Abstract
Sunflower meal represents a protein- and fiber-rich by-product of the oil industry with potential application as a natural stabilizer in food emulsions. Building upon previous findings that emphasized the role of protein content in emulsion stability, the present study further investigated the combined effect of protein level and particle size distribution of sunflower meal fractions on the formation and stability of oil-in-water emulsions. Two sets of sunflower meal fractions were prepared from finely milled material, fractionated, and blended in controlled proportions to obtain four protein-enriched (30 ± 1%) and four cellulose-rich (15 ± 1%) fractions, each defined by particle size ranges of 250/200, 200/125, 125/100, and <100 µm. Emulsion stability was evaluated through droplet size analysis, zeta potential measurements, and creaming index determination during seven days of storage. The results demonstrated that both protein content and particle size significantly affected the emulsifying and stabilizing behavior of sunflower meal fractions. For the low-protein group (15%), larger particle sizes (250/200 µm) yielded smaller emulsion droplets (D[4.3] = 66.03 µm) and higher zeta potential values (−15.53 mV), while in the high-protein group (30%), droplet size distribution was more uniform (D[4.3] from 72.13 to 76.29 µm). During seven days of storage, all emulsions exhibited a gradual increase in creaming index, followed by partial stabilization at later time points. Emulsions prepared with sunflower meal fractions of higher-protein content showed consistently lower creaming index values, indicating improved physical stability throughout storage. Overall, the study confirmed that the interplay between composition (protein level) and physical structure (particle size) governs the emulsification efficiency of sunflower meal fractions, providing insights for their potential application as plant-based stabilizers in food systems.
1. Introduction
The stabilization of oil-in-water (O/W) emulsions by natural hydrocolloids is of great interest in both food and cosmetic formulations due to growing consumer demand for “clean label” and green sustainable stabilizers [1]. Plant proteins and protein-rich by-products have particularly attracted attention for their functional amphiphilic properties, interfacial activity, and functional flexibility [2,3]. However, the application performance of such biopolymers depends critically on their conformation, chemical composition, surface charge, molecular size, and particle size distribution in dispersed phases or as stabilizer particles [4].
Sunflower meal (SFM), the largest by-product of oil extraction from sunflower seeds, is abundant but commercially underexploited, and typically used in animal feed. Yet, valorization of SFM as a functional ingredient in the production of emulsion systems could open new possibilities towards sustainable utilization [5]. Recent studies have shown that protein-enriched fractions of sunflower meal can successfully stabilize O/W emulsion, concerning relevant droplet sizes, creaming stability, and zeta potential [4]. Specifically, Bojanić et al. [4] reported that increasing the protein content from 16.8% to 42.6% in sunflower meal fractions led to a noticeable decrease in creaming and strongly affected droplet size and zeta potential values as well as droplet size distributions. The reported O/W emulsions stabilized by milled or non-milled SFM fractions stabilized droplet diameters in the range of 77–239 µm, indicating that particle size distribution played a substantial role in emulsion behavior.
Particle size distribution, particle shape, and inter-particle interactions can play significant roles in the stability of particle-stabilized emulsions [6]. Narrow and fine distributions often promote interfacial coverage and adsorption kinetics in production of O/W systems, leading to smaller droplets and stronger interfacial films [7]. Besides the impact of narrow particle size distribution, smaller particle sizes positively affect creaming stability since finer particles provide more efficient interfacial coverage and more intensive particle adsorption at the oil–water interface. As a result, droplet coalescence is reduced and upward migration during storage is hindered [8]. However, in mixtures of stabilizers including both particles and proteins, synergistic or antagonistic effects may occur depending on relative sizes, composition, and interactions [9,10].
In emulsion systems stabilized by using protein-based emulsifiers/stabilizers, the protein’s surface charge (zeta potential) and molecular flexibility greatly affect electrostatic repulsion, steric hindrance, and droplet resistance to coalescence. Proteins located on droplet surface may induce high absolute zeta potential (negative or positive) thus providing colloidal stability through electrostatic repulsion, while steric stabilization is achieved by protruding polymer chains on droplet surfaces. Sterically active protein layers around droplets reduce droplet coalescence and delay gravitational separation which improves emulsion creaming stability. The balance of the corresponding mechanisms depends mostly on particle size, macromolecular conformation, and continuous phase ionic strength [11].
Furthermore, besides the abovementioned aspects, another relevant factor is the stabilizer protein concentration and material aggregate state. When systems containing low amounts of protein are used, coverage of the oil–water interface may be incomplete, leading to larger droplets and promoted coalescence. With the increasing protein content, enhanced surface coverage, stronger interfacial films, and higher viscosity of the continuous phase are reported to improve emulsion stability in terms of both droplet size and emulsion creaming [12,13]. However, excessively high-protein levels or the presence of large insoluble particles in the emulsion system could lead to undesirable phenomena such as steric hindrance, bridging flocculation, or sedimentation.
Principle of obtaining protein-enriched fractions from sunflower meal is based on the difference between structural and mechanical properties of the particles originating from the core and from the hull of the sunflower kernel, where more elastic particles of the sunflower core are milled in greater extent comparing to particles of hull. Due to the difference in the milling patterns it is possible to separate the high-protein fraction, mainly constituted by the particles originating from the core of sunflower kernel, and the low-protein fraction, mainly constituted by the particles originating from the sunflower hull [14]. In this regard, adequate processing allows controlled design of PSD and protein content [15], which can further be correlated with emulsifying and stabilizing performance. However, conditions during the protein enrichment process must be considered, since milling parameters could influence protein physicochemical properties [16] and, consequently, protein functional properties.
