Plant-Based Bigels for Functional Delivery: Advances in Structural Design and Stabilization Strategies
Abstract
1. Introduction
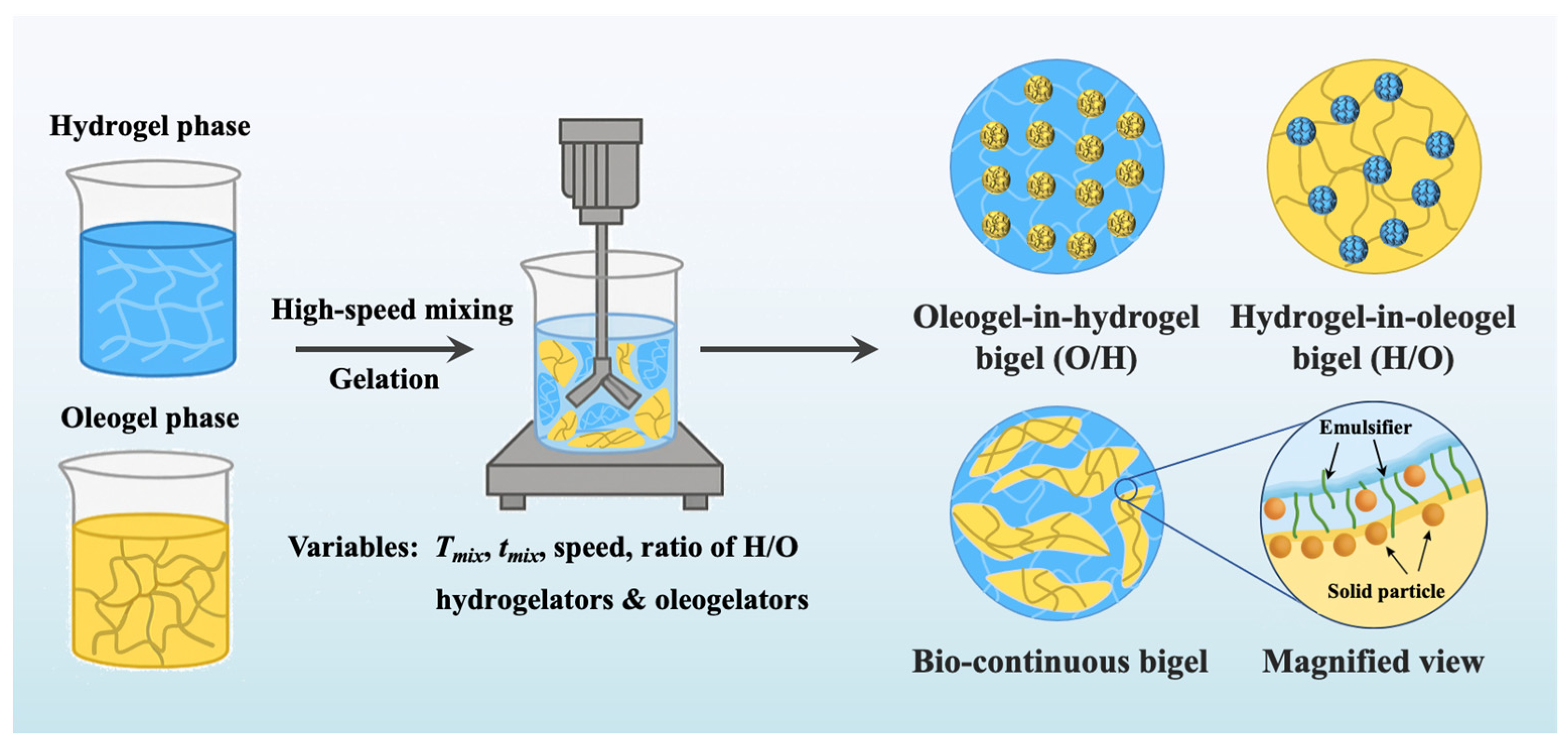
2. Structural Design and Stabilization Strategies of Plant-Based Bigels
2.1. Typical Plant-Based Gel Components
2.2. Regulation of Phase Behavior in Plant-Based Bigels
2.3. Interfacial Design for Plant-Based Bigels
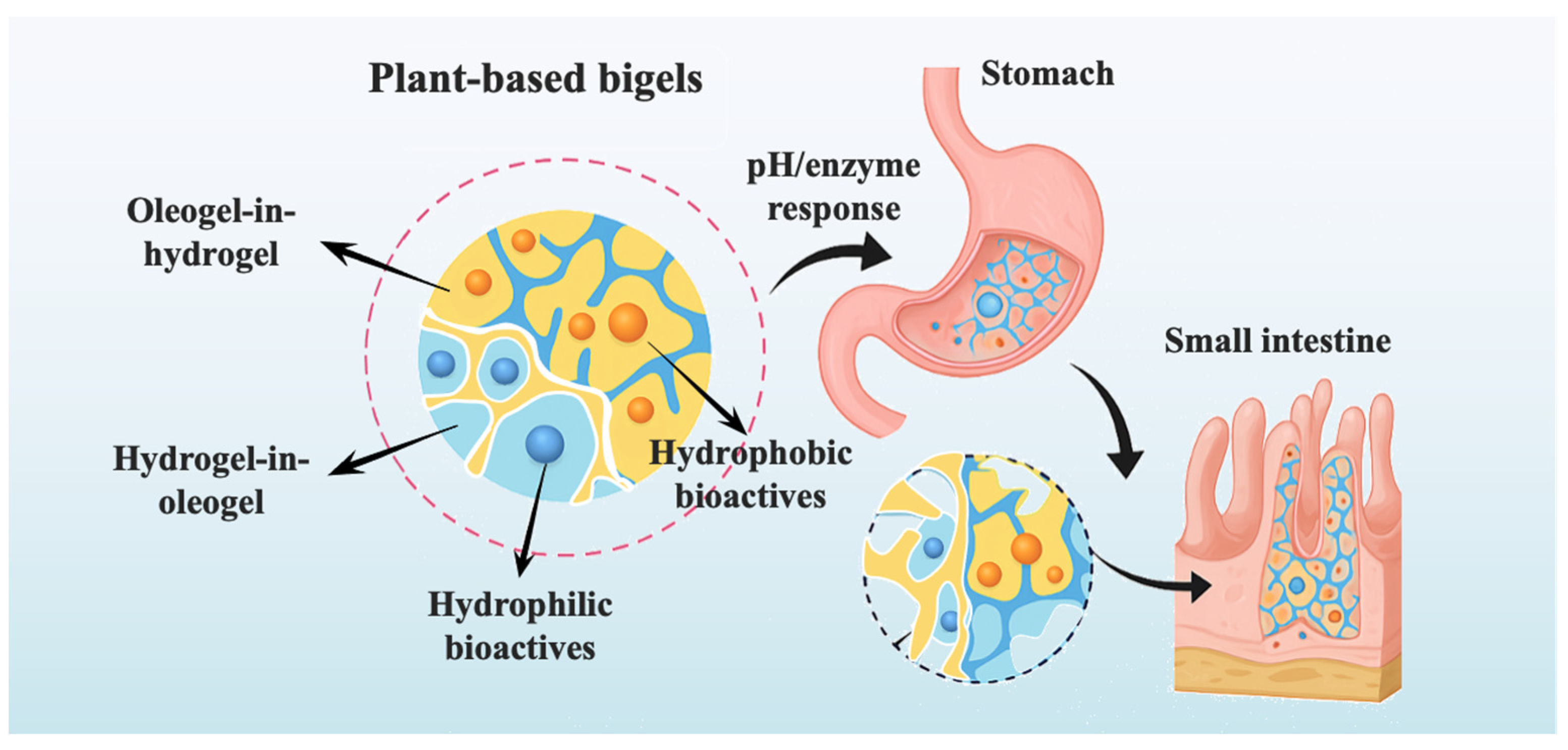
3. Functional Delivery Mechanisms and Potential Applications
3.1. Mechanistic Strategies for Bioactive Delivery
3.1.1. Encapsulation and Retention Mechanism of Bioactives
3.1.2. Controlled Release: Diffusion, Degradation, and Stimuli Response
3.1.3. Targeted Delivery in Gastrointestinal Environments
3.2. Functional Delivery Application of Bioactives in Plant-Based Bigels
3.2.1. Polyphenols and Flavonoids
3.2.2. Omega-3 Fatty Acids and Lipophilic Vitamins
3.2.3. Probiotic Encapsulation and Viability Enhancement
| Delivered Target | Oleogel | Hydrogel | Oleogel/Hydrogel Ratio (w/w) | Synthesis Parameters | Emulsifier/Additives | Key Structural Features | Key Findings | Highest Release Rate (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Lutein | Ethyl cellulose-sunflower oil oleogel (15% w/w) | Guar-xanthan gum hydrogel (1.5% w/w) | 25:75 50:50 75:25 | Oleogel at 80 °C; bigel 75 °C, 10,000 rpm, 5 min | Tween 80 (5% w/w of oil) | Hydrogel-dominant biphasic matrix with strong viscoelastic coupling | Bigels ↑ viscoelasticity & hardness; Gastric release ≈15%, intestine release up to 83%; Antioxidant activity ↑ in intestine. | 83.2 (bigel 25:75) | [103] |
| EGCG/Curcumin | GMS-corn oil oleogel (10% w/w) | 1% sodium alginate (CaCl2 crosslinked) | 10:90 20:80 30:70 | 500 rpm stirring; 0.7 mm dripping; 0.5 h crosslinking | Tween 20 (0.5%) | Transitioning from a porous to a compact and continuous network (at high oleogel fractions) | Higher oleogel ↓ swelling & oil leakage; EGCG encapsulation ↓, curcumin ↑; Curcumin retained ≈70% after 40 days. | N/A | [34] |
| EGCG/Quercetin | N/A (double emulsion W/O/W system) | Sodium alginate (SA, 3%) hydrogel beads with soybean protein isolate | 30:70 40:60 50:50 | Prepare W/O/W emulsion (3:7, 4:6, 5:5), dropwise into CaCl2 to form beads | None | Ca2+-crosslinked SA forming a porous network; Double emulsions embedded within SA network | Beads ↑ encapsulation efficiency of EGCG & quercetin; Beads slowed oil digestion; EGCG bioavailability ↑ significantly. | EE: EGCG ≈ 79–84%; Q ≈ 74–80% | [104] |
| Curcumin | 5% candelilla wax/corn oil oleogel | Potato protein hydrogel (15–25%) | 70:30 90:10 | 1300 rpm, 10 min, blending | None | A continuous hydrogel matrix with dispersed spherical oleogel droplets | Higher protein & oleogel ↑ hardness; Curcumin bioaccessibility ↓, stability ↑; Tunable textural properties for foods. | Bioaccessibility: 16.3%; Stability: 43.8% | [105] |
| Curcumin | 2% Span 60, almond oil | 2% w/v HPMC | 30:70 40:60 50:50 | Oleogel at 60 °C, 500 rpm, 10 min → add hydrogel, cooling | None | Almond oil–based oleogel droplets (OG) dispersed within a continuous HPMC hydrogel (HG) matrix | Optimal BG30 (70:30) bigel: pseudoplastic, minimal oil leaching; Enhanced skin deposition & wound healing; Non-toxic, ideal for topical sustained delivery. | N/A | [106] |
| Catechin/Curcumin | 2 wt% beeswax, algal oil | 2 wt% low acyl gellan gum | 20:80 40:60 50:50 60:40 80:20 | Mixed with 3000 rpm for 10 min at 70 °C | None | O5H5: Bicontinuous interpenetrating structure with indistinct phase boundaries | O5H5 bicontinuous structure ↑ matrix integrity; Delayed lipolysis, CUR/CAT bioaccessibility ↑; Strong rheological–release correlation. | Bioaccessibility: curcumin 68.16%; catechin 56.16% | [6] |
| β-Carotene | 20% monoglyceride/corn oil oleogel | 1.5% κ carrageenan hydrogel | 25:75 40:60 50:50 60:40 75:25 | Mixed at 80 °C, cooled rapidly | None | Bicontinuous structure with interpenetrating oil and water networks; GMS crystals stabilizing droplet surfaces | Higher oleogel ↑ strength & stability; 75% oleogel ↑ light/thermal protection; 75% oleogel gave highest intestinal release. | 80% in intestinal phase | [107] |
