Abstract
A huge amount of waste is produced annually by the food processing industry which must be valorized into high-value products. Therefore, the aim of this work involves the use of such wastes for production of β-glucans from medicinal basidiomycete strains which are powerful biological response modifiers in several clinical disorders. The production of β-glucans from basidiomycete strains in submerged fermentation was monitored by using monoclonal antibodies of the IgG and IgM classes as well as by Congo red assay in the presence of several agro-industrial waste products such as milk permeate, waste coffee grounds, orange peels and rice husks. Subsequently, these β-glucans were purified by using gel filtration and ion-exchange chromatography. FTIR analysis of several β-glucans was carried out to investigate their structural properties. The adsorption of β-glucans on microtiter plates was dependent on the temperature as well as on the time period of immobilization for ELISA. These mAbs can be used in a competitive ELISA for detection and quantification of β-glucans from basidiomycete mushrooms.
1. Introduction
The bioeconomy involves the use of renewable biological resources to produce basic precursors for energy, chemicals and material sources [1]. It may play a crucial role in the use of renewable raw materials for production of added-value substances in several fields of industrial biotechnology such as food, feed, paper and pulp and biofuel production [2]. The food processing industry and agriculture annually produce large volumes of wastes which are an environmental issue as well as a disposal problem. These wastes contain a large diversity of functional chemical substances which could potentially be used as a source of nutrients [3,4,5] for other organisms, plants or fungi/mushrooms. For instance, basidiomycete mushroom strains have been known for many centuries to grow on agricultural residues, which contain fine chemicals such as enzymes, β-D-glucans, lectins, vitamins, lipids and several secondary metabolites [6,7]. Among these fine chemicals, β-D-glucans play a major role as biological response modifiers (BRM) in various clinical disorders such as diabetes, cancer, HIV and immunological, cardiovascular and neurological disorders [8,9,10,11,12]. Submerged and solid-state cultivation of basidiomycete mushrooms in agro-industrial wastes are considered as an efficient, scalable and reproducible technique for production of biomass, metabolites and fine chemicals [6]. There is a huge range of wastes that can be used for cultivation of basidiomycete strains such as fruit peels and pomaces, agricultural straw and dairy wastes. Several basidiomycete mushrooms have been used in these bioconversion processes such as Pleurotus sp. Agaricus sp., Cordyseps sp. and Hericium sp. [6,13].
Basidiomycete medicinal mushroom strains exhibit the synthesis of a heterogeneous mixture of several β-D-glucans of different sizes which can be either intracellular or extracellular, and they can occur either bound to proteins or in their free forms [6,13]. It is of great scientific interest to investigate the purification of such β-D-glucans in homogeneous preparations as well as their structure–function relationship [14,15]. Therefore, it is of crucial interest to use specific assay methods for monitoring β-D-glucans production in solid-state and submerged fermentations from basidiomycete strains. Furthermore, purification procedures for such β-D-glucans require the use of specific assay methods to monitor their levels with chromatographic techniques. Several reports have been published in the literature about extraction and purification of mushroom β-D-glucans which involve hot or cold water, acid and alkali extractions, microwave-assisted, enzyme-assisted and ultrasound-assisted extractions and gel permeation chromatography [16,17].
Some colorimetric and fluorimetric assay methods have been published in the literature for quantification of β-D-glucans by using phenol-sulphuric acid, Congo red and alcian blue as well as aniline blue [18,19,20,21]. Regarding ELISA methods, there are few reports on β-D-glucan assays [22,23,24].
The present work involves the use of monoclonal antibodies of the IgG and IgM classes to monitor the production of β-D-glucans in basidiomycete strains by using several agro-industrial wastes. These β-D-glucans were extracted and purified by ion-exchange and gel filtration chromatography, and they were characterized by immunochemical techniques and FTIR analysis.
2. Materials and Methods
2.1. Chemicals
All chemicals and materials were supplied as described previously [24]. DEAE-cellulose anion-exchange media DE-52 GE Whatman® were obtained from Whatman, UK.
Milk permeate, orange and apple peels, pine sawdust, yellow lupine, rice husk, apple pomace, yellow lupin and waste coffee grounds were homemade.
2.2. Mushroom Samples
Ganoderma carnosum was obtained from old growth forests of the Olympic Peninsula in Port Townsend—WA (USA). Pleurotus ostreatus mushroom stems were kindly obtained from a mushroom grower in Amsterdam (Netherlands).
Agaricus blazei, Hericium erinaceus, Coriolus versicolor, Inonotus obliquus, Polyporus umbellatus, Poria cocos, Ganoderma lucidum and Lentinula edodes young fruiting body powders were supplied by Mycology Research Laboratory, Ltd. (Luton, UK).
2.3. Methods
2.3.1. Maintenance and Growth Conditions of Mushroom Strains
The strains of Pleurotus ostreatus and Ganoderma carnosum were maintained at 4 °C on PDA.
2.3.2. Production of Extracellular β-D-Glucans from Pleurotus ostreatus and Ganoderma carnosum in Culture Media Containing Agro-Industrial Wastes
Pleurotus ostreatus and Ganoderma carnosum were grown in culture media containing 1 g/L KH2PO4, 1 g/L MgSO4, 1 g/L (NH4)2SO4 and 4 g/L of agro-industrial waste (i.e., either rice husks, 40 g/L orange peels or waste coffee grounds). Actively growing mycelia of either Pleurotus ostreatus or Ganoderma carnosum were transferred in circles with diameters of 5 mm of PDA culture with a sterilized cutter to the seed culture medium. The culture was grown in 1000 mL Erlenmeyer flasks containing 400 mL medium and incubated at 28 °C on an orbital shaker at 150 rpm for 14 days. The fermentation process was monitored by using mAbs 3F8_3H7 and 1E6_1E8_B5 by indirect ELISA to detect the β-glucan in the fermentation broth as well as protein and total polysaccharide levels at 0, 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10 days and 0, 3, 5, 6, 7, 10, 11 and 14 days of fermentation with milk permeate and waste coffee grounds, respectively.
2.3.3. Production and Isolation of Extracellular β-D-Glucans from Pleurotus ostreatus
Pleurotus ostreatus was grown as described previously [24]. Mycelia biomass was separated from culture broth by vacuum filtration and extracellular β-D-glucans (EBG) in the culture filtrate were precipitated with 95% (v/v) ethanol. Precipitated β-D-glucans were dissolved in 15 mM phosphate-buffered saline pH 7.2 (PBS) [25].
2.3.4. Production of Intracellular β-D-Glucans from Ganoderma carnosum
Ganoderma carnosum was grown in culture media containing 1 g/L KH2PO4, 40 g/L orange peels and 2 g/L MgSO4 at pH 5.8, 200 rpm at 25 °C for 10 days in an orbital shaker.
2.3.5. Isolation of Intracellular β-D-Glucans
Biomass from the fermentation broth of Pleurotus ostreatus and Ganoderma carnosum was separated by filtration in a Büchner funnel [25].
The isolation of intracellular β-D-glucans (IBG) from mycelial biomass pellets from Pleurotus ostreatus and Ganoderma carnosum and from young fruiting body powder from Agaricus blazei, Ganoderma lucidum, Hericium erinaceus, Coriolus versicolor, Lentinula edodes, Pleurotus ostreatus, Inonotus obliquus, Auricularia auricula, Grifola frondosa, Polyporus umbellatus, Cordyseps sinensis and Poria cocos was performed by multistep water extraction followed by extraction with alkali and acidic solutions as described previously [20]. Briefly, FW1, FW2, FKOH, FHCl and FNaOH represent samples which were extraction with cold H2O, hot H2O, KOH, HCl and NaOH, respectively [20].
These unit operations of extraction were also performed for EBG from Pleurotus ostreatus, which was recovered by precipitation with 95% (v/v) ethanol.
2.3.6. Total Polysaccharides and Protein Assays
Total polysaccharides in several samples were assayed by the phenol–sulfuric acid method, using polygalacturonic acid as the standard [19], whereas protein determination was performed by the Coomassie blue dye-binding method with BSA as the protein standard [26].
2.3.7. Congo Red Assay for Specific Determination of β-D-Glucans with Triple Helical Structure
Congo red dye colorimetric assay was performed to quantify the levels of β-D-glucans in various samples as described previously, using β-D-glucan from barley as the standard [20].
2.3.8. mAbs Against EBG from Pleurotus ostreatus
The hybridoma cell line 3F8_3H7 secreting mAbs of the IgM class and the hybridoma cell line 1E6_1E8_B5 secreting mAbs of the IgG class against EBG from Pleurotus ostreatus were produced previously [24,25]. The mAbs were produced from 3F8_3H7 and 1E6_1E8_B5 clones in culture in vitro (RPMI 1640 + 10% (v/v) FBS) at 37 °C and 5% CO2.
2.3.9. Indirect Enzyme-Linked ImmunoSorbent Assay (ELISA)
In order to detect β-D-glucans in isolated fractions from the mushroom strains and in fermentation broths, indirect ELISA was performed by using mAbs 3F8_3H7 and 1E6_1E8_B5, as reported previously [24].
2.3.10. Study of the Effects of Temperature and Time on Adsorption of β-D-Glucans in PolySorp® 96-Well Plates
Temperature (4 and 37 °C) and time (1, 24, 48, 72, 96 and 120 h) of antigen immobilization on flat-bottom PolySorp® 96-well plates were varied as reported previously [24]. The indirect ELISA method was carried out as reported above.
2.3.11. Purification of EBG by Gel Filtration Chromatography
Extracellular β-D-glucans from Pleurotus osreatus were purified by gel filtration chromatography on a Sephacryl S-300 HR column (1 cm × 100 cm), which was eluted with PBS at a flow rate of 40 mL h−1. Column fractions were analyzed for total polysaccharides at A200 and proteins at A280 and monitored by using 3F8_3H7 and 1E6_1E8_B5 mAbs in indirect ELISA.
2.3.12. Purification of EBG by Anion-Exchange Chromatography
Extracellular β-D-glucans from Pleurotus osreatus were also purified by anion-exchange chromatography on a DEAE-cellulose column (1 × 5 cm) which was previously equilibrated with 50 mM sodium phosphate buffer at a pH of 7.8. The EBG were desorbed from the column with a linear gradient of 0–1 M NaCl in 50 mM sodium phosphate buffer with a pH of 7.8 at a flow rate of 20 mL/h. At the end of the linear gradient, the last four fractions were eluted with 2 M NaCl. The fractions (1.5 mL) were collected and analyzed for total polysaccharides at A200 and proteins at A280, as well as with mAbs 3F8 _3H7 and 1E6_1E8_B5.
2.3.13. FTIR Analysis of β-D-Glucans
In order to exploit the structural features of β-D-glucans, FTIR analysis was performed. All samples were previously lyophilized with a UNICRYO MC2L. Infrared spectra were obtained on a Bruker Vertex 70 (with OPUS 5.5 software) as KBr pellets with a total of 128 scans at a resolution of 2 cm−1 in a range of 400–4000 cm−1.
2.3.14. Interaction of mAbs with Native and Heat-Treated Antigens
The interaction of mAbs 1E6_1E8_B5 with native and heat-treated β-glucans was also investigated by indirect ELISA by using EBG from Pleurotus ostreatus and KOH extraction fractions from Coriolus versicolor and Agaricus blazei as reported previously [24]. Briefly, β-D-glucans were adsorbed either immediately in the microplates after heating or it was cooled in an ice bath for 10 min before it was adsorbed in microplates in order to study the mAb activity. In the present method, the EC50 is defined as the quantity of Mab required to give a 50% response on the dose–response sigmoid curve plotted with increasing amounts of mAbs [24]. The EC50 data were obtained by using GraphPad Prism software v.10.6.1
2.3.15. Assay of β-Glucans by Competitive ELISA
A fixed amount of mAb 1E6_1E8_B5 was incubated with different amounts (0–25 μg/mL) of selected antigens (i.e., FNaOH from Lentinula edodes and FKOH from Hericium erinaceus) at 37 °C for 1 h, as described previously [24].
2.3.16. Statistical Analysis
SigmaPlot 12.0 (2011–2012 Systat Software Inc.) was used to carry out figures in this work. Experimental results were means of three parallel measurements and the results were presented as mean values ± standard deviation (SD). All other statistical analyses were carried out with the Excel software 20132024 package (Academic License, Microsoft of Portugal). Statistical analysis was performed using one-way variance analysis (ANOVA). The significance of the p-value is indicated with asterisks (*, **, ***, ****, *****, ******) which indicate significance of the p-value less than 0.1, 0.05, 0.01, 0.005, 0.001 and 0.0001, respectively.
3. Results and Discussion
3.1. Production of β-D-Glucans by Submerged Fermentation on Agro-Industrial Wastes
Our aim was the production of β-glucans from basidiomycete mushroom strains using agro-industrial wastes. The data presented in Figure 1 and Figure 2 show that the fermentation runs were monitored both by IgG and IgM mAbs.
