Environmental Impact Assessment of New Cement Production Blending Calcareous Green Algae and Fly Ash
Abstract
1. Introduction
2. Methodology
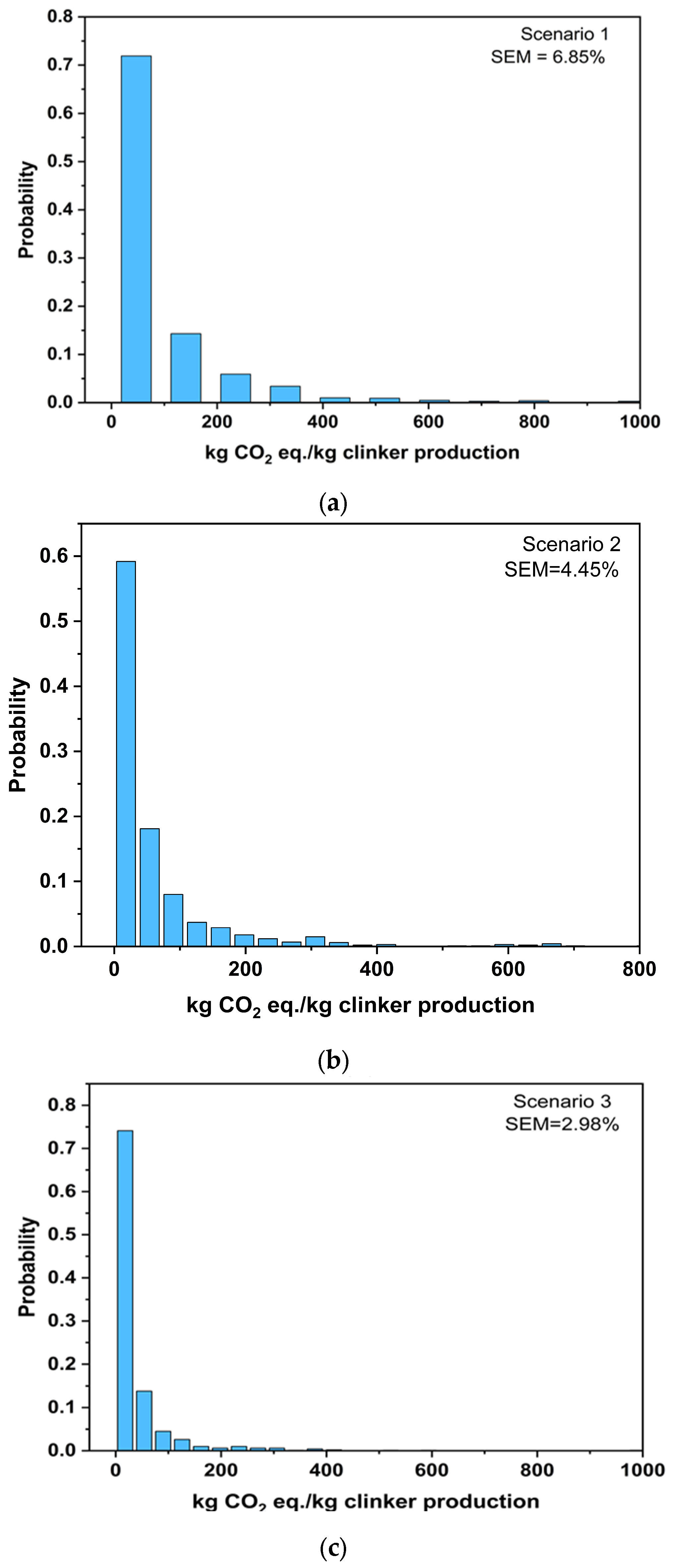
3. Validation and Sensitivity Analysis
3.1. Validation Analysis for Scenario 1
3.2. Sensitivity Analysis for Scenario 2
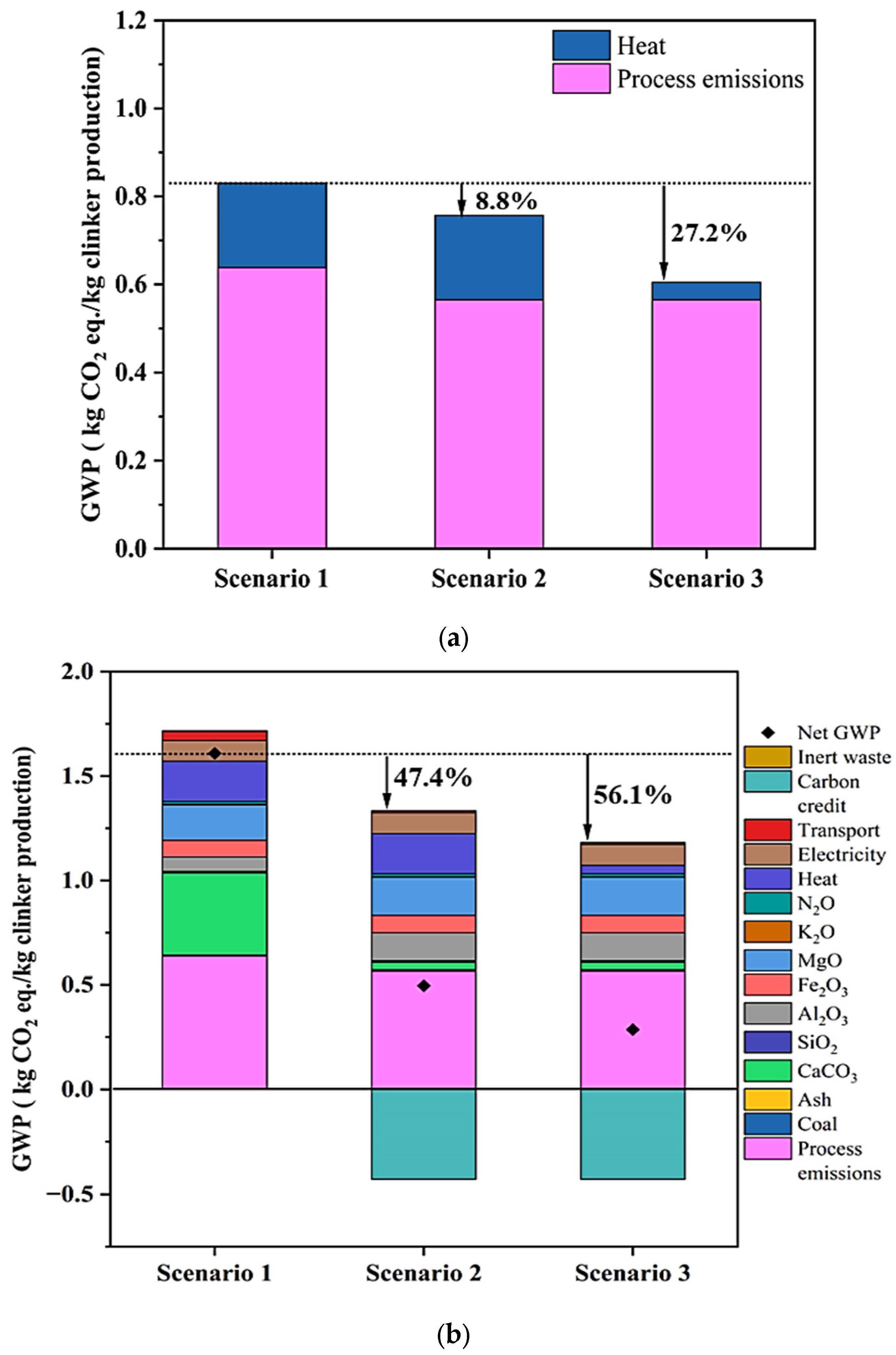
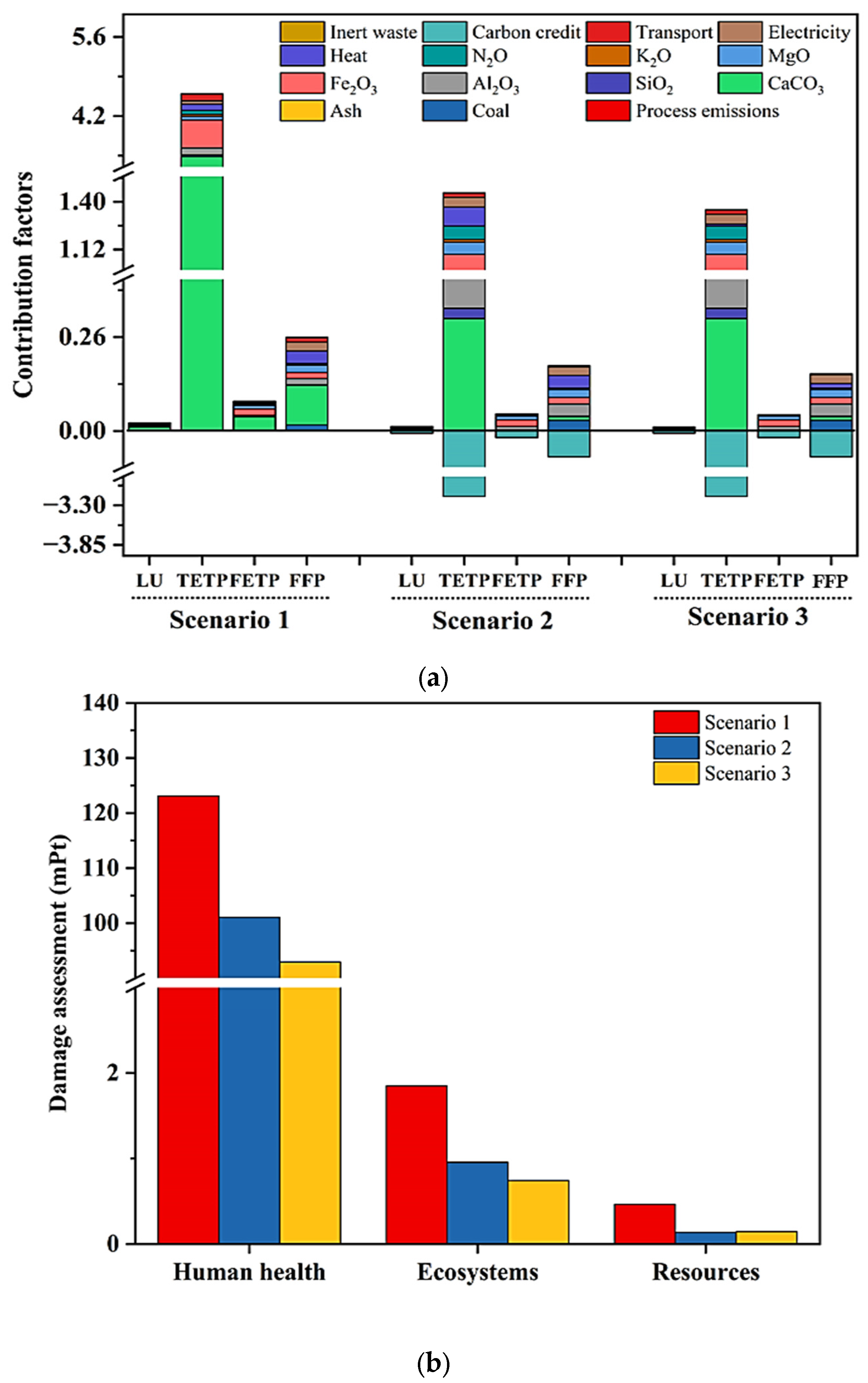
3.3. Environmental Impact Assessment
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| CaCO3 | Calcium carbonate |
| CaO | Calcium oxide |
| CKD | Cement kiln dust |
| C3A | Tricalcium aluminate |
| C3S | Tricalcium silicate |
| C2S | Dicalcium silicate |
| C4AF | Calcium ferroaluminate |
| CO2 | Carbon dioxide |
| A/F | Alumina-to-ferrite ratio |
| FCd | Fixed carbon (dry basis) |
| FGD | Flue-gas desulfurization |
| HHV | Higher heating value |
| LCA | Life Cycle Assessment |
| LCI | Life Cycle Inventory |
| LCIA | Life Cycle Impact Assessment |
| LHV | Lower heating value |
| LOI | Loss on ignition |
| Mg/Ca | Molar ratio of magnesium to calcium in seawater |
| ReCiPe | A life-cycle impact assessment method (2016 version) |
| RGibbs | RGibbs reactor block (Gibbs energy minimization reactor) |
| Rstoic | RStoic reactor block (stoichiometric reactor) |
| EIkpi | Overall environmental impact for KPI |
| KPI | Key Performance Indicator |
References
- Iqbal, A.; Swapnil, F.J. Sustainable green materials for building construction. In Proceedings of the International Conference on Mechanical, Industrial and Materials Engineering 2017 (ICMIME2017), Rajshahi, Bangladesh, 28–30 December 2017. [Google Scholar]
- Zheng, Y.; Xing, Y.; Li, G.; Gao, J.; Li, R.; Liu, Q.; Yue, T.A. Comprehensive Review of Deactivation and Modification of Selective Catalytic Reaction Catalysts Installed in Cement Kilns. J. Environ. Sci. 2025, 148, 451–467. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudkhani, M.; Kasaeian, A.; Nazari, N.S.; Afshari, F.; Esmaeili Bidhendi, M. Enhancing Sustainability in Cement Manufacturing through Waste Heat Recovery and CCHP Systems. Energy 2025, 318, 134845. [Google Scholar] [CrossRef]
- Fayomi, G.U.; Mini, S.E.; Fayomi, O.S.I.; Ayoola, A.A. Perspectives on Environmental CO2 Emission and Energy Factor in Cement Industry. IOP Conf. Ser. Earth Environ. Sci. 2019, 331, 012035. [Google Scholar] [CrossRef]
- Neerup, R.; Øbro, K.L.; Løge, I.A.; Kottaki, N.; Frøstrup, C.F.; Gyorbiro, I.; Dimitriadi, M.; Halilov, H.; Jensen, S.; Karlsson, J.L. CO2 Capture Pilot Campaign: Understanding Solvent Degradation of CESAR1 in Cement Plant Operations. Chem. Eng. J. 2025, 510, 16152. [Google Scholar] [CrossRef]
