | 2 c | 4 c | |||||||
| Lycopsamine N-oxide | 12.5–500.0 | y = 2017x + 29,990 0.999/93 | 94 ± 3 a | 86 ± 13 | 5 a | 8 a | 3.7 | 12.5 | 13 |
| 71 ± 1 b | 4 b | 5 b | |||||||
| 92 ± 3 c | 7 c | 10 c | |||||||
| Retrorsine | 5.5–500.0 | y = 1091x + 12,358 0.999/97 | 91 ± 4 a | 82 ± 9 | 7 a | 11 a | 1.7 | 5.5 | 13 |
| 73 ± 5 b | 4 b | 12 b | |||||||
| 82 ± 5 c | 2 c | 4 c | |||||||
| Retrorsine N-oxide | 3.5–500.0 | y = 543x + 13,424 0.999/95 | 79 ± 1 a | 83 ± 5 | 6 a | 8 a | 1.0 | 3.5 | 18 |
| 81 ± 8 b | 3 b | 7 b | |||||||
| 89 ± 6 c | 5 | 7 | |||||||
| Seneciphylline | 2.0–500.0 | y = 2244x + 25,457 0.999/94 | 88 ± 7 a | 90 ± 4 | 7 a | 9 a | 0.7 | 2.0 | 12 |
| 95 ± 5 b | 3 b | 10 b | |||||||
| 87 ± 7 c | 4 c | 5 c | |||||||
| Heliotrine | 4.0–500.0 | y = 4771x + 9413 0.999/92 | 80 ± 9 a | 91 ± 9 | 3 a | 8 a | 1.3 | 4.0 | 17 |
| 96 ± 4 b | 7 b | 9 b | |||||||
| 96 ± 12 c | 2 c | 6 c | |||||||
| Seneciphylline N-oxide | 1.0–250.0 | y = 2856x – 29,835 0.999/95 | 95 ± 4 a | 88 ± 10 | 3 a | 4 a | 0.4 | 1.0 | 34 |
| 77 ± 5 b | 6 b | 9 b | |||||||
| 93 ± 7 c | 2 c | 7 c | |||||||
| Heliotrine N-oxide | 3.0–500.0 | y = 684x + 13,923 0.999/95 | 81 ± 7 a | 87 ± 1 | 8 a | 9 a | 1.0 | 3.0 | 3 |
| 82 ± 5 b | 8 b | 12 b | |||||||
| 99 ± 10 c | 5 c | 12 c | |||||||
| Senecivernine | 0.5–500.0 | y = 7802x + 480,780 0.999/96 | 98 ± 7 a | 89 ± 13 | 4 a | 6 a | 0.1 | 0.5 | 25 |
| 74 ± 3 b | 4 b | 9 b | |||||||
| 95 ± 8 c | 2 c | 4 c | |||||||
| Senecionine | 0.5–500.0 | y = 7830x + 339,046 0.999/95 | 99 ± 4 a | 88 ± 12 | 4 a | 7 a | 0.1 | 0.5 | 18 |
| 76 ± 6 b | 4 b | 7 b | |||||||
| 90 ± 11 c | 4 c | 6 c | |||||||
| Senecivernine N-oxide | 6.0–250.0 | y = 2287x + 4421 0.999/97 | 95 ± 2 a | 96 ± 4 | 6 a | 10 a | 1.8 | 6.0 | 39 |
| 100 ± 6 b | 6 b | 7 b | |||||||
| 93 ± 6 c | 4 c | 8 c | |||||||
| Senecionine N-oxide | 3.0–500.0 | y = 1208x + 146,398 0.999/98 | 96 ± 8 a | 92 ± 4 | 8 a | 13 a | 0.9 | 3.0 | 28 |
| 93 ± 8 b | 3 b | 5 b | |||||||
| 88 ± 5 c | 6 c | 8 c | |||||||
| Echimidine | 7.0–500.0 | y = 4704x + 167,863 0.999/94 | 78 ± 4 a | 82 ± 10 | 5 a | 5 a | 2.0 | 7.0 | 17 |
| 75 ± 2 b | 6 b | 12 b | |||||||
| 94 ± 11 c | 4 c | 6 c | |||||||
| Echimidine N-oxide | 7.5–250.0 | y = 1005x – 48,988 0.999/96 | 87 ± 2 a | 86 ± 2 | 8 a | 12 a | 2.0 | 7.5 | 111 |
| 86 ± 10 b | 7 b | 9 b | |||||||
| 84 ± 11 c | 4 c | 12 c | |||||||
| Senkirkin | 7.5–500.0 | y = 1327x + 17,984 0.999/97 | 74 ± 5 a | 83 ± 15 | 3 a | 7 a | 2.0 | 7.5 | 8 |