| Lycopene | 1% GMS + 1% BW/soybean oil | 0.3% high acyl gellan gum | 10:90 20:80 30:70 40:60 50:50 60:40 | 85 °C melt → 50 °C cool, 10,000 rpm homogenize 1 min | None | O/H structure with oil droplets dispersed in a continuous gellan gum matrix; transition toward more complex or bicontinuous systems | ↑ Oleogel → ↑ firmness & G′; High oleogel slowed gastric release; Thermoreversible, stable ≥3 months. | 85% (10–20% oleogel) | [78] |
| Ascorbic acid | Ethyl cellulose-sunflower oil oleogel (15% w/w) | Xanthan guar gum hydrogel (0.75% each) | 25:75 50:50 75:25 | 10,000 rpm, 70 °C, 5 min, rapid cooling | Tween 80 (10% w/w of oil) | Discrete irregular gel domains with lower aggregation; phase inversion from O/H to H/O above 50% oleogel | Controlled gastric release 75–87%; Higher oleogel ↑ hardness & viscoelasticity; Bigel 75:25 had highest bioaccessibility. | 87 (bigel 75:25) | [108] |
| Coenzyme Q10 | Beeswax/fish oil oleogel (10% BW) | 1% Carbopol hydrogel | 50:50 | Oleogel: 70 °C 15 min; Bigel: 800 rpm mix | Benzalkonium chloride (preservative) | Oleogel-in-hydrogel structure combining adhesive, viscous, and lipophilic properties | Bigel ↑ permeation & flux vs. gel/oleogel; Fish oil (EPA/DHA) enhanced skin permeability; NMR/docking confirmed fatty acid-CoQ10 interaction. | 0.514 mg/cm2 (24 h) | [93] |
| L. plantarum 299 v/Metronidazole | 9% Span 40/sunflower oil | 0.5% Polysaccharides (sodium alginate/CMC/maltodextrin/starch) | 50:50 | Organogel mixed dropwise with polysaccharide solution (50 °C) → vortex mixing → cooled to 25 °C | None | Oleogel-in-hydrogel structure with oil droplets dispersed in a continuous hydrogel phase; polysaccharide-based hydrogel network | Composition-dependent, sustained drug delivery; stable over >10 months; Branched polysaccharides enhanced L. plantarum viability in gastric/intestinal conditions. | Maximum viability of 105–106 cfu/g | [82] |
4. Challenges and Future Perspectives
4.1. Current Challenges for Plant-Based Bigels in Functional Delivery
4.2. Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| LMWGs | Low-molecular-weight gelators |
| HMWGs | High-molecular-weight gelators |
| EC | Ethyl cellulose |
| GMS | Glycerol monostearate |
| O/H | Oleogel-in-hydrogel |
| SPI | Soy protein isolate |
| CNCs | Cellulose nanocrystals |
| GI | Gastrointestinal |
| EGCG | Epigallocatechin gallate |
| PUFAs | Polyunsaturated fatty acids |
| EPA | Eicosapentaenoic acid |
| DHA | Docosahexaenoic acid |
| GRAS | Generally Recognized As Safe |
References
- Francavilla, A.; Corradini, M.G.; Joye, I.J. Bigels as delivery systems: Potential uses and applicability in food. Gels 2023, 9, 648. [Google Scholar] [CrossRef]
- Hashemi, B.; Assadpour, E.; Jafari, S.M. Bigels as novel carriers of bioactive compounds: Applications and research trends. Food Hydrocoll. 2024, 147, 109427. [Google Scholar] [CrossRef]
- Almeida, I.F.; Fernandes, A.; Fernandes, L.; Pena Ferreira, M.; Costa, P.; Bahia, M. Moisturizing effect of oleogel/hydrogel mixtures. Pharm. Dev. Technol. 2008, 13, 487–494. [Google Scholar] [CrossRef]
- Shakeel, A.; Lupi, F.R.; Gabriele, D.; Baldino, N.; De Cindio, B. Bigels: A unique class of materials for drug delivery applications. Soft Mater. 2018, 16, 77–93. [Google Scholar] [CrossRef]
- Yu, J.; Qiu, R.; Zheng, W.; Wang, K.; Liu, X.; Hu, Z.; Zhao, L. Multiphase gel-based functional inks in 3D food printing: A review on structural design and enhanced bioaccessibility of active ingredients. Food Res. Int. 2025, 217, 116787. [Google Scholar] [CrossRef]
- Chao, E.; Li, J.; Fan, L. Design of bigel-based co-delivery systems for lipophilic/hydrophilic nutraceuticals: Focus on rheological properties and in vitro digestive performance. Food Hydrocoll. 2025, 162, 111017. [Google Scholar] [CrossRef]
- Dhal, S.; Pradhan, B.K.; Pal, K. Bigel-based nutraceutical delivery systems. In Advances in Biopolymers for Food Science and Technology; Elsevier: Amsterdam, The Netherlands, 2024; pp. 65–87. [Google Scholar] [CrossRef]
- Gao, Y.; Xiong, Q.; Huang, Q.; Hu, Y.; Liu, R. Bigels as novel fat substitutes: Fat simulation characteristics, quality regulation, future trends and challenges. Trends Food Sci. Technol. 2025, 163, 105156. [Google Scholar] [CrossRef]
