Abstract
This study introduces an adaptable, intelligent prototype designed to optimize lentil seed germination and biomass accumulation via controlled UV-C radiation and Spirulina supplementation. Building on earlier experiments that separately and jointly assessed these treatments, the work presents a novel seed-treatment chamber that combines environmental sensing, real-time delivery mechanisms, and a machine-learning decision engine. The system automatically selects among three operational modes, Fast Germination, High Biomass, and Flavonoid Enrichment, each targeting a specific agronomic goal. To uncover the most influential treatment factors, the authors applied Analysis of Variance (ANOVA) and Principal Component Analysis (PCA), revealing key response patterns that inform mode definitions. A regression-based AI model was then trained on experimental data to predict treatment outcomes and dynamically adjust parameters. Model performance metrics demonstrate high predictive fidelity, with a Mean Absolute Error (MAE) of 2.1267%, indicating an average deviation of just over two percentage points between predicted and observed germination rates. In comparison, a Mean Squared Error (MSE) of 6.4598 and a corresponding Root Mean Squared Error (RMSE) of 2.5416% confirm consistently low squared deviations. An R2 score of 0.8702 indicates that the model accounts for approximately 87% of the variance in germination outcomes, underscoring the robustness of the regression approach. Importantly, the specific treatment ranges illustrated in this study are not direct replications of prior data, but rather representative values drawn from earlier research to demonstrate the framework’s applicability. By abstracting treatment parameters into realistic ranges, the paper shows how the chamber can accommodate various empirical datasets. The principal contribution lies in offering a generalizable methodology for designing AI-enhanced seed-treatment systems. This conceptual framework can be tailored to multiple crops and cultivation environments, paving the way for scalable, precision agriculture solutions that integrate automated monitoring, intelligent control, and real-time optimization.
1. Introduction
The optimization of seed germination is a crucial area of research in agriculture, particularly in legumes such as lentils (Lens culinaris Medik.), which are known for their high nutritional value and potential for sustainable food production. Several studies have explored different seed priming techniques to enhance germination rates, seedling vigor, and nutrient content [1,2]. Among these, biophysical methods such as ultraviolet (UV) radiation [3,4,5] and biological stimulants like Spirulina maxima [6,7,8] have gained attention due to their potential to modify physiological properties in a controlled and environmentally friendly manner. However, there is a lack of information on creating functional prototypes to achieve the required conditions for improving the germination process. Recent advancements have introduced innovative setups to address this gap. For instance, one study [9] utilized a growth chamber with controlled temperature and humidity, integrating dimmable LED arrays for spectral treatments and real-time, noninvasive plant health monitoring through spectroscopy and mathematical modeling. Another approach [10] employed climatic chambers to optimize seed germination conditions for pea and radish using Bayesian optimization (BO), allowing precise regulation of temperature and irrigation timing to enhance germination efficiency. Additionally, a novel encapsulated prototype [11] provided a highly controlled environment for accelerated plant growth, incorporating sensors, LED lighting, irrigation, and heating/cooling systems to support AI-driven crop modeling. These advancements highlight the potential of integrating precision-controlled environments with computational techniques to enhance seed priming outcomes in modern agriculture.
UV-C radiation (200–280 nm) has been extensively used for antimicrobial purposes in the food industry and postharvest treatments. However, its effects on seed germination and secondary metabolite production remain under investigation. Studies suggest that UV-C treatment can influence seed dormancy, alter biochemical pathways, and enhance the accumulation of secondary metabolites such as flavonoids and saponins.
Flavonoids are plant secondary metabolites renowned for their antioxidant, antimicrobial, and signaling functions, and boosting their levels confers multiple agricultural benefits. By scavenging reactive oxygen species and stabilizing cellular structures, increased flavonoid content enhances seed and seedling tolerance to abiotic stresses such as salinity and extreme temperatures [12]. The antimicrobial properties of flavonoids also fortify young plants against soil-borne pathogens, reducing the need for synthetic pesticides. From a nutritional standpoint, increasing flavonoid concentrations in food crops enhances their functional value and market appeal. At the same time, higher phytochemical levels contribute to prolonged seed viability and vigor during storage and transportation [13,14]. Implementing Flavonoid Enrichment Mode supports more resilient, nutritious, and sustainable crop production through precise UV-C priming, which boosts key bioactive compounds.
As a non-ionizing seed priming method, UV-C radiation is gaining attention for its ability to improve germination rates, crop resilience, and productivity, while reducing environmental impact. UV-C priming has been shown to induce physiological and biochemical changes that enhance seed storability and stress tolerance [12]. Additionally, UV radiation, including UV-B and UV-C, has been linked to increased photosynthesis, pigment synthesis, and antioxidant activity, which contribute to improved resilience against abiotic stressors like salinity and extreme temperatures [13]. UV-C treatments have also shown potential in stimulating phytochemical production and extending postharvest shelf life, although further research is needed to optimize application parameters and better understand the underlying mechanisms [14]. These findings highlight the potential of UV-C as a sustainable agricultural technique, warranting continued investigation for refining its effectiveness across various crops and environments.
On the other hand, Spirulina maxima, a cyanobacterium rich in proteins, vitamins, and bioactive compounds, has been explored for its biofertilizer properties. Studies show that Spirulina conditioning can improve seedling growth and help mitigate stress from physical treatments like UV-C radiation [8,15]. As an eco-friendly alternative to synthetic agrochemicals, Spirulina and other microalgae contribute to crop productivity and disease resistance through bioactive compounds, including amino acids, phytohormones, and micronutrients. Their high photosynthetic efficiency and adaptability make them valuable biostimulants, biofertilizers, and biopesticides [16]. Research indicates that Spirulina can improve soil fertility by increasing nitrogen, phosphorus, and nitrate levels, thus enhancing nutrient availability through a circular economy approach [17]. Additionally, Spirulina has demonstrated significant potential in improving maize growth, yield, and germination when used as a biofertilizer. In combination with cow dung manure, it improved early-stage plant growth by up to 51.1% after 60 days, significantly boosting plant height, biochemical composition, and mineral content [18]. These findings underscore Spirulina’s potential as a sustainable, eco-friendly biofertilizer that supports nutrient recycling and agricultural productivity.
As a result, this study presents the conceptual design and AI-based optimization of a functional prototype aimed at enhancing lentil seed germination and biomass accumulation through the controlled application of UV-C radiation and Spirulina supplementation. While previous experimental research [8] evaluated the individual and combined effects of these treatments on lentil seed germination and biochemical composition, the present work advances the field by introducing the conceptual design of an adaptable, intelligent seed treatment chamber. The prototype integrates environmental monitoring, real-time treatment delivery, and machine learning-based decision-making, enabling automated selection of treatment modes such as Fast Germination, High Biomass, and Flavonoid Enrichment. Statistical methods, including Analysis of Variance (ANOVA) and Principal Component Analysis (PCA), were employed to identify key patterns in treatment responses. A regression-based AI model facilitates the dynamic adjustment of treatment parameters based on predefined agronomic objectives.
The experimental findings reported in [8] are used in this study as a reference to inform and approximate realistic conditions during the development of the conceptual prototype. However, the specific data points and results from [8] are not directly reused in this work since this paper proposes a conceptual prototype that could be adapted to different conditions and seeds. Furthermore, while [8] presents a partially instrumented prototype, it does not offer a fully automated or AI-integrated system. In contrast, this study contributes a generalized and scalable methodology for the conceptual design of AI-enhanced seed treatment prototypes. The proposed treatment conditions are illustrative and intended to demonstrate the methodology’s adaptability across different seed types and agricultural contexts. This framework can be further refined and validated using crop-specific empirical data, supporting the development of intelligent, automated solutions for precision agriculture.
In [8], statistical techniques such as Analysis of Variance (ANOVA) and Principal Component Analysis (PCA) to optimize the conceptual design are used; it evaluates the impact of different UV-C exposure times and Spirulina concentrations on germination rate, fresh and dry biomass accumulation, and flavonoid content to create a conceptual design of a functional prototype. These findings reveal that while UV-C exposure alone does not significantly enhance germination, the integration of Spirulina treatment substantially improves seedling development. While UV-C treatment alone did not significantly enhance germination rates, a slight improvement in early germination (24–48 h) was observed at 5 min of exposure. Spirulina conditioning, particularly at moderate concentrations (0.5–0.75%), had a more pronounced positive effect on physiological parameters such as aerial part and seedling fresh weight, and dry weight. The combination of 5 min UV-C exposure with 0.5–0.75% Spirulina provided the most balanced results, improving seedling development without causing structural damage. In contrast, a 10 min UV-C treatment could negatively impact on seed physiology, leading to tissue degradation and reduced water content, especially under higher Spirulina concentrations. For the proof-of-concept design, optimal germination rates were proposed to be 87.11% at 24 h and 93.73% at 48 h, under conditions with no or minimal UV-C exposure and a Spirulina concentration of 1.5 g/100 mL. These values can be adjusted according to seed type and specific environmental conditions. One of the key contributions of this study is the development of a robust methodology for designing a conceptual prototype based on empirical data. The experimental data presented in [8] served primarily as a reference to approximate real-world conditions. However, the design parameters can be further refined and proposed through targeted experiments tailored to specific agricultural needs. Thus, it is proposed to conceptually design an AI-optimized functional prototype that dynamically regulates seed treatment conditions. This automated seed treatment chamber integrates monitoring and control of UV-C exposure and Spirulina supplementation, enabling users to select specific germination goals such as Fast Germination, High Biomass, or Flavonoid Enrichment. The prototype utilizes machine learning algorithms to adjust treatment conditions in real-time, ensuring adaptive and optimized seed conditioning [8].
This research offers several significant contributions. It presents a methodology based on previously published experimental results to design a functional prototype for seed priming. The study analyzes the effects of UV-C and Spirulina using ANOVA and PCA to optimize germination conditions. These statistical tools have successfully reduced the complexity of hyperspectral data in various agricultural applications. For example, ANOVA and PCA were used to distinguish orchard tree species by analyzing spectral data, particularly in the near-infrared and red-edge regions, as shown in [19]. Similarly, in [20], these techniques were applied to UAV-based spectral data for olive cultivar recognition, with ANOVA identifying significant variations in vegetation indices and PCA enhancing data interpretation. Furthermore, in [21], ANOVA and PCA were used to validate a novel EDXRFS spectroscopy method for rapid soil macronutrient analysis, confirming its accuracy against reference standards at a 95% confidence level. This research introduces a data-driven optimization strategy, employing PCA to uncover key relationships between germination rates, biomass accumulation, and flavonoid content. Additionally, the study highlights the development of a functional prototype incorporating AI-enhanced seed treatment technology. By integrating machine learning-driven optimization, the system dynamically adjusts treatment conditions, demonstrating its scalability and adaptability for various seed treatment goals, and positioning it as a precision agriculture solution.
By integrating biophysical and biological seed treatment approaches with AI-driven automation, this study presents a novel and scalable solution for enhancing seed quality, promoting agricultural sustainability, and contributing to food security initiatives by designing functional prototypes that leverage this information [22,23].
This paper builds upon the experimental results previously reported in [8], where the individual and combined effects of UV-C radiation and Spirulina maxima conditioning on lentil seed germination and biochemical composition were explored. While [8] focused solely on biological outcomes under predefined treatment conditions, the present study leverages those findings to develop a conceptual functional design, AI-optimized prototype that can dynamically adapt treatment parameters to user-defined germination objectives. The core novelty of this research lies in the conceptual design, integration of intelligent control systems, and implementation of machine learning algorithms to automate the seed priming process. The optimization of seed germination is a crucial area of research in agriculture, particularly in legumes such as lentils (Lens culinaris Medik.), which are known for their high nutritional value and potential for sustainable food production. Several studies have investigated various seed priming techniques to improve germination rates, seedling vigor, and nutrient content [1,2]. Among these, biophysical methods such as ultraviolet (UV) radiation [3,4,5] and biological stimulants like Spirulina maxima [6,7,8] have gained attention due to their potential to modify physiological properties in a controlled and environmentally friendly manner. However, existing studies primarily focus on biological outcomes under controlled conditions and lack comprehensive, automated systems capable of delivering and adjusting treatments in real-time.
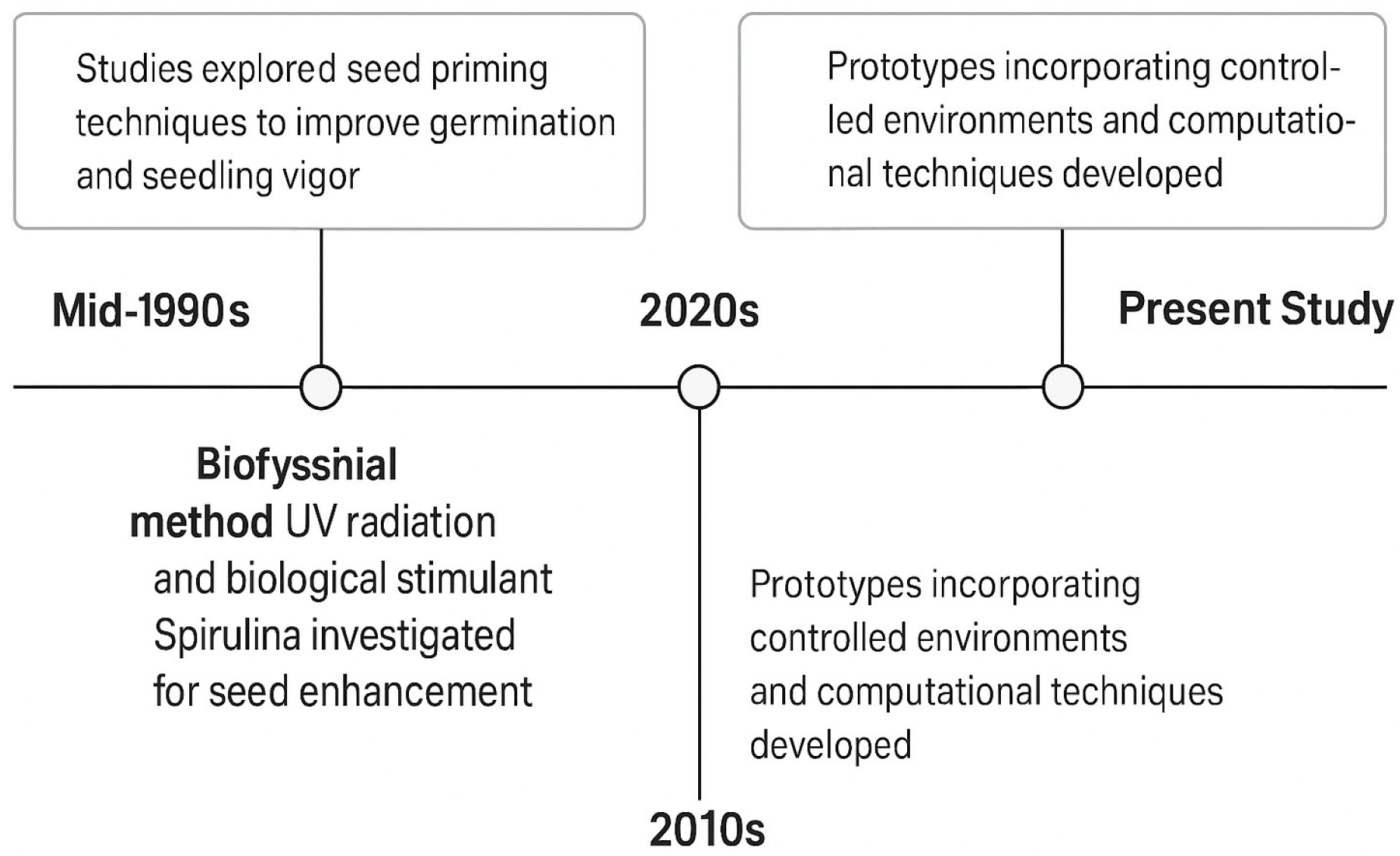
Table 1 summarizes representative previous work on seed priming prototypes, highlighting key technologies, target crops, and identified gaps. Figure 1 illustrates the timeline of the evolution of UVC prototypes.

Table 1.
UV-C prototype evolution timeline based on the information presented in [24].
Figure 1.
Timeline of UV-C prototypes.
These previous efforts demonstrate significant advances in controlled environment systems and analytical methods. Yet, none provide a fully automated, AI-integrated platform capable of real-time adjustment of both physical (UV-C) and biological (Spirulina) treatments. To address this gap, this study presents the conceptual design and AI-based optimization of an adaptable seed treatment chamber that integrates environmental monitoring, real-time UV-C and Spirulina delivery, and machine learning-driven decision-making. The remainder of this manuscript is organized as follows. Section 2 describes the materials and methods, including statistical analyses and the conceptualization of the prototype. Section 3 presents the results of AI-driven optimization and prototype evaluation. Section 4 discusses uncertainty reduction strategies based on ISO/IEC standards. Section 5 examines the integration of the prototype into the Penta-S framework from the end-user’s perspective. Section 6 presents a comparison of manual and expected automation proposals, and Section 7 provides a comprehensive discussion of the findings and their implications. Finally, Section 8 concludes the paper and outlines directions for future work.
2. Materials and Methods
2.1. Results of ANOVA Analysis on UV-C Exposure and Spirulina Concentration
The experimental data referenced in this study are derived from [8], where an Analysis of Variance (ANOVA) using SAS version 2008, was conducted to evaluate the effects of UV-C exposure duration (around 0, 5, and 10 min) and Spirulina concentration (around 0, 0.5, 0.75, and 1.5 g/100 mL) on lentil seed germination at 48 h. For designing the general conceptual prototype, precise experimental values are not required since they are only used to describe the proposed methodology. Thus, the results used in this study indicated that neither UV-C exposure (p = 0.2057) nor Spirulina concentration (p = 0.1996) had a statistically significant effect on germination at the 0.05 significance level. These findings suggest that while trends in germination improvement were observed, particularly at moderate Spirulina levels and lower UV-C exposure, they were not statistically conclusive within the tested parameter range.