| 75 ± 3 b | 4 b | 7 b | |||||||
| 101 ± 2 c | 5 c | 7 c | |||||||
| Lasiocarpine | 25.0–500.0 | y = 282x + 4928 0.999/92 | 96 ± 5 a | 94 ± 2 | 7 a | 10 a | 7.5 | 25.0 | 18 |
| 95 ± 3 b | 3 b | 10 b | |||||||
| 92 ± 10 c | 5 c | 7 c | |||||||
| Lasiocarpine N-oxide | 10.0–500.0 | y = 6252x + 2254 0.999/100 | 101 ± 6 a | 92 ± 10 | 5 a | 12 a | 3.0 | 10.0 | 13 |
| 93 ± 7 b | 8 b | 10 b | |||||||
| 82 ± 7 c | 5 c | 5 c | |||||||
| Analytes (µg/kg) | C-S1 | C-S2 | C-S3 | C-S4 | C-S5 | C-S6 | C-S7 | C-S8 | C-S9 | C-S10 | C-S11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Intermedine | n.d. | n.d. | n.d. | <MQL | <MQL | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | |
| Europine | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | <MQL | |
| Lycopsamine | n.d. | n.d. | n.d. | n.d. | <MQL | <MQL | n.d. | <MQL | n.d. | n.d. | n.d. | |
| Europine N-oxide | 14 ± 3 | <MQL | <MQL | <MQL | 25 ± 5 | 15 ± 4 | 14 ± 3 | <MQL | 16 ± 6 | 15 ± 4 | 8 ± 2 | |
| Intermedine N-oxide | 93 ± 4 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 138 ± 4 | 235 ± 5 | n.d. | n.d. | |
| Lycopsamine N-oxide | 111 ± 5 | n.d. | <MQL | n.d. | 39 ± 2 | <MQL | <MQL | 165 ± 7 | 261 ± 11 | <MQL | 17 ± 2 | |
| Retrorsine | 10 ± 2 | <MQL | n.d. | n.d. | 17 ± 1 | 10 ± 5 | 6.7 ± 0.4 | <MQL | 12 ± 4 | <MQL | <MQL | |
| Retrorsine N-oxide | 5 ± 2 | 6 ± 2 | <MQL | n.d. | 41 ± 12 | 12 ± 4 | 55 ± 5 | 19 ± 3 | 10 ± 3 | <MQL | 31 ± 4 | |
| Seneciphylline | n.d. | n.d. | n.d. | n.d. | 12 ± 2 | n.d. | <MQL | n.d. | n.d. | 5 ± 4 | n.d. | |
| Heliotrine | 5 ± 2 | 5 ± 1 | 7.9 ± 0.3 | <MQL | 22 ± 5 | 8 ± 2 | 36 ± 5 | <MQL | <MQL | 9.3 ± 0.4 | 6.1 ± 0.5 | |
| Seneciphylline N-oxide | 18.7 ± 0.8 | 82 ± 3 | 20.5 ± 0.7 | 26 ± 1 | 88 ± 18 | 19 ± 4 | 65 ± 7 | 100 ± 13 | 19 ± 2 | 53 ± 4 | 49 ± 6 | |
| Heliotrine N-oxide | 13.2 ± 0.9 | n.d. | n.d. | n.d. | 47 ± 1 | n.d. | 15 ± 6 | 6.6 ± 0.4 | n.d. | n.d. | n.d. | |
| Senecivernine | 11 ± 4 | 154 ± 3 | 102 ± 7 | 105 ± 4 | 1027 ± 58 | 385 ± 6 | 66 ± 3 | 6 ± 5 | 6 ± 3 | 225.4 ± 0.4 | n.d. | |
| Senecionine | 24 ± 3 | 166 ± 4 | 120 ± 5 | 121 ± 8 | 1103 ± 60 | 385 ± 15 | 81.8 ± 0.8 | 24 ± 5 | 24 ± 3 | 231 ± 1 | 2.4 ± 0.3 | |
| Senecivernine N-oxide | 8 ± 1 | 17 ± 3 | 10.9 ± 0.3 | 8 ± 2 | 138 ± 4 | 11 ± 1 | 24 ± 6 | 53 ± 3 | 9 ± 3 | 10.8 ± 0.7 | 70 ± 4 | |
| Senecionine N-oxide | n.d. | 16 ± 3 | n.d. | n.d. | 91 ± 9 | n.d. | 9 ± 5 | n.d. | n.d. | n.d. | n.d. | |
