Abstract
Three Schiff base fluorescent probes 3a–3c with N-heterocyclic structure were designed and synthesized by using the reaction of 4-diethylaminosalicylaldehyde with different N-heterocyclic amines, such as 2-aminobenzimidazole, 2-aminobenzothiazole, and 2-amino-6-methylpyridine. Compound 3a exhibited excellent selectivity towards Hg2+, with a detection limit of 3.21 × 10−7 M and a response time of only 30 s. It could be used as a fluorescent probe for detecting Hg2+. Meanwhile, compounds 3b and 3c exhibited excellent selectivity towards Zn2+, with detection limits of 1.61 × 10−7 M and 2.03 × 10−7 M, respectively, and response times of only 30 s. They could serve as fluorescent probes for detecting Zn2+. Using probe 3a for Hg2+ as an example, the detecting mechanism was further elucidated through 1H NMR, ESI-MS testing, and DFT calculation analysis. For compound 3a, the coordination stoichiometry between compound 3a and Hg2+ was verified to be 1:1 through a Job’s plot. After coordination with Hg2+, the molecular rigidity of compound 3a was enhanced, which inhibited the non-radiative decay process and led to the closure of the excited-state intramolecular proton transfer (ESIPT) effect. At the same time, the fluorescence intensity was significantly increased through the chelation-enhanced fluorescence (CHEF) mechanism, which was confirmed by density functional theory (DFT) calculations. In addition, compounds 3a–3c were successfully applied in practical water samples and test strips for the detection of Hg2+/Zn2+.
1. Introduction
Metal ions play different roles in various fields such as industry, medicine, and ecology [1]. While playing a role, there exists an optimal level of metal ion content in the environment. When there is an imbalance at this level, it may lead to serious pollution and harm [2]. Therefore, the selective and quantitative detection of metals is required [3]. Typically, mercury, as a highly toxic heavy metal pollutant, can be released into the environment through industrial production, polluting soil, water, and food and posing a serious threat to human health [4]. Therefore, it is crucial to develop a reliable and highly sensitive sensor to detect the presence of Zn2+ [5] and mercury ions (Hg2+) [6] in the environment.
Similarly, zinc ion (Zn2+) is one of the essential trace elements in the human body, which is crucial in biological functions such as DNA synthesis, neural signal transmission, cell apoptosis, and the regulation of multiple ion channels [7]. Meanwhile, abnormal levels of Zn2+ may lead to the disruption of corresponding biochemical processes in the body. For example, excessive intake of Zn2+ may lead to anemia, epilepsy, and Alzheimer’s disease; the lack of Zn2+ can cause phenomena such as growth arrest and diarrhea [8]. In addition, due to the widespread use of zinc in industrial production such as alloy manufacturing, battery production, and chemical industry, Zn2+, as a common industrial waste, also accumulates in the environment and causes serious water and soil pollution [9].
Due to the advantages of low synthesis cost, high sensitivity, and fast response time, fluorescent probes have been widely used in the detection of Zn2+ and Hg2+ in recent years [10,11]. Although there have been many reports on the detection of Zn2+ and Hg2+, most probes currently suffer from poor sensitivity, strong background fluorescence interference, and poor stability due to aggregation-caused quenching (ACQ), which limits their detection applications in actual samples [12,13,14]. On the contrary, the probes with aggregation-induced emission (AIE) often exhibit high photostability, good biocompatibility, and high quantum yield, solving the limitations of traditional fluorescent probes with ACQ [15,16].
In addition, excited-state intramolecular proton transfer (ESIPT) is an important mechanism for designing fluorescent probes [17,18,19]. Probes with ESIPT mechanisms often rely on intramolecular hydrogen bonding to induce fluorescence changes. When metal ions bind with hydroxyl groups, the ESIPT mechanism is turned off, and the fluorescence of the probe changes, making it suitable for detecting metal ions [20,21]. Therefore, designing Zn2+ and Hg2+ fluorescent probes with AIE and ESIPT is highly meaningful [22,23]. Given the promising performance of certain Schiff bases in metal ion detection [24], it is of significant research value to explore probes with combined ESIPT and AIE characteristics based on readily accessible Schiff base compounds for the highly selective and sensitive detection of Hg2+ and Zn2+ while also investigating their sensing mechanisms and application potential.
Based on our previous research on the synthesis of AIE-functional molecules [25] and ESIPT probes [26], we synthesized three Schiff base fluorescent probes 3a–3c (Scheme 1) by reacting 4-diethylaminosalicylaldehyde with various N-heterocyclic amines, including 2-aminobenzimidazole, 2-aminobenzothiazole, and 2-amino-6-methyl-pyridine. The test results showed that compounds 3a–3c have both AIE and ESIPT characteristics. Compound 3a can detect Hg2+, while compounds 3b and 3c can detect Zn2+ with lower detection limits in a shorter response time based on a turn-on ESIPT–CHEF mechanism.
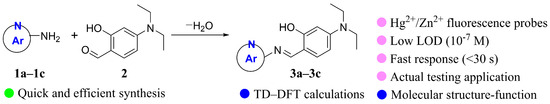
Scheme 1.
Synthesis of probes 3a–3c.
2. Results and Discussion
2.1. Design, Synthesis and Characterization of Compounds 3a–3c
The D-π-A structure refers to a molecular architecture composed of an electron donor (D), a conjugated bridge (π), and an electron acceptor (A) [27]. Owing to their advantages such as high fluorescence quantum yields, strong intramolecular charge transfer (ICT) capabilities, and tunable absorption/emission wavelengths, D-π-A type molecules are widely used in fluorescent sensors [28,29,30]. In the molecules designed herein, an N,N-diethylamino group as an electron donor is located on one side of the chromophore to extend the molecular conjugation [31]. An imine group and a benzene ring act as the conjugated bridge, while a nitrogen-containing heterocyclic fluorophore (benzimidazole/benzothiazole/pyridine moiety) on the other side serves as the electron acceptor.
Some D-π-A type molecules exhibit enhanced fluorescence in aggregated states or restricted environments due to the suppression of non-radiative transitions, a phenomenon known as aggregation-induced emission (AIE) [32]. Meanwhile, the Schiff base derivatives of salicylaldehyde often possess excited-state intramolecular proton transfer (ESIPT) characteristics [33]. Therefore, we constructed three Schiff base probes featuring both AIE and ESIPT effects by utilizing the D-π-A structure to provide the AIE effect and the proton transfer between the imine group and the phenolic hydroxyl group to facilitate the ESIPT effect.
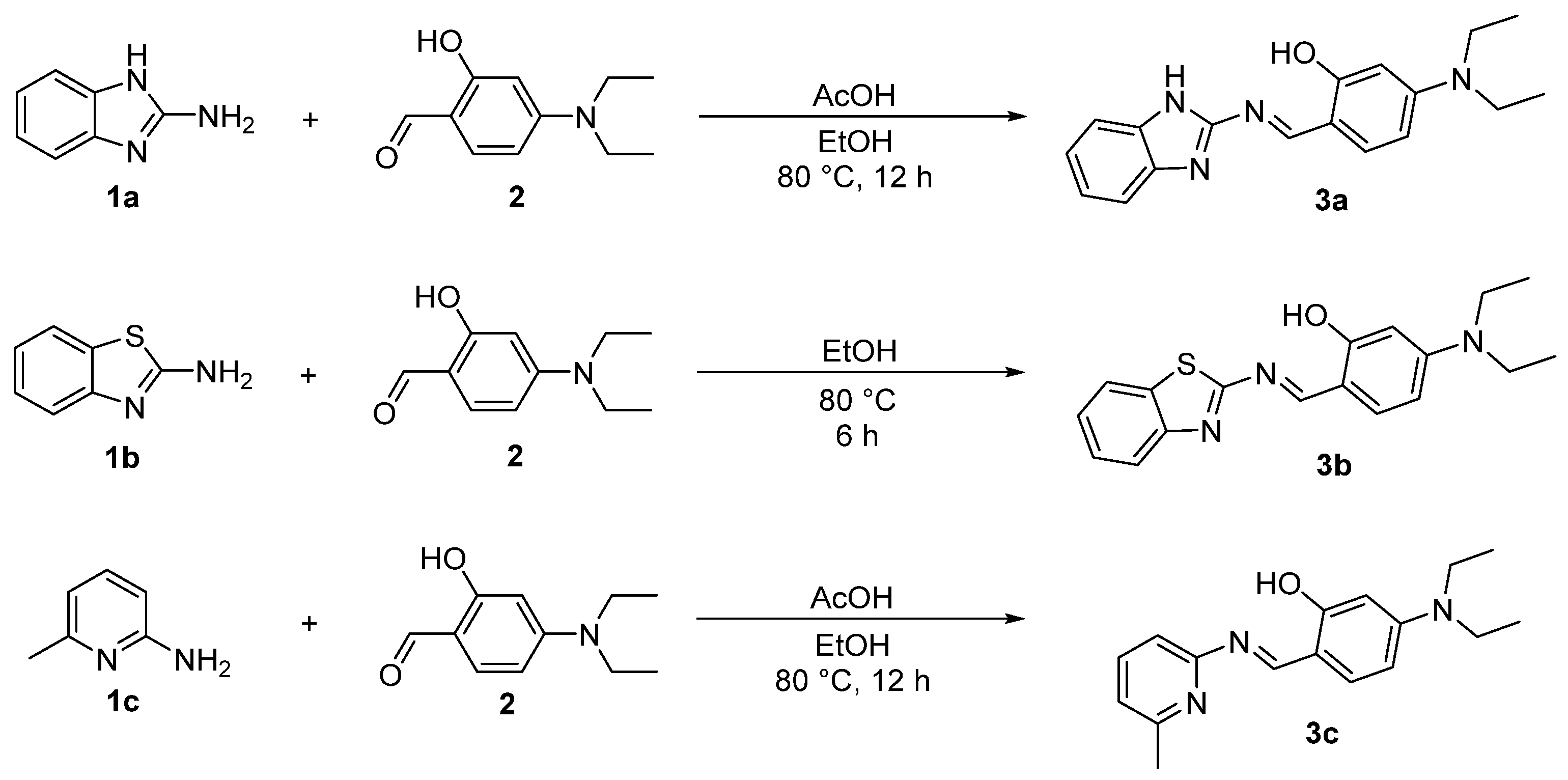
The Schiff base reaction is typically conducted under weakly alkaline conditions, as excessively low or high pH values can adversely affect its efficiency and product yield [34]. In addition to all substrates possessing a primary amino group, the imidazole structure in 1a and the pyridine structure in 1c impart the stronger basicity to the overall molecules. In contrast, for 1b, the electron-withdrawing nature of the benzothiazole stabilizes the conjugated system of the entire molecule, resulting in weaker basicity. Furthermore, the tertiary amino group of 4-diethylaminosalicylaldehyde also contributes to the basicity of the molecule. Consequently, this allows for the direct synthesis of compound 3b in absolute ethanol, whereas the synthesis of compounds 3a and 3c requires the addition of acetic acid as a catalyst [35] (Scheme 2).
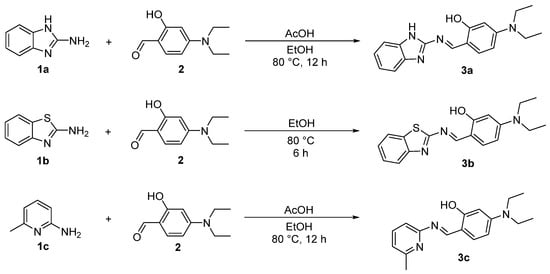
Scheme 2.
Synthesis of compounds 3a–3c.
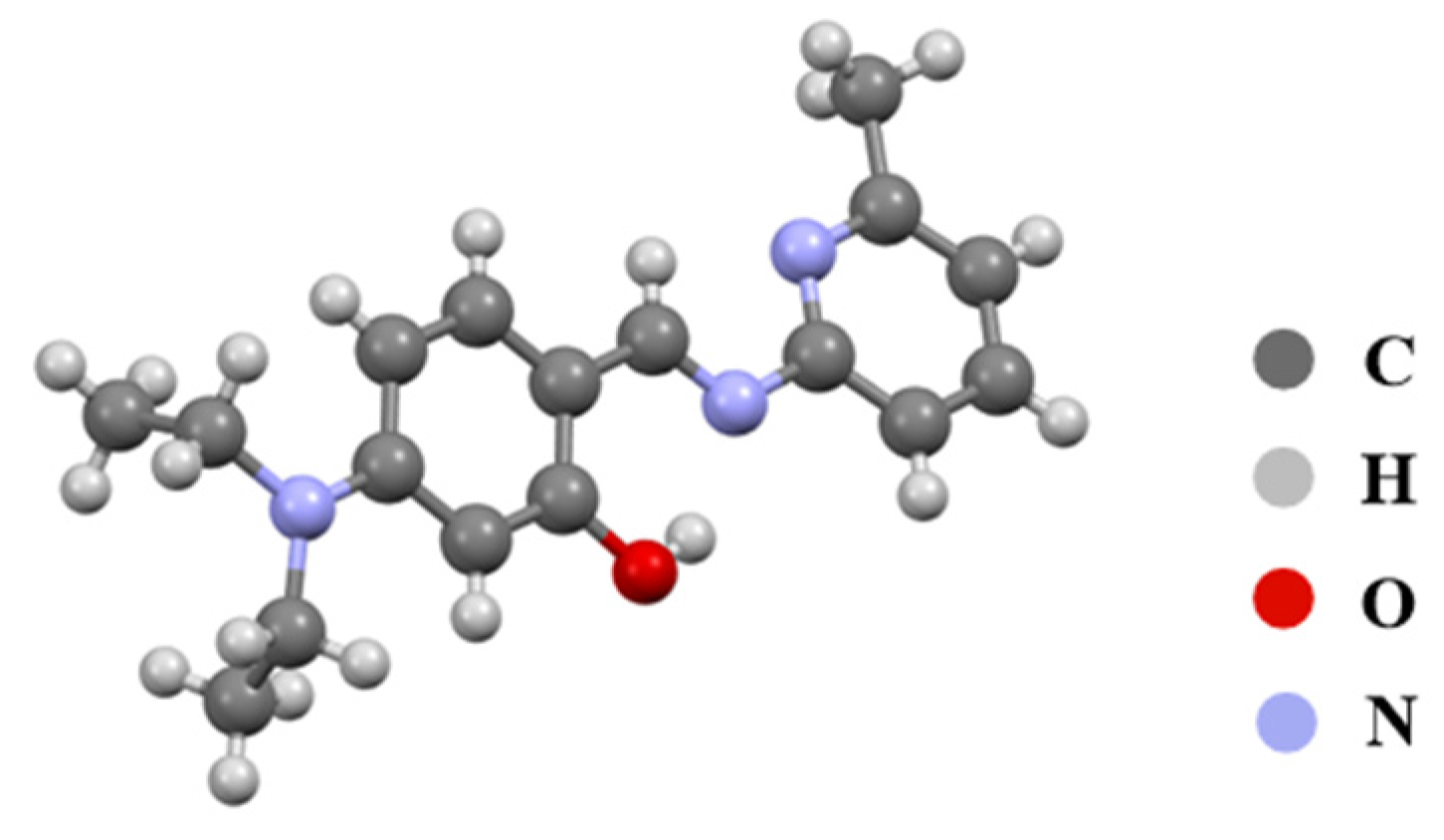
The 1H NMR and 13C NMR spectra of the synthesized compounds revealed that all characteristic signals for compounds 3a–3c appeared at the expected chemical shifts. Additionally, the structure of new compound 3c was further confirmed by HRMS and X-ray single-crystal diffraction (Table S1 and Figure 1). In short, the various characterization data confirmed that the synthesized compounds are indeed the target molecules.
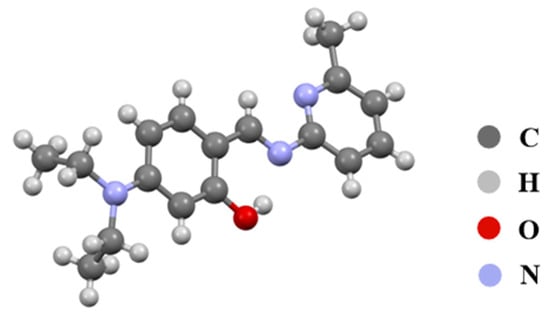
Figure 1.
Crystal structure of compound 3c.