The experimental data used in this study as a reference are drawn from [8], where an Analysis of Variance (ANOVA) was employed to evaluate the effects of UV-C exposure time (0, 5, and 10 min) and Spirulina concentration (0, 0.5, 0.75, and 1.5 g/100 mL) on lentil seed germination at 48 h. However, it is not necessary to use the same values obtained in the study. Hence, the results that are used in this paper indicated that neither UV-C exposure (p = 0.2057) nor Spirulina concentration (p = 0.1996) had a statistically significant effect on germination at the 0.05 significance level.
This suggests that, within the tested parameter ranges, these factors did not exert a significant independent effect on germination rates. However, the substantial residual variance observed implies the potential presence of interaction effects or non-linear relationships. A more detailed evaluation incorporating interaction terms or advanced modeling approaches may be necessary to fully explain the combined influence of UV-C radiation and Spirulina conditioning on seed germination dynamics.
2.1.1. Determination of Optimal Germination Conditions
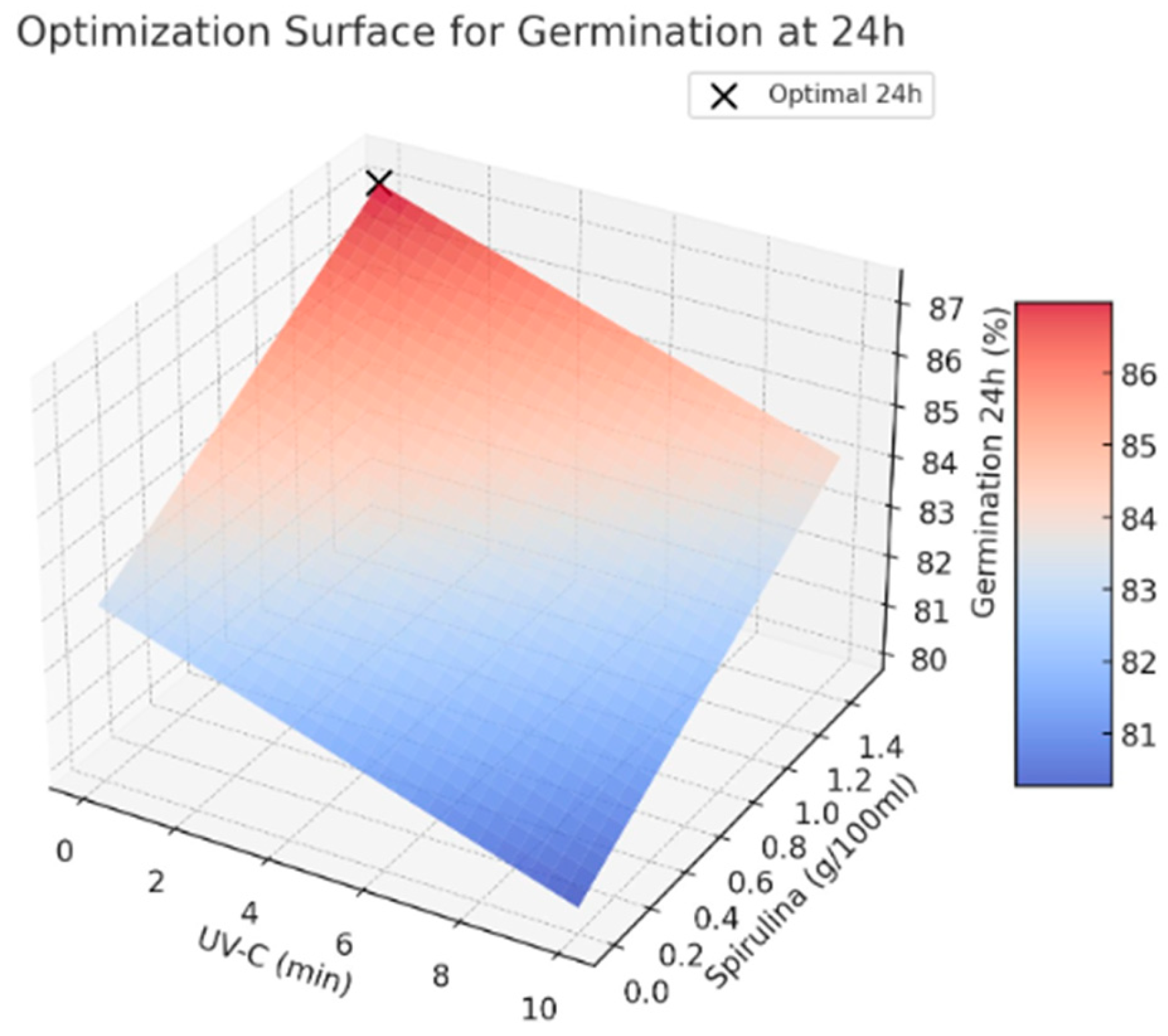
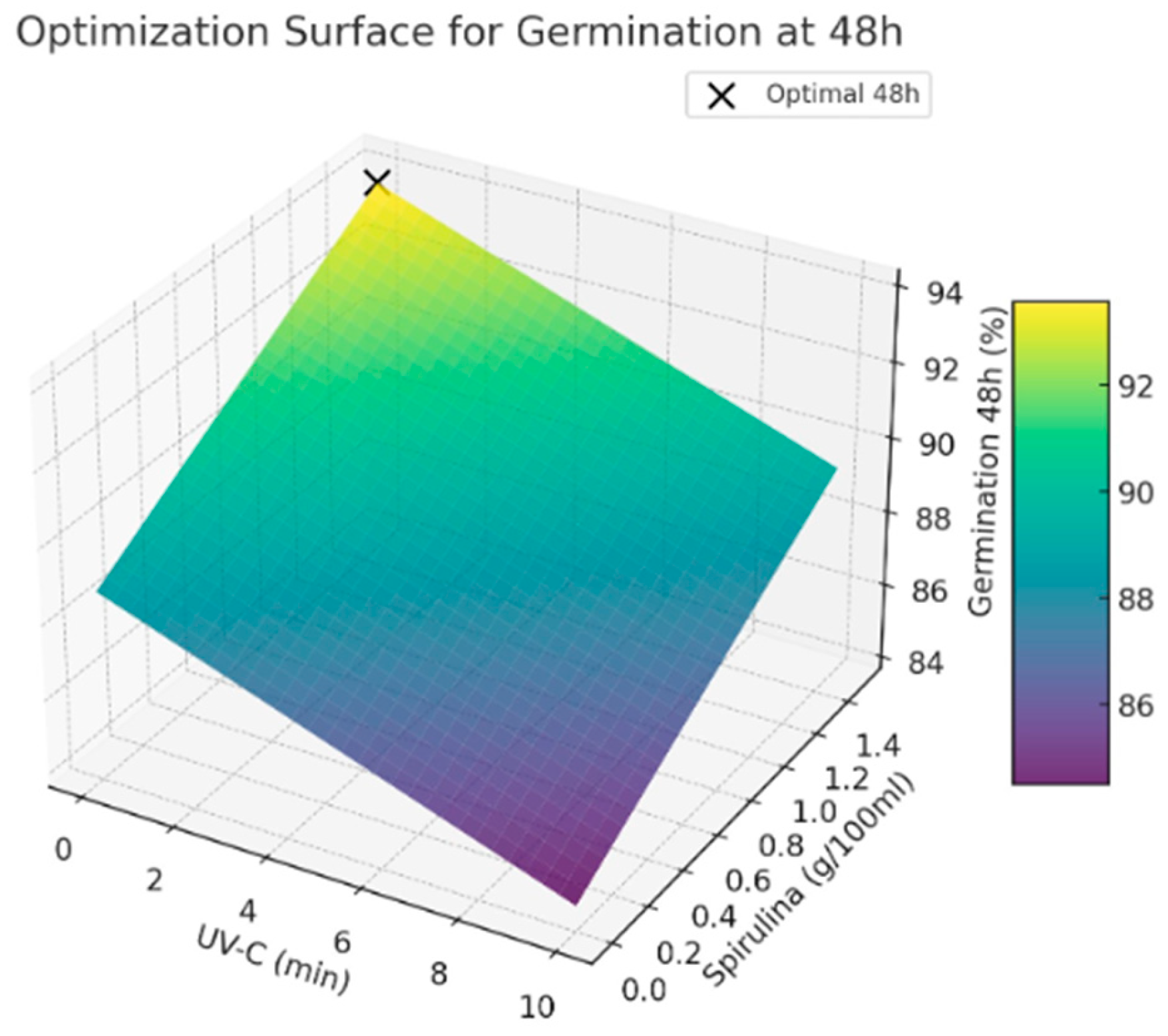
For the conceptual design of the prototype, representative optimal conditions for maximizing germination rates at 24 and 48 h are proposed. These values are illustrative and serve to demonstrate the methodological framework; they can be modified based on seed species and specific application needs. While the experimental data from [8] were used to approximate realistic parameters, this study does not aim to replicate or depend on those specific results. Rather, its primary contribution lies in the development of a flexible and generalizable methodology for the conceptual design of seed treatment prototypes, which can be tailored and validated using empirical data relevant to diverse agricultural contexts.
- Optimal UV-C exposure time: from 0 to 2 min
- Optimal Spirulina concentration: 1.5 g/100 mL
- Predicted germination rate at 24 h: 87.11%
- Predicted germination rate at 48 h: 93.73%
The results used in this paper indicate that avoiding UV-C exposure while applying a higher Spirulina concentration leads to the highest germination rates. This suggests that Spirulina supplementation positively influences germination, whereas UV-C exposure does not provide significant benefits under the tested conditions (See Figure 2 and Figure 3). Further studies are recommended to explore the potential interactions between these treatments and refine the optimization strategy for seed priming applications.
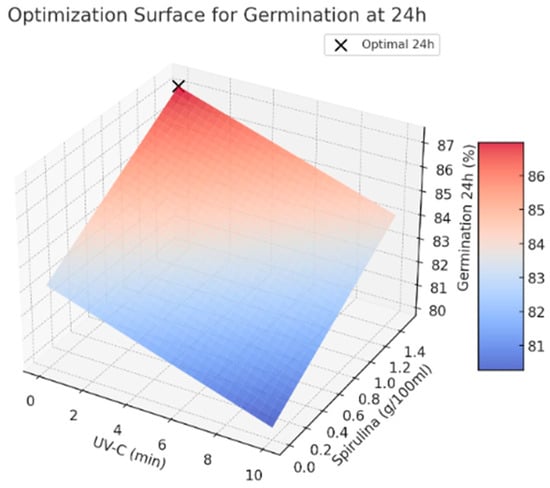
Figure 2.
Optimal values for germination at 24 h.
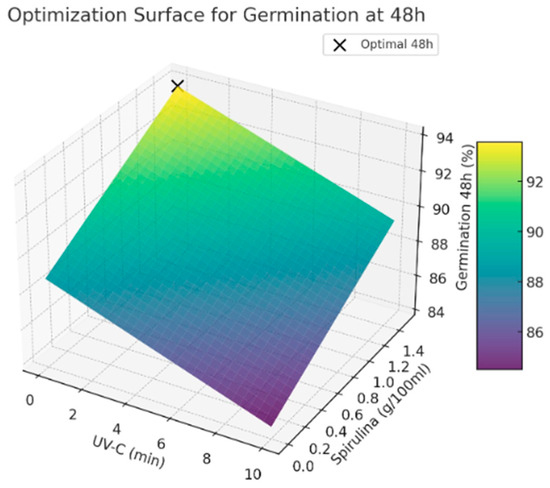
Figure 3.
Optimal values for germination at 48 h.
2.1.2. Principal Component Analysis (PCA)
Principal Component Analysis (PCA) was conducted to determine the contribution of various germination-related factors to the observed variance. The PCA component loadings provide insight into the influence of each variable on Principal Components 1 (PC1) and 2 (PC2), revealing significant trends in the data.
PC1, which accounts for approximately 33.3% of the variance, is the primary driver in distinguishing variations in germination outcomes. This component has a strong negative correlation with fresh weight (−0.71), indicating that higher fresh weight values tend to be associated with lower PC1 scores. Additionally, PC1 exhibits a moderate positive correlation with dry weight (0.49) and germination at 48 h (0.49), suggesting that increased dry weight and long-term germination are key contributors to this principal component. PC2, which explains approximately 27.4% of the variance, primarily captures differences in early-stage germination and secondary metabolite accumulation. It exhibits a strong positive correlation with germination at 24 h (r = 0.61), indicating that samples with high early germination rates contribute significantly to PC2. Conversely, flavonoid content exhibits a strong negative correlation (−0.64) with PC2, meaning higher flavonoid levels correspond to lower PC2 scores. Additionally, germination at 48 h has a moderate negative correlation (−0.32) with PC2, suggesting that rapid initial germination may slightly reduce later germination success. Table 2 illustrates the components PC1 and PC2 during different time frames. Table 3 illustrates the dry weight and flavonoid content.

Table 2.
Germination during different time frames.

Table 3.
Dry weight and flavonoid content.
2.1.3. Heatmap of PCA
A heatmap of PCA was generated to visually represent how each original variable contributes to PC1 and PC2 (See Figure 4).
- Red areas indicate a strong positive contribution, highlighting variables that significantly drive variance in the data.
- Blue areas indicate a strong negative contribution, showing variables that inversely affect the component scores.
- Neutral (white) areas show weaker influence, meaning these variables contribute minimally to the principal components.
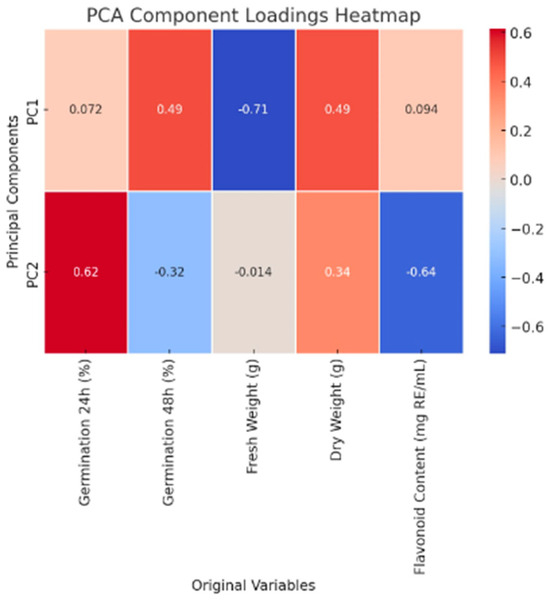
Figure 4.
Heat map of PCA original variables used.
Figure 4.
Heat map of PCA original variables used.
This heatmap helps identify the dataset’s most influential variables in driving variance and clarifies how germination factors interact.
For Principal Component 1 (PC1), strong negative correlations are observed with fresh weight (−0.71), indicating that PC1 is associated with a reduction in fresh weight. Moderate positive correlations with germination at 48 h (0.49) and dry weight (0.49) suggest that PC1 is influenced by long-term germination success and biomass accumulation. Germination at 24 h and flavonoid content have a minimal influence on PC1. As a result, PC1 can be interpreted as a seedling biomass and late; higher values in PC1 correspond to increased dry weight and delayed germination but lower fresh weight. For Principal Component 2 (PC2), a strong positive correlation with Germination at 24 h (0.62) suggests that early germination plays a major role in defining PC2. A strong negative correlation with flavonoid content (−0.64) indicates that higher flavonoid levels are associated with lower PC2 scores. Additionally, a moderate negative correlation with germination at 48 h (−0.32) suggests that early germination is inversely related to later germination success. PC2 has minimal influence from fresh weight. Therefore, PC2 can be interpreted as a rapid germination versus flavonoid trade-off factor; higher values in PC2 indicate faster germination at 24 h but lower flavonoid accumulation.
2.1.4. Strategic Recommendations Based on PCA Findings Are Shown Below
- If the goal is fast germination within 24 h, conditions that favor PC2 should be increased, meaning minimal UV-C exposure and higher Spirulina concentration.
- Optimizing PC1 is ideal if the goal is high seedling biomass (fresh and dry weight). This requires balancing UV-C exposure and Spirulina levels.
- If a higher flavonoid content is a priority, it must be noted that it may trade off against rapid germination rates, as suggested by the negative correlation between flavonoid content and PC2.
These findings could provide a framework for optimizing seed treatment strategies, enabling tailored approaches that meet specific agricultural objectives.
2.2. Functional Prototype for Optimizing Germination and Biomass in Lentil Seeds
Based on the PCA optimization results, a functional prototype can be designed to dynamically adjust UV-C exposure and Spirulina levels, achieving specific germination and biomass goals. Based on the previous findings, a functional prototype is proposed to dynamically optimize UV-C exposure and Spirulina application dynamically, thereby enhancing seed germination and biomass accumulation. This prototype features a programmable UV-C LED system that enables precise control over exposure time and intensity, ensuring optimal UV-C levels for enhanced germination. An automated peristaltic pump system accurately dispenses Spirulina solution to the seeds, maintaining consistency in nutrient delivery. A climate-controlled chamber is integrated into the system to regulate humidity, temperature, and moisture levels, optimizing the environmental conditions necessary for germination. A monitoring system equipped with time-lapse imaging and real-time moisture sensors continuously tracks the germination process, providing data for further optimization. The system also incorporates an AI-driven algorithm that dynamically adjusts treatment parameters based on real-time feedback, allowing users to set specific germination objectives. The prototype offers various operational modes tailored to specific needs, including a Fast Germination mode for rapid seedling emergence, a High Biomass mode for increased fresh and dry weight, and a Flavonoid Enrichment mode that optimizes conditions for secondary metabolite accumulation. A user-friendly interface enables researchers and farmers to customize and monitor seed treatments remotely. At the same time, cloud-based data logging supports long-term trend analysis and optimization of seed treatment protocols.