Therefore, main objectives of this study were to prepare sunflower meal fractions with controlled particle size distributions and distinct protein levels (high and low), and to characterize their physicochemical properties in relation to their emulsion stabilizing performance. Furthermore, the stability of O/W emulsions evaluated through emulsion droplet size, zeta potential, and creaming behavior was investigated to understand the mechanisms governing the influence of particle size and protein content on emulsion formation and stability.
2. Materials and Methods
2.1. Sample Preparation
For the dispersed phase, sunflower oil (Victoriaoil, Šid, Serbia) obtained from a local market was used. The sunflower meal, produced as a by-product during oil extraction, was provided by Victoriaoil (Šid, Serbia). Sunflower meal was milled using a hammer mill (ABC Engineering, Pančevo, Serbia) and a laboratory roller mill (model S-150M) equipped with four corrugated rolls. The material was then sieved through a sieve with an aperture size of 630 µm to obtain a protein-enriched fraction, mainly consisting of endosperm particles, and a cellulose-rich fraction, primarily composed of cellulose particles. To achieve particle sizes smaller than 250 µm, both fractions were subsequently milled using a planetary ball mill PM 400 (Retsch GmbH, Haan, Germany). In the case of the fraction enriched with sunflower hulls, milling was assisted by liquid nitrogen, known as cryogenic milling, due to the physical properties of the sunflower hulls that make them difficult to grind [14]. The resulting finely milled material was subsequently fractionated and blended together in controlled proportions to obtain four protein-enriched fractions (each with a protein content of 30 ± 1%) with particle size ranges of 250/200, 200/125, 125/100, and <100 µm, as well as four cellulose-rich fractions (each with a protein content of 15 ± 1%) exhibiting the same particle size distribution (Table 1).

Table 1.
Experimental plan of sunflower meal fraction with different protein levels and particle size.
2.2. Chemical Characterization of Sunflower Meal
The chemical composition of sunflower meal fractions was analyzed using standard AOAC procedures for determining moisture and protein content [17].
2.3. Sunflower Meal Fractions Hydration Properties and Oil Binding Capacity
Water holding capacity (WHC, bound water without external force application) and binding capacity (WBC, bound water after external force application) were determined as hydration properties. In addition, oil binding capacity (OBC) was determined according to the procedure of [18]. WHC was determined according to the procedure of Raghavendra et al. [19] with slight modification. All measurements were conducted in duplicates.
An accurately weighed dry sample (0.5 g) was taken in a graduated test tube; around 15 mL of water was added and it was hydrated for 18 h. The supernatant was removed by passing through a sintered glass crucible under vacuum. The hydrated residue weight was recorded and it was dried at 105 °C for 2 h to obtain the residual dry weight. WHC was obtained by equation [19]:
For WBC measurements the sample was weighted (0.5 g) and taken in a graduated centrifuged tube. Around 15 mL of water was added and it was hydrated for 18 h, centrifuged (3000× g; 20 min), and the supernatant solution was removed by passing through a sintered glass crucible under an applied vacuum. The hydrated residue weight was recorded and then the sample was dried at 105 °C for 2 h to obtain dry weight. WBC was calculated as [19]:
For OBC measurements, a similar method was applied, with modification of replacement water with sunflower oil [18].
2.4. Emulsion Preparation
Aqueous continuous phase solutions were prepared 24 h prior to the emulsification process to ensure complete hydration of the material used. Following the experimental plan (Table 1), the required amount of sunflower meal (1% of the total emulsion mass) was calculated based on the total sample mass, weighed into double-distilled water, and mixed for 24 h. Consequently, the total dry matter content of the aqueous continuous phase in all experiments was maintained within the range of 0.93–0.94%, depending on the dry matter content of the sunflower meal. Before the addition of sunflower oil, the continuous phase was homogenized for 2 min at 6500 min−1 using an Ultra-Turrax T25 basic homogenizer equipped with an S 25 N-18 G dispersing tool (IKA-Werke GmbH & Co. KG, Staufen, Germany).
Sunflower oil-in-water emulsions were prepared using a one-step homogenization process, combining 10 wt% sunflower oil with 90 wt% aqueous continuous phase. The mixture was homogenized with an Ultra-Turrax T25 basic homogenizer equipped with an S 25 N-18 G dispersing tool for 5 min at 9500 min−1 under a constant temperature of 25 ± 0.1 °C. The oil-to-water ratio was chosen based on previous studies investigating similar systems. This 10:90 (oil/water) ratio was selected to better emphasize the stabilizing and emulsifying capacity of the applied stabilizer, as it allows greater sensitivity to variations in emulsion stability [20].
2.5. Emulsion Analysis
Droplet size and droplet size distribution were determined using the laser light scattering technique with a Mastersizer Hydro 2000 (Malvern Instruments, Malvern, UK). The particle size distribution of the ground sunflower meal samples was analyzed using a Mastersizer Scirocco 2000 analyzer (Malvern Instruments, Malvern, UK). The obtained results were expressed in terms of two dependent parameters: the volume-weighted mean diameter, also known as De Brouckere Mean Diameter (VD) (µm) (D4.3), and the volume median diameter, D(0.5) (µm). All measurements were conducted in duplicates.