- Yi, H.J.; Kang, Y.-R.; Chang, Y.H. Structural and rheological properties of bigels formed with xanthan gum hydrogel and lecithin/glycerol monostearate oleogel. Int. J. Biol. Macromol. 2025, 306, 141549. [Google Scholar] [CrossRef]
- Dimakopoulou-Papazoglou, D.; Zampouni, K.; Katsanidis, E. Natural waxes as gelators in edible structured oil systems: A review. Gels 2025, 11, 656. [Google Scholar] [CrossRef] [PubMed]
- Nutter, J.; Shi, X.; Lamsal, B.; Acevedo, N.C. Plant-based bigels as a novel alternative to commercial solid fats in short dough products: Textural and structural properties of short dough and shortbread. Food Biosci. 2023, 54, 102865. [Google Scholar] [CrossRef]
- Wang, X.; Li, H.; Liu, Y.; Ding, S.; Jiang, L.; Wang, R. A novel edible solid fat substitute: Preparation of biphasic stabilized bigels based on glyceryl monolaurate and gellan gum. Int. J. Biol. Macromol. 2024, 263, 130081. [Google Scholar] [CrossRef]
- Hashemi, B.; Assadpour, E.; Wang, Y.; Jafari, S.M. Application of oleogels, hydrogels and bigels as novel edible inks for 3D/4D printing of food products. Adv. Colloid Interface Sci. 2025, 343, 103578. [Google Scholar] [CrossRef]
- Karimidastjerd, A.; Cetinkaya, T.; Tarahi, M.; Singh, L.; Konar, N.; Khiabani, A.H.; Toker, O.S. Novel approaches in food grade bigels properties and applications: A review. Int. J. Biol. Macromol. 2024, 283, 137424. [Google Scholar] [CrossRef]
- Martins, A.J.; Silva, P.; Maciel, F.; Pastrana, L.M.; Cunha, R.L.; Cerqueira, M.A.; Vicente, A.A. Hybrid gels: Influence of oleogel/hydrogel ratio on rheological and textural properties. Food Res. Int. 2019, 116, 1298–1305. [Google Scholar] [CrossRef]
- Moguiliansky, S.; Friedman, N.; Davidovich-Pinhas, M. The effect of transglutaminase on the structure and texture of plant-protein based bigel. Food Hydrocoll. 2025, 162, 110981. [Google Scholar] [CrossRef]
- Qiu, R.; Wang, K.; Tian, H.; Liu, X.; Liu, G.; Hu, Z.; Zhao, L. Analysis on the printability and rheological characteristics of bigel inks: Potential in 3D food printing. Food Hydrocoll. 2022, 129, 107675. [Google Scholar] [CrossRef]
- Machado, M.; Sousa, S.C.; Rodríguez-Alcalá, L.M.; Pintado, M.; Gomes, A.M. Bigels as delivery systems of bioactive fatty acids present in functional edible oils: Coconut, avocado, and pomegranate. Gels 2023, 9, 349. [Google Scholar] [CrossRef]
- Guo, J.; Gu, X.; Meng, Z. Customized 3D printing to build plant-based meats: Spirulina platensis protein-based Pickering emulsion gels as fat analogs. Innov. Food Sci. Emerg. Technol. 2024, 94, 103679. [Google Scholar] [CrossRef]
- Lee, G.R.; Baek, Y.; Jeong, E.; Lee, H.G. Development and characterization of a novel bigel system based on candelilla wax oleogel and guar gum hydrogel for heat-triggered release properties. Food Hydrocoll. 2024, 152, 109892. [Google Scholar] [CrossRef]
- Perera, P.; Mansouri, S.; Tang, Y. Cellulose-derivative based bigels: Stability and printability assessment for extrusion-based 3D printing. Food Hydrocoll. 2026, 170, 111642. [Google Scholar] [CrossRef]
- Xu, D.; Liu, Z.; An, Z.; Hu, L.; Li, H.; Mo, H.; Hati, S. Incorporation of probiotics into 3D printed Pickering emulsion gel stabilized by tea protein/xanthan gum. Food Chem. 2023, 409, 135289. [Google Scholar] [CrossRef]
- Fernandes, A.S.; Jacob-Lopez, E.; Zepka, L.Q.; de Rosso, V.V.; Roca, M. Bioactive compound-loaded food-grade bigels:(II) Delivery systems focusing on the bioaccessibility of chlorophylls and the effects of microstructure and 3D printing. Food Hydrocoll. 2025, 169, 111596. [Google Scholar] [CrossRef]
- Srivastava, S.; Singh, A. Bigels: A review on formulation, characterization, and application in the food industry. Trends Food Sci. Technol. 2025, 165, 105285. [Google Scholar] [CrossRef]
- Zampouni, K.; Dimakopoulou-Papazoglou, D.; Katsanidis, E. Food-grade bigel systems: Formulation, characterization, and applications for novel food product development. Gels 2024, 10, 712. [Google Scholar] [CrossRef]
- Chao, E.; Li, J.; Duan, Z.; Fan, L. Bigels as emerging biphasic systems: Properties, applications, and prospects in the food industry. Food Hydrocoll. 2024, 154, 110089. [Google Scholar] [CrossRef]