- Attia, S. Net Zero Energy Buildings (NZEB): Concepts, Frameworks and Roadmap for Nnproject Analysis and Implementation; Butterworth-Heinemann: Oxford, UK, 2018; ISBN 0128124628. [Google Scholar]
- Amorim, A.; Filipe, R.M.; Matos, H.A. Analysis of Integrated Calcium Looping Alternatives in a Cement Plant. Chem. Eng. Sci. 2025, 313, 121709. [Google Scholar] [CrossRef]
- Asgharian, H.; Iov, F.; Nielsen, M.P.; Liso, V.; Burt, S.; Baxter, L. Analysis of Cryogenic CO2 Capture Technology Integrated with Water-Ammonia Absorption Refrigeration Cycle for CO2 Capture and Separation in Cement Plants. Sep. Purif. Technol. 2025, 353, 128419. [Google Scholar] [CrossRef]
- Ghalandari, V.; Majd, M.M.; Golestanian, A. Energy Audit for Pyro-Processing Unit of a New Generation Cement Plant and Feasibility Study for Recovering Waste Heat: A Case Study. Energy 2019, 173, 833–843. [Google Scholar] [CrossRef]
- Brandoni, C.; Kaldis, S.; Lappas, A.; Snape, C.; Jaffar, M.; Rolfe, A.; Hewitt, N.J.; Martinez, J.C.; Lysiak, B.; Huang, Y. Process Design and Techno-Economic Risk Assessment of a Solid Sorbent Silica Polyethyleneimine (Si-PEI) CCS Process Integrated into a Cement Plant. Energy 2025, 322, 135482. [Google Scholar] [CrossRef]
- Kim, A.; Miller, S.A. Meeting Industrial Decarbonization Goals: A Case Study of and Roadmap to a Net-Zero Emissions Cement Industry in California. Environ. Res. Lett. 2023, 18, 104019. [Google Scholar] [CrossRef]
- Driver, J.G.; Hills, T.; Hodgson, P.; Sceats, M.; Fennell, P.S. Simulation of Direct Separation Technology for Carbon Capture and Storage in the Cement Industry. Chem. Eng. J. 2022, 449, 137721. [Google Scholar] [CrossRef]
- Nazari, N.S.; Afshari, F.; Mahmoudkhani, M.; Ghaemi, A. Optimization of Carbon Dioxide Adsorption from Industrial Flue Gas Using Zeolite 13X: A Simulation Study with Aspen Adsorption and Response Surface Methodology. Results Eng. 2024, 24, 103206. [Google Scholar] [CrossRef]
- Sobuz, M.H.R.; Shuvo Dip, D.; Akid, A.S.M. Investigating the Combined Effect of Aggregate Size and Sulphate Attack on Producing Sustainable Recycled Aggregate Concrete. Aust. J. Civ. Eng. 2023, 21, 224–239. [Google Scholar] [CrossRef]
- Hossain, M.U.; Poon, C.S.; Wong, M.Y.K.; Khine, A. Techno-Environmental Feasibility of Wood Waste Derived Fuel for Cement Production. J. Clean. Prod. 2019, 230, 663–671. [Google Scholar] [CrossRef]