2.2. Theoretical Calculation Studies of Compounds 3a–3c
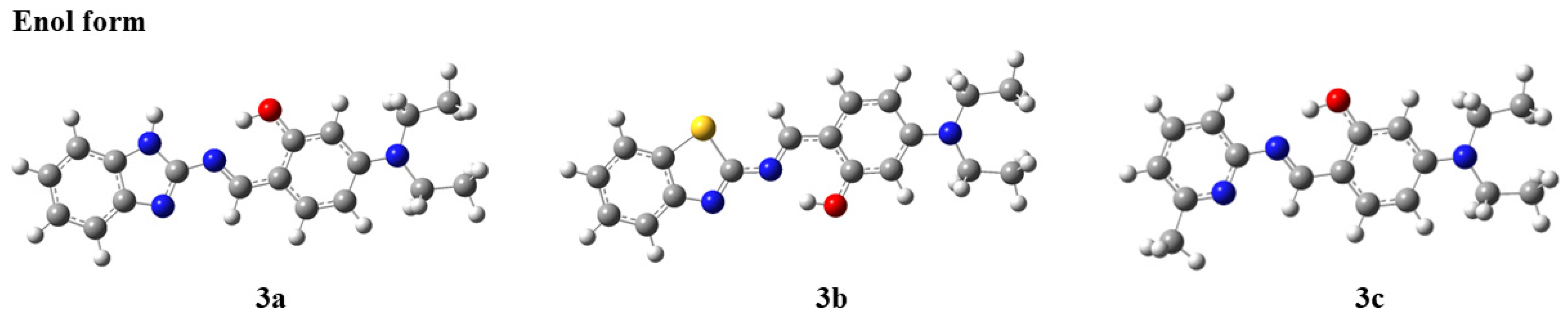
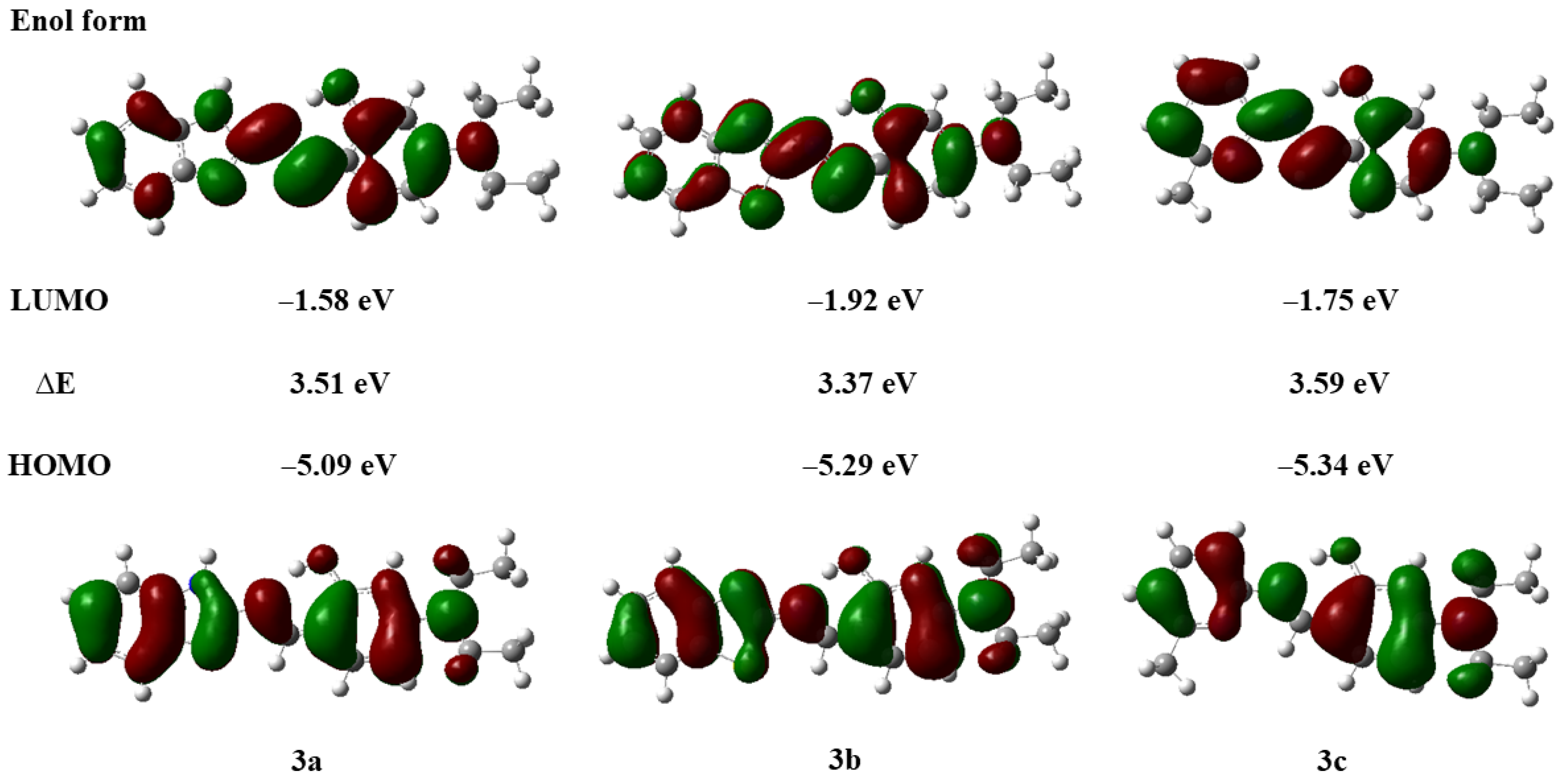
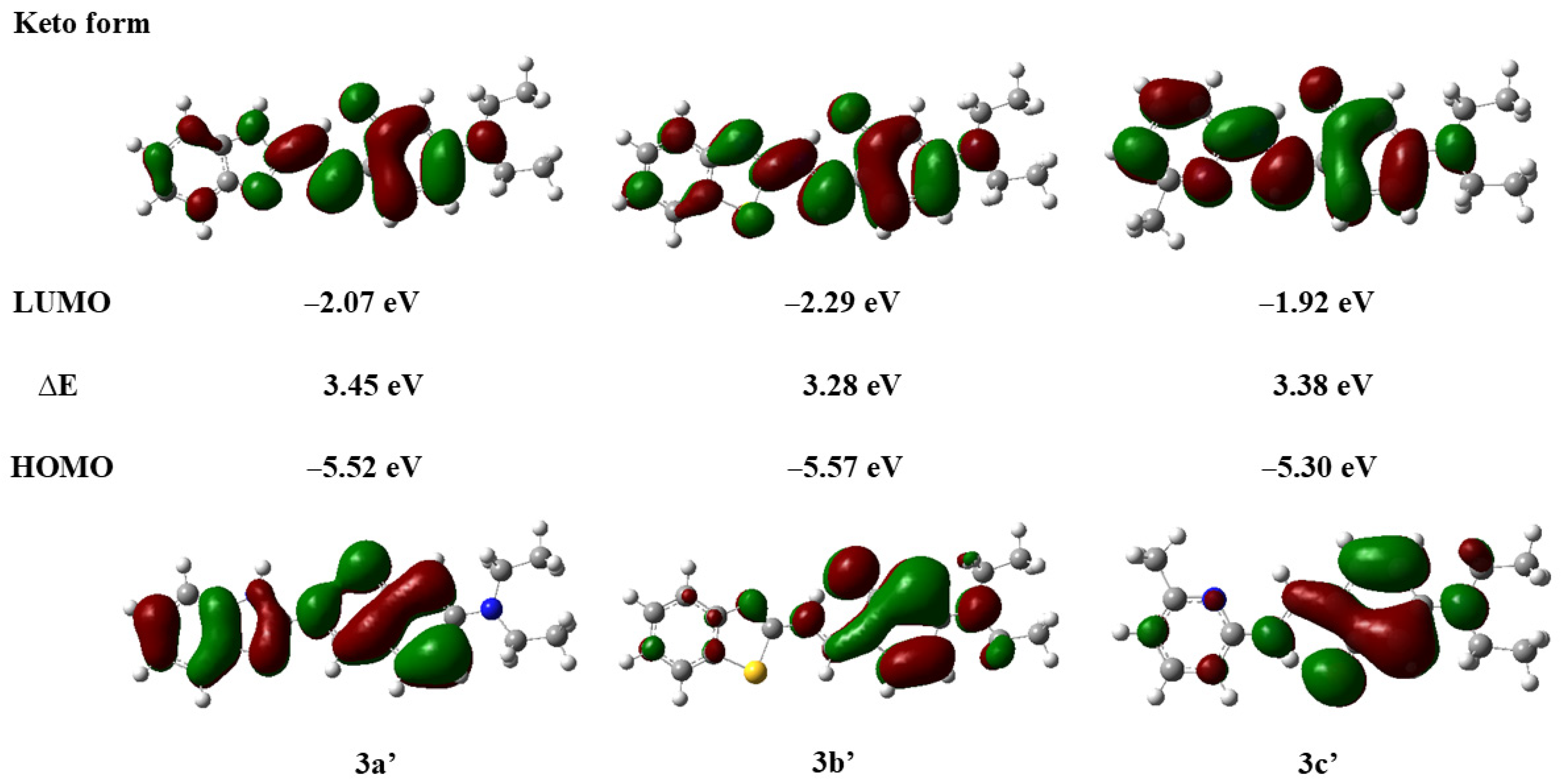
Compounds with ESIPT effects typically exist as keto and enol isomers [36]. To demonstrate the ESIPT effect of compounds 3a–3c, we performed time-dependent density functional theory (TD-DFT) calculations for compounds 3a–3c and their keto tautomers using the Gaussian 09 program at the B3LYP/6-31G(d,p) level of theory, following the literature methods [37,38]. The optimized molecular structures and the corresponding frontier molecular orbitals are shown in Figure 2 and Figure 3 (enol form) and Figure 4 and Figure 5 (keto form), respectively.
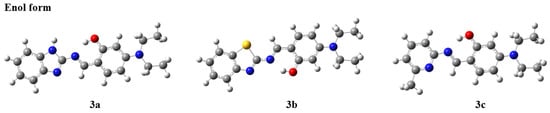
Figure 2.
Optimized molecular structures of compounds 3a–3c (enol form).
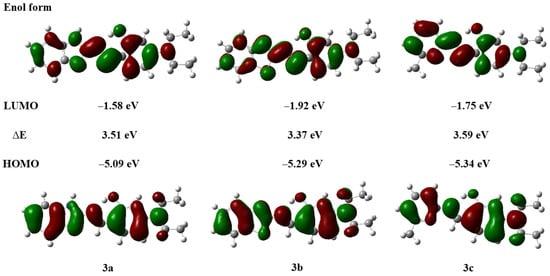
Figure 3.
Frontier molecular orbitals and energy levels of compounds 3a–3c (enol form).
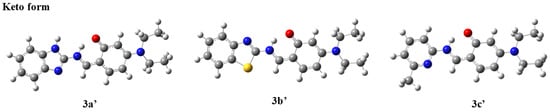
Figure 4.
Optimized molecular structures of compounds 3a′–3c′ (keto form).
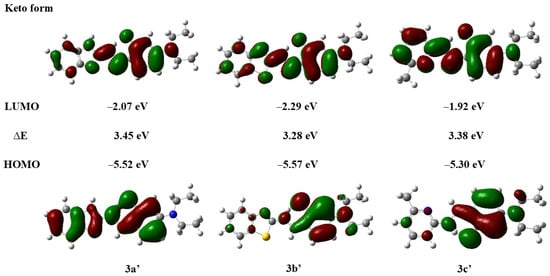
Figure 5.
Frontier molecular orbitals and energy levels of compounds 3a′–3c′ (keto form).
As shown in Figure 2, after geometry optimization, all molecules of compounds 3a–3c exhibit a nearly planar structure, which is conducive to molecular aggregation. Meanwhile, as can be seen from Figure 3, there is a significant overlap in the electron cloud densities of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) for compounds 3a–3c. These orbitals are primarily distributed over the nitrogen-containing heterocyclic fluorophore and the salicylaldehyde moiety, suggesting that compounds 3a–3c may possess good fluorescence properties [39].
As depicted in Figure 4, the optimized molecular structures of the keto isomers of compounds 3a–3c also maintain a nearly planar conformation. Furthermore, we calculated the energy states derived from the HOMO and LUMO of the keto isomers of compounds 3a–3c (Figure 5). It can be observed that the LUMO energy level of the enol form is higher than that of the keto form. This indicates that in the excited state, the proton of the phenolic hydroxyl group in the enol isomer of compounds 3a–3c can be readily transferred to the nitrogen atom of the imine group, forming the more stable keto isomer. Concurrently, the energy gaps of the keto isomers (∆E′3a = 3.45 eV, ∆E′3b = 3.38 eV, ∆E′3c = 3.38 eV) are smaller than those of the enol isomers (∆E3a = 3.51 eV, ∆E3b = 3.37 eV, ∆E3c = 3.59 eV), which further lays the foundation for the occurrence of the ESIPT effect in compounds 3a–3c [39].
2.3. Photophysical Properties of Compounds 3a–3c
2.3.1. Effect of Different Solvents on the Optical Properties of Probes 3a–3c
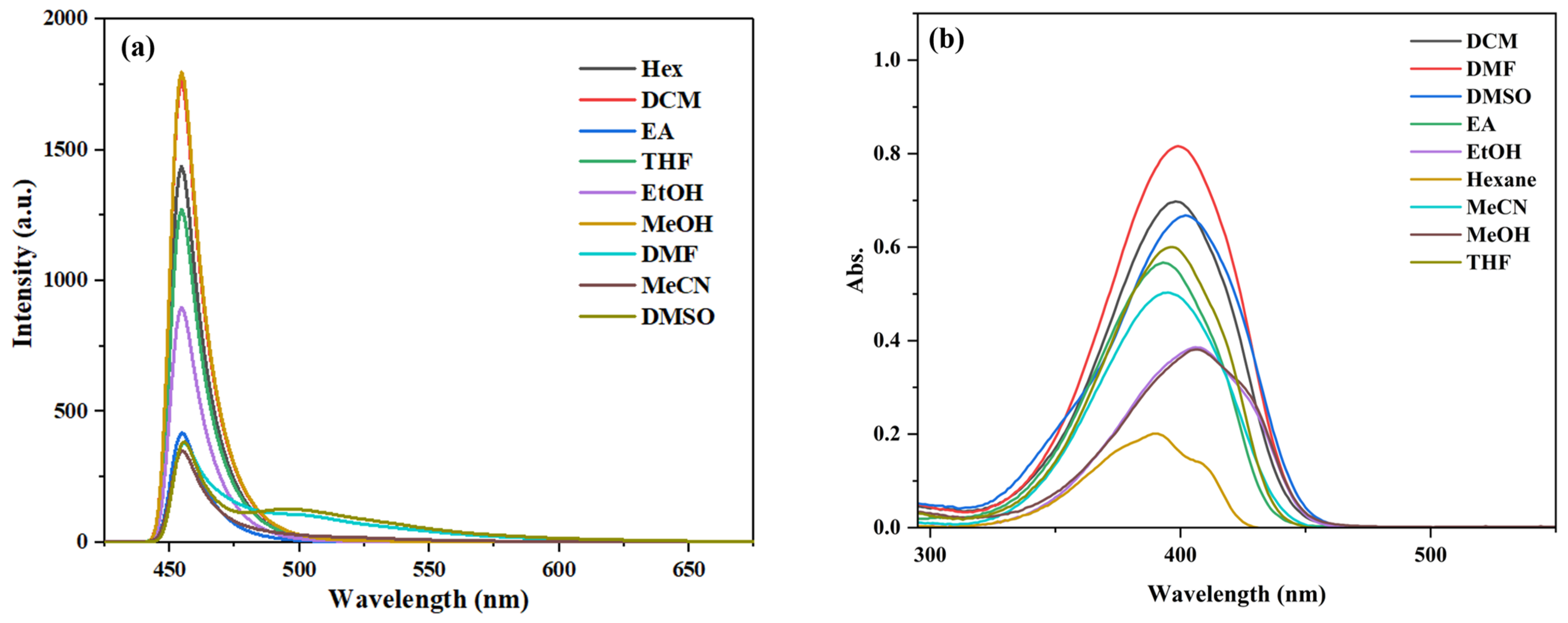
The TD-DFT calculation results suggest that compounds 3a–3c exhibit an ESIPT effect. To further investigate this effect, their fluorescence emission spectra and UV–vis absorption spectra in various solvents were studied (Figure 6, Figures S8 and S9).
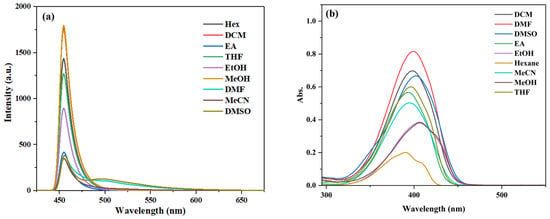
Figure 6.
(a) Fluorescence spectra and (b) UV–vis absorption spectra of compound 3c (10 μM) in different solvents.
As shown in Figure S8, compound 3a displays dual emission bands (enol emission and keto emission) in aprotic solvents, such as tetrahydrofuran (THF), dichloromethane (DCM), acetonitrile (MeCN), dimethylformamide (DMF), and dimethyl sulfoxide (DMSO). Similarly, compound 3b exhibits dual emission bands in solvents of lower polarity (DCM, EA) and aprotic solvents (THF, DCM, MeCN, DMF, and DMSO) (Figure S9). Probe 3c only shows dual emission bands in DMF and DMSO (Figure 6), which is characteristic of a typical ESIPT process [39].
In addition, a noticeable difference in fluorescence intensity was observed between methanol and ethanol for compounds 3a–3c, both being polar protic solvents. This variation may be attributed to the slightly higher polarity of methanol, which can suppress the excited-state intramolecular proton transfer (ESIPT) process, consequently leading to differences in fluorescence intensity [40].
Compounds 3a and 3c show the highest fluorescence intensity in CH3OH, while compound 3b has the highest intensity in DMSO. The characteristic fluorescence values of compounds 3a–3b are summarized in Table S2 of the Supplementary Materials. As shown in Table S2, compound 3a exhibits the largest stokes shift of 89 nm, while compounds 3b and 3c display smaller stokes shifts, with 3c having the smallest among them.
Therefore, to obtain better luminescence intensity and emission wavelengths, the AIE properties of 3a and 3c were studied in CH3OH, and those of 3b were studied in DMSO.
2.3.2. AIE Properties of Compounds 3a–3c
We investigated the fluorescence properties of compounds 3a–3c in solvent systems with varying ratios of organic solvent and water by using fluorescence emission spectroscopy. The fluorescence spectra are shown in Figure 7, Figures S10 and S11.
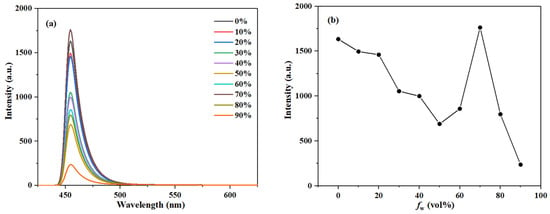
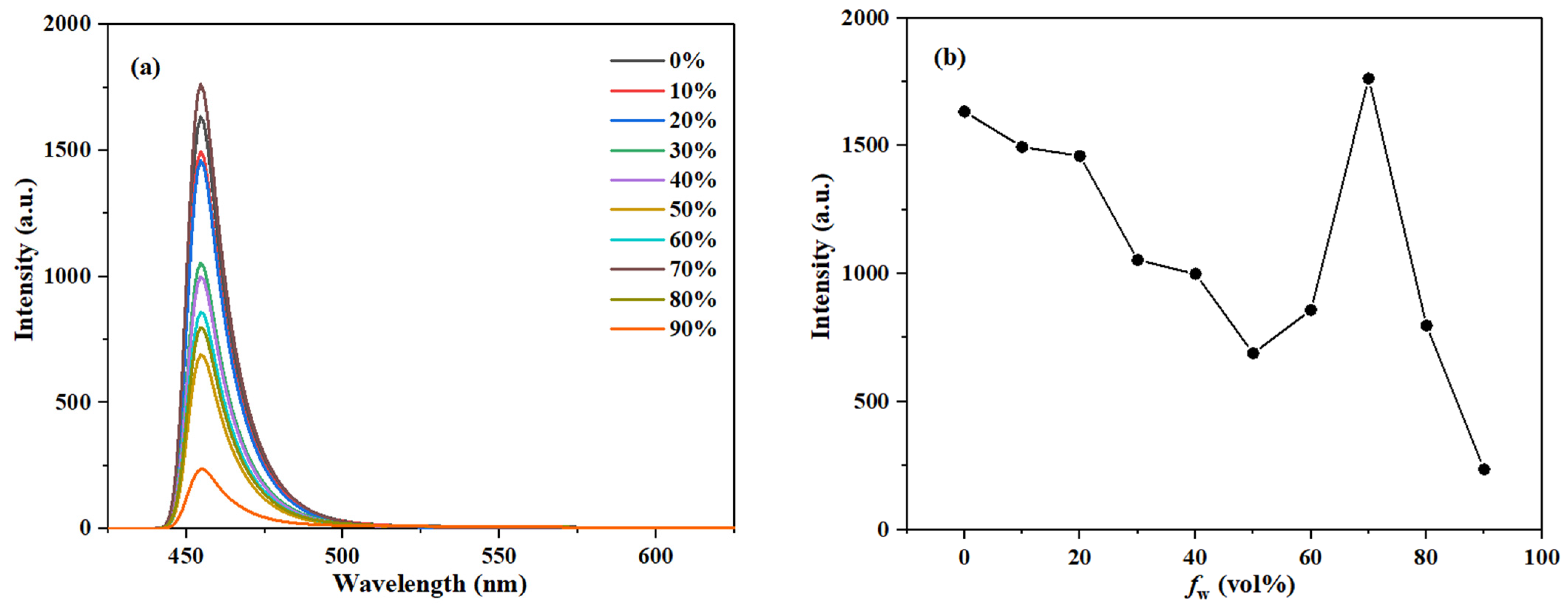
Figure 7.
(a) The fluorescence spectra of compound 3c (10 μM) and (b) plot of emission peak intensity in CH3OH/H2O systems with different water fraction (0–90% by volume).
As the water fraction gradually increased, the fluorescence emission peak of compound 3a first decreased, then increased, and finally decreased again, reaching its maximum intensity at a water fraction of 70% (Figure S10). Furthermore, the short-wavelength emission peak of compound 3a changed significantly with the water fraction, while the long-wavelength peak showed little change, indicating that the enol form of 3a is the predominantly existing species in polar protic solvents. Similarly, the fluorescence emission peak of compound 3c also shows a trend of first decreasing, then increasing, and then decreasing again, with the maximum intensity observed at a 70% water fraction (Figure 7).