For stability evaluation, emulsion samples were stored for seven days in 10 mL graduated glass cylinders at room temperature (25 °C), following the procedure described in the study by Maravić et al. [21]. Sodium propionate was added to inhibit microbial growth in the samples. The creaming behavior of the emulsions was monitored visually, and the extent of creaming was quantified using the creaming index, CI (%) calculated by equation:
where HC is the volume of the transparent serum layer at the bottom of the cylinder and HE is the total emulsion sample volume.
Zeta potential measurements were performed using a Zetasizer Nano ZS (Malvern Instruments, Malvern, UK). The analysis was carried out with a disposable folded capillary cell (model DTS1060). During the measurements, the refractive index and absorbance values were set as follows: for aqueous solutions containing SBP (Sodium Borosilicate Particles), the refractive index was 1.470 and the absorbance was 0.100; for sodium chloride aqueous solutions without SBP, the refractive index was adjusted to 1.544, with the absorbance maintained at 0.100. The zeta potential was calculated using the Smoluchowski approximation of Henry’s equation as the model. The reported values represent the mean of three independent measurements.
The main distinction between the two groups of sunflower meal fractions rises from their protein content, with protein-enriched fractions representing the endosperm-rich portion and cellulose-rich fractions corresponding to the hull-enriched material. The experimental setup, comprising protein-enriched and cellulose-rich fractions with defined particle size distributions, was designed to investigate how the particle size distribution and protein content of sunflower meal fractions correlate with their emulsifying and stabilizing behavior.
3. Results and Discussion
3.1. Sunflower Meal Fractions Hydration Properties and Oil Binding Capacity
The results of sunflower meal fractions hydration properties and oil binding capacity are presented in Table 2.

Table 2.
Hydration properties and oil binding capacity of sunflower meal fractions.
Based on the results presented in Table 2, it is evident that both particle size distribution and protein content influence WHC, WBC, and OBC. WHC ranged from 1.52 to 4.77 g/g which is in accordance with previous research [22].
In high-protein fractions (samples 1–4), WHC increased with the decrease in particle size sunflower meal fraction and range. Since the ratio of hydrophilic and hydrophobic groups and the exposure of hydrophilic groups in sunflower meal protein could be influenced by previous treatment [23], the observed trend could be attributed to the fact that in finer particles of sunflower meal (obtained by milling), increase in specific surfaces was followed with higher availability of hydrophilic protein groups that could attract water.
On the other hand, fractions of sunflower meal with lower-protein content (samples 5–8) expressed an overall higher capacity for holding water in comparison with samples 1–4, and a different trend regarding particle size. Fraction 250/200 µm expressed the highest water holding capacity among all samples. This could be due to a higher amount of fibrous particles originating from the sunflower hull in a particular fraction [24]. Those particles are not mechanically damaged, which could help to retain its fibrous structure, capable of mechanically holding water. Water binding capacity expressed a similar trend, but the measured values were lower, which could suggest that proteins do attribute to hydration properties, but not in such amounts as sample physical properties and its porosity do.
In the case of oil binding capacity (OBC), it was observed that fractions with larger particle sizes exhibited higher OBC values compared to finer fractions, regardless of protein content. The highest values were recorded for the 250/200 µm fractions (1.70–1.80 g/g), while the smallest particles showed lower values (0.79–1.61 g/g). The results are consistent with the previous research [25] and indicate that larger particles possess a more porous structure and a greater number of hydrophobic sites suitable for interaction with oil, which, as previously, could be attributed to the alteration of hydrophilic and hydrophobic groups as a consequence of processing treatment [23].
3.2. Emulsion Droplet Size
The values of the volume-weighted mean diameter (D[4.3]) are shown in Table 3, while the corresponding droplet size distributions of the examined emulsions are illustrated in Figure 1.

Table 3.
Volume-weighted mean diameter of obtained emulsion droplets.