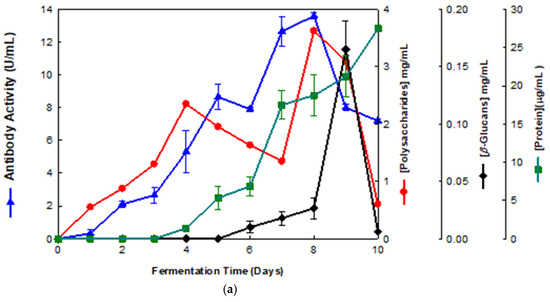
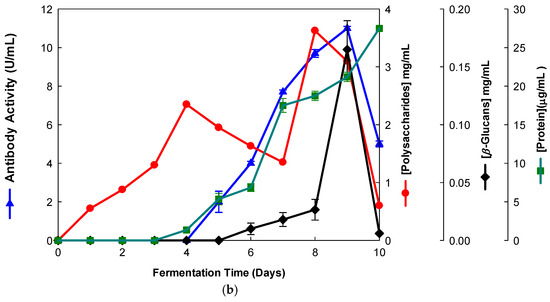
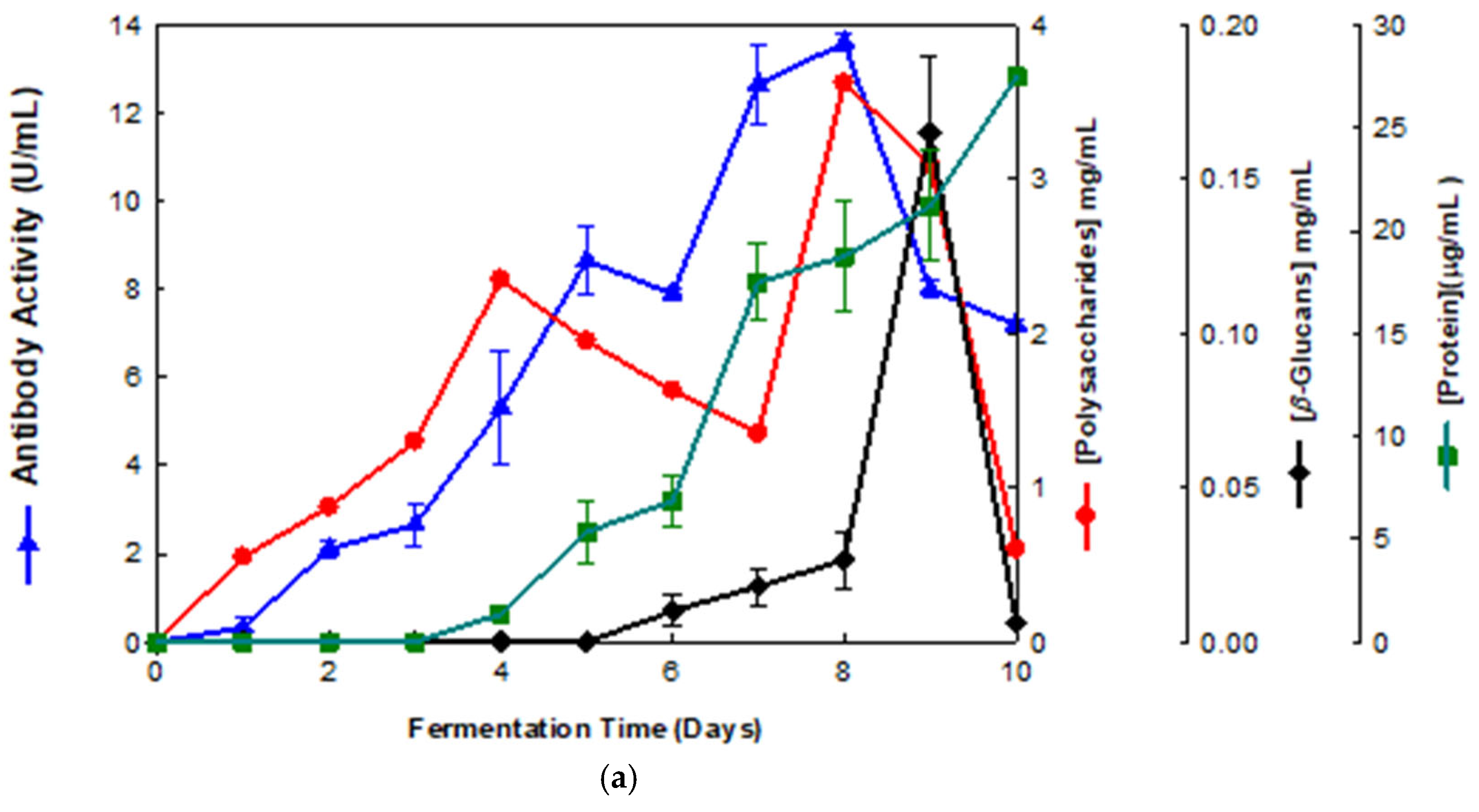
Figure 1.
Production of β-D-glucans from Pleurotus ostreatus in culture medium containing milk permeate. The fermentation broth was monitored for levels of protein, total polysaccharides, β-glucans and immunochemical detection of β-D-glucans by indirect ELISA using (a) mAb 3F8_3H7 and (b) mAb 1E6_1E8_B5.
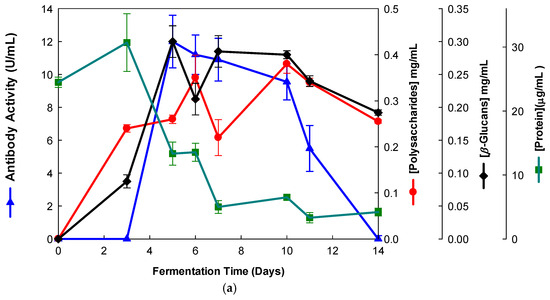
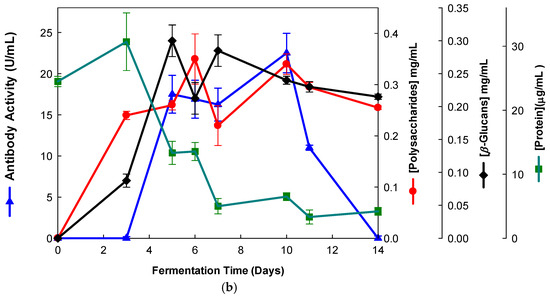
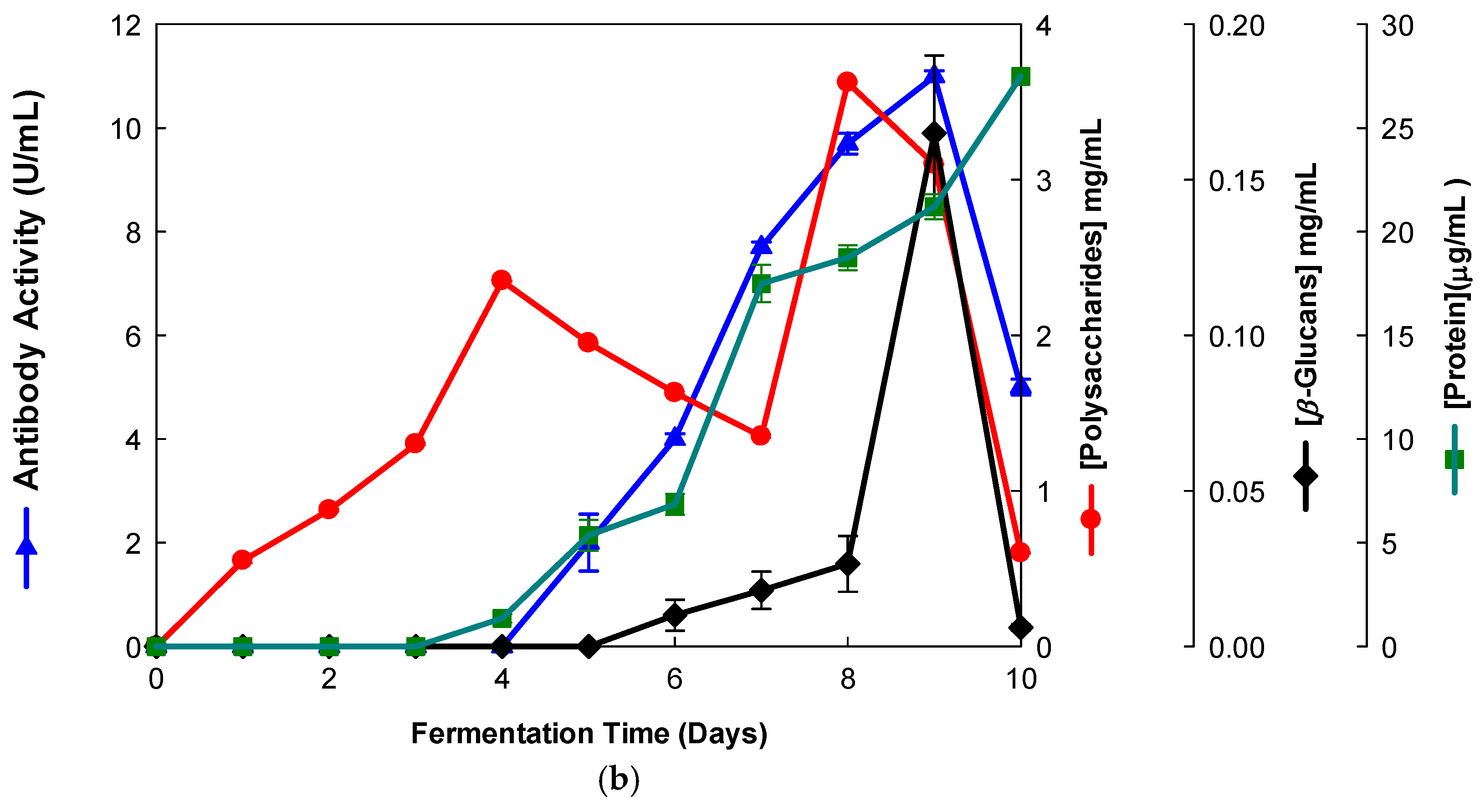
Figure 2.
Production of β-D-glucans from Ganoderma carnosum in culture medium containing waste coffee grounds. The fermentation broth was monitored for levels of protein, total polysaccharides, β-D-glucans and immunochemical detection of β-D-glucans by indirect ELISA using (a) mAb 3F8_3H7 and (b) mAb 1E6_1E8_B5.
Several fermentation runs were carried out by using both of the basidiomycete strains which were grown in several agro-industrial wastes as shown in Table 1. The peak production of β-D-glucans from Pleurotus ostreatus by submerged fermentation occurred on the 9th day of fermentation by using milk permeate (Figure 1a,b). However, the fermentation profile for Ganoderma carnosum grown in waste coffee grounds exhibited peak production of β-D-glucan on the 5th day of fermentation (Figure 2). The data in Table 1 strongly suggest that peak production of β-D-glucan occurred in the range of the 5th–10th days of fermentation for both strains grown in agro-industrial wastes. Waste coffee grounds have been revealed to be rich in nitrogen, but they may contain some growth inhibitors such as caffeine [27]. This waste has been shown to promote mushroom growth and β-D-glucan production but this is dependent on the basidiomycete mushroom strain used. Milk permeate contains high levels of lactose and protein; therefore, it is suitable for mushroom fermentation and β-D-glucan production because of its high levels of carbohydrate content. Rice husks have high levels of cellulose and silica but they contain low levels of nitrogen, and they can be used for mushroom growth and β-D-glucan production in conjunction with other growth substrates. Orange and apple peels contain pectin and cellulose and low nitrogen content which can be used for mushroom growth and β-D-glucan production mixed with other growth substrates [28,29]. In all cases, a suitable mushroom strain must be used for β-D-glucan production, as each fungal strain will behave differently for the substrate used. The comparative analysis of these data on β-D-glucan production with reports in the literature is rather difficult because the culture media, mushroom strains and growth conditions are significantly different from those of the present study.

Table 1.
Fermentation runs by using these two basidiomycete strains in the presence of several agro-industrial wastes.