For compound 3b, as the water fraction gradually increased, its fluorescence emission peak at 455 nm first decreased, then increased, and then decreased, reaching a maximum intensity at a water fraction of 50%. The emission peak at 503 nm first increased, then decreased, and then increased again, reaching its maximum intensity at a water fraction of 90% (Figure S11).
These results suggest that compounds 3a–3c likely aggregate with increasing water fractions, which restricts intramolecular rotation and intermolecular interactions, leading to the AIE effect and a consequent enhancement in fluorescence intensity [17]. Compared with our group’s previous studies [26], the current work incorporates a diethylamino functional group in the design. Thus, in the dispersed state, the flexible alkyl chains within this group undergo intramolecular rotation and vibration, effectively promoting the non-radiative energy decay and resulting in low fluorescence intensity [19]. While in the aggregated state, these motions are significantly restricted, thereby suppressing non-radiative decay channels and leading to remarkable fluorescence enhancement, which demonstrates distinct AIE characteristics as designed.
2.4. Detection of Metal Ions by Probes 3a–3c
2.4.1. Selectivity Studies of Probes 3a–3c for Metal Ions
Selectivity is a crucial factor in evaluating the performance of a fluorescent probe for practical applications [41]. Through simple experimental tests, it was found that the detection effects of compounds 3a and 3c are not significant in methanol as the solvent (Figures S12 and S13). However, in DMF, compound 3a could clearly detect Hg2+ (Figure S12); in DMSO, compound 3c could clearly detect Zn2+ (Figure S13).
Therefore, considering the aforementioned AIE properties, we further investigated the fluorescence emission spectra of compounds 3a–3c (10 μM) with various metal ions (100 μM) in DMF/H2O (v/v = 3:7), DMSO/H2O (v/v = 5:5), and DMSO/H2O (v/v = 3:7) solvent systems, respectively (Figure 8, Figure 9 and Figure S14). The tested metal ions included Na+, Mg2+, Al3+, K+, Ca2+, Mn2+, Cd2+, Cr3+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Hg2+, Pb2+, and Fe3+.
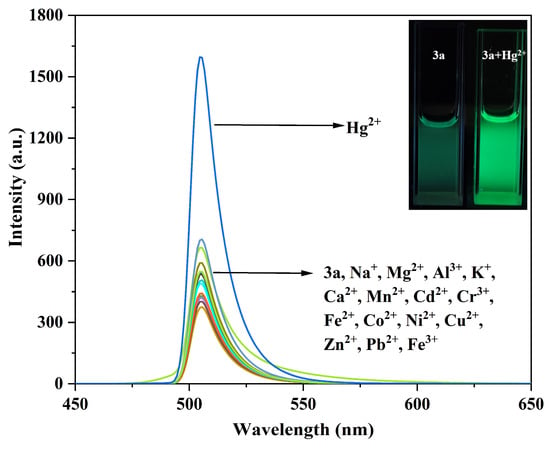
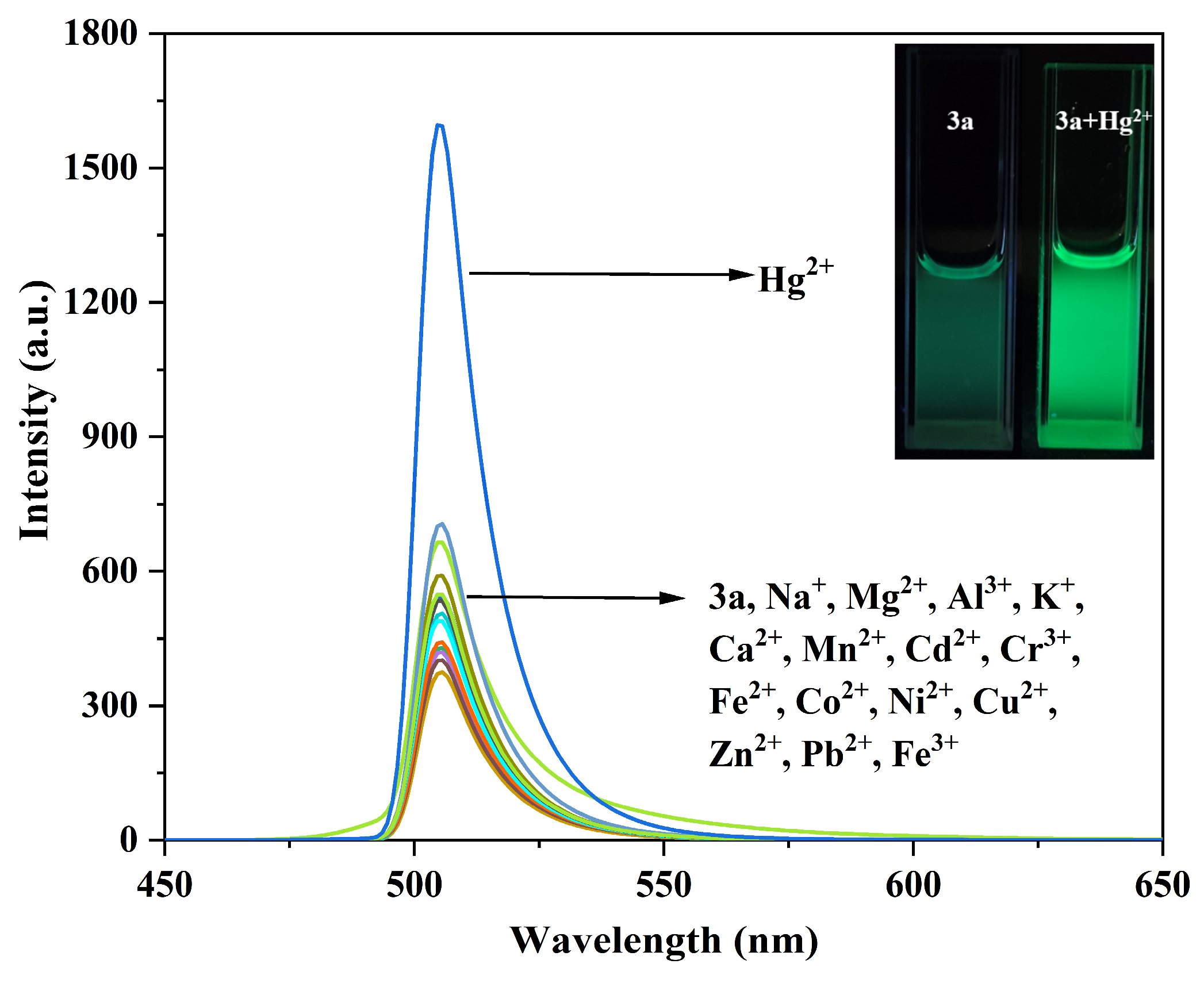
Figure 8.
Fluorescence emission spectra of compound 3a (10 μM) before and after interaction with different metal ions. The inset shows the fluorescence changes of compound 3a before and after adding Hg2+.
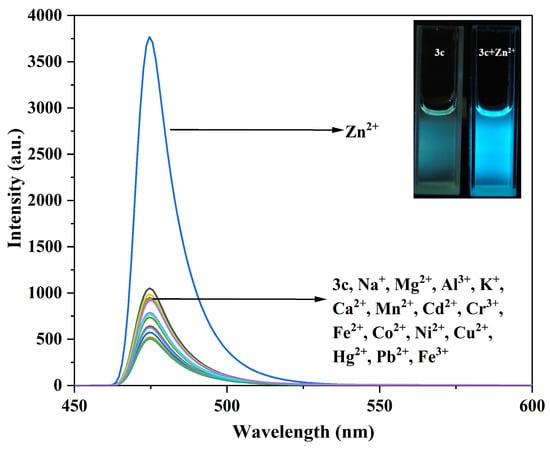
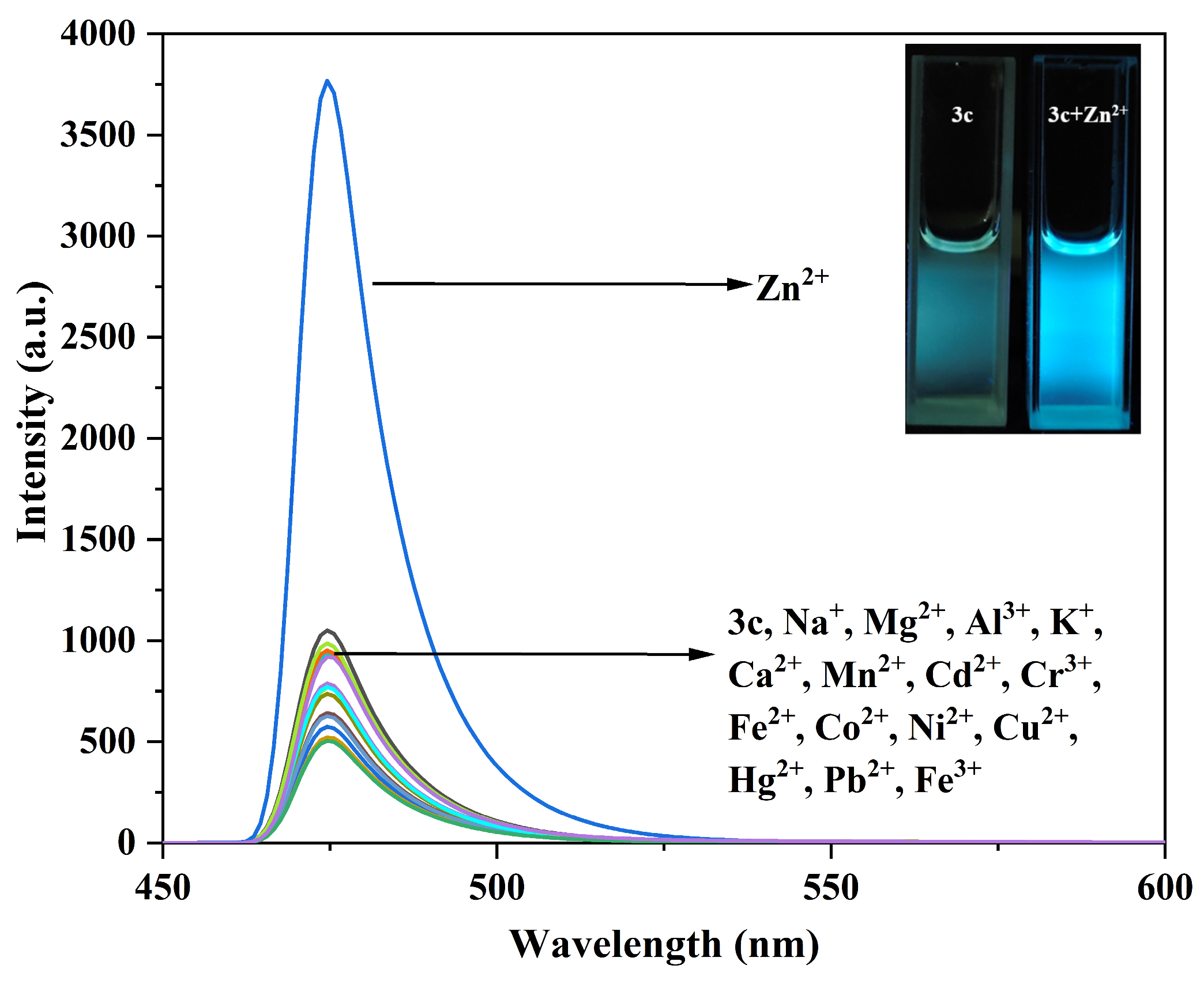
Figure 9.
Fluorescence emission spectra of compound 3c (10 μM) before and after interaction with different metal ions. The inset shows the fluorescence changes of compound 3c before and after adding Zn2+.
As shown in Figure 8, it is observed that other metal ions caused almost no spectral changes to compound 3a, whereas the addition of Hg2+ led to a significant enhancement of the fluorescence emission peak at 505 nm, with the fluorescence color changing to bright green.
Similarly, only the addition of Zn2+ to the solution of compound 3b enhanced the fluorescence intensity at 515 nm, changing the fluorescence color from blue-green to yellow-green (Figure S14). For compound 3c, only the addition of Zn2+ enhanced the fluorescence intensity at 474 nm, resulting in a bright blue color (Figure 9).
This is likely because the coordination of compounds 3a–3c with the metal ions enhances the structural rigidity of the molecules while simultaneously suppressing C=N isomerization and the ESIPT process. Furthermore, the fluorescence responses of compounds 3a–3c to other metal ions were negligible, indicating that compound 3a can serve as a promising “turn-on” probe specific to Hg2+, while compounds 3b and 3c can act as “turn-on” probes specific to Zn2+.
2.4.2. Anti-Interference Studies of Probes 3a–3c
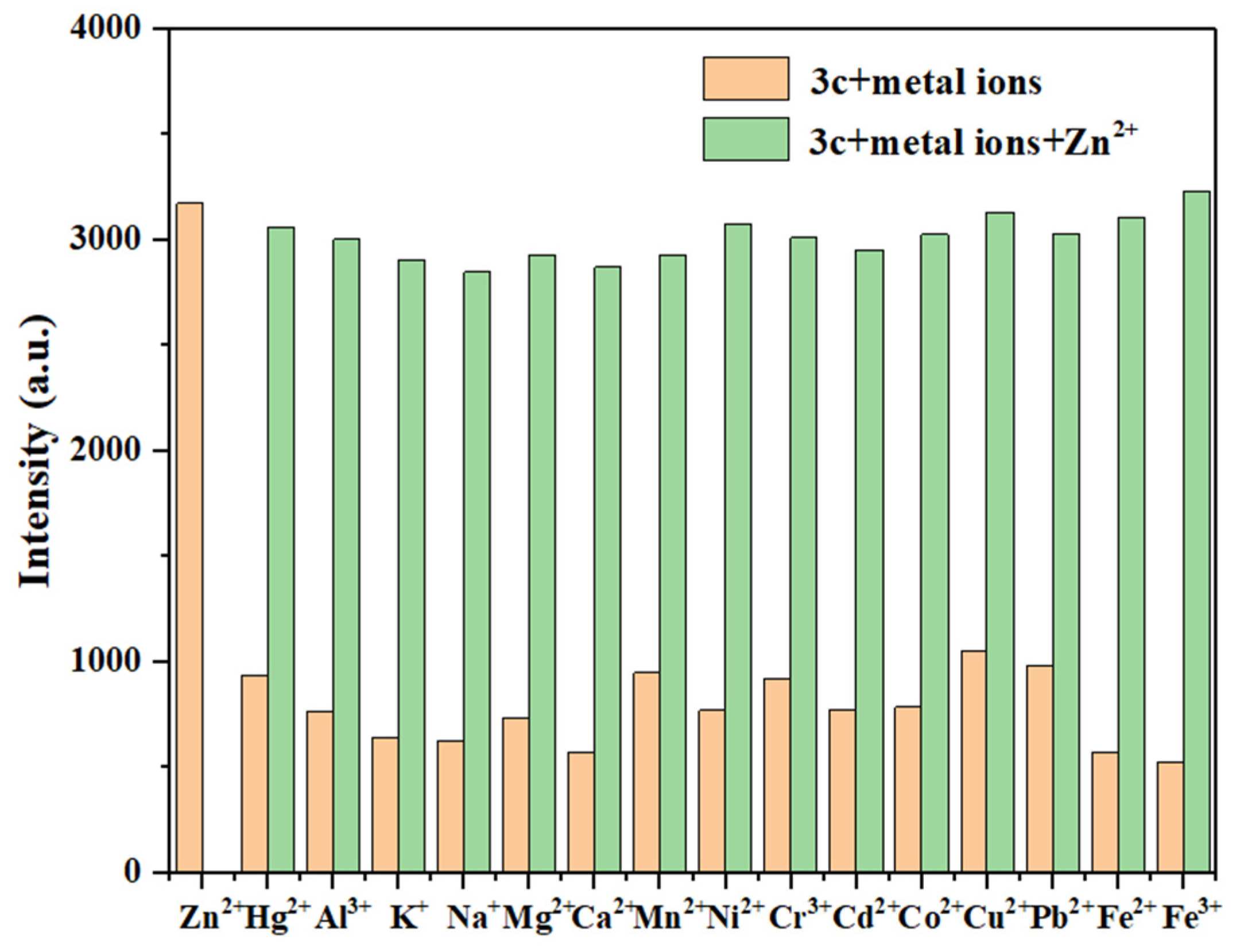
To further evaluate the specificity of probes 3a–3c towards the target ions, interference studies were conducted in the presence of competing metal ions under similar conditions.
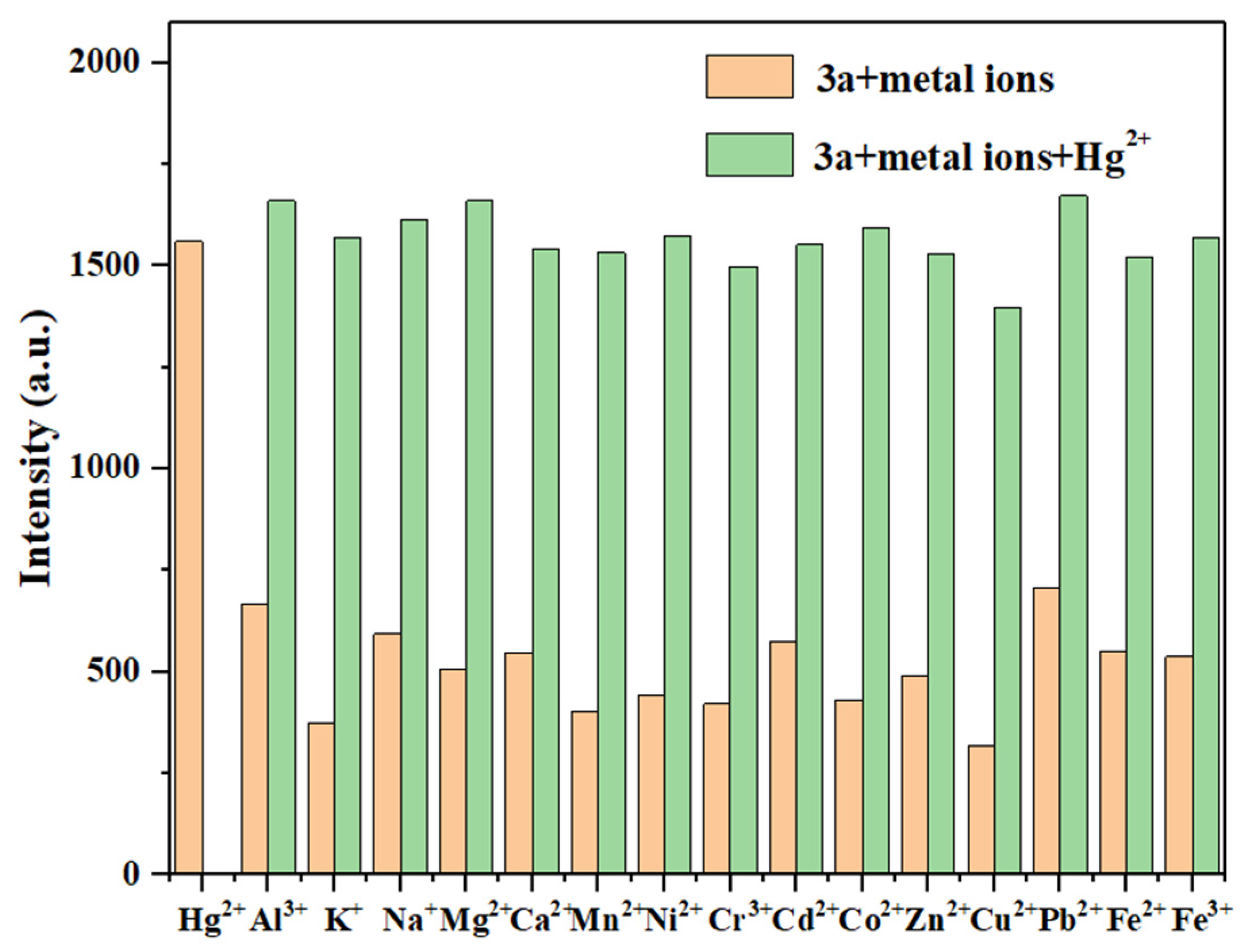
When mixing probe 3a (10 μM) with 11 equivalents of Hg2+ and 11 equivalents of other competing metal ions, as shown in Figure 10, the fluorescence intensity of 3a showed no significant changes in the presence of most competing metal ions. However, there was a significant fluorescence enhancement with the subsequent addition of Hg2+. This indicates that most competing metal ions do not significantly interfere with the detection of Hg2+ by probe 3a. Combined with its low LOD and rapid response, this indicates its potential application value in environmental monitoring.