- Dankwa, J.; Jin, C.; Afrane, S.; Li, B.; Adun, H.; Liu, H.; Yao, M.; Morrow, D. Does China’s Pathway to Carbon Neutrality Require the Integration of Land-Based Biological Negative Emission Solutions with Geochemical and Chemical Alternatives? Sustain. Prod. Consum. 2024, 45, 27–41. [Google Scholar] [CrossRef]
- Sani, M.M.; Noorpoor, A.; Motlagh, M.S.-P. Optimal Model Development of Energy Hub to Supply Water, Heating and Electrical Demands of a Cement Factory. Energy 2019, 177, 574–592. [Google Scholar] [CrossRef]
- Verbruggen, H.; Kooistra, W.H.C.F. Morphological Characterization of Lineages within the Calcified Tropical Seaweed Genus Halimeda (Bryopsidales, Chlorophyta). Eur. J. Phycol. 2007, 39, 213–228. [Google Scholar] [CrossRef]
- Dijoux, L.; Verbruggen, H.; Mattio, L.; Duong, N.; Payri, C. Diversity of Halimeda (Bryopsidales, Chlorophyta) in New Caledonia: A Combined Morphological and Molecular Study. J. Phycol. 2012, 48, 1465–1481. [Google Scholar] [CrossRef]
- Cremen, M.C.M.; Huisman, J.M.; Marcelino, V.R.; Verbruggen, H. Taxonomic Revision of Halimeda (Bryopsidales, Chlorophyta) in South-Western Australia. Aust. Syst. Bot. 2016, 29, 41–54. [Google Scholar] [CrossRef]
- Borowitzka, M.A.; Larkum, A.W.D. Calcification in the Green Alga Halimeda: III. The Sources of Inorganic Carbon for Photosynthesis and Calcification and a Model of the Mechanism of Calcification. J. Exp. Bot. 1976, 27, 879–893. [Google Scholar] [CrossRef]
- Borowitzka, M.A.; Larkum, A.W.D. Calcification in the Green Alga Halimeda. I. An Ultrastructure Study of Thallus Development 1. J. Phycol. 1977, 13, 6–16. [Google Scholar] [CrossRef]
- Mayakun, J.; Bunruk, P.; Kongsaeng, R. Growth Rate and Calcium Carbonate Accumulation of Halimeda Macroloba Decaisne (Chlorophyta: Halimedaceae) in Thai Waters. Songklanakarin J. Sci. Technol. 2014, 36, 419–423. [Google Scholar]
- McNeil, M.A.; Webster, J.M.; Beaman, R.J.; Graham, T.L. New Constraints on the Spatial Distribution and Morphology of the Halimeda Bioherms of the Great Barrier Reef, Australia. Coral Reefs 2016, 35, 1343–1355. [Google Scholar] [CrossRef]
- Prathep, A.; Kaewsrikhaw, R.; Mayakun, J.; Darakrai, A. The Effects of Light Intensity and Temperature on the Calcification Rate of Halimeda Macroloba. J. Appl. Phycol. 2018, 30, 3405–3412. [Google Scholar] [CrossRef]
- Rees, S.A.; Opdyke, B.N.; Wilson, P.A.; Henstock, T.J. Significance of Halimeda Bioherms to the Global Carbonate Budget Based on a Geological Sediment Budget for the Northern Great Barrier Reef, Australia. Coral Reefs 2007, 26, 177–188. [Google Scholar] [CrossRef]