It must be stressed that we have also used some other agro-industrial wastes for β-D-glucan production in these two strains, such as apple pomace, sawdust, yellow lupin and rice bran (raw data information and Dataset)
3.2. Purification of Basidiomycete Mushroom β-D-Glucans
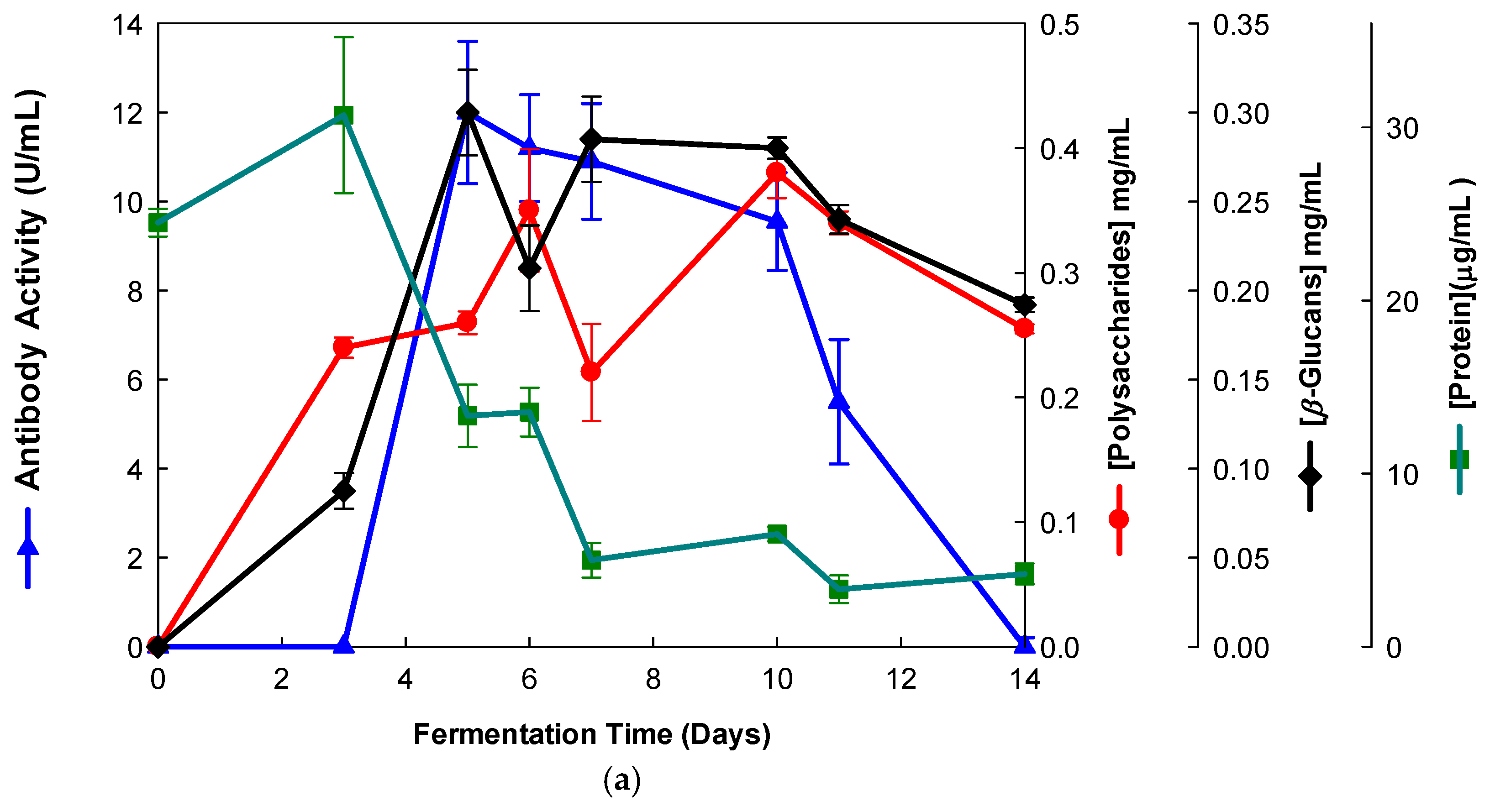
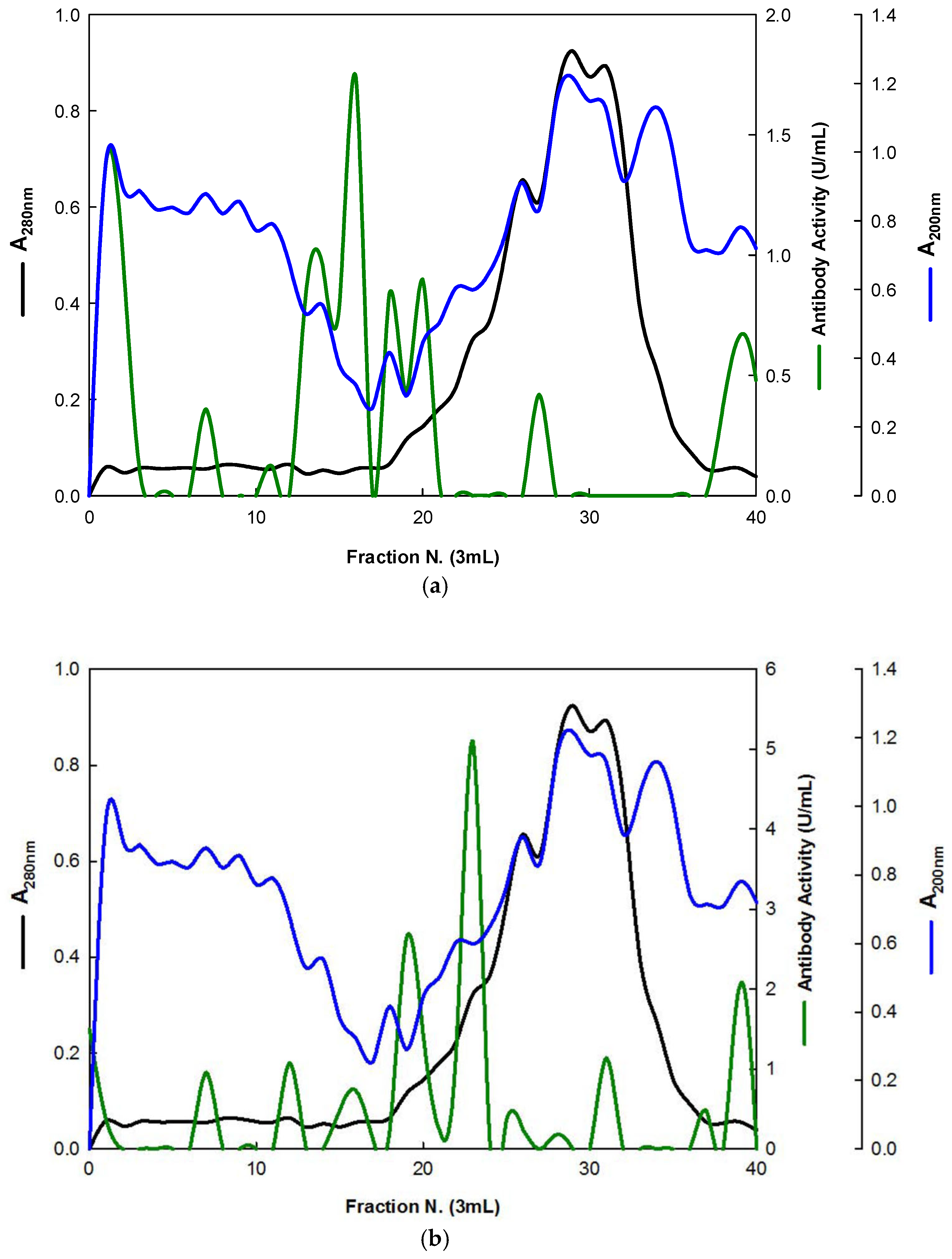
The EBG from Pleurotus ostreatus was purified by gel filtration chromatography which exhibited several peaks with different Mr values both for antibody activity as well as for polysaccharides at A200 (Figure 3). The specific detection of polysaccharides in column fractions by IgM and IgG mAbs is described in Figure 3a,b.
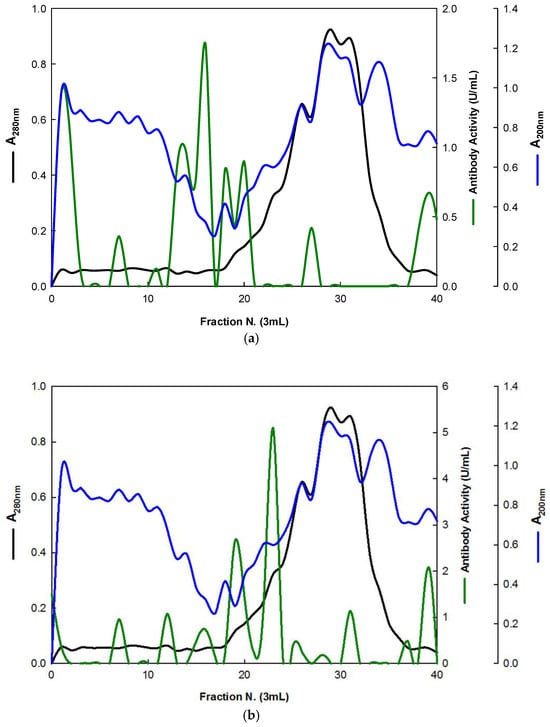
Figure 3.
Purification of EBG from Pleurotus ostreatus by gel filtration chromatography on Sephacryl S-300HR. Column fractions were analyzed for levels of polysaccharides, proteins and immunochemical detection of β-D-glucans by indirect ELISA using mAbs: (a) 3F8_3H7; (b) 1E6_1E8_B5.
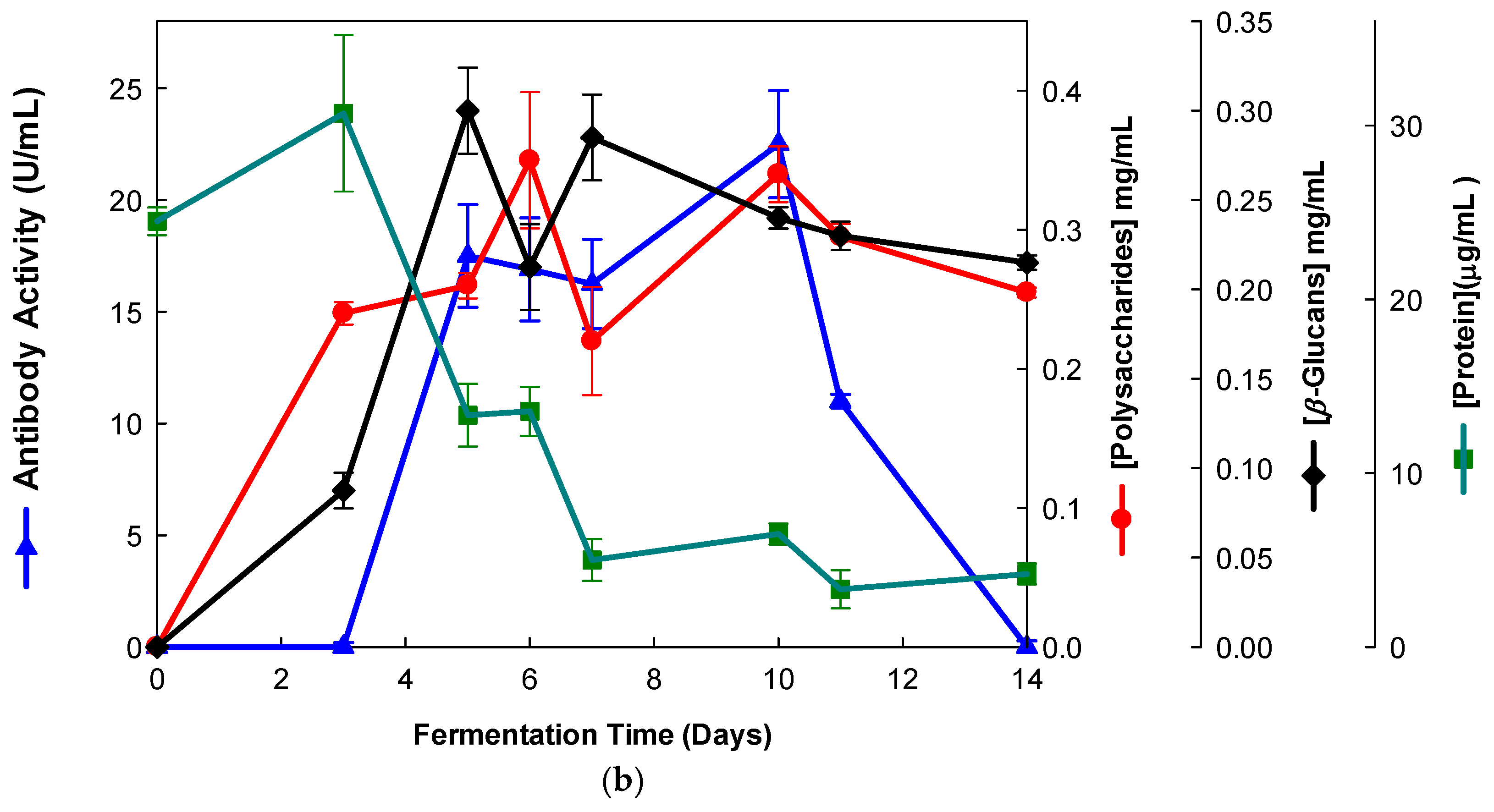
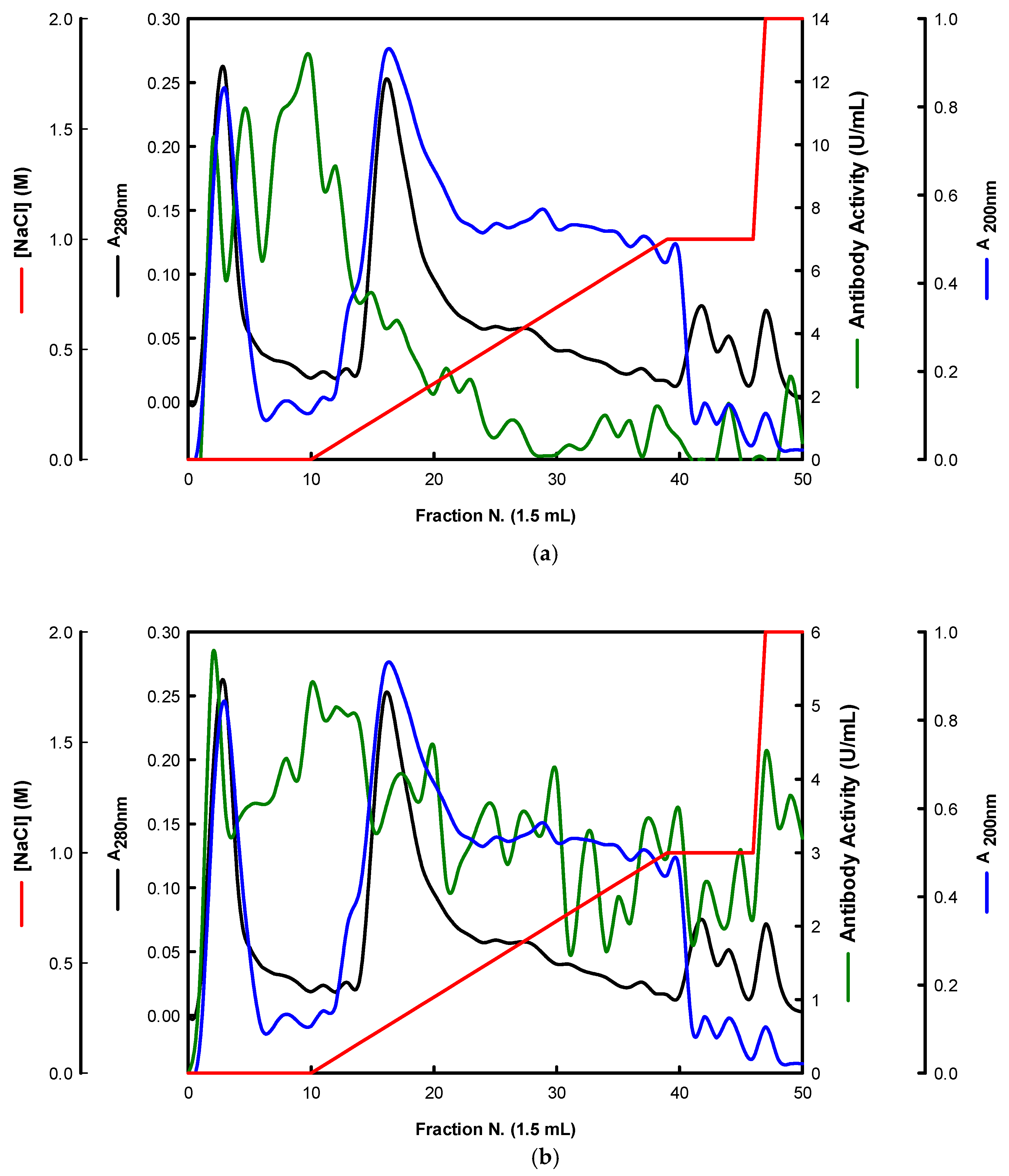
Similarly, the EBG from Pleurotus ostreatus was also purified by ion-exchange chromatography (Figure 4).