- Liu, D.; Xu, B.; Zhao, D.; Ma, Q.; Li, J.; Amjad, M.U.; Ding, Y.; Liu, L. Construction and application of a novel bigel system to prepare a nutritional cream. LWT 2024, 198, 116000. [Google Scholar] [CrossRef]
- Liu, G.; Wang, Y.; Yang, J.; Wang, Y.; He, H.; Mao, L. Roles of different polysaccharides on the structures of alginate-based Bigel beads and co-delivery of bioactives. Food Chem. X 2025, 27, 102359. [Google Scholar] [CrossRef]
- Hou, Y.; Wu, Y.; Ouyang, J. Novel bigel based on nanocellulose hydrogel and monoglyceride oleogel: Preparation, characteristics and application as fat substitute. Food Res. Int. 2024, 198, 115397. [Google Scholar] [CrossRef]
- Zheng, R.; Chen, Y.; Wang, Y.; Rogers, M.A.; Cao, Y.; Lan, Y. Microstructure and physical properties of novel bigel-based foamed emulsions. Food Hydrocoll. 2023, 134, 108097. [Google Scholar] [CrossRef]
- Shakouri, S.; Tehrani, M.M.; Koocheki, A.; Farhoosh, R.; Abdolshahi, A. The development and characterization of edible bigel a hydrogel/oleogel structure based on guar gum, walnut oil and rice bran wax for using as fat replacer. J. Polym. Environ. 2025, 33, 2058–2071. [Google Scholar] [CrossRef]
- Qiu, R.; Qiu, G.; Zhao, P.; Awais, M.; Fan, B.; Huang, Y.; Tong, L.; Wang, L.; Liu, L.; Wang, F. Regulation of rheological properties of soy protein isolate-beeswax based bigel inks for high-precision 3D printing. Food Hydrocoll. 2024, 153, 110052. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, L.; Zhang, Q.; Wang, Y.; Jiao, A.; Jin, Z. Development and characterisation of a novel bigel based on pea protein hydrogel and rice bran wax oleogel: Enhancement of rheological properties and freeze-thaw stability. Int. J. Biol. Macromol. 2024, 282, 136606. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Song, J.; Miao, S.; Gao, Y.; Mao, L. Alginate-based gel beads with bigel structures: Preparation, characterization and bioactive encapsulation. Food Hydrocoll. 2024, 146, 109294. [Google Scholar] [CrossRef]
- Zampouni, K.; Mouzakitis, C.; Lazaridou, A.; Moschakis, T.; Katsanidis, E. Physicochemical properties and microstructure of bigels formed with gelatin and κ-carrageenan hydrogels and monoglycerides in olive oil oleogels. Food Hydrocoll. 2023, 140, 108636. [Google Scholar] [CrossRef]
- Sanpedro-Díaz, M.; García-Hernández, A.B.; Gómez-Gómez, A.L.; Salgado-Cruz, J.; Ramos-Monroy, O.A.; Oliver-Espinoza, R.; Rivera-Vargas, G.A.; de la Paz Salgado-Cruz, M. Trends and future perspectives of polysaccharide-based bigels from seeds, vegetable oils, and waxes: A bibliometric review. Gels 2025, 11, 413. [Google Scholar] [CrossRef]
- Esposito, C.L.; Kirilov, P.; Roullin, V.G. Organogels, promising drug delivery systems: An update of state-of-the-art and recent applications. J. Control. Release 2018, 271, 1–20. [Google Scholar] [CrossRef]
- Davidovich-Pinhas, M. Oil structuring using polysaccharides. Curr. Opin. Food Sci. 2019, 27, 29–35. [Google Scholar] [CrossRef]
- Singh, A.; Auzanneau, F.-I.; Rogers, M. Advances in edible oleogel technologies—A decade in review. Food Res. Int. 2017, 97, 307–317. [Google Scholar] [CrossRef]
- Guo, Z.; Chen, Z.; Meng, Z. Bigels constructed from hybrid gelator systems: Bulk phase-interface stability and 3D printing. Food Funct. 2023, 14, 5078–5089. [Google Scholar] [CrossRef]
- Farzana, W.; Mahesh, S.; Sharma, S.; Syed, I.; Abdi, G.; Upadhyay, R. A comprehensive review on bigels as a potential replacement to solid fat in food applications. J. Food Qual. 2025, 2025, 2483241. [Google Scholar] [CrossRef]
- Alhasawi, F.M.; Rogers, M. Ternary phase diagram of β-sitosterol–γ-oryzanol–canola oil. J. Am. Oil Chem. Soc. 2013, 90, 1533–1540. [Google Scholar] [CrossRef]
- Manzoor, S.; Masoodi, F.; Naqash, F.; Rashid, R. Oleogels: Promising alternatives to solid fats for food applications. Food Hydrocoll. Health 2022, 2, 100058. [Google Scholar] [CrossRef]
- Lim, J.; Hwang, H.S.; Lee, S. Oil-structuring characterization of natural waxes in canola oil oleogels: Rheological, thermal, and oxidative properties. Appl. Biol. Chem. 2017, 60, 17–22. [Google Scholar] [CrossRef]
- Ramos-Souza, C.; Fernandes, A.S.; Mazzo, T.M.; Perrechil, F.; De Rosso, V.V. Fine-tuning carotenoid-enriched bigel formulations: Exploring the influence of Oleogel: Hydrogel ratio on physicochemical properties and 3D food printing. Food Hydrocoll. 2025, 164, 111202. [Google Scholar] [CrossRef]