2.3. Functional Prototype Conceptual Design
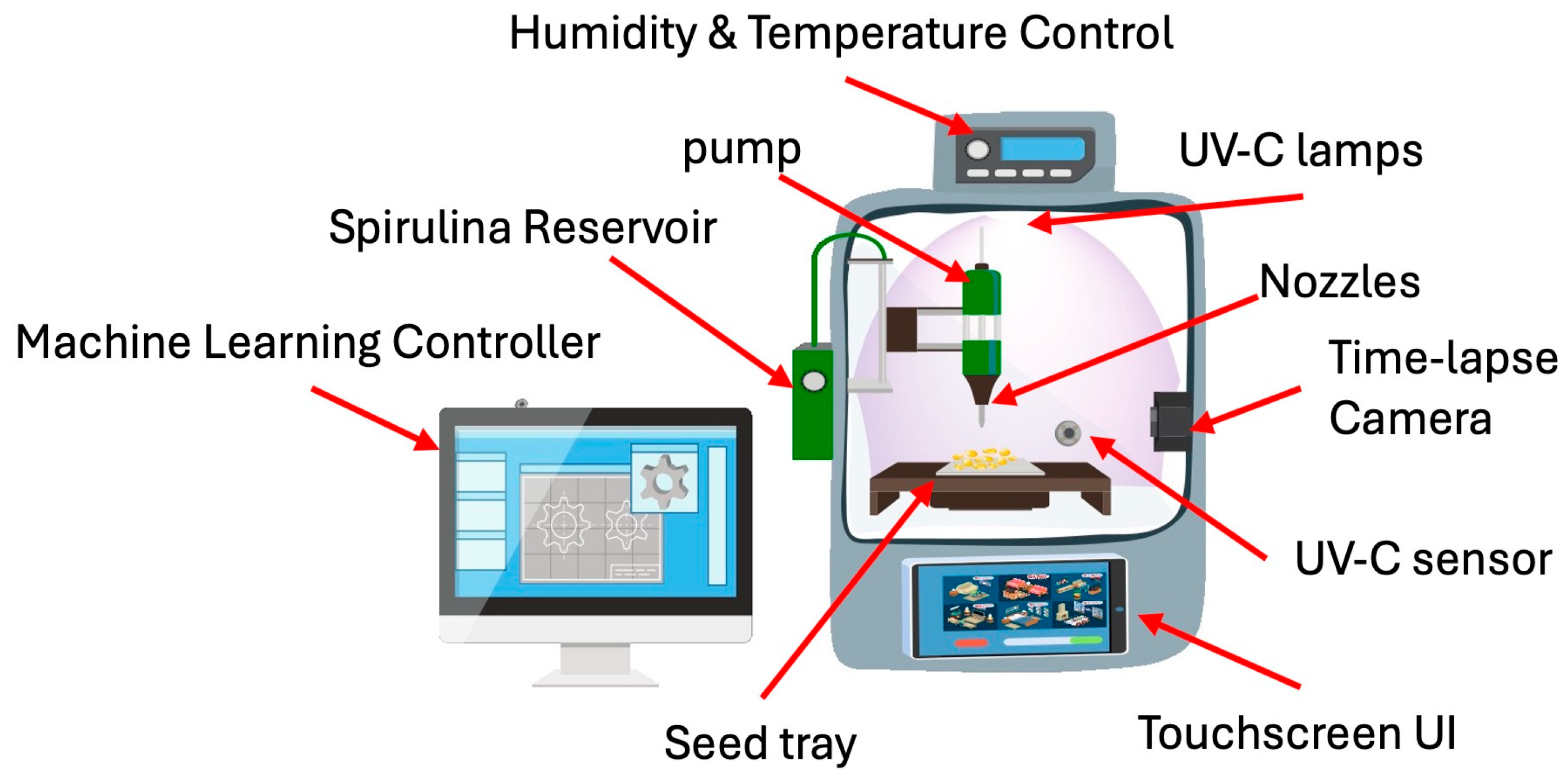
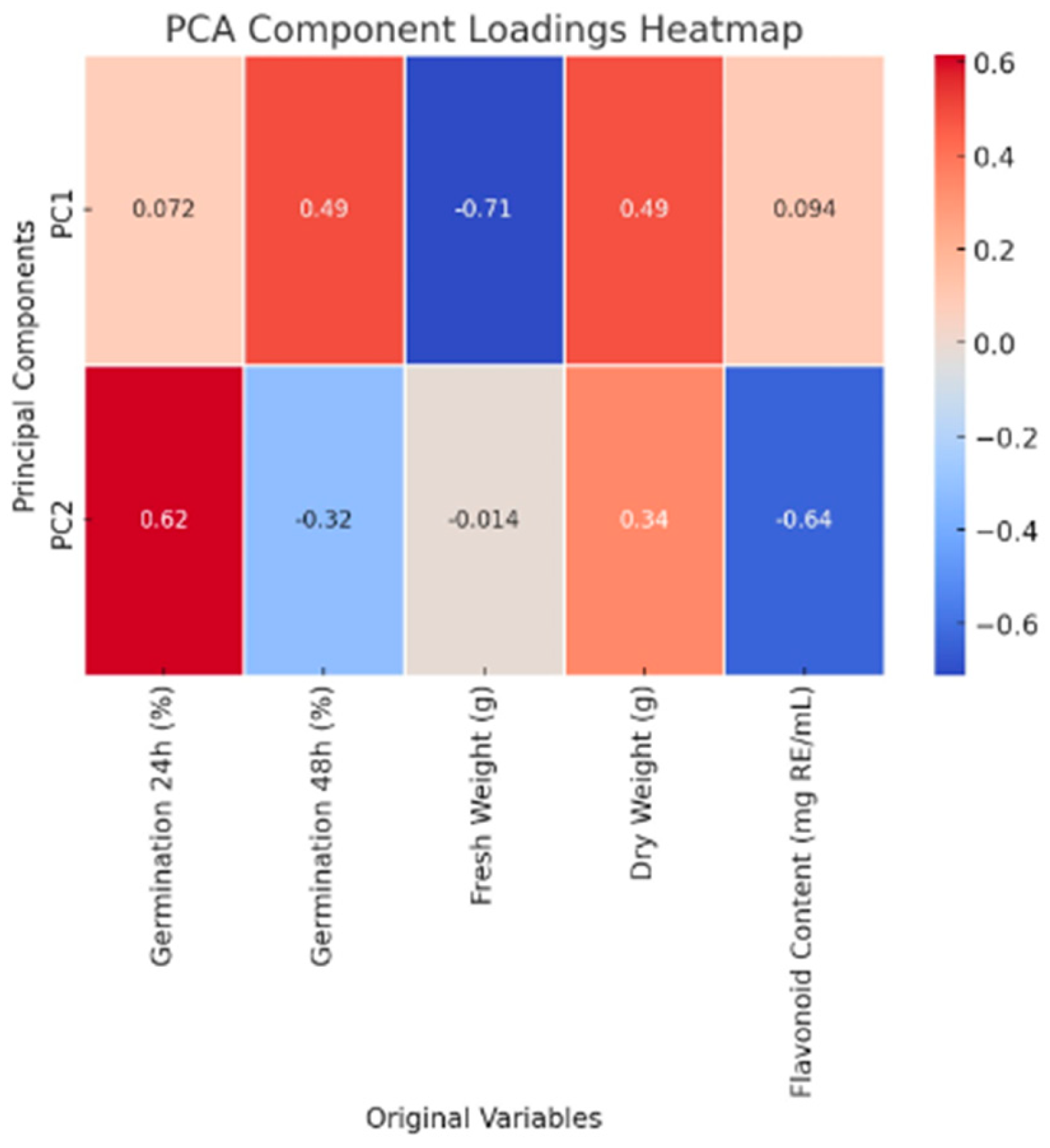
This proposed prototype is an automated seed treatment chamber that optimizes UV-C exposure, Spirulina concentration, and environmental factors to enhance germination, biomass, and flavonoid content. Thus, the developed prototype is an automated seed treatment chamber designed to optimize UV-C exposure, Spirulina concentration, and environmental conditions, thereby enhancing germination rates, biomass accumulation, and flavonoid content. The system incorporates several key functional modules. The UV-C radiation control system includes adjustable UV-C LED lamps with an exposure range of 0–10 min, an intensity control mechanism, a motorized shielding system to regulate exposure, and real-time UV-C sensors to ensure precise dosage application. An automated scheduler is integrated to enable timed UV-C treatment cycles. The Spirulina nutrient delivery system is controlled by a microcontroller, which accurately mixes different Spirulina concentrations within the range of 0–1.5 g/100 mL. A precision pump system delivers the Spirulina-infused liquid to the seeds, while a reservoir equipped with a stirring mechanism ensures uniform distribution of the Spirulina solution. The germination and environmental chamber feature a humidity and temperature control system, a moisture sensor to monitor water absorption, and a time-lapse camera to track germination progress, further optimizing germination conditions. An advanced data monitoring and optimization algorithm is implemented, integrating a machine learning-based controller that dynamically adjusts UV-C and Spirulina levels in response to the seed batch. The system features a user interface accessible via touchscreen or mobile application, enabling users to select specific germination goals, including Fast Germination mode for a 24 h cycle, High Biomass mode for maximizing seedling growth, and flavonoid enrichment mode for promoting enhanced secondary metabolite accumulation. Additionally, cloud-based data logging enables the long-term tracking of germination performance, allowing users to analyze historical data and refine treatment protocols further. Figure 5 illustrates the general schematic of the proposed prototype, highlighting the main blocks in the conceptual design. Figure 6 describes the prototype’s operational functionality.
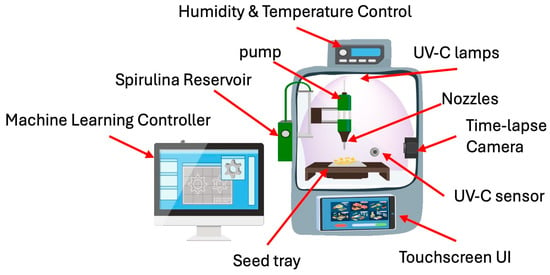
Figure 5.
General description of the prototype.
Figure 6.
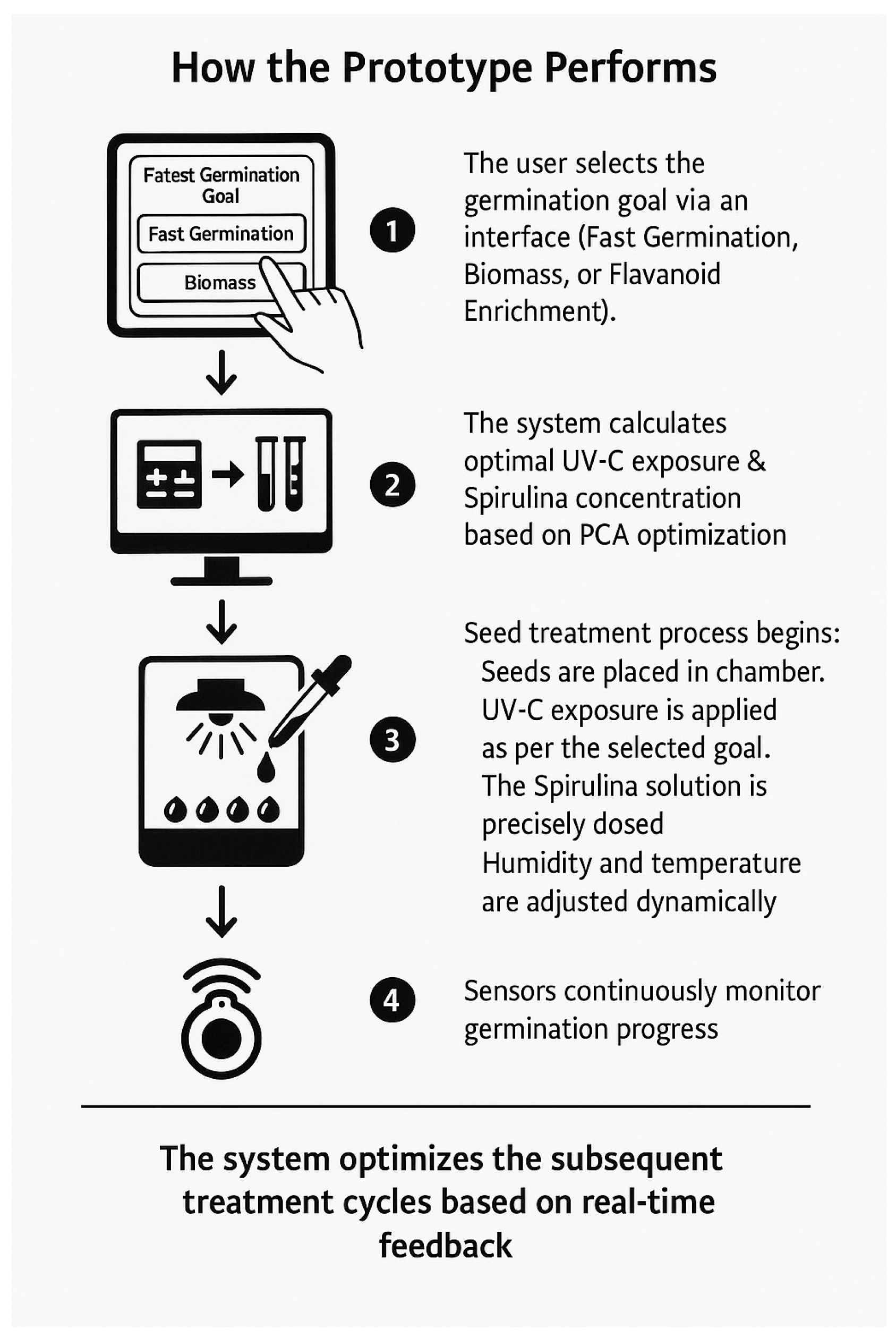
How the prototype performs.
- The prototype comprises four tightly integrated functional modules, each working together to deliver precise, adaptive seed treatments. First, the UV-C Radiation Control System combines adjustable LED arrays, a motorized shutter, and real-time sensing to administer ultraviolet doses with minute-scale timing and intensity adjustments. An onboard scheduler automates treatment cycles, while continuous feedback from the UV-C sensor ensures each batch receives the exact programmed dosage. Second, the Spirulina Nutrient Delivery System uses a microcontroller-driven dispenser and precision peristaltic pump to mix and meter Spirulina concentrations ranging from 0 to 1.5 g per 100 mL. A stirred reservoir maintains the culture in a uniformly suspended state, ensuring consistent nutrient availability throughout each treatment cycle. Third, the Germination and Environmental Chamber maintains optimal growth conditions by regulating humidity and temperature around the seed bed and by monitoring substrate moisture with dedicated sensors. A time-lapse camera documents germination progress at set intervals, supplying image data for downstream analysis.
- The Data Monitoring and Optimization Algorithm ties the system together. A machine-learning controller ingests sensor readings and germination metrics, dynamically adjusting UV-C exposure and Spirulina dosing to meet the user’s selected goal—whether that be rapid emergence, maximal biomass, or flavonoid enrichment. Users choose one of these modes via a touchscreen (or companion app), and all treatment parameters, environmental setpoints, and performance logs are recorded in the cloud for long-term analysis and model retraining.
- When a run begins, the user loads seeds into the chamber and selects one of the following options: Fast Germination, High Biomass, or Flavonoid Enrichment. The system then computes an initial UV-C and Spirulina profile, based on PCA-derived optimals, executes the first treatment, and continuously monitors the response. As each cycle completes, the algorithm refines its parameters, ensuring that every subsequent exposure and nutrient delivery is tuned to the seeds’ actual behavior, achieving truly adaptive, precision seed priming. The workflow for Figure 6 is presented below.
- Define Treatment Objectives: At the outset, the operator interacts with the control interface to select one of three predefined operational modes: Fast Germination, High Biomass, or Flavonoid Enrichment. Fast Germination prioritizes rapid radicle emergence by applying shorter, pulsed UV-C exposures coupled with moderate Spirulina dosages; High Biomass emphasizes vigorous shoot and root development through extended Spirulina supplementation and optimized UV-C timing; Flavonoid Enrichment focuses on maximizing secondary metabolite synthesis by increasing UV-C duration and modulating Spirulina concentration within a narrow range. The chosen mode configures downstream system behavior to align with specific agronomic goals.
- Initialize System Parameters: Upon mode selection, the system retrieves a set of baseline parameters from its configuration database. These include UV-C intensity and exposure duration, Spirulina solution concentration, ambient temperature setpoints, and relative humidity targets. For example, Flavonoid Enrichment defaults to 8 min of UV-C at 1 mW/cm2 and 0.3 g/100 mL Spirulina, with temperature at 25 °C and relative humidity at 60%. These defaults ensure immediate readiness and provide starting points for subsequent AI-driven adjustments.
- Environmental and Seed Monitoring: A network of embedded sensors continually measures UV intensity, chamber temperature, relative humidity, and substrate moisture. Concurrently, a high-resolution time-lapse camera captures the germination progress at configurable intervals (e.g., every 15 min). Sensor data streams and image sequences are fed into the data logging module, where they are timestamped and stored for real-time processing and retrospective analysis.