Figure 4.
Purification of EBG from Pleurotus ostreatus by ion-exchange chromatography on DEAE-cellulose anion exchange media. Column fractions were analyzed for levels of polysaccharides, proteins and immunochemical detection of β-D-glucans by indirect ELISA using mAbs: (a) 3F8_3H7; (b) 1E6_1E8_B5.
The results obtained for the purification of EBG from Pleurotus ostreatus by gel filtration chromatography are in agreement with published work which reported a heterogeneous mixture of β-D-glucans in mushroom basidiomycete mushroom strains with different Mr values and degree of ramification of β-D-glucan chains [14,15,18,30]. The specific detection of polysaccharides in column fractions by mAb IgM is apparently in agreement with mAb IgG (Figure 3a,b). The EBG from Pleurotus ostreatus was also purified by ion-exchange chromatography, which revealed two main β-D-glucan peaks (Figure 4). One of the β-D-glucan peaks was adsorbed to the column whereas the other β-D-glucan peak did not bind to the column, which suggests that the latter was an uncharged β-D-glucan. However, mAbs strongly recognized the neutral polysaccharide peak that did not bind to the column (Figure 4a,b), whereas mAb reacted weakly to the charged peak. As far as comparative analysis of these data with the literature is concerned, it is difficult as there are no published reports about the purification of β-D-glucans from Pleurotus ostreatus by these two chromatographic techniques.
3.3. Adsorption of β-D-Glucans on ELISA Microtiter Plates
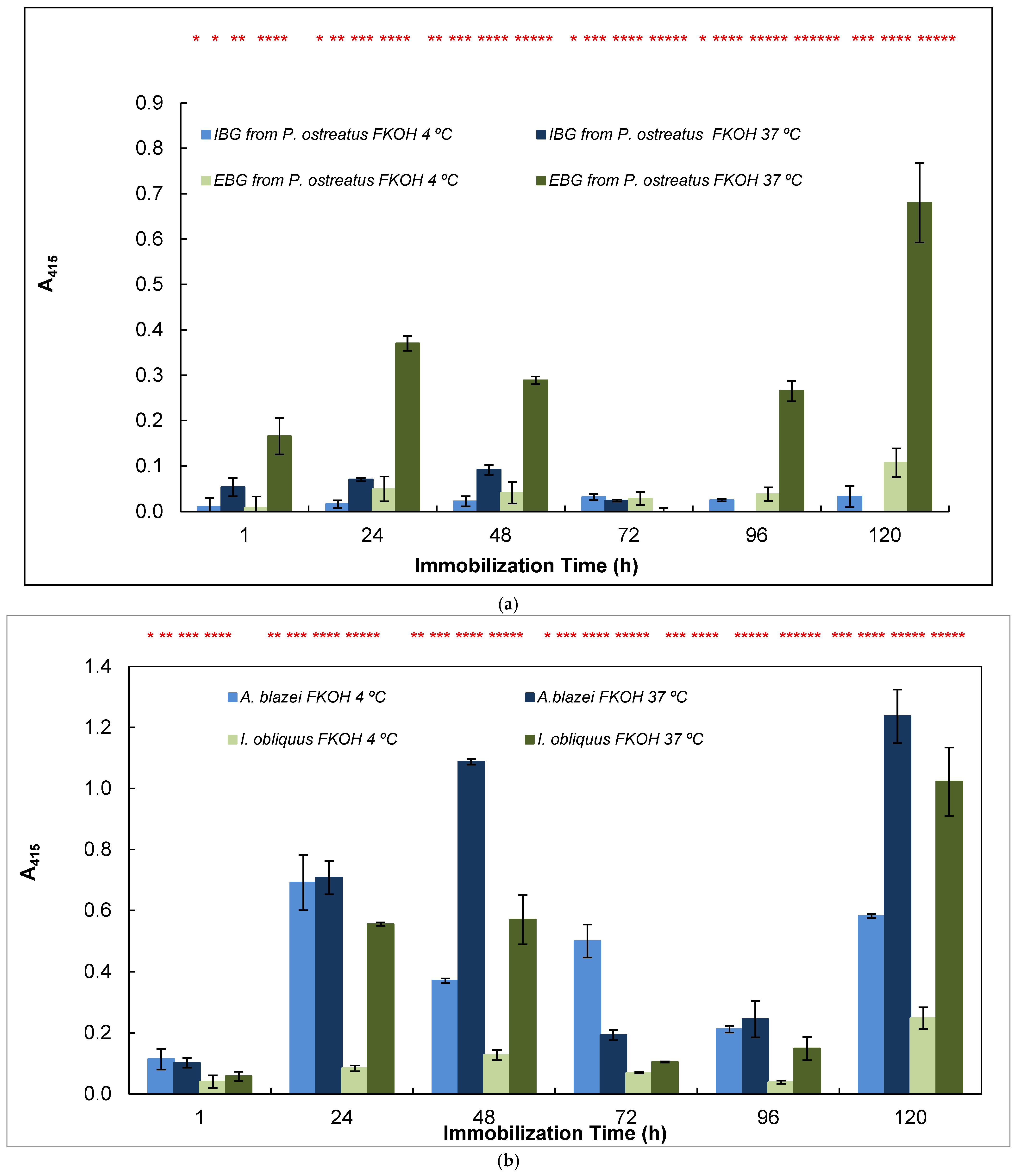
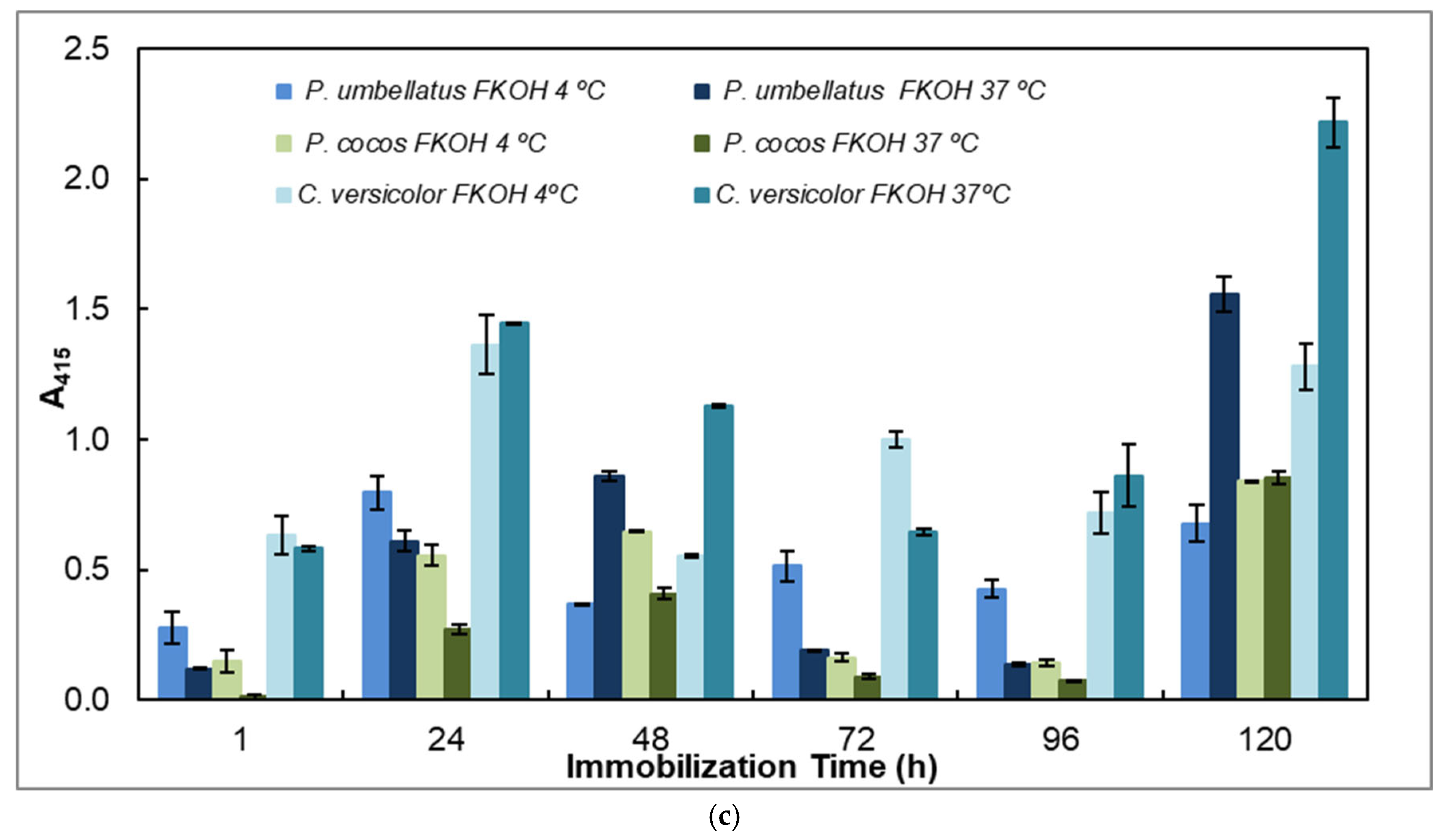
In this work, we also studied the effects of time and temperature on adsorption of β-D-glucans on microplates by using β-D-glucans from various mushroom strains (Figure 5).