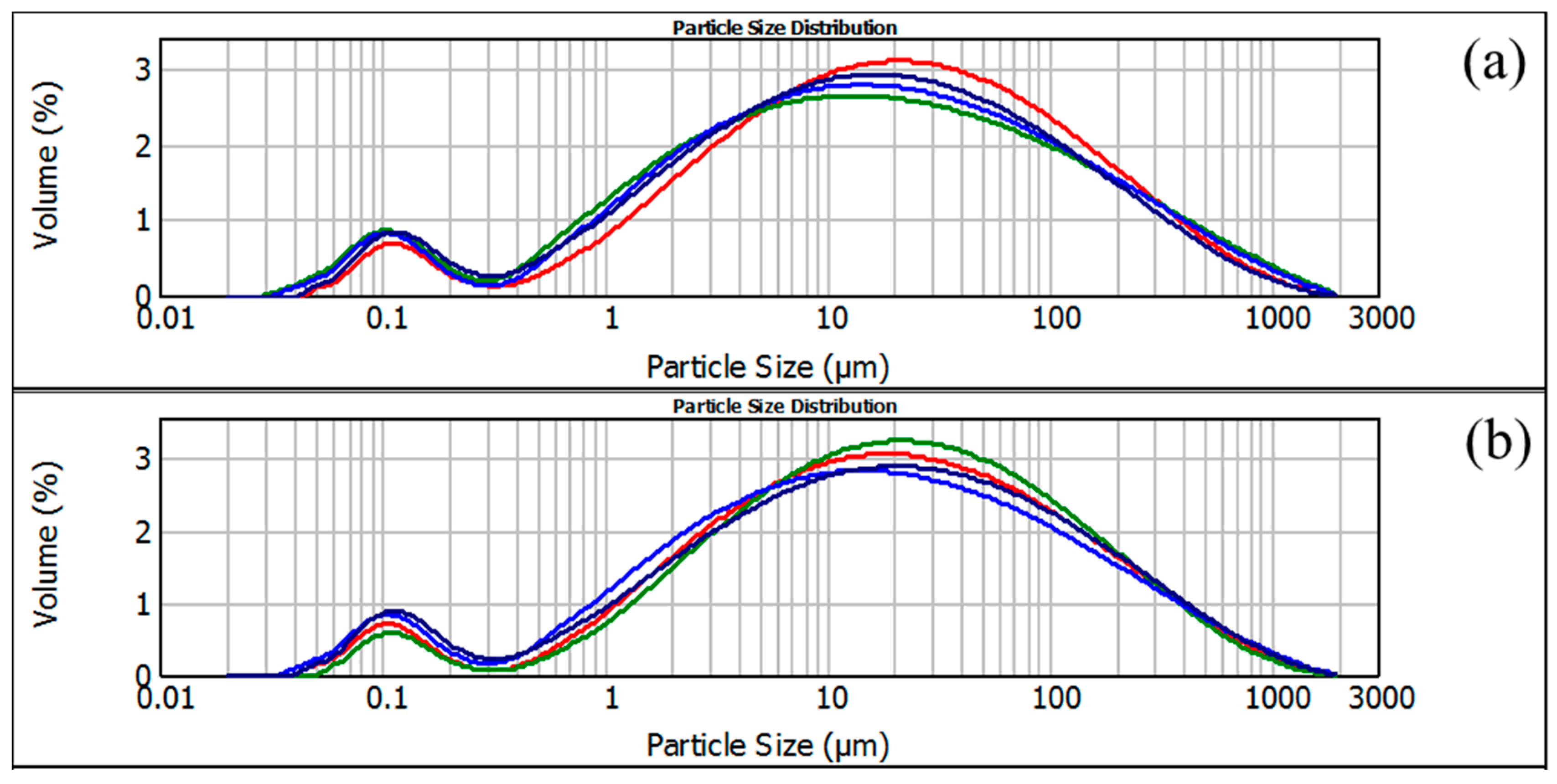
Figure 1.
Droplet size distribution of 15% (a) and 30% (b) protein content (red line fraction < 100 µm; green line fraction 125/100 µm; purple line 200/125 µm; blue line 250/200 µm).
The volume-weighted mean diameter (D[4.3]) of emulsion droplets for all investigated samples ranged between 66 and 81 µm (Figure 1). Within the group of fractions containing 15% protein, the smallest droplets were observed for Sample 5 (66.03 µm), while the mean droplet size gradually increased with decreasing particle size, reaching the highest value for Sample 7 (80.76 µm). A slight reduction in droplet size was noted again for the finest fraction (Sample 8).
In the sample group with 30% protein, droplet sizes varied in a narrower range (72.13–76.29 µm) without a clear increasing or decreasing trend. Overall, emulsions prepared using the protein-enriched fractions exhibited slightly smaller droplet sizes compared to those stabilized with the lower-protein fractions, indicating that protein content and particle size both influenced droplet formation during homogenization.
Even though tested sunflower meal fractions contained equal total protein content (15%), production of specific particle sizes through the milling process strongly affected the amount of protein available to adsorb at the oil–water interface. According to the obtained results, particle fractions in the range 250/200 µm provided optimal surface accessibility and moderate protein solubility, which enabled protein molecule migration towards the oil–water interface, quickly stabilizing the smallest droplets obtained by homogenization process. As sunflower meal particle size decreased (Samples 6–7), present proteins became more entangled within dense matrices of sunflower meal or partially denatured due to higher surface exposure and physical effects of mechanical treatment during applied ball milling process [25]. Consequently, less protein was available (soluble) in the prepared continuous phase and therefore poorer interfacial coverage was obtained. Furthermore, smaller interfacial coverage of oil droplets strongly contributed to the coalescence of smaller droplets resulting in the increase in D[4.3] values. However, sunflower meal particle fractions with sizes smaller than 100 µm showed two opposite effects. Firstly, smaller particles with corresponding sizes may act as Pickering stabilizers of emulsion droplets, by physically adsorbing at the oil–water interface due to increased surface area which allows far more contact sites. On the other side, negative effect of relatively small particle sizes could cause particle aggregation or increased viscosity of continuous phase. Nevertheless, the finest fraction (<100 µm) contained a small proportion of micron particles formed during intense milling which fragments of protein–fiber complexes could strongly attach to the droplet interface and act as Pickering particles, contributing to the improved stability and slightly smaller droplet size [4,26].
The corresponding effects are not as important when sunflower meal fractions containing 30% protein were used. Increasing protein content in tested fractions from 15% to 30% led to overall smaller droplets, indicating improved emulsification/stabilization efficiency at higher-protein content. Higher-protein content effects the stabilization process through higher accessibility of interfacially active material which could occupy the new oil–water interface and prevent instant recoalescence. Better droplet surface coverage along with higher continuous phase viscosity (due to the presence of more soluble material) reduced droplet–droplet collision frequency and slowed coalescence during and right after the homogenization process. Therefore, the obtained results suggested that limited availability of interfacially active material governed the droplet stabilization process and showed no evident significant dependence on the particle size fraction. This indicates that above the threshold of sufficient protein content, all tested fractions provided enough surface-active material (soluble proteins and/or fine protein–fiber particles) to stabilize droplets during homogenization, hence the emulsification outcome becomes less particle size-dependent and more dominated by processing conditions and chemical content [27].