- Perry, C.T.; Morgan, K.M.; Salter, M.A. Sediment Generation by Halimeda on Atoll Interior Coral Reefs of the Southern Maldives: A Census-Based Approach for Estimating Carbonate Production by Calcareous Green Algae. Sediment. Geol. 2016, 346, 17–24. [Google Scholar] [CrossRef]
- Mayakun, J.; Liao, C.-P.; Liu, S.-L. The Standing Stock and CaCO3 Contribution of Halimeda Macroloba in the Tropical Seagrass-Dominated Ecosystem in Dongsha Island, the Main Island of Dongsha Atoll, South China Sea. J. Mar. Biol. Assoc. 2020, 100, 1219–1227. [Google Scholar] [CrossRef]
- VanTussenbroek, B.I.; VanDijk, J.K. Spatial and Temporal Variability in Biomass and Production of Psammophytic Halimeda Incrassata (Bryopsidales, Chlorophyta) in a Caribbean Reef Lagoon 1. J. Phycol. 2007, 43, 69–77. [Google Scholar] [CrossRef]
- Littler, M.M.; Littler, D.S.; Brooks, B.L. Herbivory, Nutrients, Stochastic Events, and Relative Dominances of Benthic Indicator Groups on Coral Reefs: A Review and Recommendations. In Proceedings of the Smithsonian Marine Science Symposium; Smithsonian Institution Scholarly Press: Washington, DC, USA, 2009. [Google Scholar]
- Ortegón-Aznar, I.; Chuc-Contreras, A.; Collado-Vides, L. Calcareous Green Algae Standing Stock in a Tropical Sedimentary Coast. J. Appl. Phycol. 2017, 29, 2685–2693. [Google Scholar] [CrossRef]
- Beach, K.; Walters, L.; Vroom, P.; Smith, C.; Coyer, J.; Hunter, C. Variability in the ecophysiology of Halimeda spp. (chlorophyta, bryopsidales) on conch reef, florida keys, USA. J. Phycol. 2003, 39, 633–643. [Google Scholar] [CrossRef]
- Vroom, P.S.; Smith, C.M.; Coyer, J.A.; Walters, L.J.; Hunter, C.L.; Beach, K.S.; Smith, J.E. Field Biology of Halimeda Tuna (Bryopsidales, Chlorophyta) across a Depth Gradient: Comparative Growth, Survivorship, Recruitment, and Reproduction. Hydrobiologia 2003, 501, 149–166. [Google Scholar] [CrossRef]
- de Macêdo Carneiro, P.B.; de Morais, J.O. Carbonate Sediment Production in the Equatorial Continental Shelf of South America: Quantifying Halimeda Incrassata (Chlorophyta) Contributions. J. S. Am. Earth Sci. 2016, 72, 1–6. [Google Scholar] [CrossRef]
- Carneiro, P.B.D.M.; Pereira, J.U.; Matthews-Cascon, H. Standing Stock Variations, Growth and CaCO3 Production by the Calcareous Green Alga Halimeda Opuntia. J. Mar. Biol. Assoc. 2018, 98, 401–409. [Google Scholar] [CrossRef]