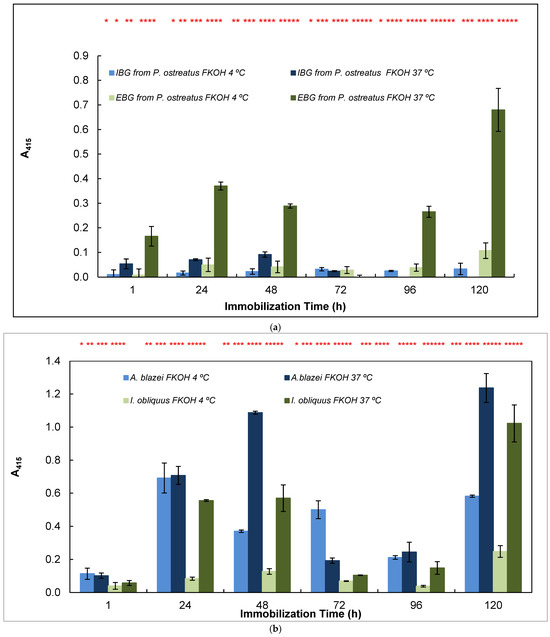
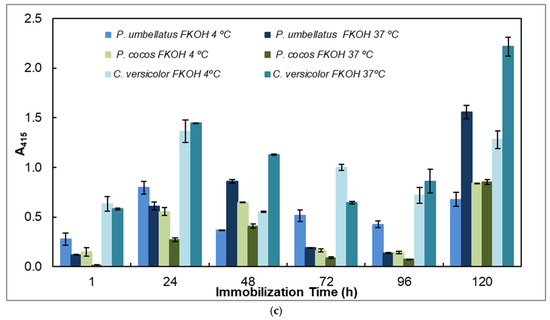
Figure 5.
Adsorption of antigens on microtiter plates by using different temperatures (4 and 37 °C) and time periods (1–120 h) for indirect ELISA. Polysorp® microtiter plates were coated with FKOH fractions of β-D-glucans (20 μg) and were incubated at 4 and 37 °C for between 1 and 120 h to perform indirect ELISA with mAb 1E6_1E8_B5; (a) EBG and IBG from Pleurotus ostreatus; (b) β-D-glucans from Agaricus blazei and Inonotus obliquus; (c) β-D-glucans from Polyporus umbellatus, Poria cocos and Coriolus versicolor. Different letters exhibit significant differences. Statistical analysis via one-way ANOVA: * p < 0.1, ** p < 0.05, *** p < 0.01, **** p < 0.005, ***** p < 0.001, ****** p < 0.0001.
The study of the effects of time and temperature on adsorption of β-glucans on microplates revealed that the values exhibited in indirect ELISA were highest at higher temperature and longer periods of adsorption for EBG from Pleurotus ostreatus whereas IBG revealed higher signals at 37 °C and shorter periods of adsorption (Figure 5a). Figure 5b presents data on Agaricus blazei and Inonotus obliquus, which exhibited the highest signal at higher temperatures and longer periods of β-D-glucan adsorption. Figure 5c reveals that the highest values were obtained either at lower or higher temperatures and longer periods of β-D-glucan adsorption for Poria cocos, whereas Coriolus versicolor and Polyporus umbellatus exhibited higher values at higher temperatures (i.e., 37 °C) and longer periods of adsorption.
3.4. mAbs as Powerful Probes for Changes in Conformations in Native β-D-Glucans
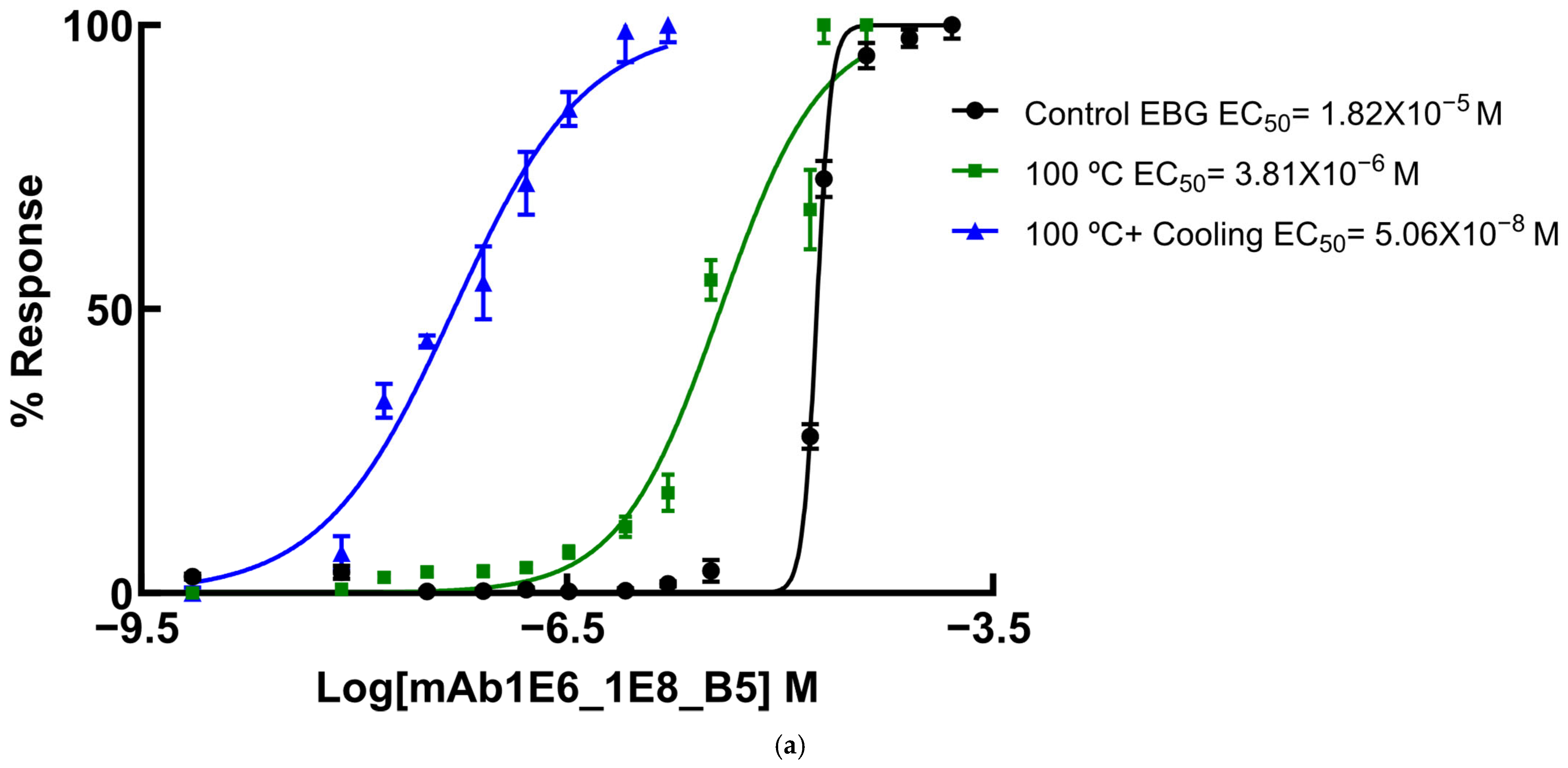
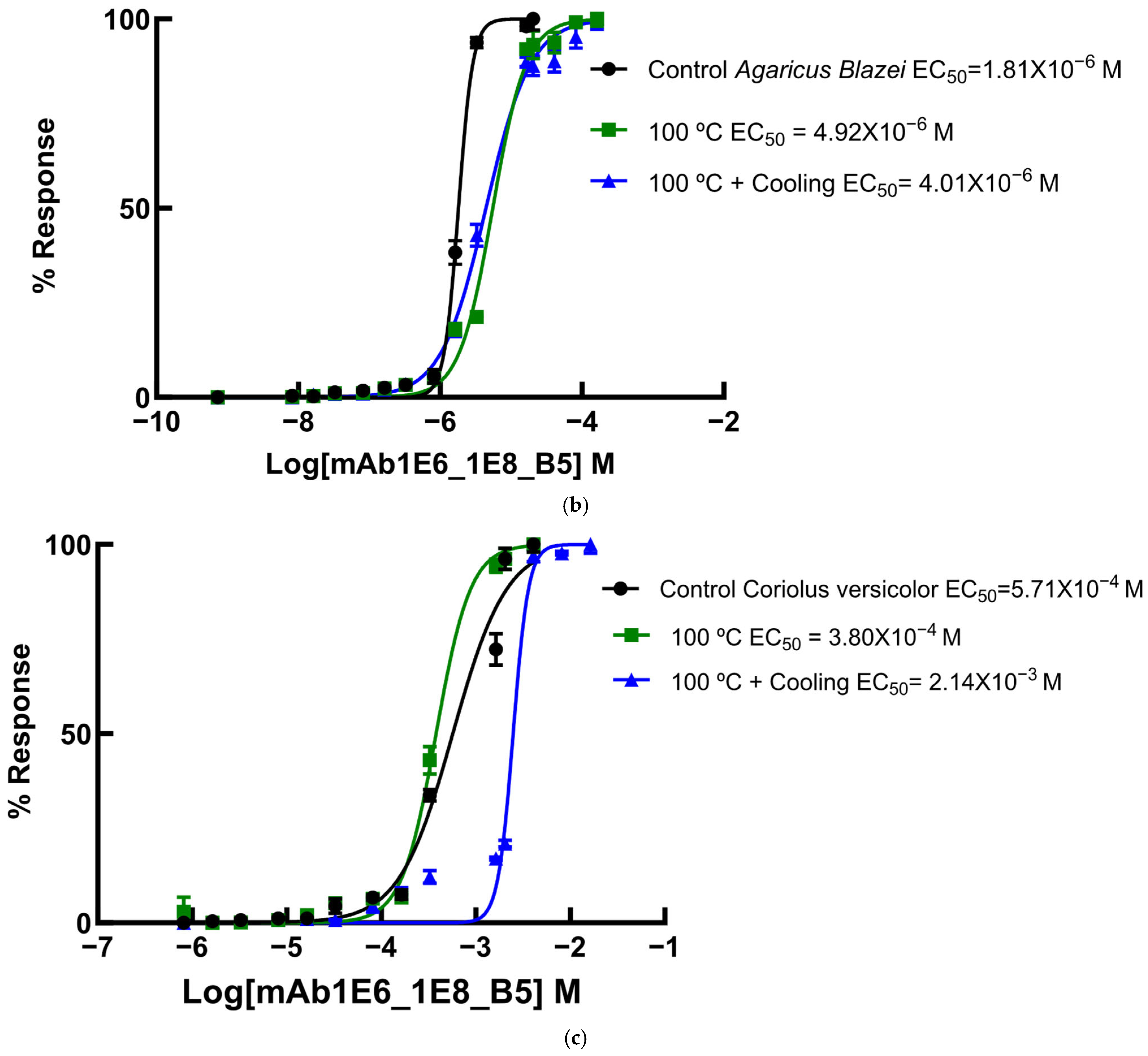
Since several β-glucans have revealed triple helix structures, mAb reactivity was studied for heat-treated β-glucans compared with native forms (Figure 6 and Figure 7). Therefore, EC50 binding curves were obtained by using GraphPad Prism software v.10.6.1 as described in Materials and Methods and EC 50 data is reported in the figure legend. The data were expressed as the fraction between EC50 (Figure 6 and Figure 7) determined on the sigmoid dose–response curves obtained with native and heat-treated β-D-glucans (i.e., percentage reactivity of mAb on the y-axis).
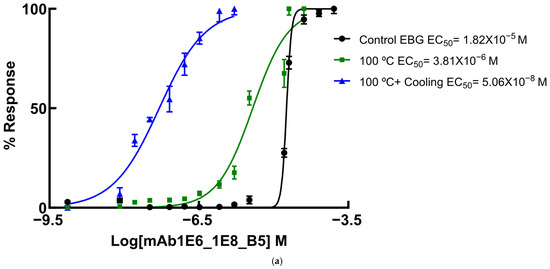
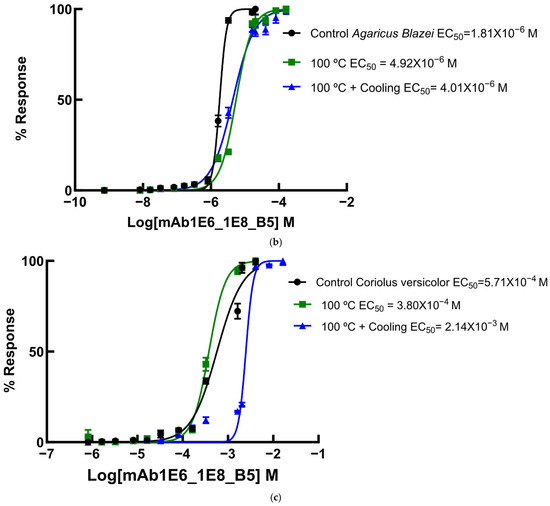
Figure 6.