- Yang, J.; Zheng, H.; Mo, Y.; Gao, Y.; Mao, L. Structural characterization of hydrogel-oleogel biphasic systems as affected by oleogelators. Food Res. Int. 2022, 158, 111536. [Google Scholar] [CrossRef]
- Liu, Z.; Hu, X.; Lu, S.; Xu, B.; Bai, C.; Ma, T.; Song, Y. Cellulose nanofiber/beeswax based bigels: Study of phase inversion and regulatory properties. Food Hydrocoll. 2025, 168, 111494. [Google Scholar] [CrossRef]
- Chen, Z.; Bian, F.; Cao, X.; Shi, Z.; Meng, Z. Novel bigels constructed from oleogels and hydrogels with contrary thermal characteristics: Phase inversion and 3D printing applications. Food Hydrocoll. 2023, 134, 108063. [Google Scholar] [CrossRef]
- Xie, D.; Hu, H.; Huang, Q.; Lu, X. Development and characterization of food-grade bigel system for 3D printing applications: Role of oleogel/hydrogel ratios and emulsifiers. Food Hydrocoll. 2023, 139, 108565. [Google Scholar] [CrossRef]
- Gao, Y.; Xiong, Q.; Zhao, S.; Huang, Q.; Hu, Y.; Liu, R. Mechanism of the influence of single-phase dynamic gelation process on the structure and properties of bigels. Food Hydrocoll. 2026, 171, 111837. [Google Scholar] [CrossRef]
- Martín-Illana, A.; Notario-Pérez, F.; Cazorla-Luna, R.; Ruiz-Caro, R.; Bonferoni, M.C.; Tamayo, A.; Veiga, M.D. Bigels as drug delivery systems: From their components to their applications. Drug Discov. Today 2022, 27, 1008–1026. [Google Scholar] [CrossRef]
- Clímaco, G.N.; Fasolin, L.H. Effect of the gelling mechanism on the physical properties of bigels based on whey protein isolate. Food Res. Int. 2024, 176, 113784. [Google Scholar] [CrossRef]
- Fasolin, L.H.; Martins, A.J.; Cerqueira, M.; Vicente, A. Modulating process parameters to change physical properties of bigels for food applications. Food Struct. 2021, 28, 100173. [Google Scholar] [CrossRef]
- Lin, X.; Liu, F.; Ma, Z.; Li, Y.; Zheng, H. Effects of different gelation mechanisms on the structural properties of bigels: A comparative study. Food Hydrocoll. 2025, 167, 111249. [Google Scholar] [CrossRef]
- Fasolin, L.H.; Vicente, A. Evaluation of process conditions in the bigels production: A multivariate analysis. In Proceedings of the 19th Gums & Stabilisers for the Food Industry Conference: Hydrocolloid Multifunctionality, Berlin, Germany, 27–30 June 2017. [Google Scholar]
- Contreras-Ramírez, J.I.; Patel, A.R.; Gallegos-Infante, J.A.; Toro-Vázquez, J.F.; Pérez-Martínez, J.D.; Rosas-Flores, W.; González-Laredo, R.F. Organogel-based emulsified systems, food applications, microstructural and rheological features—A review. Biointerface Res. Appl. Chem. 2022, 12, 1601–1627. [Google Scholar] [CrossRef]
- Su, L.Z.; Chen, Y.X.; Zhang, S.Q.; Hu, C.; He, J.B.; Zhang, W.N.; Han, L.J. Phase inversion of bigels tuned by the concentration of soy protein isolate in the binary hydrogel matrix. Food Hydrocoll. 2024, 155, 110176. [Google Scholar] [CrossRef]
- Saffold, A.C.; Acevedo, N.C. The effect of mono-diglycerides on the mechanical properties, microstructure, and physical stability of an edible rice bran wax–gelatin biphasic gel system. J. Am. Oil Chem. Soc. 2022, 99, 1033–1043. [Google Scholar] [CrossRef]
- Samui, T.; Goldenisky, D.; Rosen-Kligvasser, J.; Davidovich-Pinhas, M. The development and characterization of novel in-situ bigel formulation. Food Hydrocoll. 2021, 113, 106416. [Google Scholar] [CrossRef]
- Nutter, J.; Shi, X.; Lamsal, B.; Acevedo, N.C. Designing and characterizing multicomponent, plant-based bigels of rice bran wax, gums, and monoglycerides. Food Hydrocoll. 2023, 138, 108425. [Google Scholar] [CrossRef]
- Deng, Y.; Jiang, Y.; Lu, Y.; Zhao, Y.; Fan, F. Preparation and characteristics of TEMPO-oxidized bamboo shoot nanofibers based bigels system and steady delivery of Lactobacillus plantarum. Food Hydrocoll. 2025, 163, 111057. [Google Scholar] [CrossRef]
- Marand, M.J.; Pero, M.; Ahmadi, M.; Nayebzadeh, K.; Farhoodi, M.; Mirmoghtadaie, L.; Hosseini, S.M. Octenyl succinate starch as a hydrogelator and emulsifier for the fabrication of innovative bigels. Carbohydr. Polym. 2025, 356, 123228. [Google Scholar] [CrossRef]