- Stanley, S.M.; Ries, J.B.; Hardie, L.A. Increased production of calcite and slower growth for the major sediment-producing alga halimeda as the Mg/Ca ratio of seawater is lowered to A “calcite sea” level. J. Sediment. Res. 2010, 80, 6–16. [Google Scholar] [CrossRef]
- Castro-Sanguino, C.; Bozec, Y.-M.; Mumby, P.J. Dynamics of Carbonate Sediment Production by Halimeda: Implications for Reef Carbonate Budgets. Mar. Ecol. Prog. Ser. 2020, 639, 91–106. [Google Scholar] [CrossRef]
- Ridgwell, A.; Hargreaves, J.C. Regulation of Atmospheric CO2 by Deep-Sea Sediments in an Earth System Model. Glob. Biogeochem. Cycles 2007, 21, GB2008. [Google Scholar] [CrossRef]
- García-gusano, D.; Garraín, D.; Herrera, I.; Cabal, H.; Lechón, Y. Life Cycle Assessment of Applying CO2 Post-Combustion Capture to the Spanish Cement Production. J. Clean. Prod. 2025, 104, 328–338. [Google Scholar] [CrossRef]
- Habert, G.; Billard, C.; Rossi, P.; Chen, C.; Roussel, N. Cement Production Technology Improvement Compared to Factor 4 Objectives. Cem. Concr. Res. 2020, 40, 820–826. [Google Scholar] [CrossRef]
- Porter, H.; Mukherjee, A.; Tuladhar, R.; Dhami, N.K. Life Cycle Assessment of Biocement: An Emerging Sustainable Solution? Sustainability 2021, 13, 13878. [Google Scholar] [CrossRef]
- Rafiq, A.; Ren, J.; Laosiripojana, N.; Silalertruksa, T. Life Cycle Environmental and Economic Viability Analysis of CO2 Utilization for Chemical Production in the Cement Sector. Sustain. Prod. Consum. 2025, 58, 364–384. [Google Scholar] [CrossRef]
- Sambataro, L.; Bre, F.; Ukrainczyk, N.; Koenders, E.A.B. Environmental Benchmarks for the European Cement Industry. Sustain. Prod. Consum. 2024, 45, 429–449. [Google Scholar] [CrossRef]
- Benhelal, E.; Zahedi, G.; Hashim, H.A. Novel Design for Green and Economical Cement Manufacturing. J. Clean. Prod. 2012, 22, 60–66. [Google Scholar] [CrossRef]
- Pei, X.; He, B.; Yan, L.; Wang, C.; Song, W.; Song, J. Process Simulation of Oxy-Fuel Combustion for a 300 MW Pulverized Coal-Fired Power Plant Using Aspen Plus. Energy Convers. Manag. 2013, 76, 581–587. [Google Scholar] [CrossRef]
- Yadav, S.; Mondal, S.S. Numerical Investigation of 660 MW Pulverized Coal-Fired Supercritical Power Plant Retrofitted to Oxy-Coal Combustion. Int. J. Greenh. Gas Control 2021, 105, 103227. [Google Scholar] [CrossRef]
- Aldieb, M.A.; Ibrahim, H.G. Variation of Feed Chemical Composition and Its Effect on Clinker Formation–Simulation Process. In Proceedings of the World Congress on Engineering and Computer Science; International Association of Engineers: Hong Kong SAR, China, 2010; Volume 2, pp. 1–7. [Google Scholar]