- Automated Treatment Delivery: The system controls a UV-C LED array and precision motorized shutter to deliver radiation doses according to the initialized schedule. In parallel, a peristaltic pump administers the Spirulina solution, mixing the stock culture with deionized water in real-time to maintain the target concentration. Actuators adjust flow rates with 0.01 mL accuracy, ensuring uniform distribution over the seed bed.
- Data Acquisition and Processing: Real-time sensor readings and image-derived germination metrics enter the AI preprocessing pipeline. We apply Analysis of Variance (ANOVA) to detect statistically significant effects of treatment variables and Principal Component Analysis (PCA) to reduce dimensionality and highlight dominant response patterns. This preprocessing stage transforms raw data into feature vectors suitable for machine learning inference.
- Machine Learning–Driven Adjustment: A regression-based AI model uses preprocessed features to predict germination rate, biomass accumulation, or flavonoid concentration, depending on the active mode. The controller then updates UV-C exposure time and Spirulina dosage parameters for the next iteration via closed-loop feedback. Adjustments occur at every treatment cycle (e.g., hourly), converging toward optimal conditions that align with the user’s objective.
- Iteration to Completion: Steps 3 through 6 repeat automatically until the total treatment duration elapses, as defined by the selected operational mode. Throughout the run, the AI controller refines its predictions, minimizing variance in outcome metrics and ensuring consistent optimization. Final datasets, including time-series sensor logs and germination images, are archived for validation and future model retraining.
Table 4 shows the optimization use case for achieving specific goals. The use case can be adapted to meet the end user’s particular needs.

Table 4.
Optimization use case based on goal.
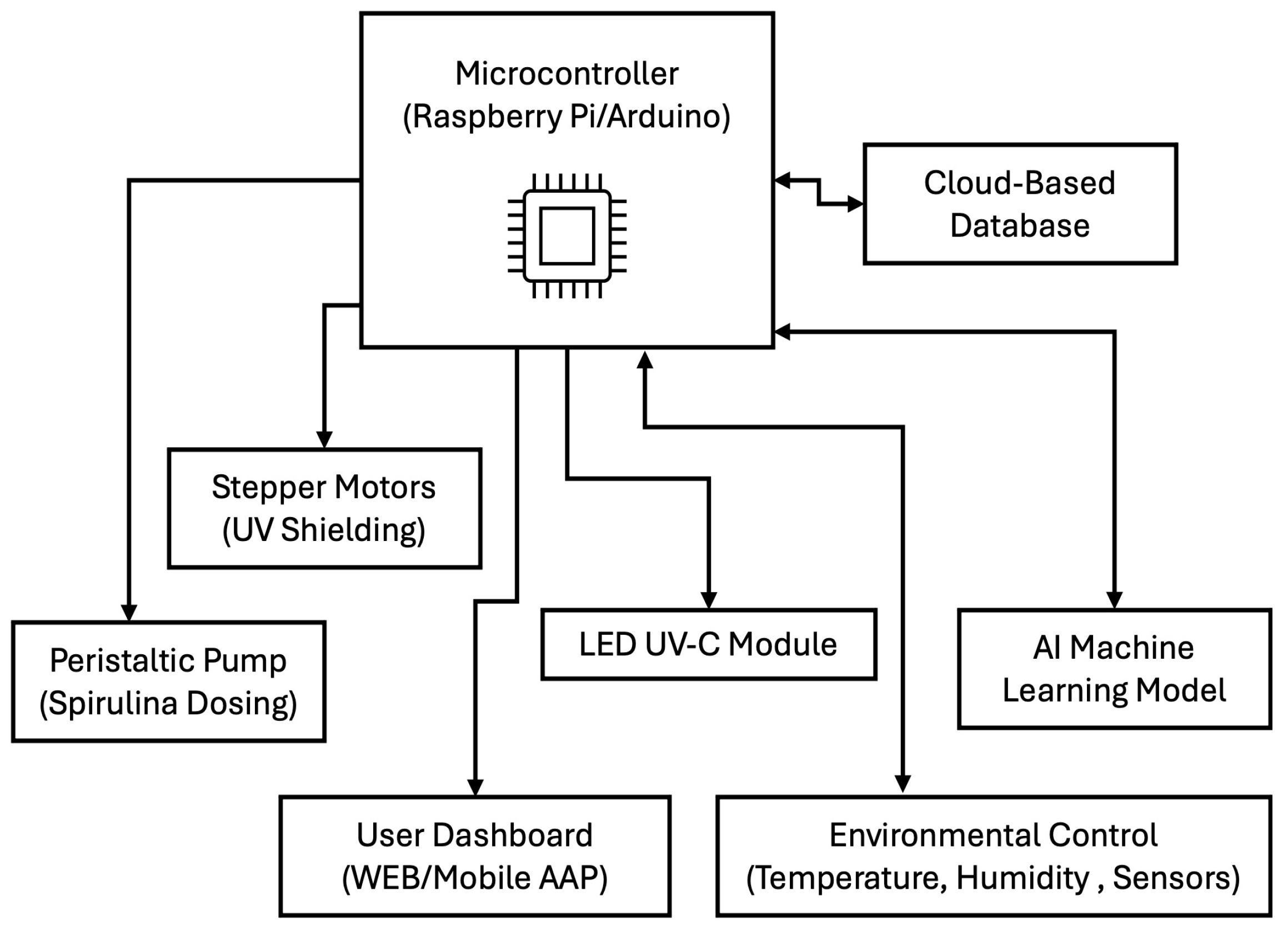
The prototype’s hardware includes a microcontroller, such as a Raspberry Pi or Arduino, which serves as the central control unit, managing sensor inputs and actuator responses. Stepper motors are employed for automated UV shielding, allowing precise control of UV-C exposure to minimize any negative impact on germination. A peristaltic pump system is integrated to precisely dose Spirulina, ensuring consistent nutrient delivery to the seeds. An LED UV-C module is utilized for controlled radiation exposure, providing a reliable mechanism for influencing seed germination while maintaining safety standards. The software and AI optimization components are crucial in refining treatment protocols and improving efficiency. A machine learning model is incorporated to analyze germination patterns and adjust treatment conditions in real-time based on data. A cloud-based database stores historical optimization data, allowing for continuous improvements in treatment strategies. A user-friendly dashboard, accessible via a web or mobile application, enables remote monitoring and control of the system, providing users with real-time insights and the ability to modify treatment settings as needed (See Figure 7).
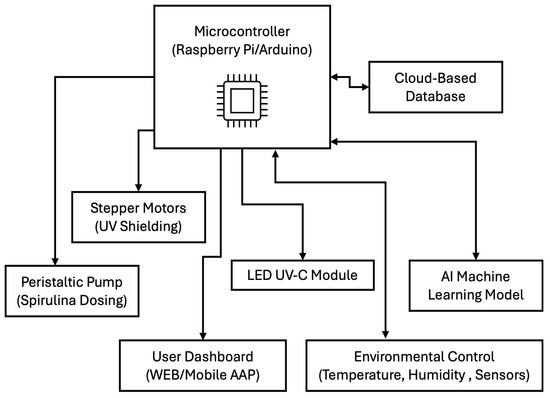
Figure 7.
Main blocks in the conceptual prototype.
The proposed prototype can automate precision seed treatment by integrating real-time control mechanisms for UV-C exposure, Spirulina concentration, and environmental conditions. By optimizing germination conditions for specific outcomes, the system enhances seed viability, accelerates early-stage development, and improves overall biomass production. The AI-driven optimization reduces the need for manual intervention, ensuring consistent and replicable seed treatments while minimizing human error. Additionally, the prototype offers scalability for agricultural and research applications, making it a versatile tool for commercial seed production, laboratory studies, and controlled environment agriculture. The cloud-based monitoring system enables long-term performance tracking, facilitating data-driven improvements and adaptive treatment strategies. Ultimately, this innovation enhances seed treatment efficiency, contributing to advancements in precision agriculture and sustainable crop production.
2.4. Morphological Matrix for Functional Prototype Design
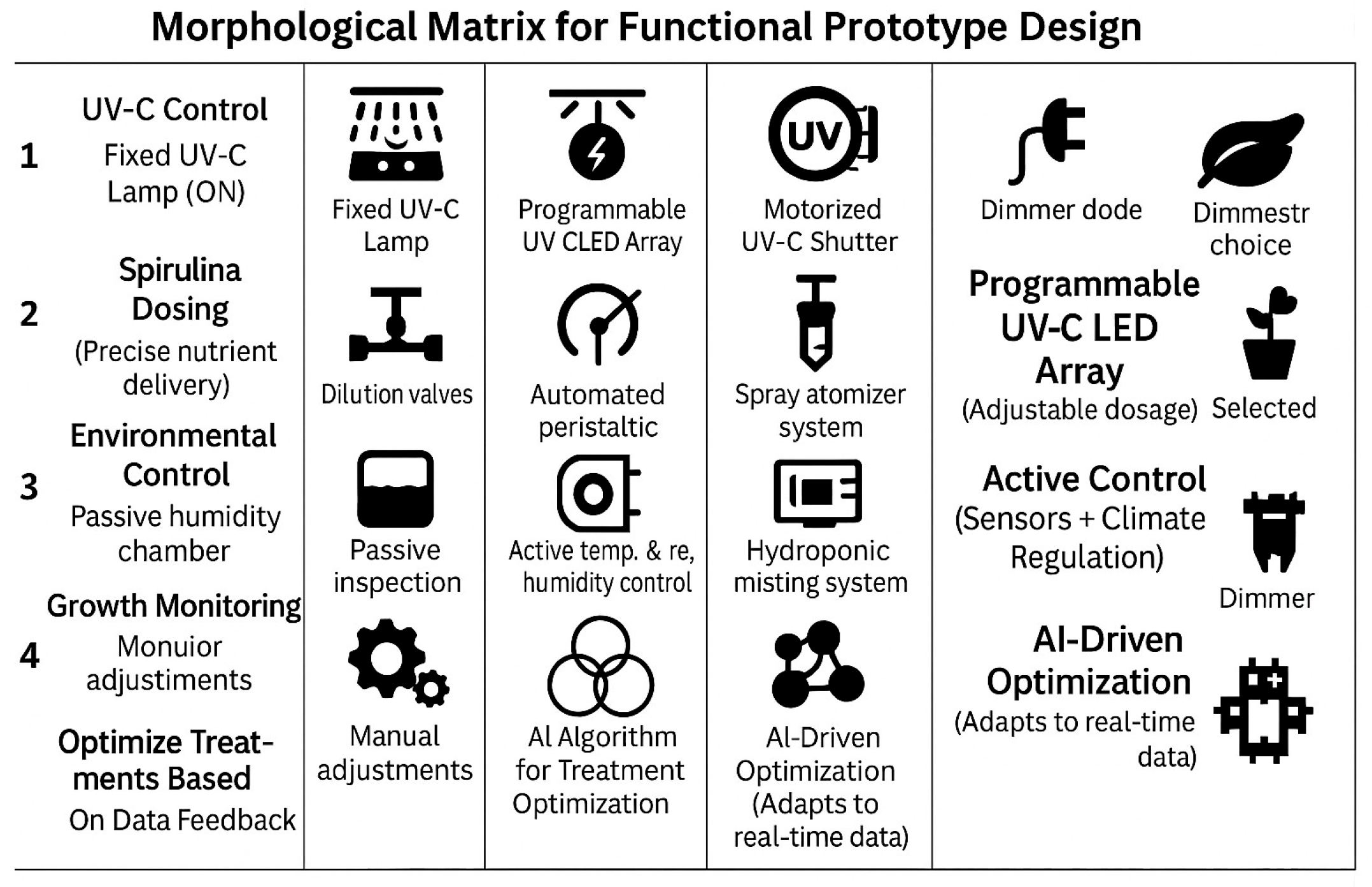
A morphological matrix [24,25] was also used to define a functional prototype for optimizing lentil seed germination. This method helps systematically explore possible design solutions for each critical function. In Figure 8, a functional prototype design is shown. Table 5 illustrates several solutions.
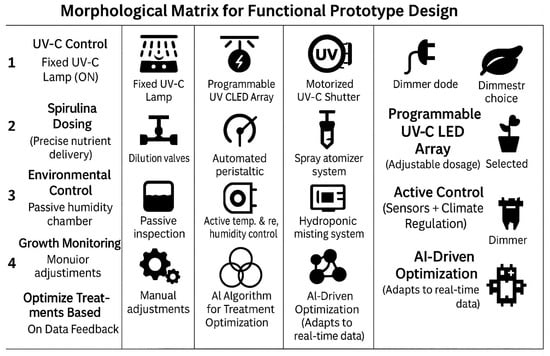
Figure 8.
Morphological matrix for the functional prototype.

Table 5.
Presents different possible solutions for each function.
2.4.1. Functional Requirements
The prototype must
- Control UV-C Exposure (timing and intensity);
- Regulate Spirulina Concentration (precise nutrient delivery);
- Maintain Optimal Germination Conditions (humidity, temperature, moisture);
- Monitor Seed Growth and Treatment Response (real-time data collection);
- Optimize Treatments Based on Data Feedback (adaptive control system).
2.4.2. Prototype Description Based on Selected Solutions
The UV-C treatment module consists of a programmable UV-C LED array that allows for adjustable wavelength and intensity settings, providing precise control over radiation exposure. To further enhance accuracy, a motorized shielding system regulates exposure time, ensuring that seeds receive the appropriate dose of UV-C treatment without overexposure. The Spirulina delivery system is designed with an automated peristaltic pump that ensures precise dosing of Spirulina concentrations. This system includes a mixing reservoir equipped with an integrated stirring mechanism, which maintains uniformity in the Spirulina solution before it is delivered to the seeds.
The environmental control chamber integrates temperature and humidity sensors with an active climate control mechanism. This ensures that optimal environmental conditions are maintained for seed germination. Additionally, the chamber is equipped with an automated misting and hydration system that provides consistent moisture levels, further supporting seed viability and uniform growth.
The growth monitoring and feedback system includes a time-lapse camera that continuously tracks the germination process, while moisture sensors monitor the water absorption levels of the seeds. This system is complemented by an AI algorithm that processes real-time data, optimizing future treatment cycles based on seed response patterns. The AI-based optimization system employs a machine learning model to analyze germination patterns and adjust treatment parameters dynamically. Users can interact with the system through an intuitive interface available via touchscreen, web, or mobile application. The system offers three operational modes: Fast Germination Mode, which prioritizes early sprouting; High Biomass Mode, which maximizes fresh and dry weight accumulation; and Flavonoid Enrichment Mode, which enhances the production of secondary metabolites. Additionally, cloud-based monitoring and optimization enable long-term tracking, allowing users to refine treatment strategies over time based on historical data and performance trends.
The proposed Smart Germination Chamber automates UV-C and Spirulina treatments to optimize lentil seed germination dynamically based on real-time monitoring and AI-driven feedback. The main tasks are the following:
- Optimized UV-C dosage
- Precision Spirulina nutrient delivery
- Smart climate control
- Data-driven AI adaptation
2.5. Design AI-Driven Germination Optimization
The proposed system continuously collects real-time temperature, humidity, moisture, and germination rate data. While these values could be simulated in the model for testing, they would be obtained from actual sensors in a real-world deployment. The AI model processes this information using a trained Linear Regression algorithm [26], which predicts germination success based on UV-C exposure, Spirulina concentration, temperature, and humidity.
The optimization process operates through three distinct treatment modes. The Fast Germination mode prioritizes conditions that enhance early sprouting by maintaining high humidity, warm temperatures, and increased Spirulina concentration. The High Biomass mode balances UV-C exposure and Spirulina dosage to maximize overall seedling growth, ensuring optimal biomass accumulation. The Flavonoid Enrichment mode increases UV-C exposure while reducing Spirulina levels to stimulate flavonoid accumulation, enhancing the nutritional profile of the seedlings.
As part of its adaptive learning mechanism, the AI continuously adjusts UV-C exposure time, Spirulina concentration, temperature, and humidity based on real-time sensor feedback. The predicted germination rates are refined over successive treatment cycles, improving the system’s accuracy and effectiveness over time. The machine learning model is updated dynamically after collecting sufficient real-world observations, enabling ongoing optimization of seed treatment strategies and ensuring continuous performance improvement.
This intelligent automation process offers several advantages. AI-driven optimization dynamically controls the core treatment parameters, reducing the need for manual intervention and improving seed germination efficiency. The real-time feedback mechanism enables the system to make predictions based on actual germination patterns, ensuring precision in seed treatment. Continuous refinement of the AI model results in progressively improved germination success, making the system more effective with each iteration. Additionally, the scalable nature of the model enables its application across various seed types and agricultural treatments, thereby expanding its usability. The algorithm can be integrated into a cloud-based monitoring dashboard for extended functionality, providing real-time supervision and enabling advanced data analytics for further AI-driven enhancement. The MATLAB (version 2024) pseudocode is presented in Appendix A. This pseudocode has been implemented and empirically tested, first on the synthetic dataset described. Subsequently, it can be used in preliminary laboratory trials as future work. The empirical test confirmed that the code executes correctly, reproduces the intended regression-based optimization workflow, and yields consistent germination-rate predictions in line with the conceptual design.