Dose–response sigmoid curves for determination of EC50 for mAb reactivity to native and heat-treated forms of β-D-glucan. Binding curves of mAbs to (a) EBG from Pleurotus ostreatus: EC50 = 1.82 × 10−5 M for control EBG; EC50 = 3.81 × 10−6 M for 100 °C and EC50 = 5.06 × 10–8 M for 100 °C + Cooling; (b) FKOH from Agaricus blazei: EC50 = 1.81 × 10−6 M for control Agaricus blazei; EC50 = 5.51 × 10−6 M for 100 °C and EC50 = 4.42 × 10–6 M for 100 °C + Cooling; (c) FKOH from Coriolus versicolor: EC50 = 5.71 × 10−4 M for control Coriolus versicolor; EC50 = 3.80 × 10−4 M for 100 °C and EC50 = 2.48 ×10–3 M for 100 °C + Cooling.
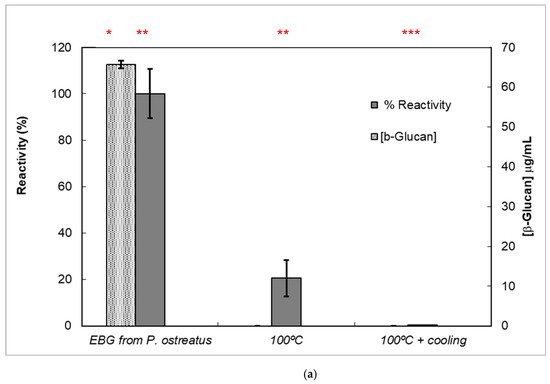
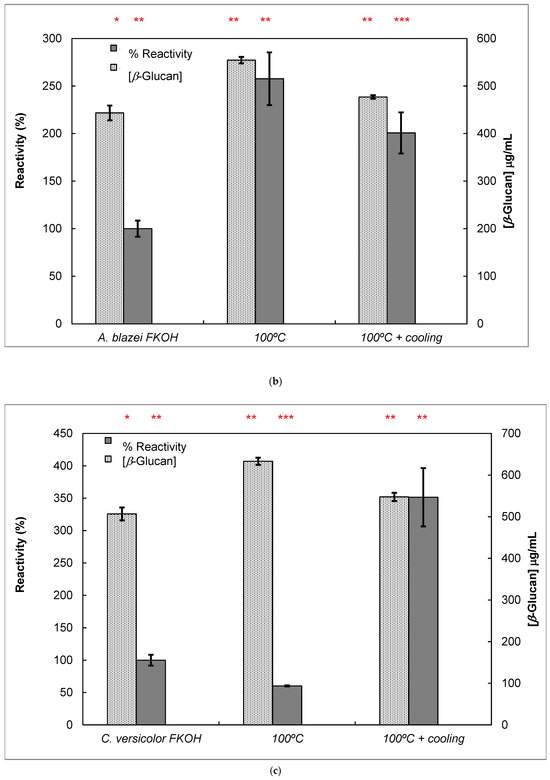
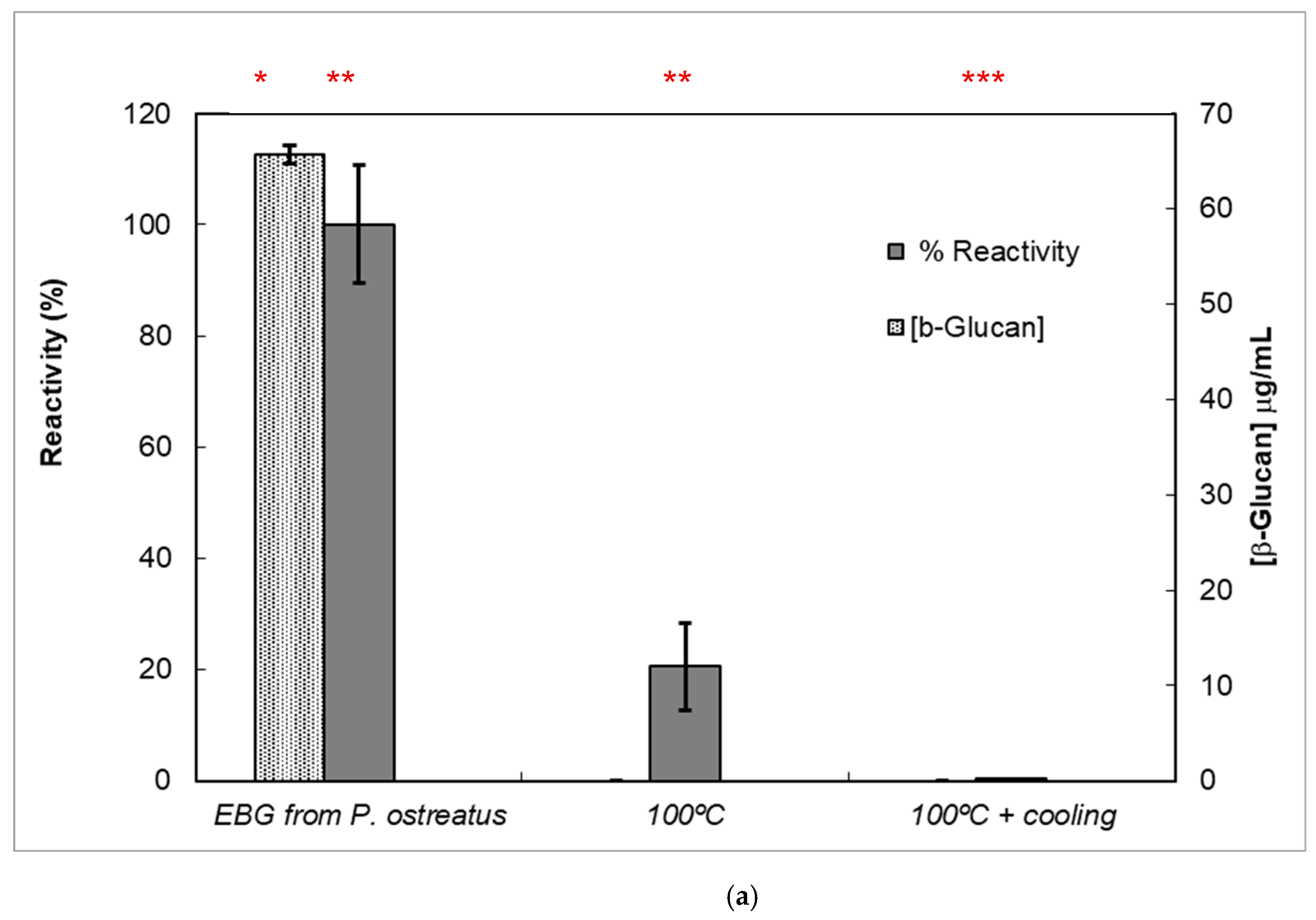
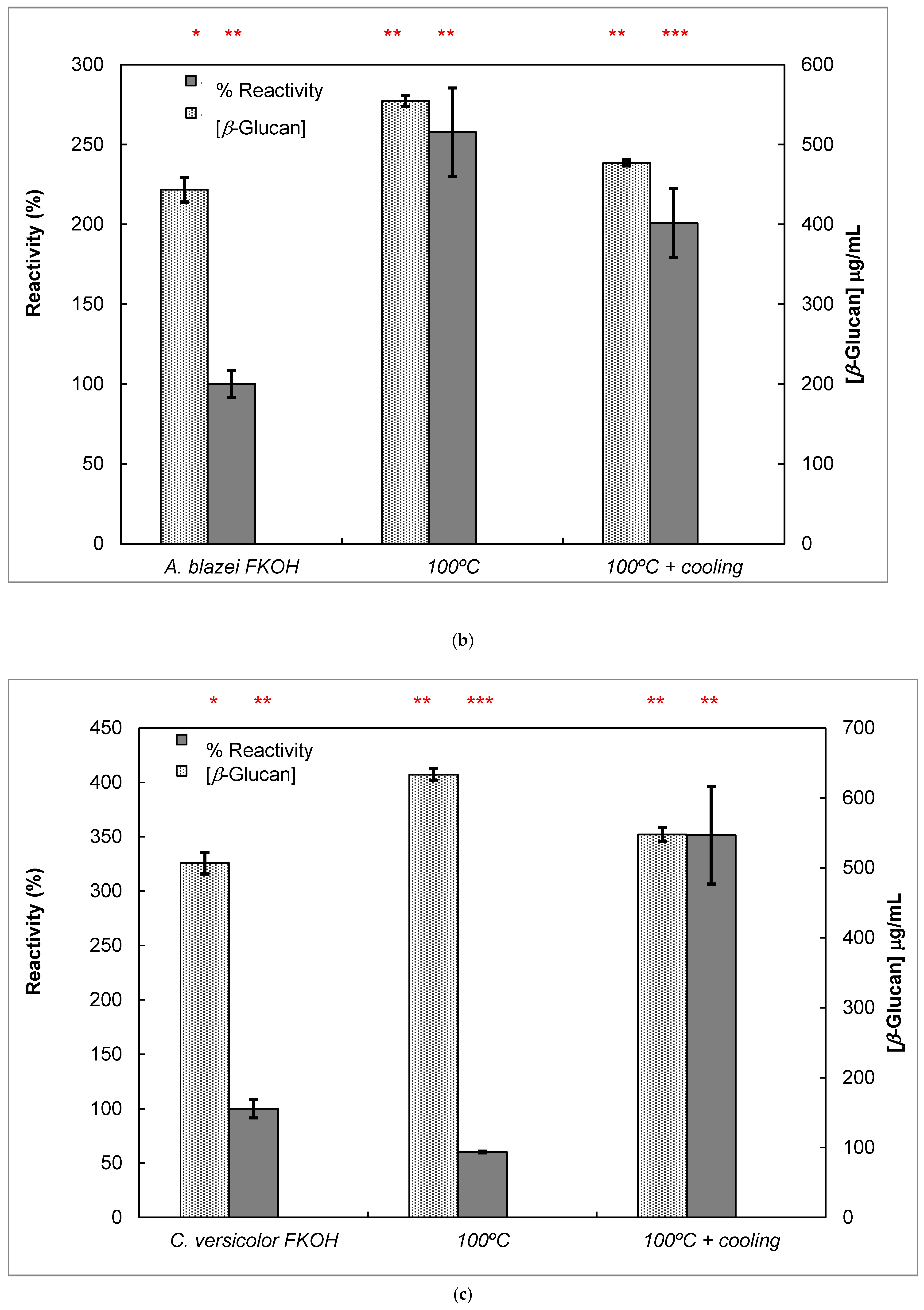
Figure 7.
Reactivity of mAb 1E6_1E8_B5 to native and heat-treated forms of β-D-glucans and Congo red assay for β-glucan with triple helix conformation. β-D-glucan samples (25 μg) from basidiomycete strains were heated at 100 °C and adsorbed to microplates as described in Materials and Methods. The antigen-binding with mAb to native and heat-treated forms of β-glucan was determined by indirect ELISA. The conformational changes in β-D-glucan were monitored by Congo red assay in both native and heat-treated β-glucans. (a) Reactivity of mAbs to EBG from Pleurotus ostreatus and Congo red assay for β-D-glucans; (b) Reactivity of mAbs to FKOH from Agaricus blazei as well as the Congo red assay; (c) reactivity of mAbs to FKOH from Coriolus versicolor as well as the Congo red assay. Statistical analysis via one-way ANOVA: * p < 0.1, ** p < 0.05, *** p < 0.01.
In fact, published reports in the literature strongly suggest that heating causes significant changes in the secondary and tertiary structures of β-D-glucans [10,30].
Various published works have presented significant evidence for conformational changes in heat-treated β-D-glucans from several basidiomycete strains because of their structural transition from triple helix to random coil [14,30,31,32,33]. Similar data have been published by other researchers on mAbs raised against porcine growth hormone and troponin subunits, which detected conformational changes on the antigen molecule [34,35]. On the other hand, some researchers have found that some mAbs raised against native proteins reveal preferential binding to the denatured forms of proteins [36]. In the present study, mAb 1E6_1E8_B5 exhibited a lower affinity for heat-treated β-D-glucans from Pleurotus ostreatus compared with native β-D-glucans (Figure 7a). However, this mAb revealed a higher and lower affinity for heat-treated β-D-glucans from Agaricus blazei and Coriolus versicolor than for their native β-D-glucans, respectively (Figure 7b,c). The effect of cooling after the heat treatment was investigated on EC50 which suggests that the mAb did not recognize cooled β-D-glucans from Pleurotus ostreatus. However, the mAb revealed a lower and higher affinity for β-D-glucans from Agaricus blazei and Coriolus versicolor, respectively (Figure 7b,c). During this investigation, the data on mAb reactivity was compared with data from Congo red assay which only recognizes and monitors β-D-glucans with a triple helix structure [29]. Congo red assay did not detect heat-treated β-D-glucans from Pleurotus ostreatus, which strongly indicates that heat treatment damaged its triple helix structure (Figure 7a). On the other hand, Congo red assay detected higher levels of heat-treated β-D-glucans from Agaricus blazei and Coriolus versicolor compared with their native forms (Figure 7b,c). The effect of cooling after the heat treatment on β-D-glucan concentrations by Congo red assay shows that it did not detect cooled β-D-glucans from Pleurotus ostreatus, whereas it revealed lower values for β-D-glucans from Agaricus blazei and Coriolus versicolor compared with their heat-treated forms (Figure 7). Therefore, the data presented in Figure 6 and Figure 7 exhibit strong evidence that mAbs detected conformational changes in β-D-glucans from basidiomycete strains as a result of heating and cooling treatments.