| Echimidine | <MQL | <MQL | <MQL | <MQL | 19 ± 5 | <MQL | <MQL | <MQL | n.d. | <MQL | n.d. | |
| Echimidine N-oxide | 160 ± 2 | 100 ± 6 | 194 ± 2 | 185 ± 2 | 140 ± 4 | 148 ± 1 | 106 ± 7 | 253 ± 6 | 302 ± 7 | 202 ± 11 | 267 ± 10 | |
| Senkirkin | 10.32 ± 0.06 | n.d. | 8 ± 2 | n.d. | 27 ± 5 | <MQL | 10 ± 1 | <MQL | <MQL | <MQL | <MQL | |
| Lasiocarpine | 81 ± 3 | 45 ± 5 | 76 ± 3 | 36 ± 2 | 66 ± 4 | 47 ± 4 | 57 ± 7 | 50 ± 5 | 65 ± 4 | 72 ± 7 | 55 ± 7 | |
| Lasiocarpine N-oxide | 12 ± 1 | 25 ± 5 | 18 ± 2 | 14 ± 1 | 38 ± 3 | 15 ± 4 | 27 ± 6 | 14 ± 2 | 15 ± 4 | 17.9 ± 0.2 | 18 ± 1 | |
| Total | 576 ± 10 | 616 ± 12 | 557 ± 10 | 495 ± 10 | 2940 ± 88 | 1055 ± 19 | 573 ± 19 | 829 ± 19 | 974 ± 18 | 841 ± 15 | 524 ± 15 | |
| Analytes (µg/kg) | C-S12 | C-S13 | C-S14-A | C-S14-B | C-S15-A | C-S15-B | C-S16-A | C-S16-B | O-S17 | O-S18 | 0-S19 | W-S20 |
| Intermedine | <MQL | 47 ± 3 | 27.7 ± 0.8 | <MQL | <MQL | n.d. | <MQL | n.d. | n.d. | <MQL | n.d. | n.d. |
| Europine | <MQL | 3142 ± 4 | 170 ± 3 | <MQL | <MQL | 11 ± 2 | <MQL | <MQL | <MQL | 25 ± 6 | <MQL | <MQL |
| Lycopsamine | <MQL | 44 ± 5 | 25.4 ± 0.8 | <MQL | <MQL | n.d. | <MQL | n.d. | n.d. | <MQL | n.d. | <MQL |
| Europine N-oxide | 9 ± 1 | 118 ± 7 | 737 ± 9 | 12.2 ± 0.2 | 10.5 ± 0.1 | 23 ± 4 | 16.6 ± 0.8 | 11.3 ± 0.2 | 17 ± 1 | 1195 ± 235 | 10.1 ± 0.2 | 10.5 ± 0.7 |
| Intermedine N-oxide | n.d. | n.d. | 22.8 ± 0.1 | n.d. | n.d. | n.d. | 152 ± 7 | 206 ± 1 | n.d. | n.d. | 602 ± 8 | 305 ± 10 |
| Lycopsamine N-oxide | 24 ± 4 | 18 ± 4 | 78 ± 4 | 104 ± 9 | <MQL | 40 ± 3 | 99 ± 5 | 99 ± 6 | n.d. | 35.1 ± 0.3 | 211 ± 10 | 163 ± 7 |
| Retrorsine | 19 ± 2 | 12 ± 2 | 19 ± 2 | 9 ± 3 | 14 ± 2 | 8 ± 2 | 11 ± 2 | 10 ± 3 | <MQL | <MQL | <MQL | <MQL |
| Retrorsine N-oxide | 12.8 ± 0.8 | 46 ± 7 | 33 ± 9 | 15 ± 3 | 5.4 ± 0.4 | 4.8 ± 0.8 | 5 ± 1 | n.d. | n.d. | <MQL | n.d. | 7 ± 2 |
| Seneciphylline | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Heliotrine | 12 ± 2 | 30 ± 4 | 45 ± 10 | <MQL | 6.2 ± 0.9 | n.d. | 12 ± 1 | 5 ± 1 | 6.6 ± 0.5 | 8.1 ± 0.7 | 7 ± 1 | 17 ± 1 |
| Seneciphylline N-oxide | 17 ± 1 | 20 ± 3 | 23 ± 4 | 22 ± 3 | 23 ± 4 | n.d. | 21 ± 2 | 20 ± 4 | 13.7 ± 0.2 | 19 ± 2 | 16 ± 3 | 20 ± 3 |
| Heliotrine N-oxide | n.d. | <MQL | 34 ± 4 | n.d. | n.d. | n.d. | <MQL | 6 ± 2 | n.d. | 75 ± 6 | 11 ± 4 | n.d. |
| Senecivernine | n.d. | n.d. | 492 ± 78 | 94 ± 5 | n.d. | 8 ± 0.7 | 19 ± 2 | 21 ± 4 | 115 ± 7 | 224 ± 7 | 57 ± 4 | n.d. |