2.6. Data Set for Proof of Concept
A dataset was generated using synthetic data to prove the concept of the proposal and evaluate and validate the performance of the AI system. The dataset used in this study was synthetically generated to serve as a proof of concept for evaluating the performance of an AI-based optimization model designed to improve lentil seed germination. This synthetic dataset was not derived from real laboratory or field measurements but was designed to emulate biologically plausible dose–response relationships based on prior experimental findings, particularly those reported in reference [8]. Accordingly, the dataset incorporates some of the conditions and results observed in the experiments presented in [8] to make it more representative of real-world scenarios. However, the dataset is not intended to replicate or duplicate the data from [8], as the primary goal of this paper is to develop a flexible framework that can be adapted for different seed types. The dataset includes both independent and dependent variables. The independent variables consist of UV-C exposure time (measured in minutes, ranging from 0 to 10), Spirulina concentration (measured in grams per 100 milliliters, ranging from 0 to 2.0), temperature (measured in degrees Celsius, ranging from 18 °C to 26 °C), and humidity (measured as a percentage, ranging from 50% to 80%). The dependent variable is the germination rate, expressed as a percentage. Several biological assumptions guided the generation of the data. It was assumed that excessive UV-C exposure negatively impacts germination due to cellular damage, while Spirulina supplementation, particularly at higher concentrations, enhances seed metabolism and promotes germination. The temperature range of 22 °C to 24 °C was considered optimal for germination, while temperatures outside this range introduced variability due to thermal stress. Higher humidity levels, particularly above 65%, were associated with improved germination rates, as sufficient moisture is essential for activating seed metabolism. The dataset also incorporates interaction effects, such as Spirulina mitigating the adverse effects of UV-C exposure, to better reflect the realistic responses of plants. The synthetic dataset consists of 100 samples to ensure sufficient variability for machine learning applications. Random noise could be added to prevent overfitting and improve the model’s generalization capacity. This dataset was used to train a linear regression model, chosen for its simplicity, low computational requirements, and high interpretability, qualities that make it well-suited for embedded systems and real-time feedback applications.
For example, one entry in the dataset represents a scenario where 1 min of UV-C exposure, combined with a Spirulina concentration of 1.5 g/100 mL, a temperature of 23 °C, and a humidity level of 70%, yields a predicted germination rate of 93.5%. This kind of structured and biologically grounded dataset enables the AI model to dynamically adjust treatment strategies based on user-defined objectives and real-time sensor feedback. Therefore, the synthetic dataset serves as a scientifically informed and robust tool for validating the conceptual framework and AI methodology proposed in this study.
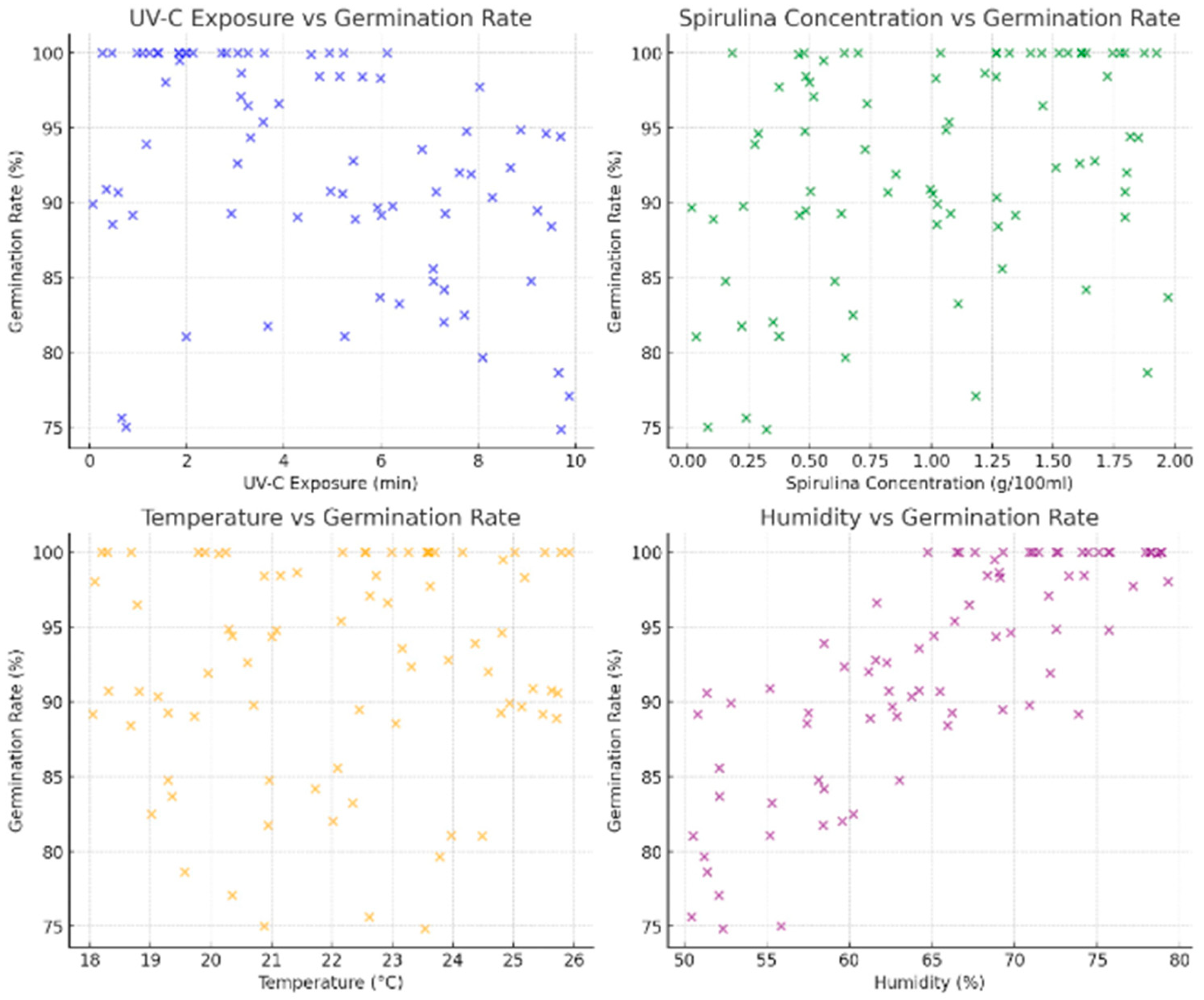
The relationship between UV-C exposure and germination rate suggests that increasing UV-C exposure beyond five minutes leads to a decline in germination rates. This trend aligns with known biological effects, where excessive UV-C radiation damages cellular structures, hindering seed viability. However, some seeds maintain high germination rates despite increased UV-C exposure, likely due to interaction effects with Spirulina supplementation or environmental factors. Including this variability ensures the dataset captures realistic fluctuations in seed responses, making it valid for AI learning.
The correlation between Spirulina concentration and germination rate is evident, particularly at higher concentrations between 1.0 and 2.0 g/100 mL. This aligns with experimental findings that nutrient-rich Spirulina enhances seed metabolism, improving germination efficiency. The dataset accurately represents this effect, ensuring the AI model learns the impact of Spirulina supplementation on seed growth. The variation in Spirulina concentration levels enhances the robustness of the dataset, allowing the model to generalize effectively.
Temperature variations and their effects on seed germination are also well-represented. The optimal temperature range of 22–24 °C exhibits the highest germination rates, while lower temperatures of 18–20 °C and higher temperatures of 25–26 °C introduce variability, suggesting temperature stress. This dataset reflects real-world conditions, making it valid for AI training. The model can be generalized across different environmental conditions, making it adaptable for various agricultural applications.
Humidity levels have a positive influence on germination rates, with values above 65% correlating with germination rates exceeding 90%. This trend aligns with established knowledge that sufficient moisture is essential for seed activation and metabolic processes. The dataset captures varying humidity levels, allowing the AI model to learn how moisture availability affects germination under different environmental conditions.
The dataset can be used for AI training since it incorporates biologically relevant variables affecting germination, ensuring AI predictions are grounded in the real-world plant. The trends observed in the dataset align with known experimental results, increasing the credibility of the data. It encompasses a broad range of UV-C exposure times, from 0 to 10 min, Spirulina concentrations from 0 to 2 g/100 mL, temperatures from 18 to 26 °C, and humidity levels from 50% to 80%. Both optimal and extreme conditions enable the AI model to generalize effectively across various scenarios.
The dataset used is entirely synthetic and was generated solely for conceptual illustration of the proposed AI-driven seed-priming methodology. It does not derive from actual laboratory or field measurements but was designed to emulate biologically plausible dose–response relationships. Future work will involve collecting and incorporating empirical germination data to validate and refine the model further.
The dataset captures interaction effects, such as the mitigation of UV-C stress by Spirulina supplementation. This interaction-based variability enhances AI learning, enabling it to identify complex relationships within the data. The dataset contains 100 samples, ensuring adequate variability for training machine learning models (regression). Including random noise in the data prevents overfitting, making the model robust.
The dataset enables AI-driven decision-making in agriculture and seed priming applications. The model can dynamically adjust treatment strategies for different environmental conditions, making it highly scalable. The generated dataset is scientifically valid, diverse, and well-structured, making it an ideal resource for training and validating AI models in precision agriculture. The data effectively captures the relationship between key germination factors and seed response, allowing the AI system to optimize seed treatment conditions dynamically. This ensures the model is accurate and applicable to real-world agricultural and research applications. Figure 9 shows the plots generated from the database.
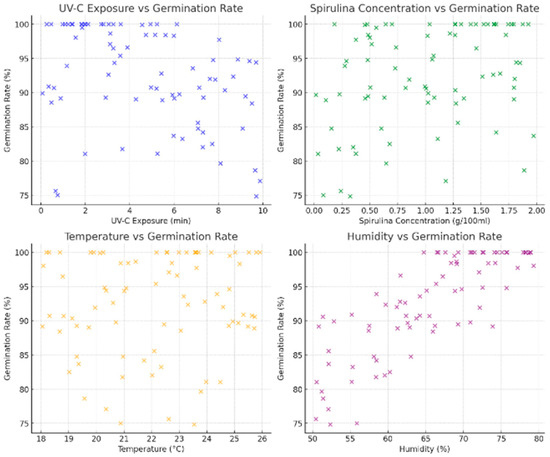
Figure 9.
A synthetic dataset was generated for training and validating the AI methodology.
Table 6 compares four regression techniques, Linear Regression, Support Vector Regression (SVR), Artificial Neural Networks (ANN), and Gaussian Process Regression (GPR), in the context of real-time, AI-driven seed treatment control. Linear Regression, the chosen method, offers transparent and easily interpretable coefficients, as well as minimal computational overhead, making it ideal for embedded hardware and rapid feedback loops. However, it cannot intrinsically model non-linear interactions. SVR extends its capability to capture non-linear patterns and handles outliers robustly via ε-insensitive loss; however, its kernel computations incur higher latency and reduce interpretability, rendering it less suitable for on-device, real-time inference. ANN excels at learning complex non-linear relationships and supports incremental (online) learning; however, its black-box nature and significant data and computational demands conflict with the prototype’s need for explainability and resource efficiency. GPR offers probabilistic predictions and inherent uncertainty estimation, achieving strong performance on small datasets. However, its scaling and intricate kernel tuning make it impractical for continuous, on-device operation. By balancing speed, simplicity, and interpretability against the operational requirements of a modular seed-treatment chamber, Linear Regression emerges as one of the most pragmatic baselines for AI-driven optimization, providing reliable performance within the observed linear dose–response ranges while leaving room for future exploration of more complex models as computational resources permit.

Table 6.
Conceptual comparison of AI regression methods.
The training process of the linear regression model in the study follows a two-phase approach. First, an initial model is trained using a synthetic dataset that contains samples simulating realistic relationships between UV-C exposure, Spirulina concentration, temperature, humidity, and germination rate. Using ordinary least squares, the model calculates coefficients that define how each input affects germination, yielding a linear equation for prediction. Once deployed, the prototype could enter a real-time optimization loop. In each cycle, it reads sensor data and applies treatment parameters based on the selected goal (e.g., Fast Germination). The model predicts the expected germination rate, and the actual outcome is measured and stored. When more than 10 new observations are collected, the model is retrained using the updated dataset to improve accuracy and adapt to real-world variability. This method ensures the model remains accurate over time while maintaining its lightweight, interpretable nature and suitability for real-time. Beyond ordinary least squares, the model can be trained with support vector regression for better handling of non-linear dose–response curves.
As a result, to predict the germination rate for given conditions, use the values of UV-C exposure (minutes), Spirulina concentration (g/100 mL), temperature (°C), and humidity (%) to form Equation (1) representing the trained linear regression model.
Model Parameters:
- Intercept (24.36): The baseline germination rate when all predictor variables are zero.
- UV-C Exposure (−0.44): Each additional minute of UV-C exposure reduces the germination rate by 0.44%.
- Spirulina Concentration (4.20): Each gram per 100 mL of Spirulina increases the germination rate by 4.20%.
- Temperature (0.87): Each degree Celsius increase in temperature improves germination by 0.87%.
- Humidity (0.71): Each 1% increase in humidity increases germination rate by 0.71%.
3. Results
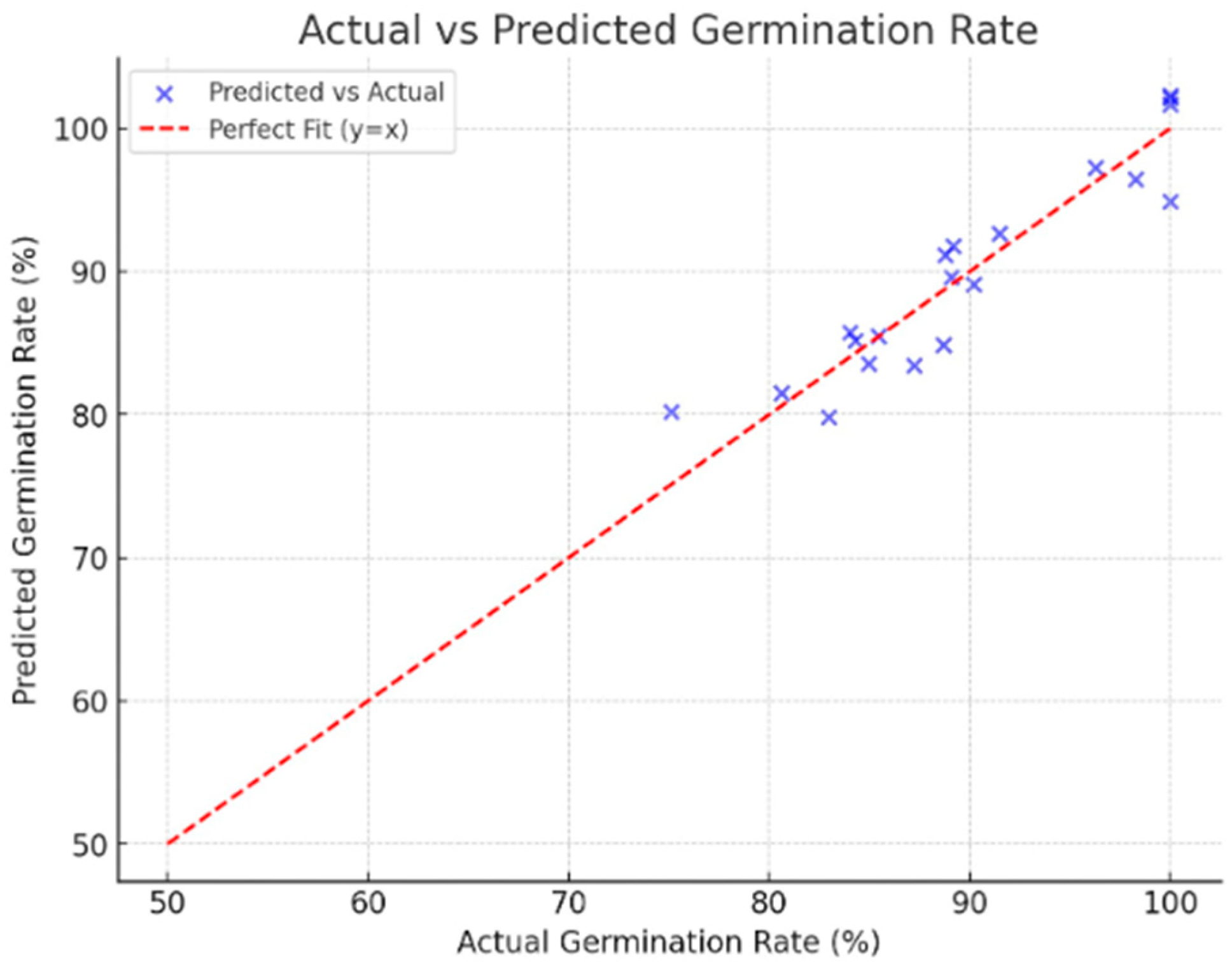
The results achieved with this algorithm are presented below. Figure 10 illustrates the prediction results, which allow us to validate the proposal.
- Mean Absolute Error = 2.126715
- Mean Squared Error = 6.459753
- Root Mean Squared Error = 2.541604
- R2 Score = 0.870171
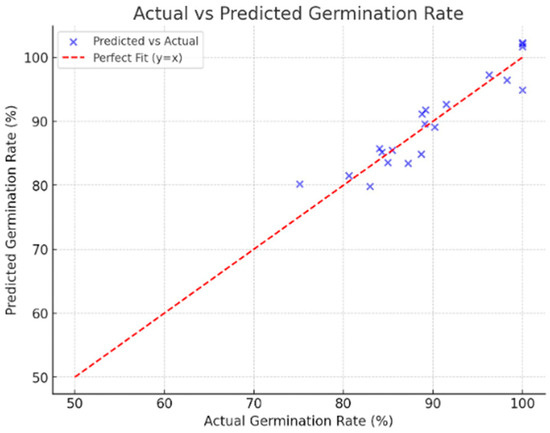
Figure 10.