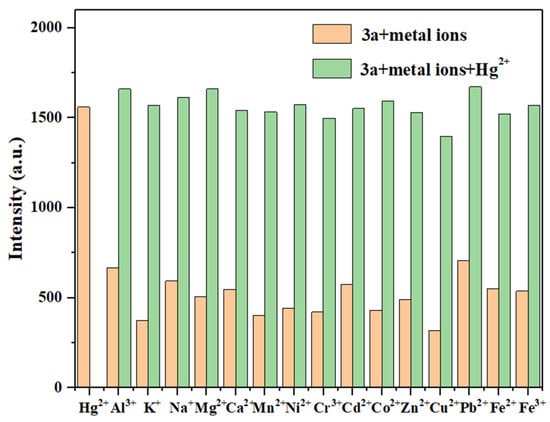
Figure 10.
The anti-interference test of probe 3a for detecting Hg2+ (Hg2+: 11.0 eq.; other metal ions: 11.0 eq.).
Similarly, there were no obvious changes for the fluorescence intensity of probes 3b (Figure S15) and 3c (Figure 11) in the presence of most competing metal ions, but a significant enhancement was observed upon the subsequent addition of Zn2+. This suggests that most metal ions do not cause significant interference in the detection of Zn2+ by probes 3b and 3c. Therefore, these results demonstrate that probes 3a–3c can selectively detect Hg2+ and Zn2+, respectively, in complex environments.
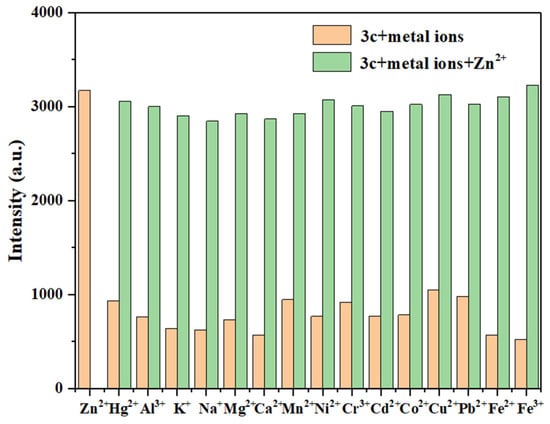
Figure 11.
The anti-interference test of probe 3c for detecting Zn2+ (Zn2+: 10.0 eq.; other metal ions: 10.0 eq.).
2.4.3. Fluorescence Titration Studies of Probes 3a–3c
Fluorescence titration is an important method for understanding the sensitivity of a probe towards an analyte. Therefore, fluorescence titration experiments were performed for probes 3a–3c to analyze the changes in their fluorescence emission spectra. The fitting curve of the maximum emission intensity versus the ion concentration was plotted, and the limit of detection (LOD) was calculated by using the formula LOD = 3δ/K [42]. Here, δ is the standard deviation of 10 blank measurements of the probe solution, and K is the slope of the linear fitting curve of the maximum emission intensity versus the analyte concentration.
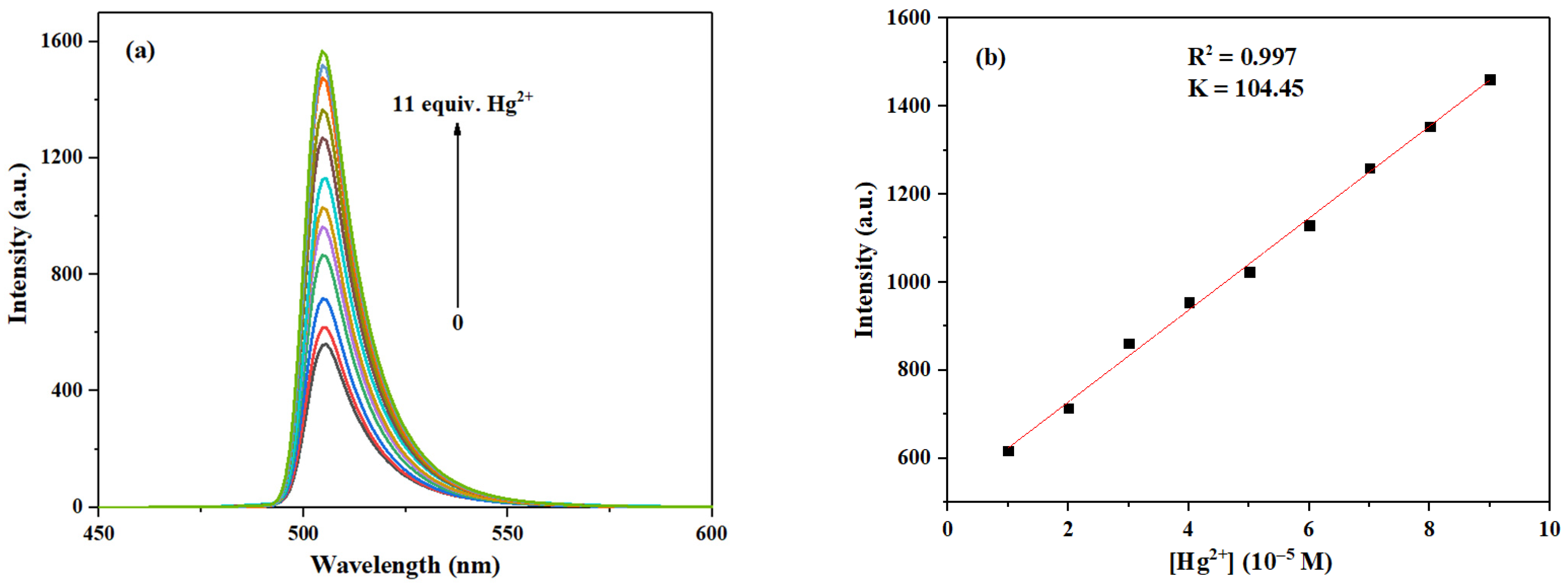
As shown in Figure 12, with the gradual increase of Hg2+ concentration from 0 to 11.0 eq., the fluorescence intensity at 505 nm was progressively enhanced. Furthermore, the fitting curve reveals a good linear relationship (R2 = 0.997) between the fluorescence intensity of probe 3a and the concentration of Hg2+. Thus, the LOD for Hg2+ can be calculated to be 3.21 × 10−7 M, which is significantly lower than those of most fluorescent probes for Hg2+ reported before [43,44] (see Table S3 for more detailed comparison).
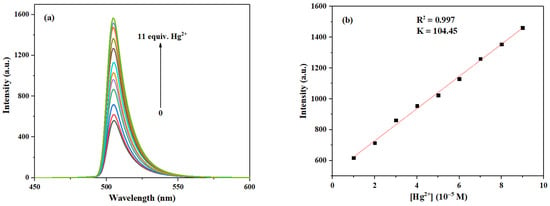
Figure 12.
(a) Fluorescence emission spectra of probe 3a (10 μM) after interaction with Hg2+ at different concentrations (0–11.0 eq.). (b) The relationship between the maximum fluorescence intensity of probe 3a and the concentration of Hg2+ (1.0–9.0 eq.).
Similarly, as shown in Figure S16, the fluorescence intensity at 515 nm increased with the rising concentration of Zn2+ from 0 to 11.0 eq. Moreover, the fitting curve shows a good linear relationship (R2 = 0.997) between the fluorescence intensity of probe 3b and the Zn2+ concentration. The LOD of probe 3b for Zn2+ can be calculated to be 1.61 × 10−7 M. Although probes with even lower LODs have been reported [24], this value remains significantly lower than those of most previously reported fluorescent probes for Zn2+ [45,46] (see Table S4 for more detailed comparison).
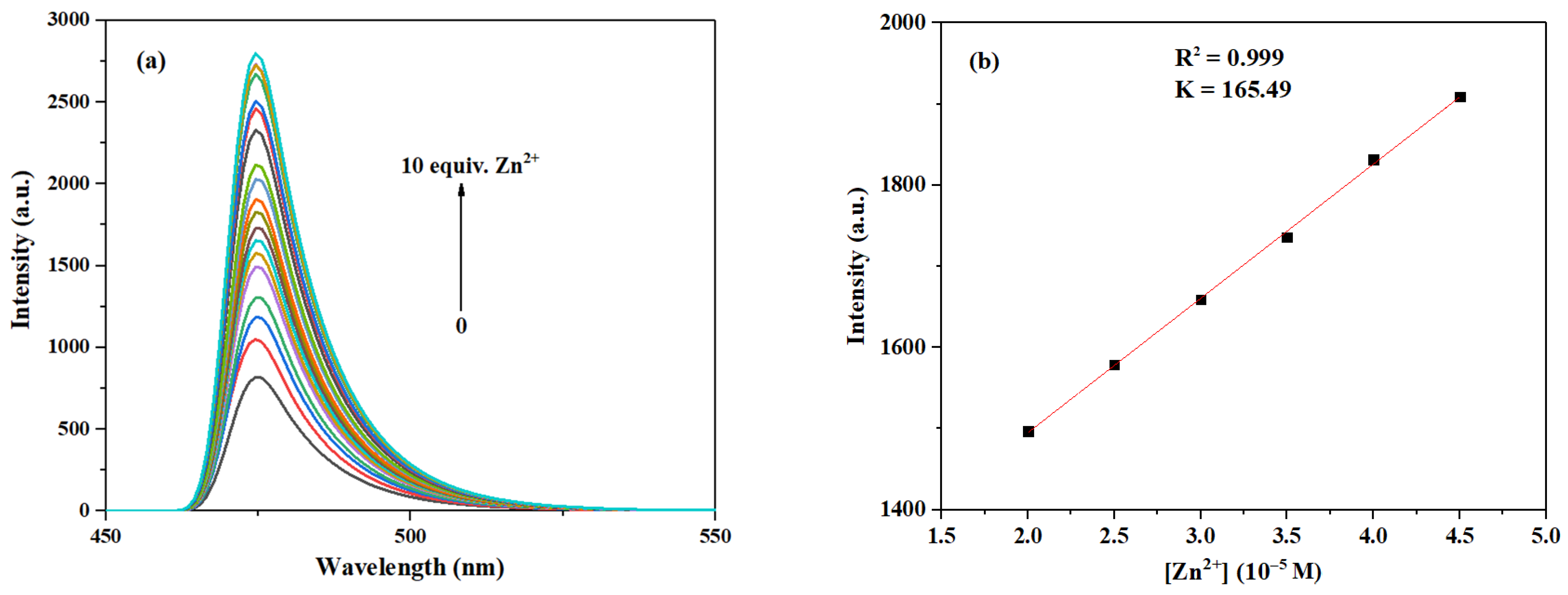
As can be seen in Figure 13, the fluorescence intensity at 474 nm was enhanced as the concentration of Zn2+ increased from 0 to 10.0 eq. Following the method described in references [15,39], and based on the results shown in Figure S17 in Supplementary Materials, we selected the range of 2.0–4.5 eq. for linear fitting because, within this interval, the fluorescence response of the probe was stable and showed optimal linearity, which is suitable for establishing a reliable calibration curve and calculating the sensitivity parameter (slope K). Although the response at lower concentrations is important for assessing sensitivity, the signal variation in the low-concentration region is relatively gentle and lacks good linearity. Hence, we employed data from the 2.0–4.5 eq. range for fitting. Thus, based on the fitting curve within the Zn2+ range of 2.0–4.5 eq., the LOD of probe 3c for Zn2+ can be calculated to be 2.03 × 10−7 M, which is also significantly lower than those of most fluorescent probes for Zn2+ reported before [45,46] (see Table S4 for more detailed comparison).
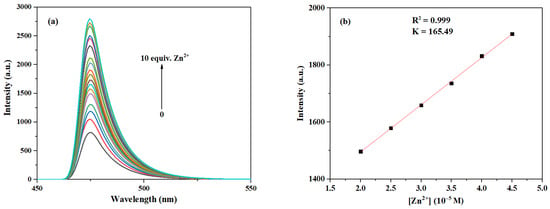
Figure 13.
(a) Fluorescence emission spectra of probe 3c (10 μM) after interaction with Zn2+ at different concentrations (0–10.0 eq.). (b) The relationship between the maximum fluorescence intensity of probe 3c and the concentration of Zn2+ (2.0–4.5 eq.).
2.4.4. Time-Response Studies of Probes 3a–3c to Hg2+/Zn2+
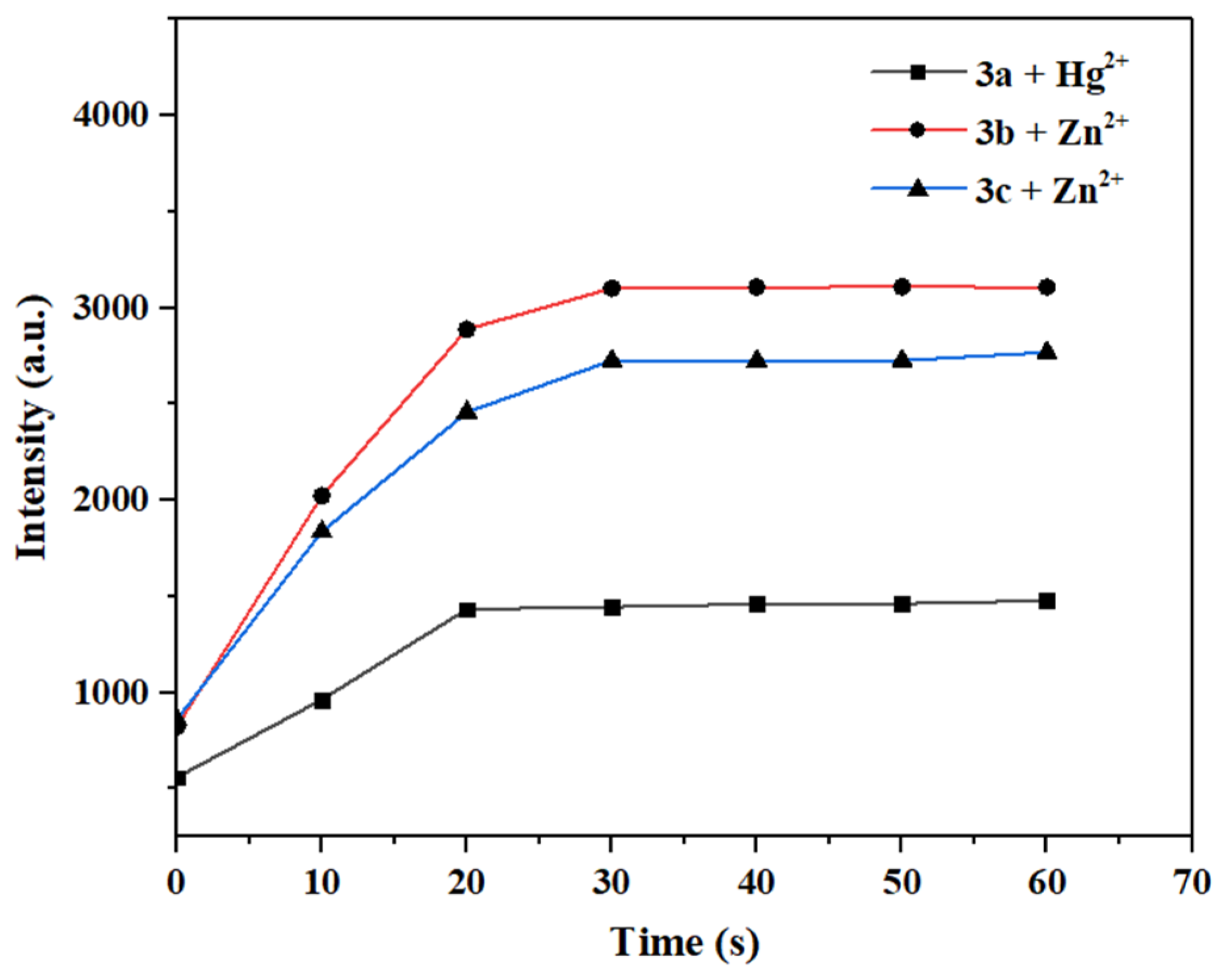
The response time of a fluorescent probe to metal ions is crucial for its application in real-time detection [47]. Therefore, 10.0 eq. of Hg2+/Zn2+ solution was rapidly added to the solutions of probes 3a–3c, and their fluorescence intensities were measured at different time intervals to investigate the relationship between maximum fluorescence intensity and time.
As shown in Figure 14, upon the addition of Hg2+/Zn2+ to the probe solutions, the fluorescence intensity increased immediately, and the action reached completion within 30 s. No significant change in the maximum fluorescence intensity was observed with further extensions of time. Thus, probes 3a–3c exhibit a rapid response to Hg2+/Zn2+, and they are obviously faster than most fluorescence sensors reported before [5,6] (see Tables S3 and S4 for more detailed comparison). Therefore, probes 3a–3c are suitable for the in situ and real-time detection of Hg2+/Zn2+.
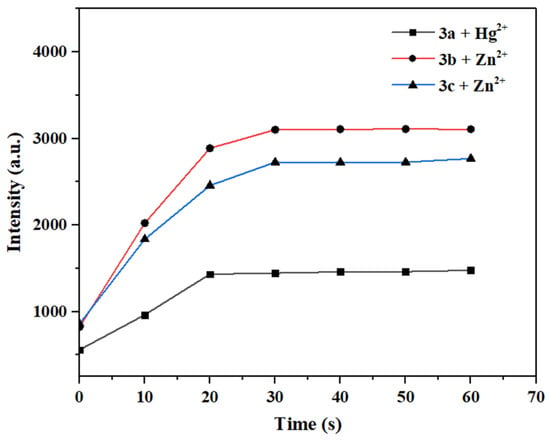
Figure 14.
Time-dependent variation of maximum fluorescence intensity after the interaction between probes 3a–3c and Hg2+ or Zn2+.