- Mcdonald, L. Precipitated Calcium Carbonate as Sustainable Supplementary Cementitious Material to Reduce the Environmental Impact of Portland Cement. Ph.D. Thesis, University of Aberdeen, Aberdeen, Scotland, 2022. [Google Scholar]
- Kosmatka, S.H.; Kerkhoff, B.; Panarese, W.C. Design and Control of Concrete Mixtures; Portland Cement Assn: Washington, DC, USA, 2008; ISBN 0893122173. [Google Scholar]
- Irfan, H.M.; Iqbal, K.; Taipabu, M.I.; You, C.Y.; Mazumdar, D.; Wu, W. Decarbonization Frameworks to Industrial-Scale Ammonia Production: Techno-Economic and Environmental Implications. Int. J. Hydrogen Energy 2024, 78, 580–593. [Google Scholar] [CrossRef]
- Riondet, L.; Rio, M.; Zwolinski, P. Emerging Technologies Upscaling: A Framework for Matching LCA Practices with Upscaling Archetypes. Sustain. Prod. Consum. 2024, 50, 347–363. [Google Scholar] [CrossRef]
- Ige, O.E.; Olanrewaju, O.A.; Duffy, K.J.; Obiora, C.A. Review of the Effectiveness of Life Cycle Assessment for Gauging Environmental Impacts from Cement Production. J. Clean. Prod. 2021, 324, 129213. [Google Scholar] [CrossRef]
- ISO14040:2006; Environmental Management-Life Cycle Assessment-Principles and Framework. International Organization for Standardization: Geneva, Switzerland, 2006.
- Irfan, H.M.; Yang, C.; Wu, W.; Lin, B.; Shiau, J. Environmental and Techno-Economic Analyses of Oxygen Blast Furnace Systems Integrated with Carbon Capture and Utilization Strategy. Fuel Process. Technol. 2025, 274, 108238. [Google Scholar] [CrossRef]
- Arias, A.; Feijoo, G.; Moreira, M.T. Macroalgae as a Sustainable Biostimulant for Crop Production According to Techno-Economic and Environmental Criteria. Sustain. Prod. Consum. 2024, 48, 169–180. [Google Scholar] [CrossRef]
- Naqi, A.; Jang, J.G. Recent Progress in Green Cement Technology Utilizing Low-Carbon Emission Fuels and Raw Materials: A Review. Sustainability 2019, 11, 537. [Google Scholar] [CrossRef]
- Medas, D.; Cappai, G.; DeGiudici, G.; Piredda, M.; Podda, S. Accelerated Carbonation by Cement Kiln Dust in Aqueous Slurries: Chemical and Mineralogical Investigation. Greenh. Gases Sci. Technol. 2017, 7, 692–705. [Google Scholar] [CrossRef]
- Chiarathanakrit, C.; Mayakun, J.; Prathep, A.; Kaewtatip, K. Comparison of the Effects of Calci Fi Ed Green Macroalga (Halimeda Macroloba Decaisne) and Commercial CaCO3 on the Properties of Composite Starch Foam Trays. Int. J. Biol. Macromol. 2019, 121, 71–76. [Google Scholar] [CrossRef]