- Luan, Q.; Zhou, W.; Zhang, H.; Bao, Y.; Zheng, M.; Shi, J.; Tang, H.; Huang, F. Cellulose-based composite macrogels from cellulose fiber and cellulose nanofiber as intestine delivery vehicles for probiotics. J. Agric. Food Chem. 2018, 66, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Han, J.; Xiao, Y.; Guo, R.; Liu, X.; Zhang, H.; Bi, Y.; Xu, X. Fabrication and characterization of novel food-grade bigels based on interfacial and bulk stabilization. Foods 2023, 12, 2546. [Google Scholar] [CrossRef]
- Wang, X.; Sun, L.; Zhang, X.; Jia, L.; Wang, S.; He, T.; Xie, F. Soy protein isolate-acylated anthocyanins self-assembled nanoparticles enhanced bigels physicochemical properties: Effect of amphiphilic nanoparticles on the bigels system. Food Res. Int. 2025, 218, 116913. [Google Scholar] [CrossRef] [PubMed]
- He, W.S.; Wang, Q.; Li, Z.; Li, J.; Zhao, L.; Li, J.; Tan, C.; Gong, F. Enhancing the stability and bioaccessibility of tree peony seed oil using layer-by-layer self-assembling bilayer emulsions. Antioxidants 2023, 12, 1128. [Google Scholar] [CrossRef]
- Wang, H.; Sun, Y.; Zhang, P.; Tu, Z.; Tan, M. Tailoring the properties of bigels by beeswax content for 3D food printing, astaxanthin delivery, and freshness sensing. Food Hydrocoll. 2026, 170, 111703. [Google Scholar] [CrossRef]
- Singh, V.K.; Banerjee, I.; Agarwal, T.; Pramanik, K.; Bhattacharya, M.K.; Pal, K. Guar gum and sesame oil based novel bigels for controlled drug delivery. Colloids Surf. B Biointerfaces 2014, 123, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lin, S.; Sun, N. Food gel-based systems for efficient delivery of bioactive ingredients: Design to application. Crit. Rev. Food Sci. Nutr. 2024, 64, 13193–13211. [Google Scholar] [CrossRef]
- Tan, C. Hydrogel delivery systems of functional substances for precision nutrition. Adv. Food Nutr. Res. 2024, 112, 301–345. [Google Scholar] [CrossRef]
- Okonkwo, C.E.; Ofoedu, C.E.; Hussain, S.Z.; Adeyanju, A.A.; Naseer, B.; Inyinbor, A.A.; Olaniran, A.F.; Kamal-Eldin, A. Application of biogels for bioactives delivery: Recent developments and future research insights. Appl. Food Res. 2022, 2, 100238. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, J.; Miao, W.; Lin, Q.; Ji, H.; Li, X.; Mcclements, D.J.; Jin, Z.; Qiu, C. Recent advances in bio-based co-delivery systems for food bioactive compounds: A review. Food Biosci. 2025, 63, 105758. [Google Scholar] [CrossRef]
- Cheng, H.; Chen, W.; Jiang, J.; Khan, M.A.; Liang, L. A comprehensive review of protein-based carriers with simple structures for the co-encapsulation of bioactive agents. Compr. Rev. Food Sci. Food Saf. 2023, 22, 2017–2042. [Google Scholar] [CrossRef]
- Misra, S.; Pandey, P.; Mishra, H.N. Novel approaches for co-encapsulation of probiotic bacteria with bioactive compounds, their health benefits and functional food product development: A review. Trends Food Sci. Technol. 2021, 109, 340–351. [Google Scholar] [CrossRef]
- Zhao, L.; Li, J.; Ding, Y.; Sun, L. Preparation and characterization of a novel longzhua mushroom polysaccharide hydrogel and slow-release behavior of encapsulated rambutan peel polyphenols. Foods 2024, 13, 1711. [Google Scholar] [CrossRef]
- Mao, L.; Lu, Y.; Cui, M.; Miao, S.; Gao, Y. Design of gel structures in water and oil phases for improved delivery of bioactive food ingredients. Crit. Rev. Food Sci. Nutr. 2020, 60, 1651–1666. [Google Scholar] [CrossRef]
- Habibullah, S.; Mohanty, B.; Babu, A.; Sivakumar, G.; Maji, S.; Pal, K.; Sagiri, S.S. Design of plant-based bigels by incorporating candelilla wax oleogel and xanthan gum hydrogel for oral curcumin delivery. Int. J. Biol. Macromol. 2025, 318, 145312. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Gao, J.; Han, L.; Han, K.; Wei, W.; Wu, T.; Li, J.; Zhang, M. Development and characterization of novel bigels based on monoglyceride-beeswax oleogel and high acyl gellan gum hydrogel for lycopene delivery. Food Chem. 2021, 365, 130419. [Google Scholar] [CrossRef]
- Santos, T.P.; Cunha, R.L. Role of process variables on the formation and in vitro digestion of gellan gels. Carbohydr. Polym. 2018, 192, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Chen, L.; McClements, D.J.; Qiu, C.; Li, C.; Zhang, Z.; Miao, M.; Tian, Y.; Zhu, K.; Jin, Z. Stimulus-responsive hydrogels in food science: A review. Food Hydrocoll. 2022, 124, 107218. [Google Scholar] [CrossRef]