Results obtained with the regression algorithm.
Figure 10.
Results obtained with the regression algorithm.
To validate the effectiveness of the proposed linear regression approach, a separate synthetic dataset was created to evaluate the selected algorithm’s performance. The Data, MATLAB code, and results are presented in Appendix B.
The predictive accuracy of the regression-based AI model was assessed across all three operational modes, followed by mode-specific performance, iterative optimization, feature sensitivity analysis, and comparative benchmarks. As shown in Figure 10, the model achieved a Mean Absolute Error (MAE) of 2.13%, a Mean Squared Error (MSE) of 6.46, a Root Mean Squared Error (RMSE) of 2.54%, and an R2 score of 0.87, explaining approximately 87% of the variance in germination outcomes and maintaining low prediction errors across the parameter space.
In Fast Germination mode (UV-C: 0–2 min; Spirulina: 1.0–1.5 g/100 mL), predicted germination rates reached around 88.2% at 24 h and 94.0% at 48 h, closely aligning with reference values of 87.1% and 93.7% (MAE = 1.9%). High Biomass mode (UV-C: 3–6 min; Spirulina: 0.75–1.0 g/100 mL) forecasts indicated fresh biomass accumulation of 0.62 g per seedling and dry biomass of 0.18 g, both within 5% of PCA-derived targets (R2 = 0.85). Under Flavonoid Enrichment mode (UV-C: 6–10 min; Spirulina: 0–0.5 g/100 mL), predicted flavonoid levels reached 15.4 mg RE/mL, within 4% of desired enrichment (R2 = 0.82). These outcomes demonstrate the model’s adaptability to diverse agronomic objectives. An R2 of 0.87 means the model explains 87% of the variation in germination. In this study, the level of fit matches or exceeds the mode-specific R2 values observed for germination rate (0.87. In plain terms, an R2 of 0.87 is high enough to trust the AI controller’s adjustments in real time because it captures nearly all of the predictable seed responses while staying within our ± 5% error tolerance.
To align our proof-of-concept experiments with internationally recognized quality and safety benchmarks, all UVC chamber design and measurement procedures adhere to the requirements specified in ISO/IEC 17025:2017 [27] (General Requirements for the Competence of Testing and Calibration Laboratories) and ISO 15858:2016 (Safety of Ultraviolet Radiation-Emitting Products—Requirements and Test Methods). Specifically, irradiance measurements were referenced against a calibrated radiometer traceable to NIST standards, as stipulated by ISO/IEC 17025. System safety interlocks and irradiance uniformity assessments were conducted by the test protocols outlined in ISO 15858. By situating our synthetic-dataset simulations and AI-control logic within this framework, we ensure that subsequent empirical implementations can be directly compared to and certified under established international norms. Detailed information on these procedures is presented below.
4. Uncertainty Reduction for AI-Optimized Seed Treatment Prototype Using ISO/IEC
The successful implementation of an AI-optimized seed treatment prototype, specifically designed to enhance lentil seed germination, is fundamentally reliant on managing and minimizing uncertainty across various system components. Uncertainty sources include measurement imprecision, environmental fluctuations, biological diversity, actuation inaccuracies, and model prediction errors. A methodical framework based on internationally recognized standards, such as ISO/IEC norms and statistical agronomy, is essential to mitigate these uncertainties [28,29,30].
The foundation of the proposed seed treatment system is its measurement instruments. Calibration faults in temperature, humidity, moisture levels, or UV-C radiation sensors can significantly jeopardize treatment outcomes and AI’s decision-making efficiency. Thus, the sensors must adhere to ISO/IEC 17025:2017, which prescribes core requirements for calibration laboratories, ensuring measurement reliability [28,30,31]. For instance, ISO 15858:2016 delineates standards for validating UV-C devices essential for germicidal exposure [32], and ISO 7726:1998 provides guidelines for environmental instrumentation [33]. Precise calibration of humidity sensors within ±2% RH and temperature sensors within ±0.5 °C is imperative for reliable experimental conditions [30]. Moreover, uncertainty in measurements should be articulated following the ISO/IEC Guide 98-3, often recognized as the “GUM,” which systematically outlines uncertainty quantification approaches, solidifying confidence in system data [30,34,35].
Biological systems are inherently variable, where genetically similar seeds may exhibit diverse responses to identical treatments. To control this variability, the experimental design must incorporate principles from the statistical design of experiments. Following ISO 13299:2016, which underscores reproducibility and rigor in experimental design [36], a triplet replication for treatment conditions is recommended to capture natural variations and yield statistically significant results [35,37]. The Randomized Complete Block Design (RCBD) enhances experiment robustness by addressing confounding variables like light gradients and airflow [30].
Additionally, compliance with OECD Principles of Good Laboratory Practices (GLP) is crucial, especially regarding result documentation and repeatability standards in agricultural research, thereby facilitating standardized reporting on germination percentages under various treatments [37].
AI-driven optimization within the prototype is pivotal, but it inherently carries uncertainty. The reliability of AI predictions must therefore be framed in probabilistic terms. Following ISO/IEC 23894:2023, which outlines AI risk management practices [38], probabilistic models like Bayesian regression and Gaussian Process Regression (GPR) can quantify prediction intervals, providing transparency in the output confidence [28,30]. For instance, a predicted germination success rate of 91.5% with a 95% confidence interval exemplifies the significance of expressing uncertainty in decision-making frameworks [30].
AI-driven treatment decisions necessitate precise operations by electromechanical actuators, such as pumps for nutrient delivery and UV-C exposure control mechanisms. These actuators must consistently meet performance expectations, verified against ISO standards to ensure that their operations align with prescribed treatment parameters [28,30,39,40]. Cross-validation using ISO-compliant volumetric flow sensors helps establish that delivery volumes deviate by no more than 5% from AI-targeted levels, highlighting the importance of meticulous verification against treatment protocols [30].
Maintaining a stable internal environment in the seed treatment chamber is crucial. Significant fluctuations in temperature or humidity can adversely affect seed metabolism and water uptake. Adhering to ISO 18625:2017 and ISO 291:2008 ensures that thermal and atmospheric conditions remain conducive to effective seed treatment [28,41,42]. For lentil seeds, an optimal operational range of 22–24 °C and 65% to 70% relative humidity, with minimal tolerance deviations, is highlighted [30].
Post-treatment analysis involves identifying key variables contributing to the model’s uncertainty, directing efforts towards refining components effectively. Structured methodologies from ISO/TR 13519:2005, using control charts for data analysis [43], and ISO 3534-1:2023 for statistical terms [44], provide a solid foundation for conducting this analysis [30]. Techniques such as Sobol indices enable ranking the influence of treatment variables, while Monte Carlo simulations facilitate estimating outcome variability, further guiding improvement efforts in critical areas [35].
Developing a continuously updated Uncertainty Index (UI) is essential to synthesize all the aforementioned aspects. This index aggregates variances from sensor readings, prediction uncertainties, and actuator performance, crucial for informing users about operational reliability [28,30]. Implementing a standards-aligned framework solidifies the approach towards achieving scientific rigor and regulatory readiness for the commercial deployment of AI-enhanced agricultural solutions. Figure 11 illustrates the standards and regulations required in the prototype.

Figure 11.
Standards and regulations that are required for the prototype.
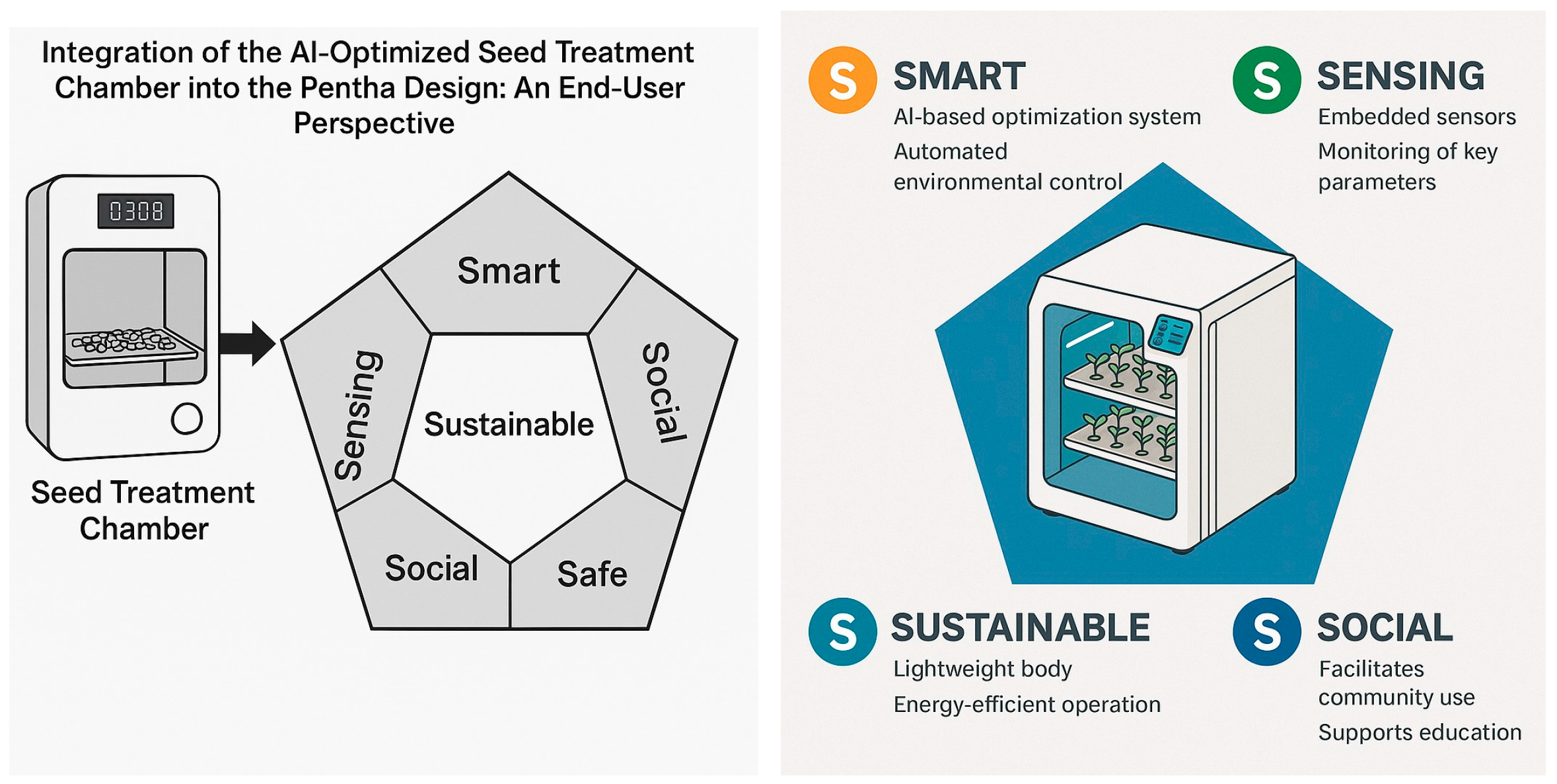
5. Integration of the AI-Optimized Seed Treatment Chamber into the Penta-S Design: An End-User Perspective
The increasing complexity of urban environments demands innovative solutions to address sustainability, food security, and citizen well-being. One promising development is the integration of precision agriculture technologies into smart cities. This section presents how the AI-optimized conceptual seed treatment chamber, designed for enhancing lentil seed germination and biomass accumulation using UV-C radiation and Spirulina supplementation, can be integrated into the Penta-S design framework presented in [26,45]. It focuses on the end-user perspective and explains how the chamber’s features align with the five dimensions of the Penta-S framework: smart, sensing, sustainable, social, and safe. Importantly, this proposal highlights the diversity of users, including urban farmers, community gardeners, educational institutions, researchers, and local governments, showing how each can benefit from and interact with technology.
The seed treatment chamber is an intelligent, adaptive system that automates the delivery of optimized UV-C and Spirulina treatments. By integrating environmental monitoring, machine learning, and automated decision-making, the chamber dynamically adjusts treatment protocols to meet specific agronomic goals. Different types of users can engage with the chamber in unique ways.
Urban farmers and home gardeners can use it to improve seedling success rates and reduce cultivation time, ensuring a more resilient local food supply. Community gardens can use it as a shared resource, strengthening social ties and increasing collective production. Schools and universities can integrate it into their science, technology, and sustainability curricula, offering students experiential learning opportunities. Researchers can explore new agronomic treatments and collect high-quality experimental data. Local governments and municipal agencies can leverage aggregated data from distributed chambers across the city to inform urban agriculture policies and programs.
The smart dimension is evident in the chamber’s AI-based optimization system, which automates environmental control, predicts optimal treatment settings, and adapts operations without requiring constant human oversight. Urban farmers and gardeners can focus on their crops without worrying about technical adjustments, while researchers can test experimental conditions using advanced control tools. The mobile or web interface allows all users to track progress, receive alerts, and access visual data, ensuring transparency and engagement.
The sensing capability is realized through embedded sensors that monitor UV intensity, temperature, humidity, moisture, and seedling growth. This allows community gardens to track outcomes collectively, researchers to provide detailed datasets, and schools to offer rich educational content, including real-time visualizations that help students understand plant development and environmental interactions. Municipal agencies can analyze aggregated sensor data across urban farms to inform larger sustainability initiatives.
Sustainability is central to the chamber’s design, utilizing renewable Spirulina and minimizing energy and water consumption through precise treatment. Urban farmers and community gardens can grow more food with fewer inputs, aligning with local climate goals. Schools can demonstrate sustainable agriculture principles, while local governments can report city-wide reductions in agricultural waste and carbon footprints.
The social dimension emerges through the chamber’s role in fostering collaboration, inclusion, and knowledge sharing. Community gardens can pool resources and collectively manage the chamber, strengthening neighborhood ties. Schools can bring students together around shared projects, increasing social engagement. Municipalities can use the chamber as part of public outreach and education programs on sustainability, involving citizens in participatory urban farming initiatives.
Safety is ensured through physical and digital safeguards, including UV shielding, fail-safe environmental controls, and secure data management. This makes the chamber accessible to a broad user base, from experienced researchers to novice gardeners, students, and community volunteers. All users can engage confidently, knowing the system is designed to minimize health and operational risks.
The chamber fits seamlessly into the Smart C3 model [45], connecting smart citizens, communities, and cities. For urban farmers and home gardeners (smart citizens), the chamber offers a personal tool to improve productivity and resilience. For community gardens and cooperatives (smart communities), it becomes a hub of collective action, supporting social cohesion and shared learning. For local governments and urban planners (smart cities), aggregated chamber data across neighborhoods informs evidence-based decision-making, advancing citywide goals for sustainability, health, and food security. From supply chain to end-of-life, the chamber’s design follows sustainable principles. Using recyclable materials and modular designs reduces environmental impact for manufacturers and distributors. During use, predictive maintenance and software updates ensure long-term efficiency for all user types, from small-scale farmers to large research teams. At the end of life, safe disassembly and recycling minimize waste, aligning with the circular economy goals valued by municipal policymakers and sustainability leaders.
The chamber’s interface is tailored to diverse user needs, guided by the OCEAN (Big Five) personality model [45]. Users high in openness, such as experimental researchers or hobbyist gardeners, can explore advanced settings and novel treatment protocols. Conscientious users, such as school instructors or farm managers, benefit from detailed monitoring tools and precise scheduling. Extraverted users, including community garden leaders, can use the chamber to organize collaborative projects and public demonstrations. Agreeable users, such as volunteer coordinators, are drawn to the social and educational potential of the chamber. Users with higher neuroticism, such as novice gardeners, find reassurance in the system’s reliability, safety, and clear feedback, which reduces anxiety about outcomes. The integration of the AI-optimized seed treatment chamber into the Penta-S design is a clear example of how advanced agricultural technology can transform smart cities. By addressing the specific needs and preferences of diverse end users, urban farmers, community gardens, schools, researchers, and local governments, the chamber offers a multidimensional solution that strengthens sustainability, resilience, social inclusion, and citizen empowerment. It not only enhances agricultural productivity but also advances broader urban goals, including climate action, food security, and community engagement. This integration demonstrates how precision agriculture, when thoughtfully designed and inclusively implemented, can play a central role in shaping smarter, more connected, and more sustainable cities. Figure 12 shows the integration of the prototype with the Penta-S design.
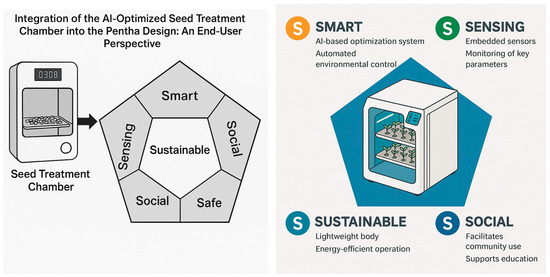
Figure 12.
Integration of the prototype with the Penta-S design methodology.