3.3. Emulsion Zeta Potential
The results of zeta potential measurements for the examined emulsions are presented in Table 4, illustrating the potential for both short- and long-term stability of the investigated systems.

Table 4.
Zeta potential of obtained emulsions.
The measured potentials ranged from −10.73 to −16.97 mV. Samples containing 30% protein (Samples 1–4) exhibited values between −10.73 and −16.97 mV, with the highest surface charge recorded for Sample 2 (−16.97 mV). The group with 15% protein (Samples 5–8) showed slightly more uniform values, ranging from −11.67 to −15.53 mV. The results of zeta potential measurements did not clearly indicate that protein content was the only factor influencing the surface charge of the emulsions. A higher zeta potential was observed for the higher-protein sample only in the fraction with particle sizes of 200/125 µm (Sample 2 vs. Sample 6). Additionally, it is worth mentioning the observed consistent inverse relationship between emulsion droplet size and zeta potential across all paired fractions. For each particle size range, the fraction with higher-protein content exhibited larger droplets but lower zeta potential values compared to the corresponding lower-protein fraction, and vice versa.
All zeta potential values obtained suggested moderate to low stability of produced emulsions (values ranging from −10 mV to 17 mV) which was also confirmed through creaming values. In general, emulsion samples with zeta potential values above ±30 mV are considered electrostatically stable, while values below ±10 mV reflect low stability [1,28]. The highest value of zeta potential, concerning samples with 15% protein content, was measured for Sample 5 where droplet size was the smallest (D[4.3] 66.03 µm). Relatively high net charge of droplets, obtained as a result of adsorbed sunflower meal fraction to the interface, improved droplet electrostatic repulsion and slowed the coalescence process of produced droplets. As the particle size of sunflower meal fraction used for emulsion stabilization decreased, zeta potential value also decreased, negatively affecting the ability of droplets to repulse each other. Furthermore, small zeta potential values suggested that droplets are forming aggregates and, as the results measured, droplet size is increasing. The corresponding effect is in correlation with the previously discussed availability and functionality of the present proteins after the applied ball milling process [25]. Further decrease in sunflower meal particle sizes confirmed the previous assumption of the combined effect of fine protein–fiber particles and residual soluble proteins which enhanced the interfacial charge (ζ = −14.33 mV compared to −11.67 mV for the coarser fraction), thereby increasing electrostatic repulsion between droplets, reducing droplet tendency to coalesce and finally resulting in a slightly smaller mean droplet size (D[4.3] 73.81 µm).
Emulsions produced by using sunflower meal fractions containing 30% protein did not show clear correlation between droplet size and zeta potential, (in contrast to 15% protein experiments, where a clear inverse relationship between zeta potential magnitude and droplet size was noticed) thus indicating that electrostatic stabilization was not the most dominant factor governing droplet formation. It can be therefore concluded that interfacial charge played a more pronounced role under lower-protein availability.
Furthermore, both the results (droplet sizes and zeta potential values) suggested higher-protein content increased interfacial saturation (higher amount of protein) and steric stabilization became sufficient to prevent extensive coalescence during the process of homogenization, so droplet size becomes controlled primarily by processing conditions rather than by droplet electrostatic repulsion alone. Therefore, measured zeta potential better addressed the surface charge of the formed emulsion, rather than possibilities of smaller droplet production and coalescence during the process of homogenization [29]. Furthermore, it can be concluded that the prevailing emulsion stabilization mechanism was highly dependent on the interfacial protein availability, shifting from predominantly electrostatic repulsion at lower-protein levels (15%) to steric stabilization as protein concentration increased (30%) and interfacial saturation was approached.