- Koetting, M.C.; Peters, J.T.; Steichen, S.D.; Peppas, N.A. Stimulus-responsive hydrogels: Theory, modern advances, and applications. Mater. Sci. Eng. R Rep. 2015, 93, 1–49. [Google Scholar] [CrossRef]
- Behera, B.; Sagiri, S.S.; Singh, V.K.; Pal, K.; Anis, A. Mechanical properties and delivery of drug/probiotics from starch and non-starch based novel bigels: A comparative study. Starch-Stärke 2014, 66, 865–879. [Google Scholar] [CrossRef]
- Huang, W.C.; Wang, W.; Wang, W.; Hao, Y.; Xue, C.; Mao, X. A double-layer polysaccharide hydrogel (DPH) for the enhanced intestine-targeted oral delivery of probiotics. Engineering 2024, 34, 187–194. [Google Scholar] [CrossRef]
- Luo, N.; Ye, A.; Wolber, F.M.; Singh, H. Effect of gel structure on the in vitro gastrointestinal digestion behaviour of whey protein emulsion gels and the bioaccessibility of capsaicinoids. Molecules 2021, 26, 1379. [Google Scholar] [CrossRef]
- Liu, L.; Tian, W.; Chen, Μ.; Huang, Y.; Xiao, J. Oral sensation and gastrointestinal digestive profiles of bigels tuned by the mass ratio of konjac glucomannan to gelatin in the binary hydrogel matrix. Carbohydr. Polym. 2023, 312, 120765. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Hu, H.; Huang, Q.; Lu, X. Influence of oleogel/hydrogel ratios and emulsifiers on structural and digestion properties of food-grade 3D printed bigels as carriers for quercetin and catechin. Food Hydrocoll. 2023, 144, 108948. [Google Scholar] [CrossRef]
- Zhai, X.; Sun, Y.; Cen, S.; Wang, X.; Zhang, J.; Yang, Z.; Li, Y.; Wang, X.; Zhou, C.; Arslan, M. Anthocyanins-encapsulated 3D-printable bigels: A colorimetric and leaching-resistant volatile amines sensor for intelligent food packaging. Food Hydrocoll. 2022, 133, 107989. [Google Scholar] [CrossRef]
- Li, L.; Feng, X.; Teng, F.; Geng, M.; Li, Y. Insights into beeswax-gelatin/carboxymethyl chitosan bigel systems: Structural-property relationships governing dual encapsulation of hydrophilic and hydrophobic bioactive compounds. Food Chem. 2025, 493, 145942. [Google Scholar] [CrossRef]
- Lu, Y.; Zhong, Y.; Guo, X.; Zhang, J.; Gao, Y.; Mao, L. Structural modification of O/W bigels by glycerol monostearate for improved co-delivery of curcumin and epigallocatechin gallate. ACS Food Sci. Technol. 2022, 2, 975–983. [Google Scholar] [CrossRef]
- Qiu, R.; Liu, X.; Tian, H.; Hu, Z.; Wang, K.; Zhao, L. Exploration of bigel 4D printing with spontaneous colour change for monitoring bio-actives kinetic behaviour based on the dual-units 3D printer. J. Food Eng. 2024, 367, 111861. [Google Scholar] [CrossRef]
- Aung, T.; Kim, C.Y.; Kim, M.J. Fabrication and characterization of novel sericin bigels for the co-encapsulation of bioactive ingredients and in vitro gastrointestinal release profile. Food Hydrocoll. 2025, 166, 111386. [Google Scholar] [CrossRef]
- Esmaeilinezhad, A.; Abbasi, H. Effect of Zein-Persian Gum Water-in-Oleogels on Quality Characteristics of Unsaturated Fatty Acid-Rich and Low-Fat Croissant. Food Sci. Nutr. 2025, 13, e4649. [Google Scholar] [CrossRef]
- Zulfakar, M.H.; Chan, L.M.; Rehman, K.; Wai, L.K.; Heard, C.M. Coenzyme Q10-loaded fish oil-based bigel system: Probing the delivery across porcine skin and possible interaction with fish oil fatty acids. Aaps Pharmscitech 2018, 19, 1116–1123. [Google Scholar] [CrossRef]
- Rehman, K.; Amin, M.C.I.M.; Zulfakar, M.H. Development and physical characterization of polymer-fish oil bigel (hydrogel/oleogel) system as a transdermal drug delivery vehicle. J. Oleo Sci. 2014, 63, 961–970. [Google Scholar] [CrossRef]
- Martinez, R.M.; Magalhaes, W.V.; da Silva Sufi, B.; Padovani, G.; Nazato, L.I.S.; Velasco, M.V.R.; da Silva Lannes, S.C.; Baby, A.R. Vitamin E-loaded bigels and emulsions: Physicochemical characterization and potential biological application. Colloids Surf. B Biointerfaces 2021, 201, 111651. [Google Scholar] [CrossRef]
- Li, C.; Xu, Y.; Zhang, Y.; Shen, Y.; Deng, X.; Wang, F. Novel bigels based on walnut oil oleogel and chitosan hydrogel: Preparation, characterization, and application as food spread. Int. J. Biol. Macromol. 2024, 260, 129530. [Google Scholar] [CrossRef]