2.4.5. Binding Stoichiometry and Association Constant of Probes 3a–3c with Hg2+/Zn2+
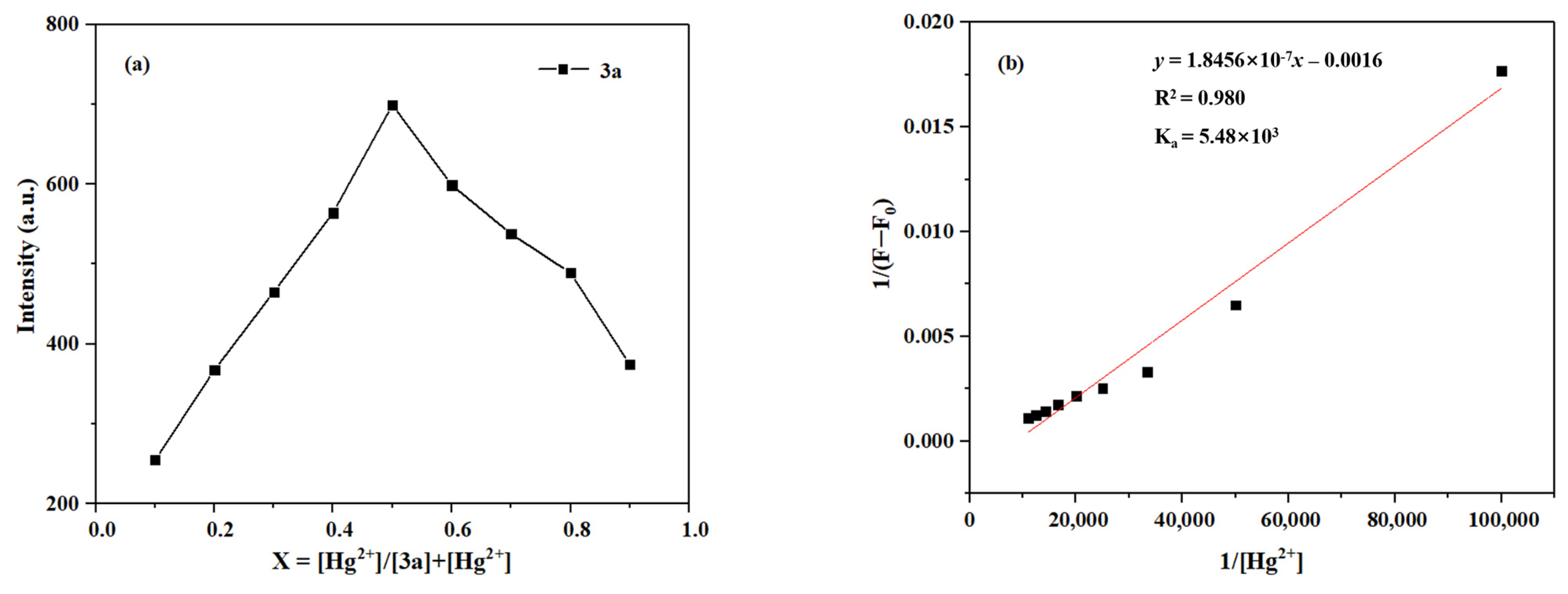
To determine the binding stoichiometry of the coordination between Hg2+/Zn2+ and probes 3a–3c, the method of continuous variations (Job’s plot) was employed. Taking probe 3a as an example, the total concentration of Hg2+ and the ligand was kept constant at 10 μM while varying the molar fraction of Hg2+ from 0.1 to 0.9. The maximum fluorescence intensity was plotted against the molar fraction of Hg2+. The molar fraction at which the intensity reaches its maximum indicates the stoichiometry of the complex [48].
As shown in Figure 15, the Job’s plot shows a maximum intensity at a molar fraction of 0.5 for Hg2+, indicating a 1:1 binding stoichiometry for the complex formed between probe 3a and Hg2+. Furthermore, the association constant (Ka) was determined via the Benesi–Hildebrand equation by linear fitting of the fluorescence titration data [49]. The Ka value for the binding of 3a with Hg2+ can be calculated to be 5.48 × 103 M.
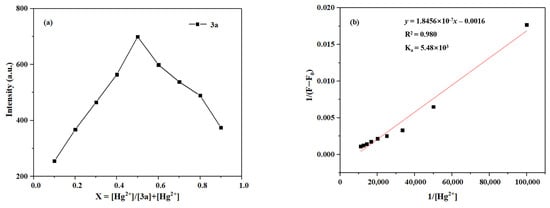
Figure 15.
(a) The Job’s plot of 3a Hg2+. (b) The Benesi–Hildebrand plot of 3a-Hg2+.
Similarly, as shown in Figure S18, the maximum fluorescence intensity was reached at a molar fraction of 0.5 for Zn2+, indicating a 1:1 binding stoichiometry for the complex formed between probe 3b and Zn2+. Furthermore, the association constant for the binding of probe 3b with Zn2+ can be determined to be 1.05 × 104 M by using the Benesi–Hildebrand equation with the fluorescence titration data.
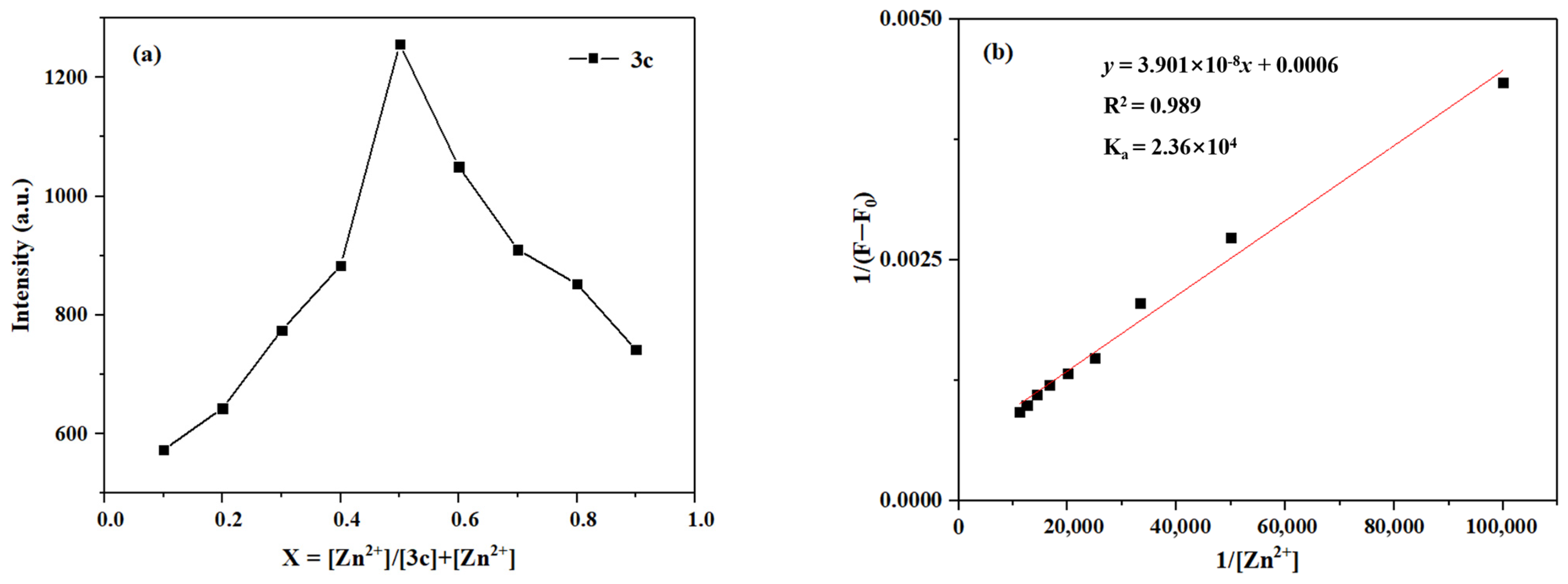
Likewise, as shown in Figure 16, the Job’s plot exhibits a maximum at a molar fraction of 0.5 for Zn2+, which indicates a 1:1 binding stoichiometry between probe 3c and Zn2+. In addition, the association constant for the binding of probe 3c with Zn2+ can be determined to be 2.36 × 104 M by using the Benesi–Hildebrand equation.
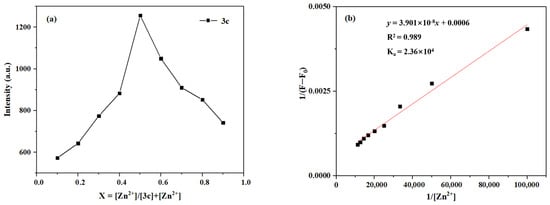
Figure 16.
(a) The Job’s plot of 3c-Zn2+; (b) The Benesi–Hildebrand plot of 3c-Zn2+.
2.4.6. pH Tolerance Studies of Probes 3a–3c for Hg2+/Zn2+ Detection
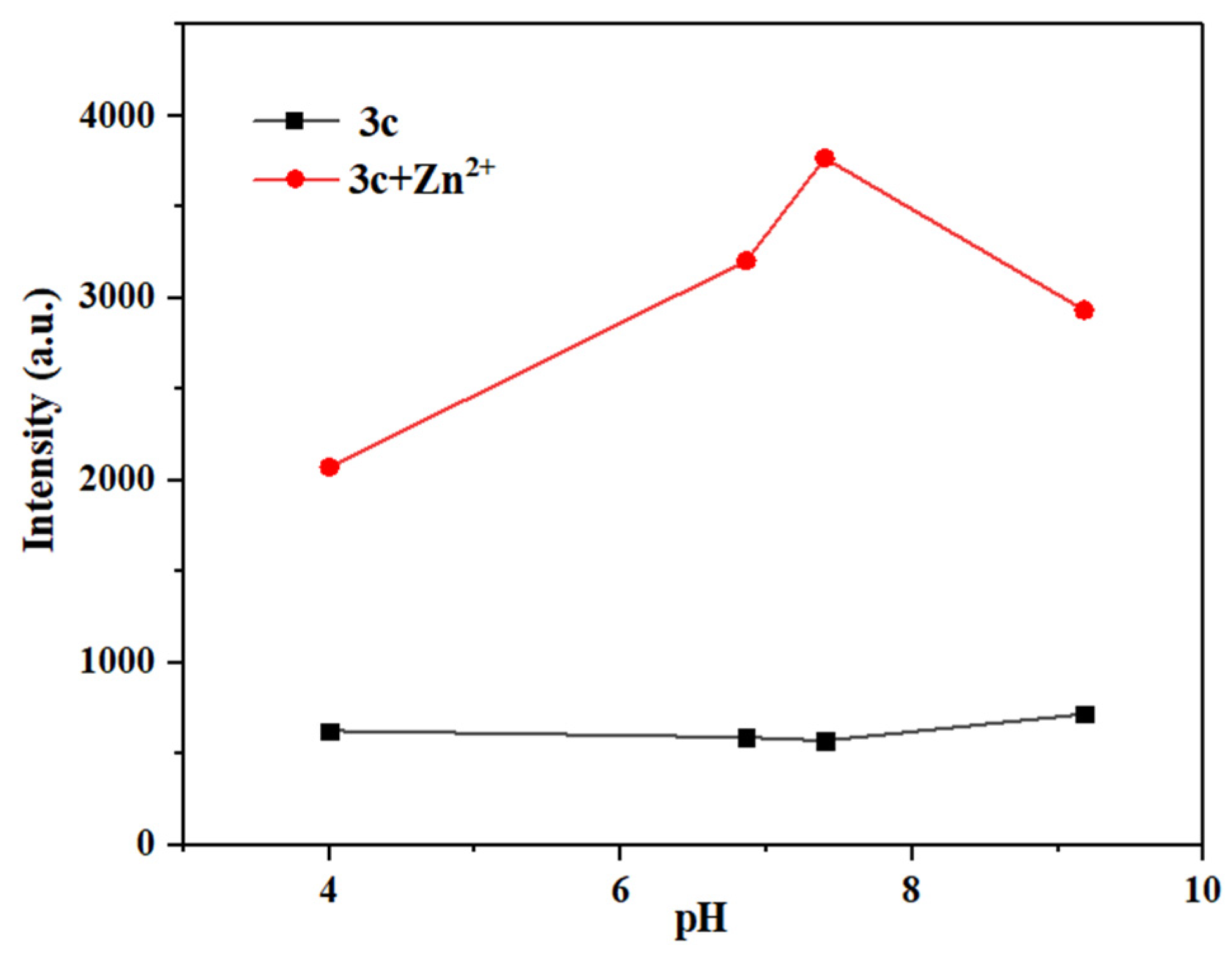
It is well known that the pH of the environment has a significant impact on the selectivity, sensitivity, and practical application of a probe [39]. Therefore, the maximum fluorescence intensity of solutions of probes 3a–3c was measured and compared before and after their interaction with Hg2+/Zn2+ under different pH solutions (pH = 4.00, 6.86, 7.40, 9.18) (Figure 17, Figures S19 and S20).
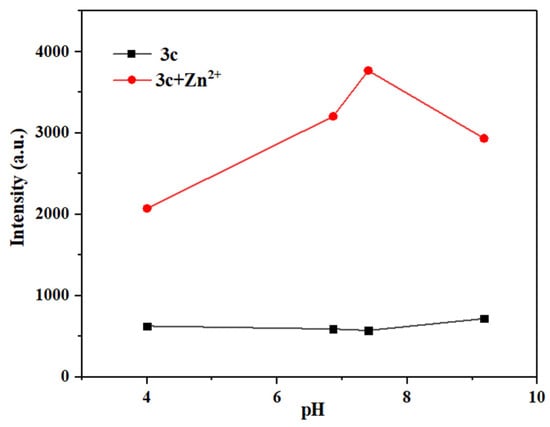
Figure 17.
Different pH values of maximum fluorescence intensity before and after the interaction between probe 3c and Zn2+.
As shown in Figure S19, probe 3a can detect Hg2+ over a wide pH range (pH = 4.00–9.18). Meanwhile, at pH 9.18, probe 3a exhibited strong fluorescence, which might be attributed to its hydrolysis under alkaline conditions. Similarly, as shown in Figure S20, probe 3b can detect Zn2+ in neutral to weakly alkaline systems (pH = 6.86–9.18).
As seen in Figure 17, probe 3c can detect Zn2+ in weakly acidic to weakly alkaline systems (pH = 4.00–9.18), also demonstrating a significantly wider detection range than that of most fluorescent probes reported before [46,50] (see Tables S3 and S4 for more detailed comparison). In short, probes 3a–3c can detect Hg2+/Zn2+ over a broad pH range, making them suitable for most real water samples.
2.5. Investigation of the Detection Mechanism Between Probe 3a and Hg2+
By using probe 3a and Hg2+ as an example, the detection mechanism between the probe and metal ion was systematically investigated from two aspects of experimental observations and theoretical calculations.
2.5.1. 1H NMR and ESI-MS Studies on the Interaction of Probe 3a with Hg2+
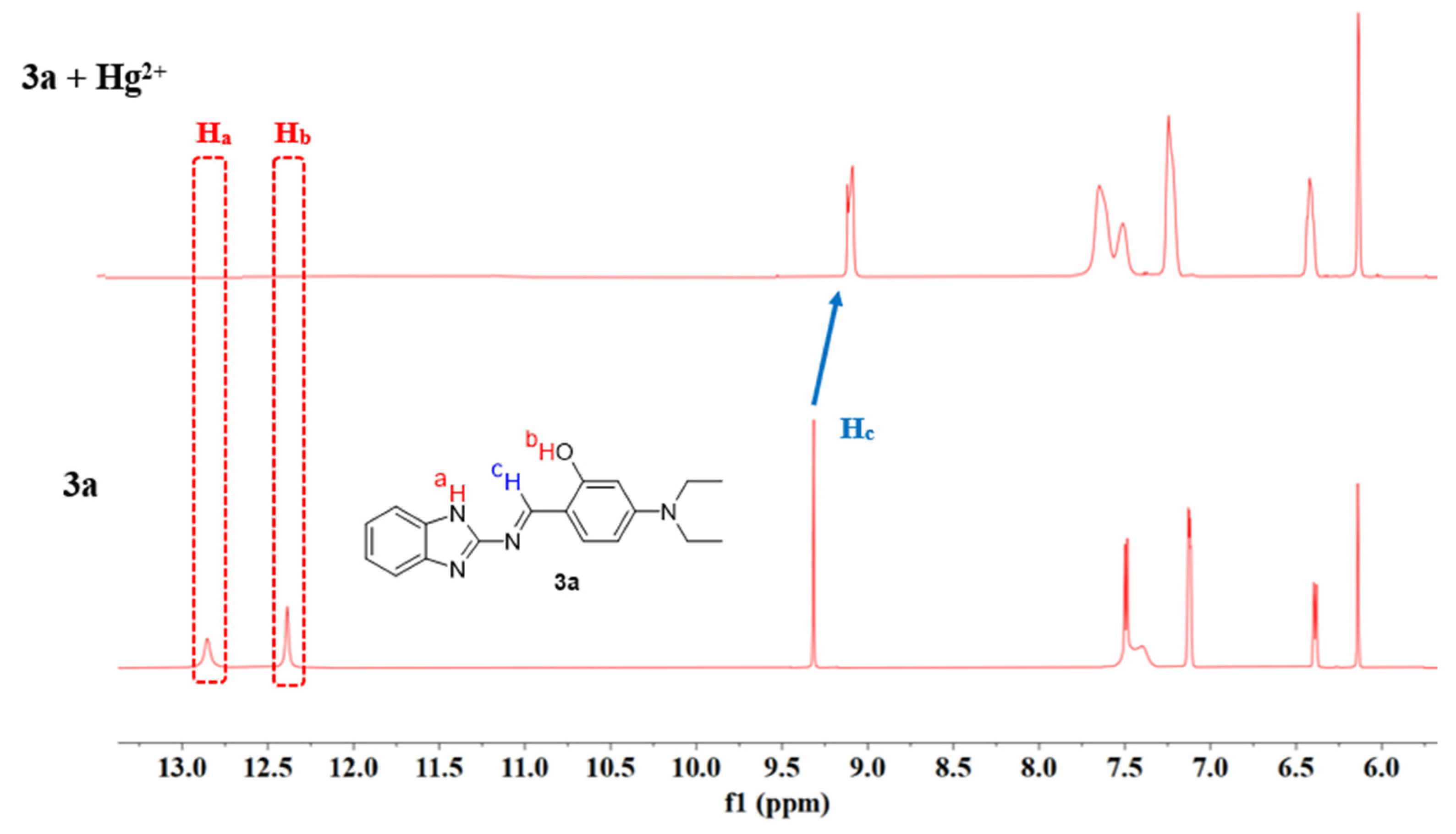
Based on the Job’s plot results, the stoichiometric ratio of the complex formed between compound 3a and Hg2+ has been determined to be 1:1. To further elucidate the recognition mechanism of 3a toward Hg2+, 1H NMR and ESI-MS studies were conducted [51].
The 1H NMR spectra of 3a in DMSO-d6 were recorded both in the absence and presence of Hg2+ (as its nitrate salt). As shown in Figure 18, upon the addition of Hg2+ (1 eq.) to 3a, the characteristic imine proton signal (Hc) at 9.31 ppm shifted to 9.16 ppm, while the signals of amino proton (Ha) and phenolic hydroxyl proton (Hb) obviously disappeared. These observations indicate that the nitrogen atom of the amino group, the oxygen atom of the phenolic hydroxyl group, and the nitrogen atom of the imine group all participate in the coordination between 3a and Hg2+.
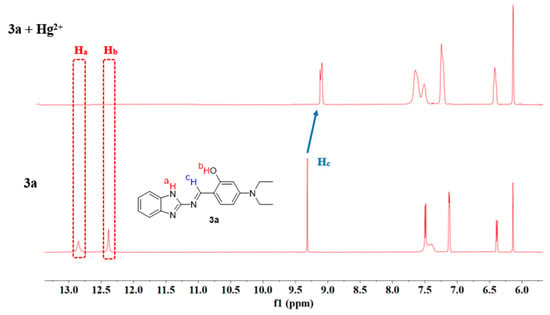
Figure 18.
1H NMR spectra of probe 3a and 3a + Hg2+ (1 eq.).