3.4. Emulsion Creaming Index
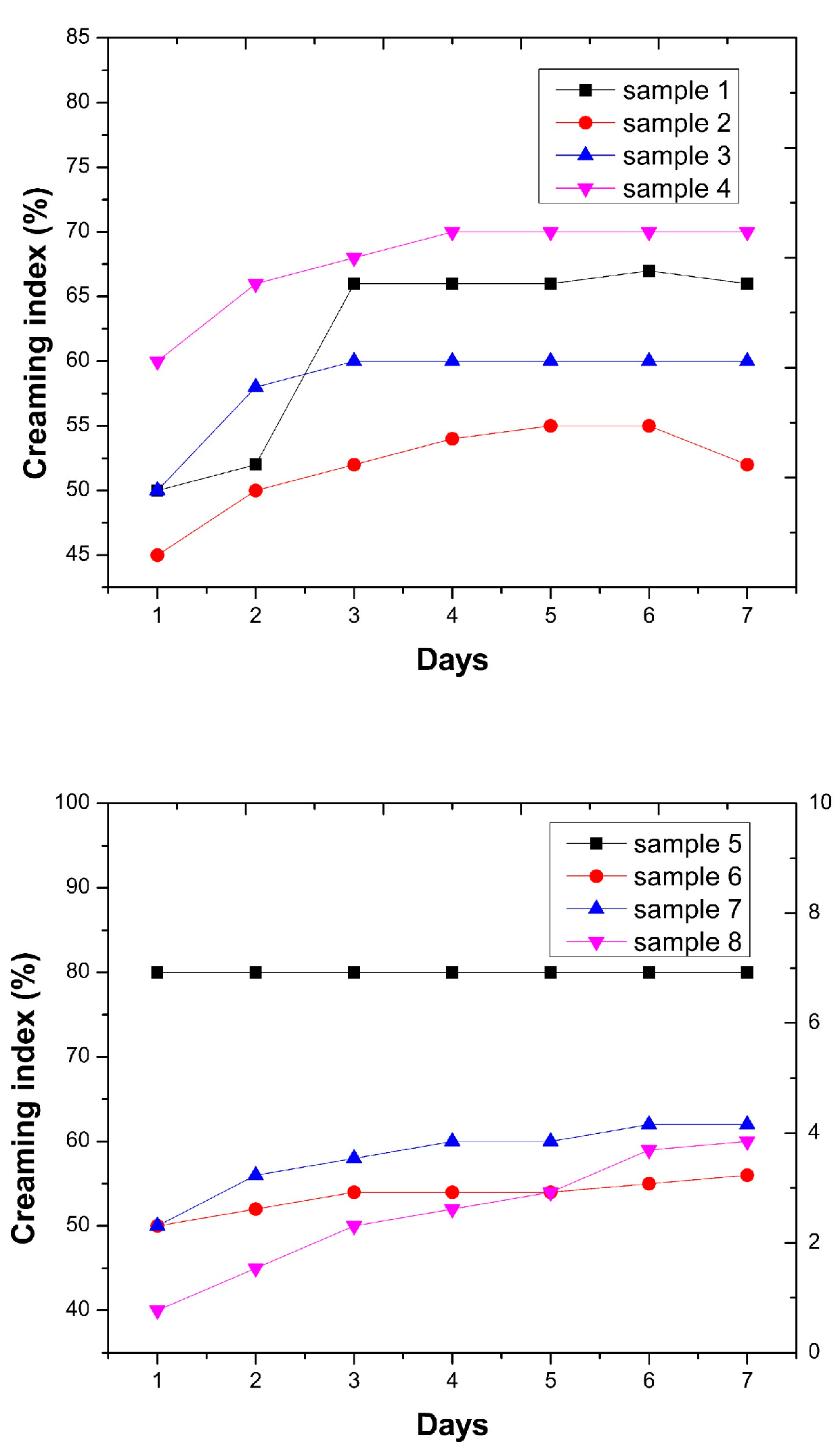
Creaming index values of the investigated emulsions are presented in Figure 2. The results showed that all samples exhibited a gradual increase in creaming index during the storage period, followed by a tendency toward stabilization at later days. For emulsions stabilized with sunflower meal fractions containing 15% protein (Samples 5–8), the highest CI values were observed for the fraction with the largest particle size (Sample 5), while smaller fractions (Samples 6–8) exhibited evidently lower CI values.
Figure 2.
Creaming index of high- and low-protein content emulsions.
For the emulsions prepared with sunflower meal fractions containing 30% protein (Samples 1–4), the fraction with the smallest particle size (Sample 4) exhibited the highest CI values (60–70%), while Sample 1 showed the lowest stability during the seven-day storage period, with its creaming index increasing from 50% to 66%.
When comparing the creaming results of emulsions stabilized with fractions of the same particle size but different protein contents, the best overall performance was observed for the fractions with particle sizes of 200/125 µm (Sample 2 and Sample 6). The fraction containing 15% protein (Sample 6: 50–56%) showed slightly higher stability, as the increase in creaming index over time was less pronounced, while the fraction with 30% protein (Sample 2: 45–52%) exhibited somewhat lower CI values. For the fractions with particle sizes of 125/100 µm, very similar CI values were obtained (Sample 7: 50–62% and Sample 3: 50–60%). However, the sample containing 30% protein (Sample 3) demonstrated slightly better stability, as its CI value increased from 50% to 58% by the second day, reached 60% on the third day, and remained unchanged until the end of the seven-day storage period.
Creaming index values of samples containing 15% of protein strongly depended on the particle size fraction used. Sample 5 (sunflower meal fraction containing particle diameters from 250 to 200 µm) showed the best physical stability with the creaming index values remaining almost constant (around 80%), indicating negligible separation of cream or serum layer. However, the corresponding sample cream layer was relatively small which could be attributed to the low amounts of oil (10%) used in the preparation of emulsions [21]. Samples 6–8 showed a constant increase in the serum layer height starting from around 40% and reaching 60–65% after seven days, which indicates a more pronounced separation of the emulsion cream layer. The corresponding trends correlate very closely with the previously observed differences in droplet size and zeta potential of the obtained emulsions. The smallest droplets (66 µm) and the most negative zeta potential (−15.5 mV) are measured in the emulsion where the sunflower meal fraction contains particle diameters from 250 to 200 µm correlating with the observed creaming values of Sample 5. Enhanced electrostatic repulsion reduced droplet aggregation and subsequent upward migration. Samples 6–7, with larger droplets and smaller zeta potential values showed faster creaming, due to weaker repulsion and larger droplets, and also phase separation (phase separation observed after 4 days, while phase separation was not observed in Sample 5 in the observed period of 7 days) on top of the measuring vessels. However, the sunflower meal fraction containing particles smaller than 100 µm, showed a slight improvement due to assumed partial Pickering-type stabilization by micron particles and a modest increase in measured zeta potential value (−14.3 mV).