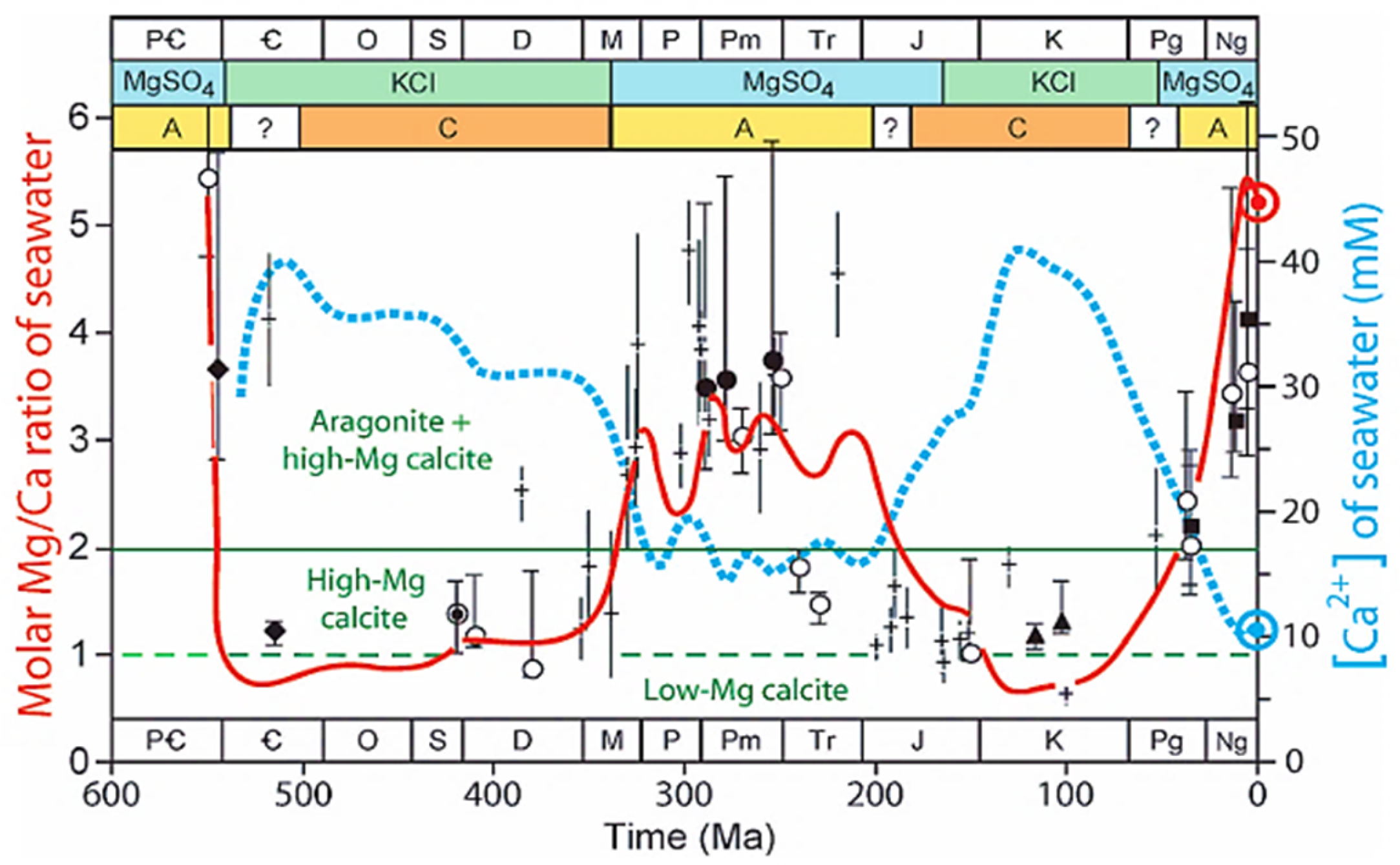

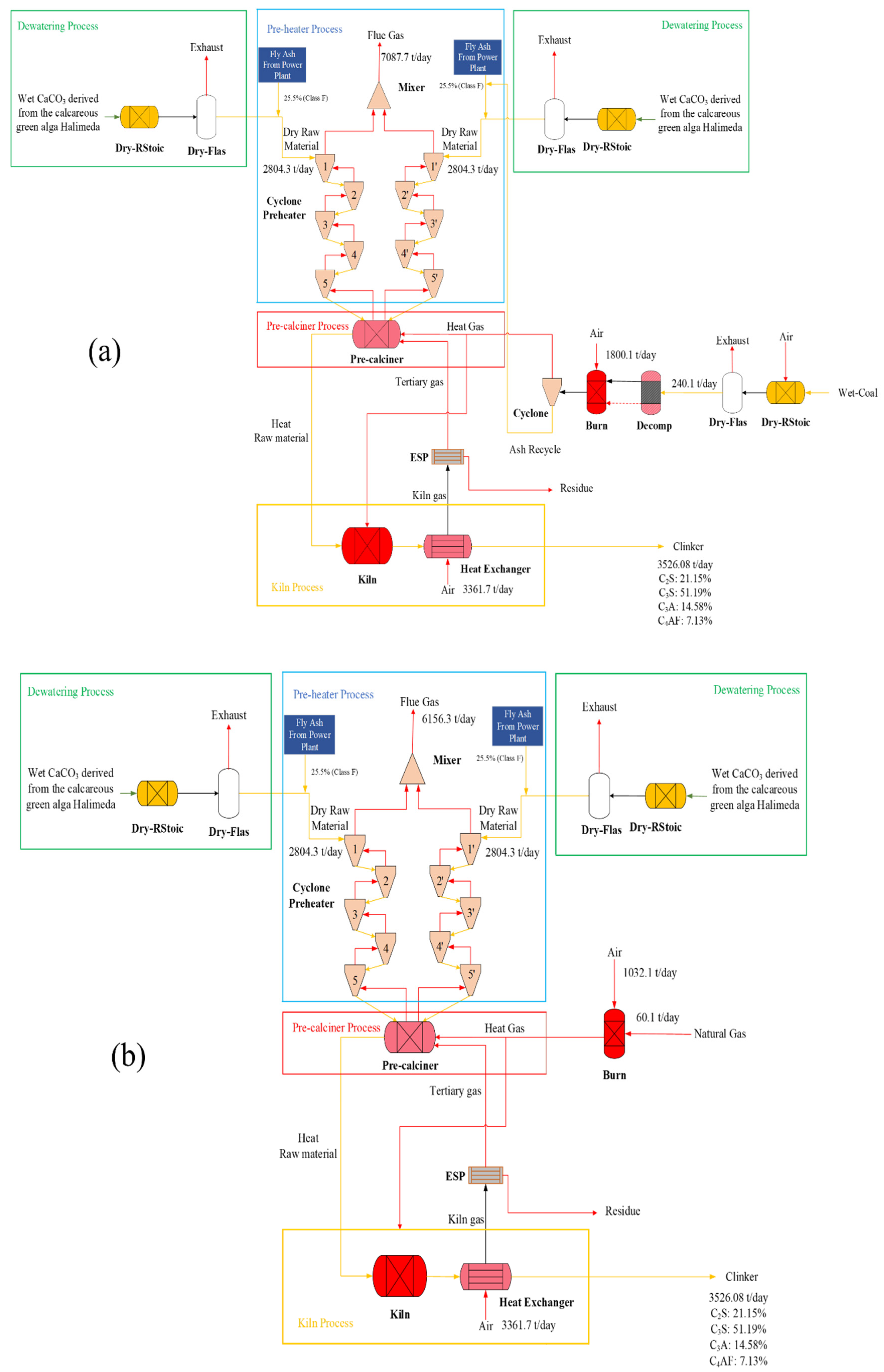
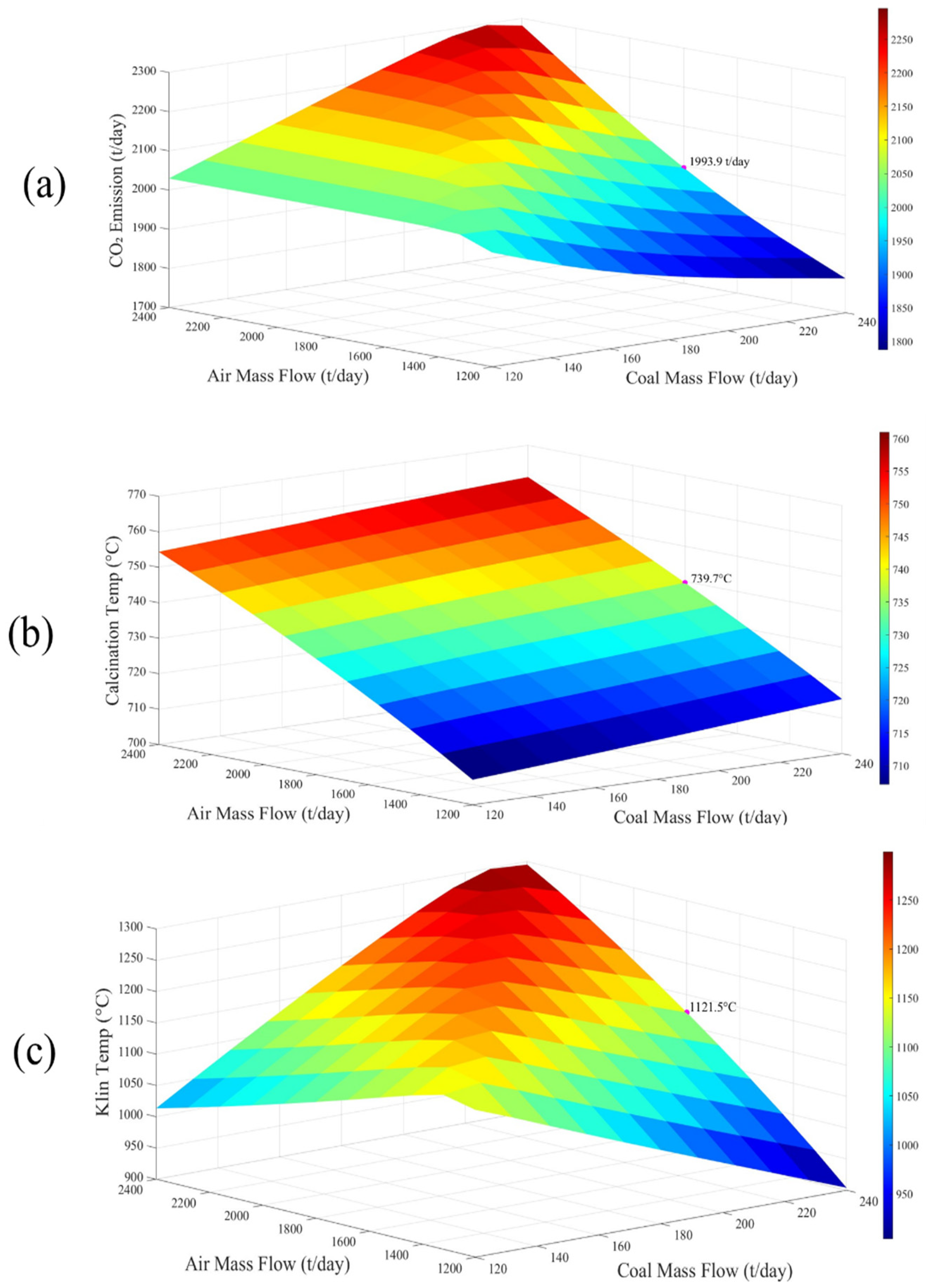

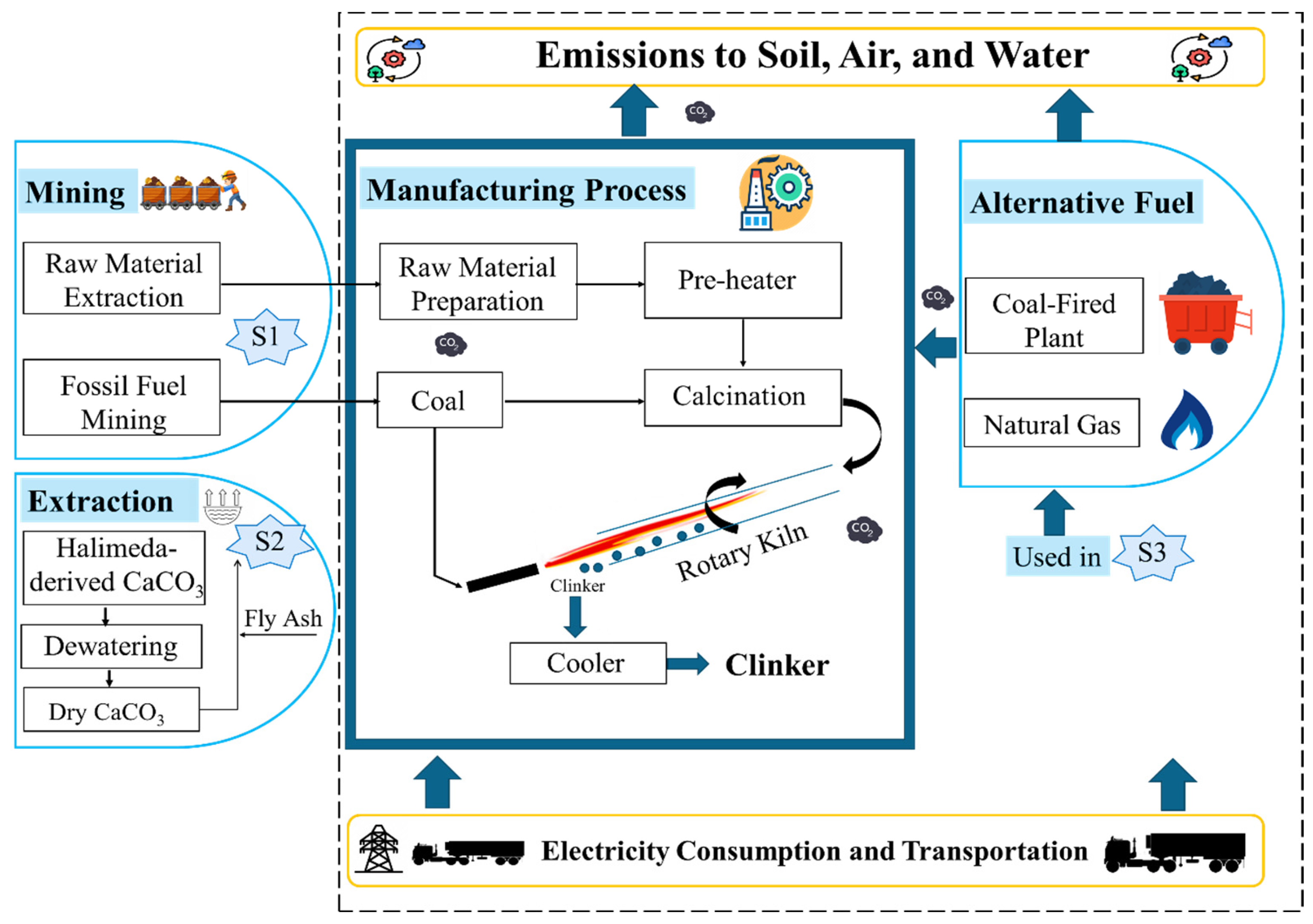
| Analysis | Value (wt%) |
|---|---|
| Proximate analysis | |
| Volatile matter (Vd) | 45.7 |
| Ash (Ad) | 9.2 |
| Moisture (Mar) | 10 |
| Fixed carbon (FCd) | 45.1 |
| Ultimate analysis | |
| Carbon (Cd) | 67.1 |
| Hydrogen (Hd) | 4.8 |
| Nitrogen (Nd) | 1.1 |
| Sulfur (Sd) | 1.3 |
| Oxygen (Od) | 16.4 |
| Chlorine (Cld) | 0.1 |
| Total sulfur | |
| Sulfate sulfur (Sp) | 0.6 |
| Sulfide sulfur (Ss) | 0.1 |
| Organic sulfur (So) | 0.6 |
| Net calorific value | (MJ/kg) |
| Qnet | 22.53 |
| Component | Composition (%wt) |
|---|---|
| CaO | 41.51 |
| SiO2 | 14.03 |
| MgO | 2.59 |
| Al2O3 | 3.39 |
| Fe2O3 | 2.54 |
| SO3 | 0.3 |
| K2O | 0.57 |
| Na2O | 0.24 |
| Loss of ignition | 34.83 |
| Reaction Name | Reaction | Temperature Range (°C) | Heat of Reaction () (mol−1) |
|---|---|---|---|
| Decalcination | 700–900 | +179.4 | |
| MgCO3 dissociation | 700–900 | +117.61 | |
| C2S formation | 1200–1280 | −127.6 | |
| C3S formation | 1200–1280 | +16 | |
| C3A formation | 1200–1280 | +21.8 | |
| C4AF formation | 1200–1280 | −41.3 |
| Case Study [44] | Simulation (Scenario 1) | Errors (%) | |||
|---|---|---|---|---|---|
| Stream | Flow Rate (t/Day) | Temperature (°C) | Flow Rate (t/Day) | Temperature (°C) | |
| Feed | 5576.64 | 50 | 5632.512 | 50 | 1% |
| Calciner Feed | 5548.8 | 760 | 5632.512 | 782 | 2% |
| Kiln Feed | 3690.48 | 890 | 3693.12 | 890 | 0% |
| Hot Clinker | 3600 | 1500 | 3665.4 | 1500 | 2% |
| Cold Clinker | 3600 | 80 | 3665.4 | 80 | 2% |
| Exhaust Gas | 7563.12 | 315 | 7364.64 | 315 | −3% |
| Mineral Composition | Bouge Calculation | Case Study [44] | Simulation (S1) | Error % |
|---|---|---|---|---|
| C2S | 21.092% | 22.42% | 22.032% | 4.45% |
| C3S | 53.856% | 52.62% | 53.332% | −0.97% |
| C3A | 7.190% | 7.34% | 7.313% | 1.71% |
| C4AF | 11.862% | 11.88% | 11.860% | 0.00% |
| SO3 | 0.68% | 0.66% | 0.68% | 0.00% |
| K2O | 0.88% | 0.87% | 0.88% | 0.00% |
| Na2O | 0.49% | 0.47% | 0.47% | 0.01% |
| MgO | 3.63% | 3.74% | 3.62% | 0.35% |
| Scenarios | 1 | 2 | 3 | |
|---|---|---|---|---|
| Stages | Materials | |||
| Input | N2 (kg/h) | 1.191 | 1.156 | 1.156 |
| O2 (kg/h) | 0.316 | 0.307 | 0.307 | |
| Coal (kg/h) Natural gas (kg/h) | 0.039 - | 0.068 - | - 0.017 | |
| Ash (kg/h) | 0.175 | 0.11 | 0.11 | |
| CaCO3 (kg/h) | 1.128 | 1.008 | 1.008 | |