- Zampouni, K.; Sideris, N.; Tsavdaris, E.; Katsanidis, E. On the structural and mechanical properties of mixed coconut and olive oil oleogels and bigels. Int. J. Biol. Macromol. 2024, 268, 131942. [Google Scholar] [CrossRef]
- Bollom, M.A.; Clark, S.; Acevedo, N.C. Edible lecithin, stearic acid, and whey protein bigels enhance survival of probiotics during in vitro digestion. Food Biosci. 2021, 39, 100813. [Google Scholar] [CrossRef]
- Sagiri, S.S.; Singh, V.K.; Kulanthaivel, S.; Banerjee, I.; Basak, P.; Battachrya, M.; Pal, K. Stearate organogel–gelatin hydrogel based bigels: Physicochemical, thermal, mechanical characterizations and in vitro drug delivery applications. J. Mech. Behav. Biomed. Mater. 2015, 43, 1–17. [Google Scholar] [CrossRef]
- Tamašauskaitė, L.; Minelgaitė, V.; Šipailienė, A.; Vinauskienė, R.; Eisinaitė, V.; Leskauskaitė, D. Bigel matrix loaded with probiotic bacteria and prebiotic dietary fibers from berry pomace suitable for the development of probiotic butter spread product. Gels 2024, 10, 349. [Google Scholar] [CrossRef] [PubMed]
- Özer, D.; Akin, S.; Özer, B. Effect of inulin and lactulose on survival of Lactobacillus acidophilusla-5 and Bifidobacterium bifidum bb-02 in Acidophilus-bifidus yoghurt. Food Sci. Technol. Int. 2005, 11, 19–24. [Google Scholar] [CrossRef]
- Zhuang, X.; Clark, S.; Acevedo, N. Bigels—Oleocolloid matrices—As probiotic protective systems in yogurt. J. Food Sci. 2021, 86, 4892–4900. [Google Scholar] [CrossRef]
- Kaimal, A.M.; Singhal, R.S. Bigels for controlled gastric release of ascorbic acid: Impact on rheology, texture, thermal stability and antioxidant activity. Food Hydrocoll. Health 2023, 4, 100171. [Google Scholar] [CrossRef]
- Hu, M.; Liu, G.; Zhang, W.; Du, X.; Qi, B.; Li, Y. Co-encapsulation of (–)-epigallocatechin-3-gallate and quercetin in double emulsion hydrogel beads: Microstructures, functional properties, and digestion behaviors. Food Chem. 2022, 373, 131427. [Google Scholar] [CrossRef]
- Gonçalves, R.F.; Zhou, H.; Vicente, A.A.; Pinheiro, A.C.; McClements, D.J. Plant-based bigels for delivery of bioactive compounds: Influence of hydrogel: Oleogel ratio and protein concentration on their physicochemical properties. Food Hydrocoll. 2024, 150, 109721. [Google Scholar] [CrossRef]
- Liston, L.S.; Phanapithakkun, S.; Pimsarn, N.; Dangkaokhia, N.; Nakasan, T.; Sae Yoon, A.; Todo, H.; Srichana, T.; Sawatdee, S.; See, G.L. Formulation development and characterization of bigels containing curcumin for topical skin delivery. AAPS PharmSciTech 2025, 26, 175. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Mao, L.; Cui, M.; Liu, J.; Gao, Y. Development of food-grade bigels based on κ-carrageenan hydrogel and monoglyceride oleogels as carriers for β-carotene: Roles of oleogel fraction. Food Hydrocoll. 2020, 105, 105855. [Google Scholar] [CrossRef]
- Kaimal, A.M.; Singhal, R.S. A bigel based formulation protects lutein better in the gastric environment with controlled release and antioxidant profile than other gel based systems. Food Chem. 2023, 423, 136304. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, C.; Yan, X.; Li, D.; Zeng, Z.; Zhao, Q.; Zhao, X.; Wang, S. Plant-Based Bigels for Functional Delivery: Advances in Structural Design and Stabilization Strategies. Foods 2025, 14, 3699. https://doi.org/10.3390/foods14213699
Cheng C, Yan X, Li D, Zeng Z, Zhao Q, Zhao X, Wang S. Plant-Based Bigels for Functional Delivery: Advances in Structural Design and Stabilization Strategies. Foods. 2025; 14(21):3699. https://doi.org/10.3390/foods14213699
Chicago/Turabian StyleCheng, Chao, Xianghui Yan, Dongze Li, Zheling Zeng, Qiangzhong Zhao, Xiujie Zhao, and Shaoyun Wang. 2025. "Plant-Based Bigels for Functional Delivery: Advances in Structural Design and Stabilization Strategies" Foods 14, no. 21: 3699. https://doi.org/10.3390/foods14213699
APA StyleCheng, C., Yan, X., Li, D., Zeng, Z., Zhao, Q., Zhao, X., & Wang, S. (2025). Plant-Based Bigels for Functional Delivery: Advances in Structural Design and Stabilization Strategies. Foods, 14(21), 3699. https://doi.org/10.3390/foods14213699