| Senecionine | 4 ± 1 | 24 ± 4 | 524 ± 80 | 52 ± 4 | 5 ± 1 | n.d. | 60 ± 6 | 62 ± 4 | 126 ± 8 | 240 ± 6 | 82 ± 5 | 5.7 ± 0.4 |
| Senecivernine N-oxide | 7.2 ± 0.6 | 17 ± 2 | 11 ± 2 | 13 ± 2 | 10.8 ± 0.2 | n.d. | 12 ± 2 | 20 ± 5 | <MQL | 6 ± 2 | 13 ± 2 | 29 ± 1 |
| Senecionine N-oxide | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Echimidine | n.d. | n.d. | n.d. | n.d. | n.d. | <MQL | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Echimidine N-oxide | 186 ± 3 | 322 ± 7 | 183 ± 11 | 297 ± 5 | 204 ± 4 | 173 ± 29 | 182 ± 9 | 491 ± 11 | 102 ± 7 | 84.6 ± 0.9 | 87 ± 6 | 211 ± 8 |
| Senkirkin | <MQL | 9.8 ± 0.8 | 17 ± 2 | 19 ± 3 | 8.6 ± 0.1 | 9 ± 2 | 16 ± 2 | 10 ± 3 | 10.5 ± 0.1 | 105 ± 2 | 13 ± 1 | 30 ± 1 |
| Lasiocarpine | 70 ± 11 | 326 ± 1 | 986 ± 98 | 48 ± 4 | 36 ± 5 | 38 ± 9 | 86 ± 3 | 67 ± 2 | 32 ± 4 | 80 ± 13 | 63 ± 10 | 58 ± 2 |
| Lasiocarpine N-oxide | 15 ± 3 | 63 ± 6 | 2947 ± 76 | 45 ± 4 | 10 ± 2 | 25 ± 5 | 30 ± 8 | 13 ± 1 | 27 ± 2 | 31 ± 1 | 27 ± 1 | 72 ± 4 |
| Total | 376 ± 13 | 4239 ± 17 | 6375 ± 168 | 730 ± 15 | 334 ± 8 | 340 ± 31 | 722 ± 17 | 1041 ± 16 | 450 ± 14 | 2128 ± 236 | 1199 ± 19 | 928 ± 16 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izcara, S.; Casado, N.; Morante-Zarcero, S.; Sierra, I. A Miniaturized QuEChERS Method Combined with Ultrahigh Liquid Chromatography Coupled to Tandem Mass Spectrometry for the Analysis of Pyrrolizidine Alkaloids in Oregano Samples. Foods 2020, 9, 1319. https://doi.org/10.3390/foods9091319
Izcara S, Casado N, Morante-Zarcero S, Sierra I. A Miniaturized QuEChERS Method Combined with Ultrahigh Liquid Chromatography Coupled to Tandem Mass Spectrometry for the Analysis of Pyrrolizidine Alkaloids in Oregano Samples. Foods. 2020; 9(9):1319. https://doi.org/10.3390/foods9091319
Chicago/Turabian StyleIzcara, Sergio, Natalia Casado, Sonia Morante-Zarcero, and Isabel Sierra. 2020. "A Miniaturized QuEChERS Method Combined with Ultrahigh Liquid Chromatography Coupled to Tandem Mass Spectrometry for the Analysis of Pyrrolizidine Alkaloids in Oregano Samples" Foods 9, no. 9: 1319. https://doi.org/10.3390/foods9091319
APA StyleIzcara, S., Casado, N., Morante-Zarcero, S., & Sierra, I. (2020). A Miniaturized QuEChERS Method Combined with Ultrahigh Liquid Chromatography Coupled to Tandem Mass Spectrometry for the Analysis of Pyrrolizidine Alkaloids in Oregano Samples. Foods, 9(9), 1319. https://doi.org/10.3390/foods9091319