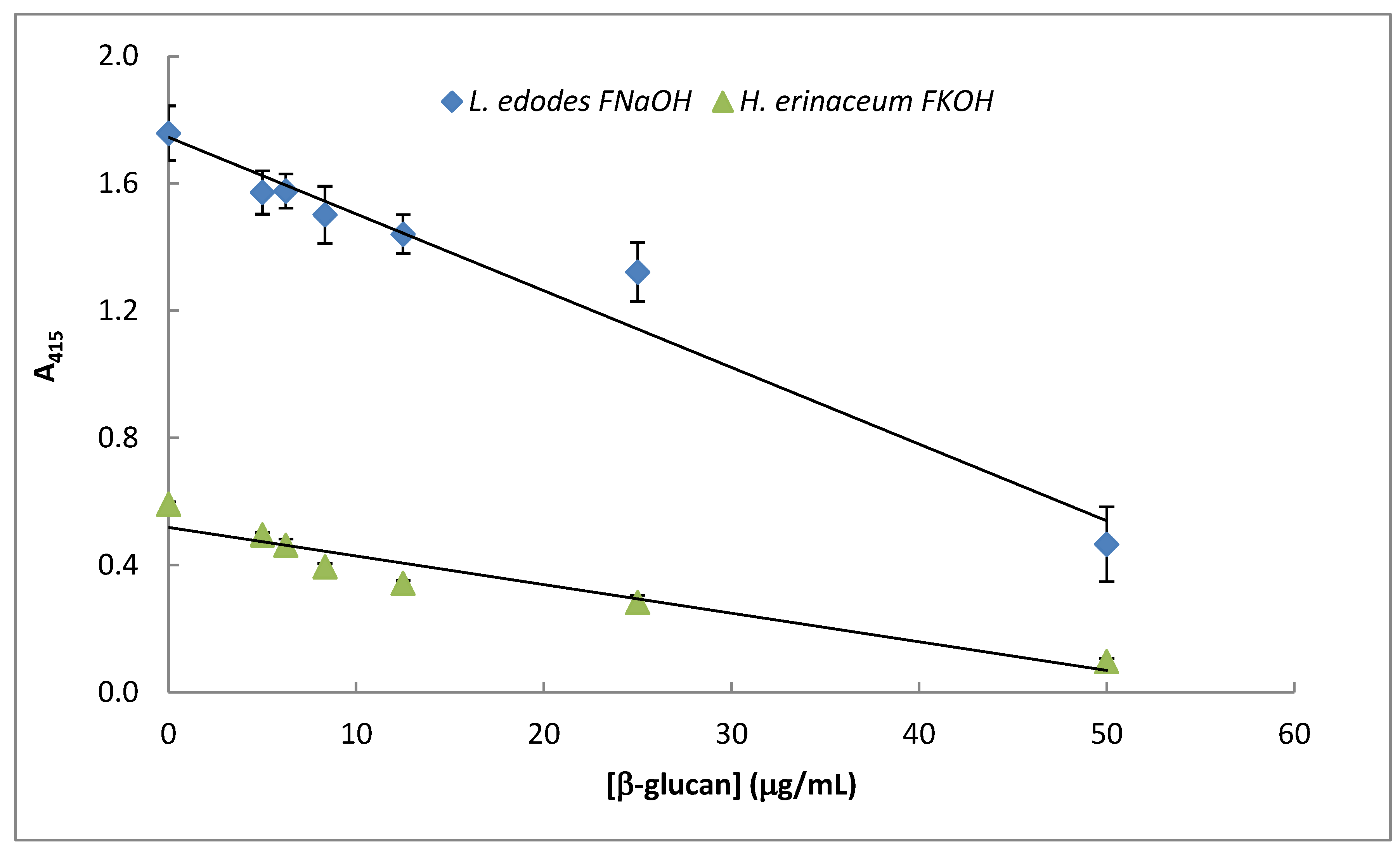
3.5. Assay of β-D-Glucans by Competitive ELISA
The data presented in Figure 8 exhibits a specific immunochemical test for assay of β-D-glucan from mushroom strains. Therefore, competitive ELISA (Figure 8) was carried out by using immobilized β-D-glucans from Lentinula edodes and Hericium erinaceus in 96-well microtiter plates, and soluble mAb was previously incubated with increasing amounts of the same soluble β-D-glucans. The data exhibited in Figure 8 strongly suggest that mAb 1E6_1E8_B5 can be used in β-D-glucan assay by competitive ELISA in mushroom strains since A415 was inversely proportional to soluble β-D-glucan concentration.
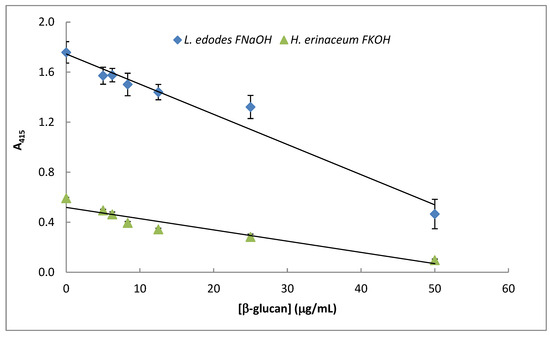
Figure 8.
Assay of mushroom β-D-glucans by competitive ELISA. A fixed concentration of mAb 1E6_1E8_B5 was incubated with different amounts (0–25 μg/ mL) of selected β-glucans (i.e., FNaOH from Lentinula edodes and FKOH from Hericium erinaceus) at 37 °C for 1 h. The resulting mAb–β-glucans complexes were transferred to PolySorp® microplate wells (50 μL) which were previously adsorbed with the same antigen for 120 h at 4 °C. Subsequently, the microplates were washed to remove unbound mAb and treated the same way as in the indirect ELISA method.
This immunochemical assay method presents some advantages over other colorimetric and fluorescence methods reported in the literature [20,21,22] since it is more sensitive and specific to an epitope on the β-D-glucan molecule. Therefore, it can be used to screen thousands of mushroom strains containing β-D-glucans with this epitope as well as to detect changes in triple helix conformation in their tertiary structure, which is responsible for their biological activity [20]. Moreover, it can be devised as a high-throughput microtiter plate of 384 wells to process hundreds of samples in a couple of hours. However, it is more expensive and slower than those published in the literature as this assay method involves slow antigen–antibody reactions and use of labeled mAb, substrates and microtiter plates.
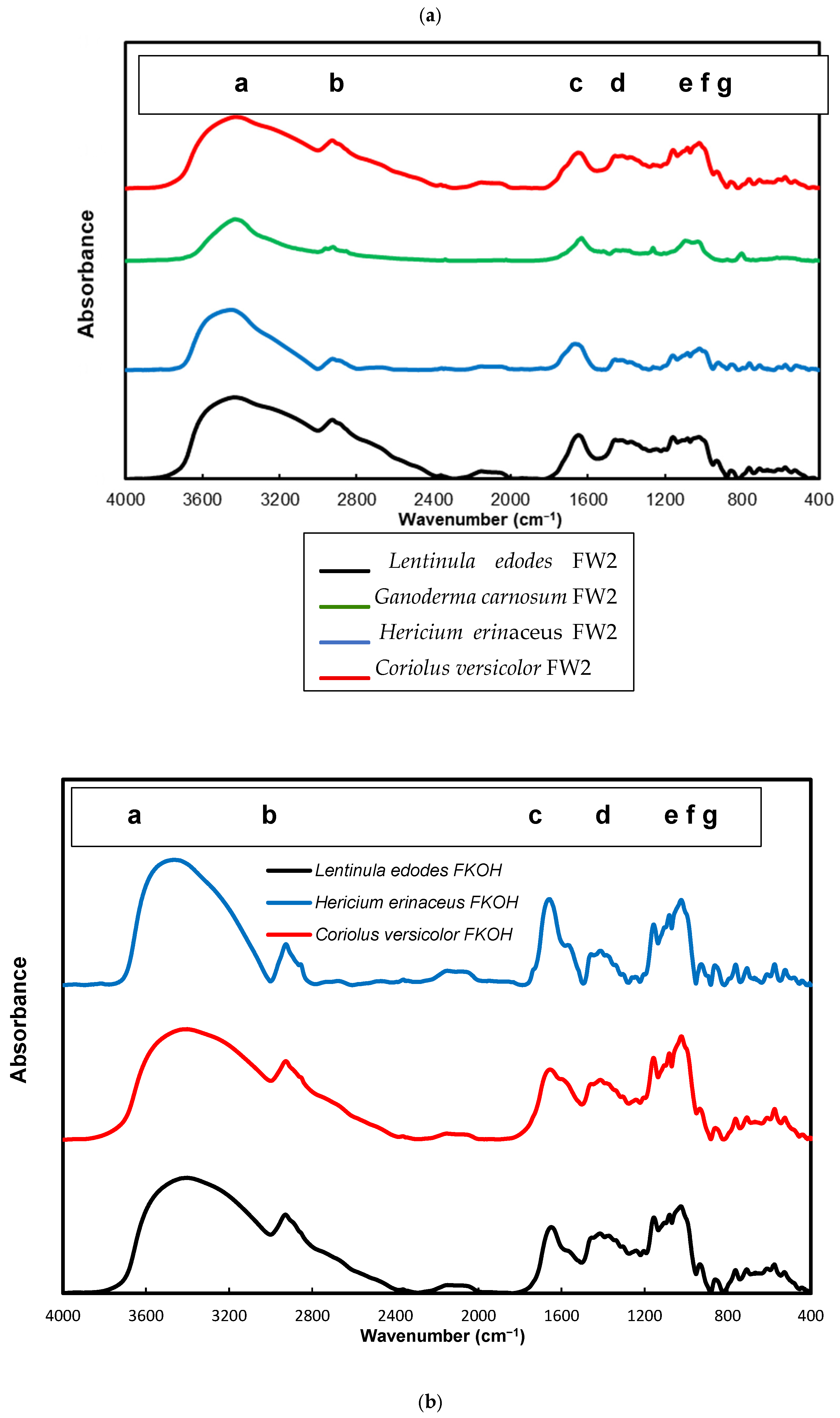
3.6. FTIR Analysis of β-D-Glucans
FTIR spectra of FW2 and FKOH from Lentinula edodes, Hericium erinaceus and Coriolus versicolor were obtained, which revealed typical absorption bands of β-D-glucan in the region of 950–1200 cm−1 (Figure 9a,b).
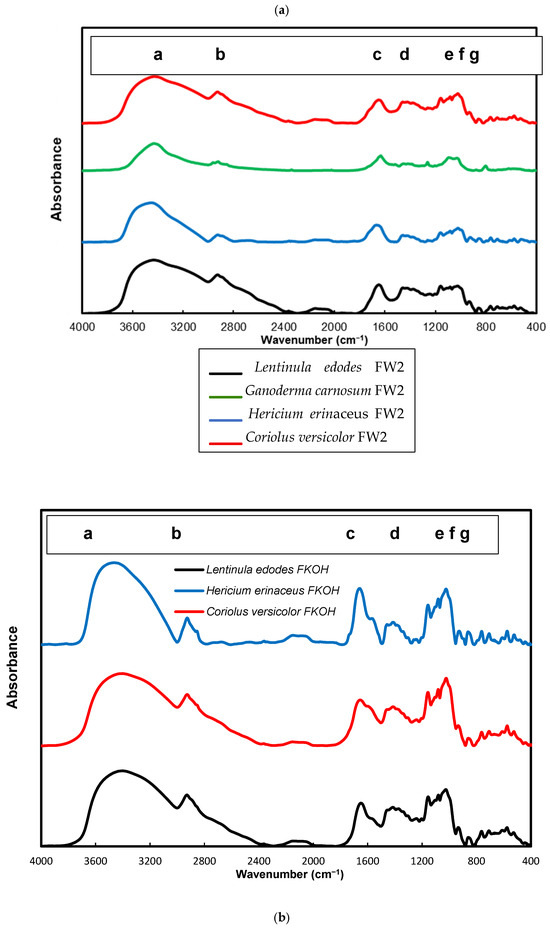
Figure 9.
FTIR spectra of FW2 and FKOH from Lentinula edodes, Hericium erinaceus and Coriolus versicolor- FTIR spectra. (a) FW2 from Lentinula edodes, Hericium erinaceus and Coriolus versicolor; (b) FKOH from Lentinula edodes, Hericium erinaceus and Coriolus versicolor. a—3430 cm−1; b—2926 cm−1, c—1640 cm−1, d—1366 cm−1, e—1183 cm−1, f—979 cm−1 and g—811 cm−1.