| SiO2 (kg/h) | 0.213 | 0.210 | 0.210 | |
| Al2O3 (kg/h) | 0.051 | 0.101 | 0.101 | |
| Fe2O3 (kg/h) MgCO3 (kg/h) K2O (kg/h) Na2O (kg/h) CaO (kg/h) | 0.038 0.082 0.008 0.003 0.004 | 0.040 0.089 0.004 0.004 0.020 | 0.040 0.089 0.004 0.004 0.020 | |
| Transport (tkm) | 0.770 | 0.159 | 0.159 | |
| Heat (MW) | 0.00035 | 0.00032 | 0.00032 | |
| Emissions to air | H2O (kg/h) | 0.016 | 0.0069 | 0.0382 |
| N2 (kg/h) | 1.191 | 1.157 | 1.157 | |
| O2 (kg/h) | 0.229 | 0.269 | 0.239 | |
| NO2 (kg/h) | 7.7 × 10−7 | 2.98 × 10−13 | 2.14 × 10−6 | |
| NO (kg/h) | 0.001 | 3.86 × 10−7 | 0.00068 | |
| S (kg/h) | 4.4 × 10−11 | 1.53 × 10−6 | 0 | |
| SO2 (kg/h) SO3 (kg/h) H2 (kg/h) CO2 (kg/h) Ash (kg/h) CO (kg/h) C (kg/h) | 0.0003 3.2 × 10−7 3.2 × 10−7 0.689 0.177 0.0005 0.00001 | 0.0008 7.87 × 10−10 0.0008 0.565 0.09 0.065 0 | 0 0 1.362 × 10−7 0.529 0 2.877 × 10−6 0 | |
| Solid output | ||||
| Residue (kg/h) | 0.201 | 0.111 | 0.004 | |
| Product | Clinker (kg/h) | 1 | 1 | 1 |
| CO2 Emission (ton/ton of Clinker) | CO2 Sequestration (ton/ton of Clinker) | Net CO2 (ton/ton of Clinker) | Ref. | |
|---|---|---|---|---|
| Portland cement | 0.75–0.9 | - | 0.75–0.9 | [56,57] |
| Scenario 1 | 0.72 | - | 0.72 | This Study |
| Scenario 2 | 0.565 | 0.600 [58] | −0.035 | This Study |
| Scenario 3 | 0.517 | 0.600 [58] | −0.083 | This Study |
| Scenarios | 1 | 2 | 3 | |
|---|---|---|---|---|
| Damage Category | Unit | |||
| Human Health | mPt | 123.11 | 101.08 | 93.80 |
| Ecosystems | mPt | 1.850 | 0.954 | 0.740 |
| Resources | mPt | 0.464 | 0.138 | 0.146 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Irfan, H.M.; Wu, C.-Y.; Hussain, M.S.; Wu, W. Environmental Impact Assessment of New Cement Production Blending Calcareous Green Algae and Fly Ash. Processes 2026, 14, 299. https://doi.org/10.3390/pr14020299
Irfan HM, Wu C-Y, Hussain MS, Wu W. Environmental Impact Assessment of New Cement Production Blending Calcareous Green Algae and Fly Ash. Processes. 2026; 14(2):299. https://doi.org/10.3390/pr14020299
Chicago/Turabian StyleIrfan, Hafiz M., Chi-Yun Wu, Muhammad Saddam Hussain, and Wei Wu. 2026. "Environmental Impact Assessment of New Cement Production Blending Calcareous Green Algae and Fly Ash" Processes 14, no. 2: 299. https://doi.org/10.3390/pr14020299
APA StyleIrfan, H. M., Wu, C.-Y., Hussain, M. S., & Wu, W. (2026). Environmental Impact Assessment of New Cement Production Blending Calcareous Green Algae and Fly Ash. Processes, 14(2), 299. https://doi.org/10.3390/pr14020299