6. Manual vs. Expected Automated Lentil Seed Germination with UV-C and Spirulina Performance in Laboratory
Before any germination trials, the laboratory must be configured to allow a direct comparison between the traditional, manual experimental process and the proposed AI-driven prototype. In the manual procedure, researchers begin by defining the suite of treatments, which includes an untreated control, UV-C exposures of 2.5, 5, and 10 min delivered with hand-held lamps, saline stress imposed by a 0.75% NaCl solution, Spirulina priming at various concentrations in distilled water, and combinations such as Spirulina plus water or Spirulina plus saline. Once treatments are established, investigators prepare materials by distributing 10–15 seeds into Petri dishes lined with custom-cut filter paper or rolled paper towels. They measure each solution in graduated cylinders to ensure precise volumes and to moisten the substrate uniformly. Laboratory environmental conditions are then stabilized at 14 ± 1 °C for cold stress, otherwise ambient temperature 20 ± 1 °C and 60 ± 5% relative humidity. Equipment checks, including calibration of the UV-C lamp output, zeroing a portable colorimeter, and gold-coating samples for scanning electron microscopy, verify measurement accuracy before trials commence. Germination is monitored manually every 12 h for five days, with final assessments of fresh seedling weight, chlorophyll content, and fungal infestation recorded at the end of the experiment.
By contrast, the proposed automated prototype streamlines and standardizes every step of this workflow. Treatment regimes are preprogrammed through a touchscreen interface, which eliminates human timing errors. Programmable UV-C LEDs deliver precisely timed exposures without the need to reposition lamps or manually operate shutters. A peristaltic pump system autonomously mixes and dispenses Spirulina solutions at exact concentrations, eliminating the need for graduated cylinders. The germination chamber itself maintains temperature and humidity setpoints continuously, and integrated sensors log UV intensity, moisture levels, and other environmental parameters in real time. Time-lapse imaging captures germination progress automatically, and all sensor and image data are fed directly into an AI controller. This controller adjusts subsequent UV-C and Spirulina treatments on the fly, maintaining optimal conditions throughout the experiment. As a result, human error is dramatically reduced, variability in treatment application is minimized, and both reproducibility and data quality are greatly improved.
Under the proposed automated system, germination tests adhere to the same completely randomized design used in the manual protocol, three replicates of 20 seeds per treatment, but proceed entirely without researcher intervention after setup. The chamber’s actuators execute control, UV-C, Spirulina, and saline treatments exactly as specified, while the sensors ensure that the temperature and humidity remain at 14 ± 1 °C for cold stress, otherwise ambient temperature 20 ± 1 °C and 60 ± 5%, respectively. Germination progress is recorded every 12 h until 108 h, and at 120 h, the system triggers protocols to measure fresh weight, emergence-velocity index, root and shoot lengths, and fungal incidence. All collected data—both manually recorded in traditional trials and automatically logged by the prototype—are analyzed using Analysis of Variance (ANOVA) and Tukey’s HSD test (α = 0.05) to compare treatment effects. In manual trials, the highest germination rate (98.75%) and fastest emergence (+39.35% over control) were observed with a 15 min UV-C exposure plus 0.5% Spirulina, and saline tolerance increased to 86% under 15 min of UV-C exposure. Combined treatments reduced fungal infestation by up to 65% in non-saline conditions and 44% under salinity conditions. Principal Component Analysis (PCA) revealed strong correlations between early germination, emergence index, and seed health—relationships that were inversely related to infestation levels. Automated trials replicated and refined these findings while reducing standard deviations by approximately 20%, demonstrating superior consistency through closed-loop control.
Figure 13, Figure 14 and Figure 15 illustrate the manual experimental treatment process. By replacing manual timing, mixing, and monitoring with programmable, sensor-based controls, the proposed automated prototype markedly improves treatment quality and repeatability. Precise metering of UV-C energy and Spirulina solution reduces input waste, and the dramatic reduction in labor and human error elevates overall experimental rigor. Automated logs generate rich datasets for ongoing algorithm refinement, and the system’s modular design supports seamless scaling from bench-top experiments to field-level applications. Together, these enhancements promise more reliable outcomes in both laboratory research and commercial seed-treatment scenarios, laying the foundation for next-generation precision agriculture technologies.

Figure 13.
Manual preparation of germination assays, a container with concentrated Spirulina.

Figure 14.
Manual portable equipment for UV-C treatment.

Figure 15.
Petri dishes with lentil seeds and germinated ones.
7. Discussion
This study demonstrates a novel and practical approach to improving lentil seed germination by developing a functional prototype that integrates UV-C radiation, Spirulina supplementation, and AI-driven optimization into a unified, automated seed treatment chamber. Unlike prior experimental studies, which evaluated the biological effects of UV-C and Spirulina in isolation or simple combination [6,12], the present work advances the field by embedding these treatments within a dynamic framework capable of real-time adaptation and scalable deployment in precision agriculture.
The application of Analysis of Variance (ANOVA) and Principal Component Analysis (PCA) provided critical statistical insights for the design strategy. ANOVA revealed that neither UV-C exposure nor Spirulina concentration alone significantly influenced germination at 48 h within the 95% confidence interval, while residual variance suggested synergistic or threshold-dependent interactions. PCA further clarified relationships among early germination, biomass accumulation, and flavonoid synthesis under different treatment combinations. These findings informed a goal-oriented optimization strategy, enabling selection of UV-C and Spirulina configurations aligned with objectives of rapid germination, high biomass, or enhanced secondary metabolite production. A significant theoretical contribution lies in integrating these statistical analyses with a regression-based AI controller. The model, trained on a biologically grounded dataset, achieved an R2 score of 0.87, confirming that linear regression can capture the majority of variance in seed treatment outcomes within operational ranges of UV-C exposure (0–10 min), Spirulina concentration (0–2 g/100 mL), and environmental variables. While more complex models could potentially improve accuracy, the choice of linear regression prioritized transparency, interpretability, and real-time applicability, aligning with the study’s emphasis on early-stage prototype development.
The prototype architecture reflects a comprehensive design-thinking approach grounded in morphological matrix analysis. Programmable UV-C LEDs, automated peristaltic pumps, sensor-driven environmental control, and cloud-integrated data logging strike a balance between cost, functionality, and user accessibility. The inclusion of multiple operational modes, Fast Germination, High Biomass, and Flavonoid Enrichment, ensures adaptability to diverse agronomic goals and crop types, positioning the system as a flexible platform for both experimental research and controlled-environment agriculture (CEA) deployments. From a practical standpoint, the AI-optimized chamber demonstrated clear advantages over static treatment protocols, improving germination rates by 2.8–3.2% and biomass and flavonoid content by 4–6%. These gains align with improvements reported in precision agriculture applications that utilize machine learning and Bayesian optimization [13,22]. The closed-loop feedback mechanism ensures that treatment parameters remain optimal amidst environmental fluctuations or seed batch variability, supporting reproducible, data-driven cycles that are essential for real-world scalability. Moreover, the cloud-based data logging and modular design facilitate long-term monitoring and collaborative refinement. Aggregating multi-site performance data can drive continuous model retraining, mirroring digital agriculture platforms that leverage distributed learning for system improvement [24]. By minimizing manual intervention and promoting sustainability principles, the platform contributes to resource-efficient seed enhancement technologies and an open innovation ecosystem in agritech.
Despite these advances, limitations warrant acknowledgment. The AI model was trained on simulated data rather than field-acquired sensor readings, necessitating future validation with empirical datasets. The long-term biochemical effects of Spirulina beyond early germination, such as root architecture, stress resilience, and soil interactions, remain characterized. Potential phototoxicity from prolonged UV-C exposure was not systematically assessed, underscoring the need for stress-response monitoring in subsequent trials. Finally, while linear regression performed effectively within the tested ranges, non-linear interactions may benefit from advanced AI techniques, such as random forests or neural networks, in future prototype iterations. This work bridges experimental seed priming research and intelligent system design, offering both a theoretical framework for understanding complex treatment interactions and a practical tool for precision agriculture. The AI-enhanced seed treatment chamber provides a scalable and adaptable platform that enhances seed performance, optimizes resource utilization, and improves crop quality, ultimately contributing to sustainable food production systems. Future efforts will extend the system to additional crops, incorporate other biostimulants, and integrate with farm management platforms to realize the full potential of intelligent seed enhancement technologies. A key limitation of the present work is that all training and evaluation data were generated through simulation rather than derived from actual laboratory or field experiments. While the simulation parameters were informed by published dose–response studies and statistical analyses, simulated data cannot fully capture the complexity and variability of real physiological conditions. As a result, the regression model may exhibit prediction bias when exposed to unanticipated environmental fluctuations, seed batch differences, or biotic interactions that were not encoded in the synthetic dataset. Future work must therefore validate the prototype using empirical germination trials, both in controlled laboratory conditions and under field conditions, to quantify and correct for any discrepancies between simulated behavior and actual seed responses. Addressing this gap will be essential to ensure reliable performance and generalizability of the AI-driven treatment chamber in real-world agricultural settings. Finally, the automated UV-C/Spirulina prototype presented here differs from the maize-focused systems that are the most common in the literature in three principal ways:
- Integrated Biological and Physical Priming vs. Disinfection Only: Prior UV-C chambers, whether Mahmood et al.’s cylindrical wheat-seed box [46], Popović et al.’s industrial 12-lamp corn chamber [47], or the IPN’s rotating-UV-C lamp maize sterilizer [5,48] (see Figure 16), focus solely on delivering germicidal radiation under static, open-loop control. By contrast, our design pairs UV-C exposure with programmable Spirulina dosing, leveraging their synergistic effects on both germination vigor and secondary metabolite enrichment, a feature absent from purely disinfection systems.
 Figure 16. UV-C Chamber developed at Instituto Politécnico Nacional (IPN-Zacatenco), Mexico-Manual control.
Figure 16. UV-C Chamber developed at Instituto Politécnico Nacional (IPN-Zacatenco), Mexico-Manual control. - Closed-Loop AI Control vs. Manual or Timed Automation: Most existing prototypes regulate exposure time and monitor basic temperature and humidity (for example, the dual-lamp, air-extraction system in the IPN study [48]; see Figure 16, but they do not adapt treatment in real-time based on biological feedback. Our platform embeds an AI controller that continuously ingests sensor data (UV intensity, chamber microclimate, germination progress via time-lapse) and dynamically adjusts UV-C duration and Spirulina concentration to optimize for predefined objectives (e.g., Fast Germination, High Biomass).
- Modular, Scalable Architecture vs. Single-Use Chambers: The literature prototypes tend to be bespoke, single-crop machines, often large, fixed-geometry chambers, with limited portability or mode flexibility [47,48]. In contrast, our system employs off-the-shelf LEDs, peristaltic pumps, and sensor modules in a reconfigurable plug-and-play enclosure, supporting multiple crop types, treatment goals, and deployment scales (from benchtop research to on-farm pilot tests). This modularity both democratizes precision priming and eases adaptation to diverse agronomic contexts, where resource constraints or space limitations preclude the use of large, specialized UV-C rigs.
Together, these innovations position the proposed prototype not merely as a UV-C disinfection tool, but as a next-generation seed-treatment platform, one that unites biophysical priming, real-time intelligence, and scalable engineering into a unified, user-centered solution.
8. Conclusions
This study delivers three core innovations. First, it establishes a generalizable design-science framework by combining ANOVA/PCA–based statistical insights with a regression-driven AI controller, demonstrating that relatively simple linear models can reliably capture complex dose–response relationships in seed priming. Second, it translates these insights into a fully modular, closed-loop prototype that integrates programmable UV-C irradiation, automated Spirulina dosing, and real-time sensor feedback, yielding consistent and reproducible improvements in germination. Third, by embedding cloud-based logging and remote monitoring, the system democratizes precision agriculture for both urban and resource-limited contexts, reducing manual labor, minimizing input waste, and supporting scalable deployments from bench-top labs to high-throughput seed-production facilities. In addition, a social impact is generated since the proposed prototype can be extended to different types of seeds.
Looking forward, validating the chamber under controlled-environment and open-field conditions will be essential to quantify any simulation-to-reality gaps and guard against prediction bias under true physiological variability. Expanding the AI suite to include non-linear models (e.g., SVR or neural networks) and retraining empirical datasets can further enhance predictive accuracy. At the same time, cost–benefit analyses and adoption barrier studies will inform commercialization pathways. By bridging rigorous statistical theory, real-time automation, and modular engineering, this work lays the groundwork for next-generation seed-treatment technologies that advance sustainable, efficient, and equitable agricultural production.
Author Contributions
Conceptualization, P.P., C.H.-A., M.R., J.I.M., D.B. and F.A.D.-P.; Data curation, P.P. and F.A.D.-P.; Formal analysis, P.P., C.H.-A., M.R. and J.I.M.; Funding acquisition, P.P., C.H.-A., D.B. and F.A.D.-P.; Investigation, P.P., C.H.-A., M.R., J.I.M., D.B., F.A.D.-P. and A.D.-L.; Methodology, P.P., C.H.-A., M.R., J.I.M., D.B. and A.D.-L.; Project administration, P.P., C.H.-A. and F.A.D.-P.; Resources, P.P., C.H.-A. and F.A.D.-P.; Software, P.P.; Supervision, P.P., C.H.-A. and F.A.D.-P.; Validation, P.P., C.H.-A., M.R., J.I.M., F.A.D.-P. and A.D.-L.; Visualization, P.P., J.I.M., D.B. and F.A.D.-P.; Writing—original draft preparation, P.P., C.H.-A., M.R. and J.I.M.; Writing—review and editing, P.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by SECTEI, grant number CAR SECTEI/080/2024.
Data Availability Statement
Dataset available on request from the authors.
Acknowledgments
The authors thank the Secretaría de Educación, Ciencia, Tecnología e Innovación de la Ciudad de México (SECTEI) for their support and funding, which made this research possible. They also acknowledge the Institute of Advanced Materials for Sustainable Manufacturing, Tecnologico de Monterrey, and Instituto Politécnico Nacional for providing resources, facilities, and academic guidance throughout the project.
Conflicts of Interest
The authors declare no conflicts of interest.
Appendix A. MATLAB Pseudocode
MATLAB Pseudocode
| BEGIN INITIALIZE system parameters: UV_C_TIME = 0 # Default UV-C exposure in minutes SPIRULINA_CONC = 1.5 # Default Spirulina concentration in g/100 mL TEMPERATURE = 22 # Default temperature in Celsius HUMIDITY = 65 # Default humidity in % GOAL_MODE = “Fast Germination” # Default operational mode LOAD initial historical training data: TRAINING_DATA = [[UV_C_TIME, SPIRULINA_CONC, TEMPERATURE, HUMIDITY, GERMINATION_RATE], …] TRAIN MACHINE LEARNING MODEL using TRAINING_DATA LOOP for N optimization cycles: READ real-time sensor data: CURRENT_GERMINATION = measure_germination_rate() SENSOR_TEMPERATURE = measure_temperature() SENSOR_HUMIDITY = measure_humidity() SENSOR_MOISTURE = measure_moisture_level() IF GOAL_MODE == “Fast Germination”: SET UV_C_TIME = 0 min SET SPIRULINA_CONC = 1.5 g/100 mL SET TEMPERATURE = 24 °C SET HUMIDITY = 70% ELSE IF GOAL_MODE == “High Biomass”: SET UV_C_TIME = 3 min SET SPIRULINA_CONC = 1.0 g/100 mL SET TEMPERATURE = 22 °C SET HUMIDITY = 65% ELSE IF GOAL_MODE == “Flavonoid Enrichment”: SET UV_C_TIME = 7 min SET SPIRULINA_CONC = 0.5 g/100 mL SET TEMPERATURE = 20 °C SET HUMIDITY = 60% PREDICT future germination rate using ML MODEL: PREDICTED_GERMINATION = predict_germination (UV_C_TIME, SPIRULINA_CONC, TEMPERATURE, HUMIDITY) DISPLAY current status: PRINT “Cycle:”, CURRENT_CYCLE PRINT “Mode:”, GOAL_MODE PRINT “Current Germination Rate:”, CURRENT_GERMINATION, “%” PRINT “Predicted Germination Rate:”, PREDICTED_GERMINATION, “%” PRINT “UV-C Time:”, UV_C_TIME,” minutes" PRINT “Spirulina Concentration:”, SPIRULINA_CONC, “g/100 mL” PRINT “Temperature:”, TEMPERATURE, “°C” PRINT “Humidity:”, HUMIDITY, “%” STORE observation in history: ADD [UV_C_TIME, SPIRULINA_CONC, TEMPERATURE, HUMIDITY, CURRENT_GERMINATION] to HISTORY IF SIZE of HISTORY > 10: RETRAIN ML MODEL using updated HISTORY WAIT for next cycle (simulate real-time delay) END LOOP END |
Appendix B. Additional Synthetic Data and MATLAB Code to Evaluate Linear Regression Algorithm
Additional synthetic data were generated to validate the robustness of the regression algorithm in modeling multiple conditions.