Furthermore, ESI–MS analysis was performed on the solution of compound 3a after interaction with Hg2+ [51]. As shown in Figure S21, a characteristic peak was observed at m/z 545.10, which corresponds to [3a + Hg2+ + Cl−] (its calculated m/z is 545.10). The result unequivocally confirms the 1:1 binding stoichiometry between 3a and Hg2+.
2.5.2. DFT Computational Studies on the Interaction Between Probe 3a and Hg2+
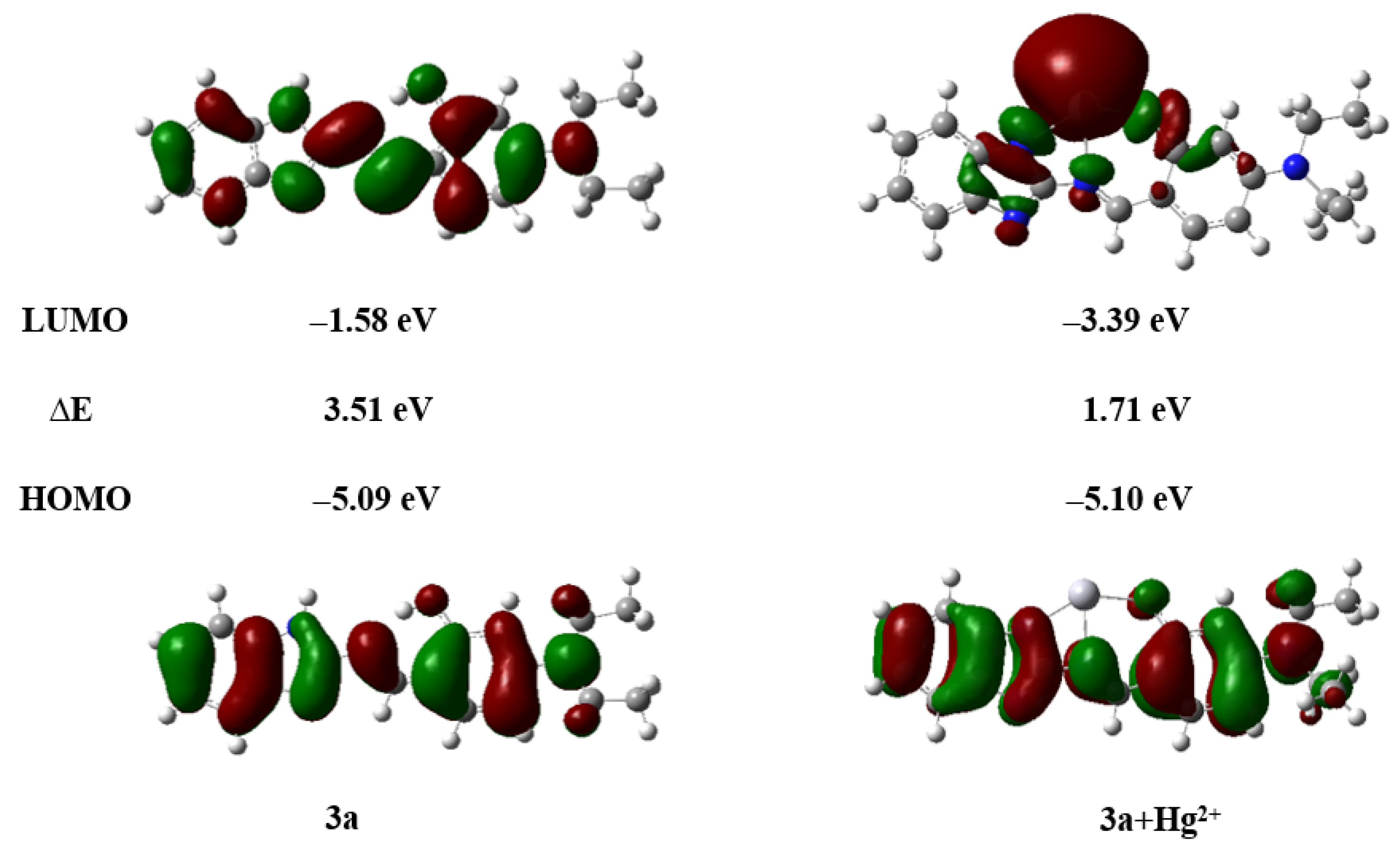
To further investigate the coordination behavior between compound 3a and Hg2+, the time-dependent density functional theory (TD–DFT) calculations were performed by using the Gaussian 09 program at the B3LYP level with the Lanl2dz basis set. The possible structure of the 3a-Hg2+ complex was geometrically optimized, and the energy levels of the LUMO and HOMO were calculated [52].
As illustrated in Figure 15, compound 3a coordinates with Hg2+ in a 1:1 stoichiometric ratio. Upon coordination, the electron density of the HOMO is primarily localized on the benzene ring and the connecting linker, while the LUMO is predominantly distributed around the mercury atom (Figure 19). The energy gap (ΔE) of the 3a + Hg2+ complex was calculated to be 1.71 eV, which is significantly smaller than that of free compound 3a (ΔE = 3.51 eV). This notable reduction in the energy gap indicates a stronger tendency for compound 3a to bind with Hg2+ and suggests the enhanced stability of the resulting complex (Figure S22).
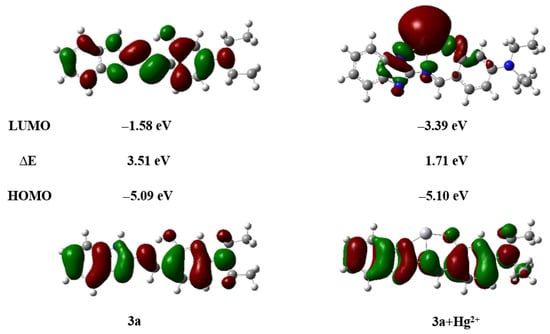
Figure 19.
HOMO–LUMO energy level diagram of compounds 3a, 3a + Hg2+.
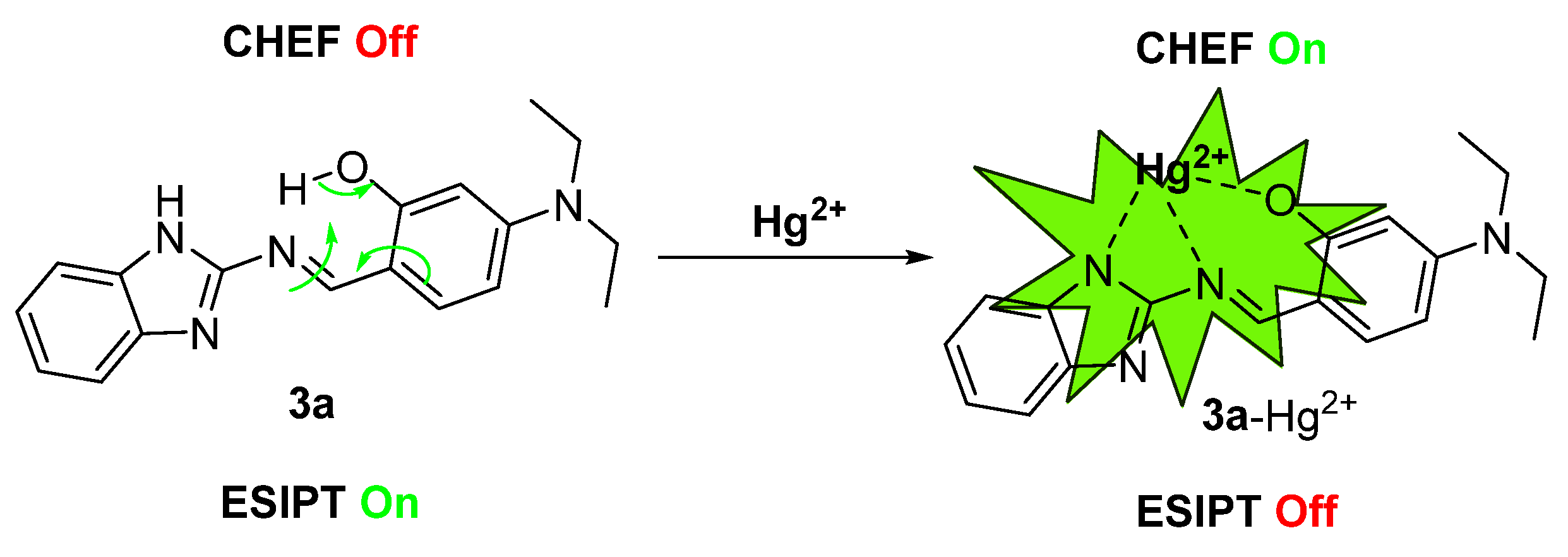
Enhanced molecular rigidity reduces non-radiative decay processes and suppresses ESIPT. Simultaneously, the chelation-enhanced fluorescence (CHEF) effect between compound 3a and Hg2+ induces a distinct fluorescence “turn-on” response, emitting bright yellow-green fluorescence [51]. Thus, the proposed detection mechanism is schematically illustrated in Scheme 3.
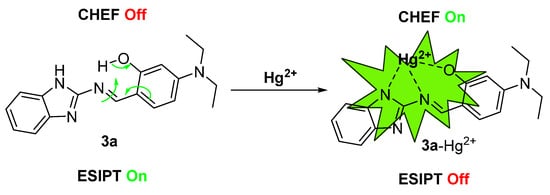
Scheme 3.
Proposed mechanism of probe 3a for the detection of Hg2+.
2.6. Practical Detection Applications of Probes 3a–3c for Hg2+/Zn2+
2.6.1. Real Water Sample Analysis Using Probes 3a–3c for Hg2+/Zn2+ Detection
To evaluate the practical applicability of probes 3a–3c for Hg2+/Zn2+ detection, real water samples, including distilled water, tap water, and river water (pearl river water containing ions such as Cl−, HCO3−, Na+, SO42−, Ca2+, K+, Mg2+, Fe2+, Al3+, and others) were analyzed. The standard addition method was employed to determine Hg2+/Zn2+ concentrations via fluorescence emission spectroscopy and subsequent quantitative analysis [53]. Recovery rates and relative standard deviations (RSD) were determined through triplicate parallel measurements [54].
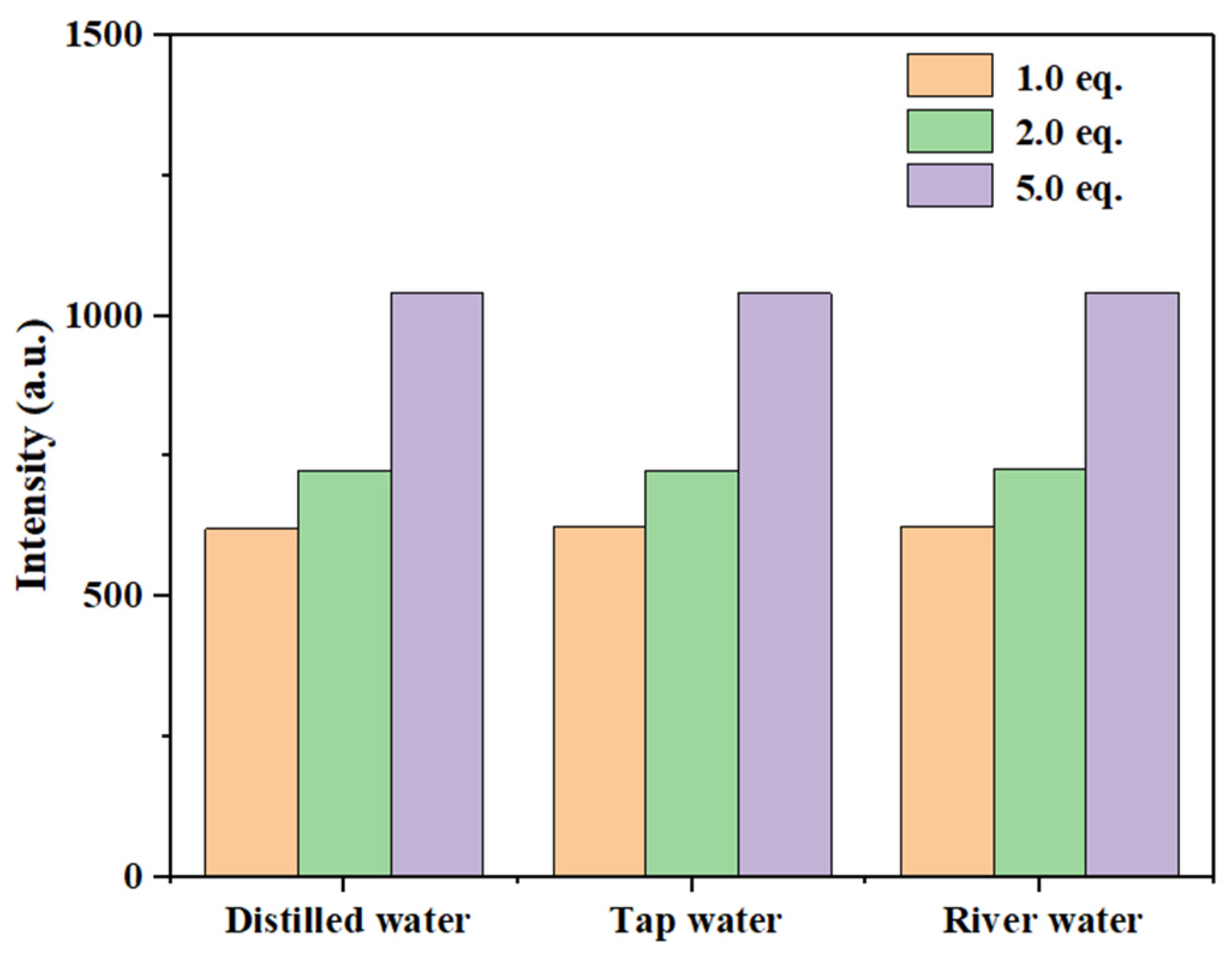
As shown in Figure 20 and Figure S23, the fluorescence intensity of compound 3a gradually increased with rising Hg2+ concentrations (1.0 eq., 2.0 eq., and 5.0 eq.) across all tested water samples. As summarized in Table 1, probe 3a demonstrated recovery rates ranging from 97% to 101% for low concentrations of Hg2+ (10−5 M), with relative standard deviations (RSD) below 1.33%. The observed recovery rates slightly exceeding 100% in some samples may originate from the slight matrix interference in real water samples affecting fluorescence signals, or from systematic measurement and data processing errors. Even so, the deviations in recovery within a similar range have been reported and deemed acceptable in analytical chemistry and practical sensor application studies [54]. Simply, these results indicate that probe 3a achieved high accuracy in detecting Hg2+ in real samples, confirming its suitability for monitoring Hg2+ in practical water systems.
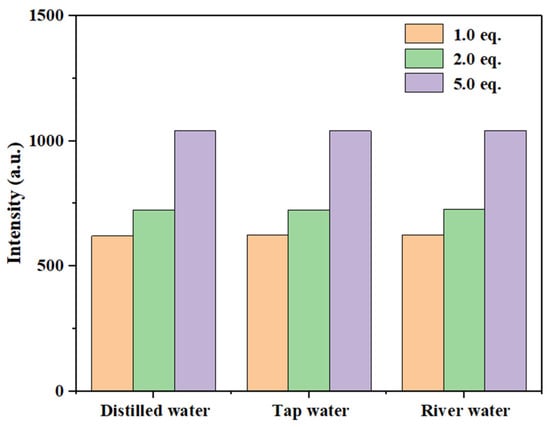
Figure 20.
Relationship between maximum fluorescence intensity of probe 3a in different water samples with different concentrations of Hg2+.

Table 1.
Determination of Hg2+ in actual water samples by probe 3a.
Similarly, as shown in Figure S24, the fluorescence intensity of probe 3b gradually increased with rising Zn2+ concentrations (1.0 eq., 2.0 eq., and 5.0 eq.) across different water samples. According to Table S5, probe 3b achieved recovery rates ranging from 96% to 104% for low concentrations of Zn2+ (10−5 M), with RSD values below 1.19%.
Likewise, the fluorescence intensity of probe 3c exhibited a gradual enhancement with increasing Zn2+ concentrations, as depicted in Figure S25. As summarized in Table 2, probe 3c demonstrated recovery rates of 97% to 101% for Zn2+ at low concentrations (10−5 M), accompanied by RSD values below 0.93%. These results confirm that both probes 3b and 3c are suitable for detecting Zn2+ in real water samples.

Table 2.
Determination of Zn2+ in real water samples by compound 3c.
2.6.2. Paper Strip-Based Detection of Hg2+/Zn2+ Using Probes 3a–3c
Paper-based testing for metal ion detection offers advantages such as simple operation and low cost [53]. To further explore the practical application of probes 3a–3c, test strip experiments were conducted [55]. Whatman filter paper was cut into appropriate strips and immersed in a DMF solution (10−3 M) of compound 3a or DMSO solutions (10−3 M) of compounds 3b and 3c for 5 min. The soaked strips were then dried in an oven to obtain test papers loaded with compounds 3a–3c.
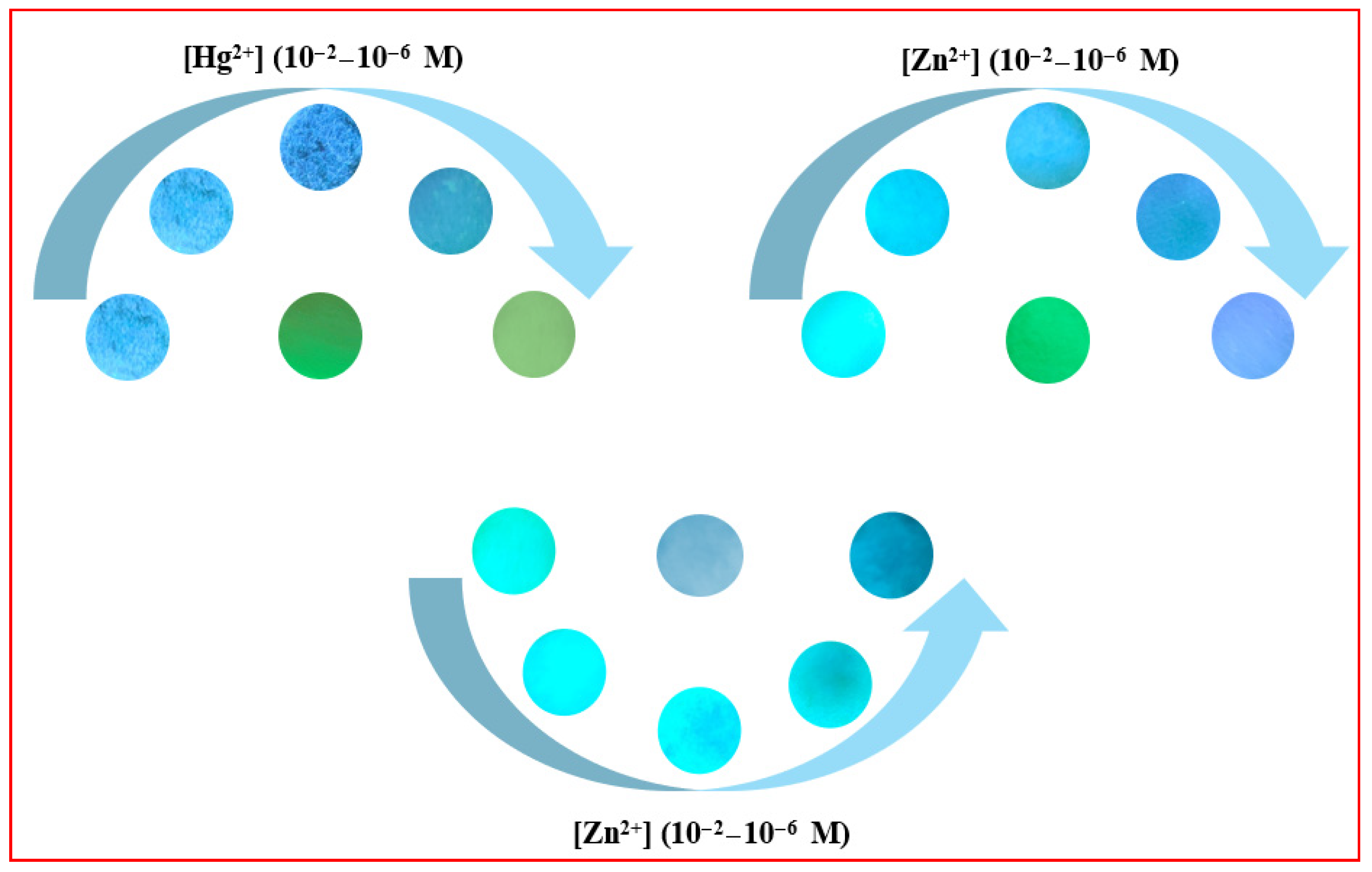
One set of strips served as a blank control, while other sets were treated with 20 μL of Hg2+/Zn2+ solutions at varying concentrations (10−2–10−6 M). After drying with a blow dryer, the fluorescence changes of the strips were observed under 365 nm UV irradiation, as shown in Figure 21.