Alkali soluble mushroom (1→3)-α-D-glucan can be detected by characteristic absorption bands near 1366, 930, 850, 822, 542, 454 and 420 cm–1. The absorption spectrum of F_KOH fraction revealed absorption bands characteristics of α-D-glucan (858 and 935 cm−1), CO stretching (1023 cm−1), anomeric C1H group vibration (1081 cm−1), C–O–C stretching (1157 cm−1), C-O bond (1640 cm−1), C–H bond (2926 cm−1) and O–H stretching (3430 cm−1). On the other hand, it has been found that absorption bands near 1160, 1078, 1044 and 890 cm–1 are characteristic for mushroom (1→3)(1→6)-β-D-glucans, while the bands of (1→4)(1→6)-α-D-glucans are located near 1155, 1023, 930, 850 and 765 cm–1. In order to identify some structural characteristics of β-D-glucans, some FTIR spectra from basidiomycete fractions were obtained, and the data are in agreement with some reports in the literature on mushroom β-D-glucans [15,37,38].
4. Conclusions
To the author’s knowledge, this is the first report about the use of mAbs of IgG and IgM to monitor β-D-glucan production from basidiomycete mushroom strains in agro-industrial wastes. The immunochemical detection of these β-glucans correlated well with Congo red assay for β-D-glucan. These mAbs are powerful tools to detect changes in conformation in β-D-glucans from basidiomycete strains due to heat and cooling treatments. Since this mAb was successfully used in competitive ELISA, it could be used to design an immunosensor for monitoring and quantification of β-D-glucans from novel basidiomycete strains. Therefore, a sandwich immunosensor could be devised to speed up the antigen–antibody reaction by immobilization of the first mAb in a suitable support. Subsequently, the addition of increasing concentrations of β-D-glucans would be carried out and detection of the immune complex with a second mAb labeled either with an enzyme or biotin which is bound to a different epitope of β-D-glucans would be performed [39]. Moreover, since it would detect changes in conformation in β-D-glucan molecules, this immunosensor would be very useful to find novel β-D-glucan with innovative biological activities.
Funding
This work was supported by European Investment Funds by FEDER/COMPETE/POCI—Operacional Competitiveness and Internacionalization Programme, under Project POCI-01-0145-FEDER-006958 and National Funds by FCT—Portuguese Foundation for Science and Technology, under the projects UID/AGR/04033/2013, PTDC/AGR-AAM/74526/2006, PEst-OE/EQB/UI0702/2012-2014 and UID/AGR/04033/2019.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the author on request.
Conflicts of Interest
There are no conflicts of interest.
Abbreviations
| BRM | Biological response modifiers |
| EBG | Extracellular β-D-glucans |
| ELISA | Enzyme-linked immunosorbent assay |
| IBG | Intracellular β-D-glucans |
| mAbs | Monoclonal antibodies |
| PBS | Phosphate-buffered saline |
| PDA | Potato dextrose agar |
References
- Khanna, M.; Zilberman, D.; Hochman, G.; Basso, B. An economic perspective of the circular bioeconomy in the food and agricultural sector. Commun. Earth Environ. 2024, 5, 507. [Google Scholar] [CrossRef]
- Virgolino, J.L.F.; Holden, N.H. Does the bioeconomy literature provide a balanced view of sustainability? Sustain. Sci. 2025, 20, 1075–1092. [Google Scholar] [CrossRef]
- Kowalski, S.; Gumul, D. The Use of Waste Products from the Food Industry to Obtain High Value-Added Products. Foods 2024, 13, 847. [Google Scholar] [CrossRef]
- Gonçalves, J.; Anjos, O.; Guiné, R.P.F. A Revisit of Plant Food Waste Along Food Supply Chains: Impacts and Perspectives. Foods 2025, 14, 1364. [Google Scholar] [CrossRef]
- Afsah-Hejri, L.; Maharlooei, M.; Ehsani, R.; Maboudian, R. Promoting sustainable food production through waste reduction and valorization: A California case study. npj Mater. Sustain. 2025, 3, 17. [Google Scholar] [CrossRef]
- Pilafidis, S.; Diamantopoulou, P.; Gkatzionis, K.; Sarris, D. Valorization of Agro-Industrial Wastes and Residues through the Production of Bioactive Compounds by Macrofungi in Liquid State Cultures: Growing Circular Economy. Appl. Sci. 2022, 12, 11426. [Google Scholar] [CrossRef]
- Barua, R.C.; Coniglio, R.O.; Molina, M.A.; Díaz, G.V.; Fonseca, M.I. Fungi as biotechnological allies: Exploring contributions of edible and medicinal mushrooms. J. Food Sci. 2024, 89, 6888–6915. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Wang, G.; Li, F.; Fang, S.; Zhou, S.; Ishiwata, A.; Tonevitsky, A.G.; Shkurnikov, M.; Cai, H.; Ding, F. Immunomodulatory Effect and Biological Significance of β-Glucans. Pharmaceutics 2023, 15, 1615. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Bhardwaj, A. β-glucans: A potential source for maintaining gut microbiota and the immune system. Front. Nutr. 2023, 10, 1143682. [Google Scholar] [CrossRef]
- Yuan, H.; Lan, P.; He, Y.; Li, C.; Ma, X. Effect of the Modifications on the Physicochemical and Biological Properties of β-Glucan—A Critical Review. Molecules 2020, 25, 57. [Google Scholar] [CrossRef]
- Singh, A.; Saini, R.K.; Kumar, A.; Chawla, P.; Kaushik, R. Mushrooms as Nutritional Powerhouses: A Review of Their Bioactive Compounds, Health Benefits, and Value-Added Products. Foods 2025, 14, 741. [Google Scholar] [CrossRef]
- Noreen, S.; Sultan, H.; Hashmi, B.; Aja, P.M.; Atoki, A.V. Mushroom marvels: Understanding their role in human health. Front. Nutr. 2025, 12, 1654911. [Google Scholar] [CrossRef] [PubMed]
- Maqsood, S.; Khalid, W.; Kumar, P.; Benmebarek, I.E.; Rasool, I.F.U.; Trif, M.; Moreno, A.; Esatbeyoglu, T. Valorization of plant-based agro-industrial waste and by-products for the production of polysaccharides: Towards a more circular economy. Appl. Food Res. 2025, 5, 100954. [Google Scholar] [CrossRef]
- Han, B.; Baruah, K.; Cox, E.; Vanrompay, D.; Bossier, P. Structure-Functional Activity Relationship of β-Glucans From the Perspective of Immunomodulation: A Mini-Review. Front. Immunol. 2020, 11, 658. [Google Scholar] [CrossRef]
- Wang, W.; Tan, J.; Nima, L.; Sang, Y.; Cai, X.; Xue, H. Polysaccharides from fungi: A review on their extraction, purification, structural features, and biological activities. Food Chem. X 2022, 15, 100414. [Google Scholar] [CrossRef] [PubMed]
- Timm, T.G.; Costa, T.M.; Alberton, M.D.; Helm, C.V.; Tavares, L. BB Mushroom β-glucans: Application and innovation for food industry and immunotherapy. Appl. Microbiol. Biotechnol. 2023, 107, 5035–5049. [Google Scholar] [CrossRef]
- Flores, G.A.; Cusumano, G.; Venanzoni, R.; Angelini, P. The Glucans Mushrooms: Molecules of Significant Biological and Medicinal Value. Polysaccharides 2024, 5, 212–224. [Google Scholar] [CrossRef]
- Zhong, Y.; Tan, P.; Lin, H.; Zhang, D.; Chen, X.; Pang, J.; Mu, R. A Review of Ganoderma lucidum Polysaccharide: Preparations, Structures, Physicochemical Properties and Application. Foods 2024, 13, 2665. [Google Scholar] [CrossRef]
- Masuko, T.; Minami, A.; Iwasaki, N.; Majima, T.; Nishimura, S.-I.; Lee, Y.C. Carbohydrate analysis by a phenol–sulfuric acid method in microplate format. Anal. Biochem. 2005, 339, 69–72. [Google Scholar] [CrossRef]
- Semedo, M.C.; Karmali, A.; Fonseca, L. A high throughput colorimetric assay of β-1,3-D-glucans by Congo red dye. J. Microbiol. Meth. 2015, 109, 140–148. [Google Scholar] [CrossRef]
- Semedo, M.C.; Karmali, A.; Fonseca, L. A novel colorimetric assay of β-D-glucans in basidiomycete strains by alcian blue dye in a 96-well microtiter plate. Biotechnol. Prog. 2015, 31, 1526–1535. [Google Scholar] [CrossRef]
- Koenig, S.; Rühmann, B.; Sieber, V.; Schmid, J. Quantitative assay of β-(1,3)-β-(1,6)-glucans from fermentation broth using aniline blue. Carbohydr. Polym. 2017, 174, 57–64. [Google Scholar] [CrossRef]
- Mizono, M.; Minato, K.-I.; Tsuchida, H. Preparation and specificity of antibodies to an anti-tumor β-glucan, lentinan. Biochem. Mol. Biol. Int. 1996, 39, 679–685. [Google Scholar] [CrossRef]
- Semedo, M.C.; Martins, S.; Karmali, A.; Fonseca, L. Novel Polyol-Responsive Monoclonal Antibodies Against Extracellular β-D-Glucans from Pleurotus ostreatus. Biotechnol. Prog. 2016, 32, 116–125. [Google Scholar] [CrossRef]
- Semedo, M.C.; Martins, S.; Karmali, A.; Fonseca, L. Generation of high-affinity monoclonal antibodies of IgG class against native β-D-glucans from basidiomycete mushrooms. Process. Biochem. 2016, 51, 333–342. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Luna, K.A.; Aguilar, C.N.; Ramírez-Guzmán, N.; Ruiz, H.A.; Martínez, J.L.; Chávez-González, M.L. Bioprocessing of Spent Coffee Grounds as a Sustainable Alternative for the Production of Bioactive Compounds. Fermentation 2025, 11, 366. [Google Scholar] [CrossRef]
- Otieno, O.D.; Mulaa, F.J.; Obiero, G.; Midiwo, J. Utilization of fruit waste substrates in mushroom production and manipulation of chemical composition. Biocatal. Agric. Biotechnol. 2022, 39, 102250. [Google Scholar] [CrossRef]
- Khatami, K.; Qazanfarzadeh, Z.; Jiménez-Quero, A. Review Fungal fermentation: The blueprint for transforming industrial side streams and residues. Bioresour. Technol. 2026, 440, 133426. [Google Scholar] [CrossRef]
- Kaur, R.; Sharma, M.; Ji, D.; Xu, M.; Agyei, D. Review Structural Features, Modification, and Functionalities of Beta-Glucan. Fibers 2020, 8, 1. [Google Scholar] [CrossRef]
- Cui, S.W. Food Carbohydrates: Chemistry, Physical Properties and Applications; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon, UK, 2005. [Google Scholar]
- Sletmoen, M.; Stokke, B.T. Review: Higher order structure of (1,3)-β-D-glucans and its influence on their biological activities and complexation abilities. Biopolymers 2008, 89, 310–321. [Google Scholar] [CrossRef]
- Nitschke, J.; Modick, H.; Busch, E.; von Rekowski, R.W.; Altenbach, H.-J.; Mölleken, H. A new colorimetric method to quantify β-1,3-1,6-glucans in comparison with total β-1,3-glucans in edible mushrooms. Food Chem. 2011, 127, 791–796. [Google Scholar] [CrossRef]
- Borromeo, V.; Gaggioli, D.; Berrini, A.; Secchi, C. Monoclonal antibodies as a probe for the unfolding of porcine growth hormone. J. Immunol. Methods 2003, 272, 107–115. [Google Scholar] [CrossRef]
- Rasmussen, M.; Jin, J.-P. Monoclonal Antibodies as Probes to Study Ligand-Induced Conformations of Troponin Subunits. Front. Physiol. 2022, 13, 828144. [Google Scholar] [CrossRef] [PubMed]
- Friguet, B.; Djavadi-Ohaniance, L.; Goldberg, M.E. Some monoclonal antibodies raised with a native protein bind preferentially to the denatured antigen. Mol. Immunol. 1984, 21, 673–677. [Google Scholar] [CrossRef]
- Silva, S.; Martins, S.; Karmali, A.; Rosa, E. Production, purification and characterisation of polysaccharides from Pleurotus ostreatus with antitumour activity. J. Sci. Food Agric. 2012, 92, 1826–1832. [Google Scholar] [CrossRef] [PubMed]
- Synytsya, A.; Novak, M. Structural analysis of glucans. Ann. Transl. Med. 2014, 2, 17. [Google Scholar]
- Djebbi, K.; Xiang, Y.; Shi, B.; Douadji, L.; Chen, X.; Liu, J.; Tlili, C.; Wang, D. A Sandwich-Type Impedimetric Immunosensor for the Detection of Tau-441 Biomarker. Bioengineering 2025, 12, 805. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.