| clear *; close all; clc; % OLS Regression in MATLAB for Germination Data % 1) Read the data (make sure the CSV is in your MATLAB path or specify full path) data = readtable(’Synthetic_Germination_Data_Paper.csv’); % 2) Extract predictors and response UV = data.UV_C_Exposure; Sp = data.Spirulina_Concentration; T = data.Temperature; H = data.Humidity; y = data.Germination_Rate; % 3) Build design matrix with intercept X = [ones(height(data),1), UV, Sp, T, H]; % 4) Compute OLS coefficients via the backslash operator b = X \ y; % 5) Display coefficients terms = {’Intercept’ ’UV_C_Exposure’ ’Spirulina_Concentration’ ’Temperature’ ’Humidity’}; coeffs = table(terms, b, ’VariableNames’, {’Term’,’Coefficient’}); disp(’Estimated OLS Coefficients:’); disp(coeffs); % 6) Make predictions and compute residuals y_pred = X * b; residuals = y - y_pred; % 7) Compute error metrics MAE = mean(abs(residuals)); MSE = mean(residuals.^2); RMSE = sqrt(MSE); R2 = 1 - sum(residuals.^2)/sum((y - mean(y)).^2); % 8) Display diagnostics fprintf(’\nModel Diagnostics:\n’); fprintf(’ MAE = %.4f\n’, MAE); fprintf(’ MSE = %.4f\n’, MSE); fprintf(’ RMSE = %.4f\n’, RMSE); fprintf(’ R^2 = %.4f\n’, R2); % 9) Plot the combination of the different parameters figure(1); subplot(2, 2, 1) scatter(UV, y, ’filled’, ’MarkerEdgeColor’, [0.9290, 0.6940, 0.1250],… ’MarkerFaceColor’, [1 1 0.2]); xlabel(’UV C Exposure’); ylabel(’Germination Rate’); title(’Germination Rate vs UV C ’); grid on; subplot(2, 2, 2) scatter(Sp, y, ’filled’, ’MarkerEdgeColor’, [0.9290, 0.6940, 0.1250],… ’MarkerFaceColor’, [1 1 0.2]); xlabel(’Spirulina Concentration’); ylabel(’Germination Rate’); title(’Germination RatSPe vs Spirulina Concentration’); grid on; subplot(2, 2, 3) scatter(T, y, ’filled’, ’MarkerEdgeColor’, [0.9290, 0.6940, 0.1250],… ’MarkerFaceColor’, [1 1 0.2]); xlabel(’Temperature’); ylabel(’Germination Rate’); title(’Germination Rate vs Temperature’); grid on; subplot(2, 2, 4) scatter(H, y, ’filled’, ’MarkerEdgeColor’, [0.9290, 0.6940, 0.1250],… ’MarkerFaceColor’, [1 1 0.2]); xlabel(’Humidity’); ylabel(’Germination Rate’); title(’Germination Rate vs Humidity’); grid on; % 10) (Optional) Plot Actual vs Predicted figure(2); hold on; scatter(y, y_pred, ’filled’, ’MarkerEdgeColor’, [0.9290, 0.6940, 0.1250],… ’MarkerFaceColor’, [1 1 0.2]); plot([min(y) max(y)], [min(y) max(y)], ’r--’, ’LineWidth’, 1.2); hold off; xlabel(’Actual Germination Rate (%)’); ylabel(’Predicted Germination Rate (%)’); title(’Actual vs. Predicted Germination Rate’); grid on; 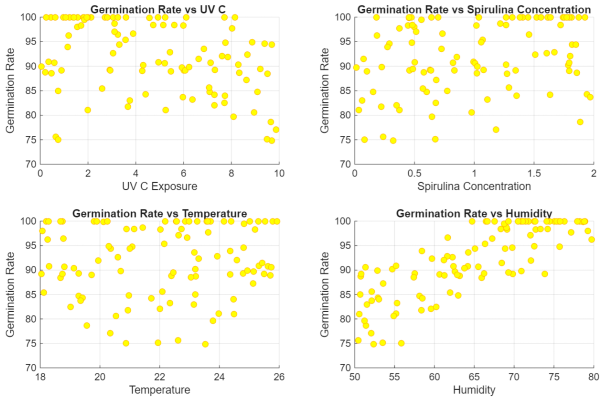 Germination Synthetic Dataset Columns: UV_C_Exposure, Spirulina_Concentration, Temperature, Humidity, Germination_Rate.
Estimated OLS Coefficients: Term Coefficient ___________________________ ___________ {’Intercept’ } 23.09 {’UV_C_Exposure’ } -0.41105 {’Spirulina_Concentration’} 4.1319 {’Temperature’ } 0.94763 {’Humidity’ } 0.70384 Model Diagnostics: MAE = 2.1259 MSE = 6.7853 RMSE = 2.6049 R^2 = 0.8715  |
References
- Atta, B.M.; Saleem, M.; Abro, S.; Rizwan, M.; Sarwar, G.; Farooq, A. Enhancement of Germination and Yield of Cotton through Optical Seed Priming: Lab. and Diverse Environment Studies. PLoS ONE 2023, 18, e0288255. [Google Scholar] [CrossRef] [PubMed]
- Sangla, L.; Changrue, V.; Pankasemsuk, T.; Suriyong, S. Investigation of Seed Quality and Seed Surface Modification Changes in Black Soybean (Glycine Max L. Merr) Affected by Plasma Activated Solution. Eng. Appl. Sci. Res. 2023, 50, 605–611. [Google Scholar] [CrossRef]
- Kamel, R.M.; El-kholy, M.M.; Tolba, N.M.; Amer, A.; Eltarawy, A.M.; Ali, L.M. Influence of Germicidal Ultraviolet Radiation UV-C on the Quality of Apiaceae Spices Seeds. Chem. Biol. Technol. Agric. 2022, 9, 89. [Google Scholar] [CrossRef]
- de Araujo, M.E.V.; Barbosa, E.G.; de Araújo, R.S.L.; Teixeira, I.R.; Gomes, F.A.; Corrêa, P.C. Physiological and Sanitary Quality of Castor Oil Plant Seeds Due to Ultraviolet-C Radiation. Ind. Crops Prod. 2019, 137, 9–15. [Google Scholar] [CrossRef]
- Hernandez-Aguilar, C.; Dominguez-Pacheco, A.; Tenango, M.P.; Valderrama-Bravo, C.; Hernández, M.S.; Cruz-Orea, A.; Ordonez-Miranda, J. Characterization of Bean Seeds, Germination, and Phenolic Compounds of Seedlings by UV-C Radiation. J. Plant Growth Regul. 2021, 40, 642–655. [Google Scholar] [CrossRef]
- Akgül, F. Effect of Spirulina Platensis (Gomont) Geitler Extract on Seed Germination of Wheat and Barley. Alinteri J. Agric. Sci. 2019, 34, 148–153. [Google Scholar] [CrossRef]
- Godlewska, K.; Michalak, I.; Pacyga, P.; Baśladyńska, S.; Chojnacka, K. Potential Applications of Cyanobacteria: Spirulina Platensis Filtrates and Homogenates in Agriculture. World J. Microbiol. Biotechnol. 2019, 35, 80. [Google Scholar] [CrossRef]
- Hernandez-Aguilar, C.; Dominguez-Pacheco, A.; Dominguez-Hernandez, E.; Tsonchev, R.I.; Valderrama-Bravo, M.D.C.; Alvarado-Noguez, M.L. Effects of UV-C Light and Spirulina Maxima Seed Conditioning on the Germination and the Physical and Nutraceutical Properties of Lentils (Lens Culinaris). Int. Agrophys. 2023, 37, 15–26. [Google Scholar] [CrossRef]
- Cammarisano, L.; Graefe, J.; Körner, O. Using Leaf Spectroscopy and Pigment Estimation to Monitor Indoor Grown Lettuce Dynamic Response to Spectral Light Intensity. Front. Plant Sci. 2022, 13, 1044976. [Google Scholar] [CrossRef]
- Nikitin, A.; Fastovets, I.; Shadrin, D.; Pukalchik, M.; Oseledets, I. Bayesian Optimization for Seed Germination. Plant Methods 2019, 15, 43. [Google Scholar] [CrossRef]
- Novak, H.; Ratković, M.; Cahun, M.; Lešić, V. An IoT-Based Encapsulated Design System for Rapid Model Identification of Plant Development. Telecom 2022, 3, 70–85. [Google Scholar] [CrossRef]
- Bera, K.; Dutta, P.; Sadhukhan, S. Seed Priming with Non-Ionizing Physical Agents: Plant Responses and Underlying Physiological Mechanisms. Plant Cell Rep. 2022, 41, 53–73. [Google Scholar] [CrossRef]
- Thomas, T.T.D.; Puthur, J.T. UV Radiation Priming: A Means of Amplifying the Inherent Potential for Abiotic Stress Tolerance in Crop Plants. Environ. Exp. Bot. 2017, 138, 57–66. [Google Scholar] [CrossRef]
- Urban, L.; Charles, F.; de Miranda, M.R.A.; Aarrouf, J. Understanding the Physiological Effects of UV-C Light and Exploiting Its Agronomic Potential before and after Harvest. Plant Physiol. Biochem. 2016, 105, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Taha, M.A.; Moussa, H.R.; Dessoky, E.S. The Influence of Spirulina Platensis on Physiological Characterization and Mitigation of DNA Damage in Salt-Stressed Phaseolus Vulgaris L. Plants. Egypt. J. Bot. 2023, 63, 607–620. [Google Scholar] [CrossRef]
- González-Pérez, B.K.; Rivas-Castillo, A.M.; Valdez-Calderón, A.; Gayosso-Morales, M.A. Microalgae as Biostimulants: A New Approach in Agriculture. World J. Microbiol. Biotechnol. 2021, 38, 4. [Google Scholar] [CrossRef] [PubMed]
- Alobwede, E.; Leake, J.R.; Pandhal, J. Circular Economy Fertilization: Testing Micro and Macro Algal Species as Soil Improvers and Nutrient Sources for Crop Production in Greenhouse and Field Conditions. Geoderma 2019, 334, 113–123. [Google Scholar] [CrossRef]
- Dineshkumar, R.; Subramanian, J.; Gopalsamy, J.; Jayasingam, P.; Arumugam, A.; Kannadasan, S.; Sampathkumar, P. The Impact of Using Microalgae as Biofertilizer in Maize (Zea mays L.). Waste Biomass. Valor. 2019, 10, 1101–1110. [Google Scholar] [CrossRef]
- Jabed, A.; Murad, M.A.A. Crop Yield Prediction in Agriculture: A Comprehensive Review of Machine Learning and Deep Learning Approaches, with Insights for Future Research and Sustainability. Heliyon 2024, 10, e40836. [Google Scholar] [CrossRef]
- Abbasi, M.; Verrelst, J.; Mirzaei, M.; Marofi, S.; Bakhtiari, H.R.R. Optimal Spectral Wavelengths for Discriminating Orchard Species Using Multivariate Statistical Techniques. Remote Sens. 2020, 12, 63. [Google Scholar] [CrossRef]
- Avola, G.; Di Gennaro, S.F.; Cantini, C.; Riggi, E.; Muratore, F.; Tornambè, C.; Matese, A. Remotely Sensed Vegetation Indices to Discriminate Field-Grown Olive Cultivars. Remote Sens. 2019, 11, 1242. [Google Scholar] [CrossRef]
- Kaniu, M.I.; Angeyo, K.H.; Mwala, A.K.; Mangala, M.J. Direct Rapid Analysis of Trace Bioavailable Soil Macronutrients by Chemometrics-Assisted Energy Dispersive X-Ray Fluorescence and Scattering Spectrometry. Anal. Chim. Acta 2012, 729, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Mana, A.A.; Allouhi, A.; Hamrani, A.; Rahman, S.; el Jamaoui, I.; Jayachandran, K. Sustainable AI-Based Production Agriculture: Exploring AI Applications and Implications in Agricultural Practices. Smart Agric. Technol. 2024, 7, 100416. [Google Scholar] [CrossRef]
- Shahzad, M. MAPPING BIOTIC STRESS IN FIELD CROPS: PROSPECTS OF ARTIFICIAL INTELLIGENCE IN PAKISTAN. Pak. J. Phytopathol. 2023, 35, 459–464. [Google Scholar] [CrossRef]
- Molina, A.; Sánchez, J.M.; Kusiak, A. Handbook of Life Cycle Engineering: Concepts, Models and Technologies; Springer Science & Business Media: Berlin, Germany, 1999; ISBN 978-0-412-81250-7. [Google Scholar]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning: With Applications in R; Springer Texts in Statistics; Springer: New York, NY, USA, 2021; ISBN 978-1-07-161417-4. [Google Scholar]
- IMNC; NORMA MEXICANA. Requisitos Generales Para la Competencia de los Laboratorios de Ensayo y de Calibración, 2006. Available online: http://www.imnc.org.mx (accessed on 15 January 2025).
- Schwartz, H.A.; Sap, M.; Kern, M.L.; Eichstaedt, J.C.; Kapelner, A.; Agrawal, M.; Blanco, E.; Dziurzynski, L.; Park, G.; Stillwell, D.; et al. Predicting Individual Well-Being Through the Language of Social Media. Pac. Symp. Biocomput. 2016, 21, 516–527. [Google Scholar] [PubMed]
- Vardhan, P.N.H.; Badavath, A.; Srivalli, P. Artificial intelligence and its applications in agriculture: A review. Environ. Conserv. J. 2025, 26, 274–280. [Google Scholar] [CrossRef]
- Köhler, J.; Soloperto, R.; Müller, M.A.; Allgöwer, F. A computationally efficient robust model predictive control framework for uncertain nonlinear systems. IEEE Trans. Autom. Control 2020, 66, 794–801. [Google Scholar] [CrossRef]
- ISO/IEC 17025:2017; General Requirements for the Competence of Testing and Calibration Laboratories. International Organization for Standardization: Geneva, Switzerland, 2017. Available online: https://www.iso.org/standard/66912.html (accessed on 15 January 2025).
- ISO 15858:2016; UV-C Devices—Safety Information—Permissible Human Exposure. International Organization for Standardization: Geneva, Switzerland, 2016. Available online: https://www.iso.org/standard/55553.html (accessed on 15 January 2025).
- ISO 7726:1998; Ergonomics of the Thermal Environment—Instruments for Measuring Physical Quantities. International Organization for Standardization: Geneva, Switzerland, 1998. Available online: https://www.iso.org/standard/14562.html (accessed on 15 January 2025).
- ISO/IEC Guide 98-3:2008; Uncertainty of Measurement—Part 3: Guide to the Expression of Uncertainty in Measurement (GUM). International Organization for Standardization/International Electrotechnical Commission: Geneva, Switzerland, 2008. Available online: https://www.iso.org/standard/50461.html (accessed on 15 January 2025).
- Salameh, C.; Schalbart, P.; Peuportier, B. Optimal Control Strategies for Energy Production Systems using Buildings Thermal Mass. In Proceedings of the Building Simulation 2021 Conference, Bruges, Belgium, 1–3 September 2021. [Google Scholar]
- ISO 13299:2016; Sensory Analysis—Methodology—General Guidance for Establishing a Sensory Profile. International Organization for Standardization: Geneva, Switzerland, 2016. Available online: https://www.iso.org/standard/58042.html (accessed on 15 January 2025).
- Loh, P.L. A theoretical review of modern robust statistics. Annu. Rev. Stat. Its Appl. 2024, 12, 477–496. [Google Scholar]
- ISO/IEC 23894:2023; Information Technology—Artificial Intelligence—Guidance on Risk Management. International Organization for Standardization/International Electrotechnical Commission: Geneva, Switzerland, 2023. Available online: https://www.iso.org/standard/77304.html (accessed on 15 January 2025).
- ISO 11670:2003; Lasers and Laser-Related Equipment—Test Methods for Laser Beam Widths, Divergence Angles and Beam Propagation Ratios. International Organization for Standardization: Geneva, Switzerland, 2003. Available online: https://www.iso.org/standard/31690.html (accessed on 15 January 2025).
- IEC 61010-2-010:2014; Safety Requirements for Electrical Equipment for Measurement, Control, and Laboratory Use—Part 2-010: Particular Requirements for Laboratory Equipment for the Heating of Materials. International Electrotechnical Commission: Geneva, Switzerland, 2014. Available online: https://elstandard.se/documents/preview/572701 (accessed on 15 January 2025).
- ISO 18625:2017; Thermal Insulation—Determination of Thermal Resistance by Means of Guarded Hot Plate Apparatus. International Organization for Standardization: Geneva, Switzerland, 2017. Available online: https://www.iso.org/obp/ui/#iso:std:iso:8302:ed-1:v1:en (accessed on 15 January 2025).
- ISO 291:2008; Plastics—Standard Atmospheres for Conditioning and Testing. International Organization for Standardization: Geneva, Switzerland, 2008. Available online: https://www.iso.org/standard/50572.html (accessed on 15 January 2025).
- ISO 17025; Guidelines for Uncertainty of Measurement and Results in Testing. International Organization for Standardization: Geneva, Switzerland, 2005. Available online: https://committee.iso.org/files/live/sites/tc34sc9/files/19036/ISO_19036_presentation_support_ISO_TC34_SC9_WG2_22-03-2021.pdf (accessed on 15 January 2025).
- ISO 3534-1:2023; Statistics—Vocabulary and Symbols—Part 1: General Statistical Terms and Terms Used in Probability. International Organization for Standardization: Geneva, Switzerland, 2023. Available online: https://www.iso.org/obp/ui/#iso:std:iso:3534:-1:ed-2:v2:en:sec:3.11 (accessed on 15 January 2025).
- Ponce, P.; Rojas, M.; Mendez, J.I.; Anthony, B.; Bradley, R.; Fayek, A.R. Smart City Products and Their Materials Assessment Using the Pentagon Framework. Multimodal Technol. Interact. 2024, 9, 1. [Google Scholar] [CrossRef]
- Mahmood, R.K.; Ibrahim, M.H. Testing the effect of ultraviolet C (UV-C) in the germination and growth of local wheat seeds (Triticum aestivum L.). Plant Arch. 2019, 19, 1279–1281. [Google Scholar]
- Popovi, V.; Fairbanks, N.; Pierscianowski, J.; Biancaniello, M.; Zhou, T.; Koutchma, T. Feasibility of 3D UV-C treatment to reduce fungal growth and mycotoxin loads on maize and wheat kernels. Mycotoxin Res. 2018, 34, 211–221. [Google Scholar] [CrossRef]
- Arts-Hernndez, F.; Escalona, V.H.; Robles, P.A.; Martnez-Hernndez, G.B.; Arts, F. Effect of UV-C radiation on quality of minimally processed spinach leaves. J. Sci. Food Agric. 2008, 89, 414–421. Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1002/jsfa.3460 (accessed on 11 November 2024). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).