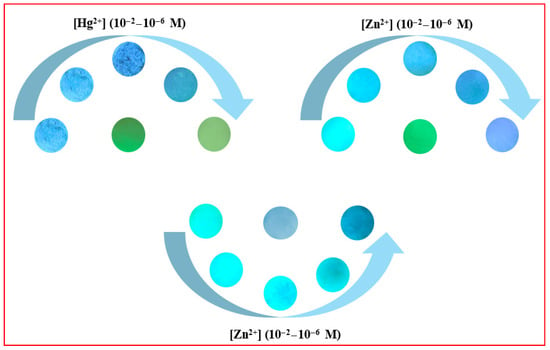
Figure 21.
Test strips loaded with compounds 3a–3c exhibit progressive fluorescence color changes across a concentration gradient of Hg2+/Zn2+ (10−2–10−6 M).
As observed, the fluorescence colors of the test strips underwent distinct changes with increasing concentrations of Hg2+/Zn2+. Therefore, compounds 3a–3c can be fabricated into test strips for the visual detection of Hg2+/Zn2+, providing a simple and rapid monitoring approach.
3. Materials and Methods
3.1. General Information
All the reagents used were domestic analytical pure, and the distilled water was used in the experiment. Nuclear magnetic resonance spectrometer, model Bruker AVANCE NEO 600 MHz, Bruker AVANCE NEO 600M model, Ettlingen, Germany; High-resolution mass spectrometer, model MAT 95XP, Thermos Fisher Q Exactive, Waltham, MA, USA; Ultraviolet spectrometer, model UV-2700, Shimadzu Company, Kyoto, Japan; Fluorescence spectrophotometer, model F-4600, HITACHI, Corporation, Tokyo, Japan.
3.2. Synthesis of Compounds 3a–3c
Synthesis of target compound 3a. As shown in Scheme 2, according to the method in reference [56], 2-aminobenzimidazole 1a (2 mmol) and 4-diethylaminosalicylaldehyde 2 (2 mmol) were dissolved into 10 mL of anhydrous ethanol in a reaction flask. After adding 50 μL of acetic acid, the reaction was carried out by heating at 80 °C for 12 h. Once the reaction was completed, the mixture was cooled to room temperature, creating solid precipitates. After filtration and washing with anhydrous ethanol, a solid crude product was obtained. Then, further recrystallization with anhydrous ethanol gave the pure product 3a as a yellow solid with a yield of 85%. m.p. 229.1–229.9 °C (228.0–230.0 °C [57]); 1H NMR (600 MHz, DMSO-d6), δ, ppm: 1.13 (t, J = 7.2 Hz, 6H, 2CH3), 3.42 (q, J = 7.2 Hz, 4H, 2CH2), 6.14 (s, 1H, ArH), 6.39 (d, J = 8.4 Hz, 1H, ArH), 7.10–7.14 (m, 2H, ArH), 7.40–7.50 (m, 3H, ArH), 9.31 (s, 1H, =CH), 12.38 (s, 1H, OH), 12.85 (s, 1H, NH); 13C NMR (150 MHz, DMSO-d6), δ, ppm: 13.02, 44.56, 97.13, 105.28, 108.75, 121.90, 135.75, 153.13, 155.23, 163.97, 164.63. The NMR spectra of compound 3a are included in the Supplementary Materials section (Figures S1 and S2).
Synthesis of target compound 3b. As shown in Scheme 2, according to the method in reference [58], 2-aminobenzothiazole 1b (2 mmol) and 4-diethylaminosalicylaldehyde 2 (2 mmol) were dissolved into 10 mL of anhydrous ethanol in a reaction flask. The reaction was carried out by heating at 80 °C for 6 h. Once the reaction was completed, the mixture was cooled to room temperature, creating solid precipitates. After filtration and washing with anhydrous ethanol, a solid crude product was obtained. Then, further recrystallization with anhydrous ethanol gave the pure product 3b as an orange solid with a yield of 89%. m.p. 168.1–168.8 °C (172.0–174.0 °C [59]); 1H NMR (600 MHz, CDCl3), δ, ppm: 1.24 (t, J = 7.2 Hz, 6H, 2CH3), 3.44 (q, J = 7.2 Hz, 4H, 2CH2), 6.20 (d, J = 2.4 Hz, 1H, ArH), 6.32 (d, J = 2.4 Hz, 1H, ArH), 7.27 (s, H, ArH), 7.29–7.33 (m, 1H, ArH), 7.42–7.46 (m, 1H, ArH), 7.79 (d, J = 7.8 Hz, 1H, ArH), 7.90 (d, J = 7.8 Hz, 1H, ArH), 8.97 (s, 1H, =CH), 12.74 (s, 1H, OH); 13C NMR (150 MHz, CDCl3), δ, ppm: 12.90, 45.05, 97.60, 105.39, 108.90, 121.71, 122.30, 124.47, 126.48, 134.04, 136.09, 152.06, 153.83, 164.77, 164.84, 170.91. The NMR spectra of compound 3b are included in the Supplementary Materials section (Figures S3 and S4).
Synthesis of target compound 3c. As shown in Scheme 2, 2-amino-6-methylpyridine 1c (2 mmol) and 4-diethylaminosalicylaldehyde 2 (2 mmol) were dissolved into 10 mL of anhydrous ethanol in a reaction flask. After adding 50 μL of acetic acid, the reaction was carried out by heating at 80 °C for 12 h. Once the reaction was completed, the mixture was cooled to room temperature, creating solid precipitates. After filtration and washing with anhydrous ethanol, a solid crude product was obtained. Then, further recrystallization with anhydrous ethanol gave the pure product 3c as a yellow solid with a yield of 92%. m.p. 119.5–120.1 °C; 1H NMR (600 MHz, DMSO-d6), δ, ppm: 1.12 (t, J = 7.2 Hz, 6H, 2CH3), 2.47 (s, 3H, CH3), 3.40 (q, J = 7.2 Hz, 4H, 2CH2), 6.05 (s, 1H, ArH), 6.32 (d, J = 9.0 Hz, 1H, ArH), 7.04–7.08 (m, 2H, ArH), 7.40 (d, J = 9.0 Hz, 1H, ArH), 7.67–7.72 (m, 1H, ArH), 9.17 (s, 1H, =CH), 13.92 (s, 1H, OH); 13C NMR (150 MHz, DMSO-d6), δ, ppm: 13.06, 24.54, 44.45, 97.26, 104.91, 108.85, 115.73, 120.97, 135.63, 139.32, 152.72, 157.22, 157.83, 161.43, 165.46; ESI-MS, m/z (%): Calcd for C17H22N3O ([M + H]+): 284.1763 (100), Found: 284.1753 (100). The NMR and HRMS spectra of compound 3c are included in the Supplementary Materials section (Figures S5–S7).
3.3. Sample Preparation
Compounds 3a–3c were respectively dissolved in organic solvents to prepare a 2 mM stock solution. The concentration of the stock solution of metal ions (10 mM, metal nitrate) was set to 2 mM by using the distilled water. After diluting 2 µL of the stock solution with water/organic solvent (pH = 7.4, v/v = 3/7) to 2.0 mL, the fluorescence emission spectrum was recorded. The selective and interference tests were conducted by adding 10.0 equivalents of metal ions to the probe solution. All samples used for spectral analysis were tested at room temperature.
4. Conclusions
Three Schiff base compounds 3a–3c featuring distinct heterocyclic fluorophores were synthesized via a straightforward one-step environmentally friendly reaction and thoroughly characterized by 1H NMR, 13C NMR, HRMS, and X-ray single-crystal diffraction. Leveraging the coordination interactions between these compounds and metal ions, 3a demonstrates excellent selectivity for Hg2+ with a detection limit of 3.21 × 10−7 M and a rapid response time of 30 s, establishing its utility as a fluorescent probe for Hg2+ detection. Similarly, compounds 3b and 3c exhibit high selectivity toward Zn2+, achieving detection limits of 1.61 × 10−7 M and 2.03 × 10−7 M, respectively, with the same 30 s response time, enabling their application as Zn2+ selective fluorescent probes. Further mechanistic investigations by using 1H NMR, ESI–MS, and DFT calculations revealed that the detection mechanism of probe 3a for Hg2+ operates via an ESIPT–CHEF process. Moreover, probes 3a–3c can be successfully applied for the detection of Hg2+/Zn2+ in real water samples and used as test strips, demonstrating their practical applicability.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/chemosensors14010009/s1, NMR spectra and HRMS for compounds 3a–3c (Figures S1–S7); Data of single-crystal X-ray analysis of probe 3c (Table S1); Studies on solvation of probes 3a–3b (Figures S8 and S9, Table S2); Studies on the AIE properties of probes 3a and 3b (Figures S10 and S11); Selective studies of compounds 3a–3c with metal ions (Figures S12–S14); Anti-interference studies of probe 3b with metal ions (Figure S15); Quantitative identification of Zn2+ by probe 3b (Figure S16); Quantitative identification of Zn2+ by probe 3c (Figure S17); Comparison of some fluorescent probes for Hg2+ and Zn2+ (Tables S3 and S4), See [3,4,18,22,23,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80]; Studies on the binding stoichiometry and association constant of probe 3b with Zn2+ (Figure S18); Studies on pH tolerance of probes 3a–3b in interaction with Hg2+/Zn2+ (Figures S19 and S20); The detection mechanism of probe 3a for Hg2+ (Figures S21 and S22); Application of probes 3a–3c in the detection of Hg2+/Zn2+ (Figures S23–S25, Table S5).
Author Contributions
Conceptualization, Z.-Y.W.; methodology, H.-Q.L.; formal analysis, H.-Q.L. and Y.L.; data curation, Y.L. and Y.-T.L.; interpretation of data, S.-W.D. and W.W.; writing—original draft preparation, H.-Q.L., S.-W.D. and W.W.; reviewing it critically for important intellectual content, Y.-T.L. and W.W.; writing—review and editing, S.-Y.L. and Z.-Y.W.; project administration, Z.-Y.W.; funding acquisition, Z.-Y.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by NNSFC (No. 20772035) and the Guangdong Basic and Applied Basic Research Foundation (No. 2021A1515012342).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data supporting the findings of this study are available within the paper and within its Supplementary Materials, published online.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Zhen, D.S.; Liu, C.L.; Deng, Q.H.; Zhang, S.Q.; Yuan, N.M.; Li, L.; Liu, Y. A review of covalent organic frameworks for metal ion fluorescence sensing. Chin. Chem. Lett. 2024, 35, 109249. [Google Scholar] [CrossRef]
- Chourasia, J.; Tohora, N.; Mahato, M.; Sultana, T.; Ahamed, S.; Maiti, A.; Ghanta, S.; Das, S.K. A sulfone-based fluorogenic probe for cascade detection of Zn2+ and PO43− ions. J. Mol. Struct. 2024, 1304, 137736. [Google Scholar] [CrossRef]
- Liu, J.F.; Wang, X.K.; Zhu, X.F.; Zhao, Y.F.; Ye, Y. Advances in dual-function bioprobes for simultaneous detection of transition metal ions (Fe, Cu, Zn) and bioactive species. Coord. Chem. Rev. 2025, 526, 216352. [Google Scholar] [CrossRef]
- Cui, B.; Yan, Z.J.; Bu, N.S.; Wang, S.R.; Yan, W.H.; Cui, J.B.; Xu, Y.M.; Shao, H.M.; Yang, L.N.; Yang, Y.J.; et al. Precise fixation of the spatial configuration of adsorption groups for removal of mercury ions from the acidic wastewater. Adv. Funct. Mater. 2025, 35, 2422894. [Google Scholar] [CrossRef]
- Chen, Y. Advances in organic fluorescent probes for intracellular Zn2+ detection and bioimaging. Molecules 2024, 29, 2542. [Google Scholar] [CrossRef]
- Gao, N.S.; Chang, X.Y.; Wang, Y.Y.; Li, N.; Guo, W.T.; Zhao, Z.W.; Liu, S.S.; Meng, G.P.; Zhang, H.; Wang, B.D. A signal amplifying MOF-based probe: On-site and ultrasensitive dual-channel portable detection of Hg2+ in groundwater through a fluorimetrically and RGB-based sensing assay. Talanta 2025, 286, 127553. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.J.; Ji, P.; Liu, X.M.; Hu, X. Peptide fluorescent probes based on aggregation-induced emission for the detection of Ni2+ and Zn2+ in different buffer systems. Luminescence 2025, 40, e70116. [Google Scholar] [CrossRef]
- Gandin, A.; Brigo, L.; Giacomazzo, S.; Torresan, V.; Brusatin, G.; Franco, A. Reversible fluorescent solid porous films for detection of zinc ions in biological media. J. Biol. Eng. 2025, 19, 17. [Google Scholar] [CrossRef]
- Yuan, Z.X.; Zhang, Y.; Qu, W.J.; Dong, J.Q.; Wang, R.Y.; Jia, L.H. A highly sensitive and colorimetric fluorescent sensor for visualizing Zn2+ in aqueous solution. Spectrochim. Acta A 2025, 311, 125793. [Google Scholar] [CrossRef]
- Song, Q.X.; Bai, C.B.; Dong, Y.J.; Chen, M.Y.; Wang, S.Z.; Hu, J.D.; Qiao, X.; Chen, J.; Li, S.Y.; Liu, X.Y.; et al. Highly selective Zn2+ near-infrared fluorescent probe and its application in biological imaging. Spectrochim. Acta A 2024, 322, 124828. [Google Scholar] [CrossRef]
- Yin, J.; Wu, Z.J.; Li, H.; Cao, B.L.; Wang, W.Z. Monitoring of mercury ion in environmental media and biological systems using a red emissive fluorescent probe with a large Stokes shift. Spectrochim. Acta A 2025, 326, 125272. [Google Scholar] [CrossRef]
- Tian, Q.Q.; Hong, T.; Zhao, Z.G.; Shi, Z.C. Synthesis and application of a novel reactive Coumarin-derived probe for the determination of Hg2+ in real samples. Inorg. Chim. Acta 2022, 537, 120937. [Google Scholar] [CrossRef]
- Chen, X.L.; Zheng, H.X.; Li, X.Y.; Ruan, Z.J.; Lu, Q.Q.; He, W.T.; Lin, J.Q.; Ran, J.W.; Liu, S.S. AIE-based ratiometric fluorescent probe for mercury ion, medium-dependent fluorescence color change and optimized sensitivity in solid state. Spectrochim. Acta A 2024, 305, 123482. [Google Scholar] [CrossRef] [PubMed]
- An, B.Q.; Yin, Z.; Yan, H.L.; Cao, W.B.; Ye, Y. A novel di-functional fluorescent probe for ONOO− and Zn2+ imaging in cells. Spectrochim. Acta A 2024, 315, 124275. [Google Scholar] [CrossRef]
- Wu, S.J.; Yang, Y.J.; Cheng, Y.; Wang, S.D.; Zhou, Z.B.; Zhang, P.; Zhu, X.Q.; Wang, B.D.; Zhang, H.; Xie, S.; et al. Fluorogenic detection of mercury ion in aqueous environment using hydrogel-based AIE sensing films. Aggregate 2023, 4, e287. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Deng, L.L.; Xue, L.; Fu, S.; Gao, Y.K.; Wang, H.B. A triphenylamine-based multifunctional fluorescent probe for Cu2+ and Zn2+ as well as mechanochromism and application in latent fingerprints. Spectrochim. Acta A 2025, 333, 125915. [Google Scholar] [CrossRef]
- Zhao, Y.; Jiang, M.H.; Xiu, X.M.; Zhang, C.Z.; Zhou, X.C.; Meng, Q.T.; Liu, Y.H. ESIPT-associated fluorescent property and AIE characteristic of multifunctional nanoprobe DPNAP. J. Mol. Liq. 2024, 407, 125291. [Google Scholar] [CrossRef]
- Wu, X.-T.; Zheng, Y.-Y.; Ma, S.-S.; Ai, S.-H.; Zhou, Q.-W.; Yang, D.; Ma, C. Synthesis, optical properties, and application of novel chalcone skeleton as pH fluorescent probe: Based AIE plus ESIPT strategy. Spectrochim. Acta A 2024, 314, 124199. [Google Scholar] [CrossRef]
- Dai, D.F.; Zhang, Z.M.; Ma, M.; Zhao, C.; Li, J.K.; Zhang, S.Q.; Ma, P.Y.; Zhang, B.; Song, D.Q. Universal low-background fluorophore platform via structural reprogramming of oxazine 1 with julolidine for activatable probe design. Anal. Chem. 2025, 97, 21071–21078. [Google Scholar] [CrossRef]
- Oguz, M.; Aydin, D.; Malkondu, S.; Erdemir, S. Specific and low-level detection of Hg2+ and CN− in aqueous solution by a new fluorescent probe: Its real sample applications including cell, soil, water, and food. Sens. Actuators B 2025, 433, 137527. [Google Scholar] [CrossRef]
- Sen Gupta, A.; Paul, K.; Luxami, V. A fluorescent probe with “AIE plus ESIPT” characteristics for Cu2+ and F− ions estimation. Sens. Actuators B 2017, 246, 653–661. [Google Scholar] [CrossRef]
- Yi, S.Q.; Liu, H.L.; Chen, Z.; Fan, C.B.; Liu, G.; Pu, S.Z. Novel fluorescent probes based on NBD-substituted imidazole amino to sequentially detect H2S and Zn2+. Dyes Pigments 2023, 214, 111211. [Google Scholar] [CrossRef]
- Huang, Y.R.; Li, Y.; Li, Y.; Zhong, K.L.; Tang, L.J. An “AIE plus ESIPT” mechanism-based benzothiazole-derived fluorescent probe for the detection of Hg2+ and its applications. New J. Chem. 2023, 47, 6916–6923. [Google Scholar] [CrossRef]
- Lu, K.; Guo, H.Y.; Jiang, Y.X.; Yang, J.Q.; Yu, S.S.; Yu, X.Q.; Pu, L. Synthesis of a binol-based C3 symmetric Schiff base and its fluorescence response to Zn2+. ChemPlusChem 2023, 88, e202300036. [Google Scholar] [CrossRef]
- Wang, B.-W.; Jiang, K.; Li, J.-X.; Luo, S.-H.; Wang, Z.-Y.; Jiang, H.-F. 1,1-Diphenylvinyl-sulfide as a functional AIEgen derived from the aggregation-caused quenching molecule 1,1-diphenylethene by easy thioetherification. Angew. Chem. Int. Ed. 2020, 59, 2338–2343. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Q.; Yang, S.-H.; Li, Y.; Ye, W.-X.; Liao, Z.-Y.; Lu, J.-Q.; Wang, Z.-Y. Schiff base compounds derived from 5-methyl salicylaldehyde as turn-on fluorescent probes for Al3+ detection: Experimental and DFT calculations. Molecules 2025, 30, 1128. [Google Scholar] [CrossRef]