Furthermore, protein content showed strong influence on creaming values. In Figure 2 is evident that increasing the protein content to 30% markedly improved emulsion physical stability. During the total observed period of seven days, creaming index values remained within a narrow range (≈50–70%) for all tested fractions, with minor variations between samples. It can be assumed that since creaming rate was controlled mainly by gravitational effects and continuous phase viscosity, steric stabilization provided by thick protein films on emulsion droplets and dispersed fine protein–fiber particles in continuous phase was sufficient to prevent extensive separation of cream and serum layer. As previously reported by Bojanić et al. [4], the creaming index values were lower in emulsions stabilized using sunflower meal fractions having higher-protein content indicating the positive impact of protein content on creaming index.
4. Conclusions
The effects of protein content and different particle sizes of sunflower meal fractions on oil-in-water emulsion droplet sizes and stability were investigated. At a sunflower meal fraction containing 15% of protein, emulsions showed clear dependence on particle size, where fractions of 250/200 µm produced the smallest emulsion droplets and the largest zeta potential values which resulted in relatively high stability. Reduction in particle size decreased protein solubility and interfacial availability, leading to larger emulsion droplets and higher creaming index values due to weaker electrostatic repulsion. The specific effect of finest fraction (<100 µm) was noticed assuming Pickering-type stabilization by micron protein–fiber particles. Emulsions stabilized by sunflower meal fractions containing 30% of protein displayed narrower mean droplet size distribution in tested samples (≈72–76 µm) and no clear correlation with obtained zeta potential values. The higher-protein concentration ensured higher interfacial saturation and enhanced droplet steric stabilization, preventing intensive coalescence during homogenization. The dominant emulsion stabilization mechanism strongly depended on the amount of protein available for interfacial adsorption and shifted from electrostatic to steric stabilization as the protein content increased. It can be concluded that both protein content and particle size of sunflower meal fractions strongly influence the emulsification and stabilization behavior of oil-in-water emulsions. The presented results highlight specific emulsification performance of sunflower meal fractions, providing a basis for cost-effective, plant-based emulsifiers in industrial formulations.
Author Contributions
Conceptualization, S.V., N.M. and N.B.; Methodology, S.V., N.M. and N.B.; Software, S.V.; Validation, S.V.; Formal analysis, S.V.; Investigation, S.V. and N.B.; Resources, Z.Š.; Data curation, N.M.; Writing—original draft, S.V., N.M. and N.B.; Writing—review & editing, Z.Š., A.F. and N.B.; Supervision, A.F.; Project administration, Z.Š.; Funding acquisition, Z.Š. and A.F. All authors have read and agreed to the published version of the manuscript.
Funding
Financial support for this research was provided by the Ministry of Science, Technological Development and Innovations of the Republic of Serbia, with grants No. 451-03-136/2025-03/200134, No. 451-03-137/2025-03/200134, and No. 451-03-136/2025-03/200222.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- McClements, D.J. Food Emulsions: Principles, Practices, and Techniques; CRC press: Boca Raton, FL, USA, 2015. [Google Scholar] [CrossRef]
- Ozturk, B.; Mcclements, D.J. Progress in natural emulsifiers for utilization in food emulsions. Curr. Opin. Food Sci. 2016, 7, 1–6. [Google Scholar] [CrossRef]
- Kim, W.; Wang, Y.; Selomulya, C. Trends in Food Science & Technology Dairy and plant proteins as natural food emulsifiers. Trends Food Sci. Technol. 2020, 105, 261–272. [Google Scholar] [CrossRef]
- Bojanić, N.; Vidosavljević, S.; Fišteš, A.; Šereš, Z.; Fodor, E.; Maravić, N. Emulsion stabilising capacity of sunflower meal depending on fraction protein content and particle size. Int. J. Food Sci. Technol. 2024, 59, 333–342. [Google Scholar] [CrossRef]
- Hadidi, M.; Aghababaei, F.; Julian, D. Food Hydrocolloids Sunflower meal/cake as a sustainable protein source for global food demand: Towards a zero-hunger world. Food Hydrocoll. 2024, 147, 109329. [Google Scholar] [CrossRef]
- Katepalli, H.; John, V.T.; Tripathi, A.; Bose, A. Microstructure and rheology of particle stabilized emulsions: Effects of particle shape and inter-particle interactions. J. Colloid. Interface Sci. 2017, 485, 11–17. [Google Scholar] [CrossRef]
- Venkataramani, D.; Tsulaia, A.; Amin, S. Fundamentals and applications of particle stabilized emulsions in cosmetic formulations. Adv. Colloid. Interface Sci. 2020, 283, 102234. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Xiong, L.; Li, M.; Liu, J.; Yang, J.; Chang, R.; Liang, C.; Sun, Q. Characterizations of Pickering emulsions stabilized by starch nanoparticles: Influence of starch variety and particle size. Food Chem. 2017, 234, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Tambe, D.E.; Shaftma, M.M. The effect of colloidal particles on fluid-fluid interfacial properties and emulsion stability. Adv. Colloid. Interface Sci. 1994, 52, 1–63. [Google Scholar] [CrossRef]
- Du, F.; Qi, Y.; Huang, H.; Wang, P.; Xu, X.; Yang, Z. Stabilization of O/W emulsions via interfacial protein concentrating induced by thermodynamic incompatibility between sarcoplasmic proteins and xanthan gum. Food Hydrocoll. 2022, 124, 107242. [Google Scholar] [CrossRef]
- Dickinson, E. Interfacial structure and stability of food emulsions as affected by protein–polysaccharide interactions. Soft Matter. 2008, 4, 932–942. [Google Scholar] [CrossRef]
- Roullet, M.; Clegg, P.S.; Frith, W.J. Viscosity of protein-stabilised emulsions: Contributions of components and development of a semi-predictive model. J. Rheol. 2019, 63, 179–190. [Google Scholar] [CrossRef]
- Hinderink, E.B.; de Ruiter, J.; de Leeuw, J.; Schroen, K.; Sagis, L.M.; Berton-Carabin, C.C. Early film formation in protein-stabilised emulsions: Insights from a microfluidic approach. Food Hydrocoll. 2021, 118, 106785. [Google Scholar] [CrossRef]
- Vidosavljević, S.; Bojanić, N.; Ilić, P.; Rakić, D.; Đuragić, O.; Banjac, V.; Fišteš, A. Optimization of Grinding Process of Sunflower Meal for Obtaining Protein-Enriched Fractions. Processes 2022, 10, 2704. [Google Scholar] [CrossRef]
- Murru, M.; Calvo, C.L. Sunflower protein enrichment. Methods and potential applications. OCL 2020, 27, 17. [Google Scholar] [CrossRef]
- Liu, B.; Wang, H.; Hu, T.; Zhang, P.; Zhang, Z.; Pan, S.; Hu, H. Ball-milling changed the physicochemical properties of SPI and its cold-set gels. J. Food Eng. 2017, 195, 158–165. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Đorđević, M.; Đorđević, M.; Maravić, N.; Teofilović, V.; Šoronja-Simović, D.; Šereš, Z. Processing of alfalfa seeds by convective hot air drying, vacuum drying and germination: Proximate composition, techno-functional, thermal and structural properties evaluation. Food Chem. 2023, 402, 134300. [Google Scholar] [CrossRef]
- Raghavendra, S.N.; Rastogi, N.K.; Raghavarao, K.S.M.S.; Tharanathan, R.N. Dietary fiber from coconut residue: Effects of different treatments and particle size on the hydration properties. Eur. Food Res. Technol. 2004, 218, 563–567. [Google Scholar] [CrossRef]
- Zang, X.; Wang, J.; Yu, G.; Cheng, J. Addition of anionic polysaccharides to improve the stability of rice bran protein hydrolysate-stabilized emulsions. LWT 2019, 111, 573–581. [Google Scholar] [CrossRef]
- Maravić, N.; Šereš, Z.; Nikolić, I.; Dokić, P.; Kertész, S.; Dokić, L.J. Emulsion stabilizing capacity of sugar beet fi bers compared to sugar beet pectin and octenyl succinate modified maltodextrin in the production of O/W emulsions: Individual and combined impact. LWT 2019, 108, 392–399. [Google Scholar] [CrossRef]
- Bárta, J.; Bártová, V.; Jarošová, M.; Švajner, J.; Smetana, P.; Kadlec, J.; Filip, V.; Kyselka, J.; Bercíková, M.; Zdráhal, Z.; et al. Oilseed Cake Flour Composition, Functional Properties Species Differences. Foods 2021, 10, 2766. [Google Scholar] [CrossRef]
- Qin, N.; Nie, J.; Hou, Y.; Shuang, Q.; Bao, X. Ultrasound-assisted macroporous resin treatment improves the color and functional properties of sunflower meal protein. Ultrason. Sonochem. 2024, 102, 106750. [Google Scholar] [CrossRef] [PubMed]
- Petraru, A.; Ursachi, F.; Amariei, S. Nutritional characteristics assessment of sunflower seeds, oil and cake. Perspective of using sunflower oilcakes as a functional ingredient. Plants 2021, 10, 2487. [Google Scholar] [CrossRef]
- Li, S.; Liu, Z.; Hei, X.; Wu, C.; Ma, X.; Hu, H.; Jiao, B.; Zhu, J.; Adhikari, B.; Wang, Q.; et al. Effect of Physical Modifications on Physicochemical and Functional Properties of Walnut Protein. Foods 2023, 12, 3709. [Google Scholar] [CrossRef]
- Chen, L.; Ao, F.; Ge, X.; Shen, W. Food-Grade Pickering Emulsions: Preparation, Stabilization and Applications. Molecules 2020, 25, 3202. [Google Scholar] [CrossRef]
- Kasprzak, M.M.; Jarzębski, M.; Smułek, W.; Berski, W.; Zając, M.; Östbring, K.; Ahlström, C.; Ptasznik, S.; Domagała, J. Effects of concentration and type of lipids on the droplet size, encapsulation, colour and viscosity in the oil-in-water emulsions stabilised by rapeseed protein. Foods 2023, 12, 2288. [Google Scholar] [CrossRef] [PubMed]
- Tadros, T.F. Emulsion Formation, Stability, and Rheology. In Emulsion Formation and Stability; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 1–75. [Google Scholar]
- Ma, J.; Pan, C.; Chen, H.; Chen, W.; Pei, J.; Zhang, M.; Zhong, Q.; Chen, W.; Zeng, G. Effects of protein concentration, ionic strength, and heat treatment on the interfacial and emulsifying properties of coconut (Cocos nucifera L.) globulins. Food Chem. X 2023, 20, 100984. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).