- Li, F.R.; Dai, X.; Zhang, L.F.; Wu, H.D.; Li, J.D.; Guo, J.; Yi, Q. Triphenylamine promoted geometric structure adjusting of the novel macrocyclic structure D-π-A conjugated microporous polymers for photocatalytic hydrogen evolution. Fuel 2024, 370, 131812. [Google Scholar] [CrossRef]
- Sun, W.; Hu, G.X.; Shen, W.L.; Xu, H.H.; Deng, Z.M.; Zhao, G.M.; Li, F.Z.; Hu, Y.H.; Yang, W.G. A series of D-π-A and A-π-A′ fluorescent probes were used to explore the influence of terminal groups on the properties of the hemicyanine probes. J. Mol. Liq. 2021, 340, 116846. [Google Scholar] [CrossRef]
- Fang, W.L.; Liang, Z.Y.; Guo, X.F.; Wang, H. A D-π-A-based near-infrared fluorescent probe with large Stokes shift for the detection of cysteine in vivo. Talanta 2024, 268, 125354. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.P.; Fu, J.; Wan, J.X.; Huang, T.S.; Zhu, W.F.; Tian, J.W.; Liu, M.Y.; Zhang, X.Y.; Wei, Y. One-step synthesis of a dual-functional AIE-active probe for ClO− detection and photodynamic therapy. Chem. Commun. 2024, 60, 984–987. [Google Scholar] [CrossRef]
- Bec, A.; Vianello, R.; Hranjec, M. Synthesis and spectroscopic characterization of multifunctional D-π-A benzimidazole derivatives as potential pH sensors. J. Mol. Liq. 2023, 386, 122493. [Google Scholar] [CrossRef]
- Rana, S.; Vaidyanathan, S.; Patel, S. Aggregation induced emission (AIE) based donor-π-acceptor fluorophores: An approach to fabricate acidochromic sensors and white light emitting diodes. J. Mater. Chem. C 2024, 12, 14148–14164. [Google Scholar] [CrossRef]
- Huang, X.; Lan, N.; Jiang, F.; He, H.F.; Zhong, J. Synthesis of a near-infrared fluorescence turn-on probe based on dicyanoisophorone for HS− detection in cancer cells and zebrafish in pure water media. ChemistrySelect 2022, 7, e202201070. [Google Scholar] [CrossRef]
- Sayed, M.; Kamal Eldean, A.M.; Ahmed, M.M.; Hassanien, R. Synthesis of some new heterocyclic compounds containing indole moiety. Eur. Chem. Bull. 2017, 6, 171–176. [Google Scholar] [CrossRef][Green Version]
- Vashist, N.; Sambi, S.S.; Narasimhan, B.; Kumar, S.; Lim, S.M.; Shah, S.A.A.; Ramasamy, K.; Mani, V. Synthesis and biological profile of substituted benzimidazoles. Chem. Cent. J. 2018, 12, 125. [Google Scholar] [CrossRef]
- Lu, C.X.; Xu, J.W.; Song, Z.; Dai, Z.Y. Advancements in ESIPT probe research over the past three years based on different fluorophores. Dyes Pigments 2023, 208, 110820. [Google Scholar] [CrossRef]
- Sharma, S.J.; Khan, Z.N.; Zambare, A.A.; Bagal, M.S.; Barshi, A.S.; Rindhe, S.M.; Sekar, N. Synthesis, spectroscopic, DFT, TD-DFT, and dyeing studies of 2-amino-3-cyano thiophene-based azo dyes on wool and nylon. Dyes Pigments 2024, 228, 112209. [Google Scholar] [CrossRef]
- Wu, W.-N.; Mao, P.-D.; Song, Y.-F.; Zhao, X.-L.; Wang, Y.; Xu, Z.-H. A simple AIE-based indole-benzimidazole probe for the ratiometric fluorescent detection of phosgene in an almost neat aqueous solution. Talanta 2025, 283, 127172. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.N.; Li, C.Q.; Wei, S.H.; Lu, C.W.; Zou, L.W. A multifunctional fluorescent probe based on Schiff base with AIE and ESIPT characteristics for effective detections of Pb2+, Ag+ and Fe3+. Spectrochim. Acta A 2023, 300, 122904. [Google Scholar] [CrossRef] [PubMed]
- Budri, M.; Naik, G.; Patil, S.; Kadolkar, P.; Gudasi, K.; Inamdar, S. An ESIPT blocked highly ict based molecular probe to sense Zn(II) ion through turn on optical response: Experimental and theoretical studies. J. Photochem. Photobiol. A 2020, 390, 112298. [Google Scholar] [CrossRef]
- Sun, J.Q.; Wang, Y.G.; Wang, M.D.; Wang, H.M. A bisalicylhydrazone based fluorescent probe for detecting Al3+ with high sensitivity and selectivity and imaging in living cells. Spectrochim. Acta A 2024, 322, 124784. [Google Scholar] [CrossRef]
- Chen, Z.-J.; Guo, J.-L.; Chen, G.-Y.; Li, Z.; Ye, Y.-Y.; Liu, Y.-T.; Wang, Z.-Y. Synthesis of novel cyanopyridine-type dual-state emission fluorophores and their sensing applications in nitro explosives. Dyes Pigments 2026, 247, 113529. [Google Scholar] [CrossRef]
- Fang, S.J.; Zhang, L.W.; Zhao, Y.; Zhang, X.; Zhang, L.; Chen, L.X.; Yoon, J.; Liu, S.D. Insight into mercury ion detection in environmental samples and imaging in living systems by a near-infrared fluorescent probe. Sens. Actuators B 2024, 411, 135768. [Google Scholar] [CrossRef]
- Zhou, H.; Jin, Y.; Liu, Y.K.; Wang, Y.X.; Pang, X.F.; Zhao, Q.; Bu, M. A near-infrared “turn-on” fluorescent probe based on dicyanomethylene-4H-pyran for the selective detection of Hg2+ in living cells. J. Photochem. Photobiol. A 2023, 447, 115294. [Google Scholar] [CrossRef]
- Jovic, M.; Markovic, O.; Newhouse, T.R.; Opsenica, I.M.; Selakovic, Z. A highly selective ESIPT-mechanism-based, ratiometric fluorescent sensor for zinc ions. Dyes Pigments 2025, 234, 112547. [Google Scholar] [CrossRef]
- Ahmad, T.; Abdel-Azeim, S.; Khan, S.; Ullah, N. Dinitrophenyl and NBD platforms based fluorescent sensors for nanomolar detection of zinc ions: Synthesis, zinc ions sensing and DFT studies. Inorg. Chem. Commun. 2024, 170, 113151. [Google Scholar] [CrossRef]
- Sidqi, M.E.; Aziz, A.A.A.; Abolehasan, A.E.; Sayed, M.A. Photochemical processing potential of a novel Schiff base as a fluorescent probe for selective monitoring of Al3+ ions and bioimaging in human cervical cancer HeLa cells. J. Photochem. Photobiol. A 2022, 424, 113616. [Google Scholar] [CrossRef]
- Zhao, C.; Xu, H.M.; Zhang, X.R.; Meng, Y.T.; Shuang, S.M.; Dong, C. Anthraquinone-metal complex fluorescence sensing platform for monitoring PPi mediated by Al3+ and bioimaging. J. Mol. Struct. 2024, 1308, 138022. [Google Scholar] [CrossRef]
- Sultana, T.; Mahato, M.; Tohora, N.; Ahamed, S.; Das, S.K. An azine-based chromogenic, fluorogenic probe for specific cascade detection of Al3+ and PO43− ions. J. Photochem. Photobiol. A 2023, 444, 114951. [Google Scholar] [CrossRef]
- Kaur, M.; Virender, V.; Singh, J.; Kumar, A.; Dubey, S.K. Smartphone-based analytical platform for real-time, reversible detection of Co2+, Ni2+, Cu2+, and Zn2+ ions. Food Chem. 2025, 492, 145388. [Google Scholar] [CrossRef] [PubMed]
- Bu, F.Q.; Zhao, B.; Kan, W.; Ding, L.M.; Liu, T.; Wang, L.Y.; Song, B.; Wang, W.B.; Deng, Q.G. An ESIPT characteristic “turn-on” fluorescence sensor for Hg2+ with large stokes shift and sequential “turn-off” detection of S2− as well as the application in living cells. J. Photochem. Photobiol. A 2020, 387, 112165. [Google Scholar] [CrossRef]
- Kaur, B.; Gupta, A.; Kaur, N. A novel, anthracene-based naked eye probe for detecting Hg2+ ions in aqueous as well as solid state media. Microchem J. 2020, 153, 104508. [Google Scholar] [CrossRef]
- Yang, S.-H.; Li, Z.; Xie, J.-Y.; Guo, Y.-T.; Chen, G.-Y.; Li, Y.; Wang, Z.-Y. Design, synthesis, optical properties, and application of benzothiadiazole fluorescent molecules for iron detection. J. Photochem. Photobiol. A 2026, 472, 116846. [Google Scholar] [CrossRef]
- Chetia, J.; Pyngrope, H.; Kharpan, B.; Chanda, S.; Jaiswar, A.; Pradhan, A.K.; Paul, P.C. Glycyl-l-phenylalanine peptide based Schiff base dual ion selective fluorescent chemosensor: Synthesis, detection of Zn2+ and Al3+, real water sample analysis and DFT study. Spectrochim. Acta Part A 2025, 342, 126489. [Google Scholar] [CrossRef]
- Chen, Z.-H.; Chen, Z.-J.; Li, W.-X.; Zeng, Y.; Lin, J.-Q.; Tao, G.-S.; Wang, Z.-Y. Synthesis of a novel N-fused ring based organic molecule probe and its detection of iron. Inorg. Chem. Commun. 2024, 163, 112381. [Google Scholar] [CrossRef]
- Ekennia, A.C.; Osowole, A.A.; Olasunkanmi, L.O.; Onwudiwe, D.C.; Ebenso, E.E. Coordination behaviours of new (bidentate N,O-chelating) Schiff bases towards copper(II) and nickel(II) metal ions: Synthesis, characterization, antimicrobial, antioxidant, and DFT studies. Res. Chem. Intermed. 2017, 43, 3787–3811. [Google Scholar] [CrossRef]
- Yeldir, E.K.; Erdener, D.; Kaya, I. Synthesis and characterization of a pyrene-based Schiff base and its oligomer: Investigation of fluorescent Cr3+ probe. React. Funct. Polym. 2022, 170, 105097. [Google Scholar] [CrossRef]
- Horak, E.; Kassal, P.; Hranjec, M.; Steinberg, I.M. Benzimidazole functionalised Schiff bases: Novel pH sensitive fluorescence turn-on chromoionophores for ion-selective optodes. Sens. Actuators B 2018, 258, 415–423. [Google Scholar] [CrossRef]
- Patil, R.; Moirangthem, A.; Butcher, R.; Singh, N.; Basu, A.; Tayade, K.; Fegade, U.; Hundiwale, D.; Kuwar, A. Al3+ selective colorimetric and fluorescent red shifting chemosensor: Application in living cell imaging. Dalton Trans. 2014, 23, 2895–2899. [Google Scholar] [CrossRef]
- Erdemir, S.; Oguz, M.; Malkondu, S. Fast tracking of Hg2+ ions in living cells, food, and environmental samples using a new mitochondria-targeted red emitting probe and its portable applications. Environ. Pollut. 2025, 367, 125637. [Google Scholar] [CrossRef]
- Zhou, J.J.; Li, J.C.; Xu, X.T.; Long, S.; Cui, N.N.; Zhang, Y.; Shi, L.H.; Zhou, J. Imaging gastrointestinal damage due to acute mercury poisoning using a mitochondria-targeted dual near-infrared fluorescent probe. J. Hazard. Mater. 2024, 470, 134269. [Google Scholar] [CrossRef]
- Pei, S.-C.; Li, C.-Z.; Pei, X.-Y.; Zeng, W.-H.; Zhang, Y.-Y.; Jiang, K.-X.; Huang, X.; Liao, X.-L.; Chen, J. Dual colorimetric and near-infrared fluorescence probe for Hg2+ detection and cell imaging. Spectrochim. Acta A 2022, 285, 121886. [Google Scholar] [CrossRef]
- Ruan, Z.J.; Fan, C.X.; Wang, X.R.; Shao, D.; Yang, X.D.; He, W.T.; Xu, T.; Lin, J.Q.; Tian, Z.F. A novel thioketal containing fluorescent dye for mercury(II) detection via Hg2+-triggered ACQ to AIE transformation. Dyes Pigments 2024, 222, 111835. [Google Scholar] [CrossRef]
- Sreedevi, A.; Shankar, R.; Subashini, G. A dual signalling on-off quinoline imidazole conjugate: A fluorescent chemosensor for Fe2+and Hg2+ ions and bioimaging studies. J. Fluoresc. 2025. [Google Scholar] [CrossRef] [PubMed]
- Janakipriya, S.; Divya, D.; Mala, R.; Nandhagopal, M.; Thennarasu, S. Wavelength specific aggregation induced emission in aqueous media permits selective detection of Ag+ and Hg2+ ions. Spectrochim. Acta Part A 2025, 325, 125085. [Google Scholar] [CrossRef]
- Meng, X.J.; Zhang, Y.R.; Li, J.J. A highly sensitive polydentate ligand-based fluorescent probe for imaging of Hg2+ in arabidopsis thaliana1. Synth. Met. 2024, 307, 117689. [Google Scholar] [CrossRef]
- Wu, C.-Y.; Ma, W.Y.; Sun, X.X.; Li, Y.; Liang, T.Y.; Tian, M.Y.; Zhong, K.L.; Tang, L.J. A smartphone-compatible coumarin-based fluorescent probe for real-time detection of Hg2+ in environmental and biological systems. Microchem. J. 2025, 219, 116056. [Google Scholar] [CrossRef]
- Kar, S.R.; Dash, P.P.; Panda, S.N.; Mohanty, P.; Mohanty, D.; Barick, A.K.; Sahoo, S.K.; Mohapatra, P.; Jali, B.R. A formyl chromone based schiff base derivative: An efficient colorimetric and fluorescence chemosensor for the selective detection of Hg2+ ions. J. Fluoresc. 2025, 35, 483–495. [Google Scholar] [CrossRef]
- Li, H.Q.; Yan, J.B.; Jiang, L.; Zhao, Y.; Song, Y.X.; Yu, J.R.; Cheng, L. Selective and sensitive detection of Hg2+ and Ag+ by a fluorescent and colorimetric probe with large stokes shift. J. Fluoresc. 2024, 34, 2793–2806. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Ge, H.J.; Sun, L.; Cheng, Y.T.; Xu, Z.H.; Gao, W.; Wang, B.L.; Rong, X.Q.; Qiu, X.Y.; Li, J.J.; et al. Novel design of near-infrared fluorescent sensors for the detection of Hg2+in living cells and real water samples. Spectrochim. Acta Part A 2024, 304, 123258. [Google Scholar] [CrossRef]
- Cai, S.-Q.; Liang, L.; Liu, Y.-Q.; Wang, J.-G.; Cai, X.-H. A dual-functional fluorescent probe based on coumarin derivative for highly sensitive and intelligent detection of Zn2+/Cu2+ and cell imaging. J. Hazard. Mater. 2025, 492, 138104. [Google Scholar] [CrossRef]
- Ranjani, M.; Keerthana, V.; Selvakumar, S.; Lynch, V.M.; Mohankumar, A.; Palanisamy, S.; Kalaivani, P.; Prabhakaran, R. Fluorescence turn on/off fluorophore- quencher complexes (Zn2+/Fe2+) of 2,3-dihydro-4-hydroxy-chromene schiff base as chemosensor, live cell imaging (in vitro/in vivo), DFT studiesand proficient cytotoxic agents. J. Mol. Liq. 2025, 433, 127935. [Google Scholar] [CrossRef]
- Pereira, T.M.; Silva Junior, H.D.C.; Da Luz, L.C.; Pereira, M.S.; Miranda, F.D.S.; Da Silva, C.O.; Rodembusch, F.S.; Kümmerle, A.E. Illuminating zinc ions in solution: Selective fluorescence detection with a simple coumarin-thiosemicarbazone sensor. Dyes Pigments 2026, 246, 113389. [Google Scholar] [CrossRef]
- Ricks, K.; Akhtar, M.; Mendez, S.; Liu, C.; Lian, I.; Guo, Z.-F. N-amidothiourea fluorescence sensor for sensitive detection of Zn2+ ions and cell imaging: A tool for plasma membrane analysis. Inorg. Chim. Acta 2025, 578, 122551. [Google Scholar] [CrossRef]
- Kumar, M.S.; Das, A.K.; Bylappa, Y.; Nag, A. Selective dual-mode detection of reactive oxygen species and metal ions by chemodosimetric vs. chelation pathways: Fluorescence ‘turn-on’ with OCl− and Zn2+/Mn2+, employing theoretical, practical, and bioimaging applications. RSC Adv. 2025, 15, 6708–6717. [Google Scholar] [CrossRef]
- Nie, Z.Q.; Wu, S.S.; Fu, Y.; Bian, Y.P.; Gai, L.Z.; Xu, H.J. A highly sensitive fluorescent probe for the detection of Zn2+ based on benzothiazole and triarylimidazole derivatives with AIEE effect. Dyes Pigments 2026, 245, 113239. [Google Scholar] [CrossRef]
- Zhao, L.X.; Shi, J.L.; Liu, Y.Z.; Han, M.F.; Li, S.L.; Cao, D.L. Novel benzothiazole-based fluorescent probe for efficient detection of Cu2+/S2− and Zn2+ and its applicability in cell imaging. Anal. Chim. Acta 2024, 1324, 343093. [Google Scholar] [CrossRef] [PubMed]
- Sevillano-Arredondo, R.M.; Desirena, H.; Juárez-Moreno, K.; Serrano, O.; Ramírez-García, G. A turn-on fluorescence sensor based on water-soluble amino-pyridyl-benzothiazole-derivatives for measuring cellular Zn2+. J. Photochem. Photobiol. A 2023, 445, 115030. [Google Scholar] [CrossRef]
- Das, G.C.; Das, A.K.; Das, D.; Maity, T.R.; Samanta, A.; Alasmary, F.A.; Almalki, A.S.; Iqbal, A.; Dolai, M. Ortho-vanillin based multifunctional scaffold for selective detection of Al3+ and Zn2+ employing molecular logic with DFT study and cell imaging with live grass pea. J. Photochem. Photobiol. A 2023, 440, 114663. [Google Scholar] [CrossRef]
- Yan, L.Q.; Lu, D.Q.; Yang, H.; Wu, X.Z. A dicyanisophorone-based probe for dual sensing Zn2+ and Cd2+ by near-infrared fluorescence. Spectrochim. Acta Part A 2023, 290, 122243. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.